Findings
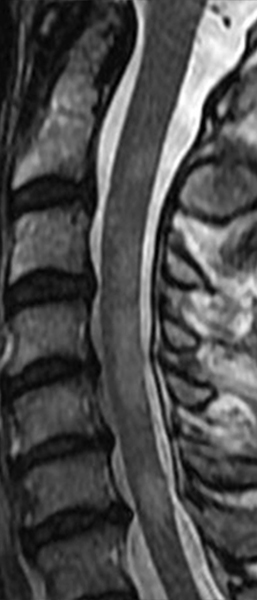
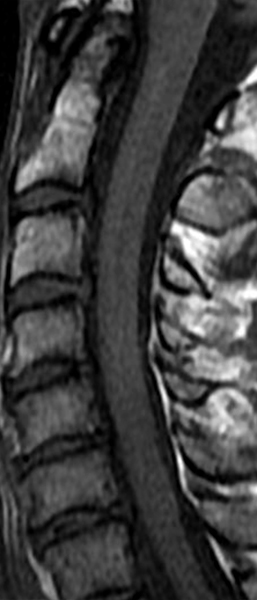
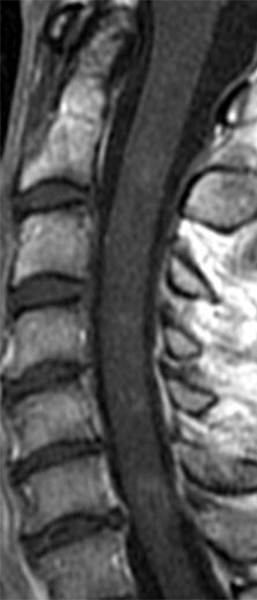
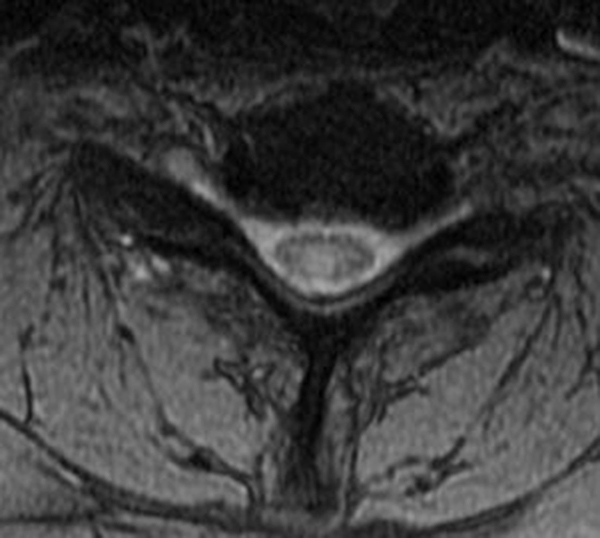
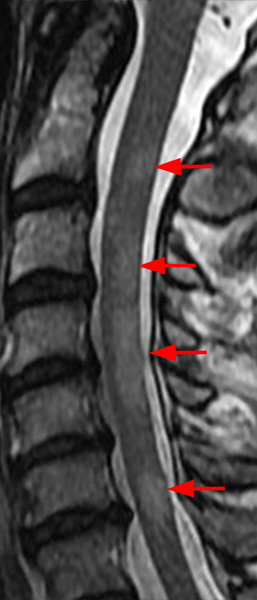
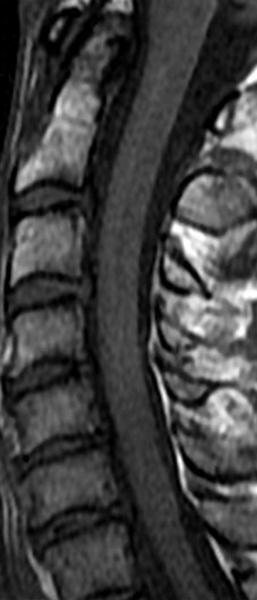
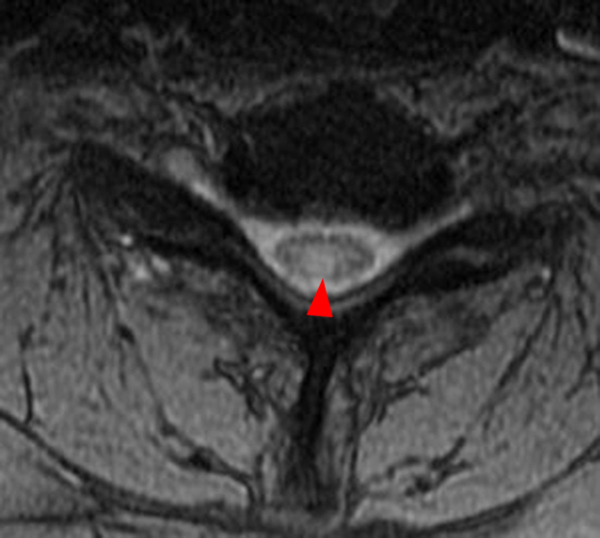
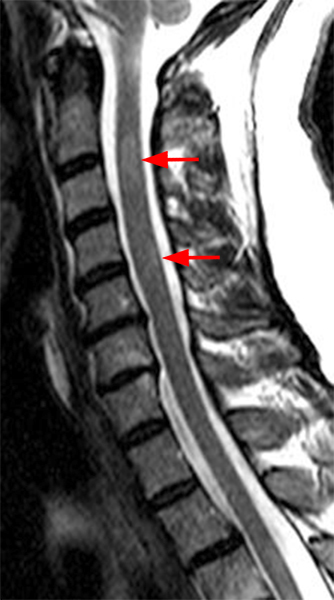
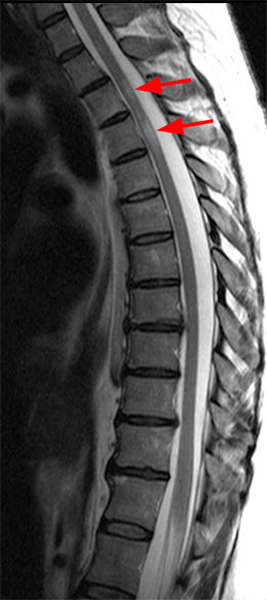
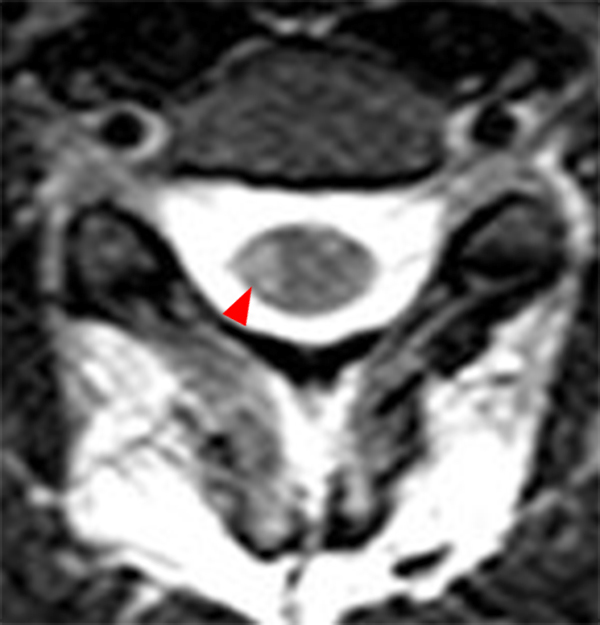
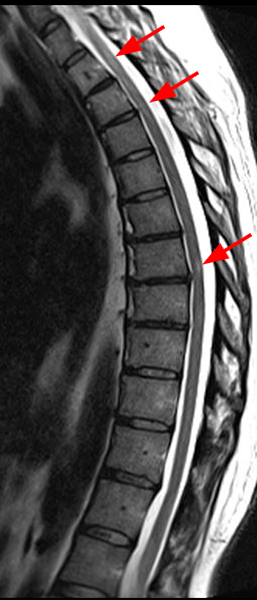
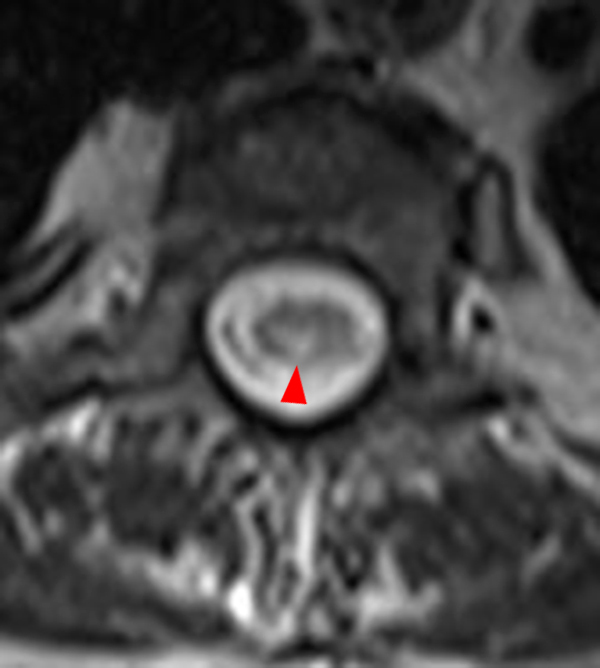
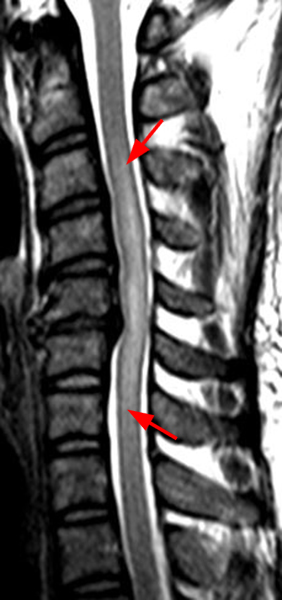
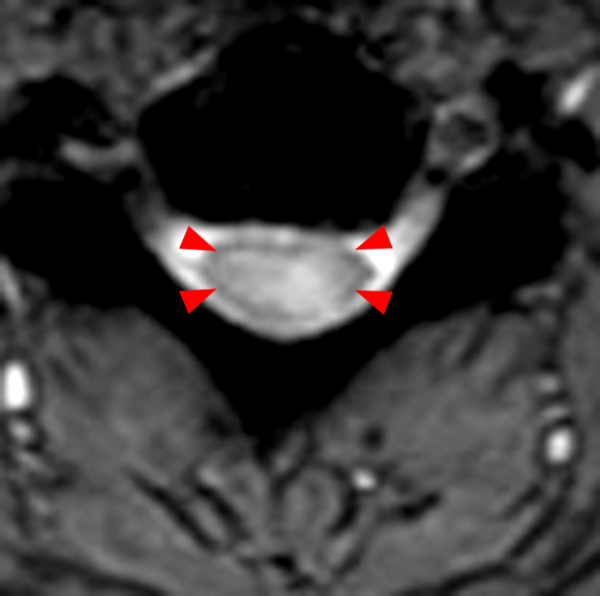
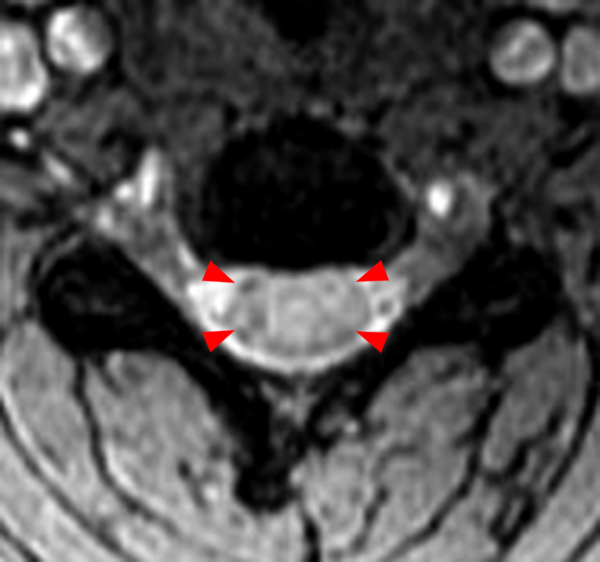
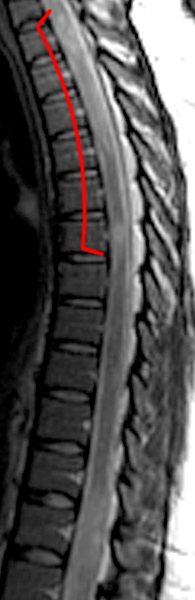
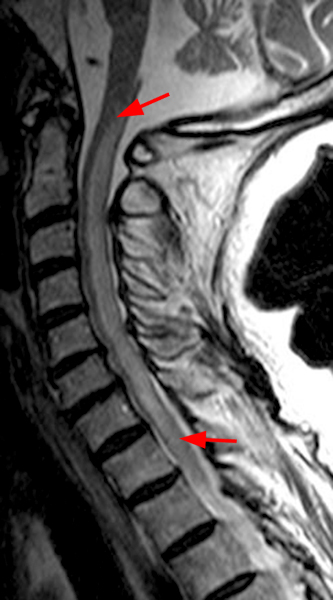
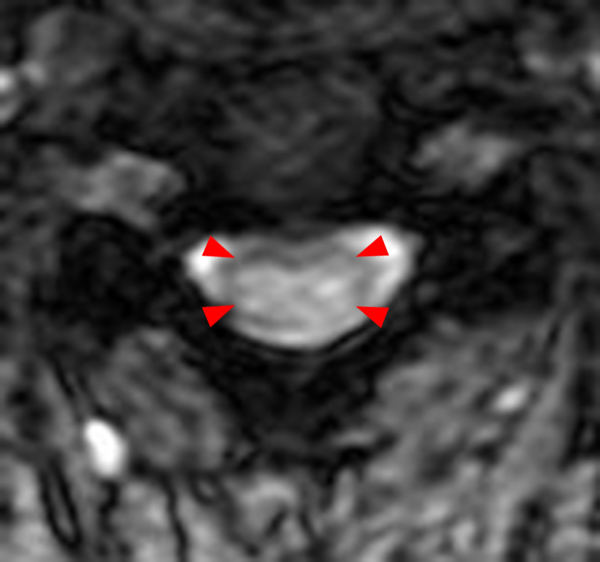
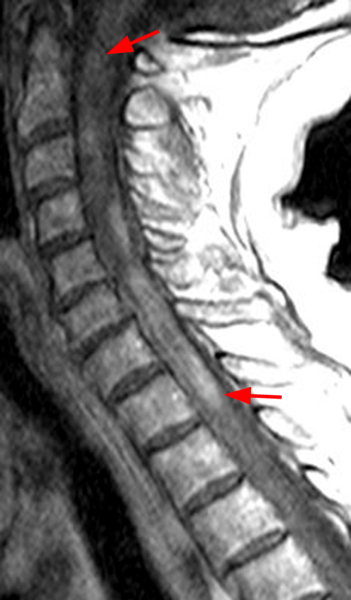
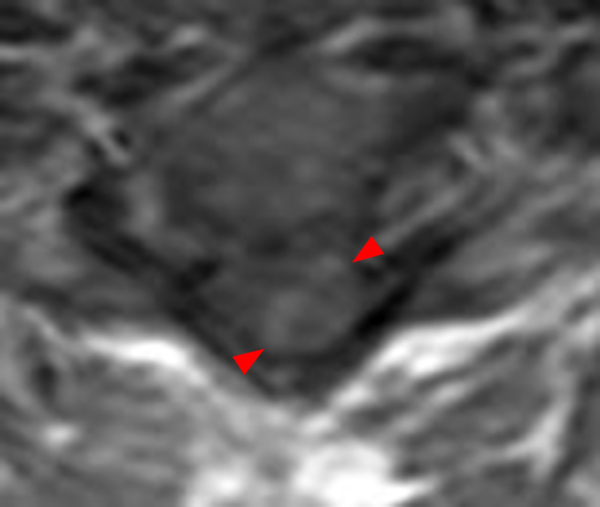
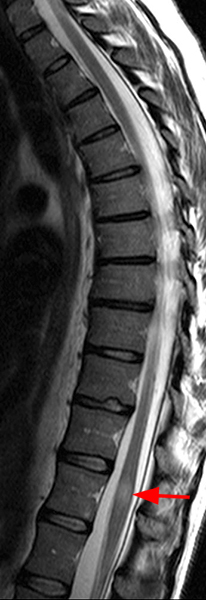
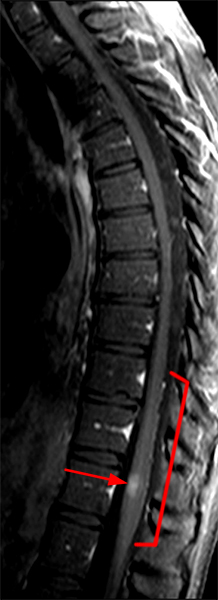
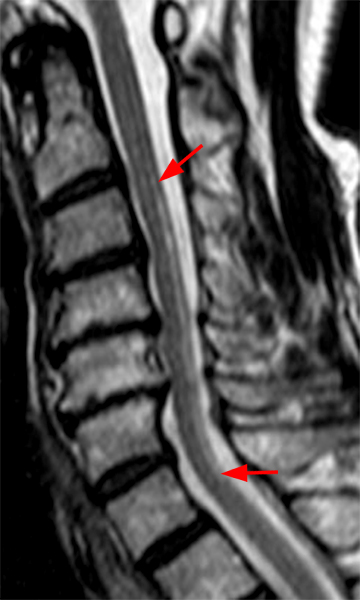
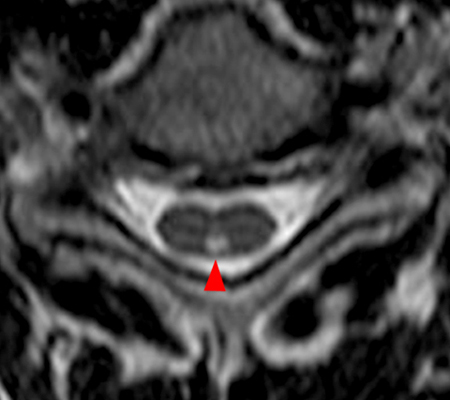
The sagittal T2-weighted image of the cervical spine (2a) demonstrates multifocal ill-defined hyperintense cord lesions centered at the inferior C2, C3-4, C4 to C5, and C6-7 levels (arrows), with no corresponding signal abnormality on pre-contrast sagittal T1-weighted image (2b), and ill-defined, incomplete enhancement (dotted arrows) on postcontrast imaging (2c). On axial T2*-weighted imaging (2d) at the C6-7 level, the increased T2 signal is centered within the dorsal cord just to the right of midline (arrowhead) and occupies less than 50% of the cross-sectional area of the cord.
Diagnosis
Acute to subacute Multiple Sclerosis (MS) demyelinating plaques.
Introduction
Demyelination is a generic term that describes the loss of normal myelin around axons in the central nervous system, which can be a cause of non-compressive spinal cord signal change on MRI.1,2,3,4,5
Demyelinating lesions of the spinal cord result from inflammation that damages myelin sheath-forming cells and may be primary, due to a condition such as multiple sclerosis (MS), or secondary, as in post-infectious acute disseminated encephalomyelitis (ADEM) or post-infectious transverse myelitis.
MS, an autoimmune cell-mediated demyelinating disorder, is the most common primary cause of demyelinating disease (approximately ten times more common than the other etiologies) and affects both the brain and spinal cord, with demyelinating plaques that are separated over both time and space.1, 2, 3, 4, 5 MS most commonly affects women, especially within the age range of 20-40 years old, and is the second most common cause of neurologic impairment in young adults after trauma. Multiple forms of MS exist, with the most common form, the relapsing-remitting form, characterized by a combination of exacerbations followed by partial or complete recovery. Neuroimaging with MRI plays a central role in diagnosis, prognosis estimation, and disease monitoring for demyelinating diseases.
Clinical Presentation
Patients may present with paresthesia, muscle weakness, gait disturbance, and bowel and/or bladder dysfunction. CSF analysis typically demonstrates oligoclonal bands.
MR Imaging
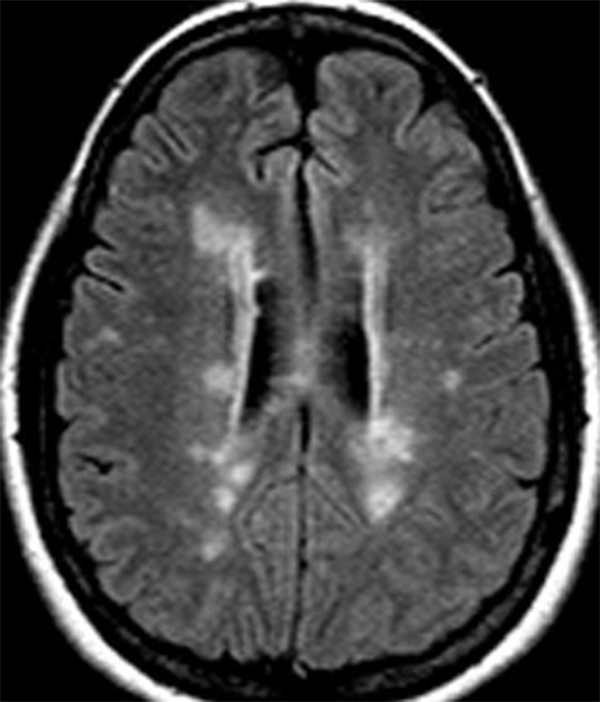
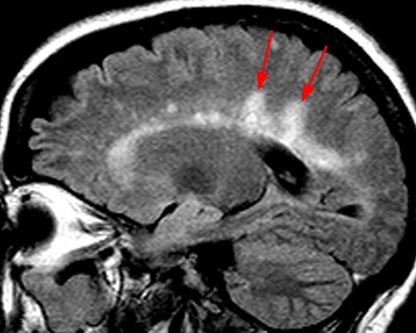
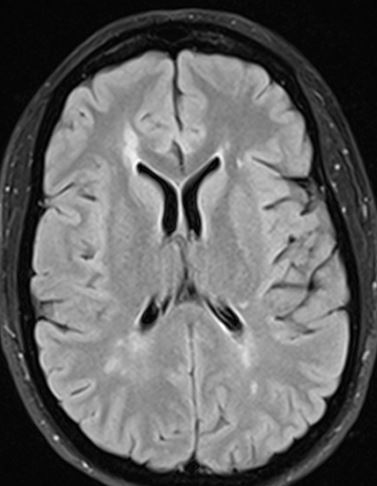
On imaging, focal discrete, or ill-defined foci (oval or wedge shaped) of T2 hypertense signal are seen within the spinal cord.2, 3, 4, 5,6 Isolated spinal cord involvement has been reported in up to 25% of cases, with the vast majority of patients (more than 90%) with clinically definite MS demonstrating spinal cord involvement.2 The cervical spinal cord is the most commonly affected region of the cord, and demyelinating plaques are most commonly peripheral in distribution, with a preference for the dorsal and lateral white matter tract, short segment (classically involving less than 1.5 vertebral body lengths), and typically affect less than half of the cross-sectional area of the cord.4, 6, 7 Occasionally, MS plaques may extend to involve central grey matter, occupying over half of the cross-sectional area of the cord. Small focal lesions may also coalesce to form more extensive ones and can extend to involve three or more spinal segments, particularly in cases of the progressive form of MS.2,3 Perilesional cord edema and marked cord expansion are not common, which may help distinguish demyelinating plaques from intramedullary tumors.4 Associated enhancement is seen with acute to subacute demyelination, and the enhancement pattern can change from focal to less well-defined as inflammation evolves. Incomplete ring enhancement, similar to MS plaques in the brain, may be present. Cord atrophy may be seen in the later stages of disease and may correlate with clinical disability. Abnormal white matter T2/FLAIR hyperintensity in a periventricular, pericallosal, cerebellar, or brainstem distribution on MR brain imaging provides support for the diagnosis (Figures 3 and 4).2, 3, 4
A single clinical event that is demyelinating in nature, where the clinical and MRI criteria for dissemination in time are not met is called clinically isolated syndrome (CIS) and prompts consideration of whether this represents a first episode of MS and what to expect in terms of prognosis and risk of relapse.7 Studies evaluating the risk of conversion to MS have contributed to the revision of some of the MRI criteria to allow earlier diagnosis of MS, and the most sensitive measure has been the presence of typical MS lesions on MRI at the time of presentation. Radiologically isolated syndrome (RIS) has been described as the entity when MRI findings typical of MS are seen in asymptomatic patients, often seen in the workup of unrelated conditions (i.e., headache, head injury, chronic back pain). Some studies have shown that some of these patients will develop clinically definite MS, and as a result, cases of RIS require follow-up clinical and imaging evaluation.
Disease-modifying therapies may include multiple pharmacologic agents, such as high-dose IV steroids and monoclonal antibodies, as well as autologous hematopoietic stem cell transplant.3
Figure 3: A 59-year-old woman presents with numbness from the nipples to the feet. Sagittal T2-weighted images of the cervical spine (3a) and thoracic spine (3b) demonstrate multifocal T2 hyperintense cord lesions (arrows). Axial T2-weighted image at the level of the lesion in the cord at the C2-3 level (3c) demonstrates a focal increased T2/FLAIR signal lesion within the right lateral cord (arrowhead). Axial (3d) and sagittal (3e) FLAIR MR brain images demonstrate multifocal T2/FLAIR hyperintense white matter lesions, several of which are periventricular and oriented perpendicular to the lateral ventricles, in a “Dawson’s fingers” configuration on sagittal imaging (long arrows). It has been postulated that this represents periventricular inflammation and is considered a relatively specific imaging sign for MS.
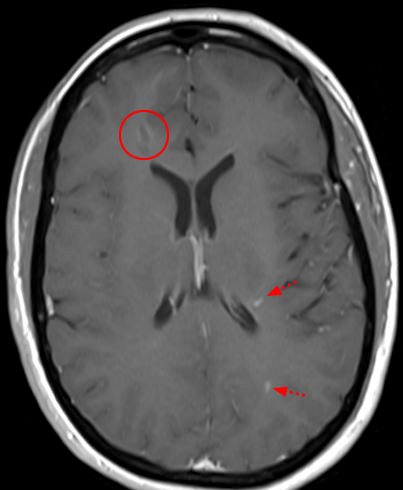
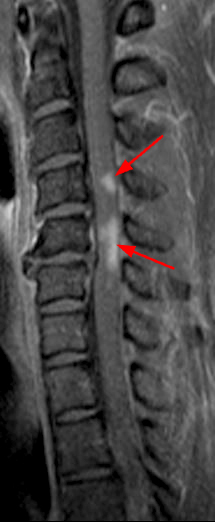
Figure 4: 42-year-old woman with tingling in the fingertips. Sagittal T2-weighted image (4a) showing multiple subtle increased cord signal abnormalities (red arrows), compatible with multiple MS plaques. A representative axial T2-weighted image at the T6 level (4b) demonstrates a T2 hyperintense plaque centered within the dorsal/right lateral spinal cord (arrowhead). On follow-up MRI brain imaging, axial FLAIR (4c) image demonstrates multiple periventricular T2/FLAIR hyperintense lesions and postcontrast T1-weighted imaging (4d) demonstrates enhancement associated with some of the lesions (short arrows), including a peripherally enhancing right frontal periventricular lesion (circled).
Differential
Imaging differentials for MS plaques in the spinal cord includes other inflammatory causes of myelopathy, such as Neuromyelitis Optic Spectrum Disorder (NMO), Acute disseminated Encephalomyelitis (ADEM), post-viral acute transverse myelitis, and idiopathic acute transverse myelitis.2, 3, 4 Vascular causes of non-compressive myelopathy, such as cord infarct and dural arteriovenous fistula (dAVF), and metabolic causes of non-compressive myelopathy, such as subacute combined degeneration of the cord may also cause similar cord signal abnormality and myelopathic symptoms.
Neuromyelitis Optic Spectrum Disorder (NMO) is an inflammatory disease of the CNS that typically demonstrates seropositivity for the astrocytic aquaporin-4 immunoglobulin G (AQP4-IgG) water channel receptor.3, 4, 5, 7, 8 Patients may present with optic neuritis, which may be bilateral, and can involve the optic chiasm, with pronounced vision loss.7 Spinal cord lesions classically clinically manifest as complete acute spinal cord syndrome. Other presenting symptoms are dependent upon the locations of any additional CNS lesions and can include oculomotor dysfunction, long tract signs, and ataxia when the brainstem is involved, or intractable nausea, hiccups, and vomiting when the area postrema is involved. A classic triad of optic neuritis, longitudinally extensive transverse myelitis involving more than three vertebral segments, and positive serologic analysis has been described.4, 6 Patients with NMO may have clinical relapses as in MS. However, there is typically no gradual decline in functional status between relapses. Many medications for MS may cause NMO to worsen, so the distinction is clinically important.3
In the acute setting on MRI, the spinal cord is often expanded, with central T2 hyperintensity, and enhancement.2, 3, 4, 5, 6, 7 The lesions are usually more extensive longitudinally than MS plaques, commonly involving three or more segments, usually involve the central gray matter of the cord, and can also extend upward to involve the brainstem (Figure 5). On MRI brain imaging, in addition to optic neuritis, T2 hyperintense lesions may be present in the areas with the highest concentrations of aquaporin-4 receptors – including the periependymal surfaces of the third and fourth ventricles, and at the area postrema. The dorsal medulla, hypothalamus, and thalamus are often involved and there may be long confluent corpus callosum lesions. Bright spotty lesions, described as T2-hyperintense foci with signal intensity similar to that of CSF within a T2-hyperintense spinal cord lesion, are suggestive of NMO.2, 4 Treatment may involve classic immunosuppressants and/or biologic agents, such as monoclonal antibodies.
Figure 5: 43-year-old man with a reported history of NMO. Sagittal T2-weighted image (5a) with long segment, non-expansile T2 cord signal abnormality (between the arrows). On the axial T2*-weighted image (5b), the T2 signal abnormality is poorly defined and involves a large cross-sectional area (arrowheads). On sagittal fat-suppressed T1-weighted postcontrast imaging (5c), there is corresponding heterogeneous enhancement within the affected segment of the cervical spinal cord (long arrows).
Acute disseminated Encephalomyelitis (ADEM), a widespread postinfectious or postvaccination inflammatory demyelinating disease affecting the brain and spinal cord, most commonly affects children and young adults.2, 3, 4, 5, 6, 7 Multiple viral and bacterial diseases have been implicated in the development of ADEM, including varicella, rubella, measles, EBV, and mycoplasma infection. The mechanism is not well understood but has been postulated to be secondary to an autoimmune response to a myelin antigen or T cell hypersensitivity reaction.1, 2, 3, 4 The onset of symptoms is usually relatively rapid – and can include myalgia, headache, nausea, vomiting, drowsiness following a multifocal neurologic disorder with encephalopathy, cranial nerve palsy, ataxia, and paresis.
On MRI, cord involvement is seen in approximately 30% of cases, classically as mild cord expansion with multifocal or segmental T2 hyperintensity, and more commonly found in the thoracic spine.2, 3, 5, 7 The T2 hyperintense cord signal lesions tend to be larger in cross-sectional area and are typically longer in CC extent than MS lesions (Figure 6). Brain involvement is common and consists of patchy asymmetric T2 hyperintense regions of the gray matter-white matter junction.2, 4 As a monophasic disorder, spinal cord and brain lesions are the same age (unlike MS).2,3 The presence of larger lesions in the subcortical white matter, involvement of the deep gray structures (basal ganglia and thalami) and brainstem, and relative sparing of the periventricular region may suggest ADEM over MS. The cranial nerves and gray matter can be involved, which is atypical for MS. Corpus callosum involvement is rare. CSF usually shows lymphocytic pleocytosis, increased protein levels, and increased pressure.2, 4 The oligoclonal bands typical of MS are rarely present. Treatment of ADEM utilizes immunosuppression, often with steroids, and recovery most frequently occurs within weeks or months, with complete recovery in 50% of cases. Follow-up imaging may show resolution compatible with a monophasic process.2, 3
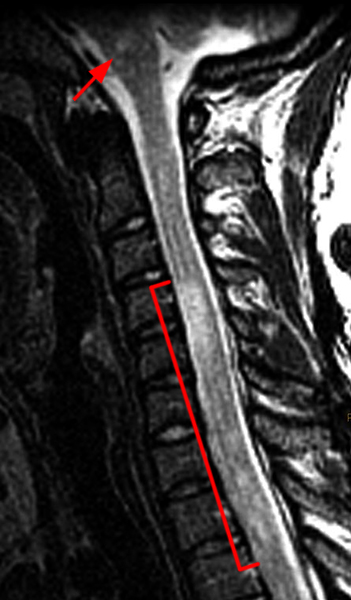
Figure 6: 30-year-old woman presenting with several weeks of left lower extremity paresis, and difficultly walking. Sagittal T2-weighted image of the cervical spine (6a) demonstrates multiple long segment T2 hyperintense cord signal lesions, with mild cord expansion (bracket), and T2 hyperintensity involving the brainstem (arrow). Axial T2*-weighted image (6b) at the C4 level demonstrates T2 hyperintense signal involving a large cross-sectional area of the cord (arrowheads). Sagittal T2-weighted image (6c) of the thoracic spine also demonstrates extensive, long segment, mildly expansive T2 hyperintense cord signal lesion (curved bracket). Given the multifocality of the signal abnormality, mild degree of cord expansion, and involvement of the brainstem, the differential of ADEM vs MS was favored. Postcontrast imaging was recommended but was not performed at our institution. This case is an example of a case in which imaging at a single point in time may not be adequate to confirm a diagnosis. In addition to correlation for a history of a preceding viral illness, CSF analysis and MRI brain imaging may contribute to diagnosis. Additional follow-up to evaluate for the resolution of clinical symptoms and imaging findings would be needed to help confirm a diagnosis of ADEM.
Post viral acute transverse myelitis is an acute inflammatory condition of the spinal cord which occurs either secondary to a direct viral infection, or as a post-viral immune response.2, 3, 4, 5, 6 Enteroviruses are the most common pathogens. Patients typically present with signs of acute myelopathy following a febrile illness. An edematous appearing spinal cord, with confluent T2-hyperintense signal in the affected regions, and variable patchy enhancement, are typical. Elevated mononuclear cells and protein levels may be present on CSF analysis.4 Brain involvement is uncommon.
Idiopathic Acute Transverse Myelitis is a diagnosis of exclusion in the setting of bilateral rapid development of myelopathy. 2, 4, 7, 8 Patients typically have a well-defined sensory level, evidence of inflammation on CSF analysis, and clinical symptoms typically progress over a period ranging from 4 hours to 21 days.4 Like post-viral transverse myelitis, there is longitudinally extensive central intramedullary T2 hyperintensity involving gray and white matter, spinal cord expansion, and variable contrast enhancement (Figure 7). While, in the absence of other definitive clues, acute transverse myelitis can be added to the differential diagnosis of most noncompressive myelopathies, it can often be excluded upon further diagnostic workup. In some studies, a more specific diagnosis, most commonly MS or spinal cord infarction, has been established in approximately 70% of cases initially diagnosed as idiopathic acute transverse myelitis.
Figure 7: Idiopathic acute transverse myelitis. 82-year-old presenting with left-sided paralysis, left-sided neck pain and increasing weakness for 1 to 2 weeks. T2-weighted sagittal (7a) and axial (7b) images of the cervical spine demonstrate long segment cord expansion and confluent hyperintense T2 signal involving the majority of the cervical spinal cord (between the arrows) and the majority of the cross-sectional area of the affected cord (arrowheads). Post-contrast T1-weighted sagittal (7c) and axial (7d) images, demonstrate corresponding heterogeneous enhancement in the affected segment of the spinal cord (between the arrows and arrowheads).
Cord infarction is relatively rare, but can result from anterior spinal artery ischemia and may cause a ventral cord syndrome resulting from involvement of the anterior 2/3 of the cord.2, 4, 5 Patients may present with acute symptoms of motor deficiency at the level below the level of infarction, which is often painful. Additional presenting symptoms can include bladder and/or bowel incontinence as well as loss of temperature and pain sensation (evidence of spinothalamic tract involvement). It should be noted that infarction of the posterior 1/3 of the cord is rare due to collateral vessels from the vascular pial plexus and paired posterior spinal arteries.2, 4
On MRI with diffusion-weighted imaging there is restricted diffusion in the region of ischemia, with a well-defined transition zone indicating an abnormality in the vascular territory, with corresponding linear, “pencil-like” T2 hyperintensity in the vascular territory.2, 4 If there is involvement of the anterior gray matter, this may result in an “owl’s eye” appearance on T2 weighted imaging. The imaging findings may not be present immediately – but are often seen a few hours after onset, with cord expansion often present on T1 weighted imaging in the acute phase. In the subacute phase, there may be corresponding enhancement.2 Hemorrhagic conversion is rare, but may also occur in the subacute phase, with intrinsic T1 shortening.4 Signal abnormality in the adjacent vertebral bodies corresponding to associated bone marrow infarction may be present and can be a helpful clue to this diagnosis.4
Dural arteriovenous fistulas (dAVF) are most commonly seen in elderly men, typically involving the thoracic spine, presenting with progressive myelopathic symptoms, which can include disturbance of gait, paresthesia, sensory loss, radicular pain (which may be exacerbated by exercise), and eventually, bowel and bladder dysfunction.2, 4 The abnormal communication between a spinal radiculomeningeal artery and radicular vein causes venous perimedullary arterialization and venous congestion.4 This causes reduced cord perfusion and cord ischemia. Subacute neurologic deterioration due to congestive myelopathy, known as Foix-Alajouanine syndrome, may be seen clinically.8
On imaging, spinal cord enlargement and edema are typically present, with central T2 hyperintensity and “flame-shaped” margins.2, 4, 8 Spinal cord edema may not be present early on and may occur distant to the site of the dAVF, and may progress in the caudocranial orientation. A T2 hypointense rim, which may represent deoxygenated blood within surrounding dilated capillaries, may also be present. An early dAVF often has slow flow, and contrast administration may be needed in order to visualize the venous plexi, which will present as dilated serpentine enhancing perimedullary vessels (Figure 8). Later, the venous plexi may be seen on T2 weighted imaging as prominent, tortuous flow voids. Ill-defined cord enhancement can also be seen in the affected region due to chronic breakdown of the blood-brain barrier. MR angiography and/or selective spinal digital subtraction angiography may also be helpful for evaluation of a dAVF.4, 5, 6 Treatment options include endovascular embolization of the dAVF, open surgical ligation, or a multi-modal approach.
Figure 8: Dural arteriovenous fistula. 65-year-old man presenting with progressive weakness. On sagittal T2-weighted imaging (8a), there is mild cord expansion at the T12 level with flame-shaped T2 hyperintensity (arrow). On sagittal T1-weighted postcontrast imaging (8b), there is corresponding enhancement (long arrow) and adjacent enhancement within perimedullary veins extending upward along the dorsal margin of the cord (bracket).
Subacute combined degeneration is most commonly secondary to vitamin B12 deficiency and can seen in patients with pernicious anemia, post bariatric surgery, and in patients following a strictly vegan diet.2, 4, 9 Copper deficiency and nitric oxide use may also cause subacute combined degeneration of the spinal cord.5, 9 Common presenting symptoms can be insidious and include sensory ataxia, paresthesia in the hands and feet, spasticity, hyperreflexia, and/or history of frequent falls. On laboratory analysis, there is typically macrocytic anemia, elevation of methymalonic acid homocysteine, and low serum vitamin B12 levels.9 On MRI, the classic finding is symmetric T2 hyperintense signal with corresponding hypointense T1 signal in the posterior columns, which may be seen in an inverted “v” configuration, most commonly involving the cervical spine (Figure 9).2, 4, 5, 9 There may be mild cord expansion. Mild enhancement may be present due to breakdown of the blood-brain barrier related to demyelination. Treatment is parenteral vitamin B12 administration, although the imaging findings may not completely resolve following treatment.4
Figure 9: Subacute combined degeneration. 61-year-old man with abnormal gait and hyperreflexia. Sagittal T2-weighted image (9a) demonstrates long segment, nonexpansile, linear T2 hyperintense signal within the dorsal cervical spinal cord from the C3 through the C7 levels (between arrows). On axial imaging, well-defined hyperintense T2 signal is present within the dorsal cord (not in the classic “v” configuration) (arrowhead). There is no associated abnormal enhancement on the sagittal T1 fat-suppressed postcontrast image (9c).
Conclusion
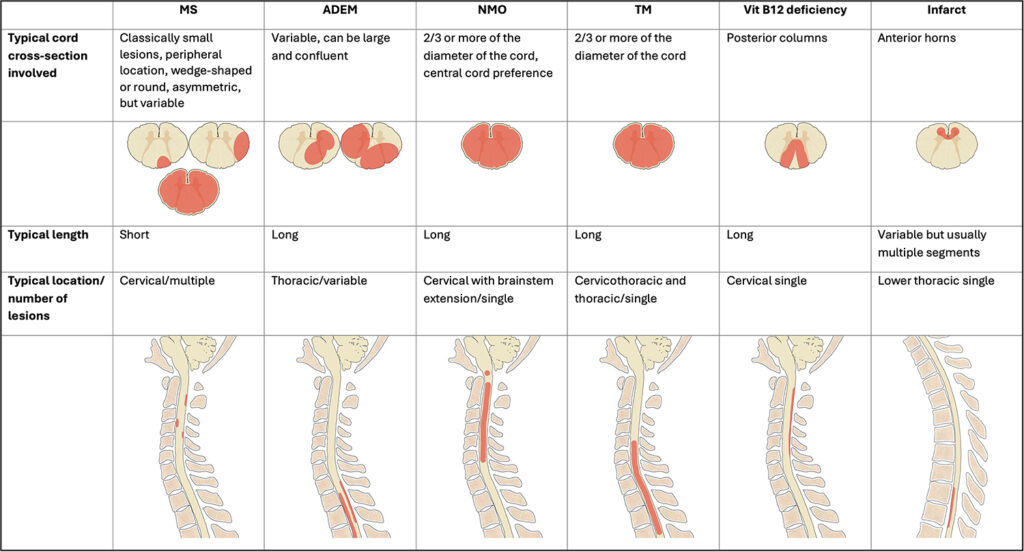
Clinical presentation and imaging findings in demyelinating diseases and non-compressive myelopathy may overlap. However, close evaluation of the length of the cord signal abnormality and the portion of the cord involved (Table 1), and the presence or absence of enhancement and/or cord expansion can help narrow the differential. In addition to clinical presentation and the time of onset (acute versus nonacute), MRI brain imaging and CSF analysis also contribute to identifying the most likely diagnosis. In some cases, follow-up, to evaluate for resolution or change in imaging findings may be required to help confirm a suspected diagnosis.
Table 1
Conclusion
Clinical presentation and imaging findings in demyelinating diseases and non-compressive myelopathy may overlap. However, close evaluation of the length of the cord signal abnormality and the portion of the cord involved (Table 1), and the presence or absence of enhancement and/or cord expansion can help narrow the differential. In addition to clinical presentation and the time of onset (acute versus nonacute), MRI brain imaging and CSF analysis also contribute to identifying the most likely diagnosis. In some cases, follow-up, to evaluate for resolution or change in imaging findings may be required to help confirm a suspected diagnosis.
References
- Love S. Demyelinating diseases. J Clin Pathol 2006, Nove; 59(11): 1151–1159. ↩
- Lee MJ et al. Diagnostic approach to intrinsic abnormality of spinal cord signal intensity. RadioGraphics 2019; 39:1824–1839. ↩
- Marrodan M, Gaitan MI, and Correale J. Spinal cord involvement in MS and other demyelinating diseases. Biomedicines 2020 May 22;8(5):130. ↩
- Laur O et al. Nontraumatic Spinal Cord Compression: MRI Primer for Emergency Department Radiologists. RadioGraphics 2019 39:6, 1862-1880. ↩
- Moghaddam SM and Bhatt AA. Location, length, and enhancement: systematic approach to differentiating intramedullary spinal cord lesions. Insights into Imaging (2018) 9:511–526. ↩
- Tillema JM and Pirko I. Neuroradiological evaluation of demyelinating disease. Ther Adv Neurol Disord 2013 Jul;6(4):249–268. ↩
- Dutra BG et al. Neuromyelitis Optica Spectrum Disorders: Spectrum of MR Imaging Findings and Their Differential Diagnosis. RadioGraphics 2018 38:1, 169-193. ↩
- Barnett Y et al. Conventional and advanced imaging in neuromyelitis optica. AJNR (2014) 35(8): 1458–1466. ↩
- Ravina B, Loevner LA, and Bank W. MR Findings in Subacute Combined Degeneration of the Spinal Cord: A Case of Reversible Cervical Myelopathy. AJR (2000); 174(3):863 – 865. ↩