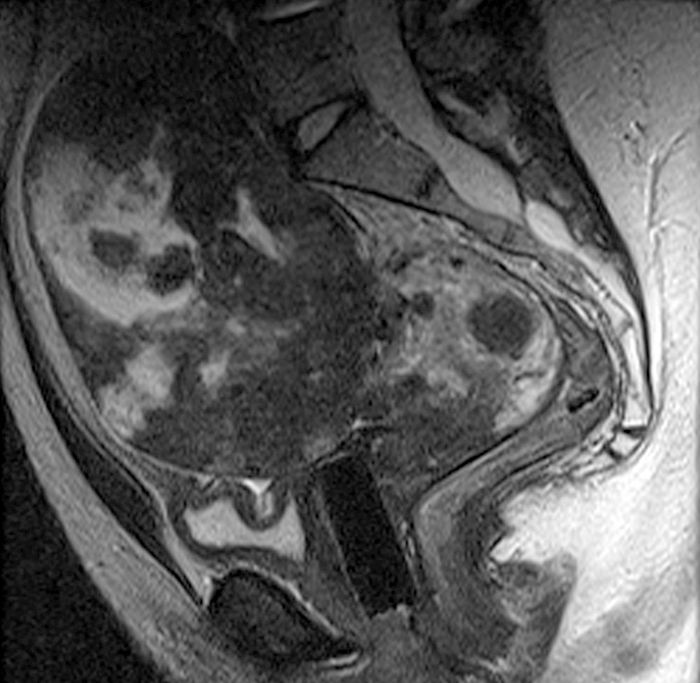
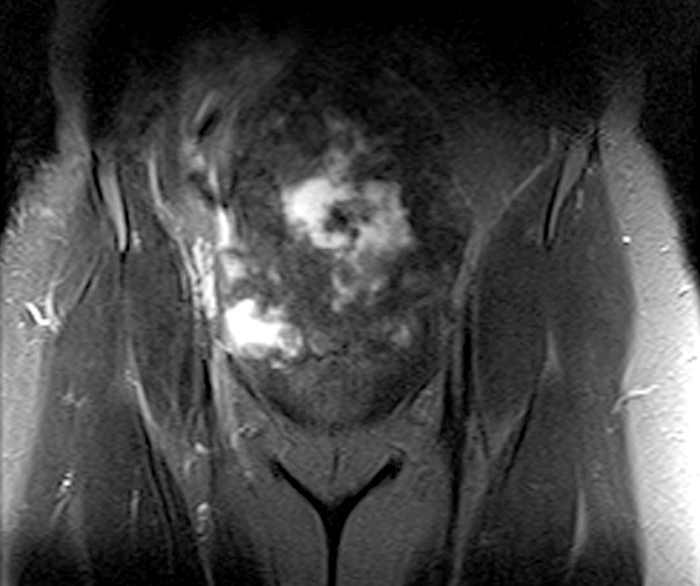
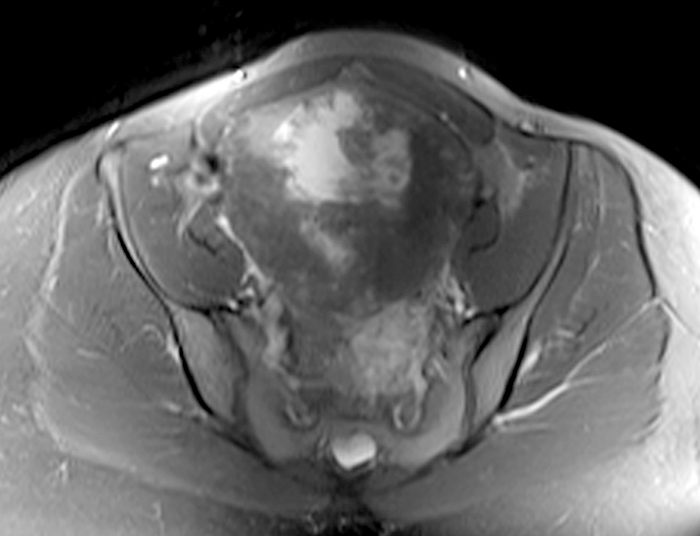
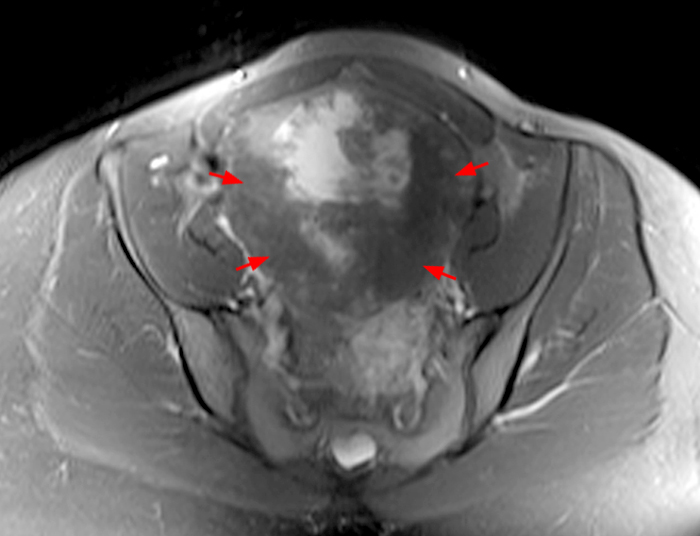
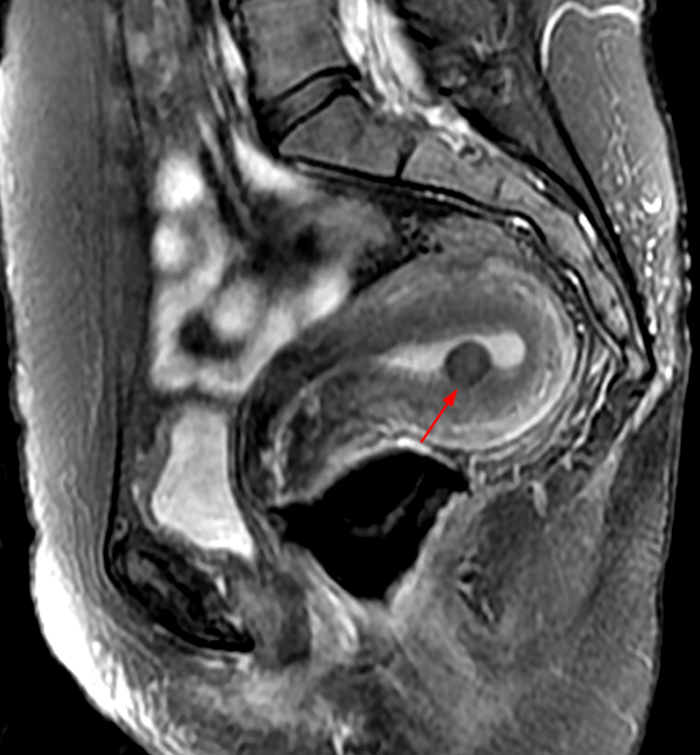
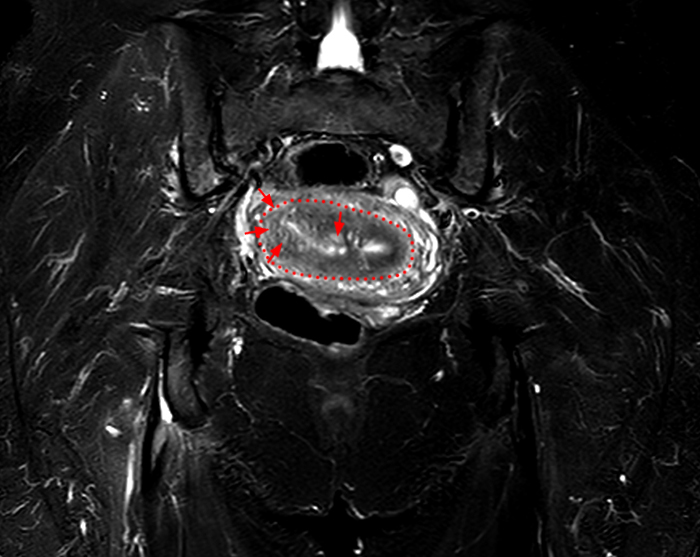
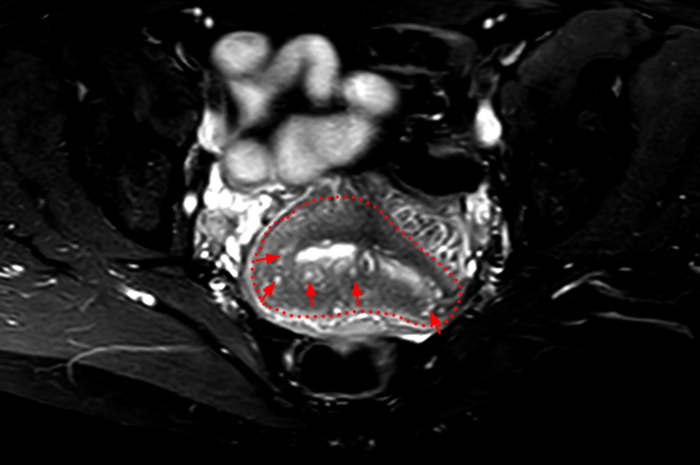
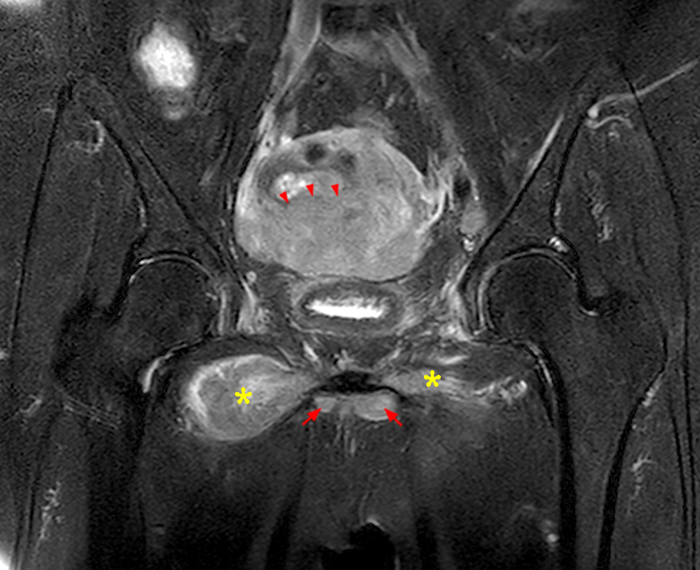
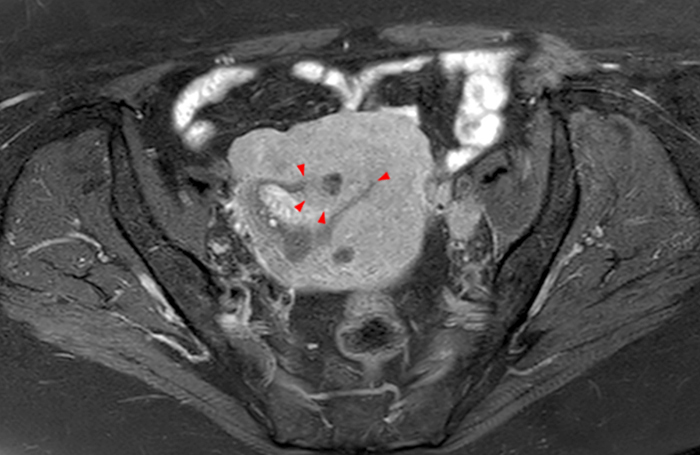
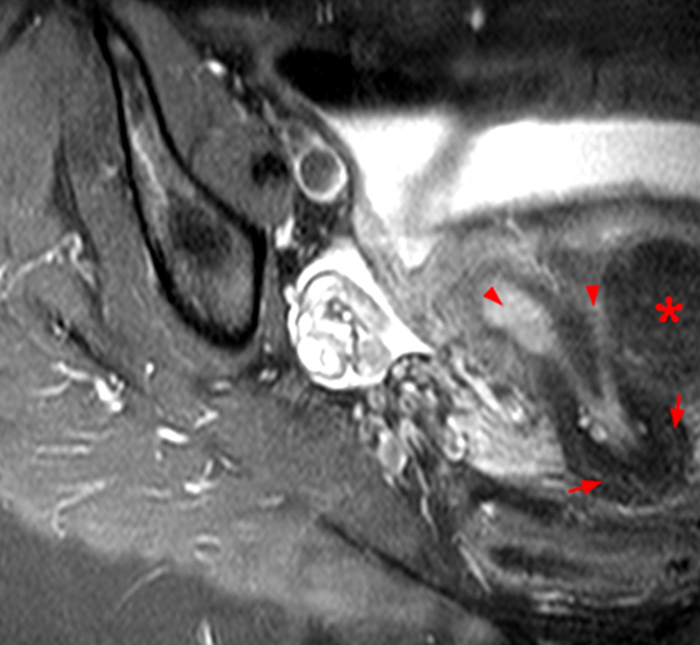
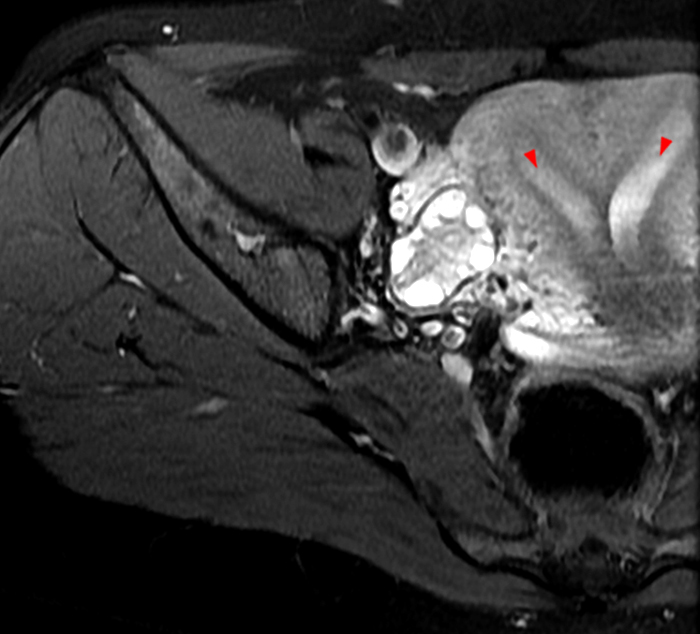
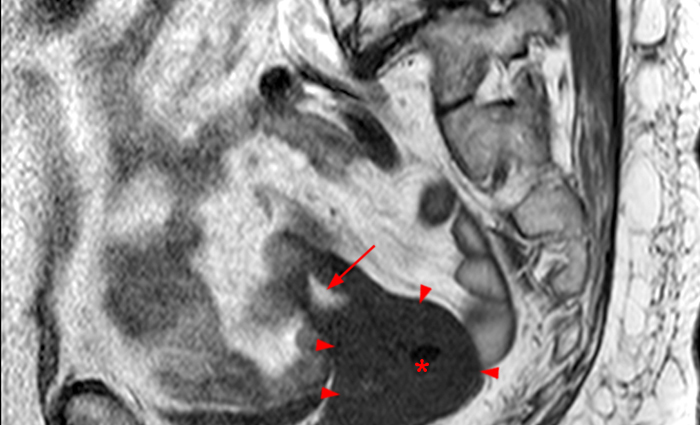
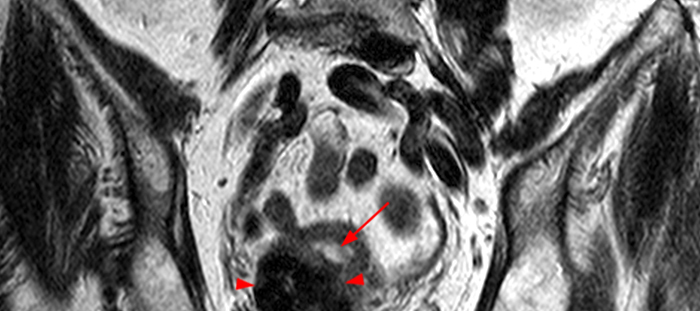
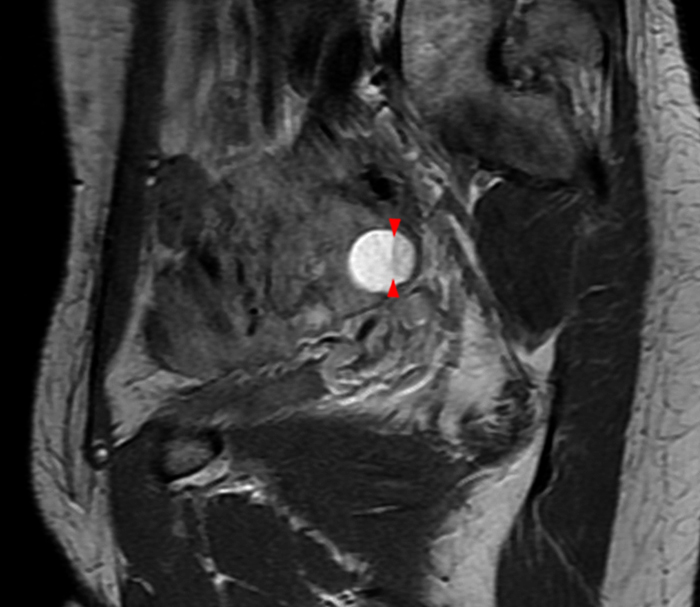
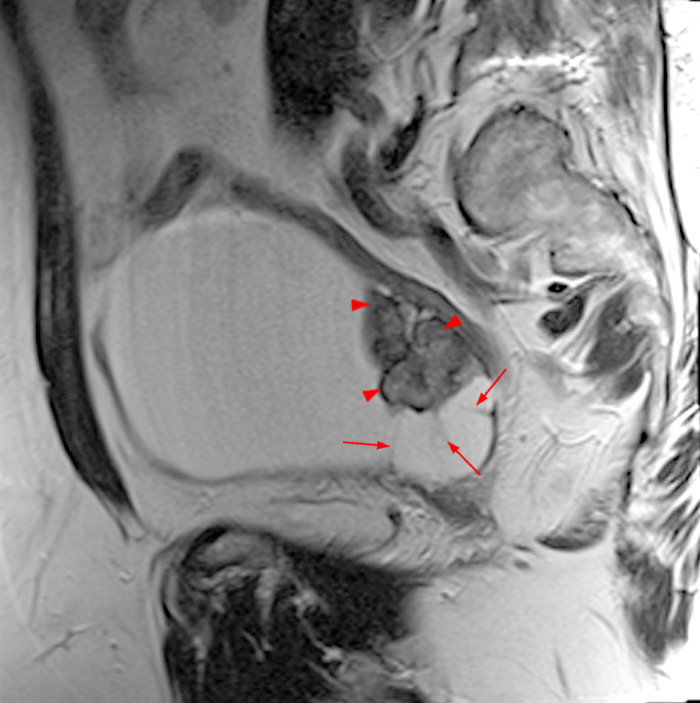
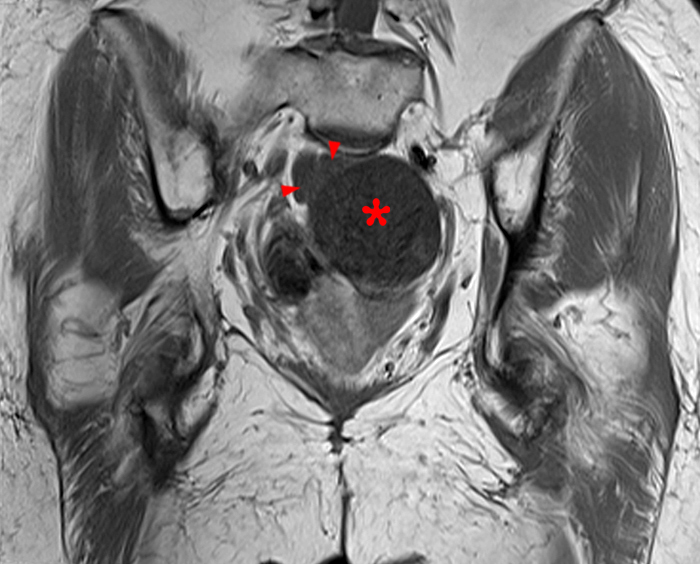
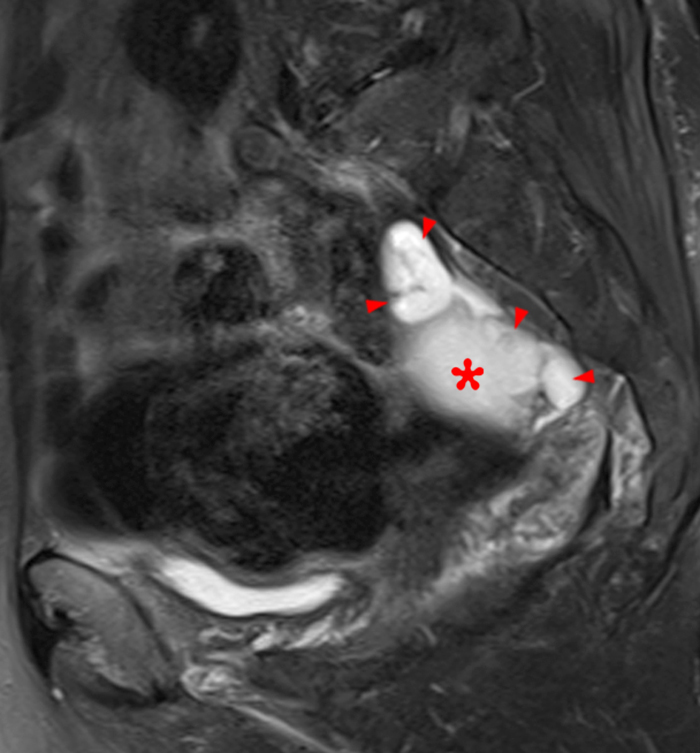
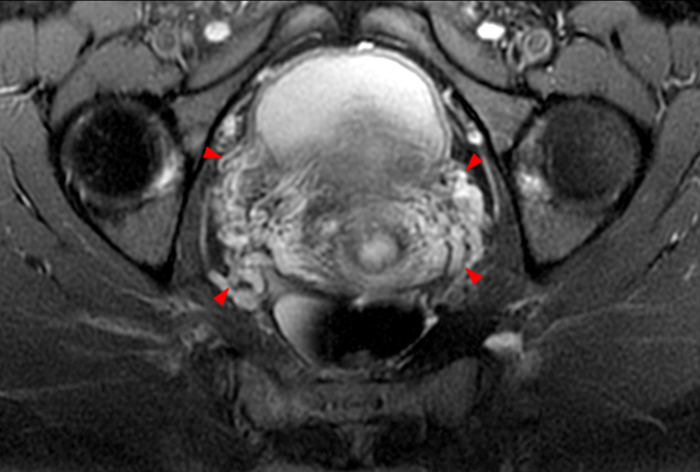
Clinical History: A 35 year-old female runner with bilateral hip pain presents for MRI of the pelvis. Sagittal and coronal T2-weighted (1a,b), axial intermediate-weighted with fat suppression (1c), and coronal post-gadolinium T1-weighted with fat suppression (1d) images of the pelvis are provided. What are the findings? What is your diagnosis?
Findings
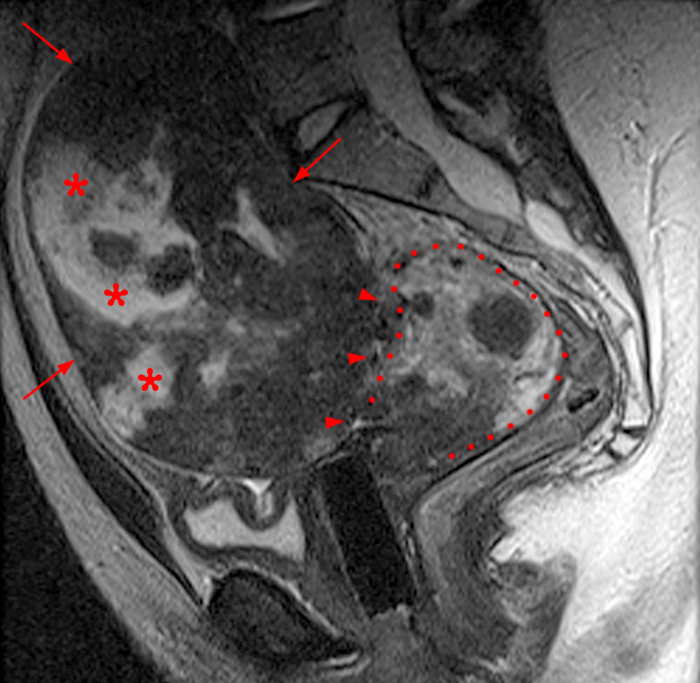
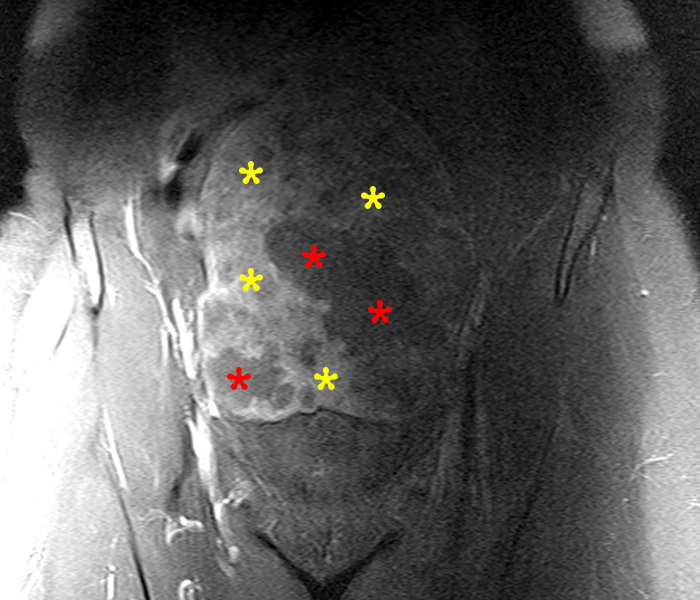
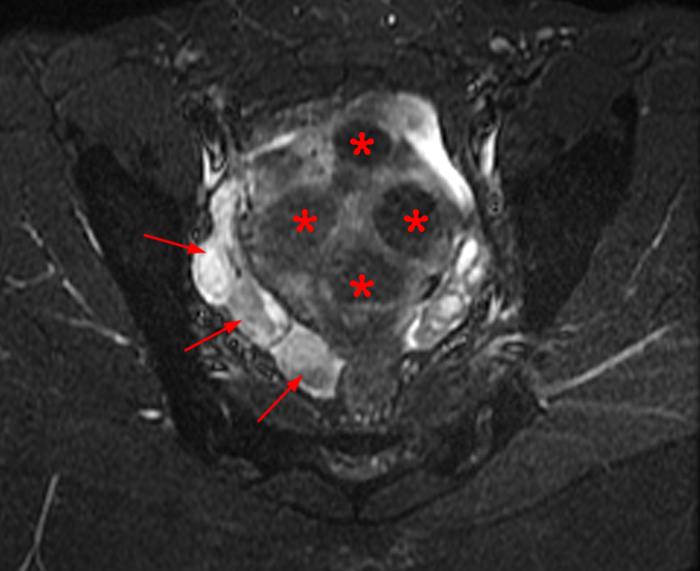
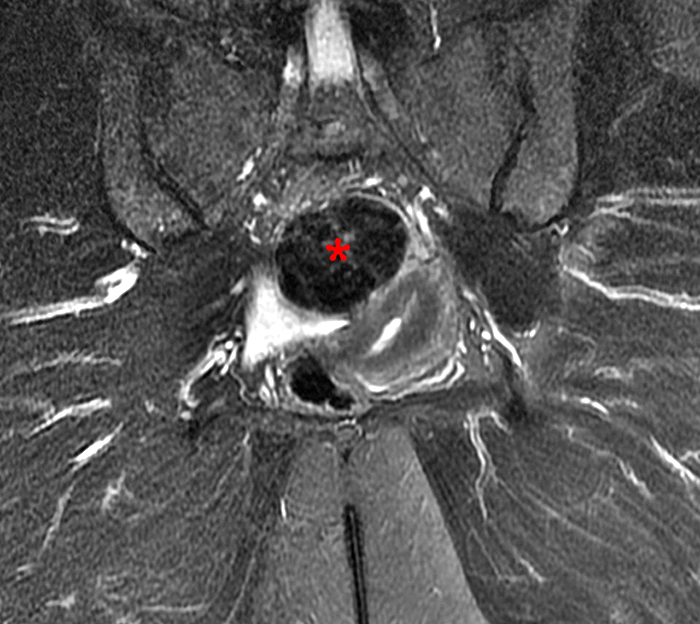
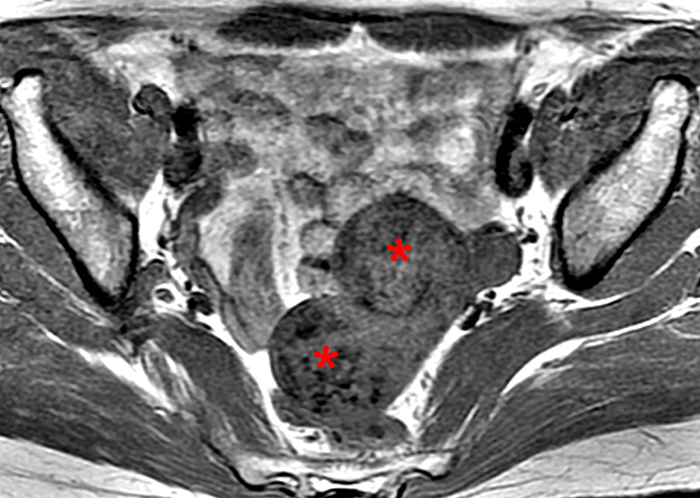
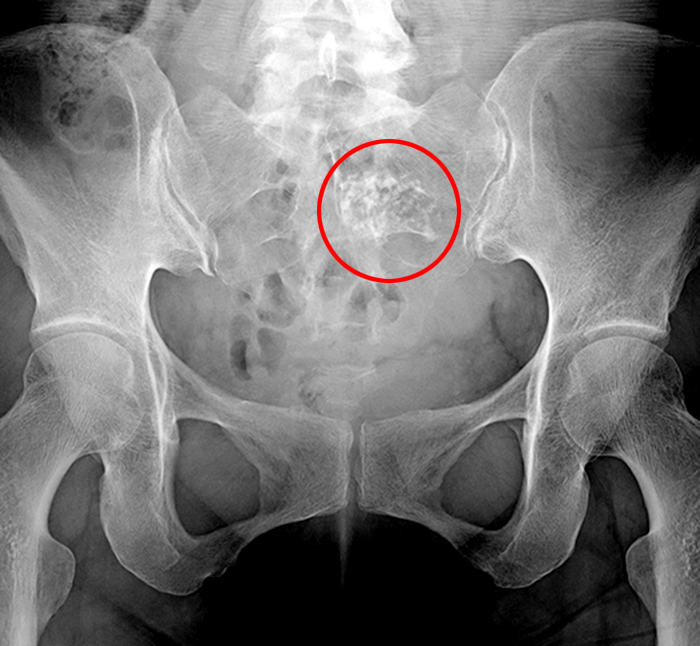
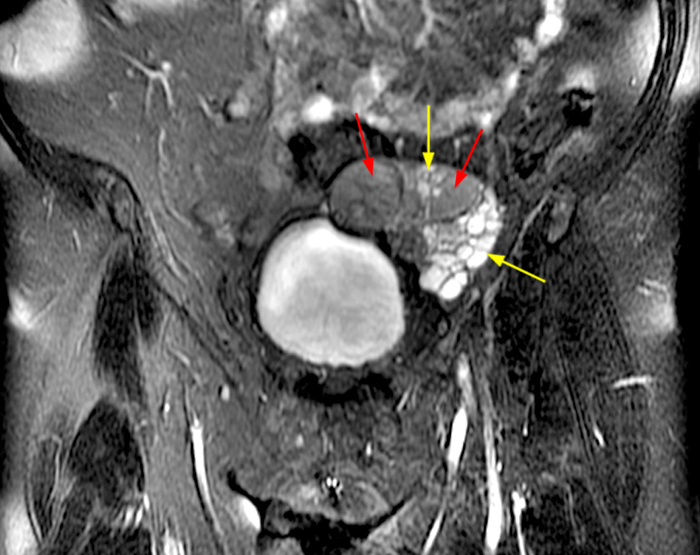
Figure 2:
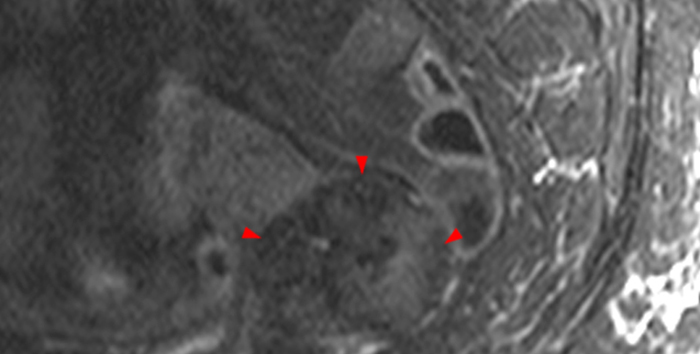
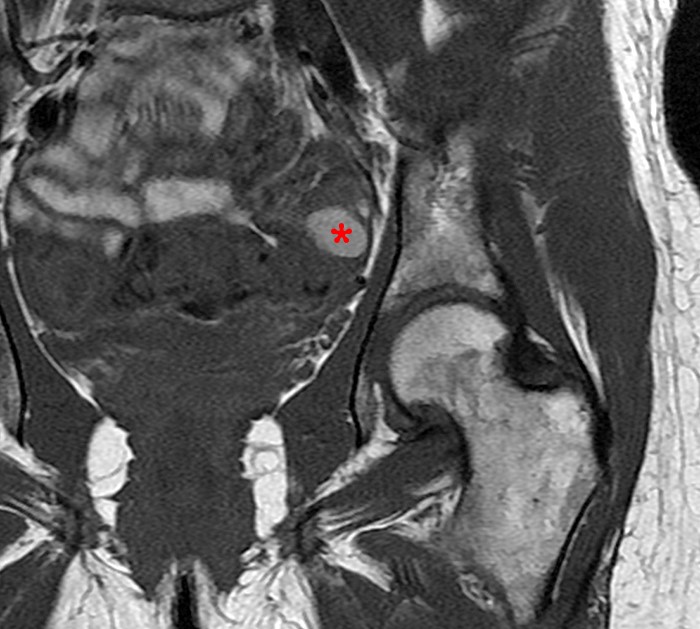
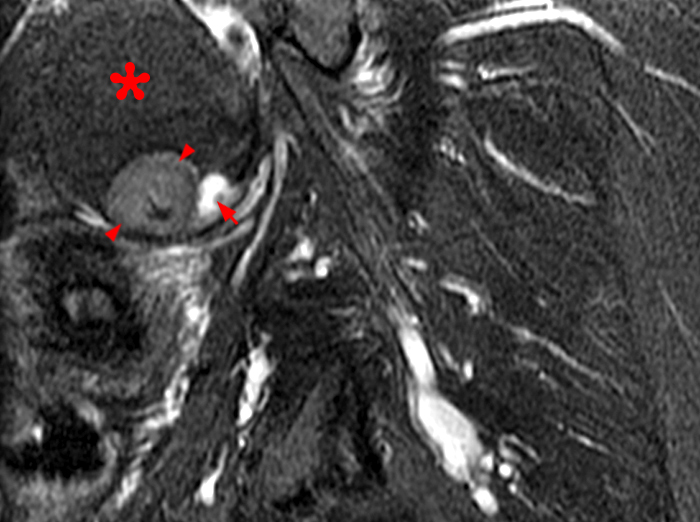
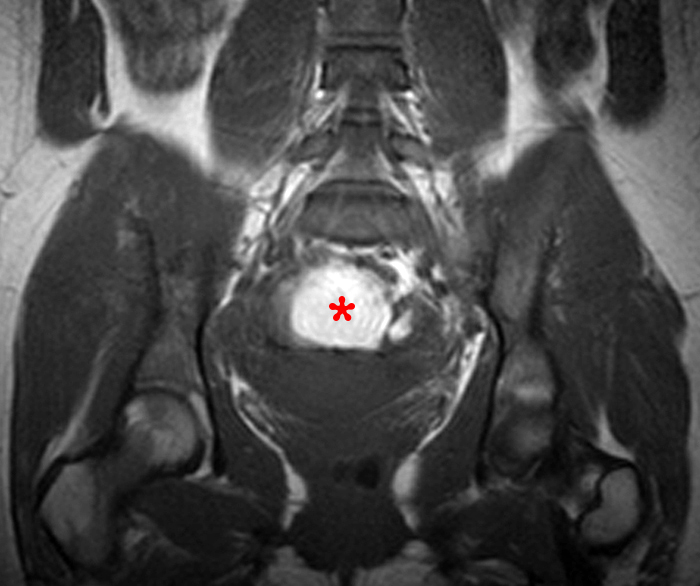
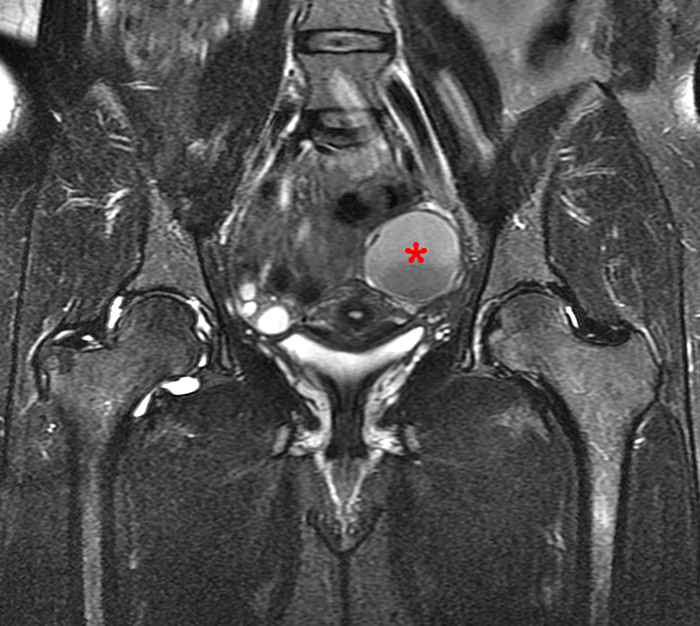
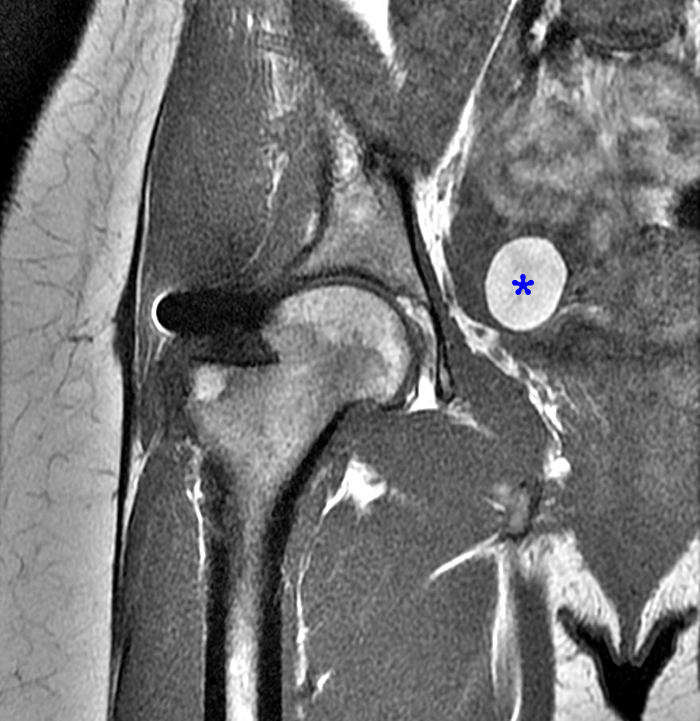
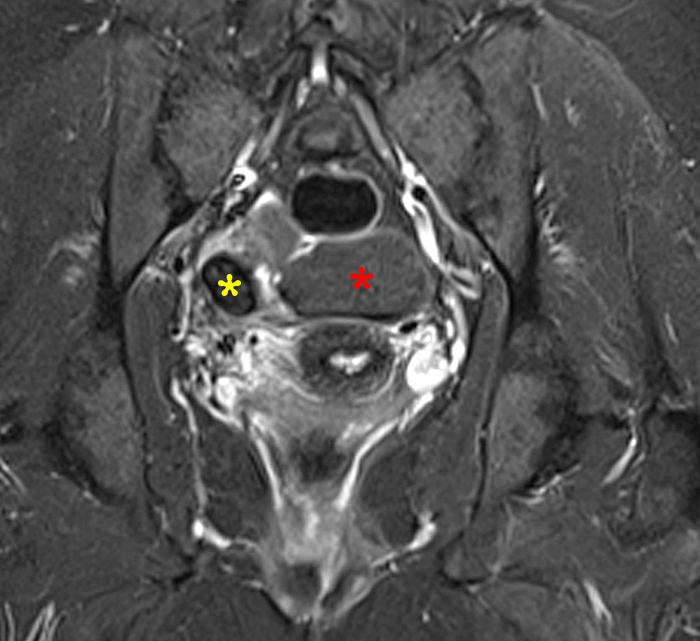
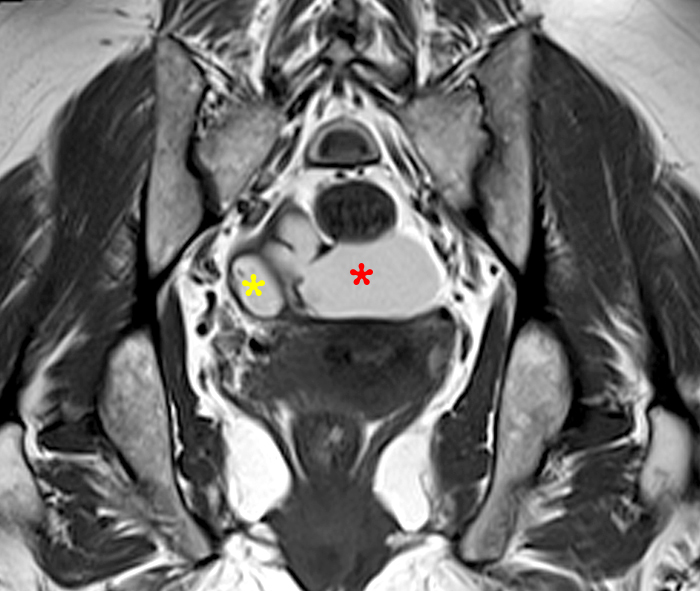
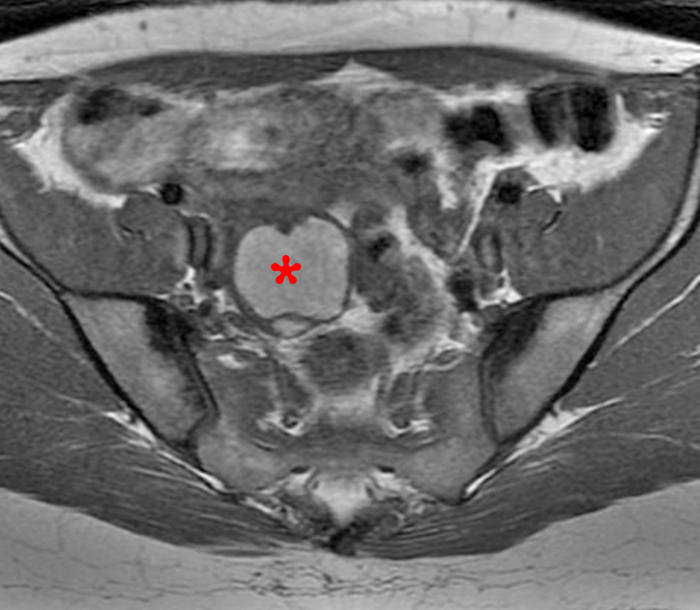
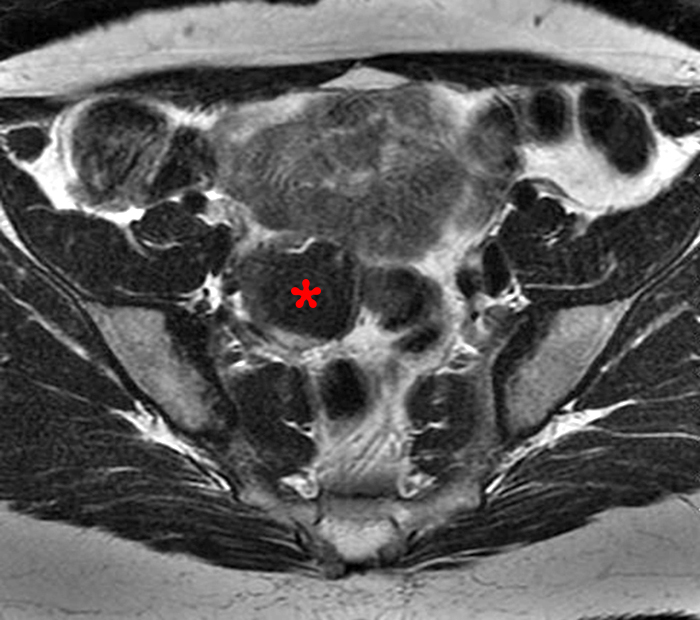
A 9 x 11 x 14 cm mass (arrows) arises from the uterine fundus (dotted outline) extending cephalad. Sagittal T2-weighted (2a) and coronal T2-weighted with fat suppression (2b) images demonstrate the mass inseparable from the myometrium (red arrowheads) with predominantly heterogenous low signal, and also large areas of irregular fluid signal (asterisks). The axial intermediate weighted sequence with fat suppression image (2c) demonstrates signal intensity similar to skeletal muscle (short arrows). The coronal post-gadolinium T1-weighted with fat suppression image (2d) demonstrates very mild enhancement of the myometrium (yellow asterisks) and lack of enhancement of the central areas (red asterisks).
Diagnosis
Degenerating uterine fibroid with large areas of necrosis.
Introduction
Many non-musculoskeletal findings on routine hip and pelvis MRI studies are diagnostic and can simply be reported without the need for further imaging. However, because musculoskeletal pelvic MRI imaging techniques differ from gynecologic pelvic MRI techniques, additional imaging is occasionally indicated for incidentally discovered female pelvic findings. The intent of this article is to help the musculoskeletal radiologist become more comfortable diagnosing and adding clinical value when unexpected female pelvis findings are discovered. This is not meant to be all-inclusive web clinic of gynecologic pathology, but a review of the most common entities upon reviewing thousands of reported cases within our musculoskeletal MRI practice.
Uterus
Leiomyoma (fibroid)
Leiomyomas are the most common uterine neoplasm, occurring in 20%–30% of women of reproductive age.1 They are composed of smooth muscle with a varying amount of fibrous tissue and often calcification. Generally, they are asymptomatic; however, they can present with abnormal uterine bleeding and pain depending upon their location and size. Approximately 30% of hysterectomies performed in the United States are for symptomatic leiomyomas; among black women, this number is as high as 50%.2,3 Some researchers find that up to 80% of women with leiomyomas are asymptomatic and require no treatment4 whereas others find that 20%–50% of women with leiomyomas present with symptoms such as menorrhagia, dysmenorrhea, pressure, urinary frequency, pelvic and back pain, dyspareunia, constipation, or obstipation.5 MRI is the most accurate method of evaluation–both for diagnosis and for potential complications. Leiomyomas can be classified as submucosal (Figure 3), intramural (Figure 4), and subserosal (Figures 1 and 2). Large fibroids can outgrow their blood supply with subsequent degeneration altering the appearance. Identifying and describing them will help the clinician appropriately address symptomatic fibroids.
Nondegenerated uterine leiomyomas have a typical MRI appearance: well-circumscribed masses with homogeneously decreased T2-weighted signal intensity compared with the normal myometrium6 and intermediate to decreased T1 signal with mild post gadolinium enhancement (Figure 4). Histologically, non-degenerated leiomyomas are composed of whorls of uniform smooth muscle cells with various amounts of intervening collagen.7 Cellular leiomyomas, which are composed of compact smooth muscles cells with little or no collagen, can have relatively increased T2-weighted signal intensity and demonstrate homogeneous enhancement on contrast-enhanced images.8
Degenerating leiomyomas (Figures 1 and 2) have a varied appearance. Calcified areas will appear as decreased T1 and T2 signal. (Figure 5) Areas of cystic degeneration will appear as fluid which doesn’t enhance. Areas of myxoid degeneration will be of increased T2 signal with minimal gadolinium enhancement. Variable T1-weighted signal intensity and low T2-weighted signal intensity are seen in necrotic leiomyomas that have not liquefied and formed cystic foci.8
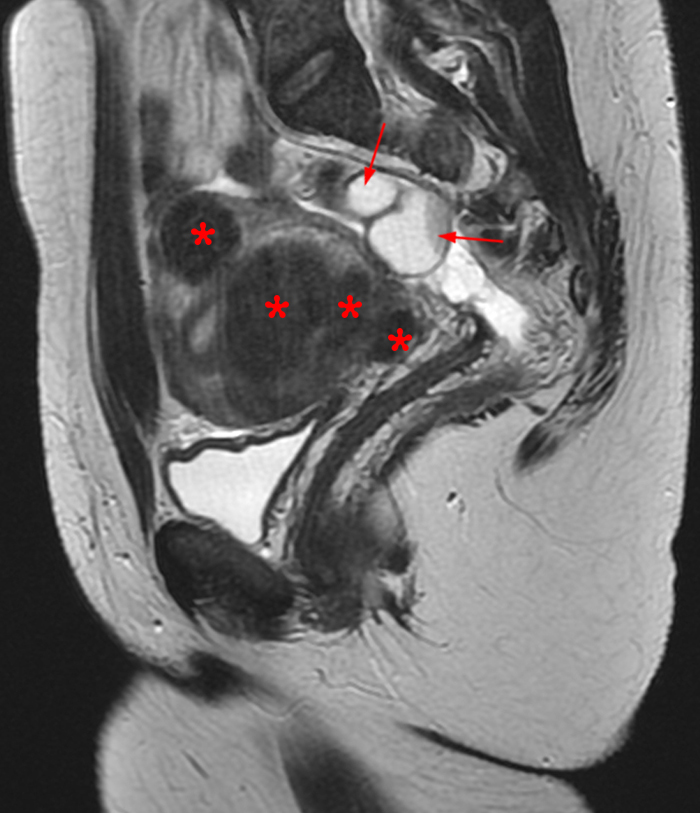
Figure 4:
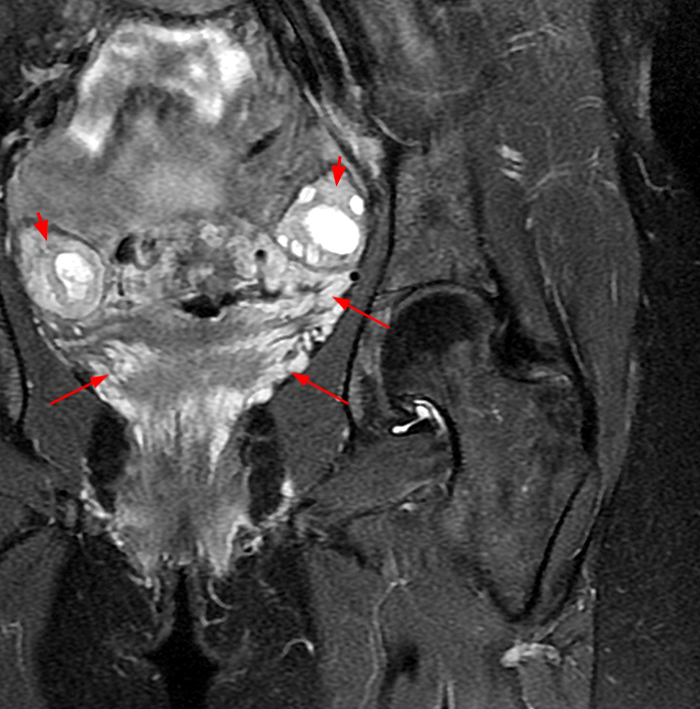
32 year-old female with known uterine fibroids presenting with pain. Sagittal T2-weighted (4a) and axial T2-weighted with fat suppression images (4b) demonstrate multiple fairly discrete foci of decreased signal within the uterine body and fundus (asterisks). The post gadolinium coronal T1-weighted fat suppression image (4c) demonstrates mild to moderate enhancement of the fibroids (asterisks) and diffuse moderate enhancement of the surrounding normal myometrium. The enlarged uterus also produces mass effect upon the right fallopian tube with resulting mild to moderate hydrosalpinx (4a and 4b arrows).
Figure 5:
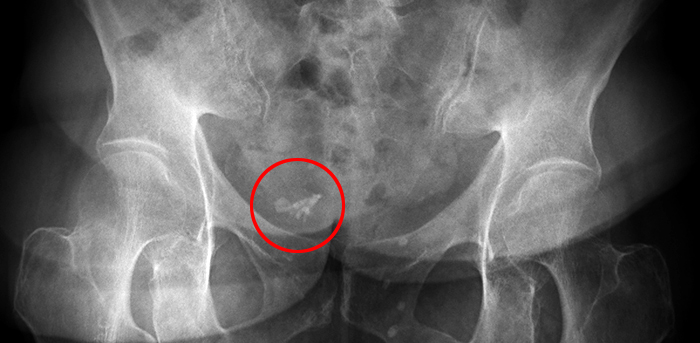
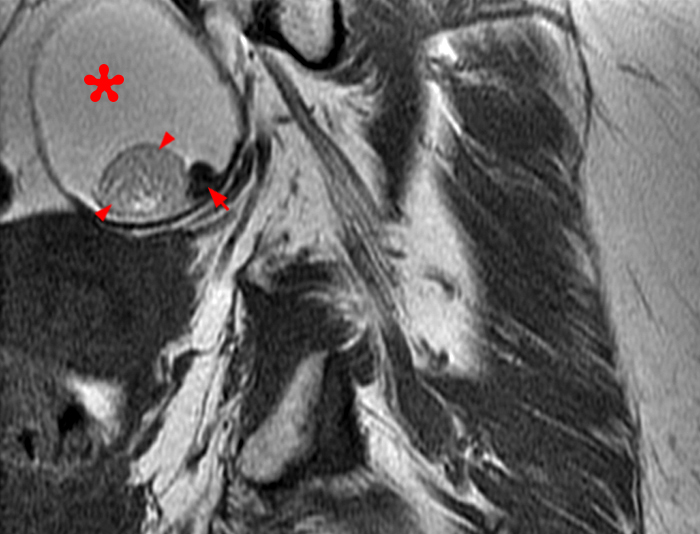
53 year-old female with known uterine fibroids. Coronal T2-weighted fat suppressed (5a) and axial T1-weighted (5b) images demonstrates two posterior fibroids (asterisks) with speckled areas of decreased signal intensity, especially visible on the T1-weighted image corresponding to calcifications (circle) demonstrated on the pelvis radiograph (5c).
Adenomyosis
Adenomyosis is ectopic endometrial gland tissue and stroma within the myometrium with reactive hypertrophy of the surrounding myometrial smooth muscle.9 Adenomyosis likely results from direct invasion of the basal endometrium into the myometrium. Adenomyosis occurs in focal and, more commonly, diffuse forms.9 Clinically, adenomyosis and leiomyomata can present similarly with dysmenorrhea or menorrhagia.
With MR imaging, the diffuse form of adenomyosis (Figure 6) appears as a thickened junctional zone (inner myometrium) on T2-weighted images.10A junctional zone measuring 12 mm thick or greater is highly predictive of adenomyosis.10 The low signal intensity of adenomyosis on T2- weighted images is due to the reactive, dense smooth muscle hypertrophy that surrounds the embedded endometrial glands.11 Small foci of high signal intensity on T2-weighted images represent the ectopic endometrial glands. Some of these ectopic endometrial glands also have corresponding high signal intensity on T1-weighted images, corresponding to functional hemorrhage.12,13
The focal form of adenomyosis appears as a poorly marginated area of low signal intensity within the myometrium on T2-weighted images.12 This has a similar appearance to a leiomyoma though less well-defined.
Figure 6:
50 year-old female evaluated for hip pain and possible labral tear. Coronal STIR (6a) and axial intermediate-weighted fat-suppressed (6b) images demonstrate a thickened junctional zone (dotted outline) with small foci of ectopic endometrial glands (small arrows) representing adenomyosis.
Neoplasm
In the United States, endometrial cancer is the most common malignancy of the female pelvis. Endometrial adenocarcinoma accounts for 90% of the endometrial cancers.14 The typical MRI appearance (Figure 7) is of a mass in the endometrium with T1 signal of or lower than normal endometrium and intermediate T2 signal, lower than the endometrium but greater than the myometrium. The mass demonstrates less post-gadolinium enhancement than the myometrium.15 Other findings suspicious for an endometrial cancer are fluid within the endometrial canal of a post-menopausal patient along with irregularity of the endometrium, an irregular mass within the canal, or disruption of the interface of the endometrium with the myometrium.14
Uterine sarcomas are rare, representing only 2-6% of uterine malignancies. Approximately one-third of uterine sarcomas are leiomyosarcomas.16 They can originate from the connective tissue of uterine blood vessels, in a preexisting leiomyoma, or de novo from uterine musculature.17 However, the prevalence of a benign uterine leiomyoma undergoing sarcomatous change is reported to be only 0.1%–0.8%.18 A leiomyosarcoma usually presents as massive uterine enlargement with irregular central zones of extensive hemorrhage and necrosis, possibly with calcific foci. The opening case in question (Figures 1 and 2) has an appearance suspicious for leiomyosarcoma. An irregular margin of a uterine leiomyoma on MRI is suggestive of sarcomatous transformation, but the specificity of this finding is unknown.17
Figure 7:
A 71 year-old female presented with a two-month history of pelvic and hip pain limiting ambulation. Coronal (7a) and sagittal (7b) T2-weighted images with fat suppression and an axial (7c) intermediate-weighted with fat suppression image demonstrate extensive endometrial adenocarcinoma involvement enlarging the uterus and disrupting the endometrium/myometrium interface (arrowheads). Extension into the cervix and vagina is present (asterisks), as well as posterior bladder wall invasion (7b arrows). Regional lymph nodes and bone metastases were also seen. Tumor extends into the bilateral ischiocavernosus muscles (7a and 7b short arrows) as well as the bilateral hip adductor musculature (7a yellow asterisks).
Septate uterus
A septate uterus (Figures 8 and 9) is one form of Mullerian duct anomaly (MDA) that arises from interruption of normal uterine development. MDAs are a broad spectrum of abnormalities that are often associated with primary amenorrhea, infertility, obstetric complications, and endometriosis. They are commonly associated with renal and other anomalies. However, MDAs are not associated with ovarian anomalies.19 The difference between a septate and a bicornuate uterus is determined by the thickness of the fundal contour. The septate uterus maintains a normal fundal contour where a bicornuate uterus has a distinct concavity. A septate uterus, the most common form of MDA, can be complete or partial.
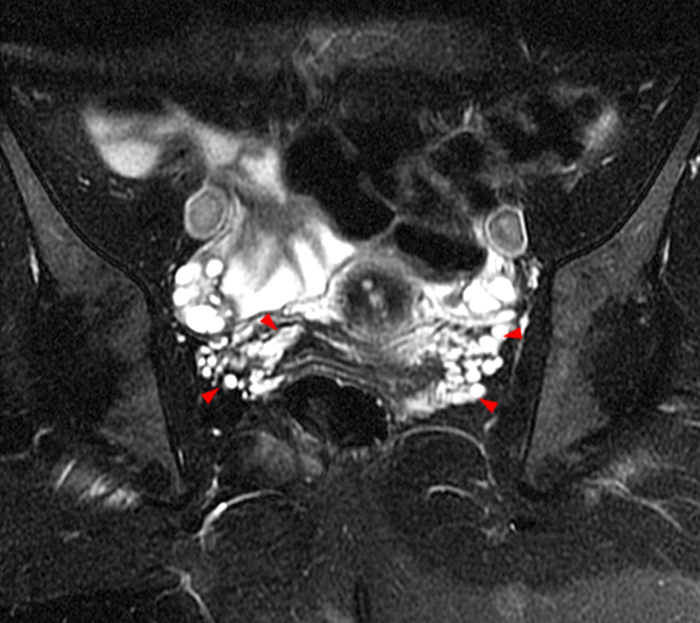
Figure 8:
40 year-old female presents for evaluation of hip pain. Incidental finding of a septate uterus as well as a left myometrial fibroid (asterisk). Coronal T2-weighted with fat suppression (8a) and axial intermediate-weighted with fat suppression (8b) images demonstrate a single cervix (small arrows) with two distinct endometrial cavities (arrowheads).
Cervix
Nabothian cyst
Nabothian cysts are very common incidental findings, representing rentention cysts of the cervical glands due to chronic inflammation and scarring.20
The fluid signal appearance is due to the accumulation of mucus. Nabothian cysts can be single or multiple within the cervical fibrous stroma, are round, and have regular borders. They are of increased T2 signal and of intermediate to slightly increased T1 signal (Figure 10).
Cervical leiomyoma
Benign leiomyomas typically occur in the uterine body and fundus. However, rarely they can arise in the cervix, accounting for 0.6-10% of uterine leiomyomata.21 The MRI appearance (Figure 11) is the same as for a uterine leiomyoma.
Figure 11:
76 year-old being evaluated with a lumbar spine MRI for radiating low back pain. Sagittal T1-weighted (11a), coronal T2-weighted (11b), and sagittal STIR (11c) images demonstrate a large, well-demarcated area of decreased signal (arrowheads) within the right side of the cervix classic for a leiomyoma. The T1-weighted image demonstrates a focus of calcification (asterisk) which is also seen (circle) on the corresponding lumbar spine radiographs (11d). The endometrial canal is also slightly distended (small arrows) with fluid of increased T1 and T2 signal, likely due to hemorrhage.
Cervical carcinoma
Cervical carcinoma typically occurs in younger women with an average patient age of 45 years.22 Cervical carcinoma has intermediate T2-weighted signal intensity, which can be seen as a disruption in the normal band-like low signal fibrous stromal ring. Cervical carcinoma can have a wide variety of morphologic features including being exophytic, infiltrating, or endocervical with a barrel shape. In young women, cervical carcinoma usually originates from the squamocolumnar junction and tends to be more exophytic; in older women it originates more often in the endocervical canal. The bulk of the tumor is centered at the level of the cervix but may protrude into the vagina or invade the lower myometrium.23
Ovaries/Adnexa
Simple ovarian cyst
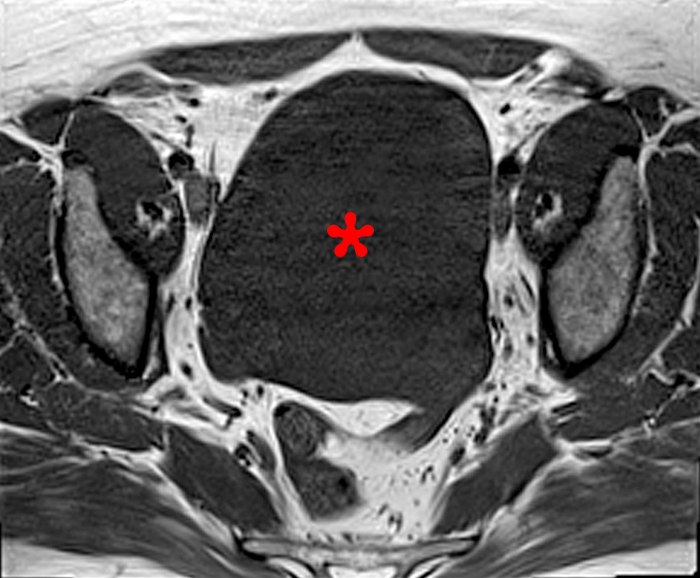
A simple, unilocular ovarian cyst (Figure 12) is a common incidental finding in both pre- and postmenopausal women. However, the postmenopausal ovary tends to contain fewer cysts of smaller size. When <3 cm in size, demonstrating a thin wall (≤3 mm), and simple fluid characteristics, these cysts can be considered benign in both populations. Studies that specifically examined the premenopausal ovary have shown the risk of malignancy in unilocular cysts less <5 cm in an asymptomatic woman approaches zero.24
Figure 12:
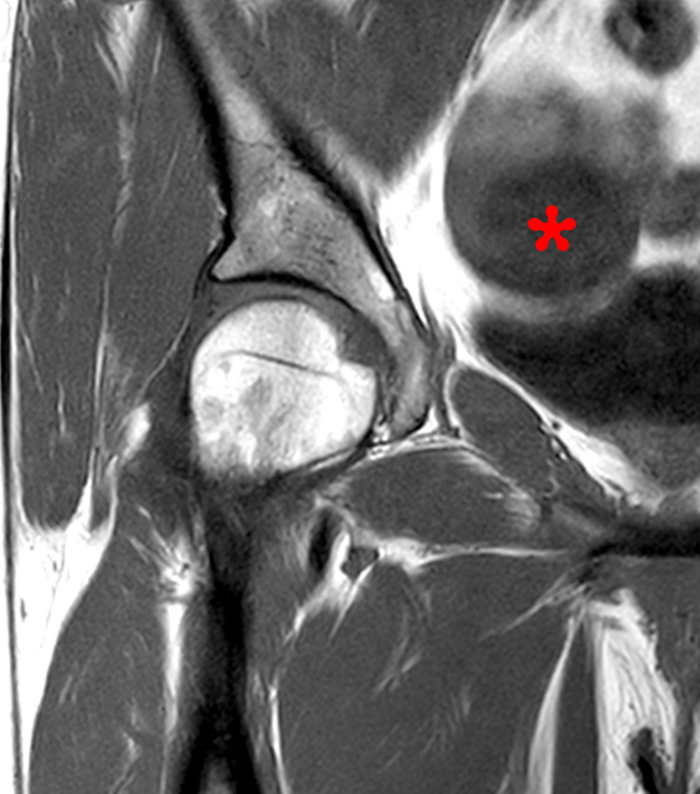
46 year-old female being imaged for right hip pain. Axial intermediate-weighted with fat saturation (12a) and coronal T1-weighted (12b) images demonstrate a well demarcated simple fluid signal focus (asterisks) within the right ovary, along with ovarian follicles, consistent with a simple ovarian cyst.
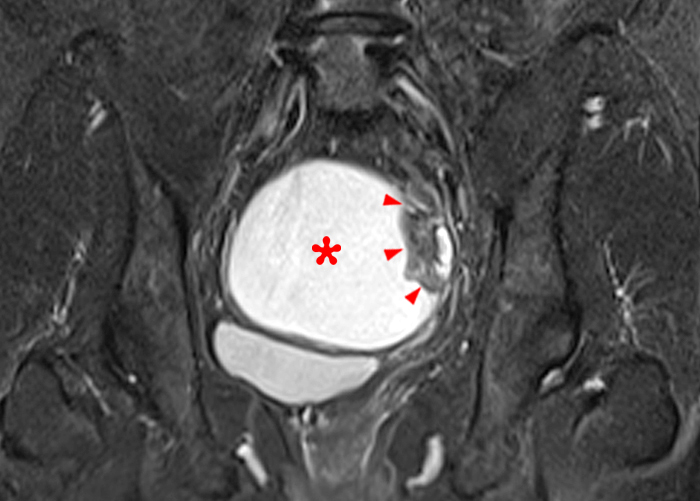
Hemorrhagic ovarian cyst
The MRI appearance of a hemorrhagic ovarian cyst (Figure 13) can be variable, depending on the age and amount of the hemorrhagic component. In general, they tend to contain high T1 signal, intermediate to high T2 signal, and frequently demonstrate a fluid-fluid level. Hemorrhagic cysts should retain high signal on fat-suppressed T1-weighted images, which differentiates them from dermoids. Hemorrhagic cysts also tend to have thicker walls than simple cysts and may exhibit post-contrast wall enhancement.24
Figure 13:
35 year-old female being evaluated for left hip and groin pain. Sagittal intermediate- weighted image (13a) demonstrates a well demarcated fluid signal focus within the left ovary with a fluid-fluid level (arrowheads). The coronal T1-weighted image (13b) demonstrates the focus to be of increased signal, consistent with a hemorrhagic cyst (asterisk). The coronal intermediated-weighted with fat suppression image (13c) demonstrates cysts within both ovaries (short arrows). Multiple prominent para-uterine and adnexal vessels (arrows) also suggest a diagnosis of pelvic venous congestion syndrome.
Dermoid
Most dermoids are unilocular, contain sebaceous fluid, and are commonly referred to as mature cystic teratomas or dermoid cysts. Although these are usually asymptomatic and are incidental findings in young women, the standard treatment is surgical removal because of their potential to cause ovarian torsion or for the cyst to rupture. There is also a rare chance of malignant degeneration to squamous cell carcinoma.24
MRI has a high sensitivity for the presence of fat within the sebaceous component, which is characteristic of nearly all these lesions. (Figures 14 and 15) The sebaceous component is of very high signal intensity on T1-weighted images and is somewhat variable on T2-weighted images.25 Fat suppression can differentiate the fat content from other hemorrhagic lesions that appear hyperintense on T1-weighted images, such as hemorrhagic cysts and endometriomas.
Figure 14:
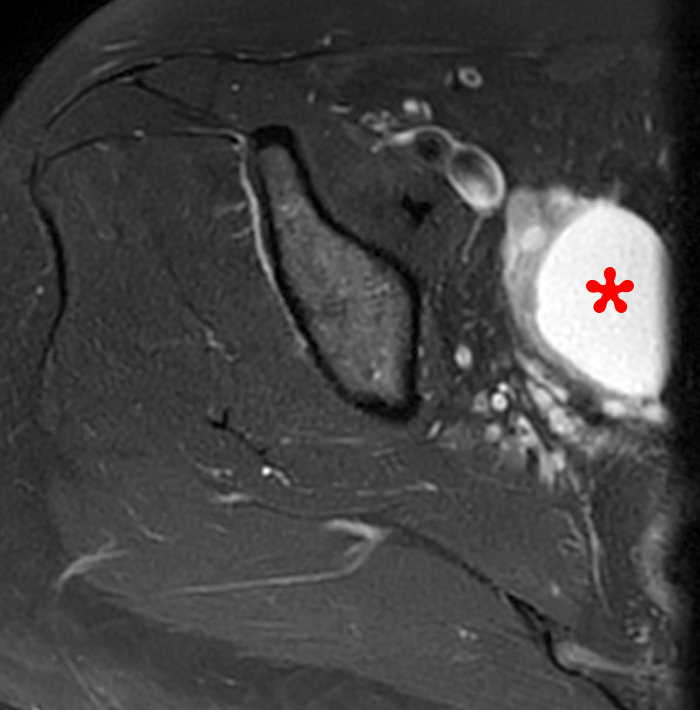
67 year-old female being evaluated for bilateral hip pain and possible avascular necrosis. Coronal T1 (14a) and fat-suppressed T2-weighted (14b) images demonstrate a large, thin-walled adnexal mass arising from the left ovary. The majority of the mass follows fat signal (asterisk). Two smaller inferior nodules are also present, one with an area of central fat (arrowheads), and one with a small cystic component (arrow). This fat containing mass is consistent with a dermoid.
Endometrioma
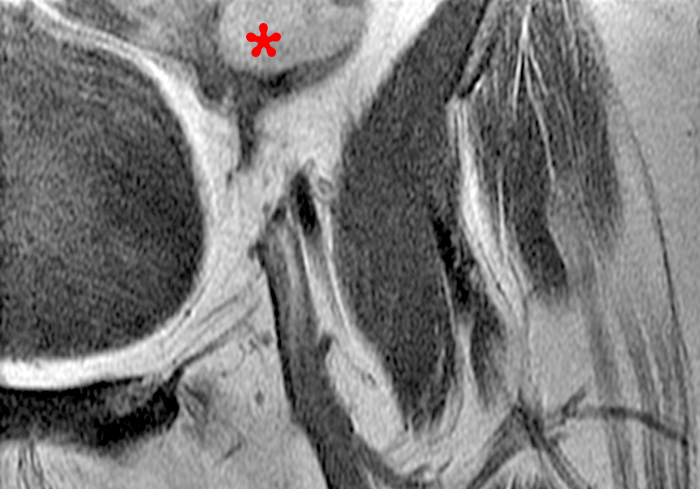
An endometrioma is a collection of uterine endometrial cells that implant, commonly on the ovary, and enlarge and repeatedly hemorrhage in response to hormonal stimulation.26 A cystic lesion with a hyperintense focus on a T1-weighted images with corresponding relative decreased signal on a T2-weighted images (termed “T2 shading”) (Figure 16) is the classic appearance of an endometrioma.26 However, this T2 shading can also be seen in other hemorrhagic adnexal lesions, particularly a hemorrhagic cyst, with a specificity for endometrioma of only 83%.27 The additional finding of a T2 dark spot along but not within the cyst wall, when not also of decreased T1 signal as can be seen with calcification, and in the absence of post-gadolinium enhancement, is specific for chronic hemorrhage and thus has higher specificity for an endometrioma (Figure 17).28 In one study, the T2 dark spot was seen in 36% of the endometriomas; thus the absence of this sign does not preclude the diagnosis of an endometrioma.28 Endometriomas can also often be strikingly dark on T2 weighted images (Figure 18).
Figure 16:
39 year-old female being evaluated for chronic hip pain. A coronal T2 with fat suppression image (16a) demonstrates a large focus in the left adnexa of classic T2 shading (red asterisk). This same focus on follow-up imaging for sports hernia evaluation 12 months later was completely gone, representing resolution of a hemorrhagic cyst. A small focus in the right ovary on the initial study increased in size between the studies and is demonstrated on the coronal T1-weighted image (16b) to be of increased signal, also consistent with a hemorrhagic cyst (blue asterisk).
Figure 17:
49 year-old female with sacral pain and known endometrial implants. A coronal STIR image (17a) demonstrates a large low signal central pelvic mass (red asterisk) and a smaller right sided mass with even lower signal intensity (yellow asterisk). Both masses demonstrate bright T1 signal (17b). A T2 dark spot is also present on sagittal imaging (17c).
Figure 18:
48 year-old female with a longstanding history of right adnexal mass, intermittently painful with menstruation. Axial T1-weighted (18a) and axial T2-weighted fat suppressed (18b) images demonstrate a right adnexal mass (asterisks). The mass is predominantly of increased T1 and decreased T2 signal intensity. Although this case is not pathologically proven, other serial studies including CT and US as well as clinical history are consistent with endometrioma.
Cystadenoma/cystadenofibroma
Serous cystadenoma (Figure 19) is a benign tumor that usually present as an unilocular cyst with a regular wall less than 3 mm thick, flat internal margins without internal septations, papillary projections or solid components, and with no significant contrast enhancement. Serous fluid shows low signal intensity on T1-weighted and high signal intensity on T2-weighted images similar to water. They are usually smaller and more often bilateral than mucinous cystadenomas. Serous cystadenomas of borderline malignancy may demonstrate small papillary projections.29
Serous cystadenofibroma (Figure 20) is a relatively rare, benign ovarian tumor. It often has a complex appearance on MRI, with both cystic and solid components, and can resemble a malignancy, due to the presence of irregular, thickened septations or nodular components. The fibrous component of the tumor demonstrates low-signal intensity (relative to skeletal muscle) on T2-weighted images.30
Mucinous cystadenoma (Figure 21) is a benign mucin-containing tumor, often larger than serous cystadenoma and usually unilateral. It usually appears as a multilocular cystic lesion with a thin regular wall and several septations, without solid components or nodularity and show no significant gadolinium enhancement. Rupture of a mucinous cystadenoma can produce pseudomyxoma peritonei.29
Figure 19:
52 year-old female being imaged for groin/buttock and left thigh pain. Fat suppressed T2 (19a) and coronal T1 weighted images demonstrate a left ovarian mass with solid (red arrows) and cystic components (yellow arrows). A coronal T1-weighted image from a hip exam partially includes the lesion and demonstrates an area of hemorrhagic or proteinaceous material within the lesion (asterisk). A nearly identical right ovarian mass is not shown. Bilateral serous cystadenomas were found at surgery.
Figure 20:
71 year-old female being evaluated with dedicated MRI pelvis imaging due to partial demonstration of a cystic mass on MRI lumbar spine imaging. Axial T1-weighted image (20a) demonstrates a large central pelvic area of homogenous intermediate signal intensity (asterisk). A coronal STIR image (20b) demonstrates the majority of the mass to be of simple fluid signal (asterisk) with a thin wall but with an area of slightly irregular intermediate to decreased signal intensity along the left wall (arrowheads). The sagittal T2-weighted image (20c) further demonstrates that area of irregular, nodular signal (arrowheads) along with thin septa (arrows). The axial T1-weighted post gadolinium with fat suppression image (20d) demonstrates enhancement of the nodular focus (arrowheads). At surgery, this was proven to be a serous cystadenofibroma.
Figure 21:
80 year-old female evaluated for right hip and leg pain following recent hip replacement. A presacral cystic structure partially imaged on a recent MR of the lumbar spine demonstrated interval growth since a MR lumbar spine two years prior. Coronal T1-weighted (21a) and fat-suppressed sagittal (21b) and coronal (21c) T2-weighted images from a follow-up MR of the pelvis demonstrate a large left presacral, thin walled focus of fluid signal (yellow asterisks). The slightly tortuous adjacent fluid signal focus (arrowheads) represents hydrosalpinx. Although this is not yet pathology proven, it is suspicious for a mucinous cystadenoma.
Pelvic venous congestion syndrome
Pelvic venous congestion syndrome (PVCS) is a challenging diagnosis and complex cause of chronic pelvic pain in female patients. PVCS can result from incompetent vein valves with gonadal vein reflux and pelvic venous engorgement. However, pelvic venous engorgement and gonadal vein reflux can be seen in patients without pelvic pain. Thus, MRI imaging alone is not specific for the diagnosis, but rather suggestive. The underlying cause of PVCS may be incompetent gonadal vein valves or structural causes such as left renal vein compression with an incompetent gonadal vein valve (nutcracker syndrome) or iliac vein compression (May-Thurner configuration) with reflux into the ipsilateral internal iliac vein.31 MRI findings that suggest PVCS are dilated parauterine varices, heterogeneous or T2-hyperintensity due to slow flow, presence of an arcuate vein crossing the midline, and vulvar and/or thigh varices.31
Figure 22:
31 year-old female being imaged for post-traumatic sacral pain. Axial intermediate-weighted fat suppressed image (22a) and coronal T2-weighted fat suppressed image (22b) demonstrate multiple very prominent adnexal, parauterine, paracervical, and paravaginal vessels (arrowheads) suggestive of pelvic venous congestion syndrome.
Ovarian neoplasm
Serous cystadenocarcinoma is the most common ovarian neoplasm and is responsible for about 40% of malignant ovarian neoplasms. Mucinous cystadenocarcinoma is less common and accounts for about 10% of ovarian malignancies. These tumors are seen as complex multilocular masses, usually with thick and irregular walls, septations, solid components and papillary projections of low signal intensity on T2-weighted images with contrast enhancement after gadolinium administration. The serous fluid of the cystic components has low to intermediate signal intensity on T1-weighted and high signal intensity on T2-weighted images. Some cases of serous cystadenocarcinoma demonstrate psammomatous calcifications. The signal intensity of mucinous content is variable depending on mucin concentration. These tumors can be very large, even greater than 12–15 cm. Serous cystadenocarcinoma is more frequently bilateral than mucinous cystadenocarcinoma.29
Pelvic floor
Bartholin gland cyst
Bartholin glands arise in the superficial perineal pouch of the urogenital triangle, and the ducts open into the posterolateral aspect of the vaginal vestibule. Ductal obstruction due to previous infection or inspissated mucus leads to retention of secretions and cyst formation. Bartholin gland cysts are the most common vulvar cysts, ranging in size from 1 to 4 cm, but can increase in size with repeated sexual stimulation.32 They are solitary round to oval cysts and are low to intermediate in signal intensity on T1-weighted sequences, depending on the mucin content of the cyst fluid, and are hyperintense on T2-weighted sequences. Simple cysts are thin-walled and unilocular, but septations can be seen. The cyst wall may be thickened and enhances if infected.33 The cysts arise within the superficial perineal pouch, inferior to the perineal membrane; on sagittal images they are seen below the inferior margin of the symphysis pubis.32
Figure 23:
38 year-old female being evaluated for pelvic pain and possible avascular necrosis. Sagittal T2-weighted fat suppressed (23a), axial intermediate-weighted fat suppressed (23b), and coronal STIR (23c) images demonstrate a very well demarcated simple fluid signal focus within the left perineum consistent with a Bartholin gland cyst (red asterisks).
Conclusion
Routine orthopaedic MR imaging of the hips, sacrum, and sacroiliac joints commonly reveals non-musculoskeletal findings in a female pelvis. Due to the techniques and field of view typically used in orthopaedic MR, these abnormalities may not always be adequately evaluated. However, it is incumbent upon the musculoskeletal radiologist to detect the pathology, make the diagnosis when possible, and guide the ordering physician when further workup is necessary.
References
- Deshmukh, SP, Gonsalves CF, Guglielmo FF, Mitchell DG. Role of MR imaging of uterine leiomyomas before and after embolization. Radiographics Oct 2012; Vol. 32, No. 6:E251-E281. ↩
- Becker ER, Spalding J, DuChane J, Horowitz IR. Inpatient surgical treatment patterns for patients with uterine fibroids in the United States, 1998-2002. J Natl Med Assoc 2005; 97(10):1336–1342. ↩
- Viswanathan M, Hartmann K, McKoy N et al.. Management of uterine fibroids: an update of the evidence. Evidence report/technology assessment no. 154, AHRQ publication no. 07-E011. Rockville, Md: Agency for Healthcare Research and Quality, 2007. ↩
- Parker WH. Myomectomy: laparoscopy or laparotomy? Clin Obstet Gynecol 1995; 38(2):392–400. ↩
- Buttram VC, Reiter RC. Uterine leiomyomata: etiology, symptomatology, and management. Fertil Steril 1981; 36(4):433–445. ↩
- Hricak H, Tscholakoff D, Heinrichs L et al.Uterine leiomyomas: correlation of MR, histopathologic findings, and symptoms. Radiology 1986; 158(2):385–391. ↩
- Prayson RA, Hart WR. Pathologic considerations of uterine smooth muscle tumors. Obstet Gynecol Clin North Am 1995; 22(4):637–657. ↩
- Okizuka H, Sugimura K, Takemori M, Obayashi C, Kitao M, Ishida T. MR detection of degenerating uterine leiomyomas. J Comput Assist Tomogr 1993; 17(5):760–766. ↩
- Novak ER, Woodruff JD. Adenomyosis (adenomyoma) uteri. In: Novak ER, Woodruff JD, eds. Novak’s gynecologic and obstetric pathology. Philadelphia, PA: Saunders, 1979; 280–290. ↩
- Mark AS, Hricak H, Heinrichs LW et al.Adenomyosis and leiomyoma: differential diagnosis with MR imaging. Radiology 1987; 163(2):527–529. ↩
- Reinhold C, McCarthy S, Bret PM et al. Diffuse adenomyosis: comparison of endovaginal US and MR imaging with histopathologic correlation. Radiology 1996; 199(1):151–158. ↩
- Outwater EK, Siegelman ES, Van Deerlin V. Adenomyosis: current concepts and imaging considerations. AJR Am J Roentgenol 1998; 170(2):437–441. ↩
- Togashi K, Ozasa H, Konishi I et al.Enlarged uterus: differentiation between adenomyosis and leiomyoma with MR imaging. Radiology 1989; 171(2):531–534. ↩
- Faria SC, Sagebiel T, Balachandran A, Devine C, Lal C, Bhosale PR. Imaging in Endometrial Carcinoma. Indian J Radiol Imaging 2015 Apr-Jun; 25(2); 137-147. ↩
- Akin O, Mironov S, Pandit-Taskar N, Hann LE. Radiol Clin North Am. 2007 Jan; 45(1):167-182. ↩
- Rha SE, Byun JY, Jung SE et al. CT and MRI of uterine sarcomas and their mimickers. AJR Am J Roentgenol 2003; 181(5):1369–1374. ↩
- Pattani SJ, Kier R, Deal R, Luchansky E. MRI of uterine leiomyosarcoma. Magn Reson Imaging 1995; 13(2):331–333. ↩
- Janus CJ, White M, Dottino P, Brodman M, Goodman H. Uterine leiomyosarcoma: magnetic resonance imaging. Gynecol Oncol 1989; 32(1):79–81. ↩
- Behr SC, Courtier JL, Qayyum A. Imaging of Mullerian Duct Anomalies. Radiographics Oct 2012; Vol. 32, No. 6:E233-E250. ↩
- De Graef M, Karam R, Juhan V, Daclin PY, Maubon AJ, Rouanet JP. High signals in the uterine cervix on T2-weighted MRI sequences. Eur Radiol 2003; 13:118-126. ↩
- Tiltman AJ. Leiomyomas of the uterine cervix: a study of frequency. Int. J. Gynecol. Pathol. 1998; 17(3): 231-234. ↩
- Reinhold C, Gallix BP, Ascher SM. Uterus and cervix.In: Semelka RC, Ascher SM, Reinhold C, eds. MRI of the abdomen and pelvis: a text atlas. New York, NY: Wiley-Liss, 1997; 585-660. ↩
- Nicolet V, Carignan L, Bourdon F, Prosmanne O. MR Imaging of Cervical Carcinoma; a practical Staging Approach. Radiographics Nov 2000; Vol. 20, No. 6:1539-1549. ↩
- Hubert J, Bergin D. Imaging the female pelvis: When should MRI be considered? Applied Radiology Oct 2011. ↩
- Outwater EK, Siegelman ES, Hunt JL. Ovarian teratomas: Tumor types and imaging characteristics. RadioGraphics 2001; 21:475-490. ↩
- Glastonbury CM. The shading sign. Radiology 2002; 224(1):199–201. ↩
- Outwater E, Schiebler ML, Owen RS, Schnall MD. Characterization of hemorrhagic adnexal lesions with MR imaging: blinded reader study. Radiology 1993; 186(2):489–494. ↩
- Corwin MT, Gerscovich EO, Lamba R, Wilson M, McGahan JP. Differentiation of ovarian endometriomas from hemorrhagic cysts at MR imaging; Utility of the T2 dark spot sign. Radiology April 2014; Vol 271: Number 1:126-132. ↩
- Foti PV, Attina G, Spadola S, Caltabiano R, Farina R, Palmucci S, Zarbo G, Zarbo R, D’Arrigo M, Milone P, Ettorre GC. MR imaging of ovarian masses: classification and differential diagnosis. Insights Imaging Feb 2016; 7(1):21-41. ↩
- Wasnik, A, Elsayes, K. Ovarian cystadenofibroma: A masquerader of malignancy. Indian J Radiol Imaging Nov 2010; 20(4):297-299. ↩
- Bookwalter CA, VanBuren WM, Neisen MJ, Bjarnason H. Imaging appearance and nonsurgical management of pelvic venous congestion syndrome. Radiographics Mar 2019; Vol. 39, No. 2:596-608. ↩
- Hosseinzadeh K, Heller MT, Houshmand G. Imaging of the female perineum in adults. Radiographics Jun 2012; 32:E129-E168. ↩
- Siegelman ES, Outwater EK, Banner MP, Ramchandani P, Anderson TL, Schnall MD. High-resolution MR imaging of the vagina. RadioGraphics 1997; 17(5):1183–1203. ↩