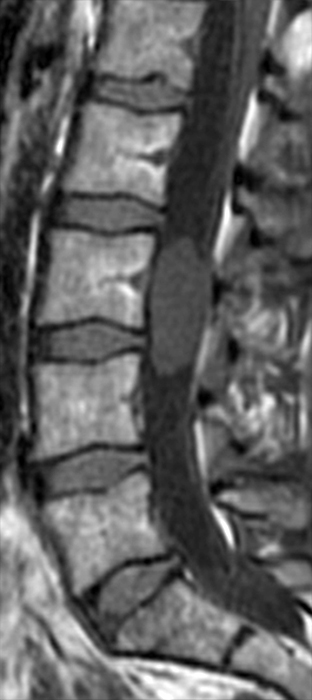
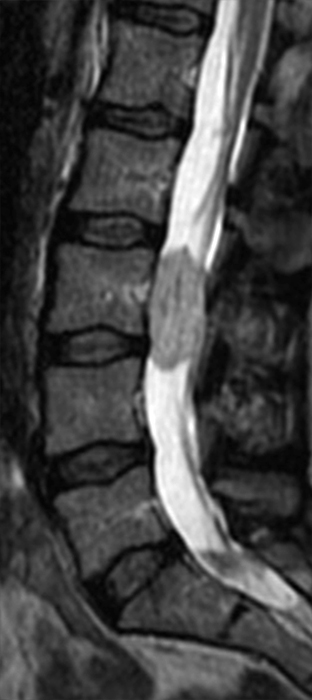
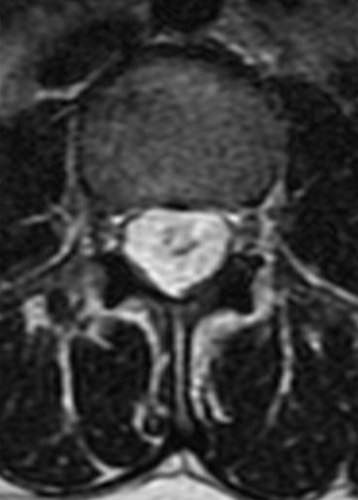
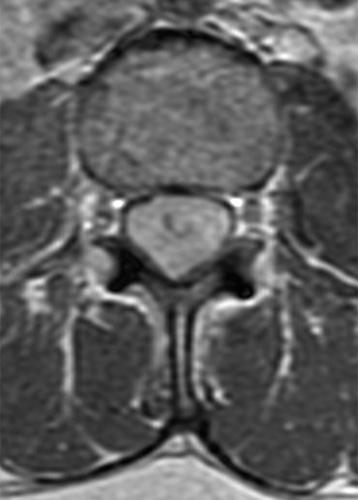
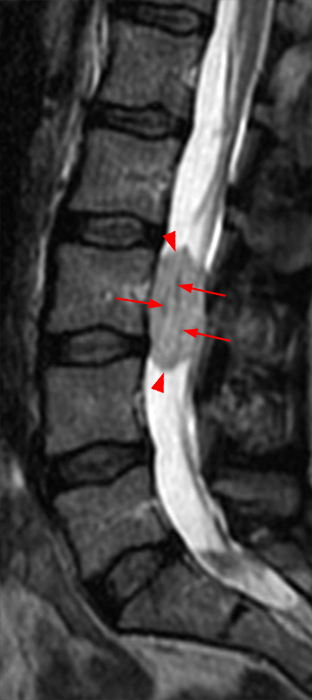
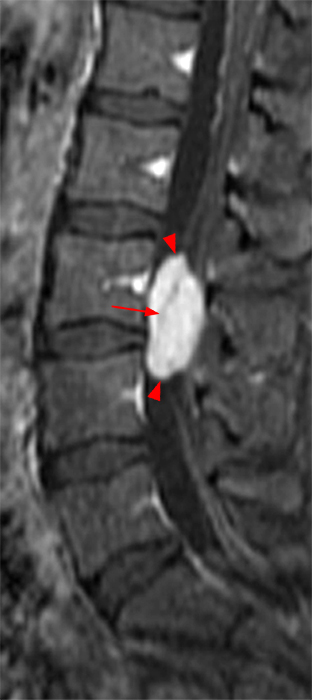
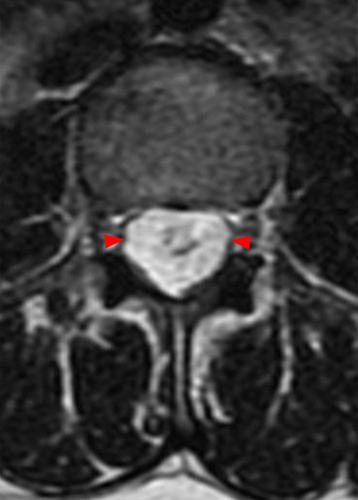
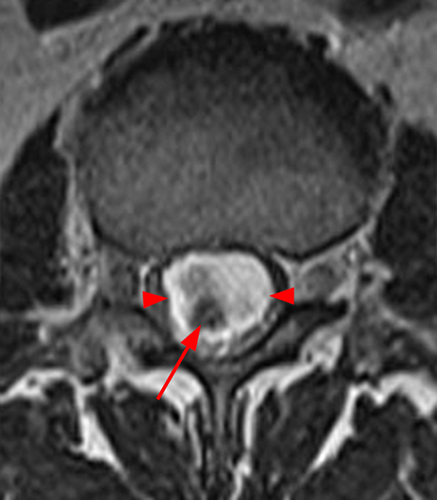
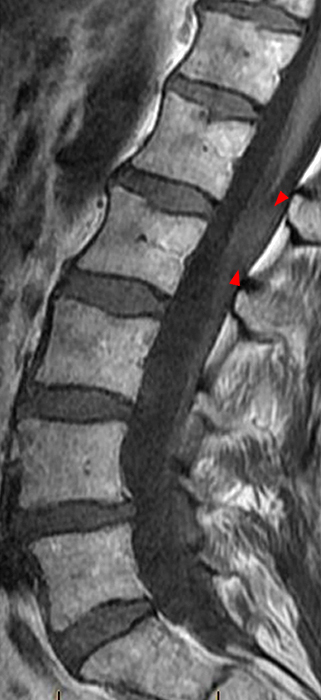
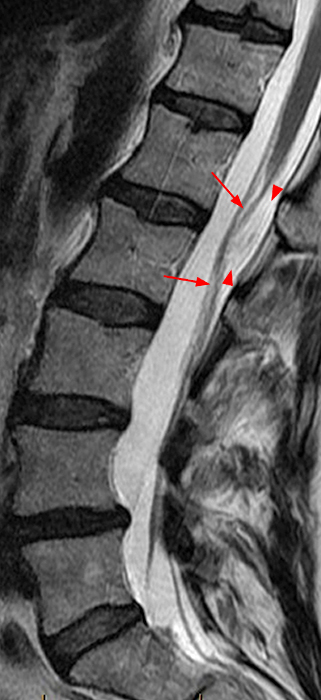
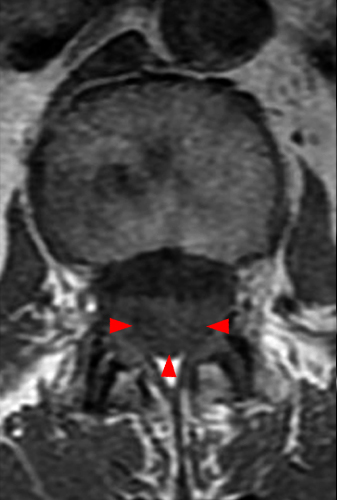
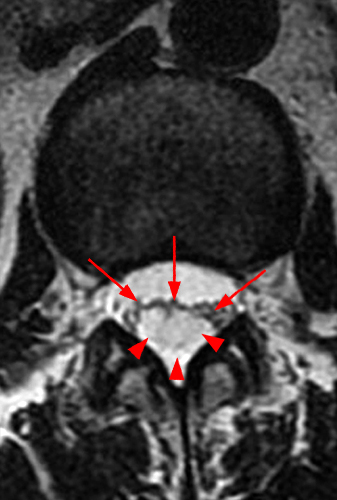
Clinical History: A 35-year-old woman presents with low back and right leg pain that began during pregnancy four years previously. Sagittal T1- and T2-weighted (1a and 1b), T1-weighted post-gadolinium (1c), axial T2-weighted (1d) and T1-weighted post-gadolinium axial images (1e) are provided. What are the findings? What is your diagnosis?
Findings
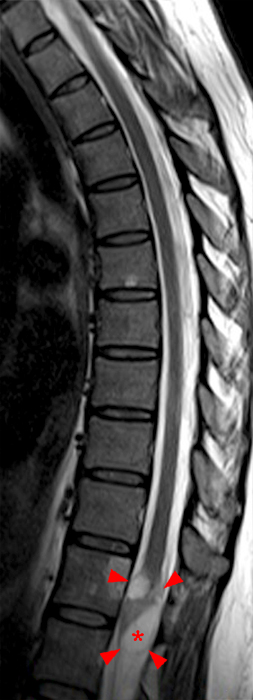
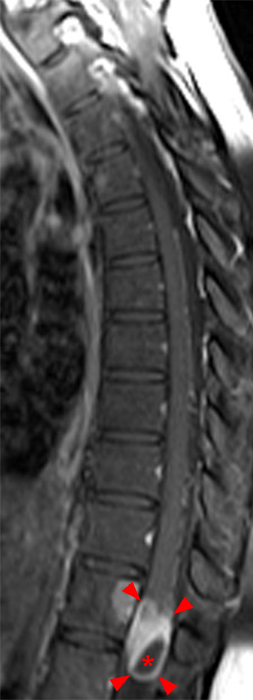
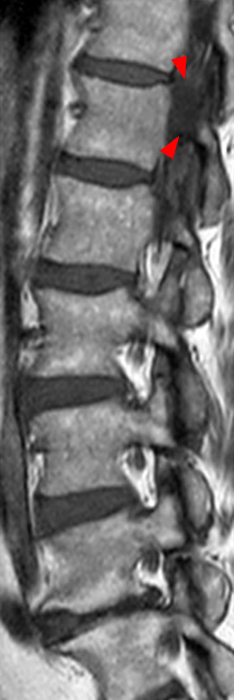
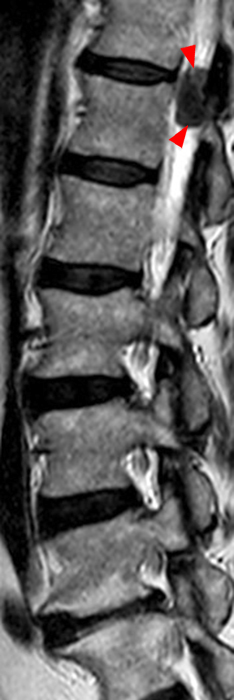
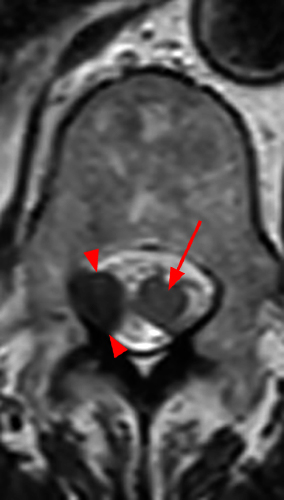
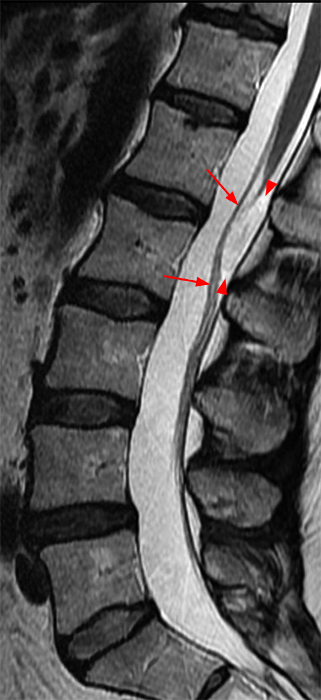
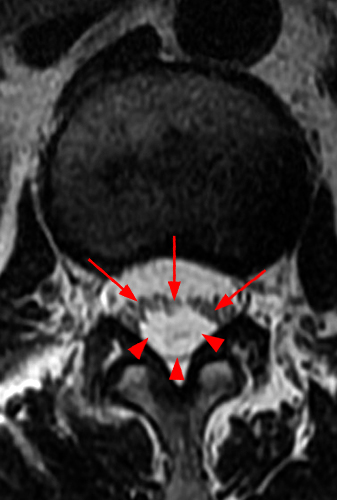
Figure 2:
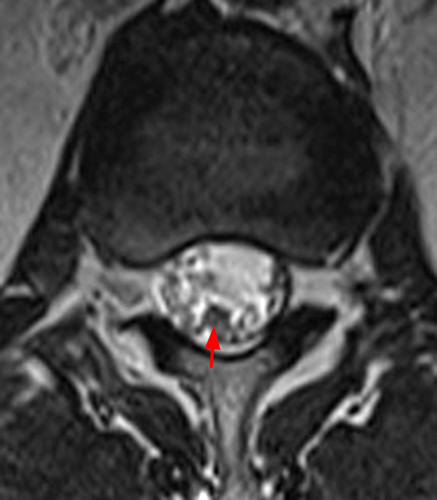
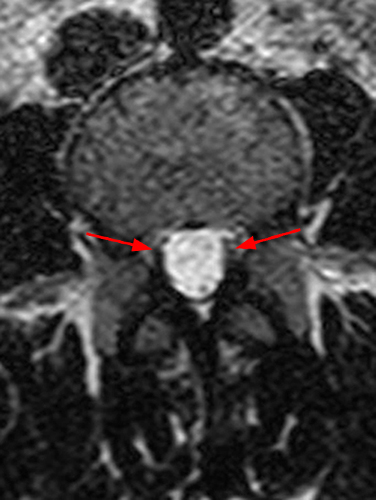
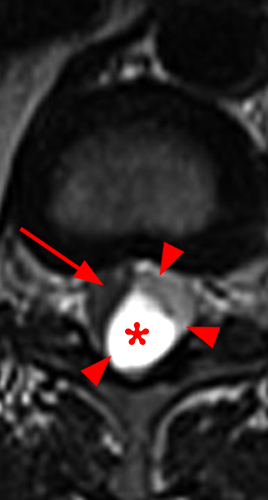
A mildly lobulated and expansile intradural extramedullary mass (arrowheads) is present at L3 and is isointense to spinal cord on T1-weighted image (2a) and increased signal intensity relative to the cord on T2-weighted images (2b, d). The mass shows diffuse, intense enhancement following the administration of gadolinium on T1-weighted images (2c, e). The thin tubular structures within the mass are encased nerve roots of the cauda equina (arrows).
Diagnosis
Myxopapillary ependymoma.
Introduction
Spinal tumors are relatively rare, accounting for only 5-10% of central nervous system (CNS) tumors. The vast majority of spinal tumors, 70-80%, are intradural extramedullary in location, with schwannomas and meningiomas most common.1 Myxopapillary ependymoma (MPE), a distinct subtype of glioma that occurs in the region of the filum terminale and conus medullaris, is a less common intradural extramedullary lesion originally described by Kernohan in 1932.2,3 It accounts for 13% of all spine ependymomas; however, it is the most common neoplasm in the conus and cauda equina, accounting for 83% of tumors in this region.1 While generally benign and slow growing, MPE demonstrates a tendency for local recurrence and may disseminate throughout the CNS with the potential for an unfavorable outcome, especially in children.1,4 Furthermore, because of the typical slow growth and insidious clinical manifestations, diagnosis may not be made until the tumor is quite large and the potential for cure limited. MR imaging can provide an early diagnosis, which is key to a favorable outcome.
Demographics
MPE occurs in all ages, with a mean age of 36 years and a range of 3 weeks to 82 years. There is a male predominance of 2.2:1.1 MPE is more common in whites than other races and in non-Hispanics compared to Hispanics.5
Clinical Presentation
Patients with MPE most commonly present with low back pain. Radicular or sacral symptoms, weakness and sphincter dysfunction are less common. Cauda equina syndrome, comprised of sphincter dysfunction, saddle anesthesia and lower extremity weakness, may occur but is not an early manifestation of MPE, reflecting this neoplasm’s slow growth.1 The usual initial presentation of low back pain and lower extremity radiculopathy is nonspecific, mimicking discogenic disease, and contributes to the frequently delayed diagnosis of MPE.3 Large tumors may be associated with an irreversible neurologic deficit.6,7
Location
MPE is typically intradural and extramedullary in the region of the conus medullaris or cauda equina. The tumor usually arises from the filum terminale and frequently infiltrates the cauda equina nerve roots and/or the conus medullaris.8,9 It most often occurs at L2, followed by L1 and L3. In Wippold’s series, none of the cases extended above T9.3 MPE frequently spans an average of 4 vertebral segments. Those tumors occurring near the sacrum may be associated with large areas of bone destruction.1 MPE is usually solitary, although multiple lesions occur in up to 43% of patients, which may correspond to drop metastases.1,3 MPE may rarely occur within the cervicothoracic cord, fourth ventricle and brain parenchyma. Other rare locations include the subcutaneous sacrococcygeal and presacral regions, where MPE is thought to arise from ectopic ependymal remnants.1 In the rare extradural location, it occurs in the sacrum, or is presacral or postsacral.4 MPE can spread through the CNS but metastases beyond the CNS are rare.9 Those tumors that do show distant metastases tend to be extradural.3
Pathology
The four types of ependymomas include typical, anaplastic, subependymoma and myxopapillary. MPE is thought to arise from ependymal cell rests normally present in the filum terminale due to the extrusion of ependymal cells before neural tube closure.4,10 The rare extradural postsacral MPE is believed to originate from a persistent portion of the neural tube.3 The tumor is highly vascular, elongated, sausage-shaped, often encapsulated and may show hemorrhagic or mucinous degeneration.3,9 Histologically, MPE is composed of cellular rosettes and pseudorosettes mixed with papillary regions containing vascular cores and an extensive intercellular mucoid matrix.9,11
MR Imaging
MPE classically presents as an intradural extramedullary, sausage-shaped, lobulated and often encapsulated thoracolumbar mass extending for several vertebral levels that may expand the spinal canal or less frequently a neural foramen. Small tumors tend to displace the lumbosacral nerve roots, while large tumors may encase them. MPE is T2 hyperintense and usually T1 isointense to cord; less often it is T1 hypointense or hyperintense, with the latter due to the presence of mucin or hemorrhage (Fig. 3).9 T1 hyperintensity is helpful in the differentiation from other subtypes of ependymoma. Superficial siderosis causing decreased signal intensity may occur, reflecting the vascularity of the tumor.
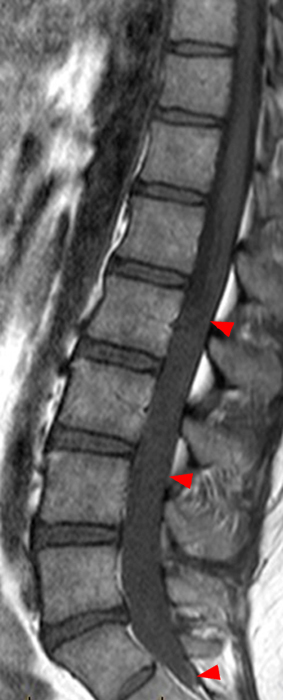
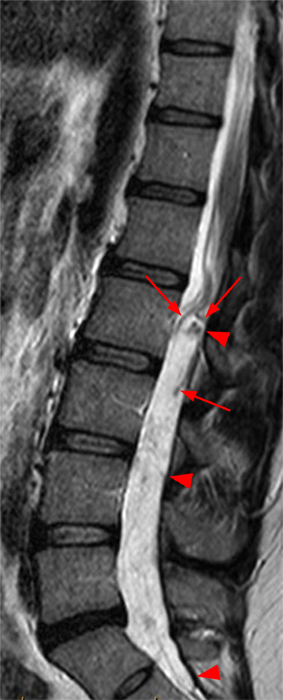
Figure 3:
Myxopapillary ependymoma in a 58-year-old man with low back and right buttock pain since being caught between two cars two years previously. Lumbar spine MR sagittal T1-and T2-weighted images (3a, b) and axial T2-weighted images (3c, d) depict a sausage-shaped mass (arrowheads) next to the conus tip that extends over 3 vertebral segments, with signal intensity predominantly hypointense to cord on T1-weighted and heterogenous though predominantly hyperintense on T2-weighted images. Note the focus of low signal intensity on the T2-weighted and corresponding increased signal intensity on the T1-weighted images, likely hemorrhage or mucin (long arrows). The tumor is anterior to the conus tip (3a, b, d, short arrows).
The vascularity also results in intense enhancement following intravenous gadolinium contrast administration (Fig 2c).1 Usually, enhancement is homogeneous, but it may be heterogeneous if hemorrhage or necrosis is present.9 Contrast administration is essential because it helps distinguish tumor from the cord and defines cysts.3 Contrast is also useful in improving detection of CNS metastases (Fig 4).
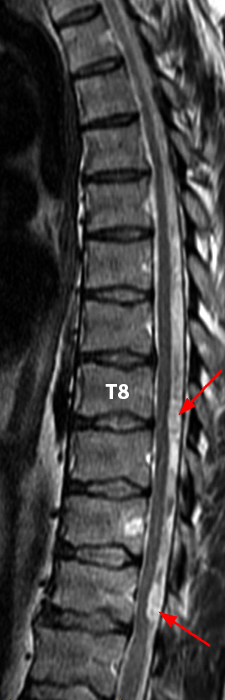
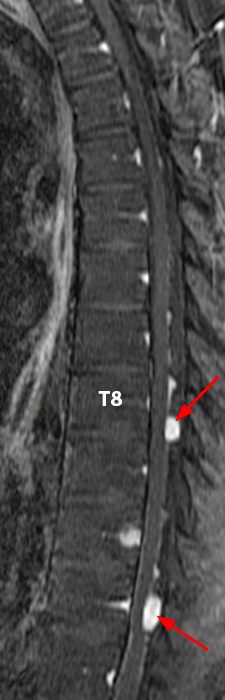
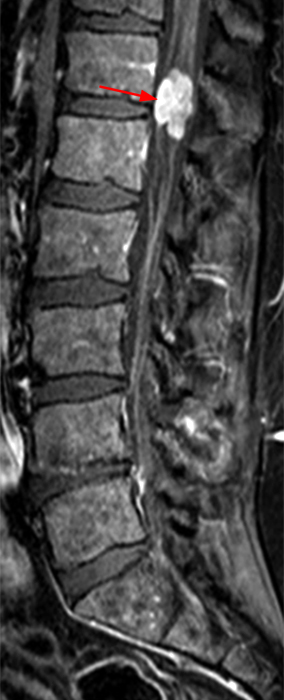
Figure 4:
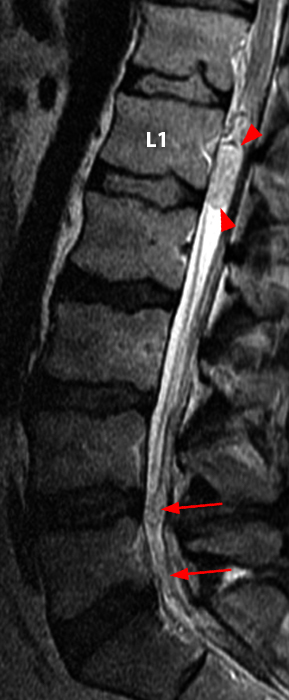
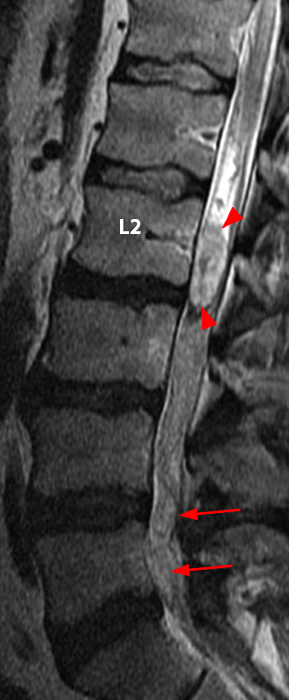
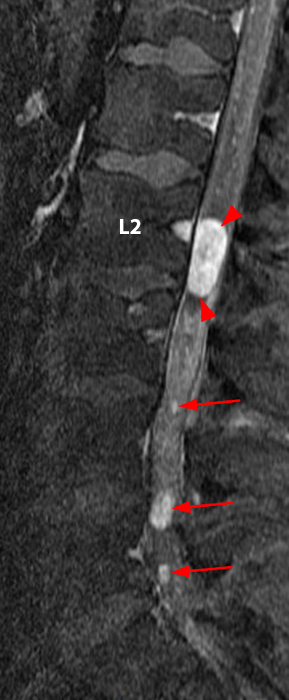
Mobile myxopapillary ependymoma with CNS metastases in a 39-year-old man with low back pain radiating to the left leg. Lumbar spine MR sagittal T2-weighted image (4a) shows a sausage-shaped mass hyperintense to cord and adjacent to the conus at L1 (arrowheads). The patient returned 9 days later for additional imaging. Sagittal T2 (4b) and T1-weighted fat-suppressed post-gadolinium (4c) images show the tumor has migrated to L2 (arrowheads). MPE may be mobile, a feature that is not specific for this tumor. 3 foci of CNS metastases at L4 and L5 are conspicuous on the contrast-enhanced image (4c) and subtle on the T2-weighted sagittal images (4a, b) (arrows). Thoracic spine MR imaging performed 2 weeks after the first study shows additional CNS metastases at T8 and T11 (4d, e) (arrows). Note the decreased conspicuity of the intrathecal metastases on the T2-weighted sequence (4d) due to signal intensity similar to CSF.
The intense enhancement of the mass makes it conspicuous on contrast-enhanced images; however, due to the frequently nonspecific clinical presentation, gadolinium contrast is not routinely administered is such cases, and careful scrutiny of the intraspinal contents on routine, unenhanced spine MR imaging is imperative for detection. A potential source of confusion is that MPE may be mobile, located at different levels on MR imaging performed at different times, a finding that is not specific for MPE, and most commonly found in schwannomas (Fig 4, a-c).12 Anatomic continuity with the conus is an important feature to assess on MR imaging for surgical planning, as adhesions may prevent complete resection, with increased risk of major neurologic deficit (Fig. 3b,d).13 A potential pitfall arises when tumor T2 signal intensity is similar to CSF, making it difficult to visualize on T2-weighted images, and stressing the importance of close inspection of the intradural contents on both T2 and T1-weighted sequences and assessing for cauda equina displacement (Fig. 5). Another potential pitfall on routine lumbar spine MR imaging is failing to scan far enough cephalad; it is important that screening sagittal lumbar sequences include the lower thoracic levels.3
Because the clinical presentation is often nonspecific, the diagnosis of MPE may be delayed until the tumor is large, potentially resulting in a less favorable prognosis. MR imaging is extremely useful as it can detect MPE when it is small, enabling a better outcome.2 A small MPE may mimic a nerve sheath tumor on MRI, and as such it should be considered in the differential diagnosis.14
Figure 5:
Myxopapillary ependymoma showing the potential pitfall of decreased conspicuity due to signal intensity similar to CSF. Routine screening lumbar spine MRI in a 37-year-old woman with low back pain and sciatica. Sagittal T1- and T2-weighted (5a, b) and T2-weighted axial (5c) images show subtle, poorly defined tumor from L1 through the caudal dural sac at the S2 level (arrowheads). On the T1-weighted sequence, the signal intensity of the tumor is slightly greater than CSF, and on the T2-sagittal sequence it is slightly lower signal intensity than CSF, with mild heterogeneity. The signal heterogeneity on the sagittal T2-weighted image and the peripheral displacement of the cauda equina on the T2-weighted sagittal and axial images (arrows) are helpful clues to the presence of a mass.
Treatment
When possible, gross total resection of MPE is the treatment of choice, providing a potential cure without the need for adjuvant therapy.4,9 85% of MPE in the cauda equina can be removed totally.7 The role of radiation therapy has not been adequately assessed. It may be necessary if there is subtotal resection or recurrence; its use in patients who have undergone total resection and for those with extradural ependymoma is controversial.15,16 Chemotherapy is indicated only in those patients with recurrent disease that is refractory to resection and irradiation.9
Prognosis
MPE typically exhibits relatively benign behavior and a more favorable prognosis compared to ependymomas occurring within the spinal cord or brain.2 The extent of surgical removal, tumor location and integrity of the tumor capsule determine prognosis.17 The five-year survival rate after complete or partial resection is 98.4%.1 Upon gross total resection, mean survival of patients with MPE is 19 years. Even after gross total resection, the recurrence rate is 10-19%.18,19 The prognosis of the rare extradural MPE is worse, and intrasacral tumors always have a poor prognosis.11,17 Metastatic spread of MPE is uncommon; when it occurs, it tends to spread rostrally within the central nervous system, with the intracranial location the most common site of metastasis. Caudal spread as drop metastases may also occur. Long term follow up is necessary since metastases have been reported up to twenty years after initial treatment. Screening of the entire CNS axis at the time of diagnosis and during follow up is recommended.10 MPE may be more aggressive in children, with distant spinal and brain metastases more common (9% and 6%, respectively) than in adults. The tendency for greater dissemination results in a less predictable outcome in children.1
Differential Diagnosis
The differential diagnosis of the typical intradural extramedullary thoracolumbar myxopapillary ependymoma is broad and includes both neoplasms and nonneoplastic disorders.
Schwannomas of the cauda equina may appear identical to small MPEs. Schwannomas involve dorsal sensory nerve roots, so clinically they present with radicular pain or signs related to nerve root or cord compression. Morphologically, schwannomas are nodular lesions that demonstrate T1 iso- or hypointensity and T2 hyperintensity relative to cord.20 T1 hyperintensity occurs in melanotic schwannomas. Larger lesions may show cystic degeneration and a target appearance. Contrast enhancement is heterogeneous or ring-like (Fig. 6). Foraminal extension is common, producing a dumbbell morphology and benign pressure erosion if there is contact with bone.1 The presence of multiple lesions should prompt consideration of neurofibromatosis type 2 and schwannomatosis.
Figure 6:
Schwannoma in a 43-year-old woman complaining of back pain and weakness and left hip and leg pain. Thoracic spine sagittal T2-weighted and T1-weighted fat-suppressed post-gadolinium (6a, b) and axial T2-weighted and T1-weighted fat-suppressed post-gadolinium (6c, d) MR images. An intradural extramedullary partially cystic enhancing mass at T11 (arrowheads) compresses the spinal cord (arrows, c and d) to a greater extent than would be expected from the clinical presentation, typical of a slowly growing neoplasm. Areas of cystic degeneration are present within the mass (asterisks). Incidentally noted is a hemangioma within the T11 vertebral body.
A meningioma’s broad dural attachments may allow differentiation from a small MPE; however, a large MPE, even though not dural-based, may broadly abut the dura, making it difficult to distinguish from a meningioma.21 The dural tail sign of meningiomas is a tapered enhancing tumor margin that is a useful clue, but it is present less often in spinal than intracranial meningiomas.[22] Meningiomas that are not dural-based are extremely rare. The vast majority of spinal meningiomas are thoracic (80%), followed by cervical (15%) and lumbar (5%) in location. The conus/ filum terminale location typical of MPE is uncommon for meningioma. The presence of multiple meningiomas should prompt consideration of neurofibromatosis type 2.21 Most meningiomas are hyper- or isointense relative to the spinal cord on T2-weighted images, intermediate to hypointense on T1-weighted images and demonstrate strong, homogeneous gadolinium contrast enhancement.22 Calcification, when it occurs, is a useful differentiating feature of meningioma that is demonstrated by signal hypointensity on T1 and T2-weighted MR images and high attenuation on CT and radiographs (Fig. 7).
Figure 7:
71-year-old female with low back and left thigh pain and meningioma at T11-12. Sagittal T1- and T2-weighted (7a, b) and axial T2-weighted (7c) images from lumbar spine MRI show a right lateral dural-based extramedullary mass that is low signal intensity on all images (arrowheads). The spinal cord (arrow) is deviated by the mass. The low signal intensity within the mass corresponds to calcification, confirmed by the characteristic increased density on the AP lumbar spine radiograph (7d) (arrowheads).
Neurofibromas are generally indistinguishable from schwannomas by imaging but are usually asymptomatic and more frequently cervical in location.1,21 Cystic change is less common than in schwannomas.20
Paragangliomas are highly vascular tumors with prominent flow voids and intense contrast enhancement, which may make them indistinguishable from MPE; however, unlike MPE, they are rare tumors of the cauda equina and filum terminale, accounting for only 3-4% of tumors in this region.1,21
Leptomeningeal metastasis is characterized by nodular intradural enhancement and while relatively uncommon, occurring in 5-15% of patients with malignancies, it should be a differential consideration of MPE in the appropriate clinical setting (Fig. 8). Leptomeningeal metastasis more commonly occurs with hematologic than solid primary malignancies. Clinical presentation includes motor weakness, gait ataxia and incontinence. The prognosis is poor.1
Figure 8:
56-year-old man with lung cancer and lower extremity weakness. Sagittal T1-weighted fat-suppressed post-gadolinium MR images of the lumbar spine (8a, b) show multiple enhancing intradural extramedullary masses proved to be leptomeningeal metastases (arrows). These findings are nonspecific, however, and MPE with drop metastases and multiple schwannomas are differential considerations. Case courtesy H. Wayne Slone, MD, The Ohio State University Medical Center, Columbus, OH.
Malignant peripheral nerve sheath tumors (MPNST) can mimic schwannomas but are more aggressive. Unlike MPE, MPNST are most commonly paraspinal in location.1
Nonneoplastic disorders including dermoid, epidermoid and disk herniations should be considered in the differential diagnosis of MPE though they are not commonly intradural (Fig. 9).1
Figure 9:
Epidermoid in a 57-year-old woman with left lower back pain radiating to left hip and groin for years. Sagittal T1- and T2-weighted (9a, b) and axial T1- and T2-weighted (9c, d) images of the lumbar spine show an L1-2 intradural extramedullary lentiform mass (arrowheads) with ill-defined margins deviating the cauda equina (arrows) anteriorly with T2 hyperintensity and T1-weighted signal intensity that is hypointense to cord but of greater signal intensity than CSF. T2-weighted sagittal and axial images from 5 years previously (9e, f) show the mass (arrowheads) is unchanged.
For the rare sacral MPE, which may be associated with considerable bone destruction, the differential diagnosis includes aneurysmal bone cyst, chordoma, plasmacytoma, metastatic disease and giant cell tumor.9
Conclusion
Myxopapillary ependymoma is a subtype of ependymoma that is typically intradural extramedullary in location and constitutes the most common tumor of the conus and cauda equina region. It is generally less aggressive than other types of ependymoma, although it has a tendency for local recurrence, and intrathecal metastases may occur. The clinical presentation often mimics discogenic disease, potentially delaying diagnosis. MR imaging is useful for determining tumor extent and relationship to intraspinal structures, and allows detection when the tumor is small, which is vital for potential cure.
References
- Koeller, K.K. and R.Y. Shih, Intradural Extramedullary Spinal Neoplasms: Radiologic-Pathologic Correlation. Radiographics, 2019. 39(2): p. 468-490. ↩
- Yamada, C.Y., G.J. Whitman, and F.S. Chew, Myxopapillary ependymoma of the filum terminale. AJR Am J Roentgenol, 1997. 168(2): p. 366. ↩
- Wippold, F.J., 2nd, et al., MR imaging of myxopapillary ependymoma: findings and value to determine extent of tumor and its relation to intraspinal structures. AJR Am J Roentgenol, 1995. 165(5): p. 1263-7. ↩
- Strojnik, T., T. Bujas, and T. Velnar, Invasive myxopapillary ependymoma of the lumbar spine: A case report. World J Clin Cases, 2019. 7(10): p. 1142-1148. ↩
- Villano, J.L., C.K. Parker, and T.A. Dolecek, Descriptive epidemiology of ependymal tumours in the United States. Br J Cancer, 2013. 108(11): p. 2367-71. ↩
- Koeller, K.K., R.S. Rosenblum, and A.L. Morrison, Neoplasms of the spinal cord and filum terminale: radiologic-pathologic correlation. Radiographics, 2000. 20(6): p. 1721-49. ↩
- Wager, M., et al., Cauda equina tumors: a French multicenter retrospective review of 231 adult cases and review of the literature. Neurosurg Rev, 2000. 23(3): p. 119-29; discussion 130-1. ↩
- Monajati, A., et al., MR of the cauda equina. AJNR Am J Neuroradiol, 1987. 8(5): p. 893-900. ↩
- Shors, S.M., et al., Best cases from the AFIP: myxopapillary ependymoma of the sacrum. Radiographics, 2006. 26 Suppl 1: p. S111-6. ↩
- Rege, S.V., et al., Spinal myxopapillary ependymoma with interval drop metastasis presenting as cauda equina syndrome: case report and review of literature. J Spine Surg, 2016. 2(3): p. 216-221. ↩
- Sonneland, P.R., B.W. Scheithauer, and B.M. Onofrio, Myxopapillary ependymoma. A clinicopathologic and immunocytochemical study of 77 cases. Cancer, 1985. 56(4): p. 883-93. MPE is reactive for GFAP and S100 protein. ↩
- Moon, K., A.K. Filis, and A.R. Cohen, Mobile spinal ependymoma. J Neurosurg Pediatr, 2010. 5(1): p. 85-8. ↩
- Al-Habib, A., et al., Myxopapillary ependymoma: correlation of clinical and imaging features with surgical resectability in a series with long-term follow-up. Spinal Cord, 2011. 49(10): p. 1073-8. ↩
- Kim, J., et al., Myxopapillary ependymoma of filum terminale mimicking nerve sheath tumor. Kor J Spine, 2008. 5(2): p. 95-98. ↩
- Biagini, R., et al., Osteolytic extra-axial sacral myxopapillary ependymoma. Skeletal Radiol, 1999. 28(10): p. 584-9. ↩
- Fassett, D.R. and M.H. Schmidt, Lumbosacral ependymomas: a review of the management of intradural and extradural tumors. Neurosurg Focus, 2003. 15(5): p. E13. ↩
- Xie, T.H., et al., Surgery for primary filum terminale ependymomas: outcome and prognostic factors. CNS Neurosci Ther, 2014. 20(2): p. 131-9. ↩
- Schweitzer, J.S. and U. Batzdorf, Ependymoma of the cauda equina region: diagnosis, treatment, and outcome in 15 patients. Neurosurgery, 1992. 30(2): p. 202-7. ↩
- Celli, P., L. Cervoni, and G. Cantore, Ependymoma of the filum terminale: treatment and prognostic factors in a series of 28 cases. Acta Neurochir (Wien), 1993. 124(2-4): p. 99-103. ↩
- Friedman, D.P., L.M. Tartaglino, and A.E. Flanders, Intradural schwannomas of the spine: MR findings with emphasis on contrast-enhancement characteristics. AJR Am J Roentgenol, 1992. 158(6): p. 1347-50. ↩
- Ross, J. and K. Moore, Diagnostic Imaging: Spine. 3rd ed. 2015, Philadelphia: Elsevier. ↩
- Yeo, Y., et al., Magnetic resonance imaging spectrum of spinal meningioma. Clin Imaging, 2019. 55: p. 100-106. ↩