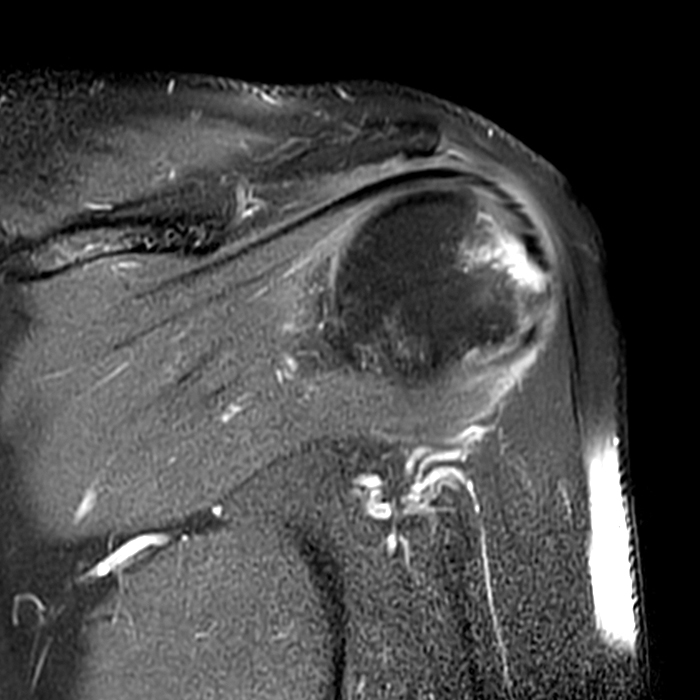
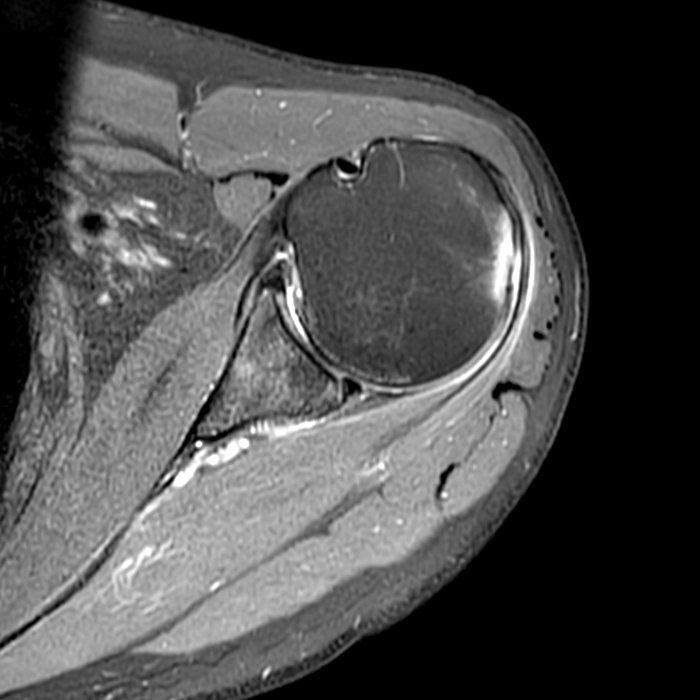
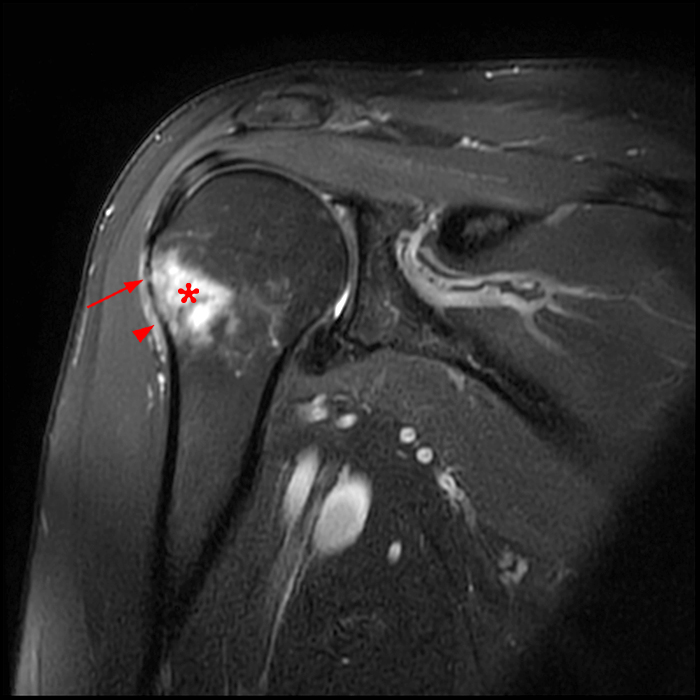
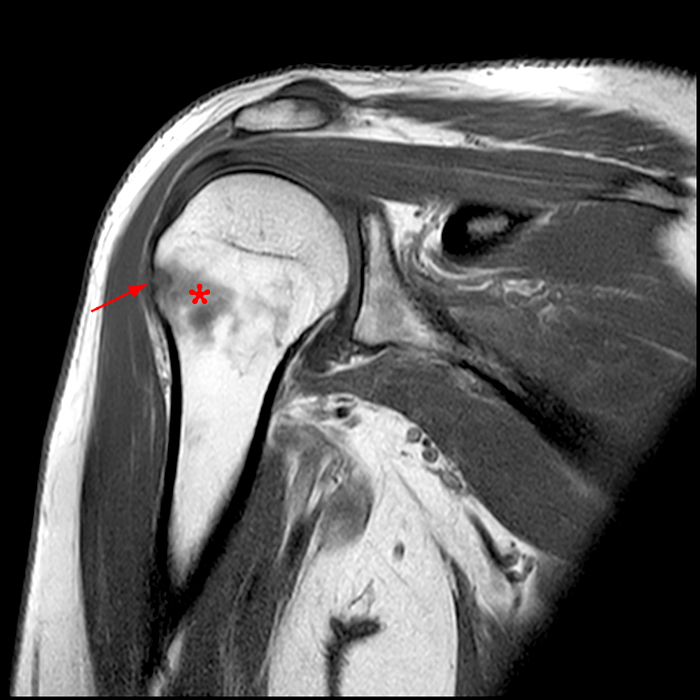
Clinical History: A 64-year-old male complains of shoulder pain and limited range of motion for six weeks after an influenza vaccination. Fat-suppressed T2-weighted coronal (1A) and proton density-weighted axial (1B) images of the left shoulder are provided. What are the findings? What is your diagnosis?
Findings
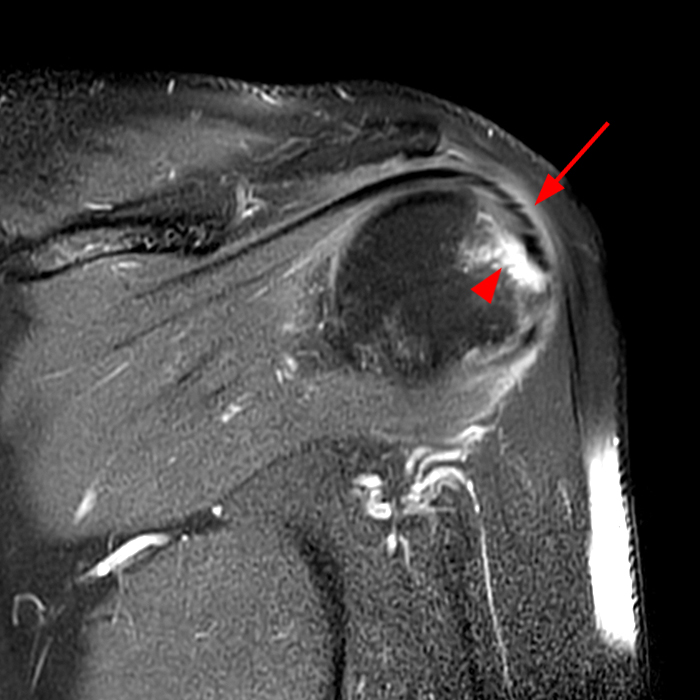
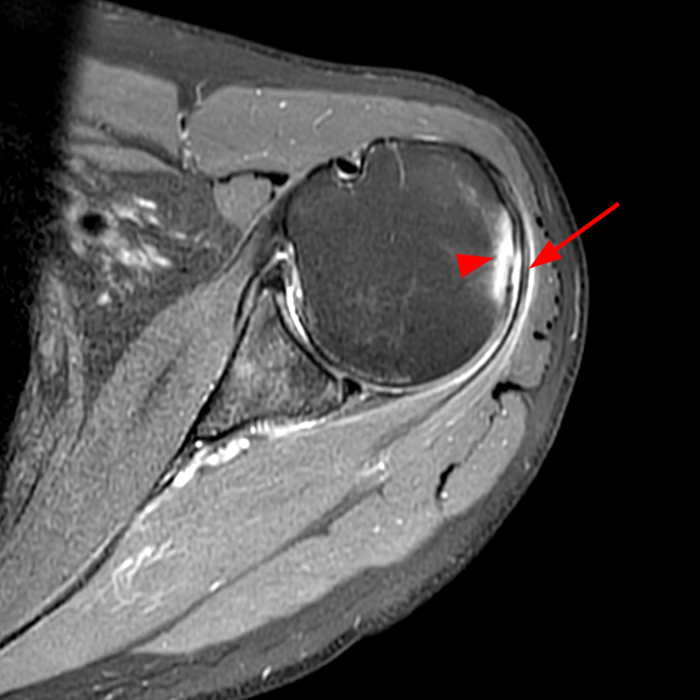
Figure 2:
The fat-suppressed T2-weighted coronal image (2a) and fat-suppressed proton density axial image (2b) demonstrate tendinopathy of the infraspinatus tendon with increased signal within the tendon and peritendinous soft tissues (arrow) and reactive marrow edema of the posterior greater tuberosity (arrowhead).
Diagnosis
Shoulder injury related to vaccine administration (SIRVA).
Introduction
Shoulder injury related to vaccine administration (SIRVA) is defined by the Health Resources & Services Administration (HRSA) as shoulder pain and limited range of motion after a vaccine injection intended for intramuscular administration in the upper arm.1
Benefits of Vaccinations
Before there is any discussion of the uncommon occurrence of vaccine complications, the benefits of vaccines should be stated. Aside from clean water practices, vaccines have done more to reduce complications of infectious diseases than any medical or public health advancement.2 Vaccines globally prevent approximately six million deaths annually.3 Vaccines have other positive effects, including cancer prevention, health care savings, preventing antibiotic resistance, extending life expectancy, promoting economic growth, and promoting political stability.4 Vaccines and public health measures have been the mainstay of controlling the current Covid-19 pandemic.5
Clinical Presentation
SIRVA is an uncommon event, although the true incidence is unknown because most cases likely go unreported. In a retrospective review of nearly 3 million vaccinated patients, the attributable risk was 7.78 additional cases of bursitis per 1 million patients.6 The proposed etiology of the symptoms is injection of the vaccine or trauma from the needle “into and around the underlying bursa of the shoulder resulting in an inflammatory reaction.” SIRVA is defined as an injury to the musculoskeletal structures of the shoulder and is not a neurological injury. Four criteria must be met for the diagnosis of SIRVA: No pre-existing condition that would cause the symptoms, pain occurs within 48 hours of the injection, pain and reduced range of motion limited to the shoulder where the intramuscular injection occurred and no other conditions that would explain the symptoms such as brachial neuritis.1
The term vaccination-related shoulder dysfunction has been suggested as a replacement for SIRVA because it better describes the clinical manifestations of the process and avoids the term “injury” which implies a traumatic etiology.7 SIRVA, in terms of etiology and scope of pathology, is not without controversy. Most cases are thought to be related to improper injection technique with injections into the subacromial bursa, rotator cuff, glenohumeral joint, or bone. Some have speculated there may be an immune response contributing to the complications of chronic bursitis, adhesive capsulitis, rotator cuff tendinopathy, osseous edema, erosive osseous lesions, and osteonecrosis.8,9,10,11,12,13,14,15,16,17
Compensation For SIRVA
To protect the manufacturers of vaccines from liability lawsuits, in 1986, Congress established the Vaccine Injury Compensation Program (VICP). VICP is a no-fault compensation program meaning the patient must show the vaccine was the causative factor of the injury regardless of who is to blame. The reduction in legal and administrative costs and lower levels of compensation, in theory, offset the number of increased claimants, and the claims can be resolved in a shorter period of time.18 VICP is funded by a $0.75 excise tax on each vaccine. The VICP is administered by government agencies, including the Health Resources and Services Administration (HRSA).1
In 2011 the National Academy of Medicine recommended adding shoulder injuries to the Vaccine Injury Table, which was done in March of 2017.19 After the addition of SIRVA to the Vaccine Injury Table, the claims for shoulder injuries went from 10 in 2011 to 711 in 2019. In 2010, SIRVA claims were 1.1% of the claims filed to VICP, and by 2017-19, SIRVA claims made up 52.6% of claims filed to the VICP. The median award for a SIRVA compensation claim is $71,355.26 compared to $22,530 for comparable claims awarded in the civil tort system. Approximately $119,154,985 has been paid out of the Vaccine Injury Compensation Trust Fund between 2016-2019 for SIRVA complaints. The majority of claimants are adults.1,20
According to the Department of Justice, 27 out of 2,214 SIRVA claims filed since 2017 were identified as having altered medical records, including changes to the site of vaccination.20 Between 1985 and 2021, the amount paid to attorneys for petitions that received financial compensation was $236,100,498.66. In the same period of time, the amount paid to attorneys for dismissed cases was $94,550,863.34, and interim attorney fees were $45,012,813.40. VICP will pay attorney fees whether or not the petition is awarded compensation if certain minimal criteria are met.1 Attorneys specializing in vaccine litigation have come to be called Vaccine Attorneys. By report, Vaccine Attorneys account for the majority of SIRVA compensation cases.19 Currently, SIRVA remains on the Vaccine Injury Table and there are proponents and opponents for whether it should be removed or kept in place.20
The concept of SIRVA has received some criticism. In a position paper, the American Academy of orthopaedic Surgeons stated that rotator cuff tendinopathy, adhesive capsulitis, and arthritis could not be caused by vaccine administration. The paper cites a lack of scientific data linking shoulder derangements to vaccinations and a high prevalence of pre-existing shoulder problems in patients who receive vaccinations which makes causation difficult to prove. The authors conclude that absent strong scientific evidence, “vaccine administration should be presumed merely coincident with these common shoulder pathologies.”19
Injection Site Issues
SIRVA is thought to be related in most patients to improper injection technique. The appropriate size and length of the needle should be chosen according to CDC guidelines. The CDC needle recommendations for intramuscular deltoid vaccine injections vary depending on age, sex, and body weight. 22-25 gauge needles are recommended for all patients. Needle length recommendations are as follows: 1” needle for 130 lbs. or less ( 5/8” if the skin is stretched tightly); 1” needle for 130-152 lbs.; 1-1.5” needle for male 152-260 lbs. and female 152-200 lbs.; and 1.5” needles for male >260 lbs. and female >200 lbs.21
The lateral deltoid muscle is the most common site for intramuscular vaccine injections. The lateral deltoid muscle is thickest in the middle one-third, approximately 2 inches (approximately 5 cm) inferior to the lateral acromion. Additionally, the superior one-third of the muscle is closer to the subacromial space and the insertion sites of the rotator cuff tendons compared to the middle one-third of the deltoid muscle.9,22,23 Bodor reported the distance from the skin to the subacromial bursa varied from 0.8-1.6 cm, and with standard 2.54 cm needles, a deep injection in the superior one-third of the lateral deltoid muscle can reach the bursa and tendon insertion sites.8
In order to properly inject the middle one-third of the deltoid muscle, it is advised that the upper arm be fully exposed and both the patient and vaccinator be seated, which will help with needle control and avoid angulation or over-penetration.13 Another method proposed to increase injection safety entails having the ipsilateral arm abducted by 60 degrees by having the hand placed on the hip. The injection site is determined by having the vaccinator place their index finger on the acromion, their thumb on the deltoid tuberosity, and injecting between those two sites.22
The lateral deltoid muscle is easily and rapidly accessible, and exposure of the muscle in public spaces is accepted in the United States. Anecdotally, the lateral deltoid muscle 5 cm inferior to the acromion is relatively avascular, relatively removed from the posterior humeral circumflex artery, and is relatively removed from major nerves such as the axillary and radial nerves.
Proposal for Posterior Deltoid Muscle Injections
Assuming improper injection techniques of the lateral deltoid are the cause for most cases of SIRVA, then alternative sites should be considered. Assuming most of the cases of SIRVA are related to overpenetration of the needle, choosing a thicker component of the deltoid muscle could possibly decrease those complications. A substitute for the lateral deltoid muscle may be the posterior deltoid muscle. To consider this hypothesis, forty consecutive shoulder MR examinations were reviewed, and measurements from axial plane images were obtained 5 cm inferior to the lateral acromion of the lateral deltoid and posterior deltoid muscles perpendicular to the overlying skin surface (Table I).

The posterior deltoid muscle was over twice the thickness of the lateral deltoid muscle in this cohort of patients: 3.55 cm compared to 1.63 cm (Figure 3). In a review of 476 patients with SIRVA recommended by VICP for compensation, 82.8% were female, and the relatively lower volume of soft tissues may predispose this population to injection site complications.24 It is unknown how the thicker posterior deltoid muscle would affect routine injection site pain and absorption of the vaccine.
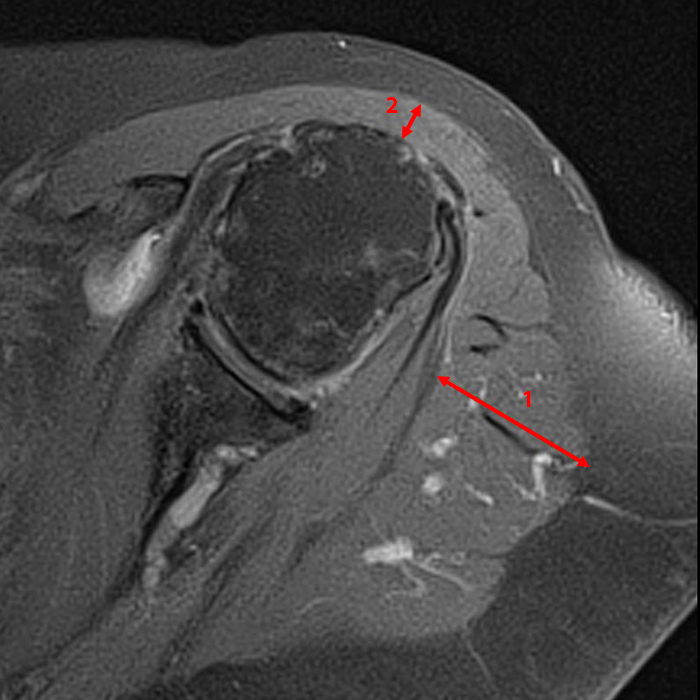
Figure 3:
Measurements of posterior and lateral deltoid muscles. The axial fat-suppressed proton density image in an 80-year-old female was used for measurements of the posterior and lateral deltoid muscles. The measurements were obtained 5 cm inferior to the lateral acromion. The plane for the posterior deltoid muscle measurement was derived from the sagittal oblique image closest to the lateral acromion. The posterior deltoid muscle thickness is 3 cm (1), and the lateral deltoid muscle thickness is 0.7 cm (2)
Moving the injection site to the posterior deltoid muscle may simplify the choice of the needle lengths and increase the margin of error if the wrong length is chosen. To further evaluate the posterior deltoid muscle as a potential injection site, in a second group of forty consecutive MR examinations, measurements in the axial plane 5 cm inferior to the lateral acromion were obtained of the subcutaneous fat thickness and the thickness of the posterior deltoid muscle. The measurements were obtained in the plane of the sagittal oblique image closest to the lateral acromion (Table II).

Most of the patients in this cohort could be safely vaccinated with a 1-inch (2.54 cm) needle. A small number of patients would require a 1.5-inch needle (3.8 cm) or manual compression of the soft tissues. Additionally, moving the injection site to the posterior deltoid muscle may decrease inadvertent subacromial bursal injections because the lateral reflection of fluid-filled subacromial bursa on MR images usually extends more caudally than the posterior reflection (Figure 4).
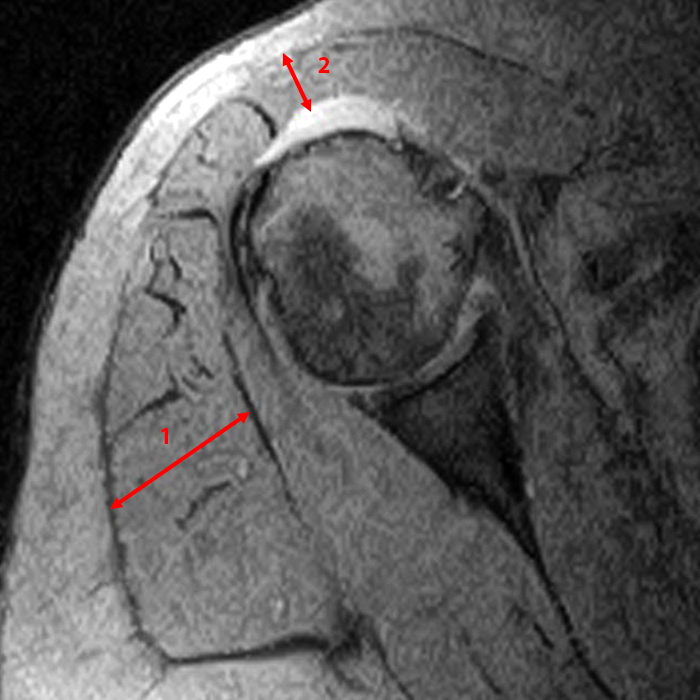
Figure 4:
Caudal extension lateral recess subacromial bursa. The axial gradient echo image in a 72-year-old female demonstrates the lateral recess of the subacromial bursa 5 cm inferior to the lateral acromion. The posterior deltoid muscle measure 3.4 cm in thickness (1), and the lateral deltoid muscle measures 1.3 cm in thickness (2). The lateral recess of the subacromial bursa is deep to the lateral deltoid muscle (arrow), and even slight over-penetration of the needle could result in a subacromial injection.
Best practice for injections includes avoiding shoulders that have acute or chronic problems or prior surgery. However, in many patients, both shoulders may be problematic, and maybe in those patients, the posterior deltoid muscle will be a better injection site. Note that the measurements in Tables I and II were obtained with the patients in the supine position with a surface coil in place, and those measurements may change in an unrestrained upright position.
MRI Findings
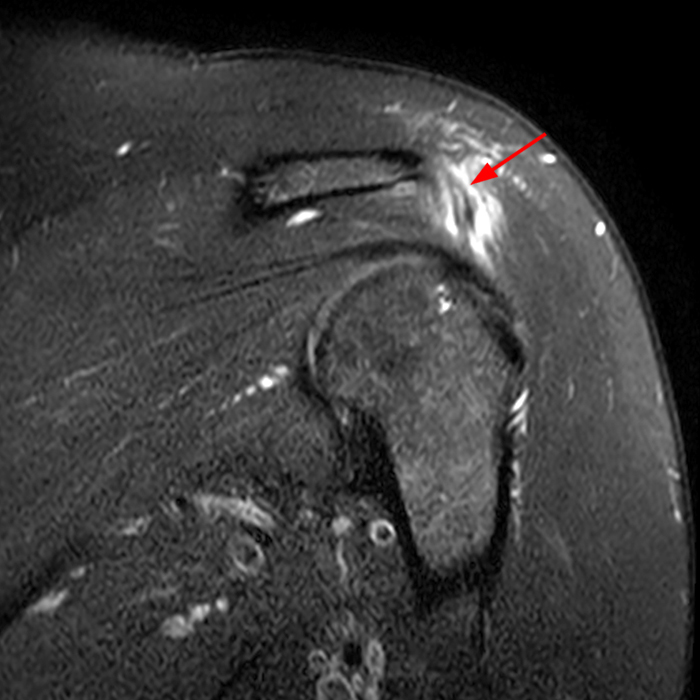
Multiple MR findings have been reported with SIRVA, and the complications are predominantly related to the site of the inadvertent injection. For the most part, the abnormalities are nonspecific and can be seen in patients without a history of vaccination. One of the more common sites of inadvertent injections are the insertion sites of the infraspinatus and teres minor tendons at the posterior tuberosity insertion site secondary to deep and high lateral deltoid injections (Figure 5).
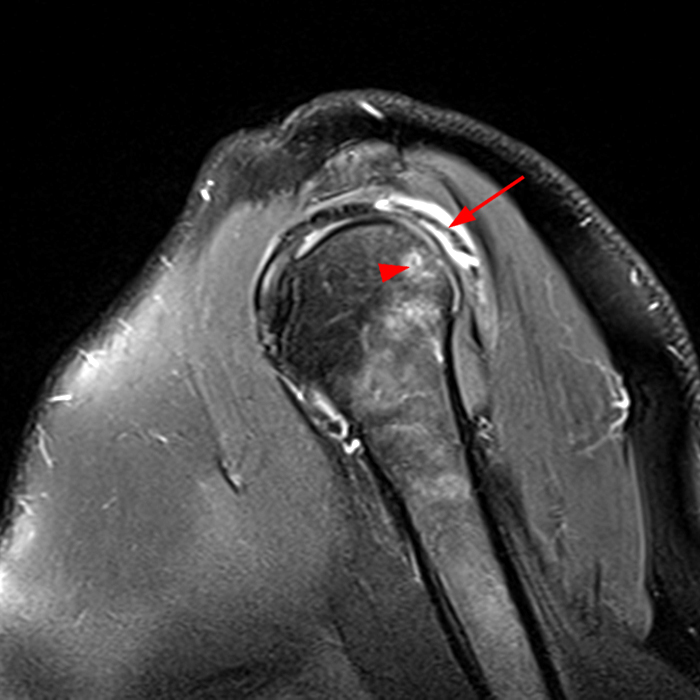
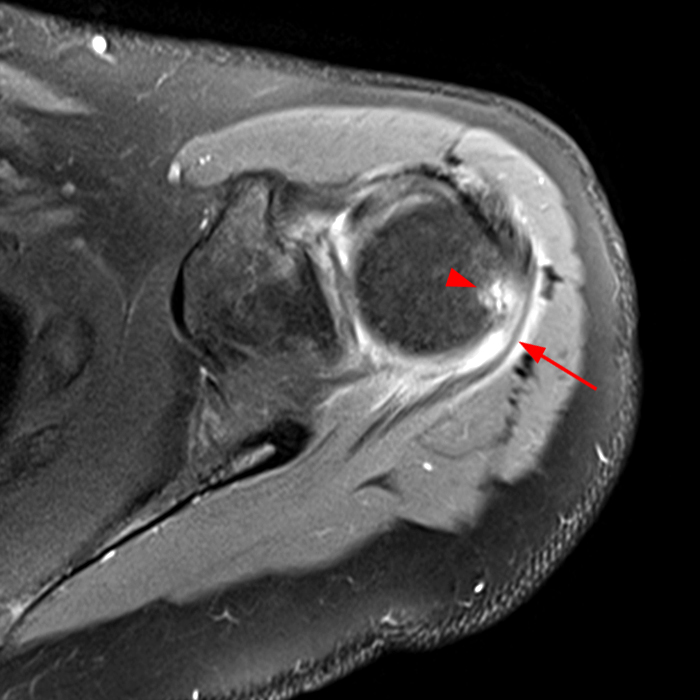
Figure 5:
Abnormalities likely related to a high and deep injection. Fifty-one-year-old female with pain and limited range of motion, flu vaccine 2 weeks prior. A sagittal fat-suppressed T2-weighted image (5a) shows fluid signal surrounding the infraspinatus tendon (arrow) and osseous edema posterior greater tuberosity (arrowhead). An axial fat-suppressed proton density image (5b) reveals fluid signal surrounding the infraspinatus tendon (arrow) and marrow edema posterior greater tuberosity (arrowhead). Images courtesy Marko Bodor, M.D., Bodor Clinic.
In some patients, a high injection will miss the subacromial bursa and instead result in symptomatic injury/inflammation of the lateral deltoid muscle origin (Figure 6).
Figure 6:
Intramuscular abnormality likely related to a high injection site. Sixty-three-year-old female with pain 5 weeks after a vaccine. A coronal fat-suppressed T2-weighted image (6a) shows interstitial fluid signal within the lateral deltoid muscle adjacent to the lateral acromial origin (arrow). An axial fat-suppressed proton density image (6b) shows the interstitial edema within the midportion of the lateral deltoid muscle origin (arrowhead).
In the presence of a full-thickness rotator cuff tear, a subacromial injection can communicate with the glenohumeral joint and possibly cause intraarticular complications (Figure 7).
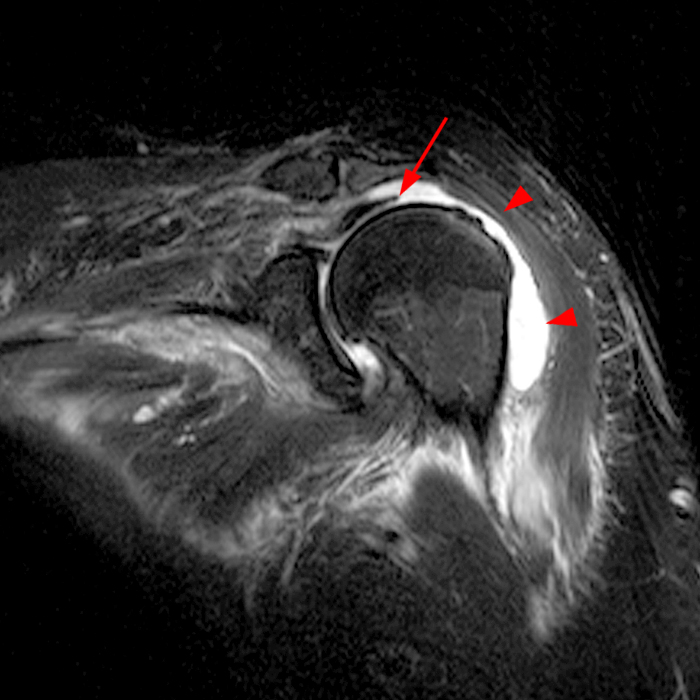
Figure 7:
Communication of the subacromial bursa and glenohumeral joint through a large rotator cuff tear. Seventy-four-year-old female with pain one week after a vaccine. A coronal fat-suppressed T2-weighted image demonstrates a large full-thickness tear of the supraspinatus tendon (arrow) and a large amount of fluid in the subacromial bursa (arrowhead). The vaccination needle may have entered the subacromial bursa and glenohumeral joint.
Most of the osseous abnormalities of SIRVA are likely secondary changes. In a smaller number of patients, the osseous abnormalities are direct osseous injuries secondary to periosteal/cortical penetration of the needle (Figure 8).
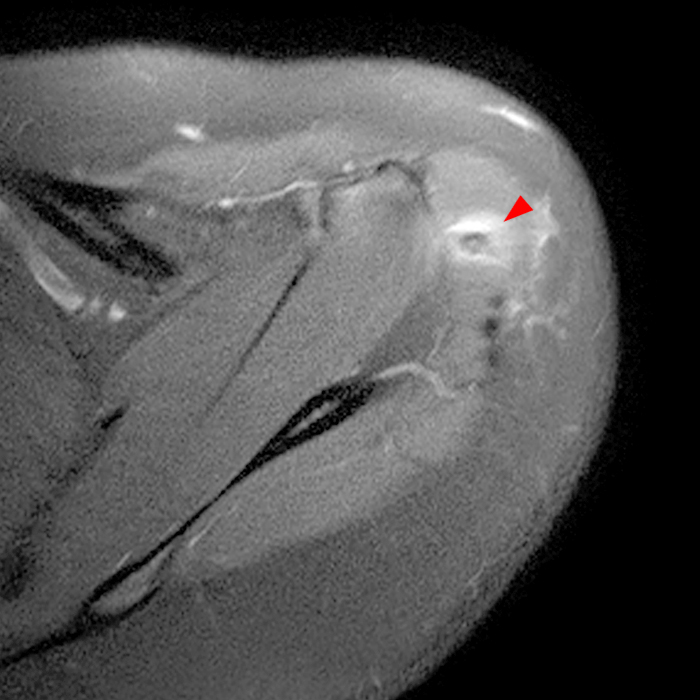
Figure 8:
Osseous abnormality likely related to periosteal/cortical penetration of the vaccine needle. Forty-eight-year-old female with pain after a recent vaccination. A coronal fat-suppressed T2-weighted image (8a) shows marrow edema (asterisk), cortical irregularities (arrow), and pericortical/periosteal fluid signal (arrowhead). A coronal T1-weighted image (8b) confirms signal changes in the marrow (asterisk) and cortical thinning (arrow).
Differential diagnosis
The clinical presentation and MR findings with SIRVA are nonspecific. Patients without vaccinations can have exactly the same clinical findings and imaging findings. The exceptions to this include osseous injuries away from the greater tuberosity in the plane of the injection site and florid inflammatory changes in younger patients with no history of shoulder problems.
Treatment and Prognosis
The mainstay of SIRVA treatment is physical therapy and corticosteroid injections, and in recalcitrant cases, surgery can be performed.8, 10-12, 25,26 Treatment using ultrasound aspiration and debridement has shown early promising results.7
The prognosis of SIRVA has not been rigorously studied. In an evaluation of 859 patients with SIRVA, 65% of patients had symptoms for more than 1 month, and 25% had symptoms lasting more than 3 months.27
Summary
SIRVA is an uncommon complication of intramuscular injections into the lateral deltoid muscle. Most of the complications are likely related to injections that are too high and/or too deep. The proposed etiology is an inflammatory reaction related to injection of the vaccine or trauma from the needle with resultant abnormalities of the deltoid muscle, subacromial bursa, rotator cuff, proximal humerus, and glenohumeral joint. It is possible that the posterior deltoid muscle, which is larger in thickness and further removed from the subacromial bursa, may be a safer injection site and that concept merits further evaluation.
References
- National Vaccine Inury Compensation Program (Internet). Health Resouces & Services Administration. 2021. (Available from: https://www.hrsa.gov/vaccine-compensation/index.html.) ↩
- Plotkin SL, Plotkin SA. A short history of vaccination. In: Plotkin SA, Orenstein WA, editors. Vaccines. 4th ed. Philadelphia: WB Saunders; 2004. p. 1-15. ↩
- Ehreth J. The global value of vaccination. Vaccine. 2003;21(7-8):596-600. ↩
- Andre FE, Booy R, Bock HL, Clemens J, Datta SK, John TJ, et al. Vaccination greatly reduces disease, disability, death and inequity worldwide. Bull World Health Organ. 2008;86(2):140-6. ↩
- CDC. Science Brief: Background rational and evidence for Public Health recommendations for fully vaccinated people. 2021. ↩
- Hesse EM, Navarro RA, Daley MF, Getahun D, Henninger ML, Jackson LA, et al. Risk for Subdeltoid Bursitis After Influenza Vaccination: A Population-Based Cohort Study. Ann Intern Med. 2020;173(4):253-61. ↩
- Bodor M. Personal Communication. Bodor Clinic; 2021. ↩
- Bodor M, Montalvo E. Vaccination-related shoulder dysfunction. Vaccine. 2007;25(4):585-7. ↩
- Cross GB, Moghaddas J, Buttery J, Ayoub S, Korman TM. Don’t aim too high: Avoiding shoulder injury related to vaccine administration. Aust Fam Physician. 2016;45(5):303-6. ↩
- Erickson BJ, DiCarlo EF, Brause B, Callahan L, Hannafin J. Lytic Lesion in the Proximal Humerus After a Flu Shot: A Case Report. JBJS Case Connect. 2019;9(3):e0248. ↩
- Kuether G, Dietrich B, Smith T, Peter C, Gruessner S. Atraumatic osteonecrosis of the humeral head after influenza A-(H1N1) v-2009 vaccination. Vaccine. 2011;29(40):6830-3. ↩
- Messerschmitt PJ, Abdul-Karim FW, Iannotti JP, Gobezie RG. Progressive osteolysis and surface chondrolysis of the proximal humerus following influenza vaccination. orthopaedics. 2012;35(2):e283-6. ↩
- Natanzi N, Hebroni F, Bodor M. Teres minor injury related to vaccine administration. Radiol Case Rep. 2020;15(5):552-5. ↩
- Okur G, Chaney KA, Lomasney LM. Magnetic resonance imaging of abnormal shoulder pain following influenza vaccination. Skeletal Radiology. 2014;43:1325-31. ↩
- Salmon JH, Geoffroy M, Ohl X. Bone erosion and subacromial bursitus caused by diptheria-tetanus-poliomyelitis vaccine. Vaccine. 2015;33:6152-5. ↩
- Shahbaz M, Blanc PD, Domeracki SJ, Guntur S. Shoulder Injury Related to Vaccine Administration (SIRVA): An Occupational Case Report. Workplace Health Saf. 2019;67(10):501-5. ↩
- Wright A, Patel R, Motamedi D. Influenza Vaccine-related Subacromial/Subdeltoid Bursitis: A Case Report. J Radiol Case Rep. 2019;13(6):24-31. ↩
- Gaine WJ. No-fault compensation systems. BMJ. 2003;326(7397):997-8. ↩
- Rotator cuff tendinopathy, adhesive capsulitus, and arthritis can not be caused by vaccine administration.: American Academy of orthopaedic Surgeons; 2017. ↩
- Register F. National Vaccine Injury Compensation Program: Revisions to the Vaccine Injury Table. 2020. p. 43794-805 ↩
- CDC. Vaccine Administration: Needle Gauge and Length 2020 (Available from: Advisory Committee on Immunization Practices General Best Practice Guidelines for Immunization. www.cdc.gov/vaccines/hcp/acip-recs/general-recs/administration.html.) ↩
- Cook IF. An evidence based protocol for the prevention of upper arm injury related to vaccine administration (UAIRVA). Hum Vaccin. 2011;7(8):845-8. ↩
- Cook IF. Best vaccination practice and medically attended injection site events following deltoid intramuscular injection. Hum Vaccin Immunother. 2015;11(5):1184-91 ↩
- Hesse EM, Atanasoff S, Hibbs BF, Adegoke OJ, Ng C, Marquez P, et al. Shoulder Injury Related to Vaccine Administration (SIRVA): Petitioner claims to the National Vaccine Injury Compensation Program, 2010-2016. Vaccine. 2020;38(5):1076-83. ↩
- Atanasoff S, Ryan T, Lightfoot R, Johann-Liang R. Shoulder injury related to vaccine administration (SIRVA). Vaccine. 2010;28(51):8049-52. ↩
- Macomb CV, Evans MO, Dockstader JE, Montgomery JR, Beakes DE. Treating SIRVA Early With Corticosteroid Injections: A Case Series. Mil Med. 2020;185(1-2):e298-e300. ↩
- Shimabukuro T. Reports of shoulder dysfunction following inactivated influenza vaccine in the Vaccine Adverse Event Reporting System (VAERS), 2010–2016. . 2017. ↩