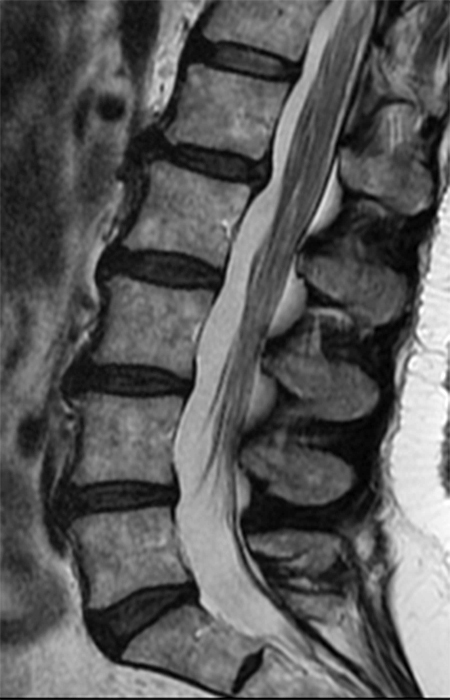
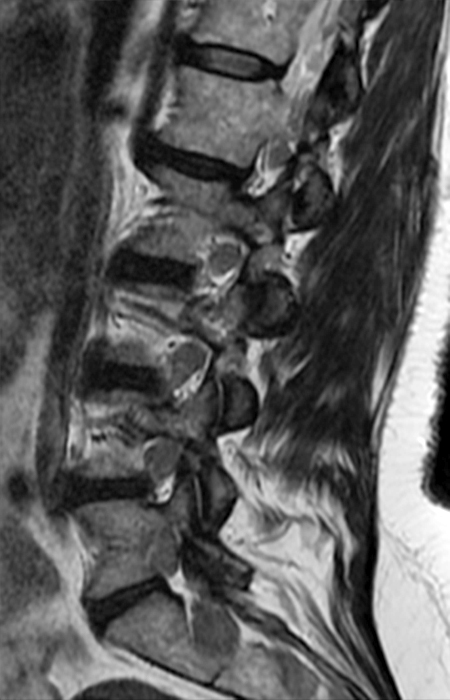
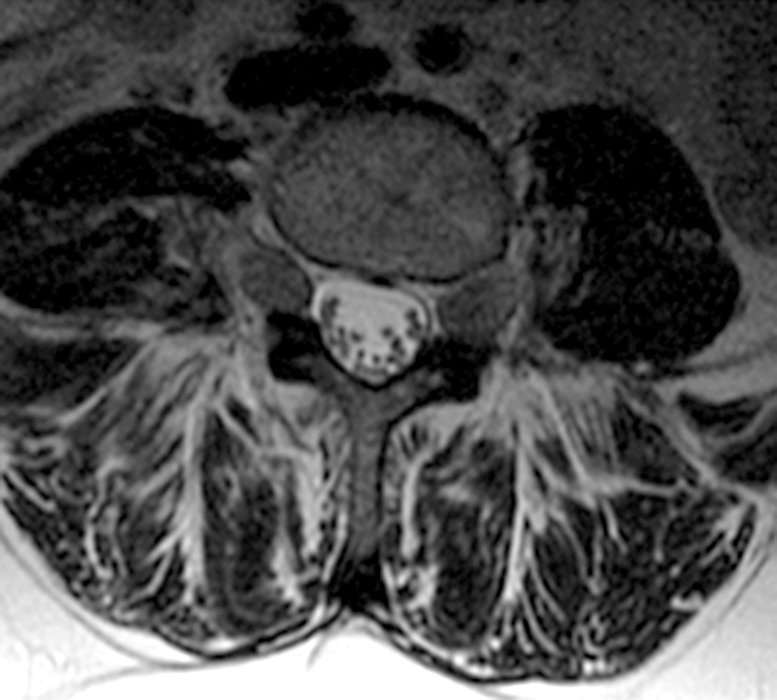
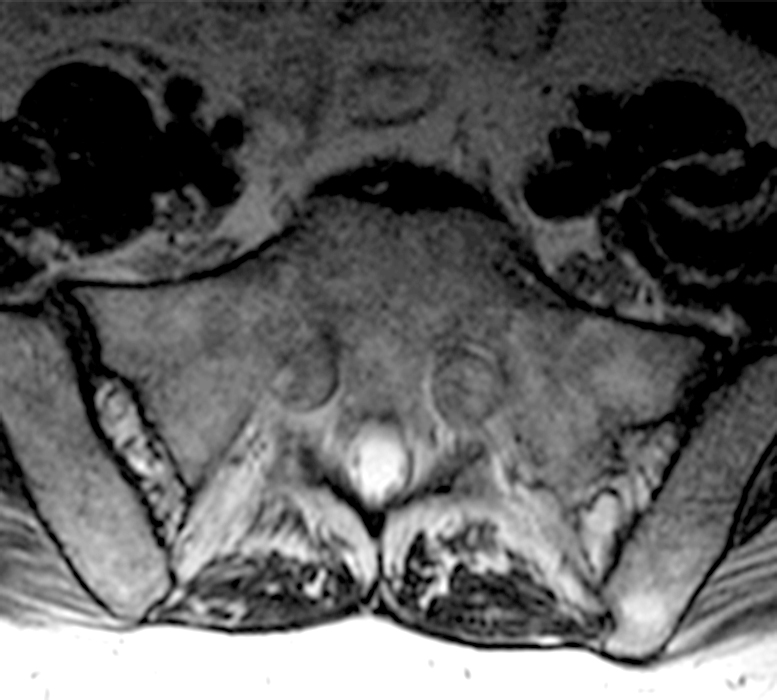
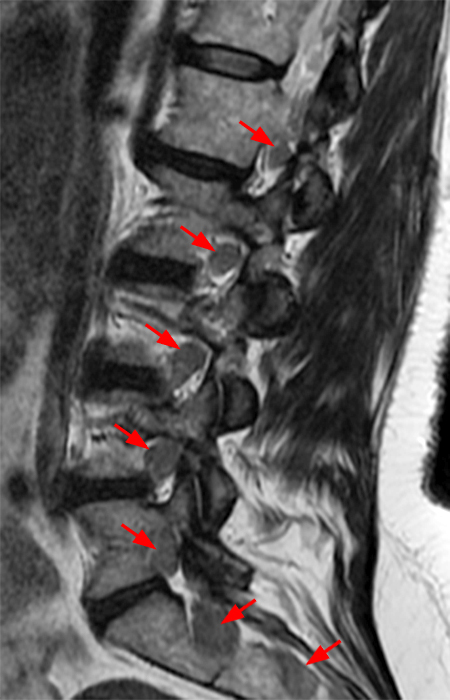
Clinical history: A 55-year-old female presents with constant lower back pain and extremity numbness. Sagittal and parasagittal T2-weighted images (1a-b) of the lumbar spine and axial T2-weighted images at the L4-5 (1c) and S1 levels (1d) are provided. What are the findings? What is your diagnosis?
Findings
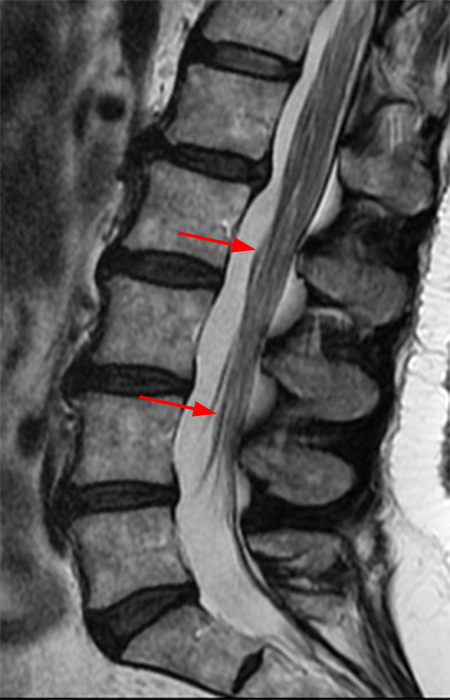
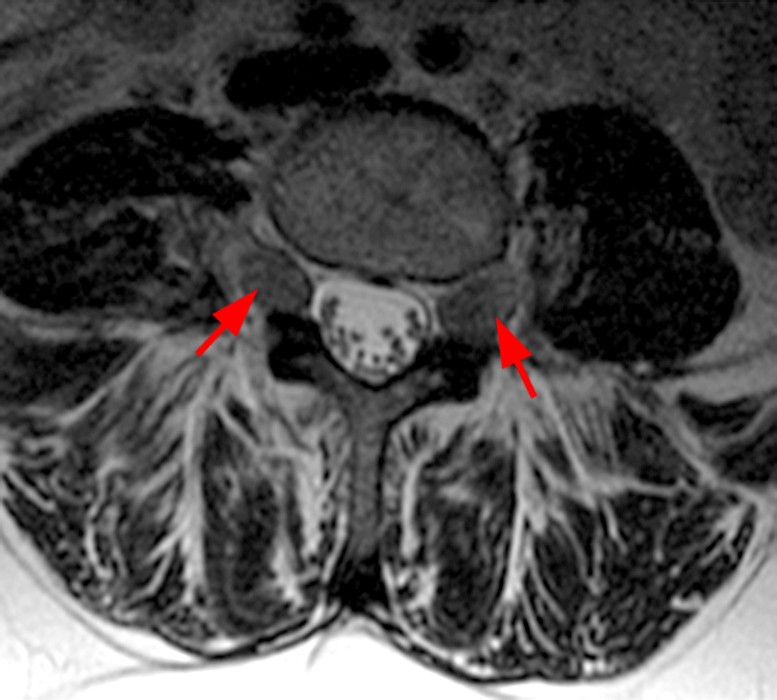
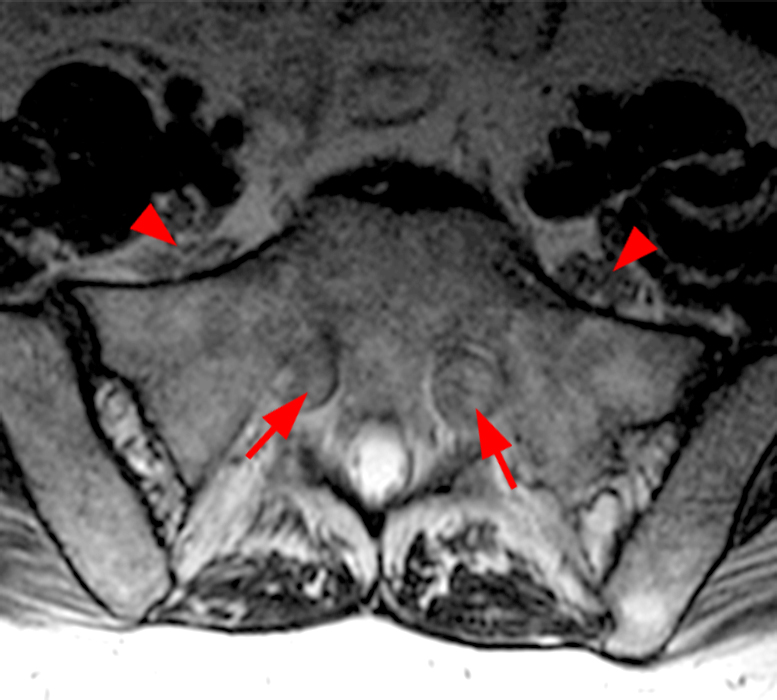
Figure 2:
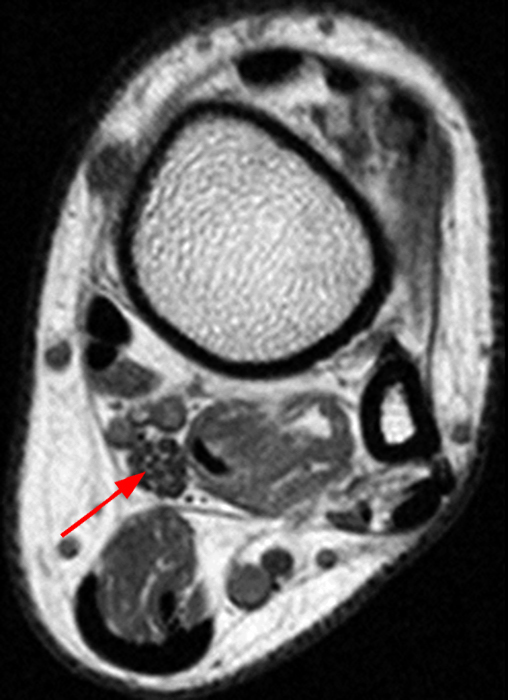
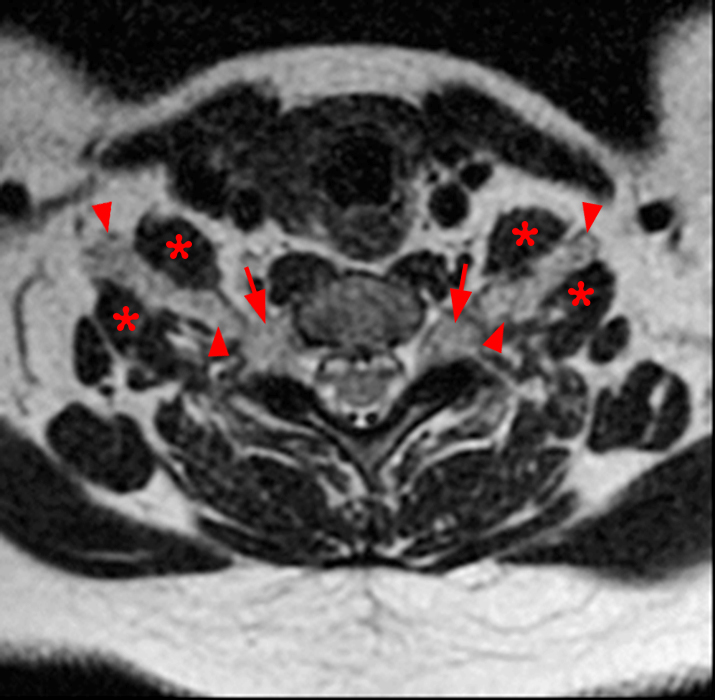
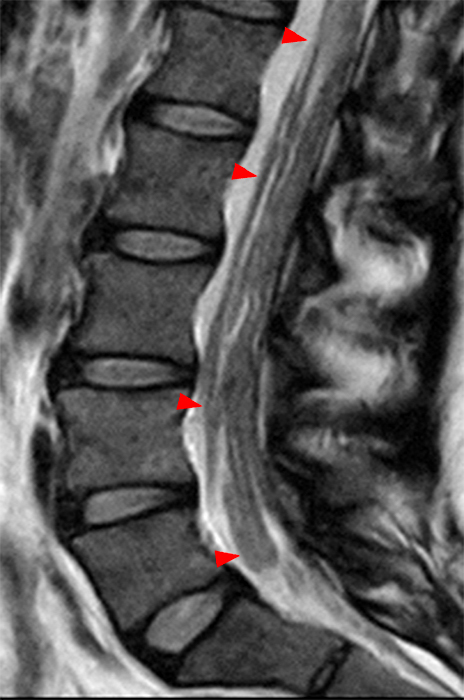
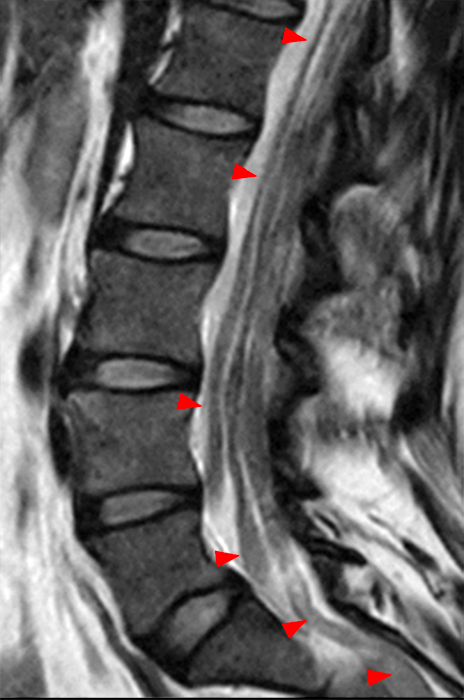
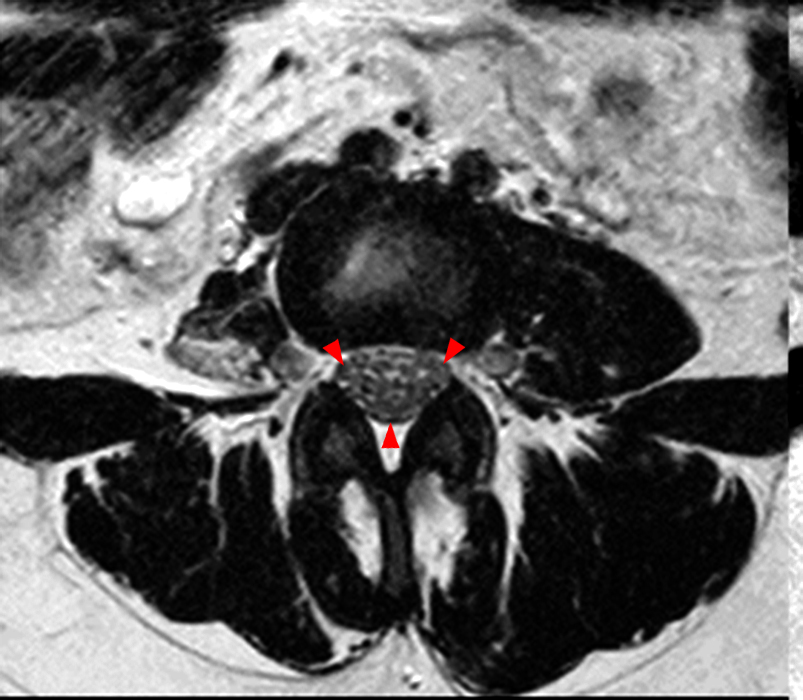
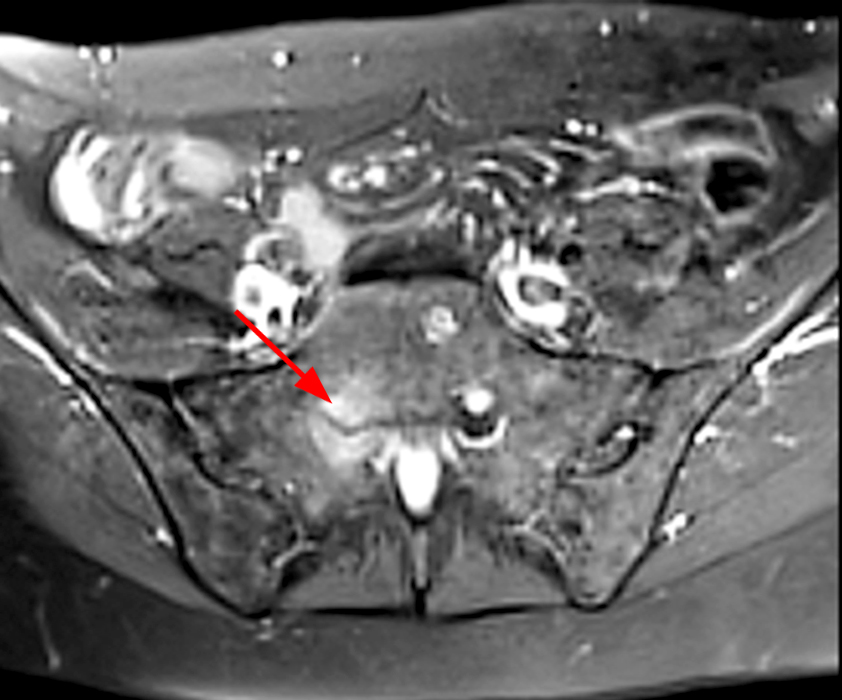
The sagittal T2-weighted image (2a) shows thickened cauda equina nerve roots (arrows) and the parasagittal T2-weighted image (2b) demonstrates enlarged L1-5 and S1-2 nerve roots (short arrows). Figure 2c, enlarged bilateral L4 nerve roots (short arrows) are seen at the L4-5 level. Figure 2d shows enlarged bilateral S1 sacral nerve roots (short arrows) within the neural foramina. Also note prominent right and left extra-foraminal L5 nerve roots (arrowheads) anterior to the sacral alae.
Diagnosis
Charcot-Marie-Tooth disease.
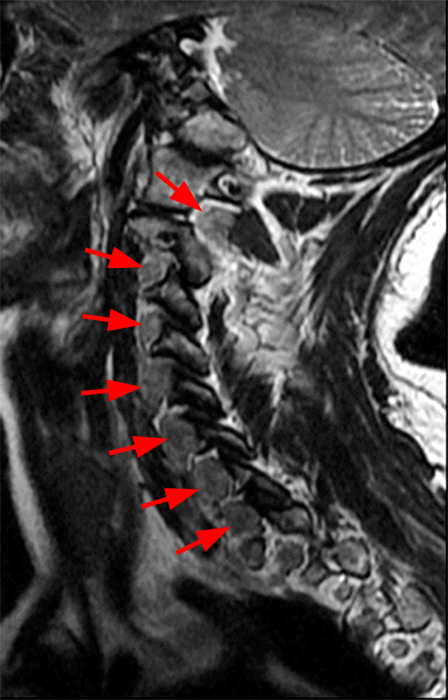
Figure 3:
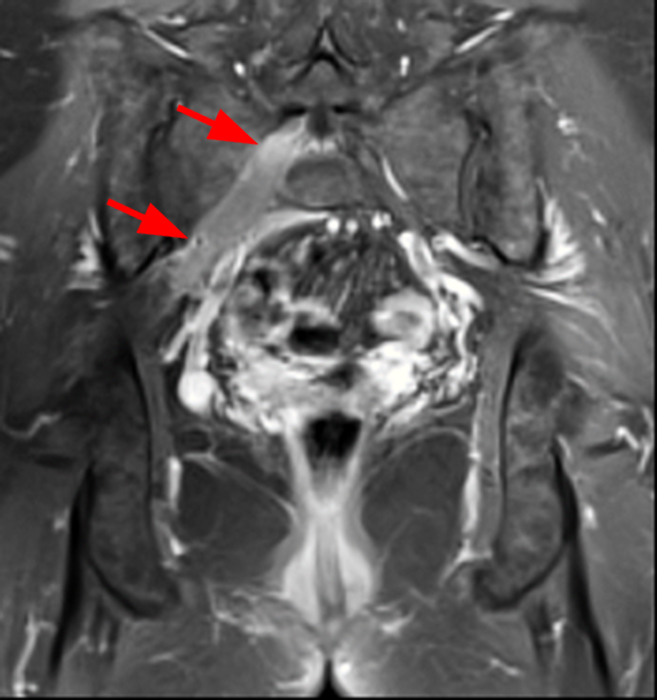
Parasagittal T2-weighted (3a) and parasagittal T2-weighted fat-suppressed (3b) images of the cervical spine of the test case patient reveal enlarged C2-T1 nerve roots (short arrows). An axial 2D MERGE sequence (3c) at the C6-7 level demonstrates enlarged bilateral C7 nerve roots at the dorsal root ganglia (short arrows) and enlarged bilateral extraforaminal C6 nerve roots (arrowheads). The internal jugular veins (blue asterisks) and common carotid arteries (red asterisks) are also seen at this level.
Introduction
Hypertrophic neuropathies include a variety of disorders with variable involvement of motor and sensory nerves. Charcot-Marie-Tooth disease (also known as hereditary motor and sensory neuropathy) is the most common inherited neuromuscular disease and Chronic Inflammatory Demyelinating Polyneuropathy (or Polyradiculoneuropathy) is likely the most recognized acquired immune-mediated peripheral neuropathy.1,2
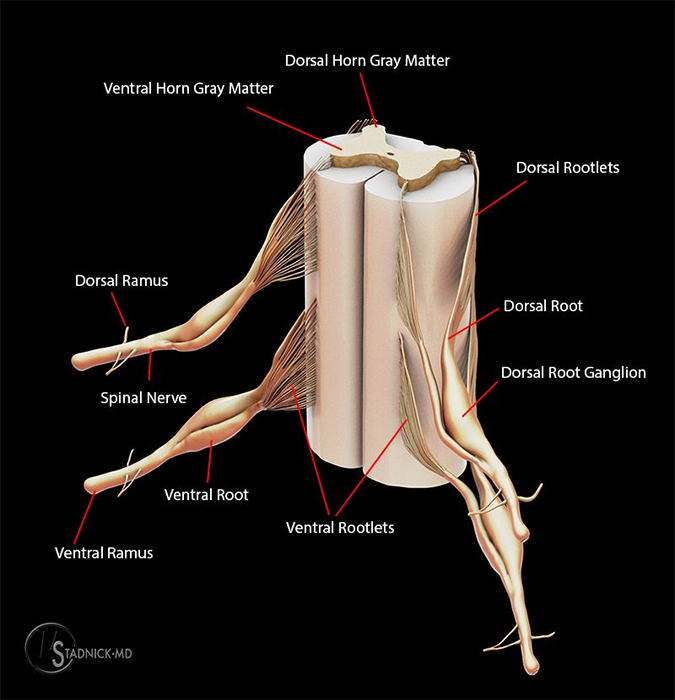
Relevant Anatomy
Spinal nerves extend from the spinal cord as rootlets to form ventral (anterior) and dorsal (posterior) nerve roots. Ventral roots carry efferent fibers from cell bodies located in the central gray matter in the spinal cord and consist of motor neurons innervating skeletal muscle and preganglionic autonomic neurons.3 Dorsal roots consist of afferent fibers carrying sensory information from the limbs and trunks to the central nervous system via cell bodies located in the dorsal root ganglion. Ventral and dorsal roots join just distal to the dorsal root ganglia, becoming the spinal nerve proper consisting of motor, sensory and autonomic fibers.3
The spinal nerve then branches into a larger ventral ramus and a smaller dorsal ramus. Ventral rami generally innervate the muscles and skin of the anterior trunk as well as the limbs whereas dorsal rami supply the musculature and skin of the back.3
Charcot-Marie-Tooth disease
Charcot-Marie-Tooth (CMT) disease is the most common hereditary neuromuscular disease affecting approximately 125,000 patients in the United States and a worldwide prevalence of approximately 1 in 2,500.4,5
CMT genetically and clinically resembles hereditary neuropathy with liability to pressure palsies (HNPP), hereditary sensory and autonomic neuropathies (HSAN) and distal hereditary motor neuropathies (dHMN).1 Nearly 100 different genes have been linked to CMT, HSAN and dHMN whereas a single gene (PMP22) is usually associated with HNPP.1 However, mutations in four genes (PMP22, GJB1, MPZ and MFN2) are disproportionately detected in patients with CMT.1 Approximately one third of CMT cases occur de novo and therefore many patients do not have a positive family history of disease.6 CMT primarily affects the peripheral nervous system, manifesting with chronic progressive motor deficits and sensory symptoms. Given the chronic nature of CMT, patients demonstrate muscle atrophy, typically beginning with the feet (resulting in pes cavus deformity) followed by hand involvement in the later stages of the disease.5 Sensory symptoms usually include loss of vibration and positional sense and subsequent loss of pain and temperature sensation.6
Figure 5:
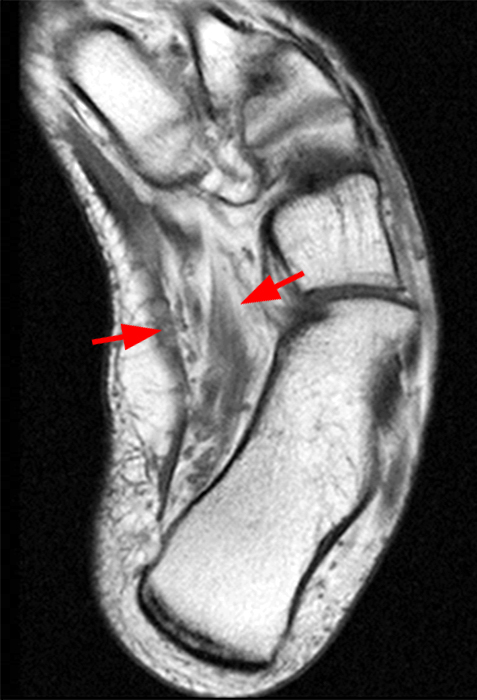
15-year-old male with a history of CMT presenting with left midfoot pain. An axial T1-weighted image (5a) at the level of the distal tibia (proximal to the tarsal tunnel) demonstrates an abnormally enlarged tibial nerve (arrow). A more distal axial T1-weighted image (5b) shows pes cavovarus deformity and atrophy of the medial musculature of the foot (short arrows).
Electrophysiological studies in patients with CMT aid in differentiating between two major subtypes: a demyelinating form (CMT1) which predominantly affects Schwann cells (typified by symmetrically slowed nerve conduction velocities) and an axonal form (CMT2) which directly targets axons (characterized by normal nerve conduction velocities but reduced compound muscle action potentials).1,6,7 Sural nerve biopsy may be performed if genetic testing fails to return a diagnosis in atypical cases of CMT or when a non-genetic inflammatory neuropathy is in the differential diagnosis.4 MRI is usually an adjunct to the diagnosis of CMT and is typically only positive in patients with the demyelinating form (CMT1). Representative MRI findings include partial or diffuse peripheral nerve thickening, iso- or hypointense nerves on T2-weighted images (occasionally with central T2 hyperintensity) and mild diffuse enhancement.8 Treatment for CMT is predominantly supportive and includes orthotics and rehabilitation to reduce contractures and improve mobility and gait.9 Pain management typically consists of nonsteroidal anti-inflammatory medications or antiepileptic agents for neuropathic pain.6,10
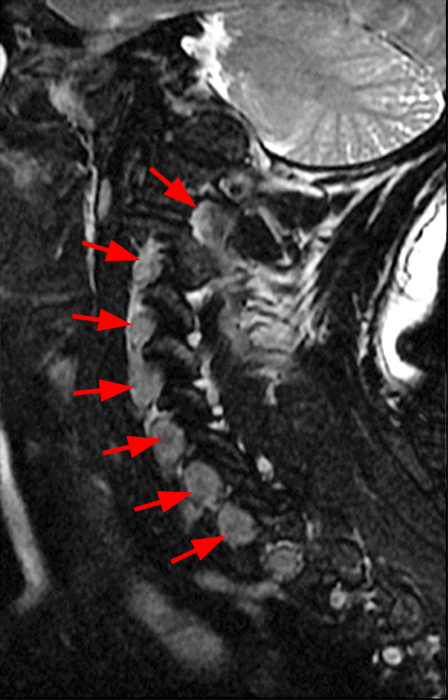
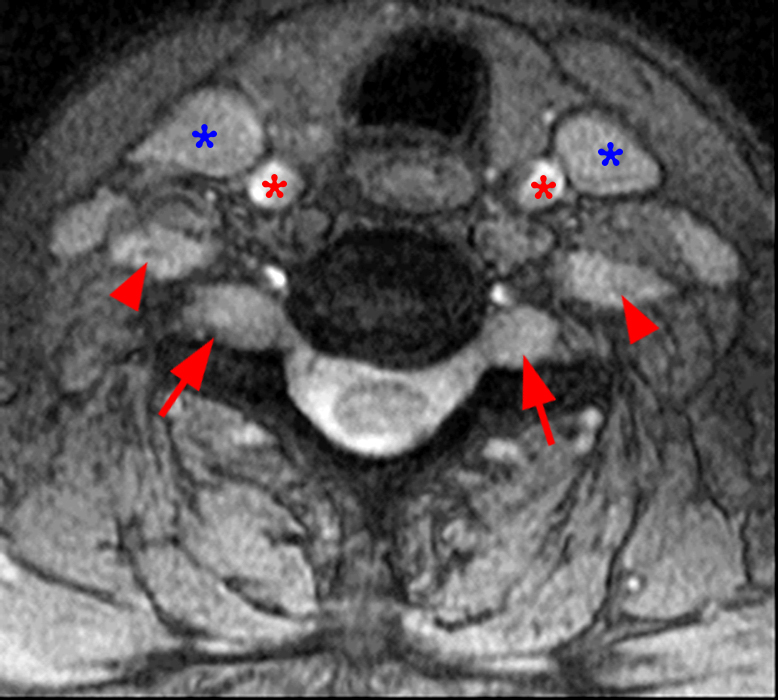
Figure 6:
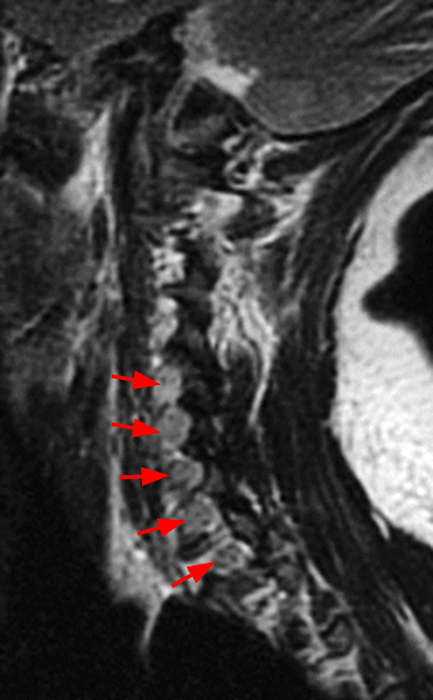
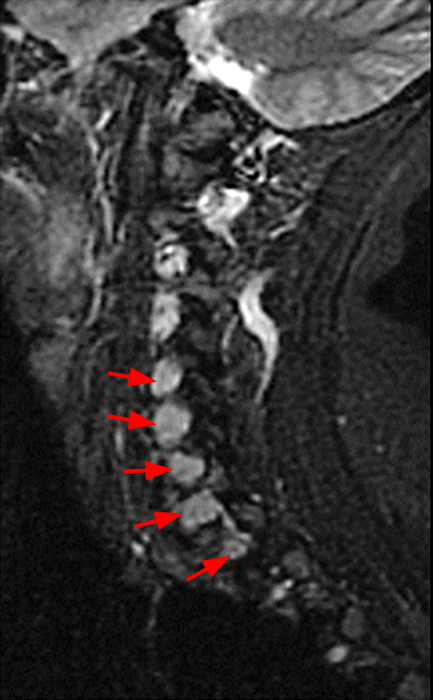
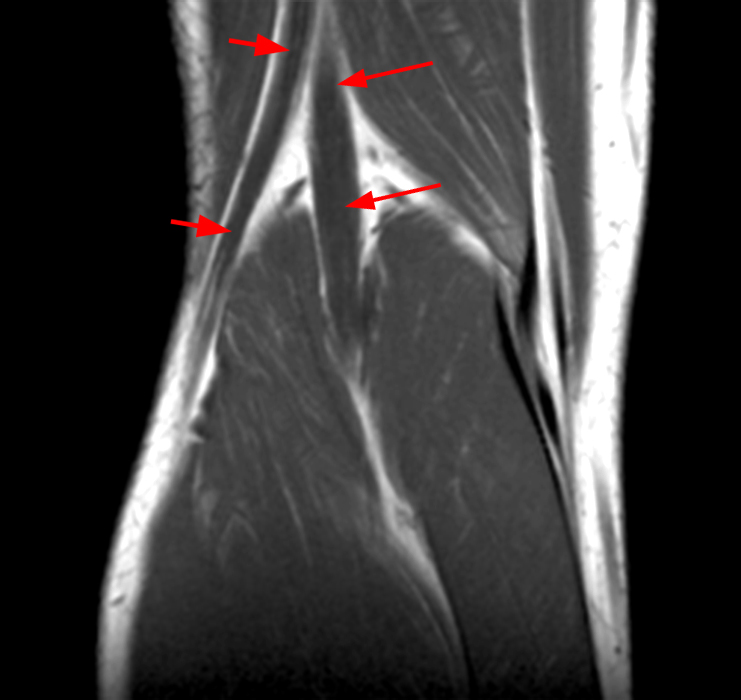
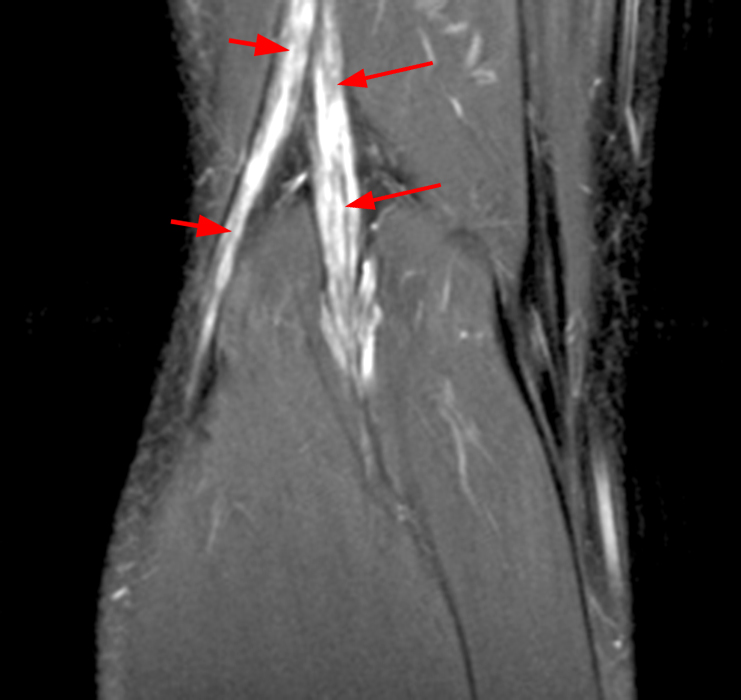
44-year-old female with a history of CMT who presents with worsening neck pain radiating to the shoulders. Parasagittal T2-weighted (6a) and Fast Inversion Recovery (6b) sequences through the cervical spine show thickened C5 through C8 and T1 nerve roots (short arrows). An axial T2-weighted image (6c) at C7-T1 demonstrates enlarged bilateral C8 roots at the level of the dorsal root ganglia (short arrows) and enlarged bilateral C7 brachial plexus nerves roots (arrowheads) coursing between the anterior and middle scalene muscles (asterisks).
Chronic Inflammatory Demyelinating Polyneuropathy
Chronic inflammatory demyelinating polyneuropathy (CIDP) is estimated to affect 1 to 2 per 100,000 adults and is one of the most prevalent acquired immune-mediated peripheral neuropathies.11, 12 CIDP manifests as progressive symmetric weakness in proximal and distal muscles and sensory ataxia.2 Diagnostic criteria for CIDP include motor or sensory dysfunction involving more than one limb and reduced or absent reflexes for greater than 8 weeks.11,13 In addition to “classic” CIDP, other subtypes exist that present with multifocal or selective involvement of motor and sensory nerve fibers. CIDP is a progressive disease with a prolonged time course distinguishing it from Guillain-Barré syndrome (which is self-limited).11 With Guillain-Barré syndrome, symptoms rapidly increase with peak deficits usually reached within one month followed by gradual improvement (although long term deficits can persist in some patients).2,13 Unlike CIDP, a preceding infection is typically identified in patients with Guillain-Barré syndrome. Electrophysiological studies of CIDP patients reveal slowed nerve conduction velocities with unequal multifocal demyelination (rather than uniform demyelination seen with CMT1 patients). Upper extremity sensory responses are typically affected more than lower extremity responses.2 Nerve biopsies reveal segmental demyelination and subsequent remyelination by Schwann cells resulting in an “onion bulb” appearance.14 MRI may reveal normal caliber or enlarged nerve roots (particularly of the brachial plexus or cauda equina). Even in patients with normal nerve root caliber on imaging, increased STIR (short tau inversion recovery) signal intensity is usually identified.15 T2/STIR signal hyperintensity of the brachial plexus is seen in approximately 50% of patients with CIDP.16 Gadolinium enhancement is more common in patients with nerve root hypertrophy on MRI and suggests active disease.17 Although central nervous system involvement is rare on imaging, MRI may reveal demyelinating lesions in the brain.18
Treatment for CIDP is primarily focused on reducing inflammation to prevent further demyelination and axonal degeneration. Mainstay therapies include corticosteroids, intravenous immune globulin (IVIG) and plasma exchange.19,20,21 Physical and occupational therapy are also employed to improve strength and mobility. In addition to Guillain-Barré syndrome and CMT1, other demyelinating neuropathies in the differential diagnosis for CIDP include monoclonal gammopathy of undetermined significance (MGUS), human immunodeficiency virus-associated neuropathy, diabetic neuropathy, osteosclerotic myeloma, lymphoma, sarcoidosis, Castleman disease and POEMS syndrome (polyneuropathy, organomegaly, endocrinopathy, M-protein, skin changes).2,11,22,23
Figure 8:
51-year-old female with biopsy proven sarcoid neuritis. An axial T1-weighted image (8a) and an axial T2-weighted image with fat-suppression (8b) demonstrate an enlarged right S1 nerve root within the neural foramen (arrows). Associated nerve root enhancement (short arrows) is seen on corresponding axial and coronal T1-weighted post contrast images with fat-suppression (8c-d). Images courtesy of David Rubin, M.D.
Figure 9:
Coronal T1 and T2-weighted images (9a-b) of the posterior knee in an asymptomatic male show enlarged tibial (arrows) and common peroneal (short arrows) nerves. Given absence of clinical symptoms, this may represent a normal variant versus a subclinical hypertrophic neuropathy. Images courtesy of David Rubin, M.D.
Conclusion
Charcot-Marie-Tooth disease and chronic inflammatory demyelinating polyneuropathy are two of the most widely recognized hypertrophic peripheral neuropathies. CMT has a genetic pathogenesis whereas CIDP is an acquired, immune-mediated disease. MRI is a valuable adjunct to support the clinical diagnosis of CMT, CIDP or similar hypertrophic peripheral neuropathies.
References
- Rudnik-Schöneborn S, Auer-Grumbach M, Senderek J. Charcot-Marie-Tooth disease and hereditary motor neuropathies. Medizinische Genetik. 2020;32(3):207-219. ↩
- Dyck PJB, Tracy JA. History, Diagnosis, and Management of Chronic Inflammatory Demyelinating Polyradiculoneuropathy. Mayo Clin Proc. 2018;93(6):777-793. ↩
- Gorman N, Mytilinaios D. (Last reviewed 2021, August 30). Spinal nerves: Anatomy, roots and function. Retrieved from https://kenhub.com. ↩
- Barreto LC, Oliveira F, Nunes P et al. Epidemiologic study of Charcot-Marie-Tooth disease: a systematic review. Neuroepidemiology. 2016;46:157–65. ↩
- Jani-Acsadi A, Krajewski K, Shy ME. Charcot–Marie–Tooth neuropathies: diagnosis and management. Semin Neurol. 2008;28:185–194. ↩
- Szigeti K, Lupski JR. Charcot-Marie-Tooth disease. Eur J Hum Genet. 2009;17(6):703-710. ↩
- De Jonghe P, Timmerman V, Van Broeckhoven C. 2nd workshop of the European CMT Consortium: 53rd ENMC International Workshop on classification and diagnostic guidelines for Charcot-Marie-Tooth type 2 (CMT2-HMSN II) and distal hereditary motor neuropathy (distal HMN – spinal CMT). Neuromuscul Disord. 1998;8:426–31. ↩
- Tatewaki Y, Nishiyama S, Kato Y, Murata T, Mugikura S, Li L, Mata Mbemba D, Takahashi S. MR Imaging of the Cauda Equina in Charcot-Marie-Tooth disease. Poster presented at: ECR 2015/C-0741. ↩
- Dimitrova EN, Božinovikj I, Ristovska S, Pejcikj AH, Kolevska A, Hasani M. The Role of Rehabilitation in the Management of Patients with Charcot-Marie-Tooth Disease: Report of Two Cases. Open Access Maced J Med Sci. 2016;4(3):443-448. ↩
- Backonja MM. Use of anticonvulsants for treatment of neuropathic pain. Neurology. 2002;59:S14–S17. ↩
- Köller H, Kieseier B, Jander S, Hartung H-P. Chronic Inflammatory Demyelinating Polyneuropathy. The New England journal of medicine. 2005;352:1343-56. ↩
- Lunn M, Manji H, Choudhary P, Hughes R, Thomas P. Chronic inflammatory demyelinating polyradiculoneuropathy: a prevalence study in south east England. J Neurol Neurosurg Psychiatry. 1999;66:677-80. ↩
- Odaka M, Yuki N, Hirata K. Patients with chronic inflammatory demyelinating polyneuropathy initially diagnosed as Guillain-Barré syndrome. J Neurol. 2003;250(8):913-916. ↩
- Krendel D, Parks H, Anthony D, St. Clair M, Graham D. Sural nerve biopsy in chronic inflammatory demyelinating polyradiculoneuropathy. Muscle & Nerve. 1989;12(4), 257–264. ↩
- Tanaka K, Mori N, Yokota Y, Suenga T. MRI of the cervical nerve roots in the diagnosis of chronic inflammatory demyelinating polyradiculoneuropathy: a single-institution, retrospective case–control study. BMJ Open. 2013;3:e003443. ↩
- Molenaar DS, Vermeulen M, de Haan R. Diagnostic value of sural nerve biopsy in
chronic inflammatory demyelinating polyneuropathy. J Neurol Neurosurg Psychiatry. 1998;64:84-9. ↩ - Duggins A, McLeod J, Pollard J, Davies L, Yang F, Thompson E, Soper J. Spinal root and plexus hypertrophy in chronic inflammatory demyelinating polyneuropathy. Brain. 1999;122-7 (7)1383–1390. ↩
- Stojkovic T, de Seze J, Hurtevent J, et al. Visual evoked potentials study in chronic idiopathic inflammatory demyelinating polyneuropathy. Clin Neurophysiol. 2000;111:2285-91. ↩
- Hughes R, Bensa S, Willison H, et al. Randomized controlled trial of intravenous immunoglobulin versus oral prednisolone in chronic inflammatory demyelinating polyradiculoneuropathy. Ann Neurol. 2001;50:195-201. ↩
- Hahn AF, Bolton CF, Zochodne D, Feasby TE. Intravenous immunoglobulin treatment in chronic inflammatory demyelinating polyneuropathy: a double-blind, placebo-controlled, cross-over study. 1996;119:1067-77. ↩
- Mendell JR, Barohn RJ, Freimer ML, et al. Randomized controlled trial of IVIg in untreated chronic inflammatory demyelinating polyradiculoneuropathy. Neurology. 2001;56:445-9. ↩
- Gorson KC, Ropper AH, Adelman LS, Weinberg DH. Influence of diabetes mellitus on chronic inflammatory demyelinating polyneuropathy. Muscle Nerve. 2000;23:37-43. ↩
- Haq RU, Pendlebury WW, Fries TJ, Tandan R. Chronic inflammatory demyelinating polyradiculoneuropathy in diabetic patients. Muscle Nerve. 2003;27:465-70. ↩