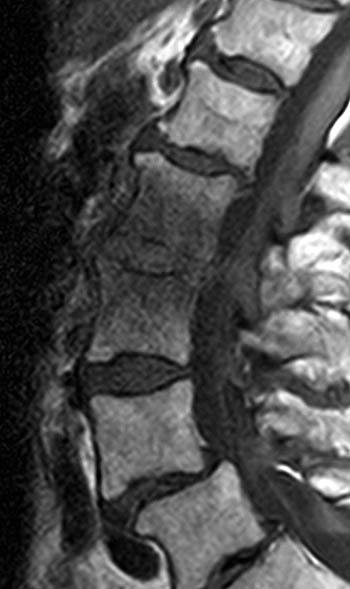
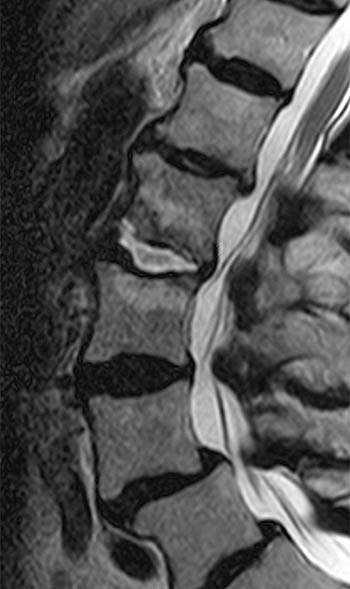
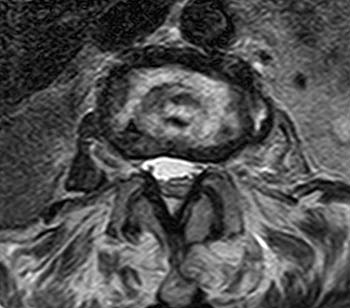
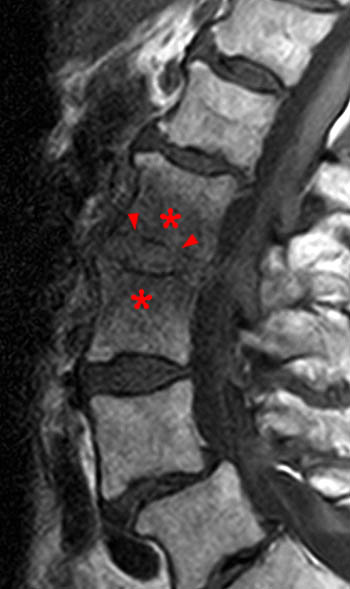
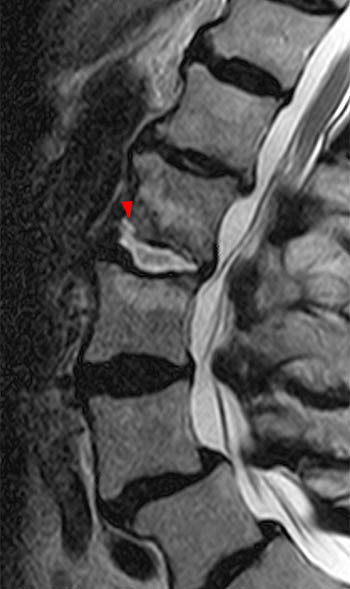
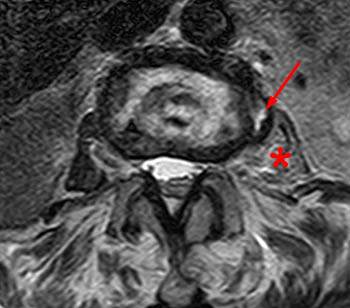
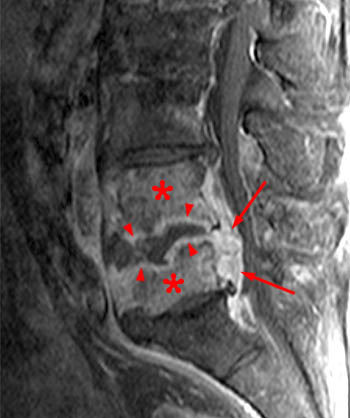
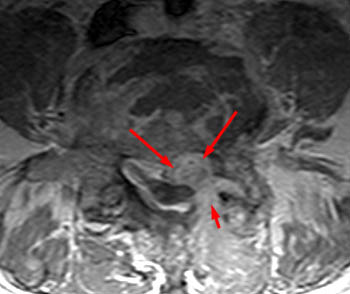
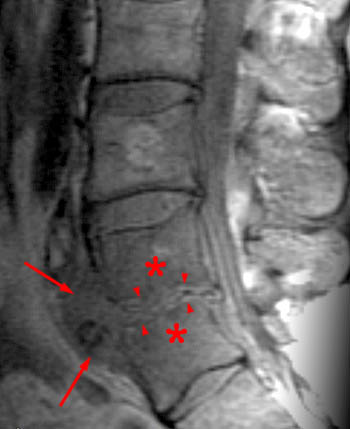
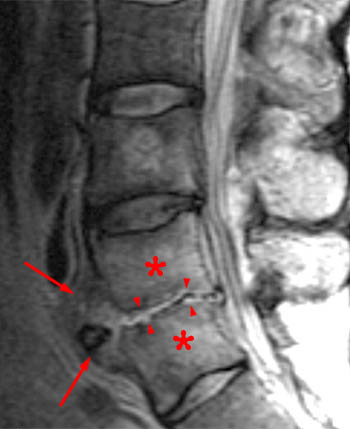
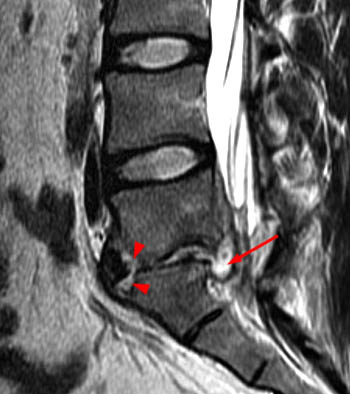
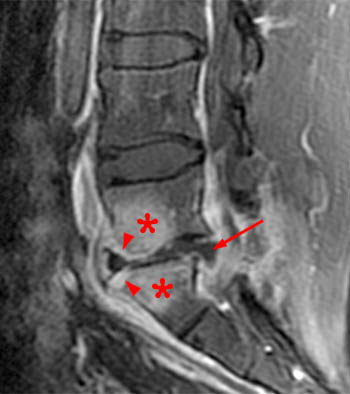
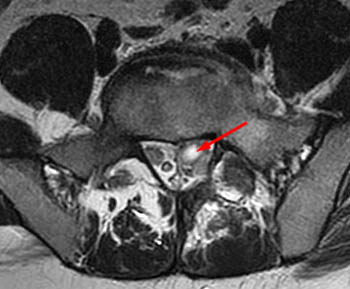
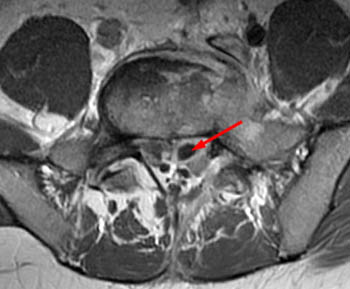
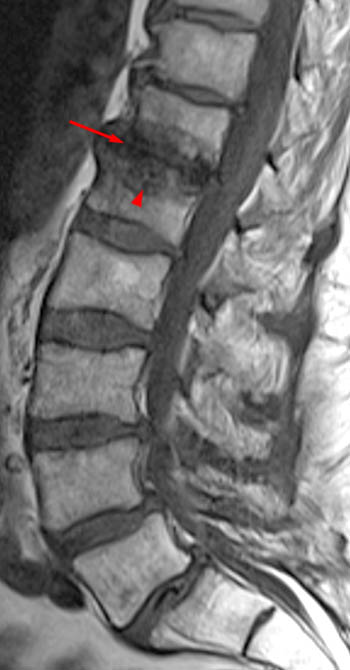
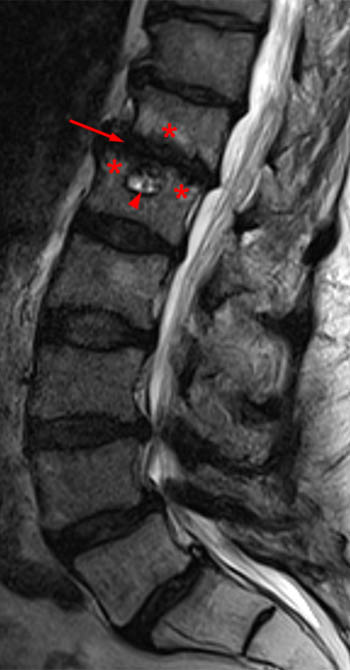
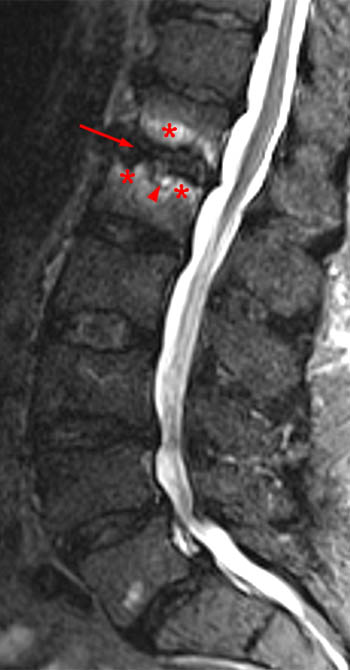
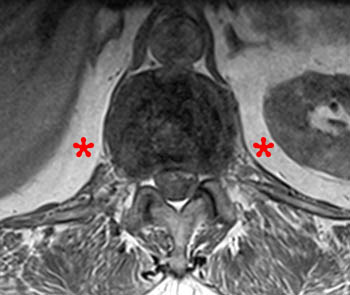
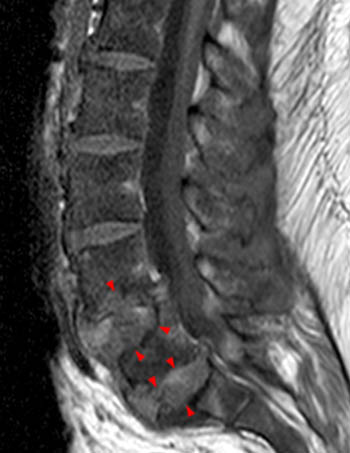
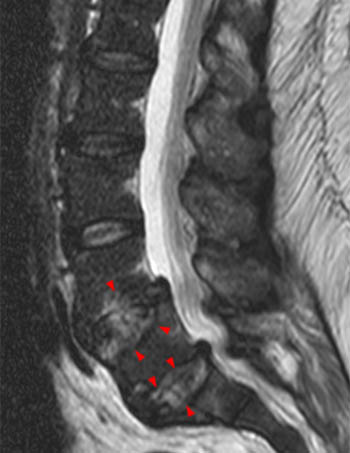
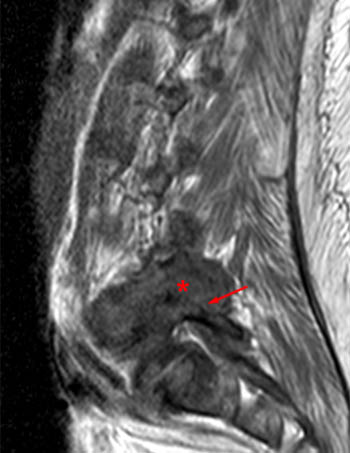
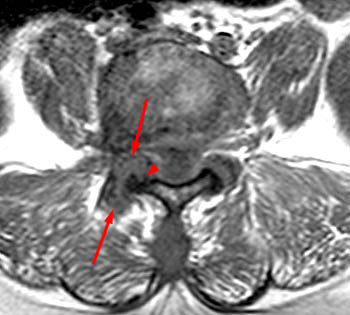
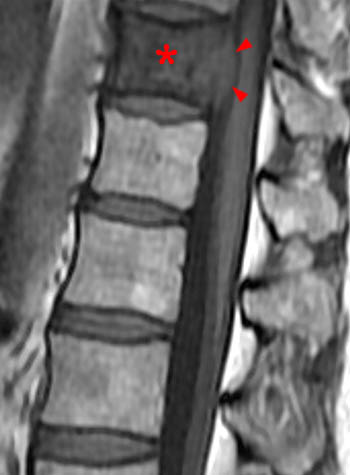
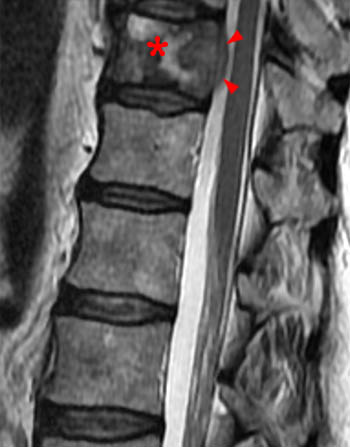
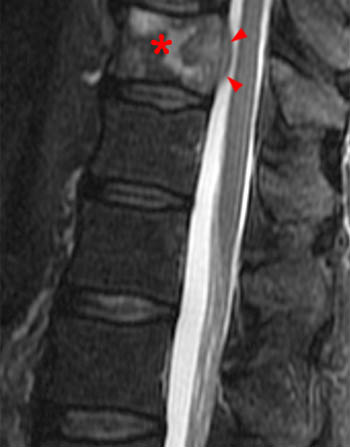
Clinical history: A 91 year-old woman complains of low back pain. A routine lumbar spine MR with (1a) T1-weighted sagittal and (1b,1c) T2-weighted sagittal and axial images was performed. What are the findings? What is your diagnosis?
Findings
Diagnosis
Spontaneous pyogenic discitis.
Introduction
Discitis, infection of the disc and vertebral endplates, has significant morbidity and mortality. Although early diagnosis and treatment leads to a better outcome, a delay in diagnosis is common due to nonspecific and inconsistent clinical signs and symptoms. A variety of imaging modalities may be used to evaluate suspected discitis with varying degrees of effectiveness.
Epidemiology
Discitis exhibits a bimodal age distribution, with peaks in early childhood and after age 50. A male predominance is seen. Risk factors for discitis include diabetes, old age, immunosuppression, IV drug use, alcoholism, and renal failure. Although rare, there is an increased risk of discitis following invasive spinal procedures, estimated at 0.5% for anterior cervical discectomy and 0.25% for lumbar discectomy, with an overall rate of 0.1 to 4% of all invasive spinal procedures. Postoperative discitis accounts for approximately 20-30% of cases of discitis.1,2
Pathophysiology
Most cases of discitis are secondary to infection elsewhere in the body, with the most common source being the genitourinary tract; other less common sources include endocarditis, pneumonia, or the oral cavity.1,3 Contrary to its name, due to the vascular anatomy of adults, discitis is a primary infection of the vertebral endplate with secondary extension into the disc. The metaphyseal arteries of the endplate are the target for septic emboli, which cause bone infarction and secondary infection. The vascular supply of the disc ends in the annulus fibrosis; the disc is largely supplied with nutrients via direct diffusion from the vertebral endplate. Infection thus spreads contiguously to the disc, spinal canal, and paraspinous soft tissues.1,2 Another postulated source of blood-borne pathogens is the epidural venous plexus; retrograde flow from the pelvis may allow the spread of infection from pelvic disease, and the pelvis is in fact the most common source of infection in discitis. However, this route of spread is not universally accepted, as pressures required to generate retrograde flow in animal models are not physiologic.2
In children, the pathophysiology of discitis differs due to differences in arterial anatomy; nutrient vessels penetrate into the nucleus pulposus, allowing direct infection of the disc.1 Childhood discitis is also less likely to be associated with a systemic infection.2
The most common organism in both adults and children is Staphylococcus aureus. Less commonly seen are Pseudomonas aeruginosa in IV drug abusers, and mycobacterial infection in immunocompromised patients.2 Blood cultures are only positive in 50% of patients, and biopsy is often necessary to isolate the causative organism.1
Clinical Presentation and Laboratory Studies
The signs and symptoms of discitis are nonspecific, commonly leading to a delay in diagnosis of 2 to 6 months on average after onset of symptoms. Over 90% of patients with bacterial discitis complain of back pain that is not relieved by rest or common analgesics. Fever is not a consistent feature of discitis, seen in only 60-70% of patients. Other non-specific symptoms such as weight loss and anorexia may be present. Localized tenderness to palpation, muscle spasm, and worsening of symptoms with movement are common. A neurologic deficit is uncommon, but when present should raise suspicion for a complicating epidural abscess. Epidural abscess is more common in chronically ill patients who are unfortunately unlikely to exhibit constitutional symptoms usually associated with abscess, such as fever and chills.1,2,3
The most common laboratory abnormalities in patients with discitis are an elevated erythrocyte sedimentation rate (ESR) and elevated levels of C-reactive protein (CRP), seen in over 90% of patients. Leukocytosis is not consistently present, occurring in less than 50% of patients; therefore a normal white blood cell count does not exclude discitis.1
In the postoperative patient, symptoms usually begin days to weeks after surgery and are similar to spontaneous discitis. As the ESR and CRP are normally elevated in the first few weeks post-operatively, laboratory studies are not reliable indicators of discitis in these patients.3 Delayed onset of discitis up to 7 years after surgery has been reported.4
Figure 5:
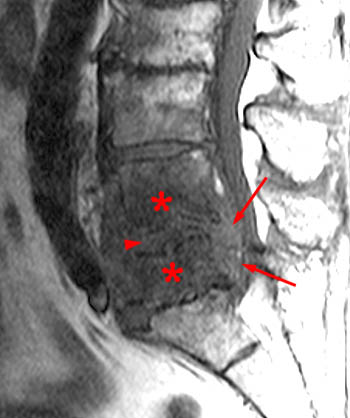
A 65 year-old male with history of L5 laminotomy several months prior, now with recurrent pain. A T1-weighted sagittal image demonstrates marked hypointensity of the L4-5 disc(arrowhead) and adjacent vertebral bodies(asterisks) with irregular, poorly defined endplates and a ventral epidural soft tissue process (arrows).
Figure 7:
T1-weighted sagittal (7a) and axial (7b) images after IV gadolinium administration demonstrate peripheral enhancement of the disc (arrowheads) and marked vertebral enhancement (asterisks). A moderate amount of uniformly enhancing soft tissue is present in the ventral spinal canal at the L4-5 level (arrows), suggesting epidural inflammatory changes. The recent left L5 laminotomy defect is demonstrated (short arrow).
The clinical presentation of discitis in children differs from that of adults and is characterized by fever, sudden onset of back pain, refusal to flex the lumbar spine, irritability, and refusal to walk.1,2
Imaging of discitis
Discitis is most commonly seen in the lumbar spine, followed by the cervical spine, and least commonly involves the thoracic spine (except for in atypical cases, such as those caused by tuberculous involvement).2 Several imaging modalities may be used in the assessment of discitis.
Plain films
Findings on plain radiographs usually require 10-14 days to develop and lag behind clinical symptoms. As a result, a negative plain film does not exclude early discitis. Eventual changes include loss of definition of the end plates and decrease in disc space height, followed by lucency of the vertebral body, loss of trabeculations, and finally frank bone destruction. Progressive kyphosis or scoliosis can develop in cases of chronic infection. Sclerosis is seen after healing with a high incidence of fusion of the disc space.2,3
Nuclear medicine studies
Gallium scans and technetium-99M scans are highly sensitive for the detection of discitis, with positive scans seen in 90% of patients that have been symptomatic for longer than 2 days. The increased uptake in the affected endplates and decrease in disc space height produces a characteristic “sandwich” appearance. However, because of the poor anatomic resolution of nuclear medicine studies and the advent of more sensitive and specific imaging modalities, these studies are usually utilized in cases of fever of unknown origin or, in the case of gallium scan, as part of treatment follow-up. Technetium-99m scans remain positive long after healing has occurred, and are thus not useful in assessing treatment efficacy.3
Computed tomography
CT scanning allows earlier detection of endplate involvement and bone destruction than plain films, and may allow identification of paraspinous inflammatory changes and psoas abscess formation, especially on contrast-enhanced scans. Post-contrast studies and sagittal and coronal reconstructed images are helpful in defining extent of disease. CT is not ideal for detection of bone marrow changes or epidural involvement.3
Magnetic resonance imaging
MR is the imaging modality of choice in suspected discitis due to its multiplanar imaging capabilities, lack of ionizing radiation, excellent anatomic resolution, and ability to assess bone marrow changes.1 It allows detection of early discitis with a reported sensitivity of 96% and a specificity of 94%. In animal studies, changes of discitis were seen as early as five days post infection.9 MR also allows accurate assessment of complications of discitis, including direct assessment of effect on neural structures. Multiplanar pre- and post-gadolinium T1- and T2-weighted images should be performed, including a STIR sequence which helps distinguish between normal and edematous marrow.6
Figure 9:
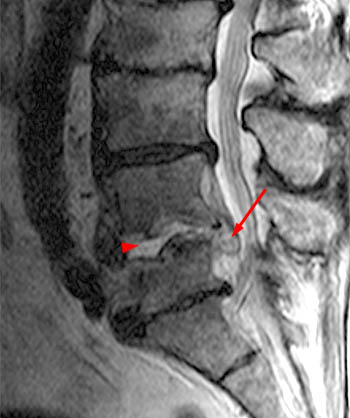
A 48 year-old male with lumbar pain, tenderness, and positive straight leg raise sign. A T1-weighted sagittal image reveals hypointensity of the L4 and L5 vertebral bodies (asterisks) with loss of disc space height at L4-5 and poor definition of the endplates (arrowheads). A contiguous soft tissue inflammatory mass is present at the anterior disc margin (arrows).
Figure 11:
The T2-weighted axial image demonstrates a large, irregular fluid collection along the left pelvic sidewall (arrowheads) extending into the left sacroiliac joint (arrow), with abnormal hyperintensity of the left iliac bone and left sacral ala (asterisks). Diagnosis: L4-5 discitis with secondary infection of the left sacroiliac joint and associated pelvic sidewall abscess.
In acute discitis, hypointensity of the disc and destruction of the endplate are often seen on T1-weighted imaging, due to inflammation and necrosis replacing the normal fatty marrow. However, these findings are not consistent. A phenomenon known as “pseudosparing” of the endplate has been described in up to 71% of patients, caused by loss of the normal chemical shift phenomenon seen with fatty marrow that typically causes the superior endplate to appear thin and the inferior endplate to appear thick. When the normal marrow is lost in discitis, the decrease in the chemical shift artifact in the inferior endplate allows visualization of the actual endplate thickness, masking endplate thinning and erosion.5
The “nuclear cleft sign” is another traditionally described MRI finding in discitis. However, Ledermann et al. found that loss of the nuclear cleft was not consistently seen in affected discs and was not a reliable indicator of the presence or absence of discitis; in fact, in many patients, the nuclear cleft could not be identified in unaffected discs.6 Loss of disc space height is another common finding in discitis that is nonspecific and found in many other conditions.
The most reliable MRI findings in discitis are hyperintensity of the disc on T2-weighted imaging (sensitivity 93%); the presence of paraspinous or epidural inflammation/abscess, (sensitivity 98%); and contrast enhancement of the disc and adjacent bone marrow (sensitivity 95%). Vacuum disc phenomenon is rarely seen in discitis and is a negative predictor, usually indicating a degenerative etiology.6
MRI is also superior to all other imaging modalities in the assessment of epidural disease, either phlegmon or abscess. Post-gadolinium images are excellent in assessing the extent of epidural involvement as well as the degree of mass effect upon adjacent structures. Uniformly enhancing soft tissue is consistent with phlegmon, while peripheral enhancement is highly suggestive of abscess. Epidural disease typically involves between two and four vertebrae and is ventral in location. MRI is also excellent in evaluating soft tissue involvement of discitis, such as paraspinous inflammation and psoas abscess.3,6
Figure 12:
A 28 year-old male three months status post L5-S1 microdiscectomy, now with progressive back pain. The T2-weighted sagittal image reveals focal edema and irregularity of the endplates at L5-S1 (arrowheads) with areas of hyperintensity in the disc and a small contiguous fluid collection in the ventral epidural space (arrow). These findings have developed in the interim since a previous MRI two months prior.
Diffusion-weighted MRI has been advocated as a method of distinguishing discitis from degenerative endplate changes. Restricted diffusion has been shown to be present in the endplates of patients with infection, but not in those with degenerative disease. Diffusion sequences are not routinely performed in the spine, but can be useful in cases where there is a specific question regarding degeneration versus infection.7
Tuberculous discitis
Tuberculous discitis is often seen in immunocompromised patients or IV drug abusers and can have a more indolent clinical course than the more commonly seen pyogenic discitis, with a subsequent longer delay in diagnosis of up to 6-8 months.1 Tuberculous discitis is more common in the thoracic spine due to spread from mediastinal lymph nodes. On imaging studies, the typical MRI signal intensity changes in the bone marrow may be absent, and bony destruction can be pronounced with relative sparing of the disc space. TB can also solely involve the posterior elements or only a portion of the vertebral body. Skip lesions, extensive paraspinous inflammation, and abscess formation are common.1,6
Differential diagnosis
Several other conditions can mimic the appearance of discitis on various imaging modalities.
Degenerative disc disease in the early, inflammatory stage can mimic discitis. Modic Type 1 changes in the endplates due to disruption of the endplate and vascularized fibrous tissue in the adjacent marrow cause hyperintensity in the endplate on T2-weighted images and enhancement after the administration of gadolinium. However, lack of disc hyperintensity on T2-weighted images and the absence of paraspinous inflammation indicate a degenerative etiology.3,10 In addition, while endplate irregularities are common in degenerative disease, actual loss of endplate definition or endplate destruction are not characteristics of Modic Type 1 changes.
Figure 15:
A 62 year-old male with back pain radiating to both legs. A T1-weighted sagittal image shows loss of disc space height at T12-L1 (arrow) with marked hypointensity of the endplates. Mild endplate irregularity is present and a Schmorl's node is seen within the superior endplate of L1 (arrowhead).
Figure 17:
The T1 weighted axial image shows well-defined paraspinous fat planes at the level of T12-L1 (asterisks). Diagnosis: Modic Type 1 degenerative endplate changes. The lack of hyperintensity of the disc on T2 weighted images, the clearly defined endplates, and the lack of paraspinous inflammation make infection highly unlikely.
Dialysis-related spondyloarthropathy is most likely related to amyloid deposition in the disc and ligamentum flavum. On plain films and CT, destruction of the end plates and severe disc space narrowing can mimic infectious discitis. However, on MRI, the T2 hyperintensity of the disc characteristic of discitis is not present. In addition, dialysis-related disease often involves the posterior elements, a rare finding in discitis. Unlike discitis, the most commonly involved area is the lower cervical spine.8 Neurogenic spondyloarthropathy caused by paralysis can cause changes similar to dialysis-related spondyloarthropathy on plain film and CT, but also fails to show disc hyperintensity on T2-weighted images.3,12
Figure 18:
A 56 year-old female with chronic back pain radiating into right hip; the patient is on dialysis for renal failure. T1- (18a) and T2- (18b) weighted sagittal images show endplate destruction at the L4-5 and L5-S1 levels (arrowheads). Of note, the discs remain relatively isointense to the unaffected discs on T2 weighted images. Marrow signal intensity is markedly decreased in all vertebral bodies, consistent with the history of renal failure.
Metastatic involvement of the spine can cause destructive changes similar to those seen in discitis. However, the disc space is rarely involved by neoplasm, and paraspinous fat planes are usually preserved. Vertebral lesions are usually hypointense to CSF on T2-weighted images, as opposed to infectious involvement, which is generally hyperintense.11 Involvement of contiguous vertebral bodies is more typical of infection.3
Figure 21:
A 67 year-old male with lower back pain radiating into the right leg. (21a) T1- and (21b) T2- weighted sagittal images and a (21c) fat saturated T2-weighted sagittal image reveal heterogeneous, abnormal signal in the T11 vertebral body (asterisk) with loss of vertebral body height and posterior epidural extension (arrowheads). The vertebral body is predominantly hypointense to CSF on T2 weighed images. The endplates are intact, and the adjacent discs are spared. Diagnosis: Metastatic disease involving the T11 vertebrae with pathologic fracture.
SAPHO syndrome (synovitis, acne, pustulosis, hyperostosis, and osteitis) can cause abnormal marrow signal intensity, paraspinous soft tissue swelling, end plate irregularity and disc space narrowing, disc hyperintensity on T2-weighted images, and disc enhancement, and can be difficult to differentiate from pyogenic discitis. The multiplicity of foci and lack of abscess formation in SAPHO syndrome can be helpful in distinguishing between the two entities.3,13,14
Treatment and follow-up
Non-operative treatment is effective in the majority of patients (up to 90%) and consists of 4-8 weeks of parenteral antibiotics and immobilization. Percutaneous CT-guided drainage of paraspinous abscesses larger than 2 cm may also be performed. Surgery is indicated in cases of spinal cord compression, instability, correction of mechanical deformity, abscess, or severe persistent pain. In cases where instability is present, the placement of fixation devices has not been shown to impede healing.1,2,3
Although MRI is frequently used to follow patients during treatment, it has not been shown to be an effective modality for assessing response to treatment. Although paraspinous inflammation and epidural enhancement improves in most patients, other findings such as disc space enhancement, marrow edema, and vertebral body enhancement can persist and actually worsen despite a favorable clinical response. In addition, post-gadolinium enhancement can persist for longer than 4 months; this may be due to development of hypervascular fibrous scar tissue during the healing phase. For these reasons, the use of MRI in routine follow-up of patients with discitis is not recommended.1,16
Monitoring of clinical symptoms and laboratory studies is the most effective method of assessing treatment. Reduction in back pain and decreased constitutional symptoms are expected. A decrease in serum ESR and CRP indicates a successful response, with CRP being the more sensitive of the two; ESR levels should fall by one third to one half, but frequently never return to pre-disease levels. Serial plain films may also be performed to determine whether bony deformity or collapse has developed. These parameters are usually followed during treatment and for one year after its completion.1,2
Morbidity and mortality
The overall mortality rate of discitis is between 2 and 11%. The presence of an epidural abscess increases mortality to 10% or greater. Up to 16% of patients with spontaneous discitis experience a recurrence, usually within 6 months of cessation of antibiotic treatment. Recurrence in cases of discitis complicating spine surgery is less common, ranging from 0 to 4%; this may be in part due to increased vigilance in the postoperative period leading to earlier diagnosis of infection.1,2,17
The morbidity of discitis is significant. Approximately one third of patients have residual disability. 15% of patients have permanent neurologic deficits. Only 45% of patients with postoperative discitis are able to return to normal daily activities.1,2,17
Early diagnosis and treatment of discitis and completion of antibiotic regimen is correlated with a better prognosis; a delay in diagnosis leads to poorer outcomes.1,2,17
Conclusion
Discitis commonly presents with nonspecific signs and symptoms and has significant morbidity and mortality, but prognosis is improved by early diagnosis. MRI is the imaging study of choice in evaluating possible discitis due to its high sensitivity and ability to define associated complications, such as epidural and paraspinous abscess, and should be performed if discitis is suspected even if plain films and CT are non-diagnostic. MRI can also distinguish between discitis and other conditions with similar presentations, allowing the clinician to make informed treatment decisions. However, because MRI findings do not correlate well with treatment response, follow up with serial plain films and monitoring of laboratory studies is recommended.
References
1 Cottle L, Riordan T. Infectious Spondylodiscitis. J Infect, Jun 2008; 56(6):401-12.
2 Jallo GI, Keenan MA. Diskitis. Medline, Feb 2011.
3 Varma R, Lander P, Assaf A. Imaging of Pyogenic Infectious Spondylodiskitis. Radiol Clin of N Amer, March 2001; 39(2):203-213.
4 Viola RW, King HA, Adler SM, et al. Delayed Infection After Elective Spinal Instrumentation and Fusion: A Retrospective Analysis of Eight Cases. Spine, 1997; 22:2444-2450.
5 Wolansky LJ, Heary RF, Patterson T, Friedenberg JS, Tholany J, et al. Pseudosparing of the Endplate: A Potential Pitfall in Using MR Imaging to Diagnose Infectious Spondylitis. AJR, 1999; 172:777-780.
6 Ledermann HP, Schweitzer ME, Morrison WB, Carrino JA. MR Imaging Findings in Spinal Infections: Rules or Myths? Radiology, 2003; 228:506-514.
7 Eguchi Y, Ohtori S, Yamashita M, Yamaguchi K, Suzuki M, Orita S, et al. Diffusion Magnetic Resonance Imaging to Differentiate Degenerative from Infectious Endplate Abnormalities in the Lumbar Spine. Spine, Feb 2011; 36(3):E198-202.
8 Kiss E, Keutch G, Zanetti M, Jung T, Schwartz A, Schocke M, Jaschke W, Czermak BV. Dialysis-Related Amyloid Revisited. AJR, 2005; 185:1460-1467.
9 Taylor DJ, Brown GC, Moore RJ, Walters R, Chapple DC, Goss DL, Fraser RJ. Early Detection of Lumbar Discitis by MRI. Proc Intl Soc Mag Reson Med, 2001; 9:249.
10 Modic MT, Ross JS. Lumbar Degenerative Disc Disease. Radiology, October 2007; 245(1):43-57.
11 Hovi I, Lamminen A, Salomen O, et al. MR Imaging of the Lower Spine. Acta Radiol, 1994;35:532-540.
12 Park YH, Taylor JA, Szollar SM, et al. Imaging Findings in Spinal Neuroarthropathy. Spine, 1994; 19:1499-1504.
13 Nachtigal A, Cardinal E, Bureau NJ, et al. Vertebral Involvement in SAPHO Syndrome: MRI Findings. Skeletal Radiol, 1999; 28:163-168.
14 Toussirot E, Dupond JL, Wendling D. Spondylodiscitis in SAPHO Syndrome: A Series of Eight Cases. Ann Rheum Dis, 1997; 56:52-58.
15 Karadimas EJ, Bunger C, Lindblad BE, Hansen ES, Hoy K, et al. Spondylodiscitis: A Retrospective Study of 163 Patients. Acta Orthop, Oct 2008; 79(5):650-659.
16 Kowalski, TJ, Layton, KF, Berbari, EF, Steckelberg, JM, Huddleston PM, et al. Follow-up MRI Imaging in Patients with Pyogenic Spine Infections: Lack of Correlation with Clinical Features. Am J Neuroradiol, Apr 2007; 28:693-699.
17 Jimenez-Mejias ME, de Dios Colmenero J, Sanchez-Lora FJ, Palomino-Nicas J, et al. Postoperative Spondylodiskitis: Etiology, Clinical Findings, Prognosis, and Comparison with Non-Operative Pyogenic Spondylodiskitis. Clin Inf Dis, 1999; 29:339-345.