Clinical History:
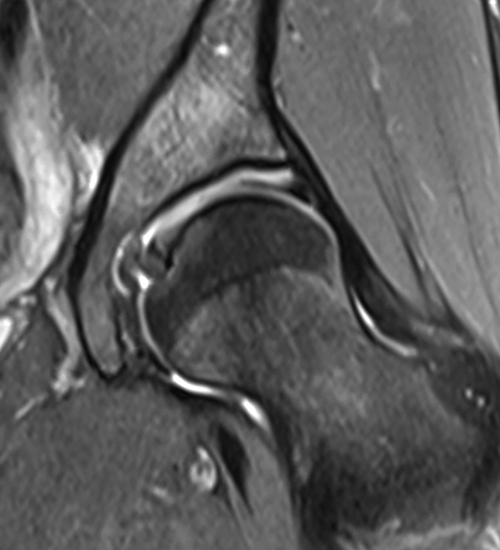
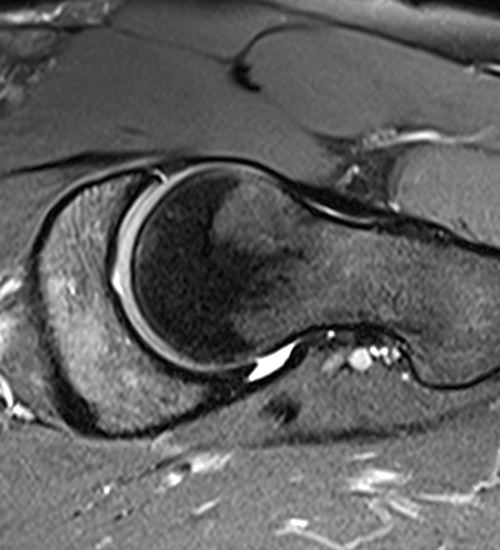
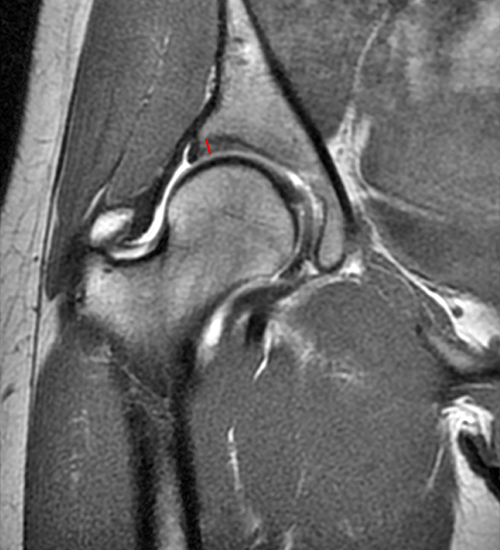
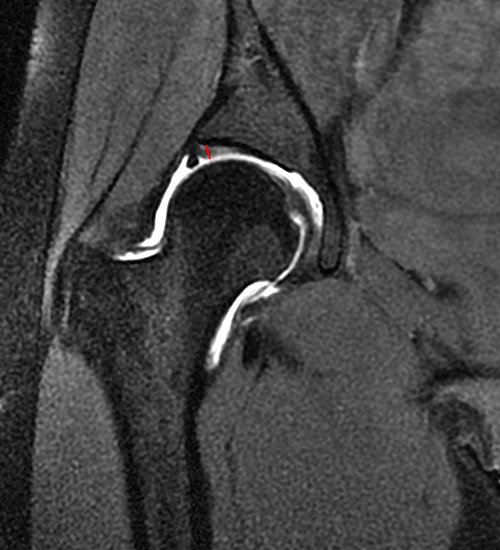
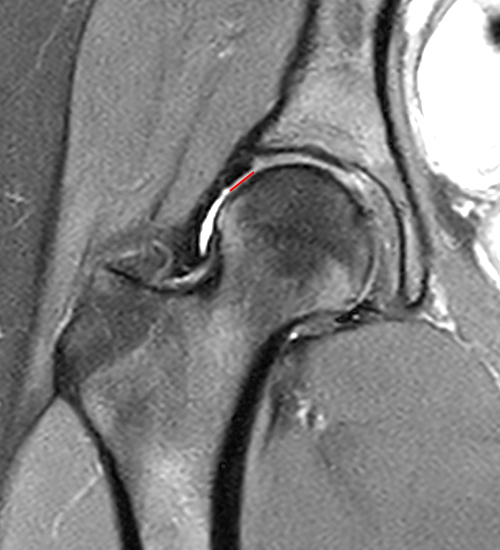
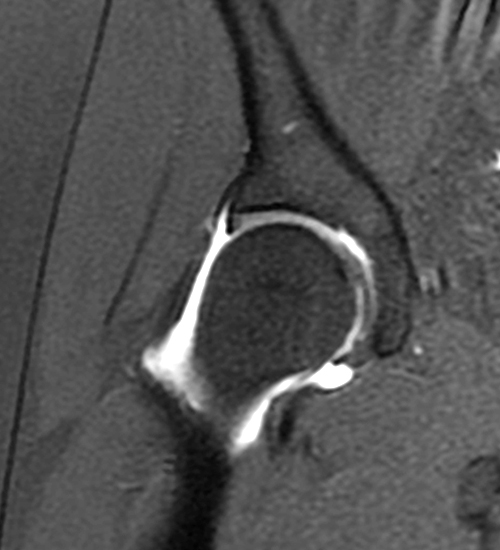
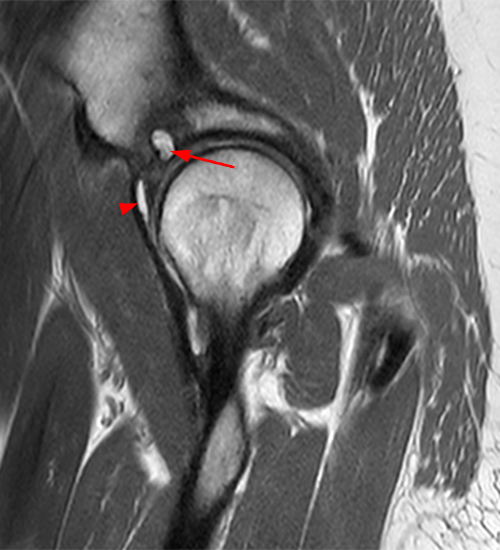
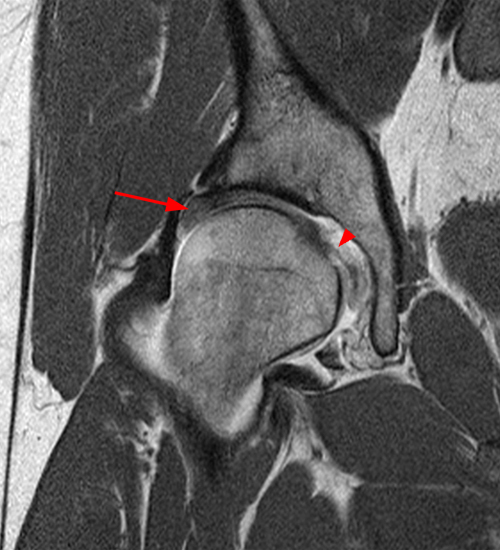
A 30 year-old female presents with chronic hip pain to evaluate for femoroacetabular impingement. Coronal (1A) and axial oblique (1B) fat-suppressed proton density-weighted images are provided. What are the findings? What is your diagnosis?
Findings
Figure 2:
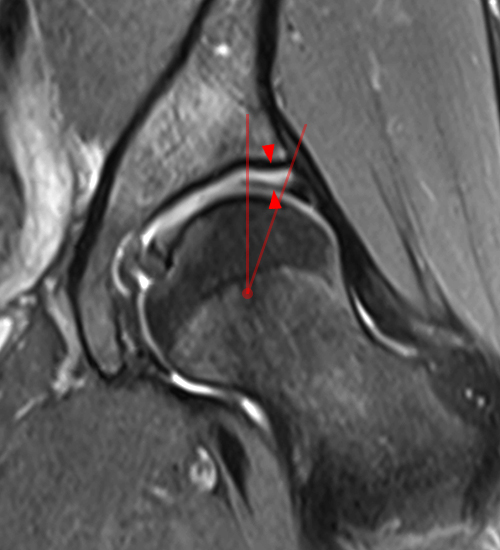
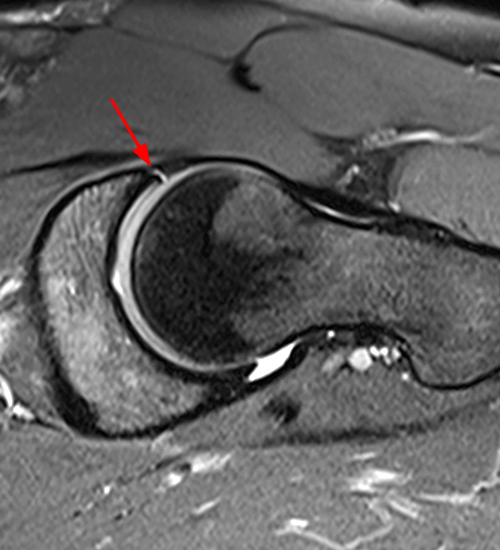
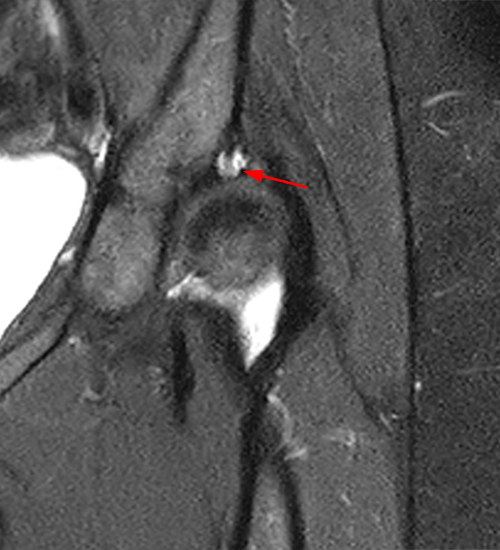
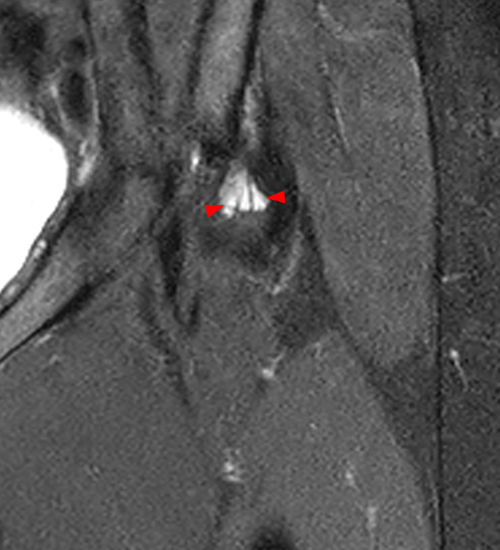
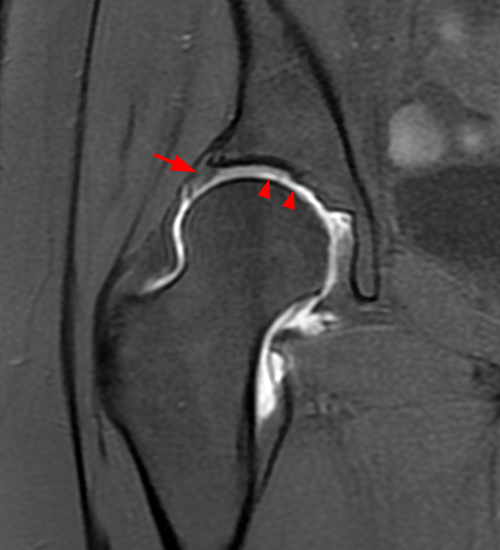
The coronal fat-suppressed proton density-weighted image (2A) demonstrates increased thickness of the superolateral acetabular cartilage (arrowheads). The lateral center edge angle measures 20 degrees. The axial oblique image (2B) demonstrates a tear across the base of the anterior labrum (arrow).
Diagnosis
Borderline hip dysplasia with features of microinstability.
Introduction
Developmental dysplasia of the hip (DDH) is present in an estimated 20 to 40% of patients with osteoarthritis of the hip.1 Despite aggressive screening for hip dysplasia in the infant, a significant number of patients are not diagnosed until adulthood with an estimated prevalence of 0.1%.2 Abnormalities associated with hip dysplasia range from subtle insufficiency of acetabular coverage to marked instability and dislocation. Surgical reorientation of the pelvis or total hip replacement is typically used to treat patients with frank dislocation or instability of the hip. A large subset of patients presents with hip pain but with milder dysplastic findings. The treatment in these patients is controversial and hinges on identifying evidence for instability.
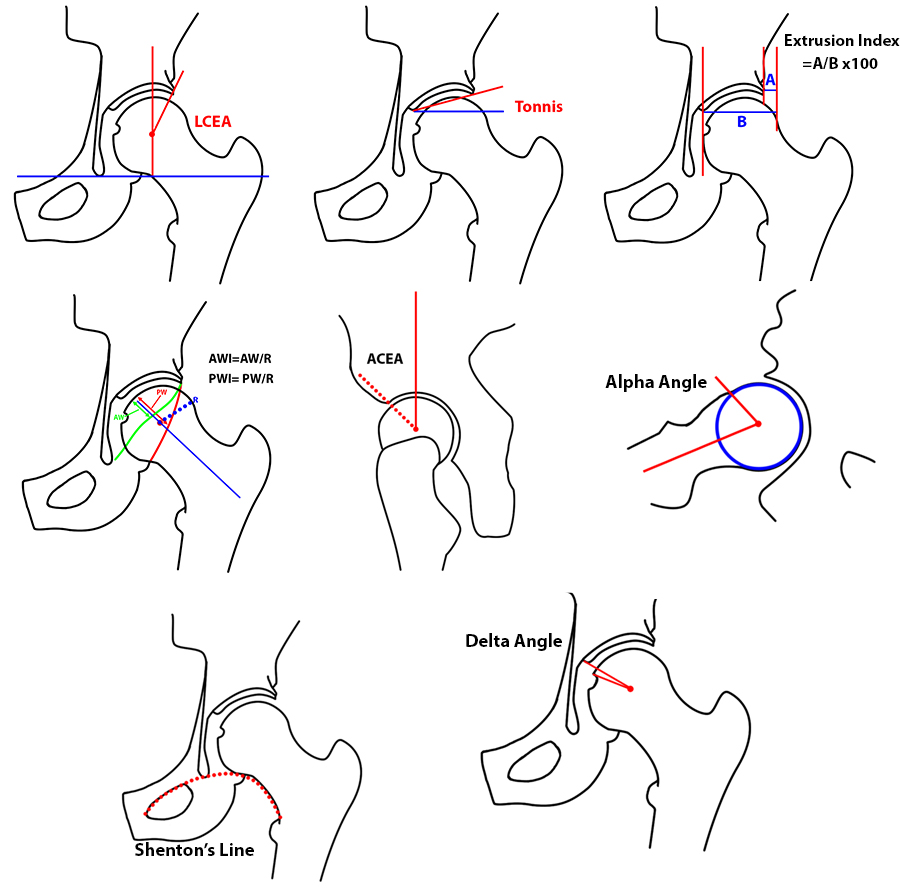
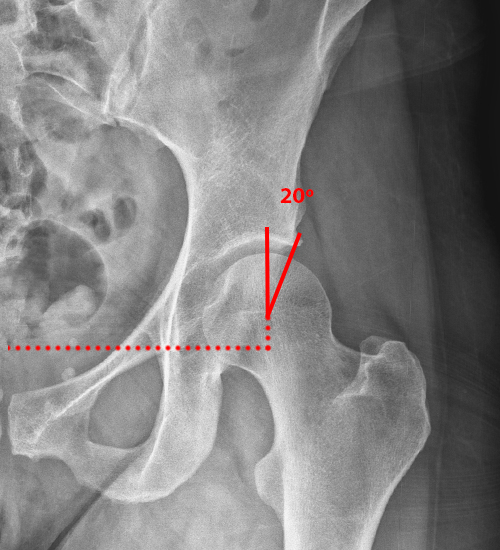
The lateral center edge angle (LCEA) is obtained from an anterior-posterior (AP) view of the pelvis and was first described by Wiberg in 1939 to quantify lateral acetabular coverage. The LCEA is the mainstay for evaluating and diagnosing DDH. Wiberg defined an angle of less than 20 degrees as being dysplastic and an angle of 20-25 degrees as “borderline dysplasia” (BDH).3 These definitions are still popular today, but over time the LCEA ranges that define DDH and BDH have become less clear with overlap, emphasizing the fact that LCEA is not sufficient to characterize the acetabular deficiencies that can contribute to hip instability. The pathophysiology, characteristic imaging features, and clinical implications of milder forms of hip dysplasia in the adult will be the focus of this Web Clinic.
Anatomy and Pathophysiology
Hip dysplasia results from an abnormal size, shape, proportion or alignment of the acetabulum or the femoral head.4 The acetabulum is most often deficient laterally or globally, but deficiencies of acetabular coverage can selectively occur anteriorly and posteriorly.5 Mechanical problems arise because of the smaller surface contact area between the acetabulum and femoral head and lateralization of the hip center of rotation. Abnormally focused stresses occur at the bony acetabular rim and acetabular labrum, and compensatory hypertrophy can occur in the capsule, labrum, and articular cartilage.4,6,7,8 If uncorrected, labral tears or detachment, acetabular rim stress fractures, and acetabular rim chondral defects can occur, further destabilizing the hip and leading to lateral subluxation of the femoral head, joint incongruence, and eventually secondary osteoarthritis.
Proximal femoral abnormalities are common. The severity of deformity of the femoral head corresponds to the severity of the acetabular dysplasia.9 The femoral head is usually smaller than normal with a decreased epiphyseal height and an ellipsoid contour which is greatest in the medial-lateral plane.10 A thicker femoral neck is common with a decreased femoral head-neck offset resulting in cam morphology. Coxa valga is common, though a normal femoral neck-shaft angle or coxa vara can be seen. Increased femoral anteversion is a frequent finding and can contribute to instability.
Clinical Findings
Early diagnosis of dysplastic hip requires a high index of suspicion and should be considered in all young adults with hip pain. Females are more often affected than males although the female preponderance is less pronounced than in infantile hip dysplasia. In addition, bilateral hip involvement is more commonly seen in the adult with DDH and BDH than in infantile DDH, leading some authors to suggest that adult hip dysplasia represents a different demographic population than the infantile variety of DDH11. A positive family history of hip problems or early age of total hip replacement is common.
Symptom onset is usually insidious with pain in the groin. Pain is commonly activity-related with associated mechanical symptoms (clicking, locking, popping), and the patient may complain of a feeling of instability. Clicking or locking is often the result of a labral tears or detachment. Clicking or snapping can also occur from motion of the iliopsoas tendon over the uncovered femoral head.11 Physical examination findings include a positive impingement test with flexion, adduction, and internal rotation (FADIR) of the hip indicating a labral abnormality.4 Musculotendinous pain is often elicited with palpation, stretching, or resistance strength testing and is most prevalent at the iliopsoas complex followed by the hip abductor muscles.12 Instability testing includes the abduction-hyperextension-external rotation (AB-HEER), hyperextension-external rotation (HEER), and prone instability tests.13
Radiographic Findings
Because of their low cost and ready accessibility, radiographs are the primary imaging modality used to evaluate patients with hip pain and to establish the diagnosis of hip dysplasia. Standardization of projection factors is imperative to prevent measurement errors. Variances in the distance of the x-ray tube from the patient, film-tube distance, patient-to-film distance, centering of the x-ray, and pelvic positioning affect the accuracy and reproducibility of the radiographic projection. Radiographs are routinely used to assess acetabular anatomy, femoral anatomy, and joint congruency. Familiarity with the variety of measurements obtained on radiographs is helpful to understand the associated osseous abnormalities and many of these measurements can be performed with CT or MRI as well. The list of measurements and angles continues to grow and evolve, and an exhaustive description of the various measurements is beyond the scope of this Web Clinic.
Acetabular anatomy
Lateral acetabular coverage as defined by the lateral center edge angle (LCEA) on the AP view of the pelvis is the most commonly used measurement to make a radiologic diagnosis of hip dysplasia. However, the LCEA only measures lateral coverage of the femoral head. Hip dysplasia is a 3-dimensional acetabular deficiency that can affect the acetabulum anteriorly, posteriorly, laterally, and globally.5 Additional radiographic measures beyond the LCEA are therefore necessary for a complete evaluation of the bony anatomy. The primary radiographic views used to evaluate acetabular coverage are an AP pelvis and the false profile view of Lequesne and de Seze.
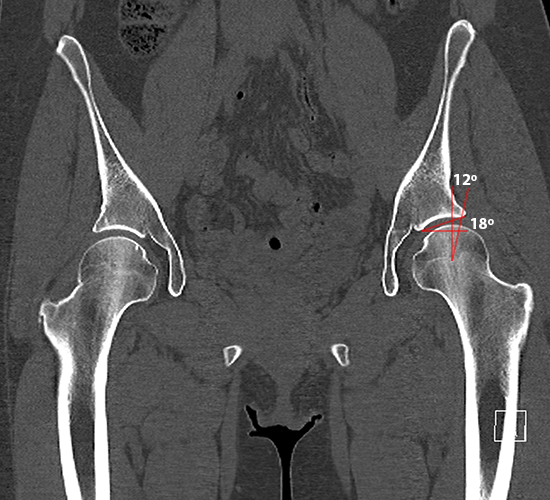
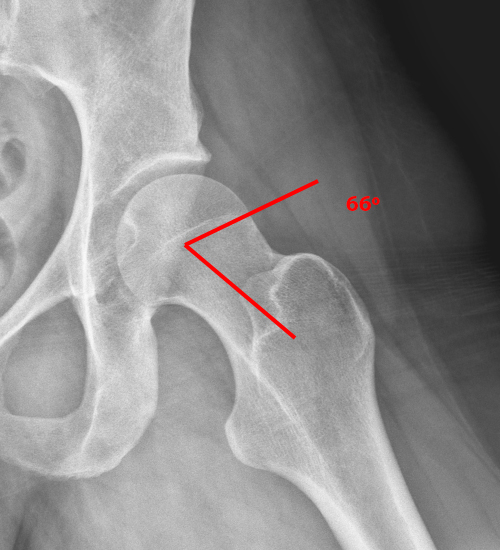
The lateral center edge angle (LCEA) (Figure 3) is obtained from an AP view of the pelvis and formed by the intersection of a line perpendicular to the teardrop line drawn through the center of the femoral head and an intersecting line extending from the center of the femoral head to the lateral-most bony prominence of the superior acetabulum. The normal center edge angle is considered to be >25 degrees. As originally defined by Wiberg, a center edge angle of less than 20 degrees is consistent with DDH, and an angle of 20-25 degrees represents borderline dysplasia of the hip (BDH).3 Currently there is a less well defined range published in the literature with a lower range of 18 degrees proposed by some surgeons. In the original description the lateral-most protruding bone was used to create the angle, however utilizing this landmark can lead to an overestimation of the extent of femoral head coverage. A refined lateral center edge angle assessment proposed by Ogata utilizes the lateral margin of the sourcil.14 The sourcil (French translation: eyebrow) is the subchondral bony condensation at the acetabular roof formed as the result of chronic compressive stresses associated with weight bearing. The normal sourcil is concave and congruent with the femoral head, but may be flattened, upsloping, or abbreviated in width in hip dysplasia. Utilizing the sourcil as a landmark to measure the LCEA is felt to provide a more reliable measure of acetabular coverage.14 When measuring the LCEA, the lateral-most cortex or lateral sourcil should be indicated as the reference point.
The Tonnis angle, Acetabular Index (AI), or Horizontal Toit Externe (HTE) (horizonal external roof) (Figure 3) evaluate the acetabular inclination obtained from an AP view of the pelvis and measures the inclination of the acetabular roof. The angle is formed by a line tangential to the medial and lateral margins of the sourcil and a line parallel to the transverse pelvic axis. A range of normal values is found in the literature with measurements greater than 10-14 degrees considered to be dysplastic.15,16,17
The extrusion index (EI) (Figure 3) measures the percentage of the femoral head which is uncovered by the acetabulum. An EI of >27% is defined as dysplastic.18
The acetabular wall index (AWI) (Figure 3) is used to quantify anterior or posterior acetabular coverage on a standardized AP view of the pelvis. The ratio of the width of the anterior or posterior acetabular wall is divided by the femoral head radius to calculate the anterior or posterior acetabular wall index. An AWI < 0.30 defines anterior undercoverage and a PWI < 0.80 defines posterior undercoverage.19 The crossover sign occurs when the contour of the anterior rim of the acetabulum lies lateral to the posterior rim and is sensitive and specific for determining retroversion or focal anterosuperior overcoverage. The posterior wall sign is present if the posterior rim of the acetabulum lies medial to the center of the femoral head, indicating acetabular retroversion and may also be seen with global acetabular dysplasia.
The anterior-center-edge angle (ACEA) (Figure 3) evaluates the anterior acetabular coverage of the femoral head and anterosuperior subluxation of the femoral head. The ACEA is measured on the standing false profile view of the pelvis. The angle is formed by a line passing from the center of the femoral head to the anterior edge of the acetabulum and an intersecting vertical line. A measurement < 20 degrees is considered diagnostic of hip dysplasia, and measurements between 20 and 25 degrees are considered borderline.16
Femoral anatomy
Proximal femoral abnormalities are common in DDH and BDH. The femoral head tends to be more ellipsoid in contour in conjunction with increasing severity of acetabular dysplasia. One study found over 70% of hips treated for symptomatic acetabular dysplasia showed an aspherical femoral head and reduced head-neck offset.20 To evaluate the proximal femur a lateral or oblique projections of the femoral head and neck (frog lateral and Dunn views) are obtained.21
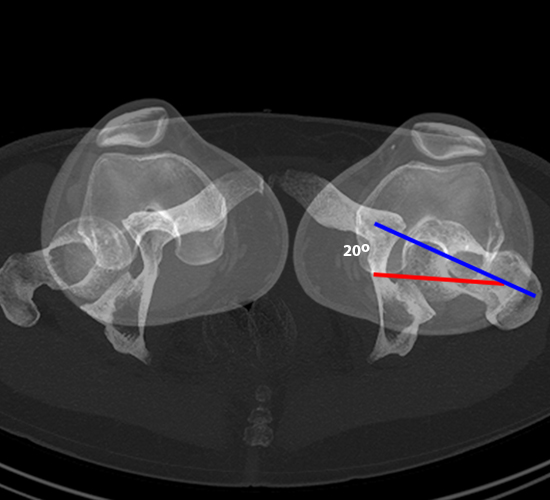
The alpha angle and femoral head-neck offset (Figure 3) can be measured on the frog-lateral and 45 degree Dunn views. These views evaluate the anterior and posterior contour of the femoral head-neck junction. The frog-lateral radiograph most closely represents the 3:00 o’clock location on the femoral head and neck, and the 45 degree Dunn view more closely reflects the 2:00 o’clock location.22 Cam morphology is most commonly seen in these locations at the anterosuperior aspect of the femoral head-neck junction. The alpha angle is calculated from a line connecting the center of the femoral head and the center of the femoral neck and a second line extending from the center of the femoral head to the point where the head-neck junction extends beyond a circle conforming to the radius of the central medial portion of the femoral head. An increased alpha angle is generally considered to be greater than 55 degrees. Asphericity of the femoral head-neck junction can also be assessed by the femoral offset ratio.
The femoral head-neck-shaft angle measures the intersection of a line along the femoral neck axis and the long axis of the femoral shaft with normal values ranging from 120 -135 degrees. An angle greater than 135 degrees represents coxa valga and less than 120 degrees is coxa vara.16 Patients with hip dysplasia most commonly have coxa valga, although normal and coxa vara measurements may be encountered. The femoral head-neck-shaft angle is limited as a diagnostic marker of hip dysplasia, though assessment of the angle is important in treatment and surgical planning as correction of femoral abnormalities may improve long-term results of acetabular reorientation surgery.
The delta angle (Figure 3) measures the position of the fovea capitis. “Fovea alta” is the abnormal superior position of the fovea capitis and is frequently seen in the adult dysplastic hip. The delta angle is calculated on the AP view by intersecting lines extending from the center of the femoral head to the medial edge of the sourcil and to the superior edge of the fovea capitis. The normal delta angle is greater than 10 degrees. In the dysplastic hip this angle is less than or equal to 10 degrees.23,16
Cranial subluxation of the femoral head is evaluated by the Shenton line (Figure 3), which represents a smooth arc formed by the medial femoral head-neck contour and the superior border of the obturator foramen. A break in this contour indicates subluxation of the femoral head from the acetabulum.
CT
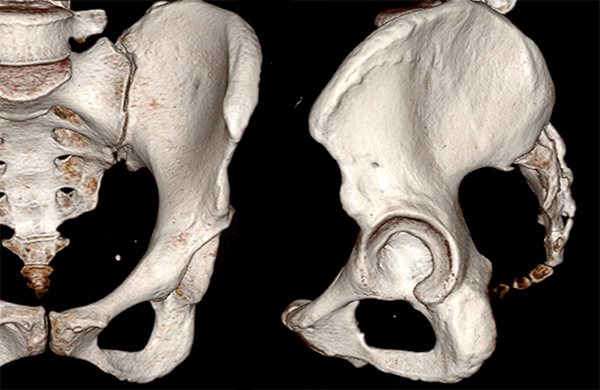
Acetabular dysplasia can present with significant anterosuperior, posterosuperior, and global acetabular deficiencies, which may be difficult to identify and accurately quantify by radiographs alone. CT with multiplanar and 3D reconstruction capabilities allows a more precise understanding of the acetabular deficiency. In addition, CT accurately depicts and allows quantification of cam morphology, femoral head-neck-shaft angle, and femoral version for accurate preoperative planning and to guide surgical corrections. Various proprietary software programs use CT data to help plan precise surgical reorientation of the acetabulum.
Figure 4:
47 year-old female with chronic hip pain. CT examination allows reformatting in multiple planes to allow precise measurements (4a). Femoral version relative to the femoral condyles is easily assessed by superimposing the femoral condyles and femoral neck (4b). 3D reformatted images allow improved visualization of the anatomy for surgical planning (4c).
MRI
After femoroacetabular impingement, hip instability represents the second most common cause for patient referral for MR imaging of the hip, and the diagnosis of underlying dysplasia is often unsuspected. While many of the radiographic measurements used to quantify the severity of hip dysplasia such as the LCEA, acetabular index, and the delta angle can be obtained from MR images, the primary value of MRI is its ability to diagnose intra-articular pathology. MRI can detect intra-articular pathology that correlate with arthroscopic findings of hip instability or microinstability including labral hypertrophy, labral tears, chondral injuries, and tears of the ligamentum teres.
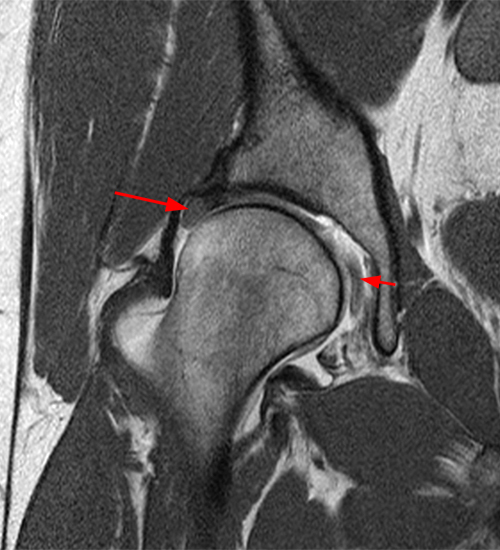
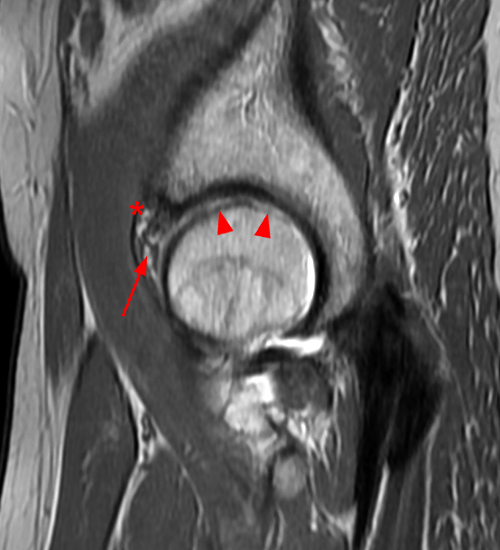
Adaptive labral and articular cartilage hypertrophy are readily visible on by MRI (Figures 1 and 5). In the normal hip, acetabular cartilage thickness is greatest laterally at the lateral sourcil. In the deficient acetabulum, greater forces are focused on the lateral acetabulum, and a compensatory response is believed to result in cartilage thickening. Ashwell et al. found that the cartilage thickness at the lateral sourcil is inversely related to the amount of lateral acetabular coverage. The mean cartilage thickness was found to be 35% greater than normal in frankly dysplastic hips. In contrast, cartilage thickness was 14% less than normal with acetabular overcoverage. For both borderline and frankly dysplastic hips, the increase in lateral acetabular cartilage thickness was significant with the frankly dysplastic hips showing the greatest cartilage thickness.24
Labral hypertrophy is often associated with hip dysplasia and microinstability. (Figure 6) 4,25,26 In one study Nwachukwu et al. used an intraoperative measurement of > 4mm to define labral hypertrophy which correlated with physical examination findings suggestive of microinstability. The LCEA angle in these patients was significantly less than the normal population, but all patients did not meet the study criteria of dysplasia (LCEA <25 degrees).25 Labral degeneration is recognized by increased signal within the labral tissue and fraying of the labral surface (Figure 8).
Figure 6:
(6a) 20 year-old female with groin pain for 6 months. LCEA of 22 degrees. A coronal fat-suppressed proton density image demonstrates enlargement of the acetabular labrum. An anterior labral tear was also present.
(6b) Comparison coronal MR arthrogram image in a 19 year-old female with LCEA of 27 degrees, demonstrates typical labral size.
Labral tears are common in patients with DDH and BDH. Anterior labral tears in the 2- 4 o’clock location are highly suggestive of instability.27 Tears in this location can also be seen with iliopsoas impingement.28 An anterior labral tear should prompt close inspection of the acetabular morphology and soft tissues for other evidence of hip dysplasia (Figure 1). Labral tears at the anterosuperior acetabular labrum commonly occur in both cam-FAI and hip dysplasia. Labral tears with intralabral and paralabral cysts are more common in the setting of instability and hip dysplasia, and the combination of labral hypertrophy and soft tissue ganglia is a positive predictor for the presence of DDH (Figure 7 and Figure 9).29,30 With repeated subluxation of the femoral head, a characteristic “inside-out” pattern of delamination of the chondrolabral complex is commonly seen at arthroscopy. The delamination begins medial to the chondrolabral junction and extending laterally with a detached continuous sleeve or flap of chondrolabral tissue. With continued lateral extension, the tear eventually undermines the labrum and can lead to labral displacement.31,32 The inside-out lesion is best detected by MR arthrography, and several authors recommend traction during the MRI to help increase contrast dispersal under the chondrolabral flap.
Acetabular rim bone cysts are also more common in DDH than in FAI patients, resulting from the increased stresses focused at the acetabular rim (Figure 7).29 Fracture of the acetabular rim can occur resulting in an osseous fragment that may hypertrophy over time.4
Figure 7:
47 year-old female with hip pain and LCEA of 16 degrees. Sagittal proton density-weighted (7a) and contiguous coronal fat-suppressed proton density-weighted images (7b, 7c) at the anterior acetabulum demonstrating subarticular cyst (arrows) at the anterior acetabular rim and paralabral cysts (arrowheads). The labral tear is not included on the images.
The ligamentum teres provides hip stability, particularly with hip flexion and external rotation (squatting) and hip extension with internal rotation (crossing one leg behind the other).33 In the patient with deficient acetabular coverage, the ligamentum teres plays a greater role in hip stability and thus is subjected to repetitive microtrauma that most commonly results in partial tears (Figure 8).34 Intrasubstance tearing at the fovea capitis is most commonly seen.35
Figure 8:
35 year-old female with chronic groin pain. LCEA of 23 degrees. Sequential coronal T1-weighted images (8a,b) from an MR arthrogram demonstrate an ellipsoid femoral head with a broad neck, a degenerated and enlarged acetabular labrum (arrows). The ligamentum teres (small arrow) (8a) is followed superiorly with partial tearing near the fovea (arrowhead).
Acetabular chondral defects are typically seen in the anterior-superior and mid-superior region, similar to cam FAI. Patients with frank dysplasia (LCEA <20 degrees) are more likely to have additional chondral defects posterosuperiorly.36 High-grade chondral defects (Outerbridge grade III and IV) of the weight-bearing surface of the femoral head are significantly more common for patients with borderline dysplasia (LCEA 20-25) compared to hips with normal acetabular coverage (Figure 9).37
Figure 9:
54 year-old female with right hip pain for 1 year for evaluation of femoroacetabular impingement. LCEA of 20 degrees. Coronal fat-suppressed T1-weighted (9a) and sagittal proton density-weighted (9b) images demonstrate a degenerated and enlarged labrum (short arrow) and anterosuperior labral tearing with intralabral (arrow) and paralabral (asterisk) cysts. Small areas of high-grade chondral loss extend over the femoral head (arrowheads).
Patients with DDH and BDH have coexistent femoral head deformities associated with the ellipsoid contour of the femoral head and from cam deformities at the anterosuperior aspect of the femoral head in up to 70% of cases.20 Detection of the cam lesion relies on evaluating radiographs, CT, or MRI images for an increased alpha angle or reduced head-neck offset ratio (Femoroacetabular Impingement). In addition, the above described findings of instability can help differentiate instability from impingement as the dominant underlying pathology.
Treatment
Activity modification, physical therapy, and postural correction are non-surgical options. Non-operative management or arthroscopy may be beneficial for pre-arthritic patients with dynamic pathologies such as microinstability and impingement. Extra-articular causes of hip pain are frequently found with hip dysplasia including iliopsoas and trochanteric bursitis and tendinopathies of the abductor, adductor and iliopsoas tendons.38,39
Osteotomy
Depending on the patient’s age and skeletal maturity, different pelvic osteotomies have been successful in treating patients with clear signs of instability. All techniques have the common goal of relieving pain and delaying or preventing progression of osteoarthritis. The Bernese periacetabular osteotomy (PAO) was developed in Bern, Switzerland by Ganz and is the most widely used osteotomy for reorientation of the acetabulum.40 The advantages that PAO offers include preservation of the posterior column, acetabular vascularity, and the hip abductors. PAO improves abductor mechanics through medialization of the hip joint. Femoral osteotomy is also performed when PAO alone is insufficient to improve hip mechanics and to help achieve a more concentric joint.
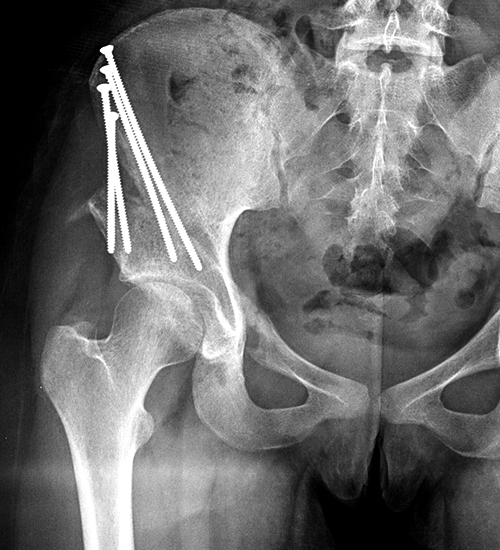
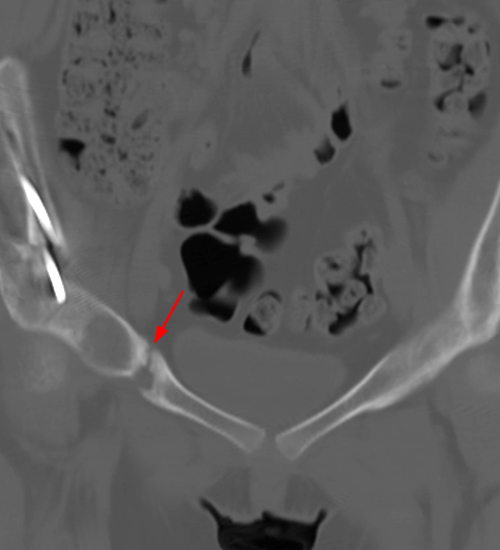
The surgery is technically demanding with a challenging learning curve and carries a significant risk of morbidity. The surgery requires 5 precise cuts to mobilize the acetabulum and allow reorientation of the acetabulum to improve femoral head coverage and joint congruency. Complications include overcorrection leading to iatrogenic femoroacetabular impingement, nerve and vascular damage, osseous non-union, and posterior column discontinuity (Figure 11).
Figure 11:
18 year-old status post periacetabular ostetomy with continued pain 4 months after surgery. AP pelvic radiograph (11a) demonstrates the post-operative changes of the PAO. Coronal CT reconstruction through the anterior pelvis (11b) demonstrates incomplete healing (arrow) across the superior pubic ramus osteotomy.
Arthroscopy
Because of the technical difficulty and surgical morbidity associated with osteotomy surgeries, the potential role of arthroscopy in the patient with less severe forms of dysplasia is expanding. However, the use of arthroscopy to treat intra-articular pathology in the setting DDH or BDH is not without controversy. Arthroscopic labral surgery can accelerate the progression of arthritis in the dysplastic hip. Worsening subluxation and even dislocation have been reported. These poor outcomes most likely resulted from excessive rim trimming, unrepaired capsulotomy, overzealous labral resection, inadequate labral repair, ligamentum teres debridement, and psoas tenotomy.41,42 Careful selection of arthroscopic candidates is mandatory with arthroscopy reserved for symptomatic patients with negative instability testing. In these patients, arthroscopic approaches must focus on preserving or restoring the chondrolabral and capsular complexes. With further understanding of the additional structures lending stability to the hip, the importance of insuring capsular integrity through capsular closure is widely recognized. Capsular plication has been proposed as an alternative procedure in highly selective cases to help stabilize the hip in patients with milder forms of instability.43 Age greater than 35 carries the greatest risk for failure in this surgical population.44 The plication procedure is technically challenging, and failure to repair the capsule can be catastrophic.
The most common indication for arthroscopic treatment of borderline acetabular dysplasia is the coexistence of cam-type femoral morphology.43 Cam-type morphology and associated intra-articular chondrolabral pathology can occur in 40-75% of cases of borderline acetabular dysplasia.43,20,38 A thorough evaluation is necessary to determine if instability or impingement is the dominant pathomechanism. In patients with symptomatic impingement, a femoral cam lesion, and negative instability testing, arthroscopy is often preferred.
Arthroscopy of the hip is frequently performed as a combined procedure with periacetabular osteotomy. Pre-emptive removal of the osseous prominence associated with the typically ellipsoid femoral head or cam lesion helps prevent femoral impingement with the reoriented acetabulum. In addition, chondrolabral injuries are present in up to 65% of patients undergoing PAO.45 Arthroscopy allows for concurrent repair to treat potential intra-articular sources of pain. The improved functional stability provided by the reoriented acetabulum reduces strain on the repaired structures and reduces the risk of iatrogenic hip instability.46
Total hip arthroplasty
In the setting of hip dysplasia, total hip arthroplasty is the treatment of choice for end-stage arthritis.
Conclusion
Hip instability is a complex problem, and dysplastic osseous anatomy at the hip is an important factor. Developmental dysplasia of the hip encompasses acetabular and femoral head abnormalities that range from subtle to severe. The decision-making for frank dysplastic hip, dislocation, and severe arthritis is usually straightforward. The diagnosis and optimal treatment planning for patients presenting with hip pain and milder forms of hip dysplasia can be challenging. The patient’s symptoms, functional impairment, age, physical examination and instability testing findings, as well as the underlying anatomical defects depicted by imaging must be carefully considered to determine the best treatment approach. Radiographs are the primary tool for imaging, and CT plays an important role in surgical planning. MRI excels at demonstrating the intra-articular pathology that often accompanies hip dysplasia. This information can provide additional clues regarding underlying instability that can inform the arthroscopic and PAO choices.
References
- Gala L, Clohisy JC, Beaulé PE. Hip Dysplasia in the Young Adult. J Bone Jt Surg. 2016;98(1):63-73. doi:10.2106/JBJS.O.00109 ↩
- Manaster BJ. From the RSNA refresher courses. Total hip arthroplasty: radiographic evaluation. RadioGraphics. 1996;16(3):645-660. doi:10.1148/radiographics.16.3.8897629 ↩
- Wiberg G. Studies on dysplastic acetabula and congenital subluxation of the hip joint: With special reference to the complication of osteoarthritis. Acta Chir Scand. 1939;83(58). ↩
- Klaue K, Durnin C, Ganz R. The acetabular rim syndrome. A clinical presentation of dysplasia of the hip. J Bone Joint Surg Br. 1991;73-B(3):423-429. doi:10.1302/0301-620X.73B3.1670443 ↩
- Jacobsen S, Rømer L, Søballe K. Degeneration in dysplastic hips. Skeletal Radiol. 2005;34(12):778-784. doi:10.1007/s00256-005-0019-7 ↩
- Armand M, Lepistö J, Tallroth K, Elias J, Chao E. Outcome of periacetabular osteotomy: Joint contact pressure calculation using standing AP radiographs, 12 patients followed for average 2 years. Acta Orthop. 2005;76(3):303-313. doi:10.1080/00016470510030742 ↩
- Hipp JA, Sugano N, Millis MB, Murphy SB. Planning acetabular redirection osteotomies based on joint contact pressures. In: Clinical Orthopaedics and Related Research. Lippincott Williams and Wilkins; 1999:134-143. doi:10.1097/00003086-199907000-00018 ↩
- Michaeli DA, Murphy SB, Hipp JA. Comparison of predicted and measured contact pressures in normal and dysplastic hips. Med Eng Phys. 1997;19(2):180-186. doi:10.1016/S1350-4533(96)00051-3 ↩
- Okano K, Yamaguchi K, Ninomiya Y, Matsubayashi S, Osaki M, Takahashi K. Femoral head deformity and severity of acetabular dysplasia of the hip. Bone Jt J. 2013;95 B(9):1192-1196. doi:10.1302/0301-620X.95B9.31503 ↩
- Steppacher SD, Tannast M, Werlen S, Siebenrock KA. Femoral morphology differs between deficient and excessive acetabular coverage. Clin Orthop Relat Res. 2008;466(4):782-790. doi:10.1007/s11999-008-0141-7 ↩
- Lee CB, Mata-Fink A, Millis MB, Kim YJ. Demographic differences in adolescent-diagnosed and adult-diagnosed acetabular dysplasia compared with infantile developmental dysplasia of the hip. J Pediatr Orthop. 2013;33(2). doi:10.1097/BPO.0b013e3182745456 ↩
- Jacobsen JS, Hölmich P, Thorborg K, et al. Muscle-tendon-related pain in 100 patients with hip dysplasia: prevalence and associations with self-reported hip disability and muscle strength. J Hip Preserv Surg. 2018;5(1):39-46. doi:10.1093/jhps/hnx041 ↩
- Hoppe DJ, Truntzer JN, Shapiro LM, Abrams GD, Safran MR. Diagnostic Accuracy of 3 Physical Examination Tests in the Assessment of Hip Microinstability. Orthop J Sport Med. 2017;5(11):232596711774012. doi:10.1177/2325967117740121 ↩
- Ogata S, Moriya H, Tsuchiya K, Akita T, Kamegaya M, Someya M. Acetabular cover in congenital dislocation of the hip. J Bone Jt Surg – Ser B. 1990;72(2):190-196. doi:10.1302/0301-620x.72b2.2312554 ↩
- Tönnis D, Heinecke A. Acetabular and femoral anteversion: Relationship with osteoarthritis of the hip. J Bone Jt Surg – Ser A. 1999;81(12):1747-1770. doi:10.2106/00004623-199912000-00014 ↩
- Beltran LS, Rosenberg ZS, Mayo JD, et al. Imaging Evaluation of Developmental Hip Dysplasia in the Young Adult. Am J Roentgenol. 2013;200(5):1077-1088. doi:10.2214/AJR.12.9360 ↩
- Lequesne M. Coxométrie: mesure des angles fonda- mentaux de la hanche radiographique de l’adulte par un rapporteur combine. Rev Rhum. 1963;30:479–485. ↩
- Tannast M, Hanke MS, Zheng G, Steppacher SD, Siebenrock KA. What Are the Radiographic Reference Values for Acetabular Under- and Overcoverage? Clin Orthop Relat Res. 2015;473(4):1234-1246. doi:10.1007/s11999-014-4038-3 ↩
- Siebenrock KA, Kistler L, Schwab JM, Büchler L, Tannast M. The Acetabular Wall Index for Assessing Anteroposterior Femoral Head Coverage in Symptomatic Patients. Clin Orthop Relat Res. 2012;470(12):3355-3360. doi:10.1007/s11999-012-2477-2 ↩
- Clohisy JC, Nunley RM, Carlisle JC, Schoenecker PL. Incidence and characteristics of femoral deformities in the dysplastic hip. Clin Orthop Relat Res. 2009;467(1):128-134. doi:10.1007/s11999-008-0481-3 ↩
- Steppacher SD, Albers CE, Tannast M, Siebenrock KA. Plain Radiographic Evaluation of the Hip. In: Nho SJ, Leunig M, Larson CM, Bedi A, Kelly BT, eds. Hip Arthroscopy and Hip Joint Preservation Surgery. New York, NY: Springer New York; 2015:33-51. doi:10.1007/978-1-4614-6965-0 ↩
- Nepple JJ, Martel JM, Kim YJ, Zaltz I, Clohisy JC. Do plain radiographs correlate with CT for imaging of cam-type femoroacetabular impingement? Clin Orthop Relat Res. 2012;470(12):3313-3320. doi:10.1007/s11999-012-2510-5 ↩
- Nötzli HP, Müller SM, Ganz R. The relationship between fovea capitis femoris and weight bearing area in the normal and dysplastic hip in adults: A radiologic study. Z Orthop Ihre Grenzgeb. 2001;139(6):502-506. doi:10.1055/s-2001-19231 ↩
- Ashwell ZR, Flug J, Chadayammuri V, Pascual-Garrido C, Garabekyan T, Mei-Dan O. Lateral acetabular coverage as a predictor of femoroacetabular cartilage thickness. J Hip Preserv Surg. 2016;3(4):262-269. doi:10.1093/jhps/hnw034 ↩
- Nwachukwu BU, Gaudiani MA, Marsh NA, Ranawat AS. Labral hypertrophy correlates with borderline hip dysplasia and microinstability in femoroacetabular impingement: a matched case-control analysis. HIP Int. 2019;29(2):198-203. doi:10.1177/1120700018807860 ↩
- Kraeutler MJ, Goodrich JA, Ashwell ZR, Garabekyan T, Jesse MK, Mei-Dan O. Combined Lateral Osseolabral Coverage Is Normal in Hips With Acetabular Dysplasia. Arthrosc – J Arthrosc Relat Surg. 2019;35(3):800-806. doi:10.1016/j.arthro.2018.10.133 ↩
- Shibata KR, Matsuda S, Safran MR. Is there a distinct pattern to the acetabular labrum and articular cartilage damage in the non-dysplastic hip with instability? Knee Surgery, Sport Traumatol Arthrosc. 2017;25(1):84-93. doi:10.1007/s00167-016-4342-4 ↩
- Domb BG, Shindle MK, McArthur B, Voos JE, Magennis EM, Kelly BT. Iliopsoas Impingement: A Newly Identified Cause of Labral Pathology in the Hip. HSS J. 2011;7(2):145-150. doi:10.1007/s11420-011-9198-z ↩
- Stelzeneder D, Mamisch TC, Kress I, et al. Patterns of joint damage seen on MRI in early hip osteoarthritis due to structural hip deformities. Osteoarthr Cartil. 2012;20(7):661-669. doi:10.1016/j.joca.2012.03.014 ↩
- Leunig M, Podeszwa D, Beck M, Werlen S, Ganz R. Magnetic Resonance Arthrography of Labral Disorders in Hips with Dysplasia and Impingement. In: Clinical Orthopaedics and Related Research. Lippincott Williams and Wilkins; 2004:74-80. doi:10.1097/00003086-200401000-00013 ↩
- Kraeutler MJ, Goodrich JA, Fioravanti MJ, Garabekyan T, Mei-Dan O. The “Outside-In” Lesion of Hip Impingement and the “Inside-Out” Lesion of Hip Dysplasia: Two Distinct Patterns of Acetabular Chondral Injury. Am J Sports Med. 2019;47(12):2978-2984. doi:10.1177/0363546519871065 ↩
- Yoon PW, Moon JK, Yoon JY, et al. A novel arthroscopic classification of labral tear in hip dysplasia. PLoS One. 2020;15(10 October):1-15. doi:10.1371/journal.pone.0240993 ↩
- Martin RRL, Palmer I, Martin HD. Ligamentum teres: A functional description and potential clinical relevance. Knee Surgery, Sport Traumatol Arthrosc. 2012;20(6):1209-1214. doi:10.1007/s00167-011-1663-1 ↩
- Kraeutler MJ, Garabekyan T, Pascual-Garrido C, Mei-Dan O. Ligamentum teres tendinopathy and tears. Muscles Ligaments Tendons J. 2016;6(3):337-342. doi:10.11138/mltj/2016.6.3.337 ↩
- Philippon MJ, Rasmussen MT, Turnbull TL, et al. Structural properties of the native ligamentum teres. Orthop J Sport Med. 2014;2(12):232596711456196. doi:10.1177/2325967114561962 ↩
- Kaya M, Suzuki T, Emori M, Yamashita T. Hip morphology influences the pattern of articular cartilage damage. Knee Surgery, Sport Traumatol Arthrosc. 2016;24(6):2016-2023. doi:10.1007/s00167-014-3297-6 ↩
- Bolia IK, Briggs KK, Locks R, Chahla J, Utsunomiya H, Philippon MJ. Prevalence of High-Grade Cartilage Defects in Patients With Borderline Dysplasia With Femoroacetabular Impingement: A Comparative Cohort Study. Arthrosc J Arthrosc Relat Surg. 2018;34:2347-2352. doi:10.1016/j.arthro.2018.03.012 ↩
- Ida T, Nakamura Y, Hagio T, Naito M. Prevalence and characteristics of cam-type femoroacetabular deformity in 100 hips with symptomatic acetabular dysplasia: a case control study. J Orthop Surg Res. 2014;9:93. doi:10.1186/s13018-014-0093-4 ↩
- Nunley RM, Prather H, Hunt D, Schoenecker PL, Clohisy JC. Clinical presentation of symptomatic acetabular dysplasia in skeletally mature patients. J Bone Jt Surg – Ser A. 2011;93(SUPPL. 2):17-21. doi:10.2106/JBJS.J.01735 ↩
- Ganz R, Klaue K, Vinh TS, Mast JW. A new periacetabular osteotomy for the treatment of hip dysplasias. Technique and preliminary results. Clin Orthop Relat Res. 1988;Jul(232):26-36. http://www.ncbi.nlm.nih.gov/pubmed/3383491. ↩
- Matsuda DK, Wolff AB, Nho SJ, et al. Hip Dysplasia: Prevalence, Associated Findings, and Procedures From Large Multicenter Arthroscopy Study Group. Arthrosc J Arthrosc Relat Surg. 2018;34(2):444-453. doi:10.1016/j.arthro.2017.08.285 ↩
- Mei-Dan O, McConkey MO, Brick M. Catastrophic failure of hip arthroscopy due to iatrogenic instability: Can partial division of the ligamentum teres and iliofemoral ligament cause subluxation? Arthrosc – J Arthrosc Relat Surg. 2012;28(3):440-445. doi:10.1016/j.arthro.2011.12.005 ↩
- Buerba RA, Murray RS, Mauro CS. Capsular Plication for Symptomatic Borderline Hip Dysplasia. Oper Tech Orthop. 2020;30(1). doi:10.1016/j.oto.2020.100781 ↩
- Hatakeyama A, Utsunomiya H, Nishikino S, et al. Predictors of Poor Clinical Outcome After Arthroscopic Labral Preservation, Capsular Plication, and Cam Osteoplasty in the Setting of Borderline Hip Dysplasia. Am J Sports Med. 2018;46(1):135-143. doi:10.1177/0363546517730583 ↩
- Ross JR, Zaltz I, Nepple JJ, Schoenecker PL, Clohisy JC. Arthroscopic Disease Classification and Interventions as an Adjunct in the Treatment of Acetabular Dysplasia. Am J Sports Med. 2011;39(1_suppl):72S-78S. doi:10.1177/0363546511412320 ↩
- Kim KI, Cho YJ, Ramteke AA, Yoo MC. Peri-acetabular rotational osteotomy with concomitant hip arthroscopy for treatment of hip dysplasia. J Bone Jt Surg – Ser B. 2011;93 B(6):732-737. doi:10.1302/0301-620X.93B6.25809 ↩