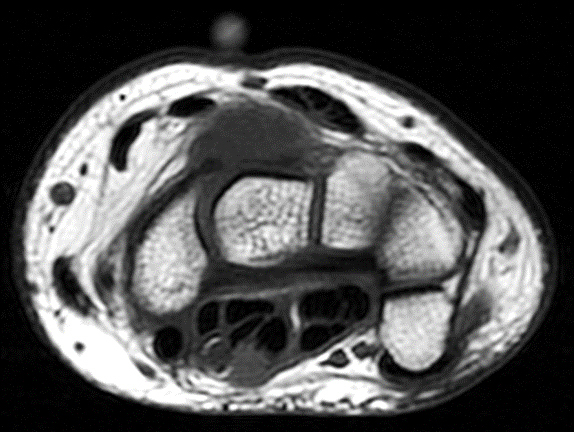
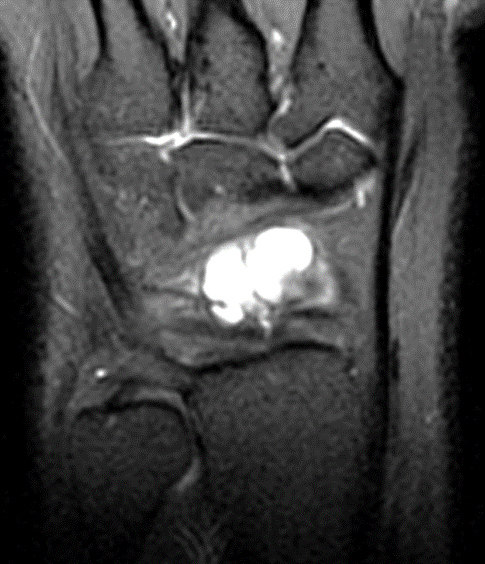
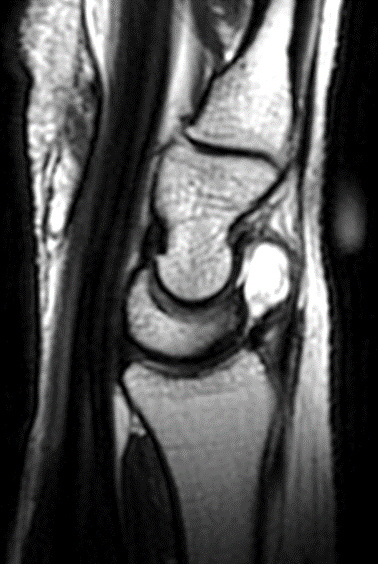
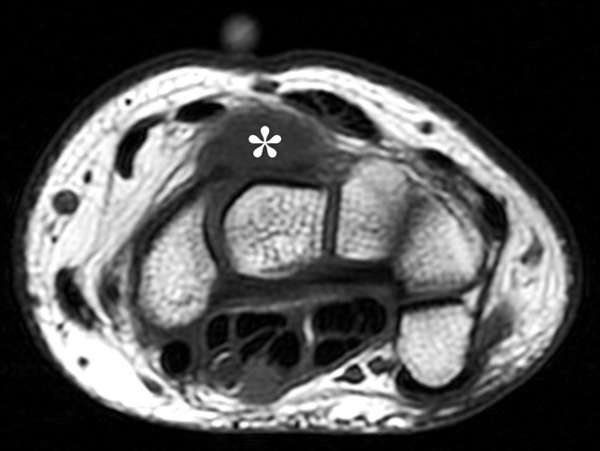
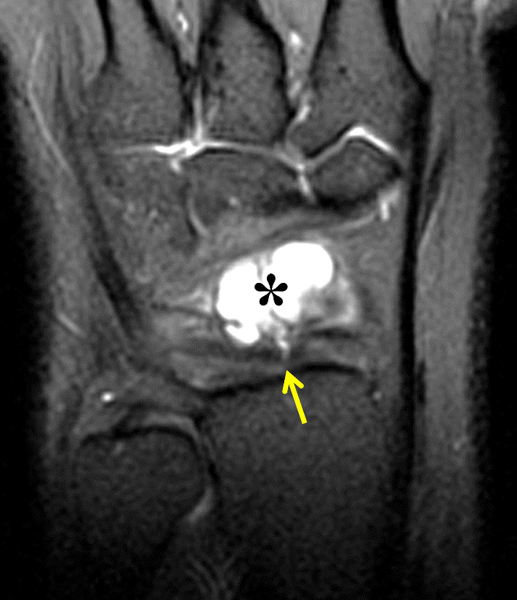
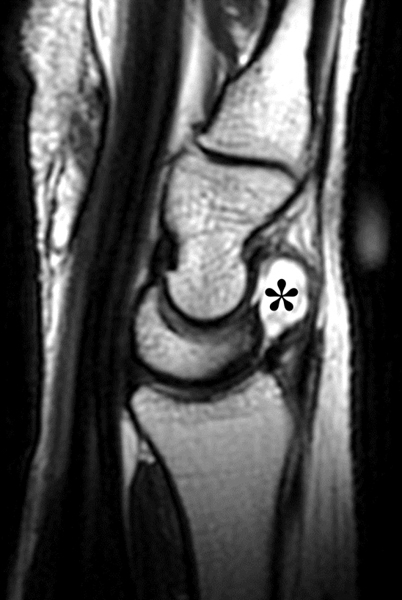
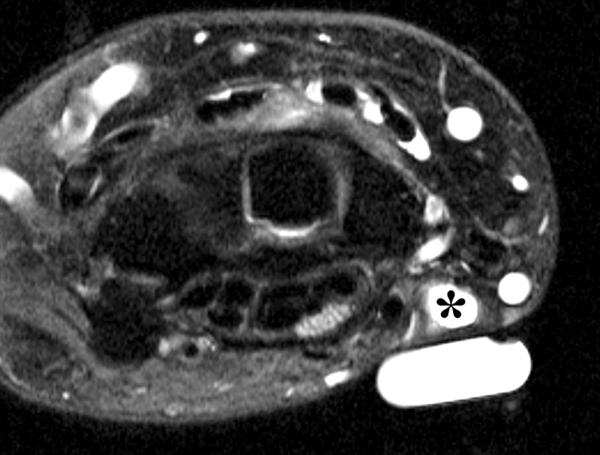
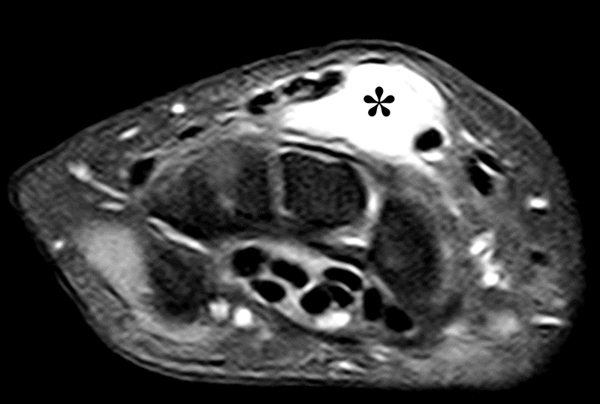
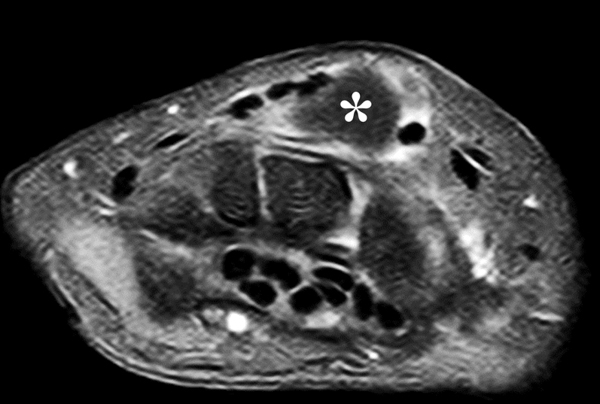
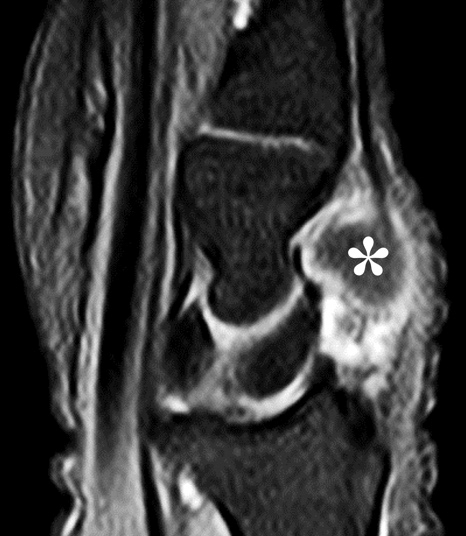
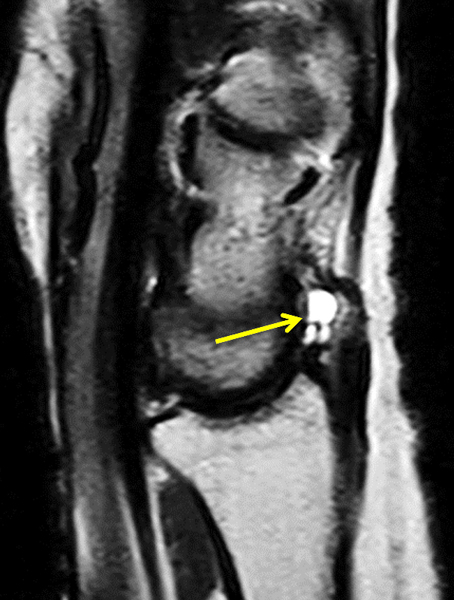
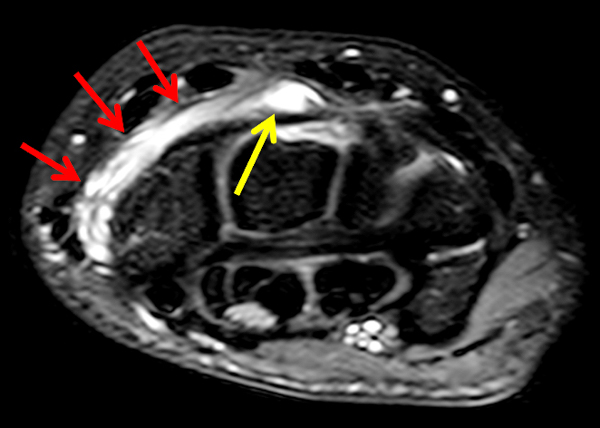
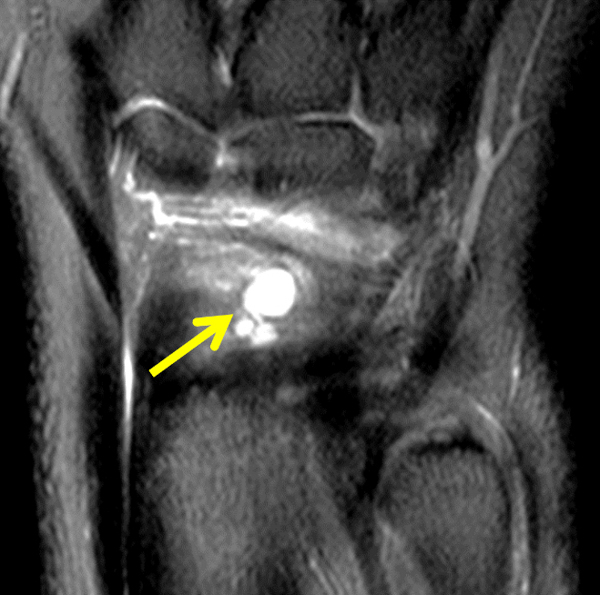
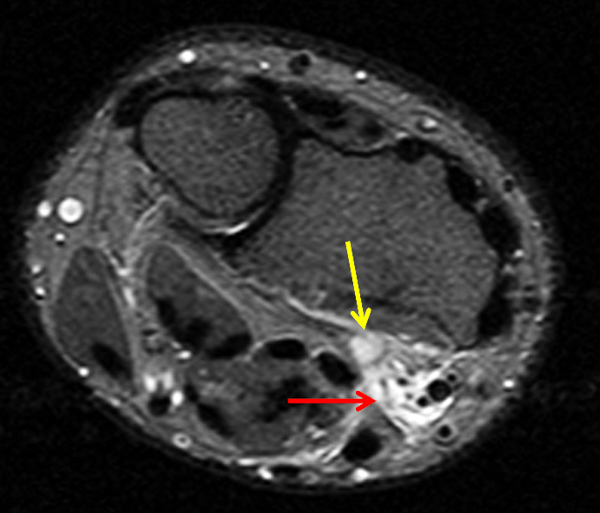
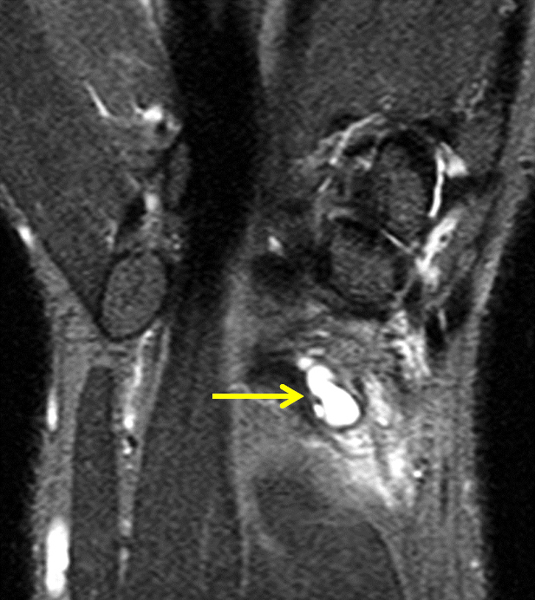
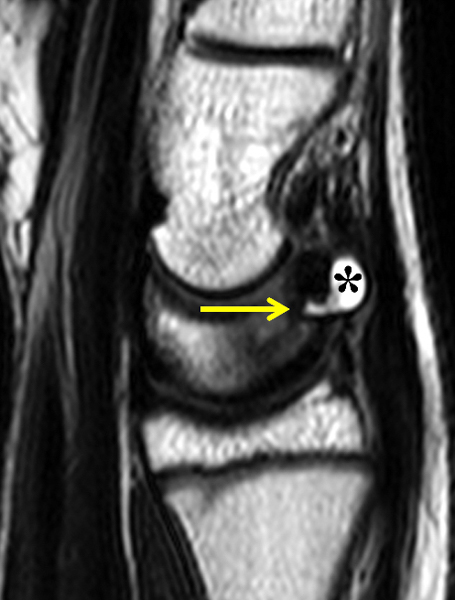
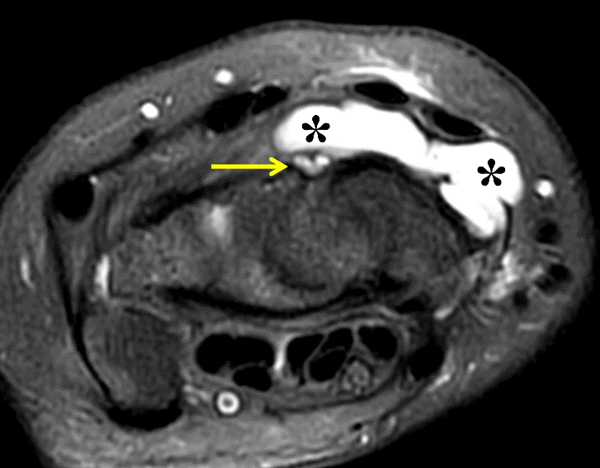
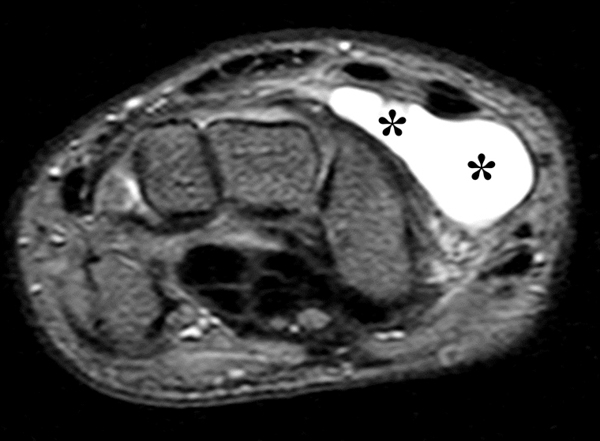
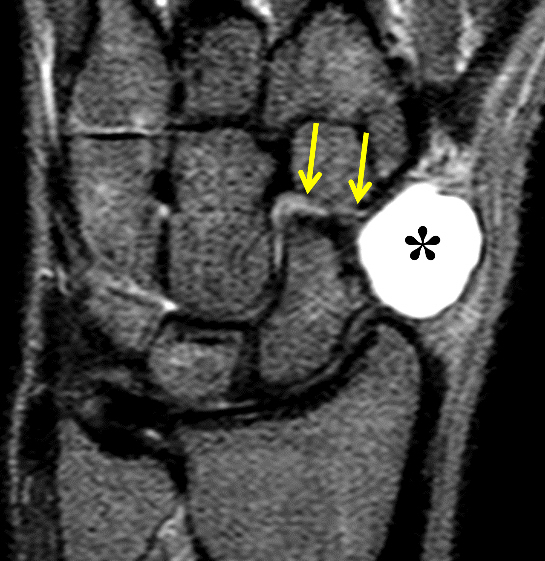
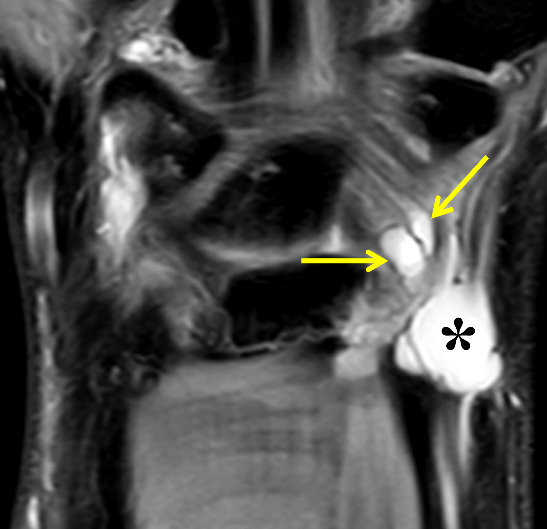
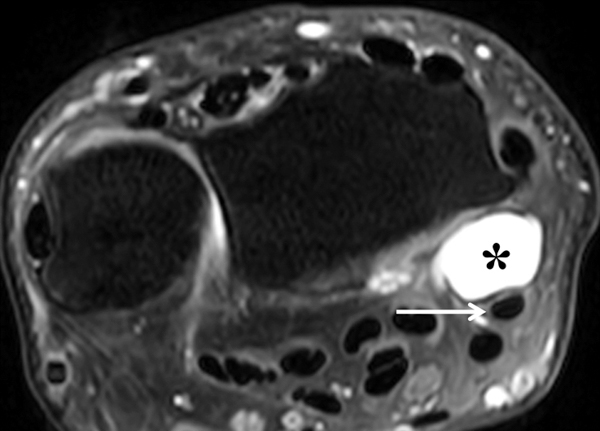
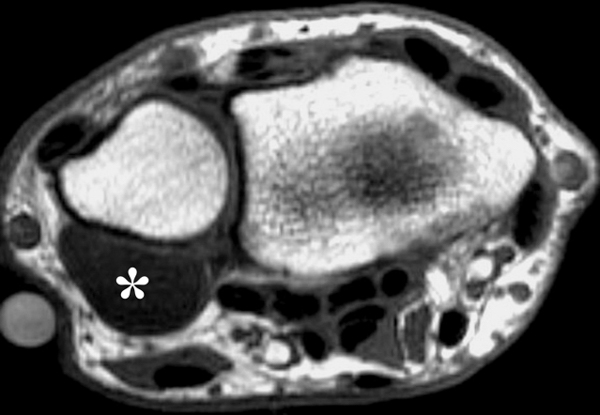
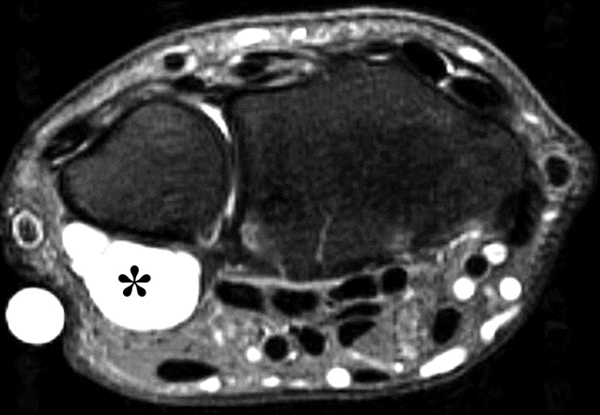
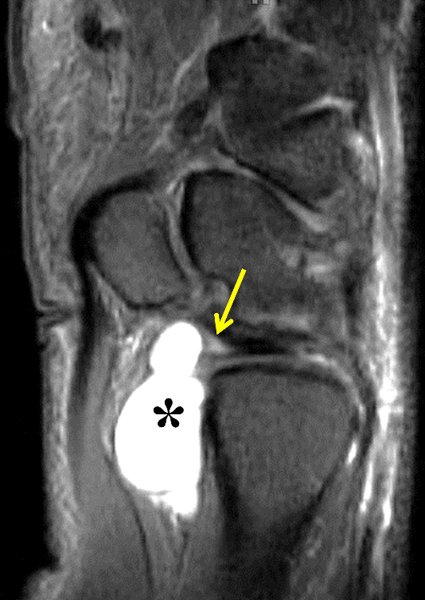
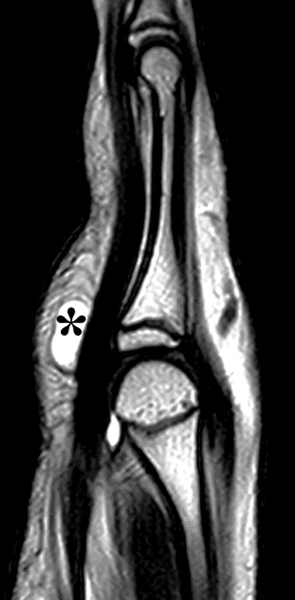
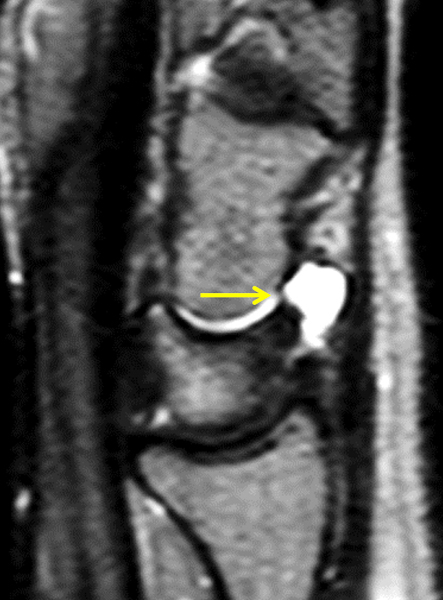
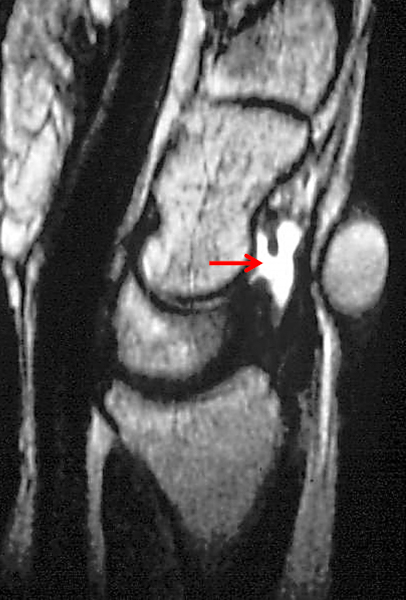
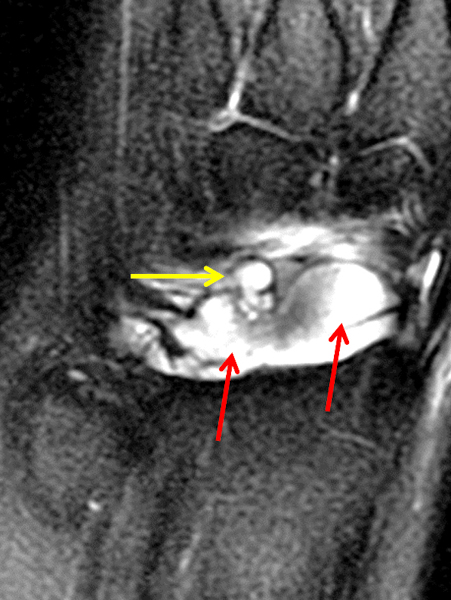
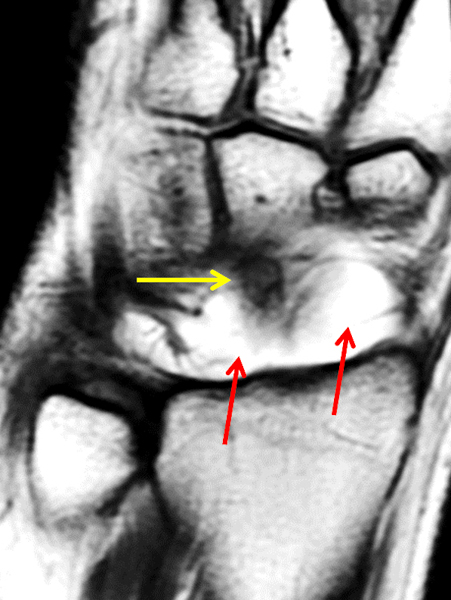
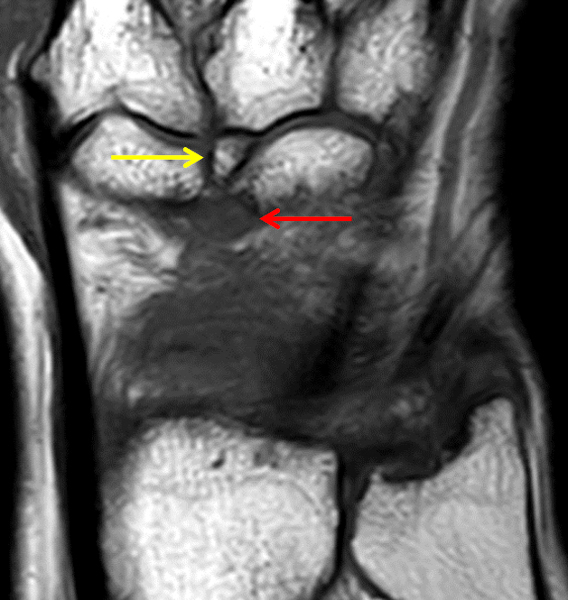
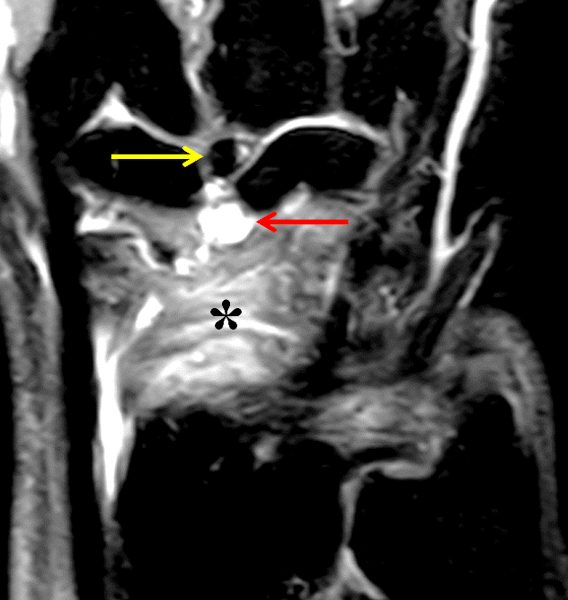
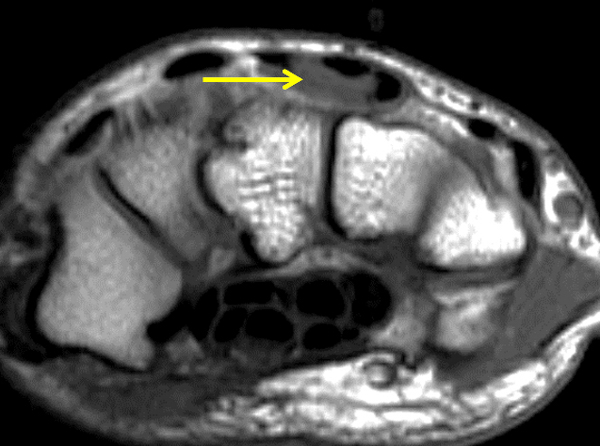
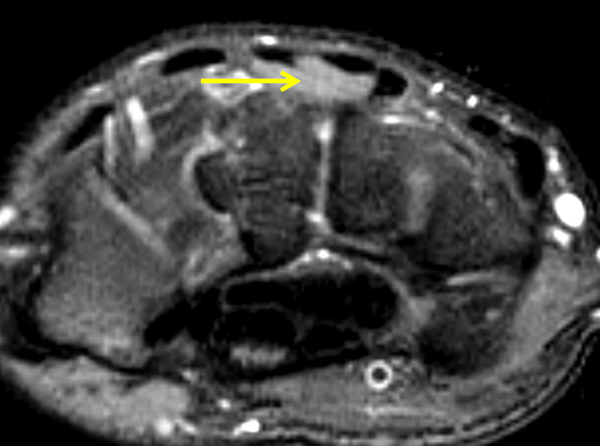
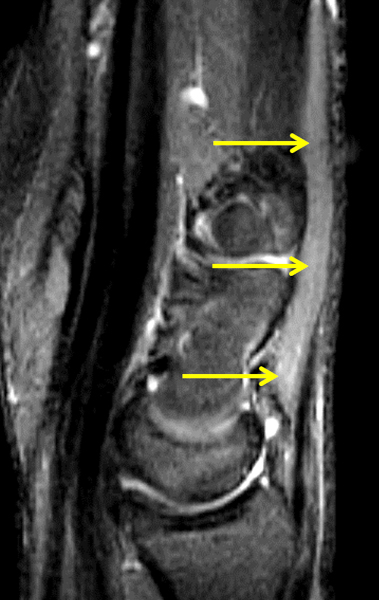
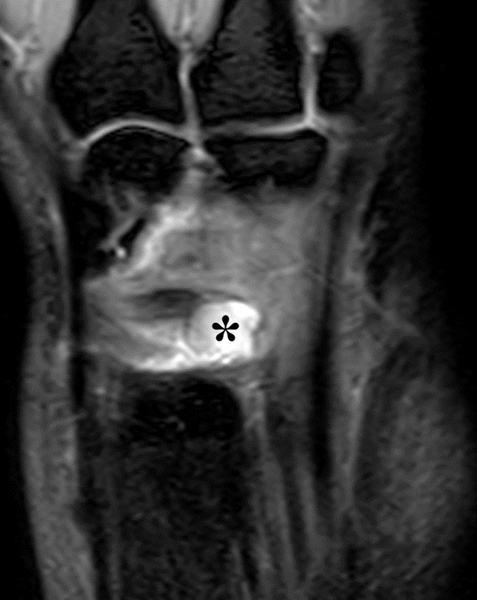
Clinical History: A 22 year-old woman first noted a painless lump on the dorsum of her wrist a few months ago. It aches when the wrist is extended and weightbearing, such as when doing a “plank” during yoga. On exam, the mass feels firm and partly mobile. Axial T1-weighted (1a) and fat-suppressed PD-weighted (1b), coronal fat-suppressed PD-weighted (1c), and sagittal T2-weighted (1d) images are shown. What are the imaging findings? What is your diagnosis?
Figure 2:
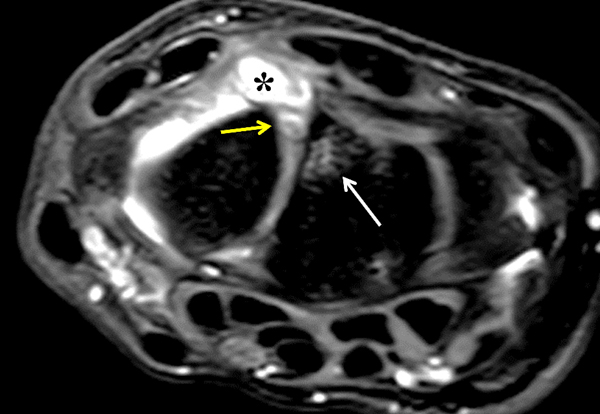
A sharply-marginated, multilocular cystic structure (*) with a thin capsule is centered over the dorsal aspect of the capitolunate articulation, with a pedicle (arrows) emanating from the distal fibers of the scapholunate interosseous ligament. The cyst contents are slightly hypointense compared to skeletal muscle on the T1-weighted images, and hyperintense with thin septations on the water-sensitive images.
Diagnosis
Dorsal carpal ganglion cyst.
Introduction
Ganglia are cysts that lack a true epithelial lining. Their walls are formed by multiple layers of compressed collagen fibers containing a few fibroblasts or mesenchymal cells. The cyst contains an acellular, highly viscous material composed of mucopolysaccharides, albumen, globulin, and hyaluronic acid.1,2 Cysts are either adherent to an underlying joint capsule, ligament, or tendon sheath, or attached via a pedicle. Grossly, ganglion cysts may be unilocular or multilocular with smooth margins, and have a white, translucent appearance.
Historically it was believed that ganglia resulted from synovial tissue that herniated out from a joint. Another concept of pathogenesis was that mucoid degeneration led to the formation of ganglion cysts. The current theory is that repeated microtrauma stretches the ligaments and joint capsule, stimulating the production of mucin, which in turn induces neighboring fibroblasts to produce hyaluronic acid. The mucin dissects through the ligaments and capsule forming droplets that coalesce into lakes and eventually form the cyst. At some point in their natural history, some ganglia will have a communication with the underlying joint (more common for volar compared to dorsal cysts) via merged microcysts in the capsular structures.2 However, an acquired “one-way valve” means that communication can only occur from the joint into the cyst so that any fluid that enters the cyst becomes trapped and eventually resorbs. The communication with the joint is often obliterated by the time of presentation, leaving only a stalk connecting the cyst to the joint.
Ganglion cysts can occur around any joint in the body but are especially common in the wrist, where they account for 60-70% of soft tissue masses. Spontaneous appearance is typical, but some patients report a preceding injury. Symptomatic cysts are most frequent (70%) in the dorsal aspect of the carpus, often arising from the scapholunate ligament. Sometimes a stalk will connect a cyst to the radial-side extensor tendons. Dorsal ganglia may be associated with partial tears of the scapholunate ligament or the radial side of the triangular fibrocartilage complex (TFCC).3,4 Approximately 20% of symptomatic ganglia are radiovolar, usually located between the first dorsal compartment and flexor carpi radialis tendon, just proximal to the radiocarpal joint, often abutting or adherent to the radial artery. Volar ganglia may have a pedicle attached to the radiocarpal, scaphotrapezial, or first carpometacarpal joint; less commonly, a palmar cyst will originate from a flexor tendon sheath.2 Rarely, a radiovolar cyst will dissect into the carpal tunnel and produce median nerve compression.1 Asymptomatic wrist ganglia are very common: an MRI study found ganglia in approximately half of wrists in subjects without symptoms, and in this cohort volar ganglia were much more common than dorsal ones.5 Palmar cysts may also be more common compared to dorsal ones in symptomatic children.6
While ganglion cysts can occur at any age, the peak incidence is 20 to 50 years old, with women affected more often than men.1 Patients typically complain of a painless lump, which may fluctuate in size over time or even spontaneously resolve. On physical exam, palpable cysts are typically firm and mobile. Large ones can often be transilluminated.2 “Occult” dorsal ganglia are lesions where a cyst is not visible or palpable but is suspected due to dorsal pain – which may be exacerbated with wrist loading and extension – and localized tenderness on physical examination.7 In these cases, pain may be due to irritation of a branch of the posterior interosseous nerve.4,8 Large volar cysts present as palpable lumps; symptoms such as weakness, numbness, and tingling may be present if a volar cyst impinges on adjacent peripheral nerve branches.2
Imaging Findings
For lesions with a classic clinical presentation, imaging may not be necessary. Radiographs may be requested to evaluate for concomitant bone or joint abnormalities, but are usually normal, and only rarely change management.9 Either ultrasound of MRI is the next appropriate imaging test.10 Ultrasound evaluation can be used to differentiate a cystic mass from a solid lesion or vascular malformation. Sonographically, ganglia typically appear sharply defined and anechoic with enhanced through transmission, and lack flow on Doppler interrogation.11 However, smaller ones may show some internal echoes due to mucin particles (Figure 3) and lack posterior acoustic enhancement.12 Conversely, larger cysts tend to have more septa and may have thicker walls, postulated to reflect prior ruptures and recurrences.13 Ultrasound guidance may also be used for aspiration or injection, especially for volar ganglia, which are often located adjacent to the radial artery.14
Figure 3:
Volar ganglion cyst. (3a) Transverse ultrasound image shows cyst (calipers), which is hypoechoic, but contains low-level internal echoes, a common finding in smaller ganglia. No flow was present on Doppler imaging (not shown). Note posterior acoustic enhancement (arrows). (3b) Corresponding fat-suppressed T2-weighted image shows the cyst (*) adjacent to the flexor carpi radialis tendon.
On MRI, a ganglion cyst presents as a well-defined, unilocular or multilocular mass with a thin capsule. The contents are typically hypointense compared to skeletal muscle on T1-weighted images and hyperintense on water-sensitive (T2 or T2*-weighted, fat-suppressed intermediate weighted, or STIR) sequences (Figure 2).4,5 It has been reported that an atypical appearance of the lesion contents may be encountered in cases of “complicated” ganglia due to hemorrhage or infection;9,11 however, in clinical practice these instances are rare. If intravenous contrast is given, the contents of the cyst should show no enhancement, while the periphery may show a thin enhancing wall (Figure 4). In small, occult ganglia either wall enhancement or diffuse enhancement may be present,9,15 and it may be hard to distinguish the two (Figure 5) possibly because of contrast material leaking from the enhancing capsule.
Figure 4:
Dorsal ganglion cyst. (4a) Axial water-sensitive (Dixon) and (4b) sagittal T2-weighted images show a typical dorsal ganglion cyst (*) extending from the scapholunate joint capsule superficially between extensor tendon compartments. (4c) Axial and (4d) sagittal fat-suppressed T1-weighted images acquired following intravenous contrast administration demonstrate no internal enhancement within the cyst (*), and a thin layer of peripheral enhancement in the cyst capsule.
While the cyst may extend superficially into the subcutaneous tissues and tent the skin, a portion of the mass should be located deep along the joint capsule or tendon sheath, which is useful to distinguish a ganglion from a lesion that arises superficially, such as an epidermoid inclusion cyst. Partial cyst rupture is suggested when there is surrounding, ill-defined, non-enhancing pericapsular soft tissue infiltration (Figures 6 and 7).9 In instances where a cyst completely ruptures, the soft tissue edema may be the sole finding and only a clinical history of a recently disappeared mass in the same location will be the clue to the correct diagnosis.
Figure 6:
Partly ruptured dorsal ganglion cyst. (6a and 6b) Axial fat-suppressed T2-weighted images show the ganglion cyst (yellow arrows). Note that the cyst capsule along the radial side of the lesion is less well defined in (6b). Ill-defined soft tissue edema tracks radially from the cyst (red arrows). (6c) Fat-suppressed coronal proton density-weighted image demonstrates the focal ganglion (arrow) together with infiltrative soft tissue edema more distally and radially.
Figure 7:
Partly ruptured volar ganglion cyst. Axial (7a) and coronal (7b and 7c) fat-suppressed proton density-weighted images show a small ganglion cyst (yellow arrows) with poorly-defined soft tissue edema extending palmarly and proximally (red arrows), abutting the radial artery and flexor carpi radialis tendon.
Small (< 1 cm in largest dimension) and occult dorsal ganglion cysts will usually be confined to the area immediately dorsal to the scapholunate ligament just proximal to the capitate; an associated tiny intra-ligamentous cyst may be present (Figure 8). Not surprisingly, very small cysts (< 4 mm) are more likely to be overlooked on MRI, especially when the imaging requisition does not specifically mention clinical suspicion of an occult ganglion.16 Larger cysts can track distally either superficial or deep to the extrinsic scaphotriquetral ligament, or can have prominent radial or even volar extension (Figure 9). Continuity with or a pedicle extending to the scapholunate joint capsule should be sought and noted because these cysts can be managed arthroscopically, while dorsal cysts arising elsewhere require open surgery (Figure 10).9
Figure 8:
Occult dorsal ganglion cyst. (8a) Axial fat-suppressed T2-weighted image and (8b) sagittal STIR image show the ganglion cyst (*), which was not palpable, directly dorsal to the scapholunate joint. Note communication with a small intra-ligamentous cyst (arrows) within the dorsal fibers of the scapholunate interosseous ligament.
Figure 9:
Large dorsal ganglion cyst originating from scapholunate joint. The cyst was palpable along the radial side of the second compartment tendons (most are located ulnar to the second compartment). (9a) Axial and (9b) coronal fat-suppressed proton density-weighted images demonstrate the cyst (*) with a prominent radial component. Despite its size and extent, note the pedicle that extends from the scapholunate ligament and the small intra-ligamentous cyst (arrows).
Figure 10:
Large dorsal ganglion cyst originating from scaphotrapeziotrapezoid joint. (10a) Axial and (10b) coronal STIR images show a cyst (*) in an almost identical location compared to the patient in Figure 9. However, the pedicle in this case originates at the triscaphe articulation (arrows), making this cyst not amenable to arthroscopic resection.
Radiovolar cysts that originate from the radiocarpal joint usually occur between the volar extrinsic wrist ligaments,5,9 and will show some interdigitation with the fibers of the radioscaphocapitate and/or long radiolunate ligaments (Figure 11). For volar cysts, identifying a radiocarpal origin is important for management because these lesions may be treated arthroscopically unlike those whose origin is the scaphotrapezial articulation.17 The relationship to the radial artery and adjacent tendon sheaths should also be noted on MRI.
Figure 11:
Volar ganglion cyst. Fat-suppressed proton density-weighted (11a) coronal and (11b) axial images show the volar cyst (*) with a loculated component interdigitating with the fibers of the extrinsic radioscaphocapitate and long radiolunate ligaments (yellow arrows). The adjacent flow void (white arrow) is within an enlarged vein; the radial artery is not visible separate from the ganglion cyst.
While dorsal and radiovolar ganglia are the most common, approximately 10% of lesions originate elsewhere in the wrist and hand. Ulnar-sided ganglia may arise from the ulnocarpal joint (Figure 12), where they are associated with TFCC tears in approximately 45% of cases,3 or from the pisotriquetral joint capsule.9 These ulnar-sided ganglia can present as either dorsal or volar masses; they may produce neurologic symptoms by compressing the ulnar nerve in Guyon’s canal, or smaller motor or sensory branches.1,9 In the fingers, ganglia often originate from the flexor tendon sheaths (sometimes called volar retinacular ganglia), most commonly in the long finger near the A1 or A2 pulleys (Figure 13).18,19 Cysts associated with osteoarthritis of the distal interphalangeal joints, called digital mucoid cysts, may present as dorsal finger masses, or may be occult and confined to the nail bed, producing fingernail changes.20
Figure 12:
Ulnar-sided ganglion cyst. Axial (12a) T1-weighted and (12b) fat-suppressed T2-weighted images show an ulnovolar ganglion cyst (*) along the palmar margin of the distal radioulnar joint capsule. (12c) A fat-suppressed T2-weighted sagittal image shows the relationship of the cyst (*) to a tear of the volar distal radioulnar ligament (arrow). (12d) A fat-suppressed T2-weighted coronal image demonstrates an associated tear involving the ulnar foveal attachment of the triangular fibrocartilage complex (arrow).
Differentiating a ganglion cyst from synovitis or a distended wrist recess (or even a diverticulum of a recess) can be challenging at times. The most useful distinguishing criteria are the shape and margins of the lesion.15 In the axial plane, ganglia tend to be round or tubular compared to the more crescentic shape of recesses.9 The margins of a ganglion cyst are usually sharp. Recognizing a characteristic location is also useful, e.g., dorsal to the scapholunate articulation for a dorsal ganglion, or directly proximal or distal to the pisotriquetral recesses for a diverticulum.9 Synovitis usually does not contain septations. Synovitis diffusely enhances following contrast administration whereas ganglia are more likely to show only peripheral wall enhancement.15 The fact that most dorsal ganglia do not have a free communication with the underlying joint can also be used to distinguish cysts from synovial recesses. On ultrasound, applied compression will decompress a recess back into the joint whereas a ganglion will be noncompressible.21,22 MRI images will show an intact capsule between a ganglion cyst and the joint space, while a communicating defect in the capsule may be visible between a recess and the joint (Figure 14). If direct MR arthrography is performed, contrast will typically not flow from the joint into a dorsal ganglion cyst, so that recesses will contain high-signal-intensity contrast on T1-weighted images unlike ganglia that will not, even though both may appear identical on water-sensitive sequences (Figure 15). These features may not always be present for volar ganglia, which more commonly communicate with the adjacent joint,12 nor after surgical treatment, where a window in the joint capsule may have been created.
Figure 14:
Dorsal ganglion cyst compared to dorsal joint recess. (14a) Sagittal STIR image shows a dorsal ganglion cyst. Note the thin low-signal-intensity capitolunate joint capsule (yellow arrow) separating the cyst from the fluid in the underlying joint. joint. (14b) Sagittal T2-weighted image in a different patient with a distended dorsal recess. A wide defect (red arrow) in the capsule allows communication of the fluid with the joint.
Figure 15:
Dorsal ganglion cyst on MR arthrogram. (15a) Coronal fat-suppressed T2-weighted image shows both a ganglion cyst (yellow arrow) and distended dorsal recesses (red arrows) as high signal-intensity structures. On coronal T1-weighted (15b) and axial fat-suppressed T1-weighted (15c) images obtained after a dilute gadolinium-based contrast mixture was instilled into the radiocarpal joint, the recesses (red arrows) contain high-signal intensity contrast because they communicate with the joint, while the ganglion cyst (yellow arrows) does not.
Treatment
Asymptomatic cysts where the patient is not concerned about cosmesis require no specific treatment; approximately 50% of these will spontaneously regress.2,23 Supportive measures including immobilization and anti-inflammatory medications may be added. Patients have been known to self-treat dorsal ganglia by attempting to rupture the cyst applying blunt force – traditionally using a heavy book, thus the colloquial term “Bible bump” applied to these cysts. While there are no controlled studies examining the effectiveness of this technique, anecdotal surveys suggest that forceful smashing of a cyst may work in some cases.24 Needle aspiration of a cyst, with or without injection (typically hyaluronidase or steroid) or cyst fenestration is typically offered to patients as an initial procedure; however, the recurrence rate is high (> 50%),24,25 especially for volar cysts.2 Furthermore, the proximity of volar cysts to the radial artery makes clinically-directed aspiration potentially more hazardous; ultrasound-guided volar ganglion cyst aspiration appears to be safe, although still hampered by a high (66%) recurrence rate.14
Surgical treatment for ganglion cysts includes both open and arthroscopic techniques. During open excision, the surgeon removes the entire pedicle and capsular attachments to decrease the risk of recurrence.2 Some surgeons will also cauterize the stalk location. The capsular tissue may be left open to reduce the incidence of post-operative stiffness. Recurrence rates are approximately 15% for dorsal ganglia and 30% or higher for volar ones.2 Dorsal cysts originating from the scapholunate ligament and volar cysts arising from the radiocarpal joint can potentially be treated arthroscopically. During arthroscopy, a small window is created in the dorsal or volar capsule, allowing the cyst contents to decompress into the joint.17,26 Reported arthroscopic recurrence rates are similar to those for open surgery (approximately 11% for dorsal ganglia).23 Recurrent ganglia have the same imaging appearances as first-time cysts.
Differential Diagnosis
Several processes can result in a mass, with or without pain, that can clinically mimic a dorsal ganglion cyst. MRI is useful in diagnosing these conditions. Extensor tenosynovitis can be related to overuse or may be a manifestation of a collagen vascular disease or primary arthritis such as rheumatoid arthritis. When the fourth dorsal compartment is involved, the associated swelling and pain may be similar to symptoms associated with a ganglion cyst. MRI demonstrates distention of the affected tendon sheaths with fluid and/or enhancing synovium; associated tendinosis or tendon tears may coexist (Figure 16).27 Carpal boss describes a dorsal ossicle separate from or attached to the second or third metacarpal base, or rarely the distal capitate or trapezoid (https://radsource.us/sesamoid-bones/). The accessory bone may be congenital, or the result of prior trauma, and can coexist with an adjacent ganglion cyst.28 MRI may show associated degenerative bone changes (marrow edema, cysts, osteophytes) in symptomatic cases (Figure 17). Rarely an accessory muscle – the extensor digitorum brevis manus – will present as a dorsal mass (https://radsource.us/accessory-muscles-of-the-hand-and-wrist/). This anomalous muscle will have the same signal characteristics as skeletal muscle on all pulse sequences, and can be recognized by following it from its origin distal to the extensor retinaculum to its insertion into the extensor hood of the index and/or long finger (Figure 18).29 The main differential diagnosis for an occult dorsal ganglion cyst, which presents with focal pain and tenderness but no palpable mass, is a tear of the scapholunate ligament, which can also coexist with a ganglion (Figure 19).23
Figure 16:
Extensor tenosynovitis. An axial fat-suppressed T2-weighted image shows high signal-intensity fluid or tenosynovitis surrounding the extensor indicis and extensor digitorum communis tendons in the fourth dorsal compartment (arrow). Intratendinous signal represents associated tendinosis.
Figure 17:
Symptomatic carpal boss and associated ganglion cyst. Coronal (17a) T1-weighted and (17b) fat-suppressed proton density-weighted images demonstrate a marrow-containing accessory ossicle (yellow arrows) at the base of the third metacarpal, with an adjacent dorsal ganglion cyst (red arrows). Generalized dorsal synovitis (*) is present more proximally. (17c) A sagittal fat-suppressed T2-weighted image shows marrow edema representing degeneration between the third metacarpal base and the carpal boss (yellow arrows), as well as the associated ganglion cyst (red arrow).
Figure 18:
Anomalous muscle presenting as a dorsal mass. Axial (18a) T1-weighted and (18b) fat-suppressed T2-weighted images demonstrate that the mass (arrows) is associated with the finger extensor tendons and has same signal characteristics and texture as normal skeletal muscle. (18c) Coronal T1-weighted and (18d) sagittal fat-suppressed T2-weighted images reveal the elongated shape of the structure (arrows), paralleling the extensor tendons and extending distally from the wrist joint capsule, diagnostic of an extensor digitorum brevis manus.
Figure 19:
Partial scapholunate ligament tear and dorsal ganglion cyst. Fat-suppressed T2-weighted coronal (19a and 19b) and axial (19c) images show a typical dorsal ganglion cyst (*) and a partial tear of the dorsal fibers of the scapholunate interosseous ligament (yellow arrows). Note the small focus of reactive marrow edema in the lunate (white arrow) adjacent to the ligament injury.
The most common condition that mimics a radiovolar ganglion cyst is a distended joint recess, as discussed above. Abnormalities of the radial artery (traumatic pseudoaneurysm, cystic adventitial disease) should also be considered when patients have complaints in this area. In the fingers, tenosynovial giant cell tumor (the second most common hand-and-wrist mass after ganglion cyst) arises within the tendon sheaths or joint synovium (https://radsource.us/pigmented-villonodular-synovitis-2/). This condition results in lobular soft tissue masses of variable signal intensity and often evidence of hemosiderin, which will appear hypointense compared to skeletal muscle on T2-weighted images and show “blooming” on gradient-echo sequences. Tenosynovial giant cell tumors enhance after contrast administration and may erode the adjacent bone, two additional features that differentiate them from flexor sheath ganglion cysts.11 Low-flow venous malformations (https://radsource.us/venous-malformations/) can occur almost anywhere in the hand or wrist. They are less sharply defined compared to ganglion cysts and appear as lobules or tubules with interposed fat on T1-weighted images and heterogeneous high-signal intensity on water-sensitive sequences, with enhancement following contrast administration. Venous malformations may also contain calcifications or hemosiderin.11 Lastly, other common masses including lipomas, epidermoid inclusion cysts, and true synovial cysts related to osteoarthritis can occur in almost any location, each with characteristic MRI appearances.
Conclusion
Ganglion cysts are the most common masses encountered in the wrist. The role of imaging in suspected cases is to (1) confirm the presence of a cystic mass as opposed to a solid lesion, (2) identify the origin of the cyst by locating its associated pedicle, (3) establish the relation of the cyst to surrounding anatomic structures, and (4) evaluate the wrist for coexistent or alternative pathologic conditions. MRI’s ability to accurately depict the anatomy and characterize lesions fulfills these criteria.
References
- Nahra ME, Bucchieri JS. Ganglion cysts and other tumor related conditions of the hand and wrist. Hand Clin. 2004;20(3):249-260, v. doi:10.1016/j.hcl.2004.03.015 ↩
- Gude W, Morelli V. Ganglion cysts of the wrist: pathophysiology, clinical picture, and management. Curr Rev Musculoskelet Med. 2008;1(3-4):205-211. doi:10.1007/s12178-008-9033-4 ↩
- el-Noueam KI, Schweitzer ME, Blasbalg R, Farahat AA, Culp RW, Osterman LA, et al. Is a subset of wrist ganglia the sequela of internal derangements of the wrist joint? Radiology PACS MR imaging findings. Radiology. 1999;212(2):537-540. doi:10.1148/radiology.212.2.r99au34537 ↩
- Goldsmith S, Yang SS. Magnetic resonance imaging in the diagnosis of occult dorsal wrist ganglions. J Hand Surg Eur Vol. 2008;33(5):595-599. doi:10.1177/1753193408092041 ↩
- Lowden CM, Attiah M, Garvin G, Macdermid JC, Osman S, Faber KJ. The prevalence of wrist ganglia in an asymptomatic population: magnetic resonance evaluation. J Hand Surg Br. 2005;30(3):302-306. doi:10.1016/j.jhsb.2005.02.012 ↩
- Zhou J, Al-Ani S, Jester A, Oestreich K, Baldrighi C, Ting JWC. Wrist Ganglion Cysts in Children: An Update and Review of the Literature. Hand (N Y). 20201558944720966716. ↩
- Nance EM, Byun DJ, Endo Y, Wolfe SW, Lee SK. Dorsal Wrist Pain in the Extended Wrist-Loading Position: An MRI Study. J Wrist Surg. 2017;6(4):276-279. doi:10.1055/s-0037-1599829 ↩
- Vo P, Wright T, Hayden F, Dell P, Chidgey L. Evaluating dorsal wrist pain: MRI diagnosis of occult dorsal wrist ganglion. J Hand Surg Am. 1995;20(4):667-670. doi:10.1016/S0363-5023(05)80288-6 ↩
- Freire V, Guerini H, Campagna R, Moutounet L, Dumontier C, Feydy A, et al. Imaging of hand and wrist cysts: a clinical approach. AJR Am J Roentgenol. 2012;199(5):W618-628. doi:10.2214/AJR.11.8087 ↩
- Expert Panel on Musculoskeletal Imaging: Rubin DA, Roberts CC, Bencardino JT, Bell AM, Cassidy RC, Chang EY, et al. ACR Appropriateness Criteria((R)) Chronic Wrist Pain. J Am Coll Radiol. 2018;15(5S):S39-S55. doi:10.1016/j.jacr.2018.03.021 ↩
- Nguyen V, Choi J, Davis KW. Imaging of wrist masses. Curr Probl Diagn Radiol. 2004;33(4):147-160. doi:10.1016/j.cpradiol.2004.01.002 ↩
- Wang G, Jacobson JA, Feng FY, Girish G, Caoili EM, Brandon C. Sonography of wrist ganglion cysts: variable and noncystic appearances. J Ultrasound Med. 2007;26(10):1323-1328; quiz 1330-1321. doi:10.7863/jum.2007.26.10.1323 ↩
- Teefey SA, Dahiya N, Middleton WD, Gelberman RH, Boyer MI. Ganglia of the hand and wrist: a sonographic analysis. AJR Am J Roentgenol. 2008;191(3):716-720. doi:10.2214/AJR.07.3438 ↩
- Gitto S, Lee SC, Miller TT. Ultrasound-guided percutaneous treatment of volar radiocarpal ganglion cysts: Safety and efficacy. J Clin Ultrasound. 2019;47(6):339-344. doi:10.1002/jcu.22724 ↩
- Anderson SE, Steinbach LS, Stauffer E, Voegelin E. MRI for differentiating ganglion and synovitis in the chronic painful wrist. AJR Am J Roentgenol. 2006;186(3):812-818. doi:10.2214/AJR.04.1879 ↩
- Versteeg G, Goyal K. Radiologist Identification of Occult Dorsal Wrist Ganglion Cysts on MRI. J Wrist Surg. 2019;8(4):276-279. doi:10.1055/s-0039-1683847 ↩
- Fernandes CH, Miranda CD, Dos Santos JB, Faloppa F. A systematic review of complications and recurrence rate of arthroscopic resection of volar wrist ganglion. Hand Surg. 2014;19(3):475-480. doi:10.1142/S0218810414300046 ↩
- Abe Y, Watson HK, Renaud S. Flexor tendon sheath ganglion: analysis of 128 cases. Hand Surg. 2004;9(1):1-4. doi:10.1142/s0218810404001930 ↩
- Kulinski S, Gutkowska O, Mizia S, Gosk J. Ganglions of the hand and wrist: Retrospective statistical analysis of 520 cases. Adv Clin Exp Med. 2017;26(1):95-100. doi:10.17219/acem/65070 ↩
- Drapé JL, Idy-Peretti I, Goettmann S, Salon A, Abimelec P, Guérin-Surville H, et al. MR imaging of digital mucoid cysts. Radiology. 1996;200(2):531-536. doi:10.1148/radiology.200.2.8685352 ↩
- Cardinal E, Buckwalter KA, Braunstein EM, Mih AD. Occult dorsal carpal ganglion: comparison of US and MR imaging. Radiology. 1994;193(1):259-262. doi:10.1148/radiology.193.1.8090903 ↩
- Chiavaras MM, Jacobson JA, Yablon CM, Brigido MK, Girish G. Pitfalls in wrist and hand ultrasound. AJR Am J Roentgenol. 2014;203(3):531-540. doi:10.2214/AJR.14.12711 ↩
- Mathoulin C, Gras M. Arthroscopic Management of Dorsal and Volar Wrist Ganglion. Hand Clin. 2017;33(4):769-777. doi:10.1016/j.hcl.2017.07.012 ↩
- Trivedi NN, Schreiber JJ, Daluiski A. Blunt Force May be an Effective Treatment for Ganglion Cysts. HSS J. 2016;12(2):100-104. doi:10.1007/s11420-016-9493-9 ↩
- Head L, Gencarelli JR, Allen M, Boyd KU. Wrist ganglion treatment: systematic review and meta-analysis. J Hand Surg Am. 2015;40(3):546-553 e548. doi:10.1016/j.jhsa.2014.12.014 ↩
- Borisch N. Arthroscopic resection of occult dorsal wrist ganglia. Arch Orthop Trauma Surg. 2016;136(10):1473-1480. doi:10.1007/s00402-016-2539-0 ↩
- Plotkin B, Sampath SC, Sampath SC, Motamedi K. MR Imaging and US of the Wrist Tendons. Radiographics. 2016;36(6):1688-1700. doi:10.1148/rg.2016160014 ↩
- Conway WF, Destouet JM, Gilula LA, Bellinghausen HW, Weeks PM. The carpal boss: an overview of radiographic evaluation. Radiology. 1985;156(1):29-31. doi:10.1148/radiology.156.1.3923555 ↩
- Rodriguez-Niedenfuhr M, Vazquez T, Golano P, Parkin I, Sanudo JR. Extensor digitorum brevis manus: anatomical, radiological and clinical relevance. A review. Clin Anat. 2002;15(4):286-292. doi:10.1002/ca.10027 ↩