Clinical History:
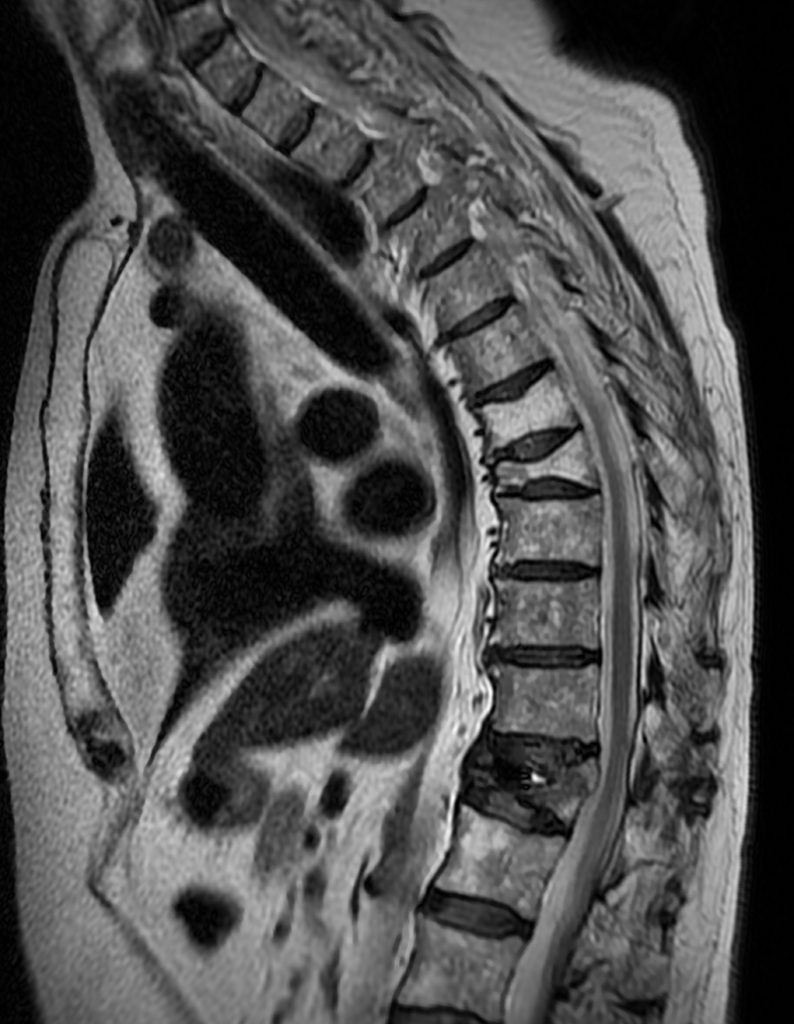
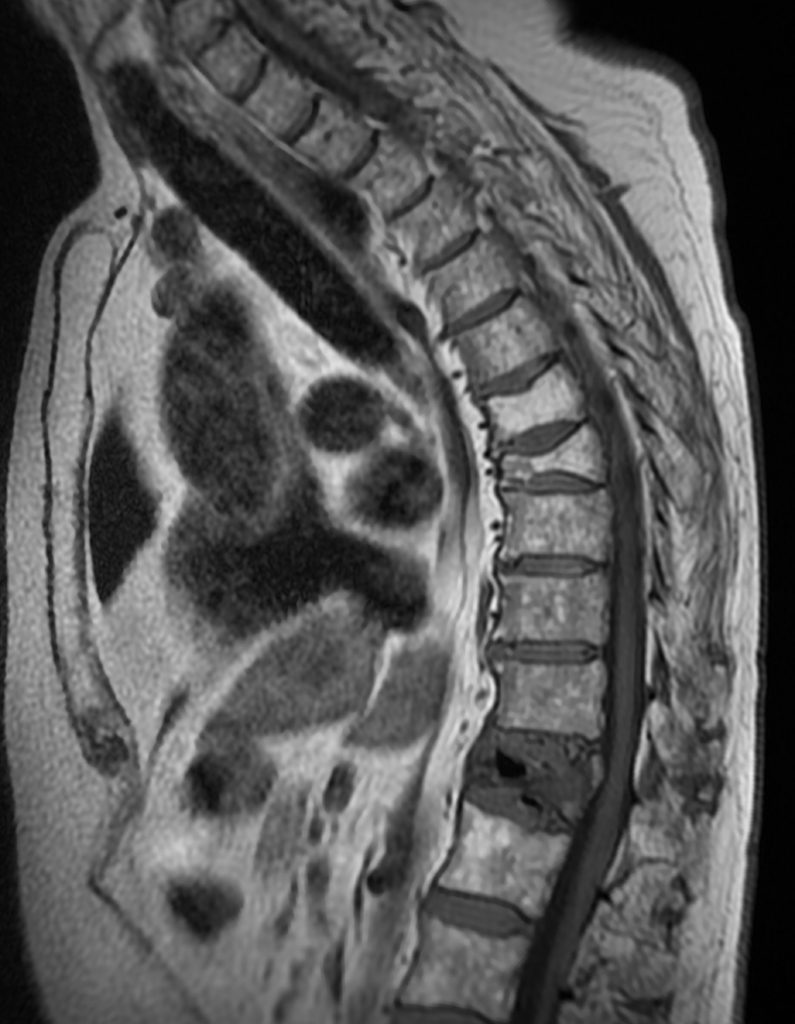
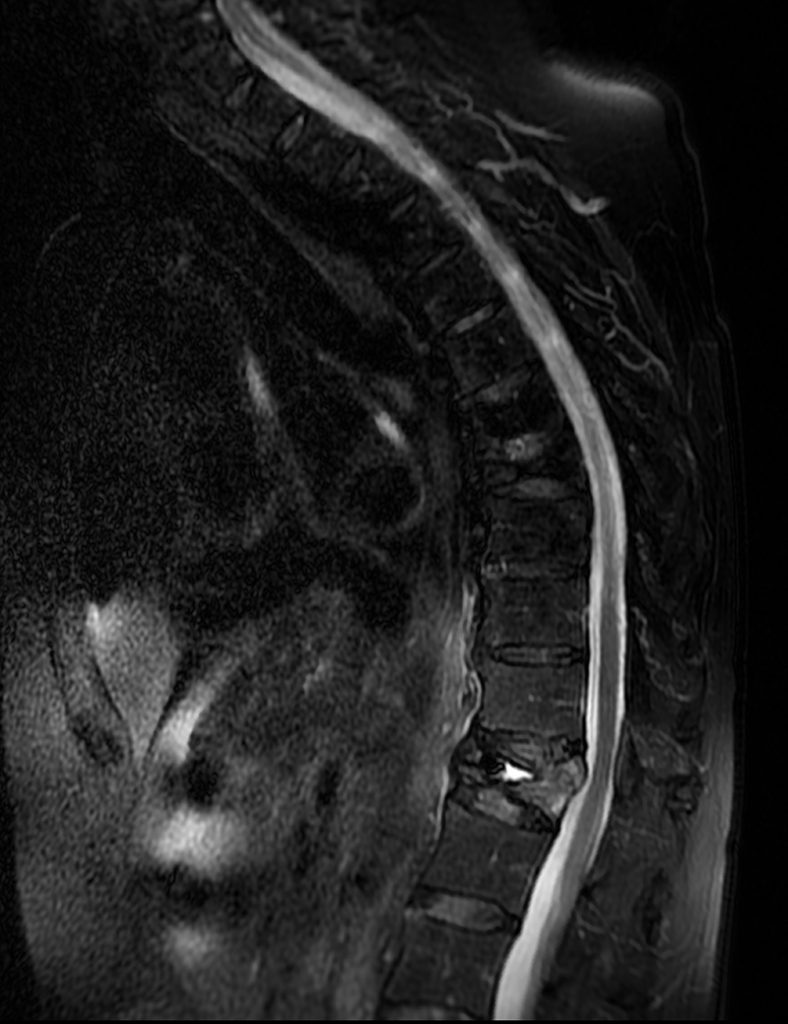
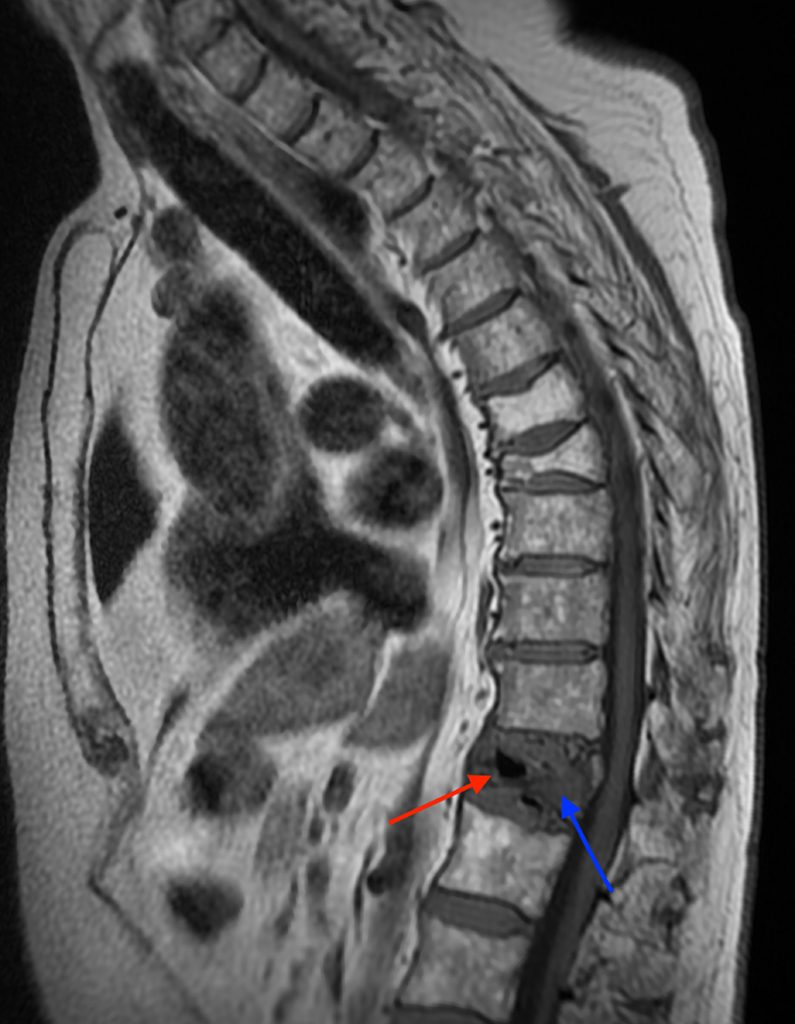
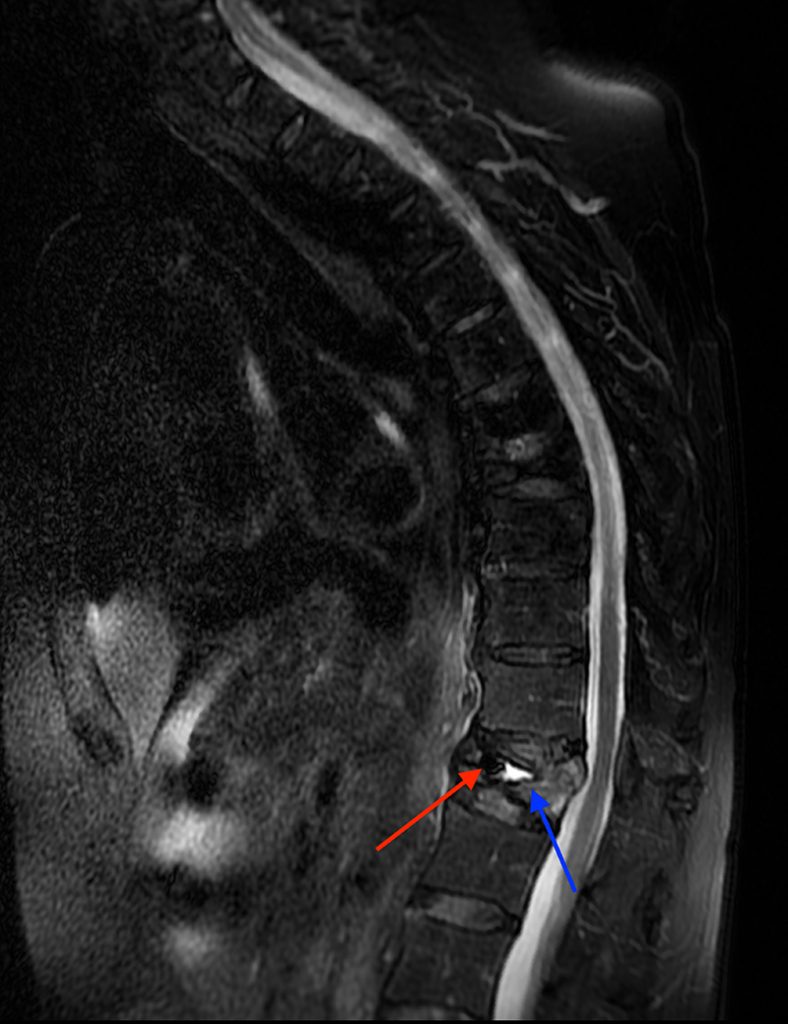
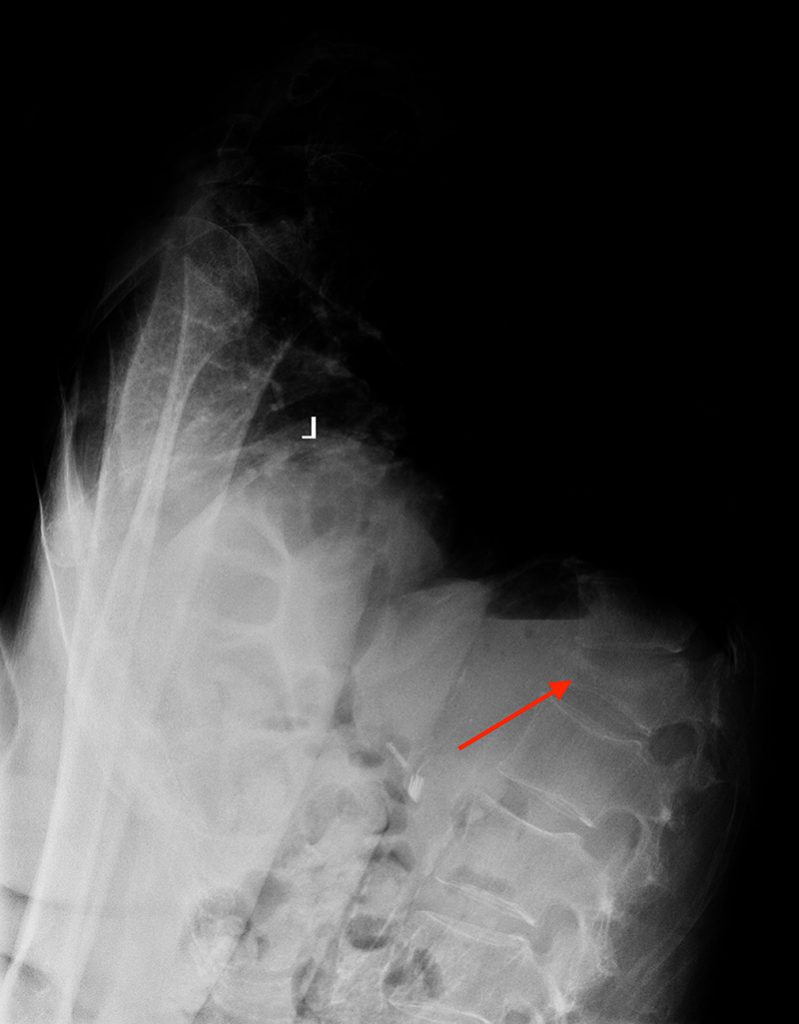
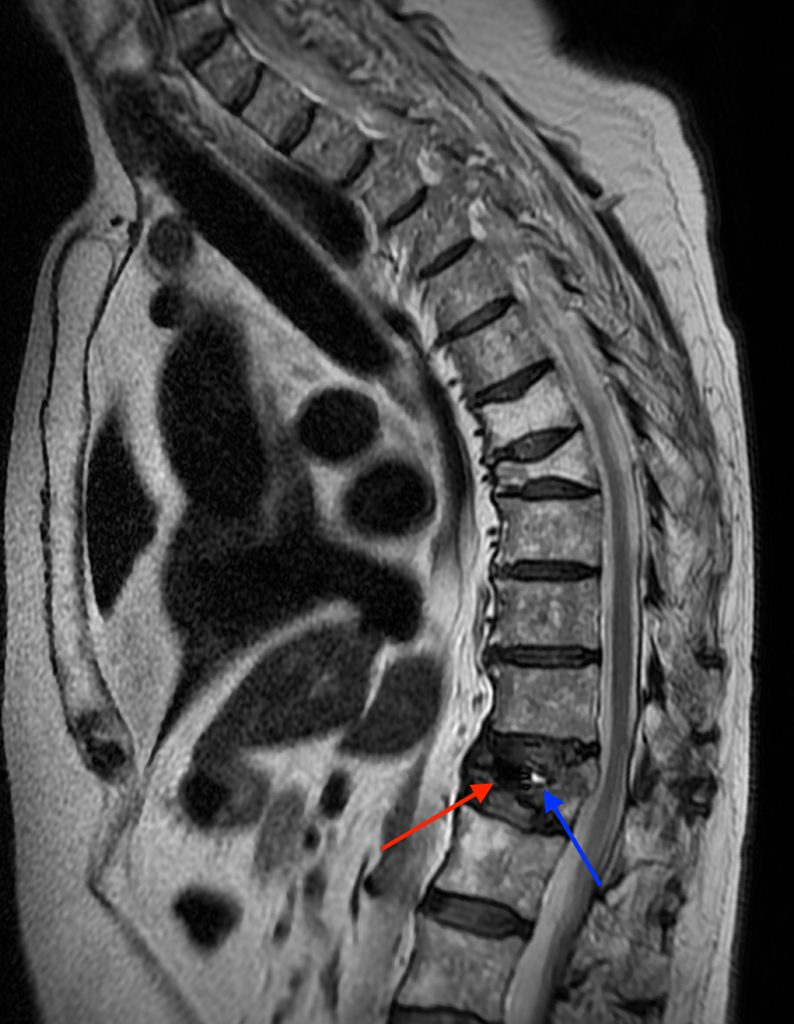
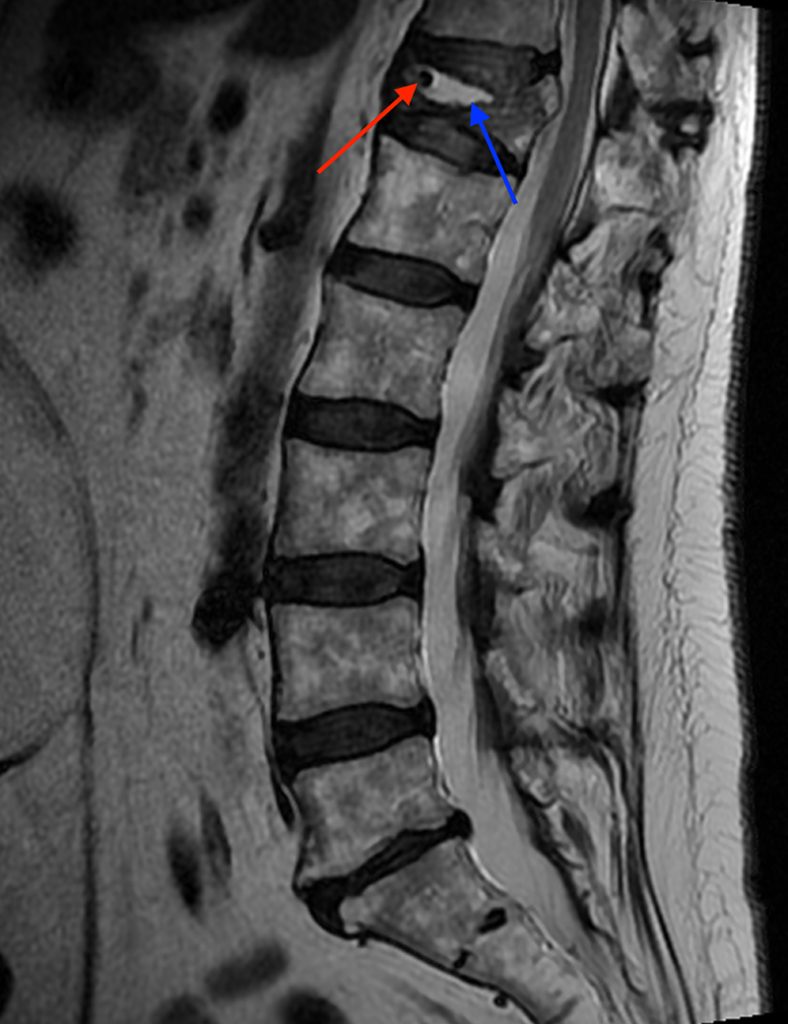
A 78 year-old male presents with low back pain radiating into the mid back for 3 months due to 4 falls. Sagittal T2-weighted (1a), sagittal T1-weighted (1b), sagittal STIR (1c) image and lateral radiograph (1d) obtained around the time of presentation for pain are provided. What are the findings? What is your diagnosis?
Findings:
Figure 2:
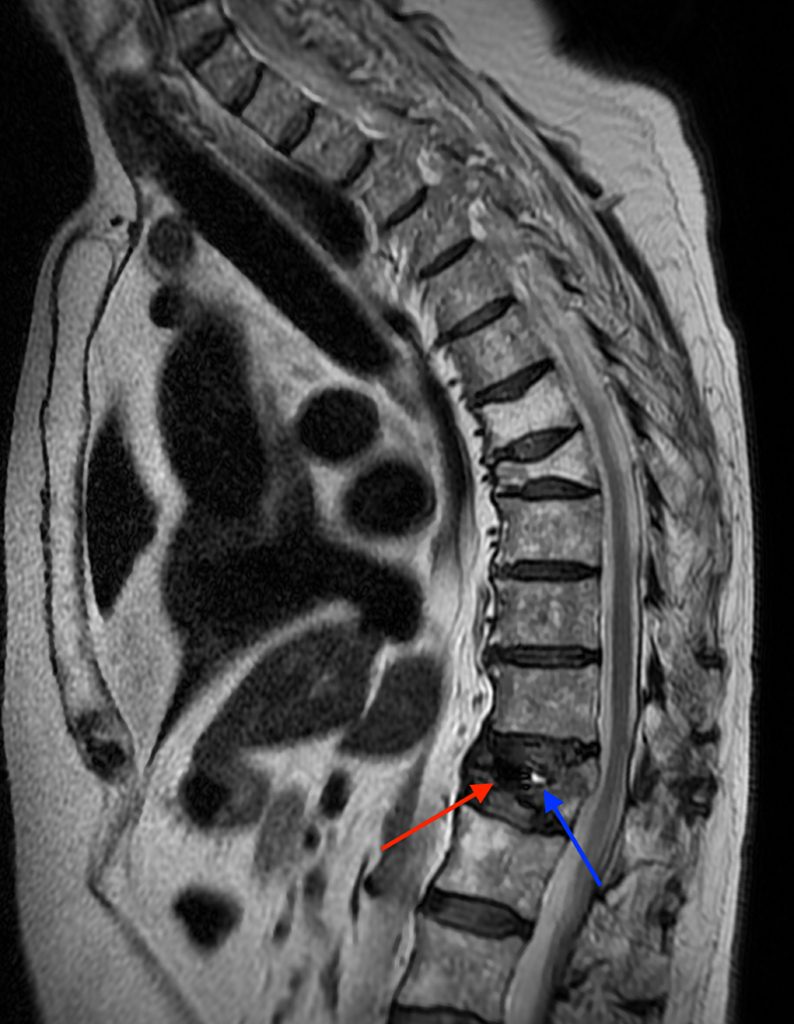
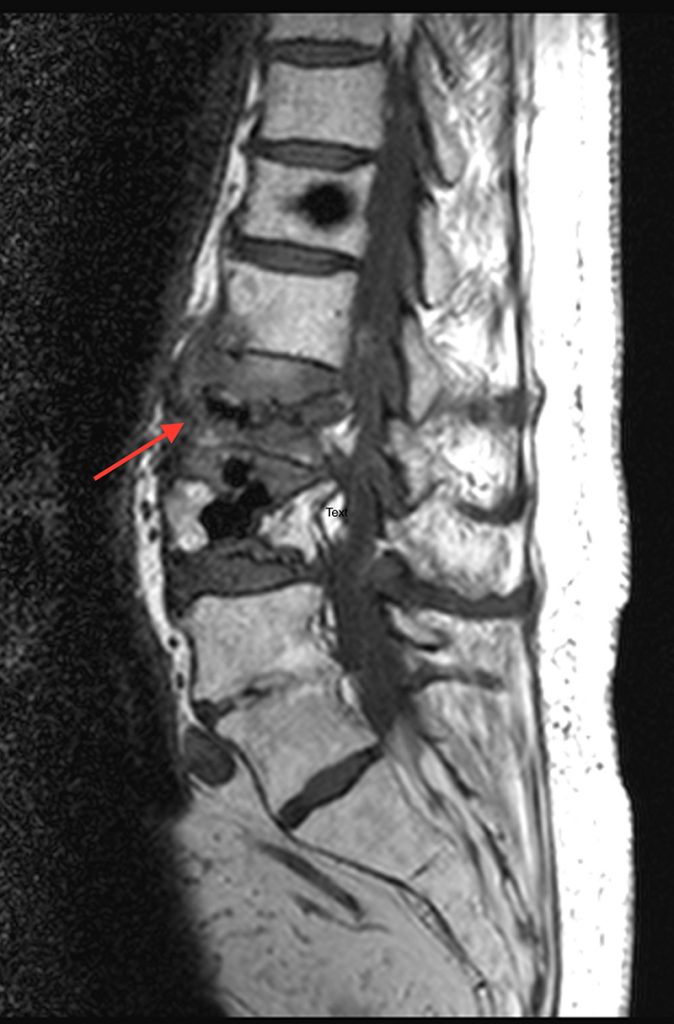
A compression fracture at the thoracolumbar junction is seen with horizontally oriented intravertebral cleft filled predominantly with hypointense air (red arrow) and a small amount of fluid (blue arrow) on sagittal T2-weighted (2a), sagittal T1-weighted (2b), and sagittal STIR (2c) images. A small intravertebral vacuum cleft (red arrow) is seen on the corresponding lateral radiograph (2d).
Diagnosis
Compression fracture with vertebral osteonecrosis and an intravertebral cleft sign.
Introduction
In 1891, Kummell described delayed painful kyphosis in the lower thoracic and upper lumbar regions developing in initially asymptomatic patients weeks to months after minor trauma. With the advent of radiographs, the kyphosis was later found to be secondary to vertebral body collapse. Later authors confirmed the sequential development of vertebral collapse on serial radiographs on which initial radiographs were essentially normal, disproving that the fractures were initially present but missed.1,2,3 Avascular necrosis was hypothesized as the cause for delay between the traumatic event and eventual vertebral body collapse by Schmorl and Junghanns4, who found minute ruptures and hemorrhage in the spongiosa of vertebral bodies at autopsy. Since the acceptance of Kummell’s disease, intravertebral vacuum clefts have been described in the literature as virtually pathognomonic of avascular necrosis.5,6,7 This has led to equating intravertebral clefts with Kummell disease although there are a limited number of reported cases in the literature which meet Kummell’s original criteria of delayed vertebral body collapse without other predisposing factors for ischemic necrosis except trauma. The intravertebral vacuum cleft was first described related to avascular necrosis with histological specimens from biopsy suggesting a reparative process following bone ischemia.5 This was confirmed with a later study documenting cleft sign sensitivity and specificity of 85% and 99%, respectively.8 Further studies with the advent of MRI, demonstrated the presence of fluid signal within these intravertebral vacuum clefts on MRI,9 changes in content of fluid and air with positioning,10 and confirmed histologic features of avascular necrosis in vertebral fluid collections associated with collapse.11
Pathogenesis and Imaging Features
There are 4 concentric zones of osteonecrosis: a central zone of cell death, zone of ischemic injury, zone of active hyperemia, and peripheral zone of normal tissue 12 In the early phase of osteonecrosis, inflammatory cells and fluid are the major components in the area of injury and hyperemia, while new bone and fibrous tissue predominate in the later phases. The variability in appearance on MRI can be attributed to the different phases of osteonecrosis at imaging.
The pathogenesis of osteonecrosis in vertebral collapse is thought to be two-fold, with traumatic and non-traumatic mechanisms13,14 The first mechanism is that of delayed collapse due to avascular necrosis, or Kummell disease3,4, related to major trauma with trauma-induced osteonecrosis and collapse in the presence of predisposing factors such as arteriosclerosis.15 The second mechanism entails fractures arising in weakened bone due to osteoporosis, replaced marrow by abnormal cells, or long-term glucocorticoid use in conjunction with minor trauma. Osteonecrosis develops at the site of fractured endplates because of strong compression of the trabecular network in that area. This explains the majority of horizontally oriented clefts seen paralleling the endplate (Figures 3 and 4).
Figure 4:
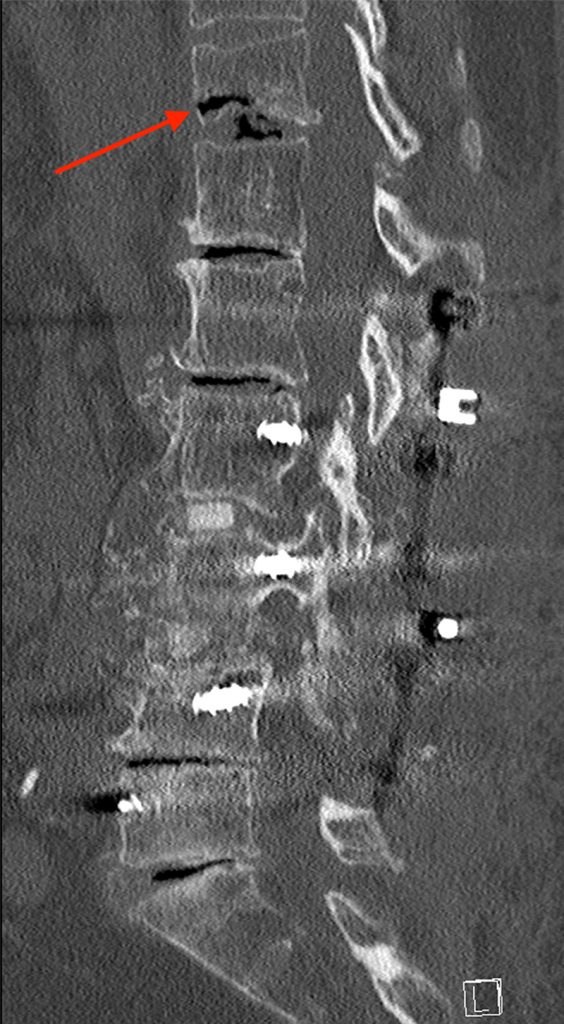
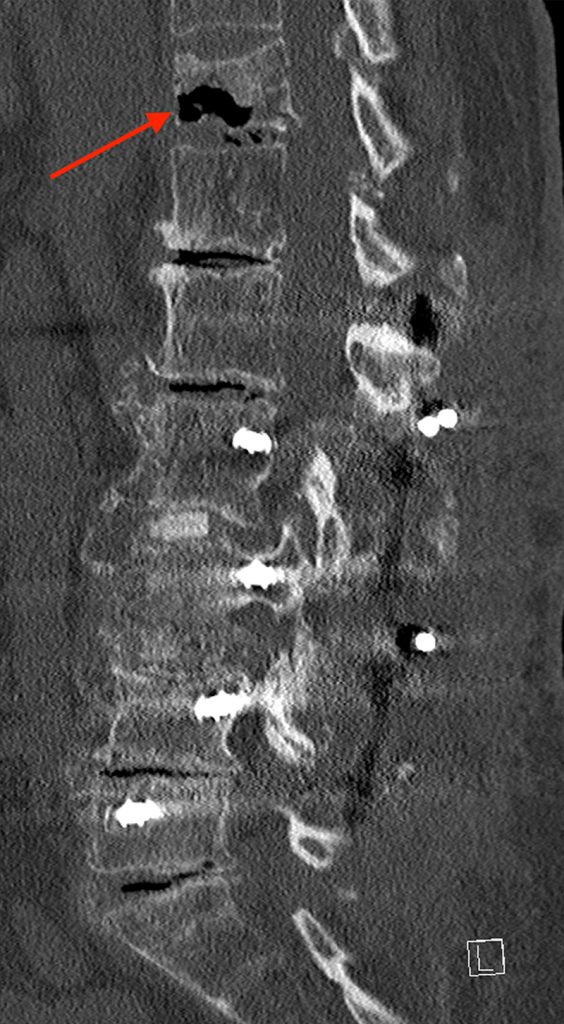
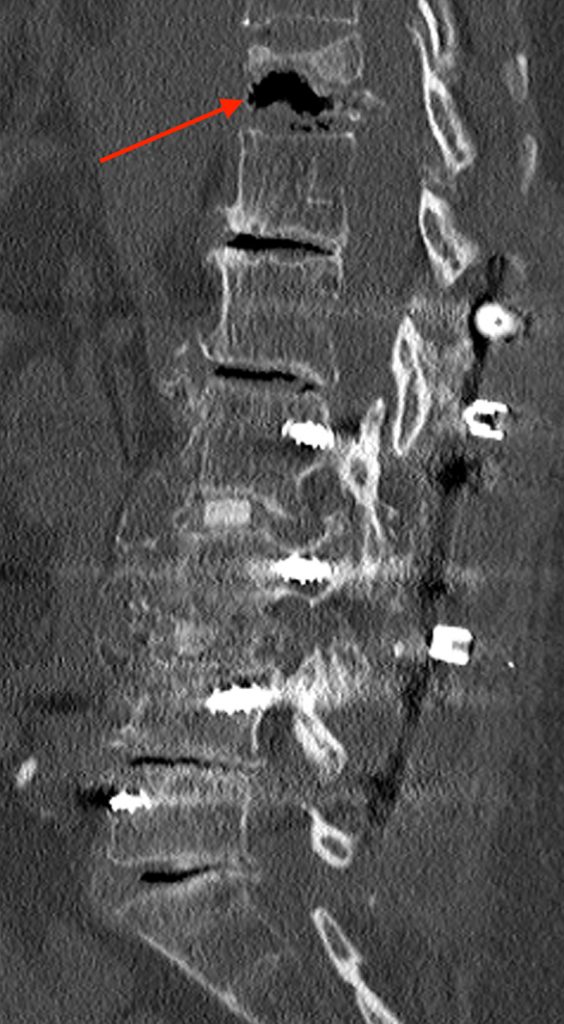
Sagittal CT images obtained at initial presentation (4a), at 2.5 months (4b), and at 5.5 months (4c) demonstrating horizontally oriented intravertebral air-filled clefts (arrows) paralleling an inferior endplate compression fracture. Note, the interval increase in size of the cleft and increased collapse with time.
Vertebral collapse most commonly occurs as a consequence of vascular insult to the anterior segment of the vertebral body which is supplied by anterior metaphyseal and peripheral arteries 16. One explanation to delayed collapse has been proposed by Benedek et al.17, that the initial trauma causes small trabecular fractures with an impaired healing process due to intrinsic (insufficient arterial supply or herniated Schmorl’s nodes) or extrinsic (normal weight-bearing on weakened bone) interference. Failure of the reparative process predisposes to ischemic necrosis and may result in collapse with progressive collapse putting the arterial supply at further risk in a vicious cycle. It has also been proposed that the anterior third of the vertebral body is a “watershed” zone with the dorsum receiving collateral supply15, and thus vulnerable to ischemic necrosis. However, there is continued debate as to whether ischemic necrosis precedes or follows vertebral collapse.
The collapse of bone with ischemic necrosis results in a decrease in bone volume. Generated low pressure allows accumulation of gas, mostly nitrogen, in the fracture line of the vertebral body.18 The vacuum cleft as a result of ischemic bone necrosis has been supported by biopsy specimens and is analogous to the radiolucent line of AVN of the hip.5,6,9 Gas within a cleft has been shown to change with position and time. The release of gas in distracted cracks of subchondral bone has been shown to appear during extension and disappear during kyphotic positioning.5 Subsequently, the first report on the MRI appearance in cases of intravertebral vacuum clefts demonstrated fluid signal.9 Although this seemed inconsistent with vacuum phenomena on radiographs at the time, this has since been explained by replacement of the fluid-like content with prolonged supine positioning.10 Clefts were best seen on extension stress views and radiographs obtained after prolonged supine positioning demonstrated a decrease or disappearance of the radiolucency. This correlated with increased T2 signal intensity seen with later scans and an increased duration of recumbency on MRI (Fig 5).
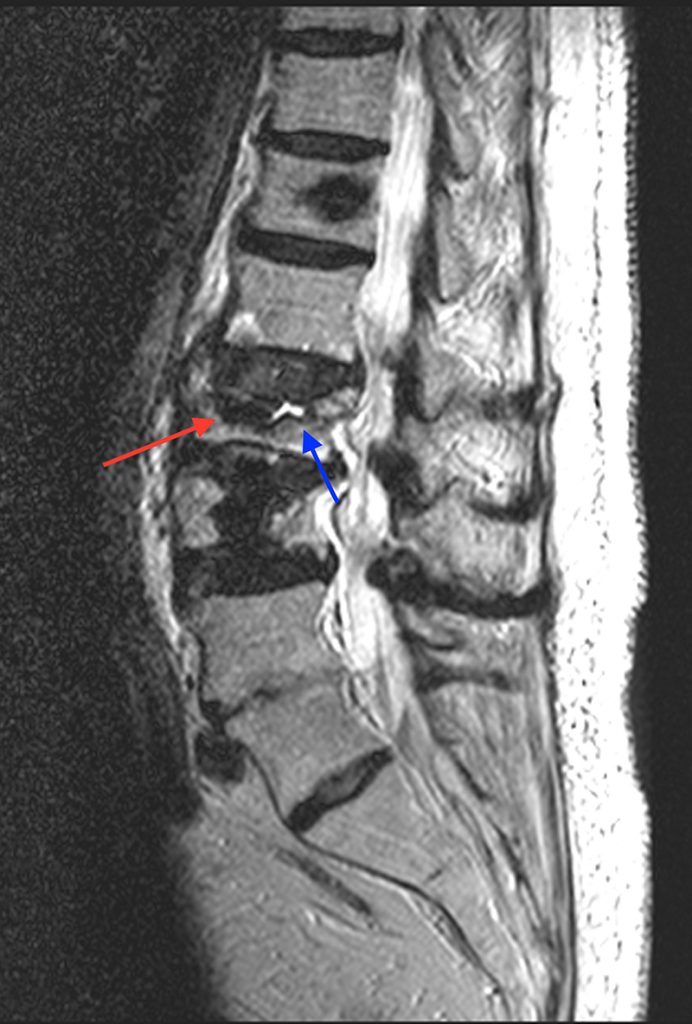
Figure 5:
Sagittal T2-weighted image of the thoracic spine (5a) from the original case and sagittal T2-weighted image of the lumbar spine (5b) obtained as part of a double study 25 minutes after the T2-weighted sequence of the thoracic spine. Note the significant increase in fluid (blue arrow) and small focus of remaining air (red arrow) within the cleft at the T12 fracture site.
The intravertebral fluid collections associated with collapse have been biopsied and have demonstrated features of avascular necrosis in patients without predisposing factors except for the presence of osteopenia.11 This had led to the thought that osteoporotic compression fractures may commonly lead to avascular necrosis and fluid accumulation.
More recent papers have demonstrated a higher incidence of clefts in osteoporotic compression fractures with a frequency of clefts at the thoracolumbar junction.19,20 Flexion and extension is most dynamic at the thoracolumbar junction and vertebral insufficiency fractures are less likely to heal compared to other levels that are less mobile. In fact, distraction of fracture fragments along the plane of the cleft with respiratory motion has been noted on fluoroscopy during vertebral augmentation.19 Several authors have proposed that motion along the compression fracture plane could prevent union of fragments and propose that clefts represent fracture non-unions.19,20,21,22 It is possible that a combination of pseudoarthrosis and ischemia could explain the existence of clefts as ischemia of a weakened osteoporotic bone could lead to fracture and the insufficient blood supply could lead to pseudoarthrosis.
The presence of an intravertebral cleft filled with air or fluid is not exclusive to vertebral osteonecrosis, however. There has been a statistically significant difference found between benign and malignant fractures demonstrating a fluid cleft sign but, rarely, the fluid-filled cleft can be seen in pathologic fractures and thus though they support a benign osteoporotic fracture they cannot definitively exclude tumor.13 A few authors have described intravertebral vacuum clefts occurring in compression fractures due to primary or secondary tumor.6,21 In rare instances, gas can be seen in the vertebral body with osteomyelitis due to gas-forming organisms6 but the pattern and distribution of gas differs with small radiolucent collections more typical as the gas is generated under high pressure.18
Treatment and Management
Management of vertebral collapse with an intravertebral cleft should include further work-up if there are additional findings or features suspicious for other causes of fracture besides trauma and osteoporosis. Treatment considerations are similar to compression fractures and should consider severity of disability and pain, comorbidities, the degree of kyphotic deformity, and neurologic deficits. In patients with collapse and back pain without neurologic impairment, minimally invasive procedures (vertebroplasty and kyphoplasty) can be effective for pain relief, decreasing motion at the fracture site, restoration of alignment, and return to mobility. Yu et al.14 found statistically significant differences in the severity of vertebral collapse in those clefts with air only as opposed to those with fluid or without air and more frequent fractures of adjacent vertebrae in those with clefts demonstrating only intravertebral air (Figure 4). They proposed that fluid could represent an earlier stage and air could represent a more advanced stage. Fluid should, therefore, be considered indicative of cavity formation or a gap between viable bones that would not spontaneously heal and aggressive treatment should be performed to prevent further collapse of affected vertebral bodies and adjacent levels. Filling the cleft would be the primary objective and is supported by the fact that patients in whom the clefts did not fill experienced return to preoperative baseline pain at 6-12 months.19 Pain relief in patients with vertebral fractures with and without clefts have been similar 23, 24 Reduction of kyphotic deformity and complications of cement extrusion, though, have yielded mixed results.
For those patients with neurologic impairment and canal compromise, decompression with instrumentation and fusion should be performed.25 The concern of decompression alone is the progression of the kyphotic deformity. Decompression can be performed anteriorly or posteriorly with greater ease of removing retropulsed fragment anteriorly. In older patients with comorbidities, however, a posterior approach is preferred to avoid complications from violating the thoracic cavity or retroperitoneal space. Manual reduction of fractures along with transpedicular insertion of vertebral body augments combined with short segment fixation has been described to treat Kummell disease with cord compromise.26
Conclusion
Avascular necrosis has been hypothesized to explain Kummell’s disease, the development of delayed vertebral collapse after trauma. Radiographs led to the discovery of intravertebral vacuum clefts in vertebral collapse after initial normal radiographs. Later, MRI demonstrated that intravertebral clefts can contain gas, fluid, or both gas and fluid. Clefts can fill with fluid with prolonged supine positioning and extension of the spine can aid in filling with gas. The presence of air only within clefts on MRI is associated with more severe vertebral collapse and more frequent fractures of adjacent vertebrae. Biopsies of these clefts have demonstrated features of avascular necrosis while the frequency of clefts at the thoracolumbar junction where motion is more dynamic has led to the proposition that the clefts could represent fracture non-unions. Although intravertebral clefts have been found in Kummell disease, they are not exclusive to Kummell disease. In fact, osteonecrosis and the development of intravertebral clefts develop in bone weakened by osteoporosis, marrow infiltration, or glucocorticoid use along with minor trauma. Intravertebral clefts are highly suggestive of osteonecrosis but do not exclude malignancy. Although uncommon, intravertebral vacuum clefts and fluid-filled clefts have been seen in vertebral collapse associated with malignancy and rarely with infection. Distinguishing these entities requires evaluation typical with any vertebral fracture. Management should include further work-up in those patients where the MRI findings suggest pathologic fracture. Treatment considerations for clefts are similar to compression fractures, ranging from conservative management, minimally invasive treatment with vertebroplasty/kyphoplasty, to surgical decompression and fusion.
References
- Steel HH. Kümmell’s disease. Am J Surg 1951; 81: 161-7. ↩
- Rigler L. Kummell’s disease with report of a roentgenologically proved case. Am J Roentgenol 1931; 35: 749-53. ↩
- Brower AC, Downey EF Jr. Kümmell disease: report of a case with serial radiographs. Radiology 1981; 141: 363-4. ↩
- Schmorl G, Junghanns H. The human spine in health and disease. 2nd New York: Grunne & Stratton 1971: 141. ↩
- Maldague BE, Noel HM, Malghem JJ. The intravertebral vacuum cleft: a sign of ischemic vertebral collapse. Radiology. 1978; 129: 23-9. ↩
- Resnick D, Niwayama G, Guera J, Vint V, Usselman J. Spinal vacuum phenomenon: anatomical study and review. Radiology 1981; 137:341– 348. ↩
- Bielecki DK, Sartoris D, Resnick D, et al. Intraosseous and intradiscal gas in association with spinal infection: report of three cases. AJR Am J Roentgenol 1986; 147: 83-6. ↩
- Libicher M, Appelt A, Berger I, et al. The intravertebral vacuum phenomenon as specific sign of osteonecrosis in vertebral compression fractures: results from a radiological and histological study. Eur Radiol 2007; 17: 2248-52. ↩
- Naul LG, Peet GJ, Maupin WB. Avascular necrosis of the vertebral body: MR imaging. Radiology 1989; 172: 219-22. ↩
- Malghem J, Maldague B, Labaisse MA, et al. Intravertebral vacuum cleft: changes in content after supine positioning. Radiology 1993; 187: 483-7. ↩
- Dupuy DE, Palmer WE, Rosenthal DI. Vertebral fluid collection associated with vertebral collapse. AJR Am J Roentgenol 1996; 167: 1535-8 ↩
- Sweet DE, Madewell JE. Pathogenesis of osteonecrosis. In: Resnick DK, Niwayama E, eds. Diagnosis of bone and joint disorders. Philadelphia: Saunders 1981; 2780-831. ↩
- Baur A,Stabler A,Arbogast S,etal. Acute osteoporotic and neoplastic vertebral compression fractures: fluid sign at MR imaging. Radiology 2002; 225:730 –35. ↩
- Yu CW, Hsu CY, Shih TT, et al. AJNR Am J Neuroradiol. 2007 Jan;28(1):42-7. ↩
- Stojanovic J, Kovac V. Diagnosis of ischemic vertebral collapse using selective spinal angiography. Rofo 1981; 135: 326-9 ↩
- Ratcliffe J. The arterial anatomy of the adult human lumbar vertebral body: a microarteriographic study. J Anat 1980; 131:57–79 ↩
- Benedek TG, Nicholas JJ. Delayed traumatic vertebral body compression fracture. II. Pathologic features. Semin Arthritis Rheum 1981; 10: 271-7. ↩
- Theodorou DJ. The intravertebral vacuum cleft sign. Radiology 2001; 221: 787-8 ↩
- Lane JI, Maus TP, Wald JT, et al. Intravertebral clefts opacified during vertebroplasty: pathogenesis, technical implications, and prognostic significance. AJNR Am J Neuroradiol 2002; 23: 1642-6. ↩
- Mirovsky Y, Anekstein Y, Shalmon E, et al. AJNR Am J Neuroradiol. 2005 Aug;26(7):1634-40. ↩
- Kumpan W, Salomonowitz E, Seidi G, Wittich GR. The intravertebral vacuum phenomenon. Skeletal Radiol 1986; 15: 444-7. ↩
- Shih TT, Tsuang YH, Huang KM, Chen PQ, Su CT. Magnetic resonance imaging of vertebral compression fractures. J Formos Med Assoc 1996;95:313–319 ↩
- Ha KY, Lee JS, Kim KW, Chon JS. Percutaneous vertebroplasty for vertebral compression fractures with and without intravertebral clefts. J Bone Joint Surg Br 2006; 88: 629-33. ↩
- Krauss M, Hirschfelder H, Tomandl B, et al. Kyphosis reduction and the rate of cement leaks after vertebroplasty of intravertebral clefts. Eur Radiol 2006; 16: 1015-21. ↩
- Kim KT, Suk KS, Kim JM, Lee SH. Delayed vertebral collapse with neurological deficits secondary to osteoporosis. Int Orthop 2003; 27: 65-9. ↩
- Li KC, Li AF, Hsieh CH, et al. Another option to treat Kümmell’s (61) disease with cord compression. Eur Spine J 2007; 16: 1479-87 ↩