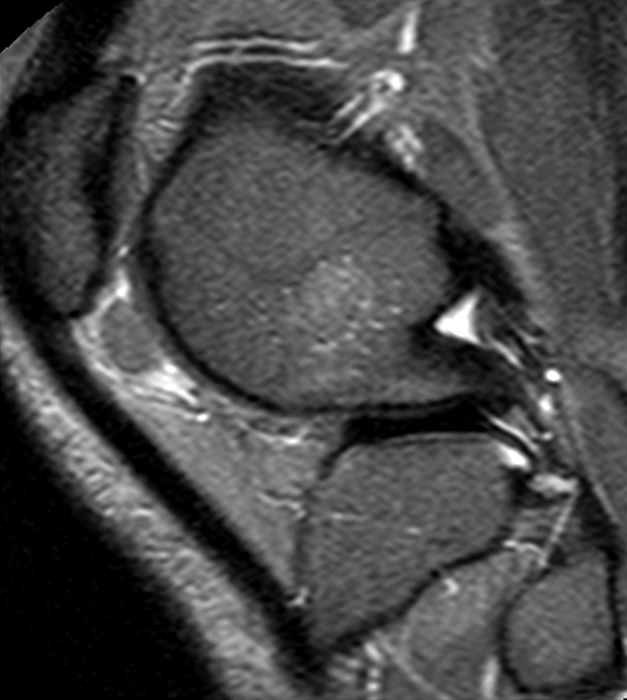
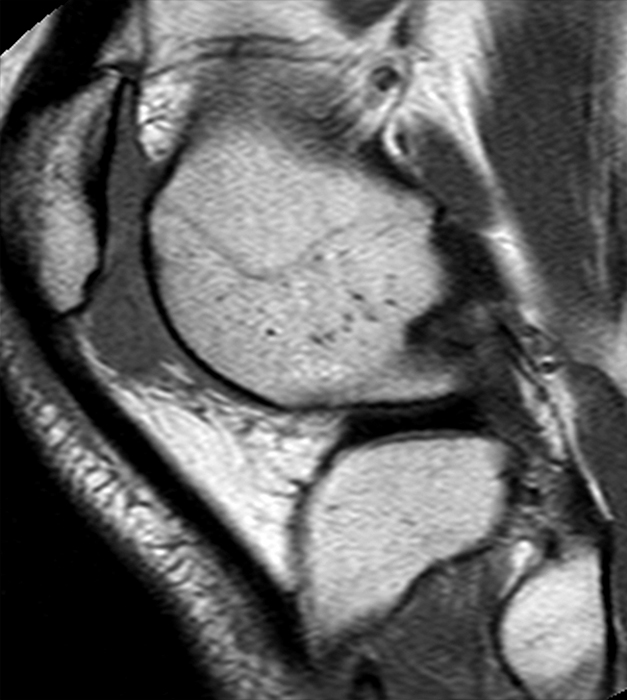
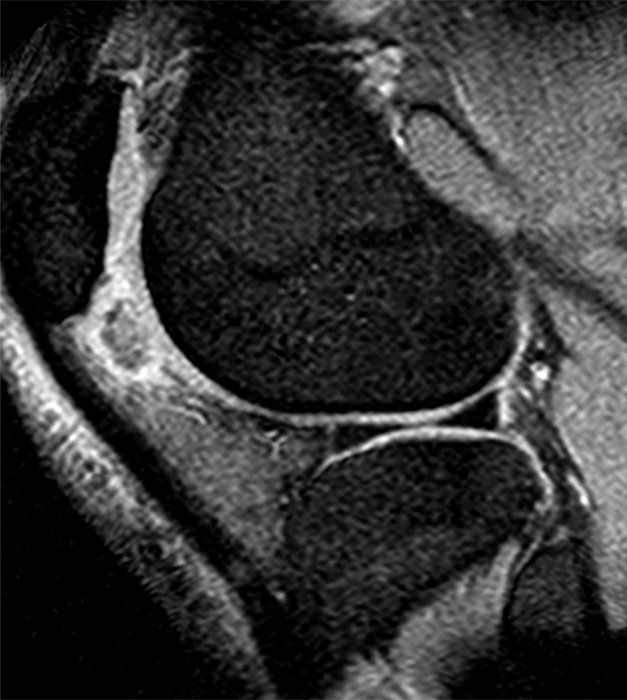
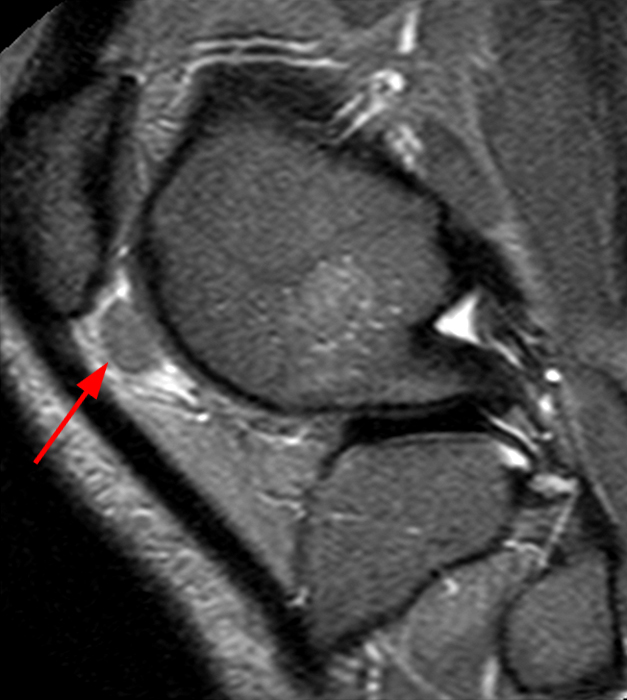
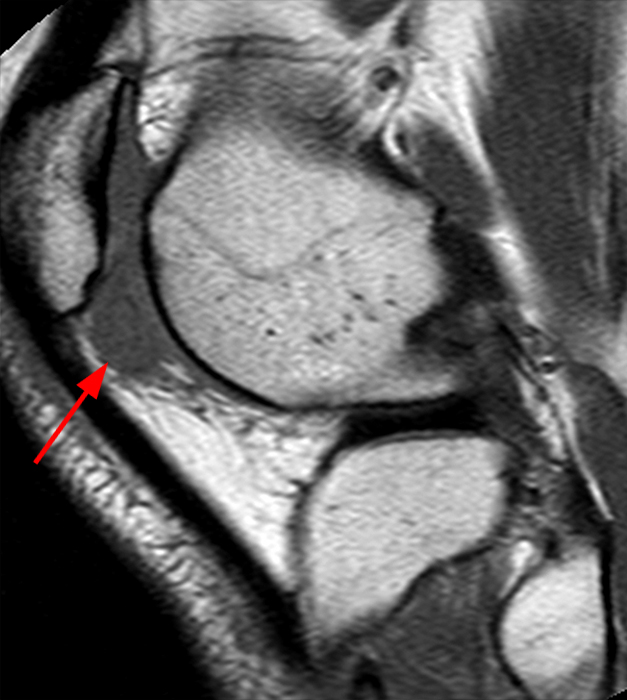
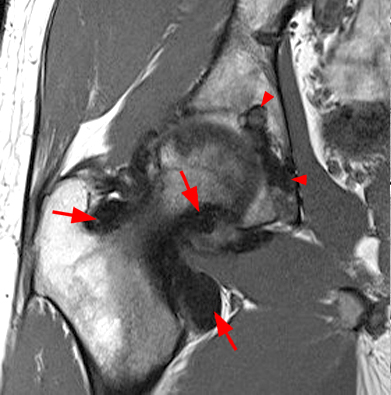
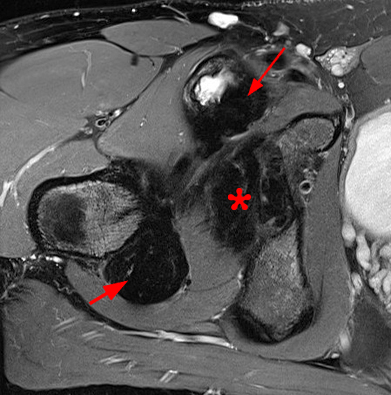
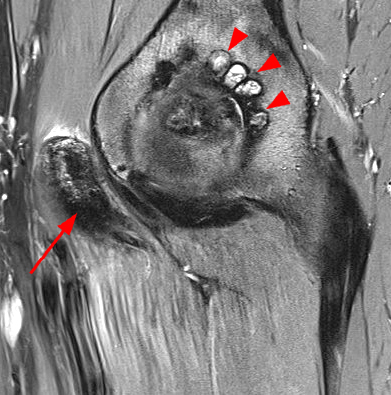
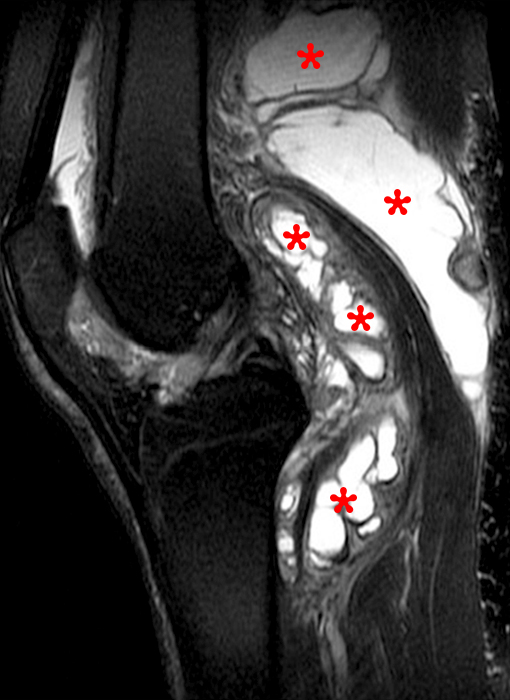
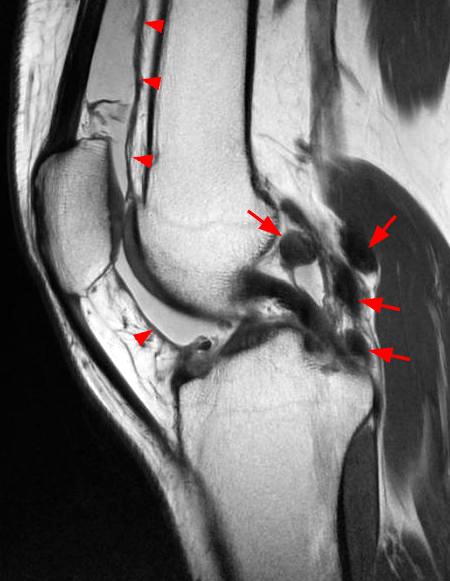
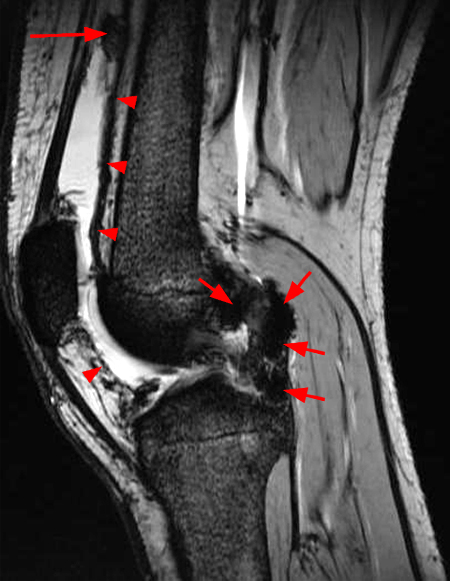
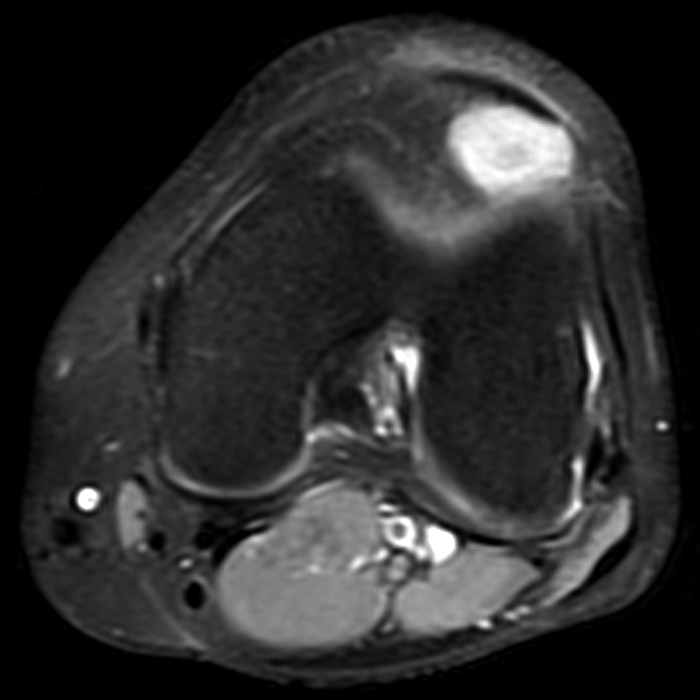
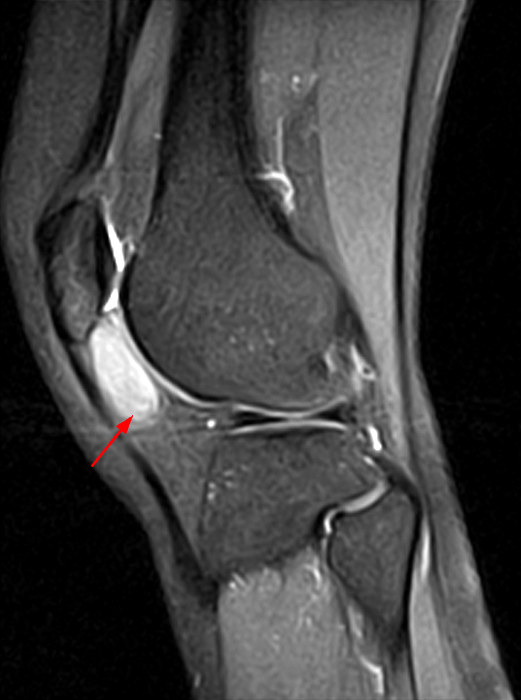
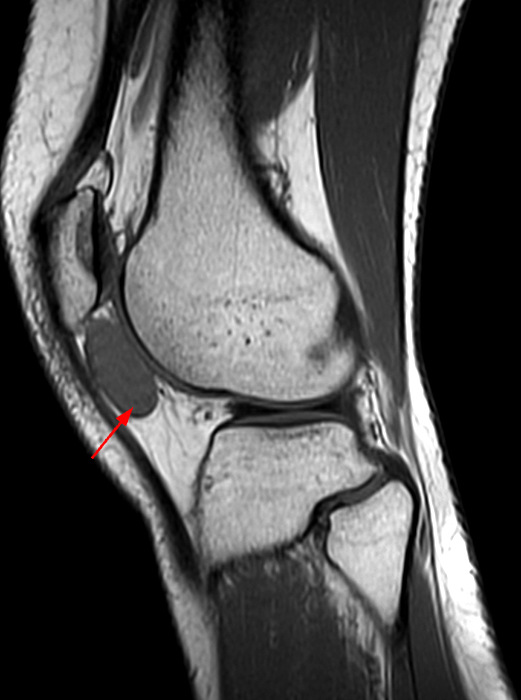
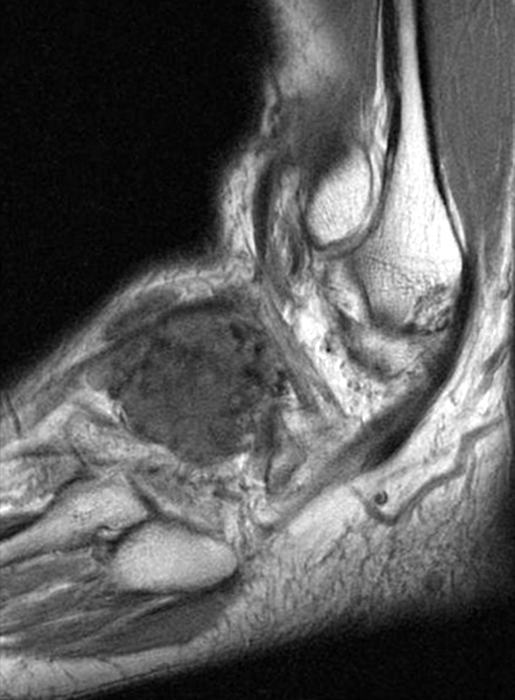
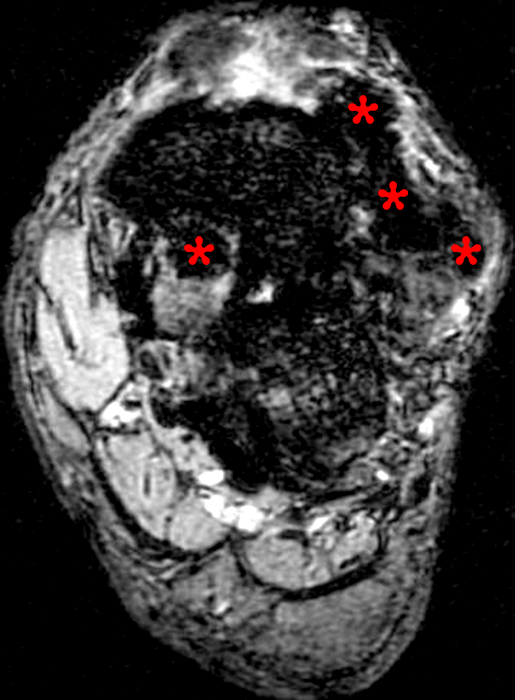
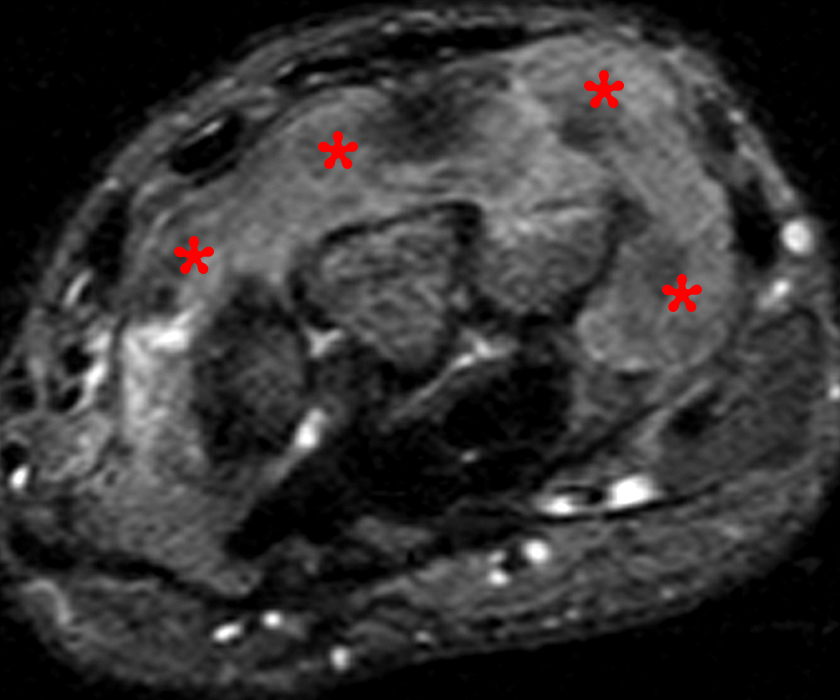
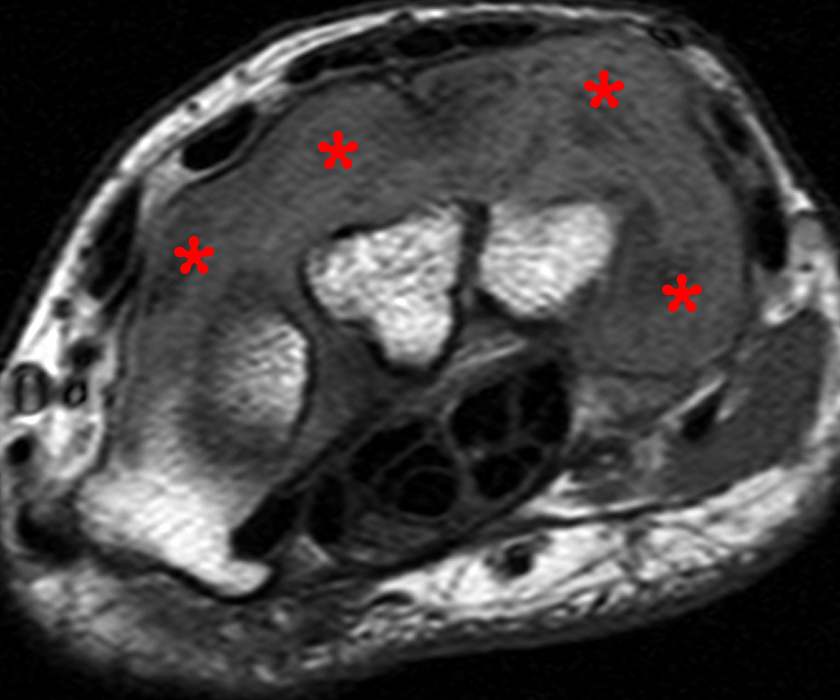
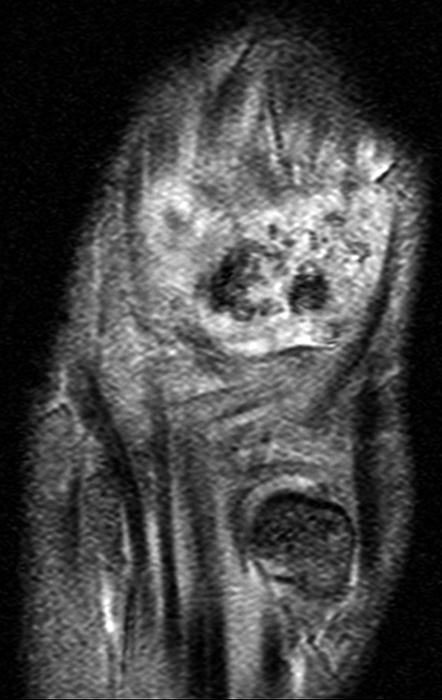
Clinical History: A 21 year-old female complains of pain, swelling, and stiffness in the knee anteriorly, status post MVA 4 months ago. (1A,1B,1C) Sagittal STIR, T1-weighted, and gradient echo sequences are provided. What are the findings? What is your diagnosis?
Findings
Figure 2:
Sagittal STIR (2A), T1-weighted (2B), and gradient echo (2C) images of the knee demonstrate a round 1.2 cm mass or mass-like structure in the anterior compartment (arrows). It is mildly heterogeneous, predominantly isointense to soft tissue on the STIR and T1-weighted sequences with very small foci of slightly low signal. Note the increased prominence of the low signal on the gradient echo sequence (2C), reflecting the presence of hemosiderin. Mass-like signal in the anterior compartment in a patient of this age that displays internal hemosiderin makes localized PVNS the favored diagnosis. Note the absence of joint effusion and nonspecific edema or edema-like signal about the PVNS in 2A. Incidentally noted is patella alta.
Diagnosis
Localized pigmented villonodular synovitis (PVNS), also referred to as localized nodular synovitis.
Introduction
PVNS is a proliferative synovial process characterized by hemosiderin deposition of varying degree. On histopathologic analysis, there is synovial proliferation, hemosiderin- and lipid-laden macrophages, as well as multinucleated giant cells and stromal and fibroblast cell proliferation.1,2 PVNS may involve a joint, bursa, or tendon sheath; when it involves a tendon sheath it is typically called giant cell tumor of the tendon sheath (GCTTS) or, less commonly, nodular tenosynovitis. In 2013 the World Health Organization reclassified all forms of PVNS as tenosynovial giant cell tumor.3 This discussion is solely focused on the process involving joints and bursae and will continue to be described as PVNS.
All forms of PVNS are felt to be a neoplastic process as there is often an overexpression of colony-stimulating factor 1 (CSF1) frequently due to a specific chromosomal translocation, overexpression of colony-stimulating factor 1 receptor (CSF1R), as well as the inconsistent presence of Trisomy 5 and 7 and extremely rare cases of malignant disease.1,3,4,5,6,7
PVNS is nearly always a monoarticular process, whether the localized or diffuse form. It most frequently affects patients in their twenties to forties.6 Males and females are affected with equal frequency. Diffuse disease usually affects large joints (knee, hip, ankle, shoulder, and elbow in decreasing order of frequency); the knee is affected in the majority (66-80%) of patients.6 The localized form of PVNS almost always involves the knee.6
The diffuse form of PVNS involves the vast majority or the entire synovium. The localized form manifests as a pedunculated or lobular mass localized to one area of synovium; in the knee, it most frequently occurs in the anterior compartment.2
Not unexpectedly, there are exceptions to the “rule”, or outliers. As a synovial process, any joint or synovial-containing structure (e.g. bursae, synovial cysts) can be affected by PVNS. For example, it can involve — rarely — the apophyseal joints in the spine, the temporomandibular joint, the sacroiliac joints, and (isolated involvement of a) bursa.1,6,8,9,10 Involvement of the spine can manifest as pain, myelopathy, and/or radiculopathy and erode, expand, and/or destroy adjacent bone, including and limited to vertebral bodies, and involve the adjacent soft tissues and often the extradural space which can simulate both benign and malignant lesions.1,8 PVNS can occur at any age; uncommonly, both localized and diffuse forms can occur in children (e.g. toddlers)11,12 and the elderly. Not surprisingly, given its rarity in pediatric patients, the diagnosis is often delayed and misdiagnosed as another arthropathy such as juvenile idiopathic arthritis.11,12 Additionally, there have been rare cases involving multiple different joints, including the contralateral joint.7,13,14 It rarely can be seen or recur after total joint replacement.15,16
While it is overwhelmingly benign, there have been rare cases (less than 40 cases) of malignant disease, an aggressive sarcoma that can metastasize typically to bone, lung, and regional lymph nodes and occurring either in newly diagnosed or recurrent PVNS.6,17 The average age of patients affected is in the fifth or sixth decade, although malignant disease has been seen in a 12 year-old patient.6,17 Extensive bone invasion and destruction as well as extensive soft tissue invasion have been identified as suggestive of malignant disease.6,17,18
Clinical Presentation
The most frequent symptoms are pain and swelling.1,6,12 Usually poorly localized, pain and swelling are more pronounced in the diffuse form.2 Stiffness/limited range of motion, warmth, and tenderness may be seen.2,5 A soft tissue mass can occasionally be palpated.6 The localized form, while also presenting with pain and swelling, more often presents with mechanical symptoms (e.g. locking, catching, instability).2,12
Joint effusions are more common in the setting of diffuse PVNS. Aspirated fluid can be serosanguinous or blood-tinged.2,12 Blood-tinged fluid can suggest the diagnosis but is nonspecific.2 It is common for aspirated joint fluid cytology to be normal.19
Symptoms of PVNS are often intermittent and are nonspecific, resulting in a lengthy time to diagnosis, often many years.1,2,6,14 The symptoms can be attributed to other conditions (e.g. early osteoarthritis, rheumatoid arthritis, meniscus tear).2
Imaging Findings
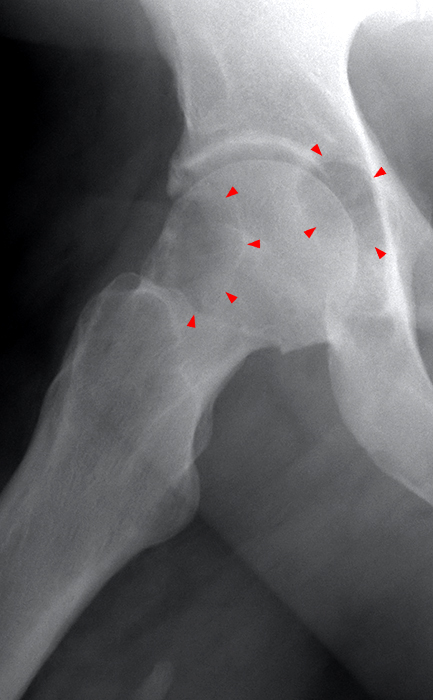
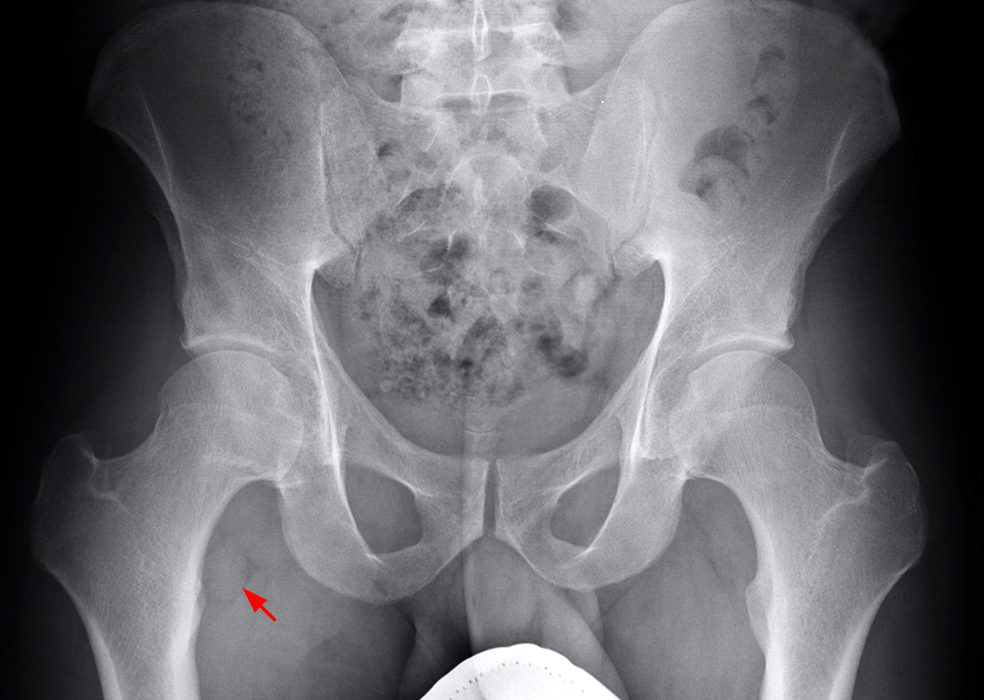
Radiographs are typically nonspecific in PVNS. Bone demineralization is not a feature of PVNS. Joint spaces are typically preserved, although diffuse PVNS can damage cartilage over time with associated joint space narrowing. Unlike typical primary osteoarthritis, osteophytes are unusual. Subchondral cysts/cyst-like areas and/or marginal erosions may be seen in diffuse PVNS, typically with thin marginal sclerosis. Although subchondral cystic lucencies can be seen in the knee, they are less common than in other joints due to the knee’s capacious capsule. The lucencies are more common and indeed may be suggestive of PVNS when seen in other joints, particularly in the hip in the setting of a preserved joint space and when present on both sides of the joint (Figures 3A and 3B). Additionally, mass-like areas which may be relatively increased in density, can be seen in and about joints (Figures 4 and 5D). While mineralization of PVNS has been reported6 it is extremely rare; if mineralization is seen, another process besides PVNS should be considered.
Figure 3:
AP (3A) and frog-leg lateral (3B) radiographs demonstrate a large lucency spanning the femoral-head neck junction and in the acetabulum (arrowheads) with normal joint space and normal bone mineralization. The lucency in the femur has a thin sclerotic margin. Axial T1-weighted image (3C) shows a large bilobed area of heterogeneous signal with partial sclerotic margin in the medial acetabulum (short arrows), corresponding to the lucency on radiographs; it contains small foci of low signal and is similar in signal to the abnormality markedly expanding the iliopsoas bursa (long arrows), reflecting foci of PVNS. Axial fat-suppressed T2-weighted image (3D) shows a subcortical area of homogeneous high signal and thin sclerotic rim spanning the femoral head-neck junction (asterisk), corresponding to the lucency on radiographs; note the extensive heterogeneous signal, much of it hypointense, expanding the iliopsoas bursa (long arrows) and extensive foci of similar signal in the joint (short arrows), representing diffuse PVNS.
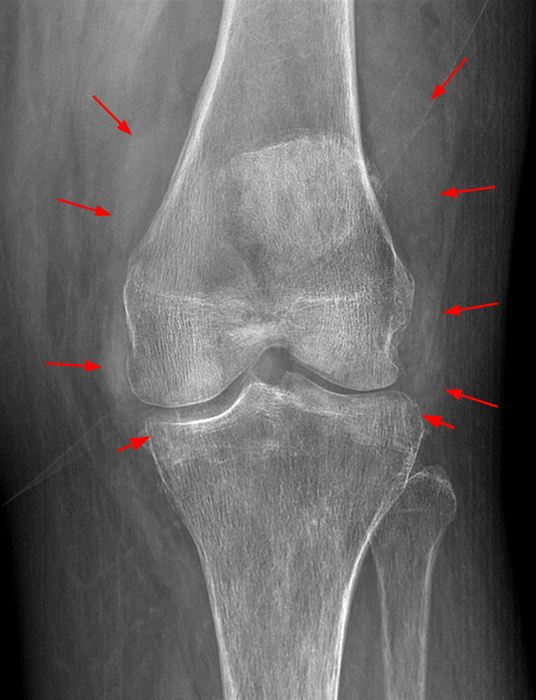
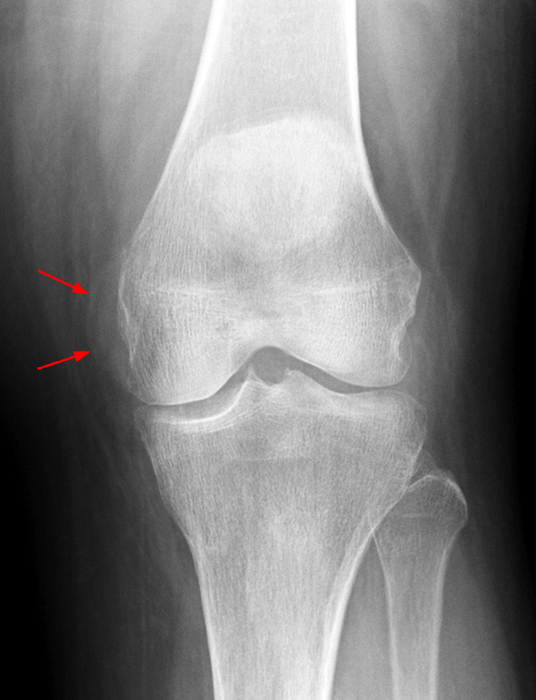
Figure 4:
69 year-old female with diffuse PVNS. AP (4A) and lateral (4B) radiographs demonstrate multiple mass-like, relatively dense periarticular and intra-articular foci (arrows), as well as marginal lucencies with thin sclerotic margins (short arrows), representing diffuse PVNS with foci of marginal intraosseous involvement. The large lobular density posteriorly in 4B represents PVNS filling a popliteal cyst as was seen on MRI (not shown). The joint spaces are preserved. Diffuse bone demineralization, not a feature of PVNS, is incidental in this elderly woman. AP radiograph of the patient’s knee 5 ½ years earlier shows prominent curvilinear opacity projecting along the medial femoral condyle (arrows), similar to that in 4A, suggesting the presence of PVNS years earlier; it is common for many years to elapse before the diagnosis of PVNS is made.
On CT, PVNS is isodense to slightly hyperdense to muscle, and synovial thickening can be seen.6 Subchondral cystic foci and/or erosions, if present, are well seen on CT. As most cases of PVNS are vascular, they can be seen with increased uptake on bone scan; uptake is more prominent on blood flow and blood pool images of three-phase bone scans than on delayed images.6 Additionally, PVNS can display hypermetabolic activity on FDG-PET with high standard uptake values which can be mistaken for malignant disease.6,14,20
The diagnosis of PVNS often can be made or strongly suggested on MRI. Heterogeneous signal is nearly always seen, usually a mixture of intermediate and low signal on fluid-sensitive and T1-weighted sequences, the low signal proportionate to the degree of hemosiderin. GRE sequences can be helpful, exaggerating hemosiderin’s paramagnetic effect (so-called low signal blooming). Hemosiderin is accentuated on sequences with longer repetition times and on higher-strength magnets. Infrequently, foci of high T1 signal have been reported; Murphey et al. believe this is likely entrapped perisynovial fat, particularly as fat is not found on histopathology.6 Although nonspecific and thus gadolinium is not recommended for evaluation, PVNS usually enhances heterogeneously.
Diffuse PVNS manifests as extensive thickening of the synovium with irregularly shaped mass-like foci of various sizes which are often scattered in various locations in the joint as well as joint effusion of variable size; it is not uncommon for it to extend into the extra-articular soft tissues through pericapsular ganglia as well as into bursae, at times marked (Figure 6). There may be subchondral cystic foci, portions often containing signal similar to that in the joint (representing frank intraosseous PVNS extension), as well as marginal erosions with adjacent mass-like signal. Particular note should be made of involvement of bursae and sites of extracapsular extension as this will usually necessitate at least a partial open approach for resection. If extracapsular extension is present, it should be noted if neurovascular structures are involved.
Localized PVNS, almost always occurring in the knee and most frequently seen in the anterior compartment, manifests as an irregularly shaped or well-circumscribed, rounded mass-like focus of signal (Figures 1,2,8). Unlike diffuse PVNS, the localized form may be only mildly heterogeneous and largely hyperintense on fluid-sensitive sequences (Figure 8). Effusions are often absent in the localized form.
Figure 5:
Coronal proton density-weighted image (5A) shows marked low signal about the femoral neck extending distally near the lesser trochanter (short arrows) as well as similar low signal in the acetabular fossa with adjacent intraosseous extension (arrowheads). Axial fat-suppressed proton density-weighted image shows similar extensive very low signal in the iliopsoas bursa (arrow) and in and distending a posteroinferior recess (short arrow) as well as similar low signal further medially (asterisk). Multiple small subchondral cystic foci containing thin curvilinear intermediate and low signal foci in the posterior acetabulum (arrowheads) and extensive heterogeneous low signal in the iliopsoas bursa (arrow) are demonstrated on the sagittal fat-suppressed proton density-weighted image (5C). While entirely nonspecific, AP pelvis radiograph (5D) reveal an asymmetric subtle round focus of density near the lesser trochanter (arrow), corresponding to the low signal as seen in 5A in this case of diffuse PVNS. This case illustrates that PVNS can manifest as very low signal, corresponding to extensive hemosiderin deposition.
Figure 6:
Sagittal fat-suppressed T2-weighted image (6A) and gradient echo image (6B) demonstrate marked posterior extracapsular extension of diffuse PVNS in very large extracapsular ganglia (asterisks). Note the accentuation of hemosiderin as “blooming” of low signal in numerous locations on the gradient echo sequence, including an extensive area adjacent to the posterior femoral metadiaphysis (arrow in 6B). It is critical to describe the extent of extracapsular extension and involvement of bursae and neurovascular structures for surgical planning.
Figure 7:
Sagittal proton density-weighted image (7A) demonstrates nodular posterior intraarticular and extracapsular foci of very low signal (short arrows) and large joint effusion with low signal (i.e. hemosiderin-laden) synovium (arrowheads). Sagittal gradient echo image (7B) demonstrates accentuation of the hemosiderin in the posterior nodular foci of PVNS (short arrows) and the hemosiderin-laden synovium (arrowheads), particularly in the patellofemoral joint and suprapatellar pouch. Note in 7B the rounded focus of PVNS in the proximal suprapatellar pouch (arrow). This case demonstrates that complete imaging of the joint and the entirety of extracapsular soft tissue involvement (if present) is necessary to evaluate the entire extent of PVNS.
Figure 8:
20 year-old female with knee pain and stiffness. Axial fat-suppressed proton density-weighted image (8A) demonstrates an ovoid, mildly heterogeneous mass-like area in the infrapatellar region. Sagittal fat-suppressed proton density-weighted image (8B) demonstrates minimal subtle low signal inferiorly (arrow). Sagittal T1-weighted image (8C) demonstrates subtle small foci of slightly low signal in the largely intermediate signal mass-like area. This case of localized PVNS shows the variability of hemosiderin deposition in PVNS. The localized form is much more likely to have minimal hemosiderin deposition. Note the similarity of location to the introductory case (Figures 1 and 2) and the normal amount of joint fluid.
Figure 9:
Sagittal proton density-weighted image (9A) of the foot demonstrates a large irregularly shaped mass-like area of largely intermediate to minimally high signal with scattered foci of low signal in the lateral hindfoot to midfoot. Coronal proton density-weighted image (9B) demonstrates extensive similar signal filling and extending laterally from the subtalar joint, extending into the subcutaneous fat laterally corresponding to that seen in 9A, with associated erosive change and osseous invagination (arrowheads in 9B). Note the prominent accentuation of hemosiderin (asterisks) on the corresponding gradient echo image (9C) in this case of diffuse PVNS.
Figure 10:
30 year-old female with radial wrist pain and swelling and dorsal mass for one year. Axial STIR (10A) and T1-weighted (10B) images demonstrate massive heterogeneous signal, some of which is minimally high signal and with amorphous foci of low signal, filling the dorsal compartment and extending ulnar and palmar in the wrist (asterisks). Coronal gradient echo image (10C) demonstrates multifocal low signal amidst the dorsal mass-like area, reflecting hemosiderin in this case of diffuse PVNS.
Treatment and Prognosis
Surgical resection is the primary treatment for all forms of PVNS. Arthroscopic resection with limited synovectomy is usually performed for localized PVNS, particularly in the knee. There is a low recurrence rate.2
Arthroscopic and/or open total synovectomy is performed for diffuse PVNS. These patients have significant rates of recurrence (up to 50%).5 Neither the arthroscopic or open approach has proven definitively better at reducing recurrence.5,19 Posterior involvement can prove more difficult to completely remove via arthroscopy. Although rare, subcutaneous contamination of a portal can occur.2 Extra-articular disease usually requires arthrotomy but may be contraindicated in some circumstances (e.g. neurovascular involvement). As expected, an open approach is associated with increased length of hospitalization and rehabilitation as well as increased rates of arthrofibrosis as compared to arthroscopy.2 Some have found a combined anterior arthroscopic and posterior open surgical approach for treatment of diffuse PVNS of the knee to be most efficacious.5
Patients with recurrent disease frequently have more extensive involvement and poorer outcomes.2 Patients with cartilage defects have greater rates of recurrence than those without; even in those without recurrence there appears to be an increased risk of osteoarthritis.19 As total synovectomy in patients with osteoarthritis may not relieve symptoms or reduce recurrence, arthroplasty may be recommended.19
Adjuncts, typically used in refractory cases or those with extensive extra-articular spread, include external beam radiation, radiosynoviorthesis (intra-articular injection of radioisotopes), and systemic therapies. No clear decrease in recurrence rate has been found using either external beam radiation or radiosynoviorthesis as adjuvant therapy in diffuse PVNS.3 Given the overexpression of CSF1 in PVNS, different tyrosine kinase inhibitors of CSF1R (imatinib, nilotinib, pexidartinib) and emactuzumab (anti-CSF1R monoclonal antibody) have been studied in clinical trials. Only one (pexidartinib) has been FDA-approved; it has a black box warning as it can cause severe and potentially fatal liver injury.3,5
Conclusion
While PVNS is well known to radiologists, the diagnosis can be elusive. Hemosiderin deposition, a key feature, can be minimal in localized PVNS. It can occur in the very young and very old. While the knee is primarily affected, PVNS can involve any joint or synovial-containing structure. For example, it can involve the apophyseal joints of the spine, the temporomandibular joint, and be isolated to a bursa; in these uncommon sites it can be mistaken for another etiology (e.g. primary bone or soft tissue lesion, metastasis). The definitive diagnosis is, as always, made by tissue sampling.
References
- Zeoli T, Mathkour M, Scullen T, et al. Spinal pigmented villonodular synovitis and tenosynovial giant cell tumor: a report of two cases and a comprehensive systematic review. Clin Neurol Neurosurg 2021 Mar;202:106489. ↩
- Tyler WK, Vidal AF, Williams RJ, Healey JH. Pigmented villonodular synovitis. J Am Acad Orthop Surg 2006 Jun;14(6):376-385. ↩
- Healey JH, Bernthal NM, van de Sande M. Management of tenosynovial giant cell tumor: a neoplastic and inflammatory disease. J Am Acad Orthop Surg Glob Res Rev 2020 Nov;4(11):e20.00028. ↩
- West RB, Rubin BP, Miller MA, et al. A landscape effect in tenosynovial giant-cell tumor from activation of CSF1 expression by a translocation in a minority of tumor cells. Proc Natl Acad Sci USA 2006 Jan 17;103(3):690-695. ↩
- Bernthal NM, Ishmael CR, Burke ZDC. Management of pigmented villonodular synovitis (PVNS): an orthopaedic surgeon’s perspective. Curr Oncol Rep 2020 Jun 4;22(6):63. ↩
- Murphey MD, Rhee JH, Lewis RB, et al. Pigmented villonodular synovitis: radiologic-pathologic correlation. RadioGraphics 2008;28:1493-1518. ↩
- Botez P, Sirbu PD, Grierosu C, et al. Adult multifocal pigmented villonodular synovitis–clinical review. Int Orthop 2013 Apr;37(4):729-733. ↩
- Roguski M, Safain MG, Zerris VA, et al. Pigmented villonodular synovitis of the thoracic spine. J Clin Neurosci 2014 Oct;21(10):1679-1685. ↩
- Safaee M, Oh T, Sun MZ, Parsa AT, et al. Pigmented villonodular synovitis of the temporomandibular joint with intracranial extension: A case series and systematic review. Head Neck. 2015 Aug;37(8):1213-1224. ↩
- Maheshwari AV, Muro-Cacho CA, Pitcher JD Jr. Pigmented villonodular bursitis/diffuse giant cell tumor of the pes anserine bursa: a report of two cases and review of literature. Knee 2007 Oct;14(5):402-407. ↩
- Karami M, Soleimani M, Shiari R. Pigmented villonodular synovitis in pediatric population: review of literature and a case report. Pediatr Rheumatol Online J. 2018 Jan 17;16(1):6.Baroni E, Russo BD, Masquijo JJ, et al. Pigmented villonodular synovitis of the knee in skeletally immature patients. J Child Orthop 2010 Apr;4(2):123-127. ↩
- Karami M, Soleimani M, Shiari R. Pigmented villonodular synovitis in pediatric population: review of literature and a case report. Pediatr Rheumatol Online J. 2018 Jan 17;16(1):6. ↩
- Soubai RB, Tahiri L, Ibrahimi A. Bilateral pigmented villonodular synovitis of the knee. Joint Bone Spine 2011;78(2):219-221. ↩
- Kim HS, Kwon JW, Ahn JH, et al. Localized tenosynovial giant cell tumor in both knee joints. Skeletal Radiol. 2010 Sep;39(9):923-926. ↩
- Oni JK, Cavallo RJ. A rare case of diffuse pigmented villonodular synovitis after total knee arthroplasty. J Arthroplasty 2011 Sep;26(6):978.e9-978.e11. ↩
- Tan YC, Tan JY, Tsitskaris K. Systematic review: total knee arthroplasty (TKA) in patients with pigmented villonodular synovitis (PVNS). Knee Surg Relat Res. 2021 Feb 25;33(1):6. ↩
- Al-Ibraheemi A, Ahrens WA, Fritchie K, et al. Malignant Tenosynovial Giant Cell Tumor: The True “Synovial Sarcoma?” A Clinicopathologic, Immunohistochemical, and Molecular Cytogenetic Study of 10 Cases, Supporting Origin from Synoviocytes. Mod Pathol. 2019 Feb;32(2):242-251. ↩
- Imakiire N, Fujino T, Morii T, et al. Malignant pigmented villonodular synovitis in the knee – report of a case with rapid clinical progression. Open Orthop J 2011 Jan 7;5:13-16. ↩
- Adelani MA, Wupperman RM, Holt GE. Benign synovial disorders. J Am Acad Orthop Surg 2008;16:268-275. ↩
- Elumogo CO, Kochenderfer JN, Civelek AC, Bluemke DA. Pigmented villonodular synovitis mimics metastases on fluorine 18 fluorodeoxyglucose position emission tomography-computed tomography. Quant Imaging Med Surg. 2016 Apr;6(2):218-223. ↩