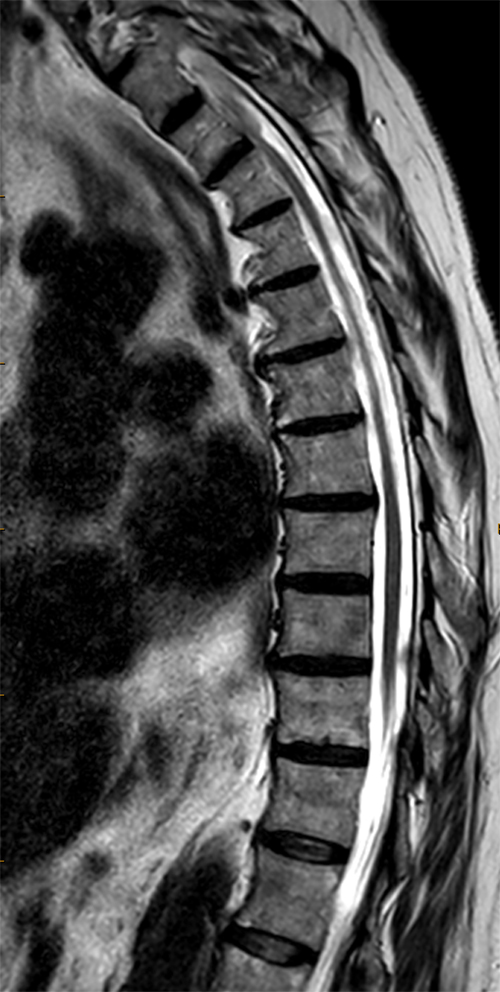
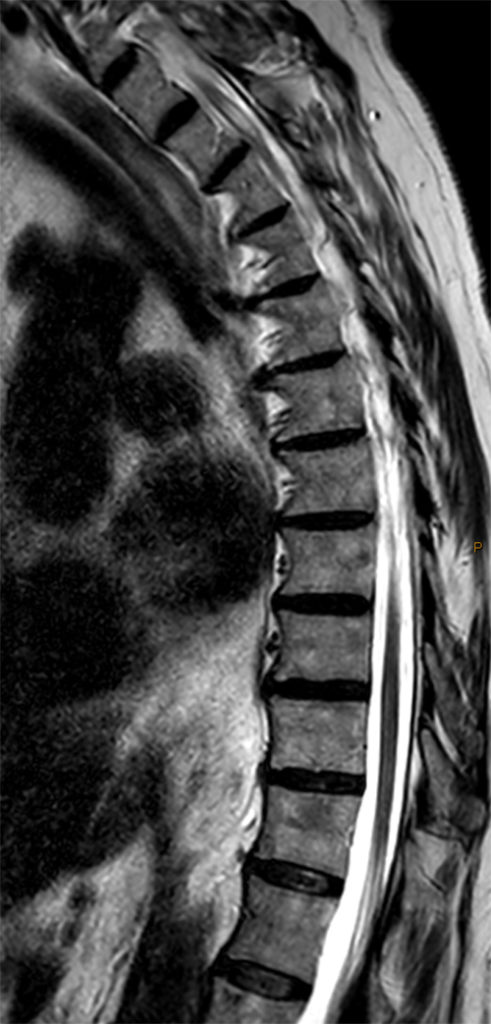
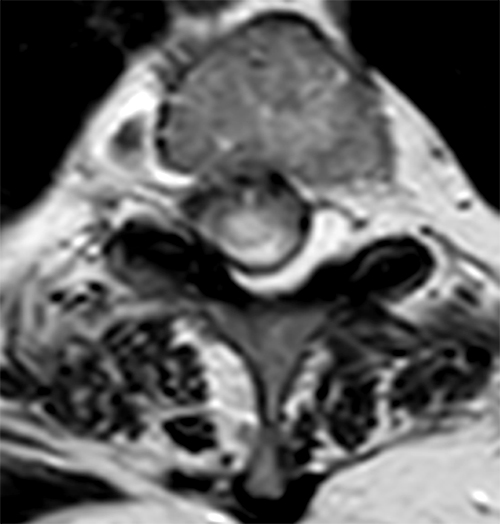
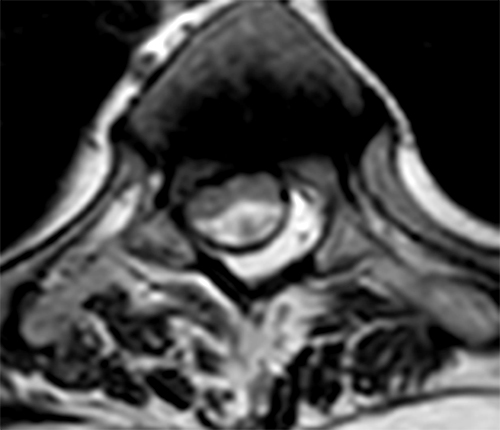
Clinical History: A 76-year-old woman presents with neck and left arm pain. Sagittal T2-weighted (1a and 1b) and axial T2-weighted (1c and 1d) images are provided. What are the findings? What is your diagnosis?
Findings
Figure 2:
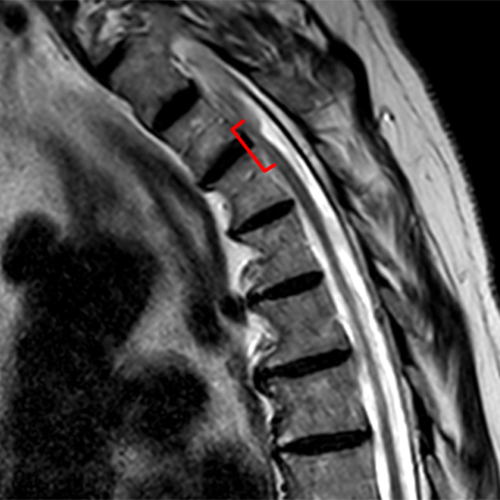
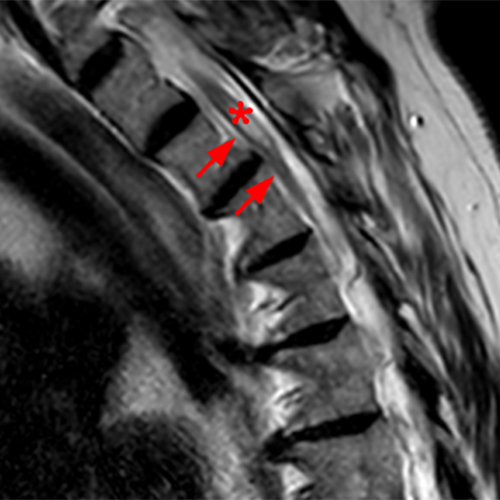
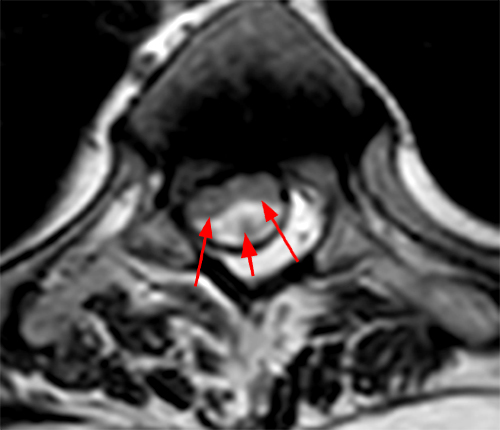
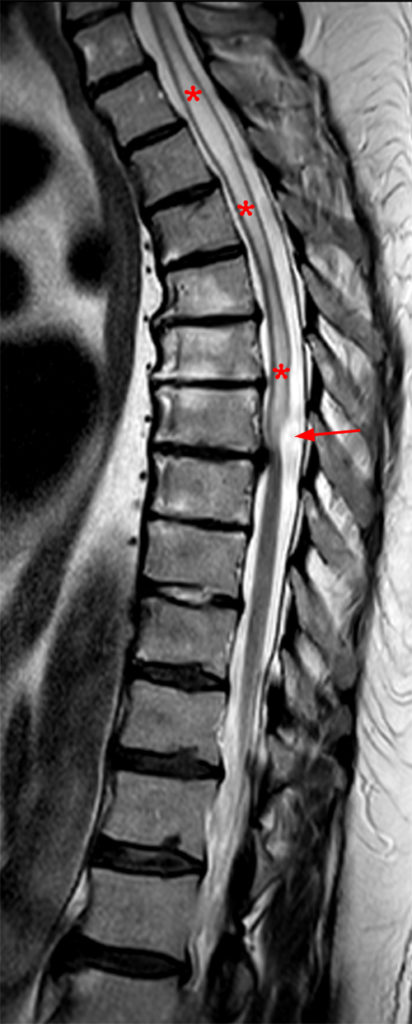
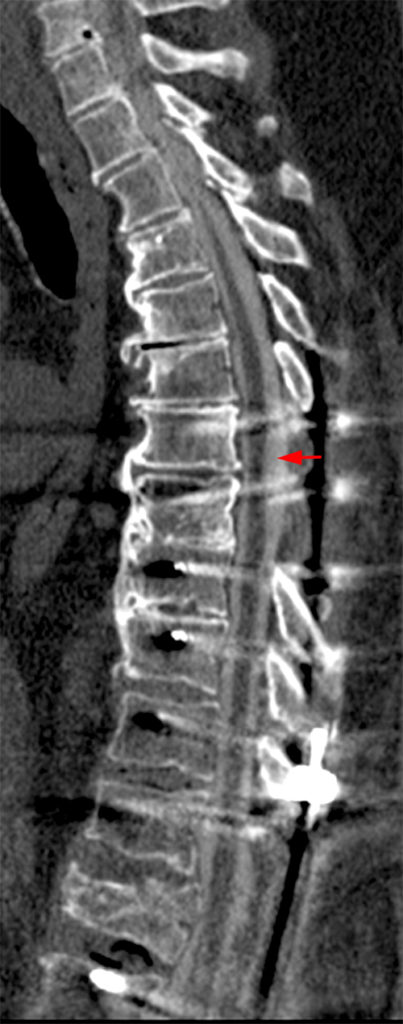
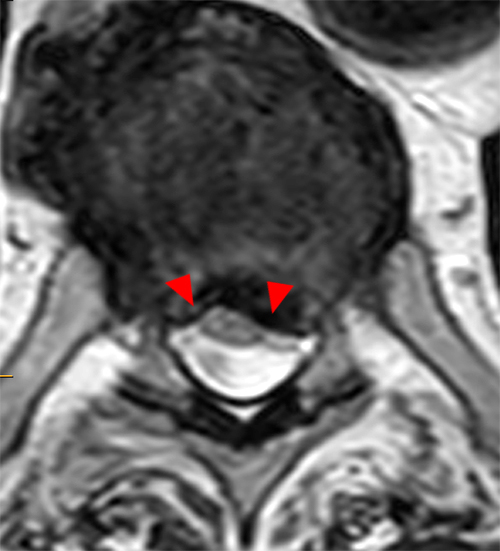
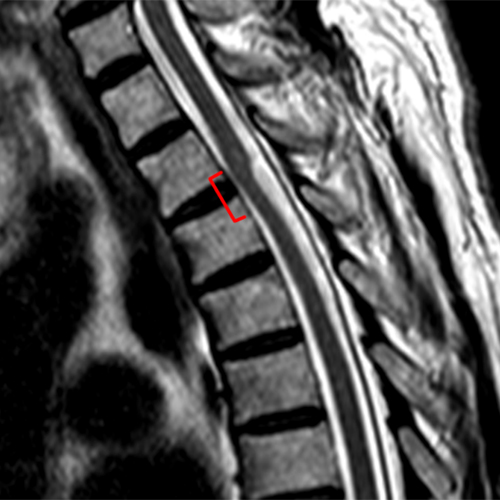
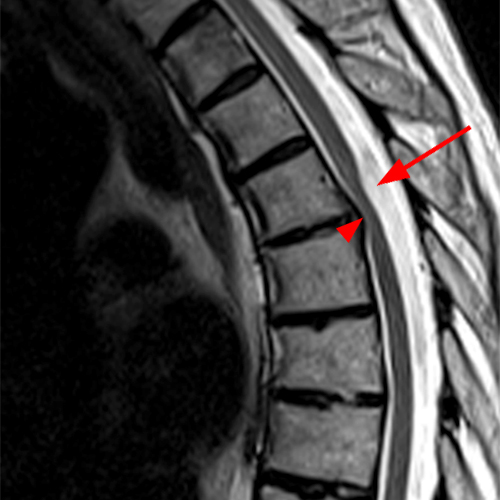
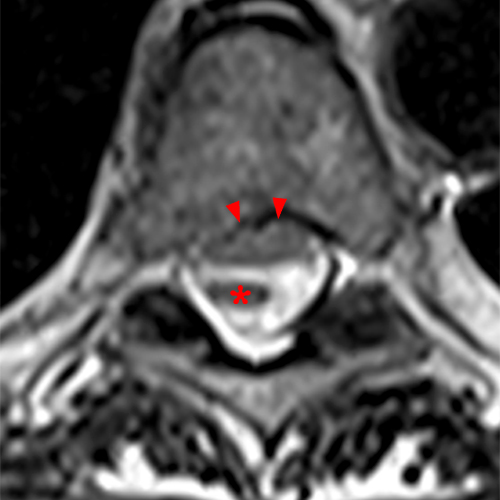
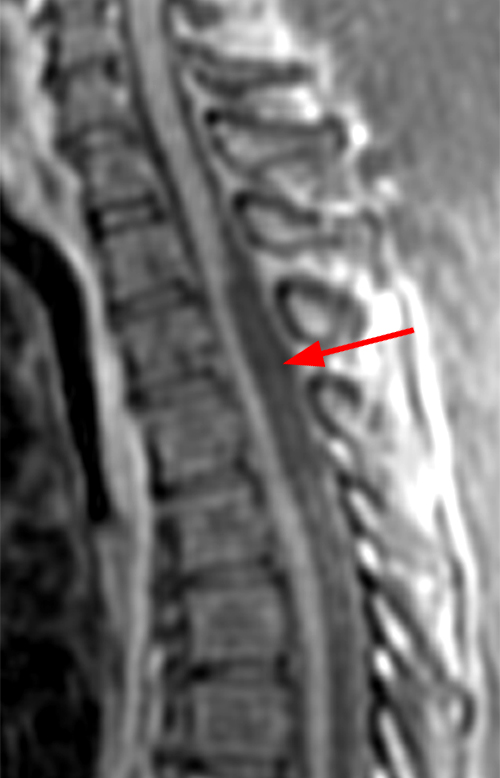
The sagittal T2-weighted images of the thoracic spine (2a) demonstrates a “scalpel sign” at T2-3 (bracket), with focal dorsal indentation of the cord (long arrows) (2d) and enlargement of the dorsal subarachnoid space. CSF pulsation artifact at the level of dorsal subarachnoid space enlargement and the ventral subarachnoid CSF are preserved (short arrows) (2b and 2d). An associated small syrinx (asterisks) is present just superior to the level of cord deviation (2b and 2c). A small central disc protrusion (arrowhead) and annular fissure are also present at T2-3.
Diagnosis
Dorsal arachnoid web (DAW) at T2-3 with an associated small syrinx.
Introduction
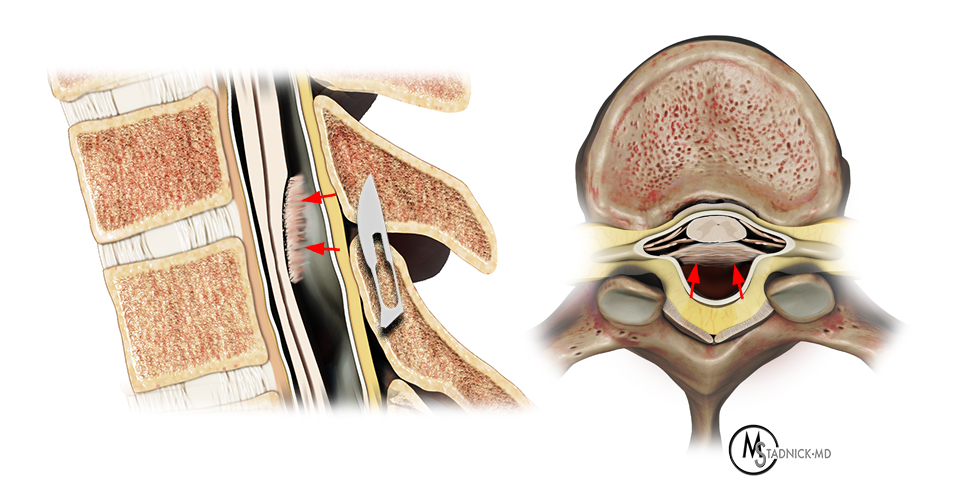
Arachnoid webs are abnormally thickened transverse bands of intradural extramedullary arachnoid tissue along the dorsal aspect of the spinal cord. These webs are believed to develop from the diverticula of the septum posticum, resulting in their dorsal location.1 The mechanism by which arachnoid webs form is not fully understood, with theories including a relationship to arachnoid cyst formation, possibly representing partial, interrupted, or collapsed arachnoid cysts, versus sequelae of localized arachnoiditis, or an idiopathic etiology. Some case reports have suggested a history of trauma. Other theories have proposed post-infectious or post-operative etiologies, as well as congenital processes and a possible association with thickening of the ligamentum flavum.1,2 Dorsal arachnoid webs (DAW) result in disruption of cerebrospinal fluid (CSF) flow in the longitudinal direction via a “one-way valve mechanism” that is not completely obstructive.3,4,5 As a result, the subarachnoid space becomes dilated, producing spinal cord deviation, cord compression, and dorsal cord deformity.2 There may be a spectrum of associated spinal cord signal changes on MRI from pre-syrinx to syringomyelia secondary to the mass effect from CSF accumulation, altered CSF flow dynamics, and CSF pressure differences.2,3,5,6 The resulting cord signal changes/syrinx have been reported to be variable- both cranial and caudal to the web, although a cranial location is reportedly more common.2,4 While reportedly rare, arachnoid webs are most commonly found in the upper thoracic spine, with a female predominance, and age range in the 4th-7th decades.1
Clinical Presentation
There are no clinical signs/symptoms specific for DAWs, and some patients may be asymptomatic with incidental findings on imaging. When symptoms are present, they can include neuropathic back pain, symptoms of compressive myelopathy or radiculopathy (including lower extremity weakness, sensory symptoms, and bowel/bladder incontinence), hyperreflexia, spastic paresis, clonus, and gait instability.1,2,7
MR Imaging
The dorsal arachnoid web itself is not visible on routine MRI imaging due to the limitations of spatial resolution and the relatively thin/small size of the webs compared to the surrounding tissues.1 Sometimes, DAWs may be identified using a CISS sequence. CT myelogram may be useful in the evaluation of DAWs given that the webs are not considered completely obstructive.2 Intraoperative studies may be utilized, including intraoperative ultrasound, on which the webs are seen in the dorsal subarachnoid space, moving in sync with the cardiac cycle, as well as utilization of gentian violet solution, which can confirm CSF flow disruption. Visually, focally thickened arachnoid membrane can be seen intra-operatively.5,8 The enlarged dorsal subarachnoid space, cord deviation, and dorsal contour deformity result in the “scalpel sign” – the appearance of a scalpel with its blade pointing anteriorly. An upside-down scalpel sign may also be identified when the area of cord expansion occurs inferior to the level of the dorsal indentation.1,2,7 As described above, there may or may not be associated myelopathy or syrinx.6
Figure 3:
A 64-year-old man presents with right upper and lower extremity pain and burning. Sagittal T2-weighted images (3a) demonstrate focal dorsal contour deformity and enlargement of the dorsal subarachnoid space (3c), with a “scalpel sign” (long arrow) at T7-8. An associated syrinx extending superiorly throughout the imaged lower cervical and upper thoracic spinal cord (asterisks). Left paracentral/posterolateral and right posterolateral disc protrusions (arrowheads) may account for loss of the ventral subarachnoid space (3c). Following laminectomy and resection of a dorsal arachnoid web, there is decreased cord deformity/displacement (thin arrow) on CT myelography (3b).
Figure 4:
74-year-old man undergoing pre-surgical evaluation for spinal cord stimulator placement for chronic low back pain and right lower extremity radiculopathy. Sagittal T2-weighted image showing a scalpel sign (bracket) at T3-4 with more subtle cord signal abnormality, possibly representing a pre-syrinx. CSF pulsation artifact in the region of subarachnoid space enlargement is preserved, and there is preservation of the ventral subarachnoid space.
Differential
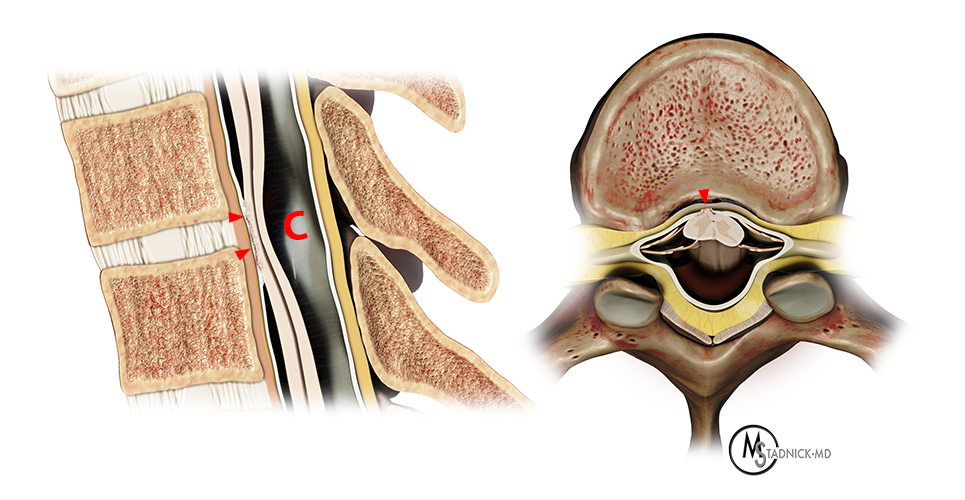
Imaging differentials for focal anterior displacement of the spinal cord with enlargement of the dorsal subarachnoid space include DAW, ventral cord herniation, and dorsal intraspinal arachnoid cyst.4,7 Intraspinal arachnoid cysts and ventral cord herniations can both be associated with cord signal changes, and patients may present with similar clinical symptoms. Idiopathic ventral cord herniation is a rare entity that typically occurs between T2 and T8 when cord tissue herniates through a ventral dural defect and is differentiated from spinal cord herniation associated with a known traumatic or postoperative etiology.9 The dural defect has been reported to be most common at the intervertebral disc level but may occur at the vertebral body level and can rarely span multiple levels. This herniation results in focal cord deformity along the ventral part of the cord and interrupts the ventral subarachnoid space. Protrusion of a segment of cord parenchyma outside of the dura may be seen.4,7,9 Increased turbulence of CSF posterior to the level of the dural defect has been reported, and a “nuclear trail” sign of hyperattenuation on CT when the nucleus pulposus leakage from a herniated disc is calcified may also be present.9 When the ventral cord is closely opposed to the anterior dura, it may be difficult or impossible to differentiate a ventral cord herniation from a dorsal arachnoid web, although the “C sign” has been described and proposed to distinguish between the appearance of the dorsal cord in ventral cord herniation compared to a DAW.5
Figure 5:
Dorsal arachnoid web (5a) versus ventral cord herniation (5b). (5a) Sagittal and axial views show focal dorsal cord deformity from a dorsal arachnoid web (short arrows) resulting in a "scalpel sign" on the sagittal view. CSF ventral to the cord is maintained, and the dorsal subarachnoid space is enlarged. (5b) Sagittal and axial views show loss of the ventral CSF with herniation of a portion of the spinal cord through a small dural defect (arrowheads). Ventral displacement of the spinal cord results in enlargement of the dorsal subarachnoid space and a "C" shaped dorsal cord contour on the sagittal view.
Figure 6:
59-year-old woman presenting with right-sided radiculitis, progressive right arm pain, numbness, and tingling. Sagittal (6a) and axial (6b) T2-weighted images with focal dorsal cord deformity (large arrow) and enlargement of the dorsal subarachnoid space at T-6. There is loss of the ventral subarachnoid space (arrowheads) at this level with a relative “C sign”. CSF pulsation artifact (asterisk) is maintained/more turbulent at the level of dorsal subarachnoid space enlargement. The findings are compatible with ventral cord herniation.
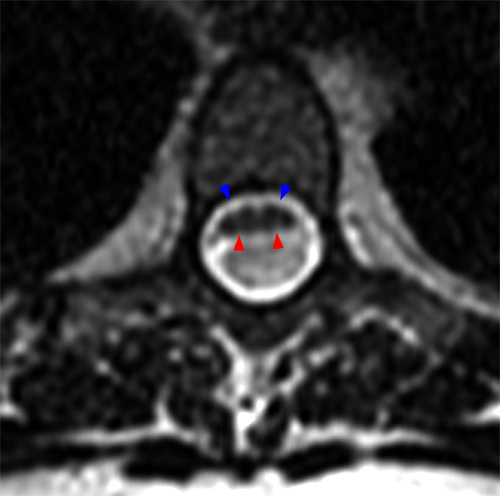
Figure 7:
64-year-old woman presenting for follow-up imaging of a thoracic abnormality identified on C spine imaging following a fall with a neck injury. Sagittal STIR (7a) image demonstrates smooth dorsal scalloping and ventral displacement of the thoracic spinal cord from T2-3 through T5 (bracket). The region of enlarged dorsal subarachnoid space demonstrates a loss (short arrows) of CSF pulsation artifact (7a). No associated abnormal enhancement is present (arrow) on postcontrast imaging (7b). There is associated dorsal cord flattening (red arrowheads) with maintenance (blue arrowheads) of ventral CSF on a corresponding axial view (7c).
Conclusion
Dorsal arachnoid webs (DAW) partially obstruct CSF flow resulting in focal dorsal cord deformity and enlargement of the dorsal subarachnoid space. Because the clinical features and imaging findings overlap, they can be difficult to distinguish from dorsal intraspinal arachnoid cysts and ventral cord herniation on routine MRI. Given that DAWs are most commonly found in the upper thoracic spine, they may be visible at the lower margin of the field of view on cervical spine imaging. Management can include surgical resection and/or fenestration, with the aim to restore normal CSF flow and prevent progression of symptoms.7 Regression of the scalpel sign, cord signal changes, and patient symptoms has been reported postoperatively. Some patients may have incidental MRI findings consistent with a DAW, with or without signal changes in the cord. In such cases, conservative treatment with clinical monitoring using routine imaging and neurophysiology is appropriate. Therefore, an individualized approach to treatment should be utilized.1,7
References
- Buttiens A, Feyen B, Dekeyzer S. Dorsal arachnoid web: a rare cause of myelopathy. Journal of the Belgian Society of Radiology 2021; 105(1):88, 1–3. ↩
- Ali HB, Hamilton P, Zygmunt S, Yakoub KM. Spinal arachnoid web—a review article. J Spine Surg 2018;4(2):446-450. ↩
- Inoue J et al. Diagnosis and surgical treatment of thoracic dorsal arachnoid web: a report of two cases. Case Rep Orthop 2020, Sep12;2022:8816598. ↩
- Aiyer R, Voutsinas L, El-Sherif Y, J Neurol Neuromed (2016) 1(6):66-68. ↩
- Voglis S et al. Spinal arachnoid web- a distinct entity of focal arachnopathy with favorable long-term outcome after surgical resection: analysis of a multicenter patient population. The Spine Journal 2022 (22):126-135. ↩
- Reardon MA et al. Dorsal thoracic arachnoid web and the “scalpel sign”: A distinct clinical-radiologic entity. AJNR 34:1104-10 ↩
- Brasil PM et al. Imaging Findings in dorsal thoracic arachnoid web and the differential diagnosis of “scalpel sign”. Neurographics 2020 March/April;10(2):96–102. ↩
- So TY et al. Dorsal thoracic arachnoid web and spinal cord compression. Applied Radiology Sept-Oct 2020, 52:52. ↩
- Parmar H et al. Imaging of idiopathic spinal cord herniation. RadioGraphics 2008; 28:511-518. ↩