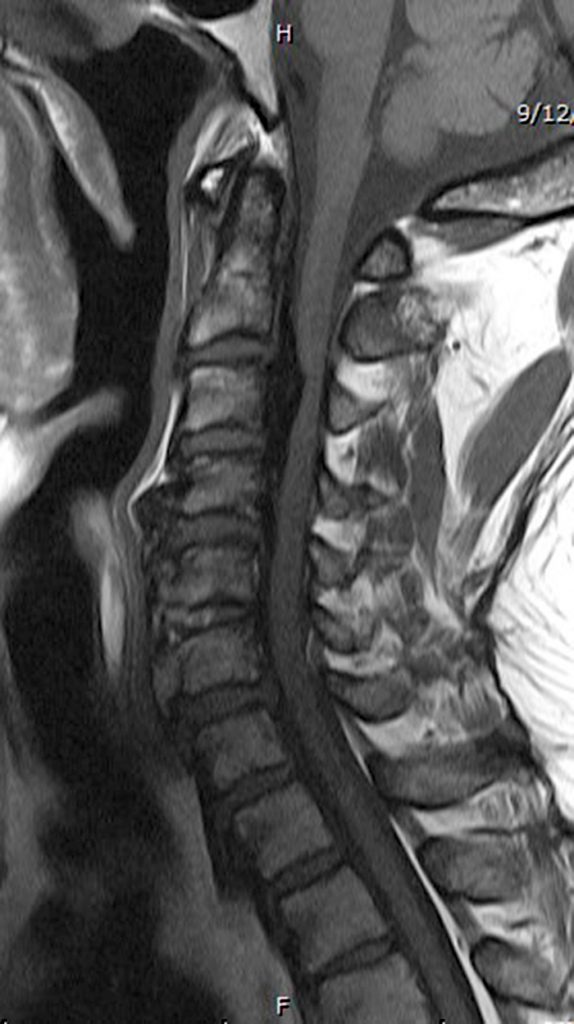
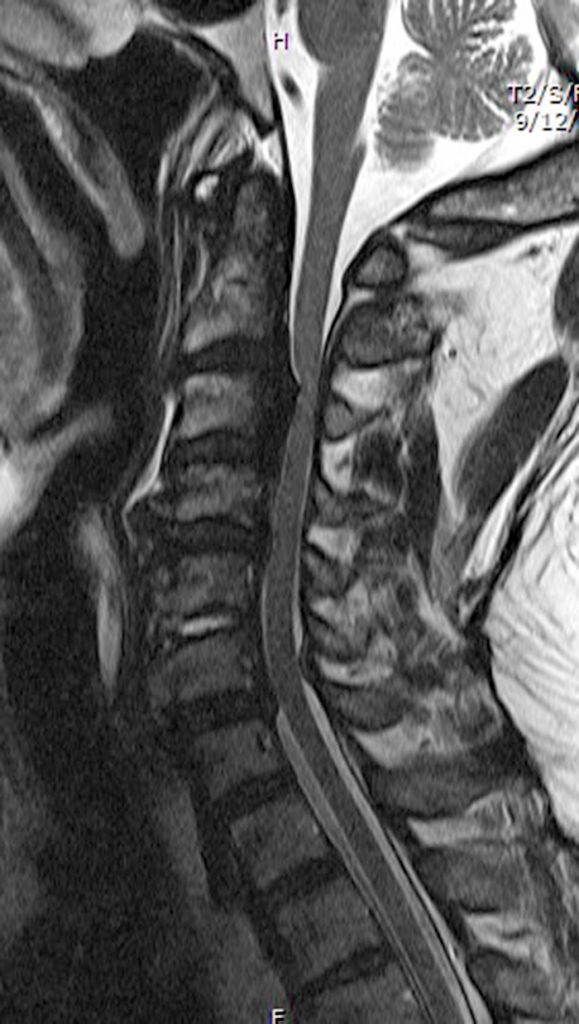
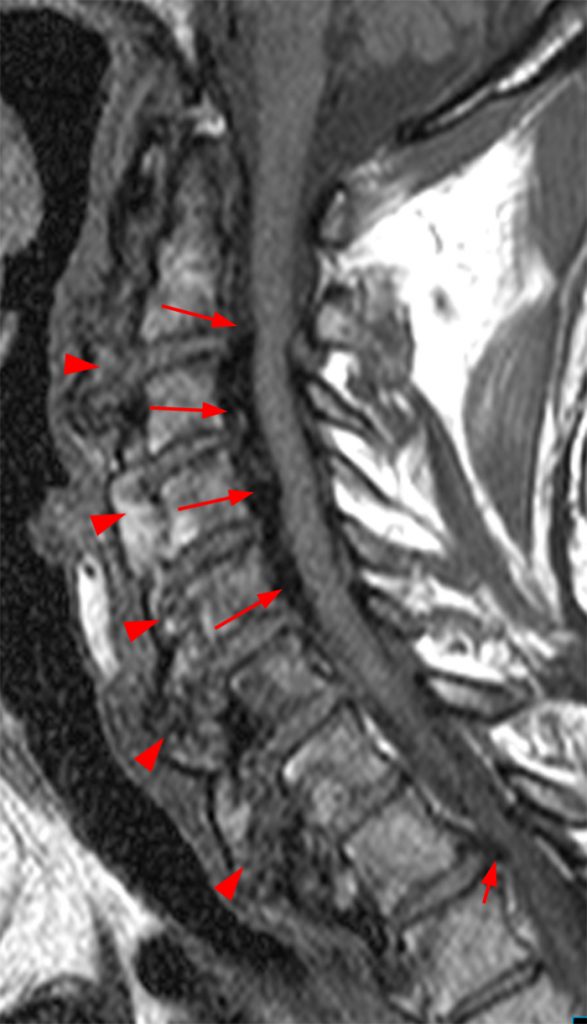
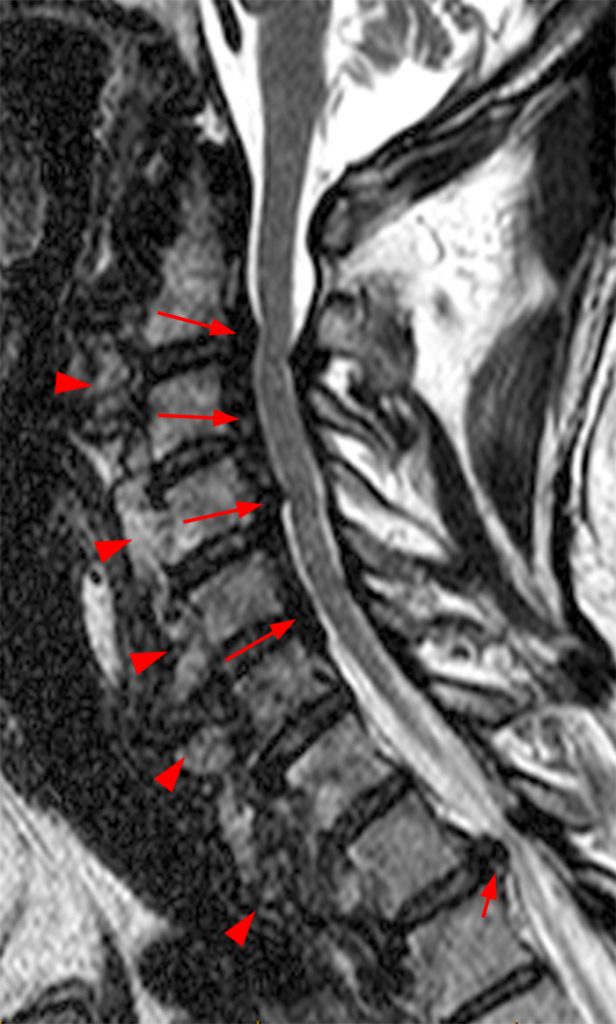
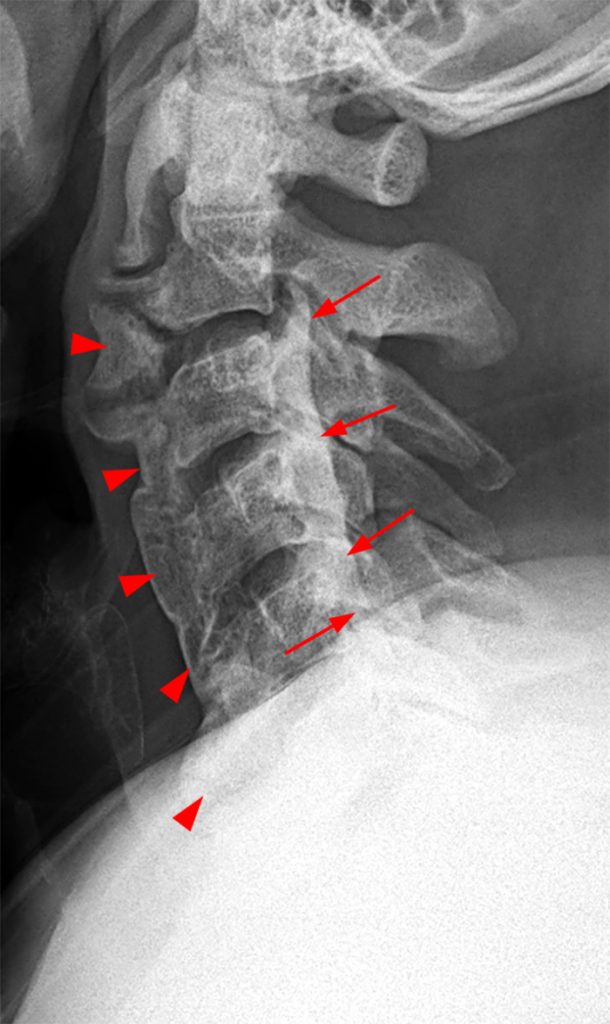
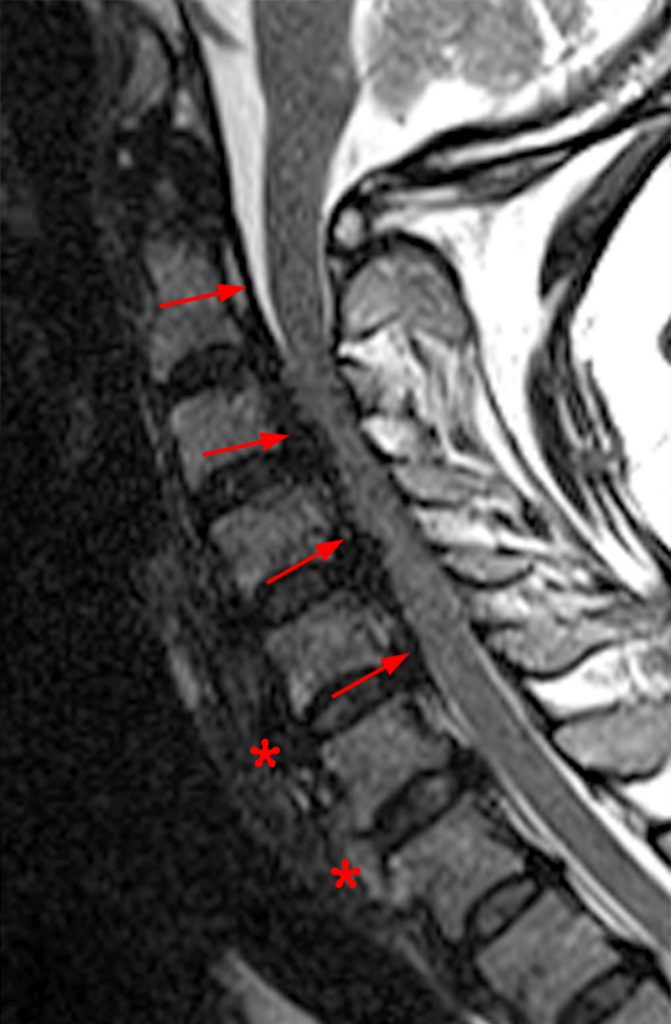
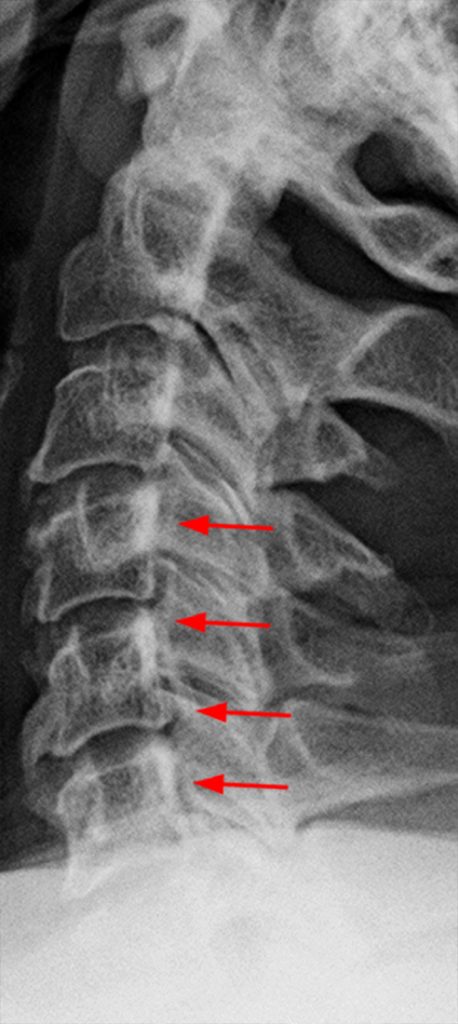
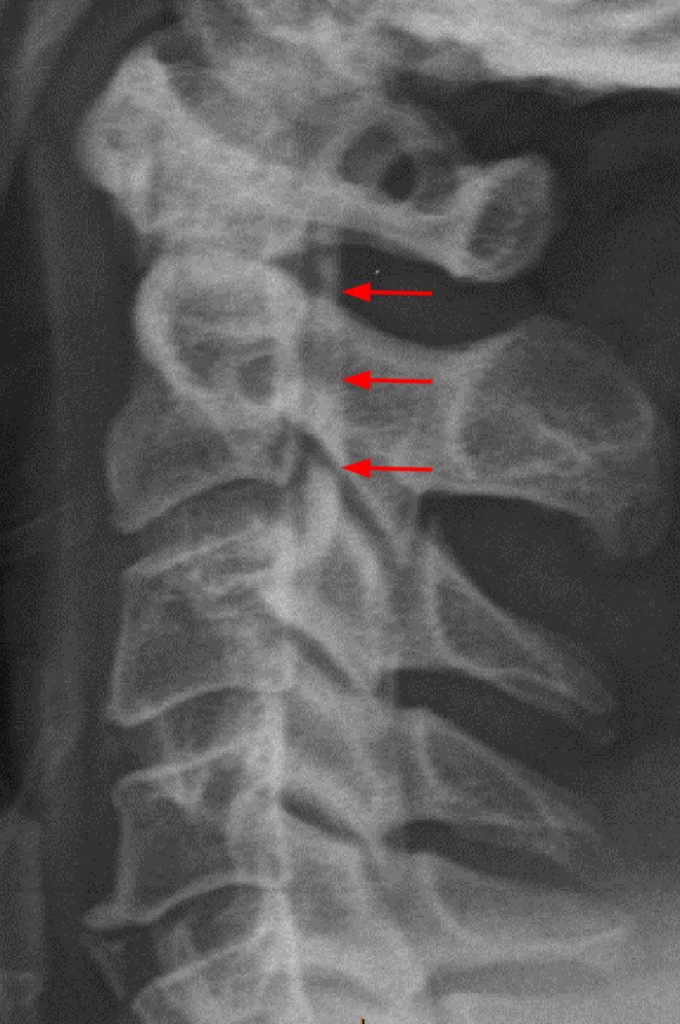
Clinical History: A 64-year-old man developed neck pain after falling off a truck one week ago. T1- and T2-weighted sagittal MR images (1a and 1b) are provided. What are the findings? What is your diagnosis?
Findings
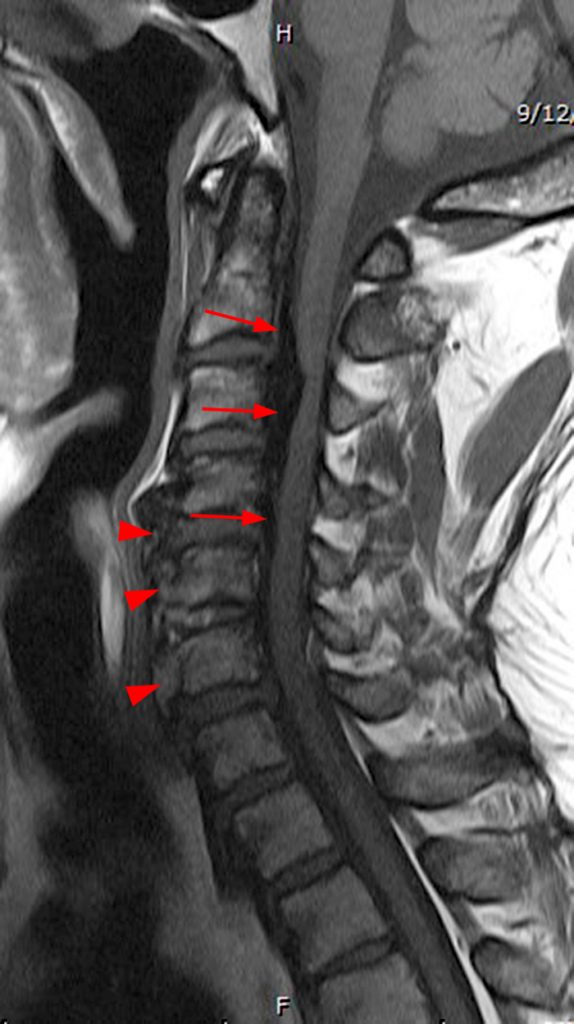
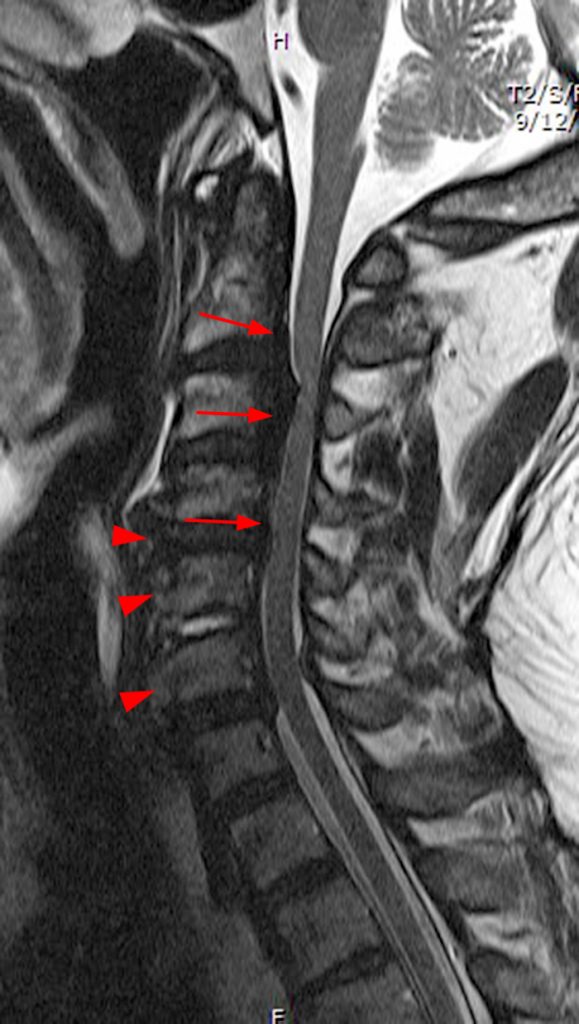
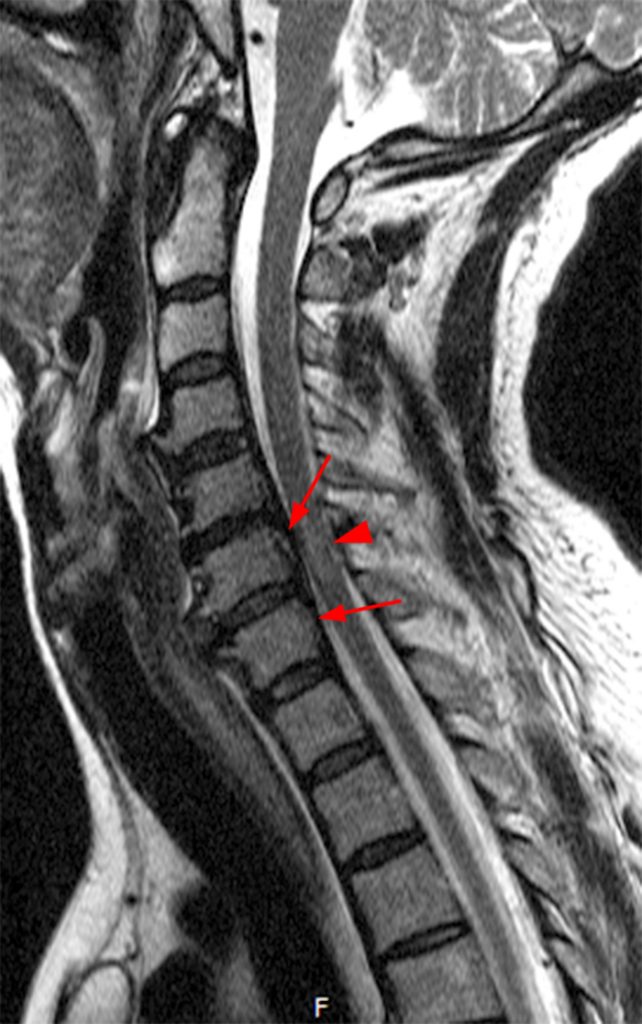
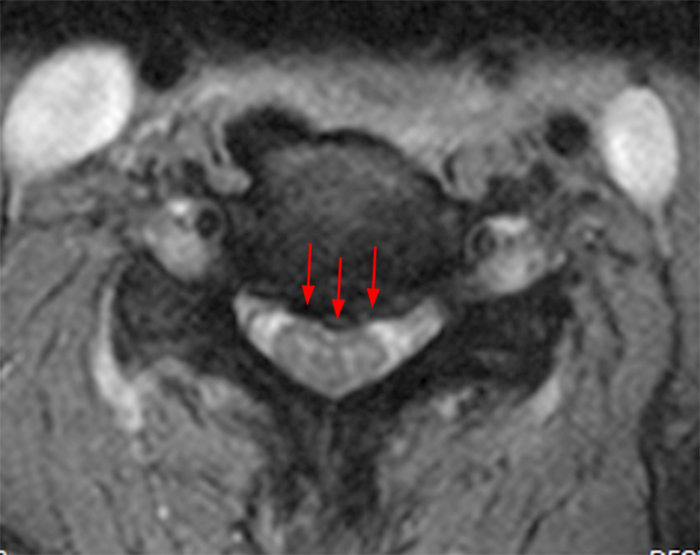
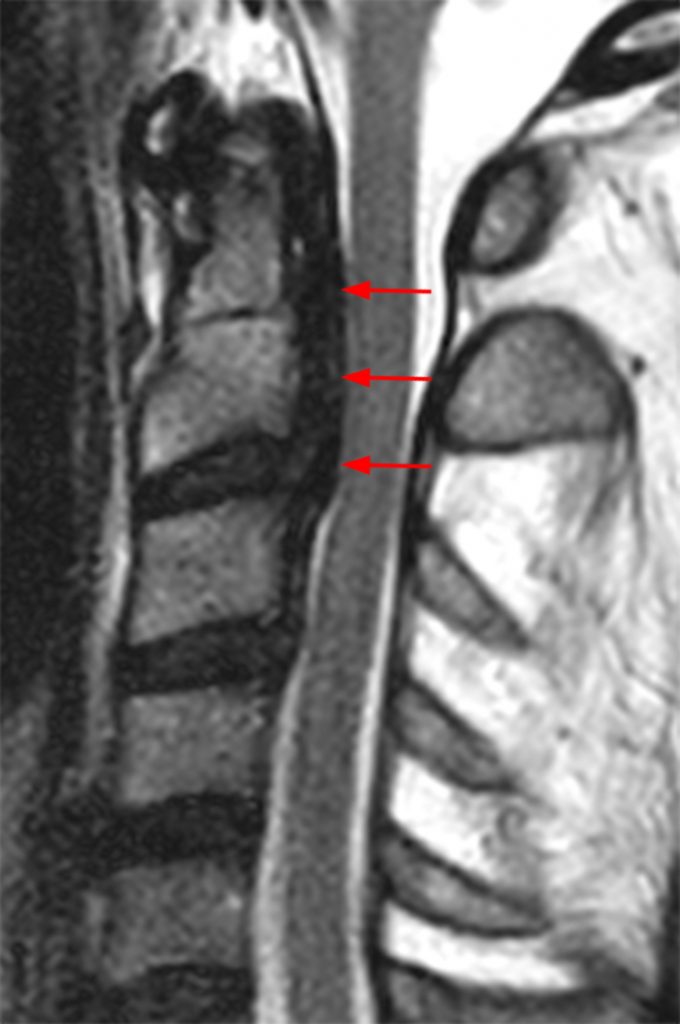
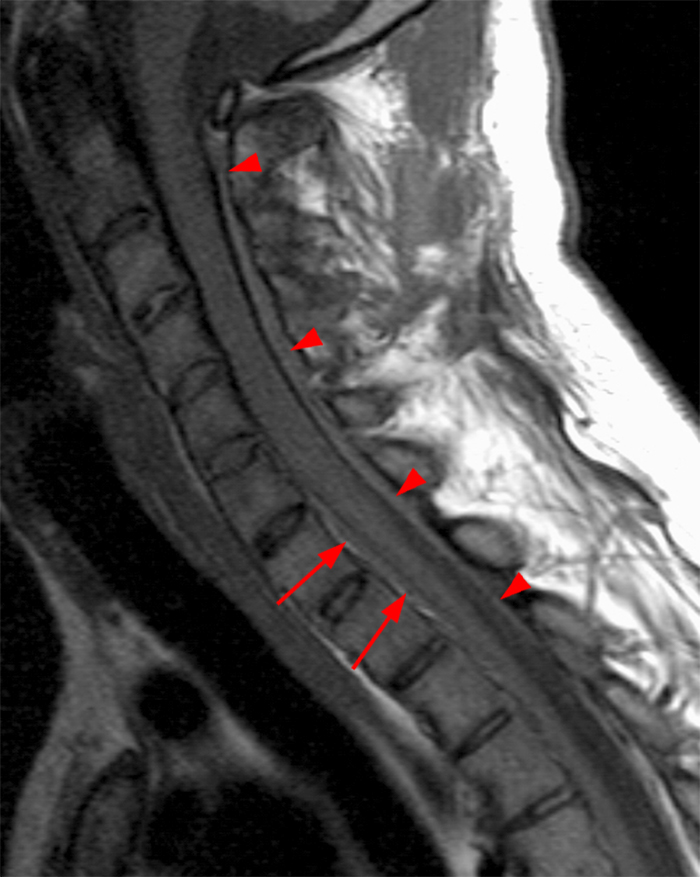
Figure 2:
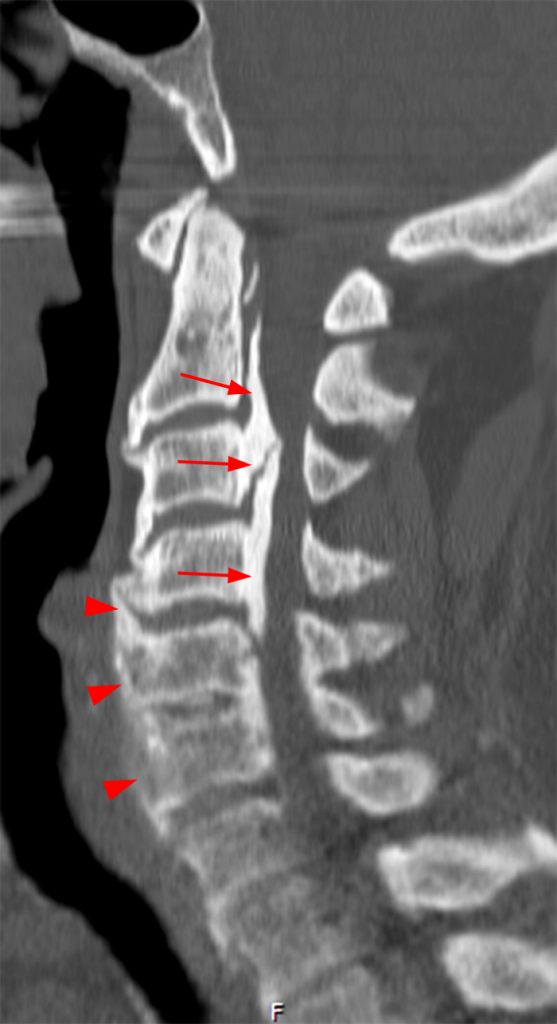
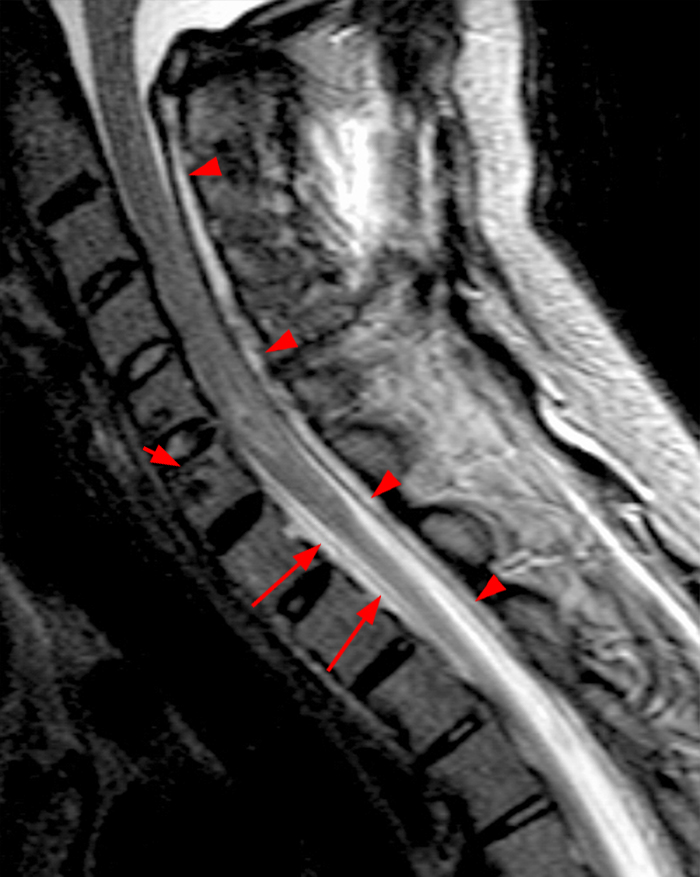
(2a, 2b) An extensive low signal intensity epidural abnormality posterior to the vertebral bodies and discs from C2 through C6 (arrows) produces spinal stenosis, most severe at C3, where there is resultant cord compression and faint T2 cord signal hyperintensity. Ossifications are present anterior to the vertebral bodies and discs (arrowheads).
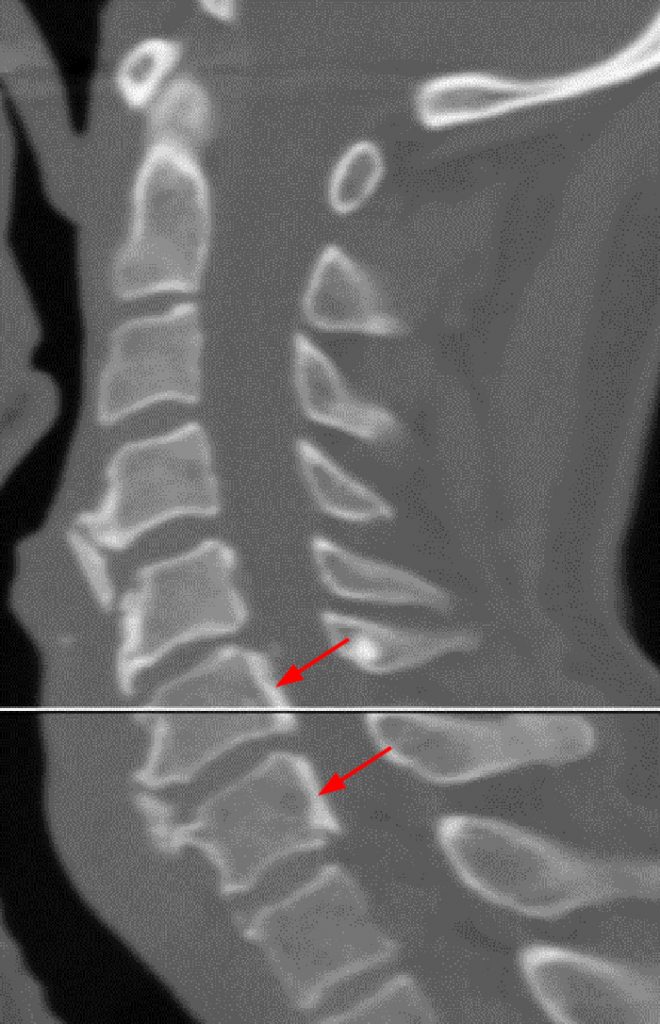
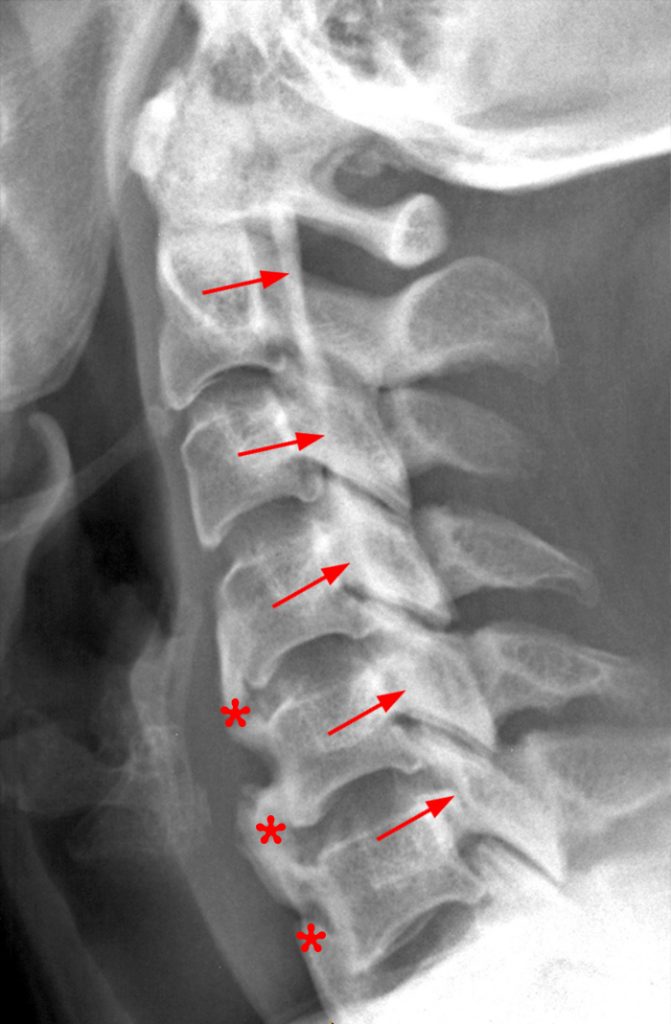
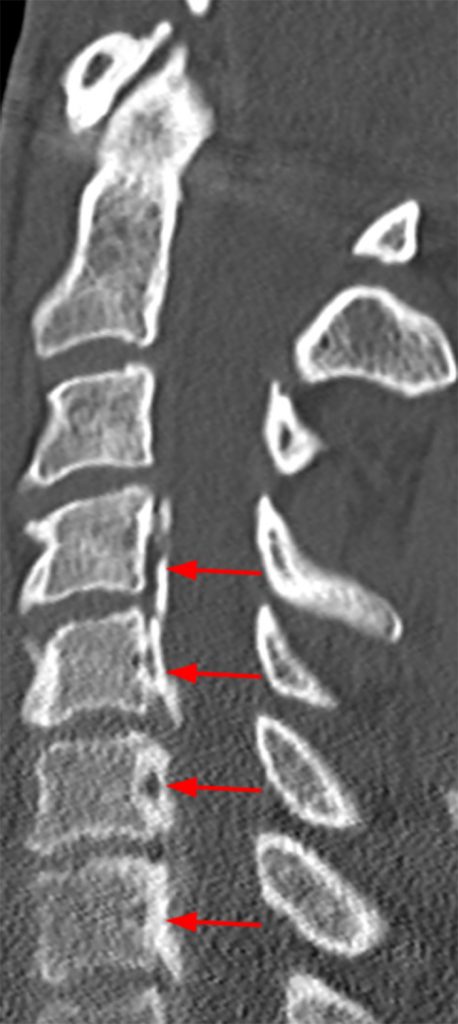
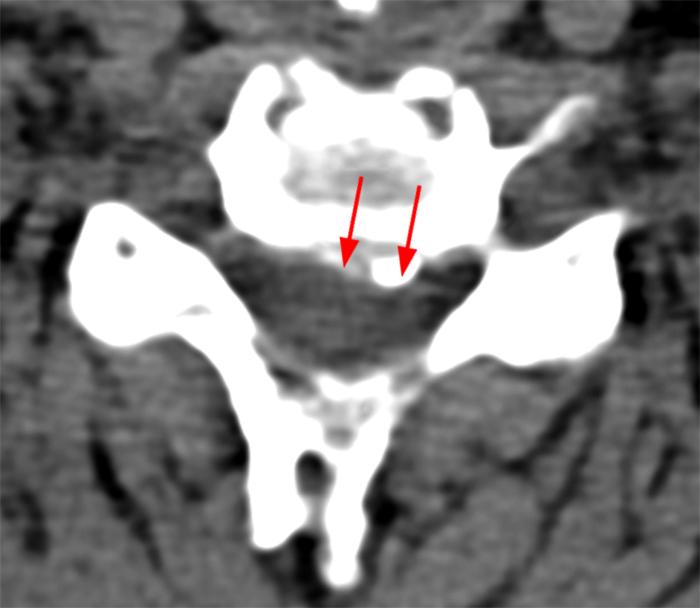
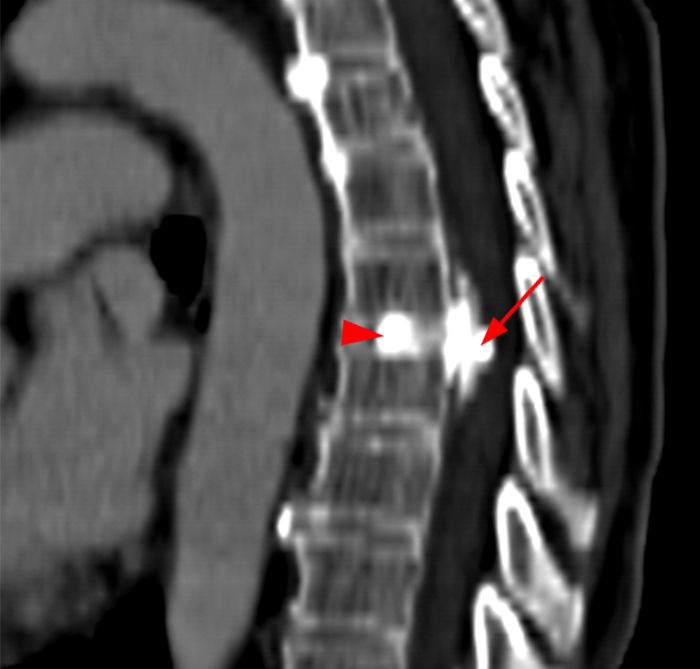
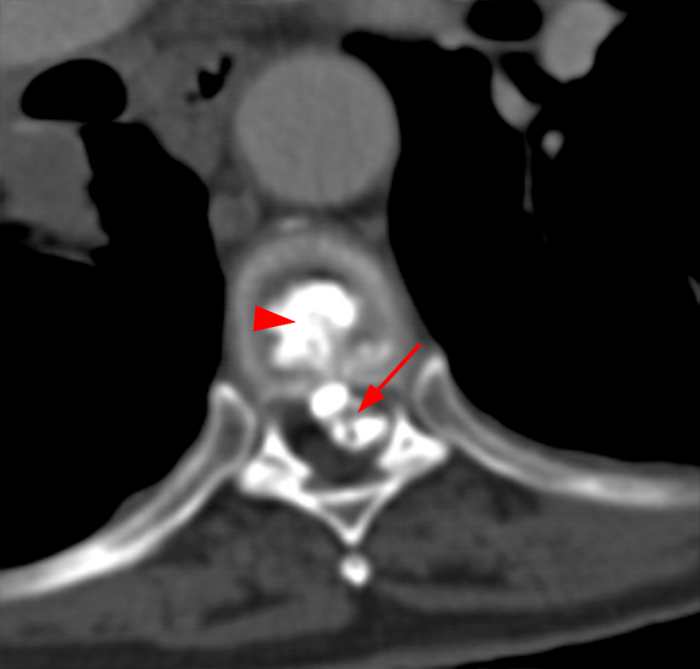
A reconstructed sagittal CT image (3) performed one week prior to the MR is available for correlation. What is your diagnosis now?
Additional Findings
Diagnosis
Ossification of the posterior longitudinal ligament (OPLL), continuous type, producing spinal stenosis, cord compression and edema. Diffuse idiopathic skeletal hyperostosis (DISH) is also present.
Introduction
Ossification of the posterior longitudinal ligament (OPLL) is an underrecognized bone-forming diathesis resulting in ectopic ossification of the posterior longitudinal ligament.1 First reported by Key in 1838, it is an important diagnosis because of its potential to compress the spinal cord, accounting for nearly twenty-five percent of cases of cervical myelopathy.2,3 Establishing OPLL as the cause of spinal stenosis and myelopathy is important for appropriate surgical management.
Pathogenesis:
The normal posterior longitudinal ligament contains collagen and elastin fibers, extends from the clivus to the sacrum. It is up to 1-2 mm in thickness, widest at the disc levels and narrowest at the mid-vertebral level. In OPLL, fibroblastic hyperplasia and increased collagen deposition produce hypertrophy of the ligament. Progressive mineralization and cartilage ingrowth occur, resulting eventually in Haversian canals and mature bone.4 OPLL has been attributed to a combination of genetic and environmental factors. Polymorphisms of collagen genes and overproduction of collagen, serving as a framework for production of ectopic endochondral ossification, have been implicated.1 OPLL is much more common in Asian populations, particularly the Japanese, with a prevalence in Japan and East Asian countries ranging from 1.9-4.3%. In contrast, its frequency ranges from 0.01-1.7% in Caucasian populations. The published incidence of the disease has increased with improved recognition.1 The male /female ratio is 2:1 and the age of onset is typically in the sixth decade with an age range of 19 to 85 years.1,5,6 OPLL is frequently associated with diffuse idiopathic skeletal hyperostosis (DISH) and diabetes mellitus (Figs. 2,3). In the North American population, the rate of concurrent OPLL and DISH was found to be 50%.1 DISH is a common condition in the middle-aged and elderly in the US, with frequencies up to 12%, so the incidence of OPLL in the US is likely much greater than reported.7 Additional factors contributing to underdiagnosis include lack of symptoms in some patients and early changes often not appreciated on lateral radiographs.8 In a study of Japanese patients, the rate of concurrency of OPLL and DISH was 25%, considerably lower than in the US.1 In contrast to its frequent association with DISH, OPLL is less often associated with ossification of the ligamentum flavum.5 The prevalence of diabetes mellitus in patients with OPLL ranges from 20 to 34%.1 Other frequent comorbid and potential risk factors include systemic hypertension, hypothyroidism, dyslipidemia and a history of meningioma.1 Increased bone mineral density has been reported in patients with OPLL.9
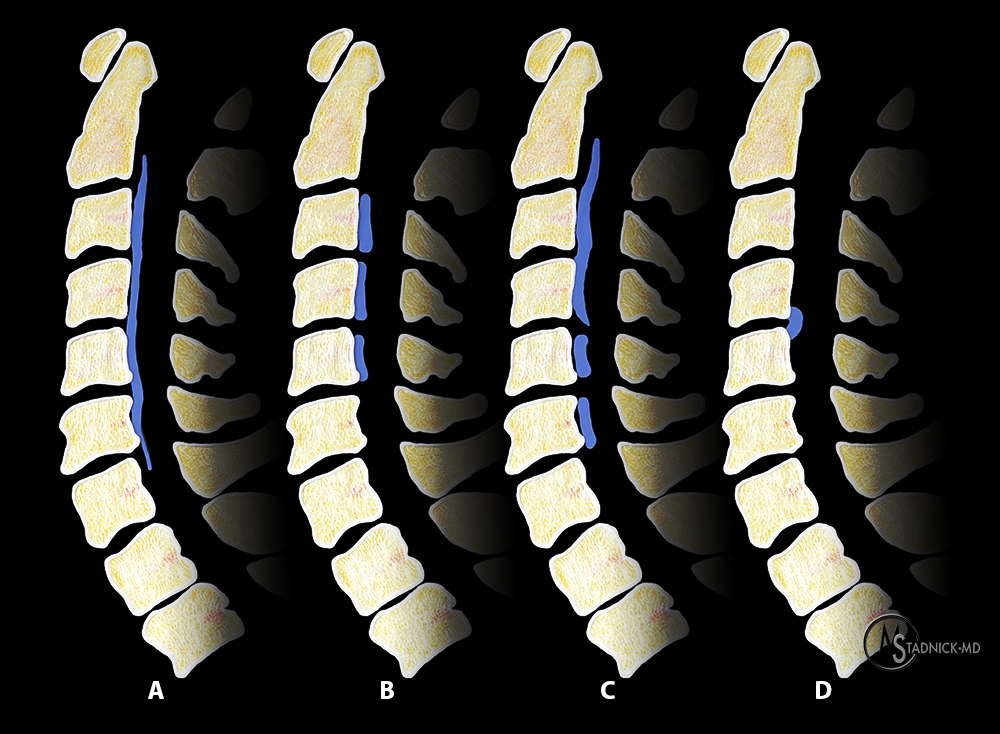
There are four subtypes of OPLL based on its location: 1) continuous, flowing ossification at several levels and their intervening discs; 2) segmental, located posterior to each vertebral body and not extending beyond the adjacent disc level; 3) mixed, a combination of segmental and continuous types; and 4) focal, occurring at the disc level (Fig. 4).1,6 Segmental or continuous types are most common, depending on the series, followed by mixed, and much less commonly the focal type (Figs. 5,6). In Epstein’s series, the segmental type was most common, occurring in 39% of cases, followed by mixed (29%), continuous (27%) and focal types (5%).10
Figure 4:
Classification of OPLL: (a) Continuous, long flowing ossification at several vertebral levels and their intervertebral discs, (b) segmental, separate lesions posterior to the vertebral bodies not extending across the adjacent disc levels, (c) mixed, a combination of continuous and segmental types, and (d) focal, occurring at the disc level.
Figure 5:
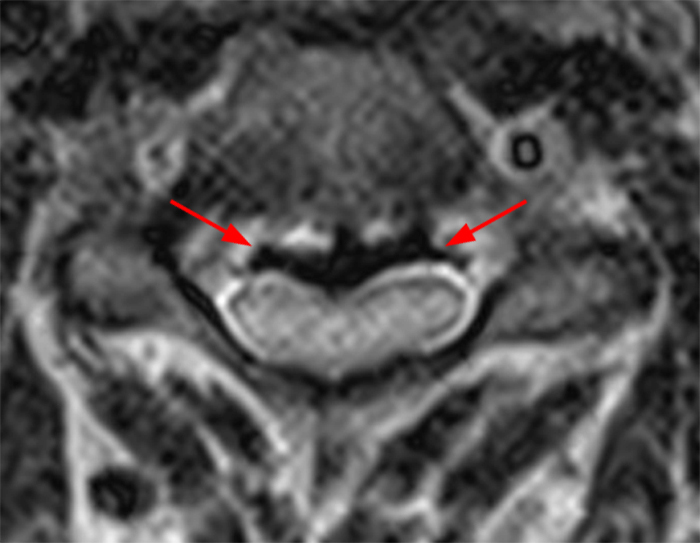
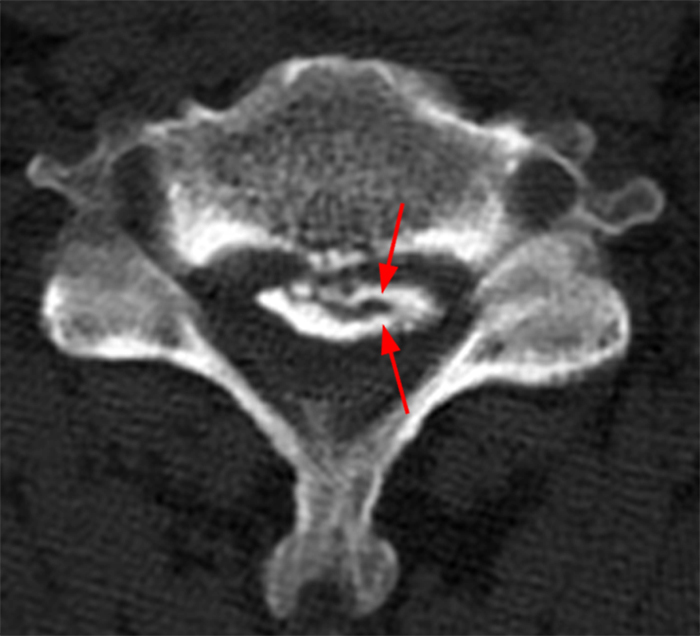
Continuous and focal types of OPLL in a 49-year-old man with neck pain. T1 and T2-weighted sagittal images (5a,b) at the midline of the cervical spine reveal the OPLL is low signal intensity and is both continuous, from C2 through C6, and focal, at T1-2 (arrows). Note also the presence of DISH (arrowheads). On the lateral radiograph (5c) the OPLL is conspicuous as a thick, radiodense, continuous anterior band. DISH present anteriorly (arrowheads). On the axial T2-weighted image at C3-4 (5d), note that the lateral margins of the OPLL are squared (arrowheads).
Figure 6:
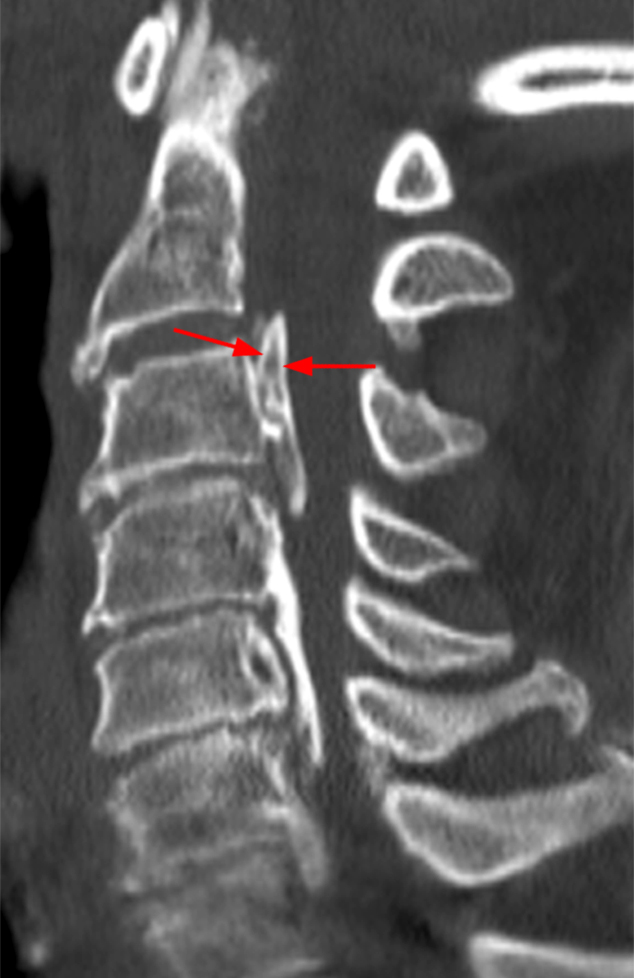
Segmental type of OPLL in a 53-year-old woman who developed neck pain and tingling in hands immediately after falling into a door. The ossification is confined to the vertebral body levels at C6 and C7 (arrows) on (6a) a sagittal reconstructed CT image of the cervical spine, indicative of the segmental type of OPLL. The ossification is less conspicuous on the corresponding (6b) sagittal T2-weighted MR image (arrows). Note the resultant spinal canal narrowing. The hyperintense area within the cord at C6 (arrowhead) is compatible with injury. The T2* gradient-echo axial MR (6c) image demonstrates the inverted “hill-shape” of the OPLL at C6 (arrows).
OPLL occurs most frequently in the cervical (70%) followed by the thoracic spine (15%), and upper lumbar spine (L1-3) (15%) (Fig. 7, 8).1 In Yamashita’s series, only four of 100 cases occurred at the lumbar level and all of these were at the disc level. 5 Within the cervical spine, ossification is most frequent at the C4-6 levels, and the average number of involved levels is 3.8 The mixed and continuous types are most often associated with myelopathy.9
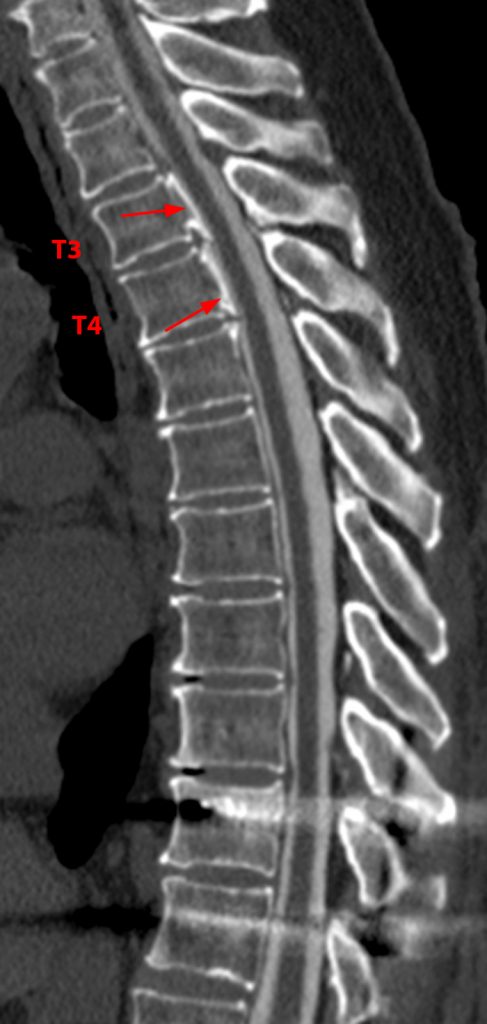
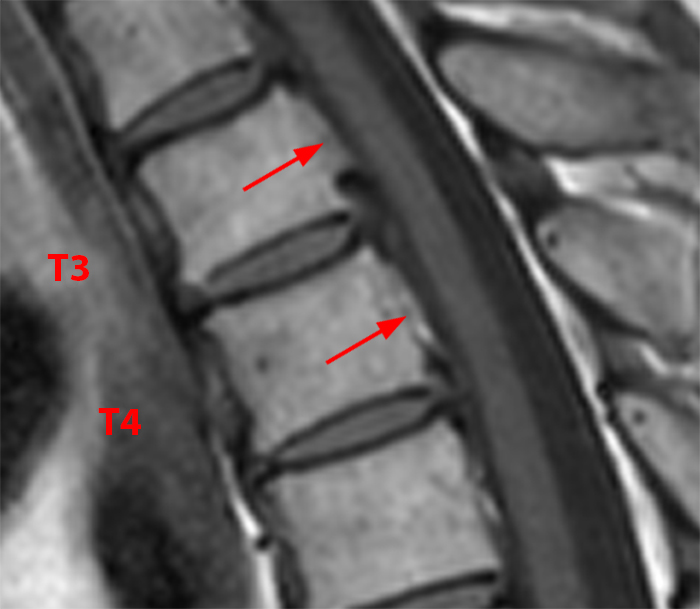
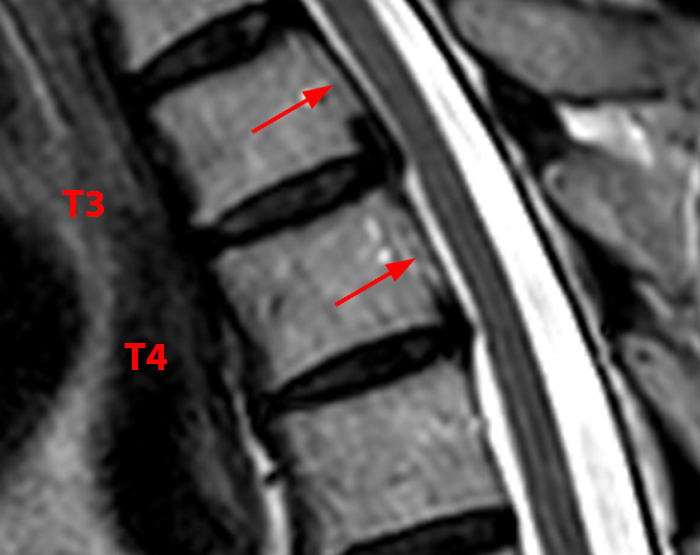
Figure 7:
Thoracic segmental OPLL in a 62-year-old woman with back pain. 2D reconstructed sagittal CT images (7a,b) demonstrate segmental ossification posterior to T3 and T4 (arrows). The T1 and T2-weighted sagittal MR images (7c,d) reveal that the ossifications (arrows) are isointense to fat, and that the findings are more subtle on MRI as compared to CT. Mild cord compression (arrow) is apparent on the corresponding (7d) T2-weighted axial image.
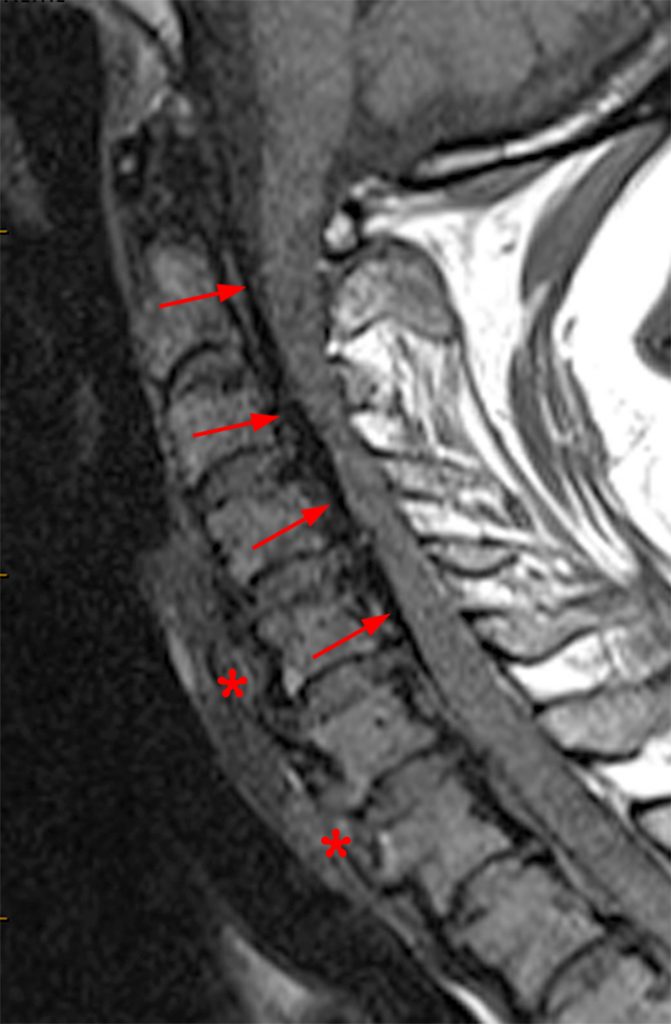
Figure 8:
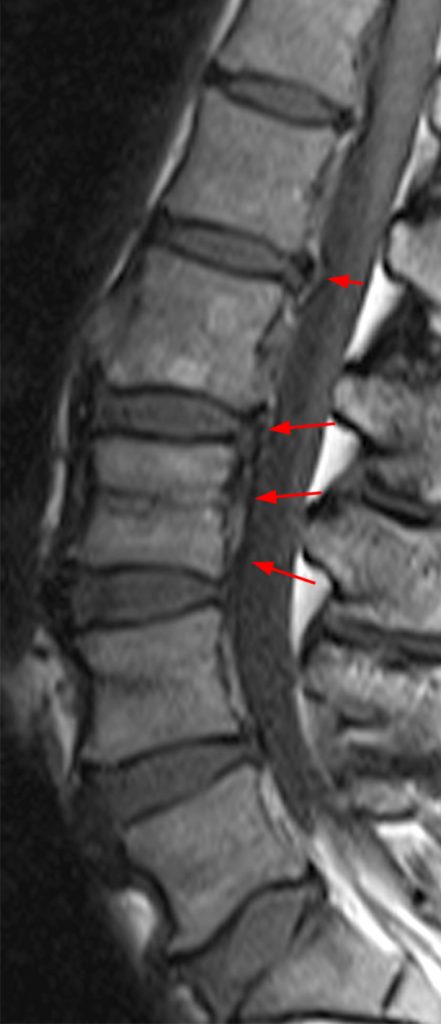
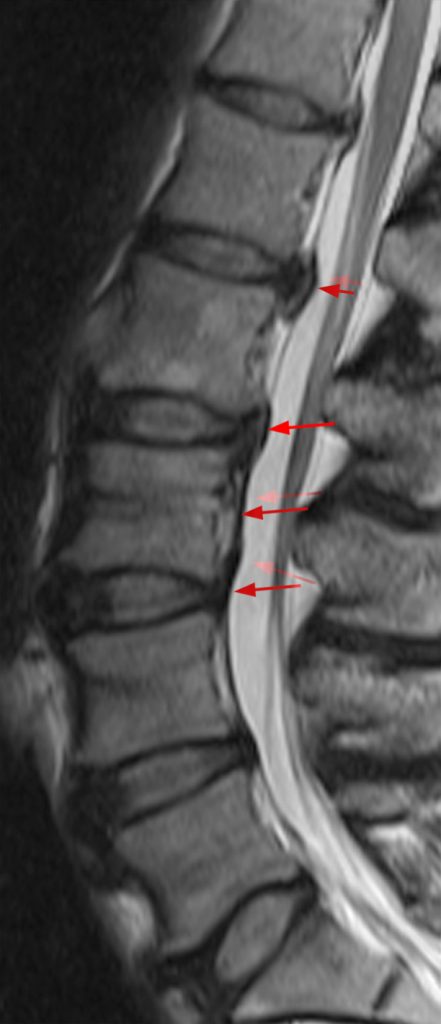
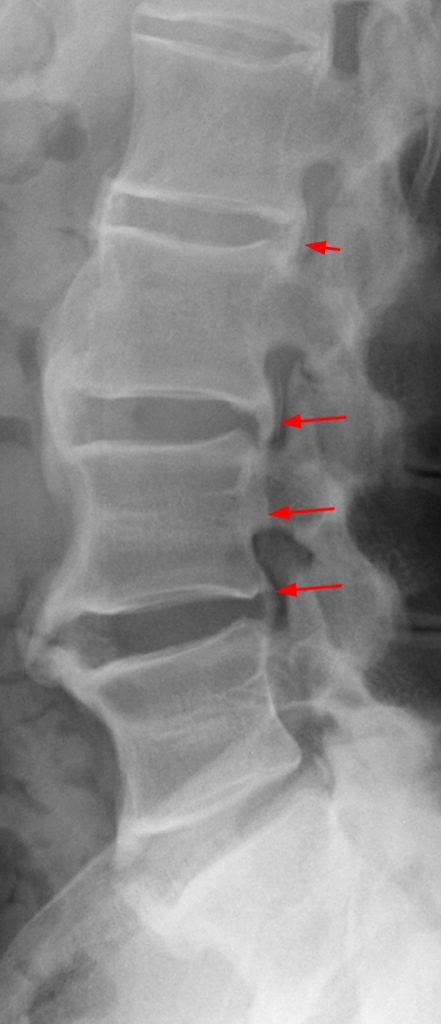
Cervical and lumbar OPLL in a 49-year-old woman with neck pain and stiffness who developed low back pain and left leg numbness after a twisting injury. T1-weighted sagittal (8a), T2-weighted sagittal (8b) and a lateral radiograph (8c) of the cervical spine demonstrate continuous type OPLL present at multiple levels of the cervical spine (arrows), resulting in spinal stenosis and cord compression. DISH is also present (asterisks). The (8d) axial T2-weighted MR image demonstrates a mushroom appearance of the OPLL (arrows). T1-weighted sagittal (8e), T2-weighted sagittal (8f) and a lateral radiograph (8g) of the lumbar spine reveal focal OPLL at L1-2 (short arrow) and continuous OPLL at L2-3 through L3-4 (arrows). Lumbar DISH is also present.
Clinical Presentation
Patients with OPLL may be asymptomatic and the diagnosis is discovered as an incidental finding.11 The clinical manifestations of OPLL are determined by the level and severity of spinal cord compression. Cervical and thoracic disease frequently manifest with myelopathy, and lumbar involvement may result in signs and symptoms of spinal stenosis.1 In approximately 21% of patients, a mild traumatic event results in acute clinical deterioration (Fig. 6).8 Some patients may present with radicular symptoms and signs such as a positive Spurling test.9 In patients who initially present with myelopathy, the disease commonly progresses over time. Less commonly, myelopathy develops in patients who are initially asymptomatic.3 Clinical progression is usually slow, explaining why marked canal compromise can sometimes be seen with relatively mild symptoms (Fig. 9).8 Rate of progression is related to age at presentation, with presentation in the fourth decade or younger associated with progressively worsening symptoms and subsequent myelopathy. In contrast, presentation in the fifth and six decades is less frequently associated with myelopathy.12,13,14
Figure 9:
69-year-old woman with neck pain radiating into the shoulders and hand paresthesias for 6 months. The T1-weighted sagittal image (9a) demonstrates intermediate signal intensity within the OPLL in, corresponding to fatty marrow (arrowheads). Compressive myelomalacia (arrow) is apparent at C5-6 on (9b) the T2-weighted sagittal view. The sagittal CT reconstruction (9c) demonstrates the double line sign at C3 (arrows), corresponding to the radiodensity of cortical bone separated by low-density fatty marrow. An axial CT image at C5 (9d) depicts the inverted T sign of the ossified mass, as well as the double line sign (arrows).
Imaging
Radiographs and CT: The diagnosis of OPLL is frequently made on radiographs and CT images.6 Ossification manifests as increased radiodensity and is present posterior to the vertebral bodies, discs or both. Radiographs are less sensitive than CT, since OPLL is often superimposed on the facet joints on lateral radiographs and may be easily overlooked (Fig. 10).8
On axial CT images, lesions range from thin to bulky, with an “upside-down T” or “bowtie” configuration (Figs. 9, 10).11 In addition to the detection of OPLL, risk factors for progression of symptoms can be identified on radiographs and CT images. The occupancy ratio is predictive of myelopathy and is defined on sagittal or lateral images as the percentage of the spinal canal diameter occupied by the OPLL.1 A ratio of 30 to 60% is predictive of the development of myelopathy, and 100% of patients with an occupancy ratio of greater than 60% develop myelopathy.9 OPLL masses that are more laterally deviated are associated with a higher risk of myelopathy than those at the midline. An anterior-posterior space available for the cord of 6-9 mm is also predictive of myelopathy. The dura may become ossified and adherent to the OPLL mass.8 Penetration of the dura by OPLL creates a challenge for surgical management due to the increased risk of a dural defect and CSF leak when the bony mass is removed. CT can be used to assess for single and double-layer signs, which predict the likelihood of dural involvement. The single-layer sign is used to describe a lesion that is uniformly hyperdense. The double-layer sign describes a lesion possessing anterior and posterior hyperdense rims separated by a hypodense area (Figs. 10b; 9c, d). The double-layer sign is associated with a greater likelihood of a dural defect.15,16 A high occupying ratio is also likely associated with dural penetration.1
Figure 10:
OPLL in a 54-year-old woman who developed neck pain and left arm radiculopathies after falling. On (10a), the lateral radiograph, OPPL from C4 to C6 is relatively thin and projects over the facets, decreasing conspicuity and making it easy to overlook (arrows). In contrast, the finding is obvious on the CT image (10b). Note the single line sign of uniform increased density of the OPLL (arrows). The OPLL (arrows) produces mass effect on the left ventral cord, readily apparent on the axial view (10c).
MR Imaging
The MR imaging findings of OPLL may be subtle or confused with disc herniations or meningioma. The ossification is demonstrated as a low signal intensity band of varying thickness between the bone marrow of the vertebral body and the dural sac on T1- and T2-weighted images, resulting in increased distance between these structures (Fig. 2, 9).5,6,8 The low signal intensity of OPLL may make it difficult to detect on MR images, particularly the segmental type (Fig. 6). Correlation with radiographs or CT is helpful for improving detection or confirming a suspected diagnosis of OPLL on MR images (Fig. 11). Intermediate or increased signal intensity bone marrow within the area of ossification on T1-weighted images may be present, reported in 56% of cases of continuous and 11% of segmental OPLL, and in 41% of the patients in Otake’s series (Figs. 7, 9).5,6 The signal increase is due to the presence of fatty marrow and is more frequent in thicker lesions.6 The continuous type is more easily recognized than segmental at MR imaging because it shows thicker ossification and therefore signal increase within the area is more common.5,6. A mean thickness of 9.1 mm in the continuous lesions of Otake’s series, compared to 4.3 mm of the segmental type was reported. The lesions may have a square, hill or mushroom shape (Figs. 5d, 6c, 8d).6 In the four cases of lumbar OPLL in Yamashita’s series, which all occurred at the disc level, differentiation from disk disease was difficult.5
Cord compression caused by OPLL is common, with moderate or severe compression occurring in up to 44% of patients in one series (Figs. 2, 5,8,9).6 Cord compression is significantly more severe and the occurrence of T2 hyperintensity in the spinal cord is greater in continuous as compared to segmental OPLL.5 Spinal cord T2 signal increase in patients with cord compression resulting from OPLL may correspond to edema, demyelination, myelomalacia and/or necrosis.17,18
Figure 11:
70-year-old man complaining of neck spasms since falling. A sagittal T2-weighted MR image (11a) A band of low signal intensity is present posterior to the C2 vertebral body and C2-3 disc (arrows). Differential considerations include OPLL and a disc extrusion. The lateral radiograph (11b) confirms the diagnosis of OPLL (arrows).
Differential Diagnosis
The differential diagnosis of OPLL includes other mass or plaque-like ventral epidural lesions. Calcified disc herniation should be considered, particularly for single level involvement, which is uncommon in OPLL (Fig. 12). The typical multilevel involvement of OPLL rarely occurs in calcified disc herniations.6 Calcified meningioma is another consideration, though it uncommonly occurs in the epidural space.5,19 Meningioma is usually round and usually does not extend longitudinally as does OPLL.6 Subacute epidural hematoma shows T1 signal increase, but unlike OPLL, there is corresponding increased signal intensity on T2-weighted images (Fig. 13). A less common lesion to be considered is epidural hemangioma, though the contrast enhancement of this lesion is not a feature of OPLL (Fig. 14).
Figure 12:
Calcified disc extrusion simulating OPLL. 2D sagittal reconstructed (12a) and axial CT images (12b) reveal a densely calcified disc extrusion at the mid-thoracic spine (arrows), with an appearance similar to localized OPLL. Note also the presence of calcification within the intervertebral disc (arrowheads).
Figure 13:
Epidural hematoma in a patient with ankylosing spondylitis who sustained a Chance fracture of the C6 vertebral body and disruption of the posterior ligaments. A T1-weighted sagittal image (13a) reveals an intermediate and high signal intensity anterior epidural process at C7 and T1 (arrows). A similar finding is noted posteriorly, from C2 to T2 (arrowheads). A corresponding T2-weighted image (13b) shows the abnormality is of high signal intensity (arrows), eliminating OPLL from consideration, and compatible with subacute hemorrhage, with similar finding posteriorly (arrowheads). A horizontal fracture line is present adjacent to the superior endplate of C6 (short arrow¬). T2 signal increase within the cord at C5 and C6 is likely due to contusion.
Treatment
Conservative treatment is appropriate if neurological signs or symptoms, mainly neck or arm pain, are minimal and there is no evidence of myelopathy.1 Immobilization and skull traction combined with steroidal or nonsteroidal agents may be utilized.
Surgery should be considered for patients with OPLL who have symptomatic myelopathy or myeloradiculopathy and for patients with OPLL and trauma who have sustained spinal cord injury.9 In the cervical spine, approaches include anterior, posterior, or a combination of both. In the anterior approach, the ossified mass is directly removed, and options include corpectomy and fusion or anterior cervical discectomy and fusion. The anterior floating decompression method allows the ossified mass to remain attached to the dura, allowing it to migrate anteriorly after decompression and decreasing the chance of dural tears and CSF leak. The posterior approach includes laminoplasty, laminectomy, or laminectomy with posterior fusion for decompression and stabilization. Rates of neurological improvement up to 92% have been reported following anterior surgery and have been associated with fewer complications compared to posterior procedures. Laminoplasty and laminectomy rates of neurological improvement up to 67% and 42%, respectively, are reported. Increasing age, misalignment of the cervical spine, a poor preoperative neurological score, intramedullary high signal intensity within the cord on T2-wieghted MR imaging, continuous and mixed OPLL subtypes and a mass occupying ratio of greater than or equal to 60% are factors associated with negative surgical outcomes.1,20 Thoracic OPLL surgical outcomes are worse than cervical.1
Conclusion
OPLL is an uncommon spinal disorder most frequently affecting the cervical spine, and is an important cause of spinal stenosis and myelopathy. The MR imaging findings may be subtle, particularly for the segmental subtype, or they may be confused with other disorders, including calcified disc herniations and meningioma. Multilevel involvement and the presence of T1 hyperintense fatty marrow within the ossification are useful differentiating features. Radiographs or CT confirm the diagnosis, and CT enables detection of a double layer sign, which is associated with an increased likelihood of dural involvement and risk of CSF leak upon surgical removal of the bony mass. In addition to helping establish the diagnosis, MR imaging is useful in detecting associated cord compression and injury. Indications for surgical treatment include symptomatic myelopathy or acute clinical deterioration after trauma. Identifying OPLL as the cause of spinal stenosis and myelopathy is important for determining appropriate surgical management.
References
- Kalb S, Martirosyan N, Perez-Orribo L, et al. Analysis of demographics,risk factors, clinical presentation, and treatment modalities for the ossified posterior longitudinal ligament. Neurosurg Focus . 2011;30(3):E:11. ↩
- Key C. On paraplegia depending on disease of the ligaments of the spine. Guys Hosp Rep . 1838;3:17-34. ↩
- Smith Z, Buchanan C, Raphael D. Ossification of the posterior longitudinal ligament: pathogenesis, management, and current surgical approaches. Neurodurg Focus . 2011;30(3):E10. ↩
- Epstein N. Ossification of the cervical posterior longitudinal ligament: a review. Neurosurg Focus. 2002;13(2):ECPI. ↩
- Yamashita Y, Takahashi M, Matsuno Y, et al. Spinal cord compression due to ossification of ligaments: MR imaging. Radiology. 2014. doi:10.1148/radiology.175.3.2111569 ↩
- Otake S, Matsuo M, Nishizawa S, Sano A, Kuroda Y. Ossification of the Posterior Longitudinal Ligament: MR Evaluation. ↩
- Resnick D, Guerra J, Robinson CA, Vint3 VC. Association of diffuse idiopathic skeletal hyperostosis (DISH) and calcification and ossification of the posterior longitudinal ligament. 1978. www.ajronline.org. ↩
- Reinsel TE, Andersson GBJ. Ossification of the posterior longitudinal ligament. Am J Neuroradiol. 1992;13(4). ↩
- Abiola R, Rubery P, Mesfin A. Ossification of the posterior longitudinal ligament: etiology, diagnosis, and outcomes of nonoperative and operative management. Glob Spine J. 2015. doi:10.1055/s-0035-1556580 ↩
- Epstein N. Diagnosis and surgical management of cervical ossification of the posterior longitudinal ligament. Spine J. 2002. doi:10.1016/S1529-9430(02)00394-7 ↩
- Shah L, Borg B, Crim J, Petersilge C, Merrow A. Diagnostic Imaging. Spine. 3rd ed. (Ross J, Moore K, eds.). Philadelphia: Elsevier; 2015. ↩
- Chiba K, Yamamoto I, Hirabayashi H, et al. Multicenter study investigating the postoperative progression of ossification of the posterior longitudinal ligament in the cervical spine: a new computer-assisted measurement. J Neurosurg Spine. 2005;3(1):17-23. doi:10.3171/spi.2005.3.1.0017 ↩
- Hori T, Kawaguchi Y, Kimura T. How does the ossification area of the posterior longitudinal ligament progress after cervical laminoplasty? Spine (Phila Pa 1976). 2006;31(24):2807-2812. doi:10.1097/01.brs.0000245870.97231.65 ↩
- Hori T, Kawaguchi Y, Kimura T. How does the ossification area of the posterior longitudinal ligament thicken following cervical laminoplasty? Spine (Phila Pa 1976). 2007;32(19):E551-6. doi:10.1097/BRS.0b013e31814614f3 ↩
- Min J-H, Jang J-S, Lee S-H. Significance of the double-layer and single-layer signs in the ossification of the posterior longitudinal ligament of the cervical spine. J Neurosurg Spine. 2007;6(4):309-312. doi:10.3171/spi.2007.6.4.4 ↩
- Hida K, Iwasaki Y, Koyanagi I, Abe H. Bone window computed tomography for detection of dural defect associated with cervical ossified posterior longitudinal ligament. Neurol Med Chir (Tokyo). 1997;37(2):173-175; discussion 175-6. doi:10.2176/nmc.37.173 ↩
- Takahashi M, Yamashita Y, Sakamoto Y, Kojima R. Chronic cervical cord compression: clinical significance of increased signal intensity on MR images. Radiology. 1989;173(1):219-224. doi:10.1148/radiology.173.1.2781011 ↩
- Takahashi M, Sakamoto Y, Miyawaki M, Bussaka H. Increased MR signal intensity secondary to chronic cervical cord compression. Neuroradiology. 1987. doi:10.1007/BF00350439 ↩
- Zhang XLH, Yuan XHS. Imaging appearances and pathologic characteristics of spinal epidural meningioma. 2017. doi:10.3174/ajnr.A5414 ↩
- Yudoyono F, Cho PG, Park SH, et al. Factors associated with surgical outcomes of cervical ossification of the posterior longitudinal ligament. Med (United States). 2018. doi:10.1097/MD.0000000000011342 ↩