Clinical History:
A 52 year-old female presents with a 4-7 month history of right shoulder pain with decreased range of motion. No prior injury or surgery. Rule out rotator cuff tear/subacromial impingement.
Coronal oblique fat-suppressed T2-weighted and T1-weighted, sagittal oblique T2-weighted, and axial fat suppressed proton density-weighted images are submitted for review. What are the findings? What is the diagnosis?
Findings:
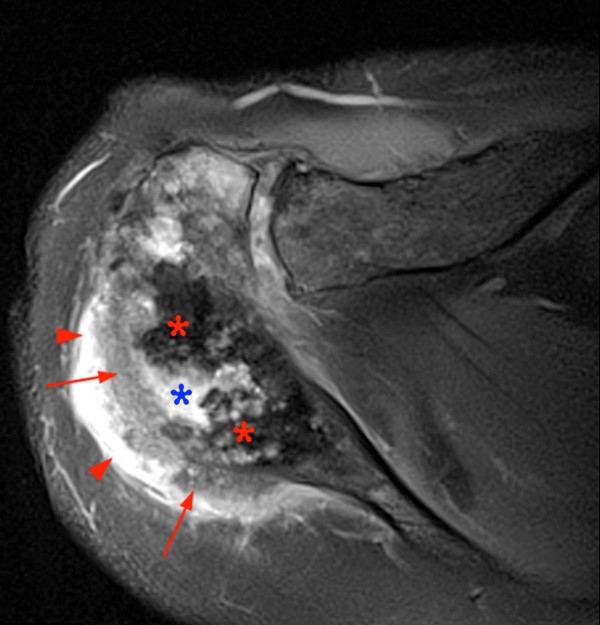
Figure 2:
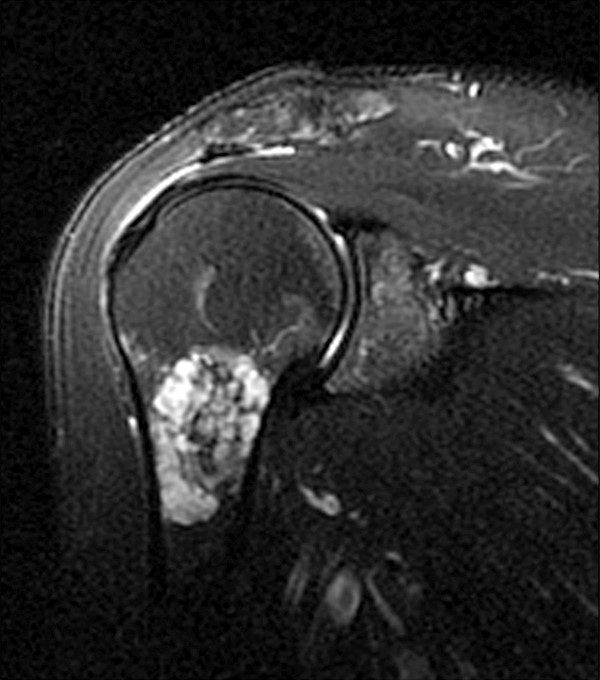
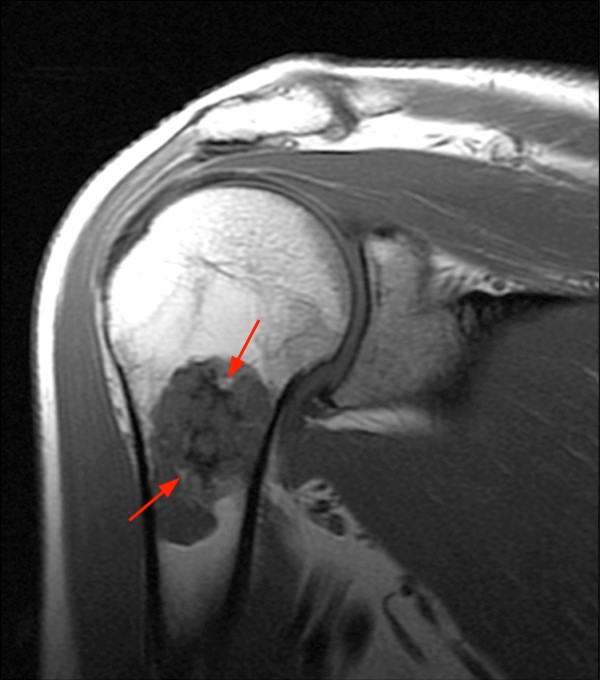
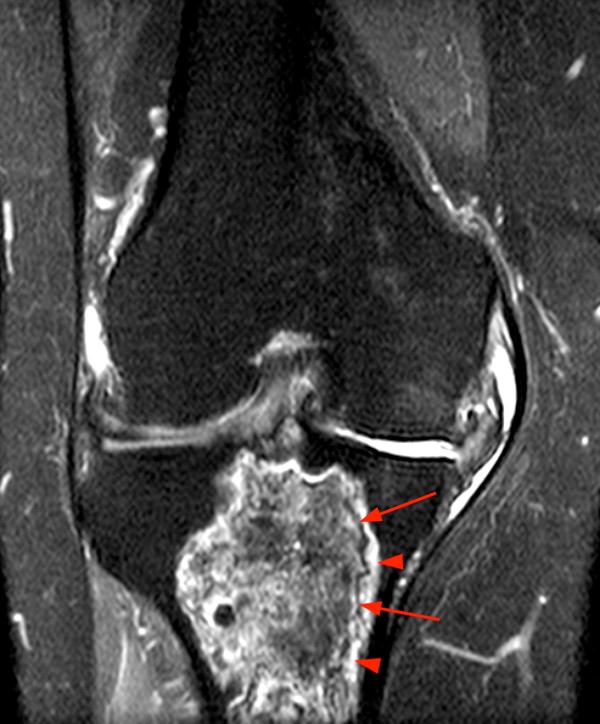
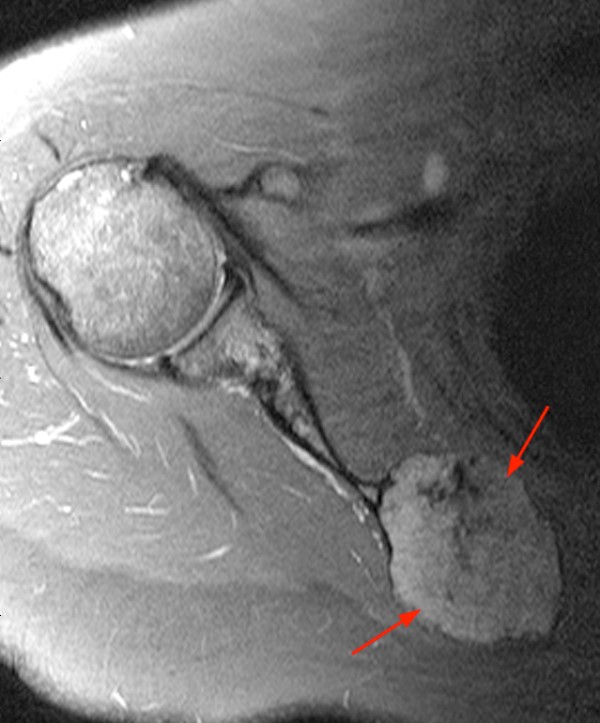
2a - The oblique coronal fat-suppressed T2-weighted image demonstrates a multilobulated predominantly fluid signal intensity lesion with sharply defined margins and irregular linear areas of low signal intensity that are hypointense to marrow (arrowheads). Note the lack of surrounding marrow edema or periostitis. Mild subacromial bursitis is also present.
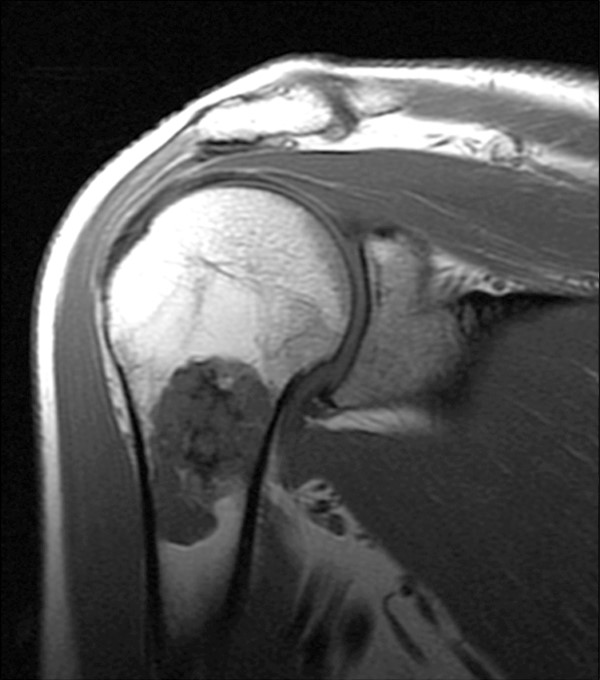
2b - The oblique coronal T1-weighted image clearly demonstrates irregular low signal intensities within the lesion and some small residual internal areas of normal fatty marrow signal intensity (arrows).
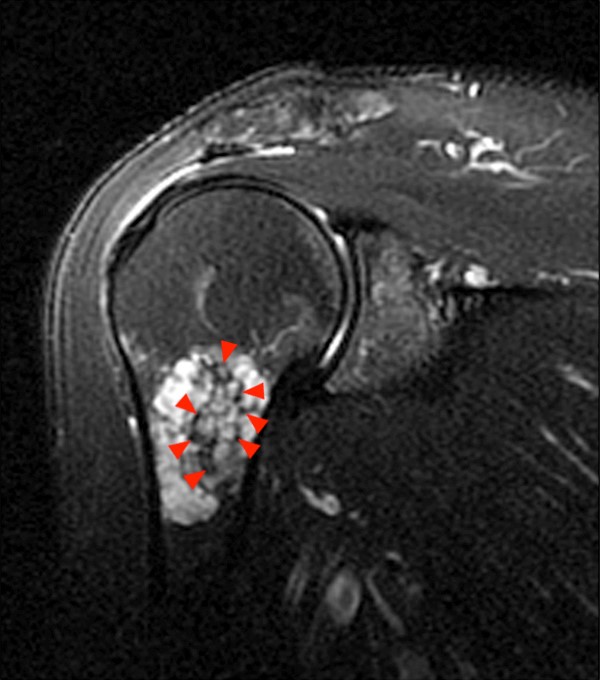
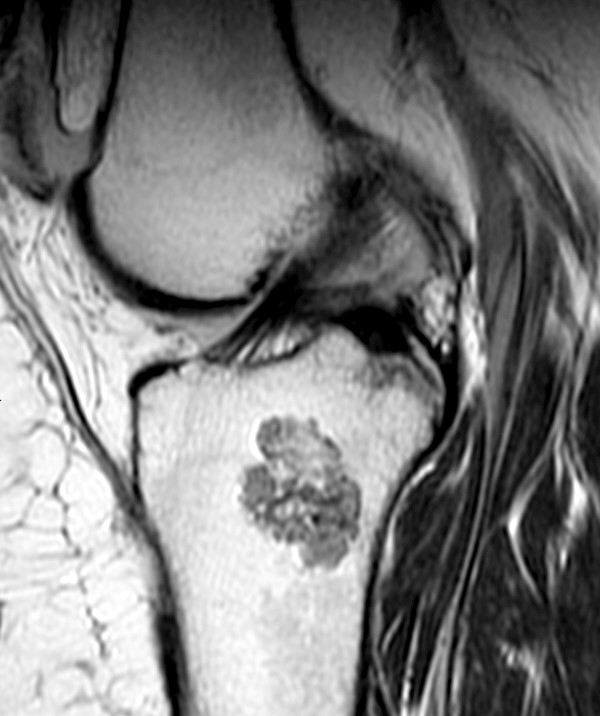
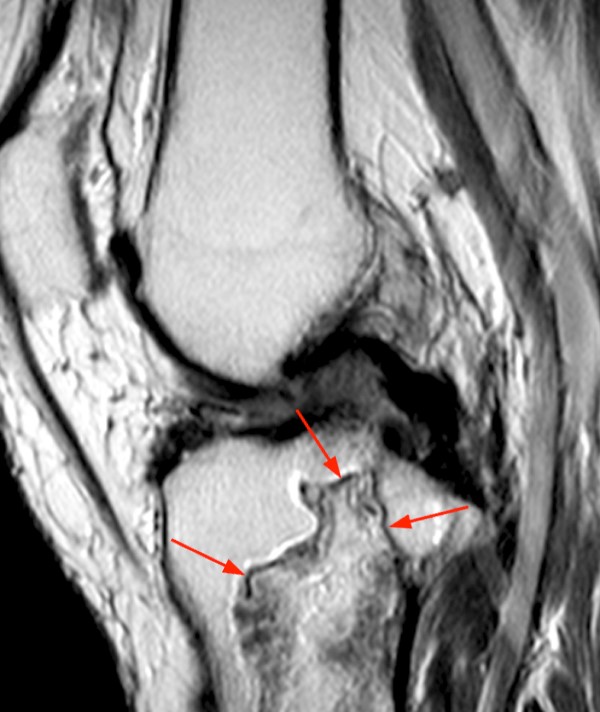
2c - The oblique sagittal T2-weighted image demonstrates the sharply defined lobulated margins, and low signal intensity “rings and broken rings” (arrowheads) characteristic of chondroid matrix.
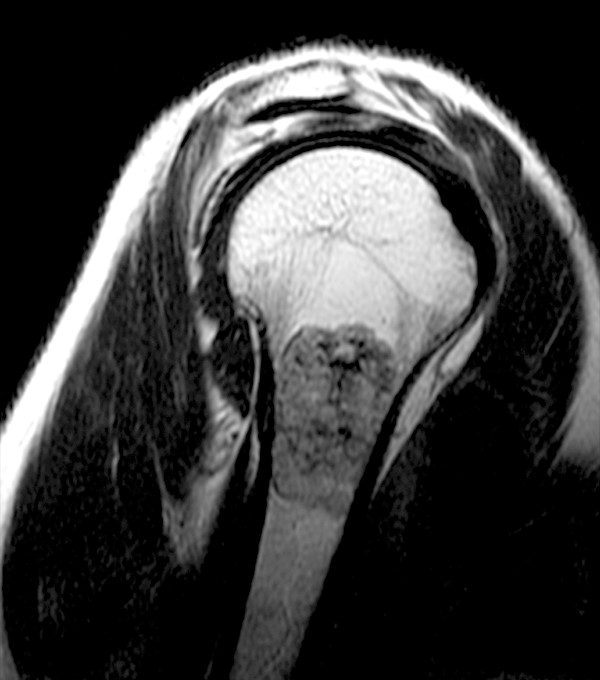
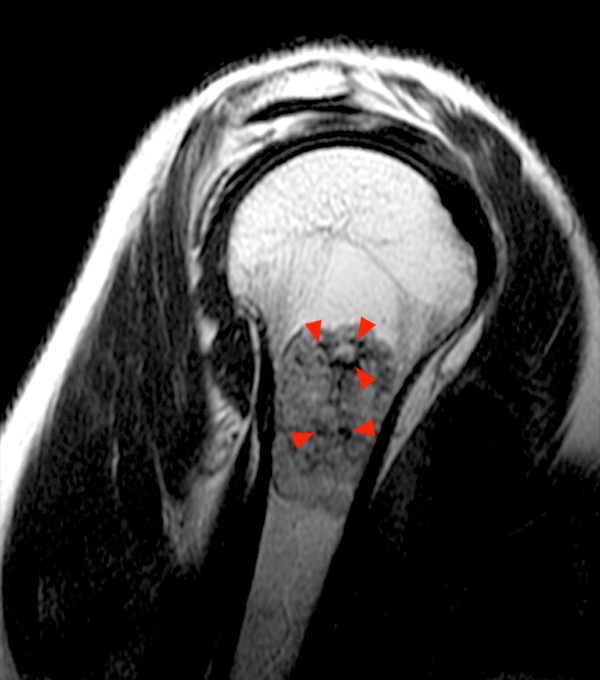
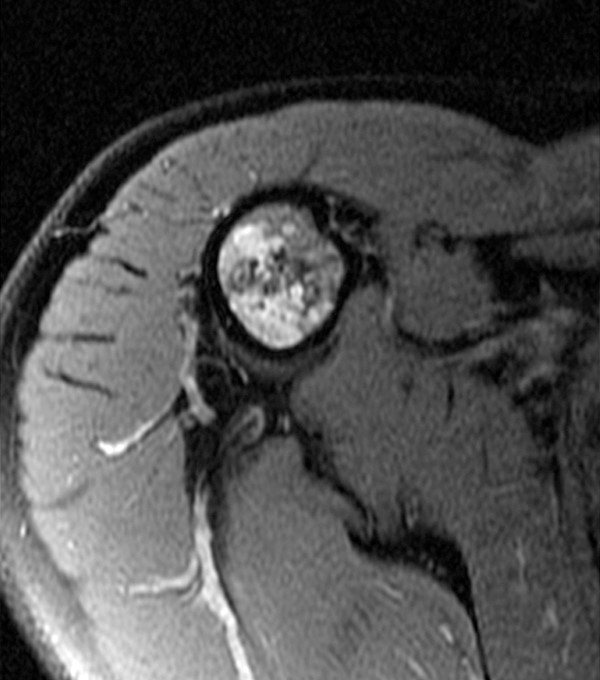
2d - The axial fat-suppressed proton density-weighted image demonstrates a lack of significant endosteal scalloping. No periostitis or cortical breakthrough is apparent.
Overall, the intramedullary lesion within the proximal humeral diaphysis demonstrates a narrow zone of transition, and well-defined lobulated margins with areas of fluid signal intensity. Areas of low signal intensity on all pulse sequences in patterns of rings and broken rings indicate areas of ossification. No surrounding marrow edema, cortical disruption or soft tissue mass is apparent.
Diagnosis
Enchondroma of the proximal humerus.
Introduction
Enchondroma and chondrosarcoma are both common osseous neoplasms. Enchondroma represents 12-24% of all benign bone tumors, and 3-10% of all bone tumors; chondrosarcoma accounts for 20-27% of primary bone sarcomas, and 8-17% of all bone tumors.1 Distinction between benign and malignant chondroid neoplasms is essential for correct patient management.
Enchondromas are most common in the small bones of the hands and feet (phalanges, metacarpals and metatarsals), while chondrosarcomas are rare in these locations. Chondrosarcomas are common in the spine and pelvis, while solitary enchondromas are very rare in these locations. The majority of chondroid lesions in the ribs and sternum are chondrosarcomas, and they are often seen in patients of a younger age. Thus, location can be a helpful discriminator. However, both lesions are extremely common in the appendicular skeleton (proximal to the metacarpals and metatarsals) where differentiation can be difficult in both imaging and pathologic evaluation.2
The most common location for both enchondroma and chondrosarcoma in the appendicular skeleton is the knee, followed by the humerus. Given the large numbers of MRIs obtained to rule out internal derangement of the knee and shoulder joints, the musculoskeletal radiologist is certain to come across chondroid lesions. The prevalence of incidental enchondroma on routine MRI of the knee for internal derangement has been found in one recent study to be 2.9%.1
MRI is not only a sensitive detector of chondroid lesions, but can be helpful in discriminating between benign and malignant lesions. It can also be used to guide the location of biopsies for the most accurate sampling.
Pathology and Imaging Findings
Enchdondromas are a benign neoplasm of the medullary canal composed of mature hyaline cartilage demonstrating small nuclei, abundant cytoplasm with absent or very rare mitotic activity. Chondrosarcoma is distinguished from enchondroma by the infiltration and entrapment of normal trabecular bone. Increasingly severe grades of chondrosarcoma demonstrate greater degrees of hypercellularity and cellular atypia. Enchondromas are thought to originate as cartilage rests derived from the physeal plate. The ring and arc pattern seen on imaging reflects the pattern of enchondral ossification around lobules of well-formed hyaline cartilage. Higher grade chondrosarcomas demonstrate larger areas of non-mineralized tissue with higher water content.2
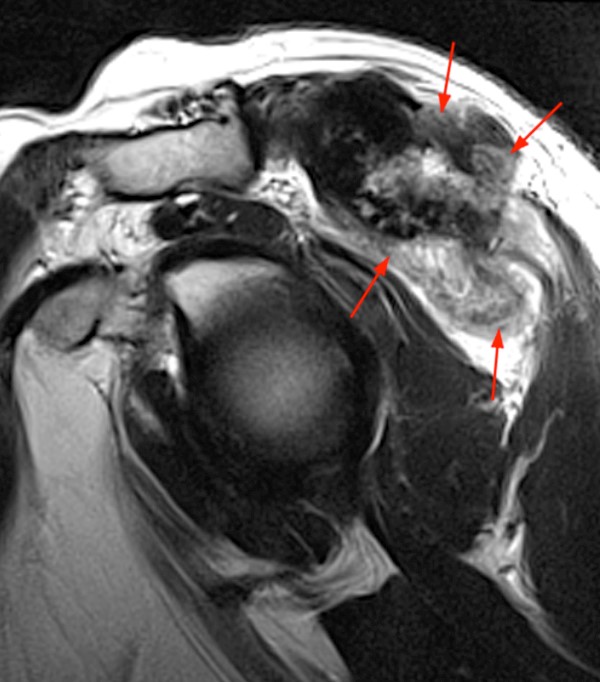
On MRI a typical enchdondroma demonstrates well-defined lobulated margins, large areas of fluid signal intensity with central linear areas of low signal intensity rings and broken rings corresponding to areas of ossification. Figure 3 demonstrates findings typical for small benign enchondroma.
Although chondroid matrix is highly recognizable, it can sometimes be difficult to distinguish from bone infarcts, especially on x-ray, since both have well-defined irregular margins with areas of fluid signal intensity and apparent calcification or ossification. The key to distinguishing between the two lesions is recognizing that the areas of mineralization are more central in chondroid lesions and peripheral in bone infarct. This is more easily appreciable on MRI than on x-ray. Figure 4 clearly demonstrates the peripheral area of calcification in a bone infarct.
Figure 4:
4a - A coronal fat-suppressed proton density-weighted image demonstrates a well-defined lesion with a serpiginous margin and a peripheral low signal intensity line (arrows) surrounded by fluid signal intensity (arrowheads). On this fat-suppressed image it is difficult to determine whether the central areas of low signal intensity correspond to areas of mineralization or fat.
4b - A sagittal non-fat suppressed T2-weighted image in the same patient more clearly reveals the predominantly peripheral location of the calcifications (arrows).
Identifying a lesion as chondroid is usually not difficult, but distinguishing between benign and malignant chondroid lesions can be a challenge. However, certain imaging findings have been found to be statistically significant in distinguishing these lesions. Findings more often associated with chondrosarcoma include the following: endosteal scalloping extending for greater than 2/3 of the cortical depth, endosteal scalloping extending for greater than 2/3 of the circumference of the lesion, (although a somewhat less reliable criterion); cortical remodeling and cortical breakthrough with associated soft tissue mass; and periostitis. (2) Peritumoral marrow edema is also more commonly associated with chondrosarcoma.3
Due to the small size of the phalanges, metacarpals and metatarsals, a high degree of endosteal scalloping is nearly always seen in chondroid lesions of the hands and feet. In those locations the most reliable indicator of malignancy is the presence of a soft tissue mass. Lesion size greater than 4.0 or 5.0 cm is more often seen in chondrosarcoma.2,3 A chondroid lesion less than 1.0 cm in size is referred to as a cartilaginous rest, an indolent lesion.
Both benign and malignant chondroid lesions are found most commonly in the diaphysis, followed by the metaphysis of long bones, with an epiphyseal location being quite unusual, and more commonly associated with chondrosarcoma. The extent of matrix mineralization and the pattern of post contrast enhancement discerned on MRI were not found to be statistically significant in distinguishing between benign and malignant lesions in earlier studies. However, more recent studies using dynamic gadolinium-enhanced MR sequences suggest a positive correlation between early and exponential enhancement and chondrosarcoma.2,4 Progressive loss of calcification in a lesion over time can be an indicator of an aggressive tumor.
Pain is more commonly associated with chondrosarcoma, and the pain is typically more intense and long standing, but it can be difficult to distinguish mechanical or inflammatory pain from tumor pain. In cases where the lesion demonstrates no imaging findings to suggest malignancy, intra-articular lidocaine injection can be used in an attempt to rule out a mechanical or inflammatory source of pain.
In our index case, the lesion measured 3.5 cm, and did not demonstrate any characteristics suggesting malignancy: no significant endosteal scalloping; there was no cortical breakthrough/soft tissue extension, no peritumoral edema or periostitis. It was read as a most likely benign lesion, unless pain could be directly attributed to the lesion. The patient had a follow up MRI 8 months later which demonstrated a small supraspinatus tendon tear for which she underwent rotator cuff repair. The patient continued to have intermittent pain for an additional 2 years, during which time she underwent 2 more additional shoulder MRI’s, all of which demonstrated a completely stable appearance of the lesion. The most recent MRI, nearly 3 years after the initial one, was obtained solely to evaluate the lesion, since the patient had no residual pain. Once again the lesion was unchanged, supporting the original impression of benign enchondroma.
Multiple Enchondroma Syndromes
Multiple enchondromas can occur in 3 different syndromes. Enchondromatosis, or Ollier’s disease, is a non-hereditary syndrome, often unilateral. It may regress or undergo malignant degeneration.5,6 Maffucci’s syndrome is another non-hereditary syndrome which consists of multiple enchondromas associated with soft tissues hemangiomas, typically located in the subcutaneous soft tissues, visible as bluish subcutaneous nodules clinically. The reported incidence of malignant transformation varies from 15-30% for Maffucci’s syndrome and has been reported at 30% for Ollier’s.7 The third syndrome is Metachondromatosis, a rare autosomal dominant inherited condition consisting of enchondromas and exostoses. However the exostoses in this condition point towards rather than away from the joint, and may regress after childhood.8 There is a predilection for the hands and feet in all 3 syndromes.
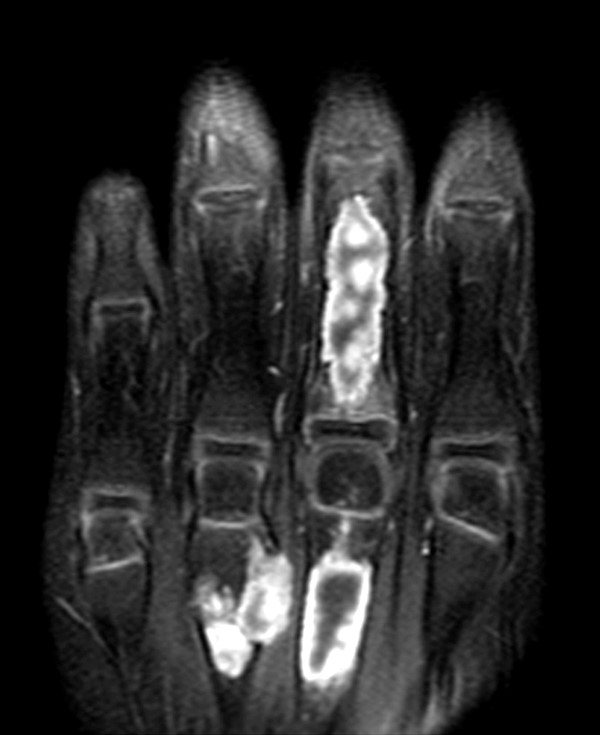
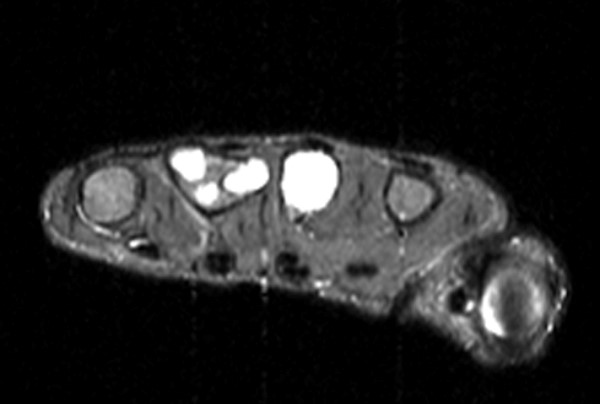
Figure 5 shows a typical appearance and distribution of Ollier’s disease in the hand, with multiple expansive, lytic deforming lesions. Recall that a greater degree of endosteal scalloping and remodeling is typical in the small bones of the hands and feet, and cannot be considered a reliable sign of malignancy.
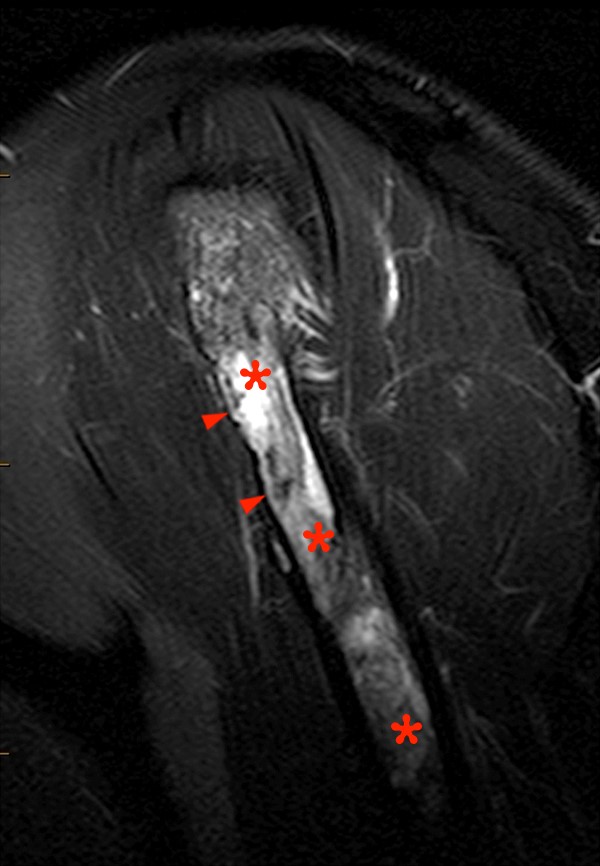
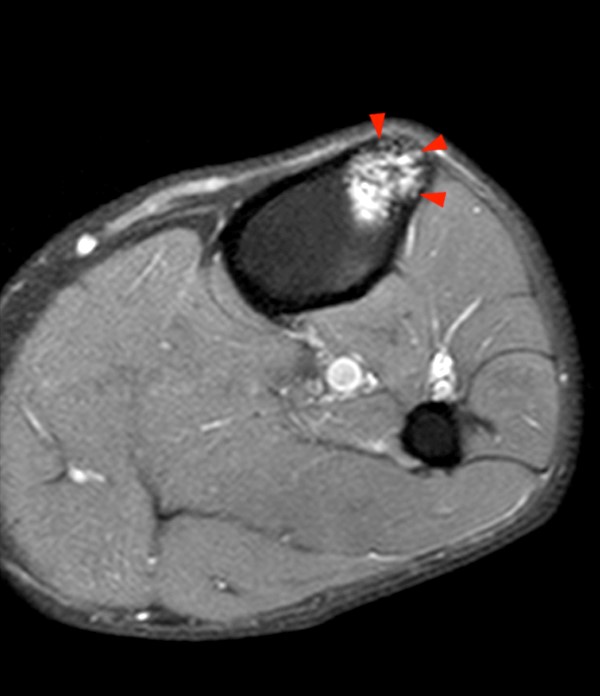
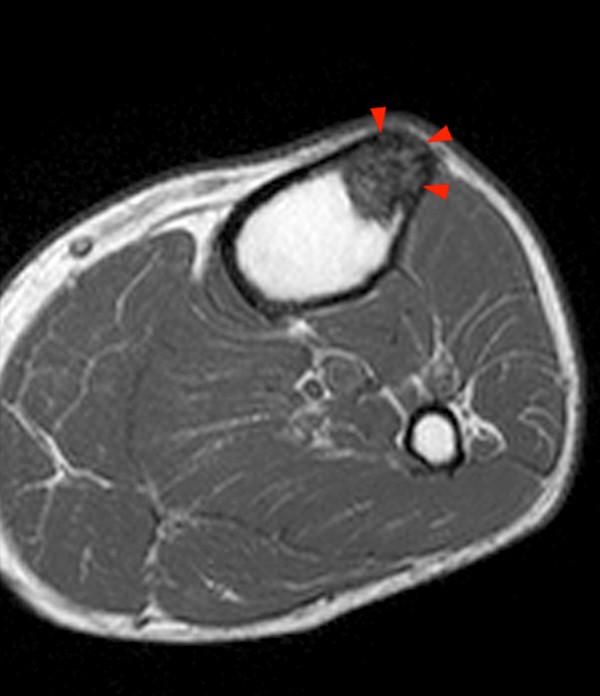
Figure 6 demonstrates multiple chondroid lesions with a large scapular lesion demonstrating cortical breakthrough and a large soft tissue mass. This is biopsy proven chondrosarcoma arising in Ollier’s disease.
Figure 6:
6a - A fat-suppressed axial proton density-weighted image in a patient with Ollier’s disease demonstrates a large soft tissue mass (arrows) arising from the scapula.
6b - An oblique sagittal fat-suppressed T2-weighted image through the humeral diaphysis in the same patient demonstrates a large chondroid lesion (asterisks) with significant endosteal scalloping (arrowheads) but no cortical breakthrough or soft tissue mass.
Management
A lesion demonstrating no signs of aggressiveness on MRI can be followed with imaging, MRI generally deemed to be the most accurate. The first follow-up examination should be performed within 3 to 6 months and yearly thereafter. There is no agreed upon duration for follow-up, but one source has suggested two decades if detected after age 25.9 Any radiologic change or new onset of symptoms related to the lesion should prompt a new imaging evaluation, and a biopsy, if significant imaging changes are confirmed. The difficulty of distinguishing benign from malignant lesions and proposing a clear management protocol can be appreciated by the fact that it is not possible to distinguish at pathological examination between an “active enchondroma” and a grade I chondrosarcoma. This has led to a further categorization of abnormal imaging findings discussed above into those which may be encountered in both active enchondroma and grade I chondrosarcoma (active findings) versus those associated with Grade 2 or 3 Chondrosarcoma (aggressive findings). Active findings include endosteal scalloping >2/3 of the cortical thickness and >2/3 of the lesion length, hyperostosis, pain related to the lesion, and early and exponential enhancement of dynamic gadolinium enhanced MRI. Aggressive findings include pathologic fracture with minimal trauma, lamellar or speculated periosteal reaction, permeative osteolysis, cortical destruction, and soft tissue mass.10 The number and degree of “active” versus “aggressive” findings, may factor into the decision to perform simple biopsy, intralesional surgery or wide resection. However, there have been no studies of sufficient size or length of follow up to definitively indicate the most successful management plan.
Figure 7a-b demonstrates a small lesion (2.5 cm) which demonstrates endosteal scalloping of greater than 2/3 cortical depth anteriorly, with questionable bony remodeling, findings which suggest an active enchondroma, with an otherwise generally benign appearance.
Most chondrosarcomas are well-differentiated low grade tumors. But up to 11% undergo anaplastic transformation into high-grade noncartilaginous sarcomas arising within a preexisting low to intermediate grade chondrosarcoma. Such tumors are highly malignant, with patient survival times ranging from 5-18 months. The non-cartilaginous component determines the rate of growth and metastases, and thus the prognosis. The most common noncartilaginous component is osteosarcoma (70%) either fibroblastic or osteoblastic, followed by fibrosarcoma (24%) and malignant fibrous histiocytoma (4%). This “tumor bimorphism” is demonstrated when a chondroid matrix exists adjacent to a markedly different area which may be purely lytic, osteoid matrix, or a large unmineralized soft tissue mass.11 This can lead to inaccurate diagnosis if only one portion of the tumor is biopsied. A central lytic or osteoblastic portion of the tumor may provide a very different pathologic diagnosis compared with a more peripheral sample, which may even demonstrate chondroid matrix interspersed between preserved trabecular bone, falsely suggestive of a benign enchondroma. MRI can be used to guide biopsy to the most representative portion of the tumor. In some cases it may be necessary to biopsy the dedifferentiated portion and the more peripheral typically chondroid appearing portion of the tumor to prove that it is a dedifferentiated chondrosarcoma. When the imaging demonstrates chondroid matrix but the biopsy demonstrates a noncartilaginous spindle cell tumor, the possibility of dedifferentiated chondrosarcoma should be considered. (11) Chondroid matrix can also be seen in osteosarcoma with chondroid dedifferentiation, and may be indistinguishable from chondrosarcoma.12
Figure 8 demonstrates an aggressive tumor in the acromion of a 65 year-old male. There is peripheral chondroid matrix, central fluid signal intensity apparently devoid of chondroid matrix, cortical destruction and a large soft tissue mass. The biopsy revealed osteosarcoma.
Figure 8:
8a - An axial fat-suppressed proton density-weighted image demonstrates areas of chondroid matrix (red asterisks), and some more centrally located predominantly lytic areas (blue asterisk), with cortical destruction, surrounding soft tissue edema (arrowheads) and a soft tissue mass (arrows).
8b - The sagittal T2-weighted image better demonstrates the associated soft tissue mass (arrows).
Conclusion
Chondroid lesions are extremely common incidental findings on musculoskeletal MRI studies, since the femur and humerus are the most common location for chondroid tumors of the appendicular skeleton. Malignant degeneration of a solitary benign enchondroma is rare, but has been reported. It is more common in syndromes demonstrating multiple enchondromas.
Chondroid matrix is not difficult to recognize, but differentiating benign enchondroma from low grade chondrosarcoma is sometimes impossible both radiographically and pathologically. However, certain MRI findings have been found to be significantly associated with active enchdondroma or low grade chondrosarcoma, so MRI is an excellent way to monitor tumor stability. In cases of more obvious radiographic malignancy, demonstrating cortical breakthrough and soft tissue tumor, MRI may detect tumor dimorphism and be useful in directing biopsy to the most active area of the tumor.
References
- Walden MJ, Murphey MD, Vidal A. Incidental enchondromas of the knee. American Journal of Roentgenology 2008; 190:1611-1615. ↩
- Murphey MD, Flemming DJ, Boyea SR, et al. Enchondroma versus chondrosarcoma in the appendicular skeleton: differentiating features. RadioGraphics 1998; 18:1213-1237 ↩
- Janzen L, Logan PM, O’Connell JX, et al. Intramedullary chondroid tumors of bone: correlation of abnormal peritumoral marrow and soft-tissue MRI signal with tumor type. Skeletal Radiology 1997; 26:100-106 ↩
- Geirnaerdt MJA, Hogendoorn PCW, Bloem JL, Taminiau AHM, and Van der Woude H-J. “Cartilaginous tumors: fast contrast-enhanced MR imaging,” Radiology, vol. 214, no. 2, pp. 539–546, 2000. ↩
- Kumar A, Jain VK, Bharadwaj M, Arya RK. Ollier Disease: Pathogenesis, Diagnosis, and Management. orthopaedics. 2015 Jun. 38 (6):e497-506. ↩
- Mainzer F, Minagi H, Steinback HL. The variable manifestations of multiple enchondromatosis. Radiology May 1971;99:377-388 ↩
- Flach HZ, MD, Ginai AZ, and J. Wolter Oosterhuis JW. Best cases from the AFIP Maffucci Syndrome: Radiologic and Pathologic Findings1 RadioGraphics 2001; 21,5: 1311-1316. ↩
- Kennedy LA. Metachrondromatosis. Radiology July 1983; 148: 117-118 ↩
- Brien, E.W., Mirra, J.M., and Kerr, R. Benign and malignant cartilage tumors of bone and joint: their anatomic and theoretical basis with an emphasis on radiology, pathology and clinical biology. I. The intramedullary cartilage tumors. Skeletal Radiology. 1997; 26: 325–353 ↩
- Parlier-Cuau C, Bousson V, et al. When should we biopsy a solitary central cartilaginous tumor of long bones? Literature review and management proposal. European journal of radiology Jan 2011: 77;1: 6-12. ↩
- Littrell LA, Wenget DE, Wold LE et al RadioGraphics, CT and MR imaging feature of dedifferentiated chondrosarcomas: A retrospective review of 174 de novo cases. Radiographics 2004; 24:5, 1397-1409 ↩
- Wadhwa N. Osteosarcoma: diagnostic dilemmas in histopathology and prognostic factors. Indian J Orthop. 2014; 48(3): 247-254. ↩