Clinical History:
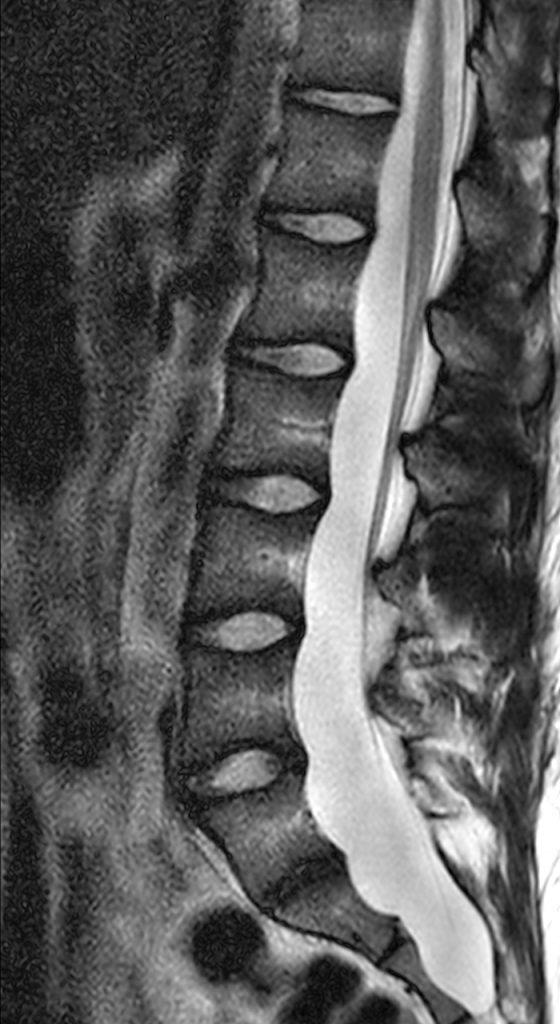
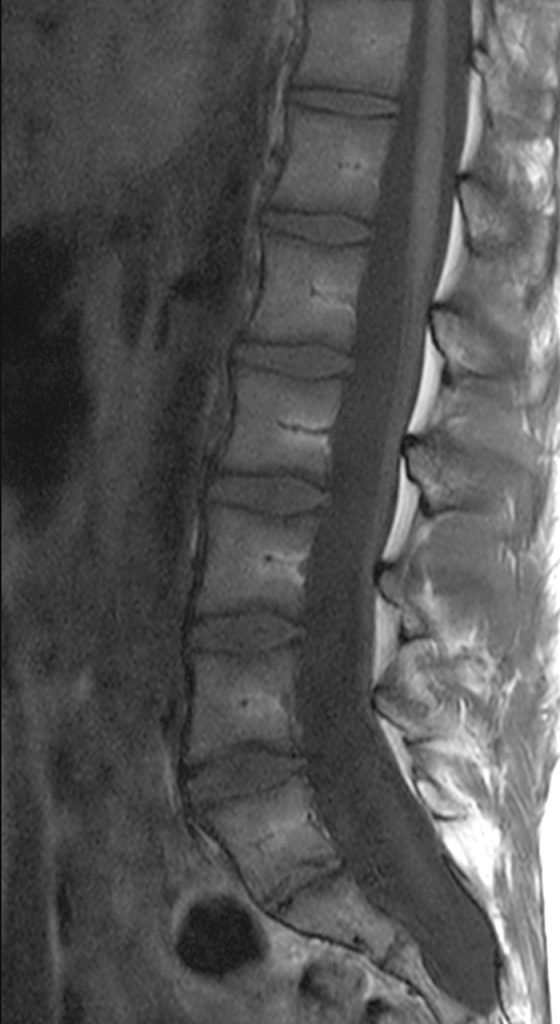
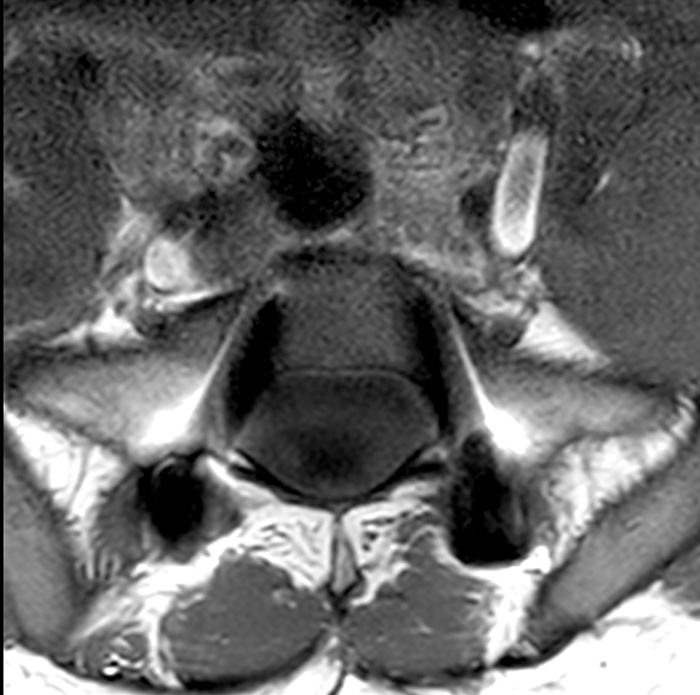
An 18 year-old male undergoes MRI for low back pain. Sagittal T2-weighted (1a), sagittal T1-weighted (1b), and axial T1-weighted (1c) and axial T2-weighted (1d) images at the level of S1 are provided. What are the findings? What is your diagnosis?
Findings
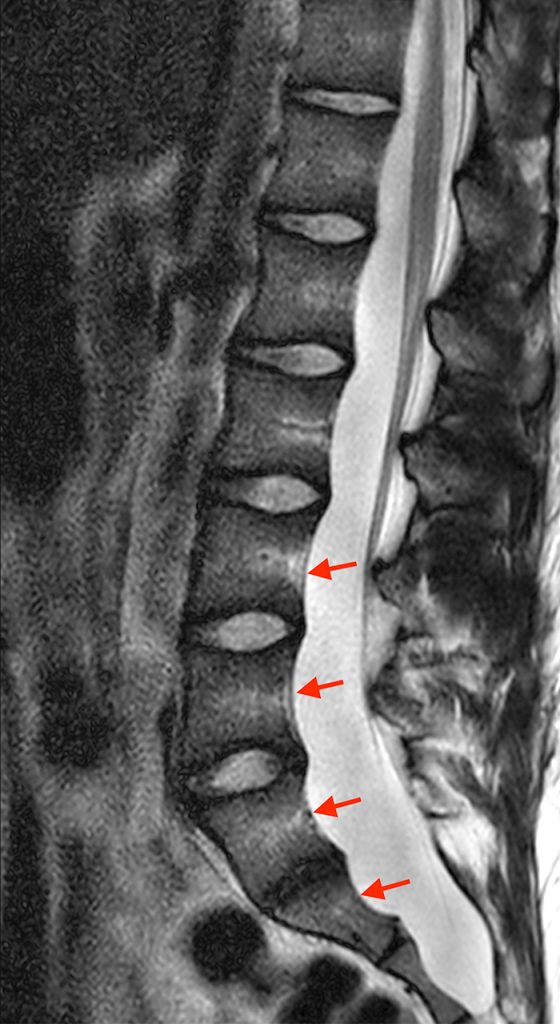
Figure 2:
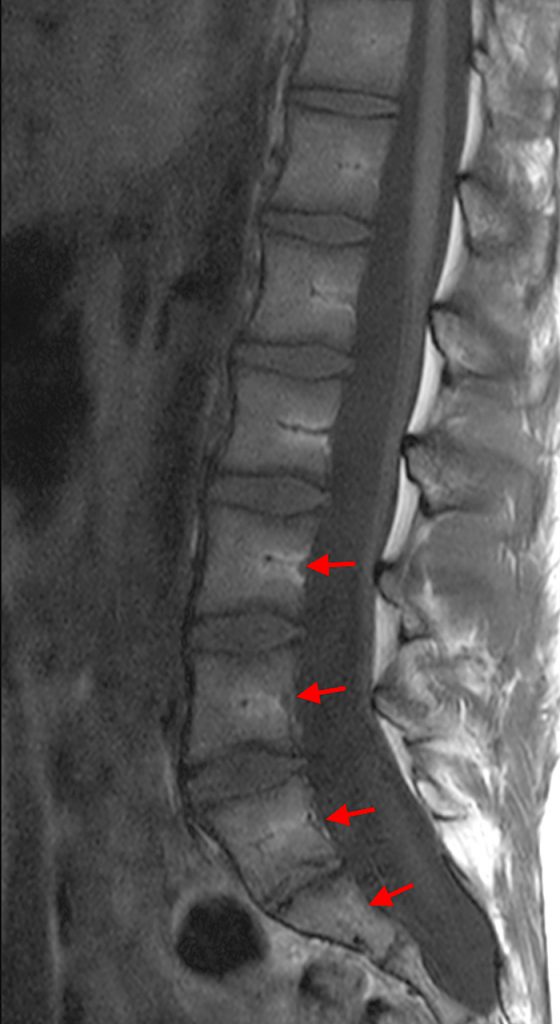
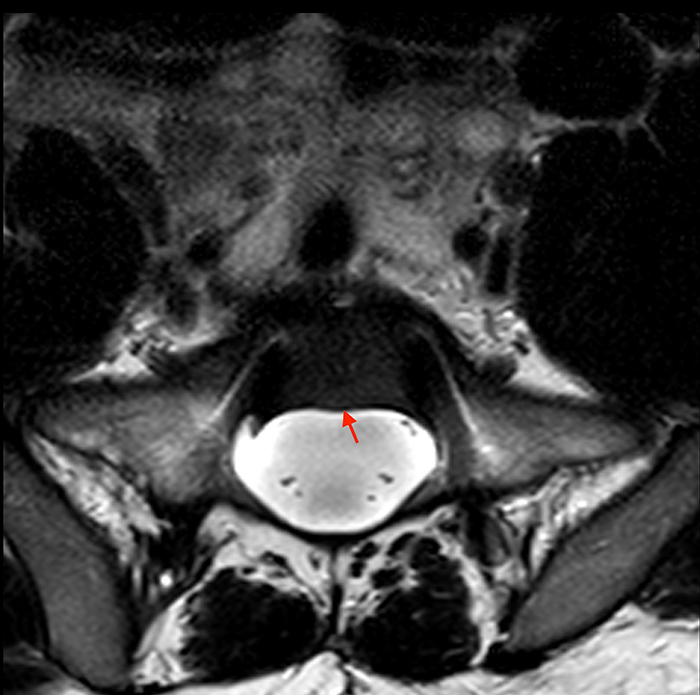
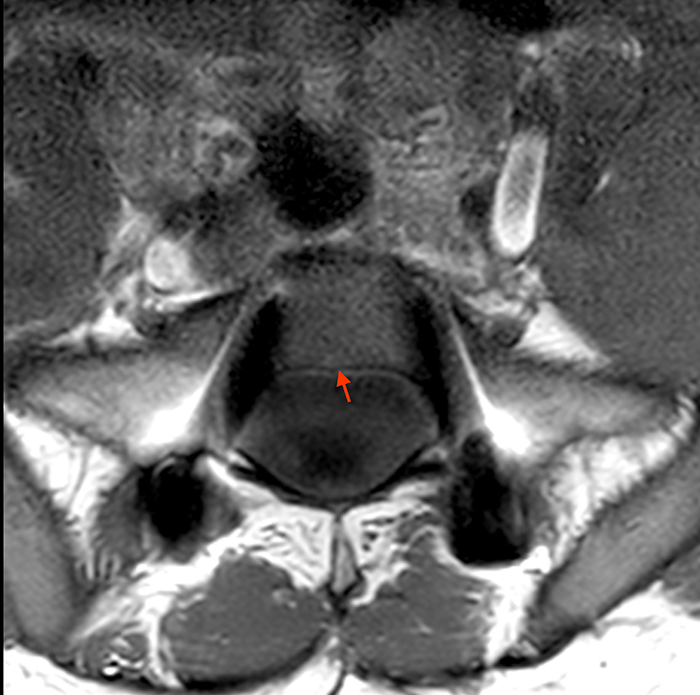
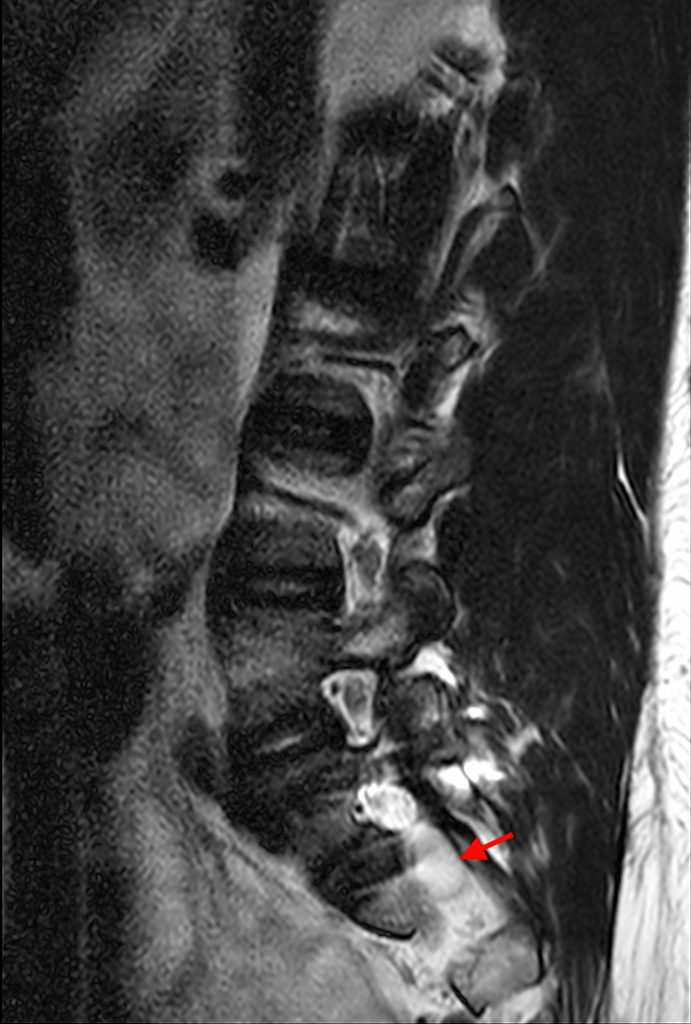
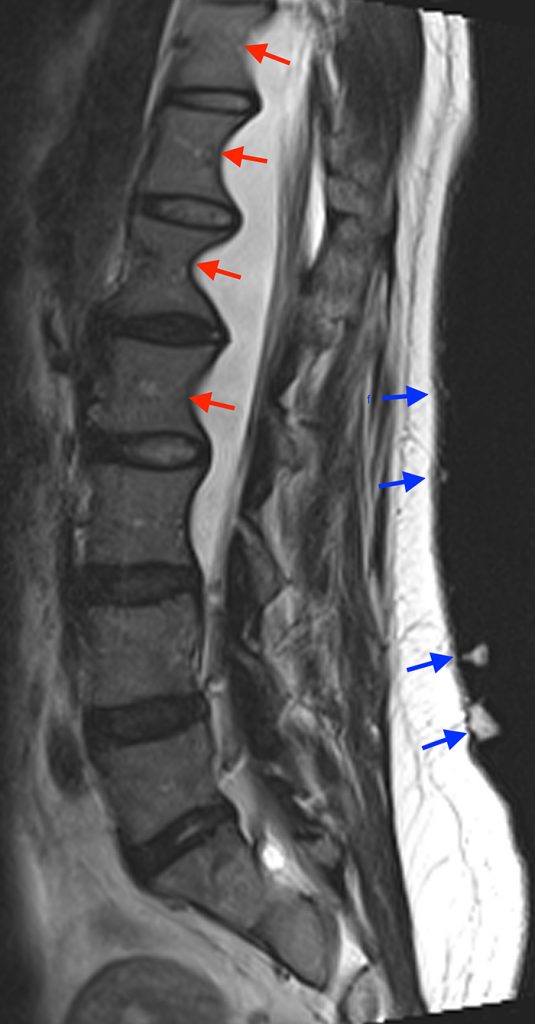
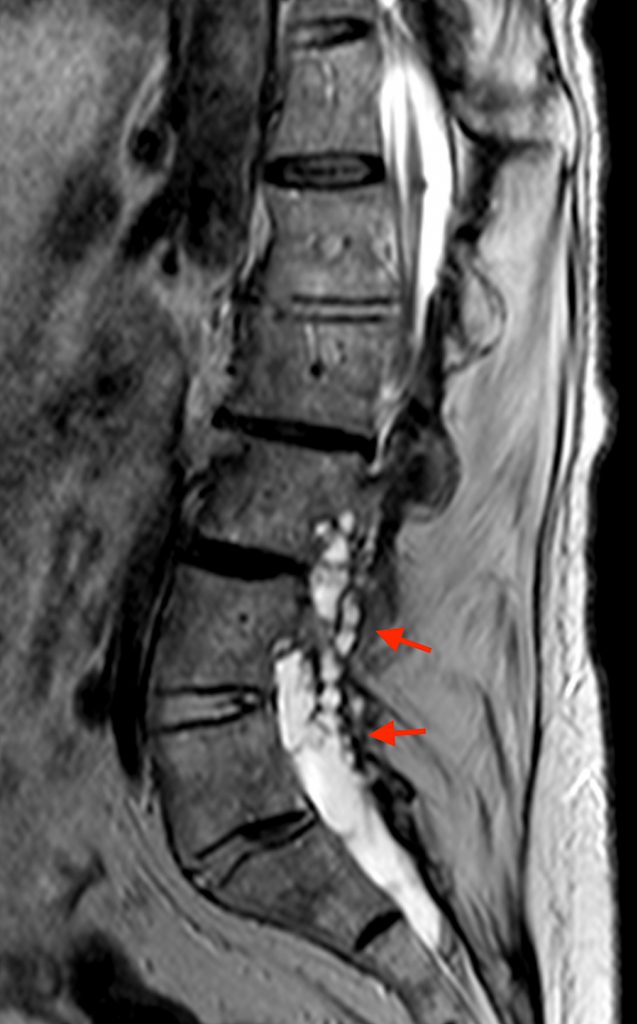
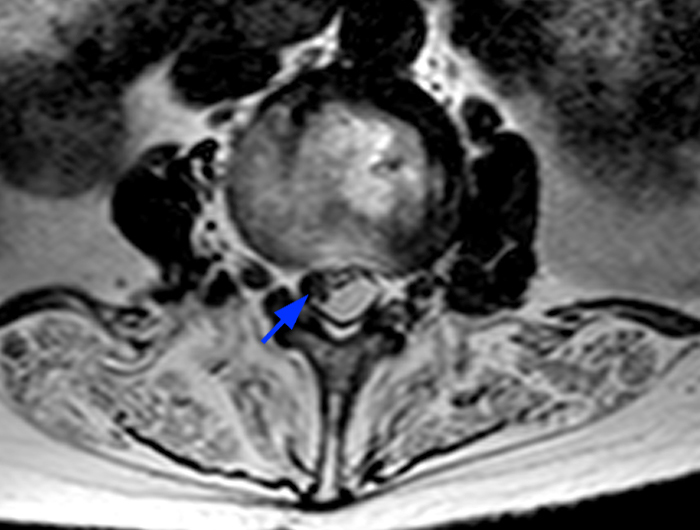
Posterior vertebral body scalloping with lack of epidural fat (red arrows) is demonstrated at the posterior wall of multiple vertebral body levels on sagittal T2-weighted (2a) and sagittal T1-weighted (2b) images, and the dural sac diameter at S1 is greater than at L4. A transitional lumbosacral vertebral body is denoted as L5. Axial T1-weighted (2c) and axial T2-weighted (2d) images demonstrate ballooning of the dural sac and again the lack of epidural fat at the posterior wall of the S1 vertebral body (red arrows).
Diagnosis
Dural Ectasia
Introduction
Dural ectasia is defined as ballooning or widening of the dural sac often associated with herniation of nerve root sleeves out of associated foramina sometimes accompanied by scalloping of the posterior vertebral body, thinning of cortex of pedicles and laminae, widening of neural foramina, meningoceles, or arachnoid cysts.1,2,3,4 Posterior vertebral scalloping can also occur in many other conditions but is also seen in normal adults. Mild posterior physiologic scalloping has been demonstrated in up to half of healthy adults.5
The diagnostic criteria for dural ectasia have been almost exclusively studied in Marfan syndrome although dural ectasia can also be associated with other inherited connective tissue disorders, including Ehlers-Danlos syndrome and Loeys-Dietz syndrome, type 1 neurofibromatosis, and ankylosing spondylitis. Dural ectasia can also be associated with trauma, scoliosis, tumors, or may be idiopathic.1 Up to 70-80% of dural ectasia is found in type 1 neurofibromatosis patients, and it has been described in 63% to up to 92% of Marfan syndrome patients.6,1,2,7
Dural ectasia can be a source of back pain, headaches, radicular pain, leg weakness, or urinary incontinence.1,5 Surgery may be needed at times to treat these symptoms or prevent complications.
Diagnostic Criteria
The diagnostic criteria for establishing dural ectasia have been most extensively studied in Marfan syndrome because it is one of several major criteria in Ghent nosology for clinical diagnosis. In fact, it has been found to be the second most common major diagnostic manifestation following aortic dilatation/dissection in Marfan patients.8 Fattori et al.7 first proposed an MRI based qualitative dural ectasia grading system with 4 grades: grade 0, normal, with no dural ectasia; grade 1, mild dural ectasia, defined by bulging of the dural sac and lack of epidural fat at the posterior wall of one vertebral body, presence of radicular cysts, or both features; grade 2, moderate dural ectasia, defined by bulging of the dural sac and lack of epidural fat at the posterior wall of 2 or more vertebral body levels and presence of radicular cysts; grade 3, severe dural ectasia, defined by the presence of an anterior sacral meningocele. Using these criteria, dural ectasia was established in 92% of patients diagnosed with Marfan syndrome and none of the control patients.
Ahn et al.1 attempted to establish simple criteria for diagnosing dural ectasia in Marfan patients based on a gold standard dural volume of > 7.0 cm3. The spinal canal was noted to be enlarged in patients with dural ectasia primarily in the most caudal portions of the spinal canal (sacral spine) and thus many measurements were obtained below L5. Dural sac diameter was measured in the sagittal plane throughout the lumbosacral spine. Nerve root sleeve diameter was measured on axial T1-weighted images at the level of the neural foramina. Scalloping was calculated by measuring the superior sagittal width, inferior sagittal width, and mid-sagittal width of sacral bodies. The superior and inferior widths were then added and divided by 2 and the mid-sagittal width was then subtracted from this value. Ahn et al. established 1 major and 2 minor criteria combining qualitative and quantitative criteria. Major criteria were sagittal diameter at or below S1 greater than the sagittal diameter at mid axis of L4 or above, and the presence of an anterior meningocele. Minor criteria were scalloping at S1 > 3.5 mm and nerve root sleeve diameter at L5 > 6.5 mm in diameter calculated based on 2 standard deviations above the mean value in control patients. The diagnosis of dural ectasia required either 2 major criteria or 1 major and 2 minor criteria. Using these criteria, CT demonstrated 82.6% sensitivity and 90% specificity compared to volumetric data and MRI demonstrated 87% sensitivity and 95% specificity.
A single quantitative measurement, dural sac ratio, was studied by Oosterhof et al.2 The dural sac ratio was determined between the midcorpus dural sac diameter and vertebral body diameter in controls from L1-S1 and the 95% confidence interval was used as a cutoff value of normal. The dural sac ratio cutoff values from L1-S1 were calculated as 0.64, 0.55, 0.47, 0.48, 0.48, and 0.57. Utilizing the cutoff values at L3 or S1, yielded a sensitivity of 95% and specificity of 98%. In their patient population, dural ectasia can be present at one or two levels, mostly at S1, and not necessarily involve the entire lumbosacral spinal canal. As found in other studies, the predominance of dural ectasia at the lowest vertebrae supports the theory that the development of dural ectasia in Marfan syndrome patients is influenced by the level of highest hydrostatic pressure in upright posture on a progressively weakened, congenitally defective, dural wall.5,7
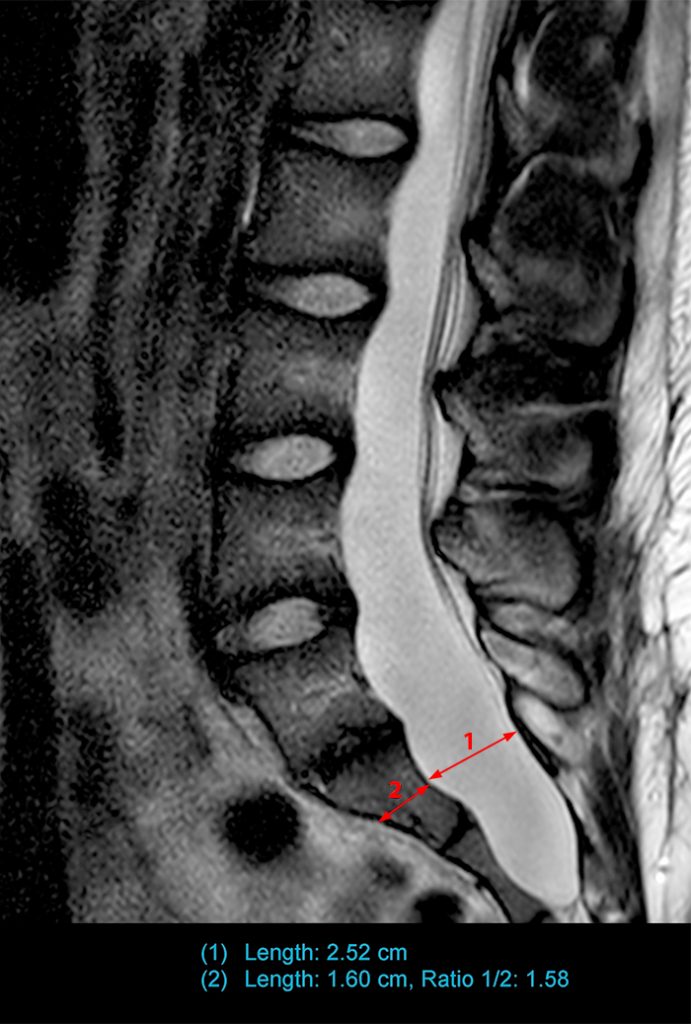
Two additional studies that followed these prior studies attempted to reassess the effectiveness of the above established criteria. Habermann et al.3 reassessed the criteria established by Fattori et al., Ahn et al., and Oosterhof et al. in children, adolescents, and young adults. They concluded that only the dural sac ratio at L5 and S1 and sagittal dural sac diameter at S1 greater than at L4 could be considered useful criteria in children, adolescents, and young adults. The dural sac ratio cutoff values used at L5 and S1 were 0.42 and 0.51 at L5 and S1, respectively, lower than in the Oosterhof et al. study. This could be age-related and related to hydrostatic pressures. Lundby et al.9 performed a case control study in adults with a larger age and sex-matched control group to better establish prevalence and the best criteria of dural ectasia. They found that qualitative signs were very useful as nerve root sleeve herniations were present in many of the Ghent positive patients and without which some of these patients would not have been diagnosed with dural ectasia (Figure 3). However, they found the dural sac ratio cutoff values proposed by Oosterhof et al. to be too low as they suggested a significant number of patients with dural ectasia in the control group similar to findings in other studies.10 The L3 level had no discriminating value and the dural sac ratio best indicated the presence of dural ectasia at L5 and S1, similar to findings supported by Habermann et al. Lundby et al. recommended a dural sac ratio cutoff value of 0.59 at S1. They also concluded that the diagnosis of dural ectasia should be based on the presence of at least one of the following: (1) anterior meningocele, (2) dural sac diameter at level S1 or below greater than at level L4, and (3) dural sac ratio at S1 > 0.59 (Figure 3).
Figure 3:
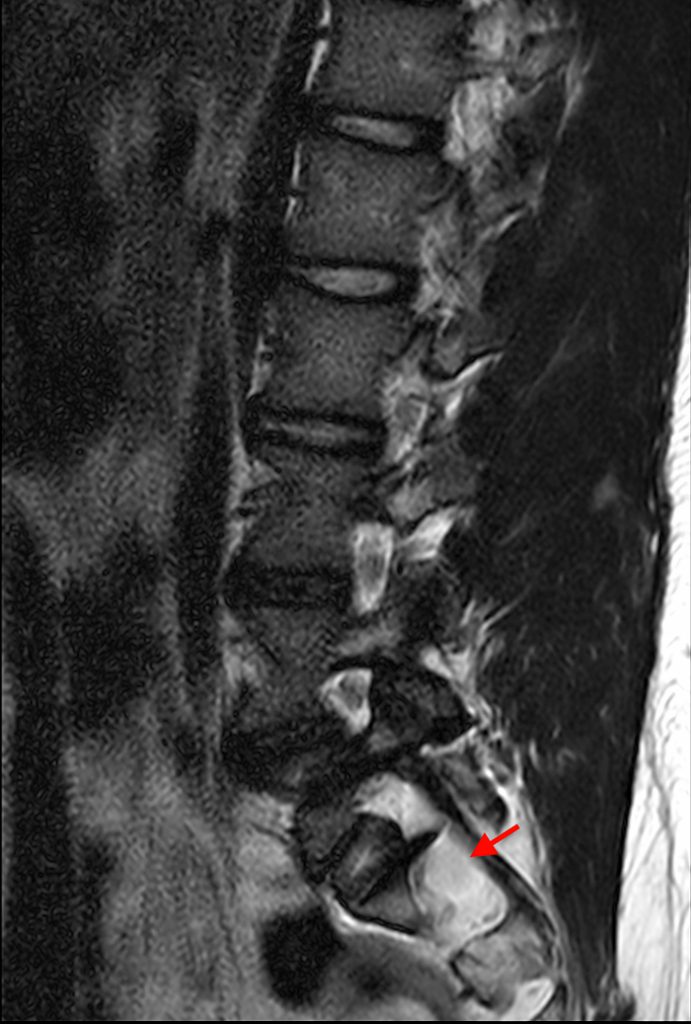
Sagittal T2-weighted images obtained towards the right (3a) and left (3b) neural foramina in the original case presentation demonstrate nerve root sleeve herniations (red arrows) at S1. In addition, sagittal T2-weighted image at midline (3c) demonstrates a calculated dural sac ratio of 1.58, more than sufficient to meet the criteria suggested by several authors.
Imaging Features & Differential Considerations
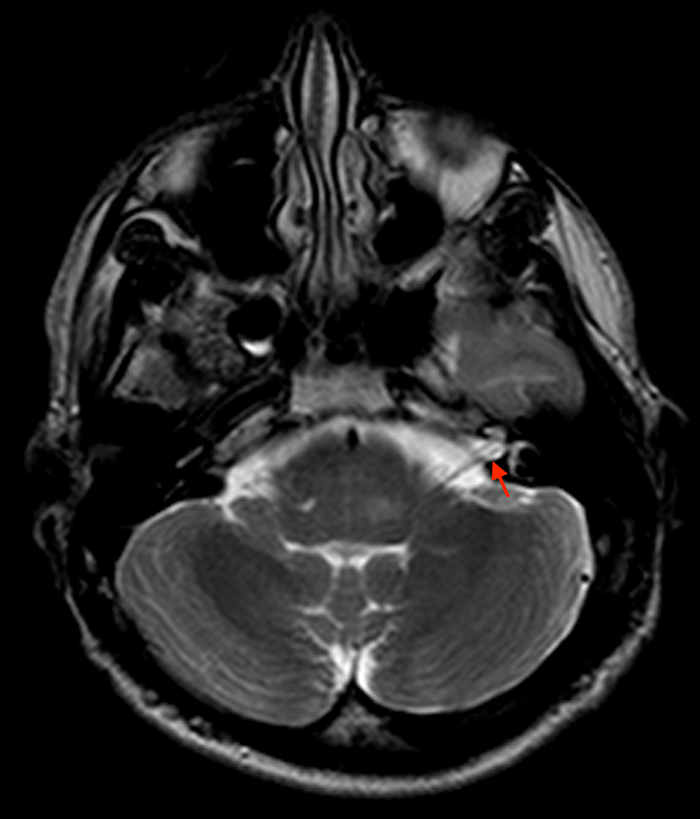
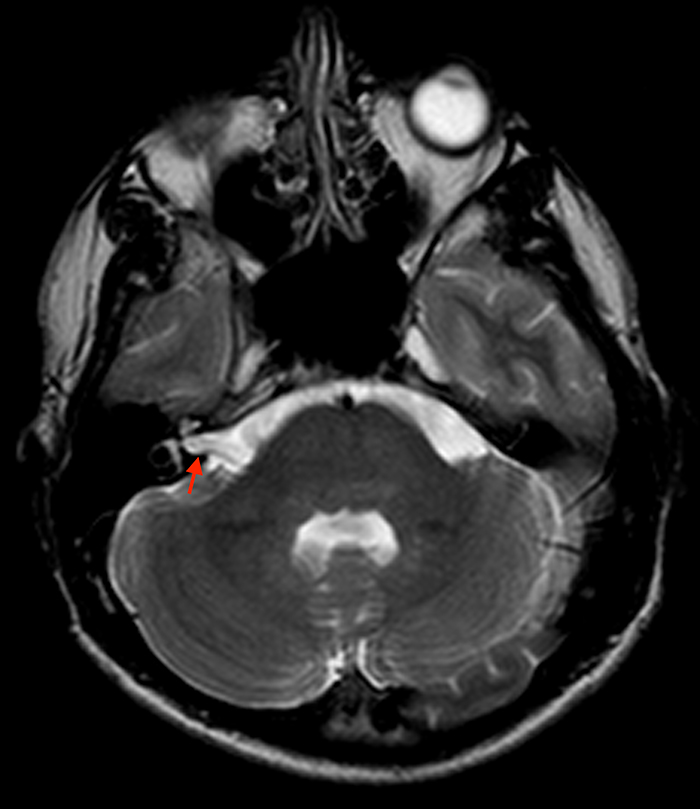
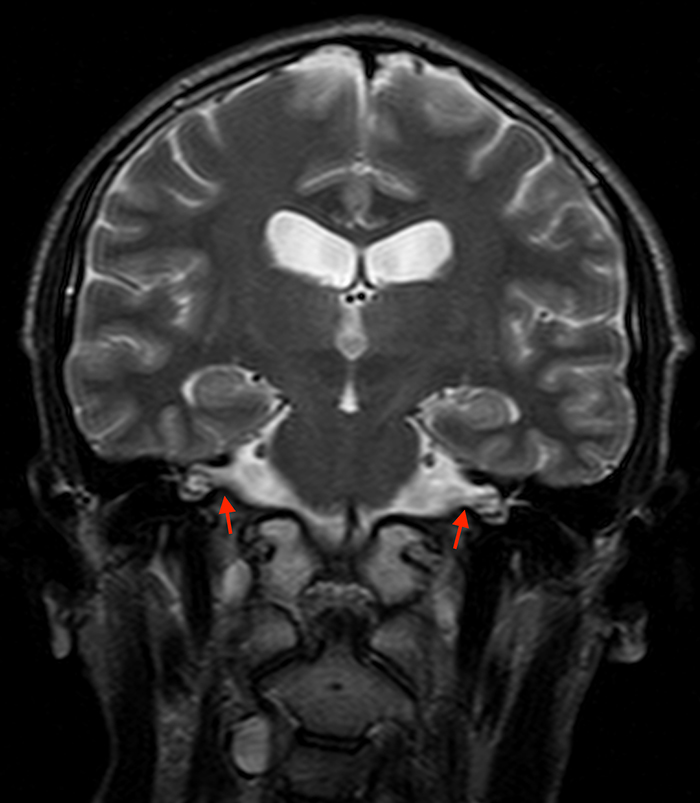
Aside from Marfan syndrome, dural ectasia is also a manifestation of type 1 neurofibromatosis and has been described in cranial and spinal findings. Dural ectasia can present as internal auditory canal ectasia without 7th or 8th cranial nerve masses (Figure 4) and can be present as dural ectasia of the optic nerve sheath.11,12 In the spine, lateral and anterior meningoceles commonly occur in the thoracic spine due to pressure differences between the thorax and subarachnoid space with posterior vertebral scalloping or bone defects.13 Meningoceles usually occur in association with thoracic scoliosis (Figure 5). Other findings include wedging of vertebral bodies, pedicle erosion, foraminal enlargement, and kyphosis.6 Sacral lesions in type 1 neurofibromatosis are commonly due to direct involvement by neurofibromas or remodeling caused by dural ectasia with foraminal enlargement.13
Figure 5:
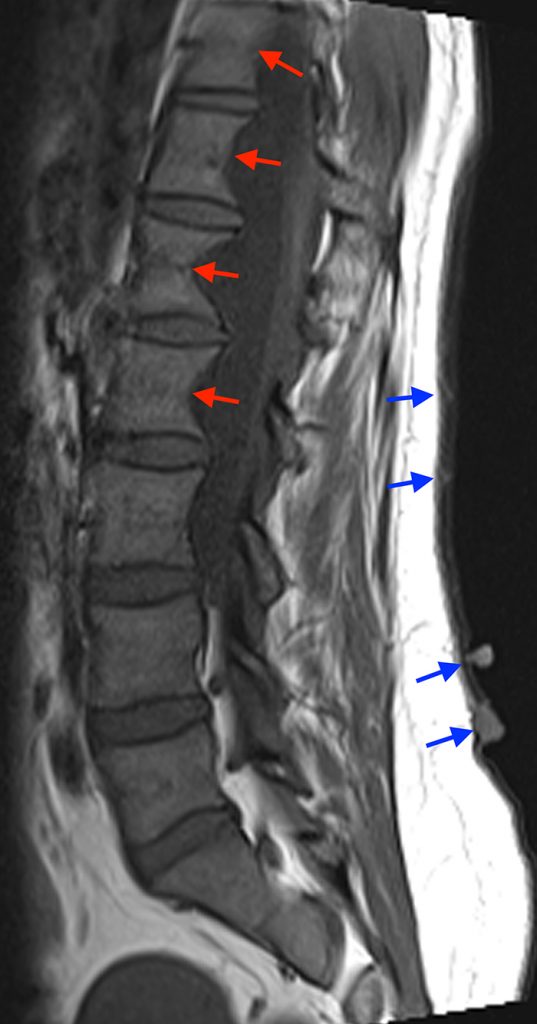
Sagittal T2-weighted (5a) and T1-weighted (5b) images demonstrating dural ectasia with prominent posterior vertebral scalloping (red arrows) in the lower thoracic and upper lumbar spine. Axial T2-weighted (5c) and T1-weighted (5d) images reveal the presence of lateral meningoceles (green arrows) with posterior vertebral scalloping. Note the multiple skin nodules (blue arrows) secondary to cutaneous neurofibromas.
Dural ectasia is also described as a manifestation of ankylosing spondylitis usually involving the lumbosacral regions but with asymmetric bony erosions predominantly involving the posterior elements including pedicles, laminae, and spinous processes.14,15 On MRI, multiple dorsal dural diverticula may be identified (Figure 6). These outpouchings are consistently associated with long-standing ankylosing spondylitis. The exact mechanism of dural ectasia is unclear but available evidence suggests local enthesitis and ligamentitis around the spinal canal contribute to arachnoiditis with fibrosis and adhesions of dura to surrounding structures, reducing elasticity and compliance of the thecal sac to pressure fluctuations of CSF.15,16 Local inflammation causes a slowly progressive process that leads to bony erosion with compression due to expansive arachnoid diverticula. These factors lead to dural sac enlargement, diverticula formation, and bony erosion.16
Figure 6:
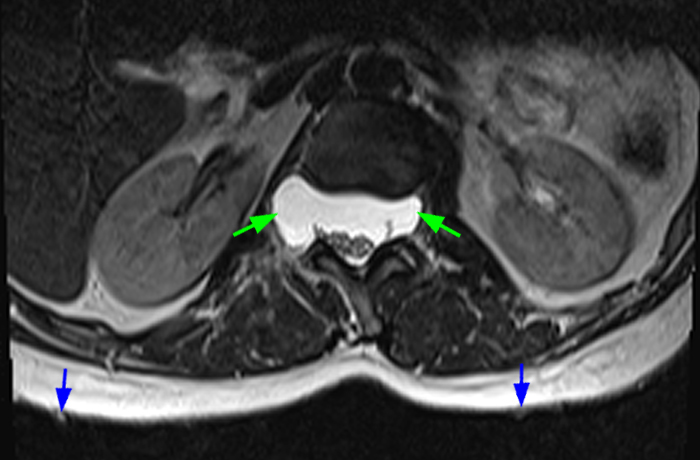
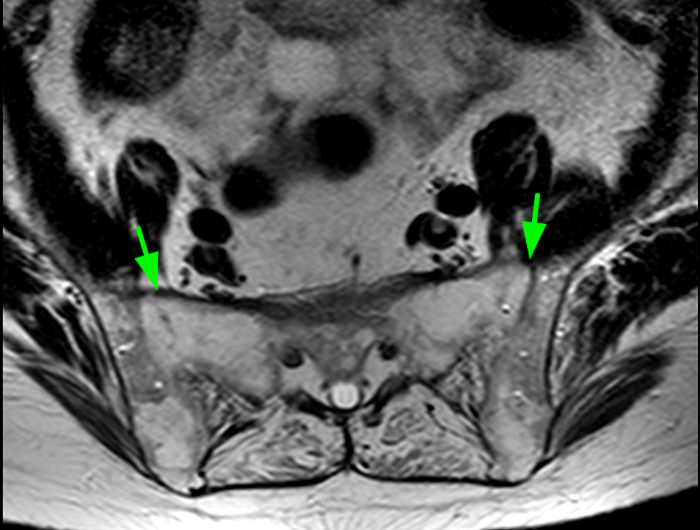
Axial T2-weighted (6a) and sagittal T2-weighted (6b) images at L4-5 level in a patient with ankylosing spondylitis demonstrates multiple arachnoid diverticula and erosions (red arrows) of the laminae and spinous processes. Note the adhesion of nerve roots in the periphery of diverticula. Axial T2-weighted image at the L2-3 level (6c) demonstrates clumping of nerve roots (blue arrows) suggestive of arachnoiditis. An axial T2-weighted image at the sacroiliac joints (6d) documents end-stage sacroiliitis with complete ankylosis (green arrows).
Complications/Symptoms
In Marfan syndrome, dural ectasia may be accompanied by spondylolisthesis, scoliosis, vertebral erosions, and/or fractures.5 Patients may be asymptomatic but can present with low back pain that can progress to radicular pain, leg weakness, or urinary incontinence.1,5
In type 1 neurofibromatosis, erosions of the posterior vertebral body can progress into angular deformities (usually less than 6 vertebral bodies) and vertebral fractures and dislocations.6 These deformities are known to progress and evolve over the disease course in a phenomenon known as “modulation” although neural deficit rarely occurs because the spinal canal is widened. Patients with angular deformities should be closely monitored for progression as early surgical intervention may be necessary to prevent severe deformity and late complications.
Dural ectasia sometimes leads to cauda equina syndrome in long standing ankylosing spondylitis (also called CES-AS syndrome), a rare complication.15,16,17 Several cases have been reported in the literature with the first cases reported dating back to 1961 by Bowie and Glasgow.18 Several possible mechanisms for CES-AS syndrome include small vessel angiitis of vasa vasorum of nerve roots, previous spinal irradiation for treatment of ankylosing spondylitis, nerve root damage due to dural ectasia with decreased elasticity of the dural sac and increased arterial pulsatile forces transmitted through CSF, and a chronic inflammatory process.15 Inflammation may spread from the peridural soft tissues to the nerve roots of the cauda equina resulting in cauda equina syndrome and arachnoiditis. There has been no effective medical or surgical treatment with most cases demonstrating little to no improvement of symptoms. Only one case showed dramatic improvement of symptoms but there were signs of active inflammation.19 The effect of treatment may be based on timing of intervention, whether there is active inflammation or irreversible nerve root damage. MRI and CSF studies may help with diagnosis and treatment planning in CES-AS syndrome patients.15
Conclusion
Dural ectasia can be assessed by qualitative and/or quantitative criteria but several studies have concluded that diagnosing dural ectasia in Marfan syndrome patients is best done using a combination of both qualitative and quantitative criteria. Useful qualitative criteria include anterior meningocele as well as nerve root sleeve herniations. The dural sac diameter at S1 or below greater than the dural sac diameter at L4 and the dural sac ratio at S1 > 0.59 are useful quantitative criteria. Aside from Marfan syndrome, dural ectasia may be found in association with other connective tissue disorders such as Ehlers-Danlos and Loeys-Dietz syndrome. Type 1 neurofibromatosis and ankylosing spondylitis are also commonly associated with dural ectasia. The knowledge of the diagnostic criteria for dural ectasia and its differentiating imaging features can help in not only diagnosing the process, but also determining the underlying etiology in those patients who do not yet have an exact clinical diagnosis.
References
- Ahn NU, Sponseller PD, Ahn UM, et al. Dural ectasia in the Marfan syndrome: MR and CT findings and criteria. Genet Med 2000; 2:173–179. ↩
- Oosterhof T, Groenink M, Hulsmans FJ, et al. Quantitative assessment of dural ectasia as a marker for Marfan syndrome. Radiology 2001; 220:514–518. ↩
- Habermann CR, Weiss F, Schoder V, et al. MR evaluation of dural ectasia in Marfan syndrome: reassessment of the established criteria in children, adolescents, and young adults. Radiology 2005; 234:535– 41 ↩
- Ha HI, Seo JB, Lee SH, et al. Imaging of Marfan syndrome: multisystemic manifestations. Radiographics 2007; 27(3):989-1004. ↩
- Ho NC, Hadley DW, Jain PK, et al. Case 47: Dural ectasia associated with Marfan syndrome. Radiology 2002; 223:767-771. ↩
- Woon CY. Dural ectasia: a manifestation of type 1 neurofibromatosis. CMAJ. 2010 Sep 21;182(13):1448. ↩
- Fattori R, Nienaber CA, Descovich B, et al. Importance of dural ectasia in phenotypic assessment of Marfan’s syndrome. Lancet 1999; 354:910–913. ↩
- Rose PS, Levy HP, Ahn NU, et al. A comparison of the Berlin and Ghent nosologies and the influence of dural ectasia in the diagnosis of Marfan syndrome. Genet Med 2000; 2:278–282. ↩
- Lundby R, Rand-Hendriksen S, Hald JK, et al. Dural ectasia in Marfan syndrome: a case control study. AJNR Am J Neuroradiol. 2009; 30(8):1534-40. ↩
- Weigang E, Ghanem N, Chang XC, et al. Evaluation of three different measurement methods for dural ectasia in Marfan syndrome. Clin Radiol 2006; 61:971–78 ↩
- Aoki S, Barkovich AJ, Nishimura K, Kjos BO, et al. Neurofibromatosis types 1 and 2: cranial MR findings. Radiology. 1989; 172(2):527-34. ↩
- Lövblad KO, Remonda L, Ozdoba C, et al. Dural ectasia of the optic nerve sheath in neurofibromatosis type 1: CT and MR features. J Comput Assist Tomogr. 1994; 18(5):728-30. ↩
- Fortman BJ, Kuszyk BS, Urban BA, et al. Neurofibromatosis type 1: a diagnostic mimicker at CT. Radiographics. 2001; 21(3):601-12. ↩
- Grosman H, Gray R, St Louis EL. CT of long-standing ankylosing spondylitis with cauda equina syndrome. AJNR Am J Neuroradiol. 1983; 4(5):1077-80. ↩
- Liu CC, Lin YC, Lo CP, et al. Cauda equina syndrome and dural ectasia: rare manifestations in chronic ankylosing spondylitis. Br J Radiol. 2011; 84(1002):e123-5. ↩
- Van Hoydonck M, de Vlam K, Westhovens R, et al. Destructive dural ectasia of dorsal and lumbar spine with cauda equina syndrome in a patient with ankylosing spondylitis. Open Rheumatol J. 2010 Aug 26;4:31-4. ↩
- Huang ZX, Deng WM, Guo X, et al. Lumbar bone erosion with anterior dural ectasia in ankylosing spondylitis: a case report. J Int Med Res. 2019; 47(3):1395-1400. ↩
- Bowie EA, Glasgow GL. Cauda Equina Lesions Associated with Ankylosing Spondylitis. Br Med J. 1961; 2(5243):24-7. ↩
- Cornec D, Devauchelle Pensec V, Joulin SJ, et al. Dramatic efficacy of infliximab in cauda equina syndrome complicating ankylosing spondylitis. Arthritis Rheum. 2009; 60(6):1657-60. ↩