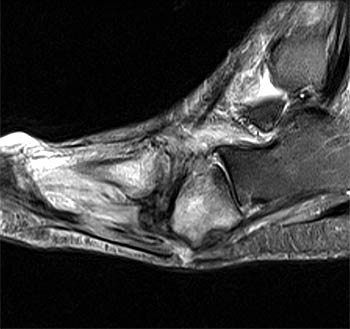
Clinical History: A 60 year-old female presents with foot and ankle pain with swelling for 3 months. No history of trauma. Axial proton density fat-suppressed and sagittal T2-weighted fat-suppressed images are provided. What are the findings? What is the diagnosis?
Findings
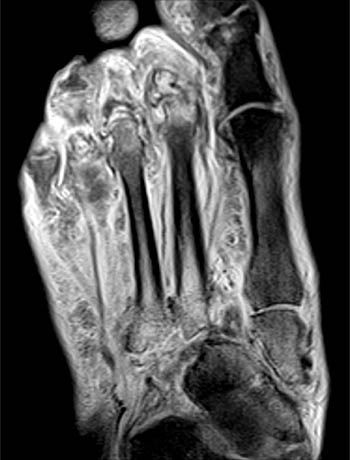
Figure 2:
The axial proton density-weighted fat-suppressed image demonstrates lateral subluxation and fragmentation of the second and third metatarsal bases with diffuse marrow edema (arrows). The navicular (N) is rotated such that its lateral cortex articulates with the second metatarsal base (asterisk), and its distal cortex with the medial cuneiform. There is also marrow edema and cortical irregularity at the metatarsal-phalangeal joints. Diffuse soft tissue edema is present.
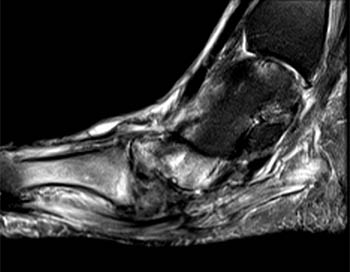
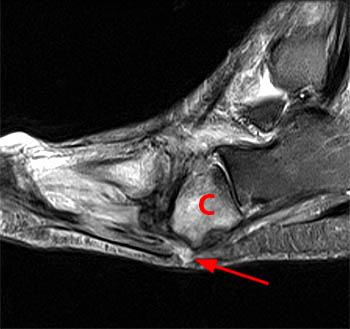
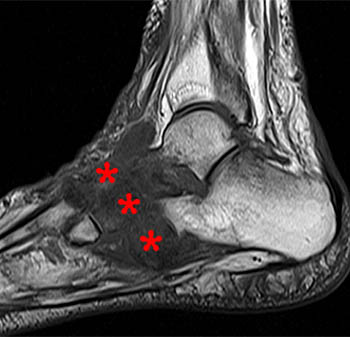
Figure 3:
A sagittal fat-suppressed T2-weighted image demonstrates disruption of the longitudinal arch of the foot, with plantar flexion of the talus (T) and navicular (N), and relative dorsal subluxation of the first metatarsal (asterisk). A fracture of the medial cuneiform (arrow) is also present.
DIAGNOSIS
Charcot foot (neuroarthropathy), with LisFranc dislocation, destruction of the tarsal-metatarsal joints, and a classic “rocker bottom foot” with increased load bearing on the cuboid and subsequent overlying bursa formation.
INTRODUCTION
In 1868, Jean-Martin Charcot gave the first detailed description of a progressive destructive arthropathy due to neuropathic disease in a patient with syphilis.1 For many years syphilis was considered to be the most common etiology of Charcot neuroarthropathy, but now diabetes mellitus is far and away the most common cause, involving primarily the joints of the foot and ankle. It can also be seen in other neurologic disorders with sensory loss in the foot such as leprosy, spinal cord injuries, alcoholic neuropathy, and congenital insensitivity to pain. The pathogenesis of Charcot neuroarthropathy is likely multifactorial. The neurotraumatic theory holds that osseous destruction results from loss of pain sensation and proprioception leading to repetitive unperceived trauma to the foot. The neurovascular theory holds that joint destruction is secondary to an autonomically stimulated vascular reflex causing hyperemia and resulting in mismatch in bone resorption and synthesis.2 Charcot arthropathy is widely thought to result from a combination of both processes.
EPIDEMIOLOGY, CLINICAL DIAGNOSIS AND TREATMENT
Neuropathic arthropathy is prevalent in 0.8 to 7.5% of diabetic patients with neuropathy, and may be bilateral in 9 to 35% of those affected.3 In the acute phase the patient presents with erythema and edema of the foot, with the foot skin temperature elevated 3-7 degrees Fahrenheit over the contralateral foot. Treatment in the acute phase involves relieving pressure by total contact casting. The clinical presentation is often indistinguishable from infection which is a common complication of Charcot arthropathy, suggested by the presence of skin ulcers or draining sinus tracts.
The lifetime risk for developing foot ulcers among diabetic patients is 25% and up to 50% may develop infections.5 Chronic stages of Charcot foot are characterized by visible foot deformity, crepitus, limited range of motion and palpable loose bodies. Surgery is contraindicated in the presence of acute inflammation or possible infection, but it can be performed in the chronic stages to stabilize the foot and reduce the risk of ulceration or further osseous destruction due to altered weight bearing from osseous deformities.
MR IMAGING
MR imaging is the modality of choice for imaging both Charcot arthropathy and infection in diabetics. In early stages MRI can demonstrate marrow edema while plain films remain normal. MRI has surpassed nuclear medicine imaging due to the greater specificity of MRI and its ability to delineate osseous anatomy as well as discrete abscesses and sinus tracts diagnostic of infection.6 MRI is commonly ordered in the diabetic patient to rule out infection in the presence of an ulcer, to evaluate the severity of Charcot arthropathy, or to distinguish between Charcot arthropathy and infection. The vast majority of diabetic foot infections are from direct inoculation rather than hematogenous spread, and are most commonly seen as contiguous spread from a skin ulcer. Infection can also arise from a foreign body or iatrogenic cause. The most reliable way to diagnose osteomyelitis is to track the ulcer or sinus tract to the underlying bone and evaluate for the presence of marrow edema, as evidenced by low signal intensity on T1-weighted images. Abnormally decreased signal intensity on T1-weighted images is a more specific indicator of marrow edema than increased signal intensity on T2-weighted images alone, which may indicate osteitis, or reactive marrow change rather than osteomyelitis.7 Figures 5a-5c demonstrate a case of osteomyelitis with an overlying ulcer and draining sinus tract at the dorsal lateral aspect of the 5th toe. Diabetic foot ulcers, and therefore underlying osteomyelitis, develop at the site of pressure points, either from weight bearing or constrictive shoes in the ambulatory patient, or at dorsal and lateral points of the externally rotated foot in the bed ridden patient.
Figure 5:
(5a) A sagittal inversion recovery-weighted image demonstrates diffuse marrow edema of the proximal phalanx and intermediate phalanx (arrows), with flexion deformity and surrounding soft tissue edema. (5b) The corresponding sagittal T1-weighted image demonstrates abnormal decreased signal intensity of the marrow of the proximal phalanx (arrow) relative to the normal fatty marrow of the metatarsal head, suggesting osteomyelitis rather than osteitis. (5c) A coronal fat-suppressed T2-weighted image shows a sinus tract (arrowheads) draining from the distal tip of the proximal phalanx out through the skin at the site of the ulcer.
The greatest challenge for the clinician and the radiologist is determining whether the red hot swollen foot is from Charcot arthropathy or from osteomyelitis. Location is the most important consideration, since osteomyelitis nearly always develops from the contiguous spread of infection. It often occurs in predictable, more peripheral locations, which tend to be at pressure points. Charcot neuroarthropathy, on the other hand, is periarticular. The most commonly involved joints in neuroarthropathy are the tarsal-metatarsal joints, and the metatarsophalangeal joints, while osteomyelitis occurs distal to the tarsal-metatarsal joints, and at the calcaneus and at the cuboid in the case of the rocker bottom foot. (Figure 6)
Figure 6:
Sagittal STIR (6a) and T1-weighted (6b) images in a patient with previous amputation and rocker bottom foot deformity, who developed a large skin ulcer (asterisk) over the load bearing cuboid (arrows). The T1-weighted image also clearly demonstrates the presence of gas (arrowheads) due to the open wound. Advanced underlying Charcot changes are evident, with osseous loose bodies and extensive fragmentation of midfoot bones and the remaining metatarsal bases.
Osteomyelitis can also occur at the tarsal-metatarsal joints, and this is a diagnostic challenge. Careful inspection will often reveal a meandering sinus tract travelling in from a more peripheral location. The multiplanar capacity of MRI is useful in this regard, since the longitudinal nature of the tract may only be appreciated in one plane, and may be too small to detect in cross section. The addition of intravenous gadolinium contrast often renders an abscess or sinus tract more conspicuous, since only the periphery of the collection will enhance, leaving a low signal intensity center on fat-suppressed T1-weighted post-contrast images. In sinus tracts this is referred to as the “tram track sign.” Another reported finding useful in distinguishing the osseous findings of Charcot arthropathy from osteomyelitis is the “ghost sign,” which is thought to be indicative of osteomyelitis. It has been describes as bones which “disappear” on T1-weighted images and then “reappear” on contrast enhanced or T2-weighted images. In the neuroarthropathic foot the ghost sign is absent because bones are destroyed, not just edematous. (Figure 7)
A recent study outlines the use of dynamic contrast enhanced MRI for the evaluation of treatment in acute Charcot foot in diabetic patients. This study revealed that a reduction in the rate of contrast uptake was a reliable indicator of improvement and that mean healing time was directly related to the baseline contrast uptake rate. While not widely used clinically, it may be valuable as a reproducible parameter to evaluate therapeutic options.8 It should also be noted that due to the presence of concomitant renal disease in some diabetic patients, contrast may be contraindicated because of potential gadolinium-induced nephrogenic systemic fibrosis. The American College of Radiology recommends that gadolinium-based contrast material not be administered to patients with a severely reduced glomerular filtration rate (<30 mL/min/1.73 m2)7,9
CONCLUSION
MRI imaging, with or without intravenous contrast, is the most specific and accurate means for diagnosing Charcot neuroarthropathy and for assessing potential complications or the presence of infections. The detailed osseous anatomy allows evaluation of the precise location and extent of bony destruction associated with both Charcot foot and osteomyelitis. The multiplanar soft tissue detail unique to MRI enables detection of abscesses and sinus tracts which are essential in the distinction between Charcot changes and superimposed osteomyelitis.
References
1 Charcot JM. Sur quelaques arthropathies qui paraissent depender d’une lesion du cerveau ou de la moele epiniere. Arch Des Physiol Norm et Path. 1868; 1:161-71.
2 Brower AC, Allman RM. Pathogenesis of the neurotrophic joint: neurotraumatic vs. neurovascular. Radiology 1981; 139:349-54.
3 Sommer TC, Lee TH. Charcot Foot: The Diagnostic Dilemma. American Family Physician 2001; 64 (9): 1591-1598.
4 Singh N, Armstrong DG, Lipsky BA. Preventing foot ulcers in patients with diabetes. JAMA 2005; 293(2):217-228.
5 Lipsky BA, Berendt AR, Deery HG, et al. Diagnosis and treatment of diabetic foot infections. Clin Infect Dis 2004; 39(7):885-910.
6 Morrison WB, Schweitzer ME, Bock GW, et al. Diagnosis of osteomyelitis: utility of fat-suppressed contrast-enhanced MR imaging. Radiology 1993; 189(1):251-257.
7 Donovan A, Schweitzer ME. Use of MR Imaging in Diagnosing Diabetes-related Pedal Osteomyelitis. RadioGraphics 2010; 30: 723-736.
8 Zampa V, Bargellini I, et al. Role of Dynamic MRI in the follow-up of acute Charcot foot in patients with diabetes mellitus. Skeletal Radiology 2011; 40: 991-999.
9 Kanal E, Barkovich AJ, Bell C, et al. ACR guidance document for safe MR practices: 2007. AJR Am J Roentgenol 2007; 188(6):1447-1474.