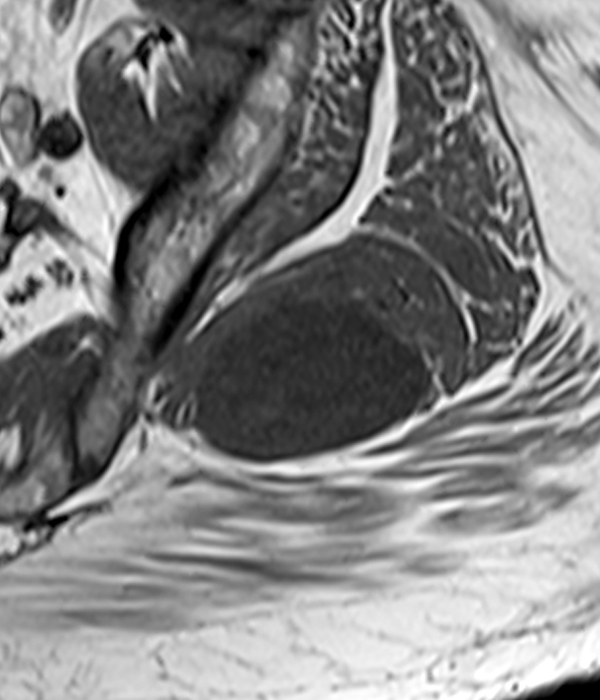
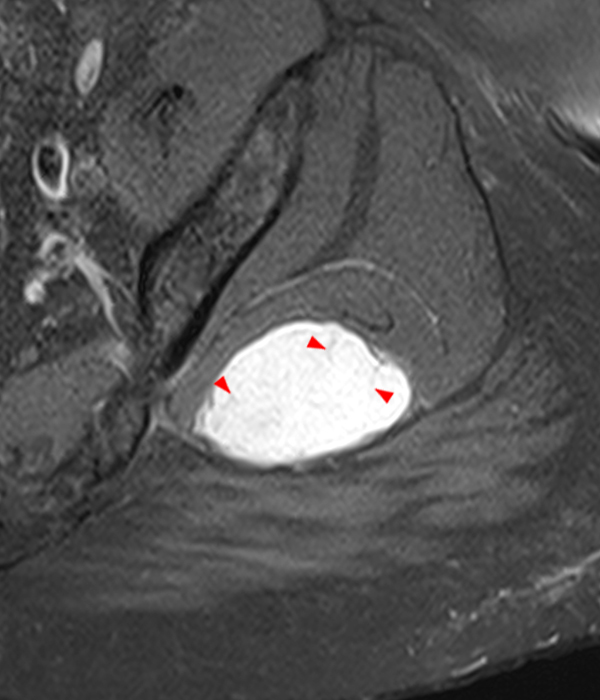
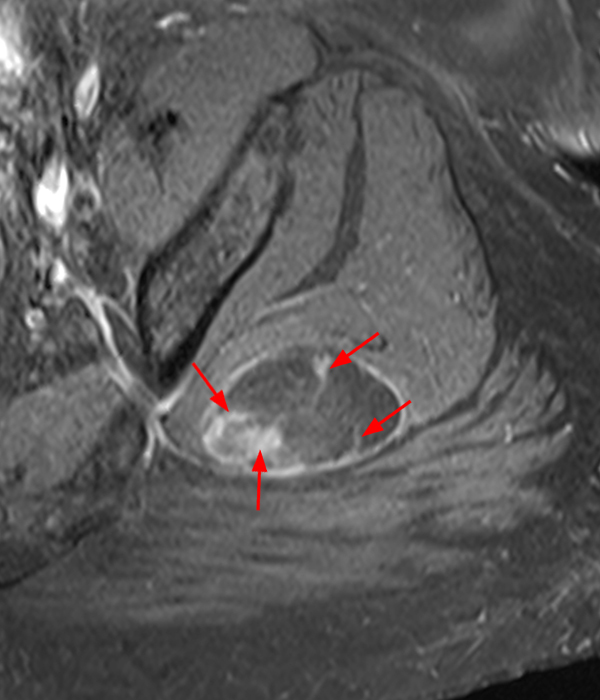
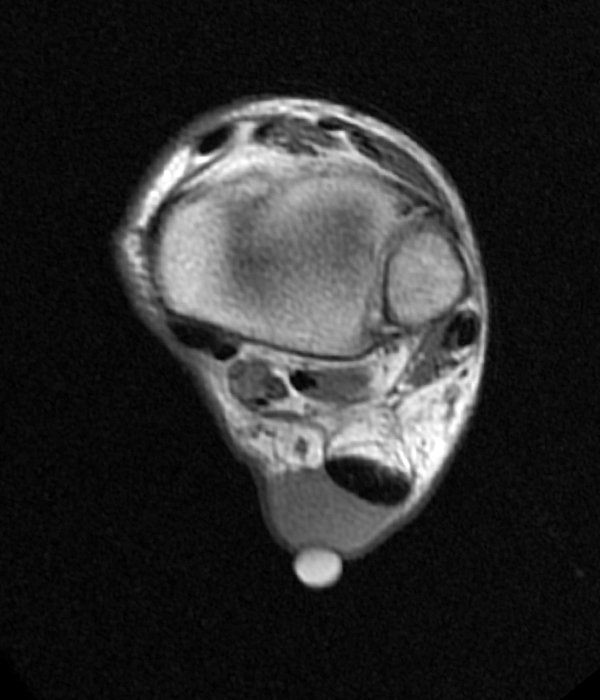
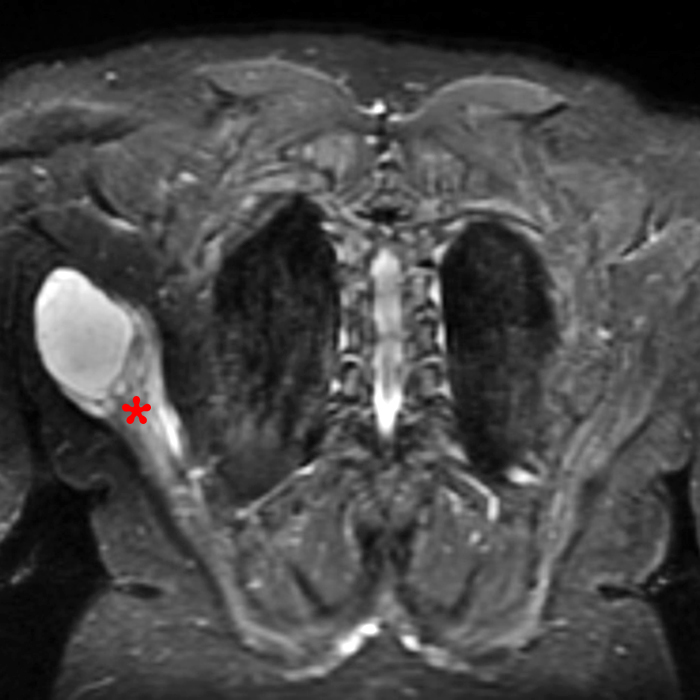
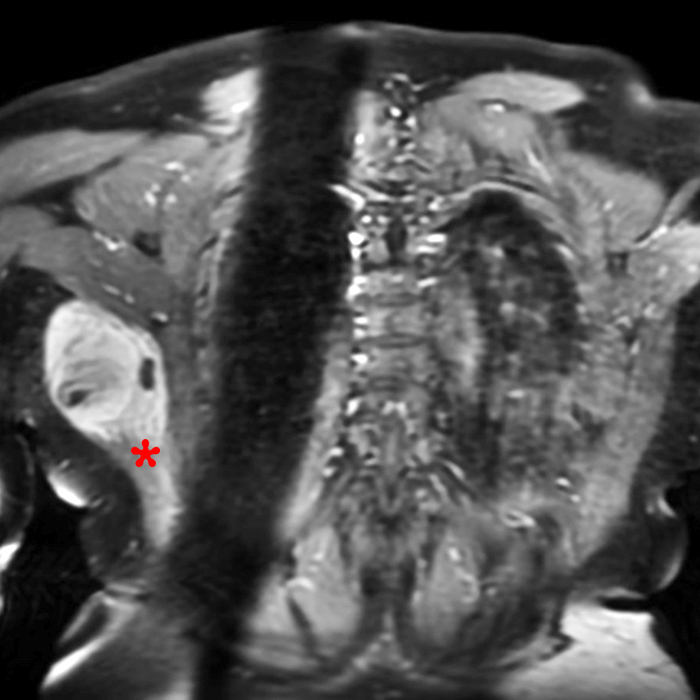
Clinical history: A 57-year-old female patient presents for MR imaging with a history of mass at the lateral aspect of her shoulder for 1.5 years, evaluate for lipoma. Coronal T1-weighted (1A), coronal fat-suppressed T2-weighted (1B), axial fat-suppressed proton density-weighted (1C) and sagittal T2-weighted (1D) images through the shoulder near the palpable abnormality are provided. What are the findings? What is the diagnosis?
Findings
Figure 2:
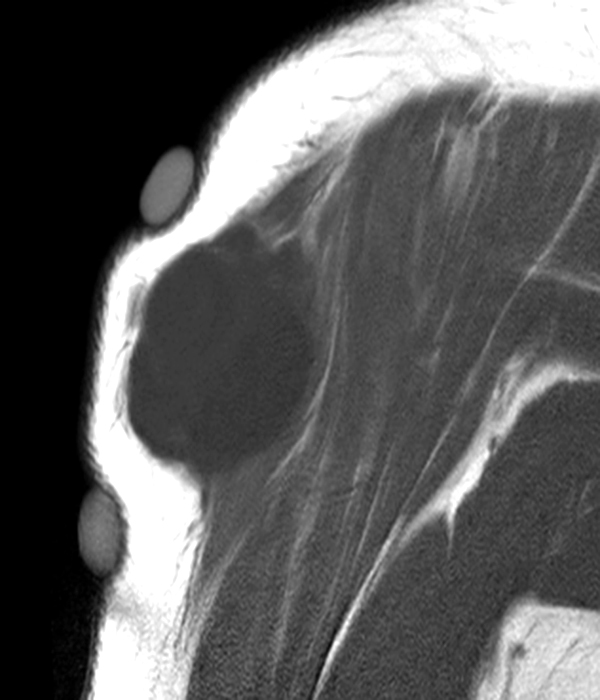
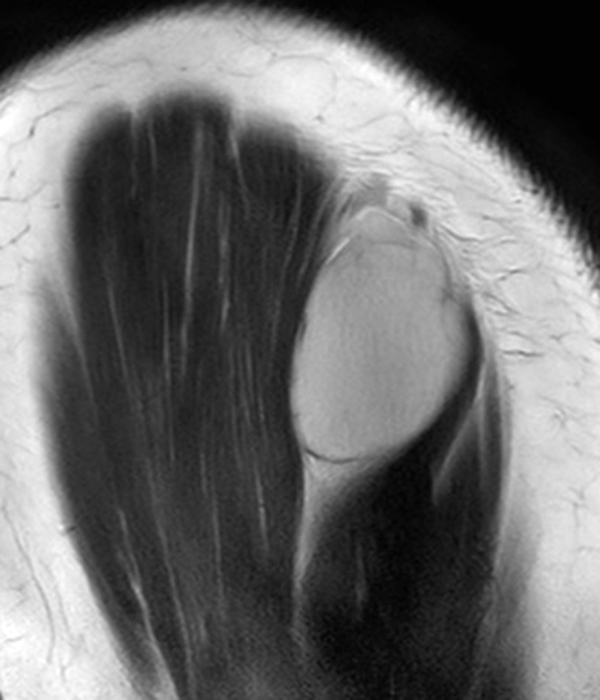
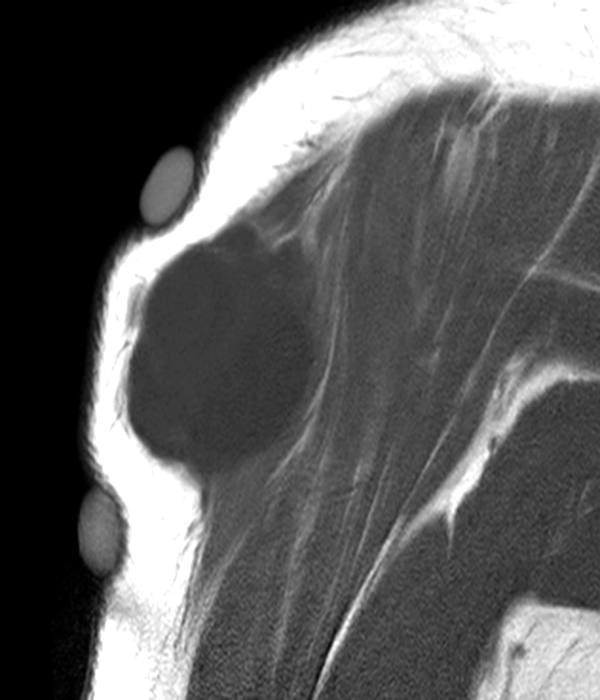
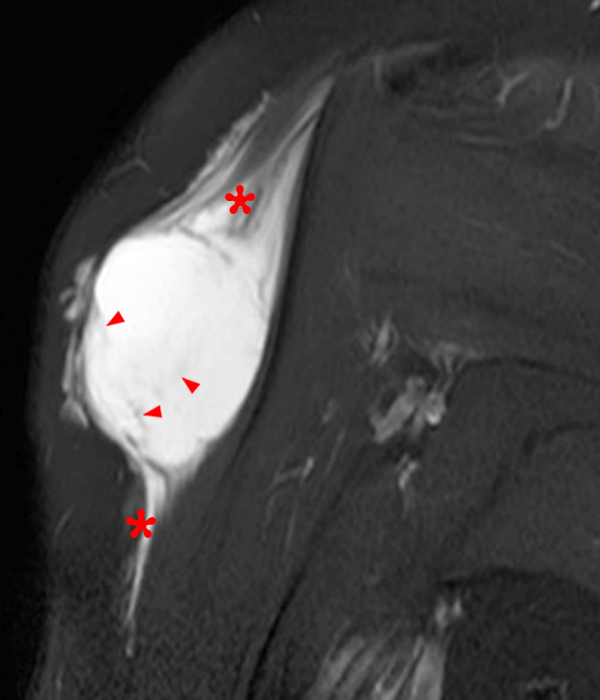
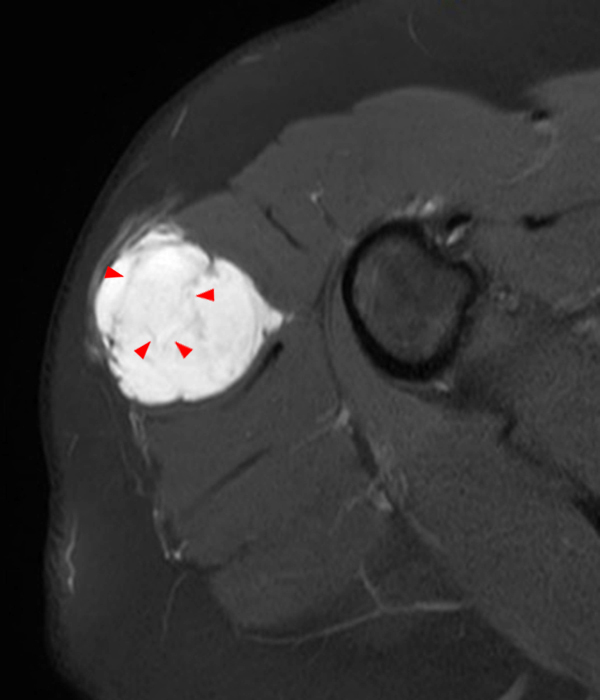
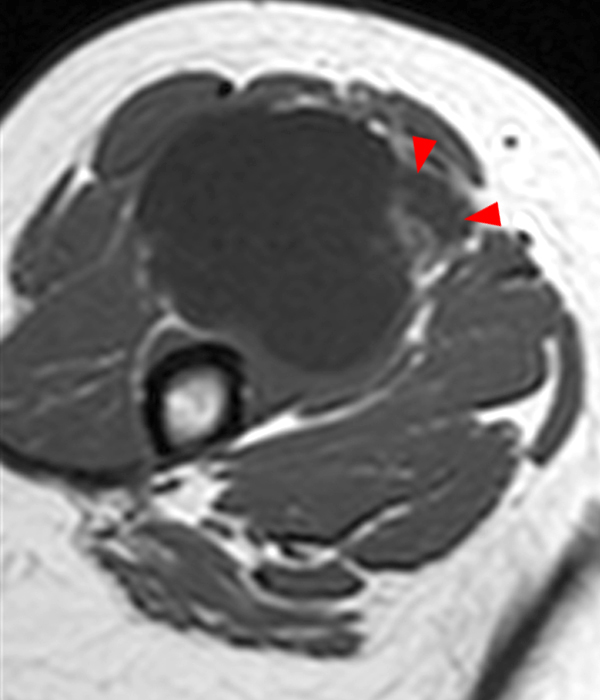
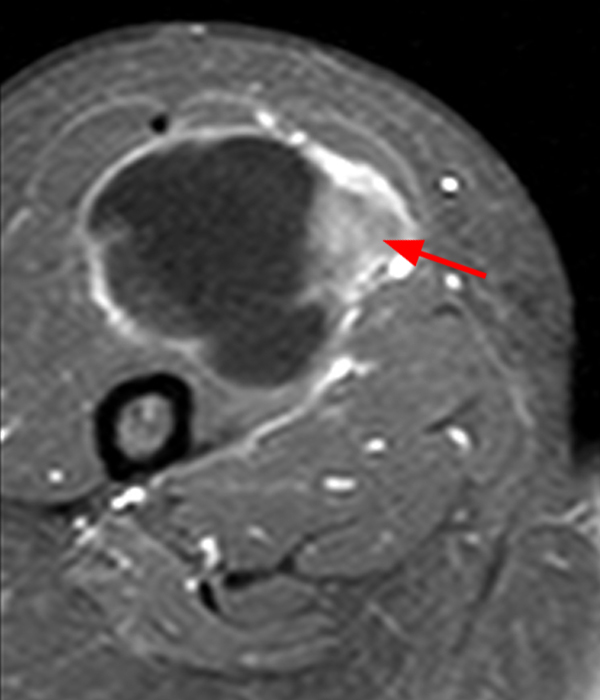
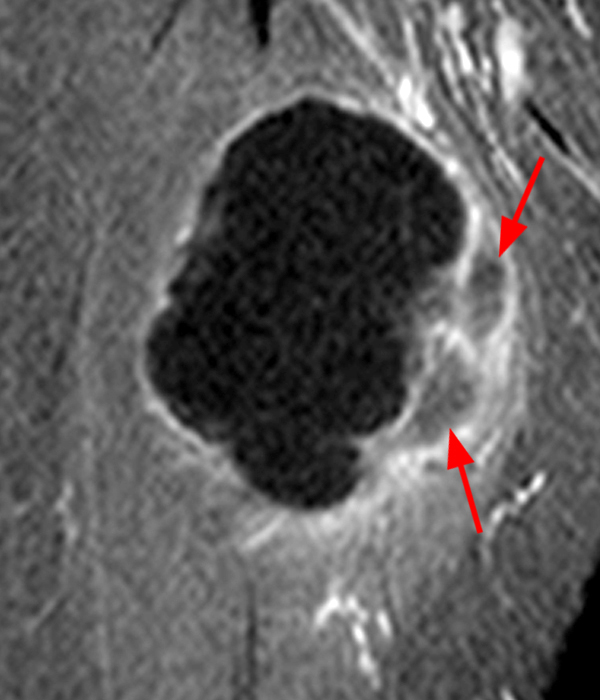
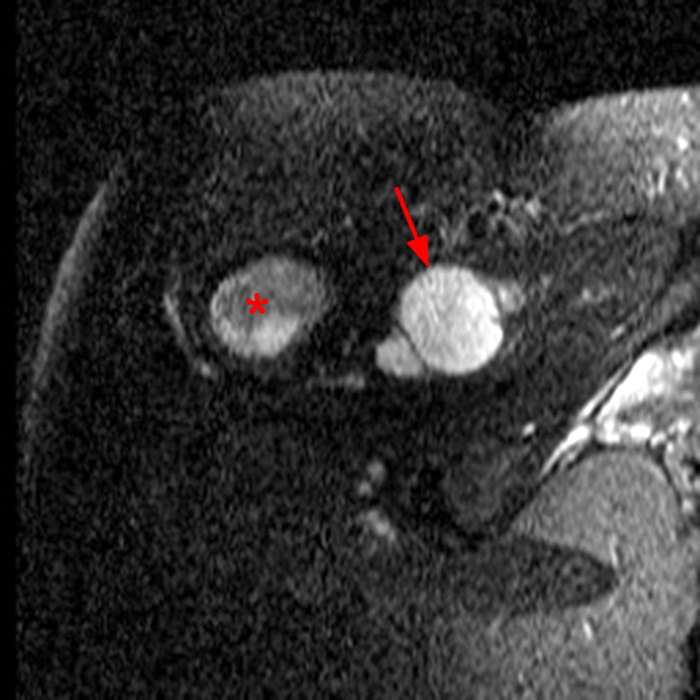
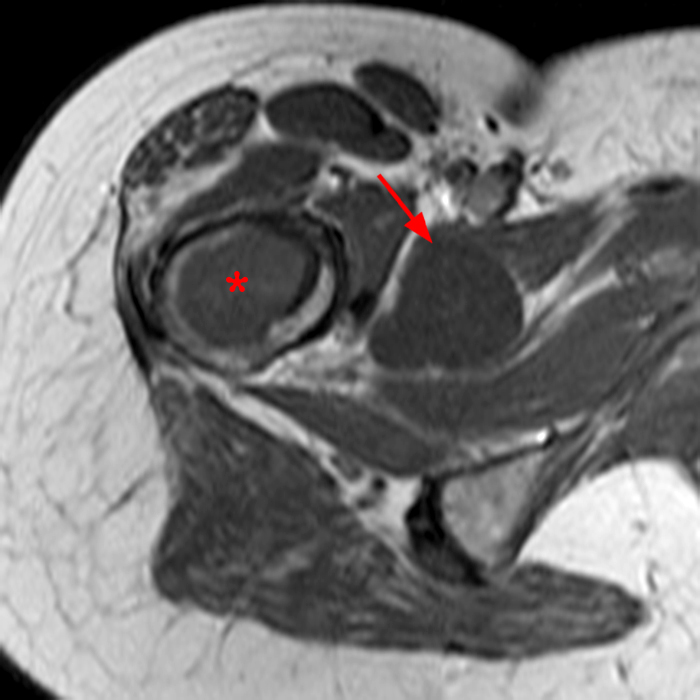
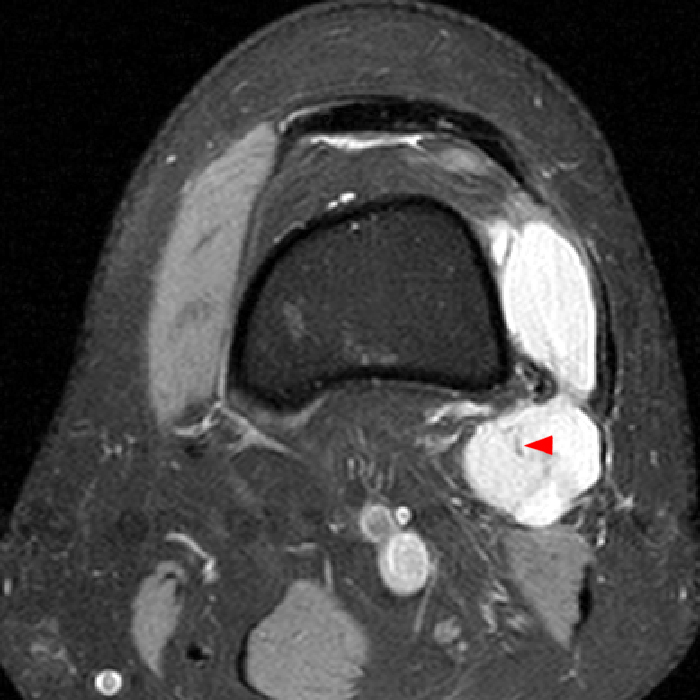
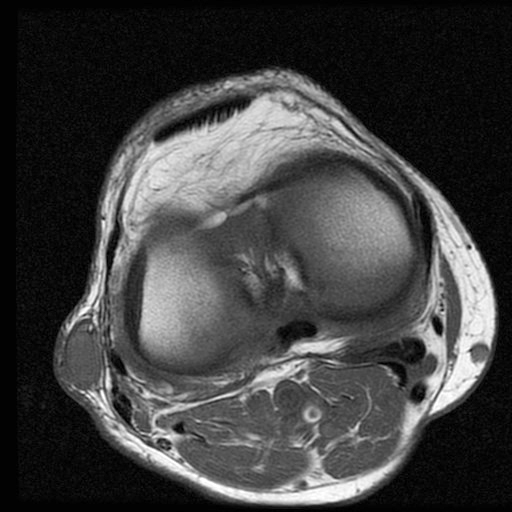
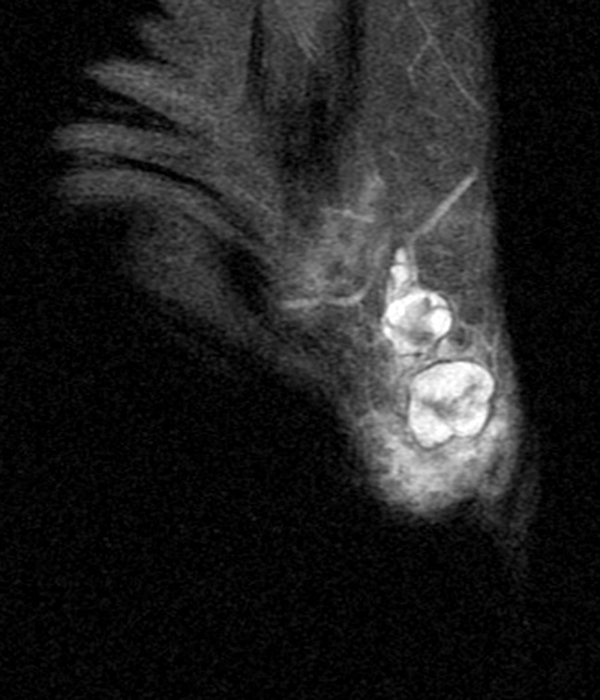
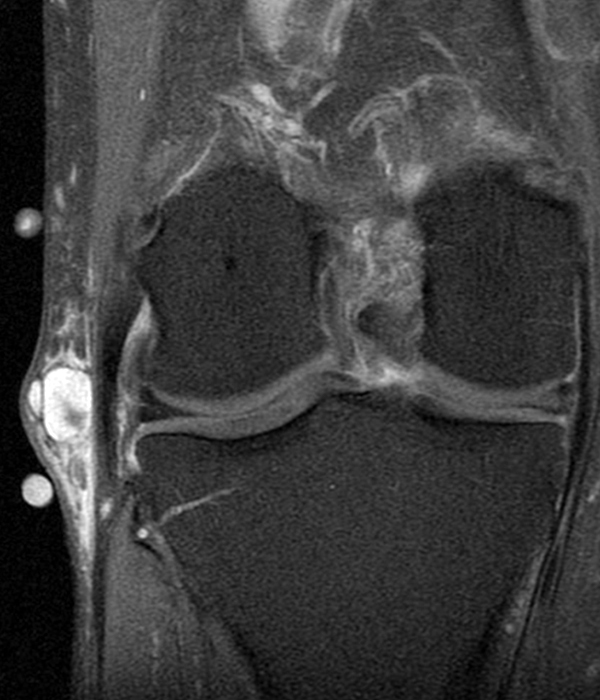
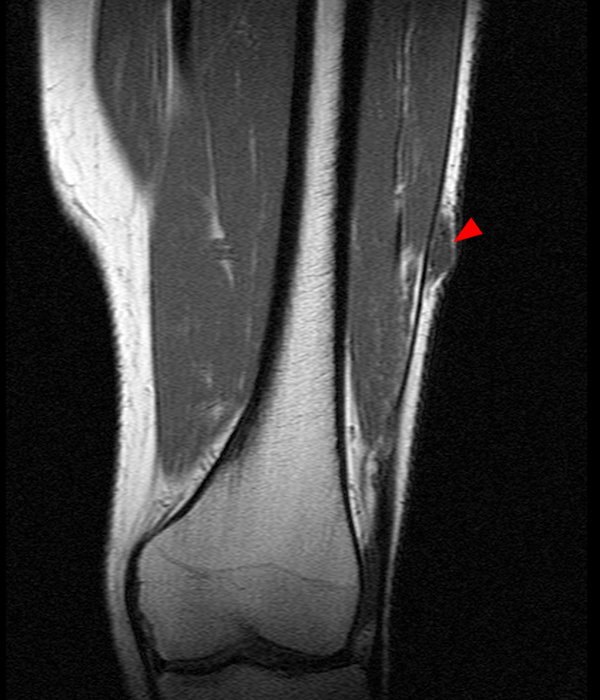
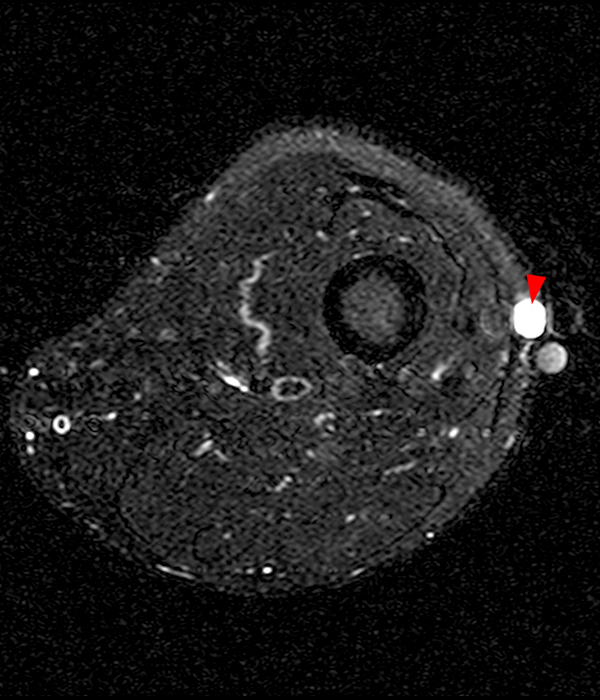
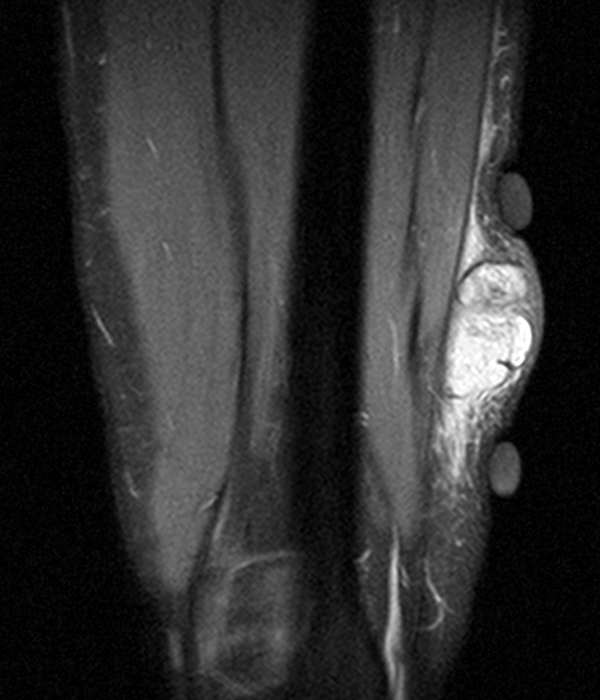
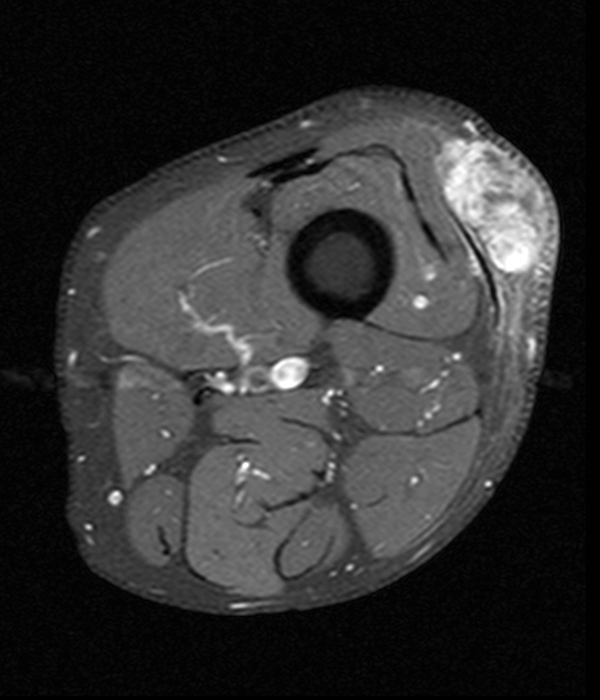
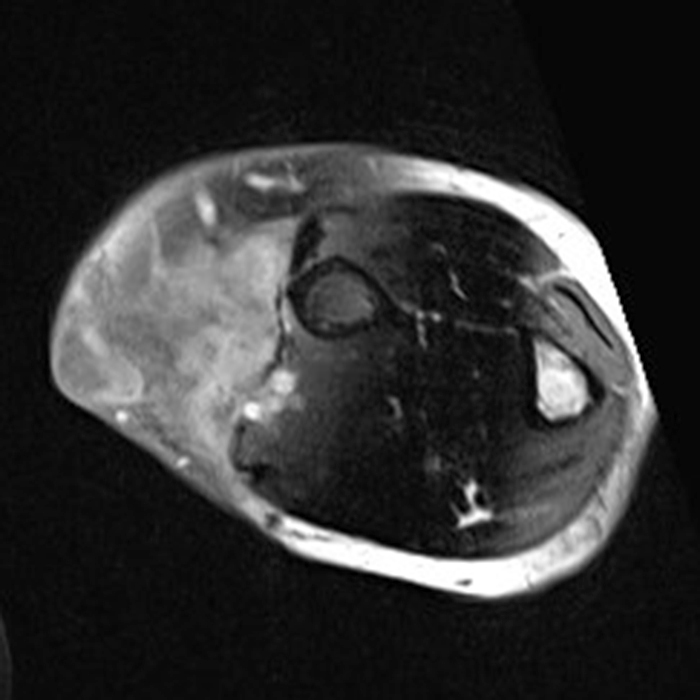
Within the superficial aspect of the deltoid muscle, there is an ovoid, well-circumscribed lesion, which is hypointense to muscle on T1-weighted images (2A) and hyperintense on fluid sensitive sequences (2B,2C,2D), measuring up to 4.2 cm, with lobulated margins, a thin peripheral capsule, and a few thin septations (arrowheads). Perilesional edema is noted within the deltoid muscle above and below the lesion on the coronal (2B) and sagittal (2D) T2-weighted images (asterisks).
Diagnosis
Benign Intramuscular Myxoma
Introduction
Myxoid soft tissue tumors are a heterogeneous group of benign and malignant lesions characterized by an abundance of extracellular mucoid (myxoid) matrix.1 Due to their high water content, myxoid lesions demonstrate high-signal-intensity on fluid-sensitive MRI sequences. Because of the homogeneous hyperintensity seen in some of these solid lesions, they may be mistaken for cysts or fluid collections.
Myxoid tumors are highly variable in their clinical behavior, ranging from completely harmless lesions, to locally aggressive lesions that recur but do not metastasize, to aggressive malignant tumors that often metastasize. There is considerable overlap clinically and radiologically between the various myxoid lesions, generating potential diagnostic problems for the clinician, radiologist, and pathologist.1
In this web clinic, we will discuss intramuscular myxomas, as well as a variety of other benign and malignant myxoid lesions. While many types of soft tissue tumors may demonstrate a degree of focal myxoid change, such as colorectal cancer, gastrointestinal stromal tumors, peripheral nerve sheath tumors, and leiomyosarcomas, we will focus this discussion on tumors in which myxoid tissue is an integral component of the lesion (Table 1).

Benign Myxoid Lesions
Intramuscular Myxoma:
Intramuscular myxoma is a benign neoplasm that may arise from fibroblasts which produce an excessive amount of mucopolysaccharide.2 Myxomas are most frequently diagnosed in patients 40–70 years of age 3, are slightly more common in females, and typically present as a slowly growing painless mass, although they can occasionally be painful.
Myxomas most frequently occur within the large muscles of the thigh (Figure 3 and Figure 4), gluteal region (Figure 5) and upper arm (Figure 2). Myxomas are generally well defined, with an average size of 6-7 cm4, with the signal intensity of fluid on MRI including low signal on T1-weighted images and markedly high signal intensity on T2-weighted images. A thin rim of fat may be seen at the superior and inferior poles of the lesion, referred to as a peritumoral fat rind or “fat cap” (Figure 4b). Non-enhancing perilesional edema is often seen on fluid sensitive sequences (Figure 2b). The non-cystic areas of the lesion usually demonstrate mild to moderate heterogeneous enhancement (Figures 3 and 5).
The differential diagnosis for intramuscular myxoma primarily includes ganglion cyst, benign peripheral nerve sheath tumor, or other myxoid neoplasms. Grossly the tumors are globular and gelatinous (Figure 6). Treatment is surgical excision with wide margins, with recurrence being rare.5 Myxomas do not metastasize.
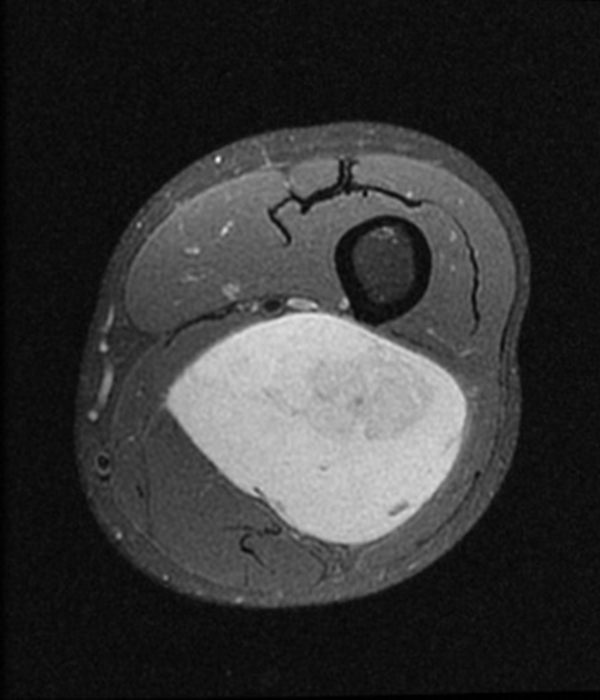
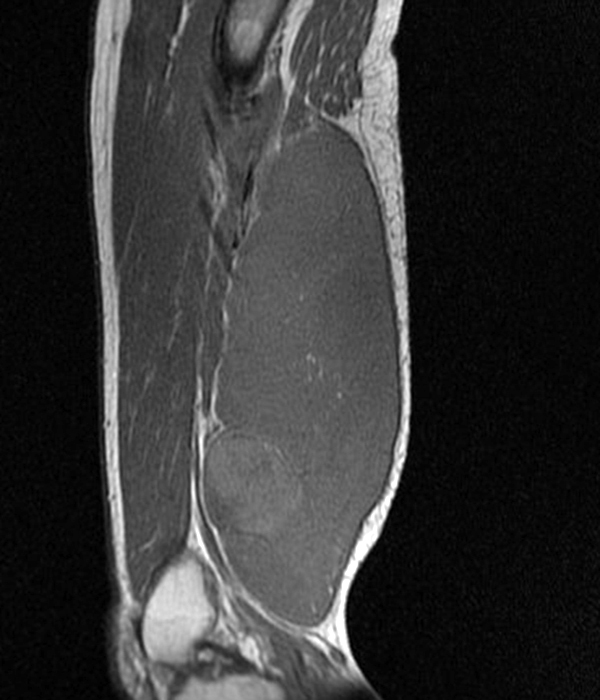
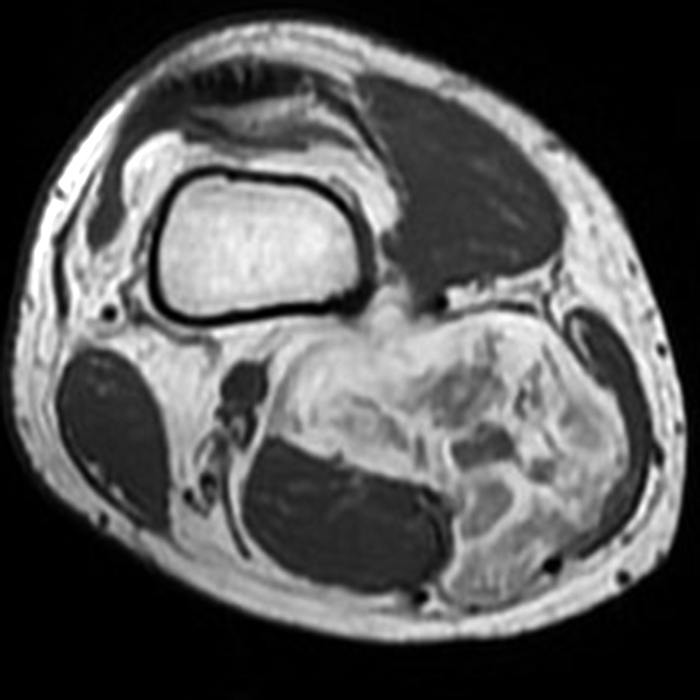
Figure 3:
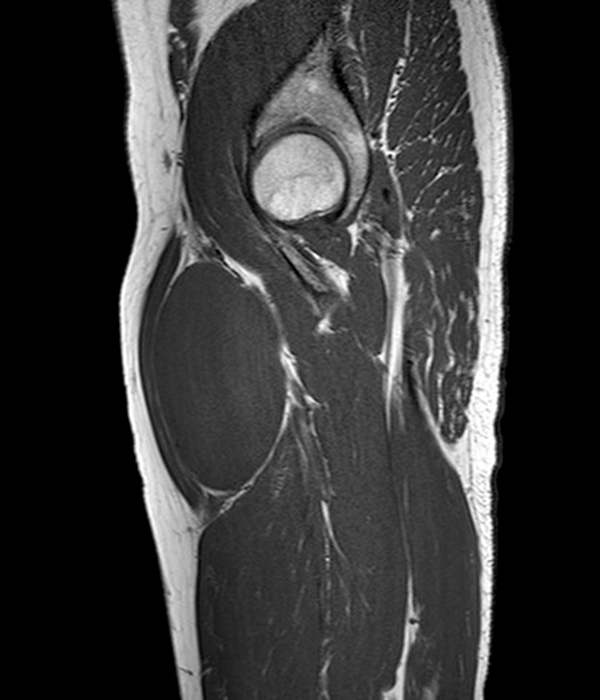
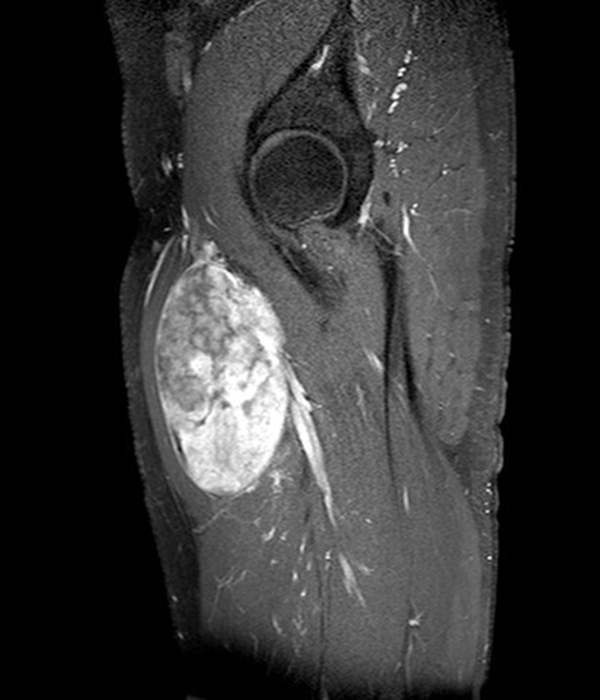
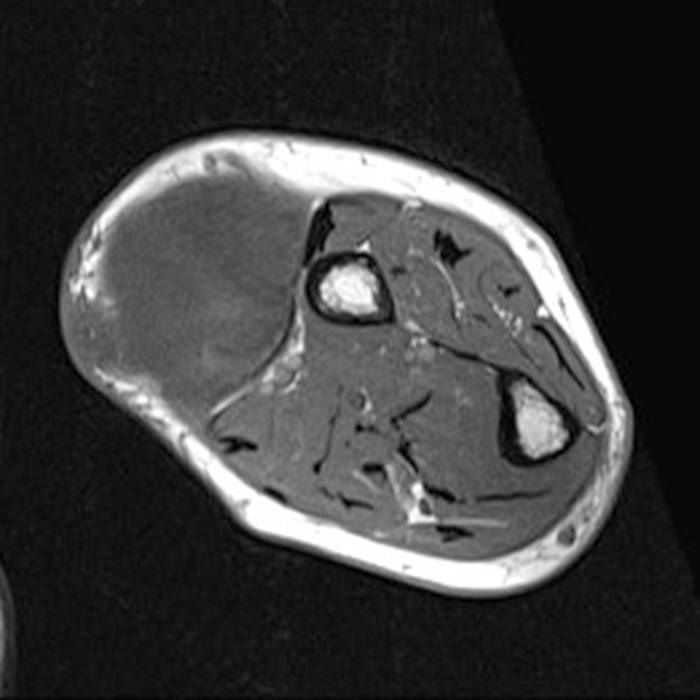
49-year-old female with an intramuscular myxoma in the right anterior thigh. Axial T1-weighted (3A) and fat-suppressed T2-weighted (3B) images show a cyst-like lesion with T1 hypointensity and T2 hyperintensity, with lobular and slightly nodular appearance anteromedially (arrowheads). Following IV gadolinium-containing contrast administration, fat-suppressed T1-weighted axial (3C) and coronal (3D) images show a predominately non-enhancing lesion, with mild peripheral enhancement of the non-cystic area medially (arrows).
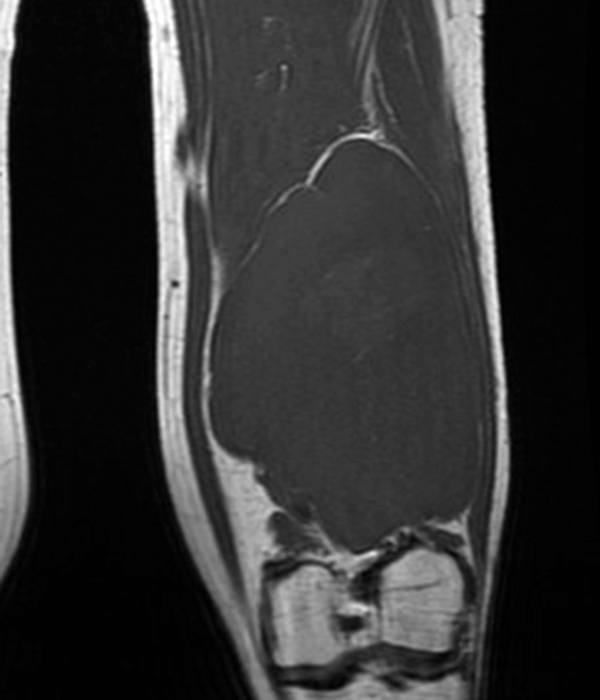
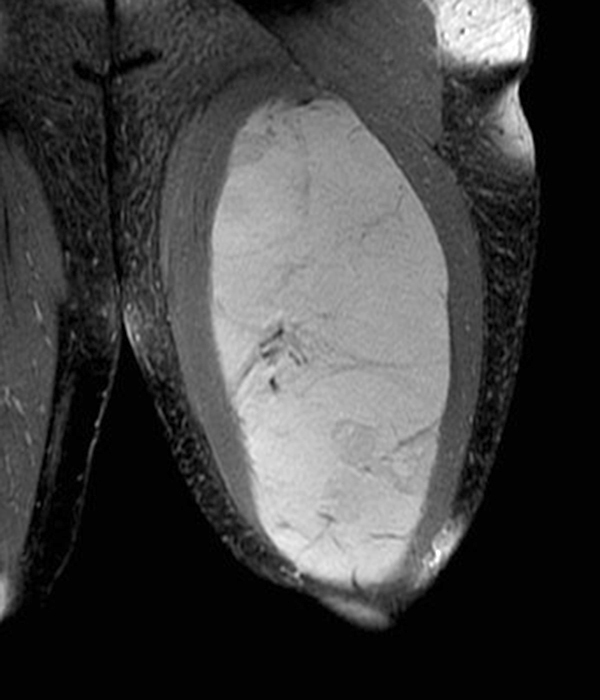
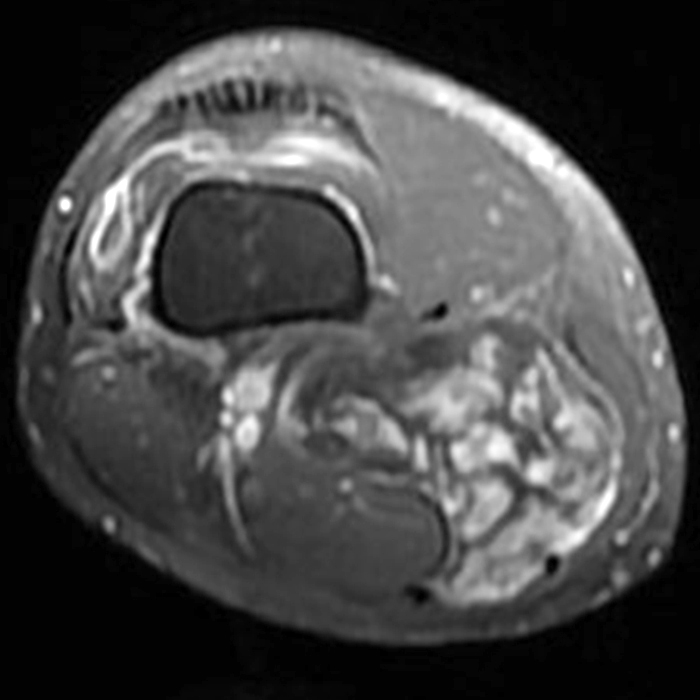
Figure 4:
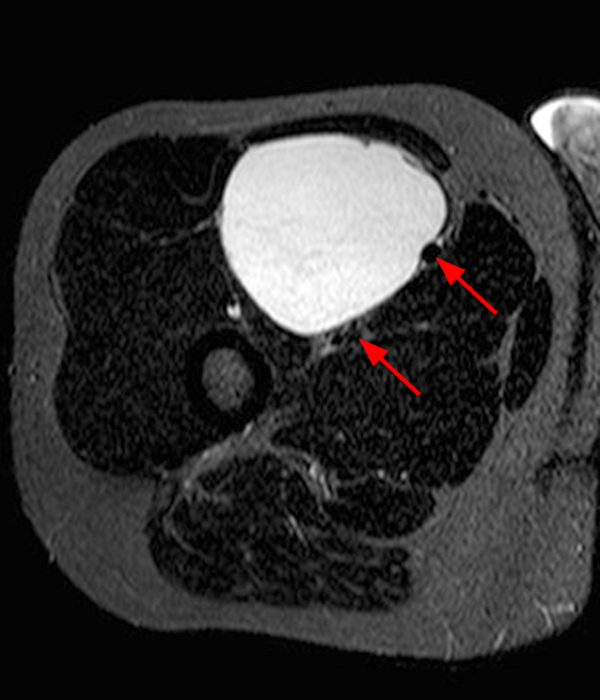
43-year-old female with an intramuscular myxoma at the left anterior thigh. Several thin septae (arrowheads) and mild perilesional edema (asterisks) are noted on the coronal fat-suppressed T2-weighted image (4A), and a thin peritumoral fat rind (arrows) is present on the coronal T1-weighted image (4B).
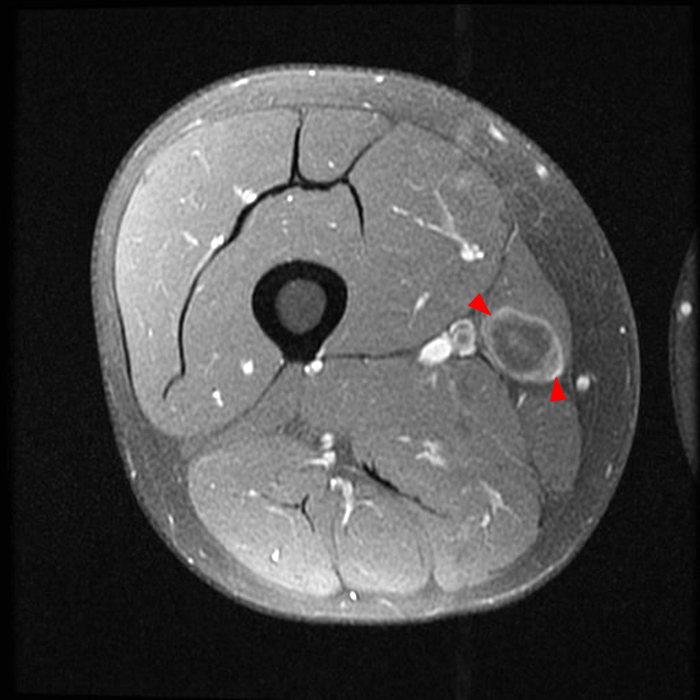
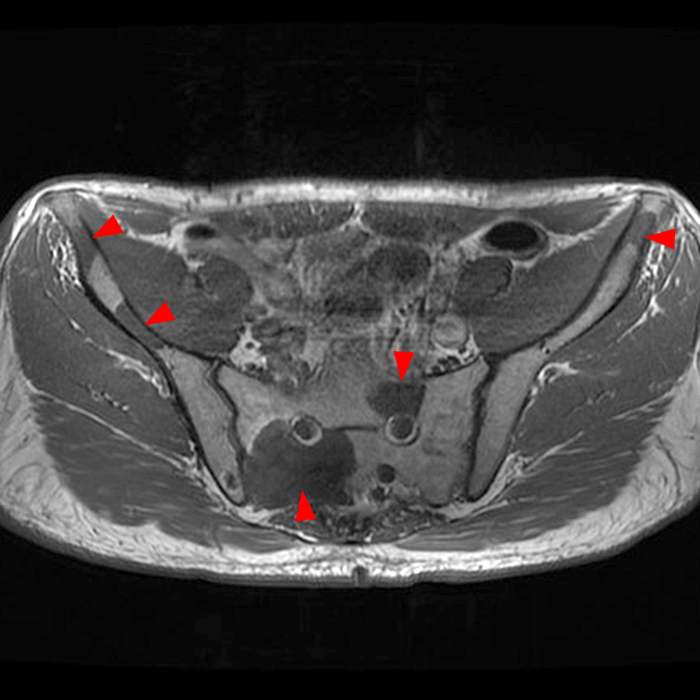
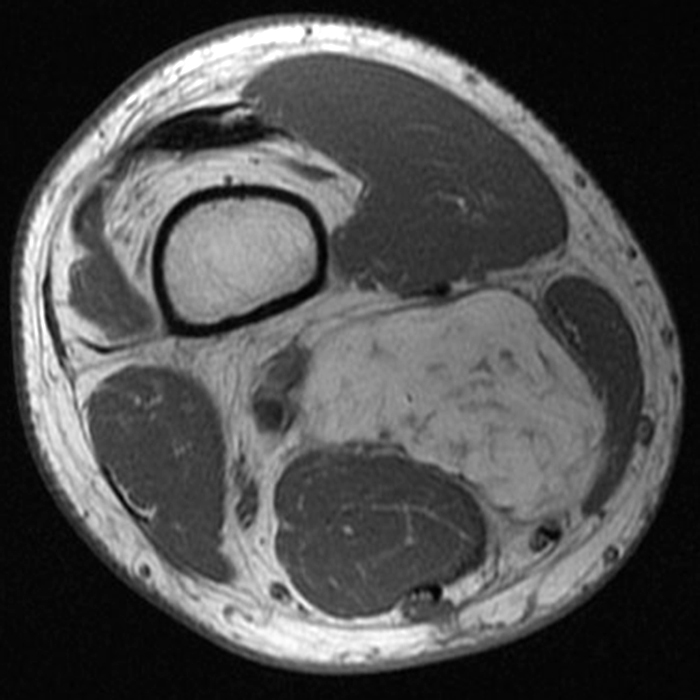
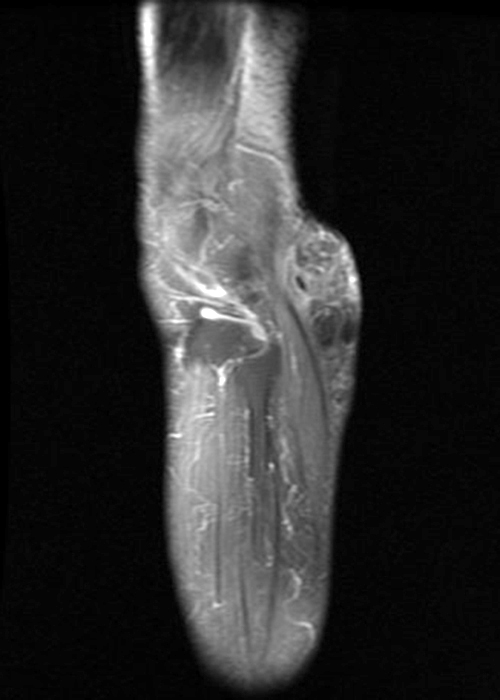
Figure 5:
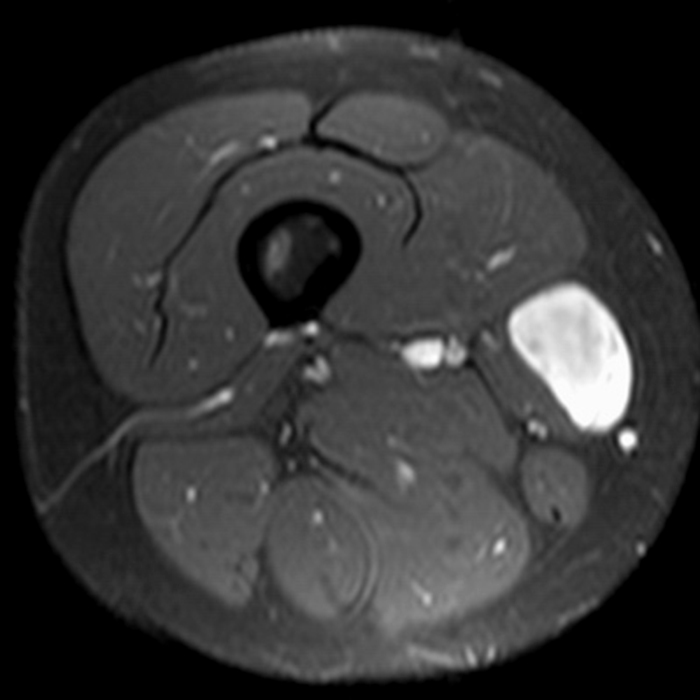
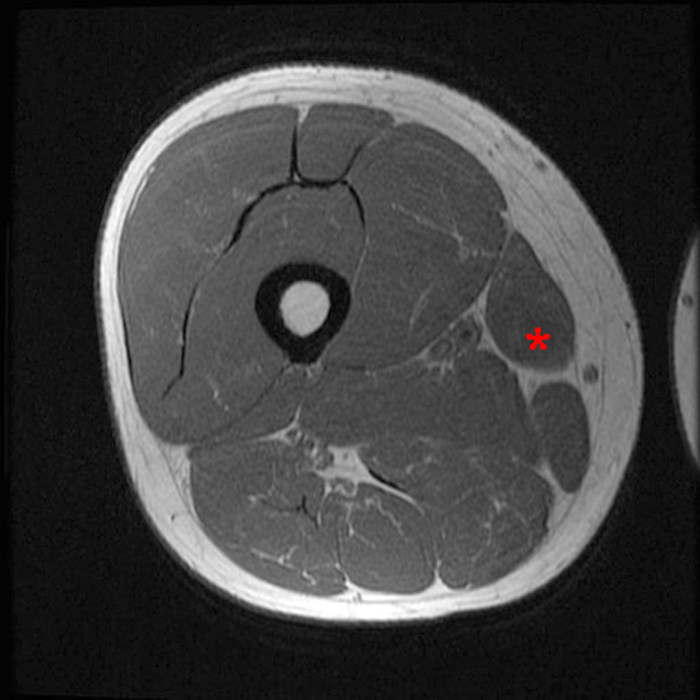
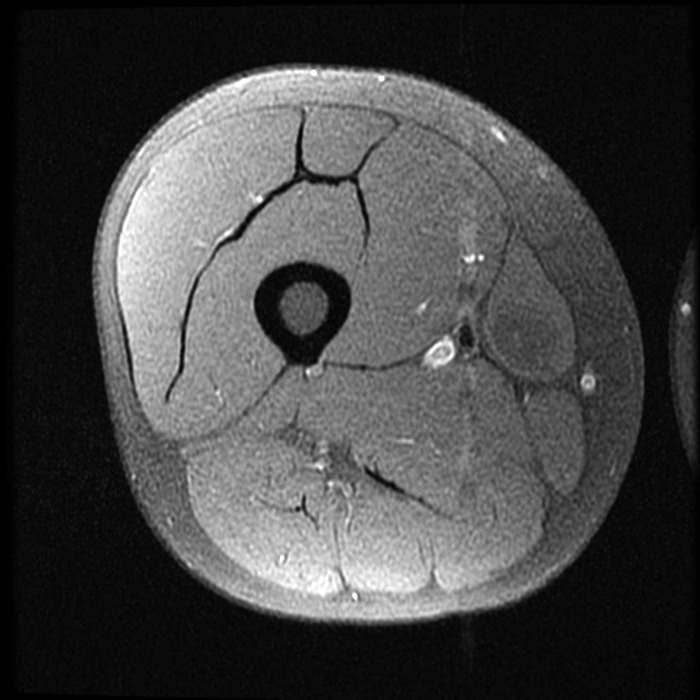
68-year-old male with an asymptomatic intramuscular myxoma within the left gluteus medius muscle, initially an incidental finding on MRI of the lumbar spine. Axial T1-weighted (5A), fat-suppressed proton density-weighted (5B), and post-contrast fat-suppressed T1-weighted (5C) images demonstrate T1 hypointensity and T2 hyperintensity with a few thin incomplete septae (arrowheads), and mild nodular and septal enhancement (arrows).
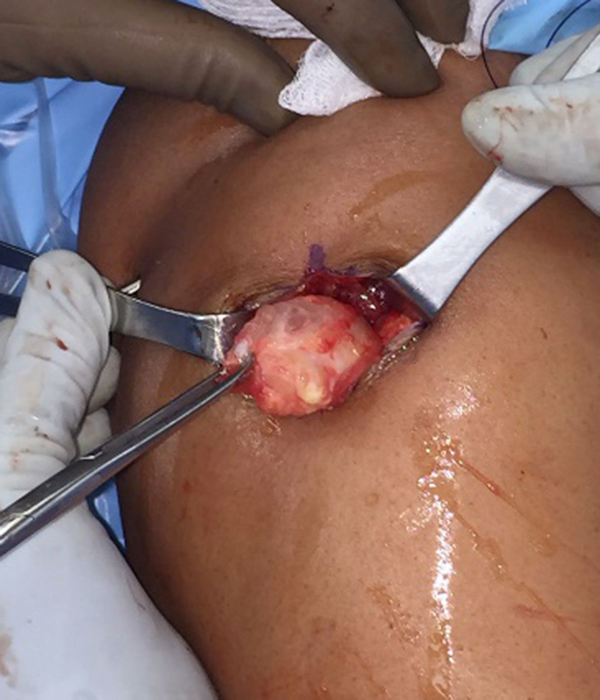
Figure 6:
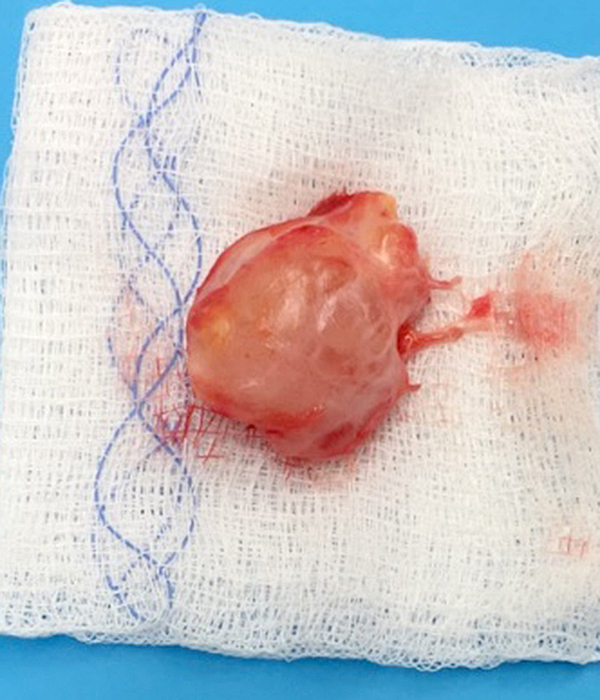
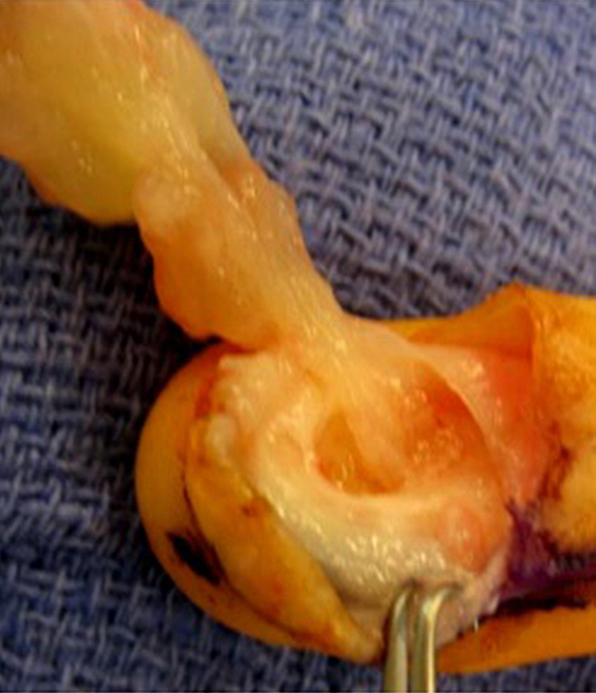
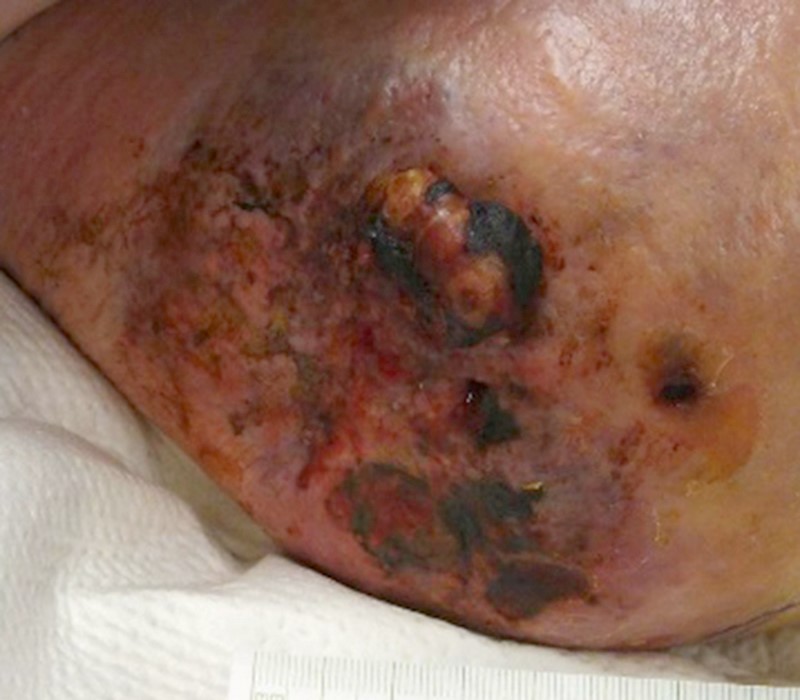
Intraoperative photos from the initial test case in Figure 1 and 2, showing an intramuscular myxoma. Pathology report: Right deltoid mass measuring 4.2 x 3.5 x 2.0 cm with clear, mucoid cut surfaces. Well-circumscribed myxoid lesion with surrounding skeletal muscle, consistent with intramuscular myxoma. No malignancy identified. Photos courtesy of Arsen Manugian, MD, OrthoSouth, Memphis, TN.
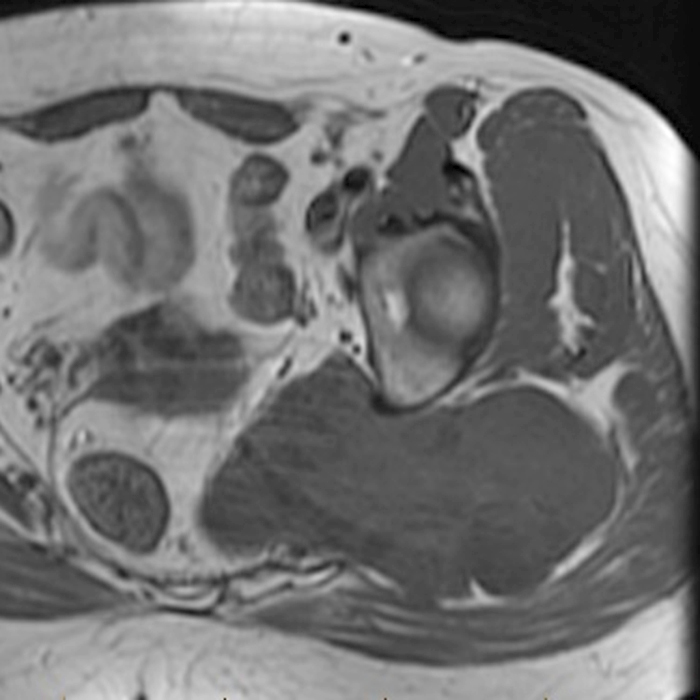
Mazabraud Syndrome:
Intramuscular myxomas are typically solitary. When multiple myxomas are present, they are almost always associated with Mazabraud syndrome, a syndrome characterized by the presence of skeletal fibrous dysplasia (FD) and associated intramuscular myxomas.6 Mazabraud syndrome is extremely rare with an estimated prevalence of 2.2 – 2.4% among patients with FD.7,8
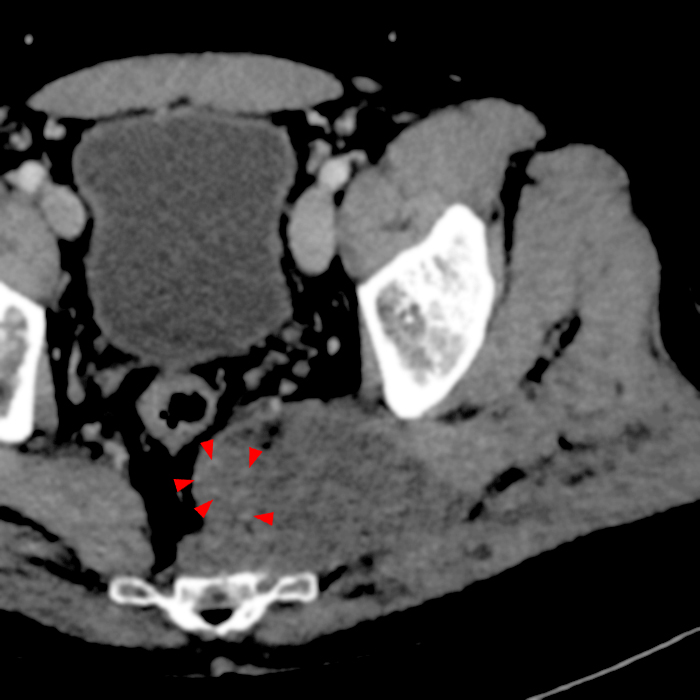
The most common localization for myxomas in patients with Mazabraud syndrome is the upper leg, including within the quadriceps muscles, the hip adductors, and the gluteal muscles. In nearly all patients, the myxomas are located near an adjacent osseous FD lesion, with the proximal femur the most common site of FD.8 (Figure 7)
Figure 7:
Mazabraud syndrome. An AP radiograph of the right femur (7a) demonstrates an expansile lucent lesion consistent with fibrous dysplasia (asterisks). STIR coronal (7B), fat-suppressed T2-weighted axial, (7C), and T1-weighted axial (D) MR images show multiple intramuscular myxomas (arrows) within the medial right thigh adjacent to the osseous fibrous dysplasia (asterisks). Images courtesy of Tudor Hughes, MD, UCSD Department of Radiology.
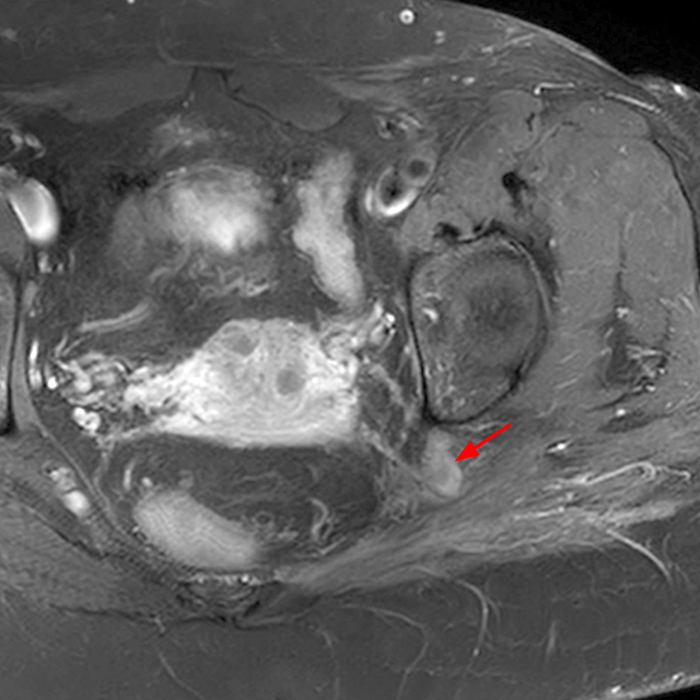
Juxta-articular Myxoma:
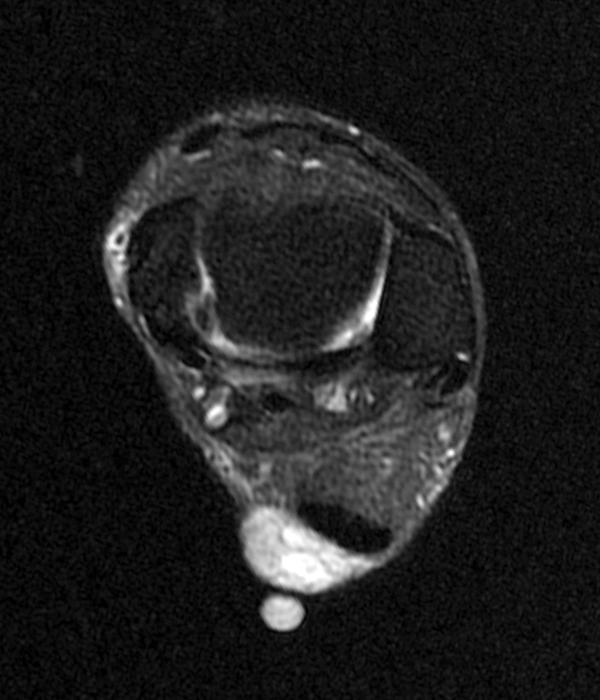
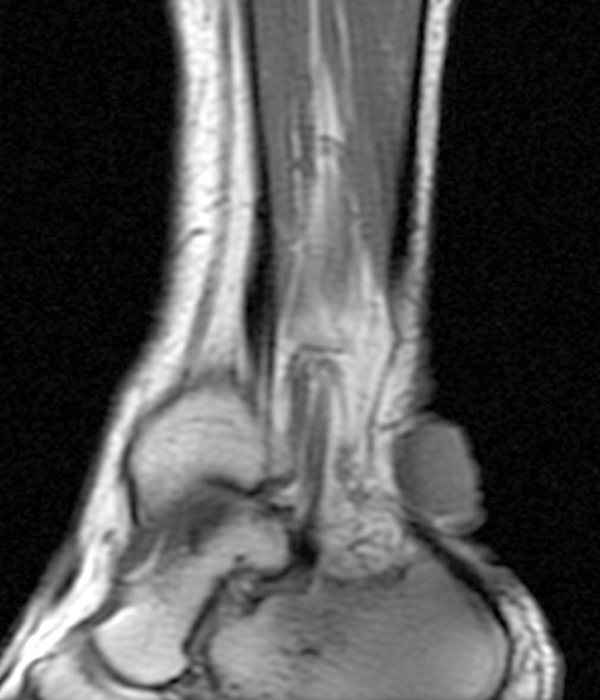
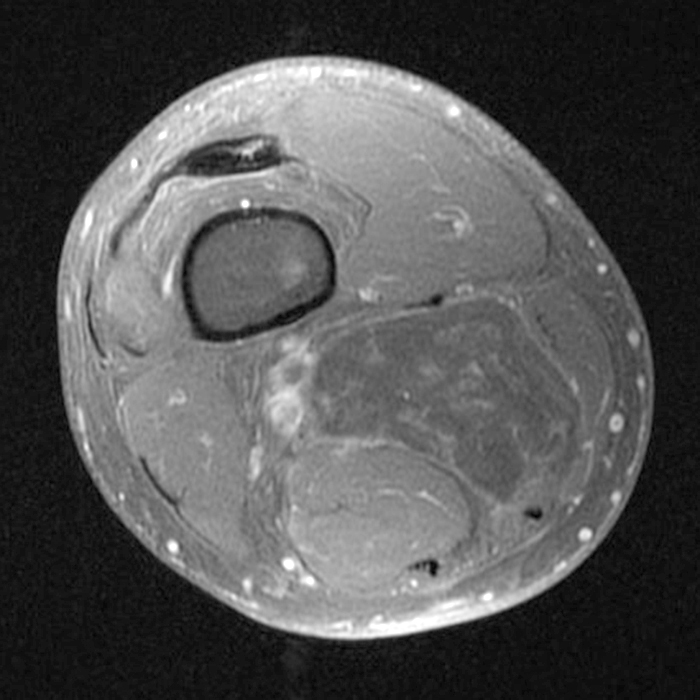
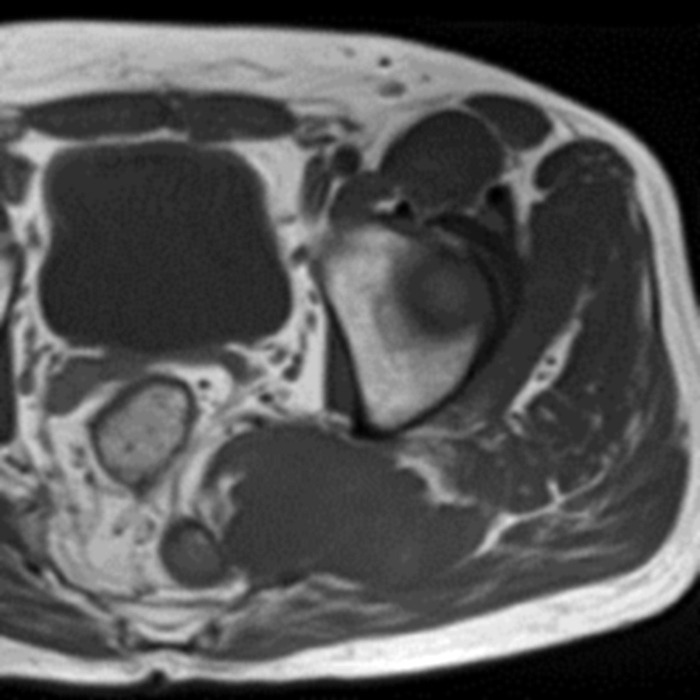
Approximately 7% of myxomas may be of the juxta-articular variety 2, which tend to be more cellular than typical myxomas. Juxta-articular myxoma represents a soft tissue myxoid lesion occurring in reasonably close approximation to major joints of the extremities, especially around the knee, and to a lesser degree near the shoulder, elbow, ankle (Figure 8), and hip. The lesions are usually less than 5 cm in diameter.1
Microscopically, the lesions resemble intramuscular myxomas but are often more cellular and vascular. Areas of hemorrhage, hemosiderin deposition, and a mild chronic inflammatory cell infiltrate may be seen. These lesions have a higher local recurrence rate than intramuscular myxomas, although this may in part be due to confusion with low-grade myxofibrosarcomas in some cases.1
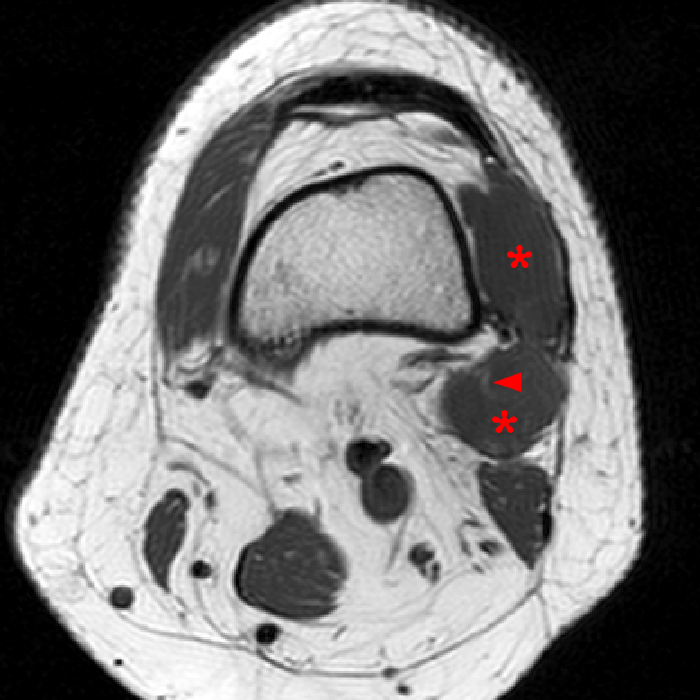
Figure 8:
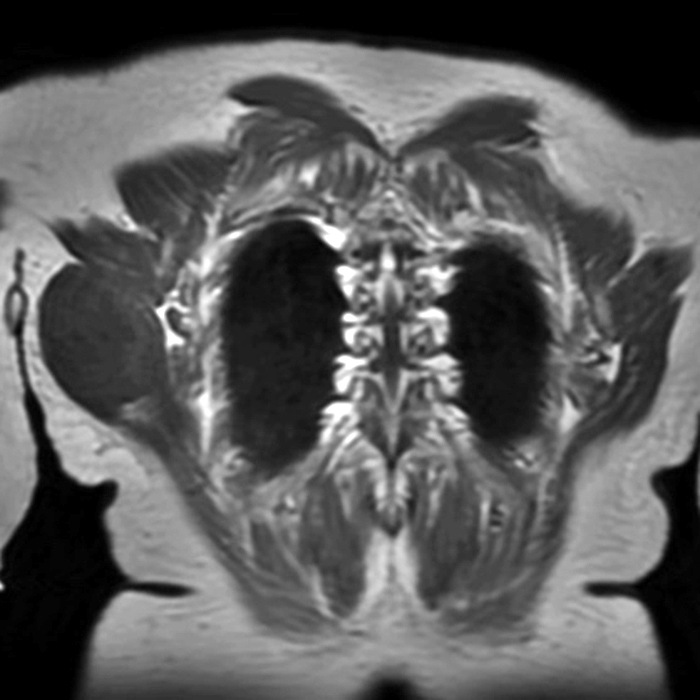
68-year-old male with pathologically proven juxta-articular myxoma with axial T1-weighted (8A), axial fat-suppressed T2-weighted (8B), sagittal T1-weighted (8C), and sagittal fat-suppressed T2-weighted (8D) images. While the vast majority of myxomas are intramuscular, a small percentage may be near a joint and/or in the subcutaneous tissues. This juxta-articular myxoma is subcutaneous, and more isointense to muscle on T1-weighted images and slightly more heterogeneous on fat-suppressed T2-weighted images than the typical intramuscular myxoma, as they are often more cellular and vascular.
Superficial Angiomyxoma:
Superficial angiomyxoma is a rare, benign myxoid soft tissue tumor which involves the dermis and subcutaneous tissues, also referred to in early literature as cutaneous myxoma. Clinically, the lesions present primarily in adults, occurring most often in the head, neck, trunk, and less frequently on the limbs.9 These lesions are usually nontender, slowly growing cutaneous nodules with an average diameter of 3-4cm.1 The tumor contains myxoid matrix with multiple blood vessels. Given the superficial nature of the lesion near the skin, imaging plays a relatively minor role in the evaluation of this lesion compared with other myxoid tumors.
The lesion is usually solitary, but when multiple is typically associated with the Carney complex, an autosomal dominant syndrome that includes the existence of cardiac, cutaneous, mammary myxoid fibroadenomas, skin hyperpigmentation and endocrine hyperactivity (Cushing’s syndrome, early sexuality, acromegaly).9
The treatment is total excision. Superficial angiomyxomas are benign, although local recurrence following surgical excision may occur if the resection is incomplete.
Nerve Sheath Myxoma:
Nerve sheath myxoma is a rare, benign, soft-tissue tumor that most often presents as a soft nodule in the extremities10, especially the fingers (Figure 9), although it also may occur in the head and neck. Also referred to as dermal nerve sheath myxoma and perineural myxoma, nerve sheath myxoma is an unusual, primarily cutaneous neoplasm of presumed nerve sheath origin.1 It occurs mainly in young and middle-aged adults, presenting as painless, slow-growing, solitary, superficial, multinodular masses from 0.5 to 2.0 cm, without any relationship to neurofibromatosis I. A benign myxoid tumor derived from Schwann cells (peripheral nerve sheath), this is probably a myxoid variant of schwannoma. Imaging characteristics include diffuse T2 hyperintensity and multi-nodularity. Immunohistochemically the cellular component is strongly S100 protein positive, which is critical in differentiating these lesions from other myxoid or nerve sheath tumors.11 Wide local excision with tumor free surgical margin is the treatment of choice for nerve sheath myxoma. Recurrence following complete resection is very uncommon.1
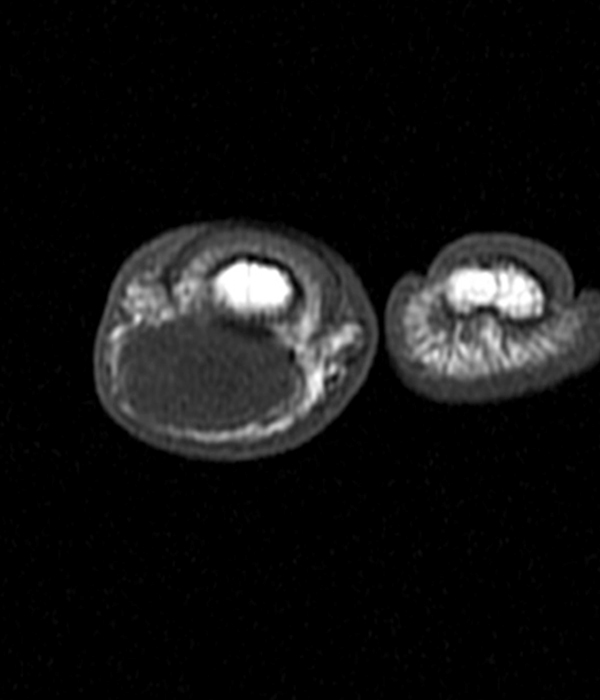
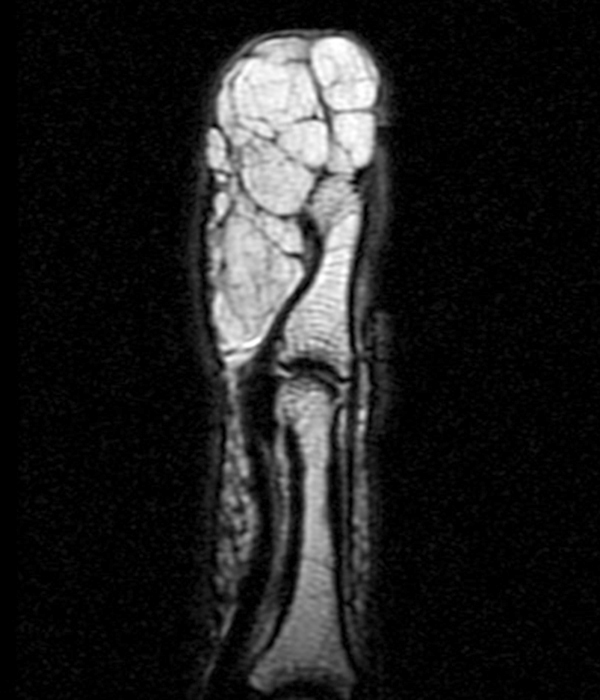
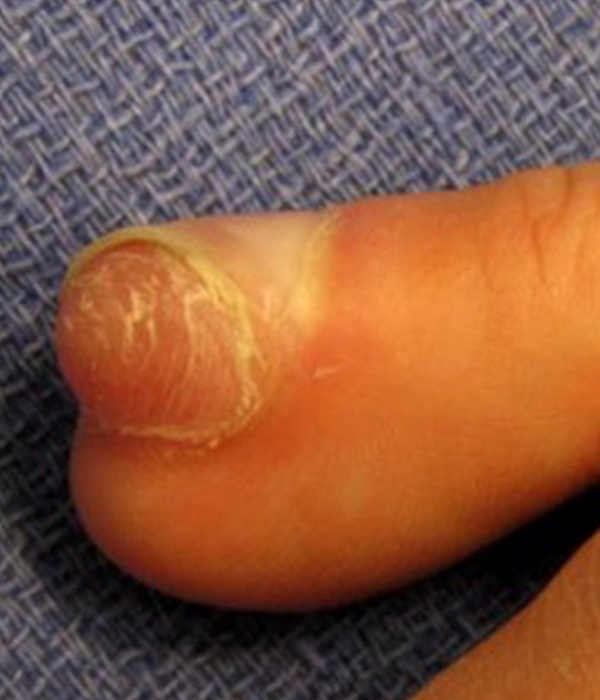
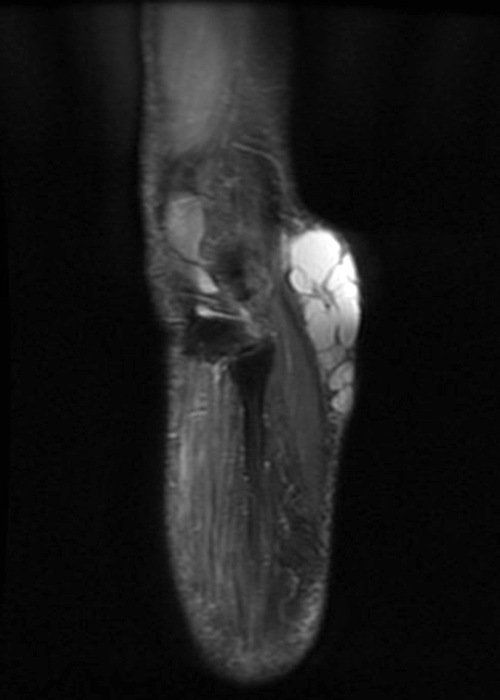
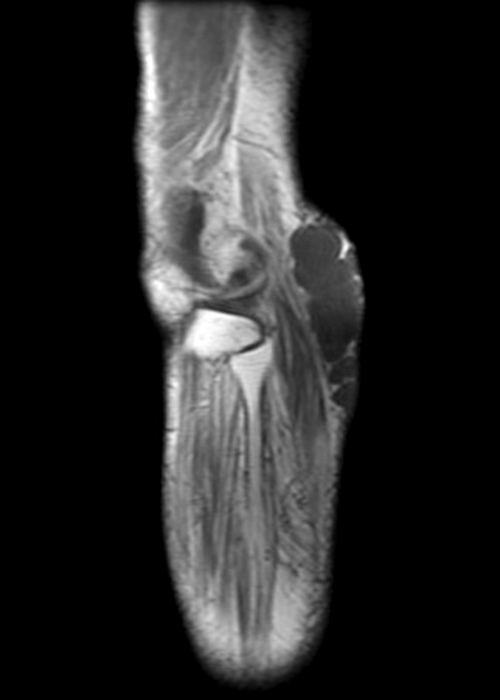
Superficial Acral Fibromyxoma:
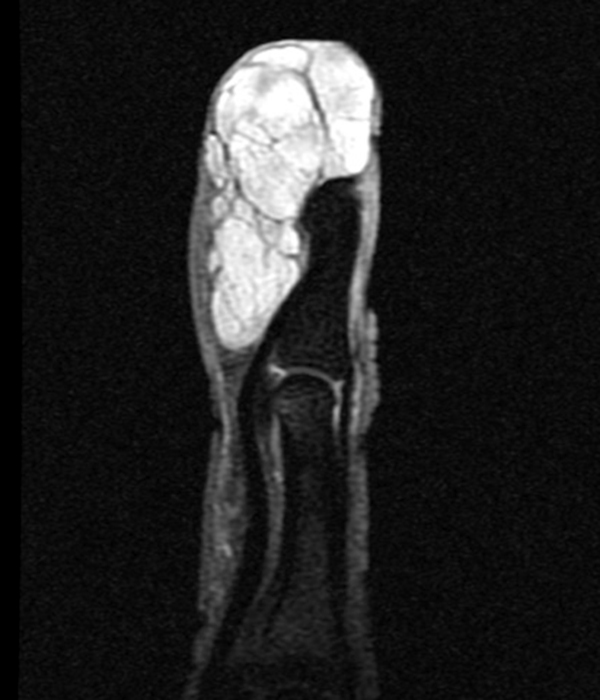
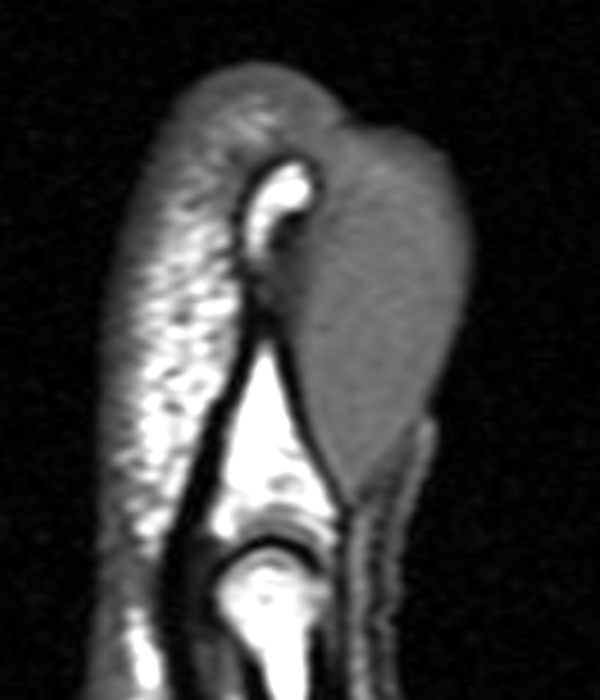
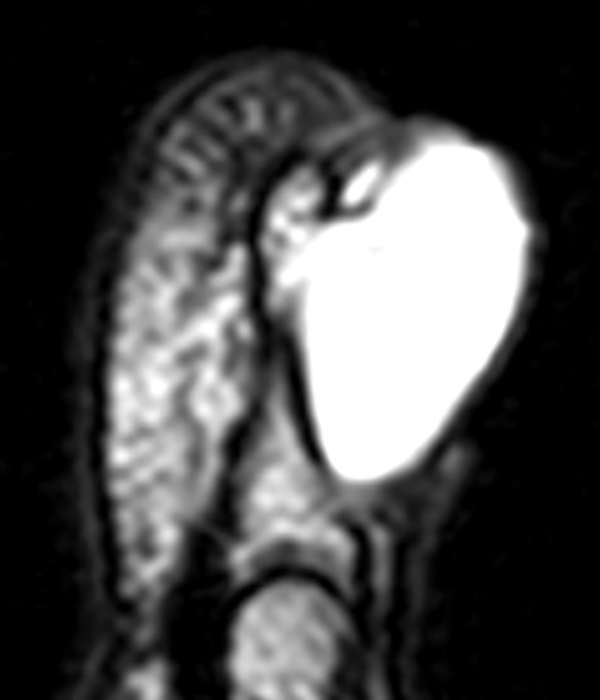
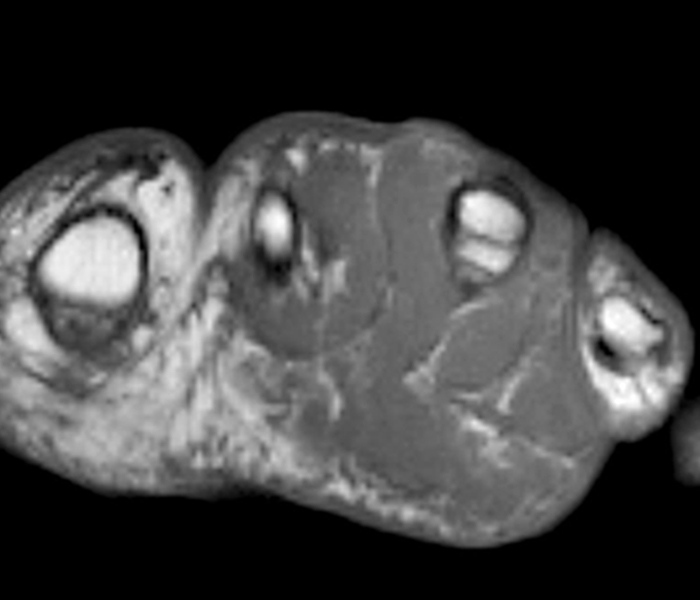
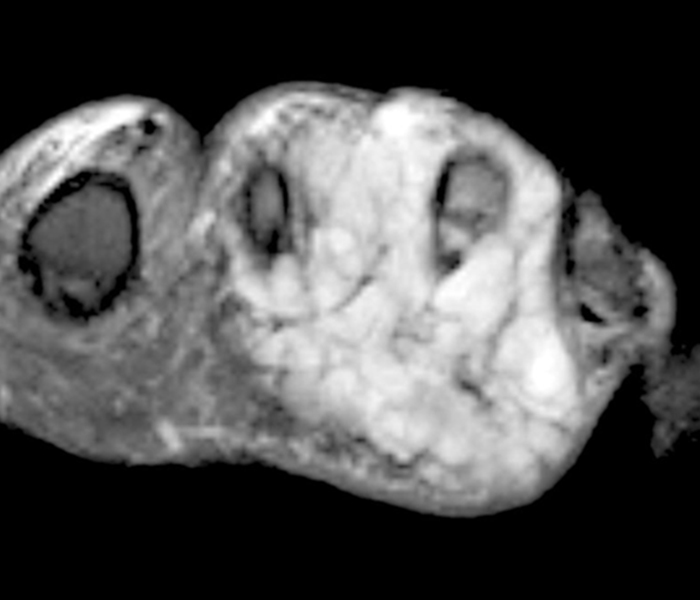
Superficial acral fibromyxoma (SAFM) (Figure 10) is a benign soft-tissue tumor which presents as a slow-growing, painless, solitary mass or nodule located over the subungal and periungal regions of the fingers and toes12,13 although it can be painful in up to 41% of patients.14 The tumor typically ranges in size from 0.6 to 5.0 cm in maximum diameter, extending throughout the entire dermis. The mean age at diagnosis is 43 years, and men are more affected than woman in a ratio of 2:1.15
On plain radiographs, SAFM typically does not calcify, although it can cause erosion of the underlying cortical bone.12 On MRI, SAFM is homogeneously hypointense on T1- and hyperintense on T2-weighted sequences due to the myxoid component. The differential diagnosis of a slow-growing mass or nodule in the periungal or subungal regions of the fingers and toes includes epidermal inclusion cyst, glomus tumor, giant-cell tumor of tendon sheath (GCT-TS), soft-tissue/periosteal ganglion, and chondroma.12 Complete excision is the treatment of choice to prevent recurrence.
Figure 10:
40-year-old female with superficial acral fibromyxoma. Axial T1-weighted (10A) and T2-weighted (10B), sagittal T1-weighted (10C) and T2-weighted (10D) images show a well-circumscribed subungal lesion with mild erosion of the underlying cortical bone. Intraoperative photos illustrate the large subungal lesion (10e) and associated gelatinous material (10f) associated with this myxoid lesion. Images courtesy of David Rubin, MD.
Aggressive Angiomyxoma:
Aggressive angiomyxoma is a rare, benign but locally aggressive lesion, most commonly arising from connective tissues of the perineum or lower pelvis in women of child-bearing age. Patients typically present with a painless, slowly growing soft tissue mass in the pelvic and perineal regions.16 The tumors are subcutaneous or deeper in location, lobulated and poorly circumscribed, typically large (occasionally more than 20 cm in diameter), but without frank invasion of perineal structures.1
At imaging, this lesion is usually low to intermediate in signal intensity on T1-weighted MR images, high signal intensity on T2-weighted images related to the myxoid matrix, and characteristically with a swirled or layered appearance. These tumors tend to displace rather than invade perineal structures. The differential diagnosis primarily includes myxoid liposarcoma.3 Wide excision is the treatment of choice. The tumors behave aggressively with a high rate of local recurrence, but without metastases.
Myxolipoma:
Myxolipoma is a lipoma with prominent myxoid areas, usually in the subcutaneous tissues. Although many lipomas may contain myxoid foci, the terms myxoid lipoma, myxolipoma and lipomyxoma have been used interchangeably to describe lipomas with extensive myxoid change. This is generally regarded as a degenerative phenomenon within a pre-existent lipoma, rather than a distinct subtype.1
Malignant Myxoid Neoplasms
Myxoid Liposarcoma:
Myxoid liposarcoma represents a third of liposarcomas and typically affects younger patients (30–50 years old) than other types of liposarcomas.5 They most often present as a painless, slowly growing soft-tissue mass, often very large. These lesions most commonly involve the lower extremity (75%–80% of cases), particularly the medial thigh (Figures 11 and 12) and popliteal locations, and most frequently are intermuscular in location (70%–80% of cases).3
Myxoid liposarcoma is an intermediate- to high-grade malignancy. Large tumor size (> 10 cm), deep location, irregular margins, lack of lobulations, and presence of thick septa (> 2 mm) are more significantly associated with intermediate-to-high–grade myxoid liposarcoma.5
At MR imaging, myxoid liposarcomas are typically large, well-defined multilobulated intermuscular lesions, and are predominantly low signal intensity on T1-weighted MR images and markedly high signal intensity on T2-weighted MR images. The pathognomonic feature of myxoid liposarcoma on MR images is a fatty component within the mass, potentially seen as small foci of high signal intensity on T1-weighted images (Figure 13, 14), usually constituting only a small volume of the overall mass (<10% of the lesion). Myxoid liposarcoma often demonstrates intense enhancement (Figure 11), although this is highly variable (Figures 12, 14, 15).
Unlike other sarcomas, myxoid liposarcomas have an unusual pattern of metastases, with extrapulmonary metastases preceding pulmonary metastases. Osseous metastases are extremely common, and other extrapulmonary sites of metastases include the abdominal cavity, paraspinal regions, retroperitoneum, and axilla.5 On MRI, osseous metastases appear as T1 hypointense and extremely T2 hyperintense with avid contrast enhancement (Figure 16).
Surgical resection is the treatment of choice for myxoid liposarcomas, with adjuvant or neoadjuvant chemotherapy or combination chemoradiation (Figure 17) used for high-risk or metastatic tumors. The survival for myxoid liposarcomas ranges from 75–83% at 5 years to 63–75% at 10 years.5
Figure 12:
35-year-old male with a myxoid liposarcoma along the right sartorius muscle. Axial T1-weighted (12A), fat-suppressed T2-weighted (12B), and fat-suppressed T1-weighted pre-(12C) and post-(12D) gadolinium contrast enhanced images. The lesion is isointense to muscle (asterisk) and demonstrates heterogeneous increased T2 signal with mild to moderate enhancement which is most prominent peripherally.
Figure 13:
58-year-old woman with low-grade myxoid liposarcoma. Sagittal (13A) and coronal (13B) T1-weighted and axial fat-suppressed T2-weighted (13C) images demonstrate a very large mass at the posterior left thigh. A few small foci of fat signal can be seen on the sagittal T1-weighted image (arrowheads).
Figure 14:
35-year-old myxoid liposarcoma. Axial T1-weighted (14A), fat-suppressed T2-weighted (14B), and post-contrast fat-suppressed T1-weighted images (14C) show a T1 hypointense mass (asterisks) with few thin striations of high T1 signal representing fat (arrowhead in 14A), which suppress on the axial fat-suppressed T2-weighted images (arrowhead in 14B). Intense heterogeneous enhancement is seen (14C).
Figure 15:
39-year-old male with myxoid liposarcoma of the anterior right thigh. 8.4 x 4.8 x 3.5 cm intermuscular soft tissue lesion deep to the sartorius muscle, which demonstrates lower attenuation than muscle on CT (15A), homogeneously T2 hyperintense (15B) and T1 isointense 15C) to muscle, and heterogeneous internal enhancement on the sagittal post-contrast fat-suppressed T1-weighted (15D) image. The mass abuts the femoral vessels but does not surround or encase them (arrows).
Figure 16:
52-year-old male with metastatic myxoid liposarcoma of the left thigh. Sagittal T1-weighted (16A) and coronal fat-suppressed T2-weighted (16B images demonstrate a large mass located posterior to the femur. The patient underwent resection with positive margins and was then treated with adjuvant radiation therapy. Subsequent axial T1-weighted (16C) and coronal post-contrast fat-suppressed T1-weighted (16D) images show recurrence with hypointense T1 (16C) and avidly enhancing (16D) bone metastases (arrowheads) in the spine, pelvis and femurs confirmed by biopsy.
Figure 17:
49-year-old male with myxoid liposarcoma of the distal right thigh, pre- and post- radiation. A lipomatous mass is seen on the radiograph (arrowheads) (17A) and the T1-weighted coronal (17B) and axial (17C) images. The mass heterogeneously enhances on the axial post-contrast fat-suppressed T1-weighted image (17D). Following radiation, the enhancing, non-fatty component has decreased in size on the axial T1-weighted (17E) and axial post-contrast fat-suppressed T1-weighted image (17F).
Myxofibrosarcoma:
Formerly known as the myxoid variant of malignant fibrous histiocytoma (MFH), myxofibrosarcoma typically presents in patients over 50 years of age with a slowly enlarging painless mass, most commonly involving the subcutaneous tissues of the extremities1 (Figures 18-20), occurring more often in the subcutaneous tissues relative to other sarcomas. Unlike other soft-tissue sarcomas which are typically well-defined lesions, myxofibrosarcomas may have infiltrating margins with a propensity for spread along fascial and vascular planes, which predisposes them to incomplete resections and high recurrence rates.
Lesions are often heterogeneous on both T1- and T2-weighted images (Figure 21), though predominately T2 hyperintense, with ill-defined infiltrative margins (Figure 22), and with heterogeneous contrast enhancement. Because of their infiltrative nature, an irregular T2-hyperintense, enhancing curvilinear “tail” may be seen extending from the primary mass into the adjacent soft tissues.
Treatment involves preoperative chemotherapy followed by wide resection. Local recurrences occur in as many as 50%–60% of cases. The 5-year survival rate for patients with myxofibrosarcoma is 60%–70%, with a 50%–70% local recurrence rate and a 23%–30% metastatic rate.3 Metastases are most commonly to the lungs and bones.
Figure 18:
72-year-old male with a high-grade myxofibrosarcoma. Axial T1-weighted (18A), sagittal fat-suppressed T2-weighted (18B), and coronal post-contrast fat-suppressed T1-weighted (18C) images demonstrate a cluster of solid enhancing nodular masses in the subcutaneous fat at the lateral knee, with intermediate T1 signal (18A), high T2 signal (18B), and enhancement of the lesion and adjacent soft tissues (18C).
Figure 19:
53-year-old male with a myxofibrosarcoma, who presented with a “knot” at the lateral aspect of the distal left thigh for several months that was tender only if bumped. Coronal T1-weighted (19A) and axial fat-suppressed T2-weighted (19B) images demonstrate a superficial lesion (arrowheads) with solid components which is heterogeneous on T1-weighted images and hyperintense on T2-weighted images, with the possibility of a sarcoma discussed in the MRI report. The lesion was not resected for unknown reasons and the patient returned 3 years later. Coronal fat-suppressed proton density-weighted (19C) and axial post-contrast fat-suppressed T1-weighted (19D) images now demonstrate significant interval increase in the size of the heterogeneous, enhancing mass.
Figure 21:
79-year-old male with a high-grade myxofibrosarcoma at the lateral aspect of the right pelvis/upper thigh. Coronal fat-suppressed T2-weighted (21A) and T1-weighted (21B) images demonstrate a superficial heterogeneous mass consistent with a sarcoma. Intraoperative photo (21D) shows the aggressive nature of the mass. Images courtesy of Brady Huang, MD, UCSD Department of Radiology.
Figure 22:
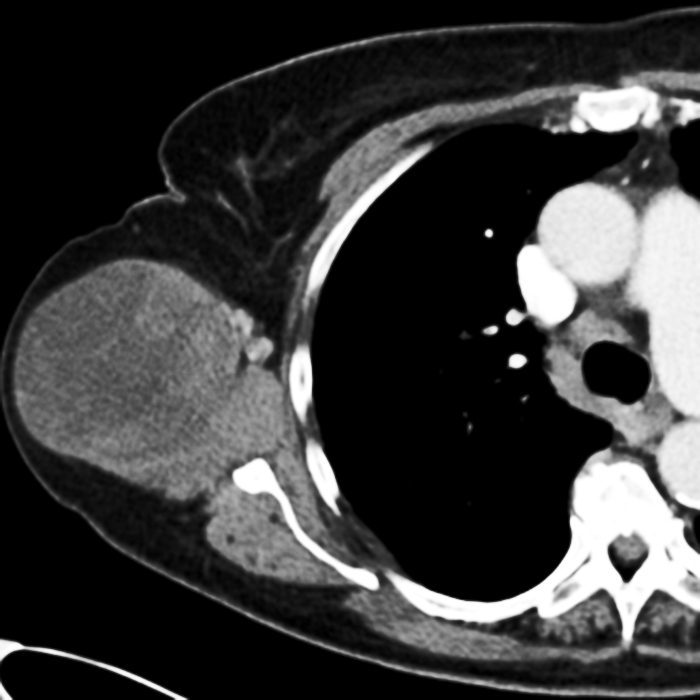
75-year-old female with a myxofibrosarcoma at the lateral right chest wall. Axial CT (22A), coronal T1-weighted (22B), coronal STIR (22C), and coronal post-contrast fat-suppressed T1-weighted mages (22D) demonstrate an infiltrative mass with enhancing perilesional edema along the inferior margin of the lesion (asterisks).
Low-grade Fibromyxoid Sarcoma:
Low-grade fibromyxoid sarcoma is a rare tumor that affects young to middle-aged adults, which may involve the extremities, trunk, or retroperitoneum, and typically involves the deep soft tissues.1 Clinically, most patients present with slowly growing soft tissue swelling over several months or years.
The tumor is characterized by relatively benign pathologic features including intermixed myxoid and fibrous stroma, yet it displays aggressive behavior with high rates of recurrence (Figures 23 and 24) and distant metastatic disease.17 There is a marked variation in tumor size at surgery, ranging from a few centimeters to 20 cm in maximal diameter.1
MRI characteristics are consistent with the tumor’s fibrous and myxoid content, with the fibrous component T1 and T2 hypointense, the myxoid component T1 hypointense and T2 hyperintense, and with heterogeneous enhancement. The tumor may show a gyriform pattern with multiple layers of hypo- or isointense signal on T2-weighted images and may contain multiple T2-hyperintense enhancing nodules. The differential diagnosis includes myxoid neurofibroma, desmoid fibromatosis, myxoid dermatofibrosarcoma protuberans, myxoid MPNST and low-grade myxofibrosarcoma.
Surgical excision with wide margins is the treatment of choice. Local recurrence is usually in the form of multiple lesions (Figure 24), and metastases are most common to the lungs. Prolonged latency between diagnosis and recurrent disease has been described, with up to 65% recurrence and 45% metastases in a period ranging from 6 months to 45 years.5 Despite this, the majority of patients are still alive 10 years after the initial surgery.1
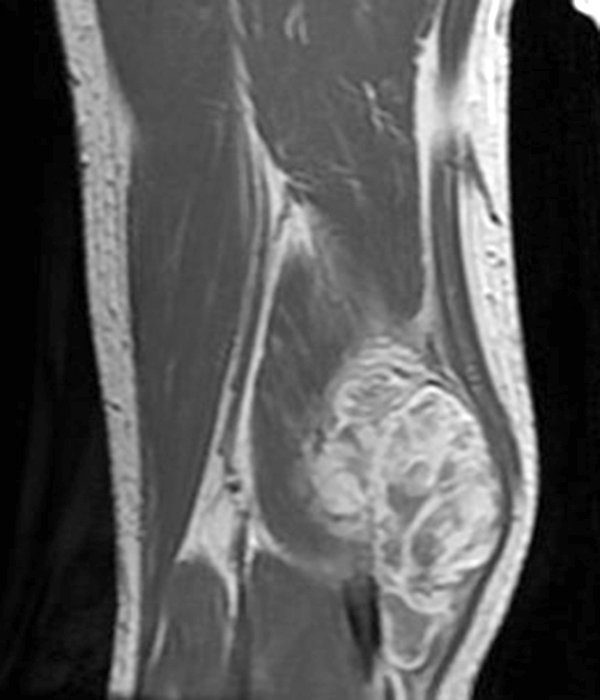
Figure 23:
86-year-old female with history of a previously excised low-grade myxofibrosarcoma, now with a recurrent mass at the posterior knee. A multilobulated mass measuring up to 9.5 cm is seen with several septations, with hyperintense signal on sagittal STIR (23A), hypointensity on sagittal T1-weighted (23B), and mild peripheral and septal enhancement on the sagittal post-contrast fat-suppressed T1-weighted images (23C), compatible with recurrent tumor.
Figure 24:
46-year-old with a low grade fibromyxoid sarcoma. Axial T1-weighted (24A) and coronal fat-suppressed T2-weighted images (24B) demonstrate a large heterogeneous and multi-nodular mass within the deep left buttock located adjacent to the sciatic nerve . Differential diagnosis at MRI included peripheral nerve sheath tumor, although biopsy confirmed low grade fibromyxoid sarcoma. Coronal STIR (24C) and axial post-contrast fat-suppressed T1-weighted (24D) 10 months later show several small nodular foci adjacent to the sciatic nerve with T2 hyperintensity and associated enhancement (arrows ), which were subsequently confirmed to be recurrent tumor.
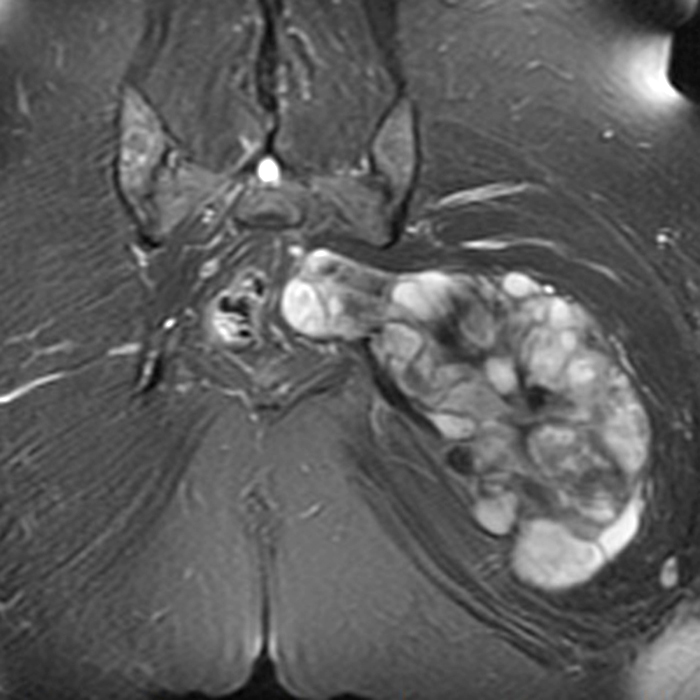
Extraskeletal Myxoid Chondrosarcoma:
An intermediate-grade tumor characterized by the presence of malignant chondroblasts in a myxoid matrix, extraskeletal myxoid chondrosarcomas typically arise in the extremities, most commonly the thigh, affecting middle-aged to older patients.18 Lesions most frequently involve the deep soft tissues, and less commonly are subcutaneous. Patients typically present with a slow growing painless mass. The mean tumor size at the time of presentation ranges from 7 to 8.9 cm, although tumors may be as large as 25 cm.1 Metastases most commonly involve the lungs, followed by the bones.
On imaging, extraskeletal myxoid chondrosarcoma presents as a large well-defined lobulated heterogeneous lesion (Figures 25 and 26), often with the presence of a pseudocapsule and intratumoral cysts and hemorrhage. Reflecting its extremely high water content, extraskeletal myxoid chondrosarcoma appears as areas of low attenuation on CT images and high signal intensity on T2-weighted MR images (Fig 9a, 9c), with varying patterns of enhancement after contrast administration. A chondroid matrix may be seen on CT (Figure 25).
Treatment includes wide surgical resection, although the tumors are prone to recurrence, and thus adjuvant or neoadjuvant chemotherapy with or without radiation may be given. The 5- and 10-year survival rates for extraskeletal myxoid chondrosarcomas are 74–100% and 60–83%, respectively.5
Figure 25:
56-year-old male with extraskeletal myxoid chondrosarcoma at the posterior left pelvis. Axial CT image (25A) demonstrates a low attenuation lesion with subtle tiny foci of increased density (arrowheads) representing chondroid matrix. Axial T1-weighted (25B) and fat-suppressed T2-weighted (25C) images show a heterogeneous T1 hypointense and T2 hyperintense mass.
Figure 26:
43-year-old male with extraskeletal myxoid chondrosarcoma of the foot. Coronal T1-weighted (26A), fat-suppressed T2-weighted (26B), and post-contrast fat-suppressed T1-weighted (26C) images demonstrate a large lobulated and heterogeneous mass, which avidly enhanced following contrast administration.
Ossifying Fibromyxoid Tumor:
Ossifying fibromyxoid tumor is a rare intermediate-grade malignant soft-tissue tumor that typically manifests as a slow-growing firm area of painless subcutaneous swelling in the extremities19, measuring from less than 5 cm to 10 cm.3
On imaging, ossifying fibromyxoid tumor appears as well-defined heterogeneous soft-tissue mass that shows foci of curvilinear ossification, best seen on CT. MRI shows heterogeneous high signal intensity on T2-weighted images and intermediate-to-low signal intensity on T1-weighted images with heterogeneous contrast enhancement, with the ossified component appearing T1 and T2 hypointense. Fatty marrow may be visualized as a T1-hyperintense component within the ossified elements.5
The differential diagnosis on imaging includes ossifying hematoma, myositis ossificans, extraskeletal or parosteal osteosarcoma, extraskeletal chondrosarcoma, and other ossifying soft-tissue sarcomas or metastases.5 After wide surgical excision, local recurrence is unusual. Metastases are uncommon and typically to the lungs.1
Conclusion
When evaluating soft tissue lesions, it is important to remember that some solid masses may be extremely T2 hyperintense, including myxoid lesions, occasionally mimicking cysts. Thus, the differential diagnosis for lesions that are predominantly T2 hyperintense includes not only fluid-filled lesions (ganglia, synovial cysts, seromas, etc.) but also solid lesions (such as myxomas, myxoid sarcomas, PNSTs, and synovial sarcomas).20 When in doubt, administration of contrast can help distinguish between true cysts and solid lesions. Larger size (greater than 7 cm), heterogeneity on T1-weighted MR images, and greater enhancement are more suggestive of malignancy.21
Imaging is extremely important for presurgical planning, and careful follow-up imaging is essential for many of these lesions to detect recurrent or metastatic disease. The ability to recognize and differentiate myxoid tumors is important and will help guide patient management.
Special thanks to Brady Huang, MD (UCSD Dept. of Radiology) for providing multiple cases of malignant tumors used in this web clinic.
References
- Graadt van Roggen JF, Hogendoorn PC, Fletcher CD. Myxoid tumours of soft tissue. Histopathology 1999;35(4):291–312. ↩
- Murphey MD, McRae GA, Fanburg-Smith JC, Temple HT, Levine AM, Aboulafia AJ. Imaging of soft-tissue myxoma with emphasis on CT and MR and comparison of radiologic and pathologic findings. (2002) Radiology. 225 (1): 215-24. ↩
- Jonelle M. Petscavage-Thomas, Eric A. Walker, Chika I. Logie, Loren E. Clarke, Dennis M. Duryea, Mark D. Murphey. Soft-Tissue Myxomatous Lesions: Review of Salient Imaging Features with Pathologic Comparison. (2014) RadioGraphics. 34 (4): 964-80. ↩
- Bancroft LW, Kransdorf MJ, Menke DM, O’Connor MI, Foster WC. Intramuscular myxoma: characteristic MR imaging features. (2002) AJR. American journal of roentgenology. 178 (5): 1255-9. ↩
- Baheti AD, Tirumani SH, Rosenthal MH, et al. Myxoid soft-tissue neoplasms: comprehensive update of the taxonomy and MRI features. AJR Am J Roentgenol. 2015;204(2):374-385. ↩
- Iwasko, N., Steinbach, L., Disler, D. et al. Imaging findings in Mazabraud’s syndrome: seven new cases. Skeletal Radiol 31, 81–87 (2002). ↩
- Benhamou J, Gensburger D, Messiaen C, Chapurlat R. Prognostic factors from an epidemiologic evaluation of fibrous dysplasia of bone in a modern cohort: the FRANCEDYS study. J Bone Miner Res. 2016 Dec;31(12):2167-72. ↩
- Majoor BCJ, van de Sande MAJ, Appelman-Dijkstra NM, Leithner A, Jutte PC, Vélez R, Perlaky T, Staals EL, Bovée JVMG, Hamdy NAT, Dijkstra SPD. Prevalence and Clinical Features of Mazabraud Syndrome: A Multicenter European Study. (2019) The Journal of bone and joint surgery. American volume. 101 (2): 160-168. ↩
- Vidu Viorica., et al. “Synchronous Superficial Angiomyxoma without Carney-Complex- A Particularly Entity”. EC Clinical and Medical Case Reports 3.3 (2020): 01-08. ↩
- Kwon HJ, Kim JH, Choi JH, Gu MJ. Nerve sheath myxoma: Unusual location and a diagnostic pitfall. Indian J Pathol Microbiol. 2017;60(4):610-611. ↩
- Bhat A, Narasimha A, C V, Vk S. Nerve sheath myxoma: report of a rare case. J Clin Diagn Res. 2015;9(4):ED07-ED9. ↩
- Bindra J, Doherty M, Hunter JC. Superficial acral fibromyxoma. Radiol Case Rep. 2012;7(3):751. ↩
- Varikatt W, Soper J, Simmons G, Dave C, Munk J, Bonar F. Superficial acral fibromyxoma: a report of two cases with radiological findings. Skeletal Radiol. 2008;37(6):499-503. ↩
- Hollmann TJ, Bovée JV, Fletcher CD. Digital fibromyxoma (superficial acral fibromyxoma): a detailed characterization of 124 cases. Am J Surg Pathol. 2012;36(6):789–98. ↩
- Lee, S., Reid, M.A.R. Superficial acral fibromyxoma: a case report with radiological review. Skeletal Radiol 47, 1021–1028 (2018). ↩
- Fetsch JF, Laskin WB, Lefkowitz M, Kindblom LG, Meis-Kindblom JM. Aggressive angiomyxoma: a clinicopathologic study of 29 female patients. Cancer. 1996;78(1):79-90. ↩
- Lee BJ, Park WS, Jin JM, Ha CW, Lee SH. Low grade fibromyxoid sarcoma in thigh. Clin Orthop Surg. 2009;1(4):240-243. ↩
- Zhang GW, Wang AJ, Zhang GH, Zhao SN, Qu JL. Extraskeletal myxoid chondrosarcoma: A report of two cases. Oncol Lett. 2014;7(4):1289-1291 ↩
- Enzinger FM, Weiss SW, Liang CY. Ossifying fibromyxoid tumor of soft parts. A clinicopathological analysis of 59 cases. Am J Surg Pathol. 1989;13(10):817-827. ↩
- Wu JS, Hochman MG. Soft-tissue tumors and tumorlike lesions: a systematic imaging approach. Radiology. 2009;253 (2): 297-316. ↩
- Harish S, Lee JC, Ahmad M, Saifuddin A. Soft tissue masses with “cyst-like” appearance on MR imaging: distinction of benign and malignant lesions. Eur Radiol 2006;16:2652–2660. ↩