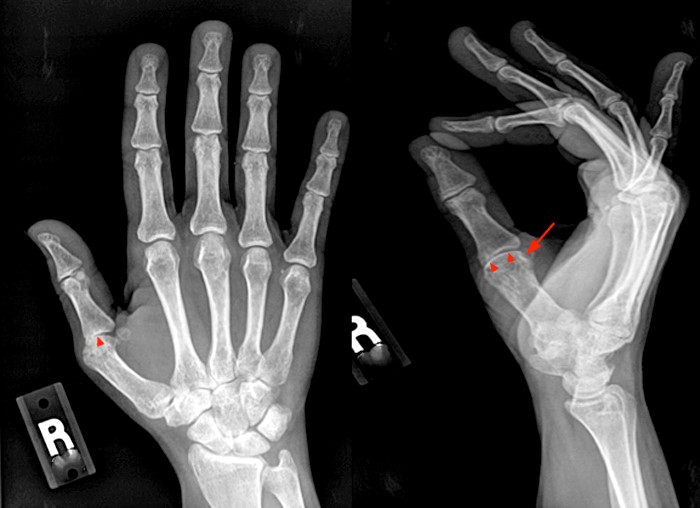
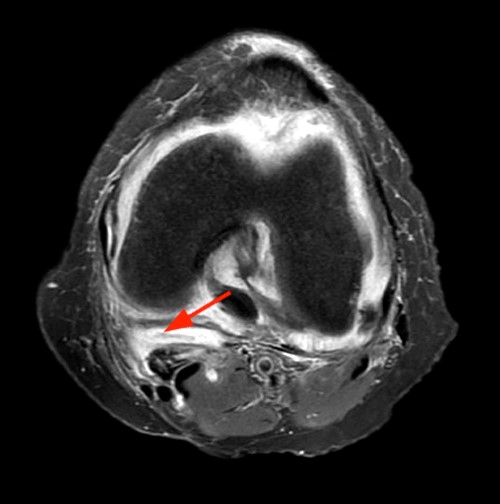
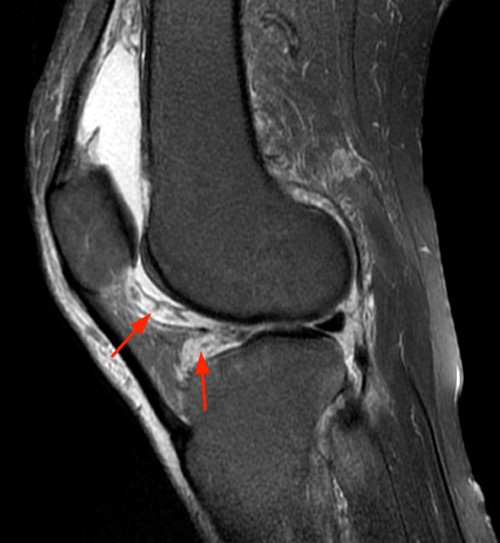
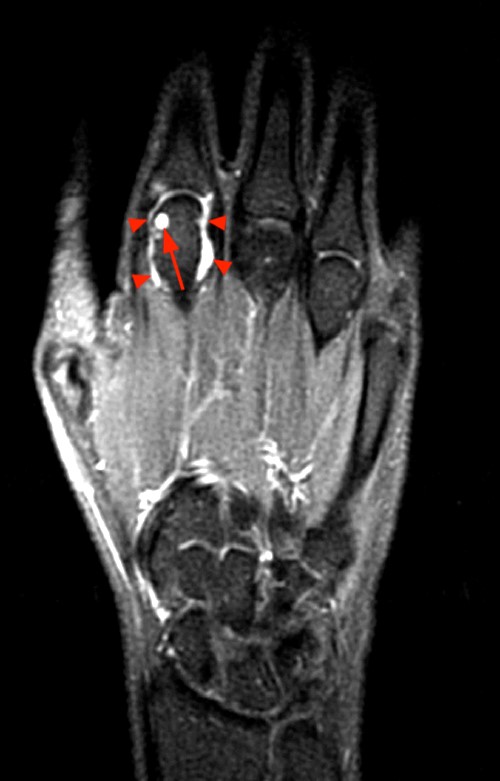
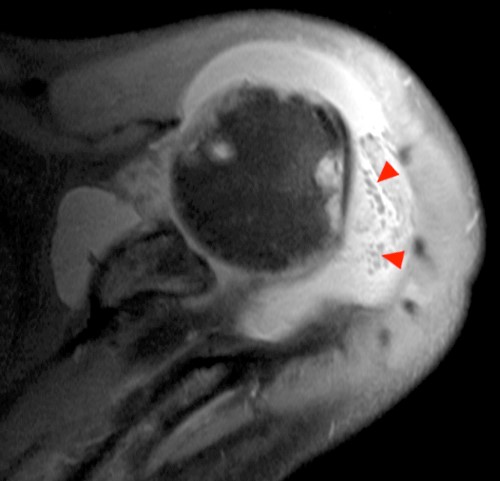
Clinical History: A 47 year-old woman presents with polyarthralgias and morning stiffness. Images provided include a coronal T1-weighted (1a), coronal fat-suppressed T1-weighted post contrast (1b), axial fat-suppressed T1-weighted post contrast at the level of the carpal tunnel (1c), and an axial fat-suppressed T2-weighted image at a level just proximal to the radiocarpal joint (1d). What are the findings? What is your diagnosis?
Findings
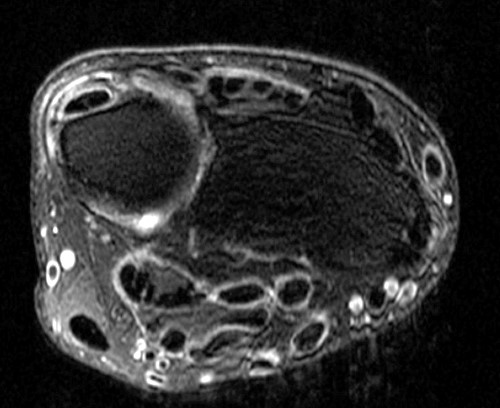
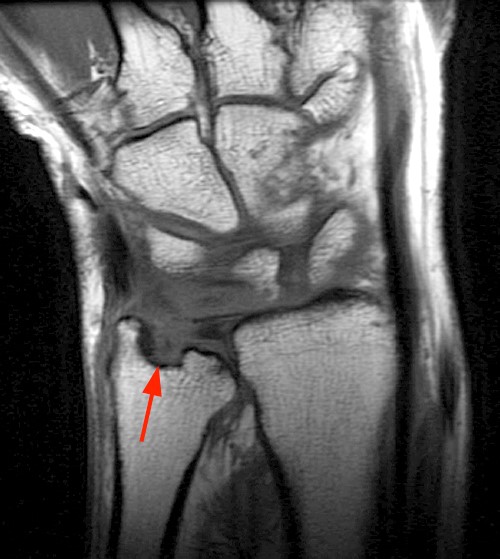
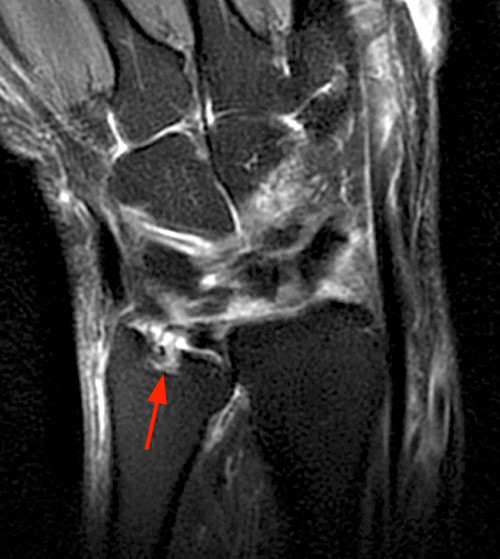
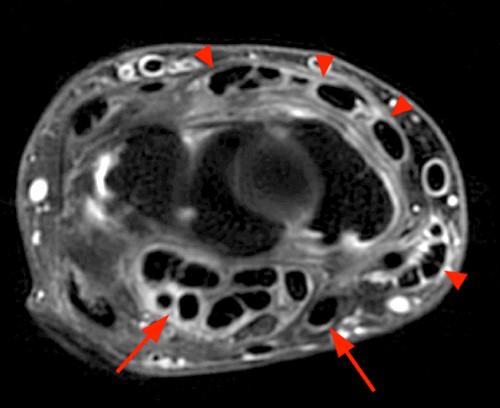
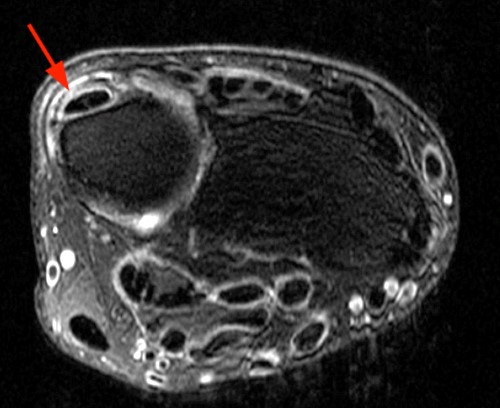
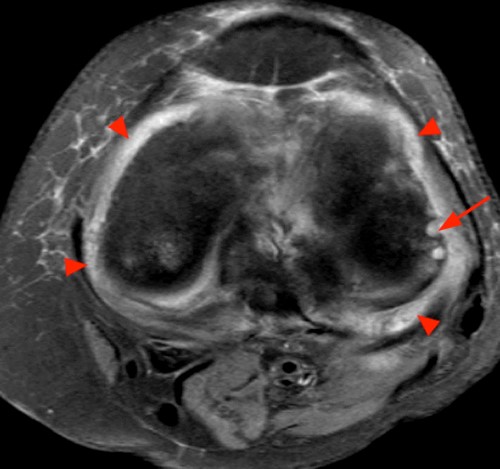
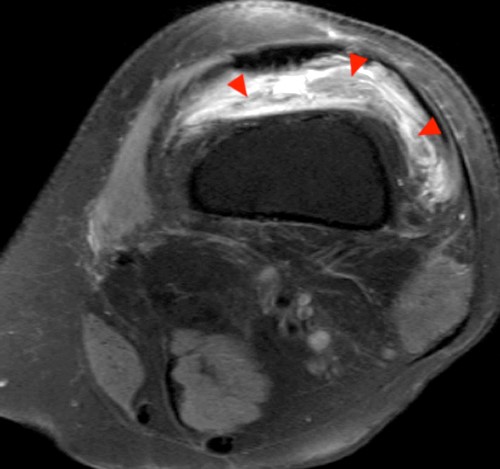
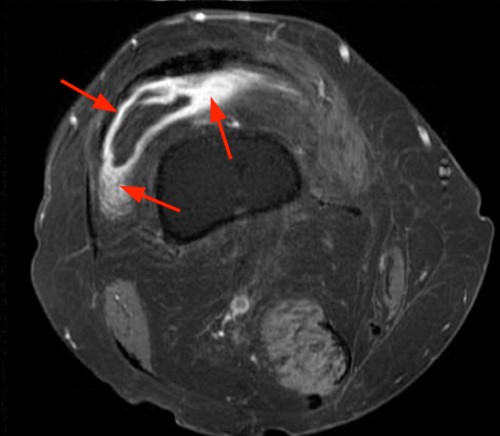
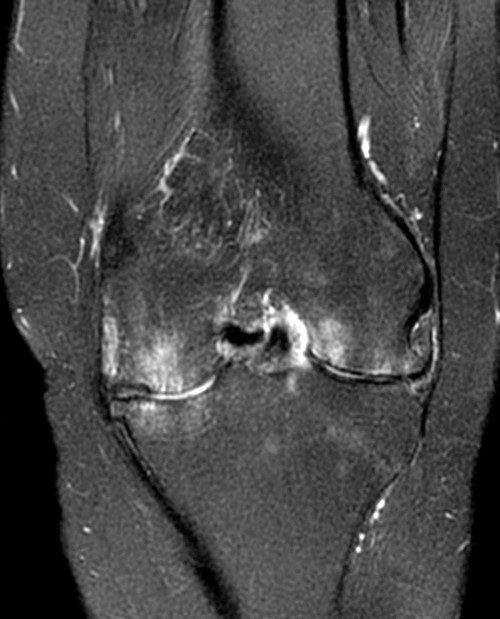
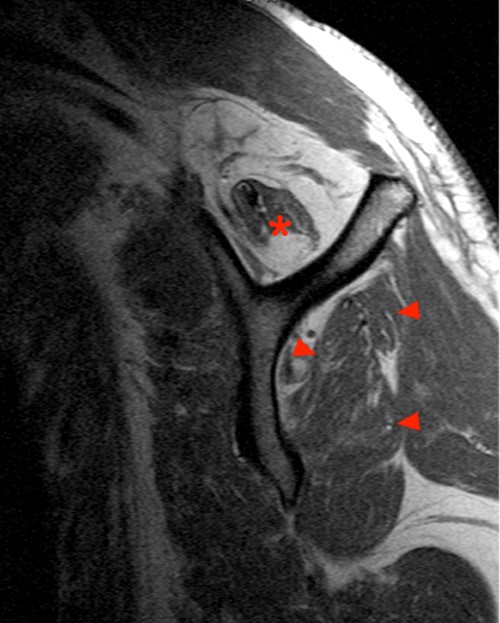
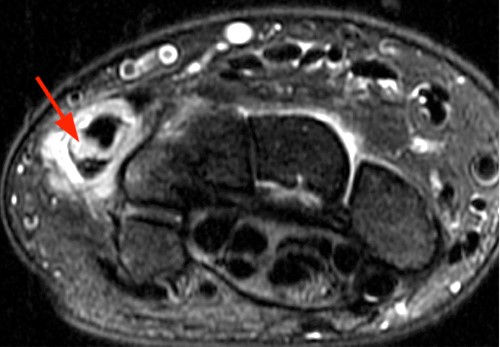
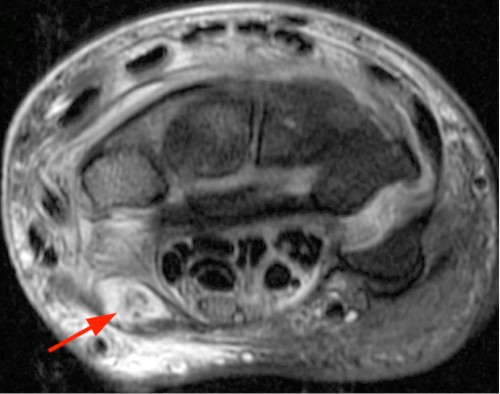
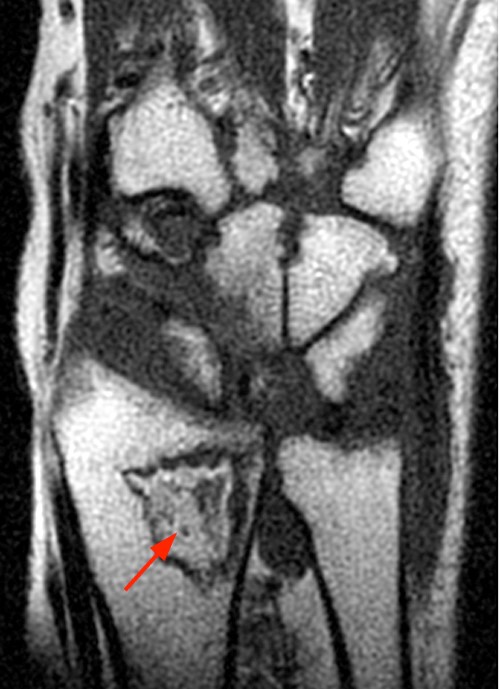
Figure 2:
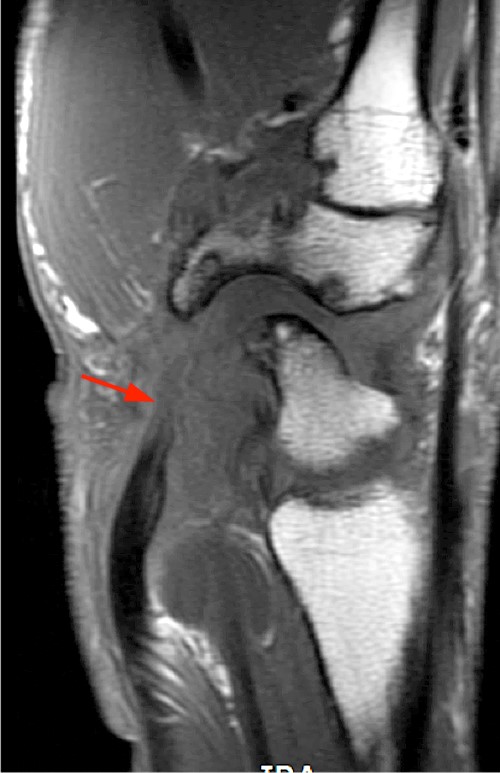
The coronal T1-weighted image (2a) and post-contrast coronal T1-weighted image with fat suppression (2b) demonstrate a large erosion in the distal ulna (arrows). The coronal post-contrast image (2b) demonstrates extensive synovitis about the carpal bones, especially in the radiocarpal and midcarpal compartments with additional synovitis in the distal radioulnar joint including the region of the ulnar erosion. The post-contrast axial T1-weighted image with fat suppression at the level of the carpal tunnel (2c) shows evidence of radial and ulnar bursitis surrounding the flexor tendons (arrows) and peritendinous enhancement surrounding the tendons traversing the 1st through 4th dorsal compartments (arrowheads). The axial T2-weighted image with fat suppression (2d) at the level of the distal radioulnar joint demonstrates a focal defect in the subsheath of the extensor carpi ulnaris tendon along its dorsoulnar aspect, consistent with a subsheath injury (arrow).
Diagnosis
The constellation of findings that includes synovitis in several compartments of the wrist, an erosion in the distal ulna, tenosynovitis in multiple dorsal extensor tendon compartments, and radial and ulnar flexor bursitis are consistent with rheumatoid arthritis.
Introduction
Rheumatoid Arthritis (RA) is a disorder that primarily affects the synovium in all areas of the body. Its incidence is highly variable. Based primarily on population-based studies, the annual incidence of RA has been estimated at 35.9 for women and 14.3 for men per 100,000 population.1 The prevalence of RA in adults varies among men and women, occurring in 0.74% and 1.34%, respectively.2 The cause of RA is not known but multiple factors have been emphasized in the recent literature. Genetic factors appear to have an important causative role in persons who develop RA; approximately 70% of patients with RA express HLA-DR4. Multiple other factors such as smoking, infectious agents, and hormones have also been implicated in the causation of RA but their exact impact upon the pathophysiology of RA is not yet clear.1,2
Anatomy and Histology of Normal Synovium
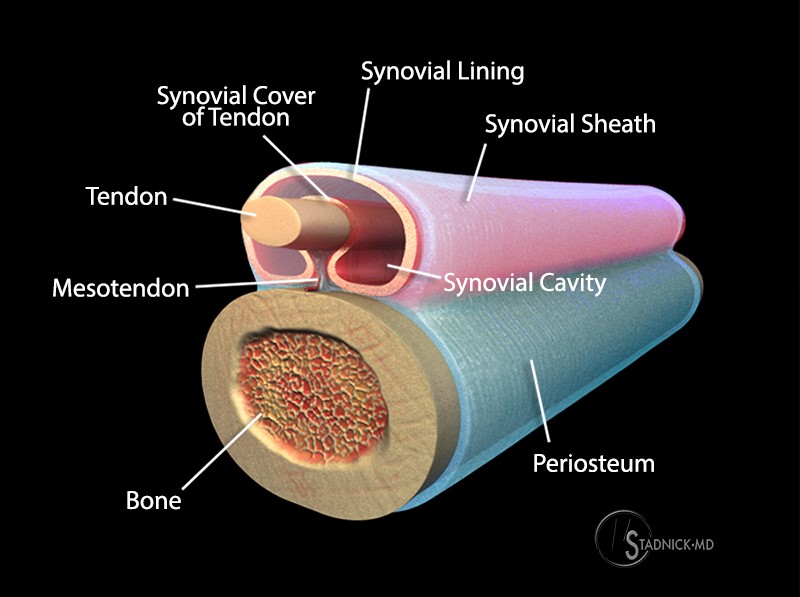
The synovium is the primary site of disease in RA, and an understanding of the anatomy and histology of the synovium will serve as the basis for a discussion of the pathophysiology of RA. The synovium is a thin, highly vascularized membrane that lines diarthrodial joints, tendon sheaths, and some but not all bursae. It contains an intimal layer that is composed of macrophages and fibroblasts and a subintimal layer that contains a blood and lymphatic supply as well as a supporting matrix.3 More specifically, there are two types of intimal cells: A cells that are derived from macrophages as well as the bone marrow; and B cells that are mesenchymal in origin and arise from fibroblasts.4,5 Macrophages are also found in the subintimal layer, but overall these cells represent a minority of the cells that are found in the synovium. Type B intimal cells, or synoviocytes, are responsible for the production of hyaluronan, which is the primary constituent of synovial fluid. Normally, the ratio of Type A/Type B synoviocytes is very low; however, in pathologic conditions such as inflammatory arthritis, type A cells predominate in the intimal lining. The normal synovium functions to allow movement of non-compliant tissues by way of synthesizing synovial fluid, allowing non-adherent motion. Further, the synovium acts to lubricate cartilage as well as to provide nutrition to chondrocytes.6
Pathophysiology
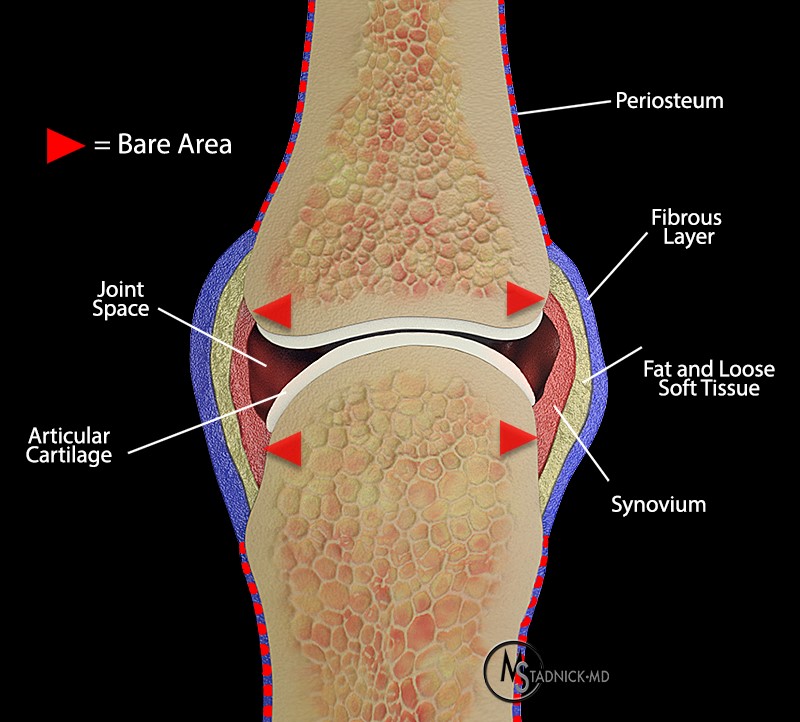
The pathophysiology of the articular manifestations of RA is based upon synovial hyperplasia. As noted earlier, the precise inciting event has not yet been clearly elucidated. Synovial hyperplasia results in significant overgrowth of the intimal layer, with reversal of the normal proportion of A and B synoviocytes. A cells (those of macrophage lineage) predominate in the intima. Various cytokines and immune modulators, including TNF-alpha and IL-1, play a pivotal role in the pathophysiology of RA. These immune modulators act upon neighboring structures, inducing the release of numerous enzymes that erode bone and cartilage and cause damage to tendons and soft tissues. The diseased synovium of RA has been likened to neoplastic tissue on a molecular level. Studies have shown that numerous mutations occur in oncogenes as well as tumor-suppressor genes such as p53, creating a molecular transformation of synovial cells. Isolated colonies of RA cells have shown independent growth and loss of contact inhibition, as well as telomerase activity. Hypertrophied synovium, the so-called “pannus”, enlarges to a point where it causes local invasion of tissue. In joints, this most commonly involves the bare areas of the articulation. These bare, or unprotected, areas are found at the margins of the articulation where no protective layer of cartilage is present. Further disease involvement results in cartilage destruction and joint space loss. In a similar fashion, tenosynovitis results from synovial lining hyperplasia about tendons, especially in the hand, wrist, ankle, and foot. When severe, tenosynovitis may lead to tendon failure. Left untreated, chronic RA affecting joints, tendons, and bursae produces joint mutilation, severe limb deformities, and secondary osteoarthritis.5, 6, 7, 8, 9
Classification Criteria
The most widely accepted criteria were established in 1987 by the American College of Rheumatology (ACR). These criteria were utilized in those patients with an established diagnosis of RA.10
[su_table]
| Criterion | Definition |
| Morning Stiffness | Morning stiffness in and around joints, lasting at least 1 hour before maximal improvement |
| Arthritis of 3 or more joint areas | At least 3 joint areas simultaneously have had soft tissue swelling or fluid observed by a physician. The 14 possible areas are right or left PIP, MCP, wrist, elbow, knee, ankle and MTP joints |
| Arthritis of hand joints | At least 1 area swollen in a wrist, MCP, or PIP joint |
| Symmetric arthritis | Simultaneous involvement of the same joint areas (as defined in 2) on both sides of the body (bilateral involvement of PIP’s, MCP’s, or MTP’s in acceptable without absolute symmetry) |
| Rheumatoid nodules | Subcutaneous nodules, over bony prominences, or extensor surfaces, or in juxtarticular regions, observed by a physician |
| Serum rheumatoid factor | Demonstration of abnormal amounts of serum rheumatoid factor by any method for which the result has been positive in <5% of normal control subjects |
| Radiographic changes | Radiographic changes typical of rheumatoid arthritis on posteroanterior hand and wrist radiographs, which must include erosions or unequivocal bony decalcification localized in or most marked adjacent to the involved joints (changes related to osteoarthritis do not qualify) |
[/su_table]
The 1987 criteria fill an important role in that they define the clinical manifestations of RA and how one may differentiate RA from other causes of joint disease. Nonetheless, the treatment of RA has been dramatically advanced by the optimal use of disease-modifying antirheumatic drugs (DMARDS) as well as by the availability of biologic agents.11, 12, 13, 14 Such treatment has necessitated the need for earlier diagnosis, as therapy in the early stages of the disease has been shown to dramatically alter the clinical course of RA. Based only on these 1987 criteria, clinical trials assessing treatment options for RA would be hampered by the lack of criteria allowing for study enrollment of patients in the early stages of disease.11 Therefore, the ACR and the European League Against Rheumatism (EULAR) created a new approach for the classification of RA. The new classification criteria can be applied to any patient or otherwise healthy person as long as two mandatory requirements have been fulfilled. First there must be evidence of active clinical synovitis in at least one joint. Any joint would qualify with the exception of the DIP joints, the first MTP joint, and the first CMC joint, as these joints are very frequently involved in osteoarthritis. Second, the criteria may be applied only to those patients in whom the synovitis is not better explained by an alternative diagnosis, such as another inflammatory arthropathy.11
[su_table]
| Classification criteria for RA (score-based algorithm; add score of categories A-D; a score of >/= to 6/10 is needed for classification of a patient having definite RA | Score |
| A. Joint involvement
1 large joint* 2-10 large joints 1-3 small joints** (with or without involvement of large joints) 4-10 small joints (with or without involvement of large joints) >10 joints (at least 1 small joint) |
0 1 2 3 5 |
| B. Serology (at least 1 test result is needed for classification)
Negative RF and negative ACPA Low-positive RF or low-positive ACPA High-positive RF or high-positive ACPA |
0 2 3 |
| C. Acute-phase reactants (at least 1 test result is needed for classification)
Normal CRP and normal ESR Abnormal CRP or normal ESR |
0 1 |
| D. Duration of symptoms
< 6 weeks >/= 6 weeks |
0 1 |
[/su_table]
*Large Joint is defined as shoulder, elbow, hip, knee, or ankle.
**Small joint is defined as the MCP, PIP, second to fifth MTP, wrist, and first IP joints.
RF=Rheumatoid Factor
ACPA=Anti-citrullinated peptid antigens
CRP=C-Reactive Protein
ESR=Erythrocyte Sedimentation Rate
Utility and Technical Considerations of MR imaging in RA
The utility of magnetic resonance (MR) imaging in the evaluation of RA has been well described in the literature.15 MR imaging clearly offers increased sensitivity to soft tissue and marrow abnormalities and provides the clinician valuable diagnostic information that may significantly alter the management of RA, particularly in the initial stages, allowing earlier treatment and significant reduction in the morbidity of the disease.16 Reported data have indicated that approximately one third of patients in clinical remission with non-tender or non-swollen joints have evidence of synovitis on MR images.17 The sensitivity of MR imaging when clinical findings are absent or subtle is important as conventional radiographs are unrevealing at such a time. Furthermore, subclinical synovitis is the predominant finding and may progress in severity, leading to bone and cartilage destruction in roughly 47% of patients.18
Bilateral joint involvement is very typical of RA, and the articulations of the wrist and hand are commonly involved in the early stages of disease. Therefore, MR imaging protocols often emphasize assessment of these typical areas. Bilateral imaging of the wrists and hands can be performed and is advocated by some authors.19 However, this significantly increases imaging time and patient cost. Furthermore, if both the wrist and hand are included in one single examination, this markedly reduces the resolution owing to a larger field of view (FOV). Narváez et al. advocated imaging only the dominant or more painful wrist.20 We also support a similar imaging strategy. In our institution, we utilize a FOV of 8-12 cm using a surface coil to optimize signal. We include the distal forearm, carpus, and carpometacarpal joints. Typically, we acquire a non-fat suppressed anatomic, short TE sequence in 2-3 imaging planes. These sequences are optimal for detecting osseous erosions. We also acquire fluid sensitive sequences with fat suppression using either inversion recovery or chemical fat suppression techniques in three planes. Fat-suppressed post-contrast gadolinium sequences are also acquired in at least two planes to optimize detection of synovitis and recent erosions. Post-contrast imaging is usually performed rapidly (within 5 minutes) after the intravenous administration of gadolinium contrast as synovial tissue enhances in the early phase following the injection.21 Imaging acquired after a delayed time interval may reduce the specificity of synovial enhancement as gadolinium begins to infuse into the joint fluid. Images are obtained sequentially with a slice thickness of 3-4 mm with an interslice gap of 0.3-0.4 mm. Our protocols are in concordance with the Outcomes Measures in Rheumatoid Arthritis Clinical Trials (OMERACT) MR imaging study group, an international, multidisciplinary group established for optimizing MR imaging protocols as well as MR scoring systems in patients with rheumatoid arthritis.20-21
MR Imaging in Early Rheumatoid Arthritis
Synovitis, Tenosynovitis, and Bursitis
Synovitis is the earliest abnormality in RA, affecting joints, tendon sheaths, and bursae.21 Within articulations, synovitis is characterized by frond-like synovial thickening along the margins of the joint. Post-gadolinium sequences demonstrate avid enhancement of acutely inflamed synovial tissue, allowing differentiation from simple joint fluid, which does not enhance.20 Fibrotic synovitis, a marker of chronic RA, will be discussed later in this article.
Tenosynovitis occurs as a result of synovial proliferation within the tendon sheath. Tenosynovitis produces a layer of fluid that is typically thicker than the diameter of the tendon itself.22 Enhancement of the inflamed tenosynovium is noted following intravenous gadolinium administration. Synovial proliferation in a tendon sheath enhances in a manner similar to that in joints. Failure of tendons, however, is uncommon, with partial tears occurring in 8% of cases and complete tendon tears occurring in 3% of cases.20, 23
Figure 6:
(6a) AP (left) and lateral (right) radiographs of the hand in a patient with RA demonstrate symmetric joint space loss in the first metacarpophalangeal joint (arrowheads) with a marginal erosion in the first metacarpal head (arrow). (6b) Sagittal T2-weighted image of the knee with fat suppression in the same patient demonstrates a large joint effusion with mild synovial proliferation (arrowheads). Note that no bone erosions are visible about the knee.
Bone Marrow Edema and Erosions
Based on a number of prior reports, subchondral and often peripheral marrow edema depicted on MR images in RA patients is a strong predictor of future erosive disease.24, 25 Further, histological studies of bone marrow edema indicate that the altered marrow signal is composed of inflammatory cellular infiltrate and a sterile osteitis, which may predispose to a molecular reaction that activates bone and cartilage cleaving enzymes.26 Although roughly half of the patients with marrow edema go on to develop frank erosions within one year, the altered marrow signal can be reversible.16 Given the histologic background of marrow edema, it is not surprising that marrow edema in RA can enhance after the administration of gadolinium.
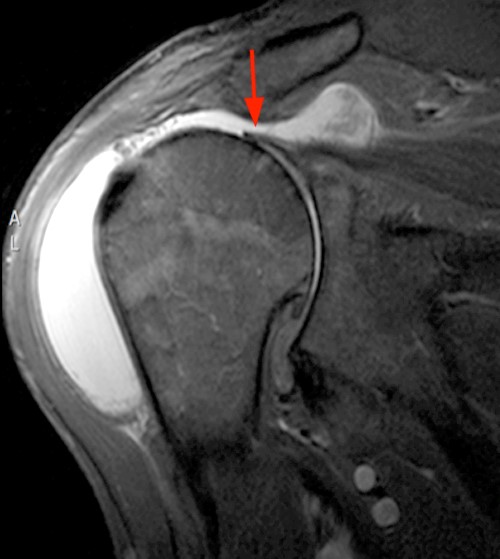
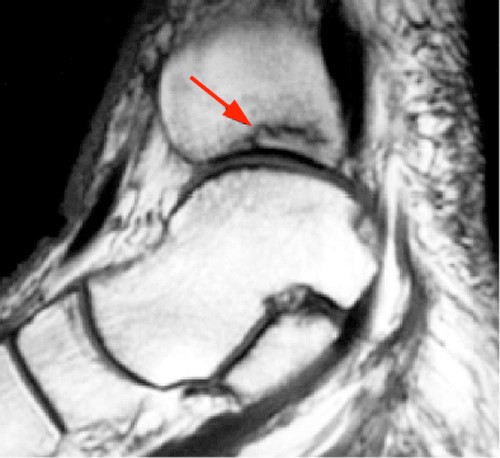
On both conventional radiographs and MR imaging, erosions are identified as a focal area of cortical loss, typically with acute margins. The “bare areas” of the joint are commonly affected, as these areas do not possess a protective layer of cartilage. On MR images, erosions are best depicted on non-fat suppressed short TE sequences. Recent erosions can be identified when there is enhancing synovial tissue within the erosion itself, as seen in the ulnar styloid in the introductory case. The detection of bone erosions in early RA correlates with a poor long-term outcome for the patient.16
Figure 8:
AP radiograph (8a) in a patient suspected to have RA is unrevealing, without evidence of joint space loss or osseous erosive disease. A coronal post-contrast MR image of the hand (8b) in the same patient demonstrates synovitis (arrowheads) in the second metacarpophalangeal joint with an enhancing erosion in the second metacarpal head (arrow), an abnormality not visible on the radiograph. Images courtesy of Dr. Evelyne Fliszar.
Figure 9:
STIR images in the axial (9a and 9b) and coronal planes (9c) demonstrate extensive synovitis (arrowheads) and multiple erosions in the medial and lateral tibial plateaus (arrows). Additionally, scattered patchy areas of bone marrow edema are seen (asterisks), suggesting a prequel to future erosive disease.
MR Imaging Findings in Longstanding Rheumatoid Arthritis
An understanding of the pathophysiology of RA helps to explain the characteristic MR imaging findings in the setting of longstanding disease.
Although synovitis is a hallmark of RA, the distribution and extent of synovitis vary with the stage of disease – early on, synovitis is more typically peripheral within the joint and its effects are usually seen in the bare area of the articulation lacking protection from cartilage, whereas later in the disease course, pannus may extend across the entire articular surface of the involved joint.27 Two types of pannus have been described – hypervascular and fibrous.28, 29 Hypervascular pannus is a feature of early and active RA and typically demonstrates high T2 signal, which can appear similar to joint fluid, thereby necessitating administration of intravenous gadolinium for clear differentiation (Figure 10).28, 29 In contrast, fibrous pannus tends to predominate in longstanding RA (particularly “burnt out” RA) and is more easily differentiated from joint fluid by virtue of its lower T2 signal. Fibrous pannus also demonstrates less avid enhancement (Figure 10).28, 29 Synovial enhancement should be assessed within five minutes of intravenous contrast administration. After this point, gadolinium leaks into the joint space, a phenomenon that obscures the distinction between synovial proliferation and joint fluid.30
Figure 10:
T2-weighted (10a) and T1-weighted fat-suppressed post-contrast (10b) images in longstanding RA reveal pannus with low T2 signal and mild to moderate enhancement (arrows). Contrast this with pannus in earlier RA – T2-weighted (10c) and T1-weighted fat-suppressed post-contrast (10d) images that show synovitis with high T2 signal and avid enhancement (arrows). Note that synovitis is difficult to clearly separate from joint fluid on T2-weighted images in early RA.
In untreated cases, extension of pannus throughout the joint eventually results in cartilage loss, which is most often concentric or diffuse (Figure 11). Concentric high-grade joint space narrowing is a feature of longstanding disease.5 MR imaging may be valuable in assessing the extent of cartilage loss, which is usually best seen on short TE sequences such as a gradient echo sequence.5
Once cartilage loss ensues, both subchondral cysts and erosions may become evident. The histological basis of subchondral cysts in the setting of RA is unclear – subchondral cysts may result from decompression of joint fluid in the setting of increased intra-articular pressure or through intraosseous extension of pannus.27 While large intraosseous cysts (>1 cm) are uncommon in inflammatory arthropathies, one study found such cysts to be more common in longstanding RA.31 The same study also found large intraosseous cysts to enhance avidly, lending support to the theory of intraosseous invasion of pannus in advanced RA (Figure 12). Erosions in longstanding RA may involve any portion of the articulation and may be extensive if disease is untreated. As one might expect, superimposed osteoarthritis is not uncommon in chronic RA.
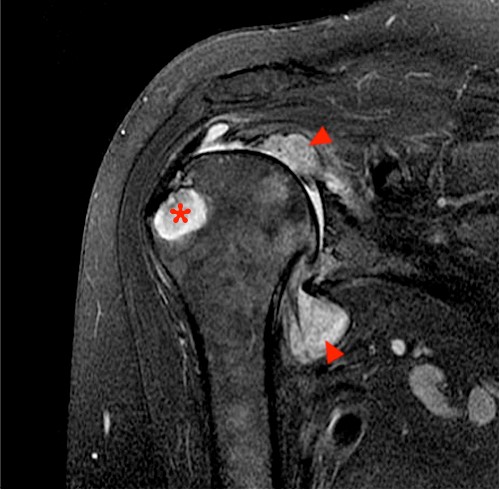
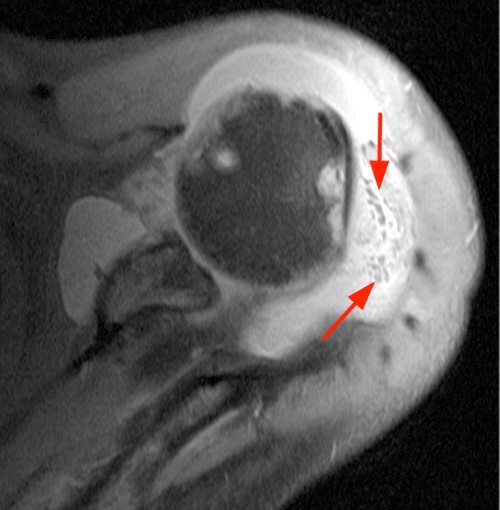
Figure 12:
Axial (12a) and coronal (12b) proton density-weighted fat-suppressed images of the shoulder reveal a large intraosseous cyst (asterisks) in the greater tuberosity. Notice the intermediate signal within the cyst, suggestive of pannus invasion. Concentric joint space loss in the glenohumeral joint is also present with extensive intra-articular pannus (arrowheads).
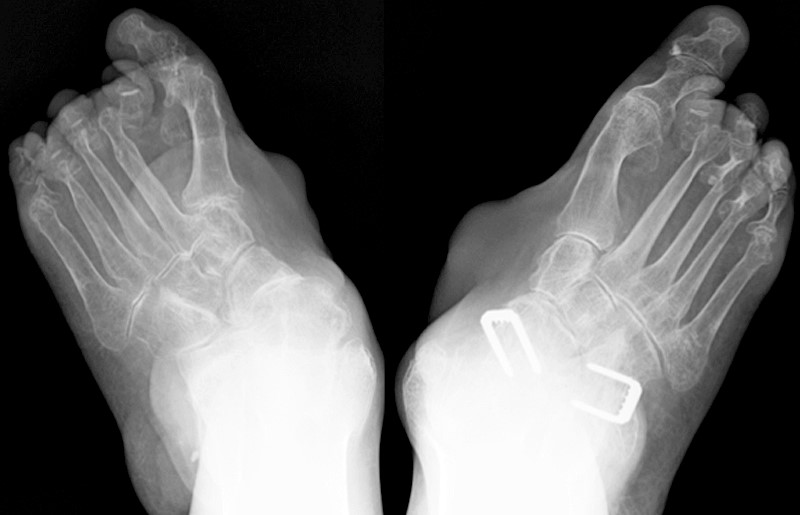
Articular deformities in chronic disease result from the damaging effects of chronic synovitis on the joint capsule and surrounding tendons.5, 27 Frequent deformities include ulnar deviation of the proximal phalanges at the metacarpophalangeal joints and fibular deviation of the toes at the metatarsophalangeal joints. In the hands, swan neck and boutonniere deformities of the fingers and a hitchhiker deformity of the thumb are well known complications (Figure 13). All are easily diagnosed on radiographs. Bone ankylosis is an uncommon complication of advanced RA, but is most commonly seen about the wrist, affecting the carpus.32 The common deformities in RA are summarized in Table 1.
Table 1. Common Deformities in Advanced RA.
[su_table]
| Deformity | Appearance | Cause |
| Swan-neck | Extended PIP, flexed DIP | Injury to volar plate and lateral slips of extensor expansion |
| Boutonniere | Flexed PIP, extended DIP | Injury to central slip of extensor expansion |
| Hitchhiker thumb | Boutonniere deformity of thumb | Injury to central slip of extensor expansion |
[/su_table]
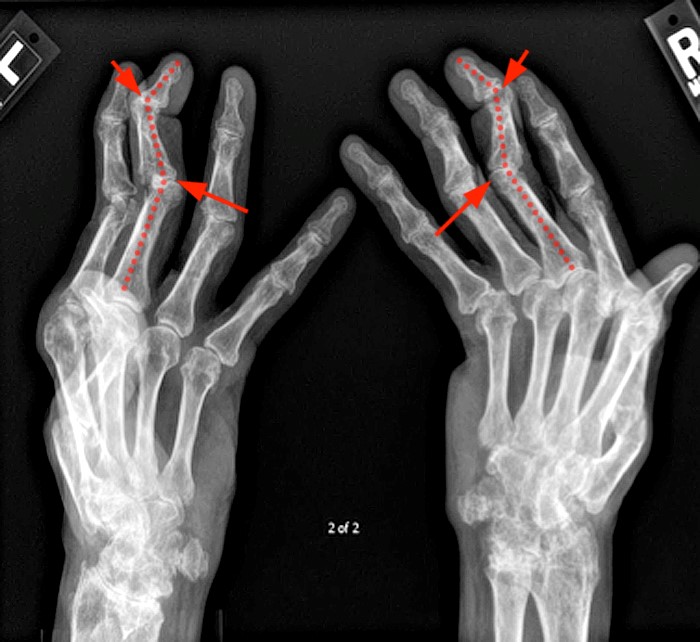
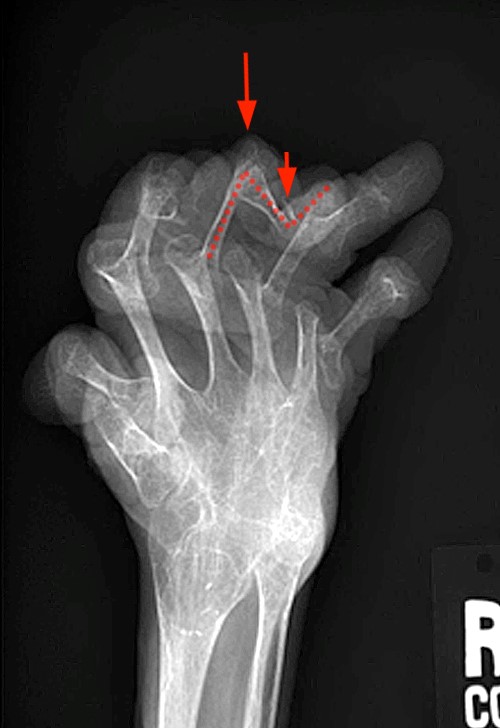
Figure 13:
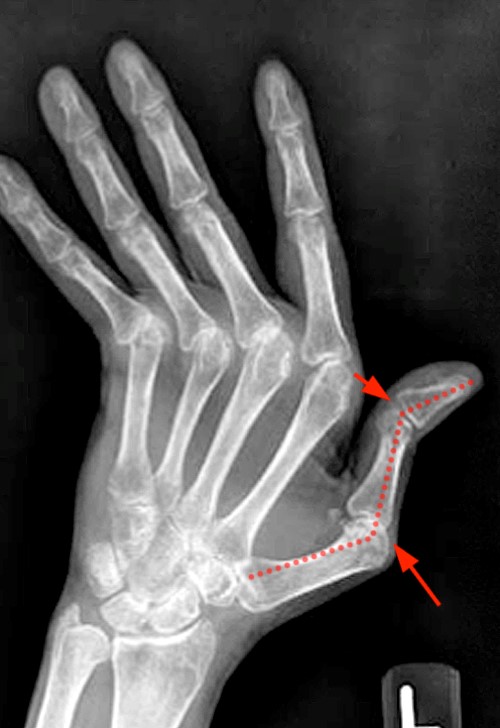
Posterior oblique view (“ball-catcher view”) of both hands (13a) demonstrate the typical appearance of the swan neck deformity characterized by extension of the PIP (long arrow) and flexion of the DIP (short arrow). In addition to multiple dislocations and ankyloses the AP views of the wrist (13b) demonstrates boutonniere deformities of the index and long fingers, best seen at the long finger characterized by flexion of the PIP (long arrow) and extension of the DIP (short arrow). The hitch-hiker thumb is illustrated in (13c) with flexion at the MP joint (long arrow) and extension of the DIP (short arrow). Images courtesy of Dr. Evelyne Fliszar.
Rice bodies are generally considered a feature of chronic synovial inflammation.33 While classically seen with tuberculosis, they are frequently seen in advanced cases of RA.34 Rice bodies may also be seen with osteoarthritis.34 Although their pathological basis is not entirely clear, rice bodies likely represent the sequela of sloughing of hypertrophied synovium.35 With imaging, rice bodies are typically small, numerous, and of similar size. They invariably demonstrate low T1 and T2 signal on MR imaging and usually demonstrate no mineralization on radiographs (Figure 14).35 Rice bodies can be differentiated from synovial chondromatosis as, in the latter condition, numerous intra-articular deposits frequently ossify (synovial osteochondromatosis) and appear much denser on radiographs (Figure 14).35
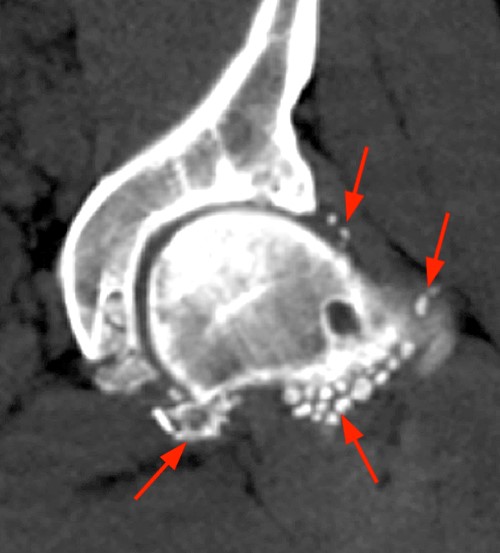
Figure 14:
An axial proton density-weighted fat-suppressed image of the shoulder (14a) demonstrates numerous rice bodies (arrows) in a patient with advanced RA. An AP radiograph (14b) and a coronal CT image (14c) of the left hip demonstrate densely mineralized deposits in synovial osteochondromatosis (arrows).
Rheumatoid nodules are more frequently seen in longstanding disease and may occur in up to 20-30% of cases.36 Histologically, these nodules are composed of fibroblasts and histiocytes surrounding a central area of fibrinoid necrosis.36 Rheumatoid nodules are subcutaneous in location and tend to occur at sites of microtrauma and traction.37 Typical sites include the olecranon region and heel pad. With MR imaging, these nodules demonstrate isointense to hypointense T1 signal relative to skeletal muscle, variable T2 signal, which is dependent on the extent of necrosis, and variable enhancement (Figure 15).37 Studies have suggested that rheumatoid nodules are associated with a more aggressive disease course.38
Complications of Rheumatoid Arthritis
There are many complications of RA, which occur predominantly in the setting of longstanding disease.
Stenosing tenosynovitis is a frequent complication of longstanding RA – this condition results in impaired tendon movement in the setting of chronic inflammation of the tendon sheath. One study found stenosing tenosynovitis to occur in one third of RA patients with an average disease duration of 5.9 years.39 Stenosing tenosynovitis is most commonly seen in the hand and wrist, particularly involving the tendons in the extensor compartments. With MR imaging, stenosing tenosynovitis manifests as bead-like dilatation of the tendon sheath with areas of synovitis.
Rotator cuff tears are a common complication of RA. Massive rotator cuff tears, characterized by full-thickness and full-width tears of two or more rotator cuff tendons, are best diagnosed with MR imaging (Figure 16).
Figure 16:
A coronal fat-suppressed T2-weighted image (16a) demonstrates a full-thickness and full-width tear of the supraspinatus tendon with retraction of torn fibers to near the glenohumeral joint (arrow). A fat-suppressed proton density-weighted axial image (16b) reveals a full thickness tear of the subscapularis tendon. Rice bodies (arrowheads) are apparent in the subdeltoid bursa. A sagittal T1-weighted image (16c) demonstrates marked atrophy of the supraspinatus muscle (asterisk) and mild atrophy of the infraspinatus muscle (arrowheads).
Extensor tendon rupture about the wrist is also a common complication in advanced RA, with reported risk factors including disease duration (> 10 years), tenosynovitis for at least 6 months, advanced Larsen radiographic score, dorsoulnar subluxation at the distal radioulnar joint (DRUJ), large erosions around the DRUJ (scallop sign), and volar dislocation of the extensor carpi ulnaris tendon.40, 41 These same studies found tears of the extensor digiti minimi (EDM) and extensor digiti communis (EDC) tendons to be most common. Flexor tendon ruptures may also occur. Chronic tenosynovitis may be treated prophylactically with tenosynovectomy, while tendon rupture requires definitive surgical treatment with tendon transfer or interposition tendon grafting.40 Examples of extensor and flexor tendon tears are shown in Figure 17.
Figure 17:
An axial T2-weighted fat-suppressed image (17a) reveals a split tear of the ECU tendon distal to the ulnar styloid process (arrow). Axial fat-suppressed proton density-weighted (17b) and sagittal T1-weighted (17c) images in a different patient demonstrate a slightly retracted tear of the flexor carpi radialis tendon (arrows).
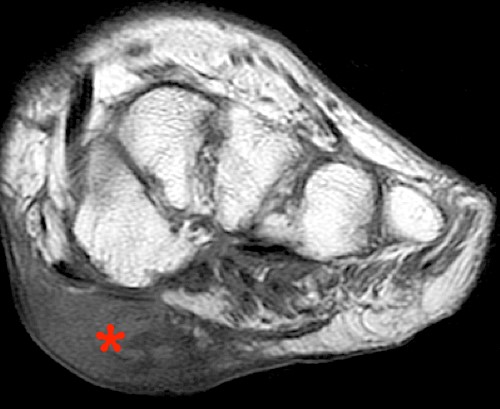
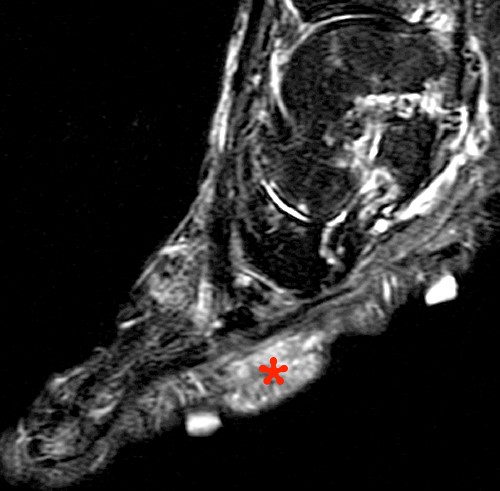
Insufficiency fractures are an important complication of RA. Classic sites include the sacrum, parasymphyseal bone, femoral neck, tibia (Figure 18), and fibula.27 When radiographically occult, MR imaging is invaluable in demonstrating these fractures.
Involvement of the craniocervical junction and cervical spine is common in RA, being seen in up to 86% of cases; however, instability in this location is usually limited to patients with longstanding disease, typically after at least 10 years.42 Sites of involvement include the atlanto-occipital joints, atlanto-axial joints, atlanto-odontoid articulation, facet joints, and uncovertebral joints. The most common complication is atlantoaxial subluxation, which is usually characterized by forward subluxation of C1 on C2.42 Atlantoaxial subluxation can result in severe narrowing of the cervical spinal canal. Cranial settling is a less common complication, which results from erosive disease between the occipital condyles and C1 as well as between the lateral masses of C1 and C2. The most feared consequence of cranial settling is indentation of the brainstem by the odontoid process (Figure 19). Erosive disease involving the facet joints and uncovertebral joints can result in multilevel step-like vertebral body subluxations42, known as the stepladder appearance (Figure 20).
Figure 19:
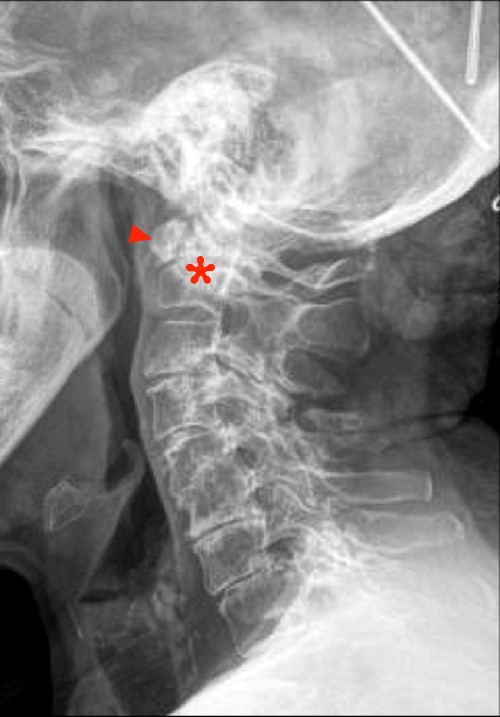
Cranial settling. Lateral radiograph (19a) shows that C1 (arrowhead) is inferiorly situated relative to C2 (asterisk) due to erosive disease between the C1 and C2 lateral masses. The corresponding sagittal STIR MR image (19b) shows indentation of the medulla by the odontoid process (arrow) projecting into the foramen magnum without associated edema.
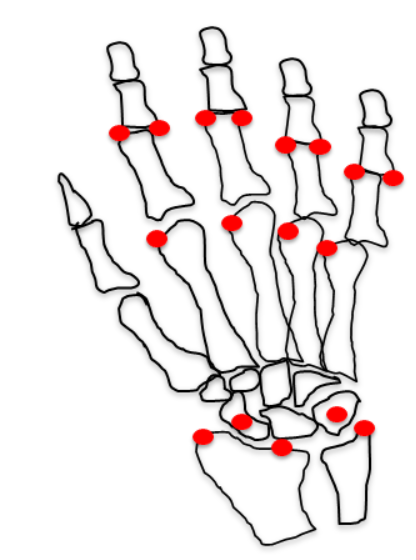
The most commonly affected sites in RA involving both the appendicular and axial skeleton are summarized in Table 2. Figure 21 highlights the typical sites of involvement in the hand and wrist.
Table 2. Typical Sites of Involvement in RA.
[su_table]
| Appendicular Skeleton | Joints | Earliest sites |
| Wrist | Distal radioulnar (DRUJ), radiocarpal, radial and ulnar styloid processes, midcarpal, pisotriquetral | DRUJ, radiocarpal, ulnar styloid |
| Hand | MCPs, PIPs | Radial aspect 2nd & 3rd metacarpal heads, 3rd PIP |
| Elbow | All articulations | |
| Shoulder | Acromioclavicular, glenohumeral (typically near greater tuberosity) | |
| Feet | MTPs, PIPs, interphalangeal joint great toe, tarsometatarsal, intertarsal, retrocalcaneal | 5th MTP |
| Ankle | Tibiotalar, subtalar | |
| Knee | All compartments | |
| Hip | ||
| Axial Skeleton | ||
| Cervical Spine | Atlanto-occipital | |
| Atlantoaxial | ||
| Facet joints and spinous processes | ||
| Thoracolumbar | ||
| Sacroiliac |
[/su_table]
Treatment and Monitoring of Rheumatoid Arthritis
Early treatment of RA, which is generally defined as within three to six months of symptom onset, has been shown to improve remission rates and delay the onset of joint destruction.
Disease modifying anti-rheumatic drugs (DMARDs) are the mainstay of treatment. Mild early onset disease may be successfully managed through monotherapy with methotrexate (MTX), while cases presenting with a larger joint burden and/or poor prognostic factors (such as elevated serum rheumatoid factor) are often treated with combination therapy comprising MTX and a biologic agent, such as infliximab 43. Corticosteroids may also be part of the combination therapy regimen.43 Joint replacement is reserved for advanced arthritis.
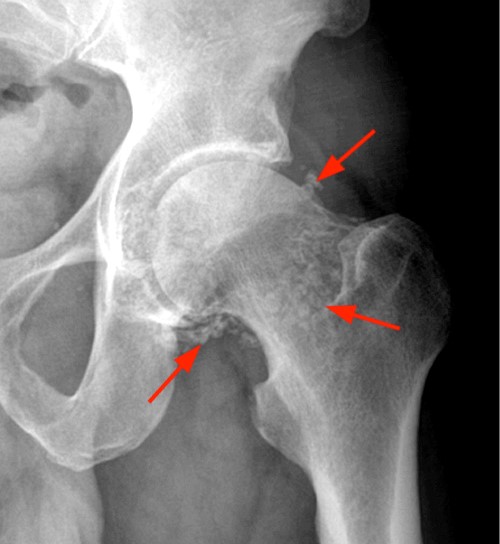
MTX is the first-line DMARD used in the treatment of RA.44 MTX may predispose to osteoporosis and insufficiency fractures.44 Osteonecrosis is not an uncommon complication of RA therapy as corticosteroids are known to be an inciting factor (Figure 22). One study demonstrated a 12% incidence of osteonecrosis involving the femoral head in patients undergoing hip arthroplasty for RA.45
Septic arthritis is also seen with increased frequency in RA.46 Predisposing factors include impaired phagocytosis 47 and increased vascularity from synovitis.46 Larger joints are more commonly affected, such as the knee, elbow, and wrist.48 Due to immunosuppressant therapy, onset is often insidious and a high degree of clinical suspicion is often required.
Ultrasonography is a useful modality for evaluating synovitis and tenosynovitis, but it is less sensitive than MR imaging for the detection of erosions and does not allow the detection of marrow edema.30
The key parameters to be assessed with MR imaging when evaluating treatment response are synovitis, marrow edema, and extent of erosions. Cartilage must also be scrutinized; this is easier in larger joints like the knee and much more difficult in smaller joints such as the wrist. A positive response to treatment includes reduction in synovial volume 17, reduction in marrow edema, and stability or minimal progression of erosions.30
Dynamic contrast enhanced MR imaging has been used to evaluate the rate of synovial enhancement and has shown strong histopathological correlation with active disease, which tends to demonstrate more rapid enhancement as a reflection of synovial inflammation and increased vascularity.49
Several studies have also documented a decrease in synovial enhancement following DMARD therapy.30
Conclusion
The principle pathophysiology of articular involvement in RA is synovial hyperplasia. This may occur in joints, bursae, or tendon sheaths. The local effects of synovitis are mediated through the release of enzymes that destroy bone and soft tissue. It is clear that imaging can be effective in documenting involvement of the joints, bones, tendon sheaths, and bursae, especially in the early stages of disease when conventional radiographs may be negative, allowing earlier diagnosis. Indeed, the institution of biologic agents early in the course of disease has had a dramatic impact upon the morbidity of RA.
Features of longstanding RA demonstrable with MR imaging include the presence of fibrous pannus, high-grade concentric cartilage loss, widespread bone erosions and prominent subchondral cysts containing pannus, intra-articular rice bodies, and rheumatoid nodules. MR imaging is valuable for assessing complications of RA, which include stenosing tenosynovitis, rotator cuff tears, tendon ruptures about the wrist, insufficiency fractures, and complications in the cervical spine such as atlantoaxial subluxation and cranial settling. MR imaging may also be used in evaluating the response to treatment – key parameters that should be monitored are the extent of synovitis, marrow edema, and erosions.
References
- Gabriel, Sherine E. “The epidemiology of rheumatoid arthritis.” Rheumatic Disease Clinics of North America 27.2 (2001): 269-281. ↩
- Gabriel, Sherine E., Cynthia S. Crowson, and Michael O’Fallon. “The epidemiology of rheumatoid arthritis in Rochester, Minnesota, 1955–1985.” Arthritis & Rheumatism 42.3 (1999): 415-420. ↩
- Smith, Malcolm D. “The normal synovium.” The open rheumatology journal 5.1 (2011). ↩
- Smith MD, Walker JG. Hochberg, Silman, Smolen, Weinblatt, Weisman, editors. The synovium. Rheumatology. (5th ed) 2011;1:51–6. ↩
- Sommer, Oliver J., et al. “Rheumatoid Arthritis: A Practical Guide to State-of-the-Art Imaging, Image Interpretation, and Clinical Implications ↩
- Edwards JCW. Functions of synovial lining. In: Henderson B, Edwards JCW, editors. The synovial lining in health and disease. London: Chapman & Hall; 1987. pp. 41–74. ↩
- Yamanishi, Yuji, and Gary S. Firestein. “Pathogenesis of rheumatoid arthritis: the role of synoviocytes.” Rheumatic Disease Clinics of North America 27.2 (2001): 355-371. ↩
- van de Sande, Marleen G., and Dominique L. Baeten. “Immunopathology of synovitis: from histology to molecular pathways.” Rheumatology (2015): kev330. ↩
- Dayer, J. M. “The pivotal role of interleukin-1 in the clinical manifestations of rheumatoid arthritis.” Rheumatology 42.90002 (2003): 3-10. ↩
- Arnett, Frank C., et al. “The American Rheumatism Association 1987 revised criteria for the classification of rheumatoid arthritis.” Arthritis & Rheumatism31.3 (1988): 315-324. ↩
- Aletaha, Daniel, et al. “2010 rheumatoid arthritis classification criteria: an American College of Rheumatology/European League Against Rheumatism collaborative initiative.” Arthritis & Rheumatism 62.9 (2010): 2569-2581. ↩
- Bijlsma, Johannes WJ, and Michael E. Weinblatt. “Optimal use of methotrexate: the advantages of tight control.” Annals of the rheumatic diseases 66.11 (2007): 1409-1410. ↩
- Pincus, T., et al. “Methotrexate as the” anchor drug” for the treatment of early rheumatoid arthritis.” Clinical and experimental rheumatology 21.5; SUPP 31 (2003): S179-S185. ↩
- Doan, Tanya, and Elena Massarotti. “Rheumatoid arthritis: an overview of new and emerging.” ↩
- McQueen, Fiona M., et al. “Magnetic resonance imaging of the wrist in early rheumatoid arthritis reveals a high prevalence of erosions at four months after symptom onset.” ↩
- McQueen, F. M. “The use of MRI in early RA.” Rheumatology 47.11 (2008): 1597-1599. ↩
- Brown, A. K., et al. “Presence of significant synovitis in rheumatoid arthritis patients with disease‐modifying antirheumatic drug–induced clinical remission: Evidence from an imaging study may explain structural progression.” Arthritis & Rheumatism 54.12 (2006): 3761-3773. ↩
- Pincus T. Longterm outcomes in rheumatoid arthritis. Br J Rheumatol 1995;34 (suppl 2):59–73. ↩
- Boutry, Nathalie, et al. “MR Imaging Findings in Hands in Early Rheumatoid Arthritis: Comparison with Those in Systemic Lupus Erythematosus and Primary Sjögren Syndrome 1.” Radiology 236.2 (2005): 593-600. ↩
- Narvaez, José A., et al. “MR Imaging of Early Rheumatoid Arthritis.”Radiographics 30.1 (2010): 143-163. ↩
- Østergaard, Mikkel, et al. “OMERACT Rheumatoid Arthritis Magnetic Resonance Imaging Studies. Core set of MRI acquisitions, joint pathology definitions, and the OMERACT RA-MRI scoring system.” The Journal of Rheumatology 30.6 (2003): 1385-1386. ↩
- Rubens, D. J., et al. “Rheumatoid arthritis: evaluation of wrist extensor tendons with clinical examination versus MR imaging–a preliminary report.”Radiology 187.3 (1993): 831-838. ↩
- Valeri, Gianluca, et al. “Tendon involvement in rheumatoid arthritis of the wrist: MRI findings.” Skeletal radiology 30.3 (2001): 138-143. ↩
- McQueen, Fiona M., et al. “Bone edema scored on magnetic resonance imaging scans of the dominant carpus at presentation predicts radiographic joint damage of the hands and feet six years later in patients with rheumatoid arthritis.” Arthritis & Rheumatism 48.7 (2003): 1814-1827. ↩
- Benton, N., et al. “MRI of the wrist in early rheumatoid arthritis can be used to predict functional outcome at 6 years.” Annals of the rheumatic diseases 63.5 (2004): 555-561 ↩
- Jimenez‐Boj, Esther, et al. “Bone erosions and bone marrow edema as defined by magnetic resonance imaging reflect true bone marrow inflammation in rheumatoid arthritis.” Arthritis & Rheumatism 56.4 (2007): 1118-1124. ↩
- Resnick D, Kransdorf MJ. Bone and Joint Imaging 3rd Elsevier Saunders 2005. Philadelphia, PA. ↩
- Konig H, Sieper J, Wolf K-J. Rheumatoid Arthritis: evaluation of hypervascular and fibrous pannus with dynamic MR Imaging enhanced with Gd-DTPA. Radiology. 1990; 176:473-47. ↩
- Lamer S, Sebag GH. MRI and Ultrasound in children with juvenile chronic arthritis. Eur J Radiol. 2000;33(2):85-93 ↩
- Hodgson RJ, O’Connor P, Moots R. MRI of rheumatoid arthritis – image quantitation for the assessment of disease activity, progression, and response to therapy. Rheumatology. 2008;47:13-21 ↩
- Taranzadeh J, Ashikyan O, Descalos J, Dennehery C. MRI of Large Intraosseous Lesions in Patients with Inflammatory Arthritis. AJR. 2004;183:1453-1463 ↩
- Manaster BJ, May, Disler. Musculoskeletal Imaging 4th ↩
- Sheybani EF, Khanna G, White AJ, Demertzis JL. Imaging of juvenile idiopathic arthritis: a multimodality approach. Radiographics. 2013;33(5):1253-73 ↩
- Bilateral recurrent wrist flexor tenosynovitis and rice body formation in a patient with sero-negative rheumatoid arthritis: A case review and review of the literature. Int J Surg Case Rep. 2011;2(7):208-11 ↩
- Raju KP, Kumar J, Shetty R. Tuberculous tenosynovitis of Ankle with Rice bodies. Foot and ankle online journal. 2013;6(10):1 ↩
- Mutlu H, Sildiroglu H, Pekkafall Z, Kizilkaya E, Cermik H. MRI appearance of retrocalcaneal bursitis and rheumatoid nodule in a patient with rheumatoid arthritis. Clin Rheumatol. 2006;25:734-736 ↩
- Starok M, Eilenberg S, Resnick D. Rheumatoid Nodules: MR Characteristics. Clin Imaging. 1998;22:216-219 ↩
- Nyhall-Wahlin, Turesson C, Jacobsson LT, Nilsson JA, Forslind K, Albertsson K, Petersson IF. The presence of rheumatoid nodules at early rheumatoid arthritis diagnosis is a sign of extra-articular disease and predicts radiographic progression of joint destruction over 5 years. Scand J Rheumatol. 2001;40(2)81-7 ↩
- Ursum J, Horsten NC, Hoeksma AF, Dijkmans BA, Knol DL, van Schaardenburg D, Dekker J, Roorda LD. Arch Phys Med Rehabil. January 2011; 92:96-100 ↩
- Hsueh JH, Liu WC, Yang KC, Hsu KC, Lin CT, Chen LW. Spontaneous extensor tendon rupture in the rheumatoid wrist: risk factors and preventative role of extended tenosynovectomy. Ann Plast Surg. 2016 March; 76(Supplement 1):S41-S47 ↩
- Egi T, Inui K, Koike T, Goto H, Takaota K, Kazuki K. Volar dislocation of the extensor carpi ulnaris tendon on magnetic resonance imaging is associated with extensor digitorum communis tendon rupture in rheumatoid arthritis. J Hand Surg Am. 2006 Nov;31(9):1454-60 ↩
- Mukerji N and Tod NV. Cervical Myelopathy in Rheumatoid Arthritis. Neurology Research International. 2011;2011:7 pages ↩
- Demoruelle MK, Deane K. Treatment Strategies in Early Rheumatoid Arthritis and Prevention of Rheumatoid Arthritis. Curr Rheumatol Rep. 2012 Oct; 14(5):472-480 ↩
- Albrecht K, Muller-Ladner. Side effects and management of side effects of methotrexate in rheumatoid arthritis. Clin Exp Rheumatol. 2010 (Suppl. 61):S95-S101. ↩
- Zabinksi SJ, Sculco TP, Dicarlo EF, Rivelis M. Osteonecrosis in the rheumatoid femoral head. J Rheumatol. 1998 Sep;25(9):1674-80 ↩
- Al-Ahaideb A. Septic arthritis in patients with rheumatoid arthritis. J Orthop Surg. 2008;3:33 ↩
- Turner RA, Schumacher HR, Myers AR. Phagocytic function of polymorphonuclear leukocytes in rheumatic disease. J Clin Invest. 1973; 52:1632-35 ↩
- Gardner GC, Weisman MH. Pyarthrosis in patients with rheumatoid arthritis: A report of 13 cases and a review on the literature from the past 40 years. Am J Med 1990; 88:503-511 ↩
- Ostergaard M, Stoltenberg M, Lovgreen-Nielsen P, Volck B, Sonne-Holm S, Lorenzen I. Quantification of synovitis by MRI: correlation between dynamic and static gadolinium-enhanced magnetic resonance imaging and microscopic and macroscopic signs of synovial inflammation. Magn Reson Imaging 1998; 16:743- ↩