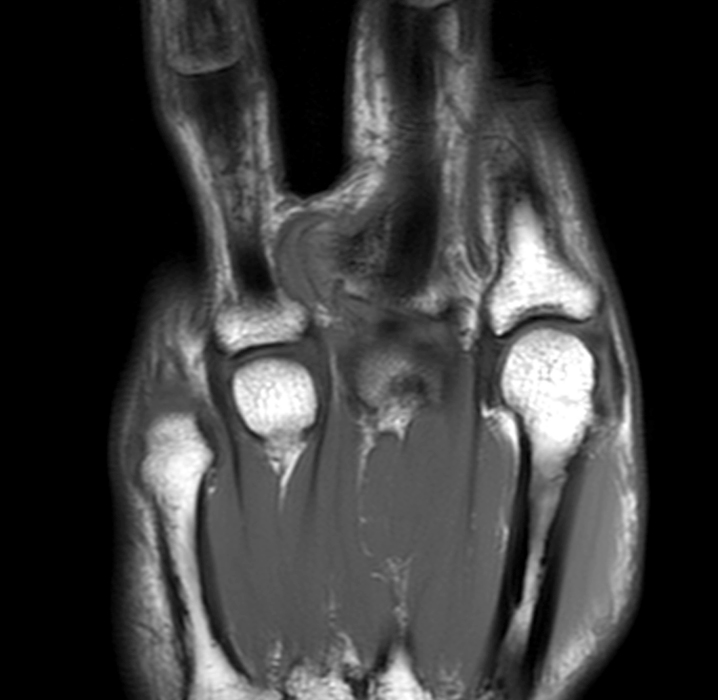
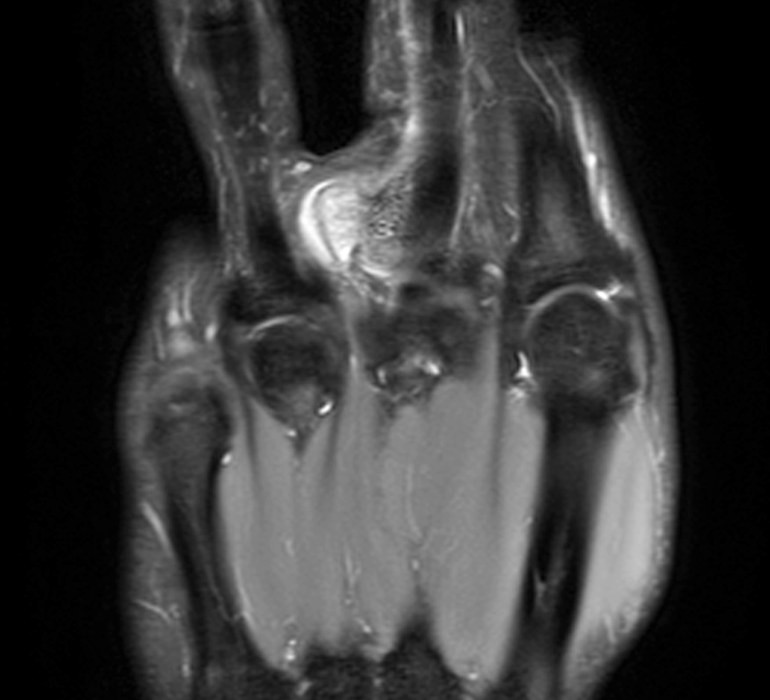
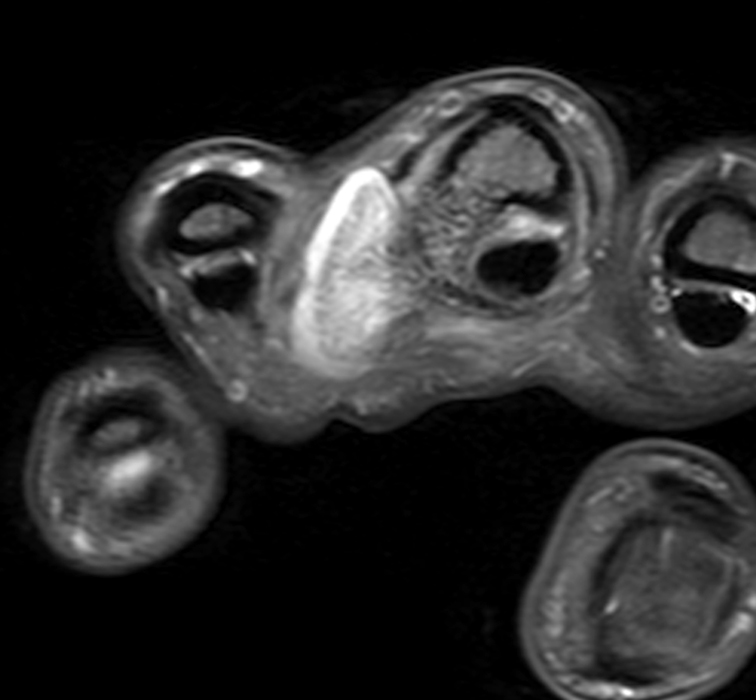
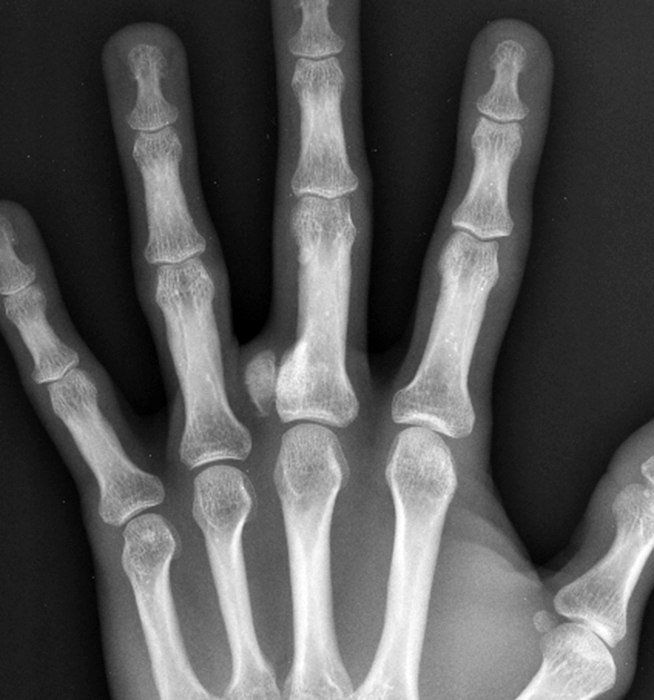
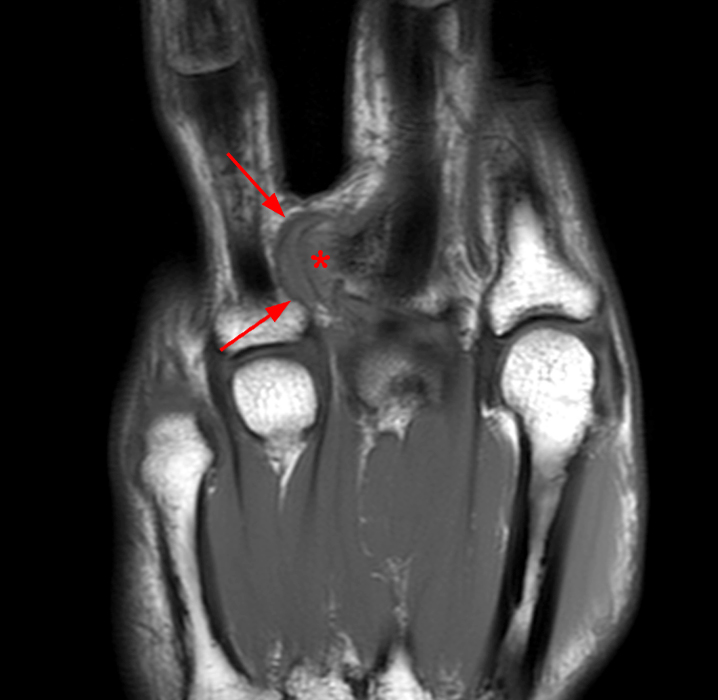
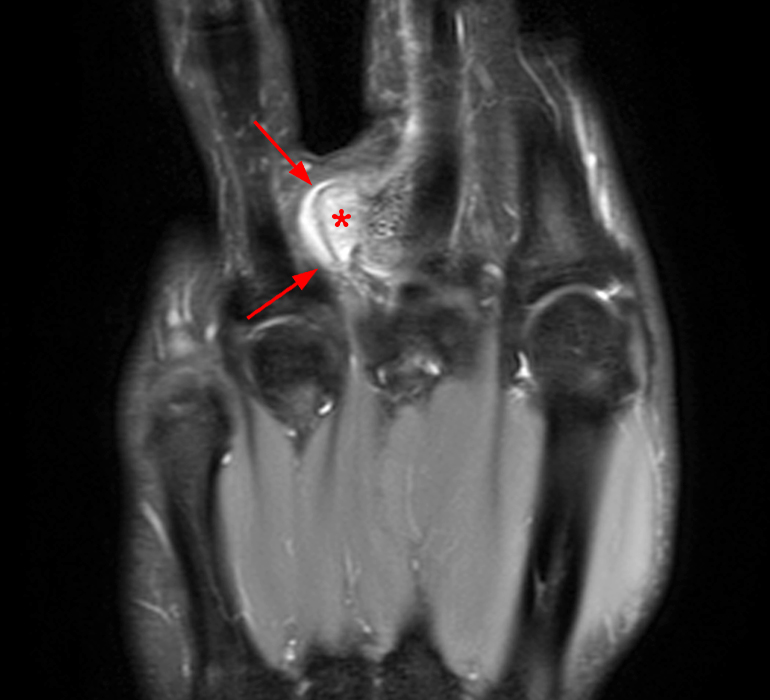
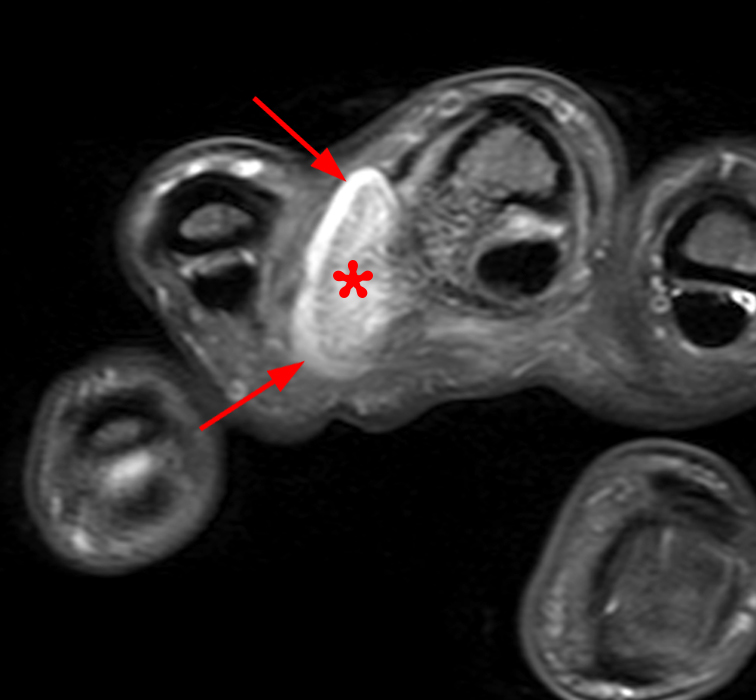
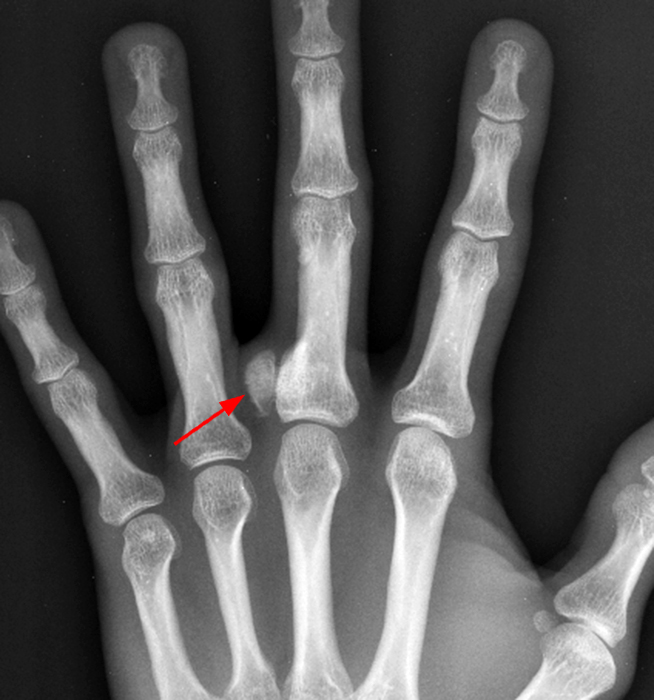
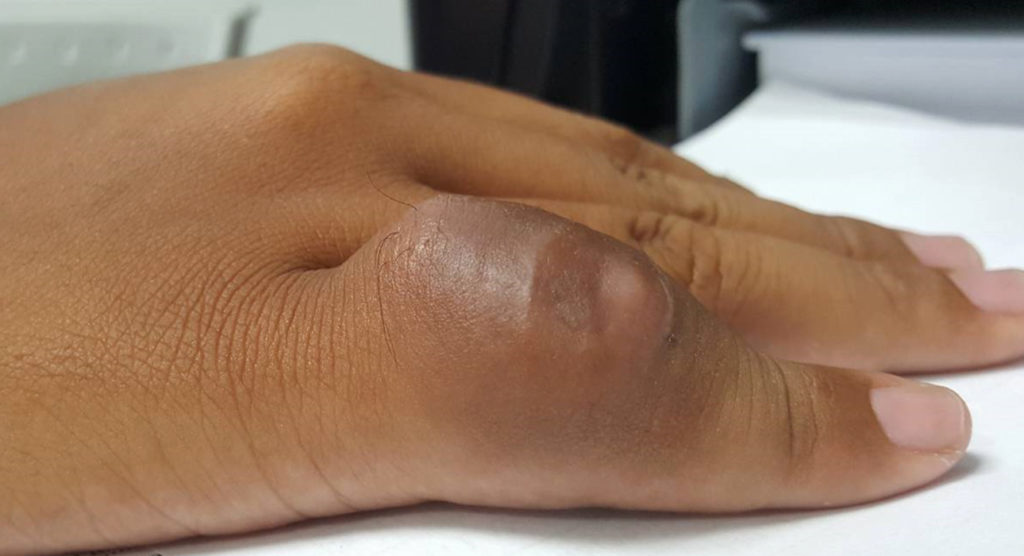
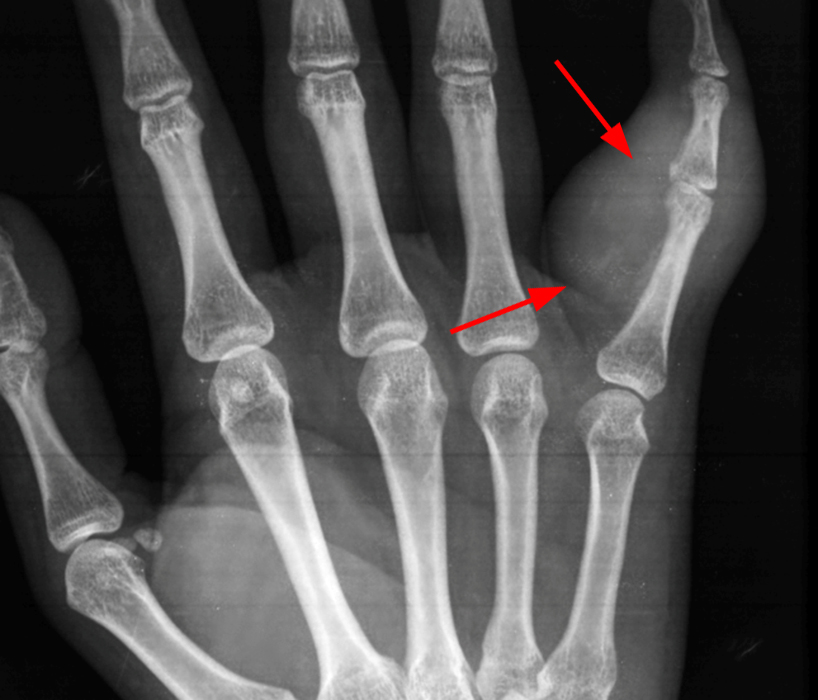
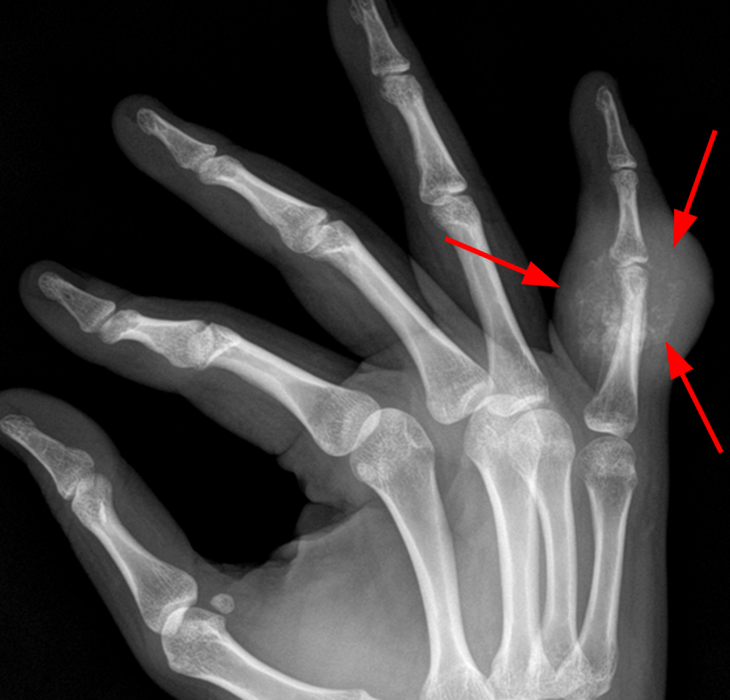
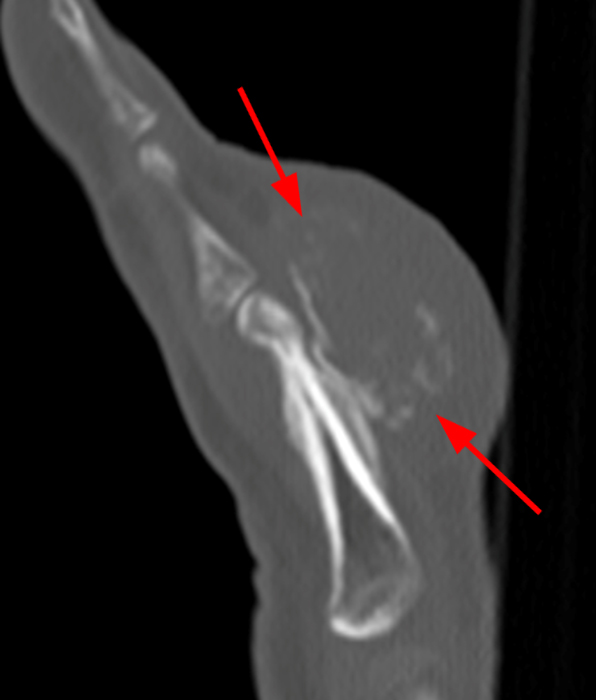
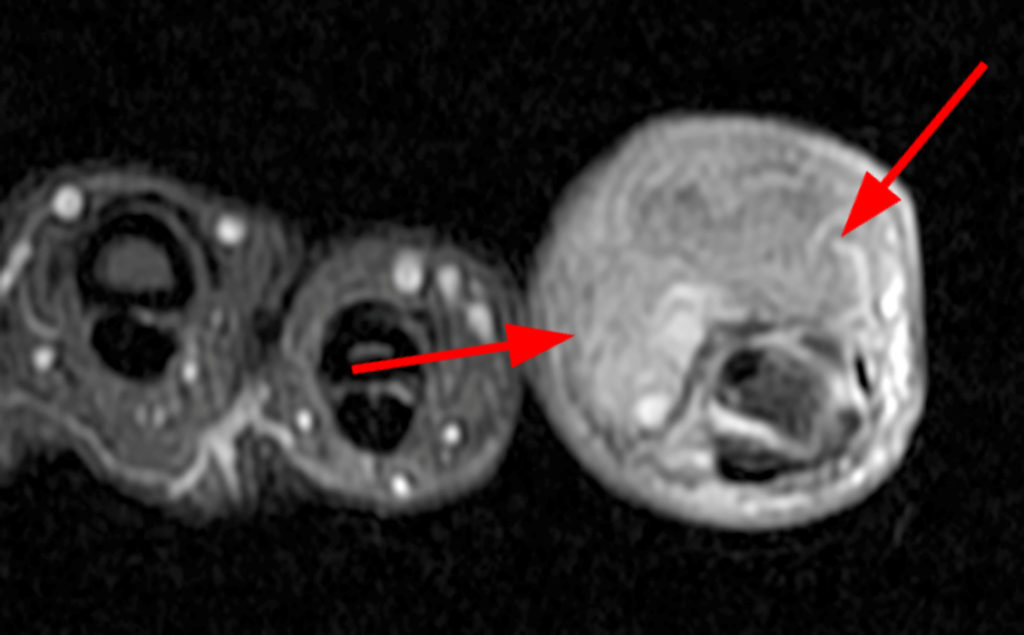
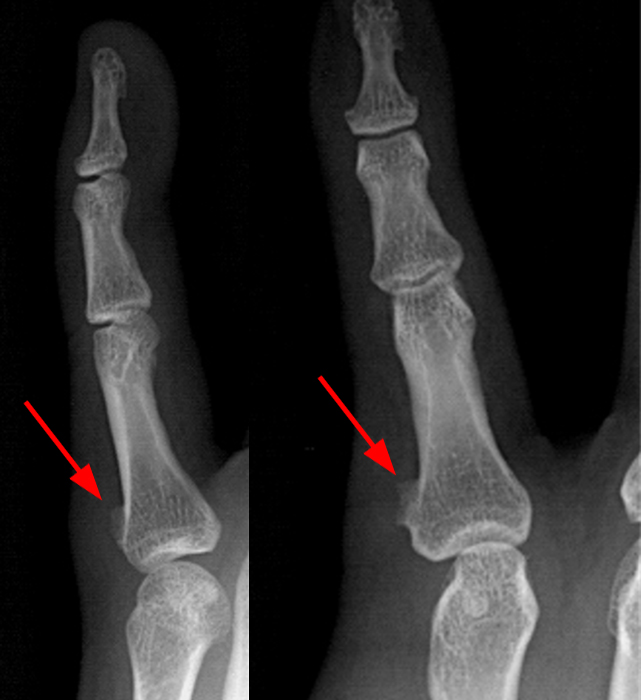
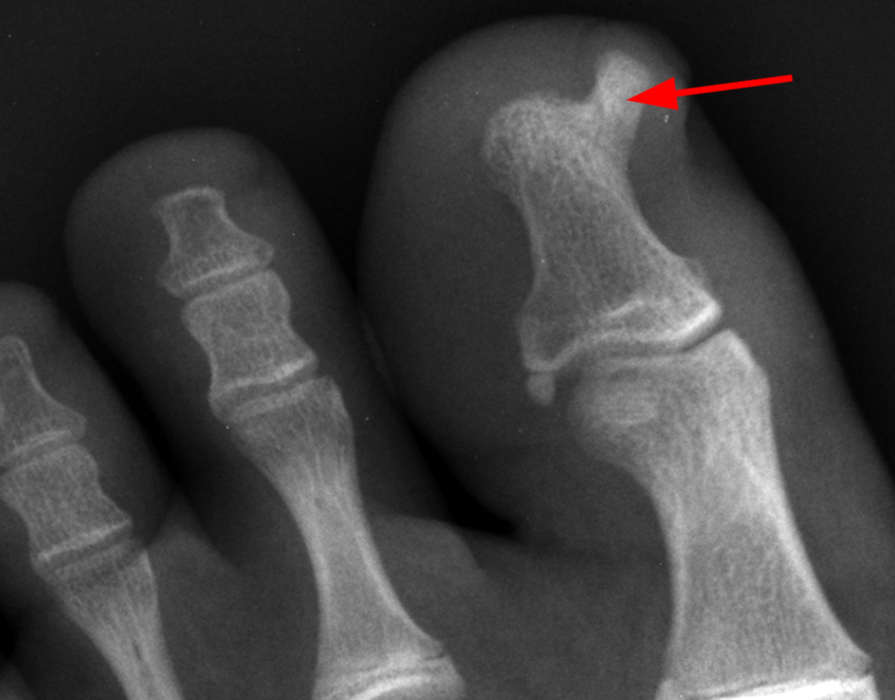
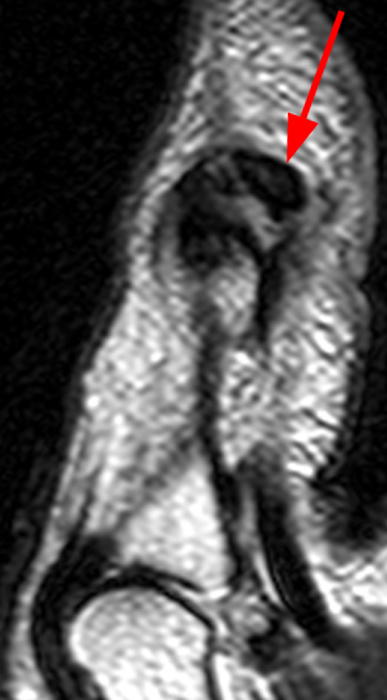
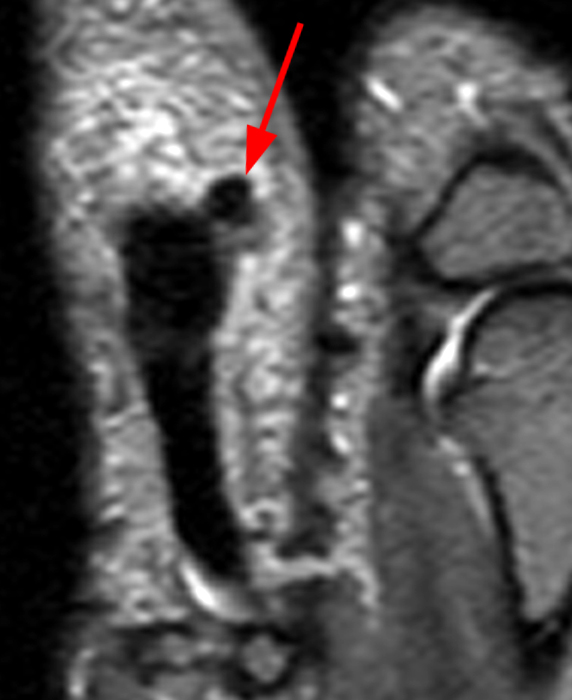
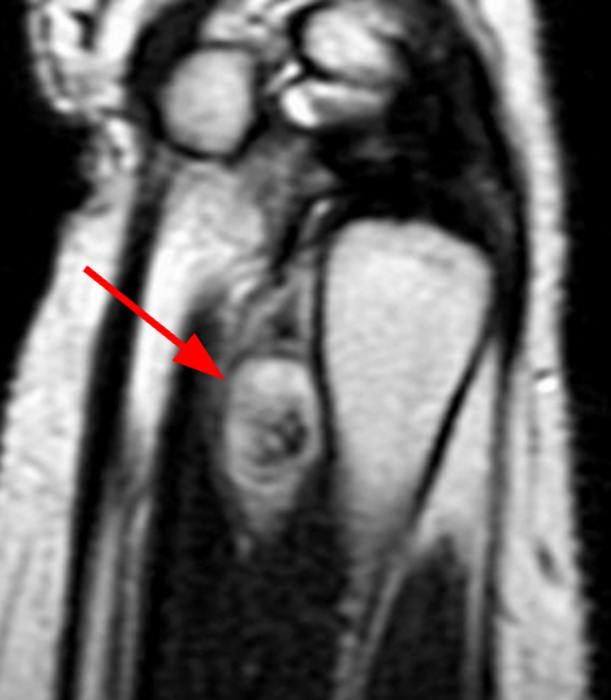
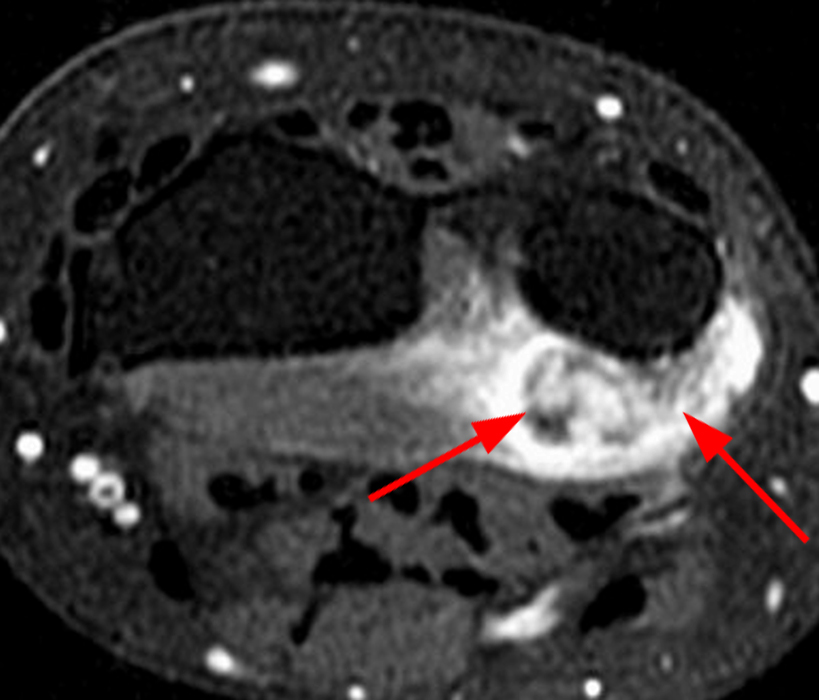
Clinical History: A 31-year-old male presents with a history of trauma two years ago and a one year history of an enlarging, mildly painful mass at the left long finger. T1-weighted (1A) and fat-suppressed proton density-weighted (1B) coronal images, a fat-suppressed proton density-weighted axial (1C) image, and an AP radiograph (1D) are provided. What are the findings? What is your diagnosis?
Findings
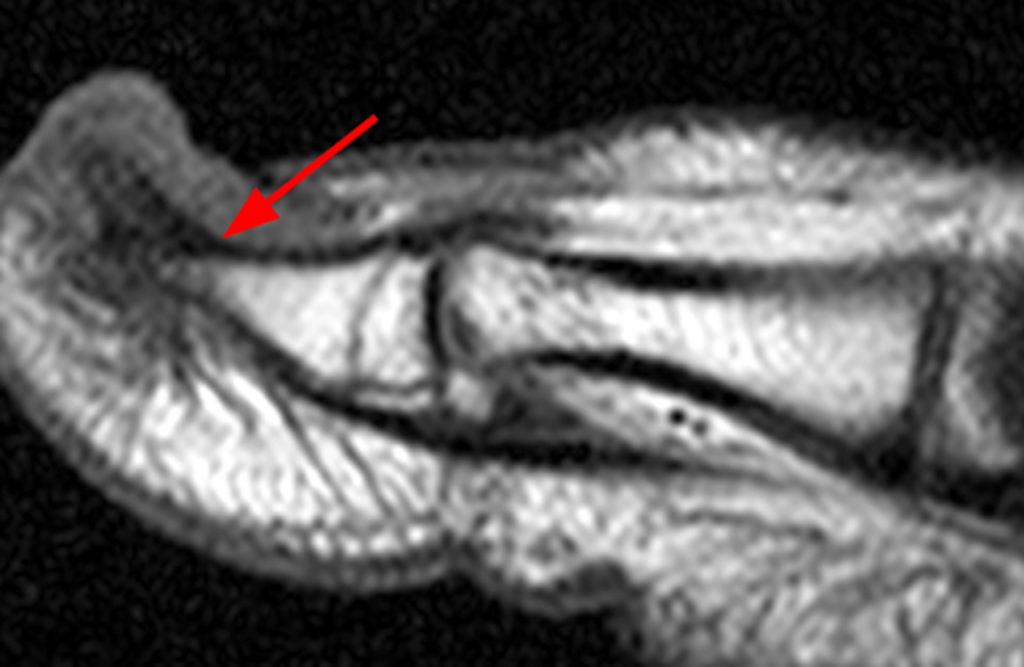
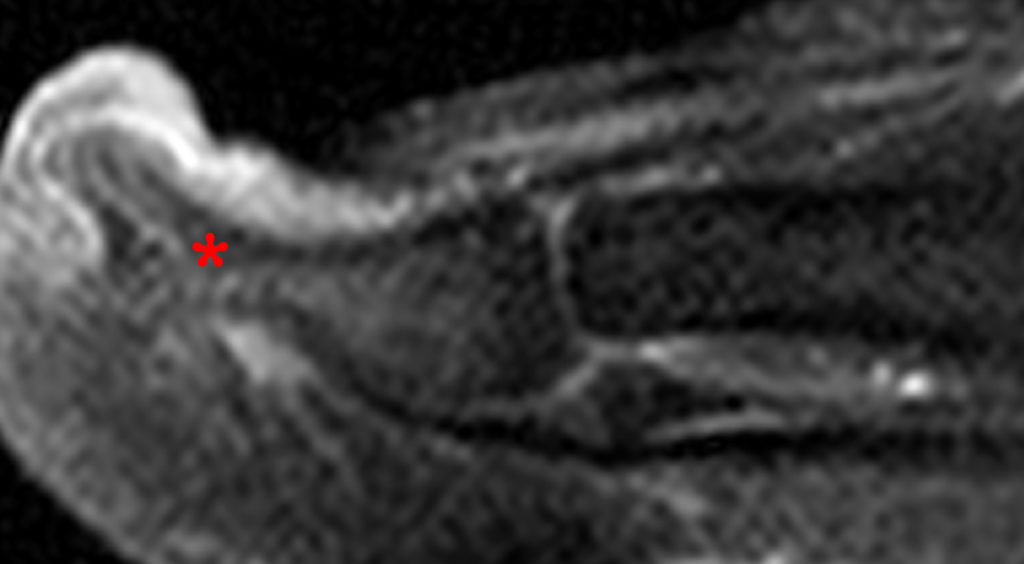
Figure 2:
A mass (arrows) is apparent at the ulnar aspect of the base of the proximal phalanx of the long finger on the T1-weighted and fat-suppressed proton density-weighted coronal and axial images (2A,2B,2C). The lesion is relatively isointense to muscle on T1-weighted images and is hyperintense on the fat-suppressed proton density-weighted images. A nodular region (asterisks) demonstrating higher signal intensity on the T1-weighted image and relatively lower signal intensity on the proton density-weighted images is present within the radial aspect of the mass. Ossification of this region is confirmed on the AP radiograph (arrow, 2D). The ossification is adjacent to cortex but demonstrates no cortical continuity or intramedullary communication.
Diagnosis
Bizarre Parosteal Osteochondromatous Proliferation (Nora’s Lesion).
Introduction
Bizarre parosteal osteochondromatous proliferation (BPOP) is a lesion first described by Nora et al. in 1983,1 and is thus also referred to as Nora’s lesion. The abnormality is relatively rare, with approximately 200 cases described in the literature.2 As a result, BPOP is unfamiliar to many radiologists and orthopaedic surgeons. It has similar features and clinical presentation to more common osteochondral tumors3,4, further exacerbating the challenge of making the correct diagnosis. Unlike other benign osteochondral masses, BPOP has a well-known propensity to recur, with some data indicating a greater than 50% chance of recurrence following resection.5 Accordingly, proper identification and diagnosis of BPOP is critical such that appropriate operative management is provided.
Demographics and etiology
BPOP is most commonly found along the surfaces of the proximal and middle phalanges of the metacarpals and metatarsals.6,7 However, it may also be found in long bones and has been reported in the mandible.8,9 Patients are most often in the third or fourth decade of life with the majority of studies noting an equal incidence between males and females. BPOP usually presents as a slowly growing mildly painful mass that enlarges over months or years.10 The etiology of BPOP remains uncertain. A history of trauma is present in up to half of cases, and it has been postulated that it is a reparative process following trauma to the periosteum. In a series of 24 proven cases of BPOP, 67% of cases were found in the right hand, supporting a possible traumatic etiology,11 and this study also demonstrated a higher frequency of BPOP in men, also thought to support a traumatic mechanism. Yuen et al. also proposed that BPOP has a traumatic etiology,12 and proposed a unitary hypothesis that suggests that BPOP is part of a continuum of disease that develops as a reaction to an initial, often traumatic stimulus. They postulated that the process begins with florid reactive periostitis, which then organizes into BPOP, and that the end stage is maturation into a turret osteochondroma. Dorman supports this contention and has described pathologic confirmation of progression of florid reactive periostitis to BPOP.13 Additional studies have demonstrated the expression of growth factors in the cartilaginous component of BPOP, suggesting that it is a reparative process following periosteal injury.14 However, it is apparent that many cases of BPOP do not report antecedent trauma. Further, consistent chromosomal rearrangements including a balanced translocation have been found in BPOP, and the specific translocation in BPOP is unique to this lesion.15,16 The known fact that BPOP has a remarkable tendency to recur following resection supports a neoplastic process and is difficult to explain if one presumes that BPOP is a reaction to trauma.
Imaging of Nora’s Lesion/BPOP
In patients in whom serial radiographs are available, the natural evolution of BPOP may be visualized. In the early stage, periosteal soft-tissue swelling is evident, and calcification may be absent or minimal. Calcifications then become more prominent and evolve to mature ossification in typically less than 6 months (Figure 3). The lesion tends to be of intermediate signal intensity on T1-weighted images, similar to muscle, and relatively hyperintense on fat-suppressed T2- or proton density-weighted images. If contrast is administered, BPOP typically demonstrates marked enhancement (Figure 4).
Figure 3:
A 16-year-old female presents with a history of trauma 3 months ago with an enlarging painful mass at the right small finger. The large mass is readily apparent on (3A) the photograph of the patient’s hand. Initial radiographs (3B) reveal swelling (arrows) about the proximal phalanx and PIP joint of the small finger with faint calcifications. On follow-up radiographs approximately 5 weeks later (3C), the mass (arrows) demonstrates considerably more calcification. A sagittal CT reconstruction (3D) confirms a partially calcified lesion (arrow) that abuts, but does not extend into, the dorsal cortex. A fat-suppressed axial T2-weighted image (3E) through the lesion (arrows) demonstrates a hyperintense mass without extension into cortex or the medullary space (asterisk). Images courtesy of Donald Resnick, MD, University of California San Diego.
Figure 4:
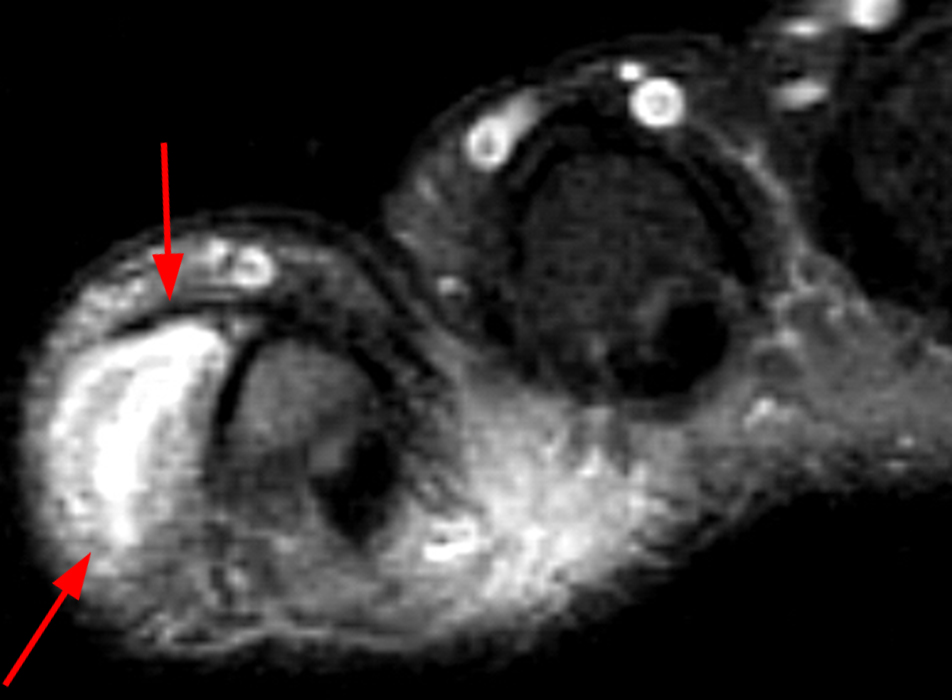
An oblique radiograph at initial presentation (left 4A) and an AP radiograph one year later (right 4A) demonstrate a mild increase in size of a calcified lesion (arrows) at the radial aspect of the proximal phalanx of the index finger. The lesion (arrows) is relatively isointense to muscle on the T1-weighted coronal image (4B), hyperintense on the fat-suppressed T2-weighted axial image (4C), and demonstrates intense enhancement on the post-contrast fat-suppressed T1-weighted coronal view (4D). BPOP was confirmed at surgery. Images courtesy of Patcharee Hongsmatip, MD, Bumrungrad International Hospital, Thailand.
Differential Diagnosis
Given its parosteal location, BPOP must be distinguished from parosteal osteosarcoma and chondrosarcoma, which are extremely rare in the hands and feet. Features of such malignancies such as cortical flaring, cortical destruction, periosteal reaction, and soft tissue invasion are not found with BPOP. Histologically, malignant features of sarcomas are absent in BPOP. Because of its surface location and pattern of ossification, BPOP may be mistaken for an osteochondroma. Osteochondromas are also rare in the phalanges of the distal extremities. Although Rybak et al. described a group of four patients with BPOP demonstrating cortico-medullary continuity,17 the vast majority of the literature has indicated that the absence of such communication is a critical feature of BPOP. A related entity to osteochondromas, the subungual exostosis, is a distal extremity lesion, but unlike BPOP it typically involves the dorsal aspect of the distal phalanx and direct continuity to bone is present in these lesions (Figure 5). The cartilaginous cap that is a feature in the majority of osteochondromas is an inconsistent feature in BPOP.
Figure 5:
An oblique AP radiograph (5A) in a 14-year-old male with pathologically proven subungual exostosis reveals an ossification at the dorsomedial aspect of the distal phalanx of the great toe (arrow), demonstrating continuity with adjacent cortex and the medullary space. On the corresponding T1-weighted sagittal MR image (5B), the lesion (arrow) is inseparable from dorsal cortex. On the fat-suppressed proton density-weighted sagittal view (5C), continuity of the lesion with the medullary canal (asterisk) is apparent.
Figure 6:
A 58-year-old female presents with a one week history of pain at the IP joint of the thumb with no prior trauma. An oblique radiograph (6A) demonstrates an ovoid periarticular calcification (arrow) at the palmar/ulnar aspect of the base of the distal phalanx of the thumb. Similar to BPOP, the abnormality is contiguous with cortex without intramedullary communication. T2-weighted sagittal (6B) and STIR coronal (6C) MR images reveal a signal void nodule (arrows) compatible with calcification in the area of interest. Surrounding soft tissue edema is present on the STIR image and there is no soft-tissue component. The findings are compatible with HADD/calcific periarthritis.
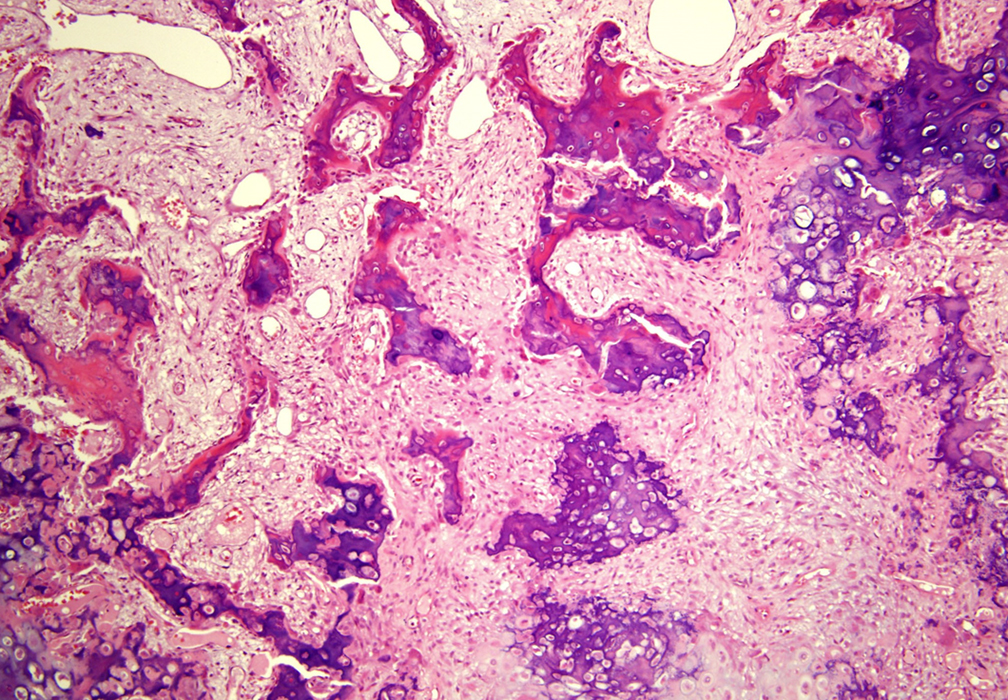
Pathological analysis
Grossly, BPOP appears as an exophytic mass without continuity with the underlying medullary cavity, containing bone and cartilage. Histologically, disorganized architecture is present with aggregates of cartilage, new bone, and fibrous tissue. A characteristic histologic finding in cases of BPOP is the presence of “blue bone” on H and E staining. Meneses first described this appearance as a “distinct blue tinctorial characteristic” and this has been utilized as a helpful feature for the diagnosis of this entity (Figure 7). The cartilaginous components of the lesion demonstrate hypercellularity with groups of binucleated and bizarre chondrocytes. Hyperchromasia and cytological atypia are not present.18
Treatment
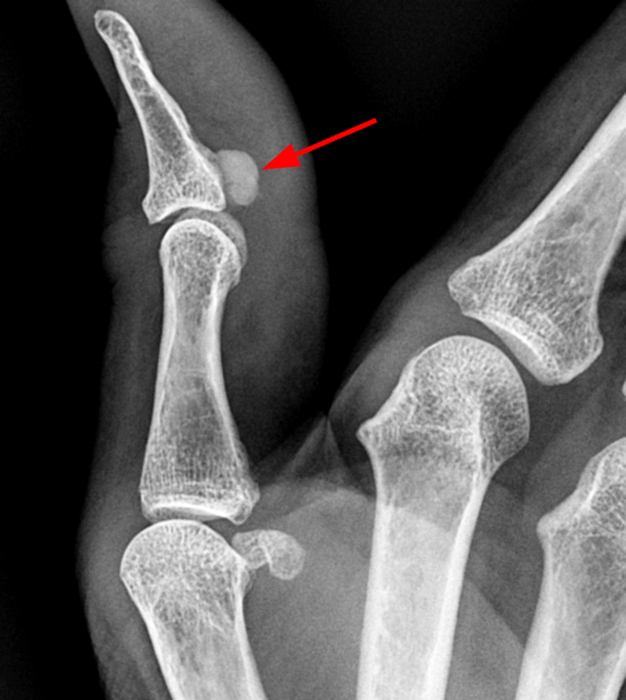
Initial treatment for BPOP usually consists of excision of the pseudocapsule and periosteal tissue with decortication of associated osseous abnormalities. BPOP is notorious for recurrence following local resection (Figure 8), with recurrence rates reported at between 35 and 54% at two years.19 Limited excision has a greater chance of recurrence than en bloc resection, but preserves stability and avoids amputation of the affected bone. Though recurrence is relatively common with limited excisions, there have been no reports of malignant transformation, metastases, or death in patients with BPOP. Therefore, repeat excision is considered preferable to more aggressive initial surgery that results in greater morbidity.20 The MR findings in patients with recurrent BPOP are similar to those seen in the initial presentation (Figure 8).
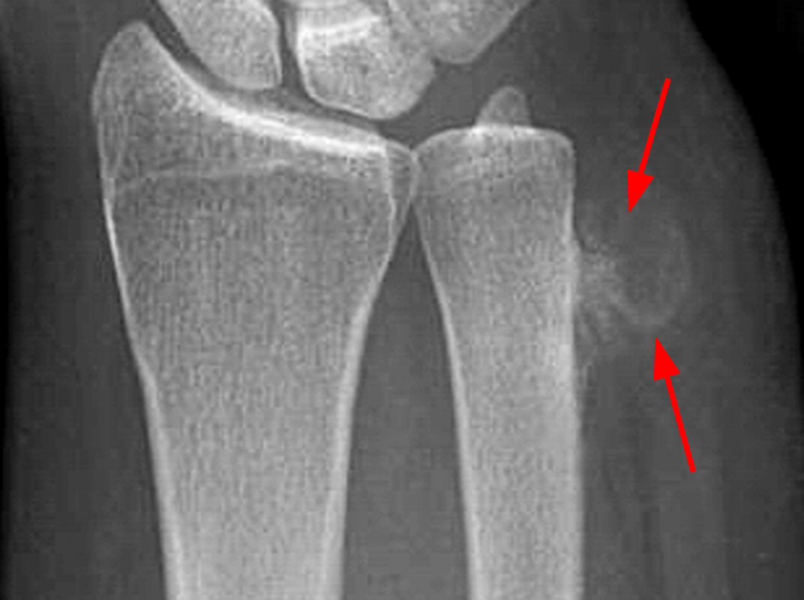
Figure 8:
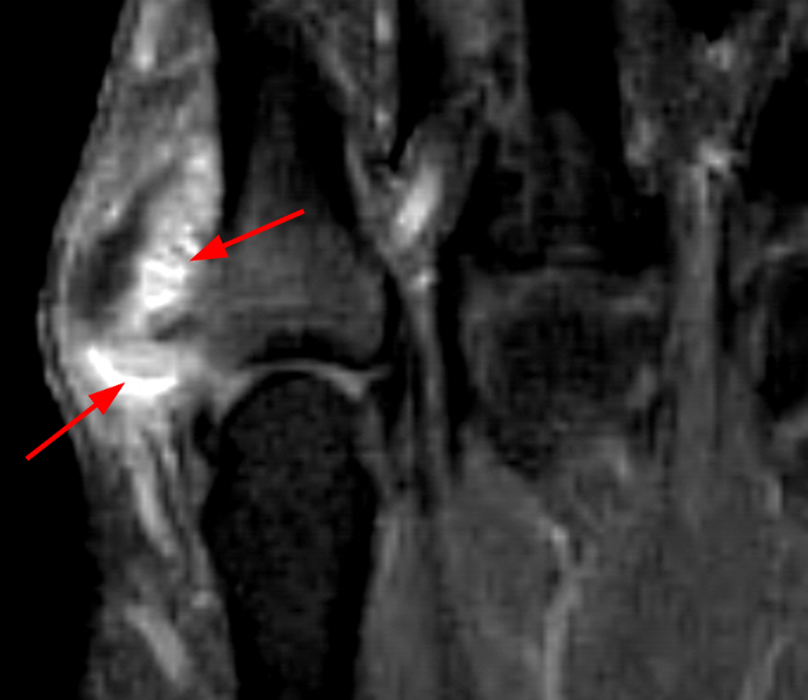
A 35-year-old female presents with a distal forearm mass with a corresponding calcification (arrow) being apparent on the initial radiograph (8A). The patient underwent local excision of the lesion, found to be BPOP on pathology. One year later the patient returned with a recurrent mass with corresponding calcification (arrow) on an AP radiograph (8B). A T2-weighted sagittal image (8C) demonstrates the recurrent ovoid mass (arrow) palmar to the distal radius. The lesion (arrows) demonstrates intrinsic and peripheral enhancement on the post-contrast fat-suppressed T1-weighted image (8D). There is no involvement of adjacent cortex or the medullary canal. Images courtesy of David A. Rubin, MD, Radsource.
Conclusion
Bizarre parosteal osteochondromatous proliferation (BPOP), also known as Nora’s lesion, is a relatively rare ossifying abnormality classically found along the proximal aspect of the fingers or toes. It is unfamiliar to many radiologists and as a result may be underdiagnosed. The etiology of BPOP is uncertain, with some authors considering it part of a continuum of post-traumatic disease that begins with florid periostitis, progresses to BPOP, and then matures into turret osteochondroma. But other authors note the lack of trauma in many cases and the presence of a chromosomal translocation indicating a neoplastic etiology. Indeed, the known high recurrence rate of BPOP is difficult to explain for a post-traumatic entity. MRI is useful for identifying the typical appearance of BPOP and to assess regional anatomy prior to resection. Importantly, the lack of direct continuity of the medullary space or cortical involvement are thought to be the key to differentiating BPOP from rare distal extremity osteochondromas. Aggressive features associated with malignancy are not found with BPOP, and the histologic findings are characteristic. Although BPOP has a high propensity to recur, it is not associated with any reports of metastatic disease or death.
References
- Nora FE, Dahlin DC, Beabout JW. Bizarre parosteal osteochondromatous proliferations of the hands and feet. Am J Surg Pathol 1983;7:245-50. ↩
- Mustafa A, Al-Zoubi A, Aldhoon M et al. Atypical Radiological Presentation of a Bizarre Parosteal Osteochondromatous Proliferation (Nora’s Lesion) Affecting the Index Finger: A Case Report. JRMS Aug 2017; 24(2):79-82. ↩
- Oviedo A, Simmons T, Benya E, Gonzales-Crussi F. Bizarre parosteal osteochondromatous proliferation: Case report and review of the literature. Ped Path 2001;4:496. ↩
- Brien EW, Mirra JM, Luck JV. Benign and malignant cartilage tumors of bone and joint: Their anatomic and theoretical basis with an emphasis on radiology, pathology and clinical biology. ↩
- Michelsen H, Abramovici L, Steiner G, Posner MA. Bizarre parosteal osteochondromatous proliferation (Nora’s lesion) in the hand. J Hand Surg Am 2004;29:520–525. ↩
- Gruber G, Glessauf C, Leithner A. Bizarre parosteal osteochondromatous proliferation (Nora’s lesion): a report of 3 cases and a review of literature. Can J Surg 2008; 51:486-489. ↩
- Davies CWT. Bizarre parosteal osteochondromatous proliferation in the hand: A case report. J Bone Joint Surg (Am). April 1985; 67-A(4):648-50. ↩
- Abramovici L, Steiner GC. Bizarre parosteal osteochondromatous proliferation (Nora’s lesion): A retrospective study of 12 cases, 2 arising in long bones. Hum Pathol 2002;33:1205. ↩
- Meneses MF, Unni KK, Swee RG. Bizarre parosteal osteochondromatous proliferation of bone (Nora’s lesion). Am J Surg Pathol 1993;17:691. ↩
- Bandiera S, Bacchini P, Bertoni F. Bizarre osteochondromatous proliferation of bone. Skeletal Radiol 1998;27:154. ↩
- Dhondt E, Oudenhoven L, Khan S et al. Nora’s lesion, a distinct radiological entity? Skeletal Radiol (2006) 35: 497–502. ↩
- Yuen M, Friedman L, Orr W, Cockshott WP. Proliferative periosteal process of phalanges: a unitary hypothesis. Skeletal Radiol (1992) 21:301 303. ↩
- Dorfman HD, Czerniak B. Bone tumors. St. Louis (MO):Mosby, 1998. ↩
- Horiguchi H, Sakane M, Matsui M. Bizarre parosteal osteochondromatous proliferation (Nora’s lesion) of the foot. Pathol Int 2001; 51:816-23. ↩
- Zambrano E, Nose V, Perez-Atayde AR. Distinct chromosomal rearrangements in subungual (Dupuytren) exostosis and bizarre parosteal osteochondromatous proliferation (Nora lesion). Am J Surg Pathol. 2004; 28:1033-9. ↩
- Nilsson M, Domanski HA, Mertens F. Molecular cytogenetic characterization of recurrent translocation breakpoints in bizarre parosteal osteochondromatous proliferation (Nora’s lesion). Hum Pathol 2004; 35:1063-9. ↩
- Rybak LD, Abramovici L, Kenan S. Cortico-medullary continuity in bizarre parosteal osteochondromatous proliferation mimicking osteochondroma on imaging. ↩
- Michelsen H, Abramovici L, Steiner G, Posner MA. Bizarre parosteal osteochondromatous proliferation (Nora’s lesion) in the hand. J Hand Surg 2004;29A:520. ↩
- Rosenberg L. Chemical basis for histological use of Safranin O in the study of articular cartilage. J Bone Joint Surg 1971;A53:69–82. ↩
- Torreggiani WC, Munk PL, Al-Ismail K et al. MR imaging features of bizarre parosteal osteochondromatous proliferation of bone (Nora’s lesion). Eur J Radiol 2001 ; 40 : 224-231. ↩