Clinical History: Four different patients presented to the orthopaedist with anterior knee pain. What are the findings? What is your diagnosis for each patient, and how are the diagnoses related?
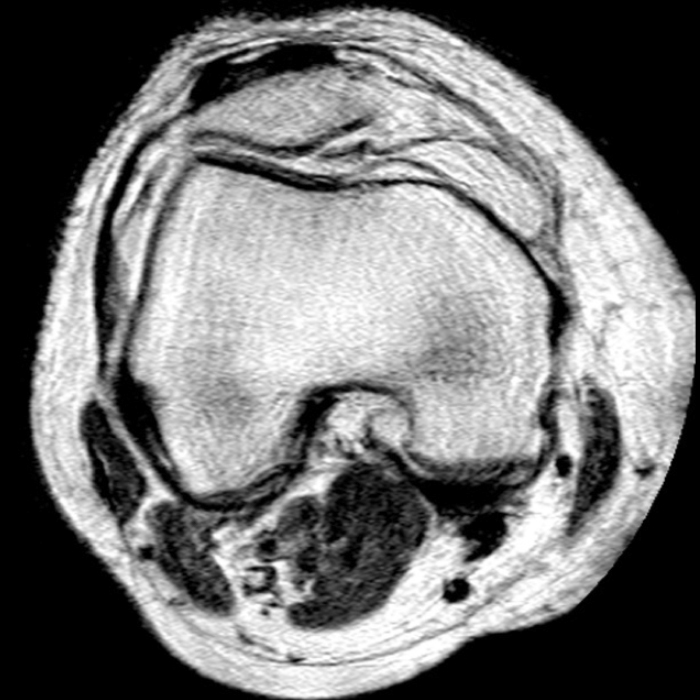
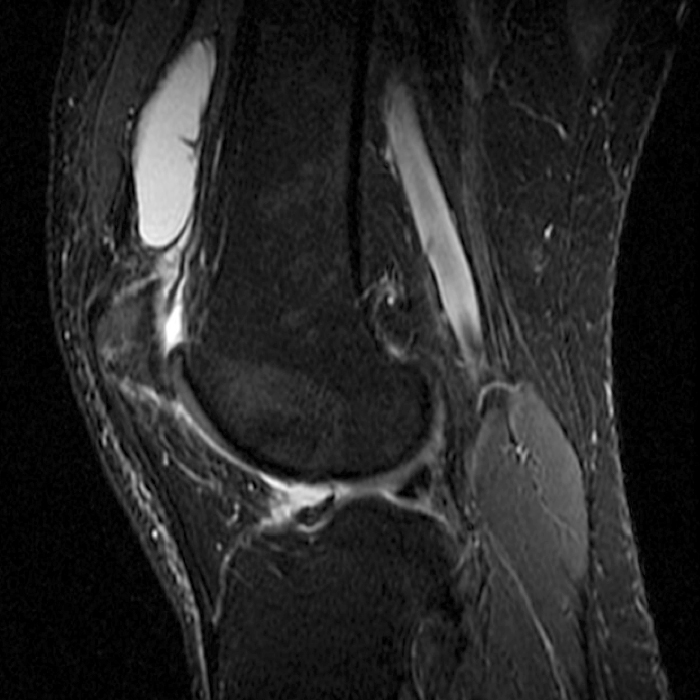
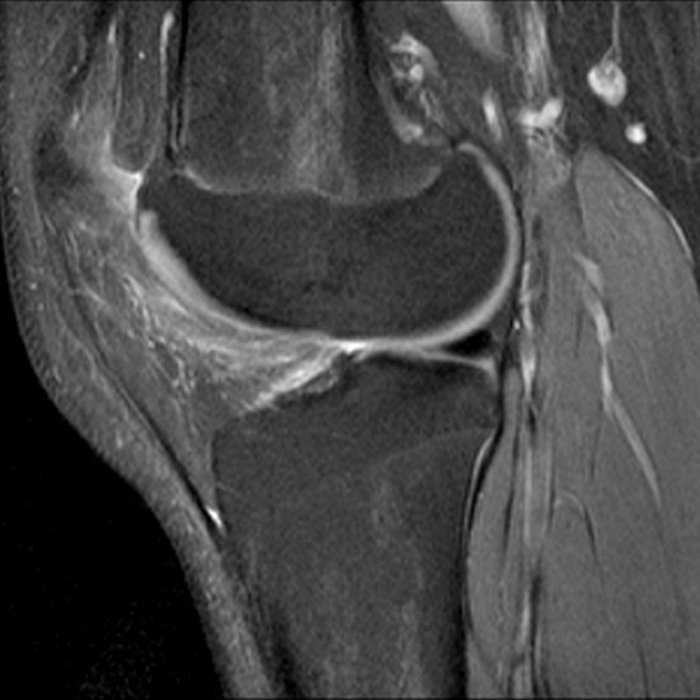
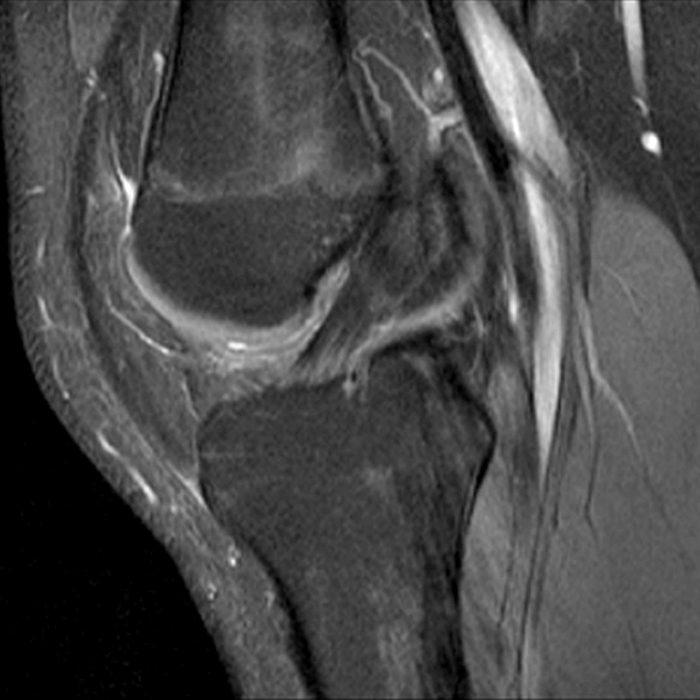
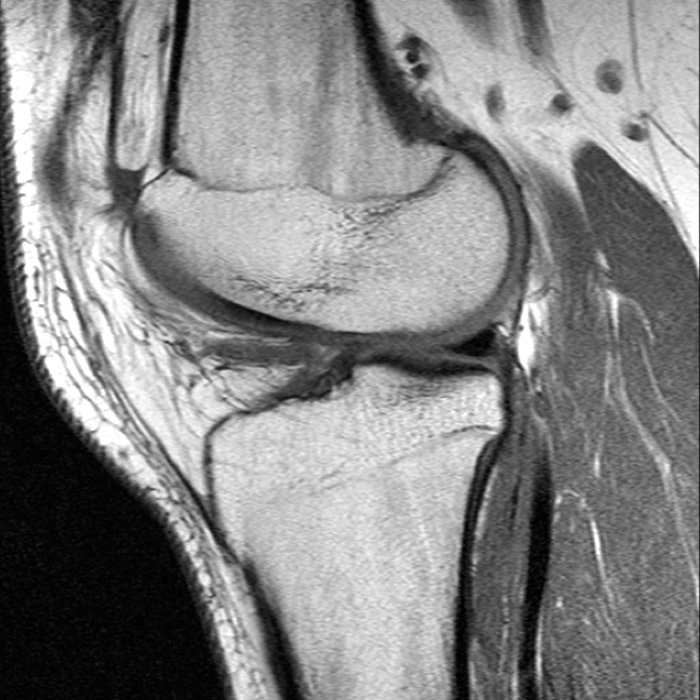
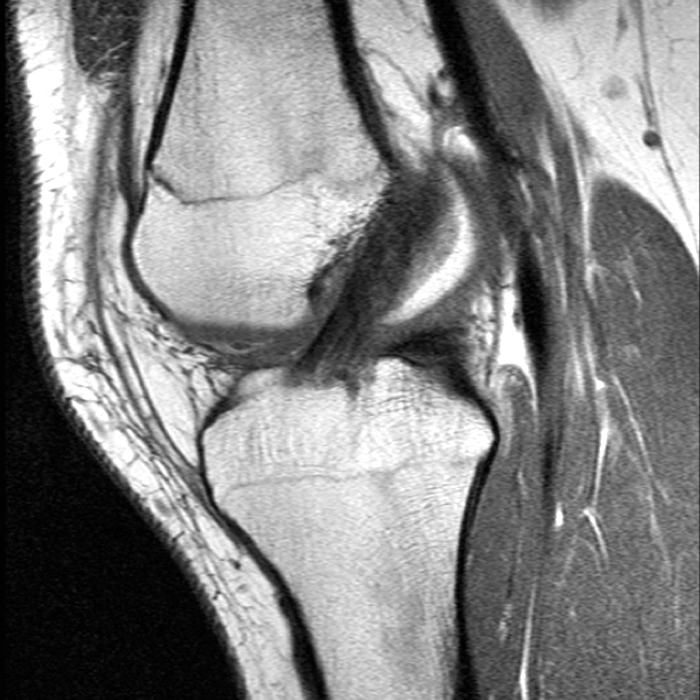
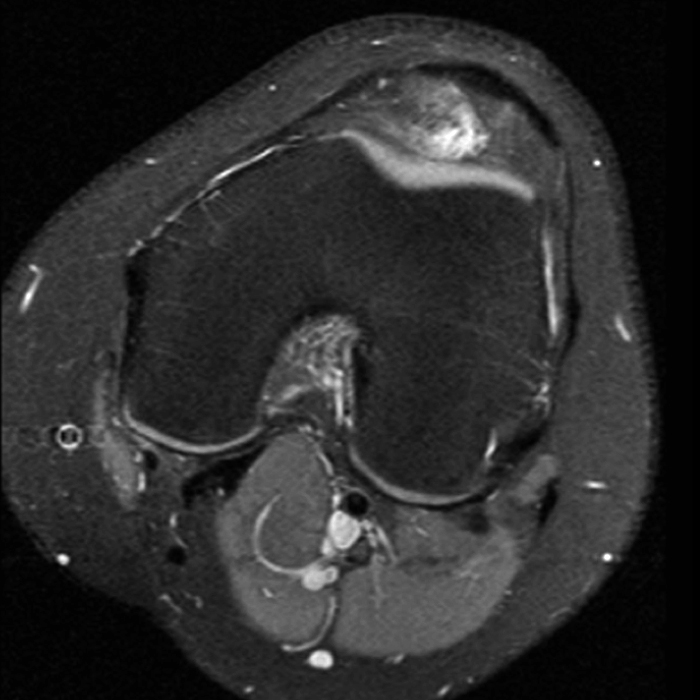
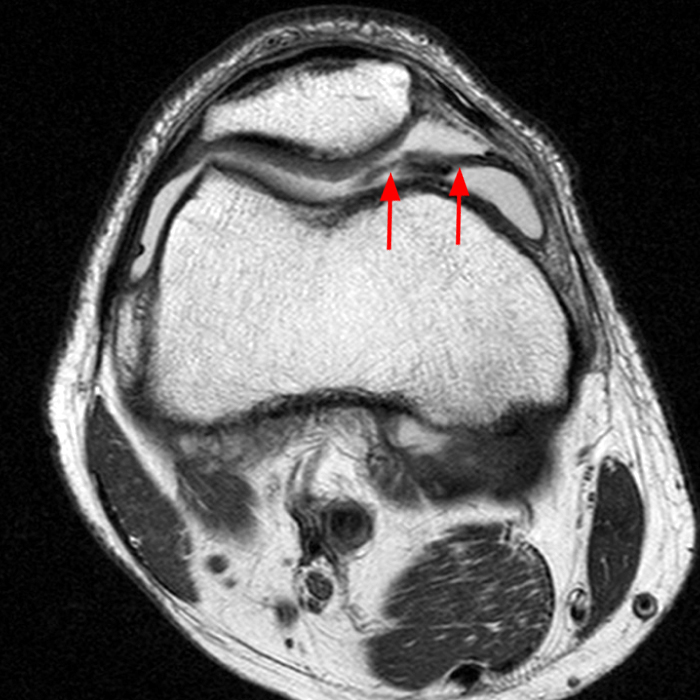
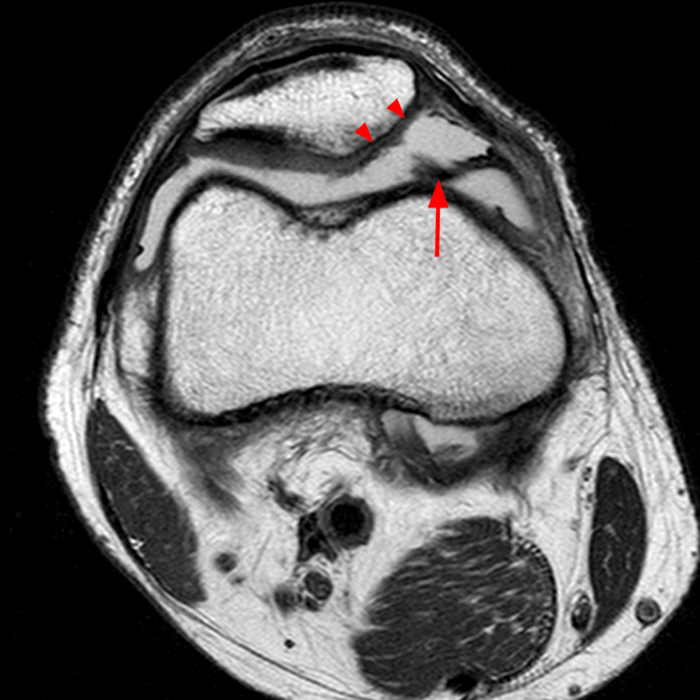
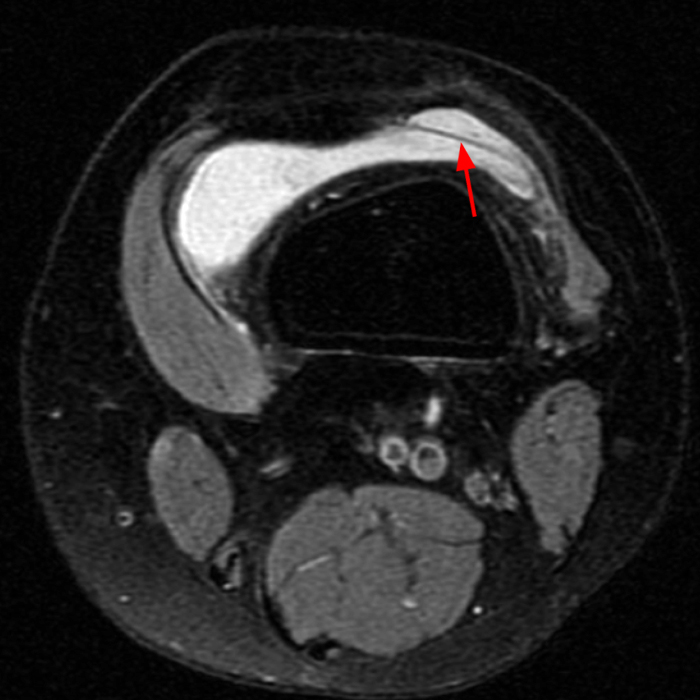
Case 1: A 47 year-old male reports anterior knee pain and swelling for 2 months. Contiguous axial fast spin-echo (FSE) proton density-weighted (1A and 1B) and sagittal FSE inversion recovery (1C and 1D) images were obtained.
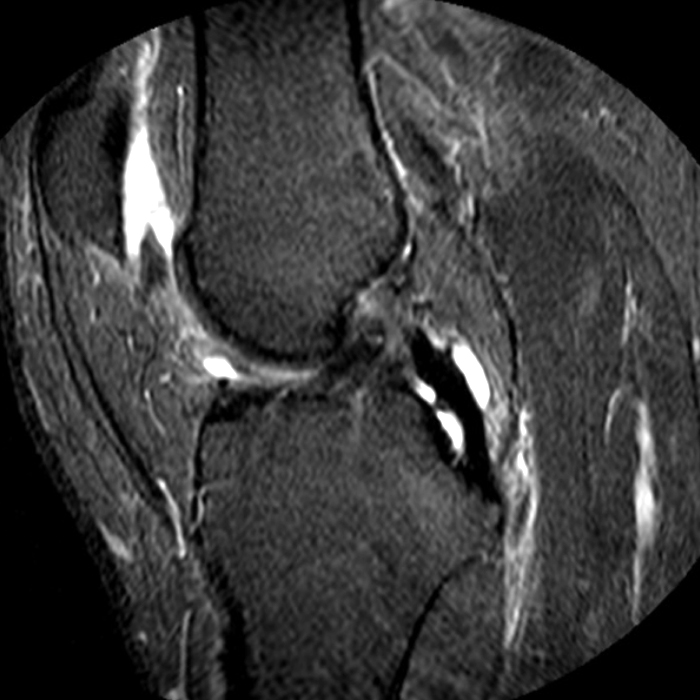
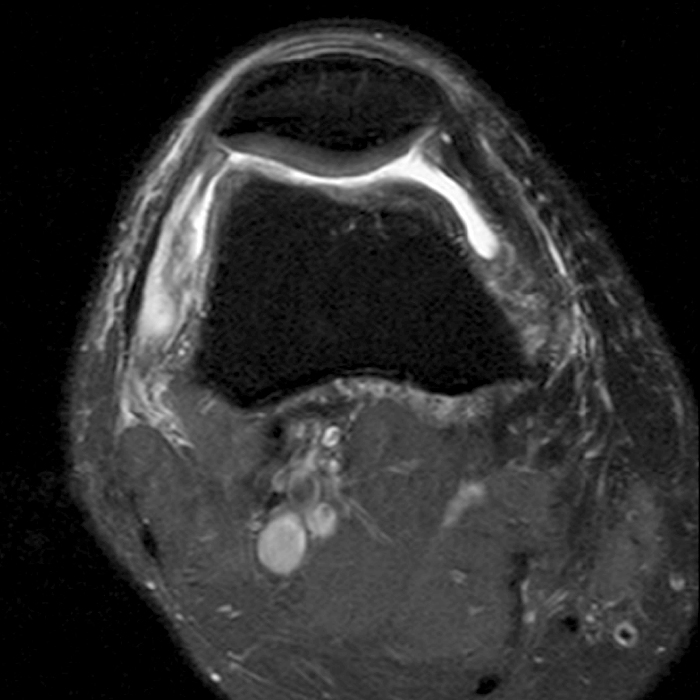
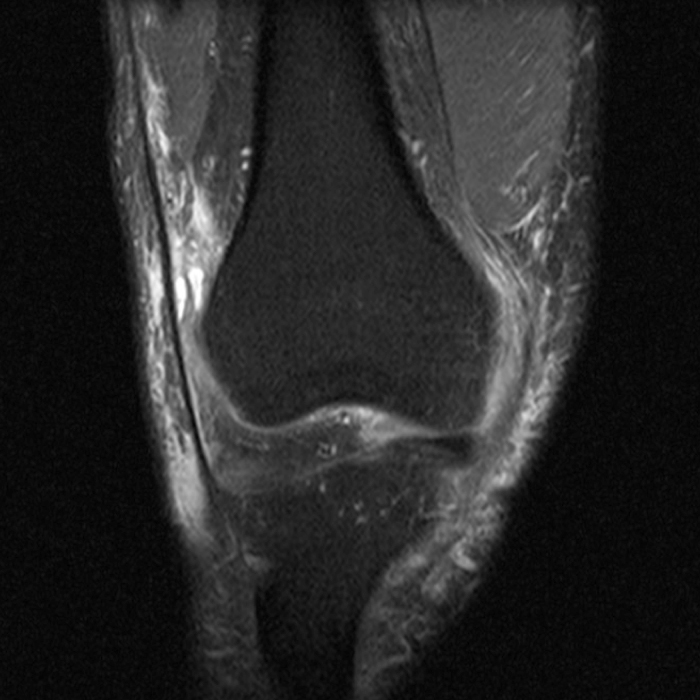
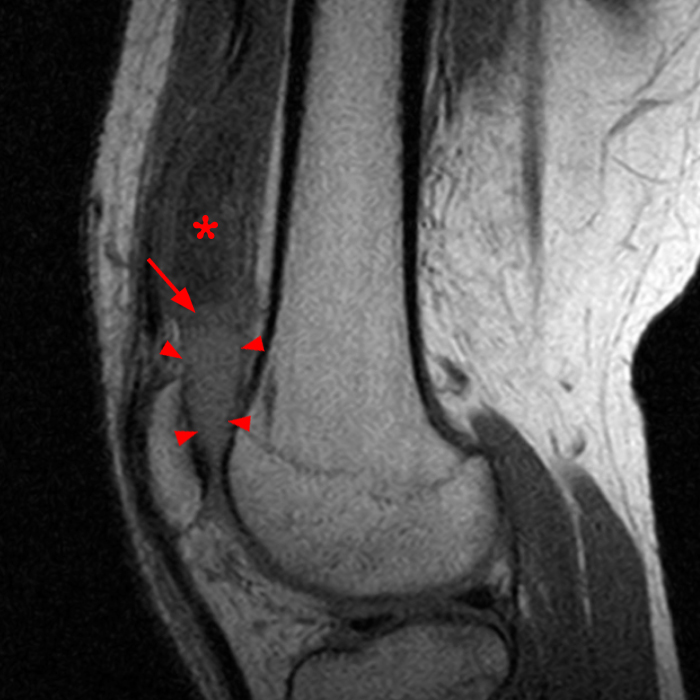
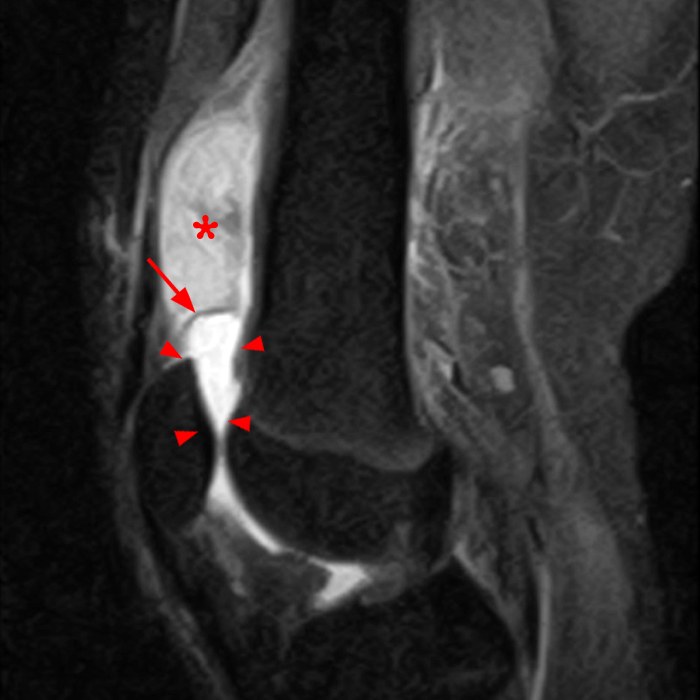
Case 2: A 58 year-old female presents with increasing suprapatellar pain and focal swelling for 1 year. Sagittal FSE proton density-weighted (Image 2A), sagittal FSE inversion recovery (Image 2B), and coronal FSE inversion recovery (Image 2C) images are available.
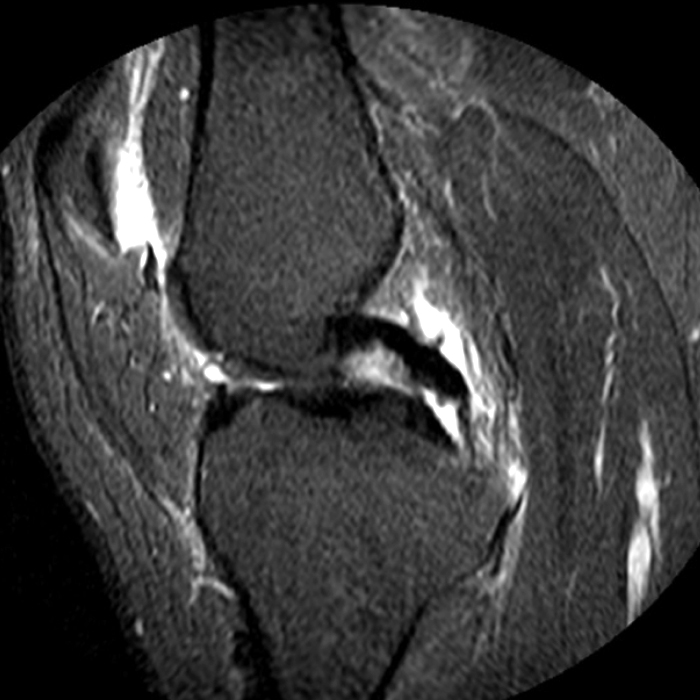
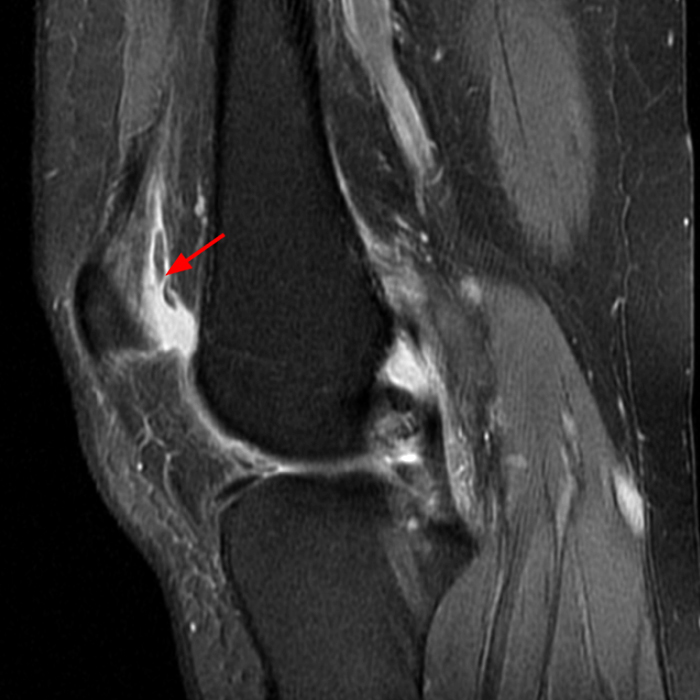
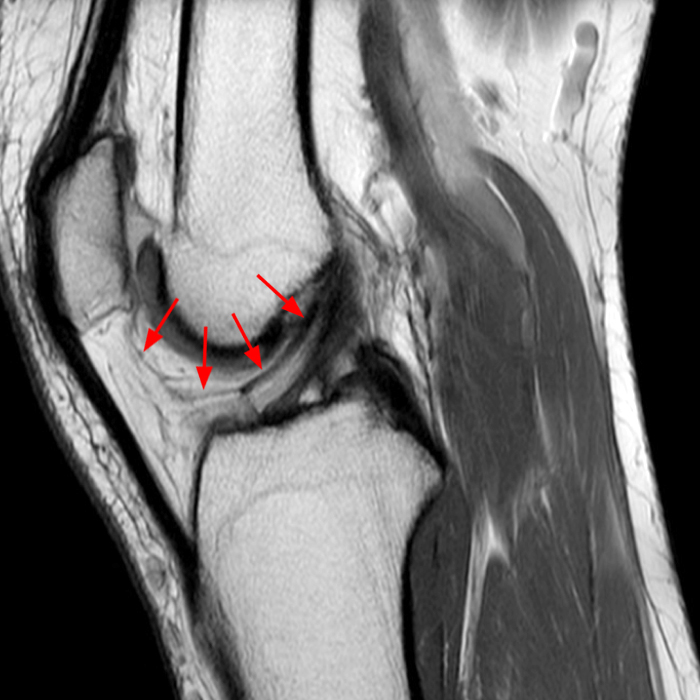
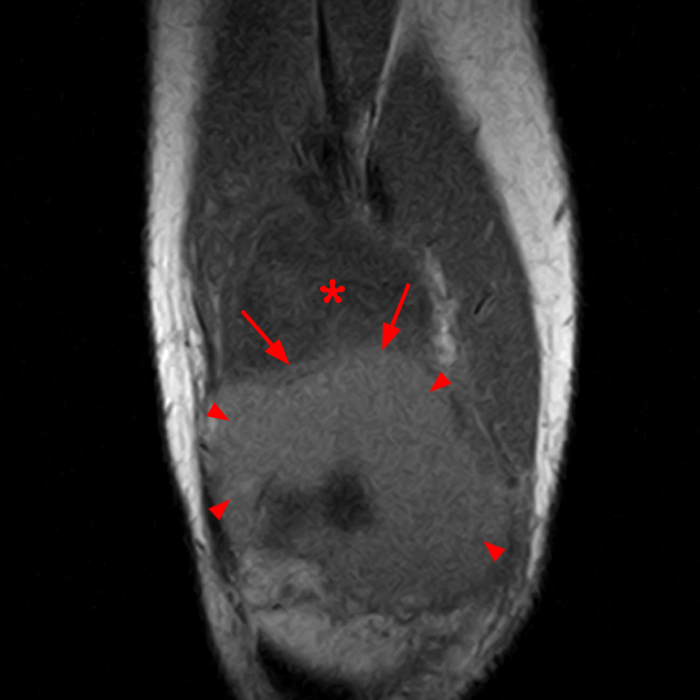
Case 3: a 15 year-old male soccer player with sudden onset of anterior knee pain during soccer practice. Sagittal fat-suppressed FSE proton density-weighted (Images 3A and 3B), sagittal T1-weighted (Images 3C and 3D), and axial fat-suppressed FSE proton density-weighted images (Images 3E) are provided.
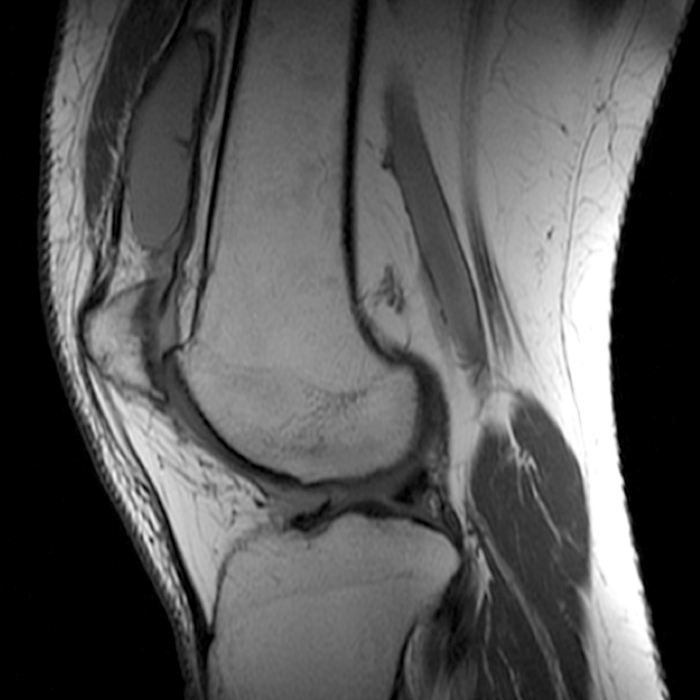
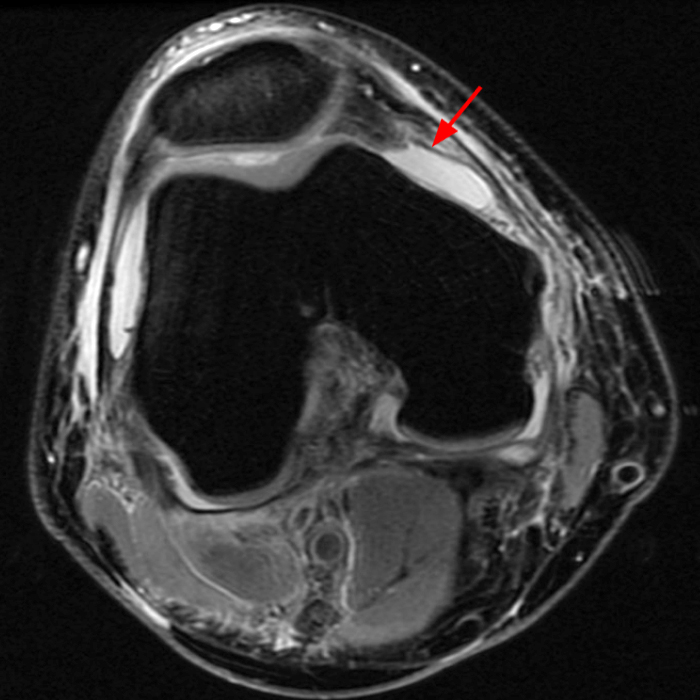
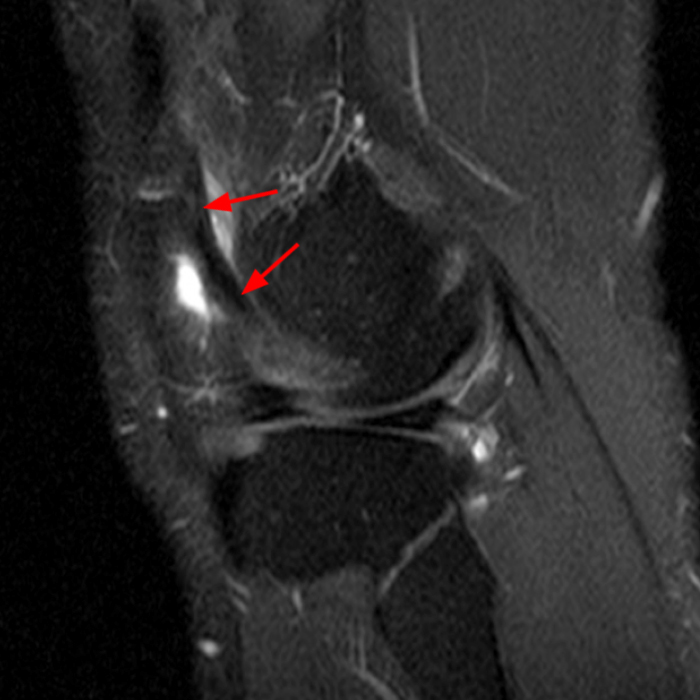
Case 4: A 55 year old male presents with anterolateral knee pain. Axial (4A) and coronal (4B) fat-suppressed FSE proton density-weighted images were obtained.
Findings and Diagnoses:
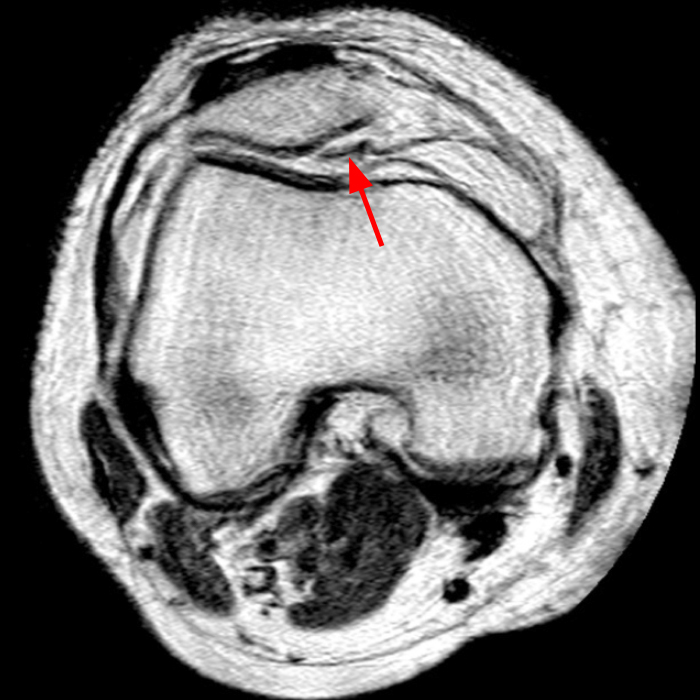
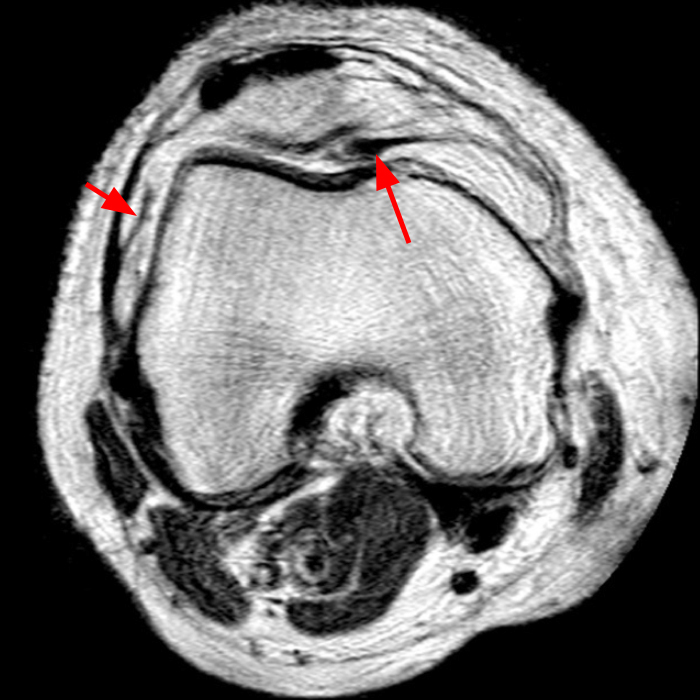
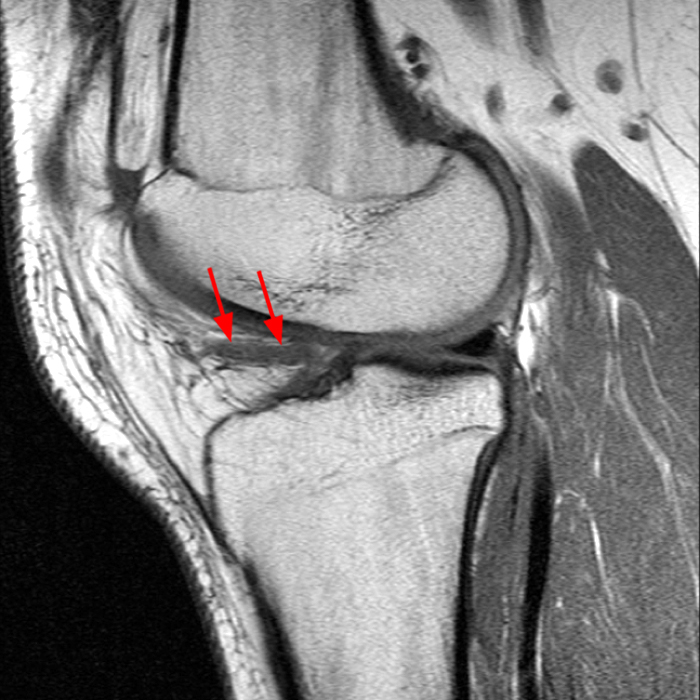
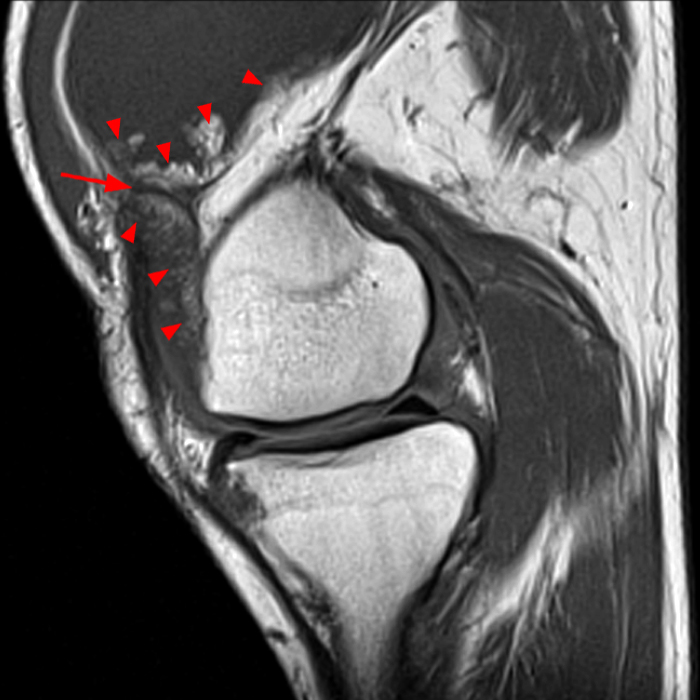
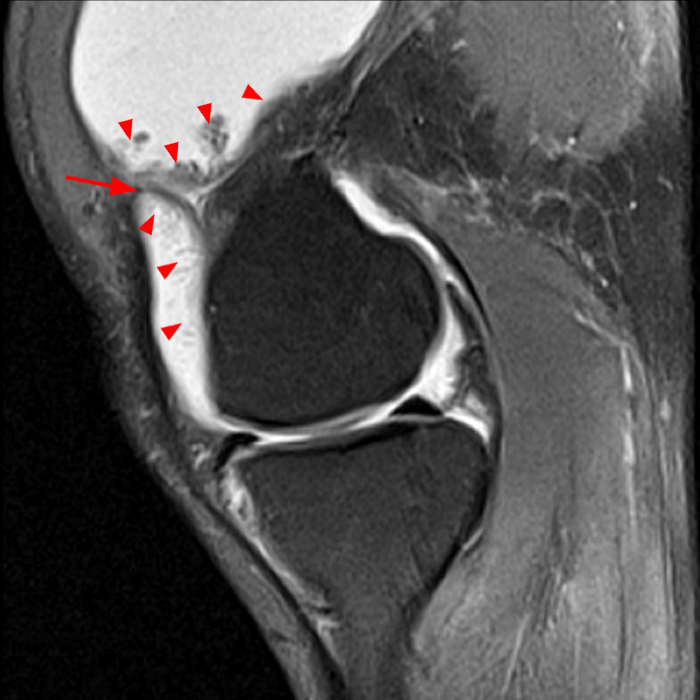
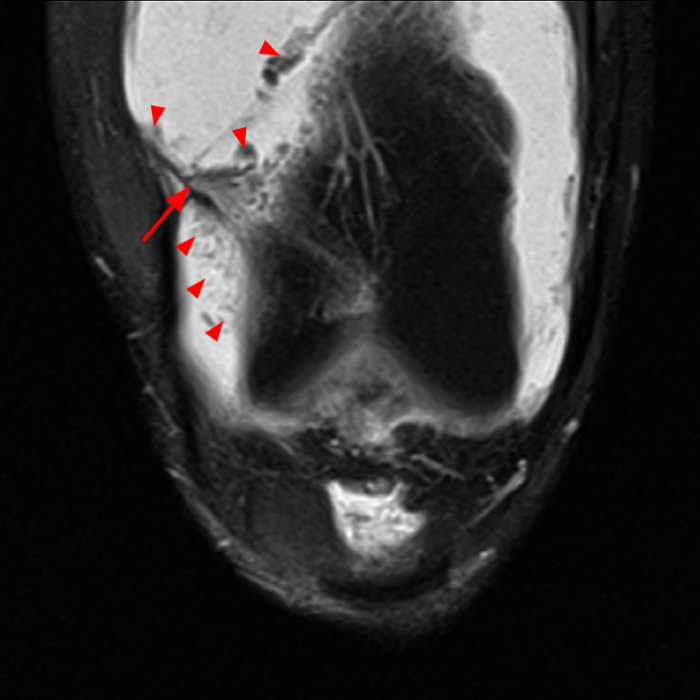
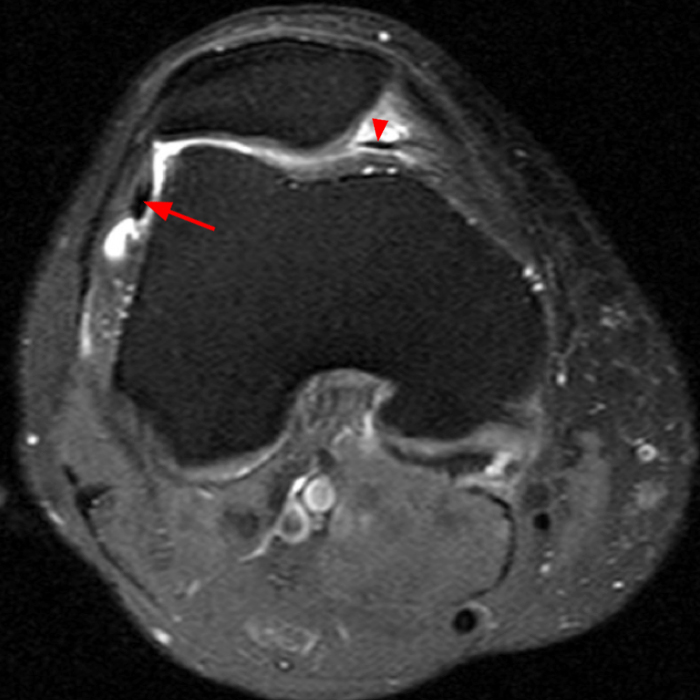
Case 1: 47-year-old male with anterior knee pain and swelling for 2 months.
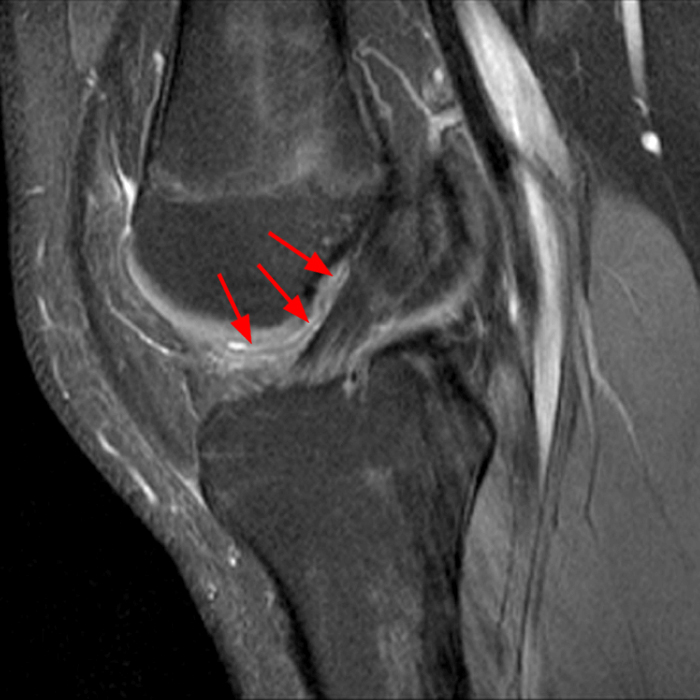
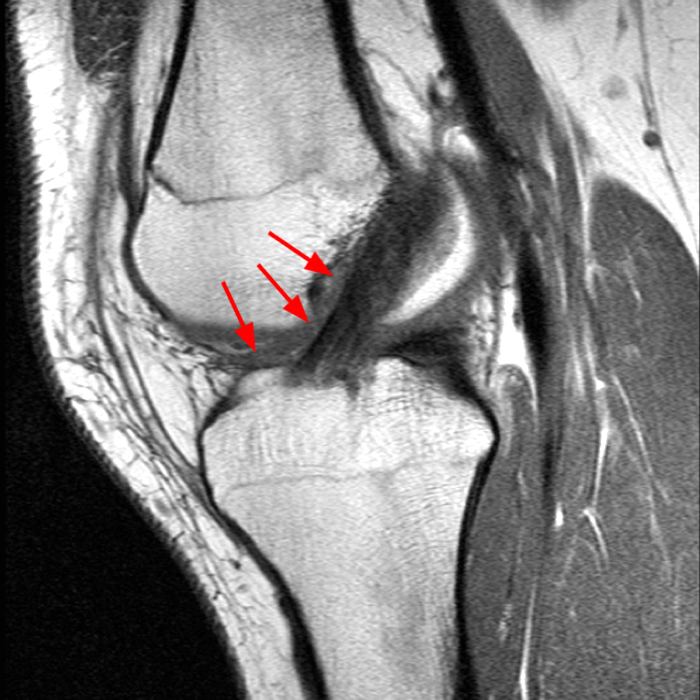
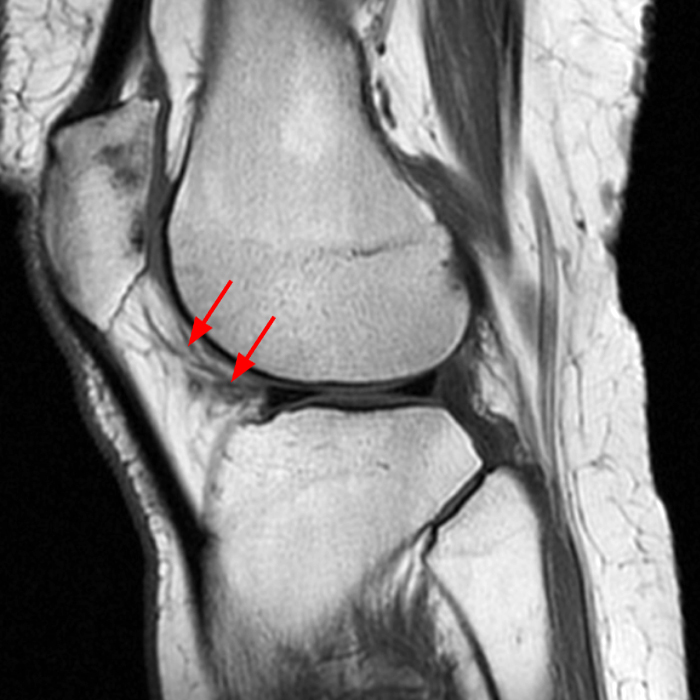
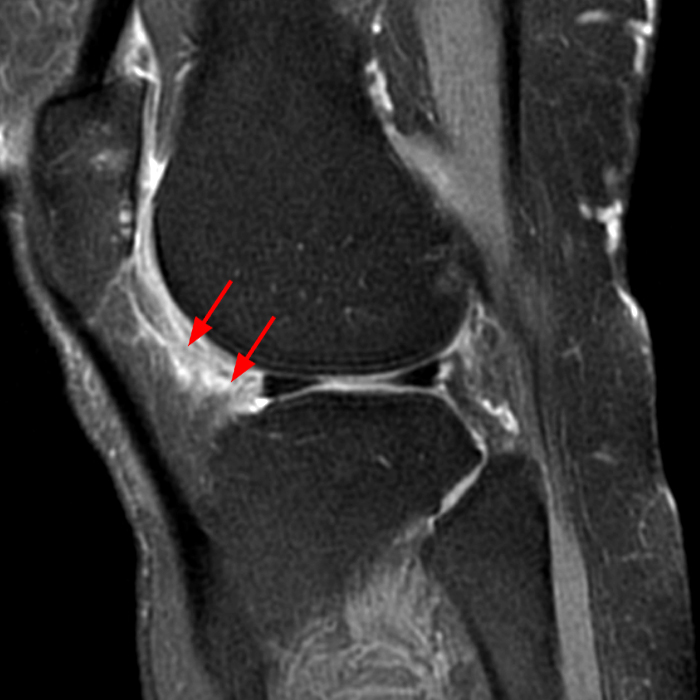
The axial and sagittal images demonstrate a thick, irregular medial synovial plica (arrows) protruding far into the medial patellofemoral joint, nearly contacting the median ridge of the patella. Severe articular cartilage loss is present along the lower medial patella (arrowheads). These findings are consistent with a chronically inflamed medial plica and erosion of adjacent articular cartilage. Incidentally noted is a normal lateral patellar plica on Image 5B (short arrow).
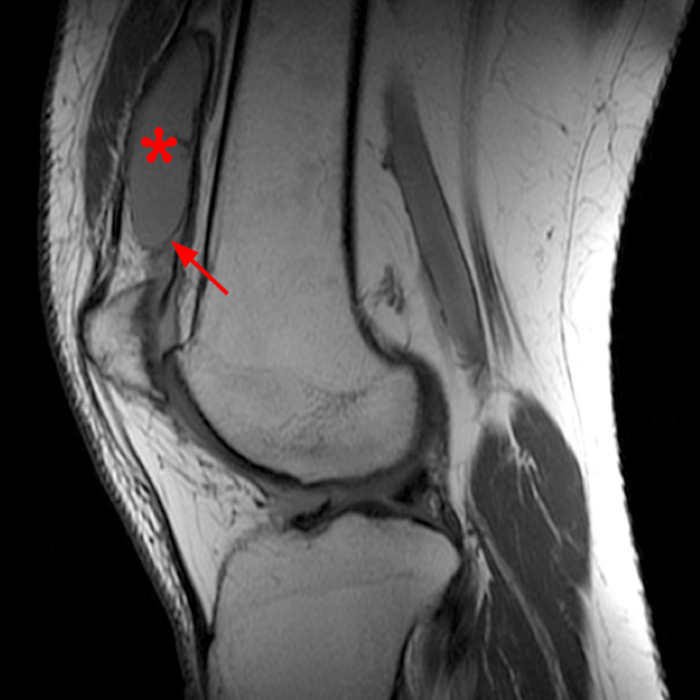
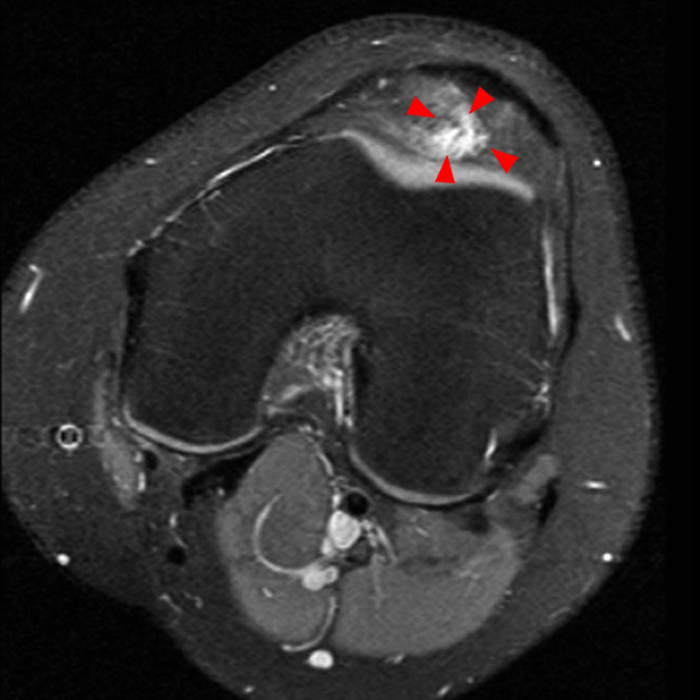
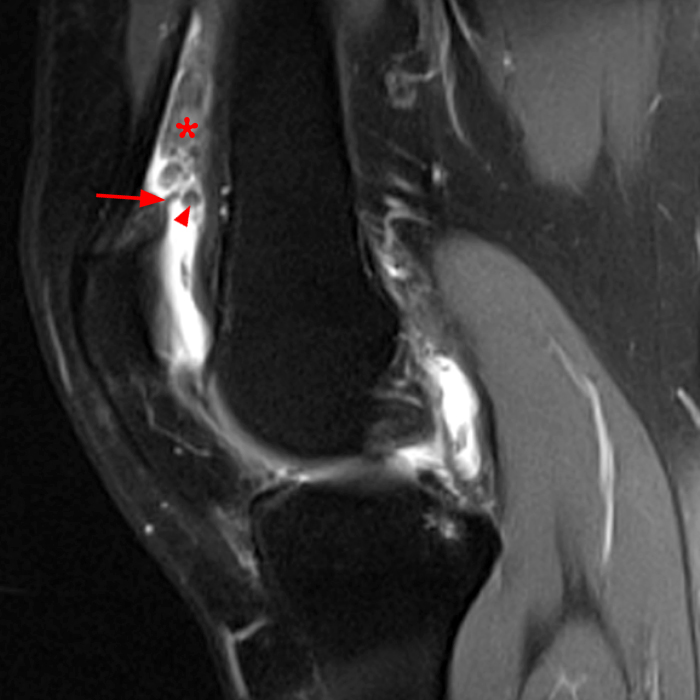
Case 2: 58-year-old female with increasing suprapatellar pain and focal swelling for 1 year.
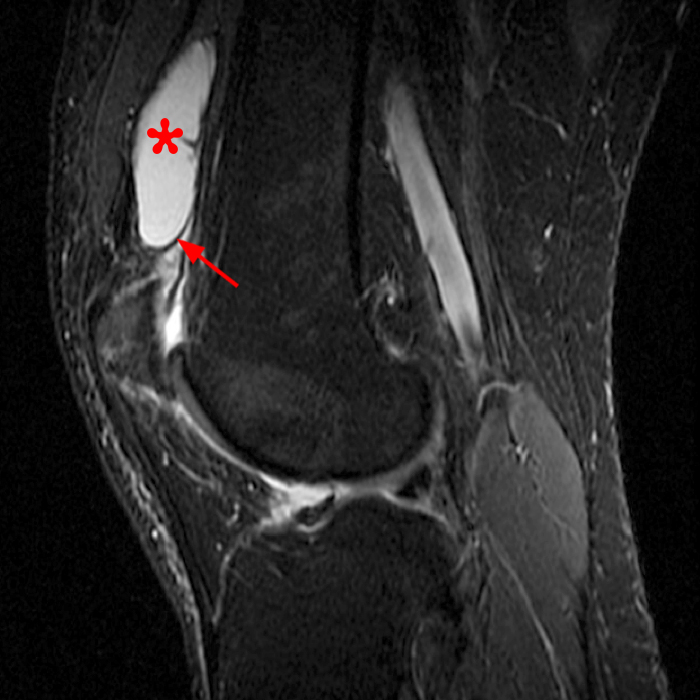
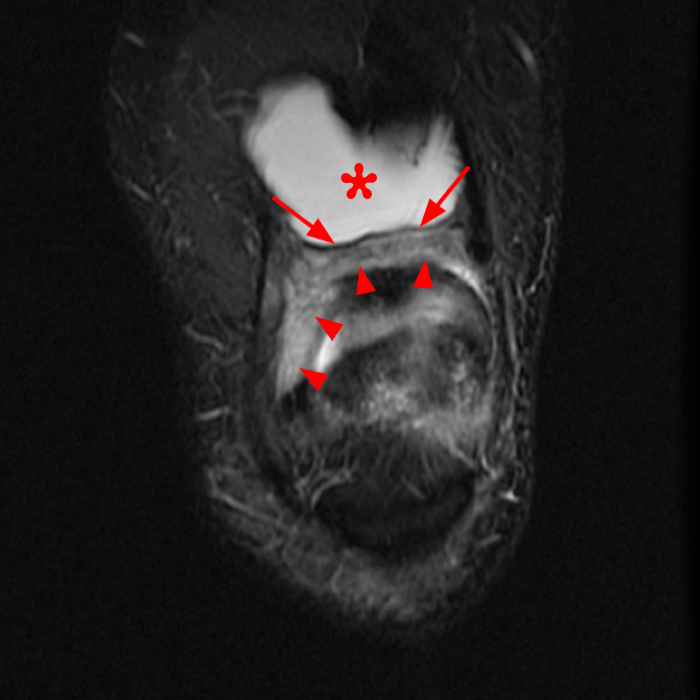
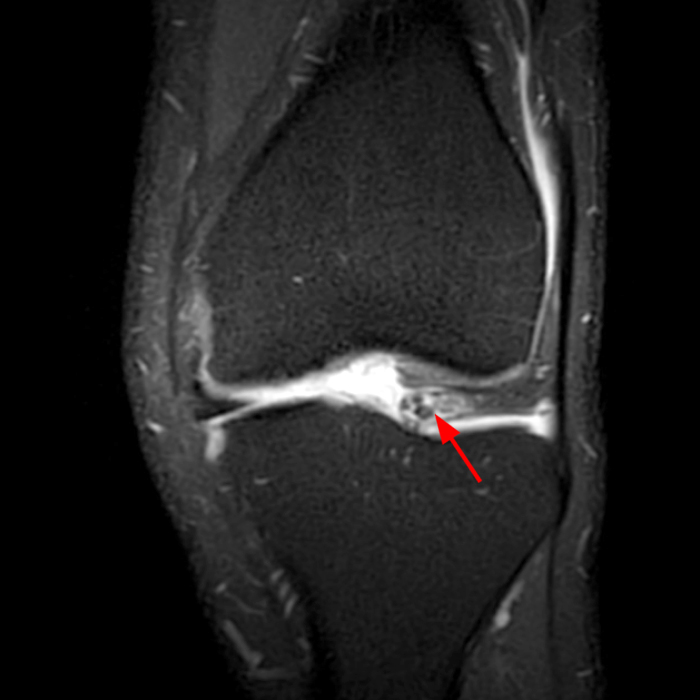
On the sagittal images (6A and 6B), a loculated fluid collection (asterisk) in the suprapatellar bursa is separated from the remainder of the knee joint by a well-defined thin hypointense septation (arrow), consistent with a suprapatellar plica causing compartmentalization. Fluid trapped above the plica distends the bursa, contributing to pain and causing a palpable abnormality on physical examination. The coronal image (6C) reveals diffuse synovitis within the joint (arrowheads), relatively sparing the suprapatellar loculation.
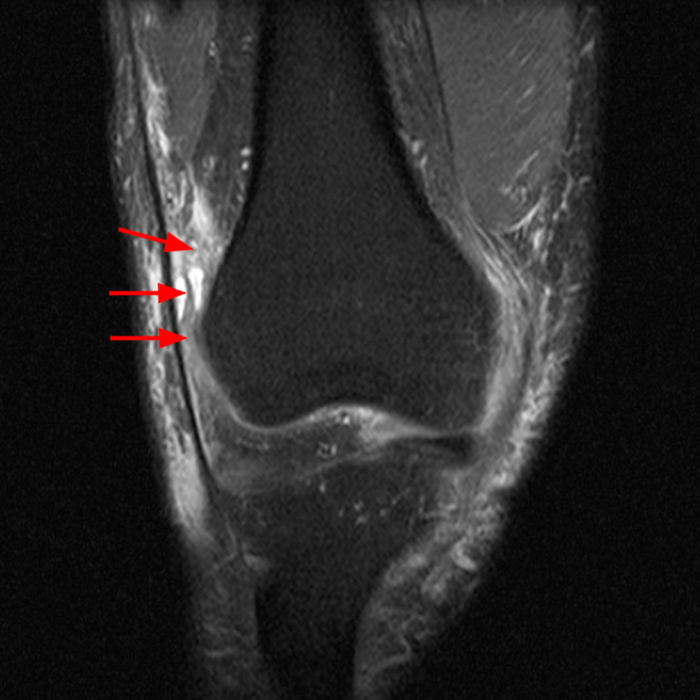
Case 3: 15-year-old male soccer player with sudden onset of anterior knee pain during soccer practice.
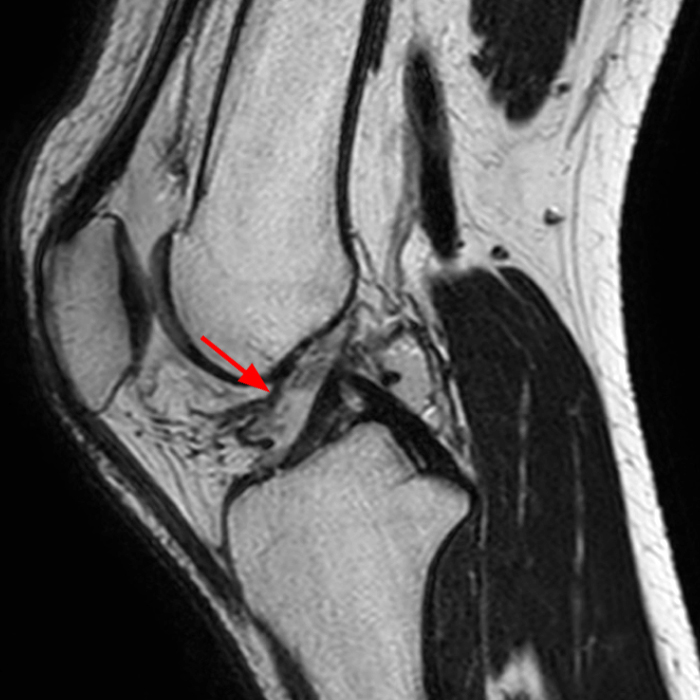
Diffuse edema is visible on the sagittal (7A and 7B) and axial (7E) fat-suppressed FSE proton density-weighted images within an irregular infrapatellar plica (ligamentum mucosum) (arrows) just anterior to the intact ACL, extending into the infrapatellar fat pad. Intermediate signal thickening is present on the T1-weighted images (7C and 7D) corresponding to the ligamentum mucosum (arrows). Edema in the fat pad is focal and curvilinear (arrowheads), located along the usual course of the infrapatellar plica. These findings are consistent with an acute sprain of the infrapatellar plica.
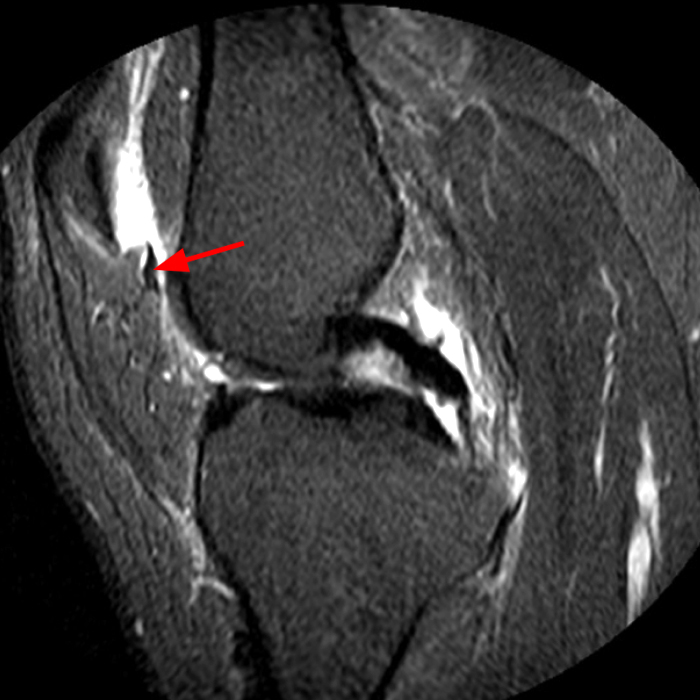
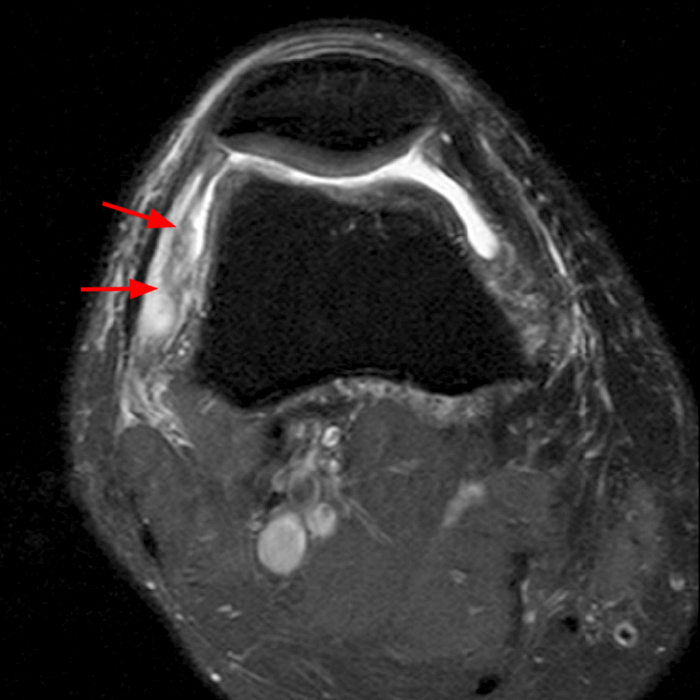
Case 4: 55-year-old male with anterolateral pain.
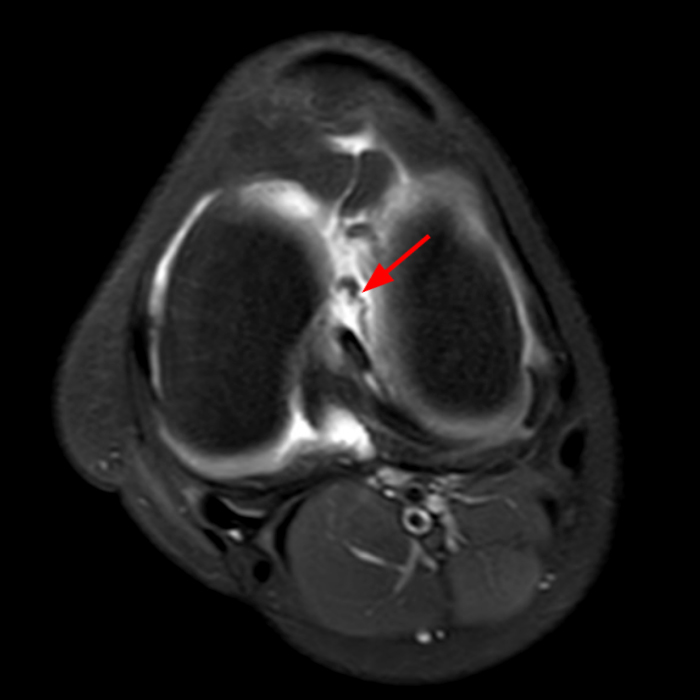
Axial (8A) and coronal (8B) fat-suppressed FSE proton density-weighted images demonstrate prominent irregular thickening of the lateral patellar plica with shaggy margins and intrasubstance heterogeneous signal, consistent with inflammation of the plica.
All four patients demonstrate abnormal synovial plicae of the knee.
Introduction
Synovial plicae of the knee are folds in the lining of the joint, thought to represent remnants from embryologic development. They are common incidental findings on cadaveric, arthroscopic and MRI examinations.1
The infrapatellar plica was first reported by Vesalius in the 16th century. Medial, lateral and suprapatellar plica were described by the early 20th century; a possible association of plicae with knee derangement was first suggested in 1918.2 Pipkin was the first investigator to focus on plicae as an etiology for knee symptoms in 1950.3
Embryology and Anatomy
The knee develops from mesodermal elements and is initially separated into three compartments, medial, lateral, and suprapatellar. The membrane separating the compartments is resorbed between the 9th to 12th week of gestation, forming a single joint compartment by the 16th week. One theory of plica formation hypothesizes that the plicae are remnants of this embryologic membrane which are not properly resorbed.4
Another theory of plica formation is based on the observation that the knee is initially filled with mesenchymal tissue at 7 weeks gestation. Cavitations within the mesenchyme coalesce to form the joint cavity by 10 weeks. In this theory, plicae are thought to represent areas of incomplete cavitation, with differentiation of residual mesenchymal tissue into synovial folds.5
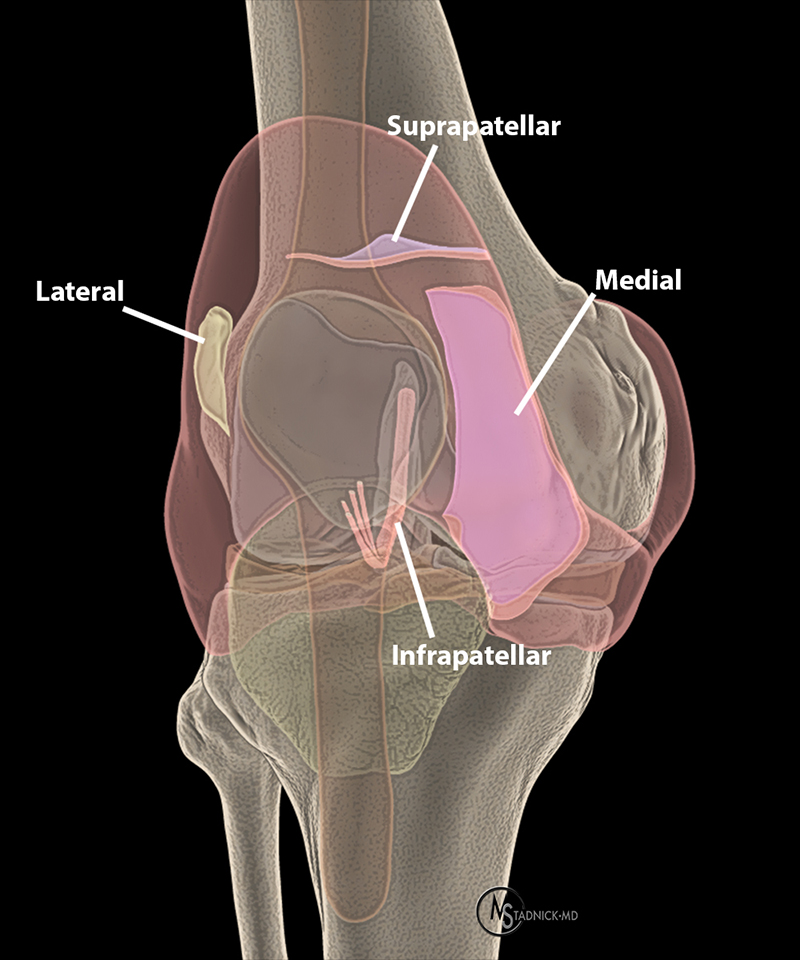
Four synovial plicae of the knee have been described, based on location: suprapatellar, infrapatellar, medial patellar, and lateral patellar. Prevalence varies widely in the literature, ranging from 14% to 100%. The arthroscopic series of Kim and Choe (1997) reported a prevalence of 87% for suprapatellar, 86% for infrapatellar, 72% for medial, and 1.3% for lateral patellar plicae.6
On gross examination, normal plicae are thin, flexible folds with a synovial lining around fibroelastic connective tissue protruding into the joint. The corresponding MRI appearance is a thin, linear hypointense object in the joint which is connected to the synovial lining, often outlined by joint fluid.7
Medial Patellar Plica
The medial patellar plica is the most frequently symptomatic knee plica, although it is less common than suprapatellar or infrapatellar plicae.8
It is known by various names in the literature, including mediopatellar plica, medial parapatellar plica, medial synovial shelf, plica alaris elongata, meniscus of the patella, Iion’s band, or Aoki’s ledge. The medial plica originates from the medial joint capsule and courses inferiorly to attach to the synovium of the infrapatellar fat pad. It may be either contiguous with or separate from a suprapatellar plica. The transverse width and lateral extent of the medial plica is variable.1
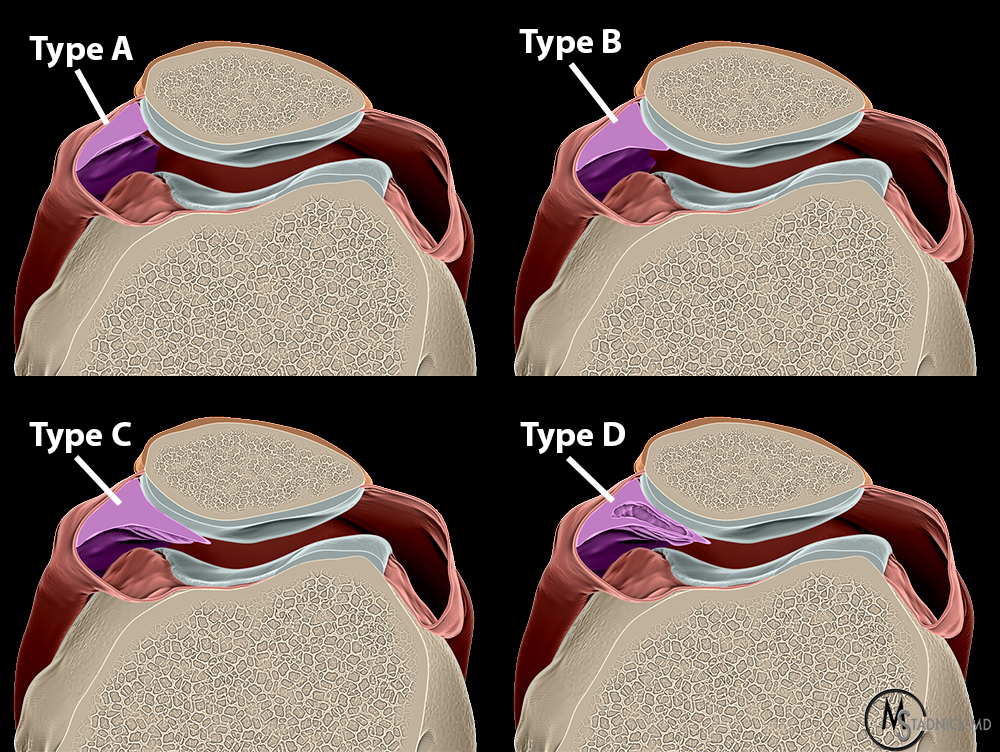
The Sakakibara classification describes 4 types of medial patellar plicae based on size and morphology. Types A and B are smaller and less likely to cause symptoms. The Type C medial patellar plica covers part of the articular surface of the femoral trochlea and is more frequently associated with symptoms. The Type D lesion is fenestrated and more likely to be symptomatic, but is relatively rare.9
Medial patellar plicae may become inflamed and symptomatic by many different mechanisms. Direct trauma, twisting injury, repetitive motion, increased activity, surgery, and various causes of intraarticular synovitis have all been reported as etiologies.2 Inflamed plicae become thickened and fibrotic, often with irregular margins. Such plicae are less flexible and more likely to cause mechanical symptoms during normal joint movement, such as snapping as it moves over the medial femoral trochlea and patella. Inflammation of the plica may be painful; as the disease progresses, associated articular cartilage loss and traction on adjacent synovium contribute to patient symptoms.1
MRI findings of pathologic medial plicae include thickening and increased intrasubstance signal on T2-weighted images, wide plica extending beyond the medial margin of the trochlear articular surface, corresponding to a Sakakibara Type C lesion, fenestration, corresponding to a Sakakibara Type D lesion, focal fluid adjacent to the plica out of proportion to the overall amount of joint fluid, and interposition between the patella and femoral trochlea on multiple contiguous images.7
Jee et al. reported overall sensitivity of 95% and specificity of 72% for MRI detection of symptomatic plicae in 55 patients. In their series, the incidence of symptomatic plicae increased when the medial plica extended beyond the medial margin of the patella into the patellofemoral joint.10 Monabang et al. reviewed 9 cases of arthroscopically significant medial patellar plicae and found that clinically significant plicae were interposed between the patella and trochlea on more axial slices than controls, and had adjacent focal fluid collections.11
Hayashi et al. reported that larger (Sakakibara Type C) medial patellar plicae on MRI were associated with a higher likelihood of medial patellar cartilage damage on review of 342 knees.12 Ultrasound has been reported to detect symptomatic medial plicae with a sensitivity of 90% and specificity of 83% in 68 knees.13
However, Boles et al. found no MR findings to be predictive of subsequent surgical resection of the medial plica in 46 cases.14 Diagnosis of symptomatic plica is primarily based on clinical findings, rather than imaging.14
Case 5 – Normal Sakakibara Type A medial patellar plica. Axial (11A) and sagittal (11B) fat-suppressed FSE proton density-weighted images in a 41 year old male demonstrate a thin uniform hypointense band in the medial joint (arrows), outlined by a joint effusion.
Case 6 – Progressive thickening of a Type B medial patellar plica over 7 months. Axial fat-suppressed FSE proton density-weighted images of a 45 year-old male initially demonstrate a normal thin medial patellar plica (arrow) (12A). Follow-up MRI 7 months later reveals prominent thickening of the medial plica (arrow) with intrasubstance heterogeneous signal. A loose body is also visible in the lateral gutter (arrowhead) (12B).
Case 7 – Thickened Type C medial patellar plica – Axial FSE proton density-weighted images (Images 13A, 13B) in a 57-year-old male with anterior knee pain reveal a prominent irregular medial patellar plica (arrows) in the medial patellofemoral joint. Articular cartilage thinning and surface fibrillation are present along the lower medial patella (arrowheads).
Case 8 – Fenestrated Type D medial patellar plica – a 56-year-old female presented with anterior knee pain. Axial (14A) and sagittal (14B) fat-suppressed FSE proton density-weighted images reveal a thickened medial patellar plica with a central perforation (arrows), protruding into the patellofemoral joint. Associated articular cartilage fibrillation with subchondral marrow edema (arrowheads) in the medial patella.
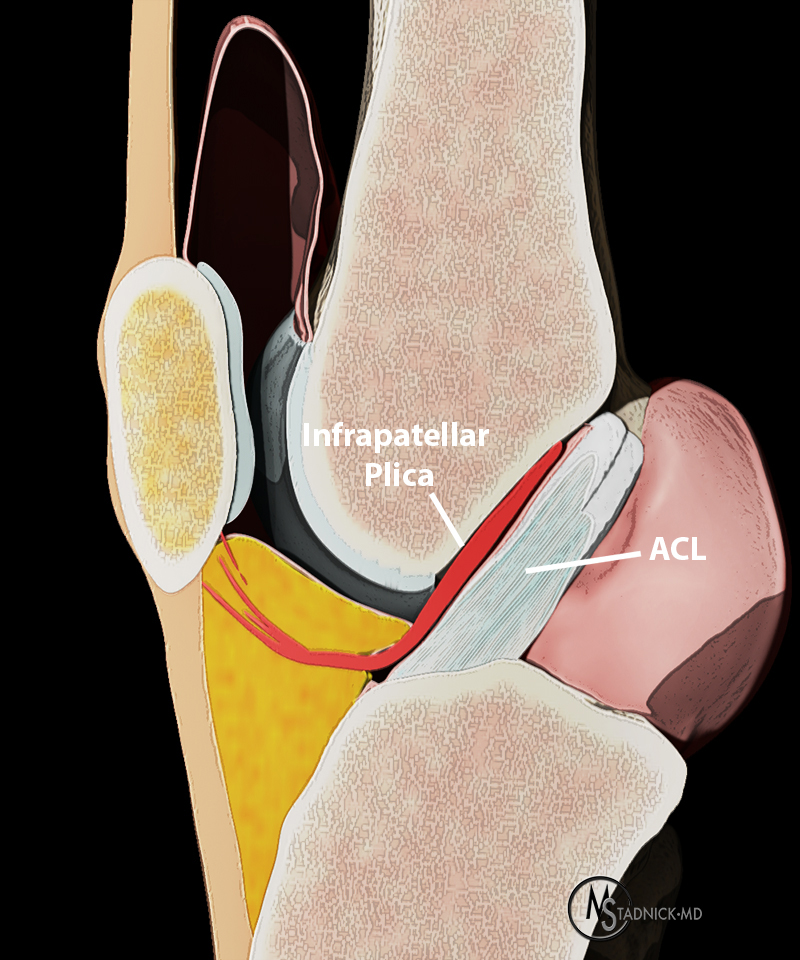
The infrapatellar plica is the most frequently seen knee plica at arthroscopy. It is also known as the ligamentum mucosum, inferior plica, infrapatellar fold, or anterior plica.15 This structure courses anteriorly and inferiorly from a small femoral origin anterior to the ACL, courses parallel to the ACL to the infrapatellar fat pad, then curves upward through the infrapatellar fat pad to attach to the inferior patella. The infrapatellar plica may also partially attach to the anterior transverse meniscomeniscal ligament, ACL or anterior horn of the lateral meniscus.16,17
The anteromedial meniscofemoral ligament may mimic the infrapatellar plica on MRI; this structure is present in up to 15% of the population, coursing anterior to the ACL in the intercondylar notch, attaching to the anterior horn of the medial meniscus and adjacent tibia. The anteromedial meniscofemoral ligament does not enter the infrapatellar fat pad or attach to the patella.18
Infrapatellar plicae have been classified by Kim et al. into four types based on morphology, including the “Separate Type” completely separated from the ACL (60.5%); the “Split Type” separate from the ACL and longitudinally divided (13.5%); a “Vertical Septum Type” attached to the ACL (10.5%); and the “Fenestra Type” similar to the Vertical Septum Type with a fenestration (1%). The infrapatellar plica was completely absent in 14.5% of their arthroscopic series.19
Acute injury causing sprain or tear of the infrapatellar plica can result in hemarthrosis, pain, and a locking sensation.17 Less commonly, the infrapatellar plica may become inflamed and symptomatic without acute injury, similar to medial patellar plicae. Compartmentalization due to a complete septum separating the medial and lateral compartments can occur, but is rare.20,21
On MR imaging, normal infrapatellar plicae are thin and often difficult to distinguish from the adjacent ACL. A normal cleft in the infrapatellar fat pad may occur in up to 90% of patients just below the infrapatellar plica, known as the horizontal cleft of the infrapatellar fat pad or ligamentum mucosum recess; this cleft may be distended by joint fluid, contain intraarticular bodies or other pathology.22 A thickened, irregular infrapatellar plica on MRI suggests inflammation or previous injury. Acute injury of the infrapatellar plica may cause edema within the infrapatellar fat pad in a characteristic curvilinear pattern, following the course of the plica. Contusion of the infrapatellar fat pad can result in concurrent Hoffa disease along with sprain of the infrapatellar plica.17
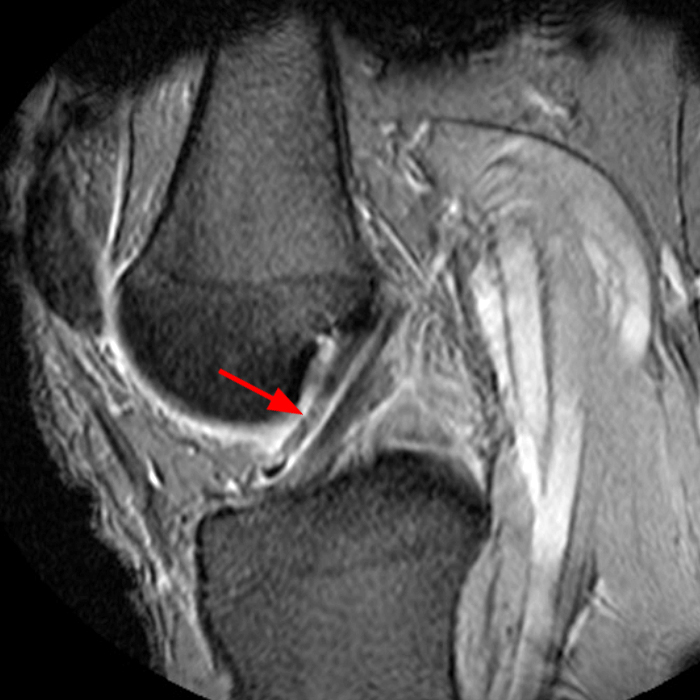
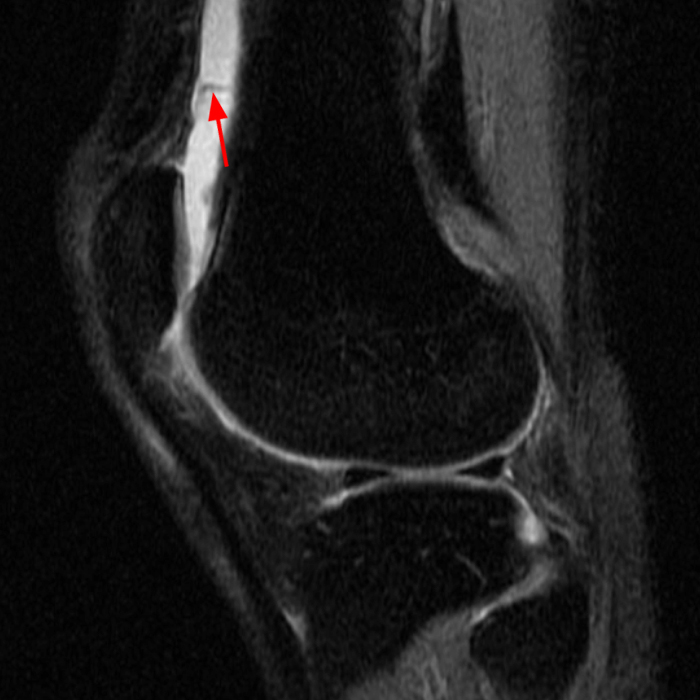
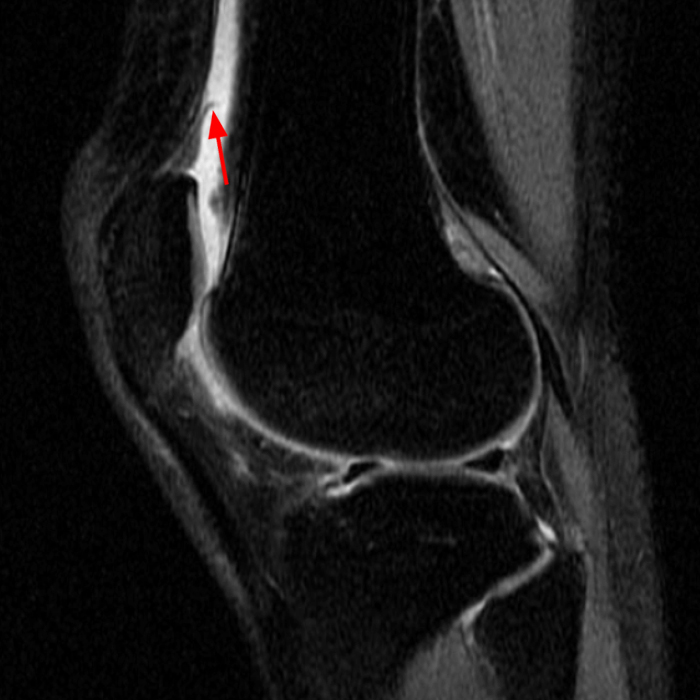
Case 9 – Normal infrapatellar plica. A sagittal FSE proton density-weighted (16) image in a 47 year-old female visualizes a normal infrapatellar plica (arrows), coursing anterior to the ACL into the infrapatellar fat pad, curving upward to the lower pole of the patella.
Case 10 – Anteromedial meniscofemoral ligament. Sagittal GRE (17A) and coronal FSE STIR (17B) images in a 72-year-old female demonstrate a bandlike structure anterior to the ACL, attaching to the anterior horn of the medial meniscus near the meniscal root (arrows). This ligament is very similar in appearance to the infrapatellar plica on sagittal images, but is more medial in location and does not extend into the infrapatellar fat pad.
Cases 11A and 11B – Two different patients with prominent horizontal clefts of the infrapatellar fat pad. A sagittal FSE T2-weighted image in a 44 year-old male with a joint effusion demonstrates joint fluid filling the horizontal cleft (asterisk) (18A). Note that the infrapatellar plica forms the roof of the cleft (arrow). Sagittal (18B) and coronal (Image 18C) fat-suppressed FSE proton density weighted images in a 71 year-old male reveal a cluster of small cartilaginous loose bodies and debris (arrows) lodged in the cleft.
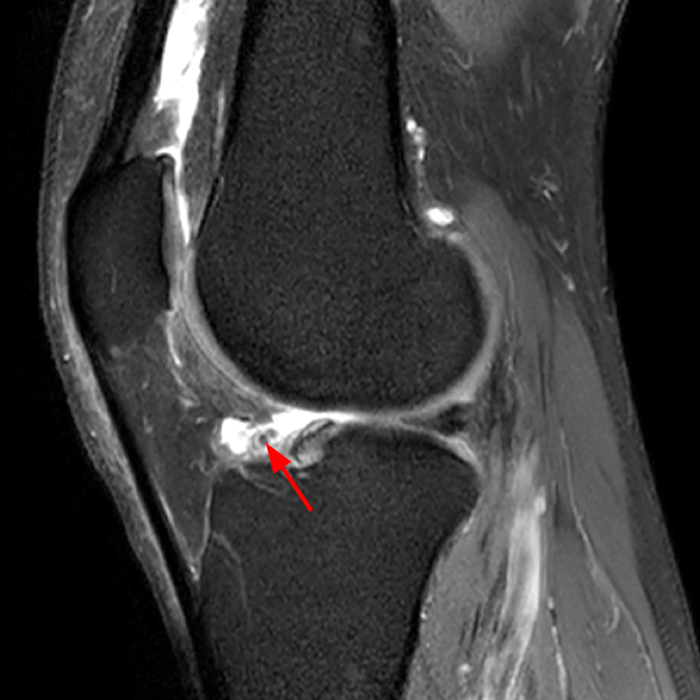
Case 12 – Infrapatellar plica injury. A 26 year-old female reports anterior left knee pain after an athletic injury. Sagittal FSE T2-weighted (19A) and axial fat-suppressed FSE proton density-weighted (19B) images reveal a thickened irregular infrapatellar plica with intrasubstance increased signal, suspicious for partial tear of the plica. The ACL remains intact.
Case 13 – Edema along the infrapatellar plica in the infrapatellar fat pad, suspicious for subacute sprain. A 54 year-old male reports persistent anterior pain for 4 months after a twisting injury at work. Sagittal FSE proton density-weighted (20A) and fat-suppressed FSE proton density-weighted (20B) images are shown. Focal edema in the infrapatellar fat pad extends in a curvilinear pattern to the lower patella along the infrapatellar plica (arrows), suspicious for sprain of the plica.
Case 14 – Edema in the infrapatellar fat pad extending along the infrapatellar plica. A 68-year-old female presents with persistent anterior pain after injury. Sagittal T1-weighted (21A) and fat-suppressed FSE proton density-weighted (21B) images reveal edema in the infrapatellar fat pad surrounding the infrapatellar plica (arrows). Edema is more extensive than in Case 13, and probably indicates contusion and inflammation of the infrapatellar fat pad (Hoffa disease) in addition to sprain of the plica.
The suprapatellar plica is the second most common plica after the infrapatellar plica, with a reported prevalence of up to 89% at autopsy. Alternative names include superior plica, supramedial plica, medial suprapatellar plica, or suprapatellar septum. The suprapatellar plica courses from the anterior femoral metaphysis to the posterior quadriceps tendon, and from the medial to the lateral wall of the joint. The medial portion may merge with a medial patellar plica, if present.1
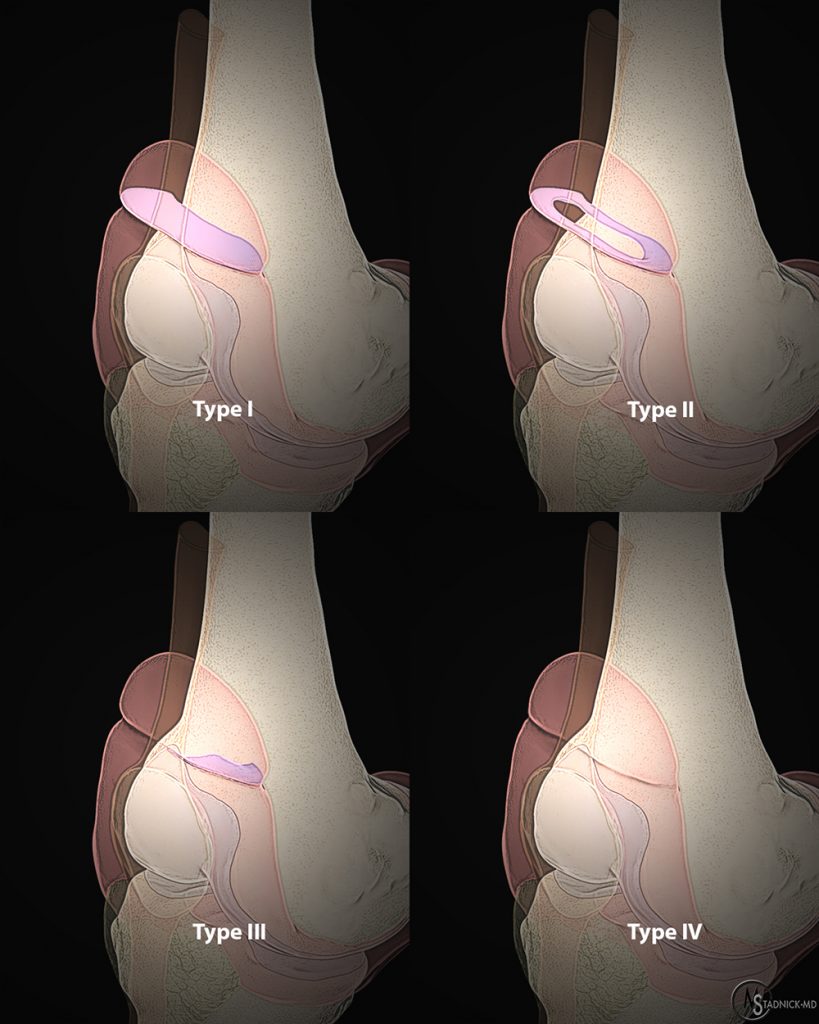
The Zidorn classification of suprapatellar plicae describes 4 types: Type I is a complete septum without communication between the knee joint and suprapatellar bursa; Type II is a perforated septum with one or multiple openings (called porta) allowing passage of fluid; Type III demonstrates a small arcuate plica, usually medial; and Type IV representing complete absence of the plica.23
Suprapatellar plicae are usually asymptomatic. Inflammation of the suprapatellar plica can occur and progress similar to the medial plica syndrome, although less frequently. Suprapatellar plicae may also impinge on the medial femoral condyle during flexion, limiting patellar mobility and causing chondral damage.3,24,25,26
Compartmentalization of the suprapatellar bursa may occur due to a complete Type I plica, or a Type II plica with a small porta. This results in a palpable suprapatellar mass and pain due to trapped fluid. Intraarticular loose bodies may become lodged in the suprapatellar bursa due to a check-valve mechanism after passing through the porta from the joint. Infection or PVNS may be confined to the suprapatellar bursa by a suprapatellar plica.27,28,29
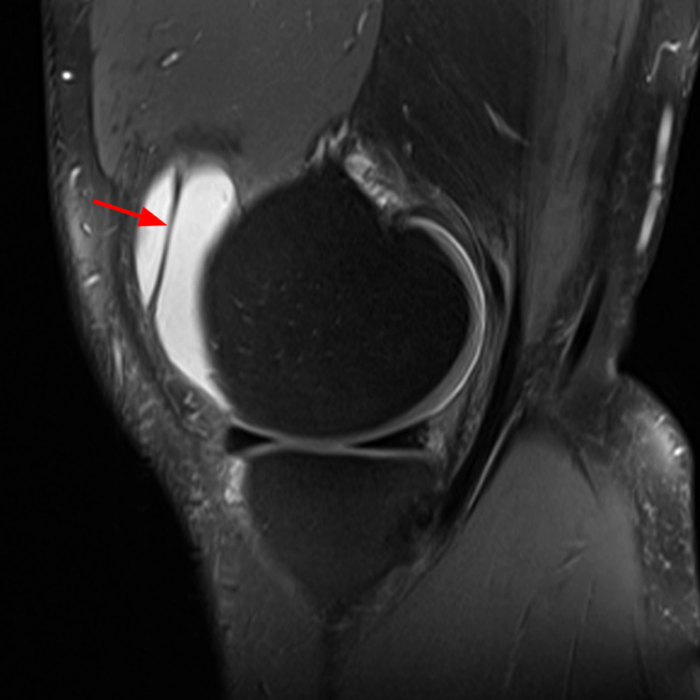
Case 15 – Normal suprapatellar plica. 33 year-old female with pain and joint effusion. Joint fluid outlines a uniformly thin, delicate suprapatellar plica (arrows) with a posterior perforation on sagittal (23A, 23B) and axial (23C) fat-suppressed FSE proton density-weighted images.
Case 16 – Thickened suprapatellar plica with cartilaginous loose body in the suprapatellar bursa. 39 year-old male with pain and swelling for 2 weeks. Sagittal FSE T2-weighted (24A) and coronal fat-suppressed FSE proton density-weighted images (24B) demonstrate a near-complete Zidorn Type II thickened and irregular suprapatellar plica (arrows), with a hypointense body lodged in the suprapatellar bursa (asterisk).
Case 17 – Compartmentalization of the suprapatellar bursa due to a complete suprapatellar plica. A 15 year-old patient presented after trauma with hemarthrosis and a palpable suprapatellar mass. Sagittal T1-weighted (25A), sagittal STIR (25B), and coronal T1-weighted (25C) images demonstrate a complex loculated collection in the suprapatellar bursa (asterisk) separated from the remainder of the knee by a thick Zidorn Type I complete suprapatellar plica (arrow). Hemarthrosis in the knee is hyperintense on the T1-weighted images and does not involve the bursa (arrowheads).
Case 18 – Lipoma arborescens involving the suprapatellar plica. 40 year-old male with chronic synovitis, knee pain and swelling. Sagittal T1-weighted (26A), sagittal fat-suppressed FSE proton density-weighted (26B), and coronal fat-suppressed FSE proton density-weighted (26C) images demonstrate villous lipomatous synovial proliferation (arrowheads) surrounding a thickened suprapatellar plica (arrows).
Case 19 – PVNS predominantly involving the suprapatellar bursa. 32 year-old male with intermittent pain and swelling for 1 year. Sagittal FSE proton density-weighted (27A), sagittal (27B) and coronal (27C) fat-suppressed FSE proton density-weighted images reveal multiple intermediate to low signal masses in the suprapatellar bursa (asterisks). A Zidorn Type II suprapatellar plica (arrows) confines most of the synovial masses to the suprapatellar bursa, but smaller lesions (arrowheads) can pass through the porta into the remainder of the joint.
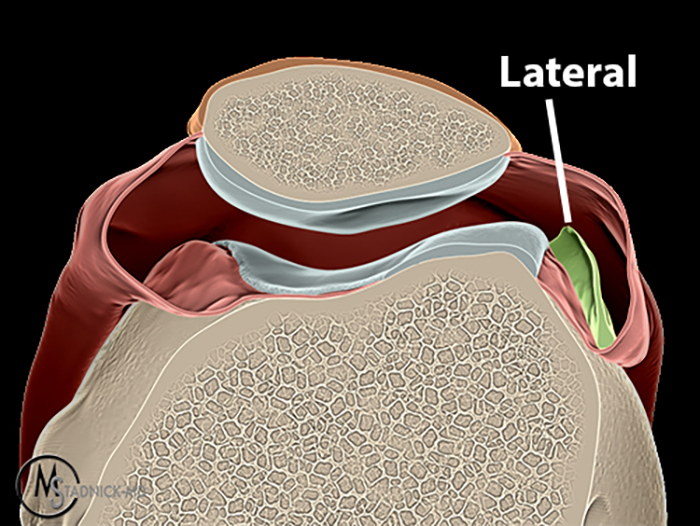
Lateral Patellar Plica
The lateral patellar plica is the least common of the four knee plicae. Most authors report a frequency of 1-3%; Gurbuz et al. found an incidence of 20.7% on arthroscopy and 50% at cadaveric dissection.1,30 Lateral plicae originate from the lateral joint capsule, coursing in the lateral gutter of the knee, attaching to the infrapatellar fat pad. It should be distinguished from other synovial folds in the lateral gutter, such as the lateral alar fold, superolateral fold, and transverse synovial arcuate fold.1,5
The lateral patellar plica can become inflamed and fibrotic, like other knee plicae. Patients with lateral patellar plica syndrome may report lateral pain and snapping; on physical examination, a palpable lateral cord may be present with associated tenderness.1,31
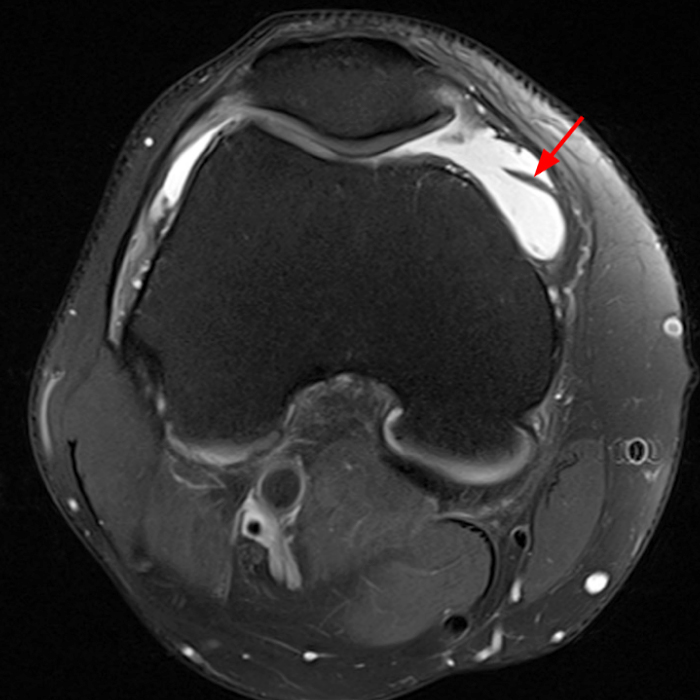
Normal lateral patellar plicae can be seen on MRI as a thin hypointense band lateral to the lateral femoral condyle on axial images.1,5 Increased signal on T2-weighted images, thickening and irregularity suggest inflammation of the lateral plica.31
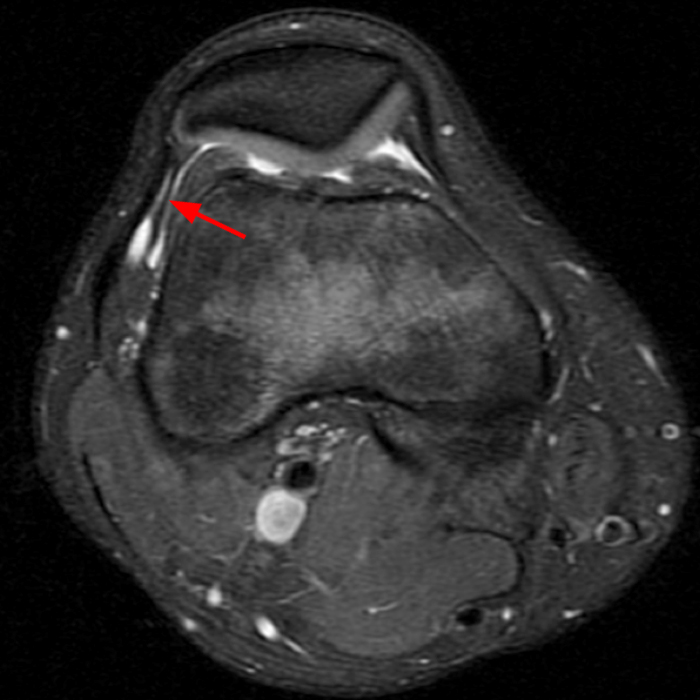
Case 20 – Normal lateral patellar plica. 16 year-old male athlete with lateral knee pain. Axial fat-suppressed FSE proton density-weighted images (29A and 29B) demonstrate a thin, uniform lateral patellar plica (arrows).
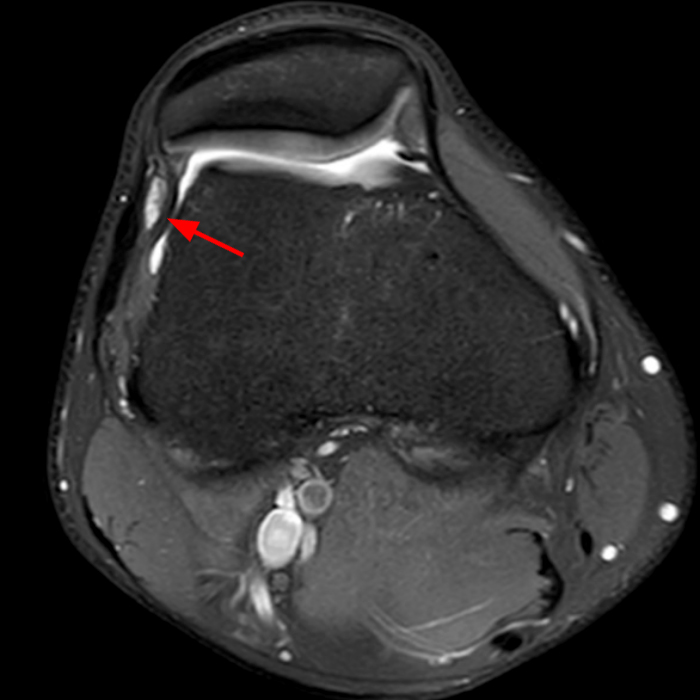
Case 21 – Thickened and irregular lateral patellar plica. 30 year-old male with knee pain. Axial (30A) and coronal (30B) fat-suppressed FSE proton density-weighted images show a thick lateral patellar plica (arrows) with irregular morphology on the coronal series.
Case 22 – Prominently thickened, cordlike lateral patellar plica. 34 year-old male with anterolateral snapping sensation and pain for 3 weeks. Coronal (31A), sagittal (31B) and axial (31C) fat-suppressed FSE proton density-weighted images reveal a prominent bandlike hypointense object in the anterolateral knee joint (arrows), consistent with a thickened lateral plica. A thin medial plica (arrowhead) is also visible on the axial image.
Treatment
Conservative treatment of symptomatic plicae includes rest, NSAIDs to reduce inflammation and pain, and physical therapy. Intraarticular or intraplica injection of medication may be useful in some patients.32,33,34,35,36
If symptoms persist, complete resection of the symptomatic plica usually results in long term relief. Division or incomplete resection of a plica may result in healing of the plica, with recurrence of symptoms. Surgery for a complete or near-complete suprapatellar plica may require careful planning to avoid contamination by pathology confined to the suprapatellar compartment.28,29,32,35,36,37
Conclusion
Synovial plicae of the knee are normal anatomic structures frequently encountered at MR imaging and arthroscopy. Most plicae are asymptomatic. Occasionally, plicae may become inflamed and thickened, causing pain, cartilage damage, and mechanical symptoms. Compartmentalization of the suprapatellar bursa due to a complete or near-complete suprapatellar plica can produce a palpable abnormality along with pain.
Diagnosis of symptomatic plicae is based on clinical findings. MRI can detect abnormal plicae, as well as other intra-articular pathology which may account for patient symptoms. The radiologist should document the presence and morphology of plicae, associated chondral abnormalities, and other pathology which may cause patient symptoms.
References
- García-Valtuille R, Abascal F, Cerezal L, García-Valtuille A, Pereda T, Canga A, Cruz A. Anatomy and MR imaging appearances of synovial plicae of the knee. Radiographics. 2002 Jul-Aug;22(4):775-84. Review. Erratum in: Radiographics 2002 Nov-Dec;22(6):1516. ↩
- Schindler OS. Synovial plicae of the knee. Current Orthopaedics 2004; 18. 210-219. ↩
- Pipkin G. Lesions of the suprapatellar plica. J Bone Joint Surg Am. 1950 Apr;32A(2):363-9. ↩
- Hoehmann CL. Plica syndrome and its embryological origins. Edorium J Orthop 2017;3:1–12. ↩
- Boles CA, Martin DF. Synovial plicae in the knee. AJR Am J Roentgenol. 2001 Jul;177(1):221-7. ↩
- Kim SJ, Choe WS. Arthroscopic findings of the synovial plicae of the knee. Arthroscopy. 1997;13:33-41. ↩
- Bui-Mansfield LT, Chen DC. Asymptomatic and Symptomatic Synovial Plicae of the Knee Joint. Contemporary Diagnostic Radiology. December 15th, 2016 – Volume 39(25):1–7. ↩
- Vassiou K, Vlychou M, Zibis A, Nikolopoulou A, Fezoulidis I, Arvanitis D. Synovial plicae of the knee joint: the role of advanced MRI. Postgrad Med J. 2015 Jan;91(1071):35-40. ↩
- Sakakibara J. Arthroscopic study on Iino’s band (plica synovialis mediopatellaris). J Jpn Orthop Assoc 1974; 50:513–522. ↩
- Jee WH, Choe BY, Kim JM, et al. The plica syndrome: diagnostic value of MRI with arthroscopic correlation. J Comput Assist Tomogr. 1998;22:814-818. ↩
- Monabang CZ, De Maeseneer M, Shahabpour M, et al. MR imaging findings in patients with a surgically significant mediopatellar plica. JBR-BTR.2007;90:384-387. ↩
- Hayashi D, Xu L, Guermazi A, Kwoh CK, Hannon MJ, Jarraya M, Green SM, Jakicic JM, Moore CE, Roemer FW. Prevalence of MRI-detected mediopatellar plica in subjects with knee pain and the association with MRI-detected patellofemoral cartilage damage and bone marrow lesions: data from the Joints On Glucosamine study. BMC Musculoskelet Disord. 2013 Oct 12;14:292. ↩
- Paczesny L, Kruczynski J. Medial plica syndrome of the knee: diagnosis with dynamic sonography. Radiology 2009;251:439-446. ↩
- Boles CA, Butler J, Lee JA, Reedy ML, Martin DF. Magnetic resonance characteristics of medial plica of the knee: correlation with arthroscopic resection. J Comput Assist Tomogr. 2004 May-Jun;28(3):397-401. ↩
- Dupont J: Synovial plicae of the knee: Controversies and Review. Clin Sports Med 16: 87-122, 1997. ↩
- Kosarek FJ, Helms CA. The MR appearance of the infrapatellar plica. AJR Am J Roentgenol. 1999;172:481-484. ↩
- Cothran RL, McGuire PM, Helms CA, et al. MR imaging of infrapatellar plica injury. AJR Am J Roentgenol. 2003;180:1443-1447. ↩
- Soejima T, Murakami H, Tanaka N, Nagata K. Anteromedial meniscofemoral ligament. Arthroscopy. 2003 Jan;19(1):90-5. ↩
- Kim SJ, Min BH, Kim HK. Arthroscopic anatomy of the infrapatellar plica. Arthroscopy. 1996 Oct;12(5):561-4. ↩
- Kim S, Choe W: Pathological infrapatellar plica: A report of two cases and literature review. Arthroscopy. 1996; 12: 236-239. ↩
- Demirag B, Ozturk C, Karakayali M. Symptomatic infrapatellar plica. Knee Surg Sports Traumatol Arthrosc. 2006 Feb;14(2):156-60. ↩
- Patel SJ, Kaplan PA, Dussault RG, et al. Anatomy and clinical significance of the horizontal cleft in the infrapatellar fat pad of the knee: MR imaging. AJR Am J Roentgenol. 1998;170:1551-1555. ↩
- Zidorn T. Classification of the suprapatellar septum considering ontogenetic development. Arthroscopy. 1992;8(4):459-64. ↩
- Pipkin G. Knee injuries: the role of the suprapatellar plica and suprapatellar bursa in simulating internal derangements. Clin Orthop Relat Res. 1971 Jan;74:161-76. ↩
- Adachi N, Ochi M, Uchio Y, Kawasaki K, Yamasaki K. The complete type of suprapatellar plica in a professional baseball pitcher: consideration of a cause of anterior knee pain. Arthroscopy. 2004 Nov;20(9):987-91. ↩
- Mine T, Chagawa K, Ihara K, Kawamura H, Kuriyama R, Date R. The Role of Complete Suprapatellar Plicae. Arthrosc Tech. 2016 Feb 29;5(1):e197-9. ↩
- Trout TE, Bock H, Resnick D. Suprapatellar plicae of the knee presenting as a soft-tissue mass. Report of five patients. Clin Imaging. 1996 Jan-Mar;20(1):55-9. ↩
- Pekmezci M, Atay OA, Kerimoğlu U, Aydingöz U, Tetik O, Doral MN. A complete supra-patellar plica with an unusual presentation. Knee Surg Sports Traumatol Arthrosc. 2006 Sep;14(9):872-4. ↩
- Ehlinger M, Moser T, Adam P et al: Complete suprapatellar plica presenting like a tumor. Orthopaedics & Traumatology: Surgery & Research 95:447-250, 2009. ↩
- Gurbuz H, Calpur OU, Ozcan M, Kutoglu T, Mesut R. The synovial plicae in the knee joint. Saudi Med J. 2006 Dec;27(12):1839-42. ↩
- Kanazawa H, Maruyama Y, Takamiya S, Kaneko, K. Bilateral Symptomatic Lateral Parapatellar Synovial Plica of the Knee in an Adolescent Athlete: A Case Report. Open Journal of orthopaedics, (2017) 7, 63-71. ↩
- Schindler OS. ‘The Sneaky Plica’ revisited: morphology, pathophysiology and treatment of synovial plicae of the knee. Knee Surg Sports Traumatol Arthrosc. 2014 Feb;22(2):247-62 ↩
- Lee PYF, Nixion A, Chandratreya A, Murray JM. Synovial Plica Syndrome of the Knee: A Commonly Overlooked Cause of Anterior Knee Pain. Surg J (N Y). 2017 Feb 15;3(1):e9-e16. ↩
- Griffith CJ, LaPrade RF. Medial plica irritation: diagnosis and treatment. Curr Rev Musculoskelet Med. 2008 Mar;1(1):53-60. ↩
- Hardaker WT, Whipple TL, Bassett FH 3rd. Diagnosis and treatment of the plica syndrome of the knee. J Bone Joint Surg Am. 1980 Mar;62(2):221-5. ↩
- Al-Hadithy N, Gikas P, Mahapatra AM, Dowd G. Review article: Plica syndrome of the knee. J Orthop Surg (Hong Kong). 2011 Dec;19(3):354-8. ↩
- Flanagan JP, Trakru S, Meyer M, Mullaji AB, Krappel F. Arthroscopic excision of symptomatic medial plica. A study of 118 knees with 1-4 year follow-up. Acta Orthop Scand. 1994 Aug;65(4):408-11. ↩