Clinical History:
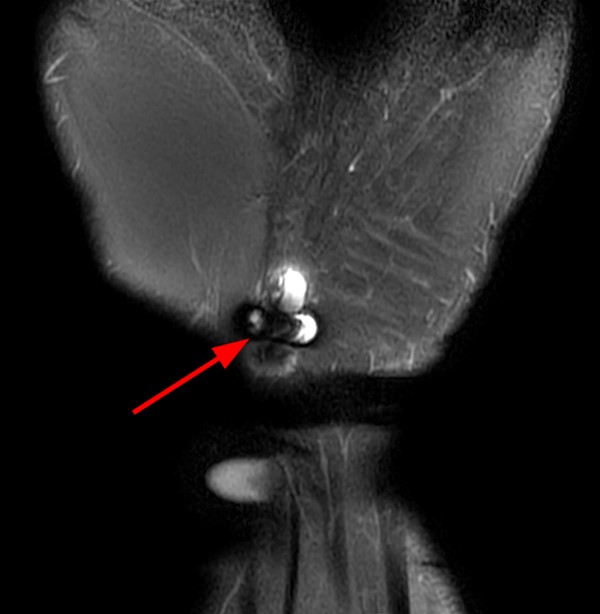
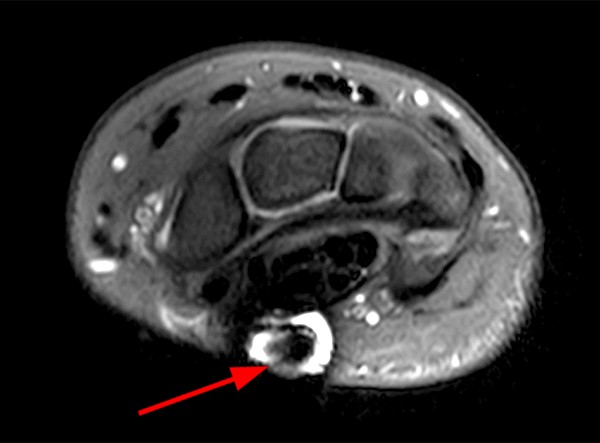
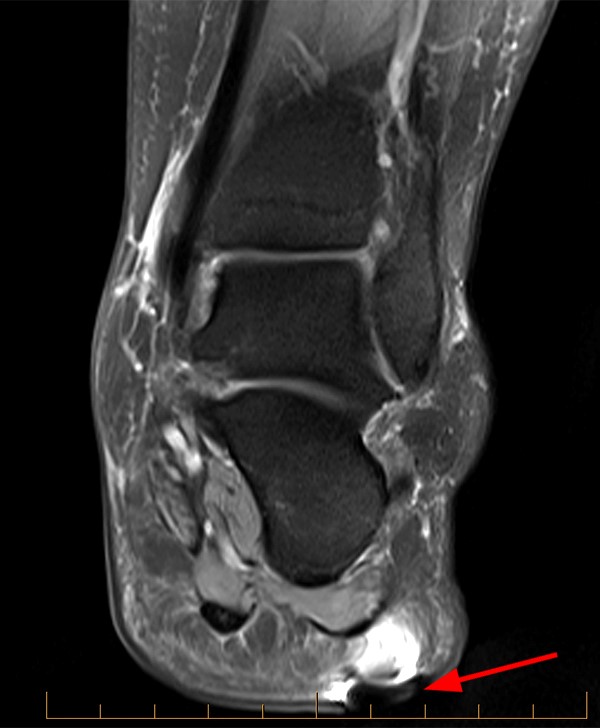
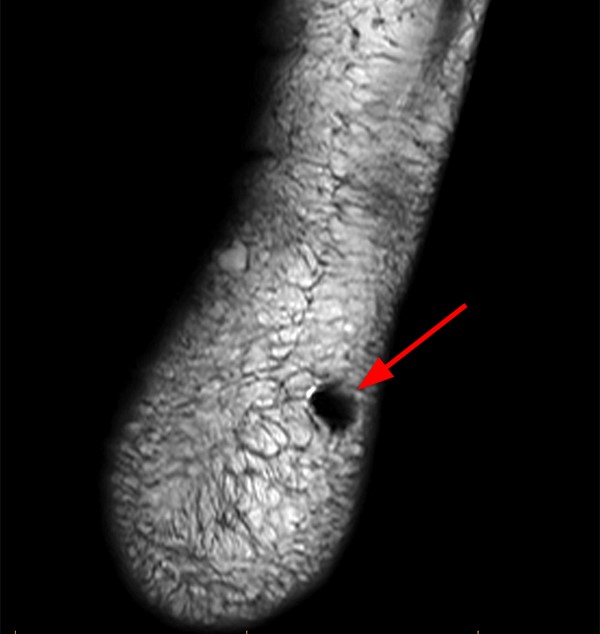
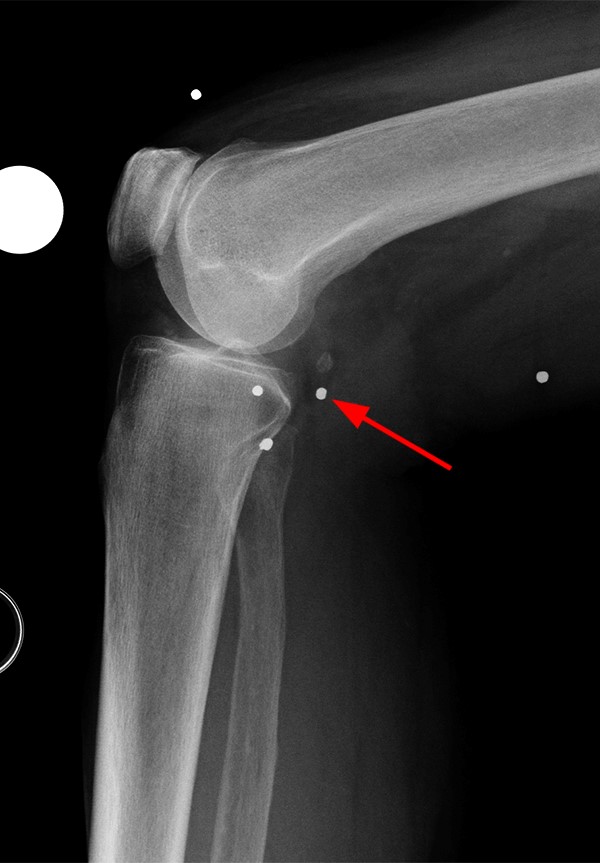
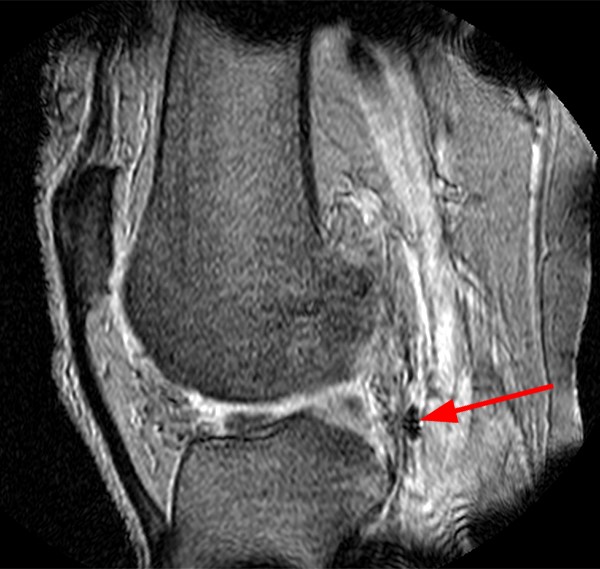
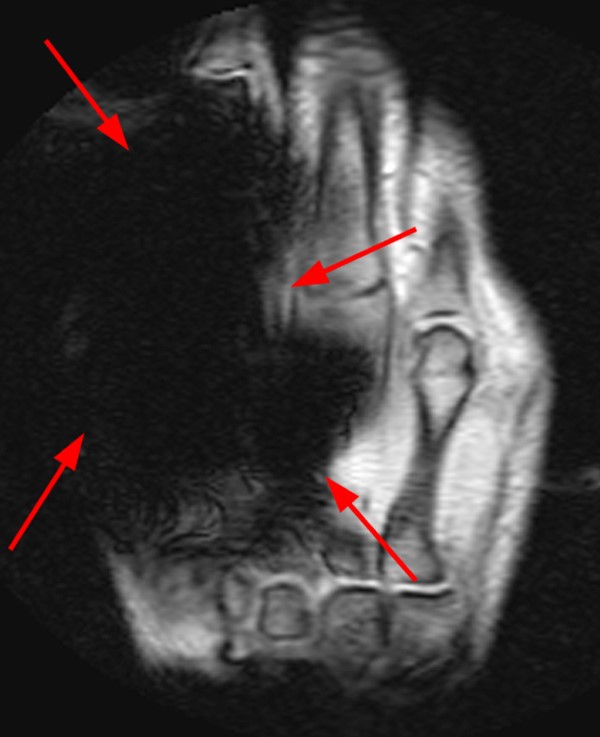
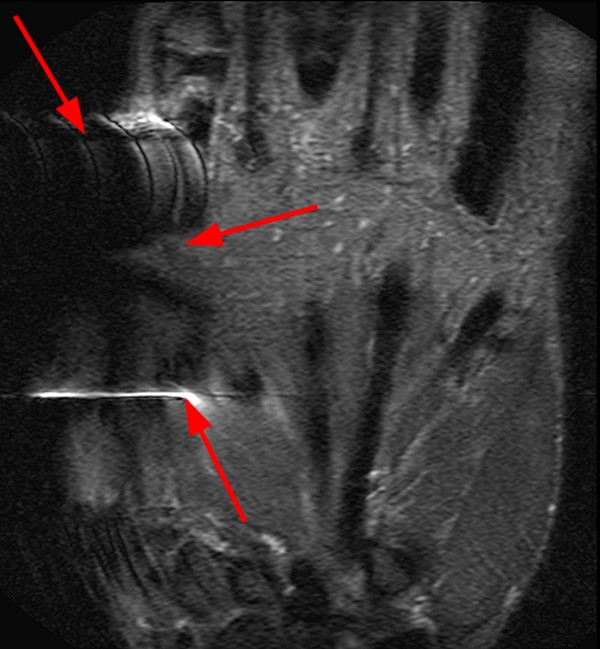
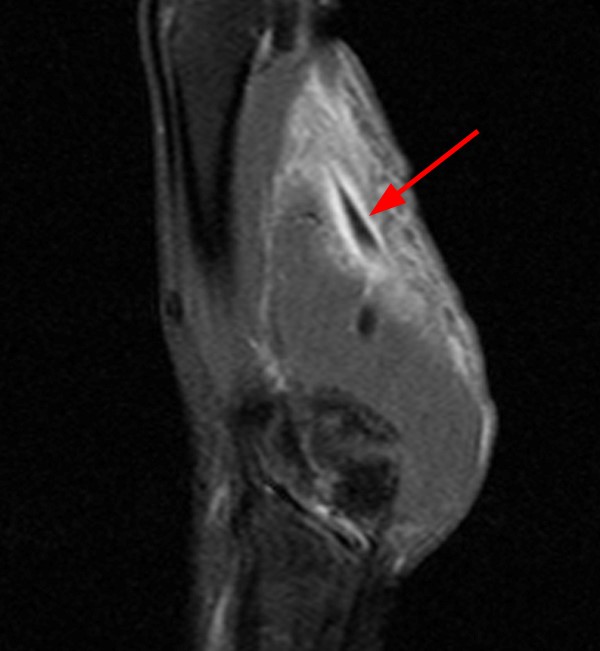
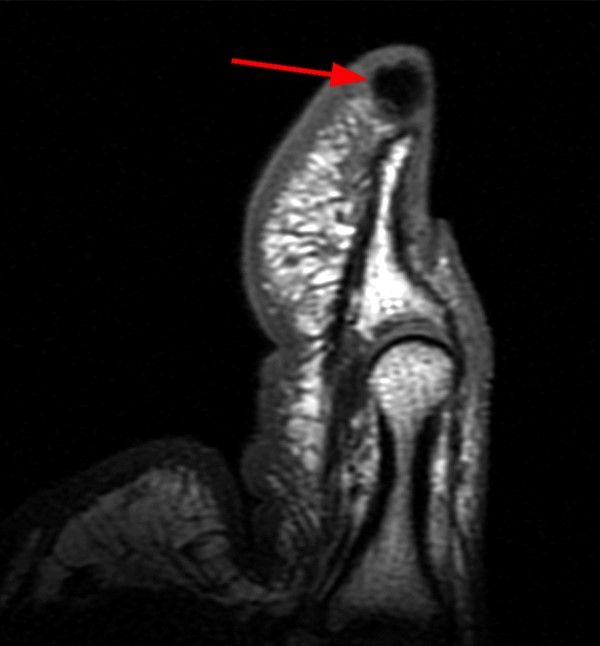
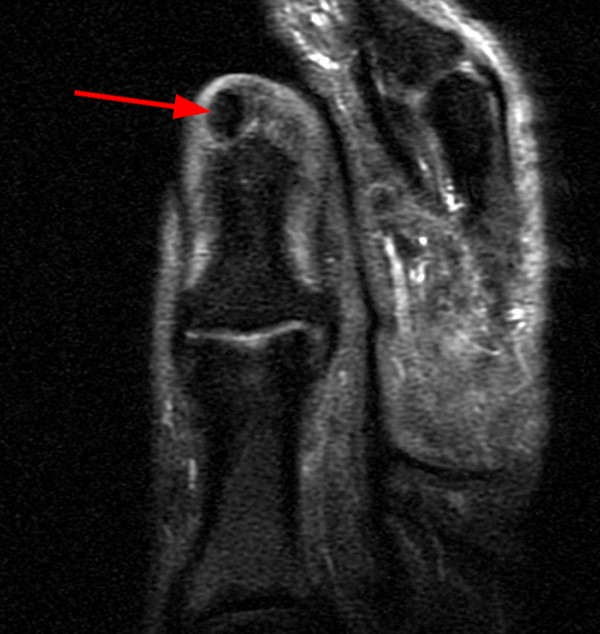
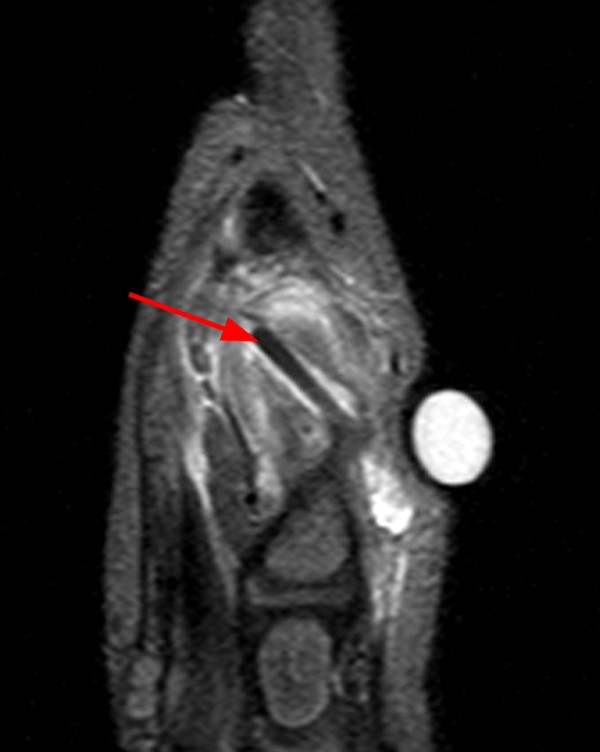
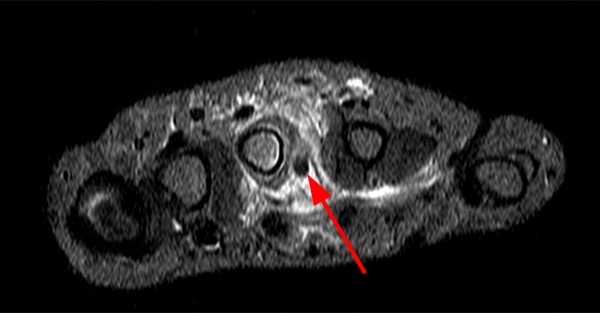
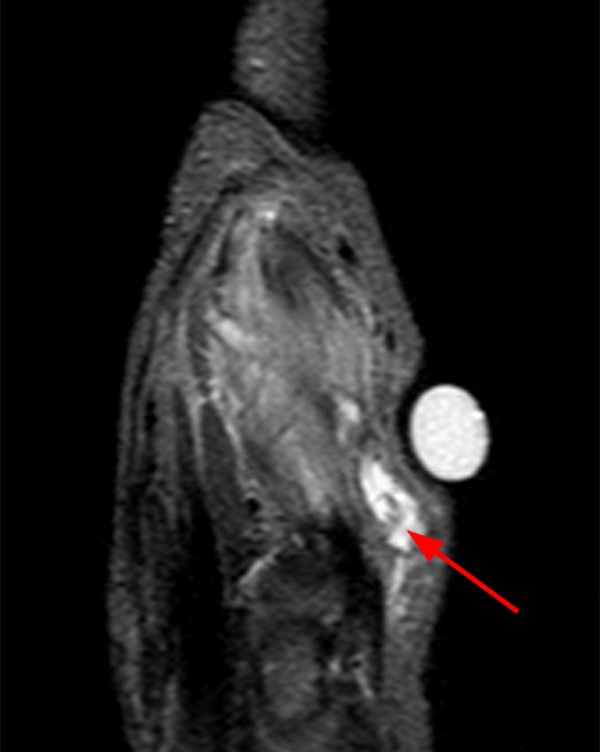
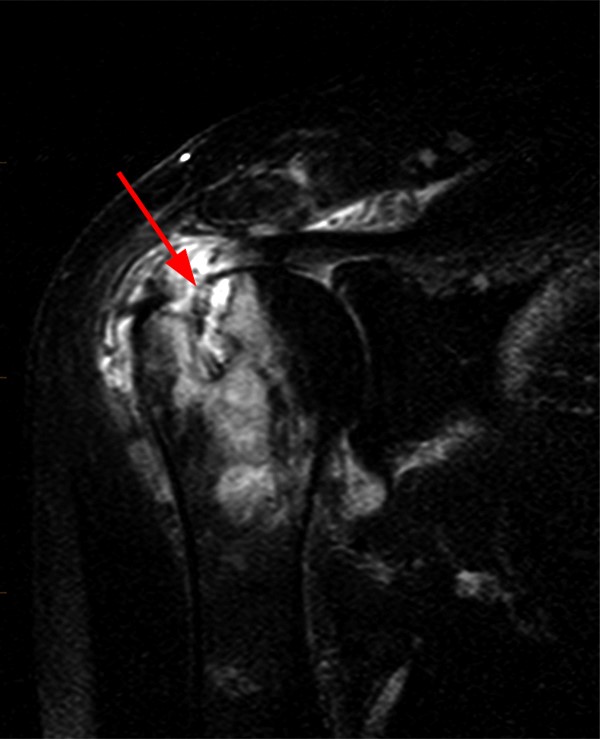
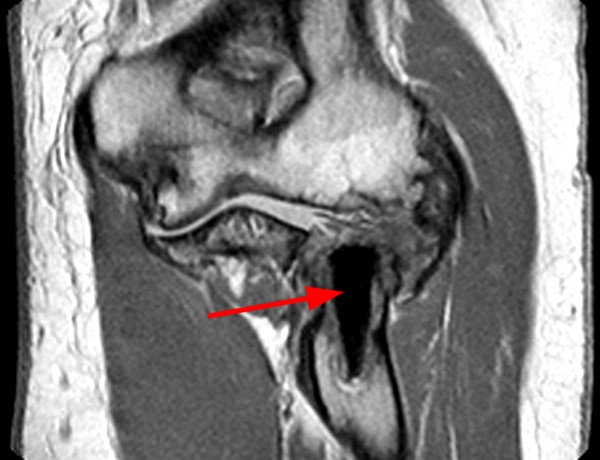
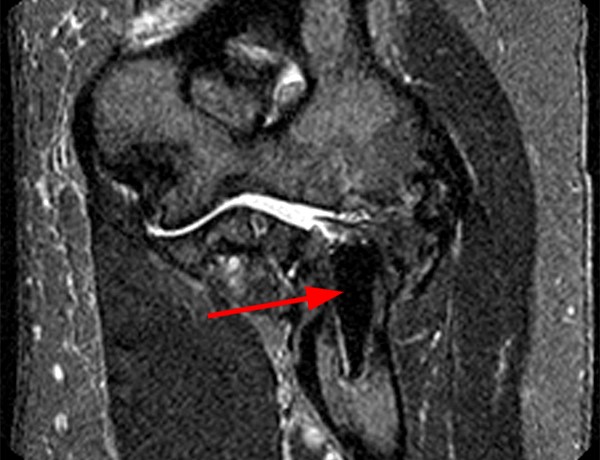
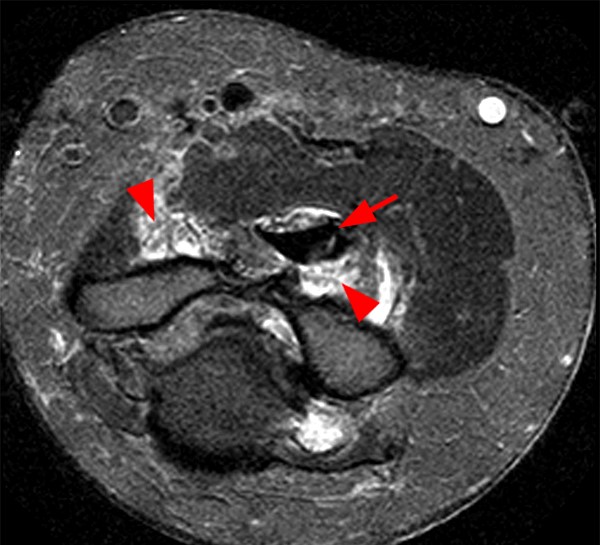
A 45-year-old female patient presents after hand trauma from a thorn bush. Fat-suppressed axial T2-weighted (1a), axial T1-weighted (1b), fat-suppressed sagittal T2-weighted (1c) and sagittal T1-weighted (1d) MR images are provided. What are the findings? What is your diagnosis?
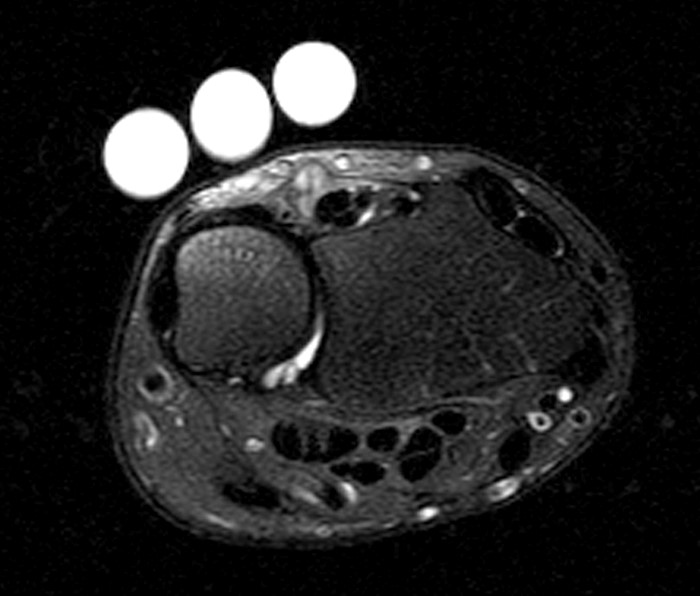
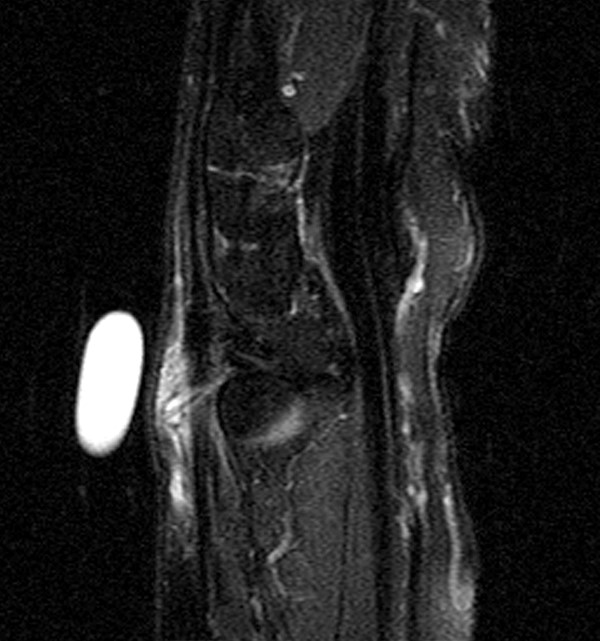
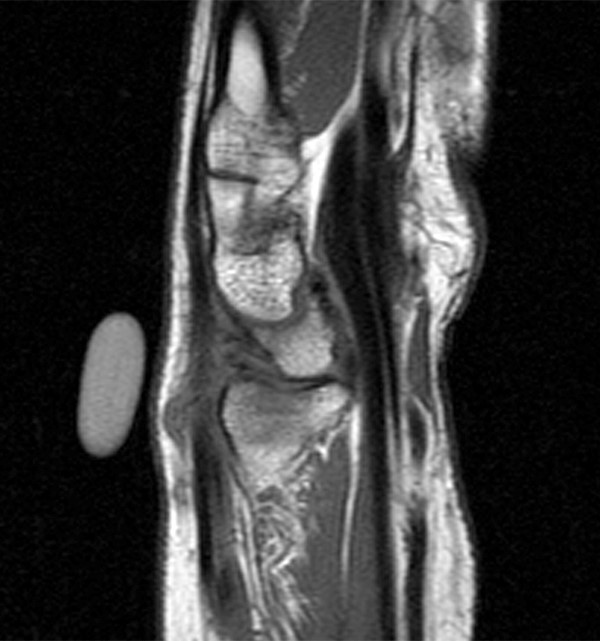
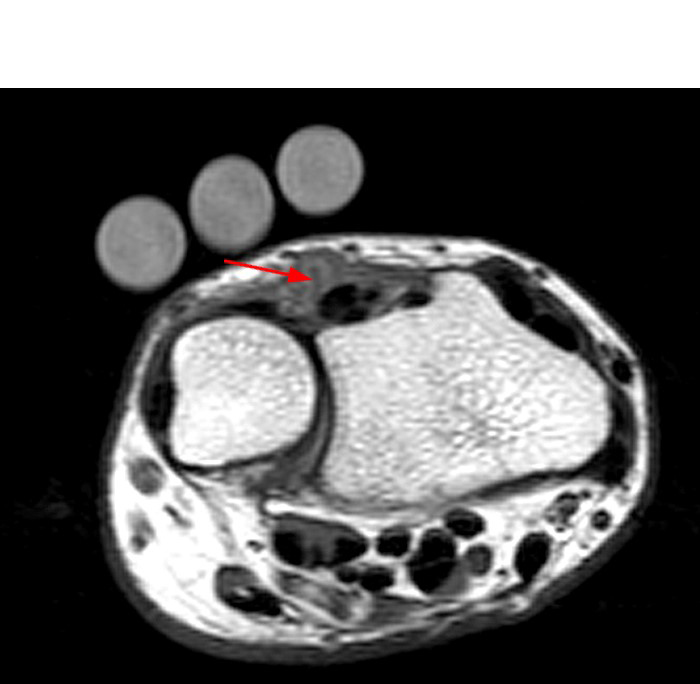
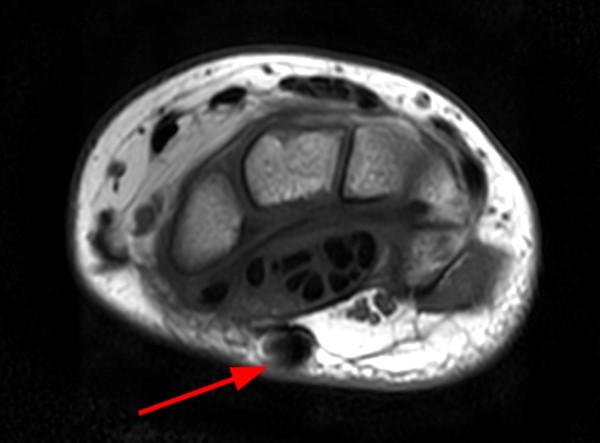
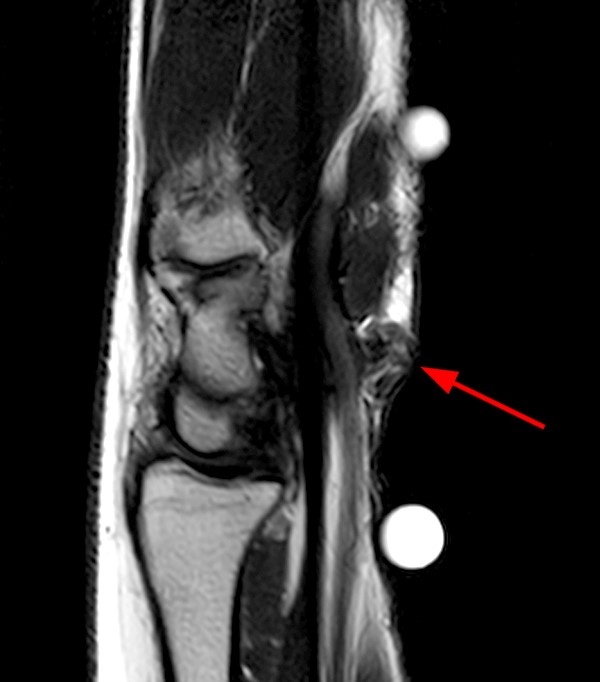
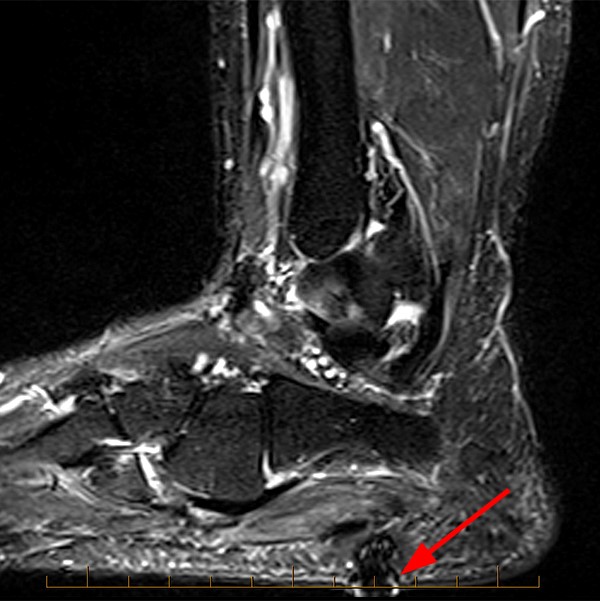
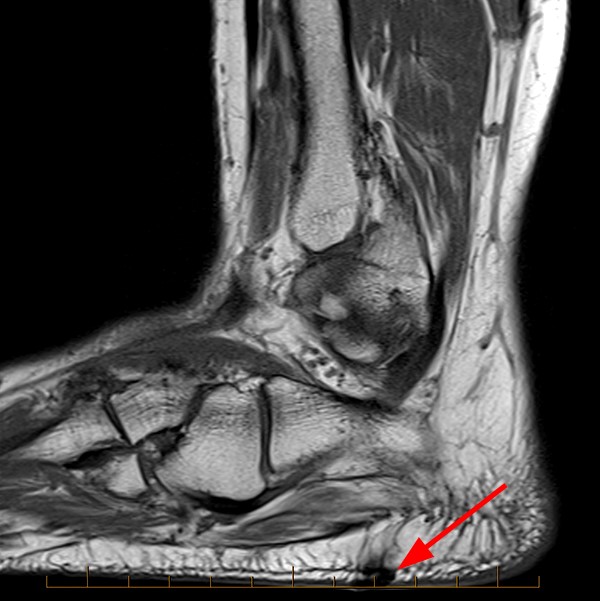
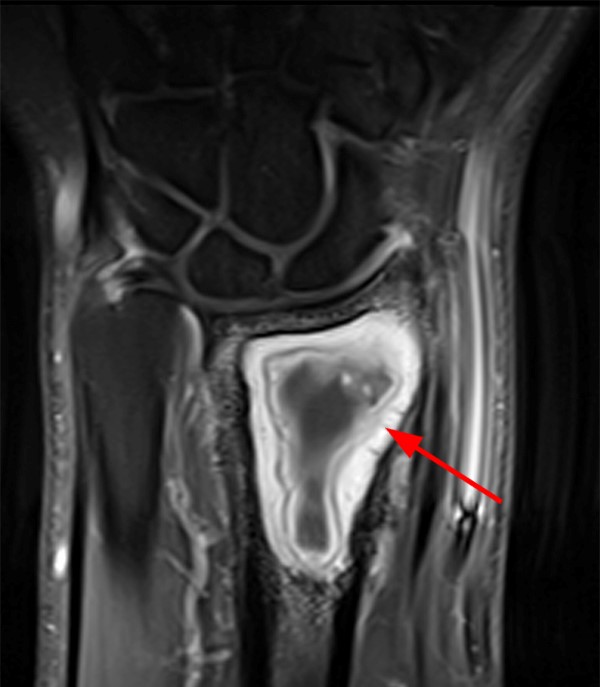
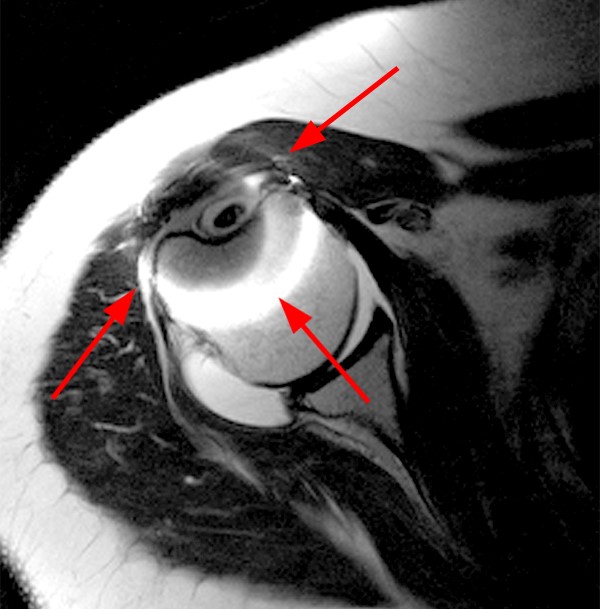
An oblique anteroposteriorly oriented low signal intensity linear structure (arrows), consistent with a thorn foreign body, is present within the soft tissues overlying the dorsal distal radius at the level of Lister’s tubercle. Surrounding edema is present, with hemorrhagic or proteinaceous debris, manifest by a halo of surrounding increased T1 signal (Figure 1b). The extensor tendons are intact and there is no osseous edema to suggest osteomyelitis. The morphology and signal characteristics of the structure, as well as the absence of overt susceptibility artifact, are most suggestive of an embedded thorn given the provided history. A hypointense, non-anatomic focus surrounded by hyperintense material on T2-weighted sequences has been described as the “target sign”, often seen with retained foreign bodies, as in this case (Figures 1a, 1c).1
Diagnosis:
Retained foreign body.
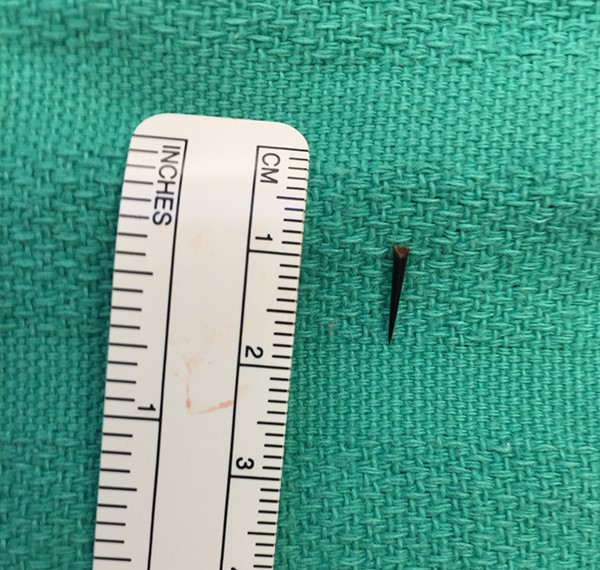
The thorn fragment was subsequently removed. Photo courtesy of Jennifer Chu, MD (orthopaedic Associates of Dallas).
Introduction:
Embedded foreign bodies are commonly encountered on imaging studies, particularly in the emergency department. The manner in which these inanimate objects find their way into the soft tissues can vary from trauma to self-embedding behavior.2,3,4 Though the incident that resulted in the retained foreign body is often known, it is not uncommon for the initial event to have been painless or the patient unaware at the time of trauma that a foreign body had broken the skin surface and become retained.3 With this in mind, close attention must be paid on all imaging studies to recognize non-anatomic structures, as well as acknowledge that an infectious of inflammatory processes could be secondary to an unsuspected foreign body. Missing such a treatable process could result in significant consequences for the patient as well as the radiologist.5,6
Confusion persists among ordering providers and radiologists which imaging modality is most appropriate for initial evaluation when a foreign body is suspected.7 Another source of confusion is what types of foreign bodies can be seen on conventional radiography, and what modality should be used if the radiographic exam is negative. As a result, the initial workup may be incomplete, increasing the risk for a missed diagnosis.5 Complications can include vascular, tendinous or ligamentous injury as well as pronounced granulomatous reactions, sinus tract formation or abscess. 1,6,8,9,10 Additionally, migration of the foreign body over time has been described in the literature, which can result in complications remote from the initial trauma.11
Foreign bodies can be loosely grouped into those that can be seen on conventional radiography and those that cannot. The basis for this visibility centers on the atomic number and density of the structure, and its ability to attenuate the x-ray beam. As a general rule, the greater the atomic number and density, the better it can be visualized.12 This would include objects such as graphite from a pencil, various metals, and stone, which are all denser than the surrounding soft tissues. A common misperception is that glass is radiolucent. Glass can be well visualized on conventional radiography, however its propensity to fragment into minute pieces can prove difficult to identify.7 Embedded wood and certain types of plastic are typically radiolucent and the only indication of their presence in the soft tissues may be its corresponding inflammatory response. 1,10,13
Radiographs represent the initial study of choice when a foreign body is suspected, but when the clinical suspicion is high and x-rays are negative, additional imaging modalities should be considered.2 This is especially true when the foreign body may reside within bone, as higher density structures may be missed in this setting.
If a radiolucent foreign body is suspected, ultrasound (US) may be useful for further evaluation.14 On ultrasound, foreign bodies typically appear hyperechoic to surrounding tissue, often with posterior shadowing which may described as “clean” or “dirty” dependent on the surface characteristics of the foreign body.2,15,16,17 Complications such as abscess formation, tendinous or vascular injury can also be identified by this modality. A potential downfall of US is the inability to identify a retained foreign body that is surrounded by air, as well as identification of foreign bodies embedded in bone. Computed tomography (CT) can also be utilized to diagnose retained foreign bodies that are radiolucent such as wood and plastic. The presence of air interdigitating the porous structure of wood requires a wide window level to adequately visualize.14 Though CT can detect most foreign bodies and provide better geographic detail of their exact location, it is less well suited to determine the degree of inflammation or associated damage to structures such as ligaments or tendons than MRI.2
The aim of this web clinic is to explore the imaging characteristics of foreign bodies on MRI. Without a guiding history, foreign bodies have the potential to be overlooked by the unsuspecting radiologist. Additionally, the sequelae of a chronically retained foreign body could be attributed to a more sinister process such as malignancy, resulting in unnecessary stress and treatment for the patient.6 We will discuss the physics behind the artifact induced by some foreign bodies, and how sequence acquisition can be optimized to reduce artifacts, or in some cases even increase artifacts to improve foreign body detection. Lastly, we will explore dedicated metallic-artifact reduction techniques when imaging for complications of prior surgery, such as hardware loosening, osteolysis or recurrent tendon tears.
Susceptibility Artifact
A well-known manifestation of embedded foreign bodies is the induction of susceptibility artifact. In many circumstances, this can limit study usefulness due to obscuration of regional soft tissues secondary to signal void and geometric distortion 18; however, it can also be used to our advantage to appreciate the presence of a foreign body that is too small to be seen on conventional imaging. In order to gain an understanding of the various imaging manifestations of susceptibility artifact, it is necessary to explore how it is created.
Susceptibility artifact results from magnetic field inhomogeneities, and magnetic susceptibility is defined as the degree a substance can affect relative magnetic field strength as a fraction of the magnetic field in which it is contained.19 Different tissues and substances within the human body induce local magnetic fields, which can either subtract from or add to relative magnetic field strength. By definition, paramagnetic substances contribute to magnetic field strength by aligning in the direction of the main magnetic field, while diamagnetic substance reduce magnetic field strength by aligning opposite to this field.20 We know that the precessional frequencies of protons, as described by the Larmor equation, are dependent on the strength of the magnetic field. A consequence of this relationship is variation in precessional frequencies of adjacent protons when variable relative magnetic fields are induced by different substances. Variation in precessional frequencies portends greater dephasing and signal loss, as well as mis-mapping of signal causing geometric distortion and signal pile-up.18 Additionally, significant frequency variations can hinder chemical fat suppression.21 Consequently, susceptibility artifacts induced by certain foreign bodies are increased when a higher field strength magnet is used, which results in even greater discrepancies in regional precessional frequencies.18 Without the use of refocusing radiofrequency (RF) pulses used in spin-echo sequences, this inhomogeneity can result in dephasing and accelerated signal decay. By understanding how a substance affects relative magnetic field strength, we can predict the degree of susceptibility artifact that it will induce.
Substances that are diamagnetic have paired electrons that can weaken the regional magnetic field. Examples of such substances are water, which has a diamagnetism of nearly zero, as well as normal tissues such as muscle and fat. Within the human body, calcium acts as the most pronounced naturally occurring diamagnetic substance. Conversely, paramagnetic materials have unpaired electrons that result in elevation of the regional magnetic field and acceleration of T1 and T2 relaxation times. An example of such a substance is the imaging contrast gadolinium, which in the appropriate concentration can shorten T1 relaxation time and produce high signal intensity useful for distinguishing pathology.22 An additional paramagnetic substance is air, which accounts for the limited usefulness of MRI when examining the face and sinuses. On the far end of the spectrum are ferromagnetic materials, such as iron or nickel, which have extreme positive magnetic susceptibility secondary to aggregation of unpaired electrons in a configuration that can result in profound signal loss.19
An important consequence of magnetic susceptibility is its effect on transverse relaxation time and resultant T2 signal. When measurement of T2 signal is influenced by both the natural T2 of the tissue in concert with the influence of magnetic field inhomogeneities, it is referred to as T2* (T2 star). If tissue heterogeneity results in relaxation times faster than the intrinsic T2, then the imaging appearance is more sensitive to the effects of magnetic susceptibility. To counteract this, spin-echo techniques with their 180° refocusing pulse were created to help negate the effects of magnetic field inhomogeneity. Conversely, gradient-echo sequences allow for dephasing of spinning protons secondary to T2* decay and therefore are more susceptible to artifact from field inhomogeneities such as metallic foreign bodies and surgical material.
Blooming artifact represents a manifestation of susceptibility artifact where an object or lesion appears larger than its true size. The more susceptibility artifact, the more pronounced the distortion. As one could infer, gradient-echo sequences are more sensitive to this artifact when strongly diamagnetic, paramagnetic, superparamagnetic, or ferromagnetic structures are present when compared to spin-echo sequences. Many sequences, such as susceptibility weighted imaging, take advantage of this artifact to help identify blood products or calcium. Despite the effectiveness of spin-echo sequences, strongly magnetic foci in soft tissues will still result in susceptibility artifact, especially when the TE is increased.
Case 2
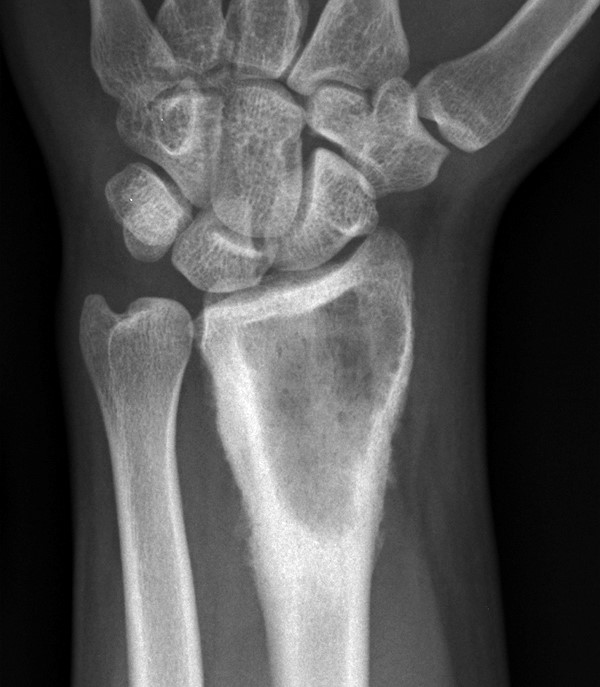
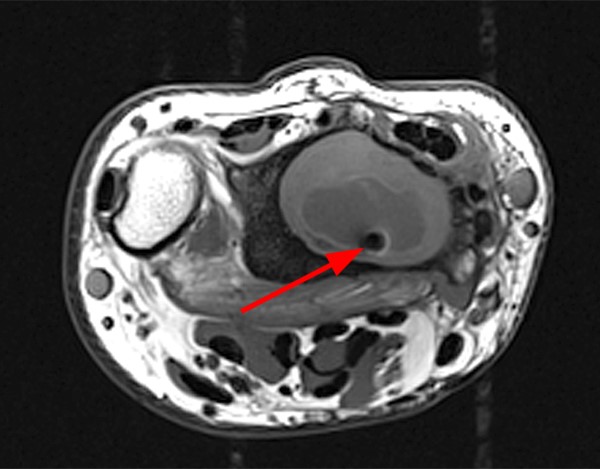
A 35-year-old female patient presents after trauma to the volar aspect of the wrist from a pencil. Presented below are: Frontal and Lateral wrist radiographs (2a, b), Coronal T1-weighted (2c), Coronal proton density-weighted with fat saturation (FS) (2d), Axial proton density-weighted FS (2e) Axial T1-weighted (2f) and Sagittal T2-weighted (2g) MRI images are provided.
Figure 2:
On the x-rays, no foreign body is seen. Further evaluation with MR reveals a region of signal void and slight blooming artifact (arrows) along the volar aspect of the wrist, superficial to the carpal tunnel. Edema surrounds the foreign body on T2-weighted sequences, which extends towards the intact transverse carpal ligament (2e).
As discussed above, susceptibility and blooming artifact are a result of magnetic field inhomogeneities which alter precessional frequencies of protons and can result in geometric distortion and regions of signal void. 23 Depending on the severity of the artifact, blooming has the potential to obscure close evaluation of adjacent soft tissues. We see in this case the degree of blooming artifact is more pronounced on the proton density-weighted sequences relative to T1-weighted sequences, with the size of the signal void closer to its actual size on the T1-weighted images. This is due to the longer TE required to obtain proton density-weighted images. It is suspected in this case that a component of the blooming artifact may be related to the presence of air (which is paramagnetic), because graphite is diamagnetic and can even be used as a shimming material to help correct magnetic field inhomogeneities.24
Case 3
This patient stepped on a piece of metal. Sagittal STIR (3a), Sagittal T1-weighted (3b), Coronal proton density-weighted FS (3c), and Axial T2-weighted (3d) MRI images are provided.
Figure 3:
The MRI reveals a region of susceptibility and blooming artifact (arrows) located within the plantar superficial soft tissues overlying the calcaneus. As expected, the degree of blooming is most pronounced on longer TE sequences (3a has a TE of 46 ms). It is important to note once again that air is a paramagnetic substance that can induce susceptibility artifact, so the presence of such an artifact in a region of recent trauma does not definitively represent a retained foreign body. This appearance can also be seen at a site of prior surgery due to air and metallic particles from tissue cauterization or use of a burr.25 Though difficult, the radiologist must attempt to “see through the artifact” and evaluate for complications such as tendinous injury.
Case 4
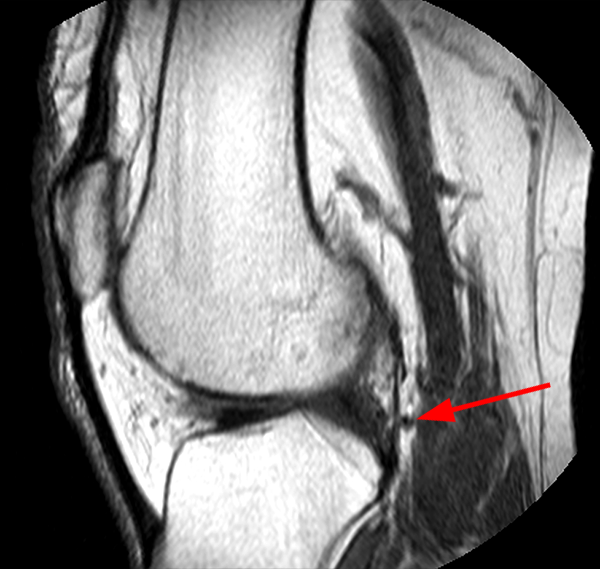
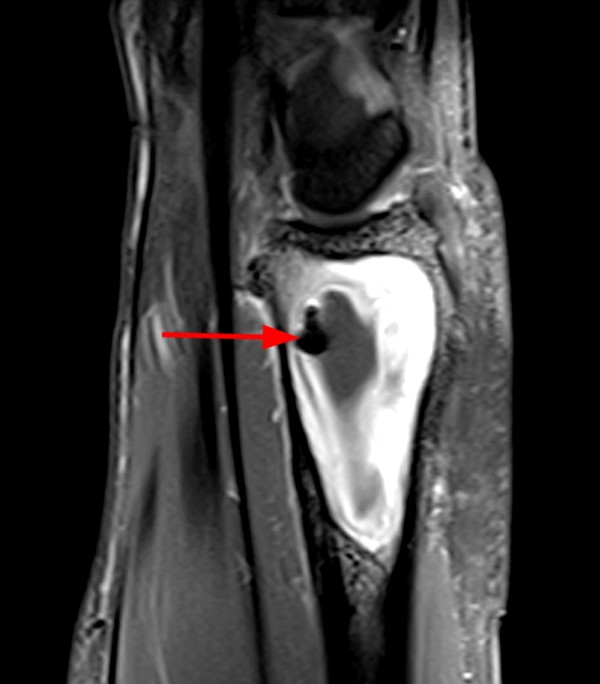
This patient presented after a shotgun injury to the knee, readily apparent on the lateral radiograph (4a).
Figure 4:
Gradient-echo imaging of the knee in the sagittal plane (4b) reveals a distorted focus of slight blooming artifact approaching, but not disrupting, the posterior oblique ligament. A more accurate representation of the true size of the foreign body is seen on the T1-weighted image (4c). Given the fact that shotgun pellets are classically composed of lead (a diamagnetic material), this accounts for the minimal amount of susceptibility artifact appreciated even on the gradient-echo sequence. A ferromagnetic structure would be expected to more significantly hinder regional soft tissue discrimination.
Case 5
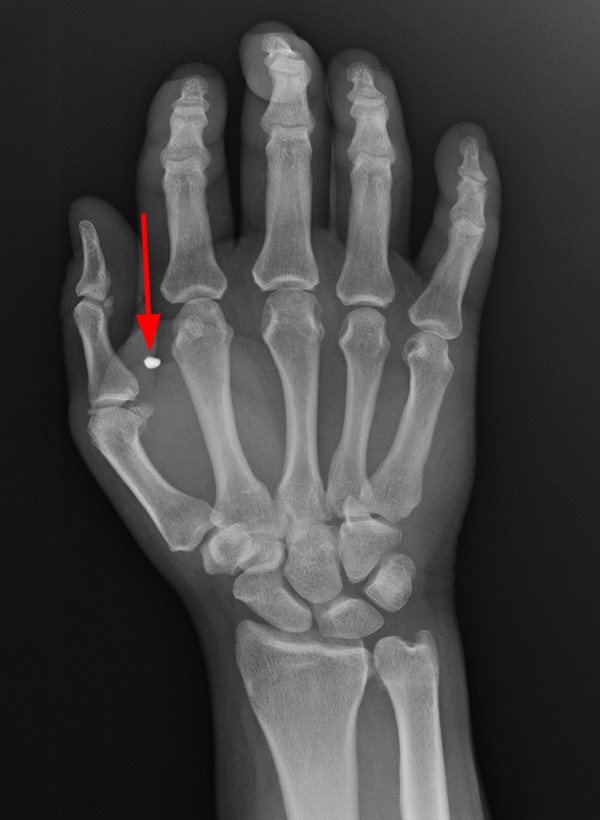
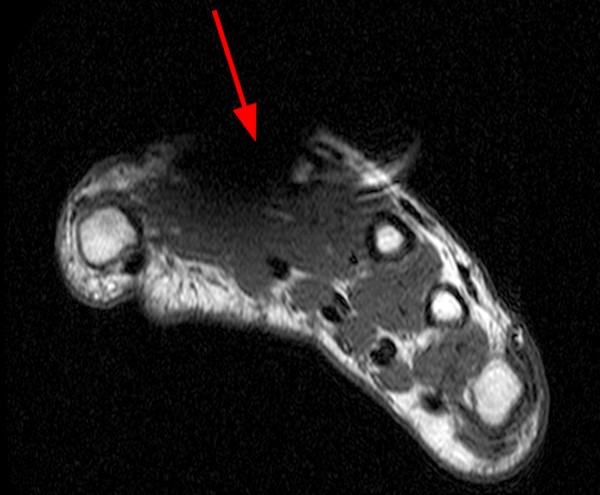
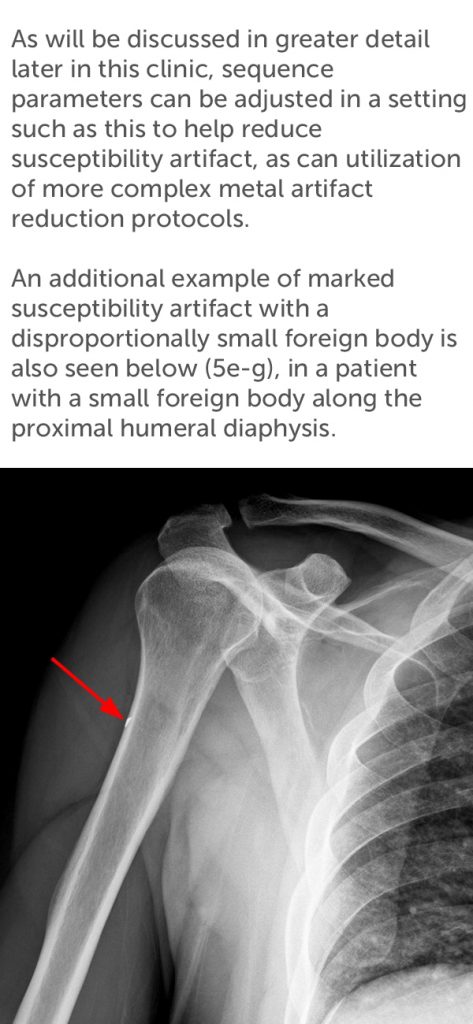
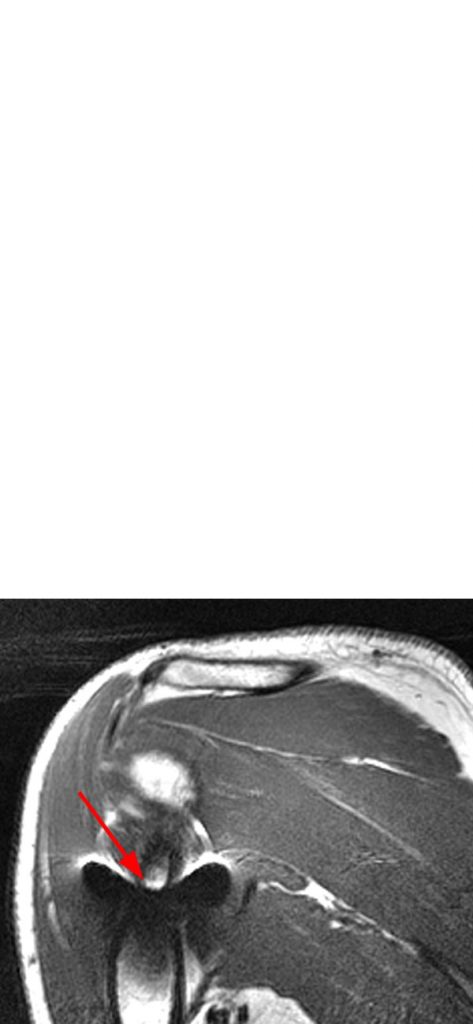
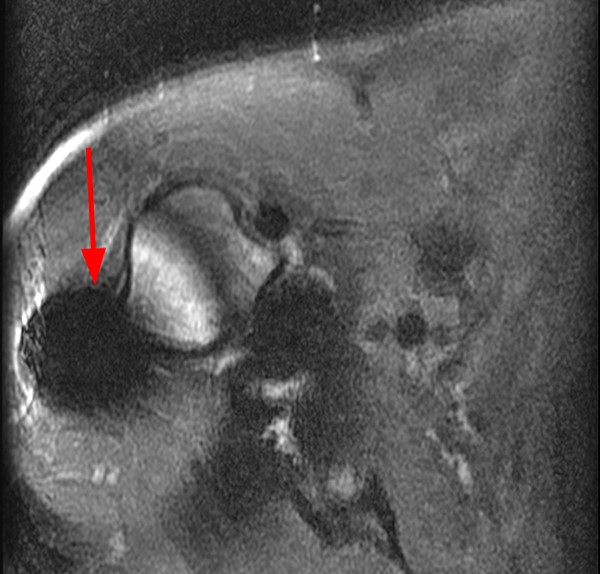
The 47-year-old male patient in case 5 presented after trauma to the hand with a metallic foreign body seen on conventional radiography (5a).
Figure 5:
MR imaging was obtained which was degraded by severe susceptibility artifact and geometric distortion despite the relatively small size of the embedded foreign body. The degree of artifact is more pronounced on the gradient echo localizer (5b) and STIR (5c) images. The T1-weighted image (5d) is more useful to show a relative decrease in susceptibility artifact and blooming.
Complications of Retained Foreign Bodies
Identification of retained foreign bodies on MRI can prove exceedingly difficult when the object does not result in significant magnetic field inhomogeneity and resultant susceptibility artifact. Examples of such objects are glass, wood and plastic. When these substances are suspected to be retained within the body, it is imperative to look closely for structures that exhibit a non-anatomic appearance and orientation. Such objects may exhibit straight edges or produce right angles. Foreign bodies are typically seen on MRI as hypointense on both T1 and T2-weighted sequences or as a region of signal void mimicking tendinous or ligamentous structures.2 It is imperative to evaluate for symmetry and to look closely for these signal characteristics in superficial soft tissues where structures such as ligaments, tendons or prominent vascular structures would not typically be seen.
In many instances, the ordering physician, as well as the patient, may be unaware that a foreign body is present. The clinical findings that may lead to obtaining an MRI include a soft tissue mass, draining wound or sinus, or simply regional erythema. An unsuspecting radiologist could then overlook a foreign body and suggest a more concerning process such as a neoplasm. A suggestion such as this could result in unnecessary stress and surgical/medical intervention for the patient.5,6 Being aware of the complications of acute or chronic foreign bodies, such as foreign body granuloma, abscess, draining sinus tract, osteomyelitis or solid/cystic mass should help the radiologist to consider the correct diagnosis.
Case 6
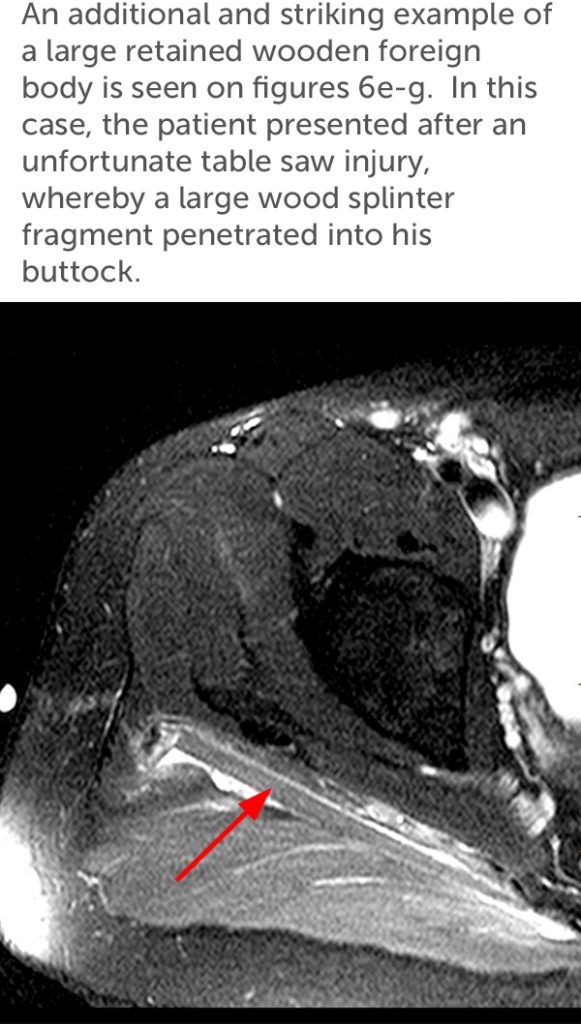
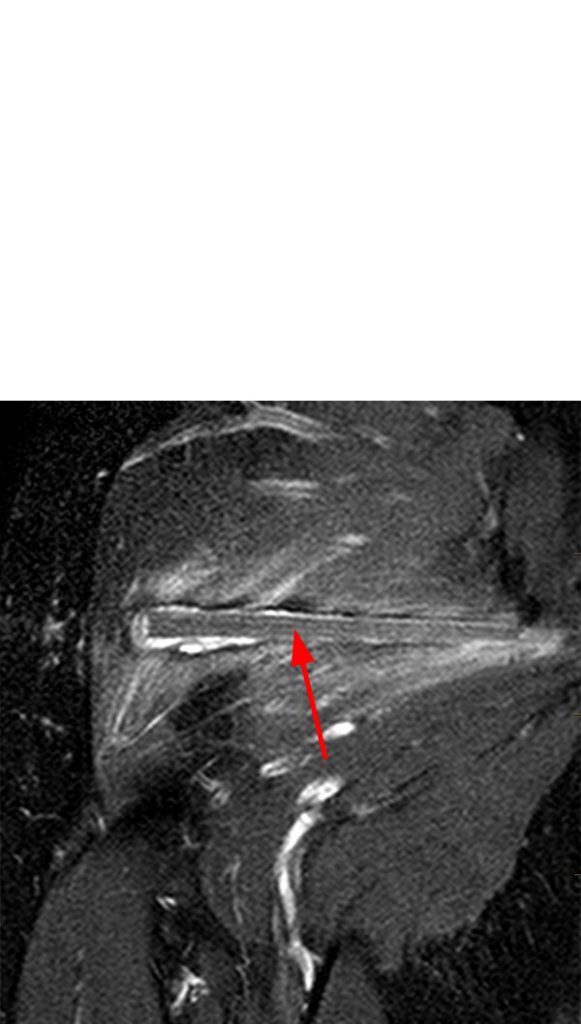
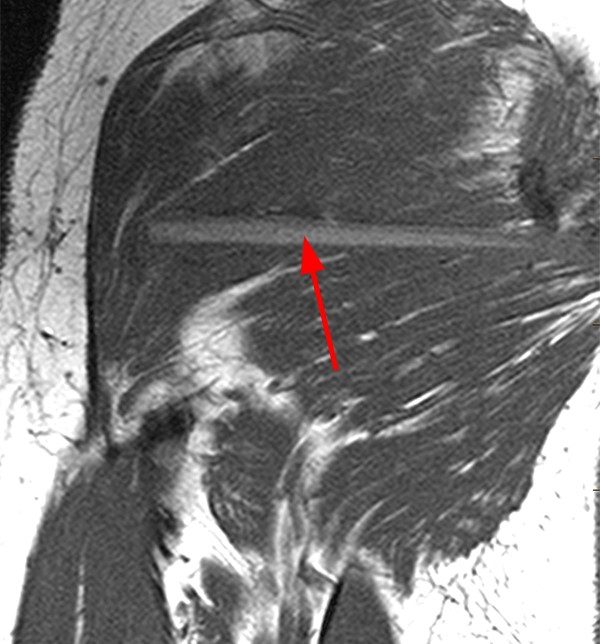
The patient below presented after trauma to the hand while handling wood, with clinical suspicion for a retained splinter. MR imaging of the hand was performed before and after intravenous gadolinium.
Figure 6:
Clearly identified is a large, triangular-shaped, T1 and T2 hypointense splinter embedded in the palmar aspect of the hand, extending from the first webspace into the flexor digitorum brevis and adductor pollicis muscles. Increased T2 signal (6a) and contrast enhancement (6b) surround the splinter compatible with granulation tissue. A pocket of increased T2 signal (6c) and rim enhancement (6d) within the adjacent web space is consistent with an adjacent abscess. On the axial image, the foreign body could be confused with a tendon, given its low signal and similar course as the adjacent flexor tendons. The secondary findings and multiplanar benefits of MRI mitigate any confusion.
The T2 weighted images (6e, 6f) reveal an intermediate signal intensity linear structure embedded in the deep aspect of the gluteus maximus muscle with surrounding edema. The T1-weighted image (6g) shows the foreign body to be hyperintense to muscle. Of note, because of the porous nature of wood, the classic T1 and T2 hypointense signal commonly seen with foreign bodies may be absent due to interdigitation of hemorrhage and fluid. This is more common with larger wooden fragments.
Case 7
The patient below presented with pain and swelling along the dorsal-ulnar aspect of the hand.
Figure 7:
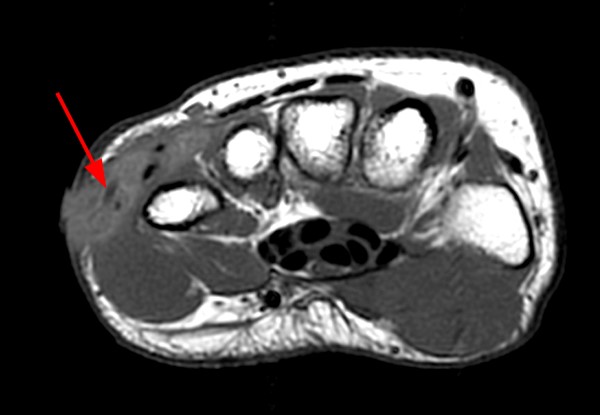
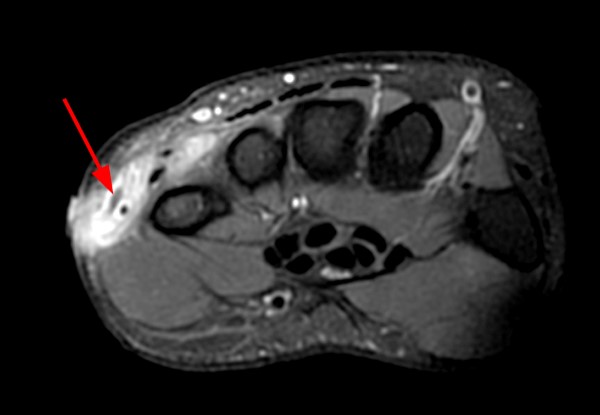
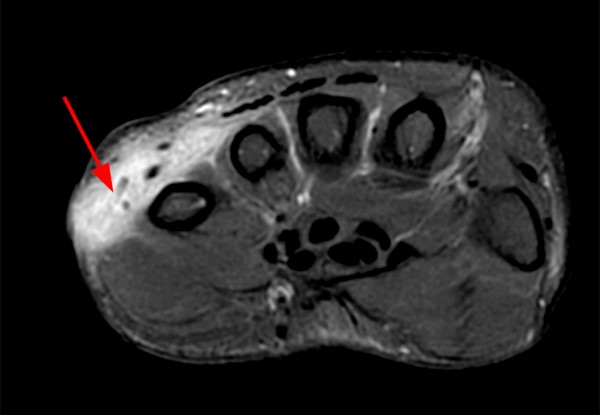
MR imaging reveals a T1 and T2 hypointense focus in the region of swelling concerning for a foreign body (arrows). No significant susceptibility artifact is identified. The T1-weighted image (7a) reveals surrounding hyperintense signal within the soft tissues compatible with hemorrhage. Increased T2 signal is also present (7b). The surrounding tissue also centrally enhances with IV contrast (7c), consistent with granulation tissue or developing foreign body granuloma. The osseous and adjacent tendinous structures appear normal.
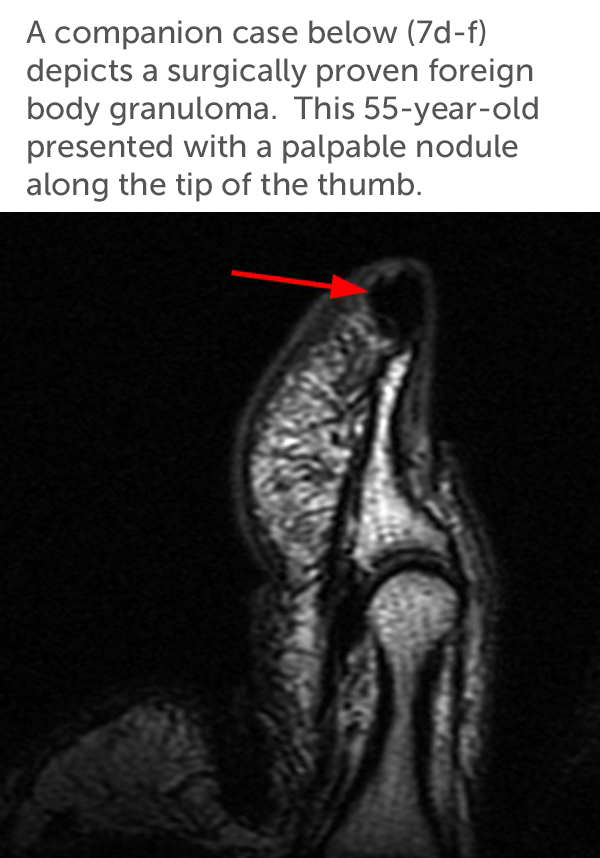
MR imaging reveals a T1 and T2 hypointense focus corresponding to the physical exam findings. On the T2-weighted image (7d), the structure exhibits slight blooming artifact, with the lesion smaller on the shorter TE/T1-weighted image (7e). No enhancement or regional edema is seen. After surgical excision and pathologic evaluation, a punctate foreign body was found surrounded by a calcified foreign body granuloma. The presence of blooming artifact is attributed to the significant diamagnetism exhibited by calcium.
Case 8
The 34-year-old male patient below presented after trauma to the hand with a wooden object.
Figure 8:
MR imaging repeals a T1 mildly low signal (8b) and T2 low signal intensity (8a), foreign body running obliquely within the second intermetacarpal interspace. Increased signal is present on both T1 and T2-weighted sequences surrounding the foreign body compatible with hemorrhage and edema. Surrounding inflammation and myositis are present. Increased T2 signal within the adjacent third metacarpal (8c), is concerning for superimposed osteomyelitis. An overlying abscess is noted in within the more superficial dorsal soft tissues adjacent to a surface marker (8d).
Case 9
Radiography of this 35-year-old male’s wrist revealed a circumscribed geographic lytic lesion within the distal radius, with surrounding sclerosis, cortical thickening and periostitis. These findings are most concerning for an infectious process with a neoplastic lesion such as a giant cell tumor considered less likely. Further characterization with MRI was performed.
Figure 9:
The lytic area contains increased T1 signal (9b), increased T2 signal (9c), and central non-enhancement on post-contrast imaging (9d), consistent with an intra-osseous hematoma and abscess, surrounded by granulation tissue. Within the abscess lies a small T1/T2 hypointense focus with mild susceptibility artifact, consistent with a foreign body (arrows). Surgical intervention revealed the foreign body to be graphite from a pencil, which accounts for its only mild susceptibility.
MR Imaging for Complications of Surgery
In a patient with a surgically-implanted foreign body, MRI becomes more challenging but remains highly useful in evaluating for complications 25,26,27 Despite its ability to provide excellent contrast resolution, MRI can be hindered by artifact secondary to metallic implants, screws, staples and metallic shavings. Depending on the metallic composition of the orthopaedic hardware, the degree of susceptibility artifact may render the MRI nondiagnostic.28 However, with optimization of sequence parameters and utilization of novel techniques designed specifically for metallic artifact reduction, MRI is now the modality of choice when previously its usefulness was limited.26
Case 10
The patient in case 10 initially presented for shoulder radiographs with clinical concern for complications of a rotator cuff repair.
Figure 10:
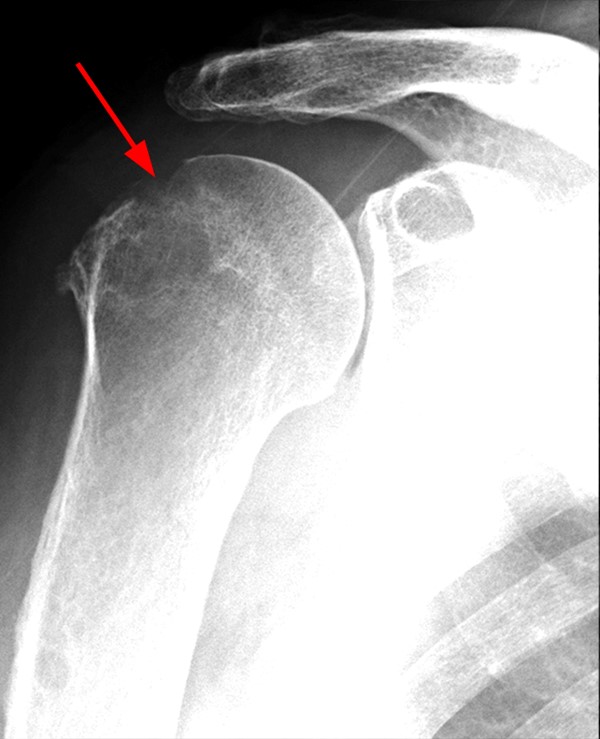
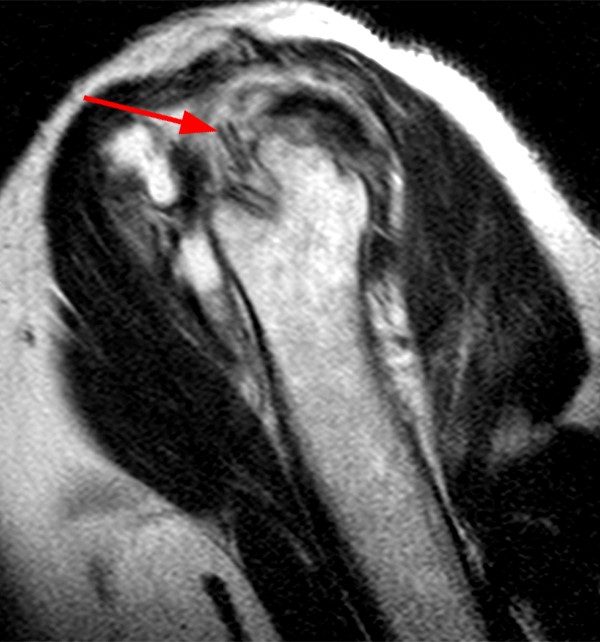
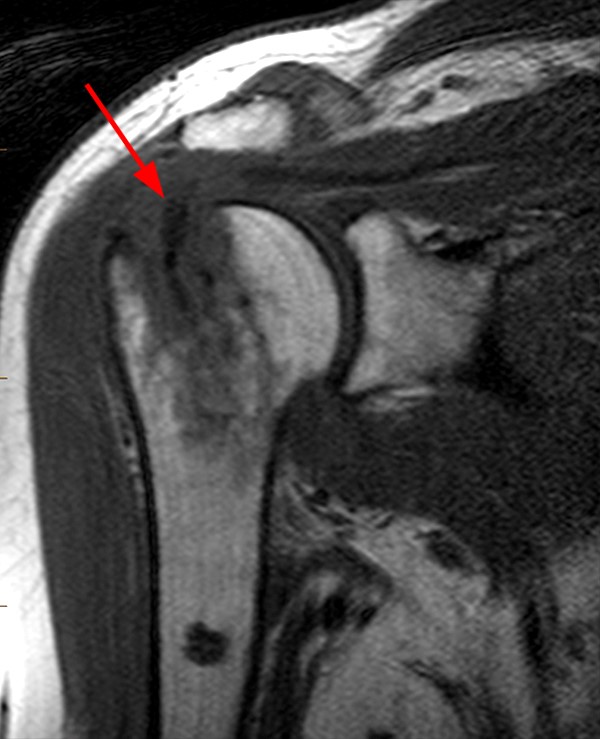
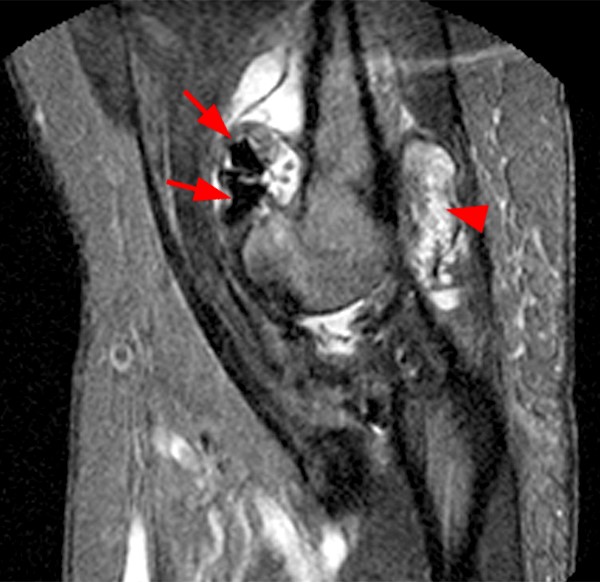
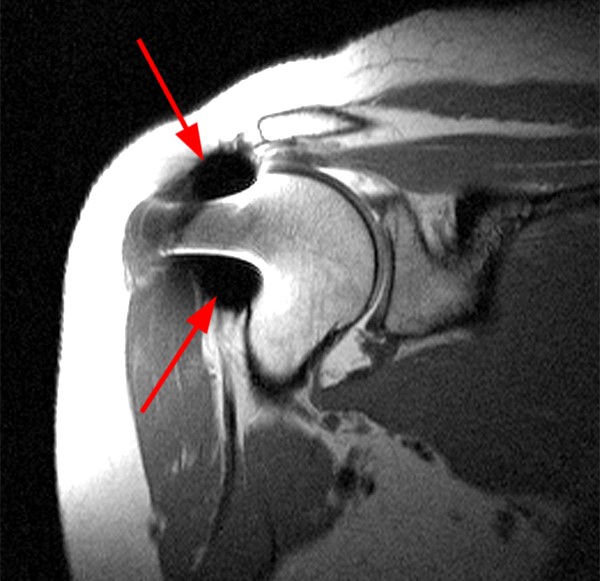
The AP shoulder radiograph (10a) reveals osteolysis with cortical destruction of the posterolateral humeral head at the greater tuberosity. The sagittal T2-weighted image (10b) reveals malposition of a suture anchor, dislodged from its original location. Coronal T1-weighted (10b) and T2-weighted fat-suppressed images (10c) reveal osteolysis surrounding the anchor, with marked surrounding osseous edema (concerning for osteomyelitis), and overlying subacromial/subdeltoid bursitis.
Case 11
The patient below had undergone a remote silicone implant of the radial head and presented with worsening pain.
MR Imaging Optimization with Known Metal
If a metallic foreign body is discovered to create problematic artifacts, techniques can be employed to decrease the artifact. To understand the metal optimization techniques, it is first helpful to further define the problematic artifacts:
In-plane distortions are a broad category of artifacts resulting from signal loss and pileup due to distortions of the local field homogeneity by the ferromagnetic foreign body. This manifests as a cloud of signal loss surrounding the body, due to T2* dephasing. This artifact is most obvious on gradient-echo sequences, which lack a refocusing pulse.29 Variable precessional frequencies induced by magnetic field inhomogeneity cause mis-registration of signal on the final image20, which accounts for the rim of increased signal surrounding the foreign body (signal pile-up).18 These artifacts tend to be less pronounced when the longest portion of the prosthesis/foreign body is aligned parallel to the B0 field.
Poor fat suppression also results from the local field variations from the ferromagnetic foreign body. These are most pronounced with chemical (spectral) fat-suppression techniques, which rely on homogeneous local fields to differentiate the relatively small differences in the chemical shifts between fat and water.
Geometric distortion is akin to chemical shift artifact, whereby the presence of both the foreign body (e.g. metallic alloy implant) and human tissue are averaged together within the same voxel, creating image distortion by spreading pixels over a wider range.
Through-plane (slice-to-slice) distortion are artifacts that extend across MR slice boundaries, and are dependent on the slices thickness and resolution utilized. Through-plane distortion occurs during slice-selective excitation due to non-linear signal mapping induced by magnetic field inhomogeneities.27
Fundamental MR image optimization techniques in a patient with metal are generically called MARS (Metal Artifact Reduction Sequences) techniques. Newer more advanced proprietary techniques such as multi-acquisition with variable-resonance image combination, (MAVRIC-GE) and slice encoding for metal artifact correction (SIEMAC -Siemens) are also available. MARS sequences are not vendor specific and are simply strategies for dealing with the artifacts:
Gradient-echo sequences should be avoided when metallic hardware is present.22 The use of fast-spin echo (FSE) is much more advantageous in decreasing in-plane distortion, due to its 180° refocusing pulse which limits the degree of intravoxel dephasing and resultant signal loss.29
Chemical fat-saturation should be avoided. If fluid-sensitive images are desired, STIR (short-tau inversion recovery) will result in more homogeneous fat-supression22.
Frequency-encoding direction should be adjusted relative to the orientation of the patient’s hardware.18,26 Local magnetic field inhomogeneities induced by hardware result in phase and frequency changes causing mis-registration of signal. Resultantly, hardware artifact is worse along the frequency-encoding direction. It is therefore beneficial to recognize the orientation of hardware and adjust the frequency and phase-encoding axis accordingly.18 This proves difficult in the setting of total shoulder arthroplasty due to the hemispheric configuration of the prosthesis.22
Wider receiver bandwidth should be utilized, as sampling a wider range of frequencies will decrease the geometric distortion.25,26,29 Additionally, a shorter echo time will allow for less dephasing and subsequent signal loss.29
Through-plane distortions can be improved by altering parameters which decrease voxel size. These can be accomplished by using a decreased of field of view (FOV), thinner slices, and higher matrix resolution.18,25,26
Decreased interecho spacing through the use of longer echo train lengths (ETL) has also been shown to decrease metal artifacts.18,22
Because many of the above techniques (increased bandwidth, smaller FOV, thinner slices, higher matrix) result in a drop in the signal-to-noise ratio (SNR), it is useful to increase the number of excitations/averages (NEX/AVG). Unfortunately, this also results in longer imaging times.
It’s also important to remember the degree of susceptibility artifact increases with MRI field strength; therefore metal artifacts are lower at 1.5T compared with 3T scanners.
Case 12
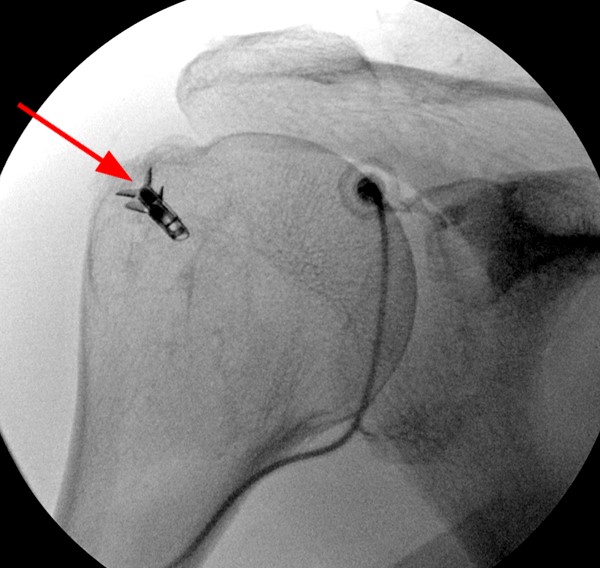
The patient below has undergone a rotator cuff repair, and a suture anchor is noted within the greater tuberosity during a fluoroscopic arthrogram injection image (12a).
Figure 12:
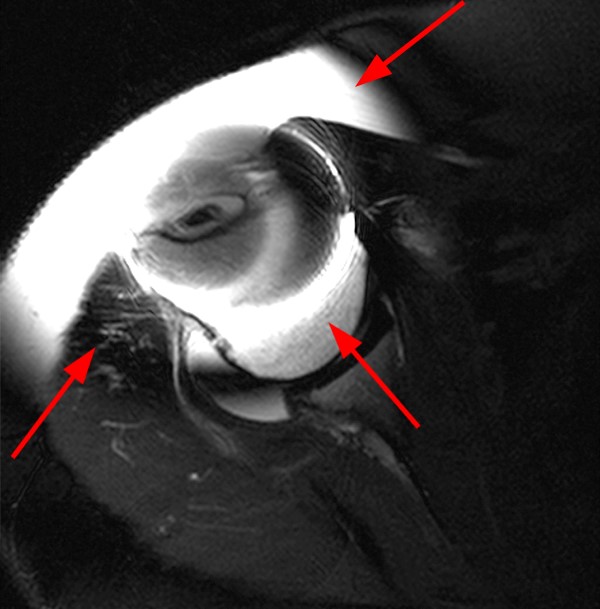
Initial T2-weighted MR images with chemical fat saturation and typical receiver bandwidth (12b, 12d) demonstrate extensive metal artifact from the suture anchor (in-plane distortion, poor fat-suppression, and geometric distortion), rendering the study almost non-diagnostic. Additional sequences were performed with MARS techniques (removed chemical fat-saturation, increased receiver bandwidth, higher resolution, decreased TE, increased ETL, increased NEX), which significantly decreased the metal artifact (12c, 12e).
It is important to recognize that magnetic field inhomogeneities induced by ferromagnetic implants often render chemical fat suppression techniques of little benefit. Chemical fat suppression takes advantage of the chemical shift between fat and water, where fat is known to resonate at a frequency approximately 220 Hz less that water (at 1.5 T). A narrow bandwidth RF pulse is administered close to that of fat followed by a spoiling gradient to null its signal. 30 When a metallic object is present, there is a shift in resonant frequency induced by local magnetic field inhomogeneity which hinders the ability to selectively excite fat. This effect, in addition to the wider bandwidth needed to reduce metallic artifact, renders chemical fat suppression nearly useless in the presence of a ferromagnetic object. When fat suppression is required, short inversion time inversion-recovery (STIR) sequences should be used. This technique does not rely on chemical shift, but instead relies on the shorter T1 relaxation time of fat relative to water.20,21 An inference one can make based on the mechanism of STIR fat suppression is its limited use when the examination is performed with contrast. The nulling RF pulse used to suppress fat signal would also null the signal induced by the T1 shortening effect of gadolinium.21
Summary
Recognition of embedded foreign bodies is possible utilizing various imaging modalities, with conventional radiography considered the initial examination of choice. MRI is typically reserved to characterize complications the foreign body might elicit. This includes, but is not limited to, abscesses, osteomyelitis, granulomas, adverse tissue reactions, as well as injury to vascular, tendinous or ligamentous structures. Being aware of these manifestations will help the observer look more closely for a foreign body, which can often be difficult to identify on MRI. An understanding of how certain foreign bodies can result in magnetic field inhomogeneities will allow for better identification and characterization of the embedded object. Additionally, a sound knowledge of the physics associated with foreign body imaging will permit the radiologist to tailor sequence acquisitions to reduce, or perhaps even enhance, associated artifact and aid in accurate diagnoses.
References
- Ando A, Hatori M, Hagiwara Y, Isefuku S, Itoi E. Imaging features of foreign body granuloma in the lower extremities mimicking a soft tissue neoplasm. Ups J Med Sci. 2009;114(1):46-51. doi:10.1080/03009730802602455. ↩
- Jarraya M, Hayashi D, De Villiers R V., et al. Multimodality imaging of foreign bodies of the musculoskeletal system. Am J Roentgenol. 2014;203(1):92-102. doi:10.2214/AJR.13.11743. ↩
- Anderson MA, Newmeyer WL, Kilgore ES. Diagnosis and treatment of retained foreign bodies in the hand. Am J Surg. 1982;144(1):63-67. doi:10.1016/0002-9610(82)90603-1. ↩
- Chaudry JL.Yousafzai AW. Self-embedding behavior (seb): a case report and revew of literature. Khyber Med Univ J. 2016;8(1). ↩
- Kaiser CW, Friedman S, Spurling KP, Slowick T, Kaiser H a. The retained surgical sponge. Ann Surg. 1996;224(1):79-84. ↩
- Pope L, Ward G. Report Masses Caused by Long-Standing Bodies in the Extremities : MR Imaging Findings Foreign. :395-397. ↩
- Madhan B, Arunprasad G, Krishnan B. Glass fragment embedded in the cheek: a missed radiographic finding. BMJ Case Rep. 2015:bcr2015210542. doi:10.1136/bcr-2015-210542. ↩
- Murakami AM, Chang A, Foo LF. Traumatic Lateral Plantar Artery Pseudoaneurysm and the Use of Time-Resolved MR Angiography. 2010:214-218. doi:10.1007/s11420-010-9170-3. ↩
- Hassan FOA. Retained toothpick causing pseudotumor of the first metatarsal: A case report and literature review. Foot Ankle Surg. 2008;14(1):32-35. doi:10.1016/j.fas.2007.07.002. ↩
- Karcnik TJ et al. Foreign body granuloma simulating solid neoplasm on MR. Clin Imaging. 21(4):269-272. ↩
- Landis BN, Giger R. An unusual foreign body migrating through time and tissues. Head Face Med. 2006;2:30. doi:10.1186/1746-160X-2-30. ↩
- Roberts W. Radiographic Characteristics of Glass. AMA Arch Ind Heal. 1958;18:470-472. ↩
- Rosenfeld R, Spigelblatt L, Chicoine R, Laverdiere M. Thorn*induced periostitis associated with Enterobacter agglomerans infection. 1978;119. ↩
- Jeffrey J.Peterson. Wooden foreign bodies. Ajr. 2002;178(March):557-562. ↩
- Rubin J, Adler R, Bude R, Fowlkes J, Carson P. Clean and dirty shadowing at US: a reappraisal. ↩
- Boyse TD, Fessell DP, Jacobson J a, Lin J, van Holsbeeck MT, Hayes CW. US of soft-tissue foreign bodies and associated complications with surgical correlation. Radiographics. 2001;21(5):1251-1256. doi:10.1148/radiographics.21.5.g01se271251. ↩
- Bray PW, Mahoney JL, Campbell JP. Sensitivity and specificity of ultrasound in the diagnosis of foreign bodies in the hand. J Hand Surg Am. 1995;20(4):661-666. doi:10.1016/S0363-5023(05)80287-4. ↩
- Lee M, Kim S, Song H, Huh Y, Han S, Suh J. Overcoming Artifacts from Metallic orthopaedic Implants at High- Field-Strength MR Imaging and Multi- detector CT. Radiographics. 2007;27:791-804. doi:10.1148/rg.273065087. ↩
- Mitchell DG. MRI Principles. 2nd ed.; 2004. ↩
- Morelli JN, Runge VM, Ai F, et al. An image-based approach to understanding the physics of MR artifacts. Radiographics. 2011;31(3):849-866. doi:10.1148/rg.313105115. ↩
- Hargreaves BA, Worters PW, Pauly KB, Pauly JM, Koch KM, Gold GE. Metal-induced artifacts in MRI. Am J Roentgenol. 2011;197(3):547-555. doi:10.2214/AJR.11.7364. ↩
- Talbot BS, Weinberg EP. MR Imaging with Metal-suppression Sequences for Evaluation of Total Joint Arthroplasty. Radiographics. 2015;36(1):209-225. doi:10.1148/rg.2016150075. ↩
- Czervionke LF, Daniels DL, Wehrli FW, et al. Magnetic susceptibility artifacts in gradient-recalled echo MR imaging. Am J Neuroradiol. 1988;9(6):1149-1155. ↩
- Leslie LK, Cohen JT, Newburger JW, et al. NIH Public Access. 2013;125(21):2621-2629. doi:10.1161/CIRCULATIONAHA.111.087940.The. ↩
- Thakkar RS, Thakkar SC, Srikumaran U, McFarland EG, Fayad LM. Complications of rotator cuff surgery-the role of post-operative imaging in patient care. Br J Radiol. 2014;87(1039):1-12. doi:10.1259/bjr.20130630. ↩
- Hayter CL, Koff MF, Shah P, Koch KM, Miller TT, Potter HG. MRI after arthroplasty: Comparison of MAVRIC and conventional fast spin-echo techniques. Am J Roentgenol. 2011;197(3):405-411. doi:10.2214/AJR.11.6659. ↩
- Lu W, Pauly KB, Gold GE, Pauly JM, Hargreaves BA. Slice encoding for metal artifact correction with noise reduction. Magn Reson Med. 2011;65(5):1352-1357. doi:10.1002/mrm.22796. ↩
- Beltran LS, Bencardino JT, Steinbach LS. Postoperative MRI of the shoulder. J Magn Reson Imaging. 2014;40(6):1280-1297. doi:10.1002/jmri.24570. ↩
- Mohana-Borges AVR, Chung CB, Resnick D. MR Imaging and MR Arthrography of the Postoperative Shoulder: Spectrum of Normal and Abnormal Findings1. RadioGraphics. 2004;24(1):69-85. doi:10.1148/rg.241035081. ↩
- Delfaut EM, Beltran J, Johnson G, Rousseau J, Marchandise X, Cotten A. Fat suppression in MR imaging: techniques and pitfalls. Radiographics. 1999;19(2):373-382. doi:10.1148/radiographics.19.2.g99mr03373. ↩