Clinical History:
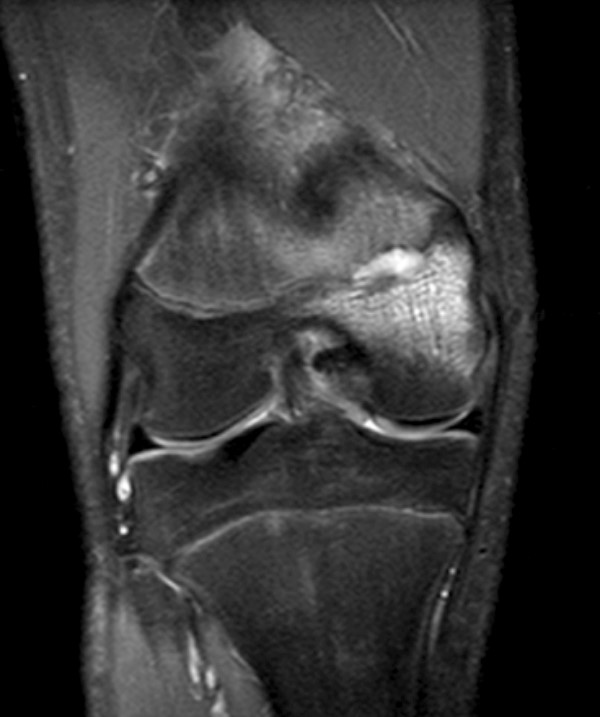
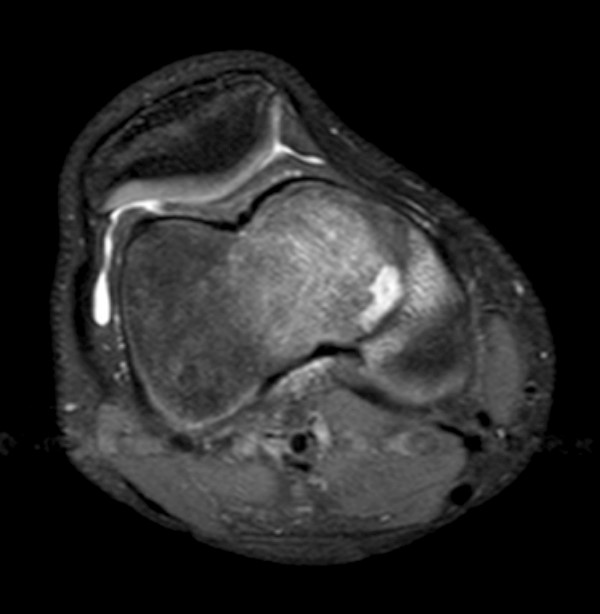
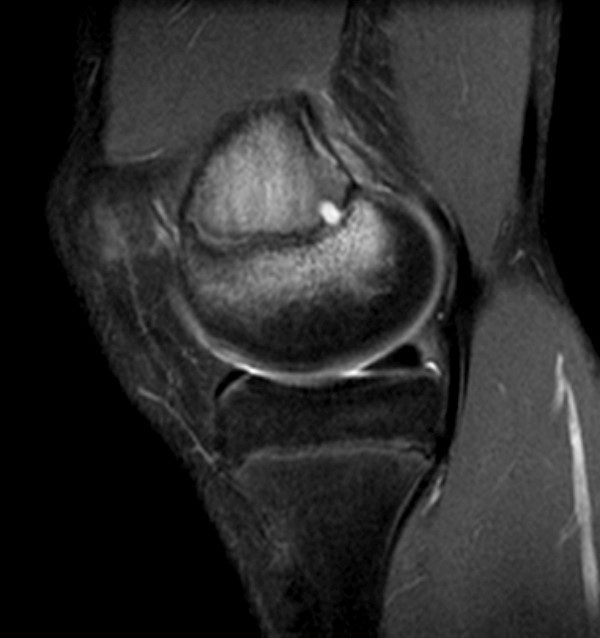
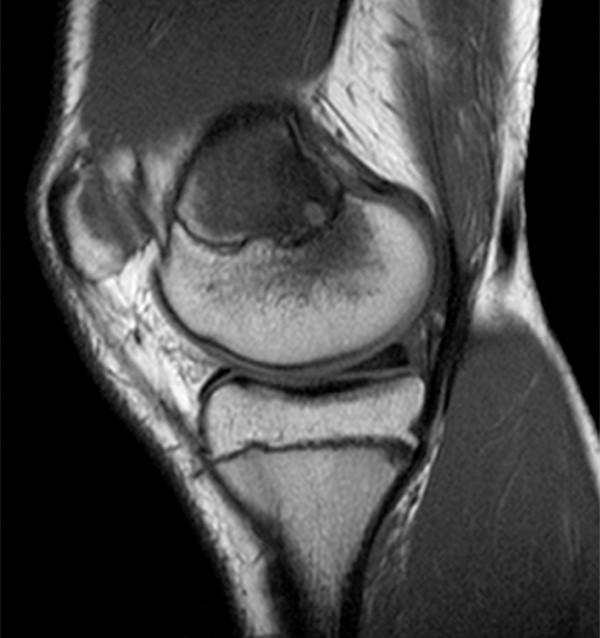
A 16 year-old male presents with pain for 2-3 weeks following a soccer injury. An MRI was performed for suspected cartilage injury. Coronal, axial, and sagittal proton density weighted and sagittal T1-weighted images of the right knee are provided (Figure 1).
What are the findings? What is your diagnosis?
Findings
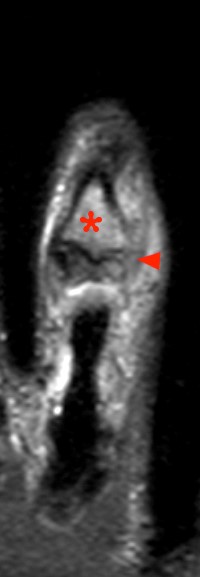
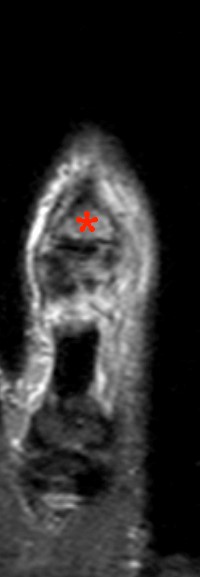
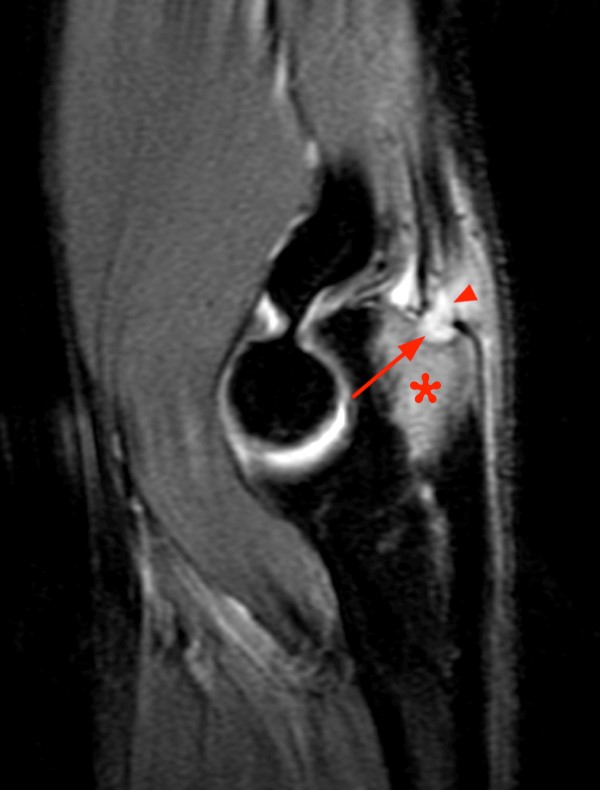
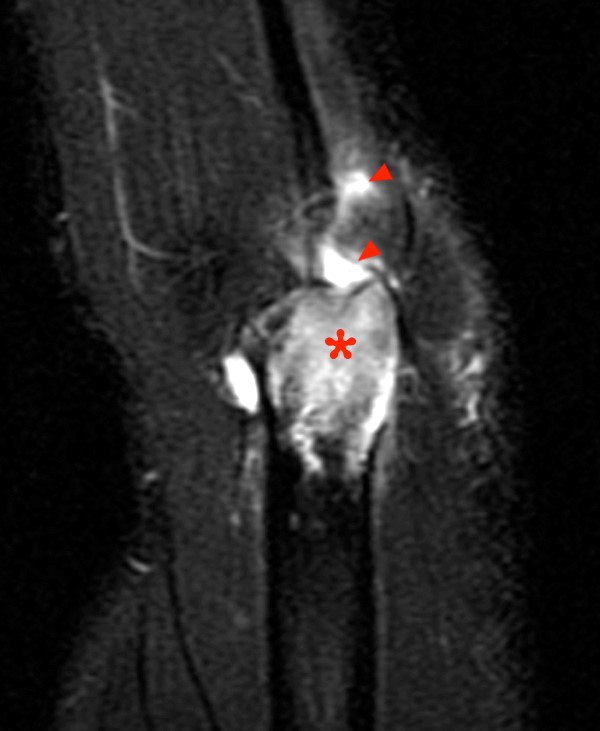
Figure 2:
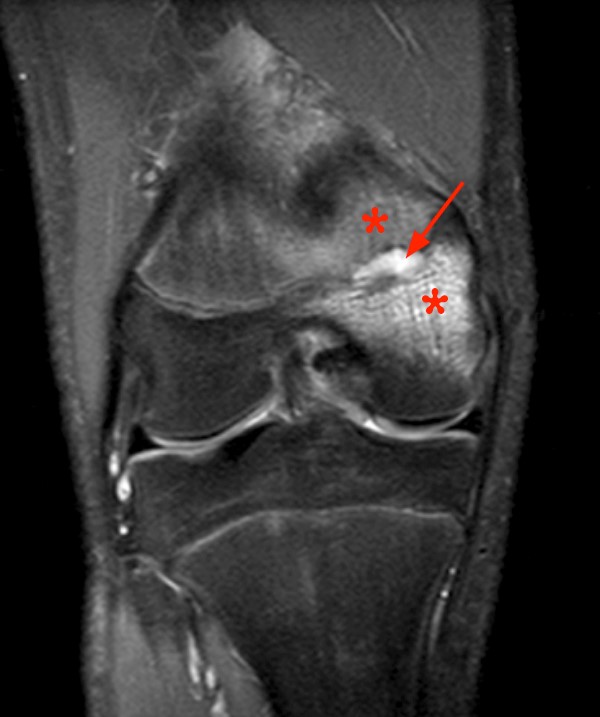
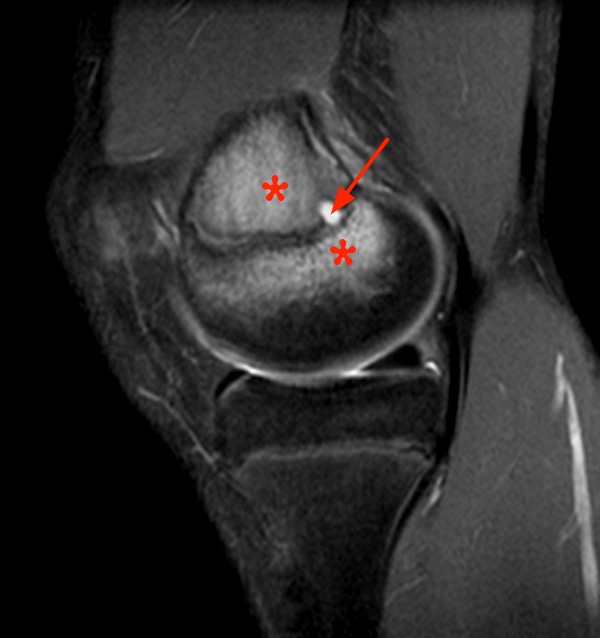
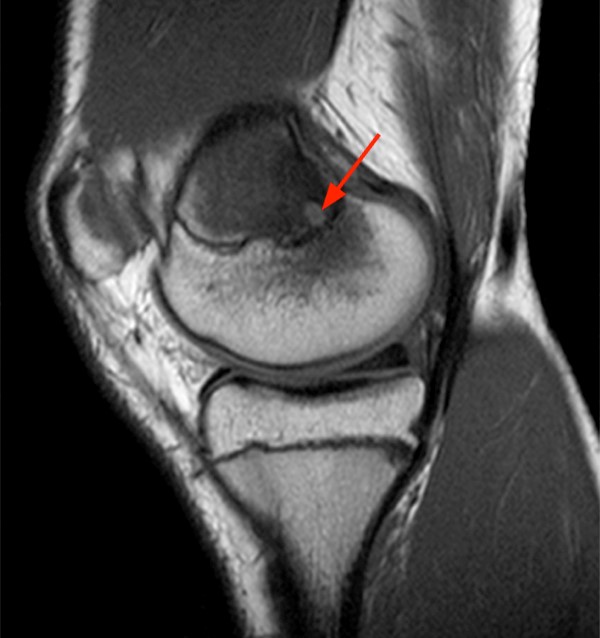
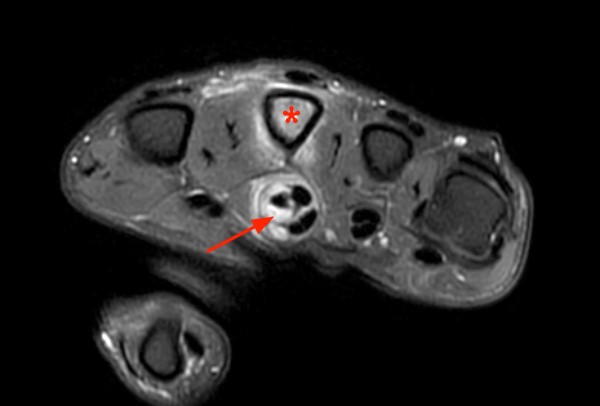
Coronal (2a), axial (2b), and sagittal (2c) proton density images demonstrate a small fluid collection in the posterior medial metaphysis (arrows), abutting the growth plate, with severe very diffuse increased signal consistent with marrow edema (asterisks). The sagittal T1-weighted image (2d) demonstrates a well-demarcated geographic zone of intermediate signal surrounding a small focus of slight increased signal (proteinaceous fluid). No fluid is demonstrated in the growth plate as would be seen with a growth plate injury.
Diagnosis
Acute hematogenous osteomyelitis with a small intra-osseous (Brodie’s) abscess.
Introduction
Osteomyelitis is commonly a diagnostic consideration in diabetic patients, particularly those with skin ulcerations and cellulitis, or the immunocompromised patient. However, it also can be found in otherwise healthy patients, presenting in the pediatric or adult population following a penetrating injury, or in the pediatric patient from metaphyseal seeding.
Imaging
The clinical presentation of infection, including pain, erythema, and edema, is nonspecific. In many cases osteomyelitis cannot be diagnosed by physical examination or laboratory studies alone, but rather requires one or more forms of imaging. Typically imaging begins with radiographs, primarily to exclude something other than osteomyelitis, such as fracture, tumor, or arthritis, but with low sensitivity for osteomyelitis. Bone scintigraphy has higher sensitivity but low specificity and doesn’t adequately visualize adjacent soft tissue infection. Computed tomography (CT) can demonstrate bony destruction and periostitis, but is not specific and often does not demonstrate adjacent soft tissue infection. Magnetic resonance imaging (MRI) is more diagnostic and is favored by most. The MRI sensitivity for the diagnosis of acute osteomyelitis has been reported to be as high as 82-100% and the specificity as high as 75-99%.1
MRI provides for not only the diagnosis of osteomyelitis, but complications associated with the bone infection, such as soft tissue abscess, tendon involvement (Figure 3), or ligament involvement (Figure 4) and is helpful in both initial treatment planning and follow up to therapy.
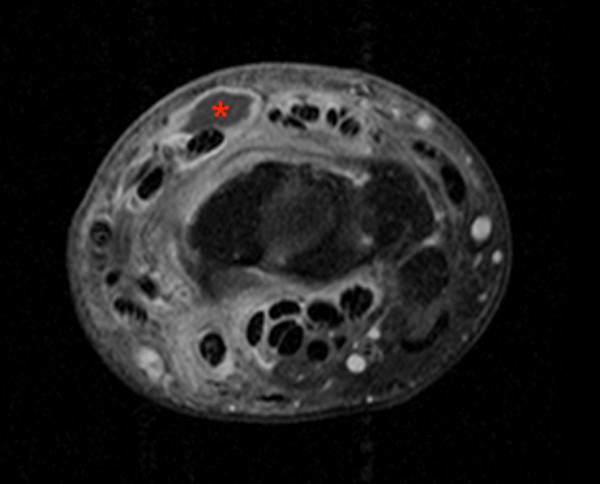
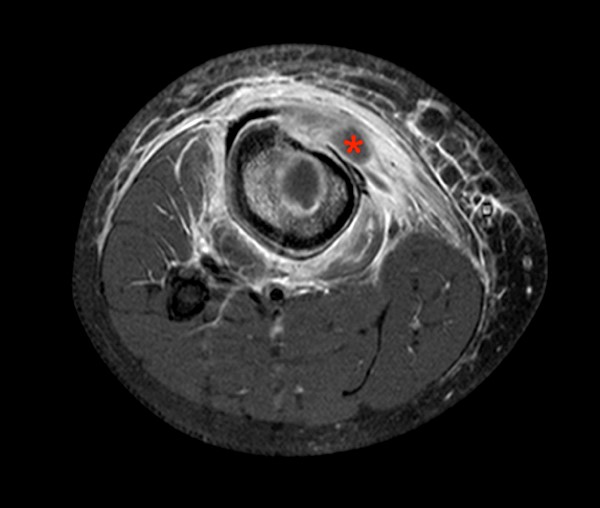
Figure 3:
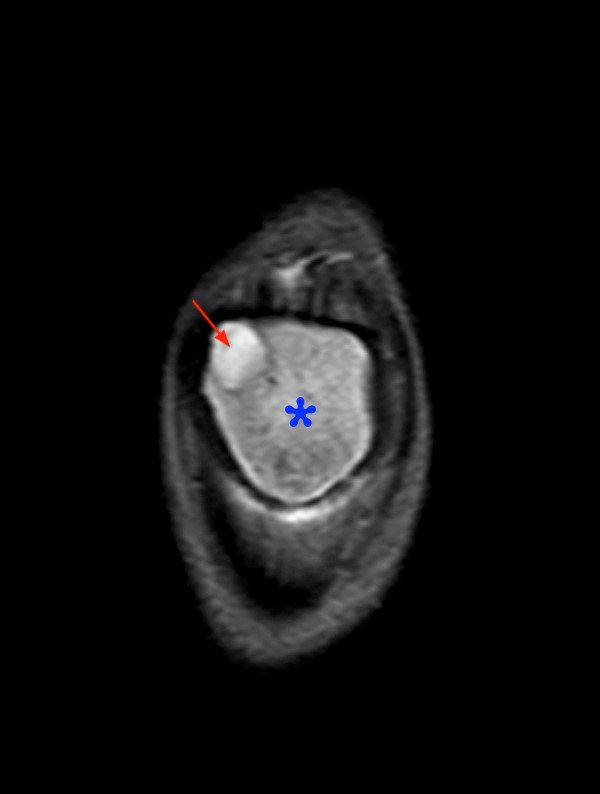
A 38 year-old with history of a puncture wound from a sea urchin 4 months prior presents with pain and swelling. An axial proton density fat-suppressed image demonstrates increased signal, representing marrow edema (asterisk) of the 2nd metacarpal and prominent irregular fluid in the flexor tendon sheath (arrow).
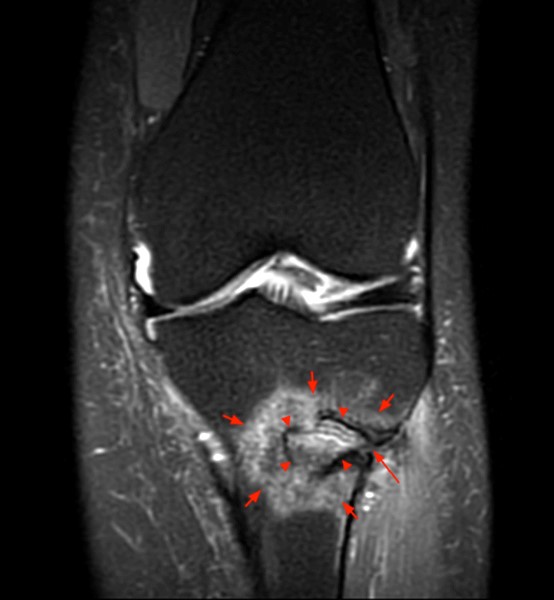
Figure 4:
45 year-old male with history of a cat bite 3 months prior. Coronal proton density with fat suppression images from dorsal (4a) to volar (4c) demonstrating increased signal of the proximal phalanx due to edema (asterisk) as well as edema and partial disruption of the proximal interphalangeal joint radial collateral ligament (arrowhead).
MRI Findings
Acute osteomyelitis can be diagnosed early in the disease process utilizing MRI. Signal change from bone marrow edema (decreased on T1-weighted and increased on T2-weighted imaging sequences) can be detected as early as 1-2 days after the onset of infection2 with the diagnosis of osteomyelitis made no later than 3-5 days after the onset of infection.3
In addition to bone marrow edema, other findings on MRI supportive of the diagnosis of osteomyelitis include cellulitis, adjacent soft tissue ulcers, soft tissue abscess, phlegmon, sinus tracts, and cortical bone destruction.4 (Figures 5 and 6)
Figure 5:
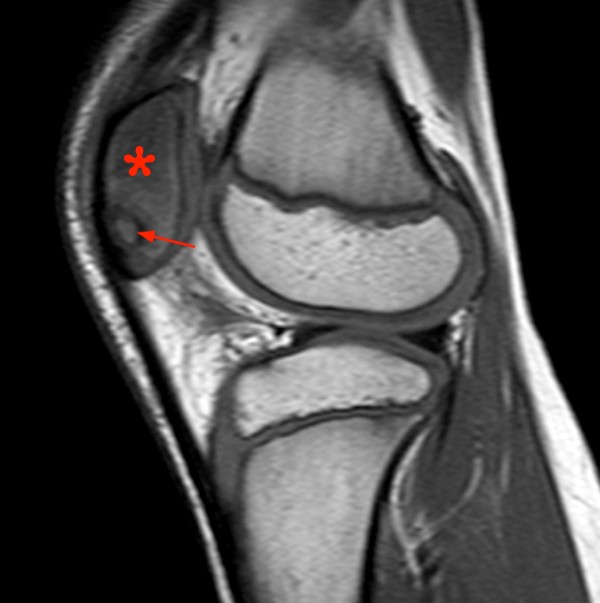
29 year-old male with a history of direct impaction injury and skin laceration. Fat-suppressed sagittal (5a) proton density and coronal (5b) T2-weighted series demonstrate intense marrow edema in a geographic pattern (asterisks), cortical loss/erosion of the tip of the olecranon (arrow) containing fluid in continuity with an extra-osseous fluid collection (arrowheads), as well as a longitudinal, fluid delineated split of the distal triceps tendon. Also note the overlying subcutaneous edema.
Typical findings of osteomyelitis seen on MRI are decreased T1 signal and increased T2 signal due to marrow edema. However, these can also be seen in the setting of stress reaction, reactive marrow, neuropathic arthropathy, and arthritis. In the absence of secondary findings, such as abscess formation (Figure 6) and phlegmon, intravenous gadolinium contrast is often utilized.
Figure 6:
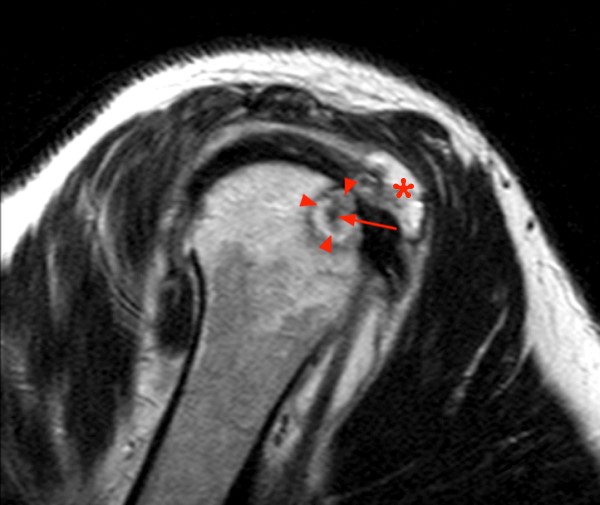
59 year-old status post rotator cuff repair 8 months ago with new onset of pain 6-8 weeks prior. Coronal T1 (6a) demonstrates a geographic area of decreased signal intensity (short arrows) surrounding the surgical anchors (arrow). The corresponding coronal fat-suppressed T2-weighted sequence (6b) demonstrates increased signal as well as fluid signal (arrowheads) surrounding the anchors (arrow). The sagittal T2-weighted image (6c) demonstrates the fluid (arrowheads) surrounding the anchors (arrow) and a contiguous, loculated fluid collection (asterisk) within the subacromial-subdeltoid bursa consistent with abscess formation.
Gadolinium contrast is often helpful in the diagnosis of osteomyelitis, demonstrating enhancement of the affected tissues. However, enhancement is not specific for osteomyelitis.
One particular study of pedal osteomyelitis found that the pattern of decreased marrow signal demonstrated on the T1-weighted images should be a geographic medullary distribution with a confluent pattern to make the diagnosis of osteomyelitis.5 (Figures 7 and 8)
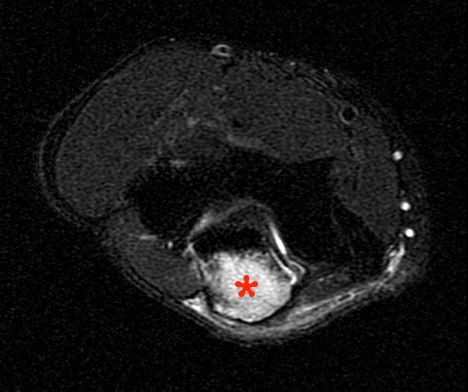
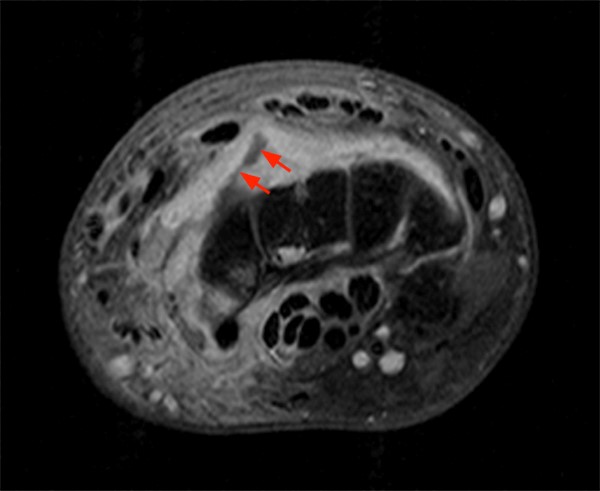
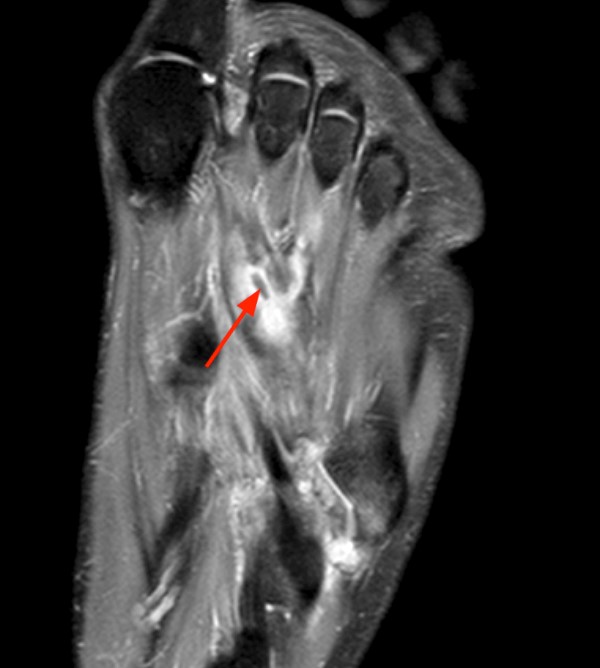
Figure 7:
8 year-old female with history of knee pain and swelling for 1 month. Sagittal T1-weighted (7a) and coronal fat-suppressed proton density-weighted (7b) images demonstrate two well-circumscribed foci of intermediate T1 and increased T2 signal (arrows), representing intraosseous abscesses, as well as diffuse decreased T1 (red asterisk) and increased T2 (blue asterisk) signal due to severe marrow edema.
Conversely, if the marrow edema pattern was ill-defined with a hazy, reticulated pattern, regardless of the appearance on T2-weighted and post-contrast images, the authors excluded the diagnosis of osteomyelitis and instead attributed the signal change to reactive marrow edema.5 (Figure 9)
Although imaging with gadolinium contrast is not always necessary for the diagnosis of osteomyelitis, it is useful for identifying intraosseous and soft tissue abscess formation, and is useful for the increase in reader confidence.6
The same study found that contrast enhancement did not add useful diagnostic information when the findings on the unenhanced series are considered definitely normal.6 Contrast enhanced imaging is also helpful in differentiating between hyperemic inflammatory tissue/phlegmon and abscess formation. (Figures 10 and 11)
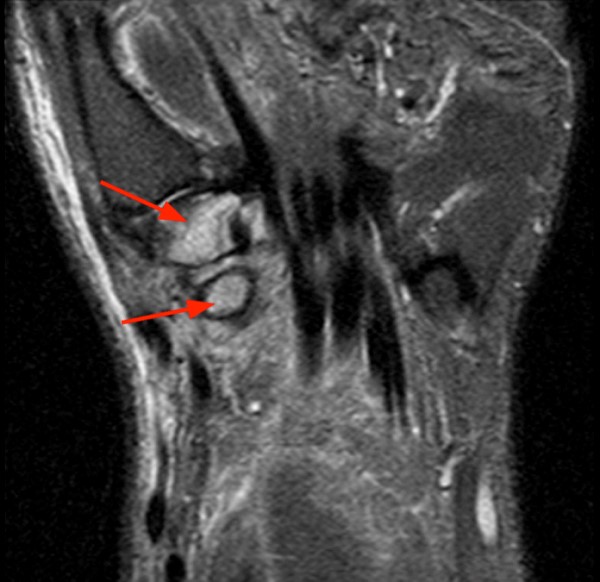
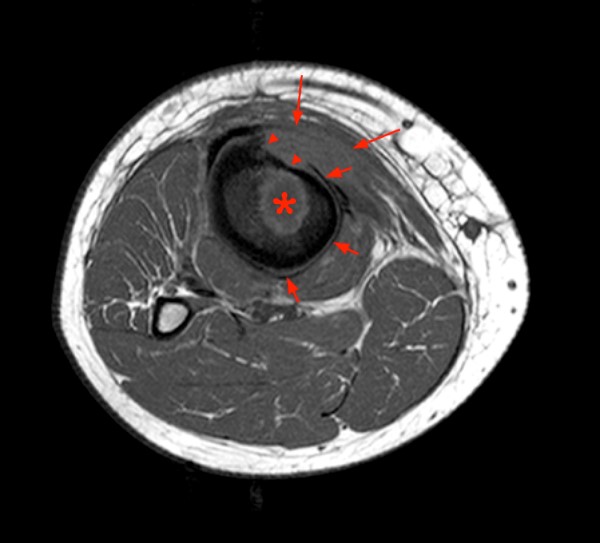
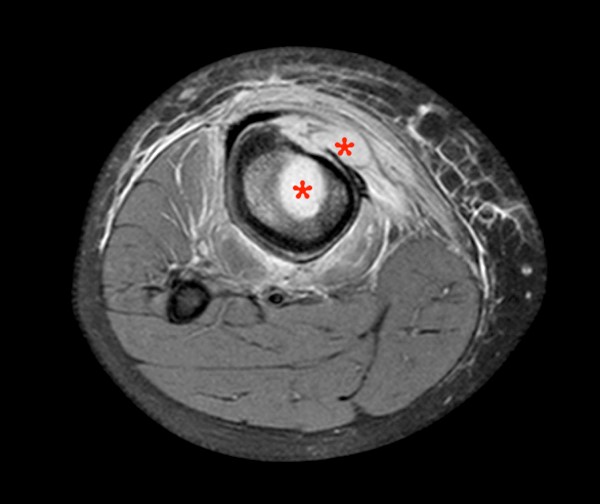
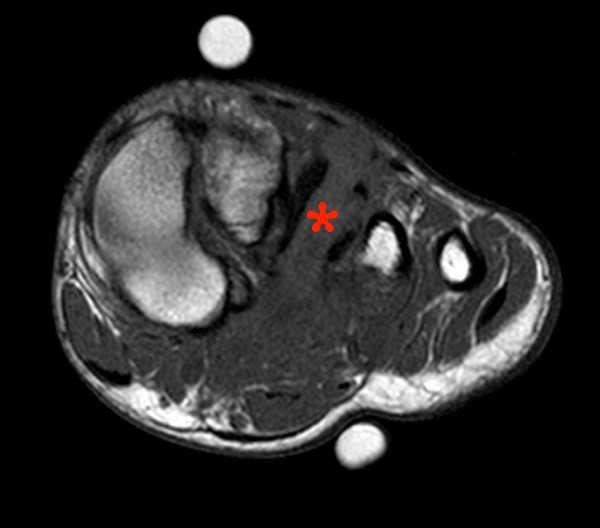
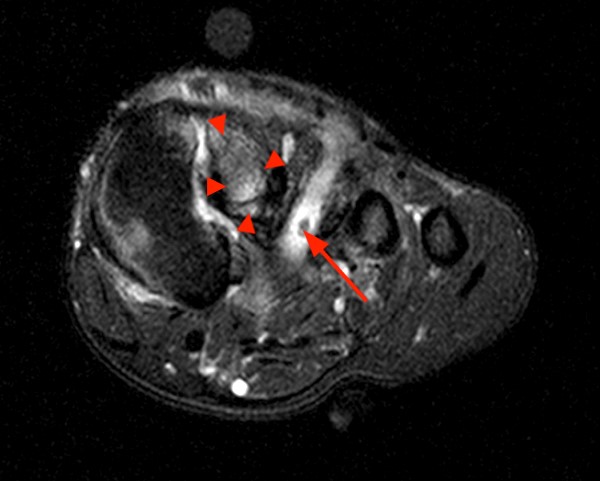
Figure 10:
46 year-old male with a history of pain and swelling following ganglion excision 4 weeks prior. Coronal fat-suppressed proton density image (10a) demonstrating increased signal of the scaphoid and trapezium (arrows) due to edema. The axial fat-suppressed proton density image (10b) demonstrates a heterogeneous area of increased signal (short arrows) in the subcutaneous space dorsal to the extensor carpi radialis brevis tendon. The post-contrast axial T1-weighted image with fat-suppression (10c) demonstrates lack of central enhancement (asterisk), thus an abscess rather than phlegmon. The axial image immediately distal to this (10d) demonstrates a smaller, deeper abscess (short arrows).
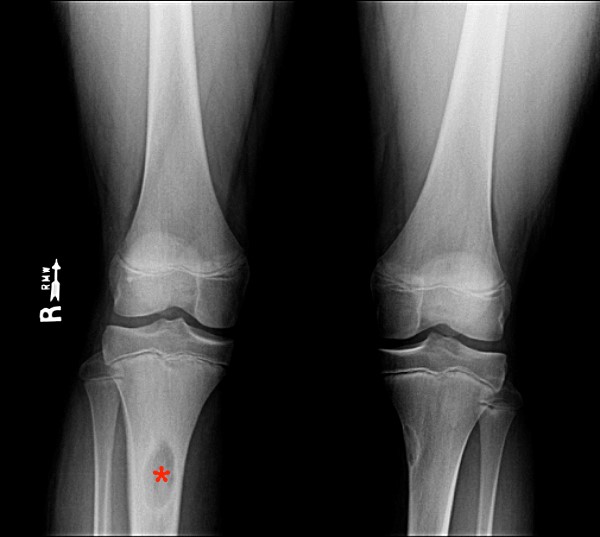
Figure 11:
13 year-old male with pain for 1-2 months with “anterior medial leg mass”. Radiographic images (11a) demonstrate a poorly defined lytic focus in the proximal right tibia (asterisk). Incidental well circumscribed lytic lesion with thin sclerotic margin in the proximal left tibia represents a non-ossifying fibroma. Axial T1-weighted image (11b) demonstrates the lytic lesion (asterisk) along with anterior medial cortical loss (arrowheads), diffuse periosteal new bone formation (short arrows), and thickening of the anterior medial soft tissues (long arrows). The axial proton density image with fat-suppression (11c) demonstrates an intraosseous fluid collection as well as fluid signal at the site of cortical loss (asterisks). The post-gadolinium enhanced, axial T1-weighted fat-suppressed image (11d) clearly demonstrates the extraosseous focus (asterisk) to be an abscess, rather than simply edematous soft tissue, with only peripheral enhancement.
Chronic osteomyelitis (Figure 12) has a much different appearance due to osteonecrosis and formation of a sequestrum. The necrotic fragment (sequestrum) will be of decreased signal on all sequences due to lack of water content. However, it can be identified due to surrounding granulation tissue of increased T2 signal, which typically enhances.7 The newly formed surrounding bone, or involucrum, is seen as a thickened shell surrounding the sequestrum and will either be of normal osseous signal or edematous.8 A cloaca, or channel, may be seen in both acute and chronic osteomyelitis as a cortical defect draining pus from within the medullary space to the surrounding soft tissues. If demonstrated, it will be of increased T2 signal due to pus content.8
Figure 12:
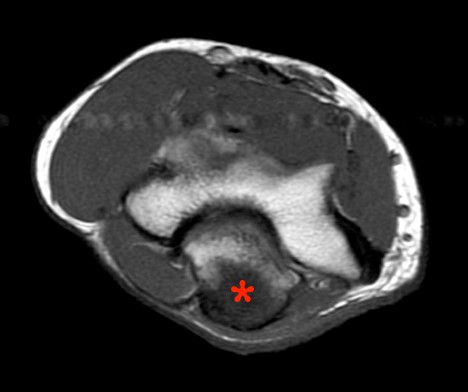
46 year-old female with history of pain for 5 months. Axial (12a) and coronal (12b) proton density images demonstrate changes of chronic osteomyelitis of the proximal tibia with a large involucrum (short arrows), with a thin margin of decreased signal consistent with new bone formation (arrowheads), and a lateral cloaca (long arrow) as well as adjacent myositis.
Discussion
Early detection and treatment of osteomyelitis can prevent complications such as abscess formation, septic arthritis with subsequent joint destruction, sepsis, and chronic osteomyelitis and can minimize sequelae such as growth arrest.9
The imaging workup can be used to determine the presence and location of bony involvement, existence of both intraosseous and soft tissue purulent collections, and involvement of important adjacent structures such as the joint and the growth plate. Imaging is also useful to guide aspiration, assess the need for drainage, and to direct the operative approach by showing the location and extent of abscesses and often the presence of a foreign body.10 (Figure 13)
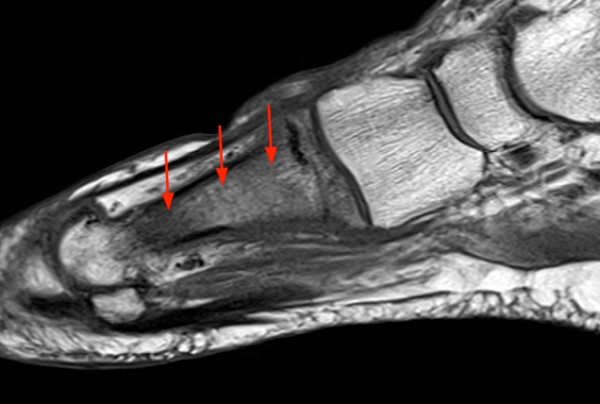
Figure 13:
20 year-old status post puncture wound 7 years prior. Coronal T1-weighted image (13a) demonstrates a complete penetrating defect through the base of the 3rd metatarsal (asterisk) with prominent bone loss. The corresponding coronal fat-suppressed T2-weighted image (13b) demonstrates fluid signal within the defect along with a small foreign body (arrow). Edema is also present in the 2nd metatarsal (arrowheads). The axial fat-suppressed proton density image (13c) demonstrates a communicating fluid collection slightly distal and plantar to the osseous lesion containing an additional foreign body (arrow).
Osteomyelitis can affect not just the immunocompromised or diabetic patient, but any patient following penetrating trauma, including puncture wounds or animal bites, or following placement of surgical metallic implants. It also occurs from deep spread of untreated cellulitis and soft tissue abscess.
Osteomyelitis can occur at any age. However, in the pediatric population it more commonly occurs from hematogenous spread. In those without specific risk factors, it is particularly common between the ages of 2-12 years of age and is more common in males (M:F of 3:1).11 In the United States, hematogenous osteomyelitis is the most common musculoskeletal infection in children and has an incidence of 1 in 5000 children per year.12
Blood-borne organisms can be deposited in the immature skeletal metaphysis of long bones due to a large volume of slow-flowing blood. This nidus of infection, usually bacteria, can then multiply and result in osteomyelitis.2
Hematogenous spread is less common in the adult population and usually presents as vertebral osteomyelitis.13 More commonly adult osteomyelitis results from mechanical introduction or contiguous spread from adjacent soft tissue infection.13
Osteomyelitis can be diagnosed either in the acute or chronic state.
Acute osteomyelitis
In acute osteomyelitis secondary to hematogenous spread or direct inoculation, bacterial growth within the bone induces an acute suppurative response. Accumulation of pus within the medullary cavity then produces an increase in intramedullary pressure and resulting vascular congestion, which can then disrupt the intraosseous blood supply. Reactive bone and hypervascular granulation tissue may form around the intramedullary pus, producing a well-circumscribed intraosseous abscess (Brodie’s abscess).14
Chronic osteomyelitis
Inadequately treated acute osteomyelitis will progress to chronic osteomyelitis.
Features of chronic osteomyelitis are a result of osteonecrosis, caused by disruption of the intraosseous and periosteal blood supply during the acute stage of disease. A fragment of necrotic, infected bone separates from viable bone producing what is termed a sequestrum. Due to lack of blood flow, the bacteria within the devascularized sequestrum are protected from antibiotics and the body’s own immune response, resulting in the formation of a nidus for chronic infection which may persist for many years.15
An inflammatory reaction of osteoclastic resorption and periosteal new bone formation occurs in an attempt to wall of the sequestrum. This surrounding pus, granulation tissue, and new bone formation is known at the involucrum. A cloaca is the communication through which pus may be discharged from the involucrum.14
Staphylococcus aureus, including more recently, methicillin-resistant S. aureus, is the primary cause of osteomyelitis in children, responsible for 70–90% of acute hematogenous osteomyelitis infections.16,17 Atypical osteomyelitis may be caused by organisms such as Mycobacterium, Bartonella, fungi (Histoplasma, Cryptococcus and Blastomyces), Candida and Coxiella, usually seen in patients with specific risk factors, such as immunosuppression, or in patients with a history of travel to or residence in geographic regions in which these pathogens are endemic.17
References
- Chiappini E, Mastrangelo G, Lazzeri S. A Case of Acute Osteomyelitis: An Update on Diagnosis and Treatment. Int J Environ Res Public Health. 2016 May 27. 13 (6). ↩
- Jaramillo D. Infection: musculoskeletal. Pediatr Radiol. 2011 May; 41 Suppl 1:S127-34. ↩
- Kocher MS, Lee B, Dolan M, Weinberg J, Shulman ST. Pediatric orthopaedic infections: early detection and treatment. Pediatr Ann. 2006 Feb; 35(2):112-22. ↩
- Unger E, Moldofsky P, Gatenby R, Hartz W, Broder G. Diagnosis of osteomyelitis by MR imaging. AJR 1988; 150:605-610. ↩
- Collins MS, Schaar MM, Wenger DE, Mandrekar JN. AJR August 2005, Volume 185, Number 2. T1-Weighted MRI Characteristics of Pedal Osteomyelitis. ↩
- Averill LW, Hernandez A, Gonzalez L, Peña AH, Jaramillo D. Diagnosis of Osteomyelitis in Children: Utility of Fat-Suppressed Contrast-Enhanced MRI. AJR 2009; 192:1232–1238. ↩
- Manaster BJ. Musculoskeletal Imaging: The Requisites, 3rd ed. Philadelphia, PA: Mosby Elsevier, 2007:545-64. ↩
- Rajashanker B, Whitehouse RW. Chapter 53: Bone, joint and spinal Infection. In: Adam A, Dixon AK, Gillard JH, et al. editors. Grainger & Allison’s Diagnostic Radiology, 6th ed. New York, NY: Churchill Livingstone, 2015:1241-2. ↩
- Morrisy RT. Bone and joint infections. In: Morrisy RT, ed. Pediatric orthopaedics. Philadelphia: Lippincott, 1990:539-561. ↩
- Dangman BC, Hoffer FA, Rand FF, O’Rourke EJ. Osteomyelitis in children: gadolinium-enhanced MR imaging. Radiology 1992:182:743-748. ↩
- Yochum TR, Rowe LJ. Essentials of skeletal radiology. Lippincott Williams & Wilkins. (1996) ISBN:0683093304. ↩
- Pugmire BS, Shailam R, Gee MS. Role of MRI in the diagnosis and treatment of osteomyelitis in pediatric patients. World J Radiol. 2014 Aug 28. 6 (8):530-7. ↩
- Calhoun JH, Manring Adult osteomyelitis. Infect Dis Clin North Am. 2005 Dec; 19(4):765-86. ↩
- Rosenberg AE. Chapter 26: Bones, joints and soft tissue tumors. In: Kumar V, Abbas AK, Fausto N, et al. editors. Robbins and Cotran Pathologic Basis of Disease. 8th ed. Philadelphia, PA: Saunders Elsevier, 2010:1221-2. ↩
- Lew DP, Waldvogel FA. Lancet. 2004 Jul 24-30; 364(9431):369-79. ↩
- Krogstad P. Osteomyelitis. In: Feigin RD, Cherry JD, Demmler-Harrison GD, Kaplan SL, editors. Textbook of Pediatric Infectious Diseases. 6th Edition. PA, USA: Saunders Elsevier; 2009. pp. 725–742. ↩
- Gutierrez K. Bone and joint infections in children. Pediatr. Clin. N. Am. 2005;52(3):779–794. ↩