Clinical History
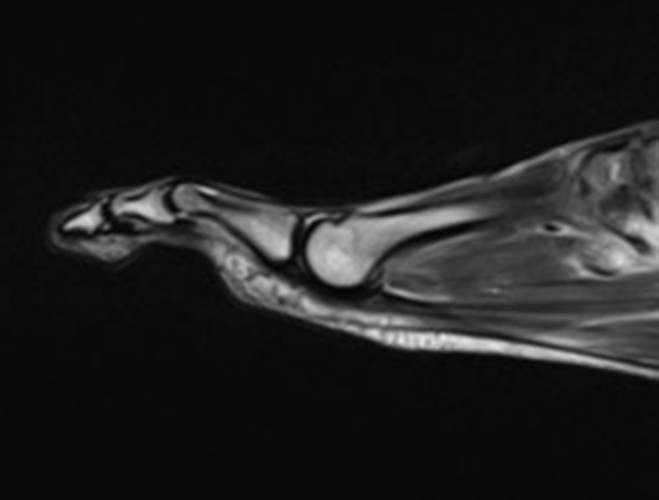
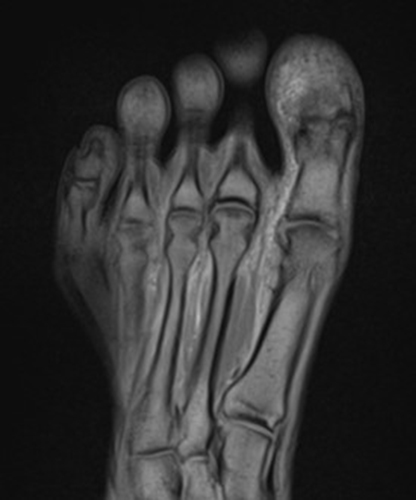
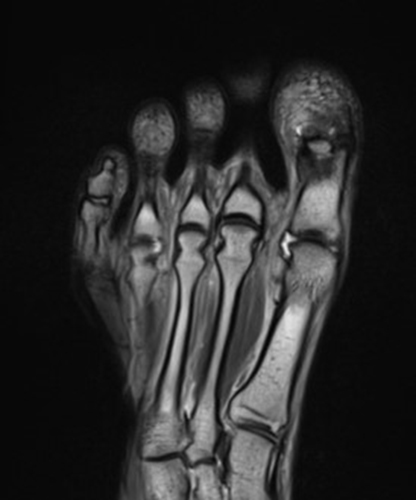
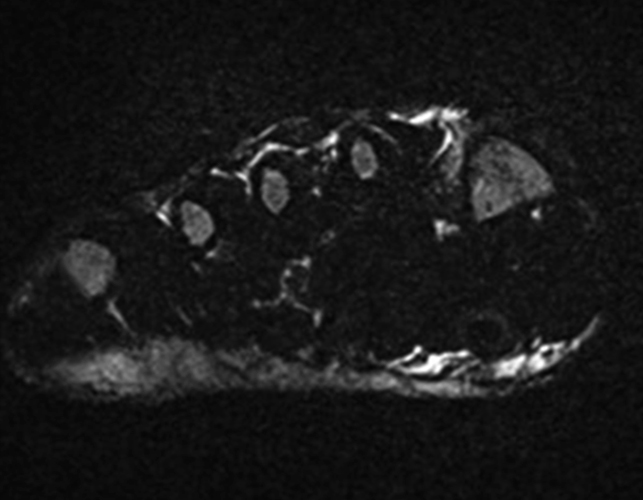
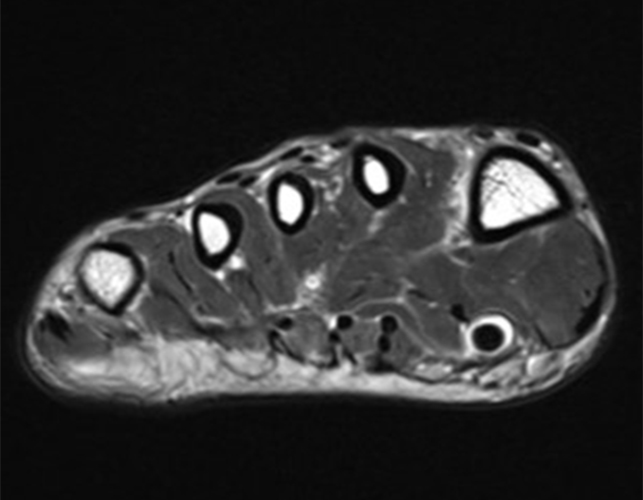
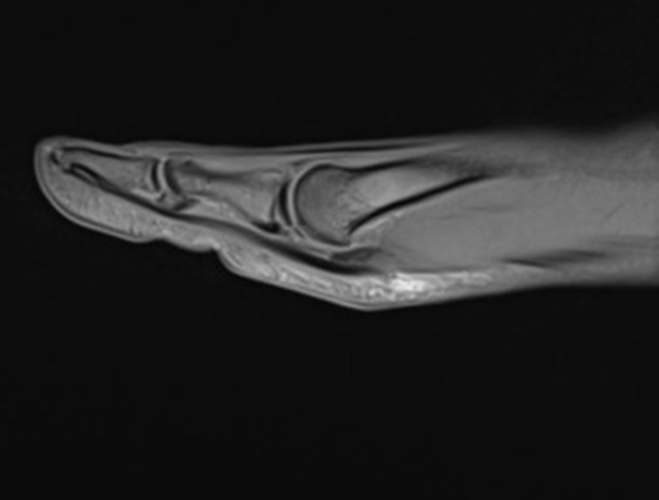
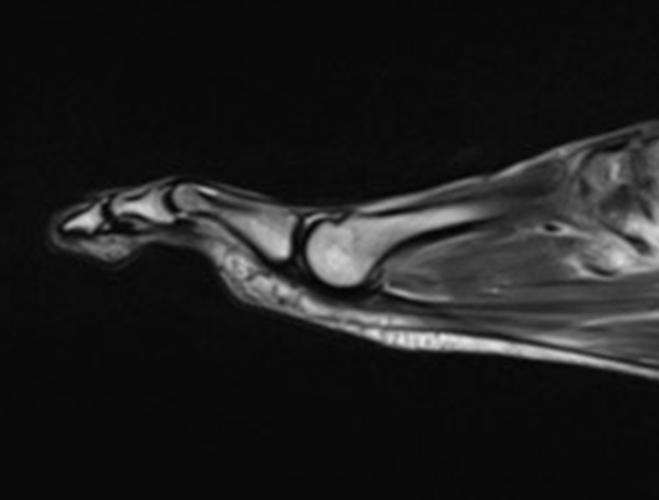
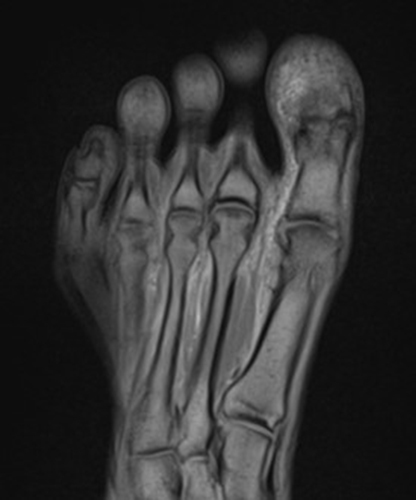
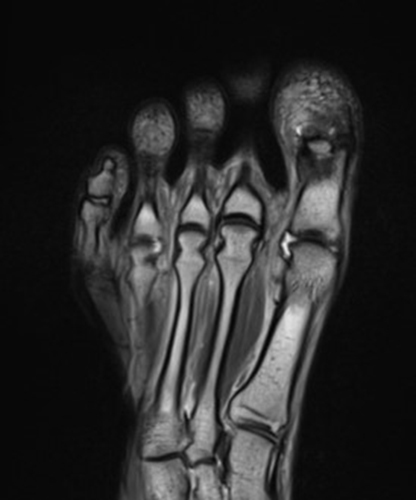
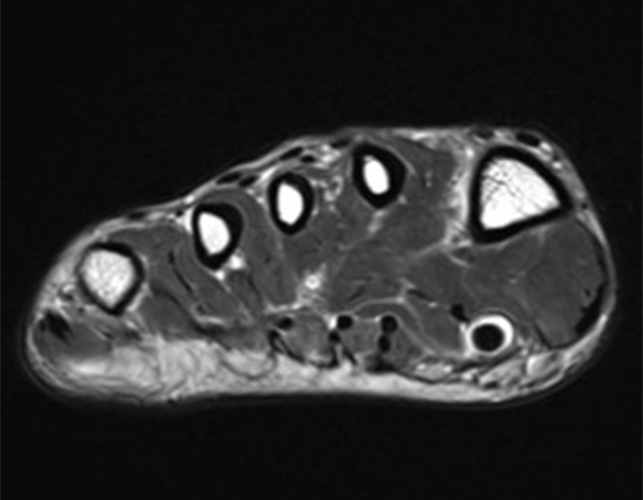
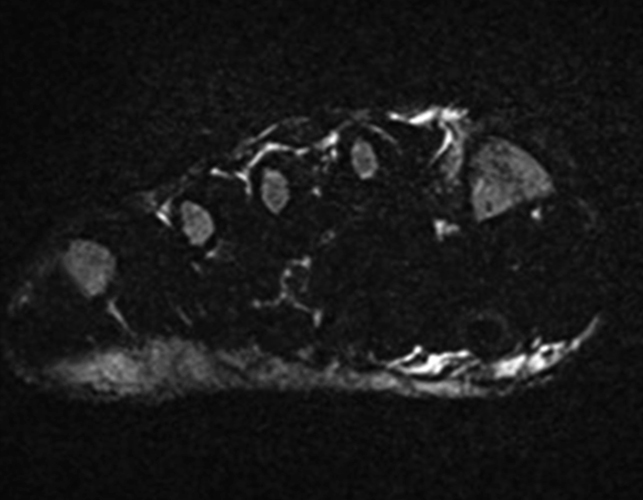
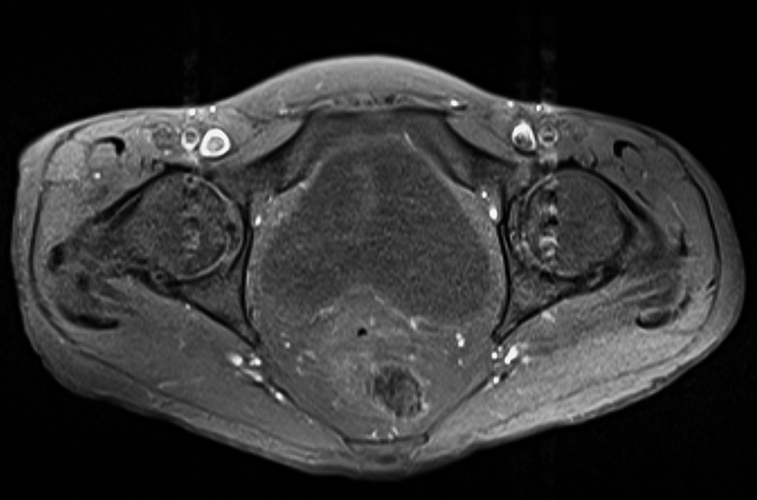
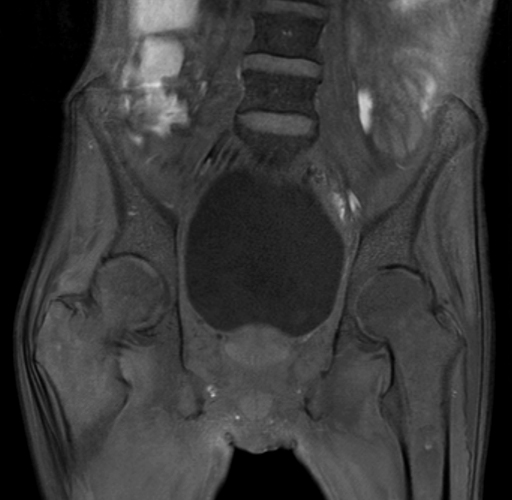
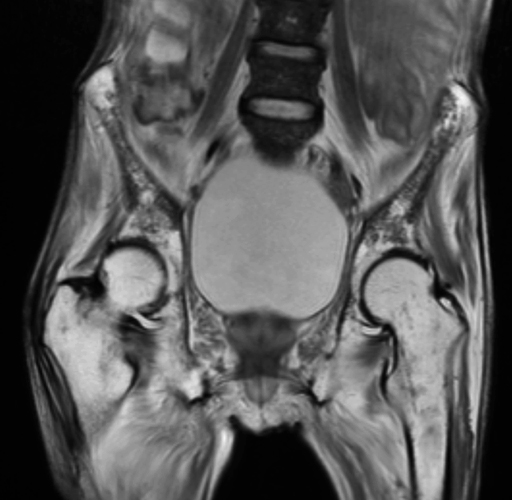
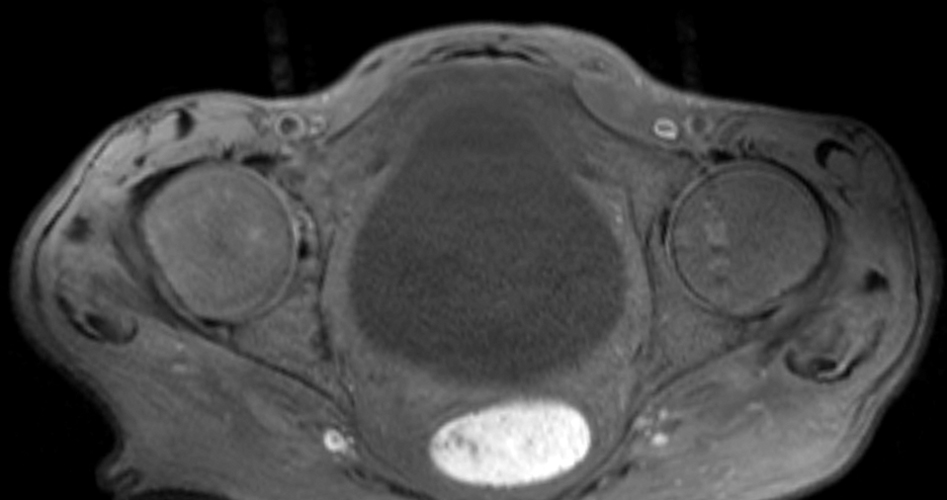
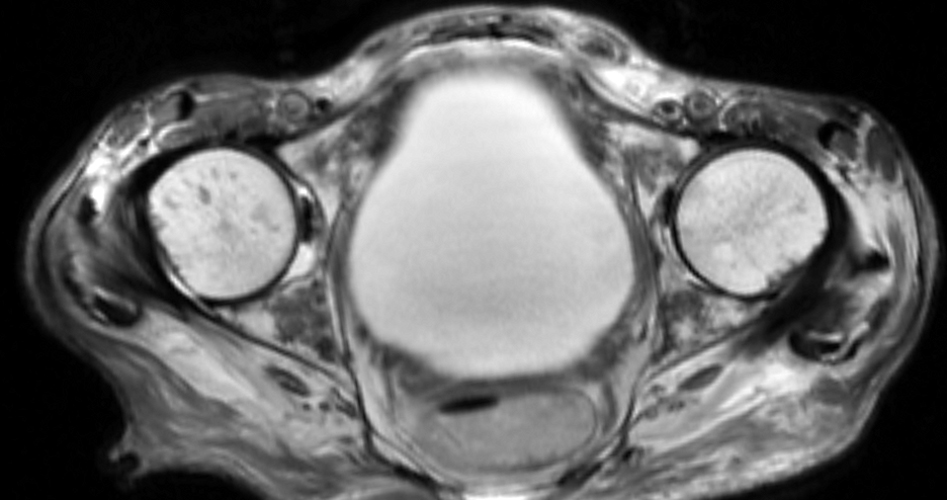
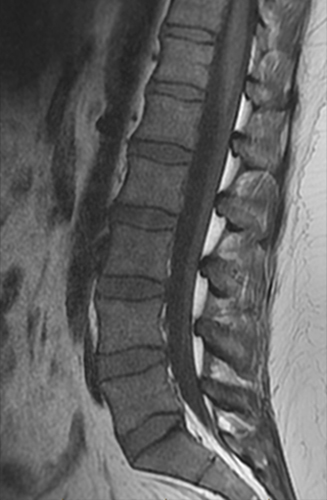
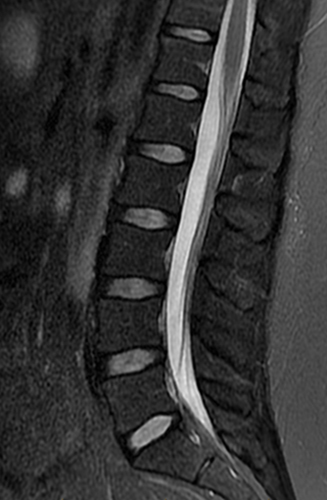
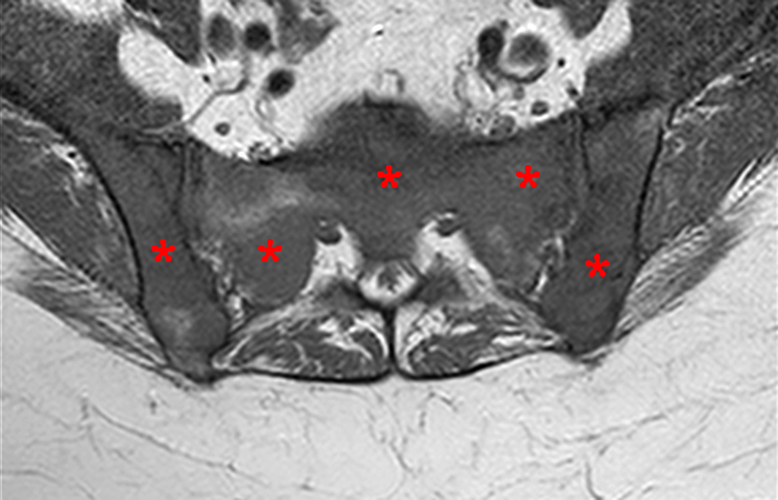
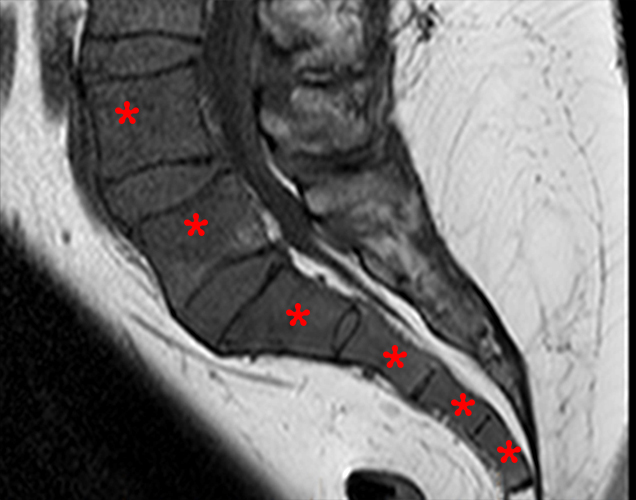
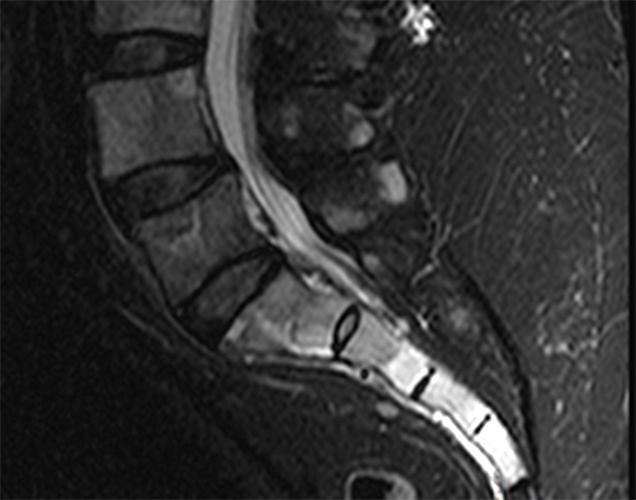
A 22-year-old female runner presents with worsening right foot pain with running and prolonged weight-bearing. Sagittal T1-weighted (1a) and STIR (1b), axial T1-weighted (1c) and fat-suppressed T2-weighted (1d), as well as coronal T2 Dixon Fat (1e) and T2 Dixon Water (1f) images are provided. What are the findings? What is your diagnosis?
Findings
Figure 2:
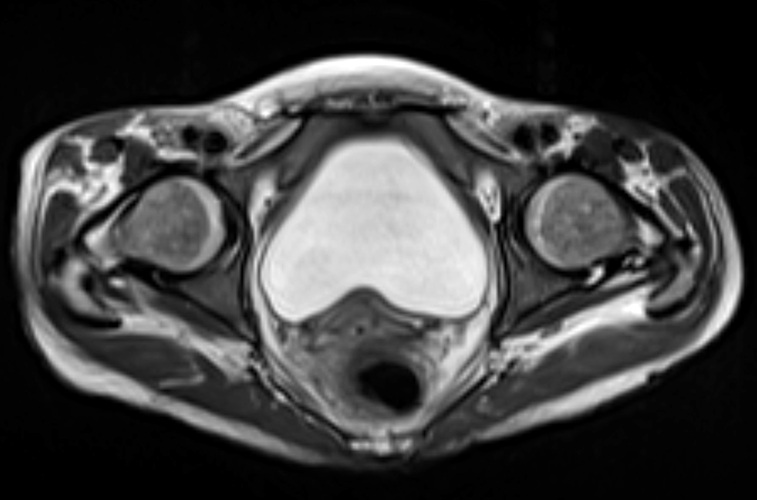
All sequences demonstrate diffusely abnormal signal within the bone marrow and subcutaneous tissues. The marrow and subcutaneous tissues are homogeneously intermediate to mildly hypo-intense in signal on T1-weighted images (2a and c), and homogeneously hyper-intense on STIR (2b) and T2-weighted fat-suppressed (2d) images. Of note, the MRI technologist added coronal T2 Dixon sequences (2e and f) for further evaluation, fearing a machine related improper fat saturation on the routine sequences. The Dixon images demonstrate intermediate signal in the marrow and subcutaneous tissues on the fat image (2e) and diffuse hyperintensity on the water image (2f). A fracture line is not identified.
Diagnosis
Serous atrophy of bone marrow, also known as gelatinous transformation of bone marrow or starvation marrow.
Introduction
Serous atrophy of bone marrow is a rare, incompletely understood, and likely underdiagnosed non-neoplastic bone marrow disorder that is associated with multiple underlying illnesses and especially with malnutrition states.1 It has been reported in anorexia nervosa, malignancy, chronic infections such as AIDS and tuberculosis, gastrointestinal disorders including malabsorption states, chronic heart failure and kidney disease, alcoholism as well as with the use of cytotoxic medications.1,2,3,4,5 Serous atrophy is not considered a specific disease, but a sign or complication of an underlying generalized serve illness and associated with weight loss and anemia.1,2,6 It is usually an incidental finding in the workup for pain and stress fractures or infection.1 Findings can be reversible with treatment of the underlying cause and with proper nutritional replenishment.7
Clinical Presentation
In the vast majority of cases, serous atrophy of bone marrow is not diagnosed or suspected prior to imaging, which is why it is important for the radiologist to be familiar with this entity. In a study of 30 patients with serous atrophy of bone marrow by Boutin et al., the most common indication for MR imaging was pain, followed by soft tissue ulcer evaluation, with the most commonly found MRI complication being fracture followed by infection.1
Serous atrophy of bone marrow is predominantly seen in adults. In a study of 155 cases of serous atrophy of bone by Bohm, all patients were adults, although pediatric cases were reported by Boutin et al., and Jain et al.1,2,3 In Bohm’s study, there was almost 2:1 male predilection, and the incidence as well as severity were the highest in young adults.2 Anorexia nervosa, acute febrile states and AIDS were the most common underlying diseases in patients under 40 years old. Alcoholism and lymphoma were the most common in the middle age group, and carcinoma, lymphoma and congestive heart failure were the most common underlying diseases in patients over 60 years old. 78% of the patients had weight loss and 81% were anemic.2
In Boutin’s study of 30 patients, the majority of patients had anorexia followed by cachexia (malignant and non-malignant) and AIDS. Common clinical findings were weight loss and anemia.1,2,6 Complications of fracture were thought to be related to bone mineral density loss and fragility which is commonly seen with anorexia nervosa and/or bone weakening from serous atrophy of bone marrow.1,8 The increased risk of infection was thought to be related to diminished hematopoiesis.1
Pathophysiology
The exact pathophysiology of serous atrophy is not completely understood but is characterized by deposition of serous (gelatinous) material composed of hyaluronic acid rich mucopolysaccharides in the bone marrow stroma. This is associated with hematopoietic atrophy, possibly due to a non-supportive marrow micro-environment and inadequate hematopoietic substrate availability.2,6,7,9
During prolonged starvation, the body mobilizes subcutaneous and visceral fat by lipolysis. However, it has been found that during this time there is actual increase in marrow fat, possibly preferential differentiation of mesenchymal stem cells into adipocytes instead of osteoblasts, and ultimately weakening of the bone.7,8,10,11 Lipolysis within the bone marrow is also thought to not occur until all other fat deposits have been mobilized, and therefore occurs in the late phase of starvation.8 During this time, the extracellular space is filled with gelatinous material.8 Serous atrophy of the bone marrow is postulated to begin in focal areas, such as the great toe, and coalesces into a more diffuse involvement with increasing severity.1,12,13 In a study by Hwang et al., histologic analysis of a focus of bone marrow abnormality in a patient status post treatment for a malignant peripheral nerve sheath tumor in an extremity demonstrated characteristic findings of serous atrophy, and 37% percent of extremity soft tissue sarcomas post radiation and/or chemotherapy treatment demonstrated focal marrow signal abnormalities in this study.12
The pattern of bone marrow changes in serous atrophy also differentiates it from other marrow converting or replacing diseases in that with serous atrophy, marrow changes first occur in fatty marrow such as the distal extremities, while marrow conversion and marrow infiltrative processes typically first occur more proximally such as in the pelvis or axial skeleton. Proximal involvement in the pelvis or axial skeleton in serous atrophy is thought to represent more severe disease.1,13,14
Histology
Histologic analysis demonstrates focal hypoplasia of fat cells and hematopoietic cells, with accumulation of extracellular gelatinous substances consisting of mucopolysaccharides rich in hyaluronic acid. There is also positive staining with Alcian blue at pH 2.5 and Periodic Acid Schiff (PAS).2,6,7,9
MRI Findings
The MRI findings of serous atrophy of bone marrow may differ depending on the underlying cause and severity of underlying disease. It is believed that marrow changes begin focally and with increased severity of disease, become more confluent to the point of diffuse abnormality.1,12,13,14,15 On imaging, early or more focal lesions may be more difficult to diagnose due its non-specific nature.
In cases of serous atrophy of bone marrow, bone marrow signal is usually intermediate to slightly hypointense on T1 sequences, hyperintense on fluid sensitive sequences such as fat-suppressed T2-weighted or STIR series, and does not demonstrate post-contrast enhancement, which is a helpful tool to differentiate from infection or marrow infiltrative processes.1,13,14 Another helpful clue to the diagnosis is the presence of significant loss or near absence of subcutaneous and visceral fat, this again is seen as intermediate to slightly hypointense T1 signal and hyperintense signal on fluid sensitive sequences. Proposed mechanisms for signal changes in the subcutaneous tissues include replacement of fat by loose fibro-vascular tissue, change in vascular permeability and pro-inflammatory cytokines.1
Figure 3:
Axial T1-weighted (3a) and axial STIR (3b) images of young woman with end-stage gastric cancer and cachexia. Note the diffuse hypointense signal in the bone marrow and subcutaneous tissues on the axial T1-weighted images, which correlates with hyperintense signal on the STIR images, completely opposite of normal marrow signal. Images courtesy of David Rubin, MD
The combined markedly irregular signal of the bone marrow and subcutaneous or visceral fat is routinely mistaken for technical error and poor fat saturation by both technologists and radiologists. 7 of the 30 cases in Boutin’s study were initially misinterpreted as technical error and a repeat MRI was performed.1 We have seen similar situations in our practice. In general, it is suggested to image with both fat-suppressed T2 and STIR sequences to eliminate failed fat suppression as a cause of the abnormal signal.1 If an indeterminate marrow process is seen more proximally in the pelvis or axial skeleton, another proposed problem-solving tool in addition to contrast is to image the distal lower extremities to look for the more characteristic findings of marrow and subcutaneous signal changes.1,13
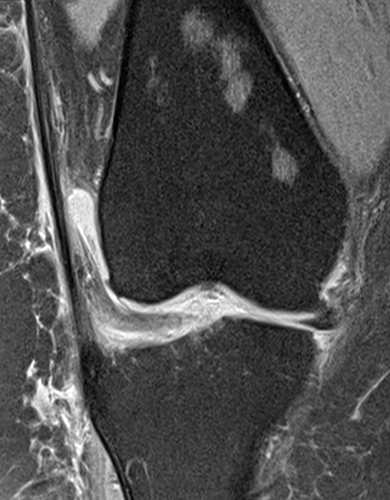
Figure 4:
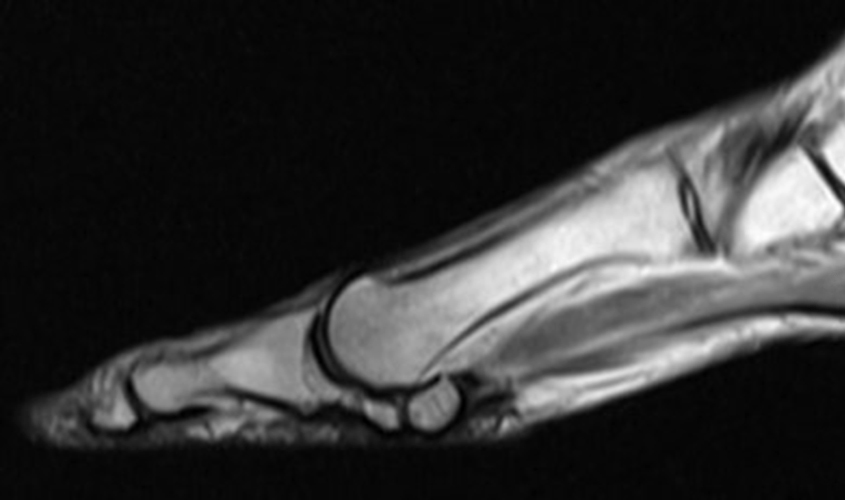
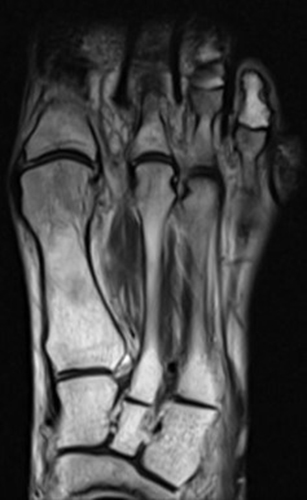
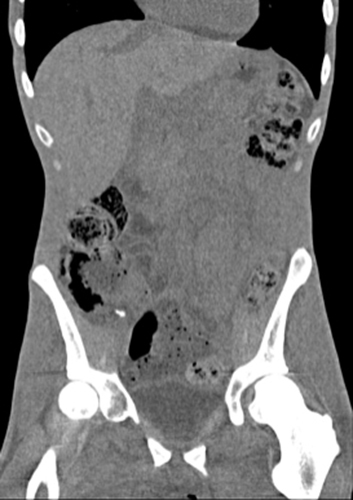
Young runner with foot pain. This case is very similar to the first example case and again demonstrates the classic appearance of serous atrophy of the bone marrow in the extremities including diffuse homogenous hyperintense bone marrow and subcutaneous fat signal on sagittal STIR (4a) and axial fat-suppressed T2-weighted (4c) images, as well as diffuse intermediate to hypointense signal on the sagittal T1-weighted image (4b). In this case the technologist called the radiologist after she had imaged the patient on two scanners and was not able to correct the “artifact”. Coronal image from a previous CT abdomen (4d) helps confirm the diagnosis and demonstrates diffuse paucity of subcutaneous and visceral fat.
Figure 5:
43-year-old male with diabetes and severe malnutrition (BMI=13). Coronal and axial T1-weighted (5a and c) and coronal STIR (5b) and axial T2-weighted fat-suppressed (5d) images demonstrate the typical appearance of serous atrophy of bone marrow with diffuse hypointense signal in the bone marrow and subcutaneous fat on T1-weighted and diffuse hyperintense signal on STIR and T2-weighted images. Please note diffuse hypo-intense signal in the lumbar spine on the coronal STIR image (5b) related to hemosiderosis from multiple blood transfusions. Images courtesy of David Rubin, MD
Figure 6:
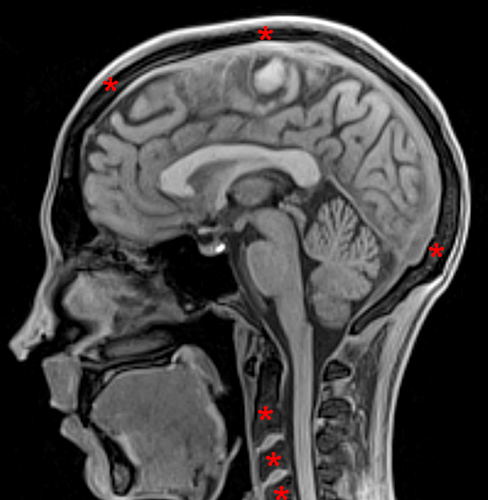
25-year-old female with anorexia nervosa with a BMI of 17. Sagittal T1-weighted image of the brain demonstrates diffuse hypo-intense signal in the calvarium and upper cervical spine (asterisks) as well as a generalized paucity of subcutaneous fat with intermediate subcutaneous signal. Images courtesy of David Rubin, MD
One important pitfall to be aware of in patients with serous atrophy of bone marrow is the potential to miss stress fractures, as fractures are a common complication seen in this patient population. The marked loss of fat saturation in the marrow on fluid sensitive sequences and decreased T1 signal may not only mask a trabecular fracture line but can also mask bone marrow edema due to the background increased signal. Correlation with radiographs and possibly CT or bone scan may be needed to see the actual signs of stress fracture.16
Figure 7:
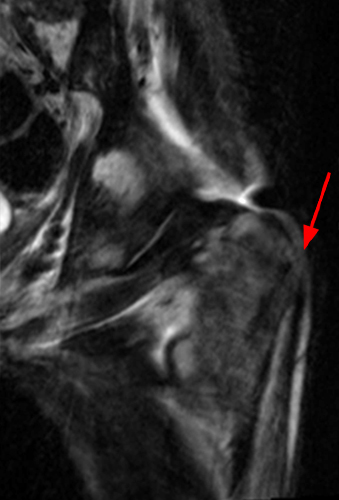
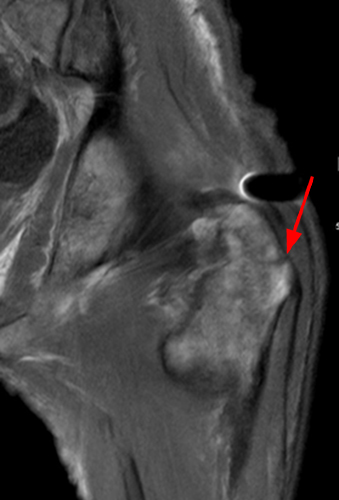
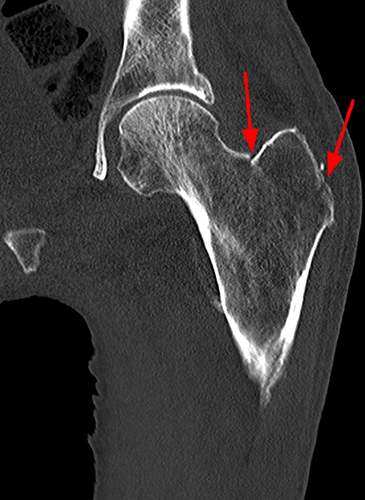
Young adult with history of partial gastric and bowel resections. Coronal STIR image of the left hip (7a) demonstrates subcutaneous hyperintense signal and patchy hyperintense bone marrow signal. The coronal T1-weighted image (7b) demonstrates intermediate signal in the subcutaneous fat and patchy intermediate signal in the bone marrow. There is a nondisplaced fracture along the greater trochanter (arrows) which is faintly seen on the T1-weighted image and is masked by the underlying hyperintense signal on the STIR sequence, where it not as well seen. CT scan (7c) confirms the diagnosis. Images courtesy of Varand Ghazikhanian, MD
Differential Diagnosis
Serous atrophy of bone marrow must be differentiated from other marrow signal altering entities including red marrow conversion and marrow infiltrative processes such as malignancy or infection.
In red marrow reconversion, the pattern is usually in reverse order of the normally seen yellow marrow conversion pattern, routinely seen with high oxygen demand athletes, smoking, obesity, respiratory related disorders, diabetes, and anemia. While yellow marrow is composed of approximately 80% fat, red marrow still contains approximately 40% fat.17 On routine T1-weighted images, red marrow is still more hyperintense than the surrounding muscle and intervertebral discs. Additional chemical shift imaging (in and out of phase imaging) may be utilized to take advantage of the microscopic fat in red marrow. This is demonstrated by visible signal drop-out on the out of phase images.18,19,20
There are many situations where one cannot diagnose serous atrophy of bone marrow confidently, especially when the findings are localized, and a biopsy may be necessary. Reported cases include focal lesions in the feet and spine.15,21
Figure 8:
Red marrow reconversion in 34-year-old female with back pain. Sagittal T1-weighted image (8a) demonstrate hypointense T1 signal throughout the vertebral bodies. Note that the signal in the vertebral bodies is still hyperintense to the disc, and bone marrow signal is normal on the sagittal STIR (8b).
Figure 9:
61-year-old female with back pain and presumed metastatic disease. Axial (9a) and sagittal (9b) T1-weighted images demonstrates diffuse T1 hypointense signal in the sacrum and iliac bones with corresponding marked STIR hyperintense signal (9c). The subcutaneous and intrapelvic fat are preserved. Note that the vertebral bodies are darker than the disc on T1 images.
Figure 10:
Red marrow reconversion in the knee. Sagittal T1-weighted (10a) and coronal T2-weighted fat-suppressed (10b) images demonstrate mild diffuse T1 hypointense signal and T2 hyperintense signal in the distal femoral and proximal tibial diaphysis with sparing of the fat in the epiphysis compatible with red marrow reconversion. Coronal T1-weighted (10c) and coronal proton density-weighted fat-suppressed (10d) images in another patient demonstrate more patchy areas of marrow reconversion. Causes of red marrow reconversion include obesity, smoking and anemia.
Treatment and Prognosis
Serous atrophy of bone marrow is a consequence of an underlying severe illness, and as such, the primary treatment for successful reversal is based on treating the underlying disease, as well as judicious nutritional support, especially in patients with anorexia nervosa.17,21 These treatments have been shown to successfully reverse serous atrophy of bone marrow. However, in patients with anorexia nervosa there is an association with increased fracture risk that may not normalize even after treatment.18 Granulocyte-colony stimulating factor has also been successfully used to aid recovery of neutropenia in anorexia nervosa.22
Summary
Serous atrophy of bone marrow is a marrow disorder characterized by atrophy of hematopoietic and fat cells in the bone marrow with accumulation of gelatinous material. This results in marked marrow signal alterations on MRI, which may cause confusion for the MRI technologist and interpreting radiologist. Many times, patients are referred for imaging to evaluate for stress fractures or infection. As it is usually not diagnosed prior to imaging, the radiologist must be familiar with this entity to not misinterpret the MRI as a technical failure of fat saturation, and to be able to differentiate it from other marrow infiltrative processes. Serous atrophy of the bone marrow does not enhance with contrast, and therefore contrast may be utilized to differentiate it from malignancy or infection.1
References
- Boutin RD, White LM, Laor T, et al. MRI findings of serous atrophy of bone marrow and associated complications. Eur Radiol 25, 2771-2778 (2015).doi:10.1007/s00330-015-3692-5 ↩
- Bohm J. Gelatinous transformation of the bone marrow: the spectrum of underlying diseases. Am J Surg Pathol. 2000;24(1):56-65. doi:10.1097/00000478-200001000-00007 ↩
- Jain R, Singh ZN, Khurana N, Singh T. Gelatinous transformation of bone marrow: A study of 43 cases. Indian J Pathol Microbiol. 2005;48(1):1-3. ↩
- Sen R, Singh S, Singh H, Gupta A, Sen J. Clinical profile in gelatinous bone marrow transformation. J Assoc Physicians India. 2003;51(JUN):585-588. http://europepmc.org/article/med/15266925. ↩
- Das S, Mishra P, Kar R, Basu D. Gelatinous Marrow Transformation: A Series of 11 Cases from a Tertiary Care Centre in South India. Turkish J Hematol. 2014;31(2):175-179. ↩
- Seaman JP, Kjeldsberg CR, Linker A. Gelatinous transformation of the bone marrow. Hum Pathol. 1978;9(6):685-692. doi:10.1016/S0046-8177(78)80051-3 ↩
- Tavassoli M, Eastlund DT, Yam LT, Neiman RS, Finkle H. Gelatinous transformation of bone marrow in prolonged self-induced starvation. Scand J Haematol. 1976;16(4):311-319. doi:10.1111/J.1600-0609.1976.TB01156.X ↩
- Bredella MA, Fazeli PK, Miller KK, et al. Increased Bone Marrow Fat in Anorexia Nervosa. J Clin Endocrinol Metab. 2009;94(6):2129. doi:10.1210/JC.2008-2532 ↩
- Cornbleet PJ, Moir RC, Wolf PL. A histochemical study of bone marrow hypoplasia in anorexia nervosa. Virchows Arch A Pathol Anat Histol. 1977;374(3):239-247. doi:10.1007/BF00427118 ↩
- Devlin MJ. Why does starvation make bones fat? Am J Hum Biol. 2011;23(5):577. doi:10.1002/AJHB.21202 ↩
- Green DE, Adler BJ, Chan ME, et al. Altered composition of bone as triggered by irradiation facilitates the rapid erosion of the matrix by both cellular and physicochemical processes. PLoS One. 2013;8(5). doi:10.1371/JOURNAL.PONE.0064952 ↩
- Hwang S, Lefkowitz R, Landa J, et al. Local changes in bone marrow at MRI after treatment of extremity soft tissue sarcoma. Skeletal Radiol 38, 11-19 (2009) doi:10.1007/s00256-008-0560-2 ↩
- Vande Berg BC, Malghem J, Lecouvet FE, Lambert M, Maldague BE. Distribution of serouslike bone marrow changes in the lower limbs of patients with anorexia nervosa: predominant involvement of the distal extremities. AJR Am J Roentgenol. 1996;166(3):621-625. doi:10.2214/AJR.166.3.8623638 ↩
- Vande Berg BC, Malghem J, Devuyst O, Maldague BE, Lambert MJ. Anorexia nervosa: correlation between MR appearance of bone marrow and severity of disease. Radiology. 1994;193(3):859-864. doi:10.1148/RADIOLOGY.193.3.7972838 ↩
- Lee J, Yoo YH, Lee S, Kim HS, Kim S. Gelatinous Transformation of Bone Marrow Mimicking Malignant Marrow-Replacing Lesion on Magnetic Resonance Imaging in a Patient without Underlying Devastating Disease. Investig Magn Reson Imaging. 2018;22(1):50. doi:10.13104/IMRI.2018.22.1.50 ↩
- Tins B, Cassar-Pullicino V. Marrow changes in anorexia nervosa masking the presence of stress fractures on MR imaging. Skeletal Radiol. 2006;35(11):857-860. doi:10.1007/S00256-005-0053-5 ↩
- Malkiewicz A, Dziedzic M. Bone marrow reconversion – imaging of physiological changes in bone marrow. Polish J Radiol. 2012;77(4):45-50. doi:10.12659/PJR.883628 ↩
- Shah LM, Hanrahan CJ. MRI of spinal bone marrow: part I, techniques and normal age-related appearances. AJR Am J Roentgenol. 2011;197(6):1298-1308. doi:10.2214/AJR.11.7005 ↩
- Hanrahan CJ, Shah LM. MRI of spinal bone marrow: Part 2, T1-weighted imaging-based differential diagnosis. Am J Roentgenol. 2011;197(6):1309-1321. doi:10.2214/AJR.11.7420 ↩
- Kung JW, Yablon CM, Eisenberg RL. Bone marrow signal alteration in the extremities. AJR Am J Roentgenol. 2011;196(5). doi:10.2214/AJR.10.4961 ↩
- Tonogai I, Nakajima D, Miyagi R, Sairyo K. Gelatinous Transformation of Bone Marrow in the Calcaneus, Diagnosed by Open Bone Biopsy in a Patient With Anorexia Nervosa: A Case Report. doi:10.1177/2473011418780325 ↩
- Muraki Y, Kobayashi T, Kojima H, et al. (Anorexia nervosa with neutropenia–response of neutrophils to G-CSF). Rinsho Ketsueki. 1992;33(3):328-332. https://europepmc.org/article/med/1374486. Accessed July 14, 2021. ↩