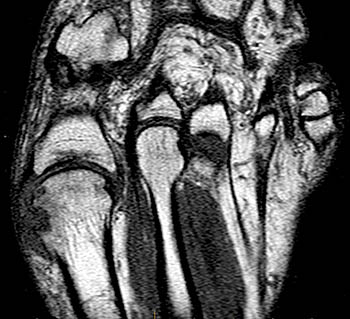
Clinical History: A 55 year-old male complains of chronic swelling and pain in the foot with inability to walk on concrete and waking at night, without relief from medication. Axial long axis T1-weighted (1a) and coronal short axis fat suppressed T2-weighted (1b) images through the forefoot are presented. What are the findings? What is your diagnosis?
Findings
Figure 2:
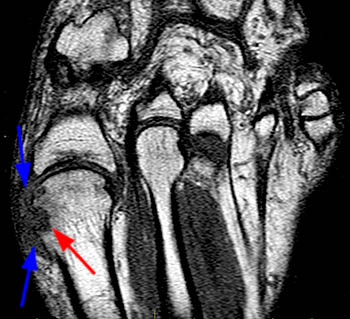
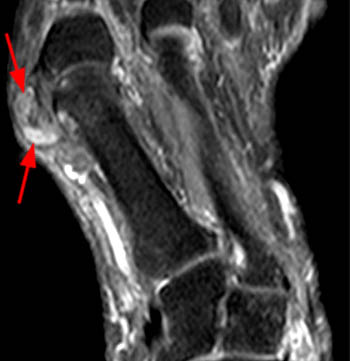
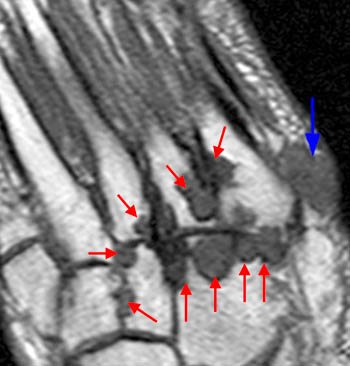
The T1-weighted long axis image reveals a well defined, marginal erosion (red arrow) with overhanging edges at the head of first metatarsal, surrounding by intermediate signal within the adjacent soft tissues (blue arrows) denoting a gouty tophus. Note the relative preservation of the joint space.
Diagnosis
First metatarsal and medial hallux sesamoid erosions with surrounding marrow edema, consistent with gout.
Introduction
Gout is a metabolic condition caused by an elevation in uric acid in the bloodstream, occurring via altered purine metabolism. The hyperuremic state may then lead to increased deposition of monosodium urate crystals in and around joints, within osseous structures, or within soft tissues. Gout is the most common form of crystalline arthritis.
Pathogenesis
Historically gout was divided into primary and secondary forms. Due to overlap in metabolic defects within the groups, a more accurate system has been derived characterizing gout into idiopathic, secondary, and drug-induced forms1. The causative factor in idiopathic gout is typically unknown (75-90 percent) but in most cases is felt to result from an unexplained impairment of uric acid excretion by the kidneys2. The remaining cases of idiopathic gout may occur due to an inborn error in purine metabolism involving its synthesis/overproduction, catabolism, or interconversion. Myeloproliferative disorders such as leukemia and lymphoma, chemotherapeutic agents, hemoglobinopathies, alcohol, chronic renal failure, dehydration, and diabetes have all been implicated in secondary gout.
In humans, the end product of purine metabolism is urate. Other mammals have the capability to oxidize urate to allantoin, which is more soluble in the bloodstream. When the local solubility limits of uric acid are exceeded in humans, monosodium urate crystal deposition occurs3. A serum urate level of approximately 6.8 mg/dl is the concentrate at which the crystals begin to precipitate and the higher the level, the more likely the crystal deposition is to occur4. This supersaturate can precipitate in the kidneys as well as in the cartilage, synovial membranes, and synovial fluid, initiating the inflammatory response seen in gouty arthritis5. Supersaturate can also precipitate into the periarticular soft tissues, leading to the typical gouty tophus.
Epidemiology
Roughly 15-25 percent of patients with hyperuricemia develop gout. Gout accounts for 5 percent of all arthritis and is found in 0.3 percent of the population, affecting males significantly more often (M:F ratio 20:1)6. In females, it typically occurs postmenopausal and in children is manifest as the inherited disorder, Lesch-Nyhan syndrome. Areas of mechanical stress/irritation are common sites of crystal deposition and these crystals also more commonly develop with lower body temperatures7. There is an association with psoriasis, and classically this condition has a negative association with rheumatoid arthritis. Gout is less common in black patients, and has a familial prevalence.
Clinical Presentation
Most patients present between the ages of 30-60, most commonly within the fifth decade of life. Most patients with hyperuricemia are asymptomatic, but crystals released into the joint space can cause acute symptomatic attacks.
In acute gouty arthritis, an inflammatory cascade occurs. The crystals are phagocytized, IL-1 beta is released and stimulates the release of chemokines, cytokines, prostaglandins, and other proinflammatory molecules8. This in turn stimulates neutrophil influx into the joint. Early in the disease, the disorder is typically monoarticular with the first MTP joint being the most common site of involvement. Clinically, patients develop pain, swelling and erythema around the involved joint. Gout can cause acute bursitis or tenosynovitis and can cause a high fever, resembling septic arthritis. Skin desquamation can occur at the affected location. Attacks typically last for 24-48 hours and usually spontaneously resolve within 5-7 days. These attacks can be precipitated by alcohol, diuretics, trauma, purine-rich foods such as meat and shellfish, and drugs such as cyclosporine to name a few9. The crystals remain within the joint, however, predisposing to subsequent attacks due to chronic low grade inflammation10 during the intercritical (between attacks) phase of the disease.
Progressive disease can ensue and patients can develop chronic gout. This stage of the disease is characterized by deposition of monosodium urate crystals, known as tophi. These tophi typically present later in the course of the disease, but rare cases of tophi presenting in acute disease have been described, making differentiation from neoplasm difficult11. Patients have usually had gout for 10-12 years before these lesions become visible. There is typically increased vascularity and inflammatory reaction in the surrounding soft tissues. Left untreated, this stage of disease can be debilitating and cause extensive morbidity.
Imaging
Radiographic changes in gout are typically not present for 6-8 years. During the acute phase of the disease, only asymmetric soft tissue swelling may be present around the affected joint on imaging studies. With resolution of the inflammation, there is reversion back to normal with no evidence of disease on plain films. Recently, however, new evidence supports the use of more advanced techniques such as MRI during the intercritical period between attacks. It has been shown that a large percentage of patients with gout and normal plain radiographs have occult destructive arthropathy that is only detected by advanced imaging such as MRI and ultrasound, with approximately half of the patients demonstrating early bone erosions and synovial pannus12.
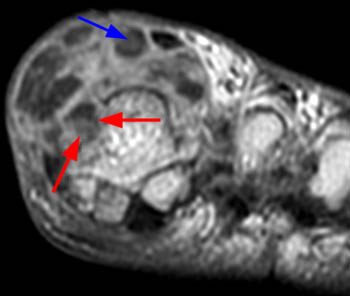
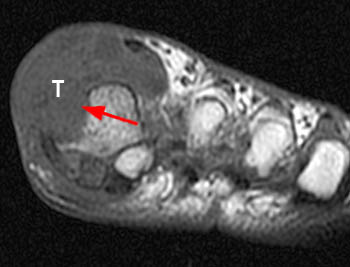
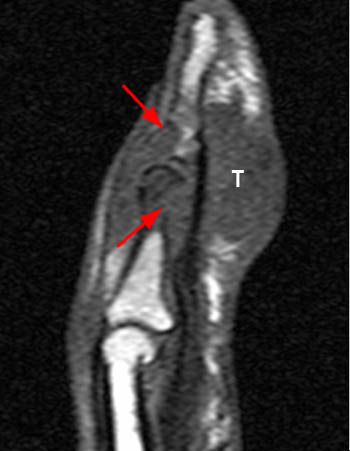
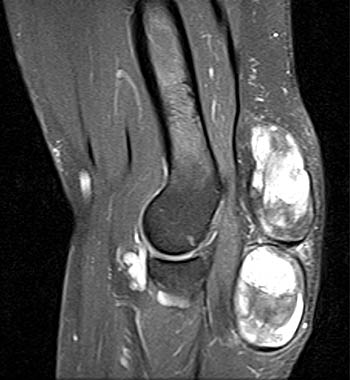
The chronic phase of gout, the tophaceous period, has several radiological hallmarks that are visualized due to the deposition of the monosodium urate crystals. The classic findings by plain film include tophi, “punched out” erosions with overhanging cortex and sclerotic margins, normal bone mineralization, and preservation of the joint spaces (4a). The overhanging edge, present in roughly 40 percent of patients, is due to bone resorption from the enlarging tophus13. The tophus, typically radiodense without or with calcification on x-ray, appears on MRI as a predominately intermediate signal mass on T1 weighted images, with variable signal on T2-weighting, depending on the calcium concentration within a tophus (5a). Tophi demonstrate fairly homogeneous enhancement after administration of IV contrast (6a)14, though this is not always the case (7a). The tophi may be intraarticular, extraarticular, or intraosseous in location (8a). The latter are typically metaphyseal and periosteal involving the long bones, with expansion and cystic changes occurring, sometimes with small circular or punctate calcifications. Osteochondral pathological fractures can occur from the bone weakening. This can then produce “cupping” from collapse of the subchondral bone15. Other late stage phenomena, such as “mushrooming” from enlargement of the ends of bone due to new bone formation, or “penciling-in” due to tapering of the shaft from osteolysis, have been described but are less common due to advances in treatment.
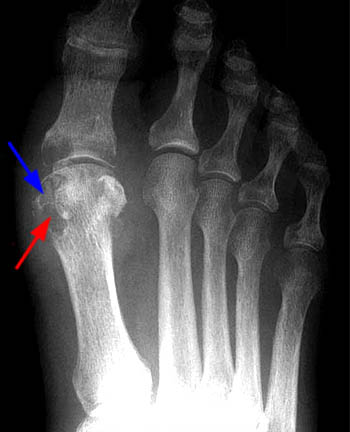
Figure 4:
AP radiograph of the foot demonstrating the radiographic hallmark findings of chronic gout. The classic marginal erosions (red arrow), overhanging cortex (blue arrow), preservation of bone density, and maintenance of the joint space are apparent, in addition to hallux valgus, which is commonly associated.
Figure 7:
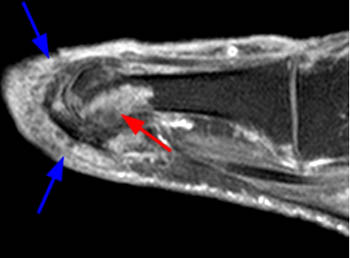
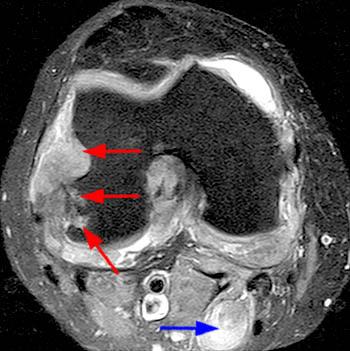
Short axis post contrast T1-weighted image through the foot reveals a large heterogeneously enhancing tophus within the dorsal soft tissues overlying the first metatarsal head with associated osseous erosion (red arrows). Some central areas of the tophus do not readily enhance (blue arrow), though this remains more the exception than the rule.
Figure 8:
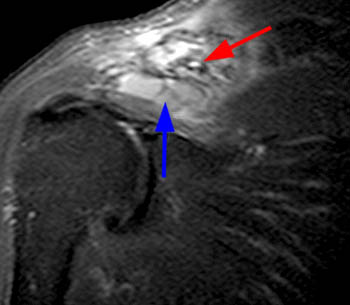
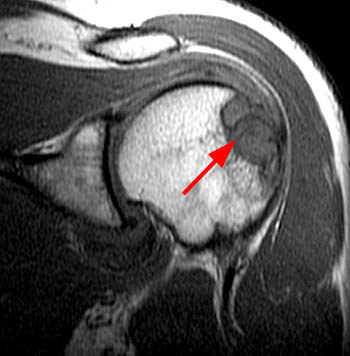
Fat suppressed T2-weighted image through the right shoulder demonstrating a biopsy proven case of distal clavicle gout with marrow replacement suggesting intraosseous disease. A marginal erosion (red arrow) is seen along the undersurface of the clavicle with adjacent extraarticular soft tissue tophus (blue arrow).
Anatomical Sites of Involvement
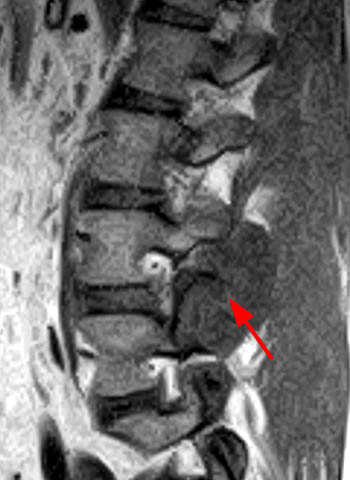
Gout is an asymmetrical polyarticular disease, most often affecting the foot (9a). There is a predilection for the first metatarsophalangeal (MTP) joint, a condition known as podagra. Erosions are typically frequent along the medial and dorsal aspect of the joint (10a), and hallux valgus deformity is often seen, though any joint can be affected. The hindfoot can also be involved. Calcaneal erosions are common, and there can be retrocalcaneal bursitis and Achilles tendon involvement (11a)16. The hands and wrist are the next most common area of involvement (12a) with elbow (13a) and knee involvement being somewhat common (14a). Less common sites of disease include the shoulder (15a), hip, sacroiliac joints and the spine (16a).
Treatment
Diagnosis is confirmed by the presence of monosodium urate crystals in synovial fluid, or within tophi, though patients can be empirically treated in appropriate clinical scenarios. Serum uric acid measurements are not utilized in diagnosis of acute disease, because these are often normal during acute attacks17.
The acute phase of the disease in which inflammation and swelling is most common is treated first by nonsteroidal anti-inflammatories or corticosteroids, with opioids used only if necessary to control pain. Risk factor modification pertaining to diet and alcohol is also important. Pharmacotherapy utilizing urate-lowering drugs such as xanthine oxidase inhibitors or uricosuric agents is recommended for patients with more than two gouty attacks per year, in patients with tophi, and in patients with joint damage seen on a radiograph18. Effective treatment in lowering the serum urate levels has been associated with the dissolving of tophi and dissolution of crystals from synovial fluid19. Hence, further attacks may be obviated and structural damage to the joints may be less severe.
Differential Diagnosis
Calcium pyrophosphate deposition disease (CPPD), also known as pseudogout, can manifest in the same way as gout clinically, but may be differentiated by the presence of chondrocalcinosis, the predilection for the hands, wrist and knee, and the lack of soft tissue masses seen in gout. There is some overlap, with 40 percent of gout sufferers also suffering from CPPD. Rheumatoid arthritis can present clinically similar, but this is a symmetrical polyarticular disease producing fusiform soft tissue swelling, without sclerosis along the margins of the erosions that occur. Erosions are only intraarticular in location with rheumatoid. Septic arthritis can also present in a similar fashion to gout, but easy differentiation can be made at the time of aspirate analysis, by the recognition of needle-shaped crystals with negative birefringence in gout. Psoriatic arthritis can also present similarly, but may be differentiated by the more extensive involvement of the sacroiliac joints and paravertebral ossification. Mimickers of the gouty tophi are rheumatoid nodules and xanthomas.
Conclusion
Gout is the most common crystalline arthropathy, responsible for extensive damage to the soft tissues and osseous structures if left unrecognized and untreated. Typically the diagnosis of gout is known at the time of radiographic analysis, but early treatment and recognition is vital to prevent advanced destruction of the joints, soft tissues, and osseous structures. Gout may be confused clinically with neoplasm, infection, and other arthritides. In such cases, characteristic findings on MR assist in making the correct diagnosis. It is important for radiologists to remember the old adage, “when in doubt think gout.”
References
1 Seegmiller JE: Diseases of purine and pyrimidine metabolism in PK Bondy, LE Rosenberg (Eds): Metabolic Control and Disease. 8th Ed. Philadelphia, WB Saunders Co, 1979 p. 780.
2 Schiller Allen in Pathology, second edition. Rubin E, Farber JL. Chapter 26 Bones and Joints. P 1335.
3 Eggebeen, AT Gout, An Update. American Academy of Family Physicians. 76(6), Sep 2007 p. 801-808.
4 Schumacher HR. The pathogenesis of gout. Cleveland Clinic Journal of Medicine. 75(5) July, 2008.
5 Stern HJ and Finkelstein JD: Heritable diseases of amino acid metabolism in Becker KL et al (Eds): Principles and Practice of Endocrinology and Metabolism 2nd Ed. Philadelphia. JB Lippincott Company, 1995. p. 1596.
6 Brower AC, Flemming DJ. Arthritis in Black and White. 2nd Ed. Philadelphia: Saunders an Imprint of Elsevier; 1997 p. 325.
7 Loeb, JN. The influence of temperature on the solubility of monosodium urate. Arthritis Rhuem 1972; 15:189-192.
8 Chen CJ, Shi Y, Hearn A et al. MyD88-dependent IL-1 receptor signaling is essential for gouty inflammation stimulated by monosodium urate crystals. J Clin Invest 2006; 116:22622271.
9 Choi HK, Atkinson K, Karlson EW, et al: Alcohol intake and risk of incident gout in men: A prospective study. Lancet. 2004, 363: 1277-1281.
10 Pascual E, Pedraz T. Gout Curr Opin Rheumatol 2004; 16: 282-286.
11 Shmerling RH, Stern SH. Gravallese EM, Kantrowitz FG,. Tophaceous deposition in the finger pads without gouty arthritis. Arch Intern Med 1988: 1830-1832.
12 Carter JD, Kedar RP, Anderson SR, et al. An analysis of MRI and ultrasound imaging in patients with gout who have normal plain radiographs. Rheumatology. 2009; 48:1442-1446.
13 Resnick D, Kransdorf MJ. Bone and Joint Imaging. 3rd Ed. Philadelphia, PA: Elselvier-Saunders; 2005.
14 Yu JS, Chung C, Recht M et al. MR imaging of tophaceous gout. AJR 1997; 168: 523-527.
15 Bloch C, Hermann G, Yu TF. A radiological reevaluation of gout: a study of 2000 patients. AJR 134: 781, 1980.
16 Berquist TH. Imaging of the Foot and Ankle, 3rd Ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2011.
17 McCarty DJ. Gout without hyeruricemia. JAMA 1994; 271: 302-303.
18 Zhang W, Doherty M, Bardin T et al. EULAR evidence based recommendations for gout. Part II: management. Report of a task force of the EULAR Standing Committee for International Clinical Studies Including Therapeutics (ESCISIT). Ann Rheum Dis 2006; 65: 1312-24.
19 Perez-Ruiz F, Calabozo M, Pijian JI et al. Effect of urate-lowering therapy on the velocity of size reduction of tophi in chronic gout. Arthritis Rheum 2002; 47: 356-360.