Clinical History:
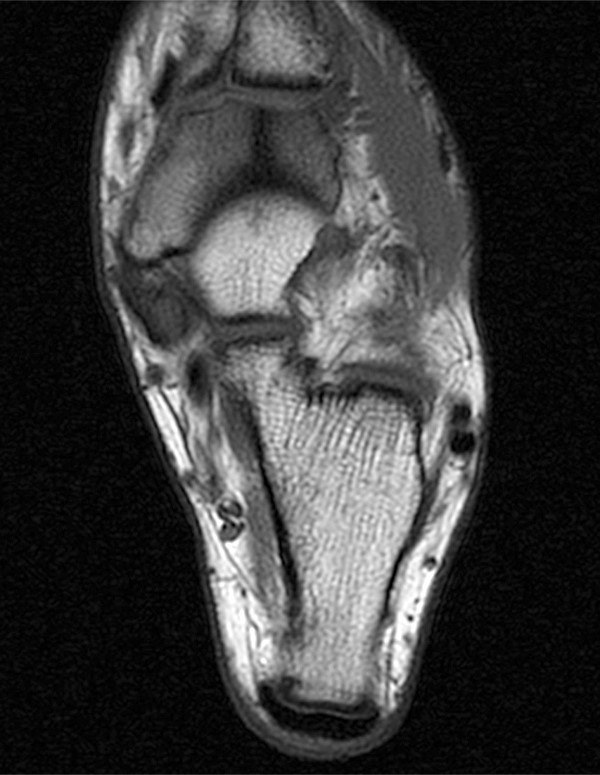
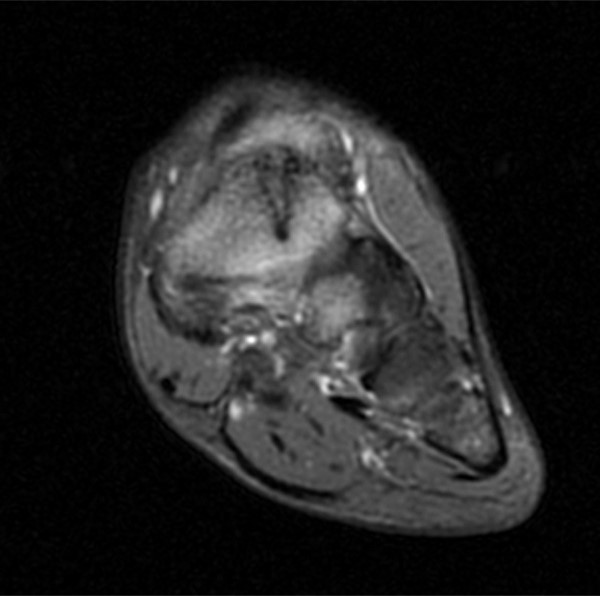
A 17 year-old female presents with three months of anterior ankle pain. (1a) Axial T1-weighted and (1b) coronal proton-density weighted fat-suppressed images are provided. What are the findings? What is your diagnosis?
Findings:
Figure 2:
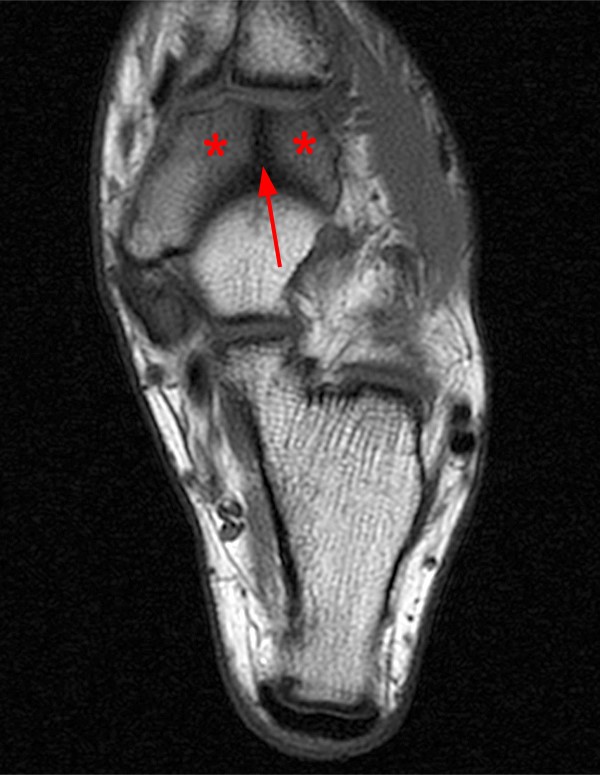
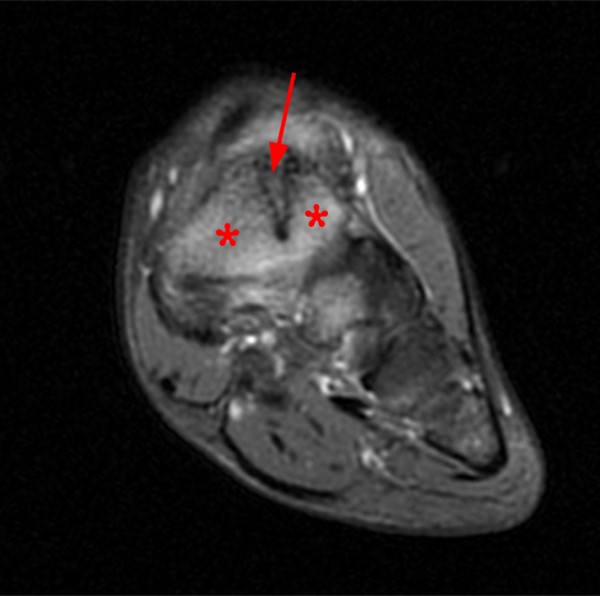
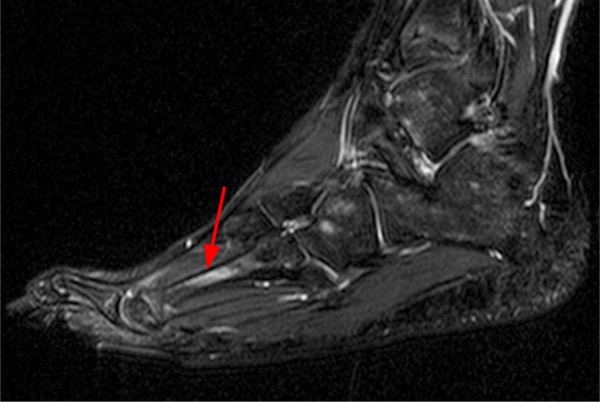
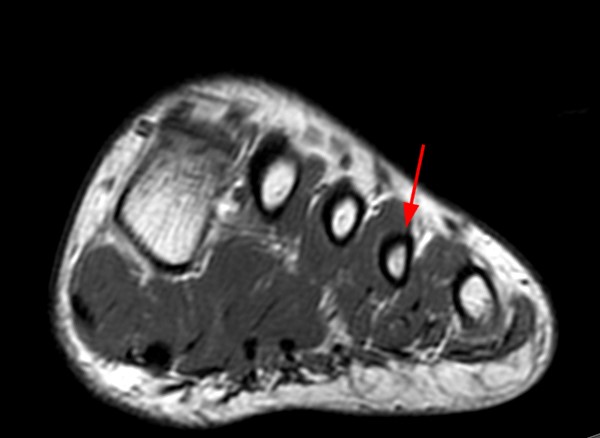
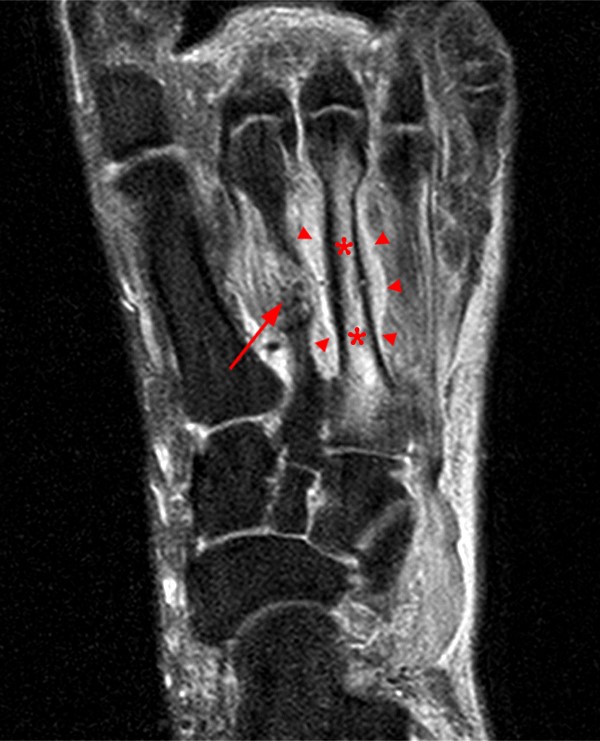
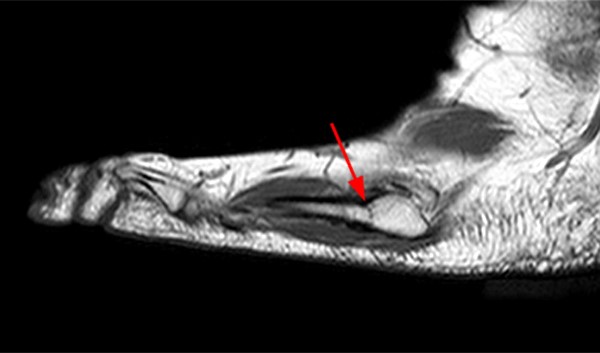
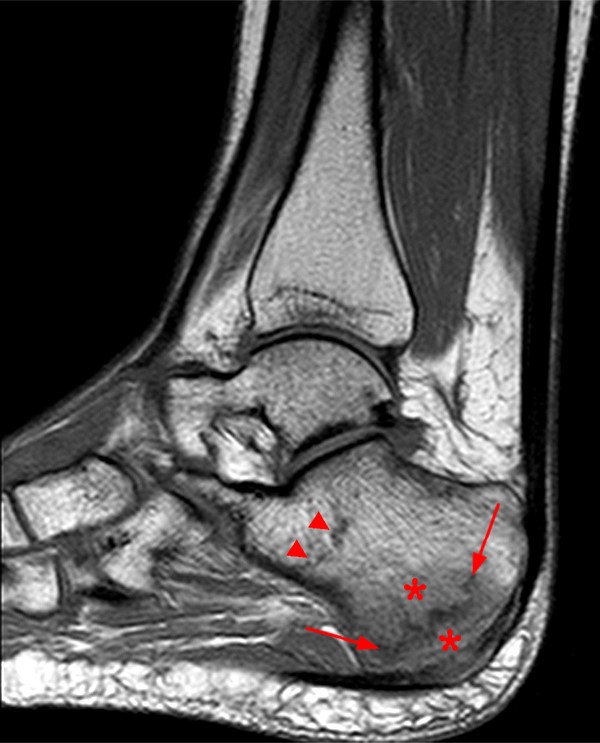
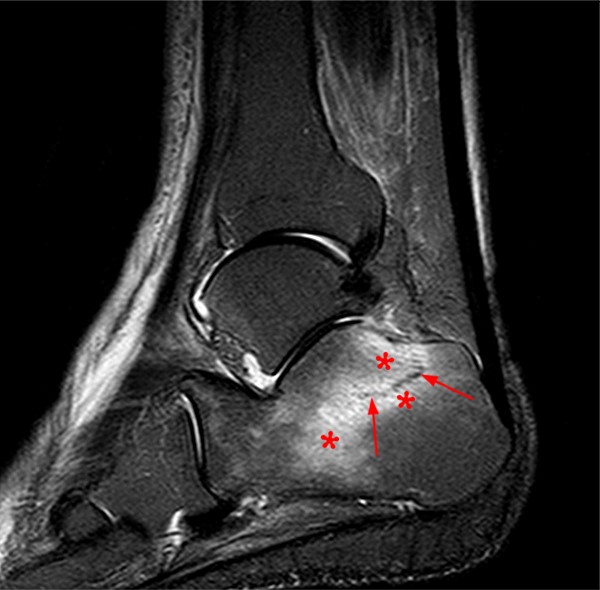
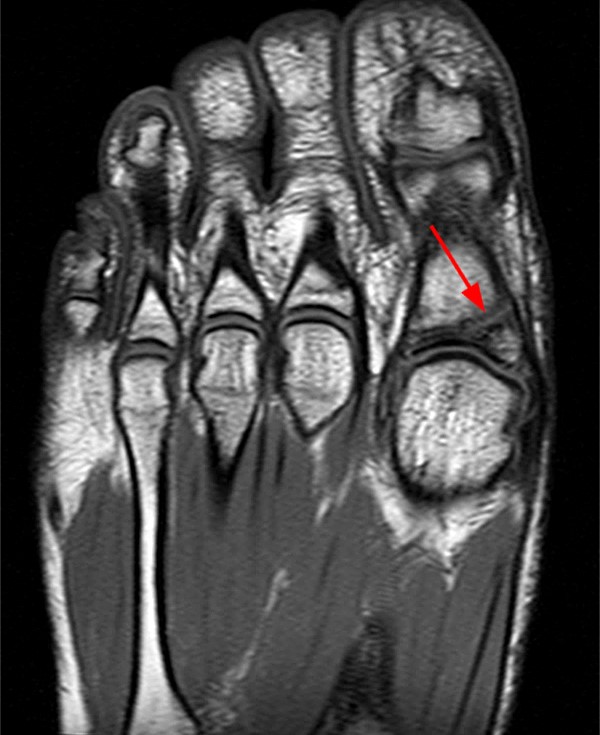
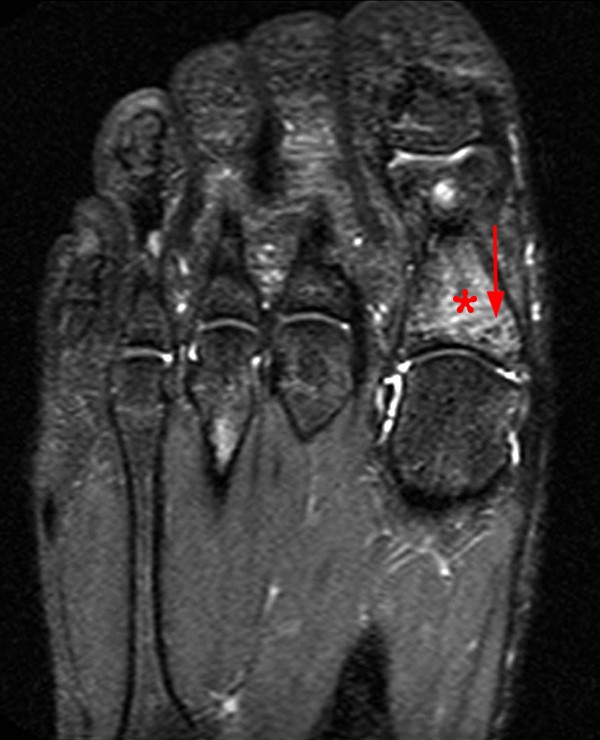
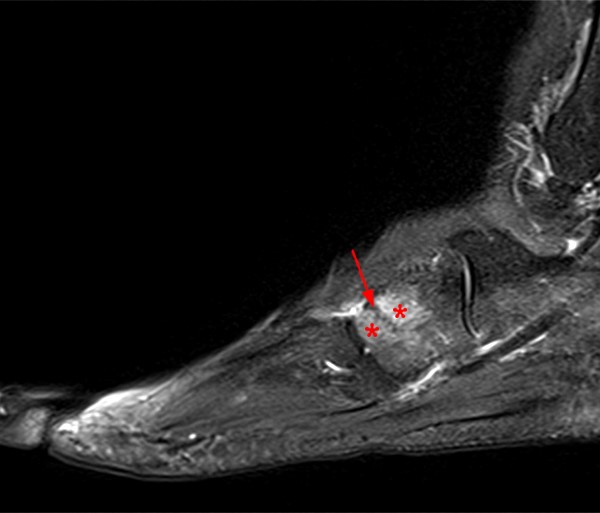
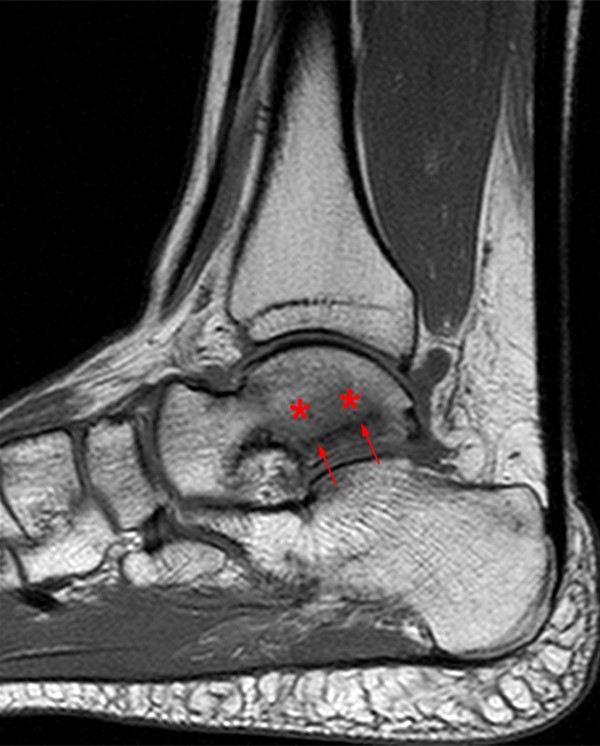
Extensive marrow edema (asterisks) lies within the tarsal navicular bone with decreased signal intensity on the axial T1-weighted image (2a) and diffusely increased signal intensity on the coronal proton density-weighted fat-suppressed, fluid-sensitive series (2b). An incomplete linear sagittal plane defect within the dorsal aspect of the bone extends from the dorsal cortex to the navicular body (arrows).
Diagnosis
Fatigue fracture of the tarsal navicular bone.
Introduction
The emphasis today on the importance of exercise and weight loss has led an increasing number of people to begin new activities, including running, cycling, aerobics and many others. Sports-related injuries are common and the current focus on fitness and competitive athletics has resulted in many individuals undertaking increased levels of physical activity. This results in greater stress upon the musculoskeletal system. Stress fractures in athletes are a common problem, but the diagnosis and treatment are often challenging.
Patients with osseous stress injuries most commonly present with the insidious onset of activity-related local pain with weight bearing. If the athlete continues to exercise, the pain may become more severe or occur at an earlier stage of exercise.1 Typically, the pain resolves when the patient is non-weight bearing. Occasionally, the patient may present with additional findings of redness, swelling, and obvious periosteal reaction at the site of stress fracture. In most cases, the diagnosis of a stress fracture is a clinical one. Occasionally, however, the diagnosis may not be as straight forward, and imaging may be indicated to differentiate among other significant etiologies.
Health care providers caring for recreational and professional athletes must be knowledgeable of the signs and symptoms of these injuries and maintain high suspicion when seeing active patients seeking care for foot and ankle pain, as the signs and symptoms are often vague and overlap with other diagnoses.
Terminology
Stress fracture. The term stress fracture is used to describe fractures that occur after repetitive stress that is insufficient to cause an acute fracture. The two recognized types of stress fracture are fatigue fracture and insufficiency fracture. A fatigue fracture is a stress fracture that results from the application of abnormal muscular stress or torque to a bone with normal elastic resistance.2 An insufficiency fracture, a subset of pathologic fractures, occurs in a nontumoral abnormal bone with a reduced elastic resistance to normal stress.2
Fatigue fracture. In 1855, Breithaupt first reported stress fractures in soldiers associated with marching, which was later radiographically confirmed in 1897.2,3 Previously reported in military recruits, sports and recreational injuries now account for up to 10% of patients in a typical sports medicine practice.4 Prospective studies indicate an incidence of stress fractures that reaches 31% in soldiers5 and 21% in athletes.6 The foot and ankle are the most commonly injured and imaged parts of the musculoskeletal system, accounting for 25% of athletic injuries,7 with runners and dancers constituting the majority.6
Insufficiency fracture. Bone strength depends on factors such as elasticity and stiffness, which are in turn related to bone mineral density, bone composition, and bone structure. Any process that can affect these parameters could also alter bone resistance and favor the development of fractures.8,9 Conditions predisposing to insufficiency fracture include metabolic disorders, inflammatory conditions, bone dysplasias, neurologic disorders and drug therapy.8 Osteoporosis and rheumatoid arthritis are the most common underlying conditions in patients with insufficiency fractures of the foot and ankle.10
Pathophysiology
There is a spectrum of osseous stress injuries that occurs, beginning with stress reaction or stress response and eventually leading to stress fracture. The pathophysiology of stress reaction and stress fractures is related to the bone response to the repetitive stresses at the cellular level. Bone undergoes a constant cycle of reabsorption and regeneration. When bone is challenged with repetitive, submaximal loading, a stress reaction will lead to physiologic and mechanical changes.
With excess stress, there is not only a failure of bone synthesis but also an increase in osteoclast activity. The osteoclasts replace the circumferential lamellar bone with dense osteonal bone. This is accompanied by the development of edema and hyperemia, which is the stress reaction or stress response that can be demonstrated by MR imaging. The relative muscle groups, which are also experiencing the repetitive stresses, respond with hypertrophy and strengthening more rapidly than bone, and this force is transmitted to the periosteum at the muscle attachments, resulting in periostitis. Mechanically, there is a decrease in ultimate strength, leading to an increased risk of microfracture. If the excessive stress on the bone continues, mechanical failure may occur through these microfracture sites, resulting in a stress fracture.11,12
Daffner13 reported that most stress fractures are caused by muscular activity rather than direct osseous trauma. Muscles normally provide biomechanical dissipation of stress from the bones, but fatigued muscle may decrease this protective contribution, and this can result in the transmission of increased stress to the bones. Most stress fractures involve the lower extremity and are actually fatigue fractures caused by muscle tension on normal bone.14,15,16 Stress fractures have been reported most frequently in military recruits and athletes, though they also occur in children and adults in the general population.17,18,19
Gender differences in athletes have not been documented though there is a perception of an increased number of stress injuries in women.20 The female athlete may have a triad of conditions that lead to stress injuries. These include a combination of aggressive training, nutritional disorders, and menstrual irregularities.21 Most stress fractures occur 4 to 5 weeks after the onset of a new exercise, are usually relieved by rest, but progress if activity is continued.
Blickenstaff and Morris22 described the phases of stress fractures that at least partially explain the changes seen with imaging. Osteoclastic resorption occurs 5 to 14 days after the initial injury. This is followed by increased vascularity, which accompanies endosteal and periosteal callus formation. With continued activity, fracture occurs. Fractures may be primarily cortical or medullary, depending on the fracture site. In the series by Greaney et al.23, 77% of fractures were cancellous and 23% cortical. Routine radiographs were more useful with cortical fractures.
Physical examination may reveal tenderness and slight swelling. Children may have slight temperature elevation. Symptoms and certain radiographic features (exuberant callus or periosteal reaction) may simulate infection, neoplasm, arthropathy, or ligament injury. With the increased use of MR imaging, the ability to detect marrow changes early has led to the concept of stress reaction and the grading of stress changes.24,25
Imaging of Stress Fractures
Detection of stress fractures in the early stages on radiographs is difficult. Radiographs are usually normal in the acute phase. Metabolic changes at the site of fracture allow isotope bone scans to detect the injury as early as 24 hours after injury. Bone changes are usually not evident on radiographs for 10 to 21 days following injury.23
MR imaging is extremely sensitive in the detection of pathophysiologic soft tissue, bone, and marrow changes associated with stress injuries.26 It allows depiction of abnormalities weeks before the development of radiographic abnormalities and has comparable sensitivity and superior specificity in relation to scintigraphy.27,28 Apart from bone response, it also provides information about the surrounding soft tissues and may demonstrate the muscular or ligamentous insults associated with or responsible for the symptoms.29,30 MR imaging is noninvasive, nonionizing, and more rapidly performed than bone scintigraphy. When evaluating a stress injury, MR imaging parameters should include a T1-weighted sequence and fluid-sensitive sequences such as STIR or T2-weighted with frequency-selective fat suppression.31 The fluid –sensitive sequences are important for detection of the earliest changes of stress reaction, such as periosteal, muscle, or bone marrow edema.31 T1-weighted sequences depict anatomy and more advanced stress-related findings.27 MR contrast enhancement studies are not necessary in the foot and ankle as unenhanced imaging studies demonstrate almost identical imaging findings.32
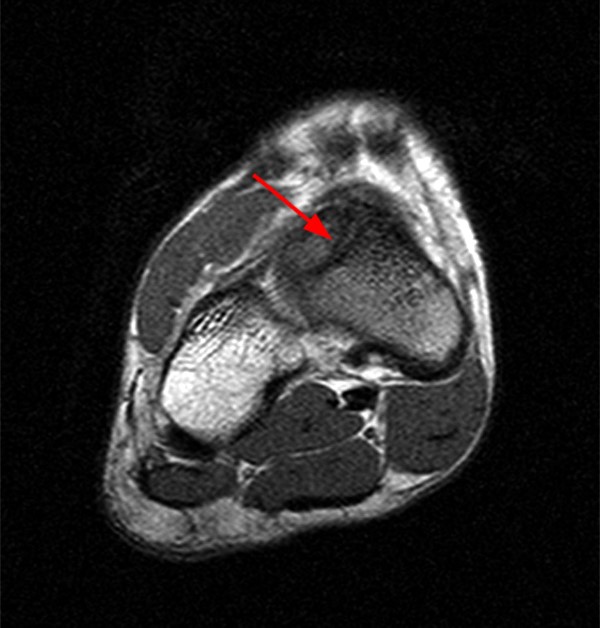
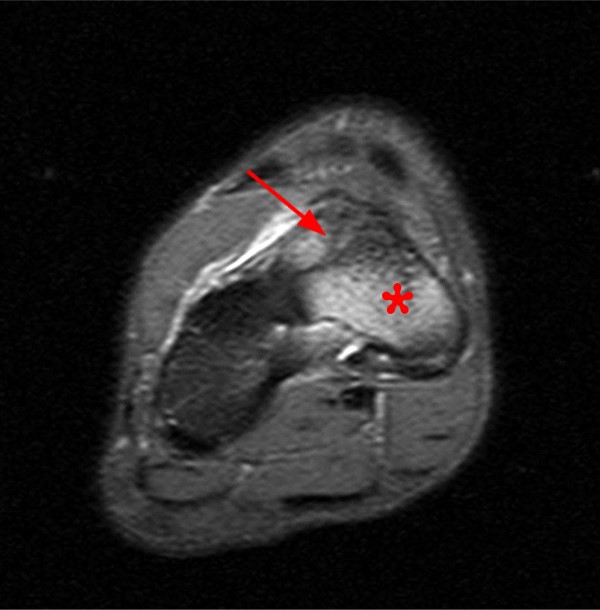
Stress injury can be subdivided into stress reaction, and stress or insufficiency fracture. Before a stress fracture takes place, a frequently painful condition termed stress reaction occurs. During this period, in which bone strengthening as a reaction to stress has reached a plateau, bone marrow edema, hyperemia, and increased osteoclastic activity develop within the stressed area of the bone, translated on MR imaging as areas of poorly defined abnormal marrow signal, similar to a bone marrow contusion [Figure 3, 4].29
Periosteal edema may be seen, manifested as high signal outside the cortex on fluid-sensitive images [Figures 5, 6, 7]. Stress reaction can be distinguished from a stress fracture by the relative absence of signal alterations on T1-weitghted images.
Figure 5:
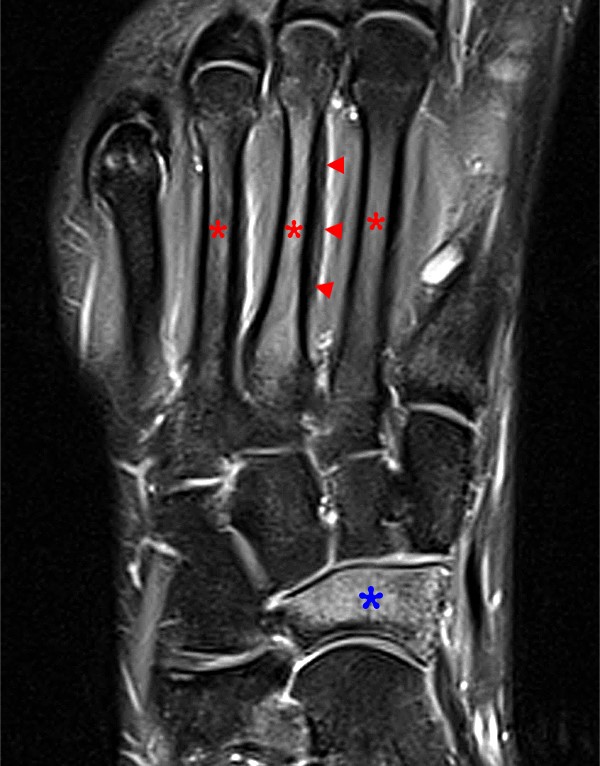
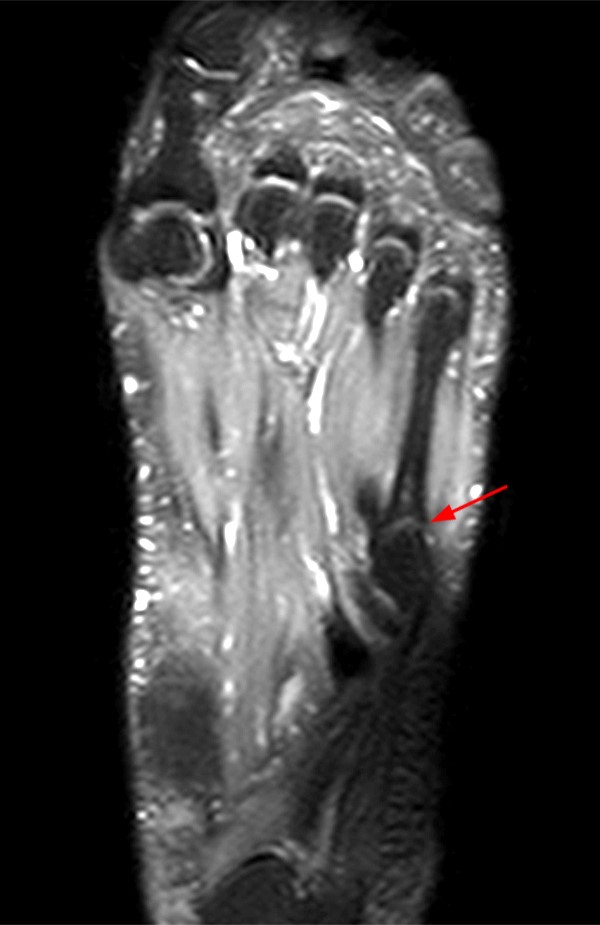
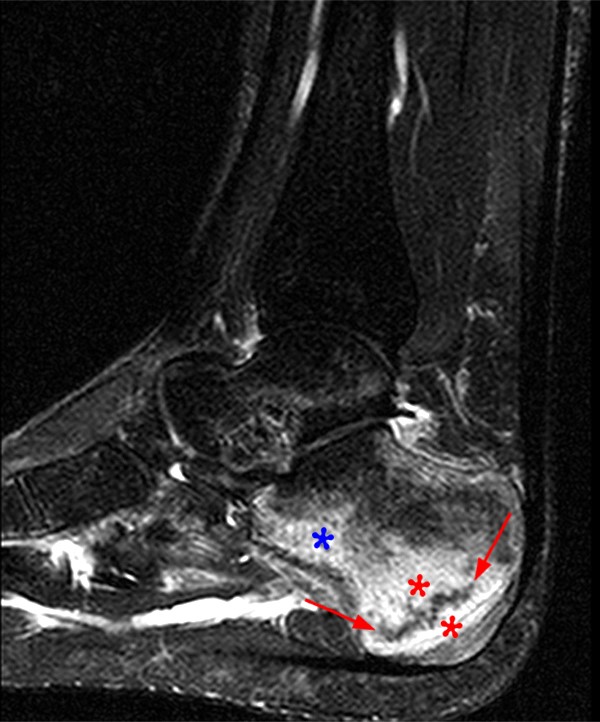
Twenty year-old male with foot pain after the start of football practice. Axial fat-suppressed proton-density weighted image of the foot. Diffuse marrow edema (red asterisks) is seen within the diaphysis of the first, second, and third metatarsals. Smooth periosteal thickening is demonstrated at the medial border of the second metatarsal (arrowheads). No distinct linear fracture. Extensive marrow edema is also seen within the navicular bone (blue asterisk).
Figure 7:
Same patient as figure 3. A coronal T1-weighted image demonstrates normal marrow signal in the second, third and fourth metatarsals. Smooth periosteal thickening (arrowhead) at the medial aspect of the third metatarsal is again shown. The appearance is compatible with grade 2 stress reaction.
As stress persists and a fracture develops, marrow signal alterations increase in extent and, unlike stress reaction, will be clearly depicted on both T1 and fluid-sensitive sequences. The marrow edema tends to be extensive and in the metatarsals often involves almost the whole shaft. Occasionally, MR imaging shows a hypointense, irregular line, typically contiguous with the cortex, within the area of edema and hyperemia consistent with a fracture line [Figure 8,9]. This line is better visualized on fluid-sensitive images and is more visible after initiation of the healing process. A discrete fracture line, however, is not always seen. Conversely, persistence of the fracture line may be noted even after complete healing has occurred. The old fracture line, however, is not accompanied by marrow edema on fluid-sensitive images.
Figure 8:
Axial fat-suppressed proton-density weighted image in a 46 year-old male with foot pain and history of a prior second metatarsal stress fracture (seen in figure 10). Extensive marrow edema is seen within the 3rd metatarsal diaphysis (asterisks) with marked surrounding periosteal and soft tissue edema (arrowheads). The fracture callous from the healing second metatarsal fracture is partially imaged (arrow).
Figure 9:
Sagittal T1-weighted image through the 3rd metatarsal of the same patient demonstrates a dorsal fracture of the cortex (arrow) with the linear low signal line extending into the medullary cavity of the bone. Extensive surrounding periosteal and soft tissue edema (arrowheads) is also demonstrated. This appearance is diagnostic of a grade 4 stress reaction or stress fracture.
Periosteal callous formation begins shortly after the onset of fracture, and can be seen on MR images as a hypointense line, peripheral to the bright signal periosteal edema and parallel to the cortex, representing the elevated periosteum and periosteal reaction [Figure 10].29 Periosteal reaction commonly accompanies stress fractures of the metatarsals but is typically not seen in the tarsal bones.
Figure 10:
Axial fat-suppressed proton-density weighted image in a 46 year-old male with foot pain. A healing stress fracture of the second metatarsal is demonstrated with extensive marrow and soft tissue edema. The fracture callous is best appreciated medially (arrowheads) where the low signal callus lies superficial to the periosteal edema.
Arendt and Griffiths 24 graded MR features of stress reactions based upon image appearance and the degree of marrow, periosteal, and cortical involvement. Grade 0 is normal. Grade 1 demonstrates subtle periosteal edema without marrow changes. Progression to Grade 2 results in periosteal and marrow edema which is only identified on fluid sensitive imaging series. Changes are more defined with Grade 3 injuries with marrow edema identified on T1-weighted images. With Grade 4 stress reactions, a discrete fracture line is identified along with the marrow and periosteal changes.
Follow-up of athletes with MR imaging is feasible, although marrow edema can take up to 6 months to resolve with healing, especially in elderly patients.33 However, resolution of MR signal abnormalities within 4 weeks has been observed if aggressive rest programs are initiated with non-weight bearing at the start of symptoms in young athletes, lending support that marrow edema in this context represents recent or ongoing injury.24
Distribution of Stress Fractures in the Foot and Ankle
Metatarsals
Metatarsal stress fractures are among the most common stress fractures seen in athletes.34 Most metatarsal stress fractures involve the second and third metatarsal. Fractures of the first metatarsal account for 7% to 8%, and fractures of the fourth and fifth metatarsals account for 3% of metatarsal stress fractures.35 Most military stress fractures are caused by marching in new recruits. In athletes, metatarsal stress fractures are most common during sprinting, hurdling, and jumping activities.20 Stress fractures in civilians are more commonly caused by new footwear or a new activity such as running or standing for long periods.13,14
Fractures of the proximal fourth metatarsal bone are less common than distal fourth metatarsal fractures, and have a longer healing time. This is similar to proximal fifth metatarsal injuries and stress fractures. Patients may continue to be symptomatic even after 3 months of rest and immobilization. Ideal treatment appears to involve a prolonged combination of non-weight bearing casting followed by weight-bearing casting.36 The fifth metatarsal stress fracture may occur in the metatarsal shaft in contradistinction to the Jones fracture, which is a fracture through the meta-diaphyseal junction of the fifth metatarsal [Figures 11,12].
Figure 11:
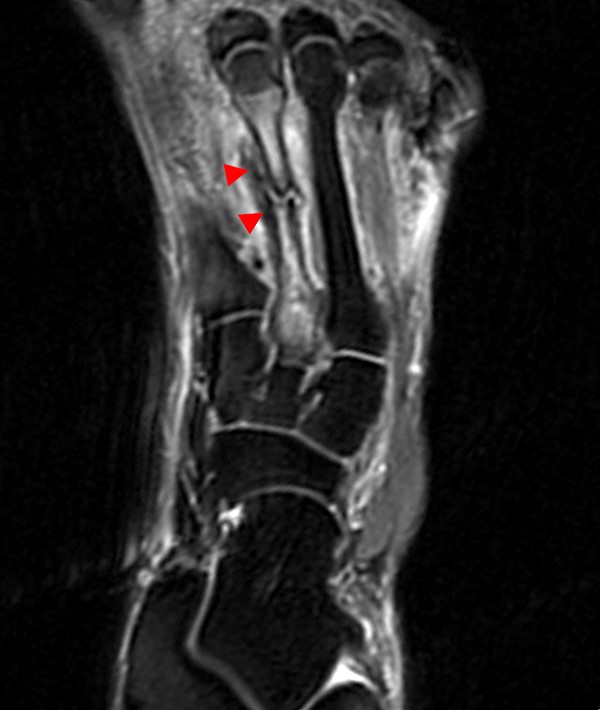
Axial fat-suppressed proton-density weighted image of a 30 year-old female with lateral foot pain. A transverse fracture (arrow) involves the meta-diaphyseal junction of the fifth metatarsal base. A fracture in this location is a true Jones fracture and may be complicated by delayed or non-union. The relative lack of adjacent marrow edema suggests this is a more chronic injury and may reflect a delayed or non-union.
Delayed union and nonunion may occur in a significant number of these injuries. Delayed union of Jones fractures may occur in up to 67% of cases treated non-operatively. Immediate intramedullary screw fixation of Jones fractures and proximal shaft fifth metatarsal fractures has been reported to have nearly 100% union rates, with an average time to union being approximately 6 to 8 weeks. Intramedullary fixation has been recommended as the treatment of choice for these fractures to achieve improved union rates.37 More recently it has been suggested that intramedullary screw fixation alone does not always adequately address the torsional stress created by the insertion of the peroneus brevis on the proximal fragment of the fifth metatarsal in fifth metatarsal fractures. It has been suggested that optimal internal fixation appears to require internal devices or fixation that also address the torsional stresses.38
Navicular
Navicular stress fractures were once considered uncommon.25,39 However, more recent studies report the incidence at 14% to 35% of all stress fractures.20 In athletes involved in track and field events, the incidence approaches 73%.39 Patients present with progressive pain over the dorsum of the midfoot. There is minimal soft tissue swelling and no discoloration or ecchymosis. Focal pressure over the navicular reproduces the symptoms.19,39,40 Initial radiographs are normal. Therefore, if there is significant clinical evidence to suggest a navicular stress fracture, additional imaging with MR imaging should be obtained.40,41,42
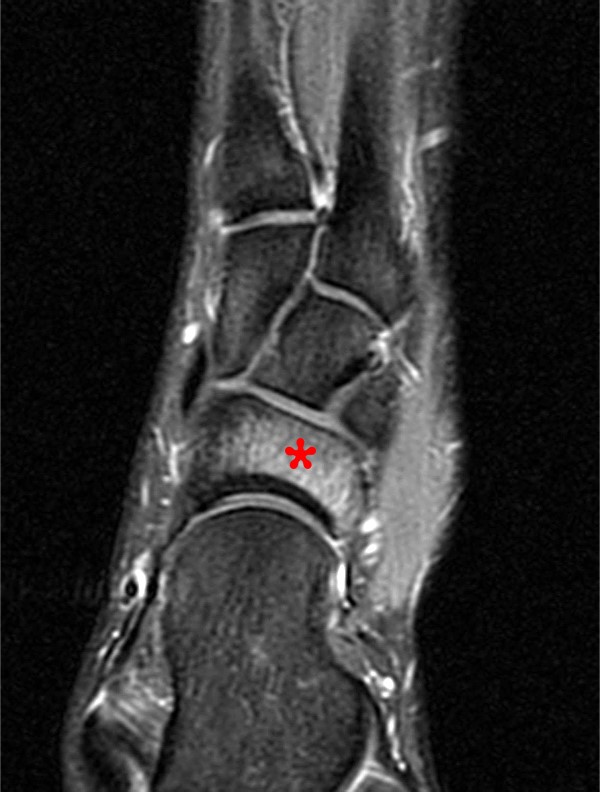
A proposed CT-based classification system separates fractures into three groups: dorsal cortical break (type I)[FIGURES 13, 14], fracture propagation into the navicular body (type II) [Figures 1, 2], and fracture propagation into another cortex (type III)[Figures 15,16]. Type III lesions have the longest average healing time with nonoperative care. These lesions may warrant more aggressive treatment and orthopaedic referral for possible open reduction and internal fixation.43
Figure 15:
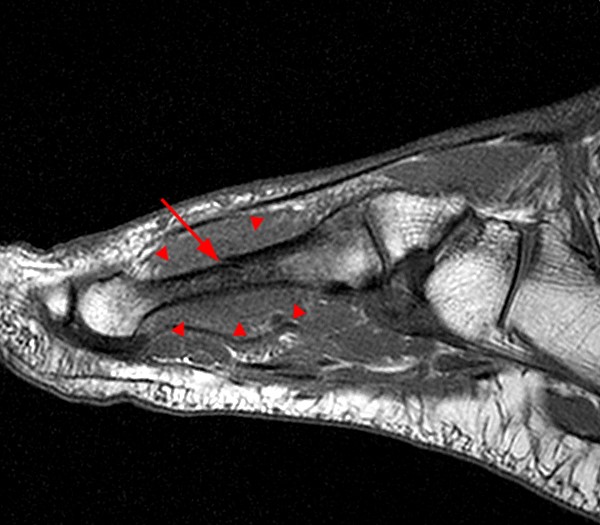
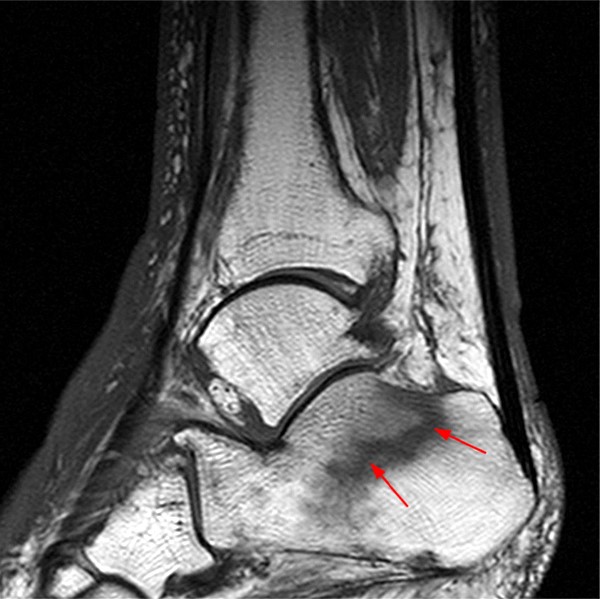
Coronal T1-weighted image obtained through the navicular bone in a 16 year-old female soccer player with 2 months of anterior and lateral foot pain. In this case, the linear fracture (arrow) extends somewhat obliquely from the dorsal cortex to the lateral cortex of the navicular bone. When two cortices are breached, operative intervention is frequently recommended.
Treatment of navicular stress fractures may present a significant challenge. Delay in diagnosis for up to 4 months is not uncommon.44 Treatment with cast immobilization may be effective for fractures involving only the dorsal cortex (type I). However, when the fracture extends to the navicular body (type II) or breaches two cortices (type III), operative intervention is recommended. 40,45 Operative intervention is also indicated for displaced and comminuted fractures. Surgical intervention also may be indicated in athletes who need quick healing to allow them to return to play. The average time for athletes to return to play after surgical intervention compared with nonoperative management using a non–weight-bearing cast is 3.8 months and 5.6 months, respectively.45 Typically, surgical intervention consists of screw fixation, with possible bone graft inlay.
Calcaneus
Calcaneal stress fractures [Figures 17 – 20] are nearly as common as metatarsal stress fractures. In fact, in the series of Greaney et al., calcaneal stress fractures were more common than metatarsal stress fractures.23 Associated upper tibial stress fractures were noted in 60% of military recruits. Bilateral calcaneal stress fractures occur in up to 24% of patients.46 Calcaneal stress fractures are less common in civilians and again related to footwear in many cases. Pain is often medial or lateral, rather than on the plantar aspect of the foot. Physical examination reveals pain when the examiner squeezes the patient’s heel from the sides.
Figure 17:
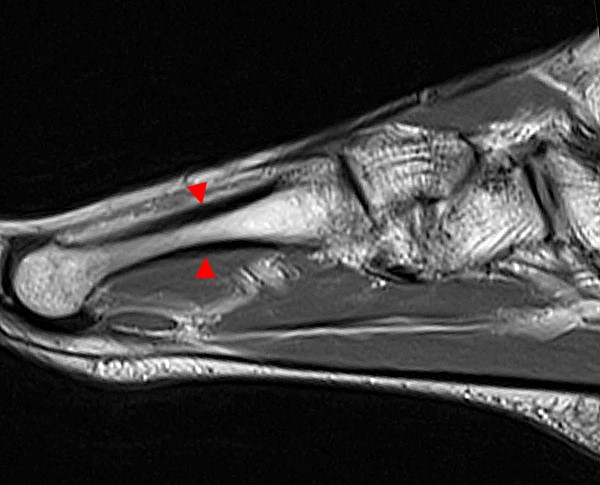
Sagittal T1-weighted image in a 19 year-old female with two weeks of ankle pain. A linear low signal fracture (arrows) parallels the posterior cortex. Adjacent low signal marrow (asterisks) is compatible with marrow edema. Posterior stress fractures are likely related to the muscle tension of the Achilles. A smaller linear fracture (arrowheads) is demonstrated more anteriorly, without significant adjacent T1 signal abnormality.
Nondisplaced and noncomminuted calcaneal fractures may be treated with non-operative management with casting and non-weight bearing for 6 weeks. Displaced or comminuted fractures are indications for surgical intervention, which may include screw fixation or autologous bone grafting, depending on the nature and age of the fracture.44 Evaluation of footwear is important to prevent recurrence.
Great toe and sesamoids
Stress fractures of the great toe and sesamoids [Figures 21 – 24] are seen less frequently than other sites of stress-related injury, but when they do occur the diagnosis may be more difficult, resulting in a delay in diagnosis if this injury is not considered. Stress fractures of the great toe have been reported in runners, soccer players, and volleyball players. Athletes who have pain in the first metatarsophalangeal joint and who are exposed to excessive running, jumping, and repeated forced dorsiflexion of the first metatarsophalangeal joint seem to be predisposed to this injury.47 As with stress-related injury in other locations, the symptoms typically occur during training without a history of trauma. Approximately 1% of running injuries involve the sesamoids; 40% of these are stress fractures and 30% are sesamoiditis.47 Sesamoiditis/osteochondritis, osteonecrosis, stress response of the synchondrosis of partite sesamoids bones, traumatic fractures, osteomyelitis, and bursitis between the tibial sesamoid and the flexor halluces brevis tendon may all occur in this location. One or both sesamoids may be involved.
MR imaging signal alterations of stress response of sesamoids and sesamoiditis overlap. Increased STIR signal intensity and low T1 signal have been described with sesamoid stress response, as opposed to increased STIR signal intensity and normal T1 signal, which favor sesamoiditis. Sesamoiditis also more commonly involves both sesamoid bones, and may be associated with bursitis, tendinosis and tenosynovitis.48,49
Treatment typically involves avoidance of physical activity and attention to predisposing extrinsic factors such as footwear.50,51 Hulkko and Orava4 reported 15 cases of stress fractures of the hallucal sesamoids in athletes. The mean patient age was 22.3 years old. Nine patients were male and 6 were female. Eight fractures involved the medial sesamoid, six involved the lateral sesamoid, and in 1 patient both sesamoids were involved. Ten patients were treated non-operatively. Five patients required surgical excision of the fragmented involved sesamoid and gradually returned to training 6 to 8 weeks after surgery. Pathology confirmed fibrotic nonunion of the stress fractures.4
Other tarsal bones
Up to 20% of stress fractures in athletes may occur in the tarsal bones.20 Stress fractures of the tarsal bones are too often a diagnostic challenge, because many providers do not consider tarsal stress fractures in the assessment of foot and ankle pain. A high clinical suspicion of stress fractures is required for an accurate and timely diagnosis. The majority of tarsal bone stress fractures occur in the navicular.34
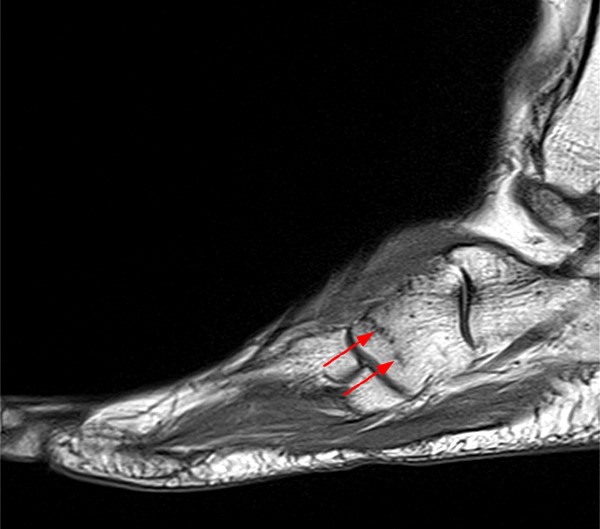
Rarely, stress fractures may occur in the cuboid bone [Figure 25, 26]. Diagnosis may again be delayed secondary to this diagnosis not being considered. It may mimic peroneal tendon pathology.52
Stress fractures of the talus [Figure 27, 28] and calcaneus also occur in athletes.53,54
Plain films will most often be normal, and MR imaging is the imaging modality of choice for detection, localization, and characterization of tarsal bone stress fractures. MR imaging most often demonstrates marrow edema or a cortical fracture line.
Nondisplaced and noncomminuted tarsal bone fractures may be treated with non-operative management with casting and non-weight bearing for 6 weeks. Displaced or comminuted fractures are indications for surgical intervention, which may include screw fixation or autologous bone grafting, depending on the nature and age of the fracture.44 Evaluation of footwear is important to prevent recurrence.
Summary
Stress fractures in the foot and ankle are a common problem, but their diagnosis and treatment are often challenging. A high level of suspicion and awareness of these injuries should be maintained when caring for physically active patients, in order to avoid misdiagnoses or delays in diagnosis. MR imaging can be particularly helpful for the diagnosis and characterization of osseous stress injuries in the foot and ankle.
References
- Brukner P, Bennell K. Stress fractures in female athletes. Diagnosis, management and rehabilitation. Sports Med 1997;24(6):419-429. ↩
- Pentecost RL, Murray RA, Brindley HH. Fatigue, insufficiency, and pathologic fractures. JAMA 1964;187:1001-1004. ↩
- Breithaupt MDS. Zur patholgie des menschlichen fusses. Med Ztg 1855;24:169-171, 175-177. ↩
- Hulkko A, Orava S. Stress fractures in athletes. Int J Sports Med 1987;8(3):221-226. ↩
- Milgrom C, Giladi M, Stein M, et al. Stress fractures in military recruits. A prospective study showing an unusually high incidence. J Bone Joint Surg Br 1985;67:732-735. ↩
- Lassus J, Tulikoura I, Konttinen Y, et al. Bone stress injuries of the lower extremity: a review. Acta Orthop Scand 2002;73:359-368. ↩
- Prokuski LJ, Saltzman CL. Challenging fractures of the foot and ankle. Radiol Clin North Am 1997;35:655-670. ↩
- Peris P. Stress fractures. Best Pract Res Clin Rheumatol 2003;17:1043-1061. ↩
- Egol KA, Koval KJ, Kummer F, et al. Stress fractures of the femoral neck. Clin Orthop Relat Res 1998;348:72-78. ↩
- Peris P. Stress fractures in rheumatological practice: clinical significance and localizations. Rheumatol Int 2002;22:77-79. ↩
- Pepper M, Akuthota V, McMarty EC. The pathophysiology of stress fractures. Clin Sports Med 2006;25:1-16. ↩
- Michael RH, Holder LE. The soleus syndrome: a cause of medial tibial stress (shin splints). Am J Sports Med. 1985;13:87-94. ↩
- Daffner RH. Stress Fractures. Skeletal Radiol. 1987;2:221-229. ↩
- Chowchuen P, Resnick D. Stress fractures of the metatarsal heads. Skeletal Radiol. 1998;27:22-25. ↩
- Eisele SA, Sammarco GJ. Fatigue fractures of the foot and ankle in the athlete. J Bone Joint Surg Am. 1993;75:290-298. ↩
- Umans H, Pavlov H. Stress fractures of the lower extremities. Semin Roentgenol. 1994;29(2):176-193. ↩
- Devas MB. Stress fractures in children. J Bone Joint Surg Br. 1963;45:528-541. ↩
- Gilbert RS, Johnson HA. Stress fractures in military recruits. A review of 12 years experience. Milit Med. 1966;131:716-721. ↩
- Goergen TG, Rossman DJ, Gerber KH. Tarsal navicular stress fractures in runners. Am J Roentgenol. 1981;136:201-203. ↩
- Arendt EA. Stress fractures and the female athlete. Clin Orthop. 2000;372:131-138. ↩
- Rossi F, Dragoni S. Talar body fatigue stress fractures: three cases observed in elite female gymnasts. Skeletal Radiol. 2005;34:389-394. ↩
- Blickenstaff LD, Morris JM. Fatigue fractures of the femoral neck. J Bone Joint Surg Am. 1966;48:1031-1047. ↩
- Greaney RB, Gerber FH, Laughlin RL. Distribution and natural history of stress fractures in U.S. Marine recruits. Radiol. 1983;146:339-346. ↩
- Arendt EA, Griffiths HJ. The use of MR imaging in the assessment and clinical management of stress reactions of bone in high-performance athletes. Clin Sports Med. 1997;16:291-306. ↩
- Zanetti M, Steiner CL, Seifert B, et al. Clinical outcomes of edema-like bone marrow abnormalities in the foot. Radiol. 2002;222:184-188. ↩
- Anderson MW, Greenspan A. Stress fractures. Radiology 1996;199:1-12. ↩
- Spitz DJ, Newberg AH. Imaging stress fractures in the athlete. Radiol Clin North Am 2002;40:313-331. ↩
- Deutsch AL, Coel MN, Mink MH. Imaging of stress injuries to bone: radiography, scintigraphy, and MR imaging. Clin Sports Med 1997;16:275-290 ↩
- Stafford SA, Rosenthal KI, Gebhardt MC, et al. MRI in stress fracture. AJR 1986;147:553-556. ↩
- Lee JK, Yao L. Stress fractures: MR imaging. Radiology 1988;169:217-220. ↩
- Rubin DA, Dalinka MK, Kneeland JB. Magnetic resonance imaging of lower extremity injuries. Semin Roentgenol 1994;29:194-222. ↩
- Schmid MR, Hodler J, Vienne P, et al. Bone marrow abnormalities of the foot and ankle: STIR versus T1-weighted contrast-enhanced fat-suppressed spin-echo MR imaging. Radiology 2002;224:463-469. ↩
- Miller JH. The radiological aspects of stress fractures and chronic stress injuries. Curr Orthop 2003;17:150-155. ↩
- Bruckner P, Bradshaw C, Khan KM, et al. Stress fractures: a review of 180 cases. Clin J Sport Med 1996;6(2):85-89. ↩
- Levy JM. Stress fractures of the first metatarsal. Am J Roentgenol. 1978;130:679-681. ↩
- Saxena A, Krisdakumtorn T, Erickson S. Proximal fourth metatarsal injuries in athletes: similarity to proximal fifth metatarsal injury. Foot Ankle Int 2001;22(7):603-608. ↩
- Portland G, Kelikian A, Kodras S. Acute surgical management of Jones fractures. Foot Ankle Int 2003;24(11):829-833. ↩
- Vertullo C, Glisson R, Nunley J. Torsional strains in the proximal fifth metatarsal: implications for Jones and stress fracture management. Foot Ankle Intl 2004;25(9):650-565. ↩
- Potter NJ, Brukner PD, Makdissi M, et al. Navicular stress fractures; outcomes of surgical and conservative management. Br J Sports Med. 2006;40:692-695. ↩
- Saxena A, Fullem B. Navicular stress fractures: a prospective study in athletes. Foot Ankle Int. 2006;27:917-921 ↩
- Kiss ZS, Khan KM, Fuller PJ. Stress fractures of the tarsal navicular bone: CT findings in 55 cases. Am J Roentgenol. 1993;160:111-115. ↩
- Koulouris G, Morrison WB. Foot and ankle disorders: radiographic signs. Semin Roentgenol. 2005;40:358-379. ↩
- Saxena A, Fullem B, Hannaford D. Results of treatment of 22 navicular stress fractures; a new proposed radiographic classification system. J Foot Ankle Surg. 2000;39:96-103. ↩
- Coris EE, Lombardo JA. Tarsal navicular stress fractures. Am Fam Phys. 2003;67:85-90. ↩
- Khan KM, Brukner PD, Kearney C, et al. Tarsal navicular stress fractures in athletes. Sports Med 1994;17:65-76. ↩
- Meurman KOA. Less common stress fractures of the foot. Br J Radiol. 1981;54:1-7. ↩
- Shiraishi M, Mizuta H, Kubota K, et al. Stress fracture of the proximal phalanx of the great toe. Foot Ankle. 1993;14(1):28-34. ↩
- Karasick D, Schweitzer ME. Disorders of the hallux sesamoid complex: MR features. Skeletal Radiol. 1998;27:411-418. ↩
- Ashman CJ, Klecker RJ, Yu JS. Forefoot pain involving the metatarsal region: differential diagnosis with MR imaging. Radiographics. 2001;21:425-440. ↩
- Richardson EG. Injuries to the hallucal sesamoids in the athlete. Foot Ankle. 1987;7(4):229-244. ↩
- Hulkko A, Orava S, Pellinen P, et al. Stress fractures of the sesamoid bones of the first metatarsophalangeal joint in athletes. Arch Orthop Trauma Surg. 1985;104(2):113-117. ↩
- Battaglia H, Simmen HP, Meier W. Stress fractures of the cuboid bone: an easy to treat rarity. Swiss Surg 2002;8(1):3-6. ↩
- Hontas MJ, Haddad RJ, Schlesinger LC. Conditions of the talus in the runner. AM J Sports Med 1986;14(6):486-490. ↩
- Norfray JF, Schlacter L, Kernahan WT Jr, et al. Early confirmation of stress fractures in joggers. JAMA 1980;243(16):1647-1649. ↩