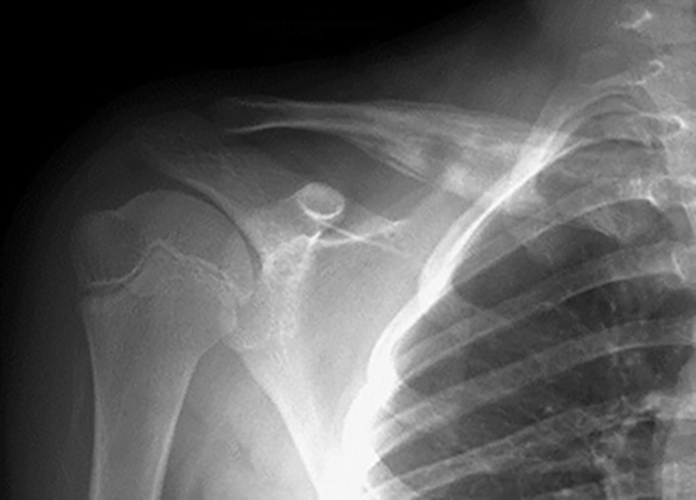
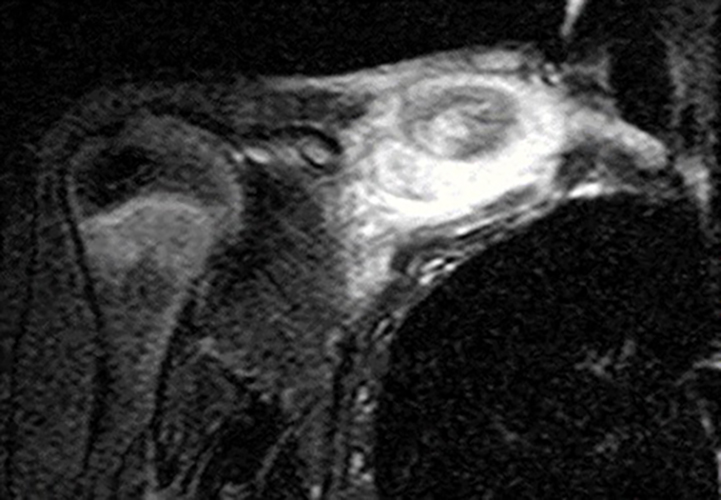
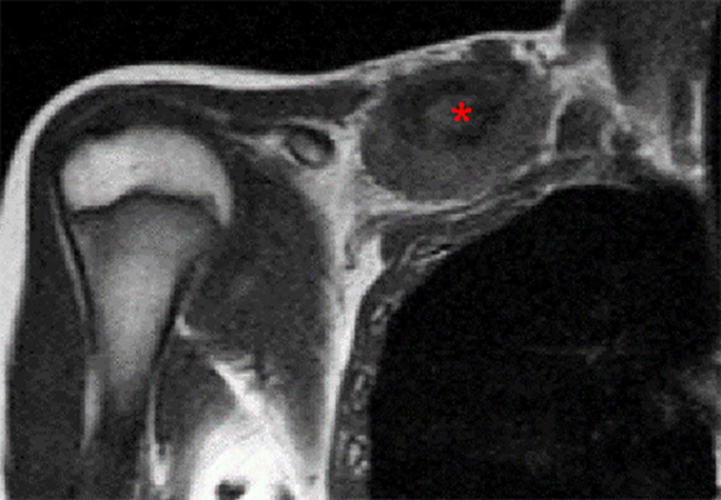
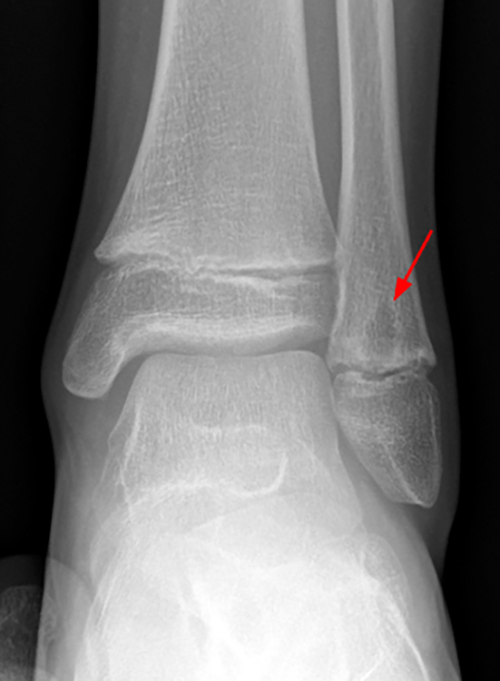
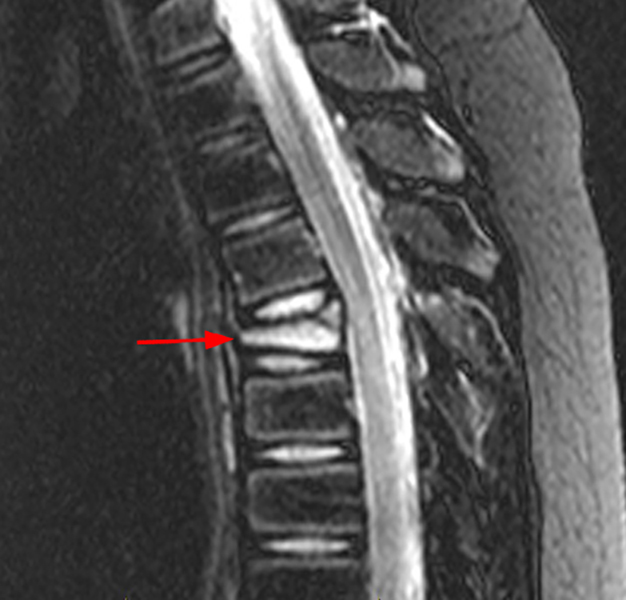
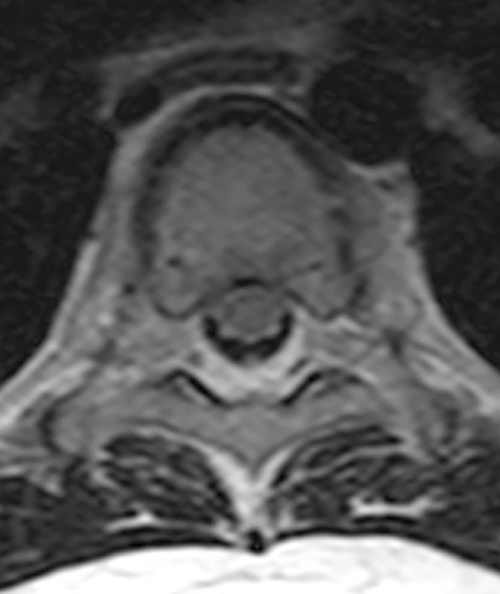
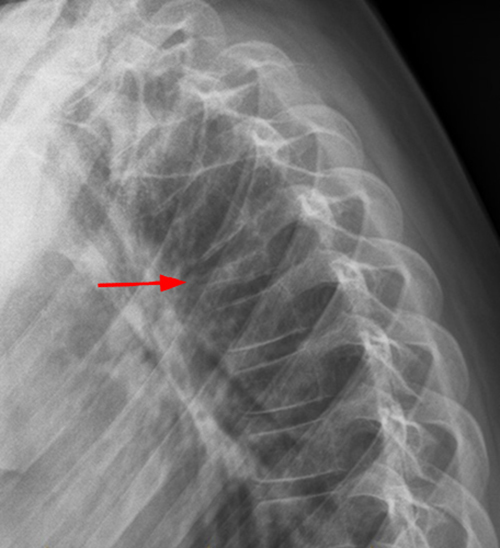
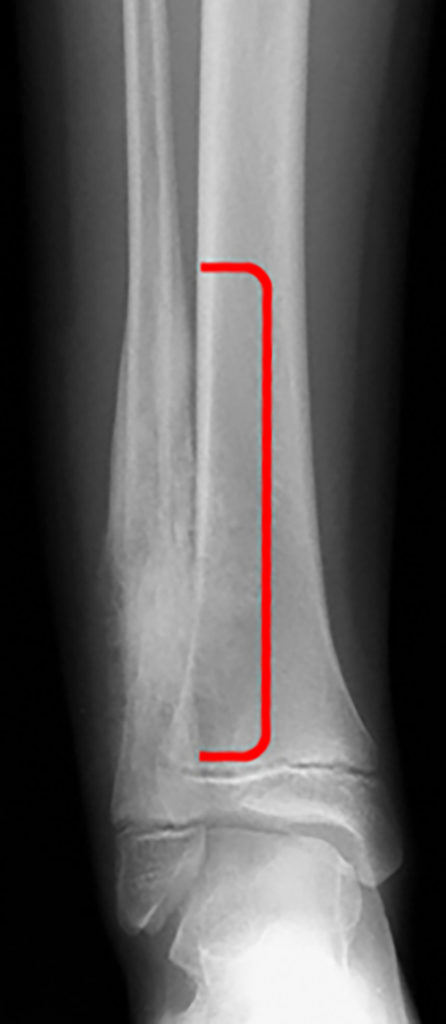
Clinical History: A 10 year-old female presents with a several-week history of atraumatic pain and swelling in the right clavicular region. A frontal radiograph with 15-degree cephalic angulation (1a) and a fat-suppressed coronal proton density-weighted image (1b) are provided. What are the findings? What are your diagnostic considerations?
Findings
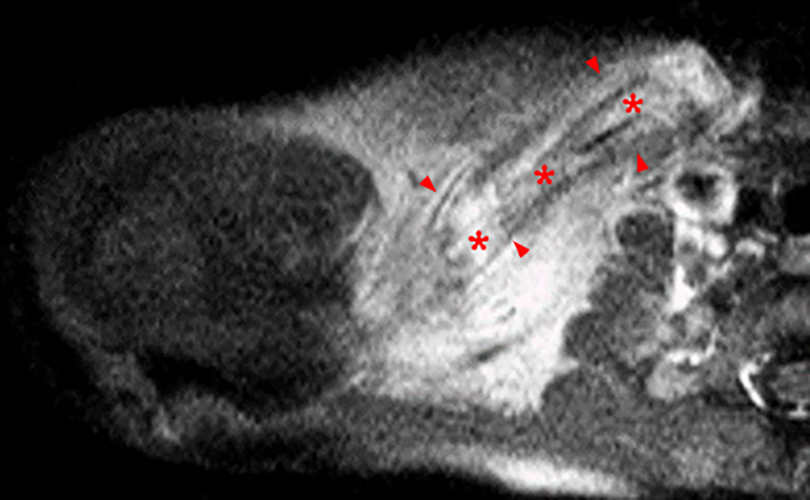
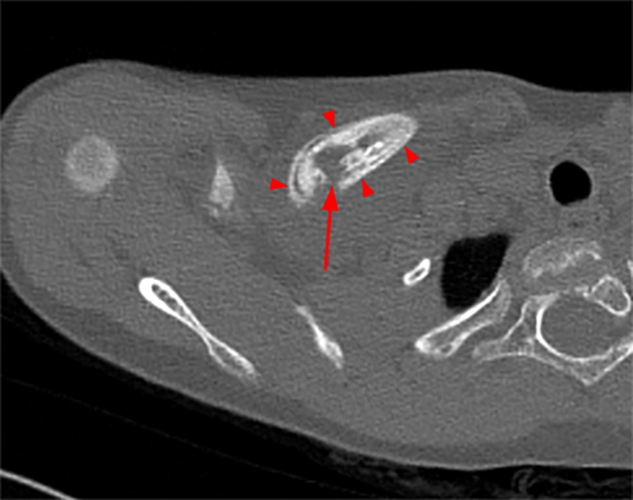
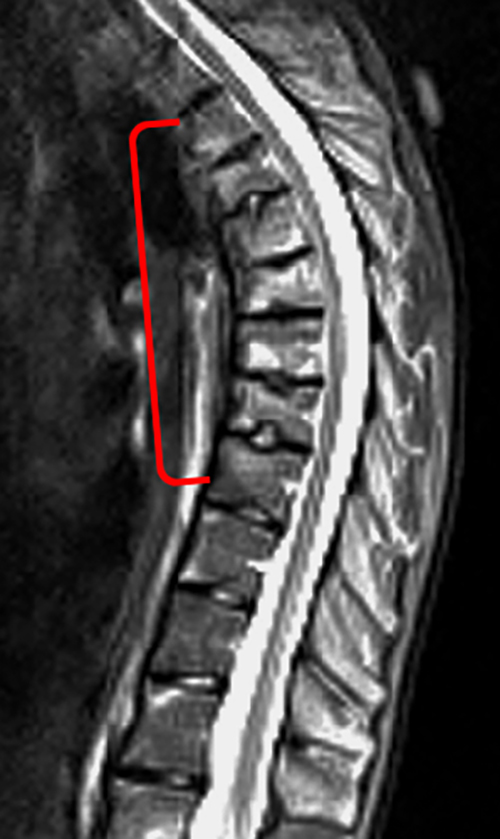
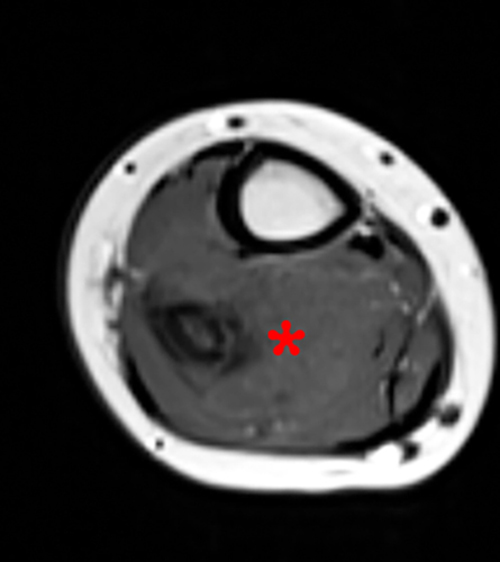
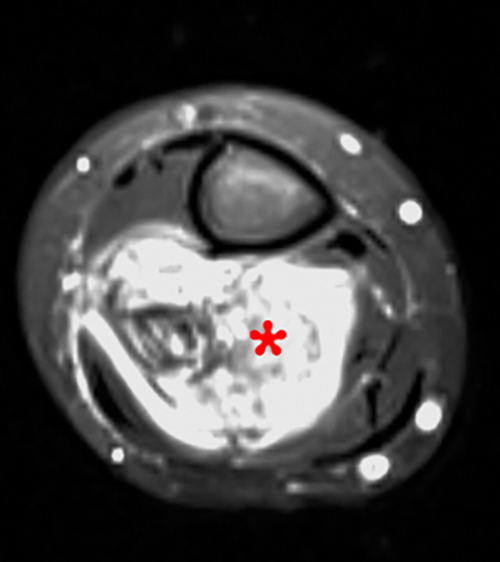
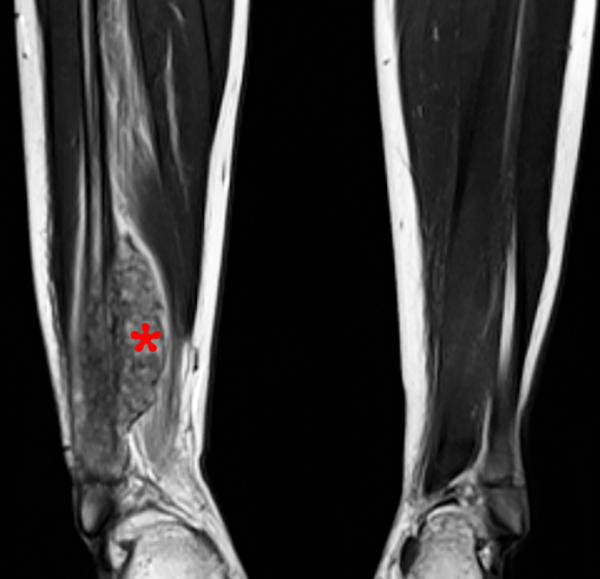
Figure 2:
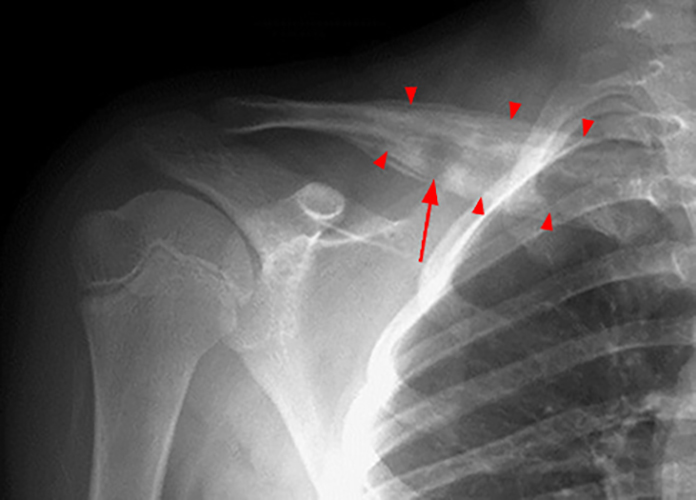
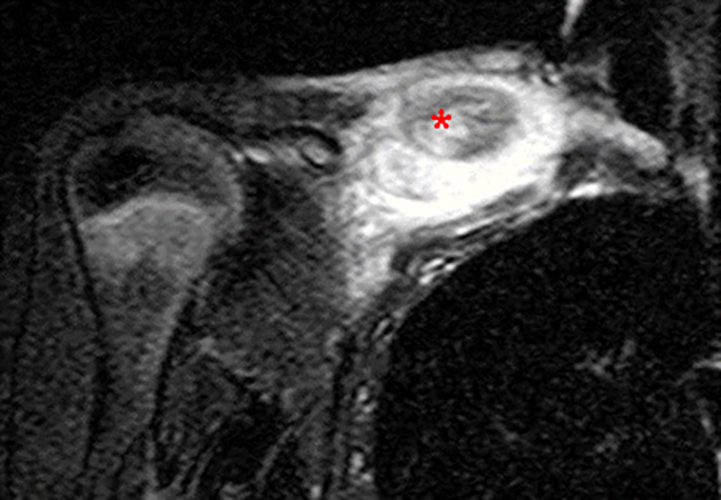
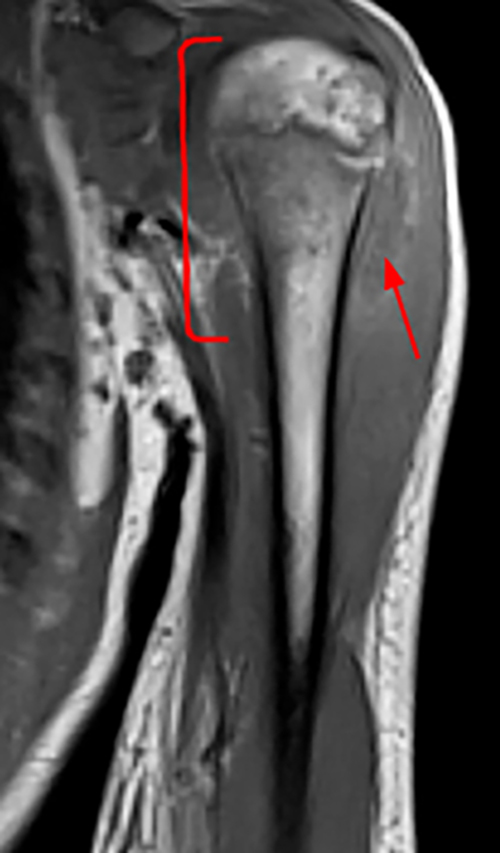
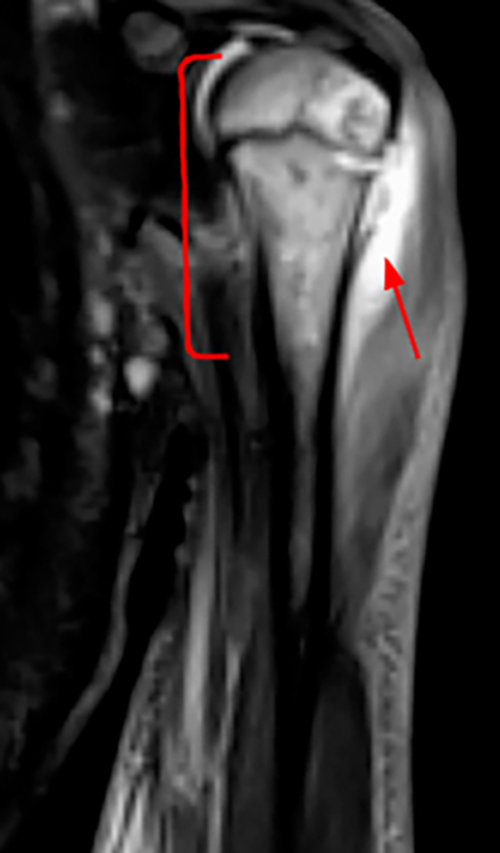
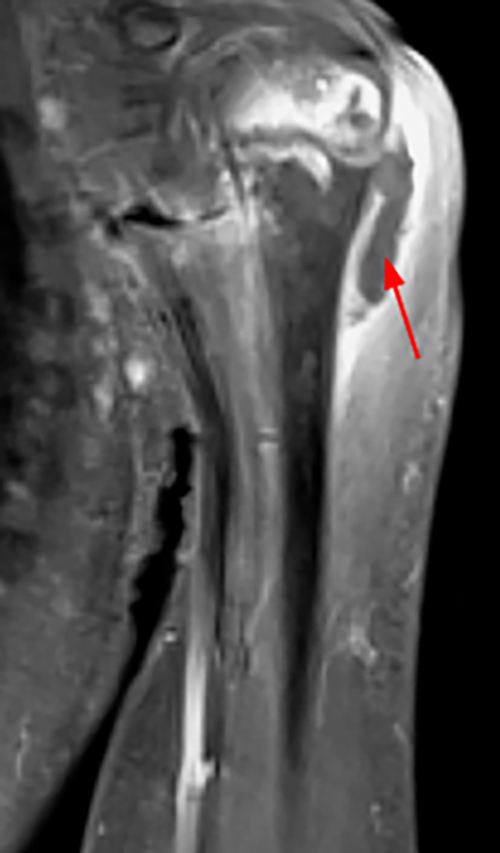
Figure 2. The right clavicular radiograph (2a) and axial CT image (2e) show cortical thickening and periosteal reaction (arrowheads) of the medial half of the clavicle with a small lytic lesion of the mid diaphysis (arrows). Coronal fat-suppressed proton density-weighted image (2b), coronal T1-weighted image (2c), and axial fat-suppressed T1-weighted image (2d) with intravenous gadolinium show abnormal intermediate marrow signal on the unenhanced T1-weighted image (asterisk), marked abnormal marrow signal hyperintensity on the T2-weighted images (asterisk) with associated periosteal reaction and surrounding soft tissue swelling. Diffuse marrow (asterisks), periosteal (arrowheads), and soft tissue enhancement are seen on the enhanced T1-weighted image.
Diagnosis
Proven Chronic Recurrent Multifocal Osteomyelitis (CRMO). Leading differential diagnostic considerations would include CRMO versus neoplasm (e.g., Ewing’s sarcoma), versus less likely infectious osteomyelitis. Notably, in contrast to CRMO which is the most common nonneoplastic process involving the clavicle in patients younger than 55 years of age, hematogenous osteomyelitis rarely involves the clavicle.1,2
Introduction
CRMO is an uncommon idiopathic musculoskeletal disorder of pediatric and adolescent individuals who develop intermittent bone pain and multifocal sites of sterile osteitis. Though the diagnosis is one of exclusion often requiring biopsy, awareness of characteristic clinical and imaging features is essential to avoid the possibility of delayed diagnosis, unnecessary biopsies, prolonged antibiotic therapy and to educate the patient and parents regarding what is a benign but potentially recurrent protracted disease.1
Overview and Clinical Features
The etiology of CRMO is not entirely clear, though recent literature postulates an autoinflammatory disorder, with genetic susceptibility. The consistent lack of identifiable causative microorganisms, failure of the disease to respond to targeted antibiotic therapy, therapeutic response to NSAIDs and steroids, and association with conditions such as psoriasis, inflammatory bowel disease, peripheral arthritis, and sacroiliitis are some disease features that have led to the hypothesis of an autoimmune etiology.1 These comorbidities have been reported in first or second-degree family members in up to 50% of patients with CRMO.3 CRMO has been identified in monozygotic twins and siblings, is a component of several rare hereditary disorders including Majeed, PAPA, and DIRA syndromes, and a CRMO-like autoinflammatory condition has been found to be linked to an autosomal recessive defect in the murine pstpip2 gene. 1,4,5,6 The syndrome was first described in a series of four children in 1972 by Giedion et al.7 Since that time there have been numerous case series and clinical studies describing the disorder, some using slightly different nomenclature. Given the inconsistent terminology, limited clinical awareness, and nonspecific findings it is suspected that the incidence of CRMO has been underestimated, though it has been reported as ranging from 0.4-10.0/100,000.1,5,8,9 CRMO usually occurs in children and adolescents with an average age of onset of 7-14 years of age and a female to male ratio of approximately 2:1.1,10 Most patients present with the insidious onset of bone pain, tenderness, with or without swelling, decreased range of motion, fever, and malaise. Though the majority of patients present with a single symptomatic site, additional sites of involvement become evident in up to 70-98% of cases with whole-body screening or continued clinical follow-up.1,8,11,12 While a course of waxing and waning symptoms with multifocal often symmetric bilateral disease is most common, a small percentage of patients have truly unifocal disease with identical histologic, imaging, and clinical features termed chronic nonbacterial osteomyelitis (CNO).1,13 Initial laboratory findings are nonspecific, often with mild to moderately elevated inflammatory markers (ESR, CRP), normal white blood cell count, and differential. Serologic tests including rheumatoid factor and ANA are usually negative.1,13 Though the disease course is variable and generally considered to be self-limited, it is often protracted, with cyclic episodes of symptom recurrence either at the site of initial involvement or at a new site, followed by periods of quiescence. While most cases resolve spontaneously within months to years, symptomatology and evidence of disease activity have been reported to last up to 25 years after initial presentation.1 Several conditions have been reported in association with CRMO, including but not limited to pustulosis palmoplantaris, acne, pyoderma gangrenosum, Sweet syndrome, psoriasis, inflammatory arthritis, and inflammatory bowel disease.1,10,14 Cutaneous conditions have been reported to be associated with CRMO in 4-25% of patients and inflammatory bowel disease in 3-7% of patients. Cutaneous and bowel-related manifestations may become apparent prior to, concurrent with, or several years following the initial presentation with musculoskeletal pain. 1,3,6,12,15,16
Imaging Findings and Distribution of CRMO
Like hematogenous bacterial osteomyelitis, lesions of CRMO most commonly affect the metaphysis or metaphyseal equivalent of the lower extremity long tubular bones though may extend to involve the metaphyseal-epiphyseal regions and may less commonly affect the diaphysis. The spine, clavicle, and less commonly the upper extremities, pelvis, ribs, sternum, mandible, hands, and feet may be affected.1,11–14 The clavicle and mandible are uncommonly involved in hematogenous bacterial osteomyelitis and lesions there should raise suspicion of CRMO.1,10,14 Mandibular and clavicular lesions have a higher propensity to present and remain as unifocal lesions during the disease course.17 Incidence of medial clavicular involvement is higher in patients with associated acne fulminans and palmopustulosis.1 Like hematogenous bacterial osteomyelitis, CRMO of the pelvis occurs at metaphyseal equivalents, e.g., near the sacroiliac joints and near the synchondroses (Figure 4). CRMO may affect vertebral body marrow throughout the spine, most commonly the thoracic spine and thoracolumbar junction, with or without extension into the pedicles, but in contrast to infectious spondylodiscitis (Figure 9), CRMO usually does not cross the disc space (Figure 6). Multiple vertebral body involvement sparing the disc space should raise concern for CRMO.1,10,18 Multifocal lesions are often present and may be underappreciated if whole-body screening is not performed. One study of fifty-three children found that 98% had 2 or more lesions on whole-body MRI screening at the time of initial presentation.11,12 Symmetric bilateral disease is common, though lesions usually do not present with clinical and temporal symmetry.1,13,14 Radiographs are insensitive to early disease, though may show one or several oval or columnar lytic lesions in the metaphysis abutting the physis, which with time develop a thin rim of peripheral sclerosis (Figure 3a and b). Lesion size and sclerosis typically progress with time, such that mixed lytic-sclerotic or purely sclerotic lesions may be seen. Serial imaging showing progressive lytic abnormality without the development of surrounding sclerosis should raise suspicion of a diagnosis other than CRMO1. Lesions demonstrating a nidus-like appearance with surrounding sclerosis or “pseudo-widening” of a physis (Figure 3a) have been described.19 As healing occurs, radiographs may return to normal over several years or varying degrees of sclerosis, and hyperostosis with metaphyseal under-tubulation may persist.1,10,14 Periosteal reaction can occur and tends to be more prevalent and prominent, sometimes with a multilamellar (“onion-skin”) appearance in smaller diameter tubular bones (e.g., clavicle, fibula, metacarpals, and metatarsals) and with greater disease chronicity.1 Recurrent disease will show new lytic foci at previously affected sites and or additional sites. CT typically shows findings paralleling those seen radiographically. In the setting of mixed lytic sclerotic disease and in areas of overlapping bone, CT may better show the disease extent, but given associated radiation and insensitivity to early disease, CT has a limited role in the imaging of CRMO. MRI is much more sensitive than radiographs for the detection of the early findings of CRMO and is useful to show the extent of local disease including transphyseal spread, to optimize biopsy site selection, and to monitor local disease activity. Whole-body MRI has been increasingly utilized to survey for asymptomatic additional lesions, monitor disease activity, and assess therapeutic response, with the advantage of minimizing pediatric radiation exposure in what may be a prolonged disease course.1,10,13 Disadvantages of MRI include expense, exam duration, and motion sensitivity, though screening whole body protocols have been developed to optimize clinical yield and minimize exam time. Technetium 99m bone scans are more widely available than whole-body MRI, and remain useful for whole-body screening, though symmetric periphyseal disease may occasionally be masked by physiologic periphyseal uptake.1,20 Active lesions demonstrate hyperintensity on T2-weighted images or STIR sequences, intermediate to low signal on T1-weighted images, and enhancement on post-contrast imaging, though intravenous gadolinium is usually unnecessary as it does not often significantly alter diagnosis or management. As lesions progress in chronicity, intralesional and perilesional T1 hypointensity corresponding to sclerosis and hyperostosis may progress. Periosteal reaction and perilesional soft tissue swelling may occur, often being greater in smaller diameter tubular bones and with chronicity, though periosteal reaction and perilesional soft tissue edema are generally less prominent than in the setting of infectious osteomyelitis and aggressive bone lesions. Although small fluid collections and areas of necrotic bone may be seen on MRI and histology, large fluid collections, fistula, and sequestrum formation do not typically occur in CRMO and should raise suspicion of infectious osteomyelitis.1 MRI may demonstrate radiographically occult joint effusion, synovitis, chondral or subchondral erosion, and transphyseal spread.1,21 The latter is important to note (Figure 3), as it may increase the risk of the development of a premature physeal bridge and subsequent growth deformity. In the reparative phase, varying degrees of sclerosis may persist, or marrow signal may return to normal.1,13 Marrow edema and cortical and or periosteal edema may be seen at sites of relapsing disease.1,13,22 CRMO of the spine may occur in a spondylodiscitis-like pattern or as a geographic area of vertebral body marrow replacement with corresponding lytic, or mixed lytic-sclerotic abnormality on radiographs and CT. In the more common spondylodiscitis type lesion, MRI shows erosion of the endplates with adjacent marrow T1 hypointensity and T2/STIR hyperintensity, sometimes with mild disc height loss and decreased intradiscal signal on T2-weighted images.1,13,14 Multiple vertebral segments may be involved (Figure 6) but unlike in bacterial or atypical infectious osteomyelitis, in CRMO transdiscal spread, prominent soft tissue swelling, and abscess usually are not seen.1,10,13 Pathologic fracture (Figures 5 and 6) and vertebra plana with associated irreversible kyphosis may occur.1,13,14
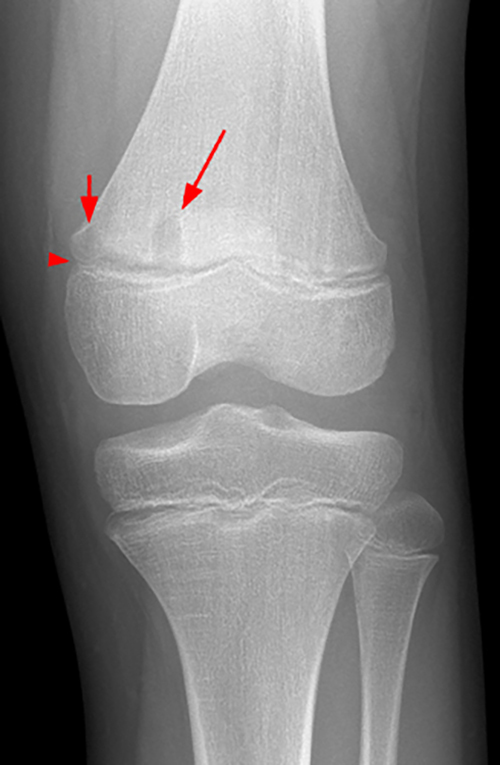
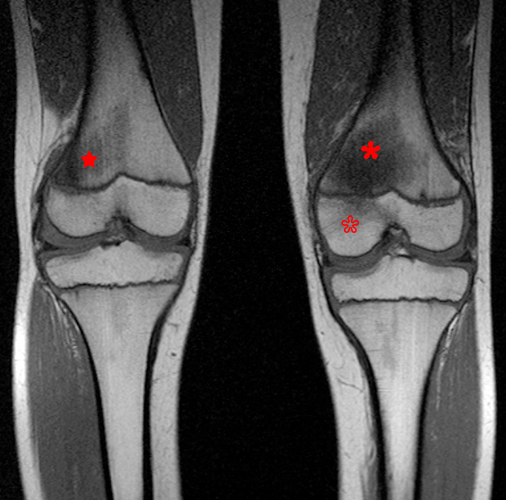
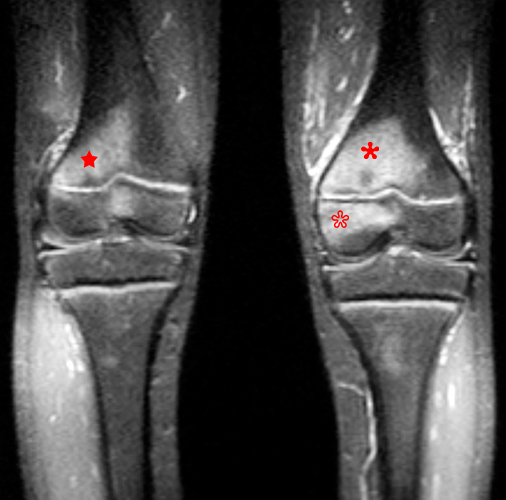
Figure 3:
A frontal radiograph (3a) of the left knee in a 10 year-old male with a three-week history of medial left knee and lateral left ankle pain shows an oval lytic lesion with a subtle thin rim of surrounding sclerosis involving the medial periphyseal left distal femoral metaphysis (arrow), a second smaller far medial periphyseal lytic lesion (short arrow) and “pseudo-widening” (arrowhead) of the medial distal femoral physis. A frontal view of the left ankle (3b) shows a similar oval lytic lesion (arrow) with a subtle thin rim of surrounding sclerosis of the distal left fibular metaphysis. Coronal T1-weighted (3c) and coronal STIR images (3d) of bilateral knees show abnormal intermediate signal on T1-weighted images and hyperintensity on STIR images involving the distal medial left femoral metaphysis (red asterisks) with transphyseal spread and radiographically occult involvement of the medial distal femoral epiphysis (empty asterisk). An additional asymptomatic focus is present in the contralateral (right) distal femoral metaphysis (stars) and central epiphysis.
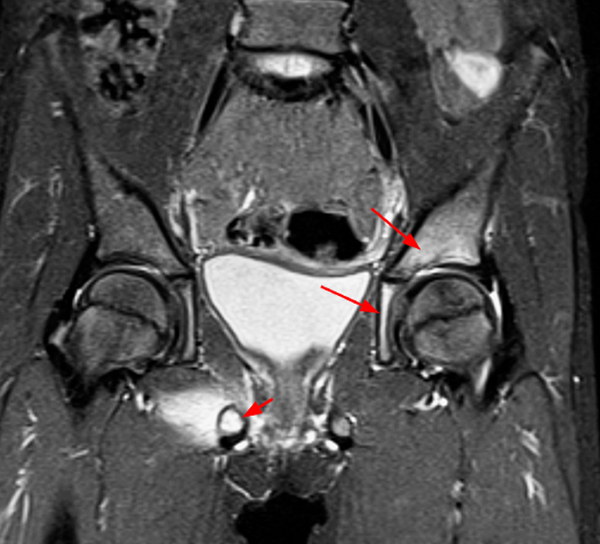
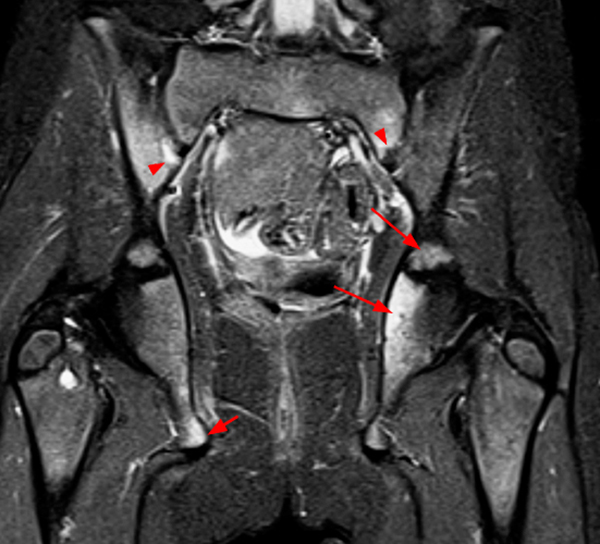
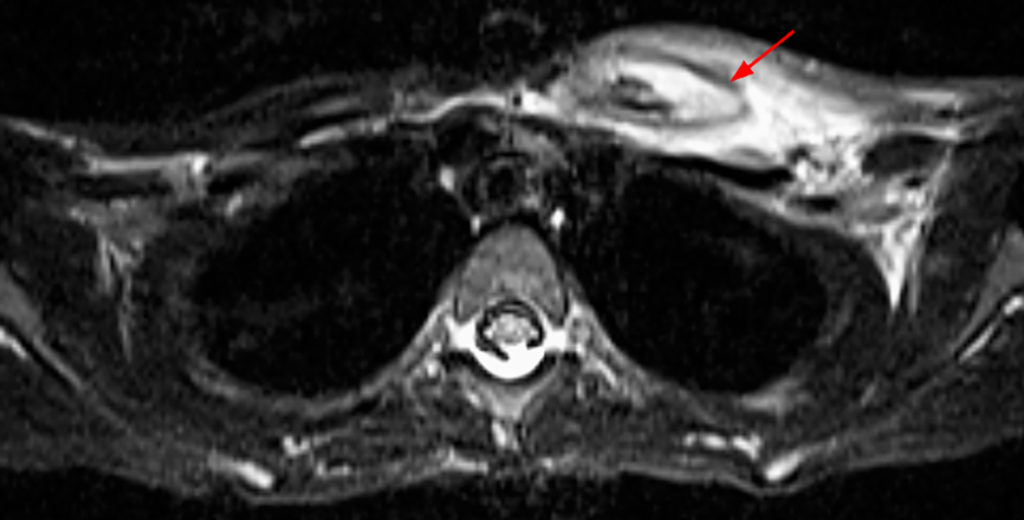
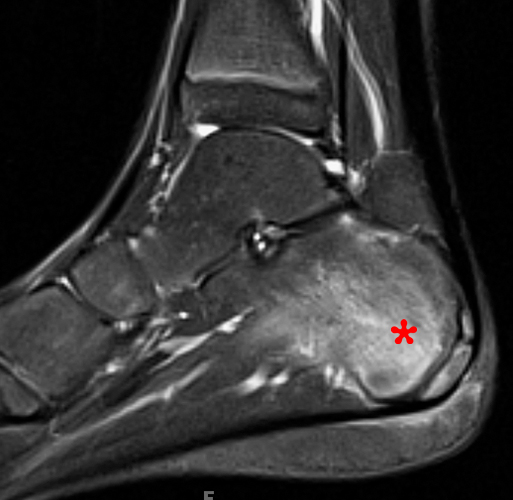
Figure 4:
An 8 year-old female complaining of pelvic pain, diagnosed with CRMO and psoriasis with erosive sacroiliitis. Coronal (4a and 4b) STIR images of the pelvis at the time of presentation show multifocal marrow edema involving the left medial and supra-acetabular ilium centered near the ischioilial synchondrosis (arrows), the right inferior ramus near the right ischiopubic synchondrosis (short arrows) with surrounding edema, subchondral edema bordering bilateral sacroiliac joints and erosive change of bilateral sacroiliac joints (arrowheads). The latter is confirmed on axial CT image of the pelvis (4c) obtained at the time of biopsy, shortly after presentation. The patient developed left clavicular pain and swelling 12 months after presentation. Axial STIR image at that time (4d) shows intense marrow edema and surrounding soft tissue edema of the medial half of the left clavicle (arrow). Nineteen months after presentation the child developed left heel pain, and a sagittal STIR image obtained then (4e) shows marrow edema (asterisk) in posteroinferior calcaneus with mild adjacent periosteal edema.
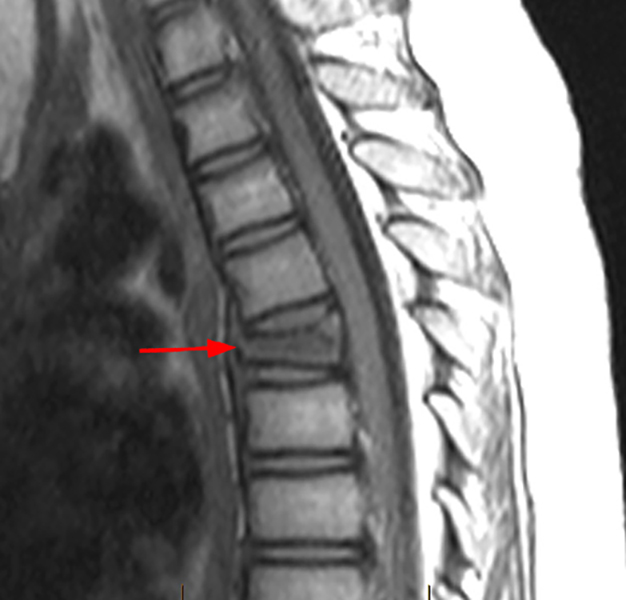
Figure 5:
A 9 year-old female with 2 weeks of worsening atraumatic mid-back and mid-chest pain around the T4-5 dermatome. Sagittal T1-weighted image (5a) and sagittal STIR (5b) images show diffuse abnormal T1 intermediate, T2 hyperintense signal in the T5 vertebral body with a compression fracture and very minimal kyphosis (arrows). No soft tissue mass, paraspinous phlegmon, abscess, or transdiscal abnormality is noted on axial T1-weighted (5c) or sagittal images. The T5 compression deformity (arrow) is also seen on a lateral thoracic radiograph (5d). A biopsy revealed no evidence for malignancy and findings of chronic osteomyelitis. Blood and bone cultures were negative for bacterial and atypical organisms, with a diagnosis of CRMO/CNO.
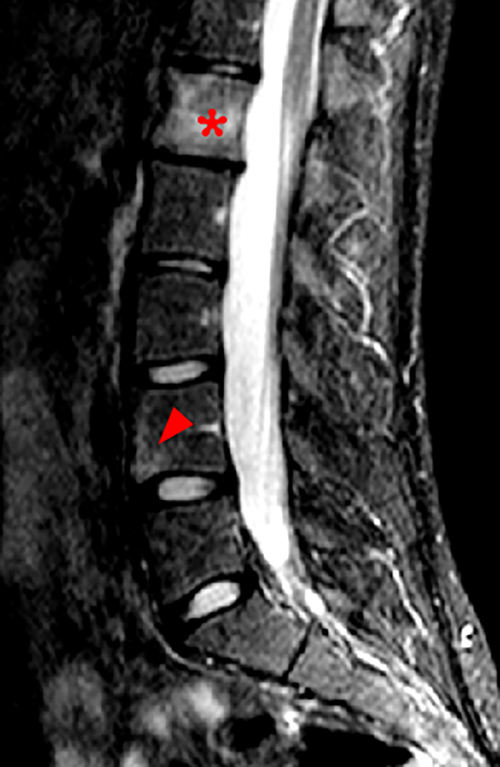
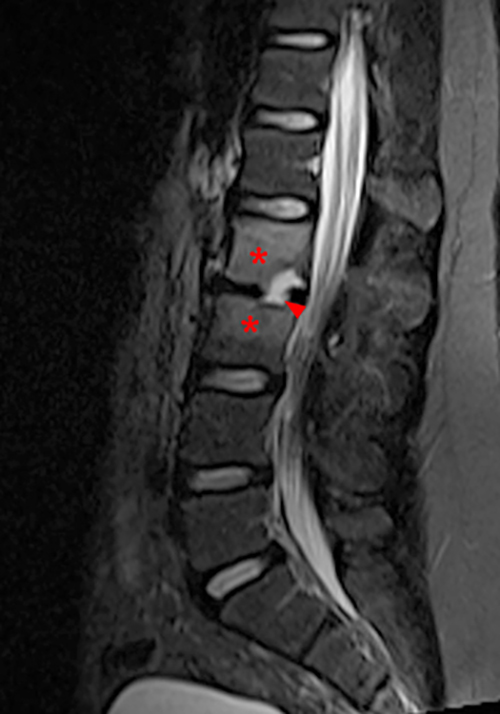
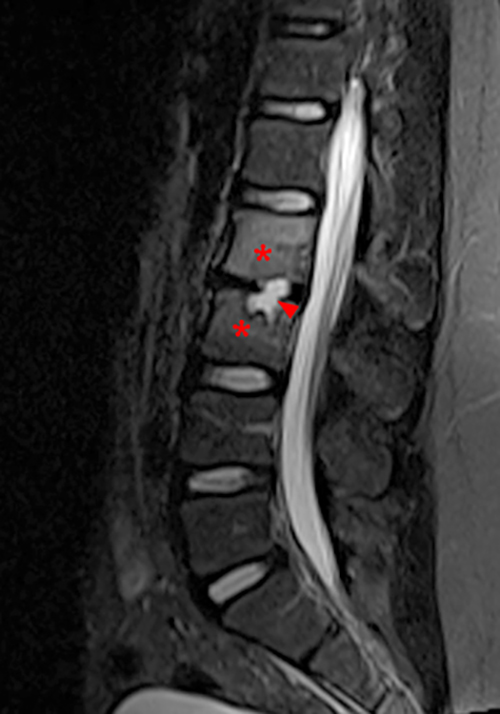
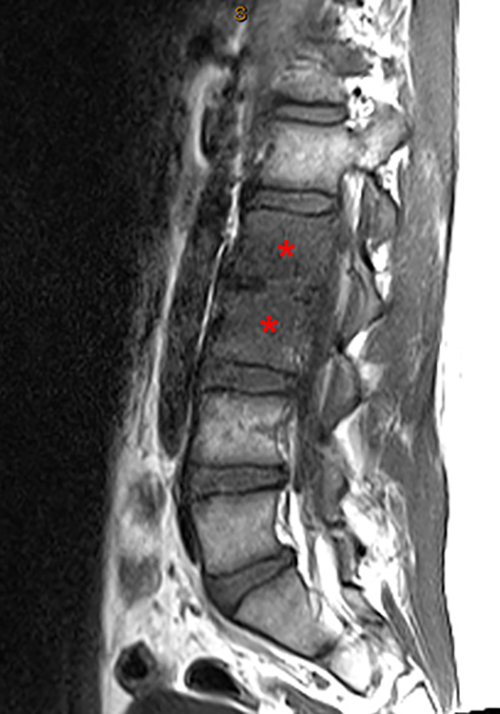
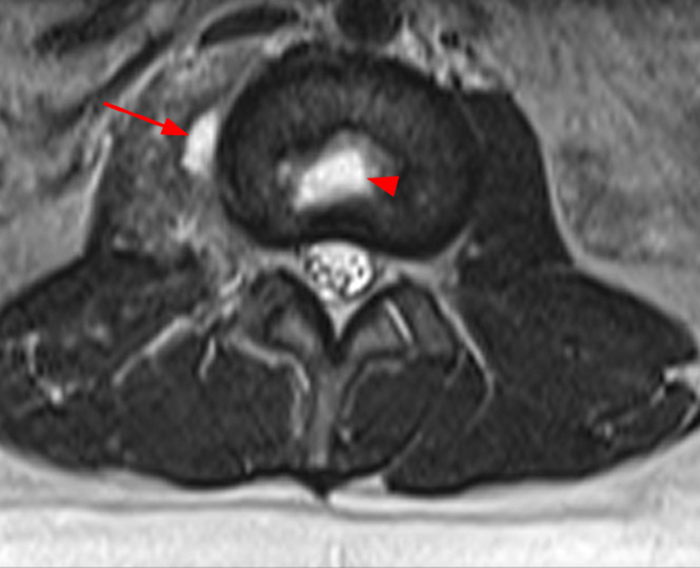
Figure 6:
A 23 year-old female with longstanding CRMO. Sagittal T1-weighted (6a) and STIR (6b) images of the thoracic spine and sagittal T1-weighted (6c) and STIR (6d) images of the lumbar spine demonstrate multifocal thoracic and lumbar areas of marrow edema/osteitis (brackets, asterisks, and arrowheads) with multiple thoracic compression fractures and kyphotic deformity (brackets). Note the absence of transdiscal spread, paraspinous mass, or abscess.
Differential Considerations
The leading differential diagnostic etiologies are infectious (bacterial or atypical) osteomyelitis (Figure 7), tumor and tumor-like lesions including Langerhans cell histiocytosis, Ewing’s sarcoma (Figure 8), osteosarcoma, leukemia, lymphoma, neuroblastoma metastases, and some more rare rheumatologic and metabolic conditions.1,10,13,23 While there is some overlap in the clinical and imaging features of CRMO and infectious osteomyelitis, highly elevated inflammatory markers, transdiscal spread in the spine (Figure 9), or monostotic disease with the presence of soft tissue abscess (Figures 7 and 9d), fistula, or sequestrum should suggest an infectious etiology. A progressively enlarging lytic lesion lacking a developing rim of surrounding sclerosis and cortical destruction are not typical for CRMO and suggest a more aggressive etiology. Aggressive periosteal reaction and prominent delineated soft tissue mass should raise suspicion of malignancy. A soft tissue mass accompanying a lytic spinal lesion with or without pathologic fracture, suggests a neoplastic etiology, while a vertebral lytic lesion and or vertebra plana without associated soft tissue mass may be seen in the setting of either CRMO or Langerhans cell histiocytosis, often requiring biopsy.6,10,13,24
Figure 7:
A 9 year-old male with a 10-day history of arm pain, limiting use. Diagnosed with hematogenous bacterial osteomyelitis. Coronal T1-weighted non-contrast image (7a), coronal STIR (7b), and coronal T1-weighted fat-suppressed post-Gd contrast (7c) images show metaphyseal marrow edema, extending into the epiphysis (brackets) with adjacent rim enhancing subperiosteal abscess (arrows).
Figure 8:
A 9 year-old female with one-month history of worsening right lower leg pain. A frontal radiograph of the distal right tibia, fibula, and ankle (8a) shows subtle permeative abnormality of the distal fibular diametaphysis with associated aggressive appearing periosteal reaction (bracket). Axial T1-weighted (8b), axial STIR (8c) and coronal FSE T2-weighted images (8d) of the distal right lower extremity show segmental marrow replacement of the distal right fibular diaphysis and metaphysis with peripheral T1 and T2 hypointense signal due to aggressive periosteal reaction and osteoid formation, and a prominent surrounding soft tissue mass (asterisk). Biopsy revealed Ewing’s sarcoma of the right fibula.
Figure 9:
13 year-old male with 3 week history of back pain. Sagittal STIR (9a and 9b) and sagittal T1-weighted (9c) images show findings of infectious discitis and osteomyelitis with intradiscal-transdiscal T2 hyperintensity at L2-3 (arrowheads) and surrounding marrow edema (asterisks). Axial fast spin-echo T2-weighted image (9d) shows intradiscal T2 hyperintensity (arrowhead) and a small right psoas abscess (arrow).
Diagnosis and Bone Biopsy
Jansson et al. developed a scaled clinical scoring model and others have proposed diagnostic criteria to facilitate diagnosis and minimize superfluous biopsies in the setting of suspected CRMO. Though these have not yet been prospectively validated and universally accepted, in a large retrospective study, Wipff et al. found that a significant number of bone biopsies (27 bone biopsies in a cohort of 178 patients with CRMO) could have been avoided if the Jansson clinical CNO scoring tool had been implemented. 12,18,24,25,26,27 A biopsy allows microbiologic testing and excludes potential differential diagnoses such as malignancy and eosinophilic granuloma. Cellular infiltrates reflective of varying stages of nonspecific subacute to chronic inflammation with associated areas of marrow fibrosis and osteonecrosis or more acute inflammatory infiltrates in sites of nascent disease may be seen.12,13,28 No causative micro-organism is found. Isolation of Propionibacterium/Cutibacterium acnes in biopsy cultures is usually considered to be skin contamination and may confound the diagnosis.1,12,28,29
Treatment and Outcomes
Historically, the treatment of CRMO has largely been based upon expert opinion and retrospective studies. NSAIDs are usually the first line of therapy and are used during symptomatic exacerbation and as preventative therapy. Several studies have shown that more than 50% of patients respond within the first 1-2 years of treatment, though additional studies have shown that more than half of patients had relapsing symptoms within 5 years of remission. Long-term symptomatic remission is rarely achieved with corticosteroids and prolonged glucocorticoid therapy in childhood and adolescence is generally avoided given potential adverse effects upon growth, though they may be utilized in limited dose and duration as they may quickly quell active inflammation.29,30 Potential second-line therapies include anti-TNF agents and bisphosphonates. There may be a delay in the resolution of clinical versus imaging findings. Thus, treatment is usually targeted toward symptomatic relief and normalization of inflammatory markers, though some advocate for treatment escalation in the setting of active spinal lesions even in the absence of symptoms, to minimize the risk of pathologic fracture and chronic deformity. It has been suggested that anti-TNF agents or bisphosphonates be considered for refractory disease and spinal involvement although continued investigations are needed to optimize and standardize management.25,31,32 In a large retrospective study including 178 patients, Wipff et al. identified three subgroups of CRMO patients (severe, intermediate, and mild phenotypes) with distinct prognostic implications.6 The severe phenotype was seen to involve male patients, the majority of whom had inflammatory syndrome during disease onset (elevated CRP >10mg/liter) and multifocal disease. These patients had the worst prognosis with a 22% remission rate even in the setting of anti-TNF and or bisphosphonate therapy. In addition to the male sex, presence of inflammatory syndrome and multifocality, a long period of time between symptom onset and diagnosis was found to be a poor prognostic indicator. The mild phenotype included mostly females with normal CRP values, unifocal disease, and a higher prevalence (up to 43%) of clavicular involvement. The best prognosis was seen in this subgroup despite the infrequent implementation of second-line therapeutic agents.6 Pathologic fractures occur with vertebral lesions more commonly than at other sites and may be associated with irreversible kyphosis.1,12 While many CRMO lesions may revert to normal, particularly in skeletally immature patients in whom osseous remodeling remains active, sclerotic lesions may persist, particularly at sites that undergo hyperostosis, for example, the medial clavicle. Clavicular hyperostosis may lead to a palpable deformity and uncommonly thoracic outlet syndrome.1,33 Growth deformities related to the development of a premature physeal bridge may occur, with limb length discrepancy (and shortening of the affected limb) being the most frequently reported deformity in those with a history of CRMO.1,12,29,34 Some consider CRMO to be within the clinical spectrum of juvenile spondyloarthropathies including juvenile psoriatic arthritis and juvenile enthesitis-related arthritis, and some patients with CRMO go on to develop inflammatory arthritis.29,31 HLA-B27 positivity has been found in between 6-7.5% of CRMO patients in several series, though this has not been found to correlate with more severe disease, arthritis, or axial involvement.35 SAPHO syndrome (synovitis, acne, pustulosis, hyperostosis osteitis) is postulated by some to be the adult counterpart of CRMO, also within the spectrum of spondyloarthropathies, sharing some similar clinical features and associations, though it is unclear whether these two entities are phenotypical variations of the same disease process or disparate diseases.1,31
Summary
CRMO is an inflammatory non-infectious condition of childhood characterized by painful bone lesions.
- Lesions most commonly affect the metaphysis or metaphyseal equivalents of the lower extremity tubular bones. The spine, clavicle, upper extremities, pelvis, ribs, sternum, mandible hands, and feet may be affected.
- The clavicle and mandible are uncommonly involved in hematogenous bacterial osteomyelitis and lesions there should raise suspicion of CRMO.
- Metaphyseal lytic lesions with a thin peripheral rim of sclerosis are initially seen on radiographs and peripheral or intralesional sclerosis often progresses with time. An enlarging lytic lesion without surrounding sclerosis or cortical destruction suggests a more aggressive etiology. Lamellated periosteal reaction and hyperostosis may occur, most often in small tubular bones. Healed lesions may return to normal, or sclerosis and hyperostosis may persist.
- Lesions show findings of marrow edema on MRI, with or without periosteal reaction and surrounding soft tissue swelling. Soft tissue abscess, fistula, sequestra, and transdiscal disease are not features of CRMO and suggest an infectious etiology. Aggressive periosteal reaction or prominent soft tissue mass should raise suspicion of malignancy.
- MRI is more sensitive than radiographs for early and asymptomatic disease and better detects local disease extent, including transphyseal spread, which increases the risk for growth deformity. MRI may be useful to select the optimal biopsy site and to monitor disease activity.
- Multifocal lesions often occur and may be underappreciated if screening with whole-body MRI or bone scan is not performed.
- Though benign, a protracted disease course with episodic symptomatic exacerbation and remission is common. Potential complications have been observed in up to 33.3% of children and include chronic pain, kyphosis related to pathologic vertebral fracture, growth deformity, and uncommonly thoracic out syndrome related to clavicular hyperostosis.35
A special thanks to David Rubin, MD for contributing multiple cases for this clinic.
References
- Khanna G, Sato TSP, Ferguson P. Imaging of chronic recurrent multifocal osteomyelitis. Radiographics. 2009;29(4):1159-1177. doi:10.1148/rg.294085244 ↩
- Suresh S, Saifuddin A. Unveiling the “unique bone”: A study of the distribution of focal clavicular lesions. Skeletal Radiology. 2008;37(8):749-756. doi:10.1007/s00256-008-0507-7 ↩
- Zhao Y, Ferguson PJ. Chronic Nonbacterial Osteomyelitis and Chronic Recurrent Multifocal Osteomyelitis in Children. Pediatric Clinics of North America. 2018;65(4):783-800. doi:10.1016/j.pcl.2018.04.003 ↩
- Ferguson PJ, Bing X, Vasef MA, et al. A missense mutation in pstpip2 is associated with the murine autoinflammatory disorder chronic multifocal osteomyelitis. Bone. 2006;38(1):41-47. doi:10.1016/j.bone.2005.07.009 ↩
- Taddio A, Zennaro F, Pastore S, Cimaz R. An update on the pathogenesis and treatment of chronic recurrent multifocal osteomyelitis in children. Pediatric Drugs. 2017;19(3):165-172. doi:10.1007/s40272-017-0226-4 ↩
- Wipff J, Costantino F, Lemelle I, et al. A large national cohort of French patients with chronic recurrent multifocal osteitis. Arthritis and Rheumatology. 2015;67(4):1128-1137. doi:10.1002/art.39013 ↩
- Giedion A, Holthusen W, Masel LF, Vischer D. Ann Radiol (Paris). Ann Radiol (Paris). 1972;15(3):329-342. ↩
- Schnabel A, Range U, Hahn G, Siepmann T, Berner R, Hedrich CM. Unexpectedly high incidences of chronic non-bacterial as compared to bacterial osteomyelitis in children. Rheumatology International. 2016;36(12):1737-1745. doi:10.1007/s00296-016-3572-6 ↩
- D’angelo P, Tanturri De Horatio L, Toma P, et al. Chronic nonbacterial osteomyelitis — clinical and magnetic resonance imaging features. Pediatric Radiology. 2021;51:282-288. doi:10.1007/s00247-020-04827-6/Published ↩
- Sato TS, Watal P, Ferguson PJ. Imaging mimics of chronic recurrent multifocal osteomyelitis: avoiding pitfalls in a diagnosis of exclusion. Pediatric Radiology. 2020;50(1):124-136. doi:10.1007/s00247-019-04510-5 ↩
- von Kalle T, Heim N, Hospach T, Langendörfer M, Winkler P, Stuber T. Typical patterns of bone involvement in whole-body MRI of patients with chronic recurrent multifocal osteomyelitis (CRMO). RoFo Fortschritte auf dem Gebiet der Rontgenstrahlen und der Bildgebenden Verfahren. 2013;185(7):655-661. doi:10.1055/s-0033-1335283 ↩
- Zhao DY, McCann L, Hahn G, Hedrich CM. Chronic nonbacterial osteomyelitis (CNO) and chronic recurrent multifocal osteomyelitis (CRMO). Journal of Translational Autoimmunity. 2021;4. doi:10.1016/j.jtauto.2021.100095 ↩
- Jurik AG. Chronic recurrent multifocal osteomyelitis. Seminars in Musculoskeletal Radiology. 2004;8(3):243-253. ↩
- Iyer RS, Thapa MM, Chew FS. Chronic recurrent multifocal osteomyelitis: Review. American Journal of Roentgenology. 2011;196(6 SUPPL.). doi:10.2214/AJR.09.7212 ↩
- Job-Deslandre C, Krebs S, Kahan A. Chronic recurrent multifocal osteomyelitis: five-year outcomes in 14 pediatric cases. Joint Bone Spine . 2001;68:245-251. ↩
- Girschick H, Finetti M, Orlando F, et al. The multifaceted presentation of chronic recurrent multifocal osteomyelitis: A series of 486 cases from the Eurofever international registry. Rheumatology (United Kingdom). 2018;57(7):1203-1211. doi:10.1093/rheumatology/key058 ↩
- Zhao Y, Ferguson PJ. Chronic nonbacterial osteomyelitis and chronic recurrent multifocal osteomyelitis in children. Pediatric Clinics of North America. 2018;65(4):783-800. doi:10.1016/j.pcl.2018.04.003 ↩
- Wipff J, Adamsbaum C, Kahan A, Job-Deslandre C. Chronic recurrent multifocal osteomyelitis. Joint Bone Spine. 2011;78(6):555-560. doi:10.1016/j.jbspin.2011.02.010 ↩
- Falip C, Alison M, Boutry N, et al. Chronic recurrent multifocal osteomyelitis (CRMO): A longitudinal case series review. Pediatric Radiology. 2013;43(3):355-375. doi:10.1007/s00247-012-2544-6 ↩
- Acikgoz G, Averill LW. Chronic recurrent multifocal osteomyelitis: Typical patterns of bone involvement in whole-body bone scintigraphy. Nuclear Medicine Communications. 2014;35(8):797-807. doi:10.1097/MNM.0000000000000126 ↩
- Manson D, Wilmot DM, King S, Laxer RM. Pediatric Radiology Physeal Involvement in Chronic Recurrent Multifocal Osteomyelitis. Vol 20.; 1989. ↩
- Jurik AG, Egund N. MRI in chronic recurrent multifocal osteomyelitis. Skeletal Radiology. 1997;26:230-238. ↩
- Iyer RS, Thapa MM, Chew FS. Chronic recurrent multifocal osteomyelitis: Review. American Journal of Roentgenology. 2011;196(6 SUPPL.). doi:10.2214/AJR.09.7212 ↩
- Jansson AF, Müller TH, Gliera L, et al. Clinical score for nonbacterial osteitis in children and adults. Arthritis and Rheumatism. 2009;60(4):1152-1159. doi:10.1002/art.24402 ↩
- Zhao Y, Wu EY, Oliver MS, et al. Consensus treatment plans for chronic nonbacterial osteomyelitis refractory to nonsteroidal anti-inflammatory drugs and/or with active spinal lesions. Arthritis Care and Research. 2018;70(8):1228-1237. doi:10.1002/acr.23462 ↩
- Roderick MR, Shah R, Rogers V, Finn A, Ramanan A v. Chronic recurrent multifocal osteomyelitis (CRMO) – advancing the diagnosis. Pediatric Rheumatology. 2016;14(1). doi:10.1186/s12969-016-0109-1 ↩
- Jansson A, Renner ED, Ramser J, et al. Classification of non-bacterial osteitis: Retrospective study of clinical, immunological and genetic aspects in 89 patients. Rheumatology. 2007;46(1):154-160. doi:10.1093/rheumatology/kel190 ↩
- Hu HI, Krauspe R, Konrad Muller-Hermelink H, Papadopoulos T. Chronic Recurrent Multifocal Osteomyelitis in Children: Diagnostic Value of Histopathology and Microbial Testing. Human Pathology. 1999;30(1):59-65. ↩
- Hofmann SR, Kapplusch F, Girschick HJ, et al. Chronic recurrent multifocal osteomyelitis (CRMO): Presentation, pathogenesis, and treatment. Current Osteoporosis Reports. 2017;15(6):542-554. doi:10.1007/s11914-017-0405-9 ↩
- Iyer RS, Thapa MM, Chew FS. Chronic recurrent multifocal osteomyelitis: Review. American Journal of Roentgenology. 2011;196(6 SUPPL.). doi:10.2214/AJR.09.7212 ↩
- Sinnappurajar P, Roderick M, Ramanan A v. The neglected and untreated pains of CRMO and SAPHO syndrome . Rheumatology. Published online March 4, 2022. doi:10.1093/rheumatology/keac128. doi:10.2214/AJR.09.7212 ↩
- Ferjani HL, Makhlouf Y, Maatallah K, et al. Management of chronic recurrent multifocal osteomyelitis: review and update on the treatment protocol. Expert Opinion on Biological Therapy. 2022;22(6):781-787. ↩
- Pelkonen P, Ryoppy S, Jaaskelainen J, Rapola J, Repo H, Kaitila I. Chronic osteomyelitis like disease with negative bacterial cultures. Published online November 1988. http://archpedi.jamanetwork.com/ ↩
- Schnabel A, Range U, Hahn G, Berner R, Hedrich CM. Treatment response and longterm outcomes in children with chronic nonbacterial osteomyelitis. Journal of Rheumatology. 2017;44(7):1058-1065. doi:10.3899/jrheum.161255 ↩
- Koryllou A, Mejbri M, Theodoropoulou K, Hofer M, Carlomagno R. Chronic nonbacterial osteomyelitis in children. Children. 2021;8(7). doi:10.3390/children8070551 ↩