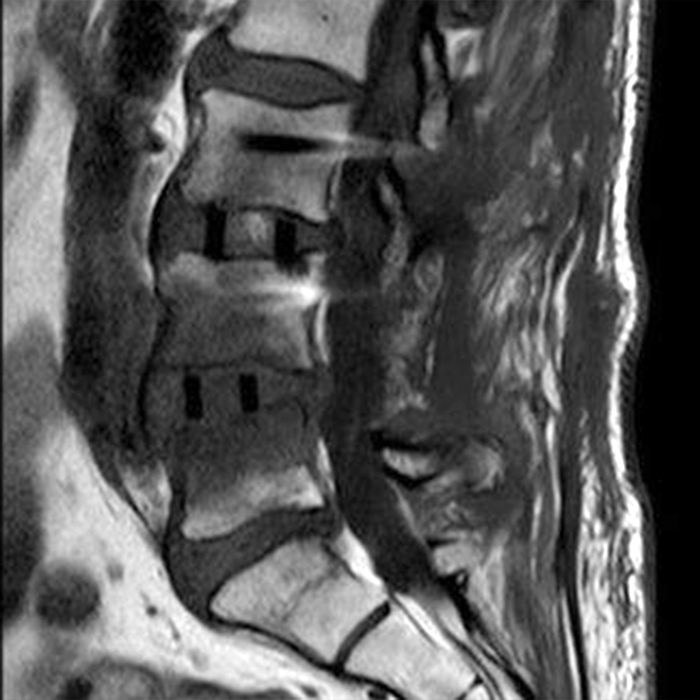
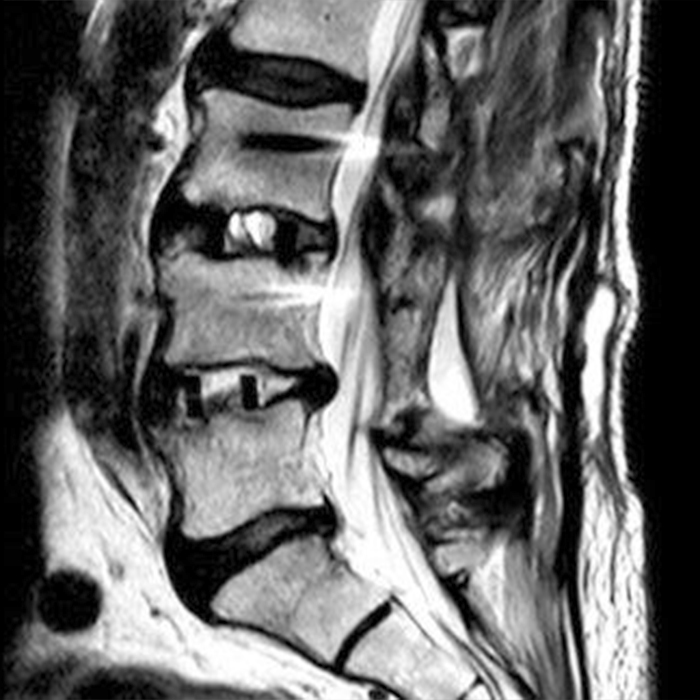
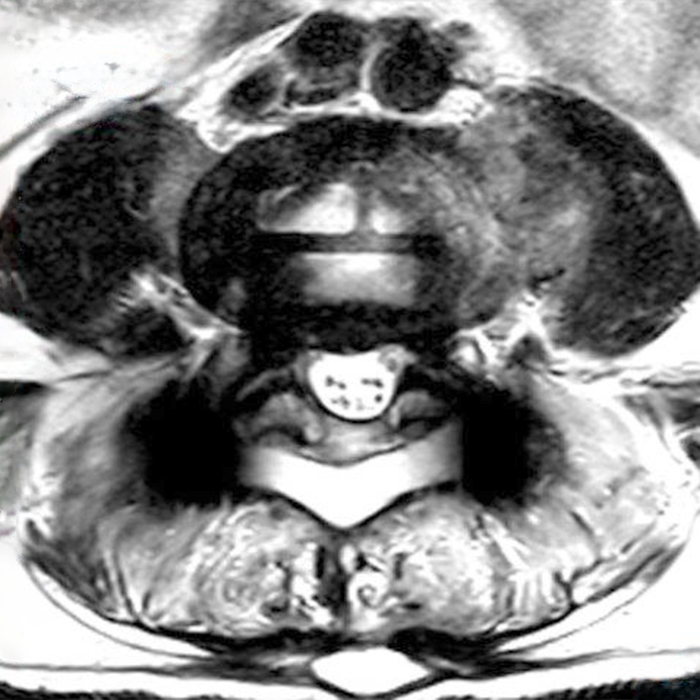
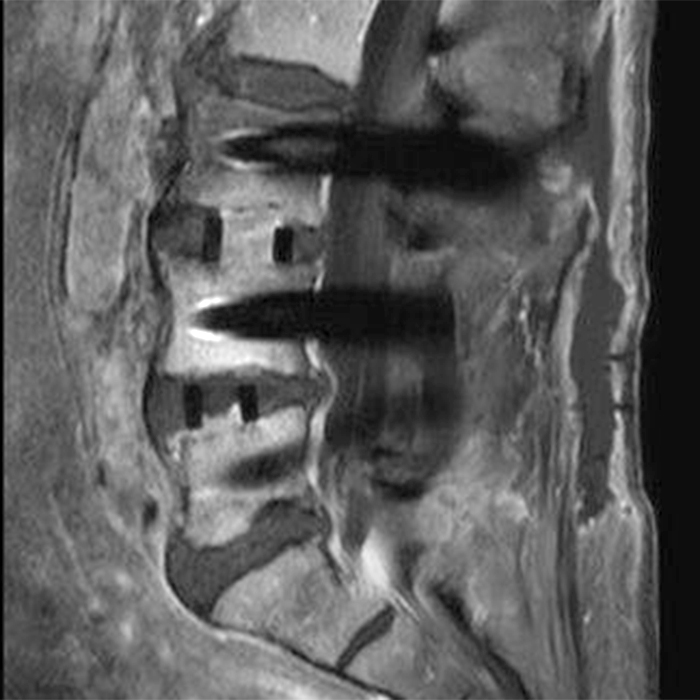
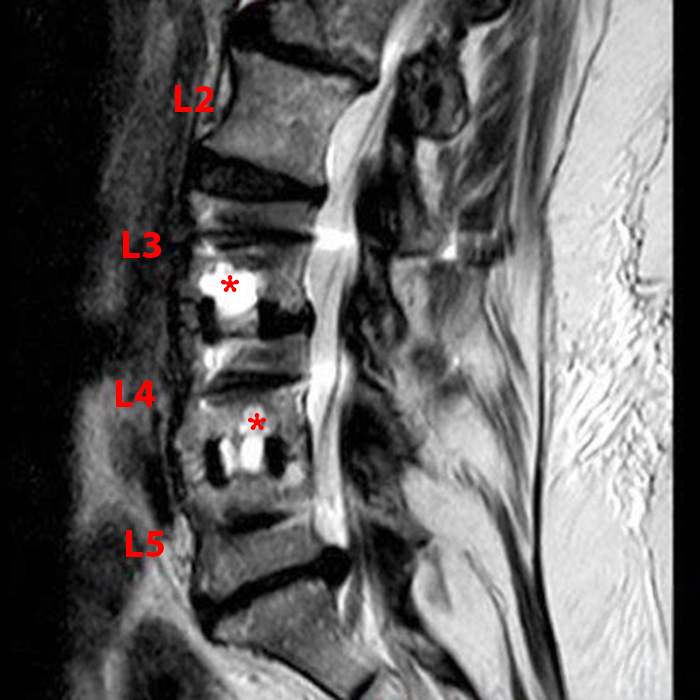
Clinical history: An 88-year-old man presents two months status post anterior lumbar interbody fusion at L3-4 and L4-5 with back pain. Sagittal T1-weighted (1A), sagittal T2-weighted (1B), axial T2-weighted (1C), sagittal post-contrast fat-suppressed T1-weighted (1D), and axial post-contrast T1-weighted (1E) images are provided. What are the findings? What is your diagnosis?
Findings:
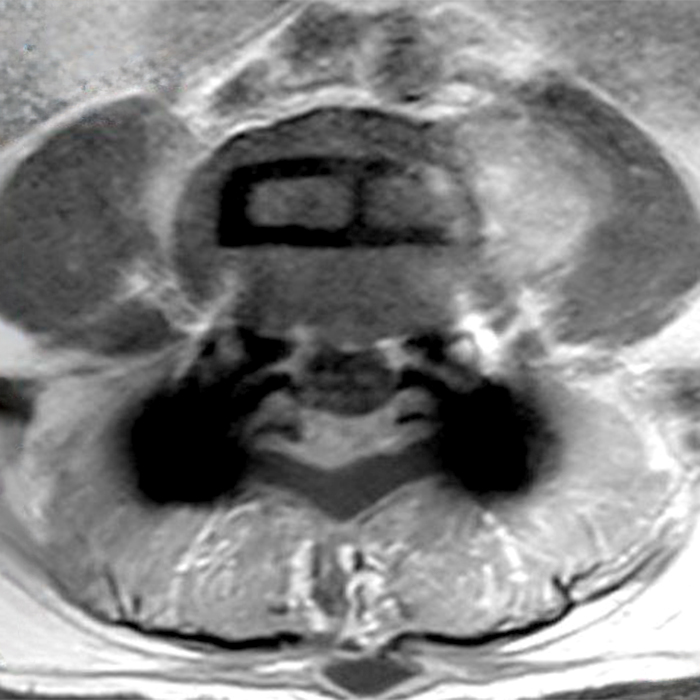
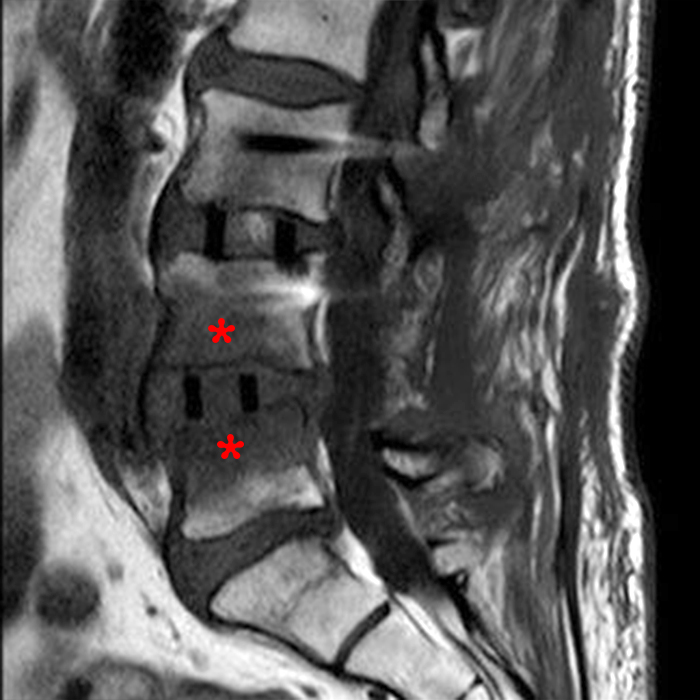
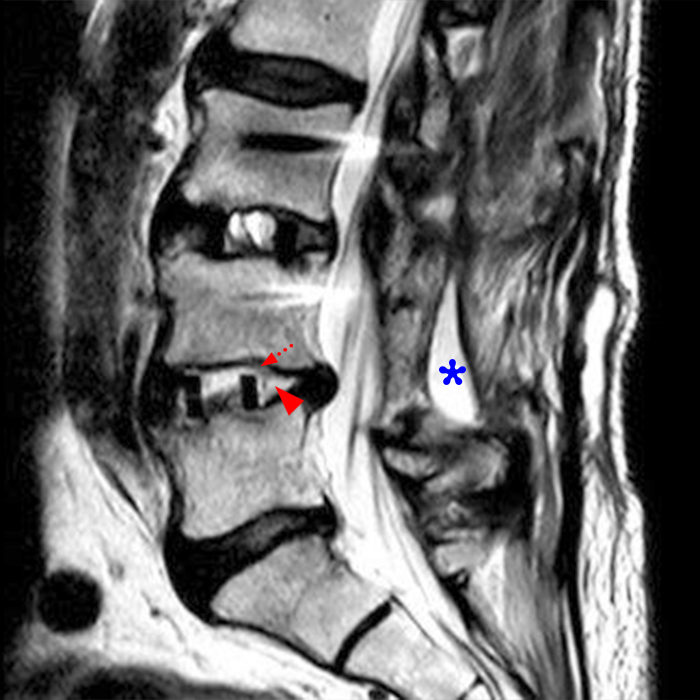
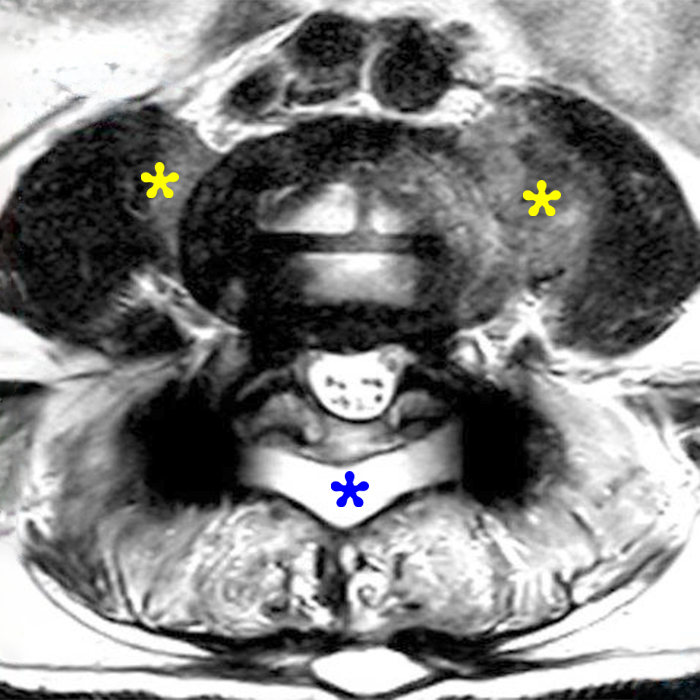
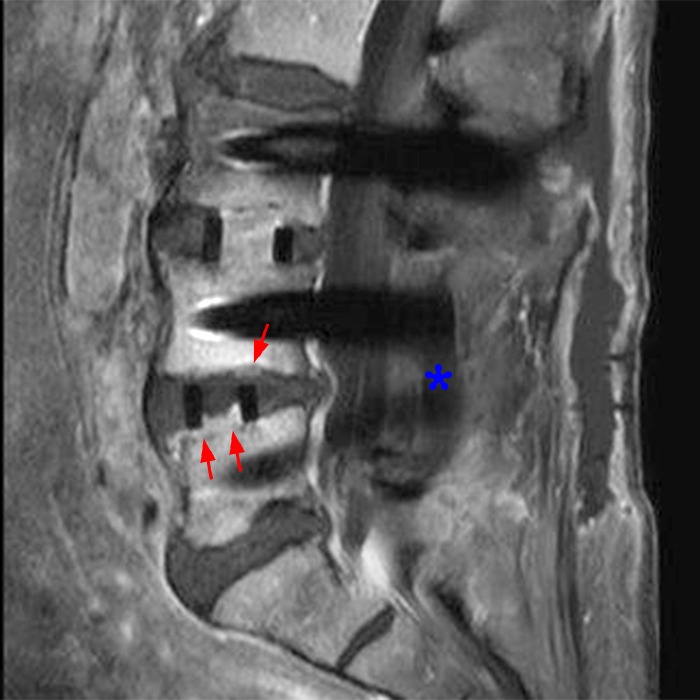
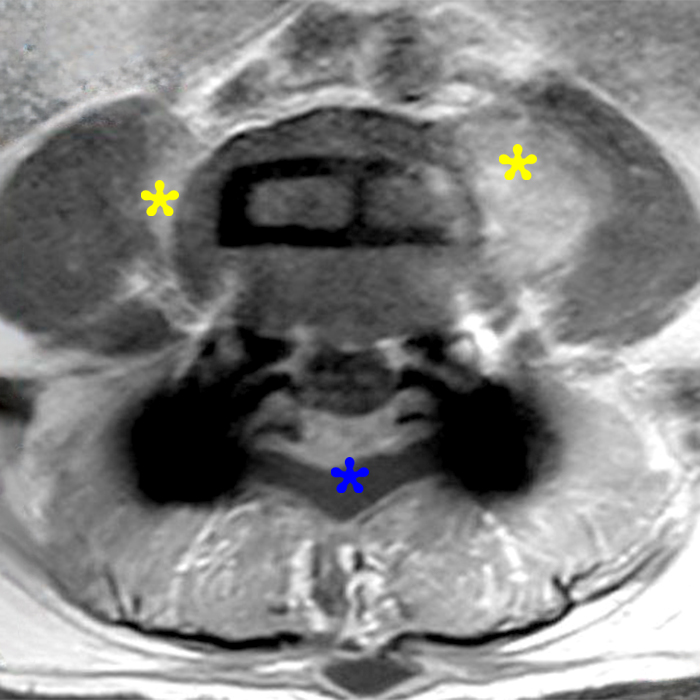
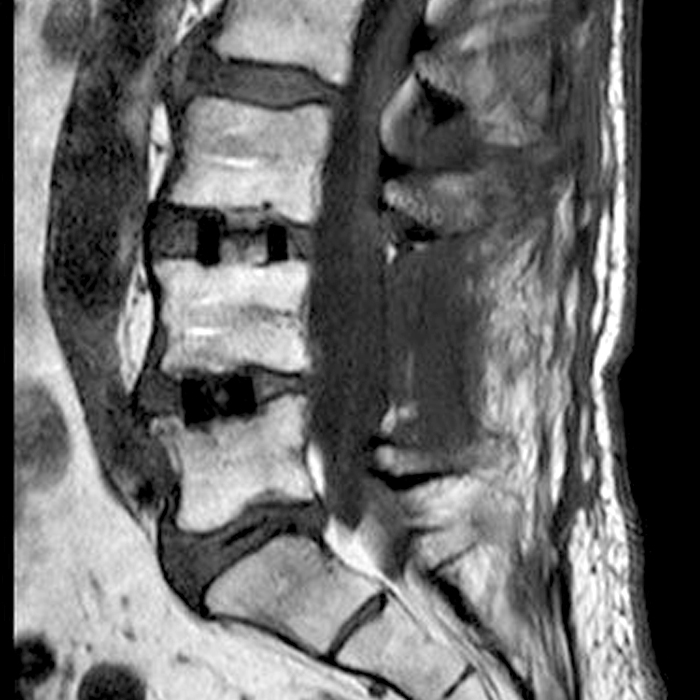
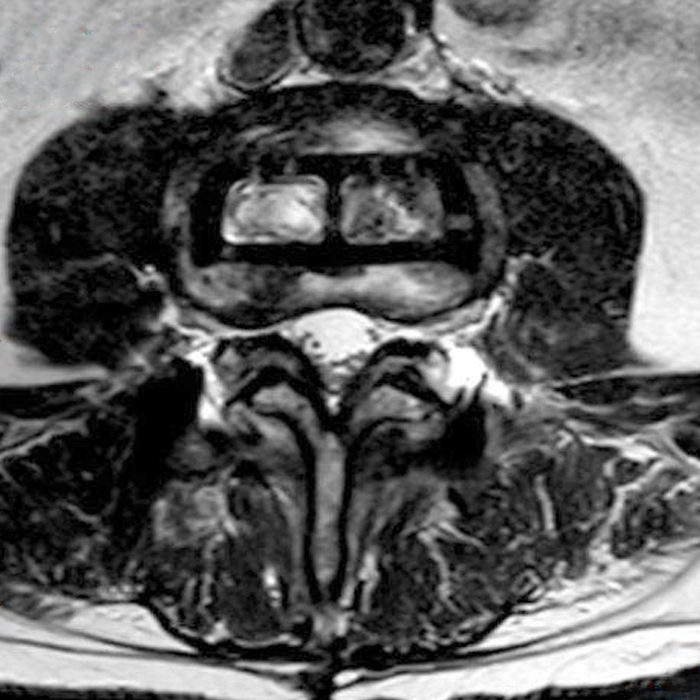
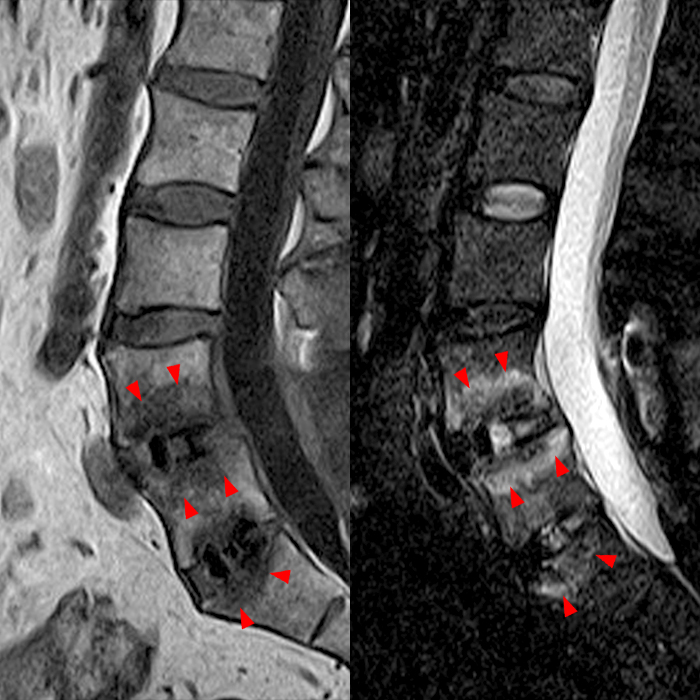
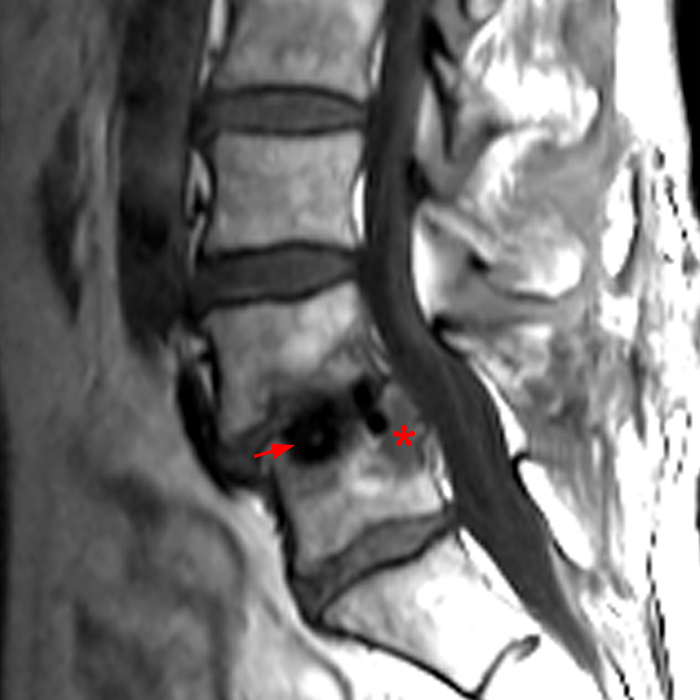
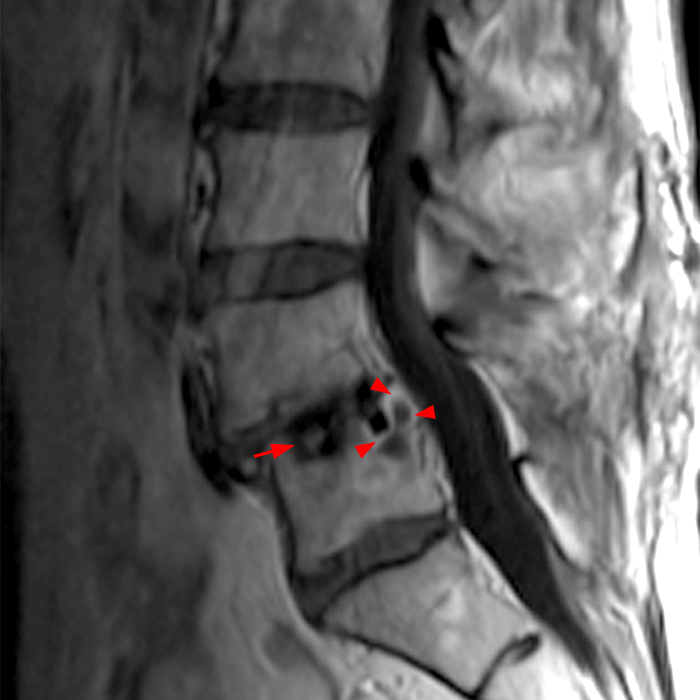
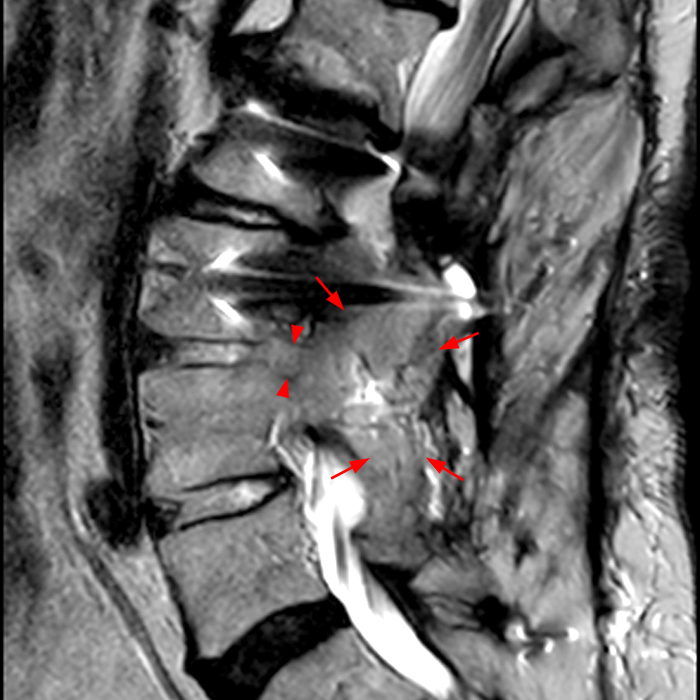
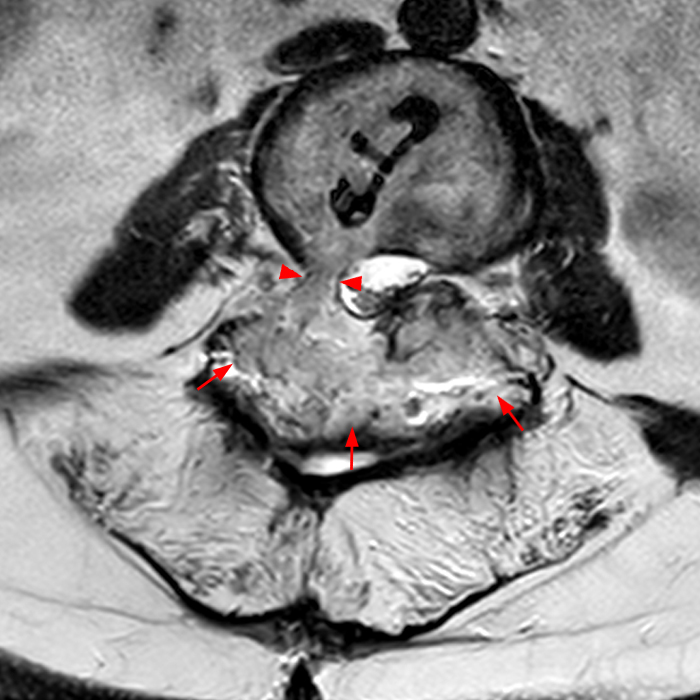
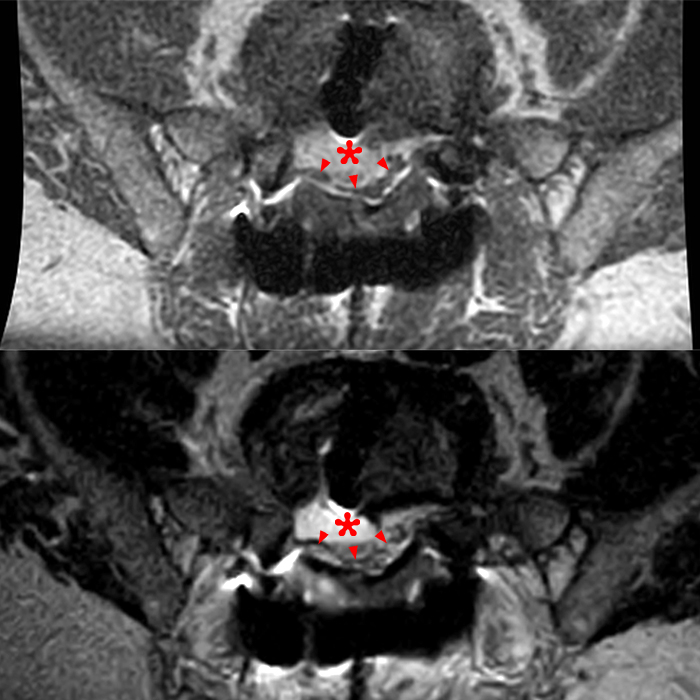
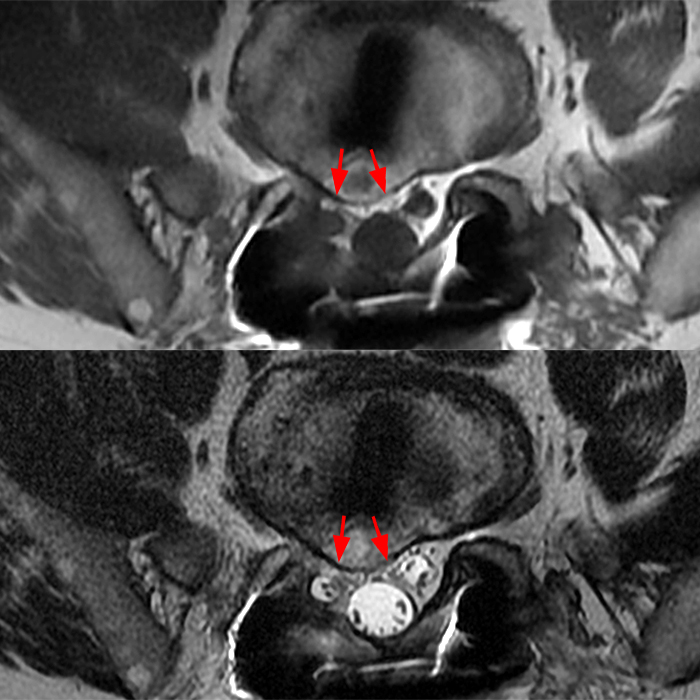
Figure 2: On the sagittal T1-weighted image (2A), reduced marrow fat signal (red asterisks), representing marrow edema, borders the L4-5 disc, which contains an L4-5 interbody cage/spacer. Intradiscal fluid signal (arrowhead) extends between the fusion cage and inferior endplate of L4 (dotted arrow) on the corresponding T2-weighted sagittal image (2B), and a dorsal fluid-signal collection is seen in the laminectomy defect (blue asterisk). A dorsal subcutaneous postoperative seroma is unmarked. The axial T2-weighted image (2C) reveals edema in the psoas muscles bilaterally (yellow asterisks) and redemonstrates the dorsal fluid signal collection overlying the laminectomy defect (blue asterisk). T1-weighted post-contrast enhanced images with fat suppression (2D) demonstrate endplate reabsorption with enhancement (short arrows) and unremarkable enhancement in the region of the post-surgical dorsal soft tissue fluid collection (blue asterisk). Post-contrast T1-weighted axial image (2E) demonstrates enhancement in the psoas muscles (yellow asterisks) and mild peripheral enhancement of the dorsal fluid collection and paraspinal musculature. Sagittal T1-weighted (2F) and axial T2-weighted (2G) images obtained 18 months following surgery demonstrate resolution of L4-5 vertebral and adjacent psoas muscle edema without therapy.
Diagnosis
Anterior lumbar interbody fusion (ALIF), synthetic polyetheretherketone (PEEK) cage with recombinant human Bone Morphogenetic Protein-2, mimicking post-operative discitis and osteomyelitis on hospital admittance MRI. No therapy was instituted with an 18-month follow-up MRI showing resolution of abnormal imaging findings.
Introduction
Recombinant Human Bone Morphogenetic Protein-2, rhBMP-2, hereafter referred to as BMP, has a long and storied history in spine fusion surgery. Initially described by Urist in 19651, multiple BMP osteoinductive proteins were identified, with BMP-2 being the most osteoinductive growth factor,2,3,4attaching to the receptor sights on osteoblasts and inducing osteogenesis. Medtronic released FDA-approved (rhBMP-2 INFUSE Bone Graft) in 2002, approved for ALIF for lumbar degenerative disease with early industry-sponsored literature noting excellent fusion results and minimal complications. BMP allowed bypassing the traditional iliac crest bone graft (ICBG) harvest, avoiding an additional surgical site and its complications with complication rates that vary widely in the literature. Industry-sponsored literature reported complication rates of 40% to 60% compared to historical incidence of 3% to 30%.5 Use of BMP-2 eliminated the need for ICBG, decreased surgical time and blood loss, and allowed for improved fusion rates in patients with diabetes, osteoporosis , tobacco use, and previous fusion failures. The Medtronic product includes powdered BMP to be reconstituted and applied to an included collagen sponge acting as a superstructure and placed within the supplied 2 conical titanium cages for ALIF. Off-label use of BMP is performed with all types of cages/spacers except for ring femur allografts, which have been found susceptible to the osteolysis stage of BMP bone formation.6
Complications
With the high fusion rate and low risk profile reported in the industry-sponsored literature, BMP use proliferated in ALIF, growing from 0.7% in 2002 to greater than 50% of cases in 2007 and growing to 43% of off-label spine fusion use in 20077,8with reported up to 85% usage for posterior lumbar interbody fusion (PLIF) and the more recent modification of PLIF, transforaminal lumbar interbody fusion (TLIF) as well as cervical fusion.9 BMP application peaked in 2010 with mounting reports of adverse events and a warning issued by the FDA in 2008 related to off-label anterior cervical spine fusion surgery with cases of severe prevertebral soft tissue swelling and airway compromise initiating a trend reversal.10
With the highly osteoinductive properties of BMP, use has continued with safety modifications of surgical techniques with reduced doses of BMP and decreased per-procedure costs. Many studies have led to changes, including decreasing the volume of the BMP placed, procedural care to position the BMP within the disc space, particularly important with posterior approaches, while not allowing BMP to enter the foramina, epidural space, or paraspinal space.11,12According to Glassman6, BMP use has rebounded to 90% of the frequency reported in 2011, but with lower doses per case used. BMP use associated imaging findings to be aware of include bone osteolysis, heterotopic ossification, and epidural and foraminal hematomas/seromas.13
Clinical Presentations
End plate osteolysis and epidural or foraminal fluid collections may present with nonspecific back pain weeks to months post interbody fusion. Patients may present to a physician other than their operating surgeon or to an ER with clinical findings and imaging appearance raising the specter of discitis/osteomyelitis in a post-operative spine fusion case. Tests for inflammation such as C-reactive protein and erythrocyte sedimentation rate are typically elevated in post-operative patients, therefore of limited help distinguishing infection from post-operative changes, and the spine surgeon then must allay fears and the calls for urgent intervention. Appropriate therapy is conservative with clinical monitoring of the patient. Sethi demonstrated bone-end plate resorption-osteolysis with BMP was seen as early as 2 weeks post interbody fusion on CT, with the largest transition between bone resorption and new bone formation occurring between 3 and 6 months after surgery.14
The 2008 black box warning from the FDA regarding risks of BMP use in anterior cervical interbody fusion (ACDF) secondary to the soft tissue swelling compromising the patient airway has dramatically reduced the use of BMP in the cervical spine, but some surgeons continue to use small doses of BMP, adding a sealant over the anterior disc space. The risk profile in posterior cervical fusion appears more benign and is weighed against fusion failure risk factors.6
Heterotopic ossification (HO), also known as ectopic bone formation, was studied by Niu15 in a recent large cohort of lumbar TLIF (transforaminal lumbar interbody fusion) with unilateral posterior annulotomy with reduced BMP dosage and care to place the cage and BMP sponge away from annulotomy and compared with a control group with TLIF using the traditional ICBG (iliac crest bone graft) and evaluating for occurrence of heterotopic ossification. Patients were followed with a 6-month CT of the spine and monitored for back pain and radicular symptoms. A 92.5% fusion rate for BMP and a 71.4% fusion rate for ICBG control groups were obtained. Heterotopic ossification occurred more frequently in the BMP group versus the control group, with a grading system noting a much higher rate of prominent HO in the BMP group. Only one patient with HO in the BMP group developed pain occurring 2 months post-operatively, with no symptomatic HO in the control group. The pain was treated conservatively without revision surgery. The authors concluded that rhBMP-2 can be safely used with minimally symptomatic HO and high fusion rates.15
A potential complication, cancer induction by this ontogenetic protein, with early anecdotal reports of cancer induction by BMP protein has been disproven by more recent multiple large cohort studies.16,17
Imaging Findings
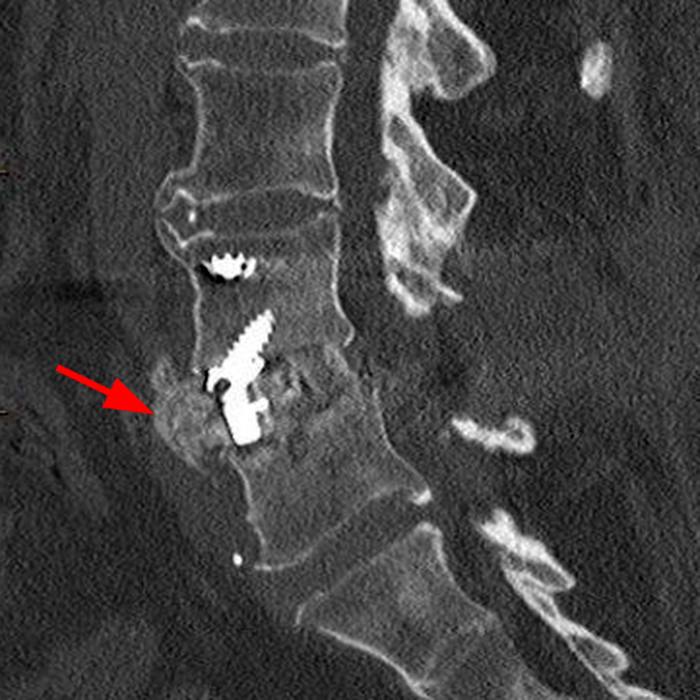
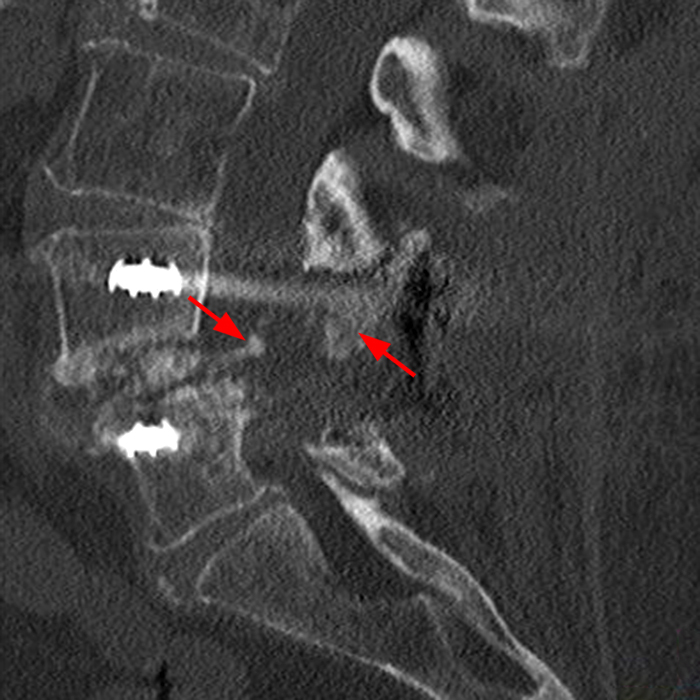
Osteolysis is readily appreciated with MRI and CT and on radiographs in more severe presentations and when associated with cage displacement. Osteolysis presents as bone defects on CT and loss of endplate cortical definition and fluid collections with marrow edema on MRI (Figures 3, 4, and 5). As noted above osteolysis can present as early as 2 weeks and as late as 6 months after BMP use.
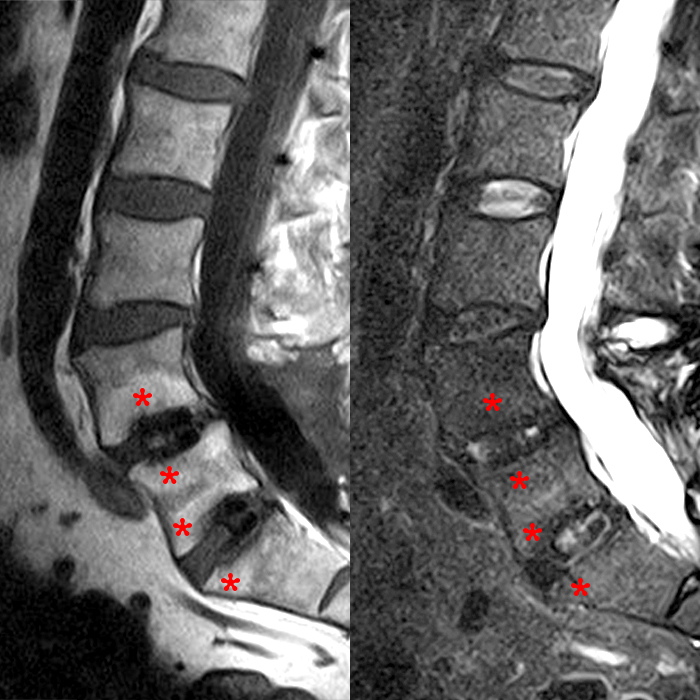
Figure 3: Sagittal T2-weighted image 6-months post ALIF with high T2 fluid signal foci of osteolysis (asterisks) after BMP use.
Figure 4: Sagittal T1-weighted (left) and STIR (right) images 5 months post ALIF with BMP show dramatic vertebral marrow edema (arrowheads). Sagittal T1-weighted (left) and STIR (right) images from a follow-up MRI at 2 years post-surgery show conversion to Modic 2 fatty marrow changes (asterisks) with mild cage subsidence at both levels secondary to the osteolytic phase of BMP bone formation induction.
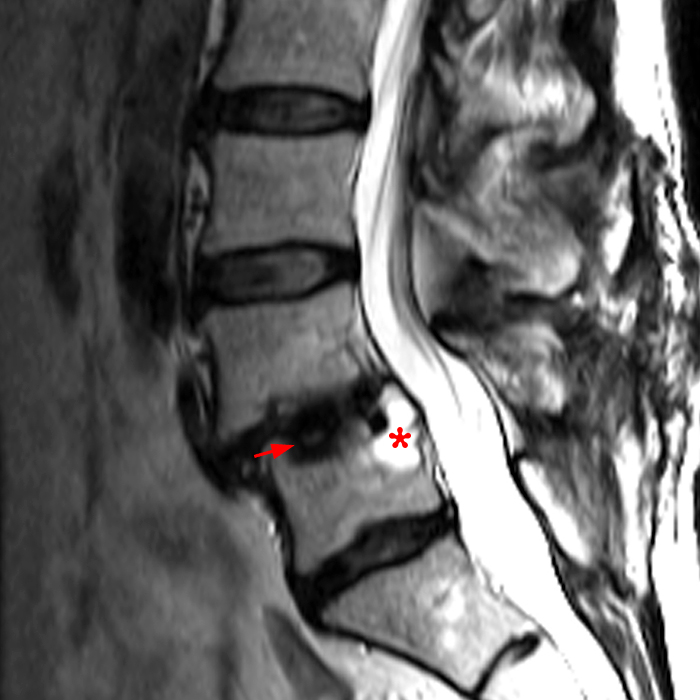
Figure 5: Sagittal T2-weighted (5A), T1-weighted (5B), and post-contrast T1-weighted (5C) images in a patient who is 10 months post-ALIF with BMP demonstrate osteolysis and intraosseous fluid (asterisks) with rim enhancement (arrowheads) and mild cage subsidence (arrows).
Figure 6: Sagittal CT image with bone windowing demonstrates changes of a remote ALIF with BMP and prevertebral heterotopic ossification (arrow).
Figure 7: Sagittal CT image with bone windowing demonstrates a remote PLIF with BMP and heterotopic ossification extending into the foramen and lateral epidural space (arrows).
Figure 8: T2-weighted sagittal (8A) and axial (8B). Remote right TLIF with BMP with exuberant heterotopic ossification involving the neural foramen (arrowheads) and posterolateral bone graft (arrows).
Figure 9: Axial T1-weighted (upper) and T2-weighted (lower) images 2 months post-op PLIF with BMP with anterior epidural seroma/hematoma (asterisks) appearing hyperintense to muscle on T1- and T2-weighted images severely flattening and displacement of the thecal sac and traversing nerve roots (arrowheads). Follow-up axial T1-weighted (upper) and T2-weighted (lower) images at 18 months show resolution of fluid and a smaller area of heterotopic ossification with visible bone cortex (arrows).
Treatment and Management
Osteolysis is primarily treated conservatively, allowing the osteoclastic phase to turn to osteoblast formation. Revision surgery is reserved for symptomatic graft malposition. Post-operative seromas are treated like all other post-operative seromas, with revision surgery reserved for the significantly symptomatic patient. Likewise, heterotopic ossification is only resected in the symptomatic patient.
Conclusion
Recombinant Human Bone Morphogenetic Protein-2 will continue to have a prominent role in interbody spine fusion, particularly lumbar fusion in both FDA-approved and off-label use with improved techniques and diminished doses providing excellent fusion results equal to if not slightly better than Iliac crest bone graft. Diminished operative time, decreased blood loss, one less operative site and its potential complications with decreased costs of lower BMP doses will fuel future use. Abnormalities related to the use of BMP should be considered when evaluating the post-operative back demonstrating findings suggesting discitis/osteomyelitis or when we see large post-operative fluid collections and/or heterotopic ossification in the post-interbody fusion spine.
References
- Urist MR Bone: formation by autoinduction. Science 1965;150: 893-99 Cross Ref Medicine ↩
- Israel DI, NoveJ, Kerns KM, Moutsatsos IK, Kaufman RJ: Expression and characterization of bone morphogenetic protein-2 in Chineze hamster ovary cells. Growth Factors 1992;7(2):139-150. ↩
- Wosney JM, Rozen V: Bone morphogenetic protein gene family in bone formation and repair. Cli Orthop Relat Res 1998;346:26-37. ↩
- Wozney JM, Rosen V, Celeste AJ, et al: Novel regulators of bone formation: Molecular clones and activities. Science 1988:242(4885):1528-1534. ↩
- Mirza SK, Folly of FDA-approval studies for bone morphogenetic protein. Spine J2011;11(6):492-492. ↩
- Glassman S, Current evidence and future uses of BMP in spinal surgery. Spinal News International3/19/2020 ↩
- Carragee EJ, Bono CM, Scuderi GJ,: Pseudomorbidity in iliac crest bone graft harvesting: The rise if rhBMP-2 in short-segment posterior lumbar fusion.Spine J2009;9(11):873-879. ↩
- Howard JM, Glassman SD, Carreon LY, Posterior iliac crest pain after posterolateral fusion with or without iliac crest graft harvest.Spine J2011 11(6):534-537. ↩
- Ong KL, Villarraga ML, Lau E, Carreon LY, Kurtz SM, Glassman SD: Offf-label use of bone morphogenetic proteins in the United States using administrative data.Spine (Phil Pa 1976)2010; 35(19):1794-1800. ↩
- Carragee EJ, Hurwitz EI, Weiner BK. A critical review of recombinant human bone morphogenetic protein-2 trials in spinal surgery: emerging safety concerns and lessons learned. Spine J 2011;11:471-91 CrossRef Medline ↩
- Personal communication and Miller A. Complications of bone morphogenetic protein-2 use in open transforaminal lumbar interbody fusion. Thesis presented to The Faculty of the School of Medicine Yale University 5/2021 ↩
- Chadi AT, Howard SA. Complications with the use of bone morphogenetic protein-2 in spine surgery. Spine J 2014;14:552-559 ↩
- Rihn JA, Patel R, Makda J, et al: Complications associated with single level transforaminal lumbar interbody fusion. SpineJ2009;9(8):623-6291 ↩
- Sethi A, et al; Radiographic and CT evaluation of recombinant human bone morphogenetic protein-2—assisted spinal interbody fusion.AJR 2011;197:W128-W133. ↩
- Niu S, Anastasio A, Faraj RR, Rhee JM; Evaluation of heterotopic ossification after using recombinant human bone morphogenetic protein-2 in transforaminal lumbar interbody fusion: a computed tomography review of 996 disc levels. Global Spine J2020 May; 10(3): 280-285 ↩
- Beachler DC, Yanik EL, Martin BL, et al. Bone morphogenetic protein use and cancer risk among patients undergoing lumbar arthrodesis: A case-cohort study using the SEER-Medicare database. J Bone Joint Surg Am.2016; 98(13): 1064-72. ↩
- Kelly MP, Savage JW, Bentzen SM, et al. Cancer risk from bone morphogenetic protein exposure in spinal arthrodesis. J Bone Joint Surg Am. 2014; 96(17): 1417-22. ↩