Clinical History:
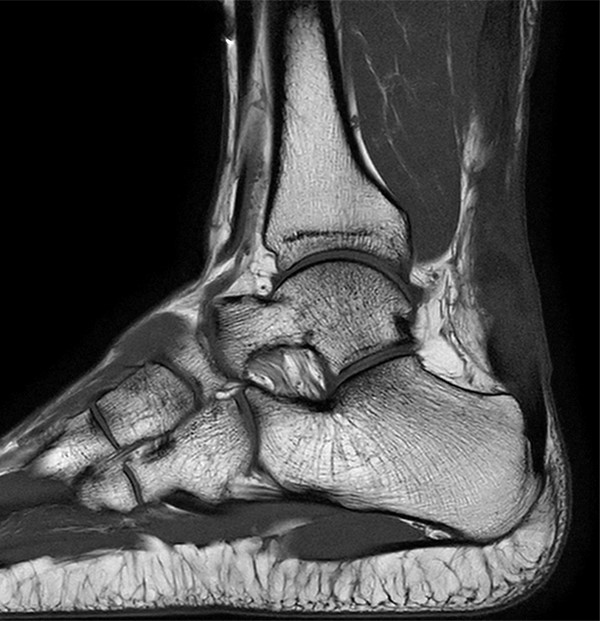
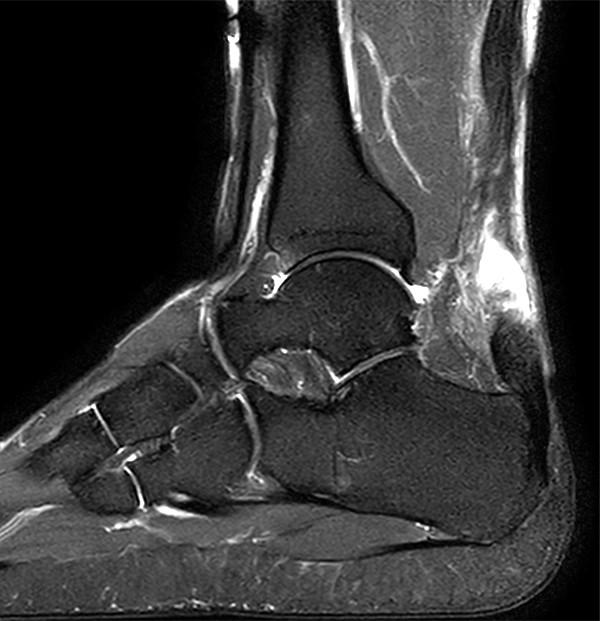
A 38-year-old male presents after feeling pain and a pop at the posterior left ankle while doing box jumps 1 month ago. He has weak plantar flexion and is unable to stand on his toes with the left foot. Sagittal T1-weighted (1A) and fat-suppressed T2-weighted (1B) images of the ankle are provided. What are the findings? What is your diagnosis?
Findings:
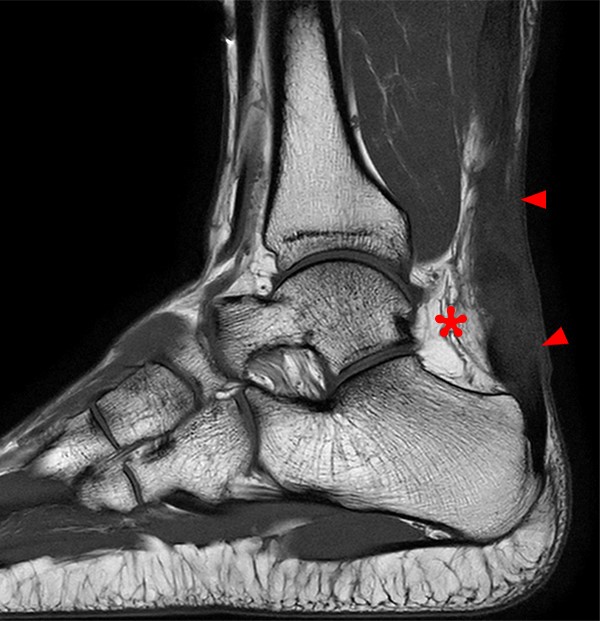
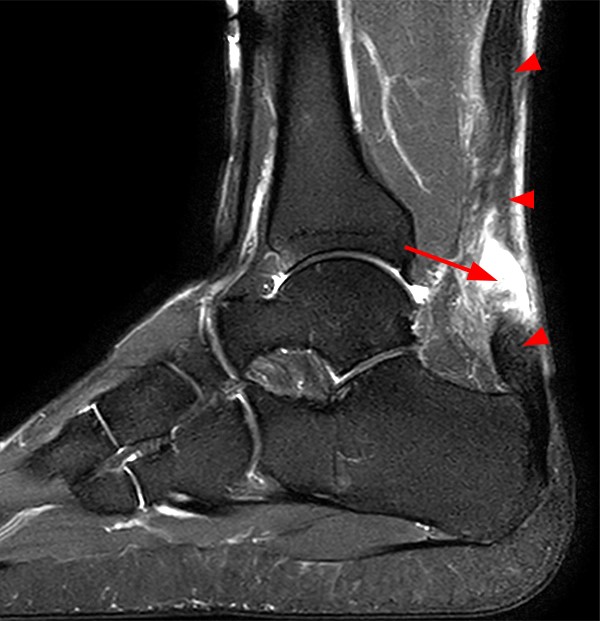
Figure 2:
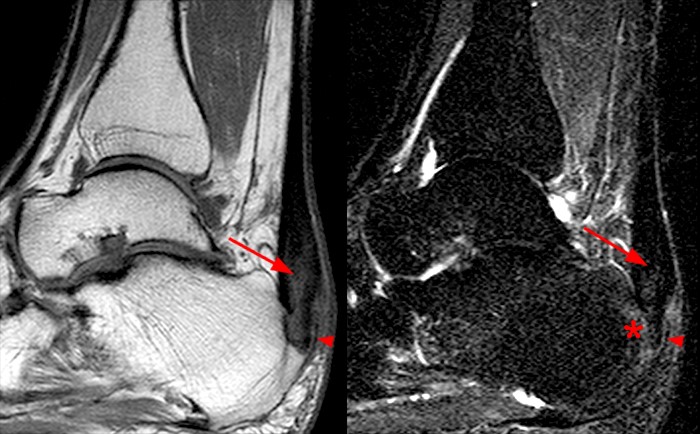
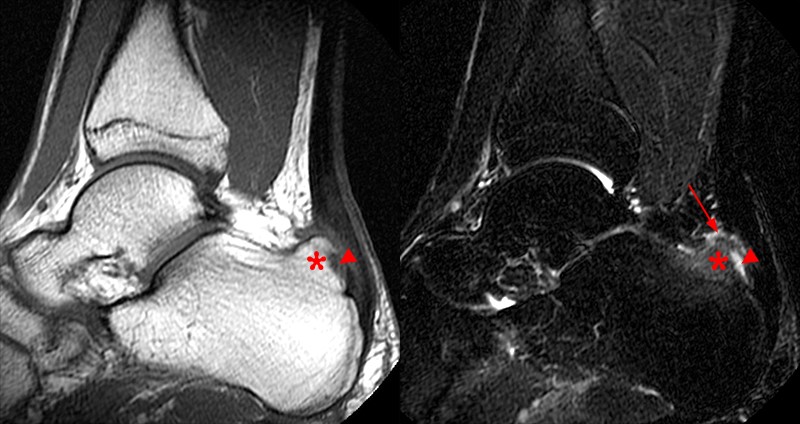
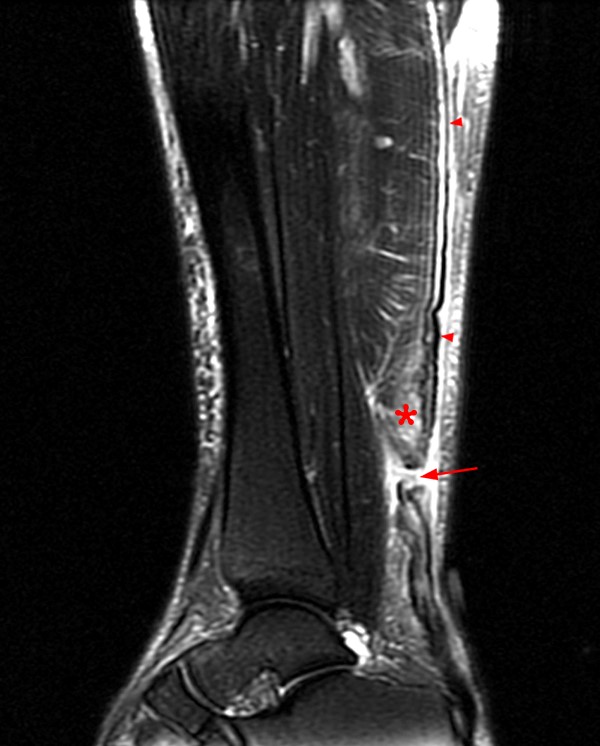
2A – The sagittal T1-weighted image demonstrates heterogeneous intermediate and low signal of the Achilles tendon. The tendon demonstrates irregular and ill-defined margins posteriorly with subcutaneous soft tissue thickening posteriorly (arrowheads) and anteriorly with pre-Achilles fat (asterisk).
2B – The fat-suppressed T2-weighted sagittal image demonstrates a rupture of the Achilles tendon 3.5 cm proximal to the calcaneal attachment with a 2-cm fluid-filled gap (arrow) between the torn ends. The proximal and distal segments of the Achilles tendon are prominently thickened (arrowheads) suggesting underlying Achilles tendinosis. Edema is demonstrated within the adjacent subcutaneous and pre-Achilles fat.
Diagnosis
Subacute Achilles tendon rupture with moderate Achilles tendinosis.
Introduction
Although the Achilles tendon is the largest and strongest tendon in the body 1,2 it is frequently injured, accounting for 20% of large tendon injuries. Achilles tendon injuries are common in both athletes and non-athletes and often result from a combination of factors that include overuse, altered biomechanics, and tendon degeneration. These injury pathways can variably affect the tendon and surrounding structures. Consequently, injuries of the Achilles tendon commonly exhibit a combination of underlying pathologies.
Anatomy
The Achilles tendon forms from the fusion of the aponeuroses of the medial and lateral heads of the gastrocnemius and the soleus muscles. This musculotendinous unit crosses three joints (knee, tibiotalar, and subtalar) making it more susceptible to injury. The Achilles tendon internally rotates by a variable amount such that at the insertion the tendon fibers from the soleus are located medially, the tendon fibers from the medial head of the gastrocnemius muscle are located posterolaterally, and the fibers arising from the lateral head of the gastrocnemius muscle are located anterolaterally 3,4. In the newborn, thick fibers extend from the Achilles tendon insertion to the plantar fascia. With increasing age, the fibrous continuity between the Achilles tendon insertion and the plantar fascia gradually diminishes resulting in clear separation in the elderly foot.5
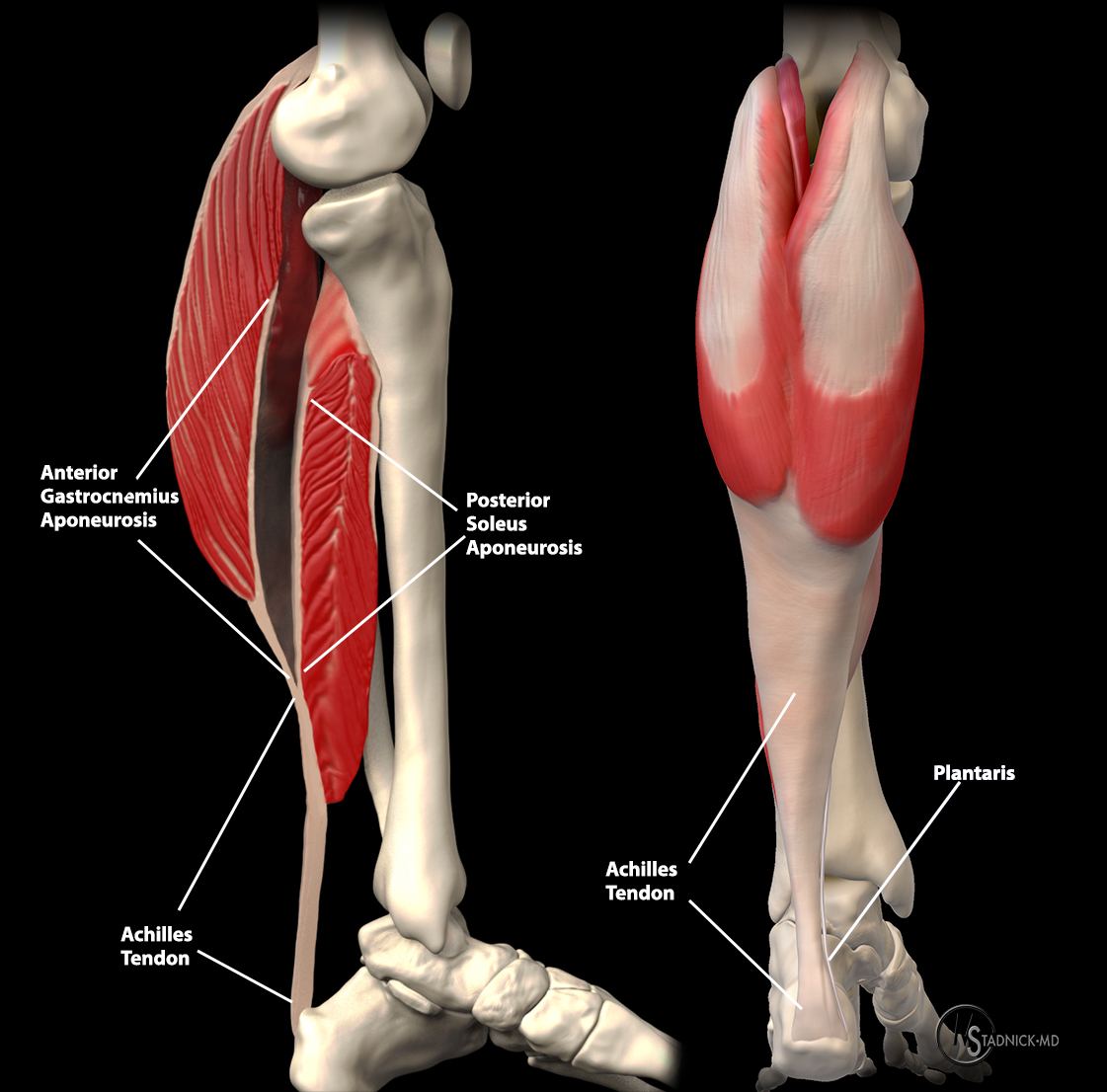
The Achilles tendon forms from the aponeuroses of the gastrocnemius and soleus muscles. The plantaris tendon extends to the medial surface just proximal to the level of fusion and extends distally to its insertion.
The Achilles tendon does not have a true synovial sheath. Instead, a richly vascularized paratenon composed of a single layer of cells and a membrane of loose areolar tissue rich in mucopolysaccharides, surrounds the Achilles tendon. The paratenon is contiguous with the muscular fascia proximally and blends with the periosteum of the calcaneus distally 6,7,8.
The paratenon allows the tendon to glide 1.5 cm cephalocaudal relative to the surrounding tissues1. The blood supply to the Achilles tendon forms a network of arteries within the paratenon covering the tendon surface 9. The majority of the Achilles tendon is supplied by branches of the posterior tibial artery which are located medial to the tendon and supply the proximal and distal portions of the tendon9. The mid-portion of the tendon is relatively hypovascular and supplied by branches from the peroneal artery located lateral to the tendon9. A 3-6 cm segment proximal to the calcaneal attachment is more susceptible to rupture due to its small cross-section and relative hypovascularity 10.
The plantaris arises medial to the origin of the lateral head of the gastrocnemius muscle at the lowest part of the lateral supracondylar ridge and the knee joint capsule. The plantaris tendon extends obliquely from lateral to medial as it passes distally between the gastrocnemius muscle and soleus muscle. The plantaris tendon most commonly emerges medially between the aponeuroses of the gastrocnemius and soleus muscles just proximal to their fusion to form the Achilles tendon. Variable insertions can be seen, including a fan-shaped insertion just medial to the Achilles tendon insertion, anterior to the medial border of the Achilles tendon insertion, a broad insertion at the dorsal and medial surface of the tendon, and least commonly an insertion into the medial Achilles tendon proximal to its insertion11. The plantaris tendon is absent in approximately 7.05% of the population11. Anatomic variations of the soleus are rare. The accessory soleus muscle (MRI Web Clinic November 2008) is separate from the soleus muscle and surrounded by its own fascia. It is found anterior or anteromedial to the Achilles tendon and can have a variety of tendinous or muscular insertions12. An association between an accessory soleus muscle and Achilles tendinopathy has been suggested13.
Function
The gastrocnemius and soleus muscles acting via the Achilles tendon are the primary flexors of the ankle joint and provide the primary propulsive force during walking, running, and jumping7,14. In addition the soleus muscle helps to prevent lurching or falling forward while standing7. Contraction of the triceps surae also acts to flex the knee and to supinate the subtalar joint7. During walking the Achilles tendon experiences dynamic loads up to 2 to 3 times the body weight and forces can rise to 12.5 times the body weight with activities such as running15.
Normal MRI Appearance
The normal Achilles tendon is of low signal intensity on all imaging sequences. The normal fascicular anatomy may be visible as linear signal on short TE (T1-weighted and proton density-weighted) sequences but is not apparent on T2-weighted images. Because of the internal twisting of the fascicles of the Achilles tendon, magic angle artifact can be seen on short TE sequences. Punctate areas of high signal can be seen in the distal tendon on axial images representing interfascicular septa containing intratendinous vessels16. The anterior and posterior margins of the Achilles tendon are normally parallel with a concave anterior margin over most of its course (Fig. 3). At the junction of the soleus and gastrocnemius aponeurosis to form the Achilles tendon the anterior margin is typically convex. In the coronal plane, the medial and lateral sides of the tendon are straight and gradually widen distally17. Several variations in contour have been identified including a localized anterior convexity that shifts from lateral to medial on sequential axial images from proximal to distal18. The average thickness of the normal Achilles tendon is 6mm. The tendon is thicker in tall individuals and the elderly, and generally slightly thicker in men19.
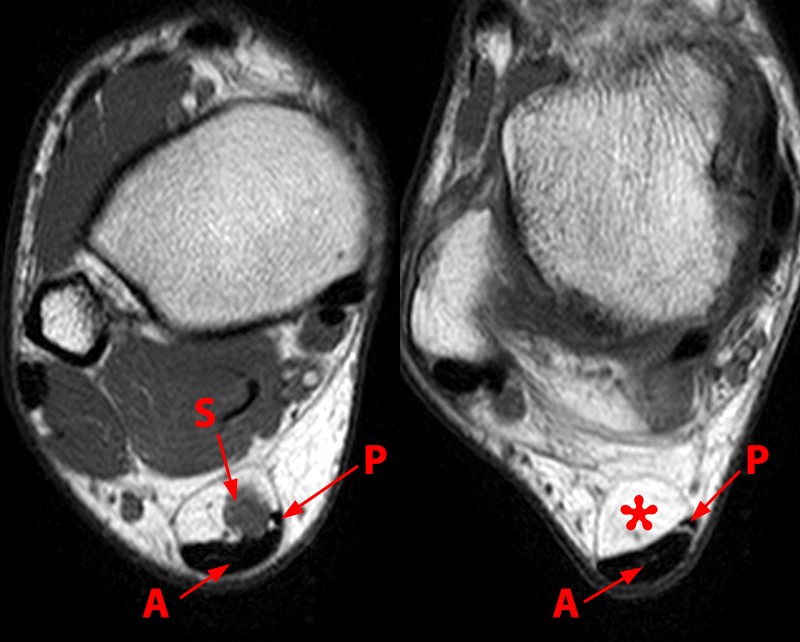
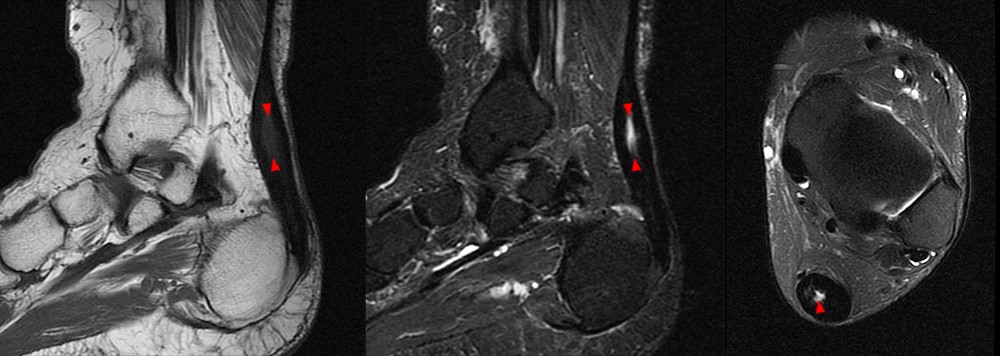
Figure 3:
Axial T1-weighted images at the proximal (left) and mid- (right) Achilles tendon (A) demonstrates the distal extent of the soleus muscle (S) and the plantaris tendon (P) located medially. Further distally at the mid-Achilles the Achilles-associated portion of Kager’s fat pad (asterisk) is surrounded by a thin septum. Note the normal concave contour of the anterior Achilles tendon.
The retrocalcaneal bursa is a saddle-shaped structure located posterosuperior to the calcaneus situated between the paratenon and the pre-Achilles fat. The retrocalcaneal bursa is normally visible on fluid-sensitive MRI sequences as a focus of high signal intensity and measurements up to 1 mm anterior to posterior, 11 mm transverse, and 7 mm superior to inferior are normal20. Fat is seen anterior to the tendon within the pre-Achilles (Kager’s) fat pad. Vessels are normally seen within the fat pad and should not be mistaken for edema on fluid-sensitive sequences based on their tubular morphology.17 The pre-Achilles fat pad can be divided into 3 portions typically separated by thin fascia into a deep FHL-associated part, a calcaneal bursal component, and a superficial Achilles-associated part which is seen immediately anterior to the Achilles tendon21. This portion of the pre-Achilles fat commonly shows edema and inflammatory changes associated with Achilles pathology22. Subcutaneous fat is normally seen between the Achilles tendon and the skin. Absence of fat in this region may be caused by callus or retro-Achilles bursitis17.
Pathology of the Achilles tendon
The terminology used to describe Achilles tendon pathology is confusing with differing descriptions based on anatomic location, histopathology, or generic names23. Conceptually, Achilles pathology can be divided into abnormalities surrounding or adjacent to the tendon, pathology within the mid-portion of the tendon, and the constellation of abnormalities that can occur at the tendon insertion. Achilles rupture can occur with or without these underlying tendon pathologies.
Peritendinitis and Paratendinitis
Confusing terminology persists in describing the inflammatory and overuse changes of the structures surrounding the Achilles tendon. Paratenonitis is popularly used to indicate inflammation of the paratenon. To better reflect the location of the abnormality, which surrounds the Achilles tendon, some authors prefer peritendinitis as a more accurate descriptor24,25,26. Peritendinitis can accompany tendon degeneration, but can also occur without underlying tendon pathology. This condition is activity related and is most commonly seen in active, athletic patients. It often results from overuse injuries or abnormal biomechanics and is commonly an overtraining injury, especially in marathon runners7,27. Fibrinous exudate, fibroblastic changes, and infiltration by inflammatory cells results in a thickened paratenon. The diagnosis is typically made clinically based on the findings of swelling, pain, crepitus, tenderness and warmth28. On MRI, peritendinitis demonstrates hyperintense signal on STIR and fat-suppressed T2-weighted images surrounding or partially surrounding the Achilles tendon (Fig. 4)17,29. In the setting of acute peritendinitis, fluid signal intensity can be seen27.
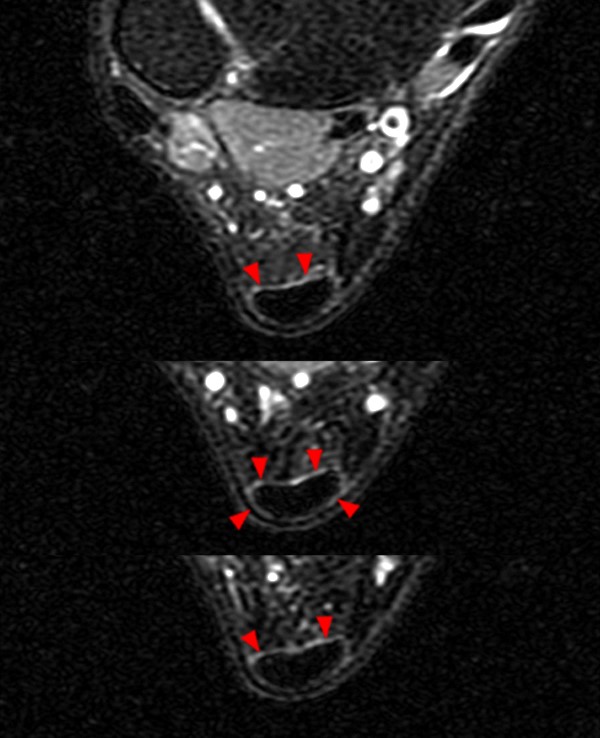
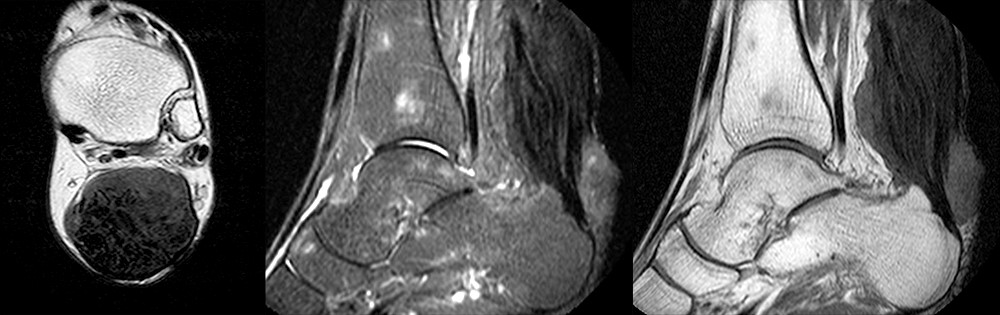
Figure 4:
25-year-old cross-country runner with pain and swelling with tenderness to palpation above the Achilles tendon insertion. Sequential axial T2-weighted fat suppressed images from superior (top) to inferior (bottom) demonstrate a thin rim of increased signal (arrowheads) surrounding a normal appearing Achilles tendon, compatible with mild peritendinitis.
Inflammation can also be seen within the pre-Achilles fat pad, with or without underlying Achilles tendon pathology. Based on its location adjacent to, but not surrounding the tendon, this has been described as Achilles paratendinitis26,30. On MRI, the fat signal of the pre-Achilles fat pad immediately adjacent to the Achilles tendon is replaced by increased signal on fluid-sensitive sequences, and the normal high signal of fat on T1-weighted images is replaced by intermediate signal (Fig. 5).
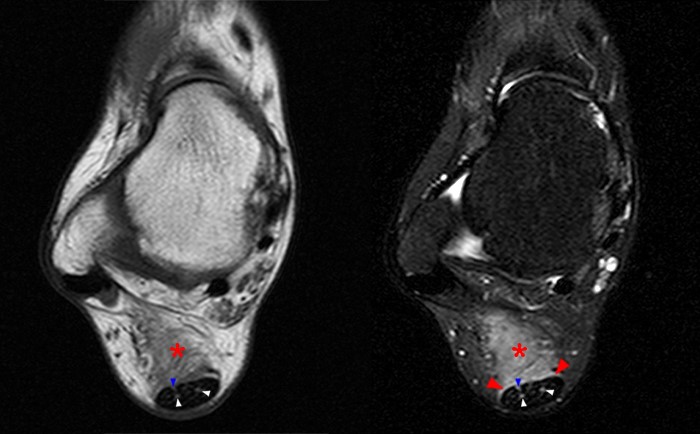
Figure 5:
32-year-old female runner with posterior heel pain. Axial T1-weighted (left) and T2-weighted fat-suppressed (right) images demonstrate changes of tendinosis with mild convexity of the ventral tendon surface and intratendinous punctate foci that are hyperintense on both series compatible with small interstitial splits seen in cross-section (white arrowheads), with a small split seen along the anterior lateral tendon surface (blue arrowheads). In addition to peritendinitis which is best seen on the fluid sensitive T2-weighted fat-suppressed image (arrowheads), edema extends into the pre-Achilles fat pad (asterisks) compatible with paratendinitis.
Mid-substance Achilles Pathology
The most common abnormality involving the Achilles tendon mid-substance is tendinosis. Tendinosis is a degenerative, noninflammatory process, most closely associated with aging31. Combinations of hypoxic, mucoid, calcific, and fatty degeneration occur with aging or with overuse, resulting in a disorganized collagen structure and increased tendon stiffness. With progression, collagen fiber separation and disruption occur with coalescence of mucoid material between the collagen fibers, which can progress to microtears, intrasubstance tears, surface partial tears, and even complete tears29,32. Although all types of degeneration are frequently found, hypoxic and mucoid degeneration are the most prevalent degenerative findings in ruptured Achilles tendons31. Hypoxic degenerative changes are felt to be caused by ischemia related to relative hypovascularity in the critical zone of the tendon31,33,17. Mucoid degeneration results in large myxoid patches and vacuoles between the degenerated tendon fibers31. Patients with predominant hypoxic changes often present with multiple symptomatic episodes while mucoid changes of the Achilles tendon often remain clinically silent17.
A typical presentation of tendinosis is the middle-aged athlete who presents following an increase in training duration, intensity, and frequency with pain in the mid portion of the tendon. On physical examination, the patient presents with swelling after activity and pain with active and passive motion34. Visible or palpable tendon enlargement or a knot, which is painful to compression are common findings34. With MRI, the changes associated with hypoxic degeneration tend to result in tendon enlargement without internal signal changes (Fig. 6). Mucoid degeneration leads to enlargement with altered T2-weighted signal, reflecting mucoid deposits and interstitial tears17. When present, interstitial tears present as longitudinally oriented linear increased signal on T1-weighted, proton-density-weighted, and T2-weighted and STIR sequences, with intact surrounding fibers (Fig.7)35,30.
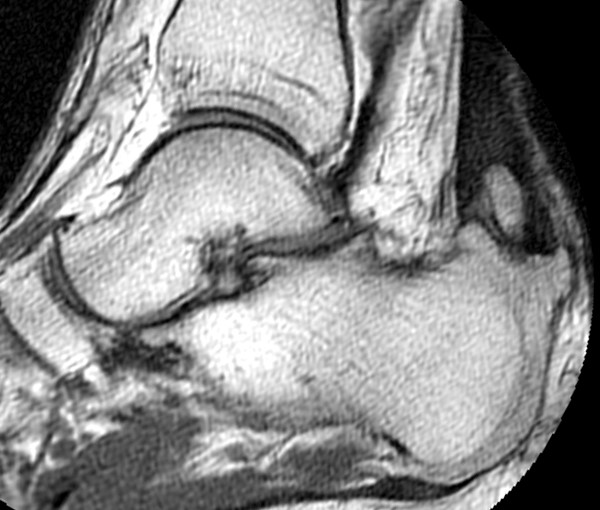
Figure 6:
52-year-old female with chronic pain and a palpable lump over the Achilles tendon. Sagittal T2-weighted fat-suppressed (left) and axial T1-weighted (right) images demonstrate thickening of the Achilles tendon with normal low signal of the tendon and loss of the normal concave contour of the Achilles tendon compatible with Achilles tendinosis. A thin layer of increased signal along the anterior surface of the Achilles tendon (arrowheads) on the fluid-sensitive series is compatible with peritendinitis.
Figure 7:
45-year-old male with chronic Achilles pain. Sagittal T1-weighted (left), STIR (middle) images and an axial fat-suppressed T2-weigted image (right) demonstrate a diffusely thickened Achilles tendon compatible with tendinosis. Internal increased signal on the T1-weighted images corresponds to fluid-signal on the STIR and the fat-suppressed T2-weighted images. These findings are compatible with an interstitial tear.
Achilles tendon enlargement may also be seen in rheumatoid arthritis and gout17. Xanthomas are the result of lipomatosis and are frequently correlated with a family history of coronary artery disease and certain metabolic diseases, in particular type IIa and type III hyperlipidemia36,37. Typically, there is internal fat or intermediate signal enlarging the tendon and spreading the tendon fascicles (Fig. 8), resulting in increased T1 and T2-weighted signal and a stippled pattern on cross-section, but occasionally, internal signal may be subtle38,39,17.
Figure 8:
53-year-old female with bilateral enlargement of the Achilles tendon and a history of hyperlipidemia. Axial T2-weighted FSE (left), sagittal STIR (middle), and sagittal T1-weighted (right) images of the left ankle demonstrate severe diffuse thickening of the Achilles tendon with intermediate signal material infiltrating and separating the tendon fibers and extending anterior and posterior to the tendon consistent with an Achilles tendon xanthoma. Similar changes were present in the right Achilles tendon.
Insertional Pathologies
Insertional Tendinopathy
Insertional Achilles tendinopathy frequently coexists with adjacent soft tissue and osseous pathology. In contrast to tendinosis that is typically found in the tendon mid-substance, insertional tendinopathy is thought to have an inflammatory component initially, thus the frequent use of the term “insertional tendinitis” in describing this process. However, in its chronic form, histopathologic changes of inflammation are commonly no longer present and some authors prefer to use the term insertional tendinopathy or even tendinosis28,34. Overuse is contributory with a reported prevalence of up to 18% in runners and 9% in dancers7. Posterosuperior boney prominence of the calcaneus (Haglund’s deformity) is felt to contribute to the development of insertional Achilles tendinopathy. Insertional Achilles tendinopathy can also occur in association with inflammatory arthropathies, corticosteroid use, diabetes, hypertension, obesity, gout, and fluroquinolone antibiotics28. Patients typically present with stiffness and a palpable, irritated, painful bump at the Achilles tendon insertion34. Enthesophyte formation at the Achilles tendon insertion or calcification/ossification within the distal Achilles tendon are common and may lead to palpable abnormalities (Fig. 9)40. MRI depicts distal tendon thickening with ill-defined tendon signal heterogeneity. Partial tears demonstrate hyperintense defects on fluid sensitive sequences. Edema within enthesophytes correlates with more acute symptoms (Fig. 10). Retrocalcaneal bursitis is a frequent accompanying finding.
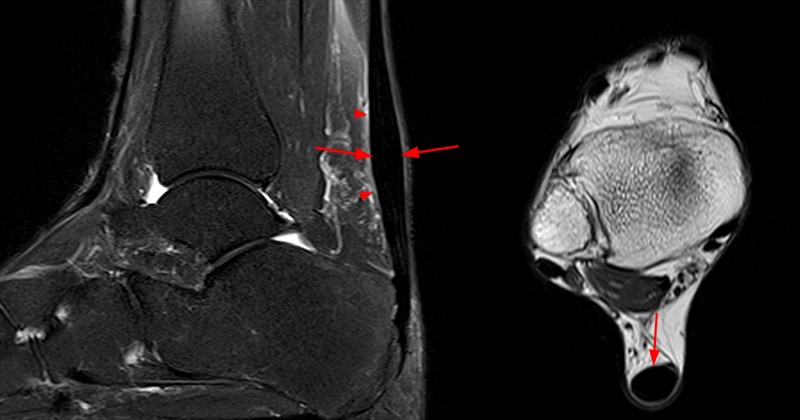
Figure 10:
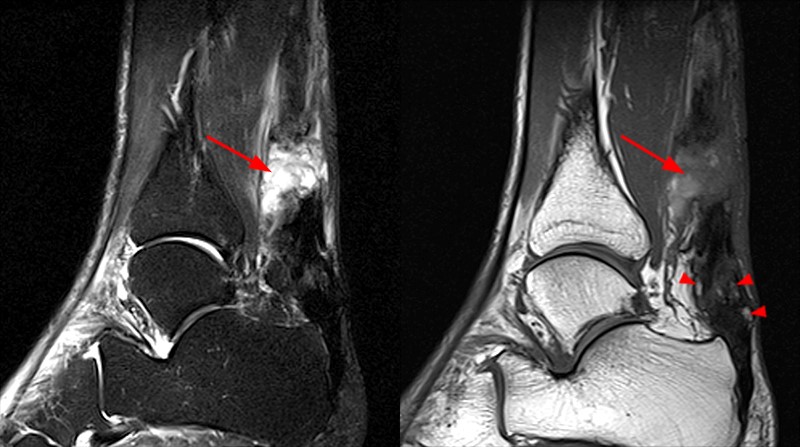
52-year-old male with chronic posterior heel pain worsening for 4 months. Sagittal T1-weighted (left) and STIR (right) images demonstrate distal tendon thickening at the insertion with heterogeneous internal signal (arrow). An enthesophyte (arrowheads)is seen posterior to the distal Achilles insertion. Edema is seen within the enthesophyte and within the posterosuperior calcaneus (asterisk) subjacent to the Achilles insertion on the STIR image.
Haglund’s Deformity
Prominence of the posterosuperior angle of the calcaneus, referred to as a Haglund’s deformity, is commonly described as a contributor to retrocalcaneal bursitis and insertional Achilles tendon pathology (Fig. 11)41. While prominent contours of the posterosuperior angle of the calcaneus are recognizable as pathologic, studies have failed to demonstrate radiographic criteria that quantify the size at which the convex posterosuperior angle of the calcaneus becomes symptomatic, and using various methods of radiographic measurement, Haglund’s deformities are found equally in symptomatic and asymptomatic individuals7,40. Nevertheless, in symptomatic patients, the prominence of the posterosuperior angle of the calcaneus is felt by many to play a role in retrocalcaneal bursitis and distal Achilles tendinopathy, and when surgical intervention is necessary, relatively small bony convexities are often removed42.
Figure 11:
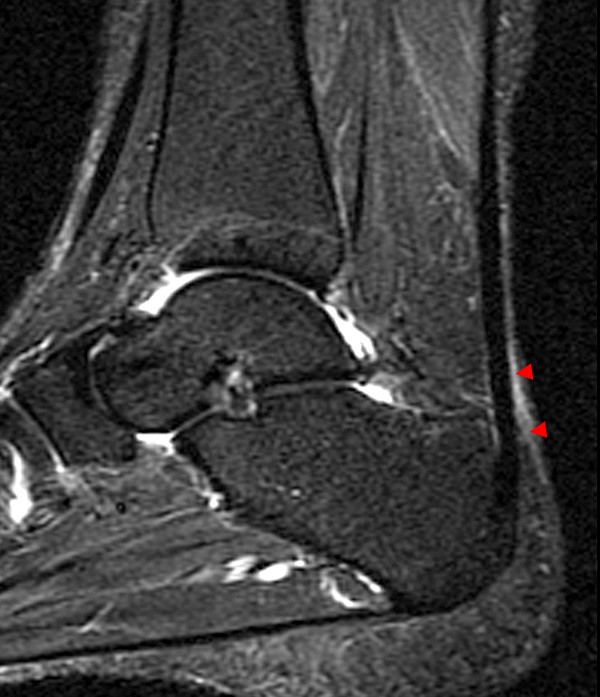
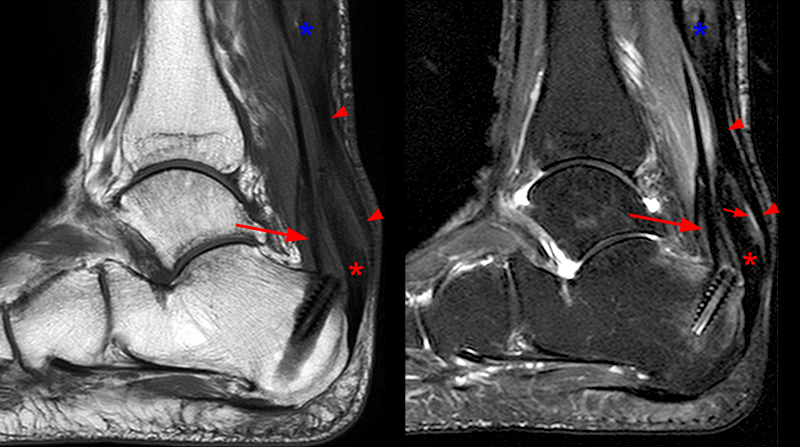
42-year-old male with heel pain. Sagittal T1-weighted (left) and fat-suppressed T2-weighted (right) images demonstrate focal prominence of the posterosuperior calcaneus (asterisk) with marrow edema, a small dorsal erosion (arrowheads) and a small amount of fluid (arrow) in the overlying retrocalcaneal bursa.
Retrocalcaneal Bursitis
In retrocalcaneal bursitis, the bursa between the posterosuperior calcaneus and the Achilles tendon becomes inflamed, hypertrophied, and adherent to the Achilles tendon7. Haglund’s deformity has been associated with retrocalcaneal bursitis41. Retrocalcaneal bursitis is commonly seen in runners resulting from repeated compression between the calcaneus and Achilles tendon with dorsiflexion of the ankle and often occurs without any calcaneal deformity27. The mechanism of irritation is further exacerbated by uphill running, which results in increased and repetitive ankle dorsiflexion7. Hindfoot varus and a rigid plantar flexed first ray are also contributory to the development of retrocalcaneal bursitis7. Bilateral involvement should alert the clinician to the possibility of a systemic inflammatory process, such as rheumatoid arthritis, which may result in retrocalcaneal bursitis in up to 10% of patients43. The clinical presentation is similar to insertional tendinitis with pain near the calcaneal insertion. A positive two-finger squeeze test indicates retrocalcaneal bursitis and is performed with pressure applied by two fingers medially and laterally just superior and anterior to the Achilles insertion7. On MRI, the retrocalcaneal bursa is distended with fluid signal and appears hyperintense on fluid-sensitive sequences. Enlargement of the bursal anteroposterior dimensions to greater than 2mm is consistent with bursitis20. There is often an associated element of inflammation in the adjacent paratenon and Achilles tendon insertion7. Associated partial Achilles tendon tear, peritendinitis, tendinopathy, or ossification is common (Fig. 12).
Figure 12:
76-year-old male with chronic heel pain. A Sagittal fat-suppressed T2-weighted image demonstrates a longitudinal partial tear (arrow) at the insertion with subjacent marrow edema of the calcaneus (red asterisk), retrocalcaneal bursitis (small arrow), retro-Achilles inflammation (arrowheads), and inflammation of the pre-Achilles fat (blue asterisk).
Retro-Achilles Bursitis
The retro-Achilles bursa is an adventitious bursa that develops as a response to friction between the posterior aspect of the calcaneus or the Achilles tendon and the skin (Fig. 13). It is most often seen at the posterolateral aspect of the calcaneus and is frequently associated with tight or poorly fitted shoes with a rigid heel counter23.
Haglund’s syndrome
The term Haglund’s syndrome was originally described as a radiographic combination of retrocalcaneal bursitis, insertional Achilles tendinitis, and retro-Achilles bursitis. This may be associated with a prominent bursal projection of the posterosuperior calcaneus (Haglund’s deformity)44,45. Ventral tears of the Achilles tendon and insertional enthesophytes may also be seen (Figure 12)46,47. This condition was attributed to poorly fitting or tight low-back shoes44, and the palpable soft tissue and osseous prominence that develops at the lateral aspect of the Achilles insertion was referred to as a “pump bump”44.
Achilles Tendon Tears
Achilles tendon ruptures occur with a bimodal age distribution, with the first peak in 25-40-year-old males and the second peak in those older than 60 years. It is rare to tear a normal tendon27. Risk factors associated with Achilles tendon ruptures include diffuse tendon degeneration, inflammatory conditions, and previous tendon repair on the contralateral side. In one study fluoroquinolone usage resulted in a 4-fold increased risk of rupture, and concomitant use of corticosteroids resulted in a 10-fold increased risk48. Haglund’s deformity may result in mechanical irritation and degeneration, raising the risk of rupture49. The most common presentation is a 40 year-old weekend warrior who experiences a sudden pop in the distal leg after pushing off from a dorsiflexed position while running or jumping 49. In the US, basketball accounts for the most injuries, followed by tennis and football50. In older patients, rupture may occur at sites of chronic degeneration during routine daily activities31. Physical findings include bruising around the posterior ankle, weakened plantar flexion, limp, and inability to bear weight. The diagnosis of Achilles rupture can be made with 2 or more of the following physical examination findings: positive Thompson test, decreased plantar flexion strength, palpable defect roughly 2 to 6 cm proximal to the insertion, and increased passive ankle dorsiflexion49,51. A positive Thompson test occurs when compression of the calf does not result in passive plantar flexion of the foot49. Approximately 25% of acute ruptures are misdiagnosed due to false-negative Thompson test, a large hematoma, or plantar flexion secondary to extrinsic foot flexors52. Partial tears or tendon ruptures can occur anywhere along the Achilles tendon. On MRI, partial tears demonstrate alteration in architecture with interruption of tendon fibers. Heterogeneous T2 hyperintense signal fills the tendon defect and outlines the ends of the torn fibers, which can partially retract (Fig. 14)30.
Complete tears of the Achilles tendon results in tendon discontinuity with fluid or heterogeneous signal hematoma filling the tendon gap, and torn tendon fibers are distracted or overlapping30. Peritendinous hemorrhage and edema are present with recent tendon tears. As the tear ages, organization of the hematoma, granulation tissue, and eventual scar tissue can make detection of the torn tendon edges difficult27. Chronic ruptures often demonstrate muscle atrophy with further retraction of the tendon27. Fatty infiltration portends a poorer prognosis for surgical repair53. Thus, when reporting tears, it is essential to describe the size of the tendon gap, the quality of the torn tendon, an estimation of cross-sectional involvement, and the presence of atrophy of the soleus and gastrocnemius muscles27.
Most Achilles tendon tears occur in the relatively avascular zone located 2-6 cm from the insertion (Fig. 2)27. Insertional tears are less common but partial tears or ruptures can occur (Fig. 15) as a result of incompletely treated insertional tendinitis27. Rarely they may be iatrogenic or associated with systemic diseases like diabetes27.
Figure 15:
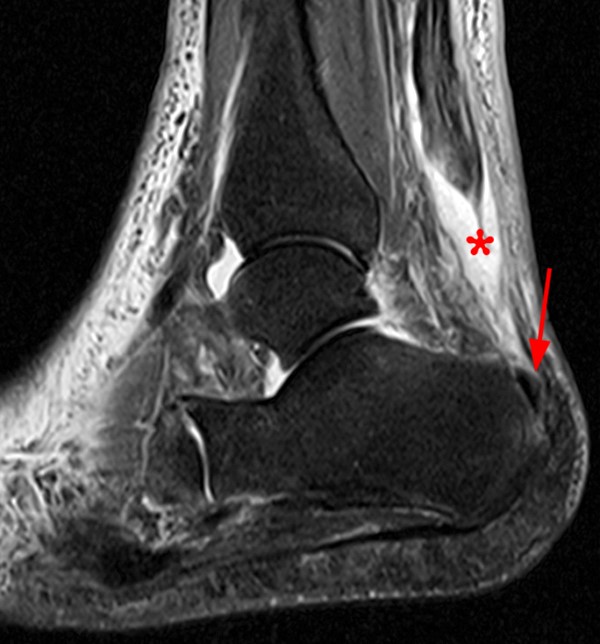
76-year-old female with pain posteriorly and laterally with no known injury. A sagittal fat-suppressed T2-weighted image demonstrates a distal Achilles rupture with edema and hemorrhage filling the gap (asterisk) between the retracted tendon and the small tendon stump (arrow) at the calcaneal insertion.
Proximal tears are most commonly partial tears involving the medial head of the gastrocnemius myotendinous junction (MRI Web Clinic October 2016). Proximal ruptures (complete tears) at the myotendinous junction are less common (Fig. 16).
Figure 16:
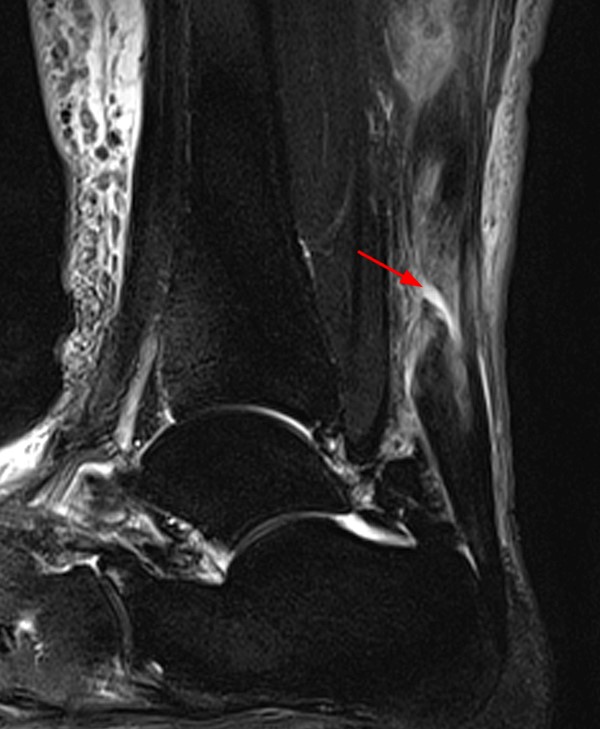
41-year-old male injured playing basketball. A sagittal fat-suppressed T2-weighted image demonstrates a proximal Achilles tendon rupture (arrow) at the myotendinous junction with a small gap. Edema is demonstrated within the distal soleus muscle (asterisk), and the gastrocnemius aponeurosis (arrowheads) is outlined by edema and fluid signal.
Treatment
Tendinopathy and peritendinitis usually respond to conservative measures including rest, ice, compression, training modifications, NSAIDs, heel lifts, orthotics, and night splints28. Local injections of pharmacologic agents into the tendon, including corticosteroids have not proven to be efficacious and raise the risk of additional weakening of the tendon28. In recalcitrant cases of peritendinitis, brisement (injection of fluid between the tendon and the paratenon) has been advocated54. Tendonoscopy can be performed to assist in the debridement of the thickened paratenon and to release the plantaris55. Open surgical techniques for peritendinitis include longitudinal incision and excision of the thickened paratenon. For patients with tendinopathy requiring surgery, debridement of the degenerated portions of the Achilles tendon and repair is performed56. Augmentation with allograft or tendon transfer are commonly performed for patients with diseased tendon affecting more than 50% of the tendon cross-section34. Retrocalcaneal bursitis is usually successfully treated conservatively including NSAIDs, modification of training regimens, and eccentric strengthening7. A heel lift and brief immobilization may be beneficial28. Surgical intervention consists of decompression of the bursa, debridement of the tendon, repair of the tendinous insertion, and resection of the osseous prominence of the calcaneus7,57. Insertional tendinopathy is most often treated conservatively with ice, NSAIDs, and brief immobilization followed by gradually increasing load-bearing activities58. Platelet-rich plasma has been proposed for refractory cases, however there is little evidence to support this procedure in the Achilles58,59,60. Surgery is reserved for refractory cases and consists of resection of the abnormal portion of the distal Achilles, resection of an irritating osseous prominence, and resection of the retrocalcaneal bursa28. In some cases, Achilles tendon lengthening may be needed58.
Treatment of acute Achilles ruptures remains controversial. In general, operative treatment allows improved functional outcome with a better chance of returning to the same performance level in athletes, faster return to work, and a decreased rerupture rate with some protocols28,61. Disadvantages include complications associated with surgery and greater cost28,61. To help mitigate the risk of infection and skin complications, surgery may be delayed to allow resolution of acute swelling62. Open, percutaneous, and mini-open repairs are used successfully with similar outcomes in returning to functional activities, patient satisfaction, and rerupture rates63,64. While percutaneous techniques have a lower incidence of wound complications, there is greater concern for sural nerve injury63,65,66. Mini-open repairs can allow a quicker return to activity than the standard open repair and with less serious wound complications67,68.
Nonoperative management lacks surgical complications, but there is a greater likelihood of patient dissatisfaction, and outcomes are extremely dependent on patient compliance and adherence to the rehabilitation protocol28. Nonoperative treatment most commonly begins with a period of casting and non-weight-bearing, which is often followed by functional bracing that allows a progressive decrease in plantar flexion69. Compared to casting and fixed immobilization, functional bracing allows an increased range of motion, earlier return to activity, and improved patient comfort69. An even more aggressive approach of immediate, full weight-bearing has been shown to result in a higher quality of life score than those allowed to weight-bear after 6 weeks.
Chronic Achilles ruptures often require surgical treatment because of poor healing potential, which leads to greater complication rates and poorer functional results28,70,71. The definition of a chronic tear varies in the literature with ranges from older than 4 weeks to 2.5 months72. Direct repair may be successful for tendons gaps of 3 cm and less72. Augmentation is frequently required for larger tendon gaps. Augmentation may be performed using soft-tissue, synthetic grafts, and allografts. The flexor hallucis longus tendon is often used for augmentation supplying vascularity to the tendon ends. Augmentation with soft tissue is often performed with aponeurotic fascial turndown flaps, advancement of the gastrocnemius-soleus complex aponeurosis, fascia lata, and plantaris tendon. Acellular human dermal tissue matrix has also been used73,74. Careful review of the surgical history and an understanding of the type of procedure performed can help to prevent confusion while interpreting the MRI examination (Fig. 17).
Figure 17:
65-year-old female following debridement and secondary repair of the Achilles tendon utilizing a gastrocnemius recession (lengthening) and gastrocnemius aponeurotic turn-down flap with FHL transfer to bridge the tendon defect following debridement. Sagittal T1 (left) and fat-suppressed T2-weighted (right) images demonstrate an intact FHL transfer (arrows). The distal flap created by the aponeurotic turn-down (arrowheads) overlies the distal Achilles tendon stump which is degenerated at is proximal aspect. A small amount of fluid (small arrow) is seen between the aponeurotic flap and the native distal Achilles tendon stump (red asterisk). On additional images continuity across the anastomosis was demonstrated medial and lateral to this slice. The proximal degenerated portion of the Achilles tendon is seen at the superior extent of the field of view (blue asterisk).
Recurrent Achilles tear
Recurrent tears of the Achilles are not uncommon following recurrent trauma (Fig. 18) or with incomplete healing of a prior tear or repair (Fig. 19)27.
Figure 18:
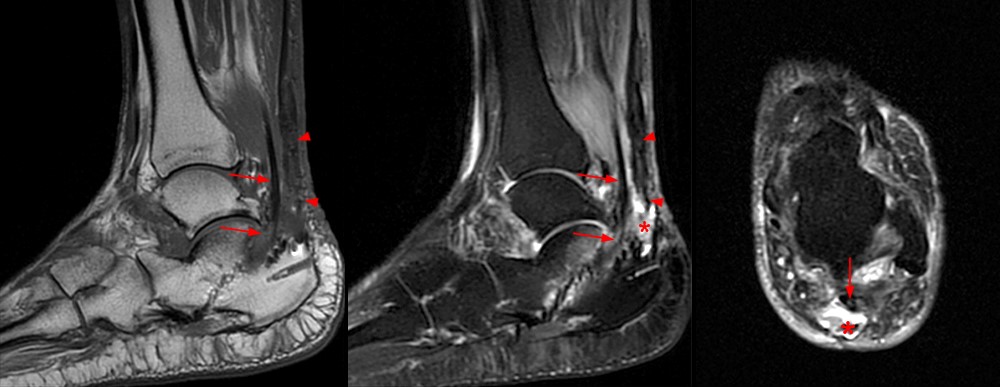
85-year-old male with a history of old healed Achilles tear and new onset pain and weakness after tripping. Sagittal T2-weighted fat-suppressed and T1-weighted images demonstrate a recurrent complete tear of the Achilles tendon with fluid signal filling the tendon gap (arrows). The Achilles tendon is diffusely scarred and thickened with small foci of heterotopic ossification in the distal tendon(arrowheads).
Figure 19:
64-year-old female with prior insertional Achilles rupture repair with gastrocnemius recession and FHL transfer presents with recurrent pain and loss of strength following a difficult recovery. Sagittal T1-weighted (left) and fat-suppressed T2-weighted sagittal (middle) and axial (right) images demonstrate an intact FHL transfer (arrows) with post-operative changes in the calcaneus. The Achilles tendon demonstrates heterogeneous intermediate and low signal with a recurrent distal complete tear filled with heterogeneous T2 hyperintense fluid and material (asterisks).
Conclusion
Achilles tendon injuries are common and increasing in frequency due to their association with an aging and active population. Many of the pathologies of the Achilles tendon share common pathways and frequently coexist at the time of patient presentation. The diagnosis of Achilles pathologies is largely clinical but MRI is valuable for evaluating patients who do not respond to conservative measures and for patients requiring surgical intervention. In these patients, MRI clearly depicts the variety of underlying pathologies encountered about the Achilles tendon. Proper assessment of tendon tears with MRI provides critical information to guide the treatment approach.
References
- O’Brien M. Functional anatomy and physiology of tendons. Clin Sports Med. 1992;11(3):505-520. ↩
- O’Brien M. The anatomy of the Achilles tendon. Foot Ankle Clin. 2005;10(2):225-238. doi:10.1016/j.fcl.2005.01.011. ↩
- Edama M, Kubo M, Onishi H, et al. The twisted structure of the human Achilles tendon. Scand J Med Sci Sport. 2015;25(5):e497-e503. doi:10.1111/sms.12342. ↩
- Ballal MS, Walker CR, Molloy AP. The anatomical footprint of the Achilles Tendon. Bone Jt J. 2014;96B(10):1344-1348. doi:10.1302/0301-620X.96B10.33771. ↩
- Snow SW, Bohne WH, DiCarlo E, Chang VK. Anatomy of the Achilles tendon and plantar fascia in relation to the calcaneus in various age groups. Foot ankle Int. 1995;16(7):418-421. doi:10.1177/107110079501600707. ↩
- O’Brien M. The Anatomy of the Achilles Tendon. Foot Ankle Clin. 2005;10(2):225-238. doi:10.1016/j.fcl.2005.01.011. ↩
- Schepsis A a, Jones H, Haas AL. Achilles tendon disorders in athletes. Am J Sports Med. 2002;30(2):287-305. doi:10.1097/BLO.0b013e3180e79c6a. ↩
- Luscombe K, Sharma P, Maffulli N. Achilles tendinopathy. Trauma. 2003;5(4):215-225. ↩
- Chen TM, Rozen WM, Pan W, Ashton MW, Richardson MD, Taylor GI. The arterial anatomy of the Achilles tendon: Anatomical study and clinical implications. Clin Anat. 2009;22(3):377-385. doi:10.1002/ca.20758. ↩
- Hess GW. Achilles Tendon Rupture: A Review of Etiology, Population, Anatomy, Risk Factors, and Injury Prevention. Foot Ankle Spec. 2010;3(1):29-32. doi:10.1177/1938640009355191. ↩
- Cummins EJ, Anson BJ. The structure of the calcaneal tendon (of Achilles) in relation to orthopaedic surgery, with additional observations on the plantaris muscle. Surg Gynecol Obstet. 1946;83:107-116. http://www.ncbi.nlm.nih.gov/pubmed/20988044. ↩
- Sookur PA, Naraghi AM, Bleakney RR, Jalan R, Chan O, White LM. Accessory Muscles: Anatomy, Symptoms, and Radiologic Evaluation. RadioGraphics. 2008;28(2):481-499. doi:10.1148/rg.282075064. ↩
- Luck MD, Gordon AG, Blebea JS, Dalinka MK. High association between accessory soleus muscle and Achilles tendonopathy. Skeletal Radiol. 2008;37(12):1129-1133. doi:10.1007/s00256-008-0554-0. ↩
- Cohen JC, Cummins JE, Anson JB, et al. Anatomy and biomechanical aspects of the gastrocsoleus complex. Foot Ankle Clin. 2009;14(4):617-626. doi:10.1016/j.fcl.2009.08.006. ↩
- Komi P V. Relevance of in vivo force measurements to human biomechanics. J Biomech. 1990;23:23-34. doi:10.1016/0021-9290(90)90038-5. ↩
- Mantel D, Flautre B, Bastian D, Delforge PM, Delvalle A, Leclet H. (Structural MRI study of the Achilles tendon. Correlation with microanatomy and histology). J Radiol. 1996;77(4):261-265. ↩
- Marshall H, Larkman DJ, Herlihy AH, Bydder GM. MR Imaging of the Achilles Tendon. 2002;(September):187-192. ↩
- SoiIa K, Aronen HJ, Pihlajam HK, Tirman PJ. High-Resolution MR Imaging of the Asymptomatic Achilles Tendon: New Observations. 1999;173. ↩
- Koivunen-Niemelä T, Parkkola K. Anatomy of the Achilles tendon (tendo calcaneus) with respect to tendon thickness measurements. Surg Radiol Anat. 1995;17(3):263-268. http://www.ncbi.nlm.nih.gov/pubmed/7502192. ↩
- Bottger1 BA, Schweitzer1 ME, Ei-Noueam1 ’ KI, Desai1 M. MR Imaging of the Normal and Abnormal Retrocalcaneal Bursae. 1998;170. ↩
- Theobald P, Bydder G, Dent C, Nokes L, Pugh N, Benjamin M. The functional anatomy of Kager’s fat pad in relation to retrocalcaneal problems and other hindfoot disorders. J Anat. 2006;208(1):91-97. doi:10.1111/j.1469-7580.2006.00510.x. ↩
- Pingel J, Petersen MCH, Fredberg U, et al. Inflammatory and Metabolic Alterations of Kager’s Fat Pad in Chronic Achilles Tendinopathy. Screen HR, ed. PLoS One. 2015;10(5):e0127811. doi:10.1371/journal.pone.0127811. ↩
- van Dijk CN, van Sterkenburg MN, Wiegerinck JI, Karlsson J, Maffulli N. Terminology for Achilles tendon related disorders. Knee Surgery, Sport Traumatol Arthrosc. 2011;19(5):835-841. doi:10.1007/s00167-010-1374-z. ↩
- Narvaez J, Narvaez J, Ortega R, Aguilera C, Sanchez A, Andia E. Painful Heel: MR Imaging Findings. RadioGraphics. 2000;20(2):333-352. ↩
- Berquist TH. Imaging of the Foot and Ankle. 3rd ed. (Berquist TH, ed.). Philadelphia: Wolters Kluwer/ Lippincott Williams &Wilkins; 2011. ↩
- Stoller D, Ferkel R, Li A, Mann R, Lindauer K. Magnetic Resonance Imaging in Orthopaedics and Sports Medicine – Google Books. 3rd ed. (Stoller DW, ed.). Baltimore: Lippincott Williams & Wilkins; 2007. ↩
- Pierre-Jerome C, Moncayo V, Terk MR. MRI of the Achilles tendon: a comprehensive review of the anatomy, biomechanics, and imaging of overuse tendinopathies. Acta radiol. 2010;51(4):438-454. ↩
- Uquillas C a., Guss MS, Ryan DJ, Jazrawi LM, Strauss EJ. Everything Achilles: Knowledge Update and Current Concepts in Management: AAOS Exhibit Selection. J Bone Jt Surg. 2015;97(14):1187-1195. doi:10.2106/JBJS.O.00002. ↩
- Paavola M, Kannus P, Järvinen TAH, Khan K, Józsa L, Järvinen M. Achilles tendinopathy. J Bone Joint Surg Am. 2002;84-A(11):2062-2076. ↩
- Lawrence DA, Rolen MF, Abi Morshed K, Moukaddam H, Moukaddam H. MRI of Heel Pain. AJR. 2013;200:845-855. doi:10.2214/AJR.12.8824. ↩
- Kannus P, Jozsa L. Histopathological Changes Preceding Spontaneous Rupture of a Tendon. J Bone Join Surgery Inc. 1991;73(10). ↩
- James SL, Bates BT, Osternig LR, Oregon E. Injuries to runners*. Am J Sports Med. 1978;6(2):40-50. ↩
- Fox JM, Blazina ME, Jobe FW, et al. Degeneration and rupture of the Achilles tendon. Clin Orthop Relat Res. 1975;(107):221-224. http://www.ncbi.nlm.nih.gov/pubmed/1132178. ↩
- Burns P, Hyer CF, Neufeld S, Penner M. 〈 Roundtable Discussion 〉 Chronic Achilles Tendon Disorders. 2008;(October):300-304. ↩
- Schweitzer ME, Karasick D. MR Imaging of Disorders of the Achilles Tendon. Am J Roentgenol. 2000;175(3):613-625. doi:10.2214/ajr.175.3.1750613. ↩
- Rodriguez CP, Goyal M, Wasdahl D a. Best cases from the AFIP: atypical imaging features of bilateral Achilles tendon xanthomatosis. Radiographics. 2008;28(7):2064-2068. doi:10.1148/rg.287085001. ↩
- Fahey JJ, Stark HH, Donovan WF, Drennan DB. Xanthoma of the Achilles Tendon SEVEN CASES WITH FAMILIAL HYPERBETALIPOPROTEINEMIA*. J Bone Jt Surg. 1973;55-A(6):1197-1212. ↩
- De Schepper AM, De Beuckeleer L, Vandevenne J, Somville J. Magnetic resonance imaging of soft tissue tumors. Eur Radiol. 2000;10(2):213-223. doi:10.1007/s003300050037. ↩
- Liem MS, Leuven JA, Bloem JL, Schipper J. Magnetic resonance imaging of Achilles tendon xanthomas in familial hypercholesterolemia. Skeletal Radiol. 1992;21(7):453-457. http://www.ncbi.nlm.nih.gov/pubmed/1439898. ↩
- Kang S, Thordarson DB, Charlton TP. Insertional Achilles tendinitis and Haglund’s deformity. Foot ankle Int. 2012;33(6):487-491. doi:10.3113/FAI.2012.0487. ↩
- Sella EJ, Caminear DS, McLarney EA. Haglund’s syndrome. J Foot Ankle Surg. 1998;37(2):110-4; discussion 173. doi:10.1016/S1067-2516(98)80089-6. ↩
- Johnson KW, Zalavras C, Thordarson DB. Surgical management of insertional calcific achilles tendinosis with a central tendon splitting approach. Foot ankle Int. 2006;27(4):245-250. doi:10.1177/107110070602700404. ↩
- Turlik MA. Seronegative arthritis as a cause of heel pain. Clin Podiatr Med Surg. 1990;7(2):369-375. http://www.ncbi.nlm.nih.gov/pubmed/2189542. ↩
- Pavlov H, Heneghan MA, Hersh A, Beth Goldman A, Vigorita V. The Haglund Syndrome: Initial and Differential Diagnosis1. ↩
- Yodlowski ML, Scheller AD, Minos L. Surgical treatment of Achilles tendinitis by decompression of the retrocalcaneal bursa and the superior calcaneal tuberosity. Am J Sports Med. 2002;30(3):318-321. ↩
- Johansson K, Lempainen L, Sarimo J, Laitala-Leinonen T, Orava S. Macroscopic Anomalies and Pathological Findings in and Around the Achilles Tendon: Observations From 1661 Operations During a 40-Year Period. Orthop J Sport Med. 2014;2(12):2325967114562371. doi:10.1177/2325967114562371. ↩
- DeVries JG, Summerhays B, Guehlstorf DW. Surgical Correction of Haglund’s Triad Using Complete Detachment and Reattachment of the Achilles Tendon. J Foot Ankle Surg. 2009;48(4):447-451. doi:10.1053/j.jfas.2009.03.004. ↩
- Corrao G, Zambon A, Bertù L, et al. Evidence of tendinitis provoked by fluoroquinolone treatment: a case-control study. Drug Saf. 2006;29(10):889-896. http://www.ncbi.nlm.nih.gov/pubmed/16970512. ↩
- Gross C. Acute Achilles Tendon Ruptures. Foot Ankle Int. 2016;37(2):233-240. doi:10711007. ↩
- Raikin SM, Garras DN, Krapchev P V. Achilles tendon injuries in a United States population. Foot ankle Int. 2013;34(4):475-480. ↩
- Academy A, Board OS, December D, et al. THE DIAGNOSIS AND TREATMENT OF ACUTE ACHILLES TENDON RUPTURE – GUIDELINE AND EVIDENCE REPORT. Vol 22.; 2009. doi:10.1016/j.injury.2014.06.022. ↩
- Ufberg J, Harrigan RA, Cruz T, Perron AD. orthopaedic pitfalls in the ED: Achilles tendon rupture. Am J Emerg Med. 2004;22(7):596-600. http://www.ncbi.nlm.nih.gov/pubmed/15666269. ↩
- Read JW, Peduto AJ. Tendon Imaging. Sports Med Arthrosc. 2000;8(1):32-55. http://www.scopus.com/inward/record.url?eid=2-s2.0-0002707999&partnerID=40&md5=0e320ee67f767e942d7531e338b32301. ↩
- Saglimbeni A, Fulmer CJ. Achilles tendon injuries. J Am Acad Orthop Surg. 1998;6(5):316-325. doi:10.1097/00004728-198905000-00021. ↩
- Steenstra F, van Dijk CN. Achilles Tendoscopy. Foot Ankle Clin. 2006;11(2):429-438. doi:10.1016/j.fcl.2006.02.001. ↩
- Leach RE, Schepsis a a, Takai H. Long-term results of surgical management of Achilles tendinitis in runners. Clin Orthop Relat Res. 1992;(282):208-212. ↩
- Ortmann FW, McBryde AM. Endoscopic bony and soft-tissue decompression of the retrocalcaneal space for the treatment of Haglund deformity and retrocalcaneal bursitis. Foot ankle Int. 2007;28(2):149-153. ↩
- McGarvey WC, Palumbo RC, Baxter DE, Leibman BD. Insertional Achilles tendinosis: surgical treatment through a central tendon splitting approach. Foot ankle Int / Am Orthop Foot Ankle Soc (and) Swiss Foot Ankle Soc. 2002;23(1):19-25. doi:10.1177/107110070202300104. ↩
- de Vos RJ, Weir A, van Schie HTM, et al. Platelet-rich plasma injection for chronic Achilles tendinopathy: a randomized controlled trial. JAMA. 2010;303(2):144-149. doi:10.1001/jama.2009.1986. ↩
- de Jonge S, de Vos RJ, Weir A, et al. One-year Follow-up of Platelet-Rich Plasma Treatment in Chronic Achilles Tendinopathy: A Double-Blind Randomized Placebo-Controlled Trial. Am J Sports Med. 2011;39(8):1623-1629. doi:10.1177/0363546511404877. ↩
- Erickson BJ, Mascarenhas R, Saltzman BM, et al. Is Operative Treatment of Achilles Tendon Ruptures Superior to Nonoperative Treatment?: A Systematic Review of Overlapping Meta-analyses. Orthop J Sport Med. ↩
- Beskin J, Sanders R, Hunter S, Hughston J. Surgical repair of Achilles tendon ruptures. Am J Sport Med. 1987;15(1):1-8. doi:10.1007/s004020050267. ↩
- Lim J, Dalal R, Waseem M. Percutaneous vs. open repair of the ruptured Achilles tendon–a prospective randomized controlled study. Foot ankle Int. 2001;22(7):559-568. doi:10.1177/107110070102200705. ↩
- Ng ES, Ng YO, Gupta R, Lim F, Mah E. Repair of acute Achilles tendon rupture using a double-ended needle. J Orthop Surg (Hong Kong). 2006;14(2):142-146. http://www.ncbi.nlm.nih.gov/pubmed/16914777. ↩
- Cretnik A, Kosanovic M, Smrkolj V. Percutaneous versus open repair of the ruptured Achilles tendon: a comparative study. Am J Sports Med. 2005;33(9):1369-1379. doi:10.1177/0363546504271501. ↩
- Tomak SL, Fleming LL. Achilles tendon rupture: an alternative treatment. Am J Orthop (Belle Mead NJ). 2004;33(1):9-12. ↩
- Bhattacharyya M, Gerber B. Mini-invasive surgical repair of the Achilles tendon-does it reduce post-operative morbidity? Int Orthop. 2009;33(1):151-156. doi:10.1007/s00264-008-0564-5. ↩
- Kakiuchi M. A combined open and percutaneous technique for repair of tendo Achillis. Comparison with open repair. J Bone Joint Surg Br. 1995;77:60-63. ↩
- Saleh M, Marshall PD, Senior R, MacFarlane a. The Sheffield splint for controlled early mobilisation after rupture of the calcaneal tendon. A prospective, randomised comparison with plaster treatment. J Bone Joint Surg Br. 1992;74(2):206-209. ↩
- Kosanović M, Brilej D. Chronic rupture of Achilles tendon: Is the percutaneous suture technique effective? Arch Orthop Trauma Surg. 2008;128(2):211-216. doi:10.1007/s00402-007-0514-5. ↩
- Pintore E, Barra V, Pintore R, Maffulli N. Peroneus brevis tendon transfer in neglected tears of the Achilles tendon. J Trauma. 2001;50(1):71-78. ↩
- Porter DA, Mannarino FP, Snead D, Gabel SJ, Ostrowski M. Primary repair without augmentation for early neglected Achilles tendon ruptures in the recreational athlete. Foot ankle Int. 1997;18(9):557-564. ↩
- Lee DK. Achilles Tendon Repair with Acellular Tissue Graft Augmentation in Neglected Ruptures. J Foot Ankle Surg. 2007;46(6):451-455. doi:10.1053/j.jfas.2007.05.007. ↩
- Barber FA, McGarry JE, Herbert M a, Anderson RB. A biomechanical study of Achilles tendon repair augmentation using GraftJacket matrix. Foot ankle Int / Am Orthop Foot Ankle Soc (and) Swiss Foot Ankle Soc. 2008;29(3):329-333. doi:10.3113/FAI.2008.0329. ↩