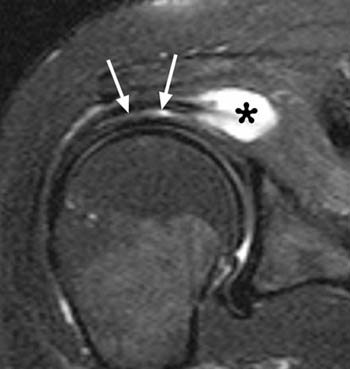
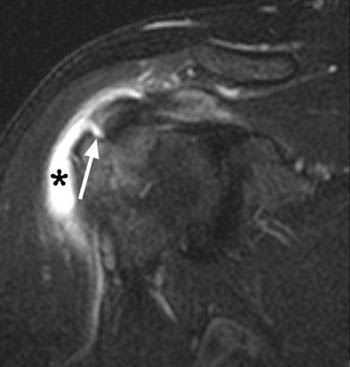
Clinical History: A 53 year-old female presents with shoulder pain. A fat-suppressed coronal T2-weighted image (1a) and sagittal T2-weighted image (1b) are provided. What are the findings? What is your diagnosis?
Findings
Figure 2:
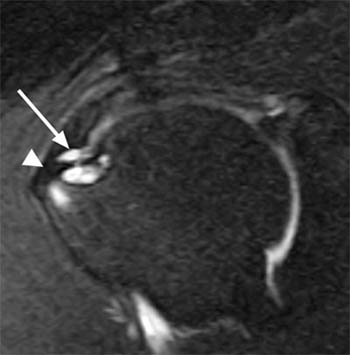
(2a) The fat-suppressed coronal T2-weighted image demonstrates a focal bursal surface fluid-filled defect of the supraspinatus portion of the rotator cuff (arrow). (2b) The sagittal T2-weighted image also demonstrates focal fluid signal within the cuff defect (arrow) extending to the cortex of the greater tuberosity (arrowhead).
Diagnosis
Bursal-sided partial-thickness tear of the supraspinatus tendon.
Introduction
Partial-thickness rotator cuff tears are estimated to be twice as common as full-thickness tears.1 Partial-thickness tears of the rotator cuff are frequently symptomatic and may increase in size or progress to full thickness tears if left untreated. Partial-thickness tears can be more difficult to detect than full-thickness tears with MRI, but careful analysis of the imaging findings and an understanding of cuff anatomy allow a precise description of partial thickness tears. Accurate reporting of such tears is important in the choice of operative or non-operative therapy in these patients.
Anatomy and Histology
The rotator cuff is formed by the confluence of tendons, the joint capsule, and coracohumeral ligament and glenohumeral ligament complexes, all of which blend before inserting onto the humeral tuberosities. The supraspinatus and infraspinatus tendons join 15 mm proximal to their insertion and are not readily separated by blunt dissection. Anatomic studies have detailed the surface area of tendon insertion, known as the rotator cuff footprint (3a, 4a). The supraspinatus portion of the cuff inserts on and covers the anterosuperior aspect of the greater tuberosity. The typical medial to lateral insertion width of the supraspinatus tendon is 12.7 mm, covering the width of the superior facet of the greater tuberosity. The distance between the articular-sided inserting fibers of the cuff and the articular cartilage of the humeral head is less than 1mm over an AP dimension of 2 cm, corresponding to the insertion of the supraspinatus and anterior infraspinatus tendons. This distance gradually widens over the more posteriorly inserting cuff, increasing to approximately 13.9 mm at the inferior aspect of the teres minor insertion.2
Figure 4:
(4a) A coronal T1-weighted image demonstrates the normal supraspinatus insertion, covering the superior facet of the greater tuberosity (line). This area of coverage is often referred to as the rotator cuff footprint. Note the lateral extension of the joint space and the close proximity of the humeral head articular cartilage to the medial insertion of the cuff (arrowhead).
The supraspinatus and infraspinatus portions of the rotator cuff demonstrate 5 distinct histologic zones.3 The tendon contributions to the cuff are contained in layers II and III, with each layer demonstrating different collagen fiber orientation and biomechanical properties (E). Indeed, the stress failure point of the articular half of the rotator cuff is approximately half of the bursal half. These differences, particularly between layers II and III, are felt to contribute to the pathogenesis of certain types of partial tears.
The articular surface of the rotator cuff tendon is relatively hypovascular. Arterioles are larger and more prevalent on the bursal surface, branching and decreasing in size between layers II and III. A “critical zone” of hypovascularity has also been described, just proximal to the cuff insertion. This region of the cuff lies between the blood supply to from the musculotendinous junction and from the bone at the tendon-bone interface.
Figure 5:
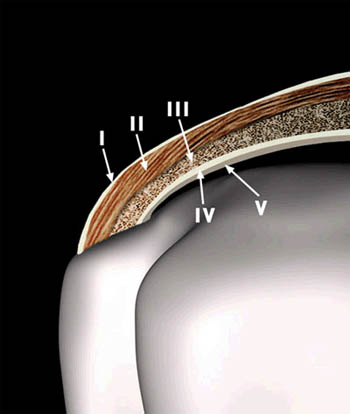
(5a) A 3D graphic of the supraspinatus insertion corresponds to the coronal imaging plane and depicts the histologic layers of the cuff. Layer I is the most superficial layer, measuring 1 mm in thickness and composed of fibers from the coracohumeral ligament which extend posteriorly and obliquely. Layer II is composed of densely packed fibers that parallel the long axis of the tendon and measures 3-5 mm in thickness. Layer III is 3 mm thick and composed of smaller bundles of collagen which are loosely organized at an approximately 45 degree angle to the long axis of the tendon. Layer IV is composed of loose connective tissue and thick collagen bands and merges with fibers from the coracohumeral ligament. Layer V represents the shoulder capsule and measures approximately 2 mm in thickness.
Mechanism of Injury
Several theories have been postulated to explain the complex pathogenesis of rotator cuff tears. Extrinsic causes include subacromial impingement, shoulder instability, internal impingement, shoulder instability, a single traumatic event or repetitive microtrauma. Intrinsic causes include age-related degenerative changes associated with metabolic and vascular alterations. In reality, partial-thickness rotator cuff tears are most frequently due to a combination of causes.
The extrinsic theory of rotator cuff tears, postulated by Neer, emphasizes narrowing of the supraspinatus outlet as the cause of rotator cuff tears. Abnormalities of the coracoacromial arch lead to mechanical impingement upon the cuff and may eventually lead to rotator cuff tears. While most clearly associated with bursal surface partial-thickness tears, subacromial impingent may lead to articular and intrasubstance tears by the creation of differential shear stresses which affect the layered anatomy of the cuff.4
Trauma may lead to rotator cuff tears, either from a single event such as a direct fall, or as a result of repetitive microtrauma often encountered in overhead athletes and laborers. Glenohumeral joint instability and traction stresses, particularly in throwing athlete, can lead to articular-sided tears.
The intrinsic theory of rotator cuff tears attributes the development of tears to tendinopathy and a failure of collagen fibers. Age-related degenerative changes such as decreased cellularity and fascicular thinning lead to weakening of the cuff. The relative lack of vascularity on the articular surface and in the “critical zone” predisposes to degenerative tears with aging. In addition, lack of uniformity of the collagen bundles in layer III are felt to predispose to articular surface tearing and delamination tears.
Natural History
Partial thickness rotator cuff tears increase in frequency with increasing age and are often compatible with normal painless functional activity. However, when symptomatic, up to 80% of partial thickness tears will progress if treated nonoperatively.
Articular-sided partial thickness tears are 2-3 times more frequent than bursal surface tears. In general, the functional prognosis is poorer with increasing age, larger initial tear size, and an atraumatic presentation.
Intratendinous partial thickness tears are felt to be approximately twice as common as articular and bursal-sided tears partial thickness tears.4 These tears represent a delamination of the tendon fibers, most commonly between layer II and III and can be entirely contained within the tendon or communicate with the cuff surface. When the latter occurs, the tear most commonly involves the articular surface of the cuff. Intratendinous tears can lead to altered stress concentration on the articular side of the supraspinatus tendon, contributing to the development of articular-sided tears.4 Intratendinous tears may develop a synovial lining, propagating motion across the tear and inhibiting healing. Thus to most fully restore normal biomechanics to the cuff and reduce the risk of repair failure or a poor operative outcome, the intratendinous component of partial thickness tears must be repaired.
Bursal sided partial thickness tears are more often associated with impingement. These tears are commonly seen at the tendon-bone interface. Though less common than articular-sided tears, bursal-sided tears are frequently the most symptomatic.
MRI
The normal rotator cuff tendon is of low signal on T1 and T2-weighted images. Partial- thickness rotator cuff tears most commonly appear as interruption of the normal cuff contour, resulting in a cuff defect filled with fluid signal. The multiplanar capability afforded by MRI allows assessment of the articular, bursal, and intratendinous components of the tear. Because many partial-thickness rotator cuff tears occur at or near the cuff insertion, utilizing the concept of the rotator cuff footprint allows a more accurate determination of the thickness of the tear.
Articular-sided partial thickness tears appear as linear fluid signal defects at the articular surface. These frequently occur near the supraspinatus tendon-bone interface in young patients or athletes. In these patients the margins of the tear are often sharply defined and the tear is felt to produce pain because of the unstable edge (6a). Articular-sided partial thickness tears at the tendon-bone interface may also occur in the elderly population as a degenerative process. These tears may be more difficult to identify, because frequently the tendon contour is smooth along the torn surface. These tears can be recognized by the cuff attaching only to the more superficial portion of the greater tuberosity, leaving the deeper portion of rotator cuff footprint on the greater tuberosity bare (7a).
Figure 6:
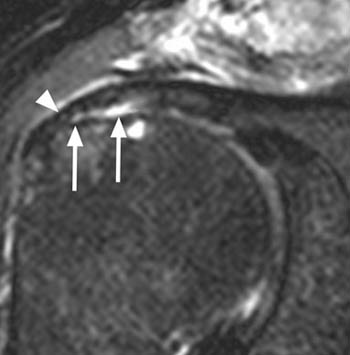
(6a) This coronal fat-suppressed T2-weighted image through the supraspinatus insertion demonstrates a fluid filled defect at the articular surface (arrow), extending through greater than 50% of the cuff thickness. The tear involves the tendon-bone interface at the articular surface and extends slightly into the distal substance of the supraspinatus tendon. The bursal sided fibers remain intact and attached at the rotator cuff footprint (arrowhead).
Figure 7:
(7a) A coronal fat-suppressed T2-weighted image demonstrates an articular-sided partial thickness tear of the supraspinatus. This tear is less well-defined but fluid signal can be seen extending over the exposed medial portion of the greater tuberosity (arrows). The remaining cuff is abnormally thinned and the bursal-sided fibers are seen attaching to the greater tuberosity (arrowhead).
Intratendinous tears are recognized as fluid-signal defects that parallel the long axis of the tendon. These tears are also known as delamination tears and typically occur between the superficial and deep layers of the tendon. Delamination tears may remain entirely within the substance of the cuff without extension to the articular or bursal surface (8a). Frequently, these are associated with extension to the articular surface (9a). Because detection of these tears requires fluid signal in the cuff defect, MR arthrography with or without ABER positioning has been advocated as a means for improved detection of this type tear. The delamination tear may extend over great distances, presenting as a cystic collection in the muscle or at the musculotendinous junction (10a). The intratendinous tear associated with such a cystic collection most commonly communicates with an articular-sided tear.1
Figure 8:
(8a) A coronal fat-suppressed T2-weighted image through the supraspinatus portion of the cuff demonstrates linear fluid signal paralleling the long axis of the tendon representing an intratendinous tear (arrow). This intratendinous tear does not communicate with the articular or bursal surface.
Figure 9:
(9a) This coronal fat-suppressed T2-weighted image demonstrates a partial thickness articular-sided tear with a delamination tear (arrowheads). Note the thinned remaining bursal half of the supraspinatus tendon (arrows) and the retracted articular-sided torn cuff fibers (short arrow). Acromioclavicular joint hypertrophy and arthritis (asterisk) are readily apparent.
Bursal surface tears most commonly occur at the tendon-bone interface as a sharply defined linear fluid filled cuff defect (11a). Because these tears are frequently associated with impingement, associated bursitis is often encountered, and should not be misinterpreted as decompression of joint fluid into the subacromial-subdeltoid bursa via a full-thickness cuff tear.
Figure 11:
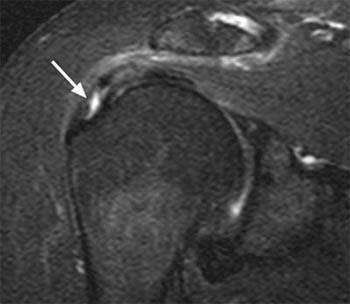
(11a) A fat-suppressed coronal T2-weighted image demonstrates a linear fluid filled defect extending to the bursal surface of the cuff at the tendon-bone interface (arrow) compatible with a bursal-sided partial tear. The more medially inserting fibers of the cuff remain attached at the greater tuberosity. Subdeltoid bursal fluid is present compatible with bursitis (asterisk).
Treatment
The treatment approach to partial-thickness tears is complex and requires assessment of not only the rotator cuff but also the age, health, and desired level of function of the patient. In general, tears greater than 50% of the cuff thickness are repaired. Many surgeons now advocate a more aggressive repair for active individuals using a 30% thickness threshold to preserve the footprint and biomechanics of the cuff.5 Double row suture techniques have improved the tendon-bone surface contact area providing a more stable cuff repair.6 Bursal-sided tears are repaired more aggressively because of a more favorable outcome over debridement alone. Intratendinous tears can significantly alter the surgical approach to rotator cuff repair. These tears are repaired by opening the tear on the bursal surface and debriding the devitalized tissue before the tendon defect is repaired by mattress sutures.5
Conclusion
With a better understanding of the biomechanics of the rotator cuff and of the importance of restoring the footprint of the cuff tendons, orthopaedic surgeons are becoming more aggressive in their approach to rotator cuff repair. MRI provides vital information about the location, size, and depth of rotator cuff tears and the quality of the surrounding tendon, allowing more comprehensive surgical planning and improved outcomes.
References
1 Resnick D, Kang HS, Pretterklieber ML. Shoulder. In: Resnick D, Kang HS, Pretterklieber ML. Internal Derangements of Joints, 2nd Ed. Philadelphia, Pennsylvania: Elsevier; 2007: 713-1122.
2 Dugas JR, Campbell DA, Warren RF, Robie BH, Millett PJ. Anatomy and dimensions of rotator cuff insertions. J Shulder Elbow Surg. 2002;11:498- 503.
3 Clark JM, Harryman DT. Tendons, ligaments, and capsule of the rotator cuff. Gross and microscopic anatomy. J Bone Joint Surg. Am. 1992;74:713-725.
4 Matava MJ, Purcell DB, Rudzki JR. Partial-Thickness Rotator Cuff Tears. Am J Sports Med. 2005;33:1405-1417.
5 Stetson WB, Ryu RKN, Bittar ES. Athroscopic Treatment of Partial Rotator Cuff Tears. Operative Techniques in Sports Medicine. 2004;12:135-148.
6 Lo IK, Burkhart SS. Double-row arthroscopic rotator cuff repair: re-establishing the footprint of the rotator cuff. Arthroscopy. 2003;19:1035-1042.