Clinical history
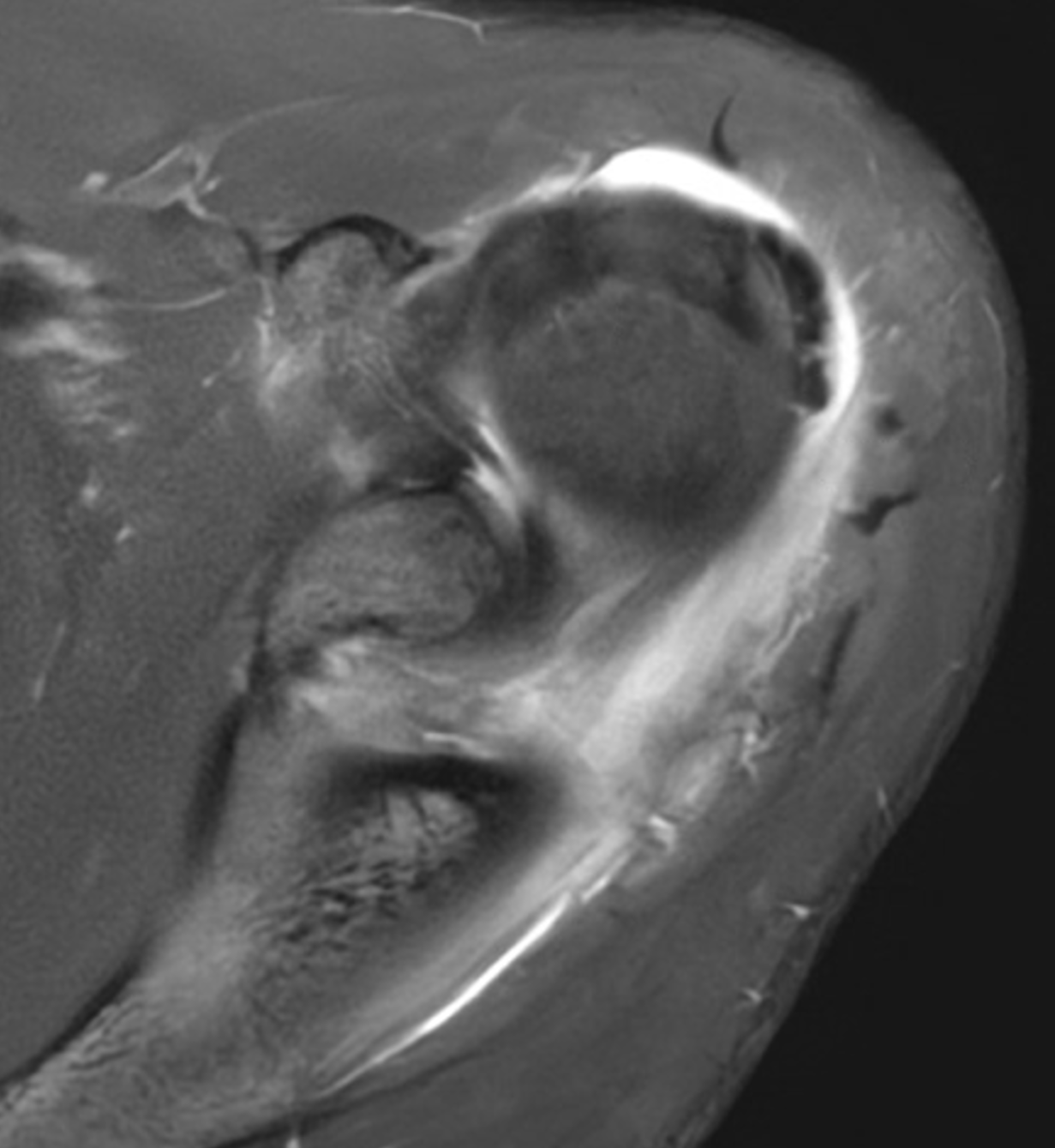
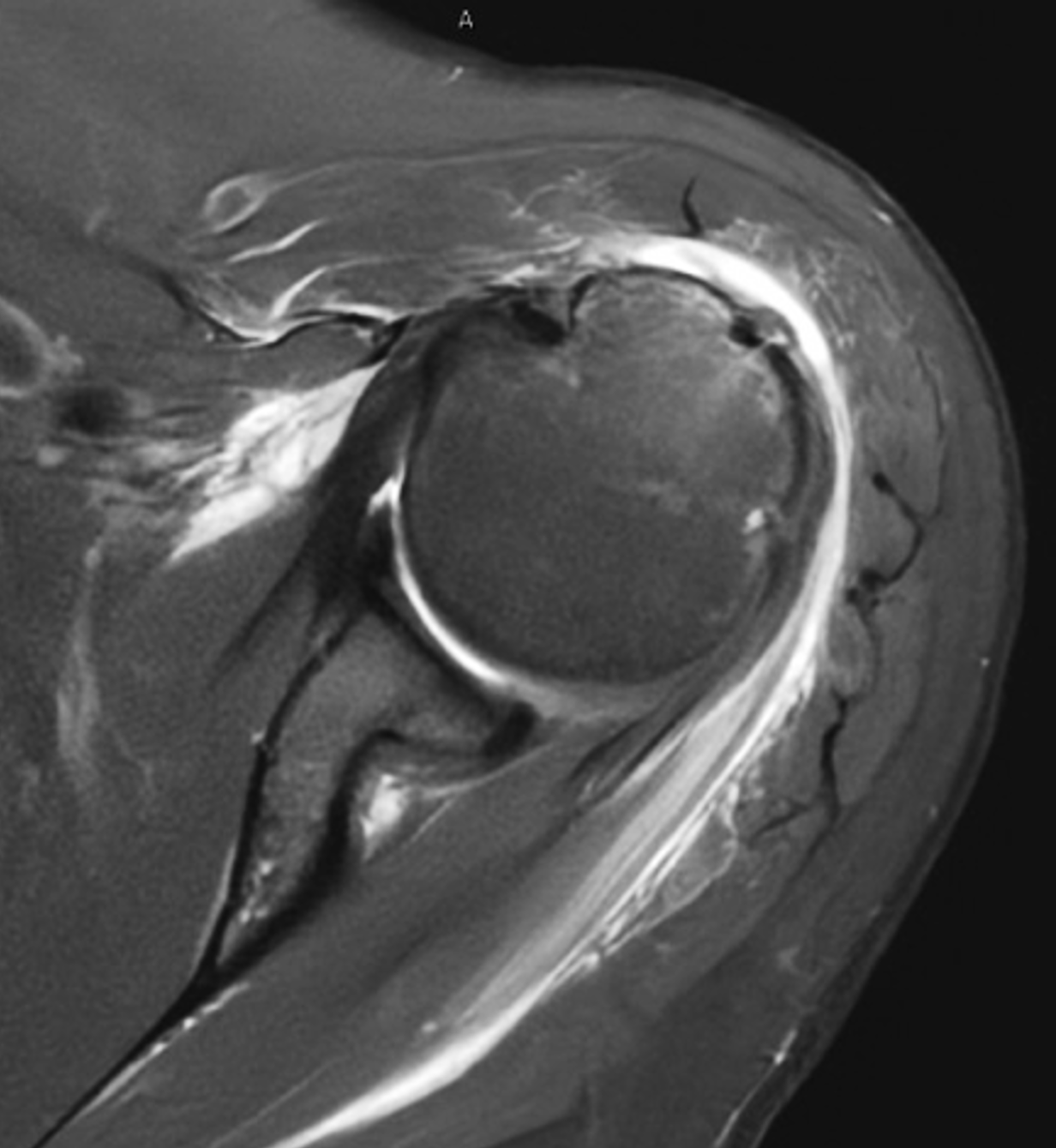
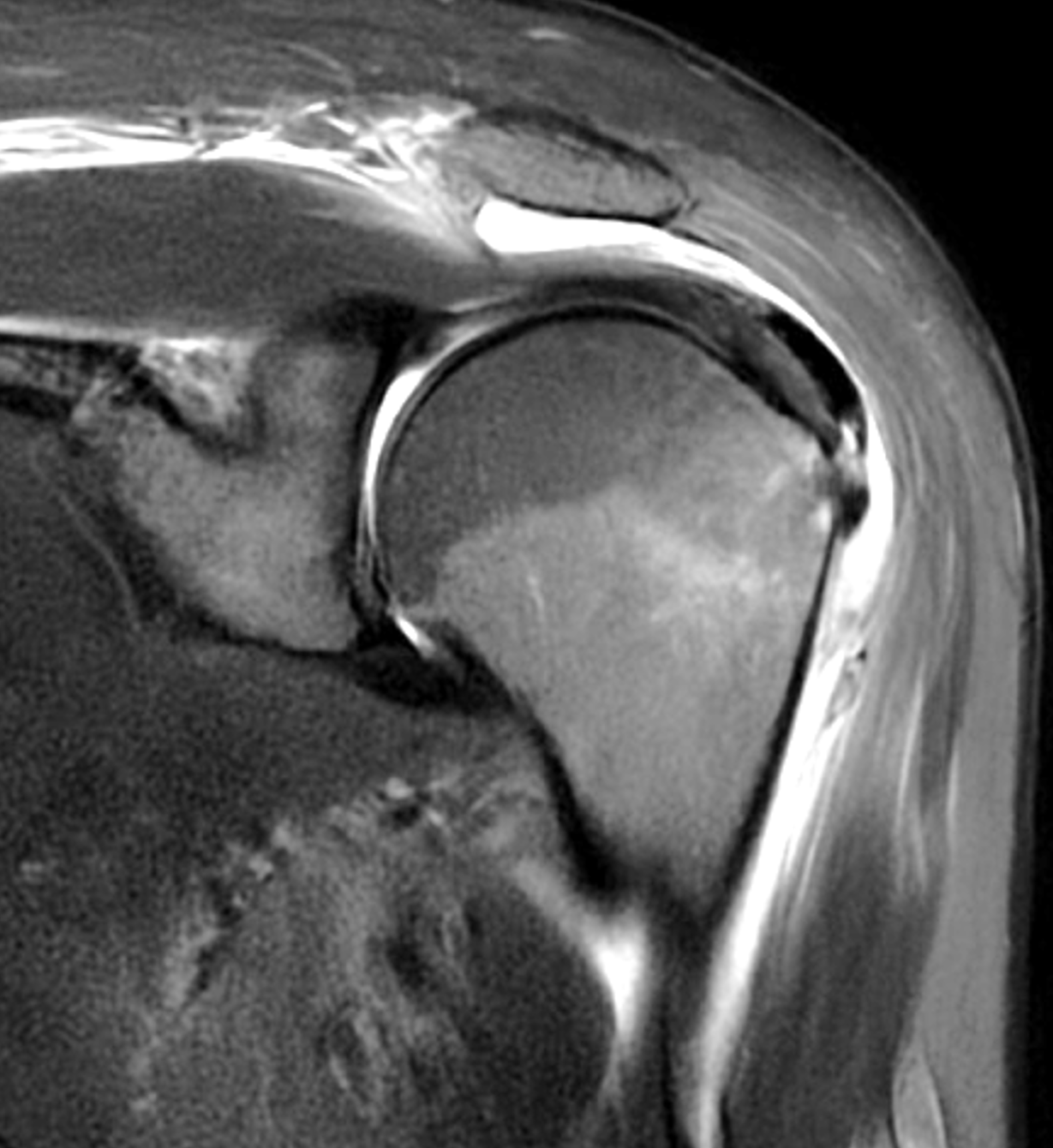
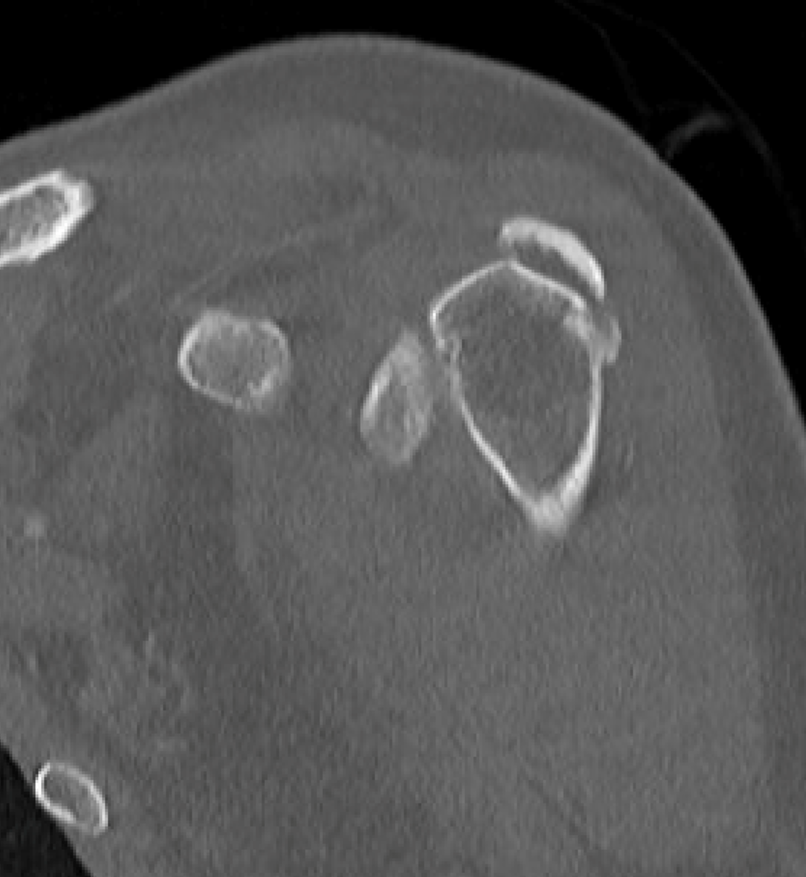
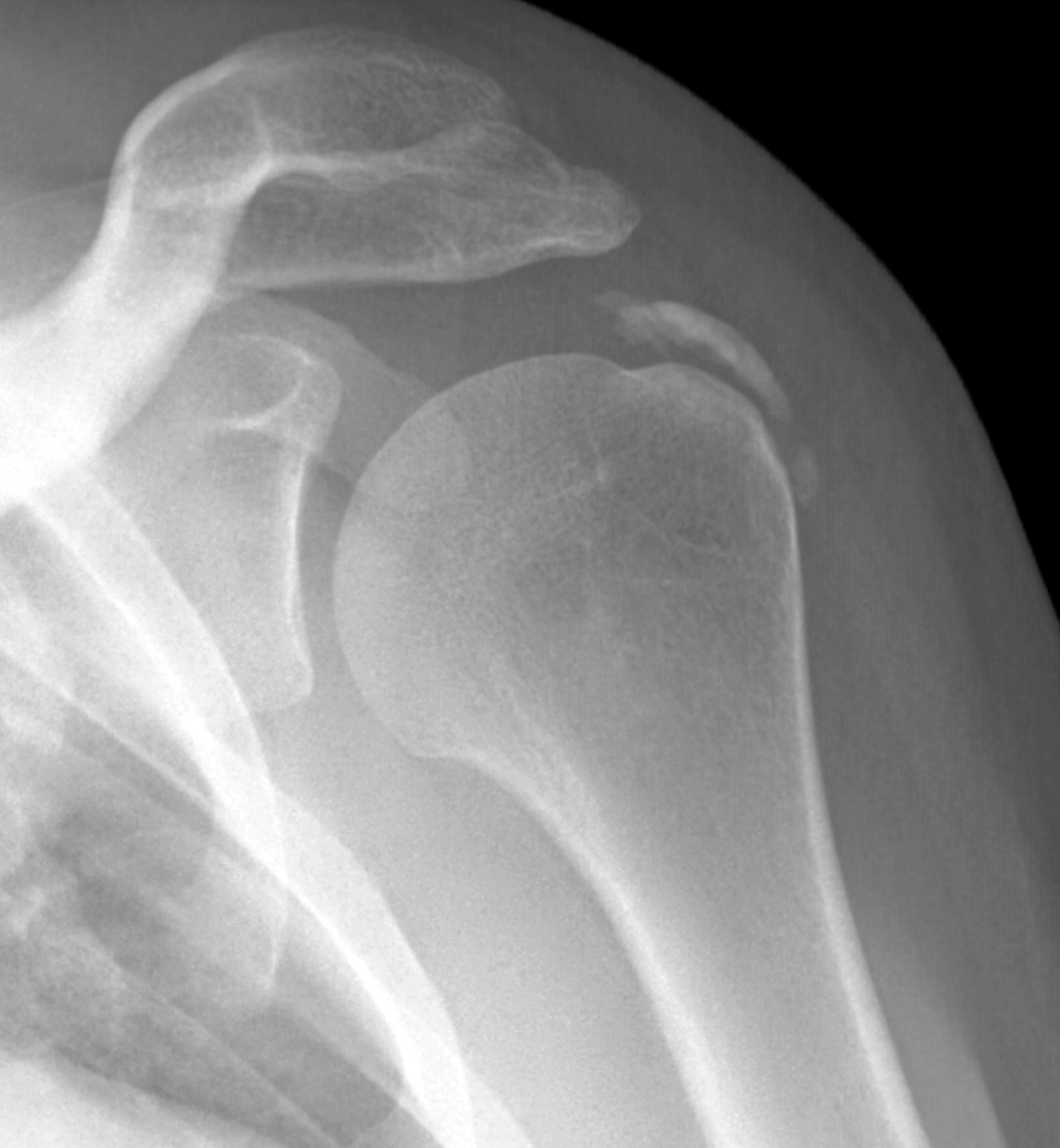
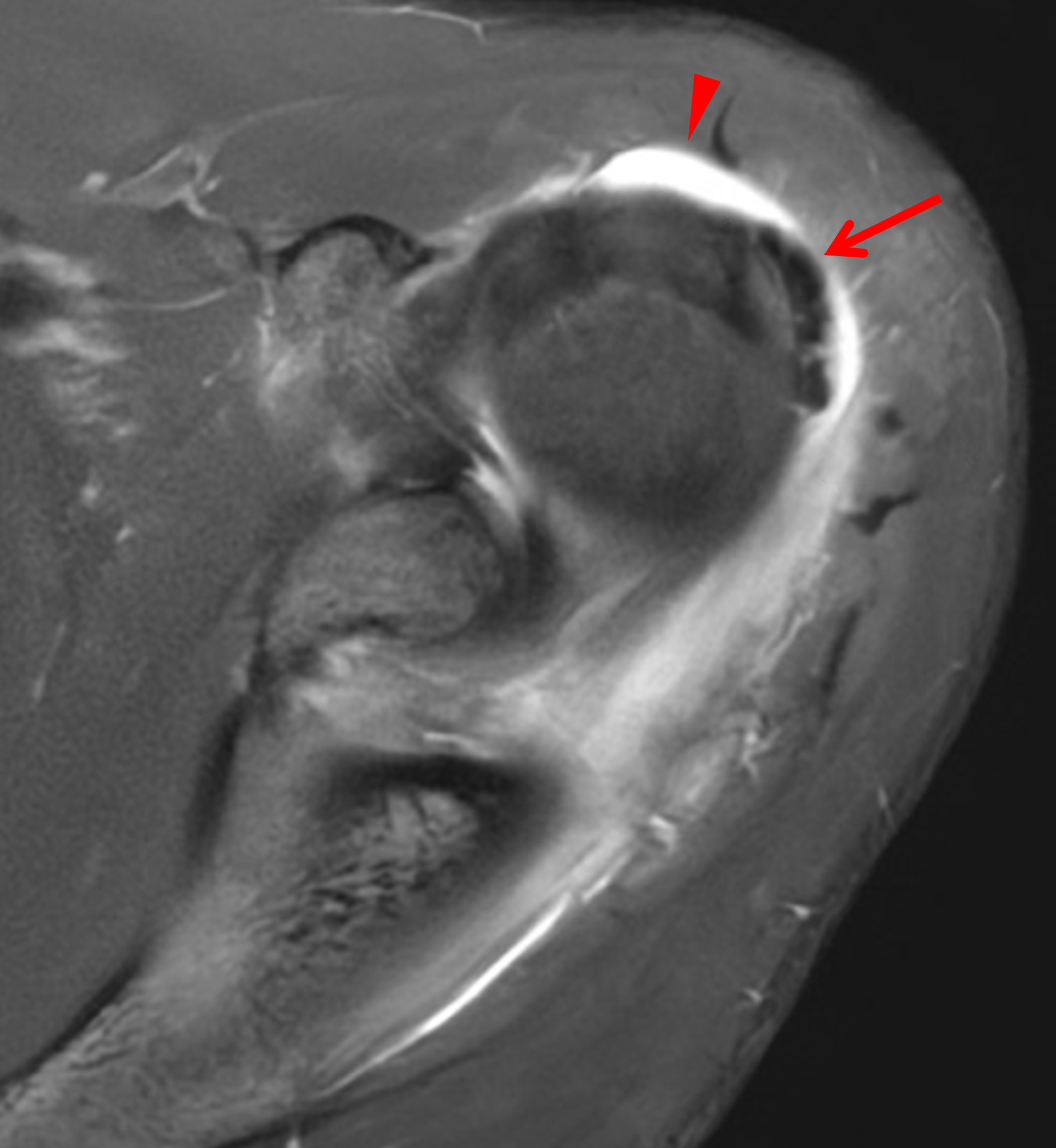
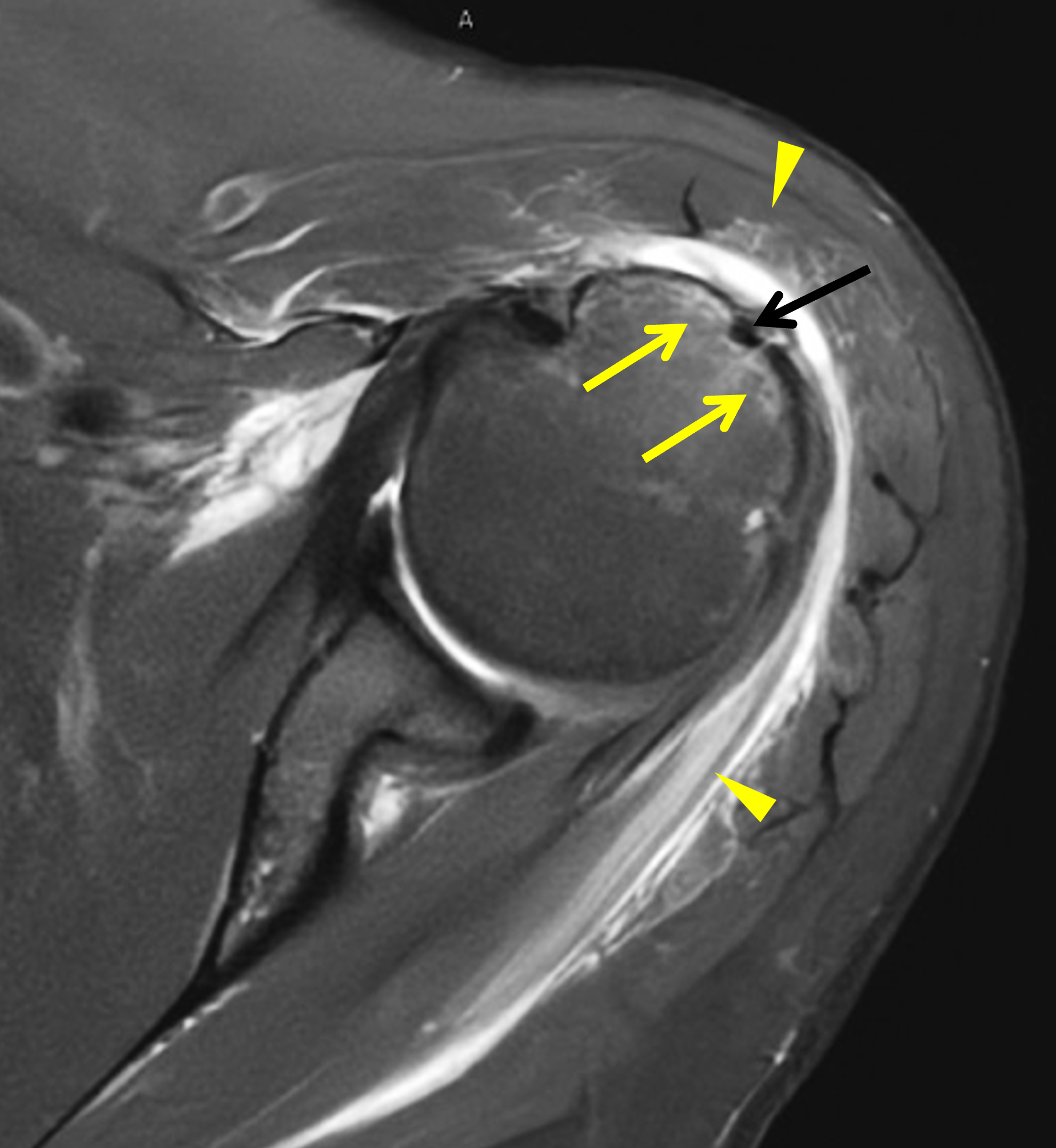
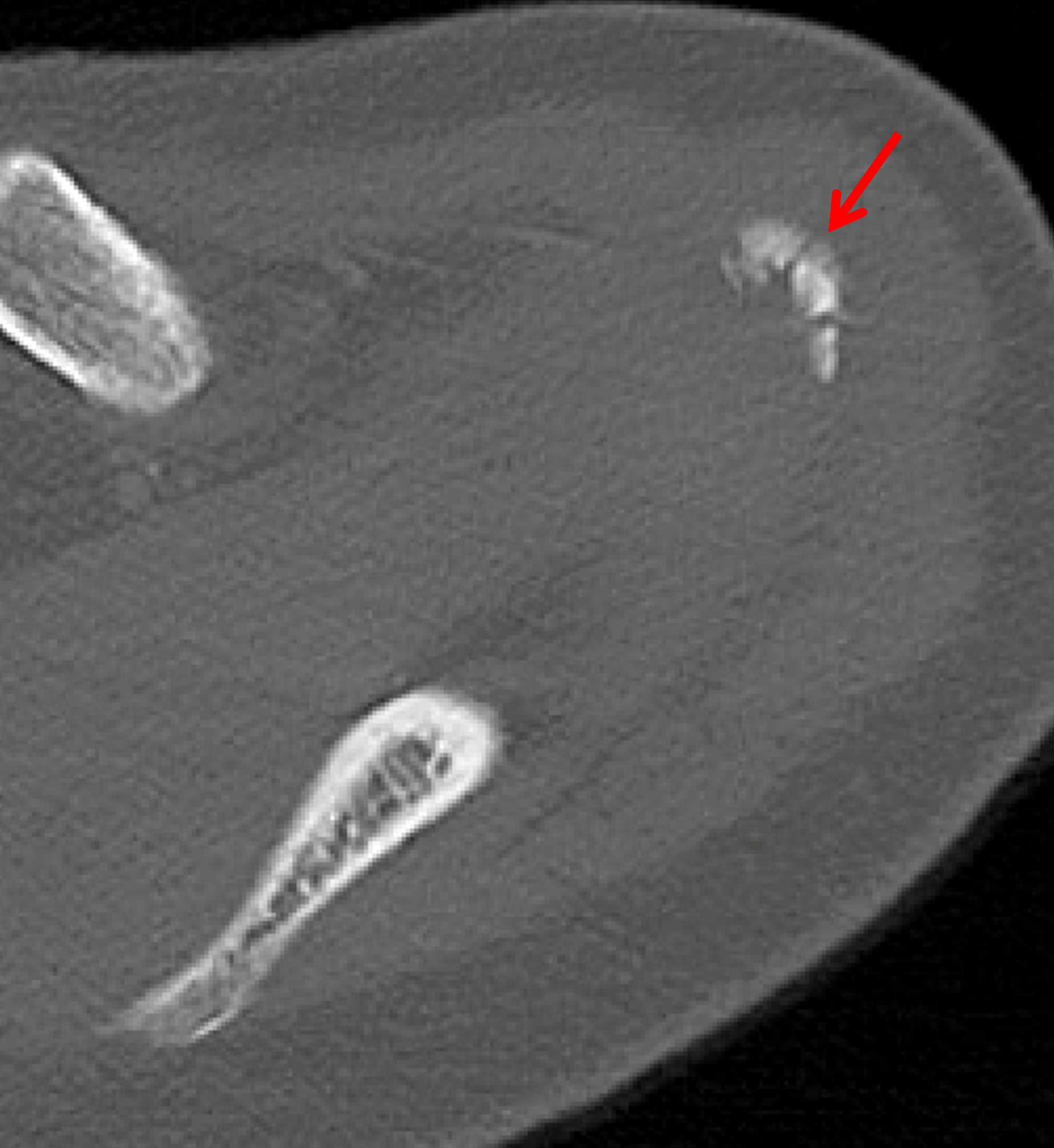
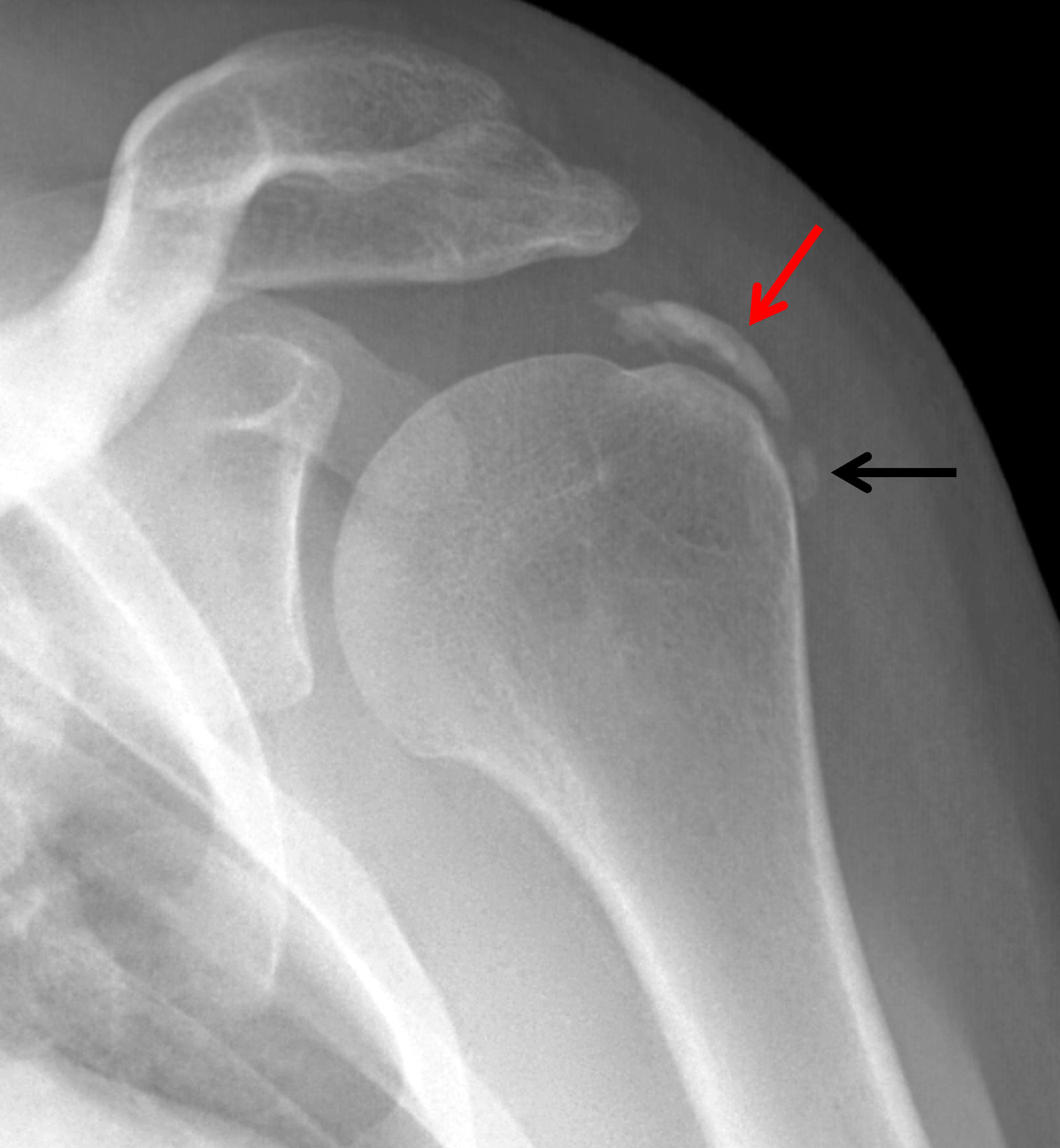
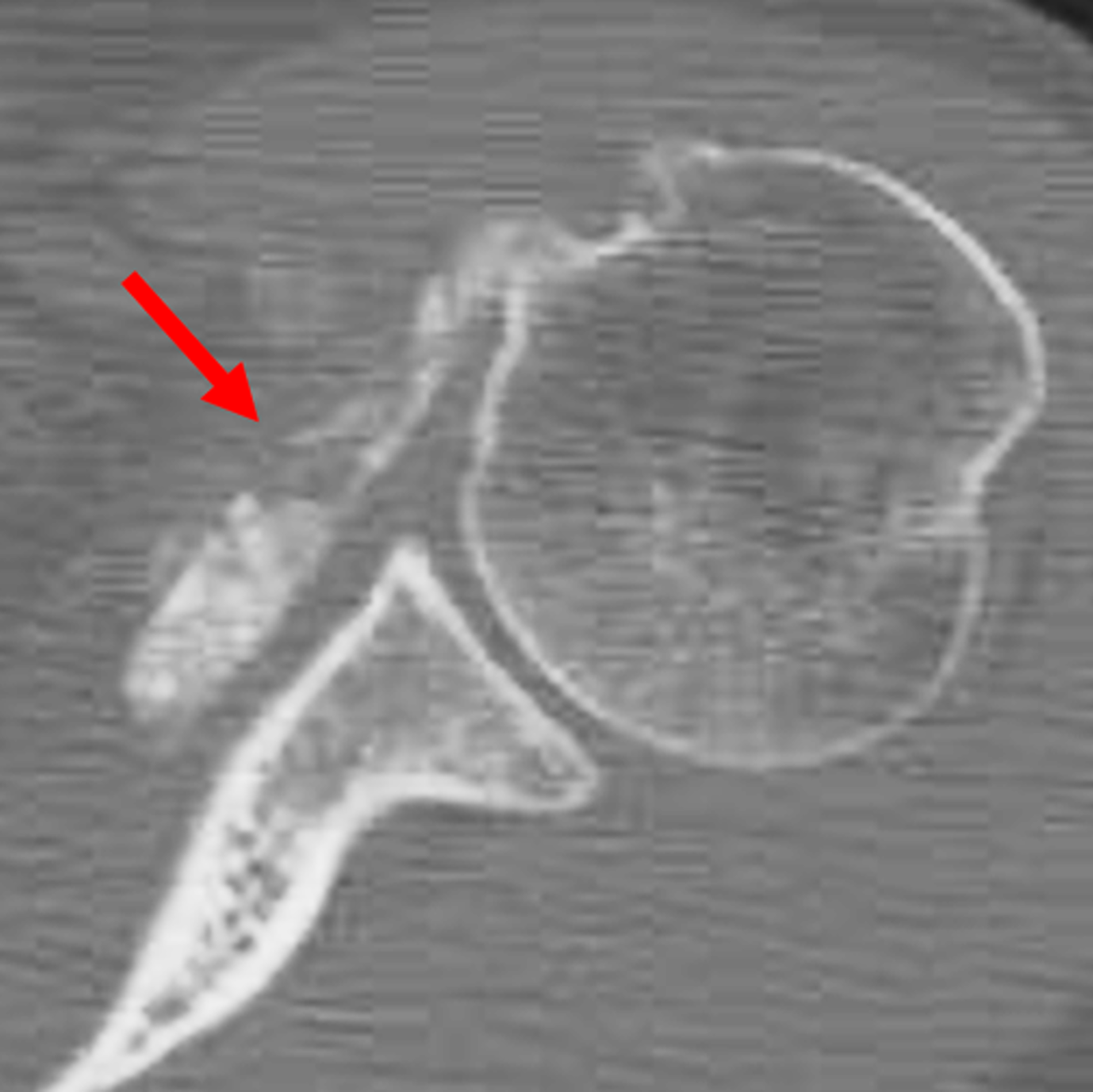
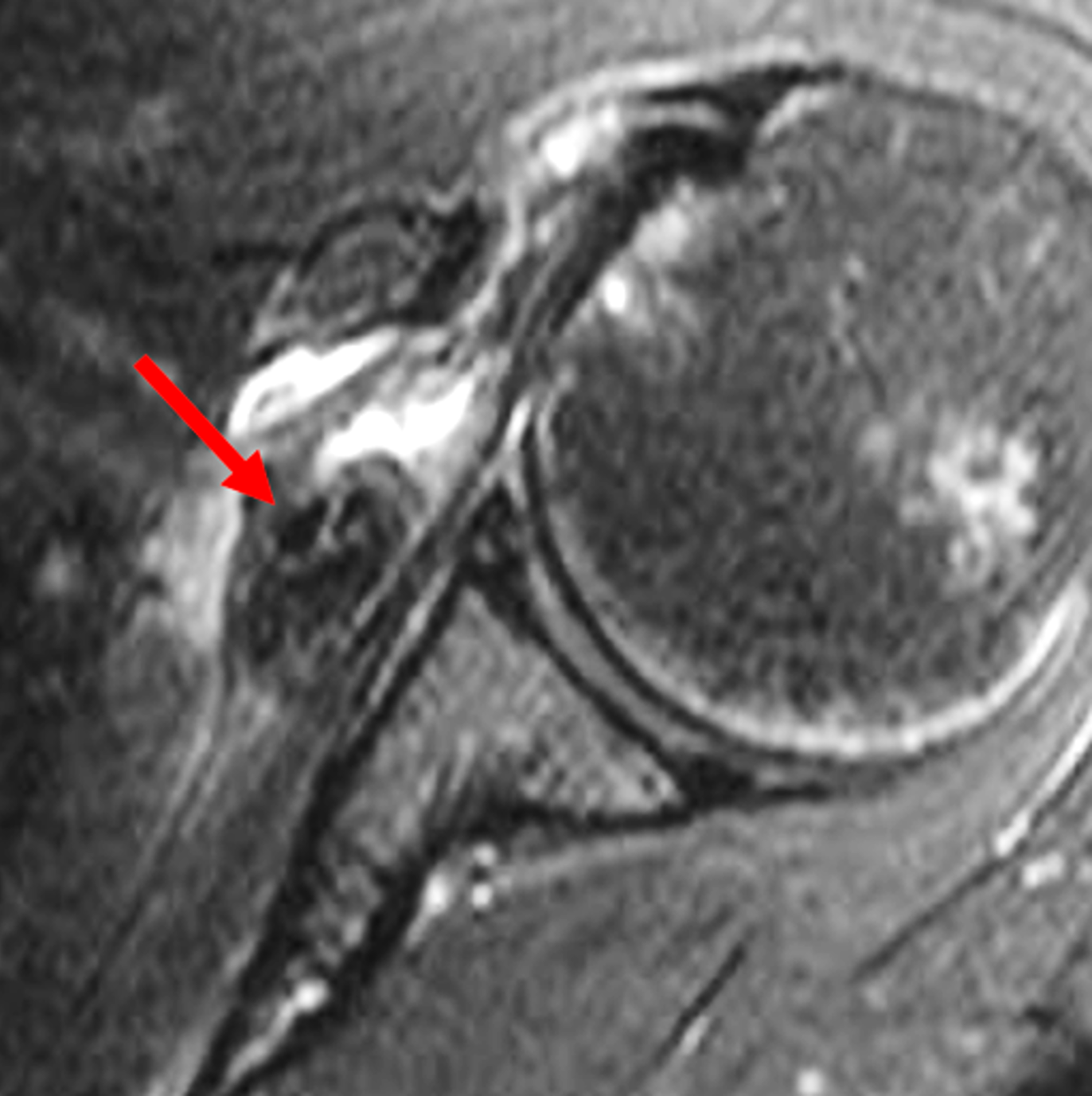
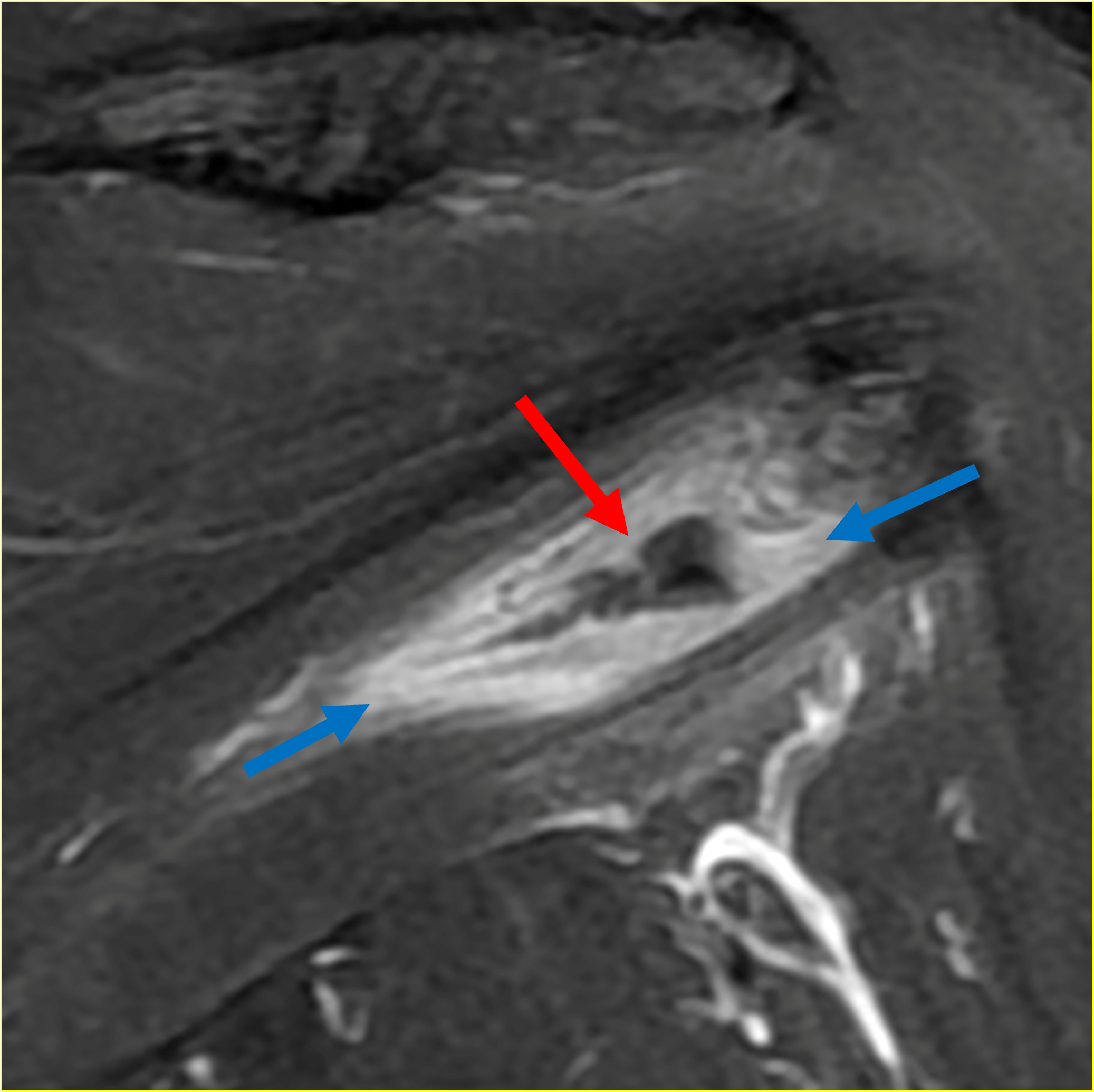
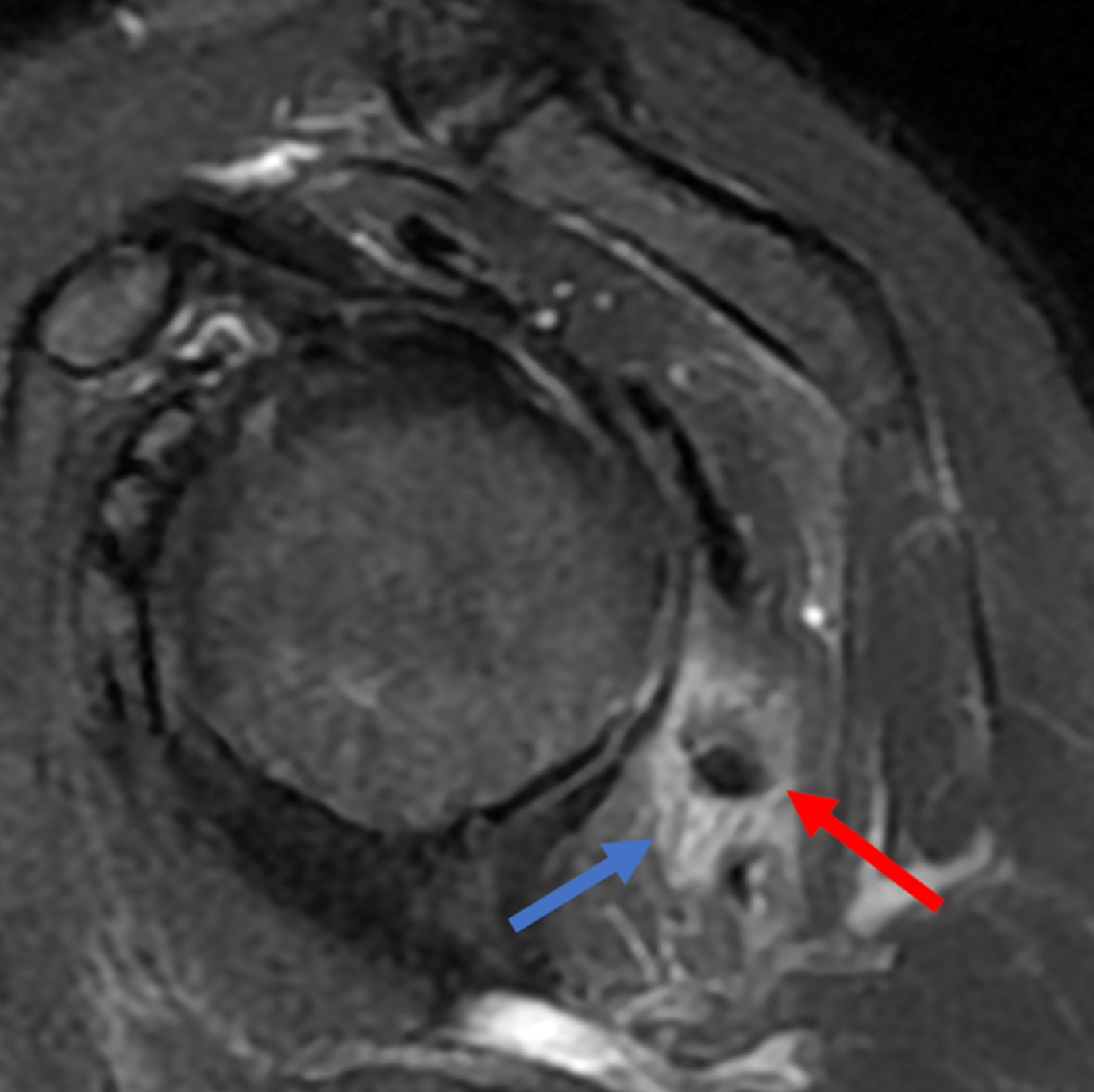
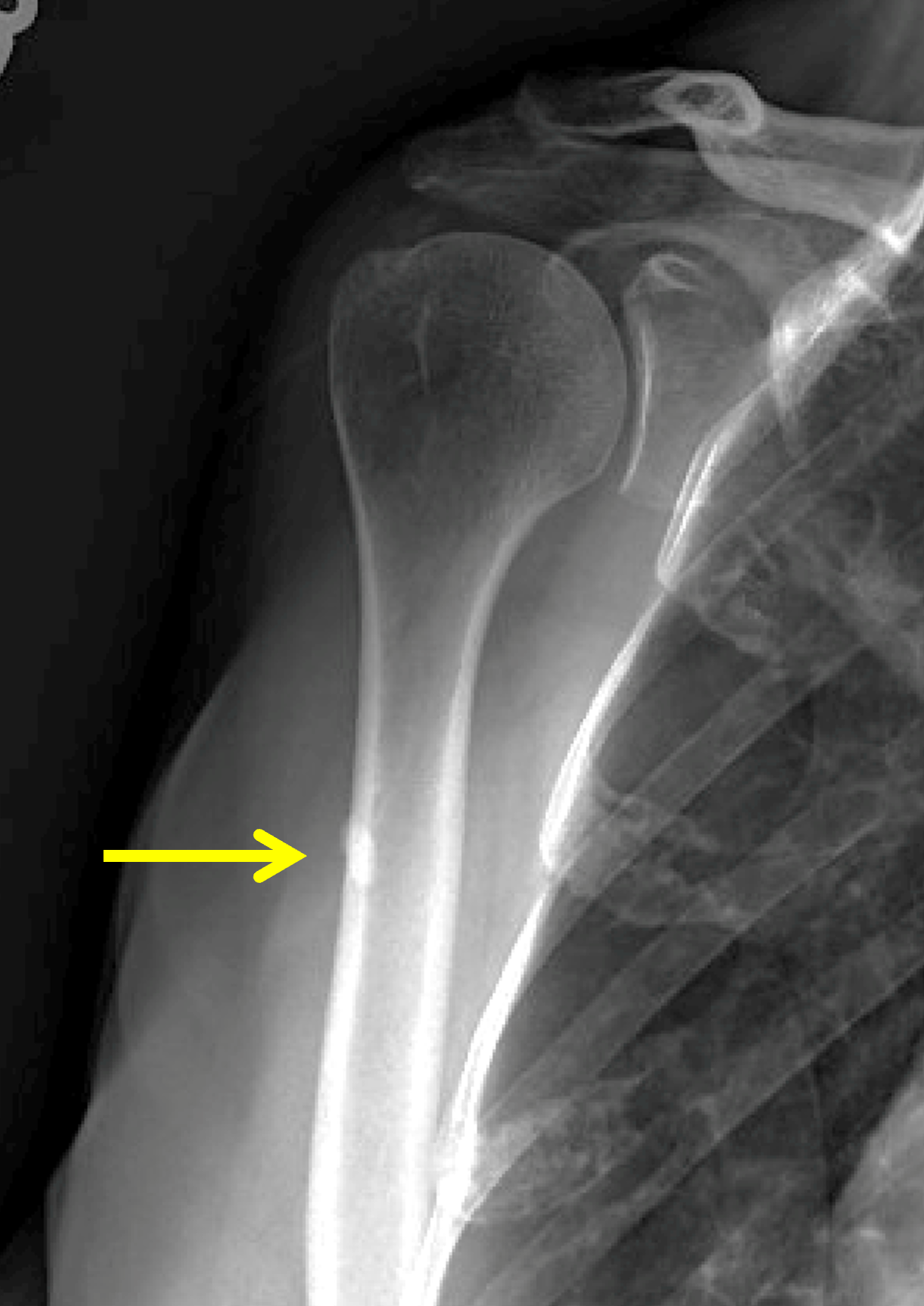
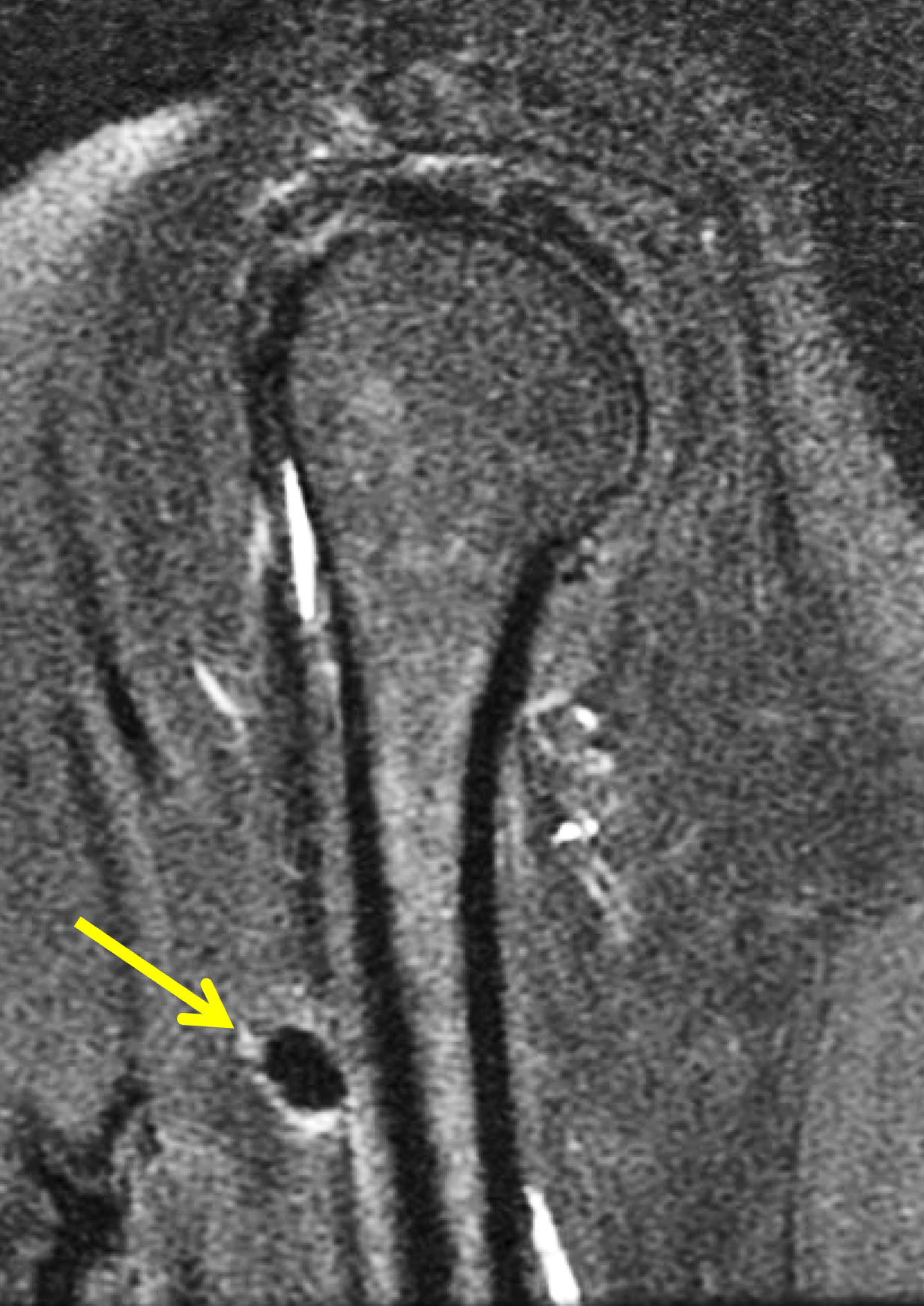
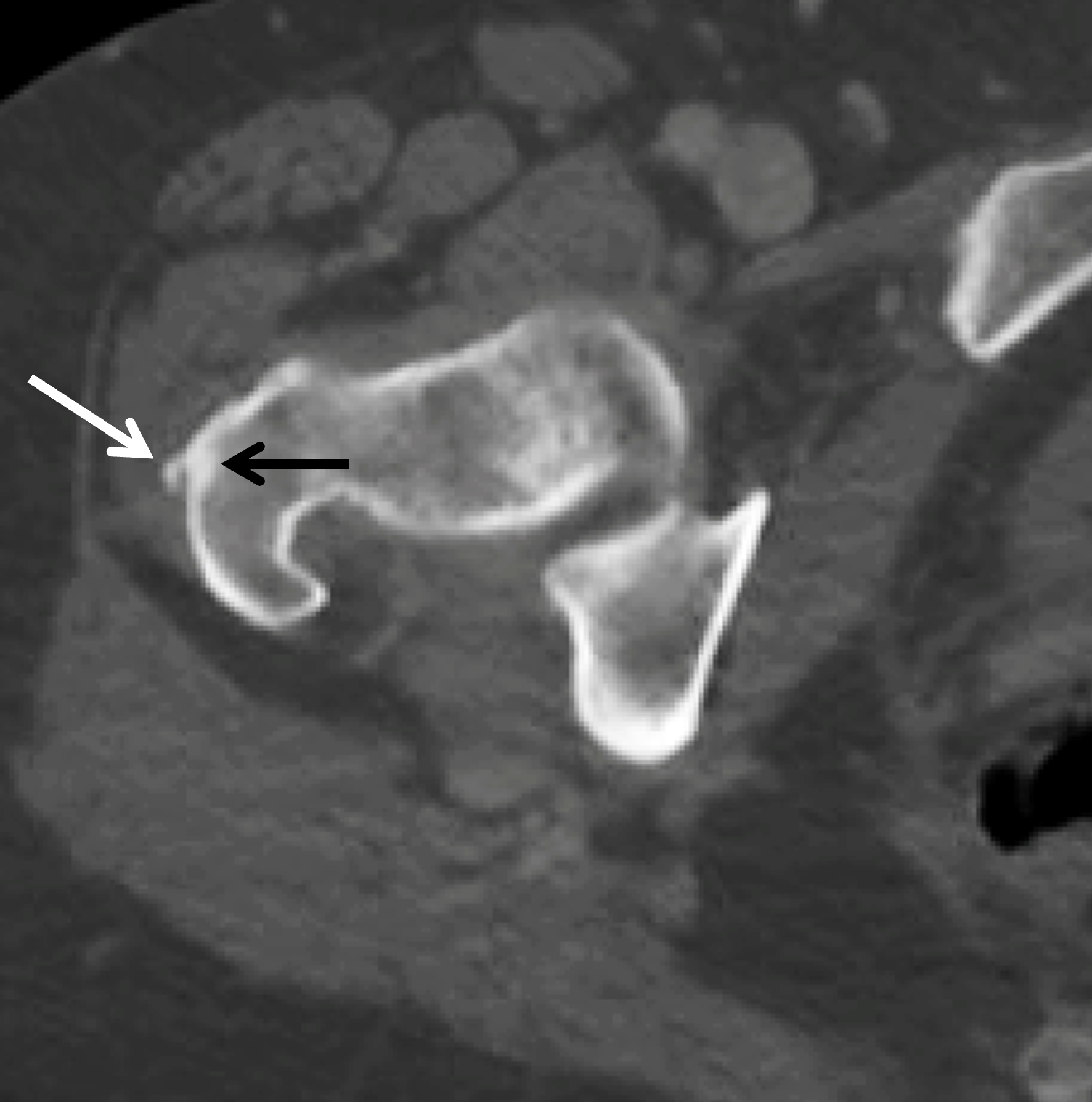
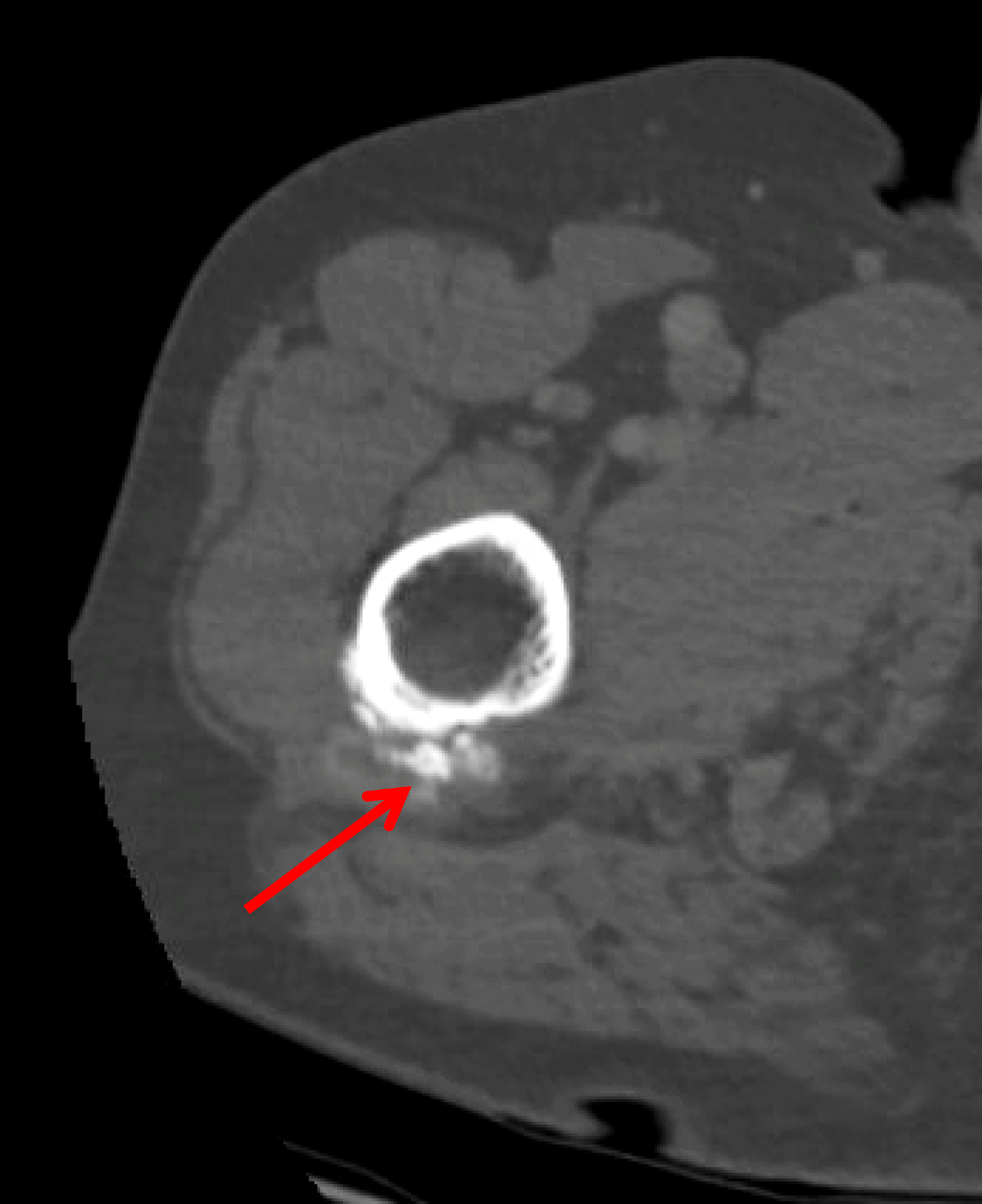
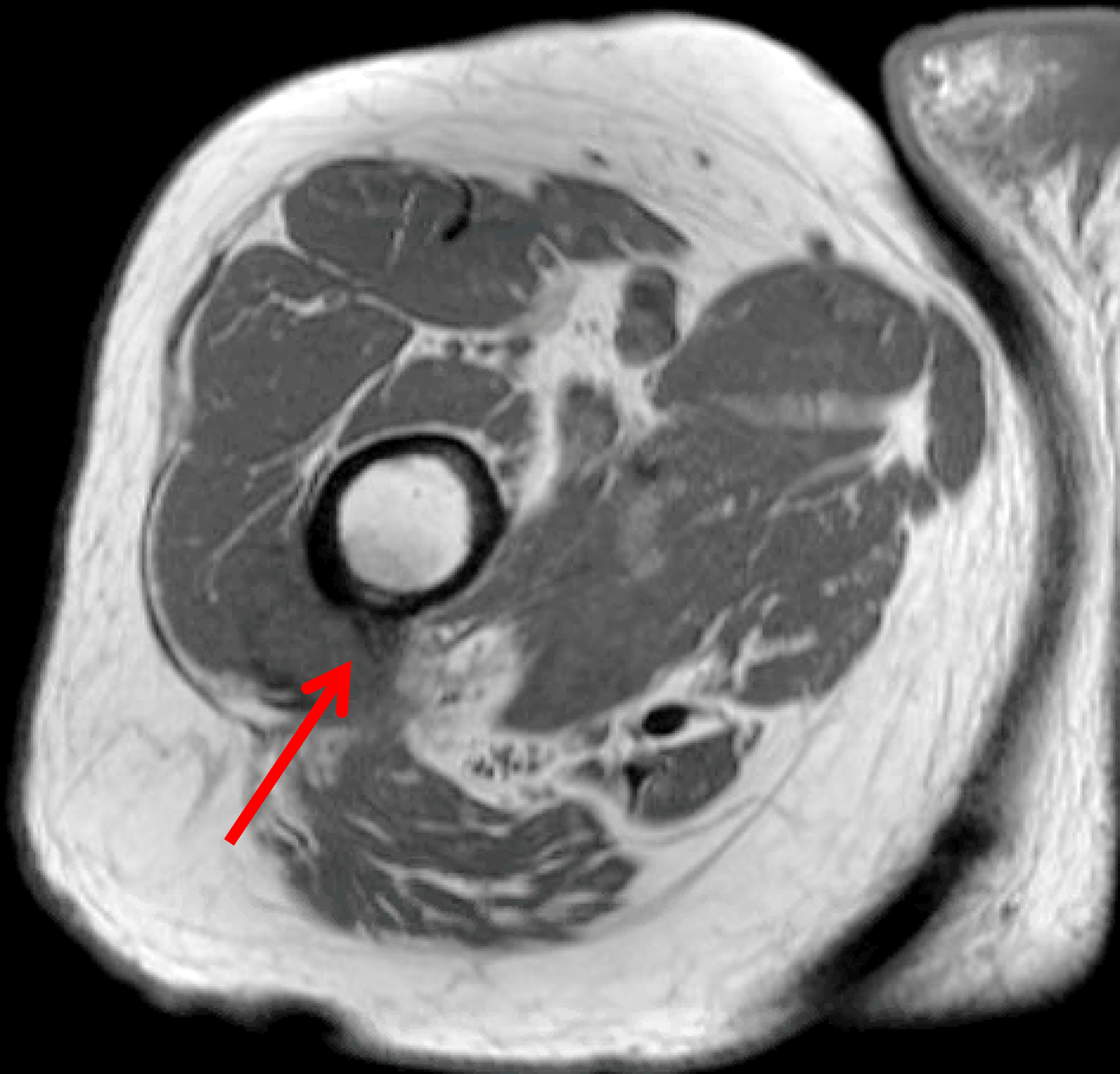
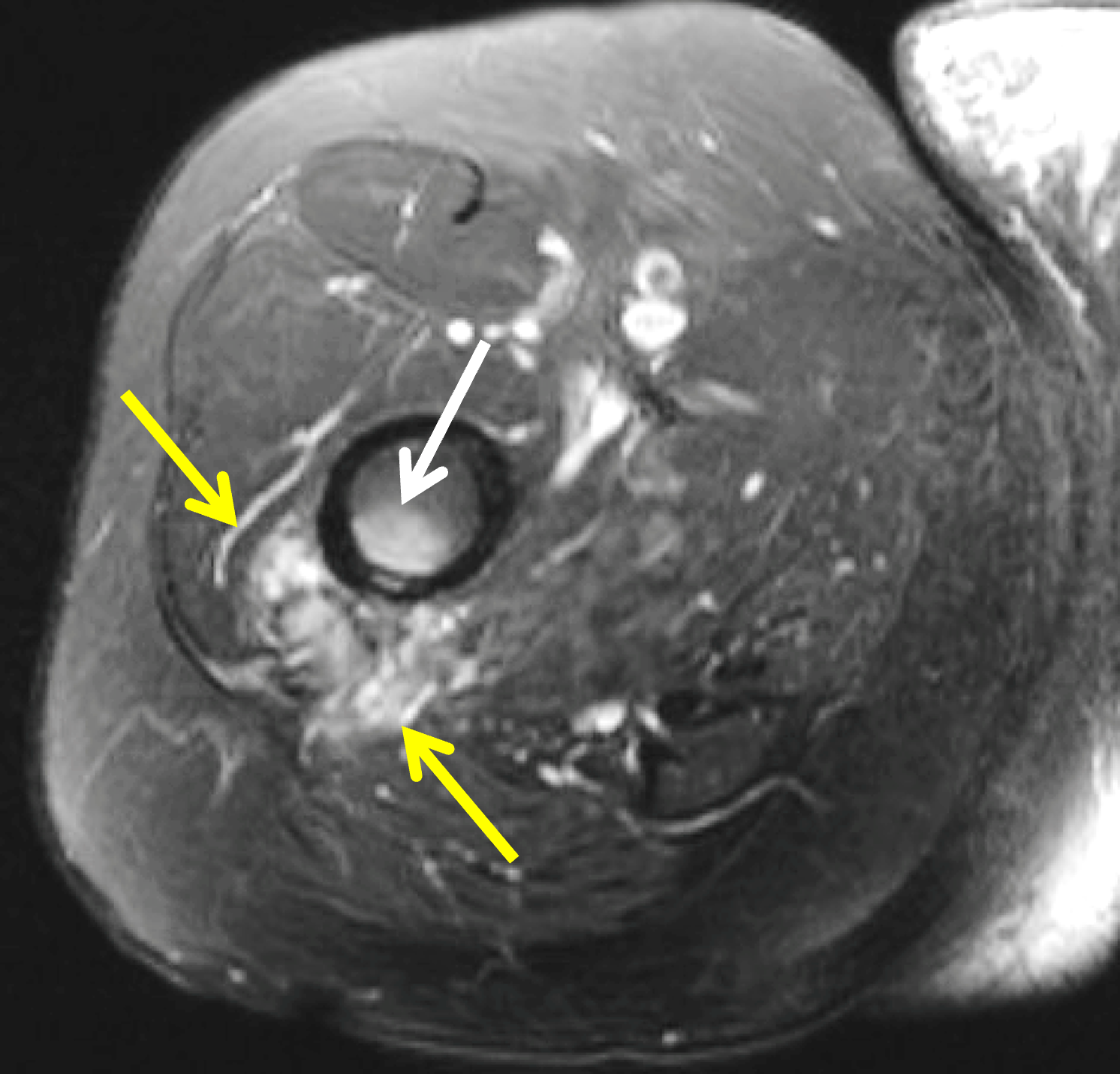
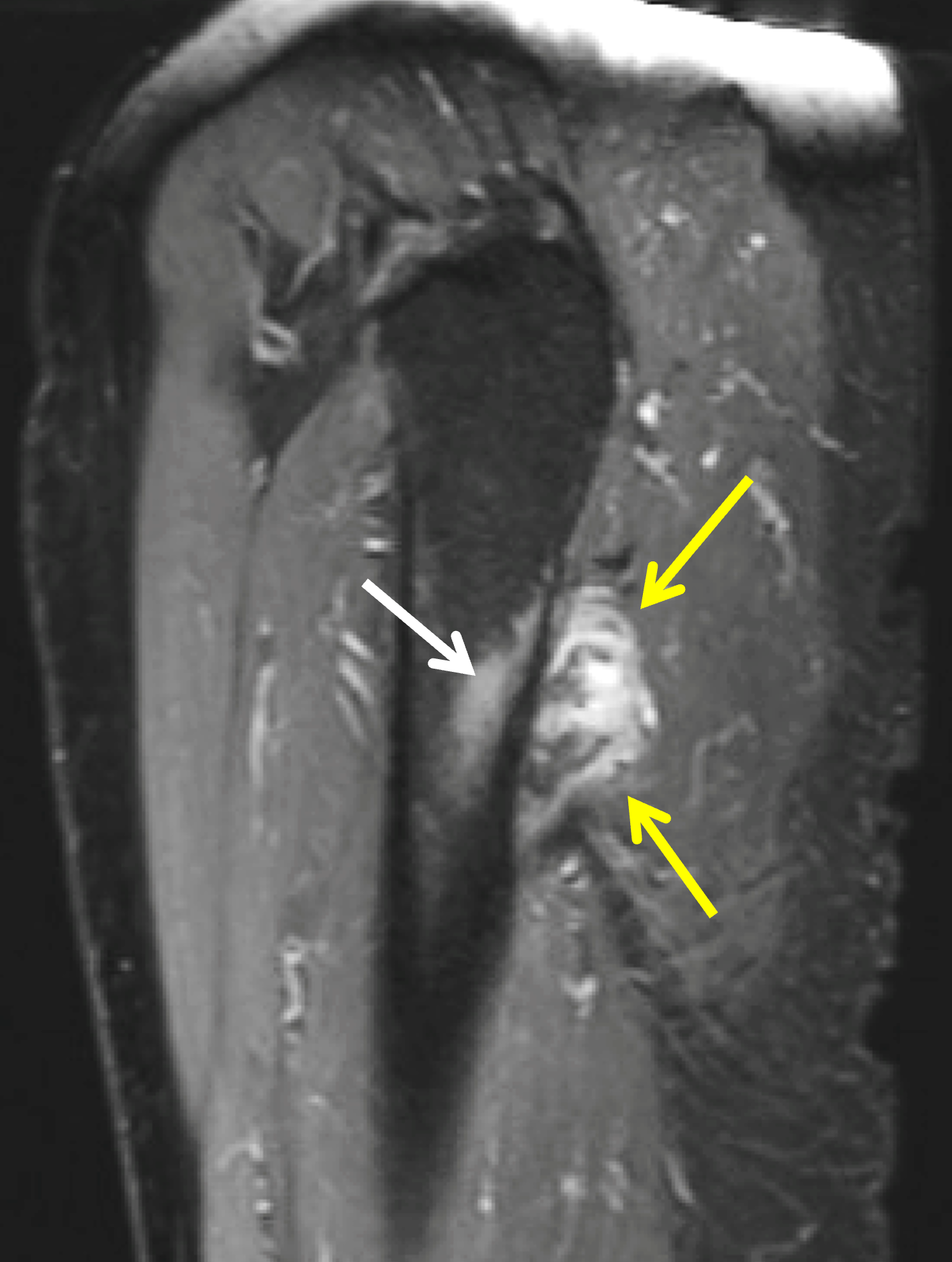
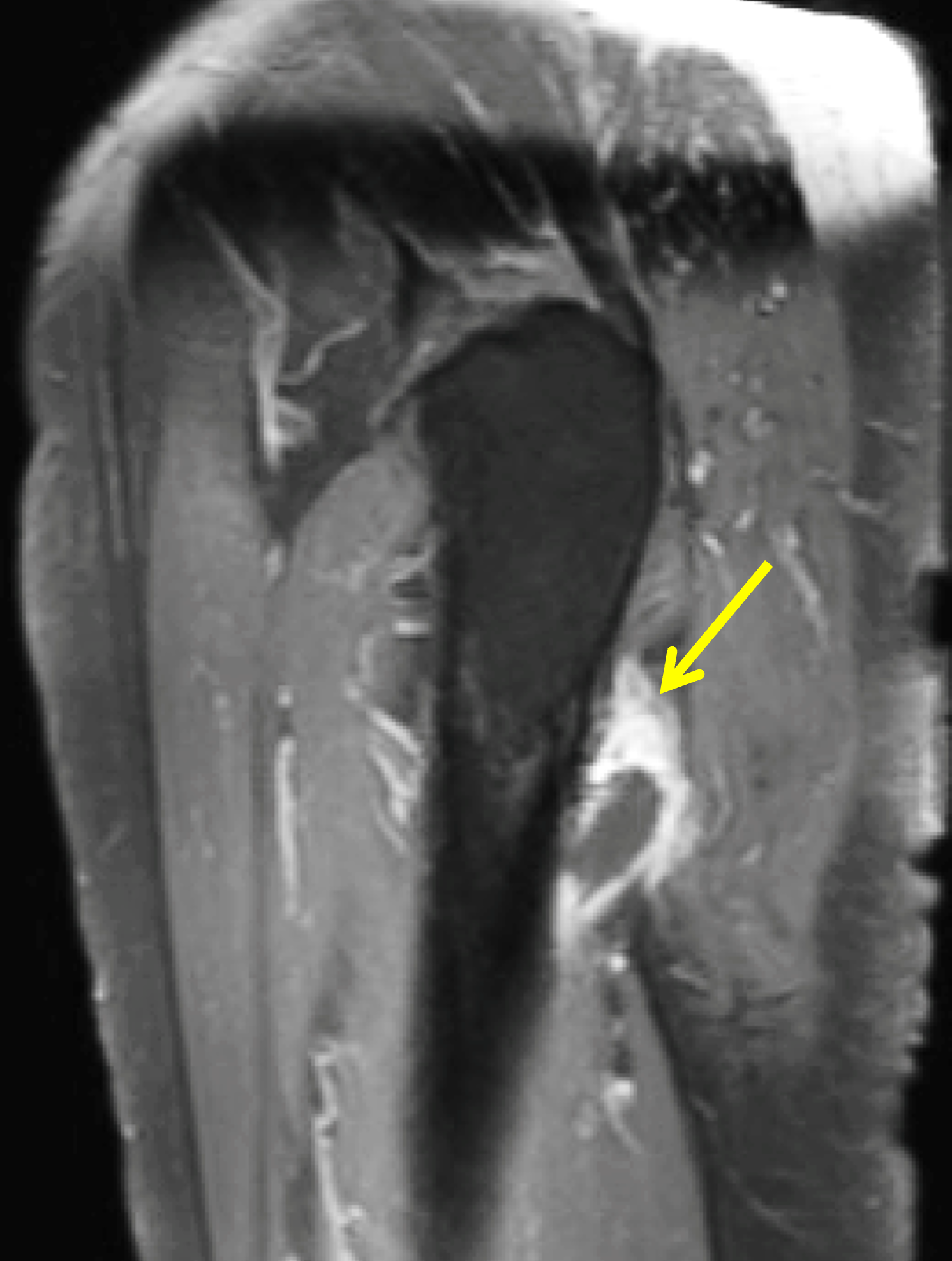
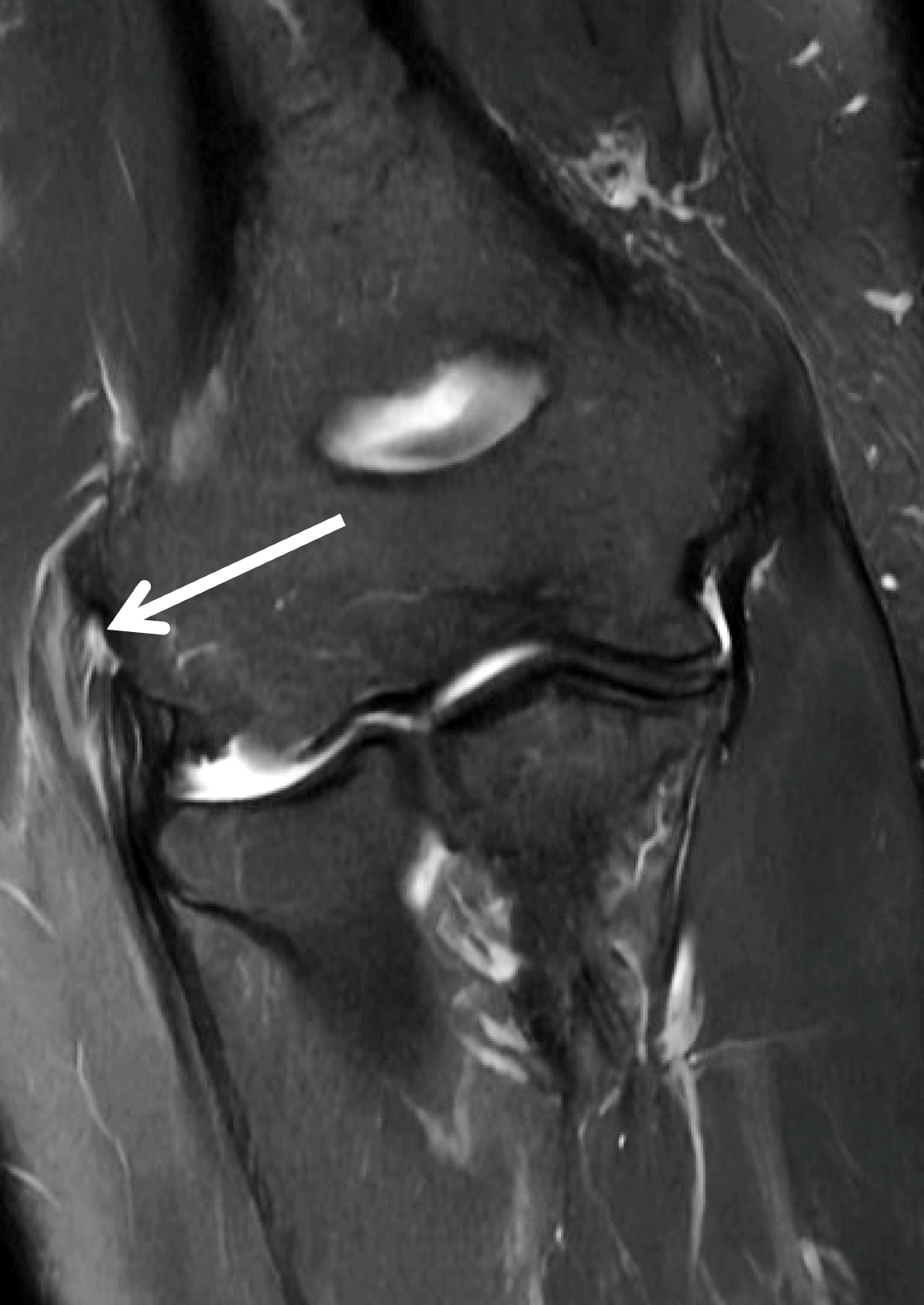
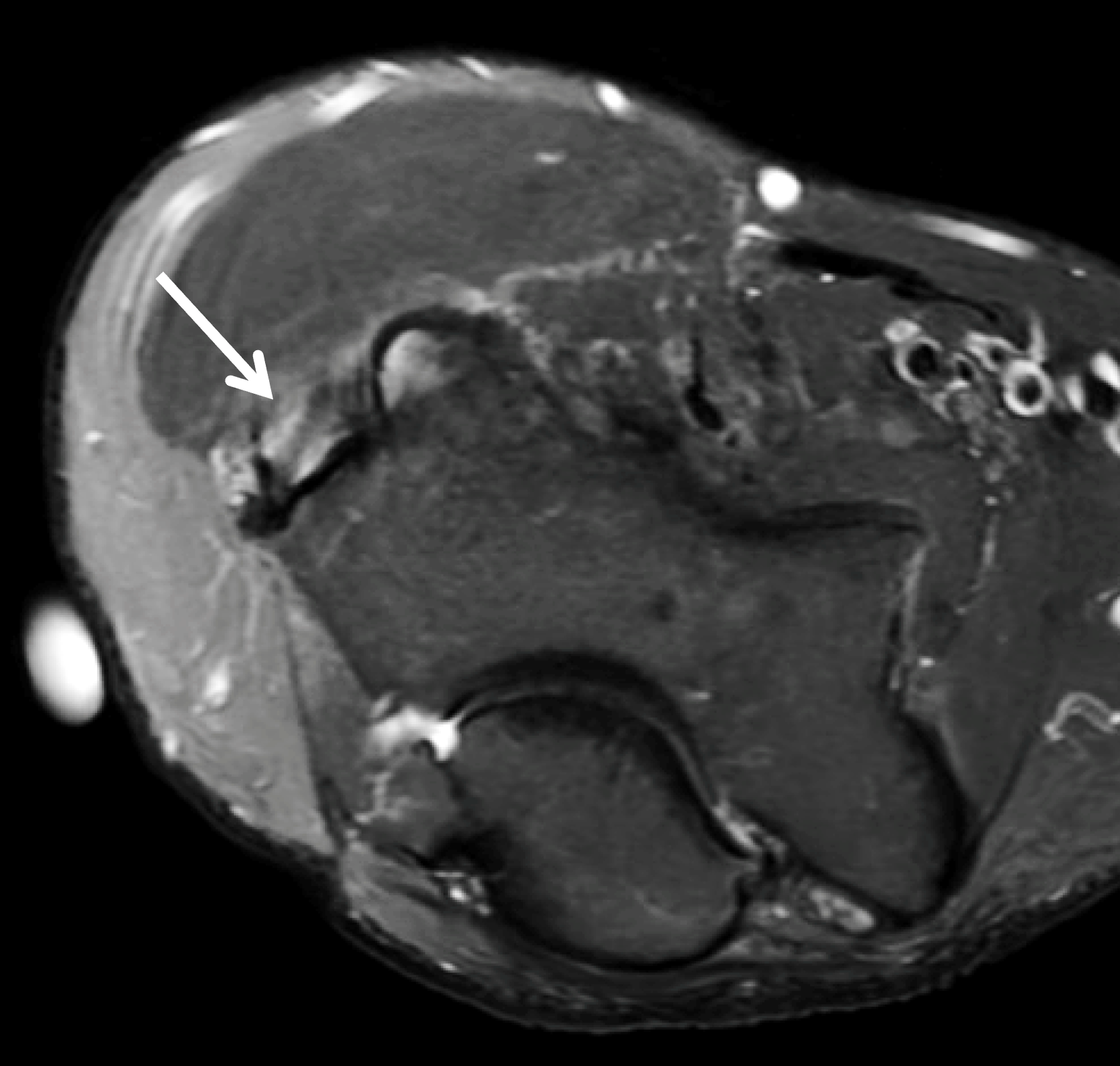
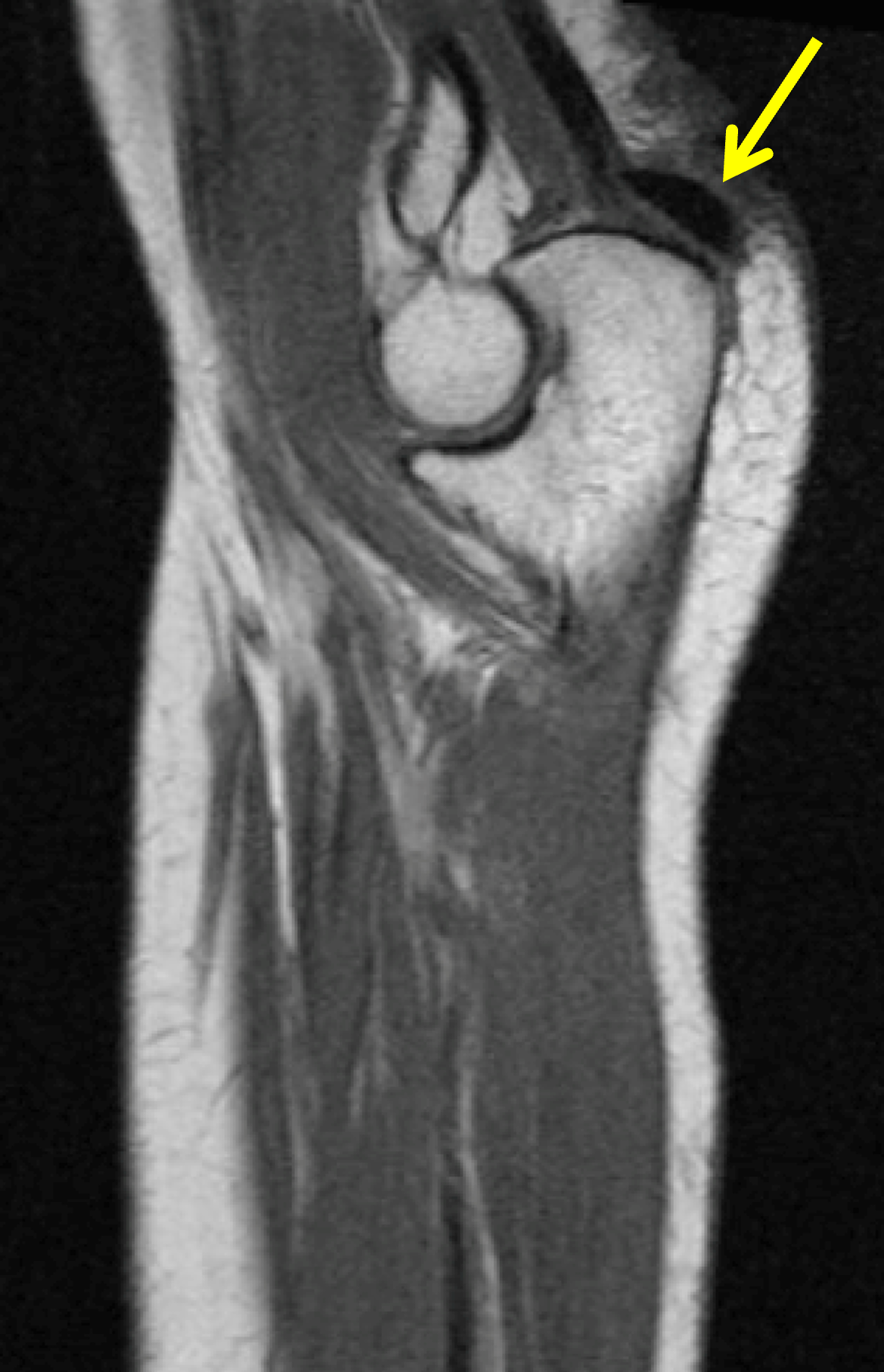
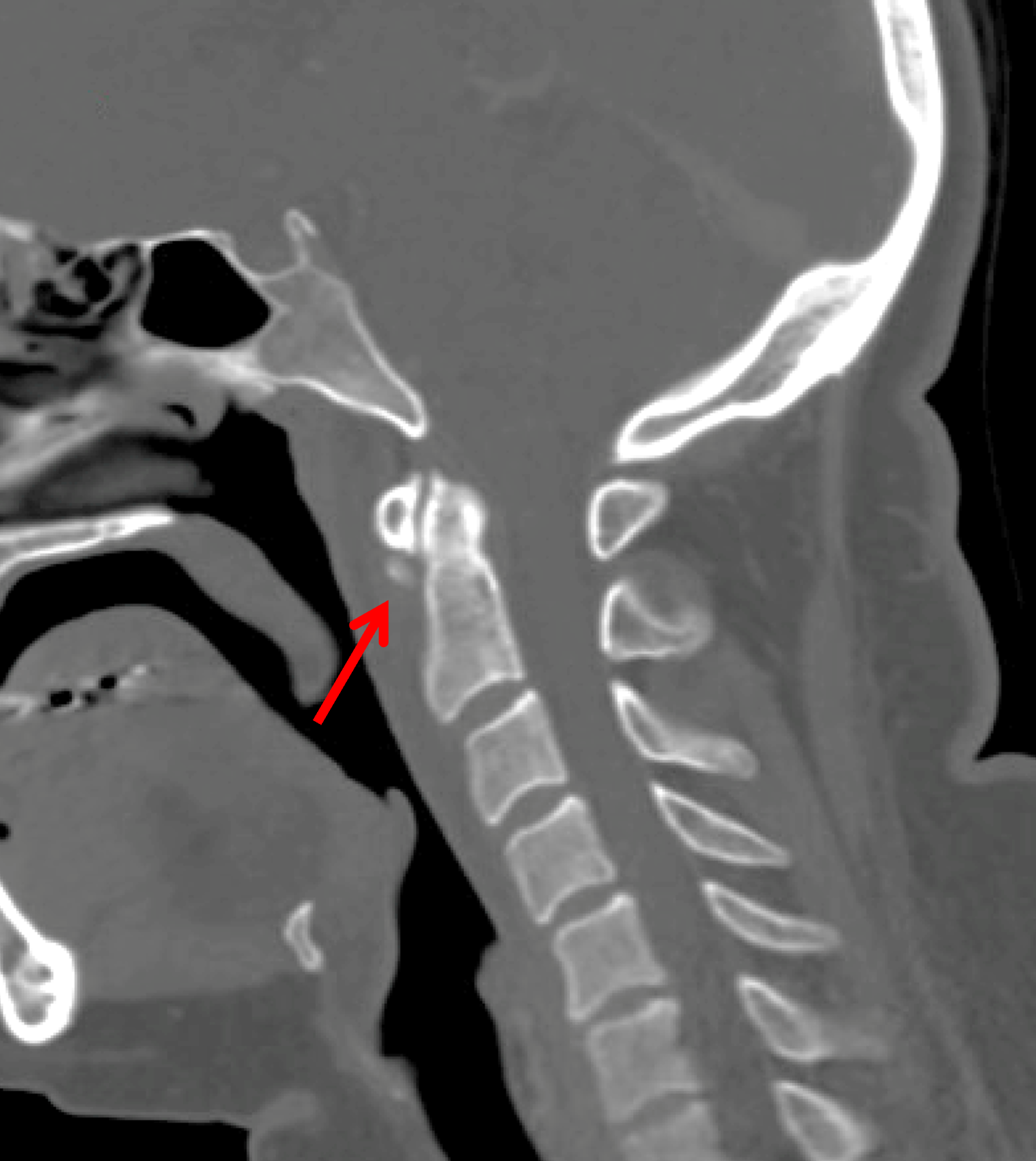
A 40-year-old woman presents with four days of atraumatic left shoulder pain. Physical examination reveals limited ability to abduct the arm. Vital signs and laboratory studies were within normal limits. (1A and 1B) Axial fat-suppressed, proton density (PD)-weighted, (1C) coronal T1-weighted, and (1D) coronal fat-suppressed T2-weighted MR images are provided, in addition to (1E) axial and (1F) reconstructed coronal CT images, and an apical oblique radiograph (1G). What are the findings? What is your diagnosis?
Findings
Figure 2: (2A) Axial fat-suppressed, PD-weighted image through the superior humeral head shows a 20 mm low signal intensity deposit (arrow) within the subacromial-subdeltoid bursa that overlies the junctional fibers of the supraspinatus and infraspinatus tendons, with a moderate amount of fluid in the subacromial-subdeltoid bursa (arrowhead). (2B) Axial fat-suppressed, PD-weighted image more inferiorly shows a 3 mm low signal intensity focus with intraosseous extension into the greater tuberosity (black arrow) with associated mild bone marrow edema (yellow arrows). Intramuscular edema is present within the deltoid and rotator cuff musculature (arrowheads). (2C) Coronal T1-weighted image shows the larger deposit within the subacromial-subdeltoid bursa overlying the supraspinatus-infraspinatus tendon junction (arrow). (2D) Coronal fat-suppressed, T2-weighted image redemonstrates the 20 mm low signal intensity deposit (red arrow), as well as the fluid in the subacromial-subdeltoid bursa (red arrowheads) and intramuscular edema (yellow arrowhead). Note the 3 mm low signal intensity focus with intraosseous extension into the greater tuberosity (black arrow) and associated mild bone marrow edema (yellow arrows). (2E) Axial CT image shows a 20 mm calcific deposit (arrow) corresponding to the MRI findings. (2F) Coronal reconstruction of the CT shows the calcification in the bursa (red arrow) and the second calcification extending into the greater tuberosity (black arrows). (2G) Apical oblique radiograph shows the larger (red arrow) and smaller (black arrow) calcific deposits; however, the intraosseous extension into the greater tuberosity is difficult to appreciate on the radiograph.
Diagnosis
Basic calcium phosphate crystal deposition disease, with calcific bursitis as well as intraosseous extension involving the greater tuberosity.
Introduction
The three major categories of crystal-induced arthropathies are basic calcium phosphate (BCP) crystal deposition disease, calcium pyrophosphate dihydrate (CPPD) crystal deposition disease, and monosodium urate (MSU) crystal deposition disease (gout). This Web Clinic will primarily focus on BCP crystal deposition disease and its imaging manifestations – emphasizing the MRI findings – with additional discussion of the differential diagnosis at various sites.
Basic Calcium Phosphate Crystal Composition
BCP crystal deposition disease results from deposition of carbonate-substituted hydroxyapatite, octacalcium phosphate, tricalcium phosphate, and magnesium whitlockite crystals,1,2 often about and sometimes within articulations, and associated with a variety of soft tissues including tendons, ligaments, joint capsules, and even the menisci of the knee. BCP crystals are predominantly composed of calcium hydroxyapatite,3 which is why the accompanying disease entity is commonly referred to as calcium hydroxyapatite crystal deposition disease (HADD). Octacalcium phosphate and tricalcium phosphate, which are present in smaller quantities, are known to be precursors to calcium hydroxyapatite formation at physiological pH levels.4,5 It has been suggested that magnesium whitlockite may be less inflammatory than the other BCP crystals.6
Crystal Deposition as a Precursor to Osteoarthrosis
Crystal deposition (BCP crystals, CPPD crystals, or both) in hyaline cartilage and fibrocartilage is a precursor to osteoarthrosis and results in degradation of cartilage through multiple intracellular processes.1 The concentration of BCP crystals in synovial fluid of osteoarthritic joints ranges from 20 µg/mL to 100 µg/mL.3 To our knowledge, there is not a reported normal range of BCP crystals in synovial fluid. In studies of 120 patients with primary knee osteoarthrosis and 80 patients with primary hip osteoarthrosis who were undergoing total arthroplasties, synovial fluid analysis revealed that 100% of synovial fluid samples from these patients contained BCP crystals and 20% contained CPPD crystals.7,8 Another three-year prospective analysis of the synovial fluid of 330 patients with knee osteoarthrosis showed that crystal deposition was associated with a more severe radiographic appearance of osteoarthrosis.9
The severity of structural joint changes and the speed of disease progression in osteoarthritic joints with crystal deposition are markedly pronounced when compared to those without crystal deposition.1 Crystal-induced stress affects chondrocytes in a similar fashion to oxidative, mechanical or biochemical stresses, and results in up to four different chondrocyte phenotypes (mitogenic, pro-inflammatory, catabolic, and apoptotic chondrocytes). In addition, these crystals create a stiffness imbalance within the cartilage that alters its biomechanical properties and further contributes to progression of degenerative changes.1
The inhibitors and promoters of calcification that are involved in regulating physiologic calcification are also observed in areas of pathologic calcification. Imbalances in these components and changes in the local environment result in pathologic calcification in the tissues of osteoarthritic joints.1
Potential Therapeutic Targets
Additional studies have shown that patients with crystal deposition disease (involving BCP crystals, CPPD crystals, or both) have higher levels of inorganic phosphate (PPi) in their joint fluid, along with increased activity of enzymes involved in phosphate metabolism,10 suggesting that PPi and these enzymes may contribute to the formation of intra-articular crystals. Other research has shown that when human immune cells, such as macrophages and dendritic cells, are exposed to BCP crystals, certain intracellular signaling enzymes become activated, resulting in the production of interleukin-1, a key inflammatory molecule. BCP crystals also increase the production of other damage-related inflammatory signals through these same enzyme pathways, suggesting that these signaling enzymes may be potential targets for future therapies.3
Synovial Fluid Analysis
Synovial fluid analysis is critical in diagnosing and distinguishing among the crystal deposition diseases. Unlike MSU and CPPD crystals, which can be readily identified with polarized light microscopy owing to their characteristic strong negative and weak positive birefringence, respectively, BCP crystals are not well depicted with this technique due to their sub-micron size.4
The Alizarin red S stain can assist in microscopic analysis and identification of BCP crystals (predominately calcium hydroxyapatite) as well as CPPD crystals, with sensitivities for the detection of these crystal groups depending on the concentration of Alizarin red S stain used and the pH of the solution.11 Unfortunately, Alizarin red S stain cannot definitively distinguish between CPPD and calcium hydroxyapatite crystals that may coexist within a single sample of synovial fluid. Different microscopy, spectroscopy, and diffraction techniques have shown potential in the diagnosis and differentiation of crystal-induced arthropathies and are beyond the scope of this Web Clinic.4,12,13,14,15,16,17,18,19,20,21,22,23
Major Crystal Deposition Diseases
In addition to BCP crystal deposition disease, the two other major categories of crystal deposition diseases are CPPD crystal deposition disease and MSU crystal deposition disease (gout).
CPPD crystal deposition disease is the most common crystal-induced arthropathy and is commonly encountered in middle-aged and elderly persons.24 The clinical manifestations and imaging findings of CPPD crystal deposition disease are discussed in great detail in the June 2024 Web Clinic.
MSU crystal deposition disease, or gout, is the oldest recognized crystal-induced arthropathy and occurs secondary to hyperuricemia, which can result from overproduction or underexcretion of uric acid.24 The clinical manifestations and imaging findings of gout are discussed in great detail in the July 2011 and July 2025 Web Clinics.
BCP crystal deposition disease can manifest in three major ways: calcific periarthritis, tumoral calcinosis, and a destructive arthropathy that primarily affects large joints, often with a rapid clinical and imaging course. Each of these patterns will now be addressed.
Calcific Periarthritis
The term calcific periarthritis is generally applied to the appearance of calcification related to BCP crystals near an articulation, often involving tendons and most frequently described in the rotator cuff and other tendons about the glenohumeral joint. The accompanying imaging findings within the tendons are in part based on the chronicity of the process in afflicted patients. Research done by Hamada and colleagues demonstrated that carbonate apatite is the most frequent cause of tendinous and soft tissue calcification in cases of calcific periarthritis.25 DePalma and Kruper categorized BCP crystal deposits occurring in calcific periarthritis into two groups, with the radiographic appearance of BCP crystal deposition in acute cases denoted by globular fluffy deposits and in subacute or chronic cases characterized by more discrete crystal deposits.26
Uhthoff and Loehr further divided the disease chronology into three stages: pre-calcific, calcific, and post-calcific.27,28,29 In the initial pre-calcific stage, fibrocartilaginous transformation secondary to diminished perfusion and subsequent hypoxia occurs at the site of future crystal deposition. Patients typically do not experience pain in this stage. BCP crystal deposition during the calcific stage is classified into formative, resting, and resorptive phases. In the formative phase, crystals replace the fibrocartilaginous matrix. This phase may or may not be associated with pain. Subsequently, during the resting phase, fibrocartilaginous tissue surrounds the crystal deposits and signals the end of crystal deposition at that site. Patients may or may not experience pain during this resting phase. During the final resorptive phase of the calcific stage, thin-walled vascular channels surround the crystal deposits, followed by phagocytosis of the crystals by macrophages and multinucleated giant cells. During this resorptive phase, patients often experience pain due to rupture of the crystal deposits into adjacent tissues. In the final post-calcific stage, granulation tissue, scarring, or both will infiltrate the site of prior crystal deposition.
Calcific Periarthritis about the Shoulder
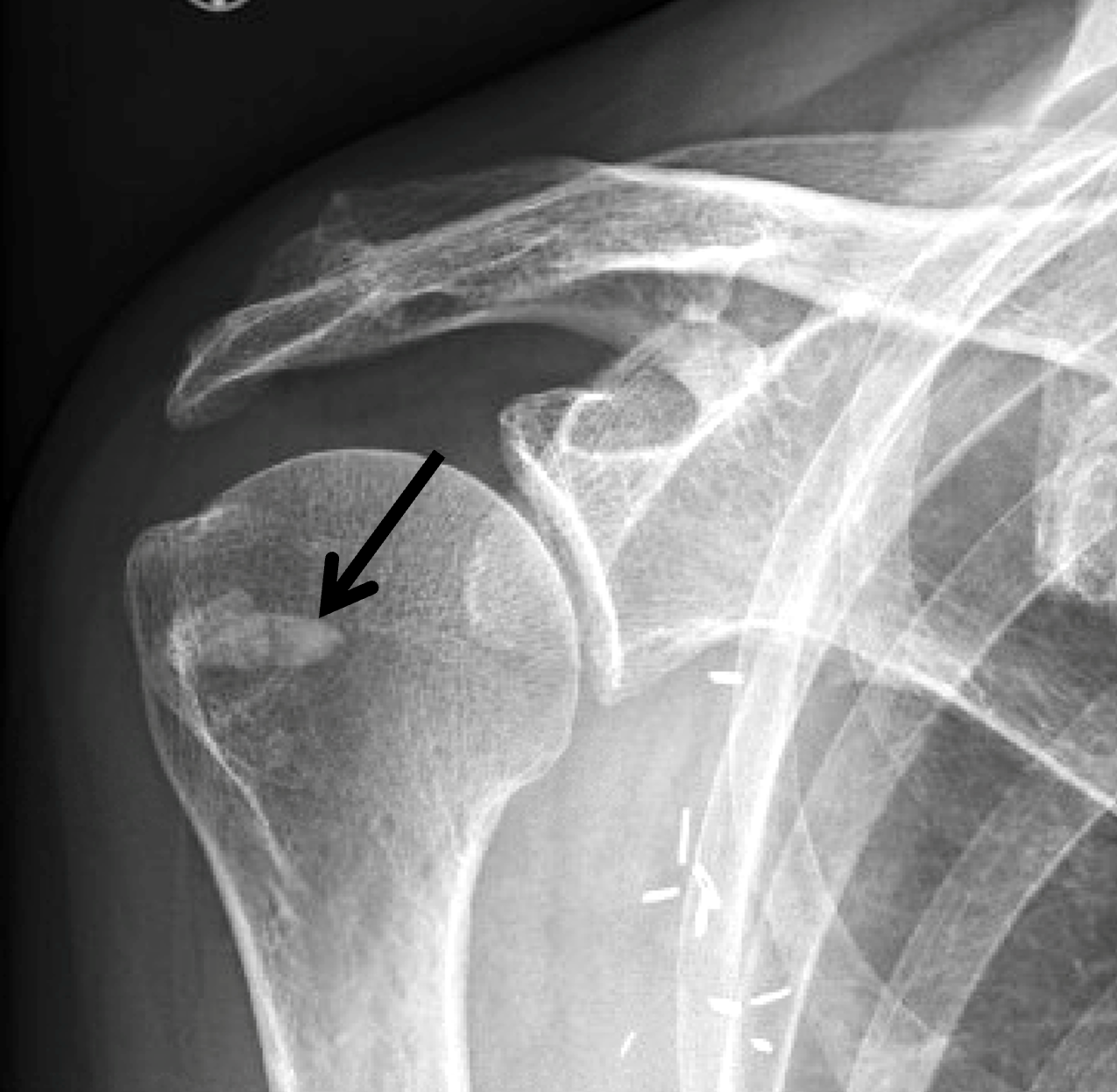
The shoulder is the most commonly affected joint in cases of calcific periarthritis related to BCP crystal deposition (Figure 3).24 In the shoulder, BCP crystals are typically deposited at or near the footprint of the involved rotator cuff tendon (Figures 1 and 2), most commonly the supraspinatus tendon. The one exception to this rule relates to the tendon of the long head of the biceps brachii muscle as calcification may appear at the proximal myotendinous junction along the humeral shaft.30
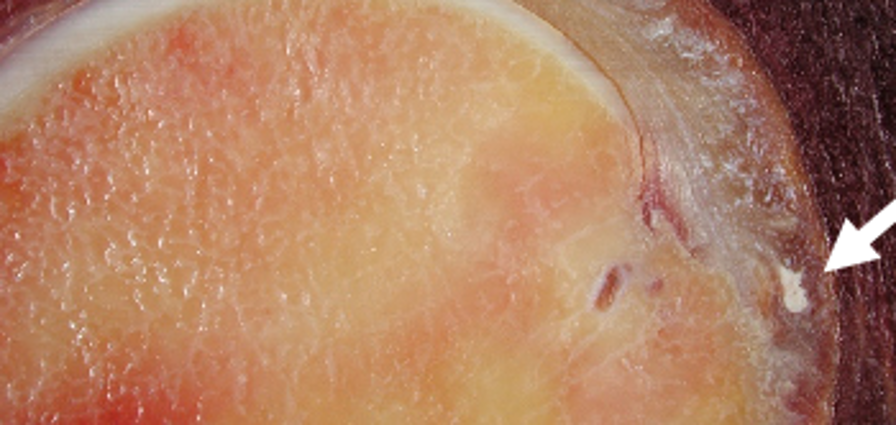
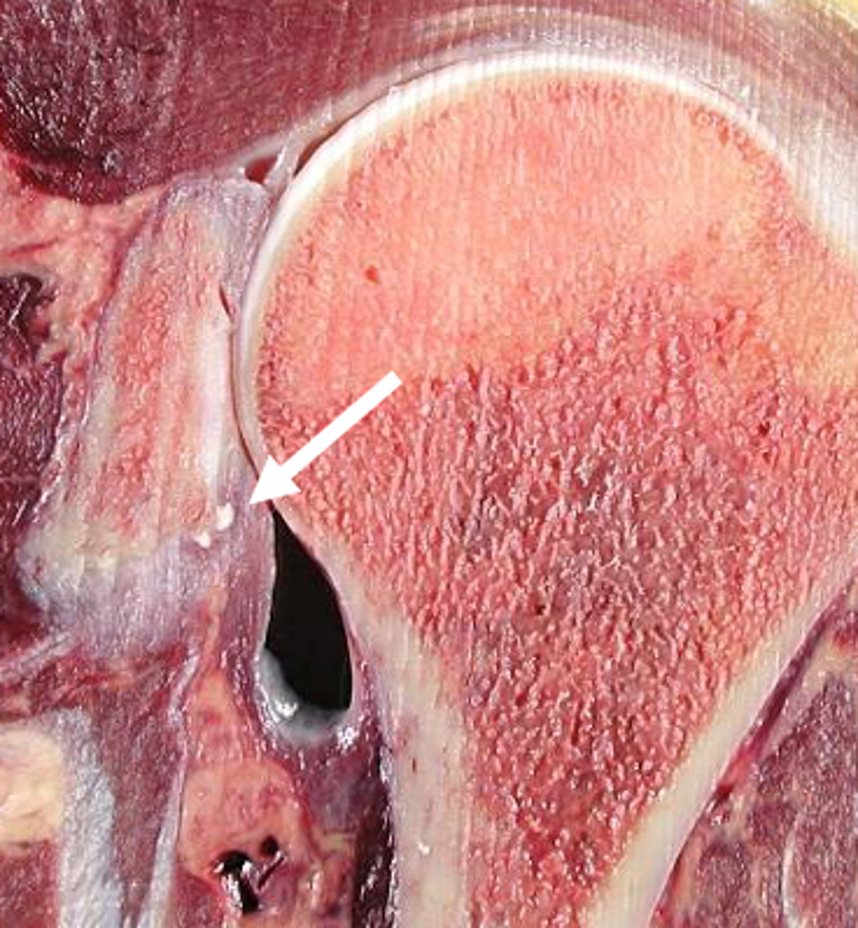
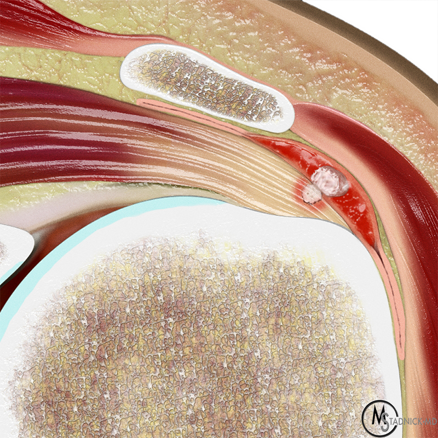
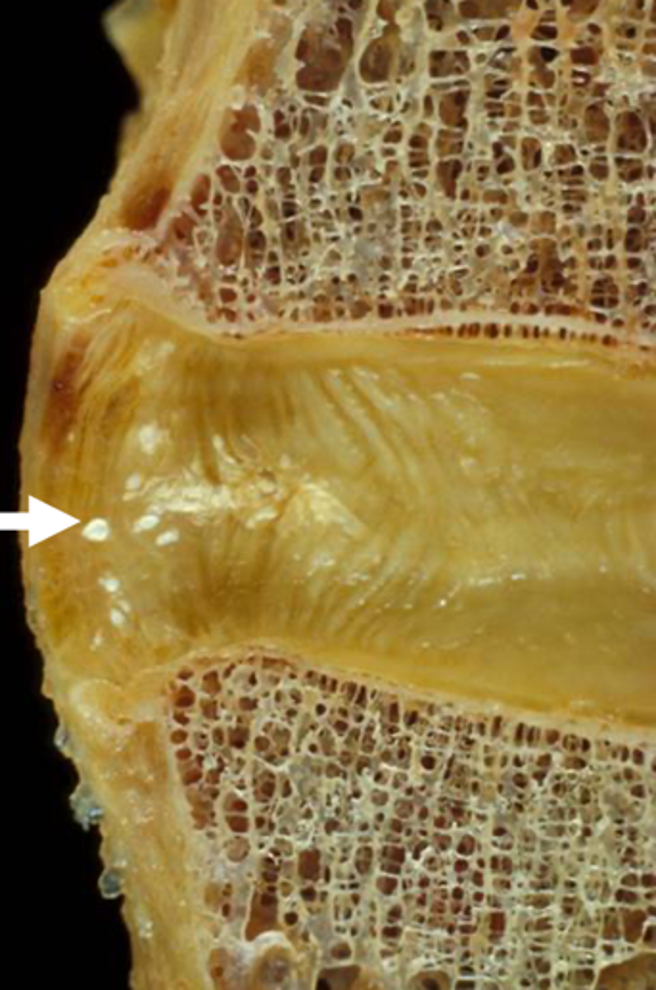
Figure 3: BCP crystal deposition affecting the shoulder. (3A) Illustration depicts BCP crystal deposition within a rotator cuff tendon at its footprint, without surrounding inflammation. (3B) Specimen photograph demonstrates BCP crystal deposition in the supraspinatus tendon at its footprint (arrow). (3C) Specimen photograph shows BCP crystal deposition of the long head of the biceps brachii tendon at the supraglenoid tubercle of the scapula (arrow). (3D) Specimen photograph shows BCP crystal deposition in the distal infraspinatus and teres minor tendons (arrows). (3E) Specimen photograph shows BCP crystal deposition in the proximal long head of the triceps tendon (arrow).
On conventional radiographs, well-defined calcification, especially when unchanging from one examination to another, is often asymptomatic. With MRI, such deposits are generally seen as well-defined globular areas of low signal intensity on all imaging sequences.
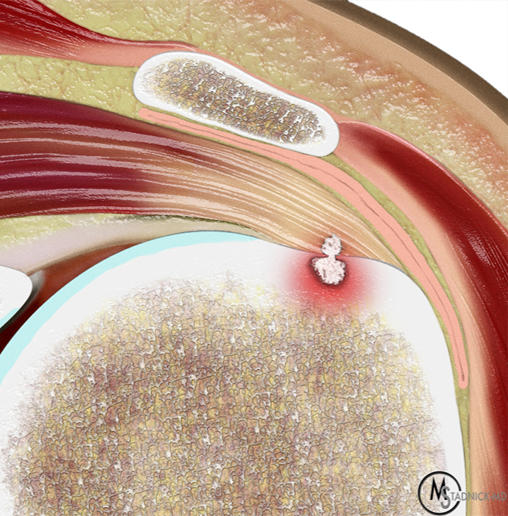
Pain is often associated with the migration of these calcific deposits, with three classic patterns of migration. The first of these relates to migration of the crystalline deposits into the adjacent subacromial–subdeltoid–subcoracoid bursa or beneath the floor of the bursa, often associated with bursitis and soft tissue edema (Figure 4).
Figure 4: Sub-bursal extrusion of BCP crystals in the shoulder. (4A) Illustration showing BCP crystals extruding through the bursal fibers of a rotator cuff tendon deep to the overlying bursa, with associated bursal inflammation. (4B) Axillary radiograph demonstrates an approximately 15 mm focus of BCP crystal deposition adjacent to the lesser tuberosity, in the region of the subscapularis tendon footprint (arrow). (4C) Sagittal fat-suppressed, T2-weighted image demonstrates calcific tendinitis of the inferior fibers of the subscapularis tendon (yellow arrow) with severe bursitis in the inferior portion of the subacromial-subdeltoid bursa (red arrows).
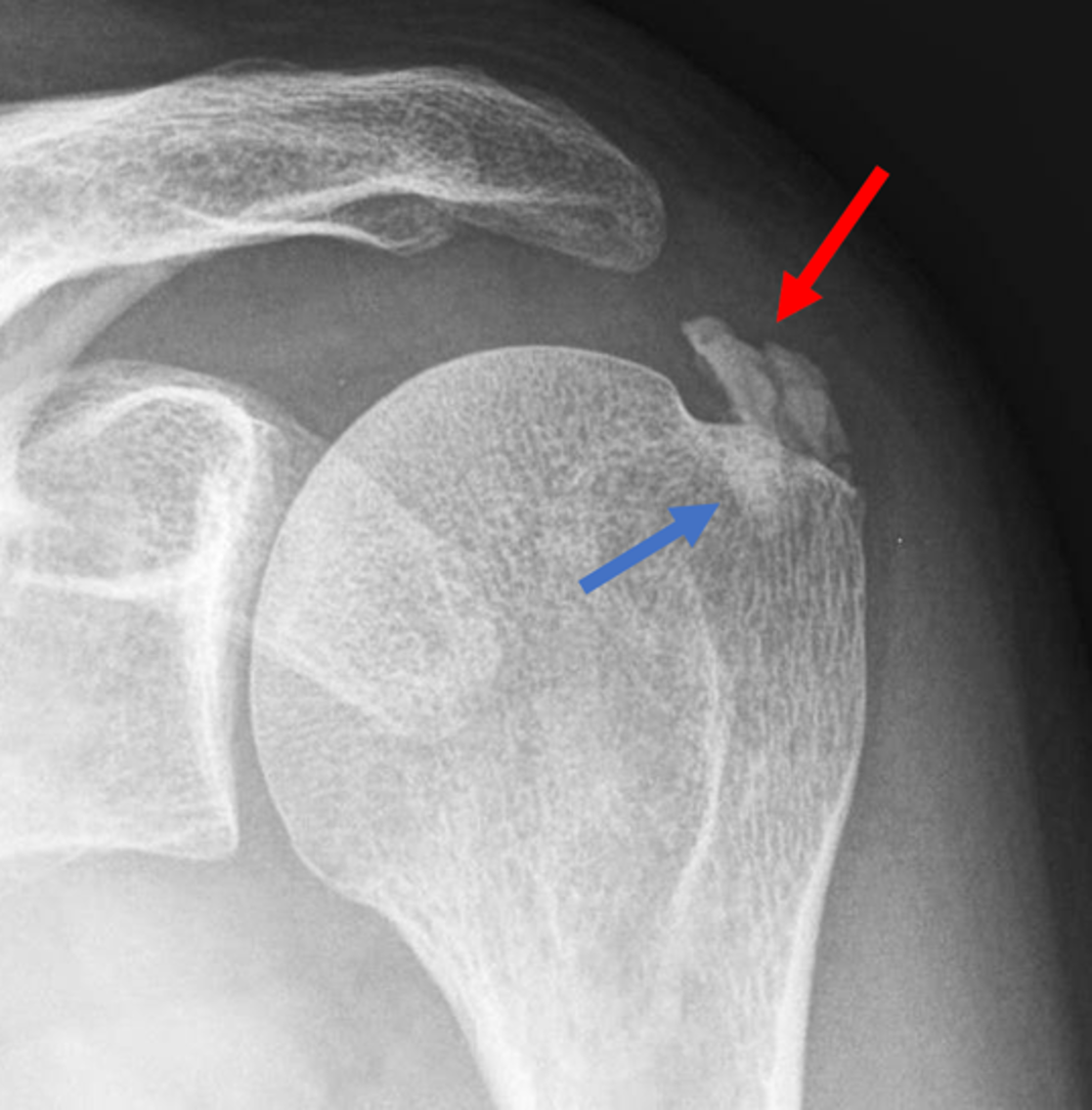
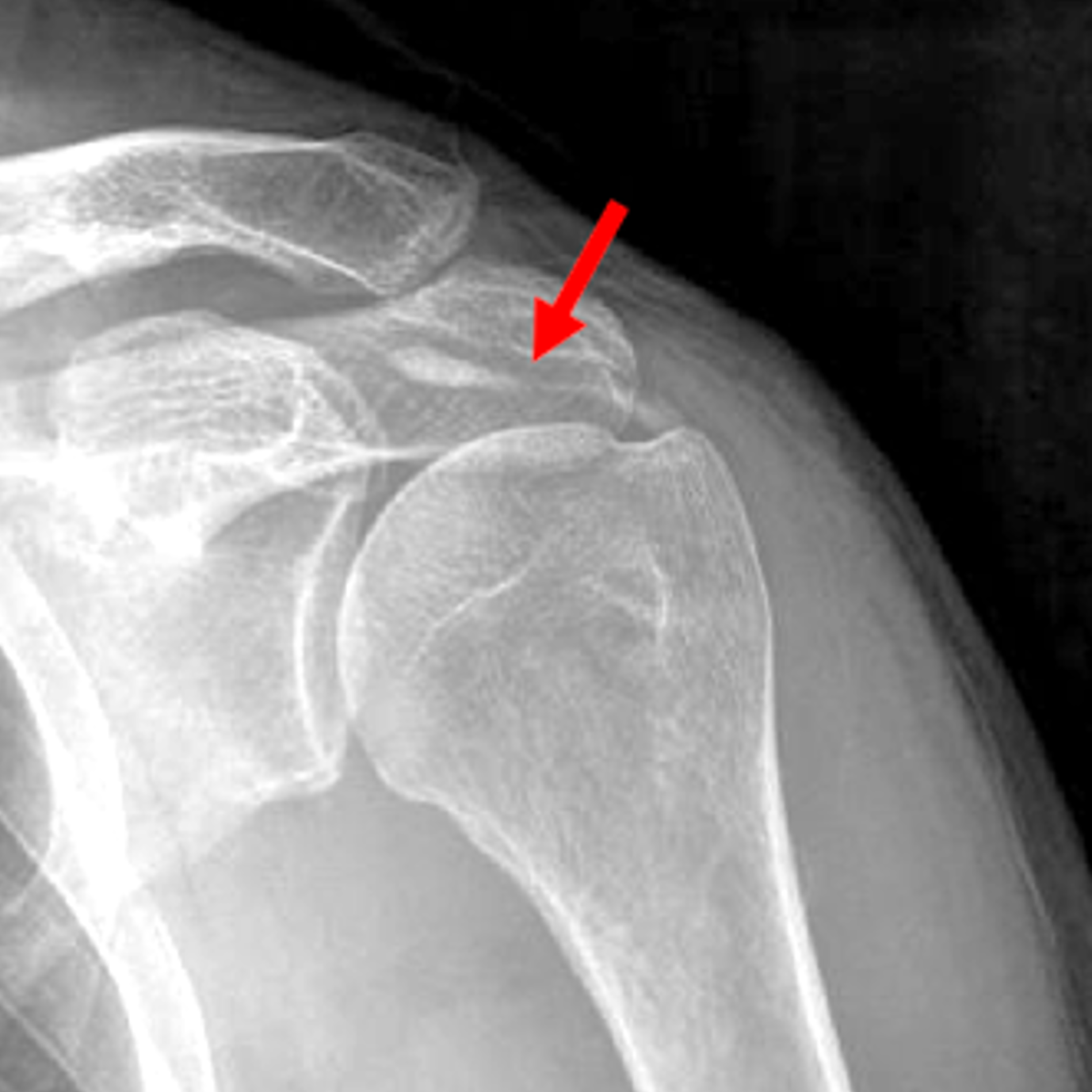
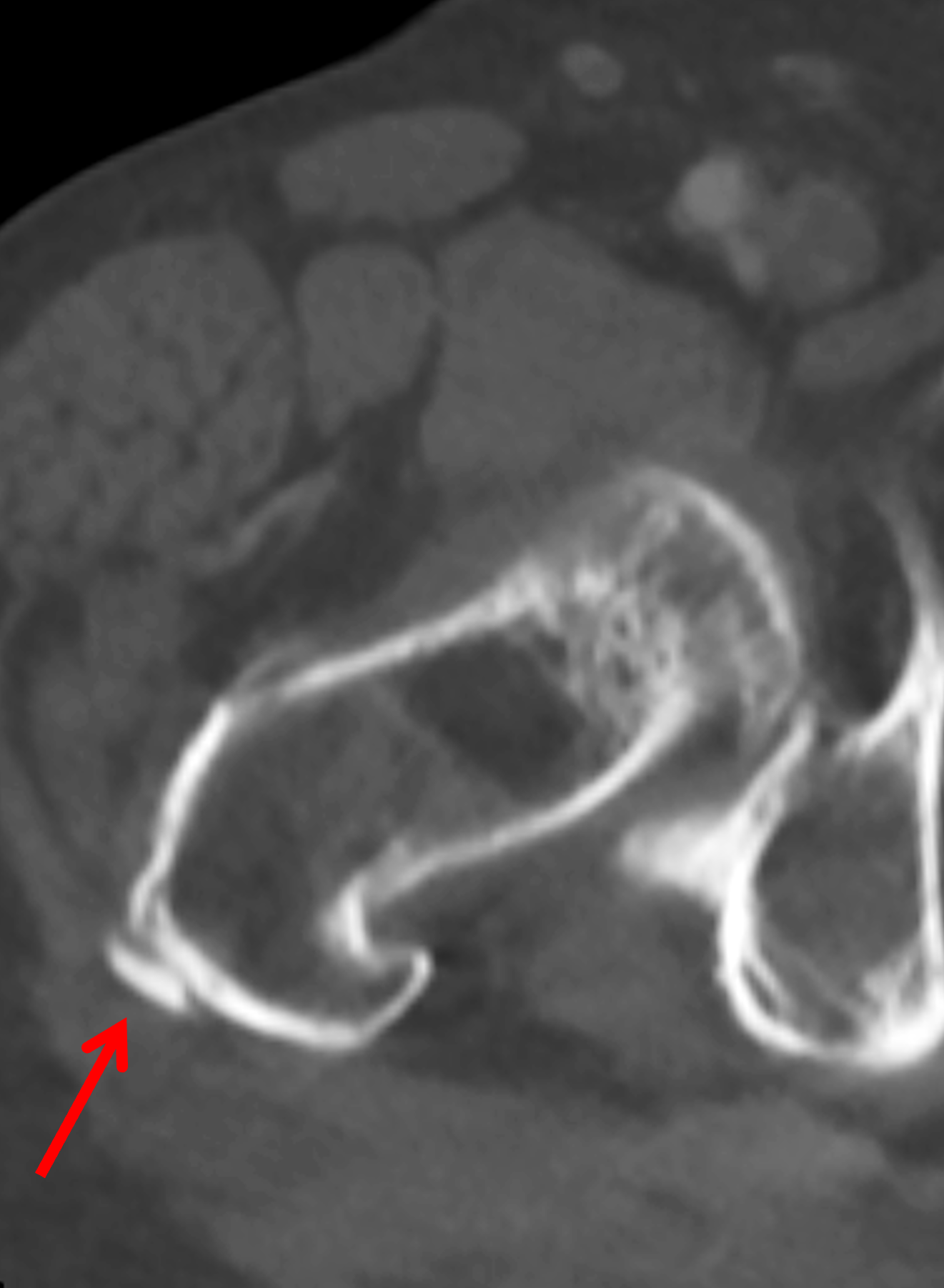
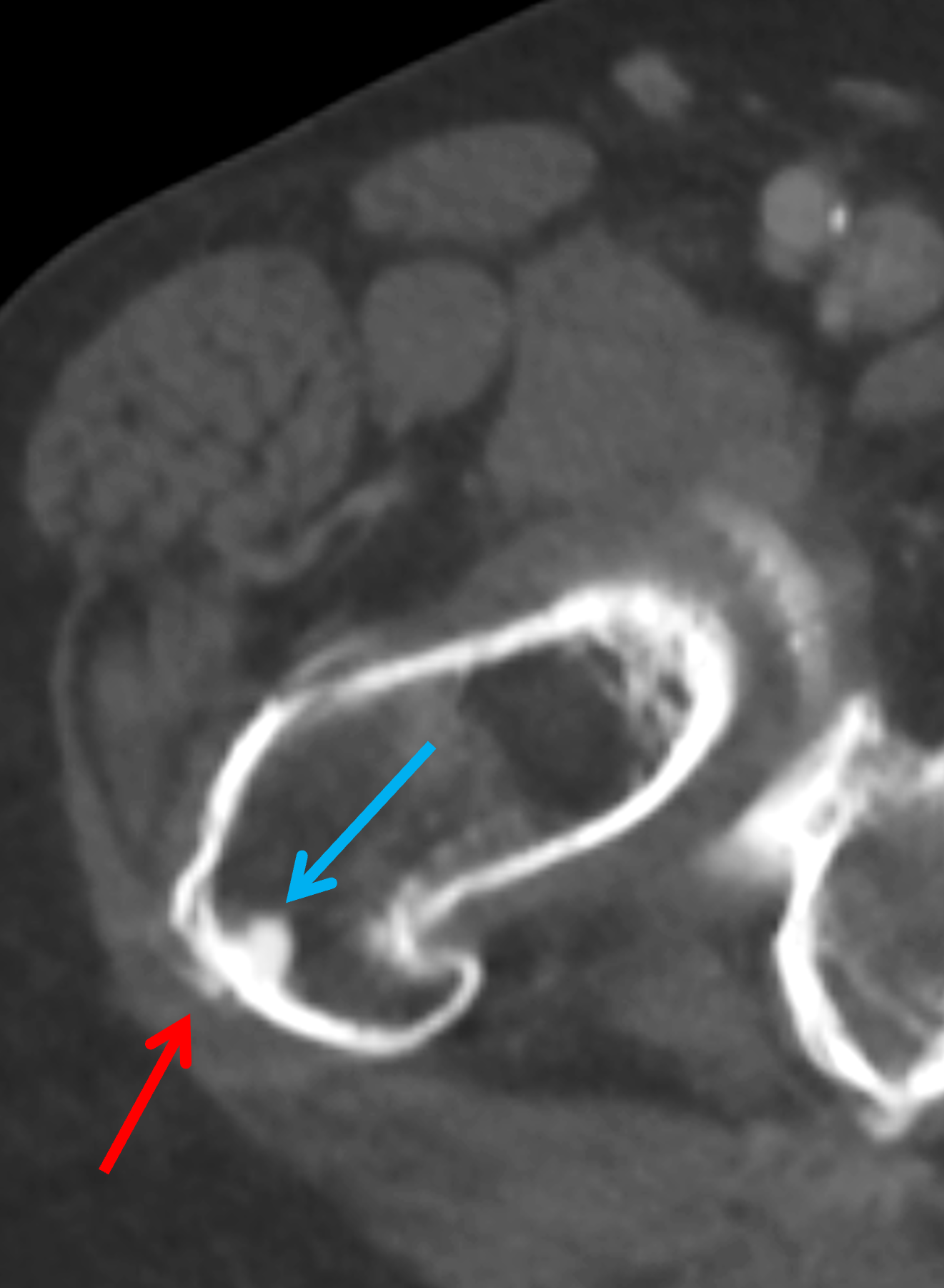
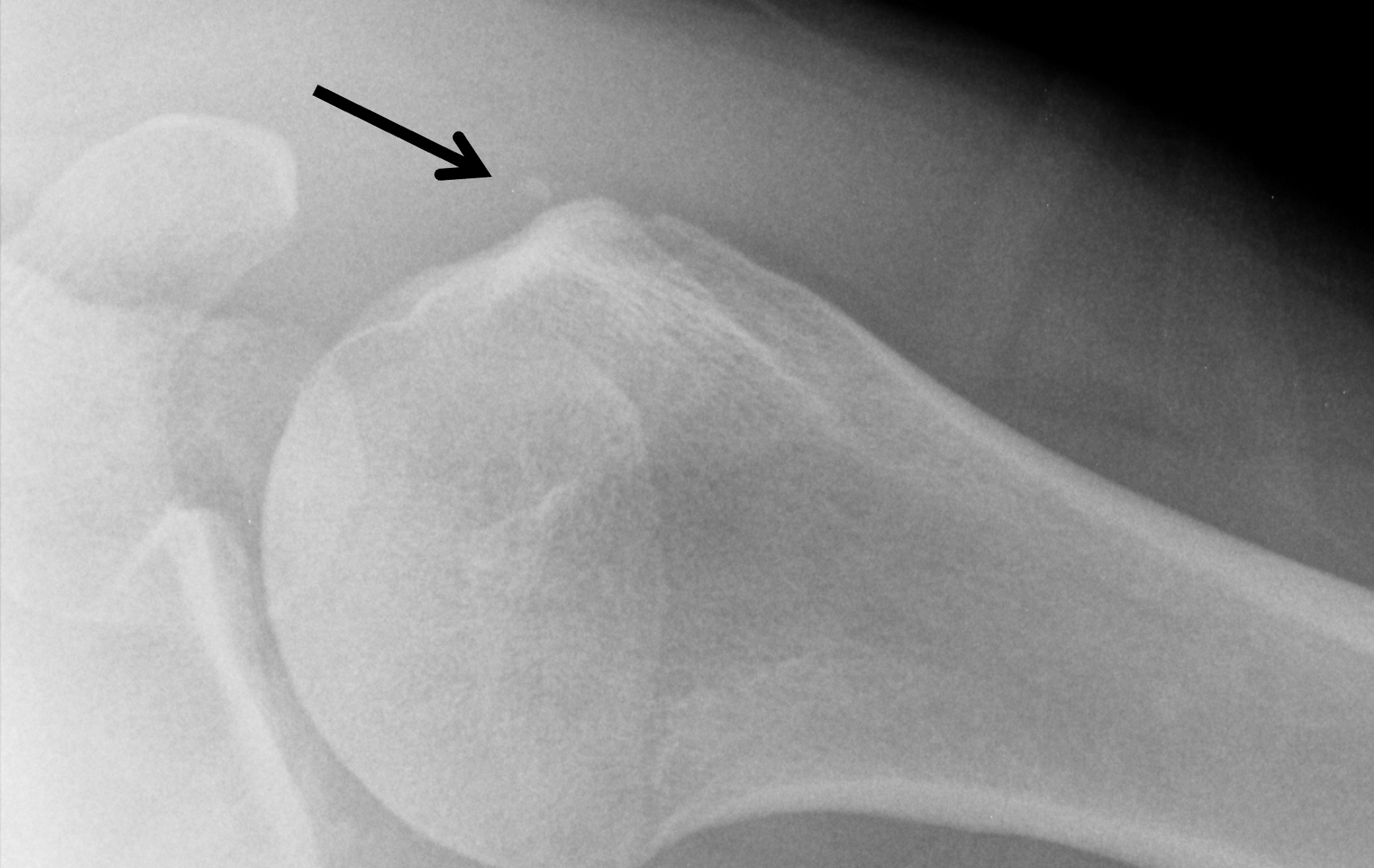
The second pattern of migration relates to intraosseous extension, usually at the greater tuberosity but, in cases of subscapularis tendon calcification, also the lesser tuberosity. MRI shows a low signal intensity intraosseous focus with associated bone marrow edema when the calcific deposit penetrates the adjacent bone (Figure 5), sometimes simulating the appearance of a neoplasm, such as a chondroblastoma, clear cell chondrosarcoma, or metastatic disease.
Figure 5: Intraosseous extension of BCP crystals in the shoulder. (5A) Illustration depicts BCP crystals extending through the articular surface fibers of a rotator cuff tendon and extending into the greater tuberosity, with surrounding marrow edema. (5B) Radiograph shows BCP crystal deposition in the rotator cuff footprint (red arrow) extending into the greater tuberosity (blue arrow). (5C) Coronal T1-weighted image shows low signal intensity BCP crystal deposition in the supraspinatus tendon (red arrow) extending into the greater tuberosity (blue arrow). (5D) Coronal fat-suppressed, T2-weighted image shows low signal intensity BCP crystal deposition in the supraspinatus tendon (red arrow) extending into the greater tuberosity (blue arrow) with surrounding bone marrow edema (arrowheads). Figures 5B, 5C, and 5D courtesy of E. Bosch, MD.
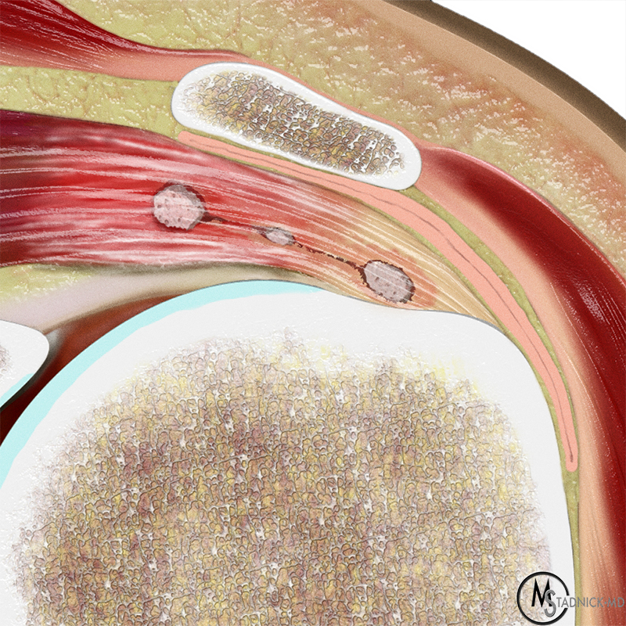
The final pattern relates to intratendinous extension of the calcification toward the myotendinous junction, typically associated with a delaminated tear of the tendon. It is not clear if the tear precedes the migration of the calcification or is caused by that migration. In these cases, foci of low signal can be seen in the tendon on MRI, often with associated muscle edema (Figure 6).
Figure 6: Intratendinous delamination of BCP crystals in the shoulder. (6A) Illustration showing BCP crystals extending medially within a rotator cuff tendon from the tendon footprint towards the myotendinous junction, with surrounding inflammation. (6B) AP radiograph demonstrates BCP crystal deposition (arrow), likely within the supraspinatus tendon, with medial intratendinous extension. In a second patient, (6C) axial CT image demonstrates BCP crystal deposition within the subscapularis tendon dissecting medially (arrow). (6D) Corresponding axial fat-suppressed, T2-weighted image demonstrates an intratendinous delaminating tear of the subscapularis tendon containing a low signal intensity focus of BCP crystal deposition (arrow). In a third patient, (6E) coronal and (6F) sagittal fat-suppressed, T2-weighted images demonstrate an intratendinous delaminating tear of the infraspinatus tendon associated with low signal intensity focus of BCP crystal deposition (red arrows), and regional soft tissue and muscle edema (blue arrows). Figures 6E and 6F, courtesy of I. Kjellin, MD.
In addition to the rotator cuff tendons, BCP crystals can also be deposited in other tendons about the shoulder, including the long and short head biceps brachii tendons, the triceps brachii tendon, or the pectoralis major tendon insertion muscle at the lateral lip of the humeral intertubercular groove (Figure 7).
Figure 7: BCP crystal deposition in the pectoralis major tendon. Shoulder radiographs with the humerus in (7A) internal rotation and (7B) external rotation demonstrate a focus of BCP crystal deposition at the proximal humeral shaft (arrows), either in the distal pectoralis major tendon or the myotendinous junction of the long head of the biceps brachii. (7C) Axial T1-weighted and (7D) sagittal fat-suppressed, T2-weighted images localize the low signal intensity BCP crystal deposit to the distal pectoralis major tendon near its humeral insertion (arrows).
Calcific Periarthritis about the Hip
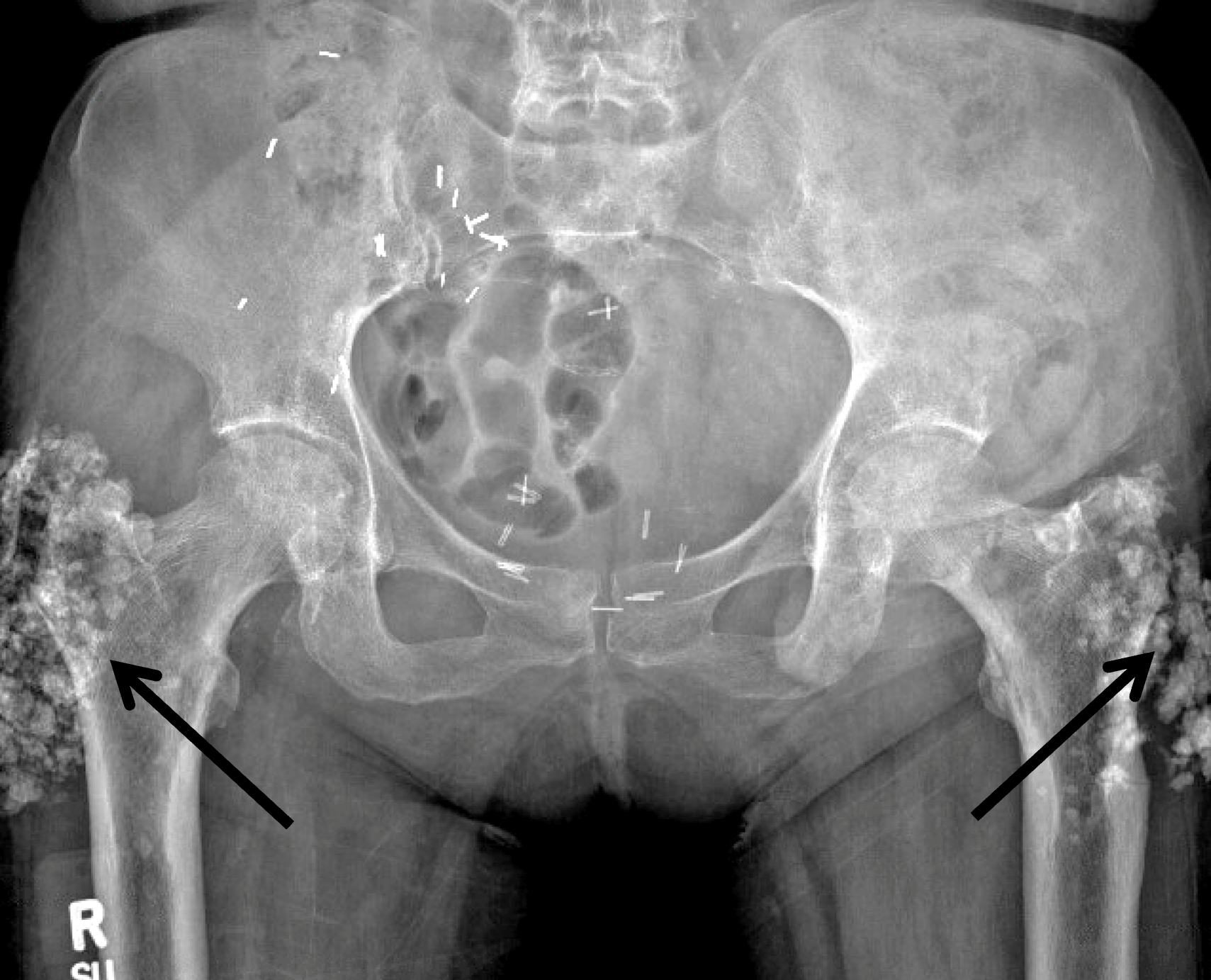
About the hip, globular calcific deposits at the gluteus minimus and gluteus medius tendon insertions at the greater trochanter as well as in the adjacent trochanteric bursa may be encountered (Figure 8).24 An additional common site of BCP crystal deposition is the gluteus maximus tendon insertion at the gluteal tuberosity on the posterior surface of the proximal femur (Figure 9). In some cases, erosion of the femoral cortex and adjacent soft tissue abnormality, or intraosseous extension of calcification, can simulate the appearance of a neoplasm (Figure 10).
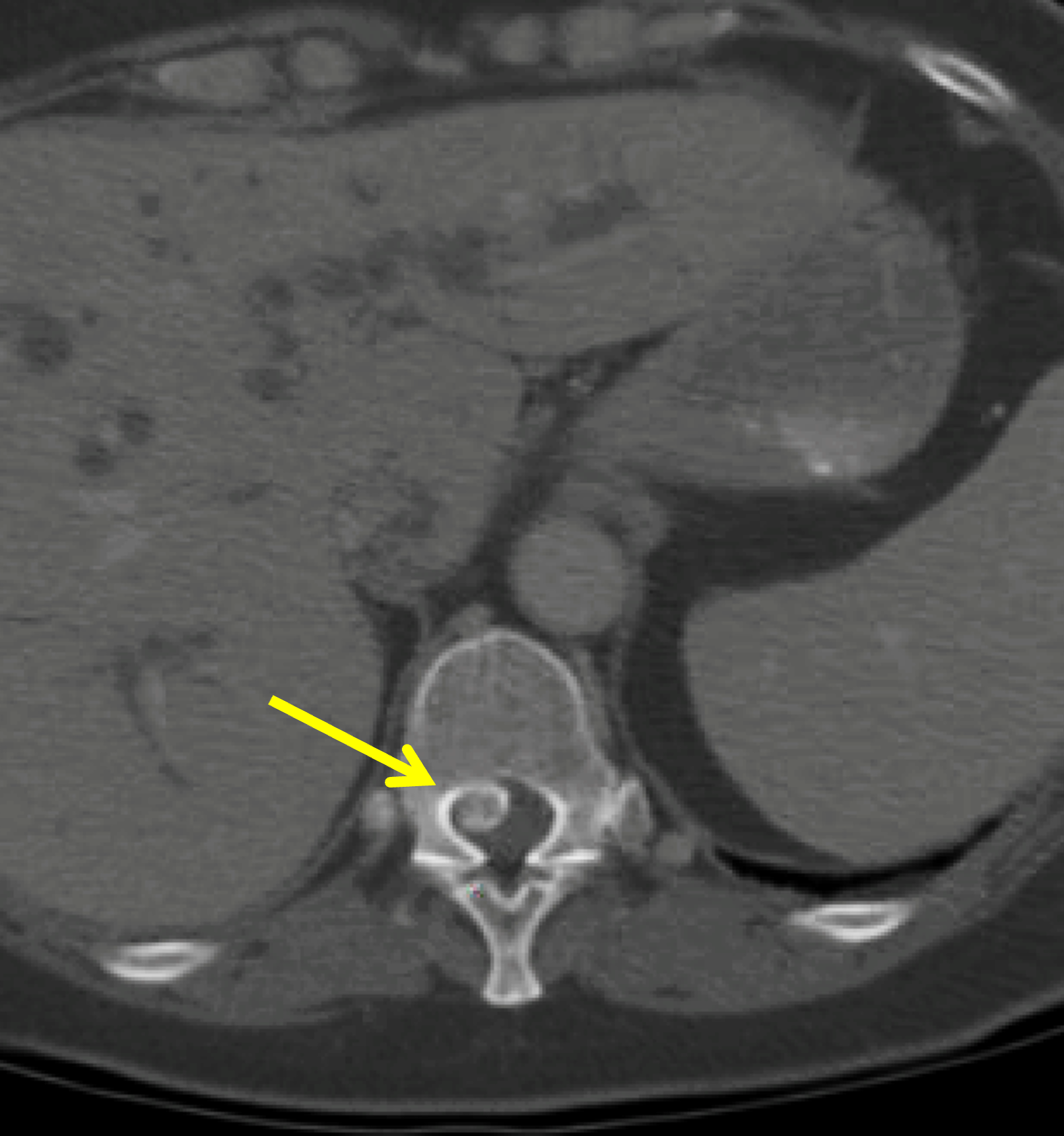
Figure 8: BCP crystal deposition involving the gluteus medius tendon insertion at the hip. (8A) Axial CT demonstrates BCP crystal deposition at the insertion of the gluteus medius tendon (white arrow) with intraosseous extension (black arrow). (8B) Axial fat-suppressed T2-weighted image demonstrates findings compatible with active calcific tendinitis from BCP crystal deposition within the gluteus medius tendon with surrounding soft tissue inflammation (white arrow), and marrow edema within the greater trochanter (yellow arrow).
Figure 9: BCP crystal deposition involving the gluteus maximus tendon insertion at the hip in a patient with history of prostate cancer. (9A) AP radiograph demonstrates BCP crystal deposition at the tendinous insertion of the gluteus maximus muscle (arrow), initially interpreted as cortical irregularity and periosteal reaction with associated mild cortical thinning/saucerization of the lateral cortex. Given the history of prostate cancer, a CT and MRI were requested for further evaluation. (9B) Axial CT confirms BCP crystal deposition in the distal gluteus maximus tendon insertion (arrow). (9C) Axial T1-weighted image shows a low signal intensity focus of BCP crystal deposition at the tendinous insertion of the gluteus maximus muscle (arrow). (9D) Axial fat-suppressed T2-weighted image demonstrates surrounding soft tissue edema (yellow arrows) as well as edema in the adjacent bone (white arrow). (9E) Sagittal STIR image redemonstrates soft tissue edema (yellow arrows) surrounding the calcification and associated marrow edema (white arrow). (9F) Sagittal fat-suppressed T1-weighted image acquired following intravenous contrast administration shows enhancement (arrow) representing surrounding soft tissue inflammation.
Figure 10: BCP deposition around the hip simulating metastatic disease in a patient with a history of colon cancer. (10A) Initial CT image shows a focus of BCP crystal deposition at the tendinous insertion of the gluteus medius muscle (arrow). (10B) CT obtained one year later demonstrates a residual focus of BCP crystal deposition at the same site (red arrow) as well as new intraosseous extension of the BCP crystals (blue arrow), which was interpreted as a possible sclerotic metastasis. (10C) CT obtained one month later demonstrates complete resorption of the BCP crystal deposition in the tendon and partial resorption of the sclerotic focus in the bone (arrow), confirming that these findings were due to the evolution of calcific periarthritis, and thus biopsy was deferred. (10D) CT obtained three years later demonstrates near complete resolution of the intraosseous calcification (arrow).
Other well-known sites of BCP crystal deposition about the hip and pelvis include the tendons of the sartorius, rectus femoris, and hamstring muscles, as well as those of the iliacus and psoas muscles. Migration of calcific deposits into the greater trochanteric or iliopsoas bursa can result in marked bursitis with associated focal areas of low signal intensity.
Calcific Periarthritis about the Knee
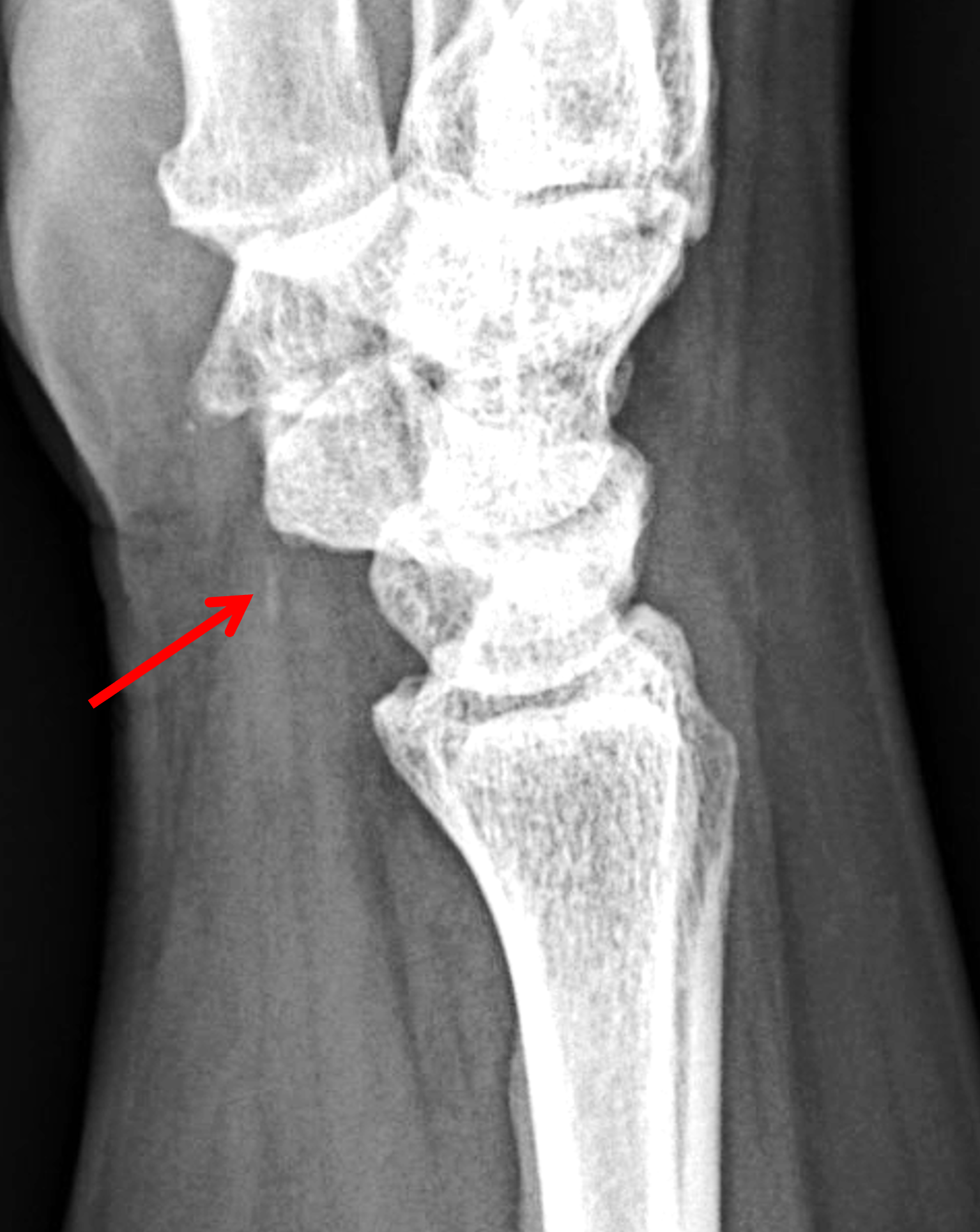
About the knee, BCP crystal deposition within the adjacent tendons can manifest on MRI as globular foci of low signal intensity on all MRI sequences. BCP crystal deposits within the popliteus tendon at the popliteus sulcus can mimic the appearance of tophaceous gout (Figure 11). A thorough history, physical examination, and laboratory studies can help distinguish these two entities.31
Figure 11: BCP crystal deposition involving the popliteus tendon insertion at the knee. (11A) AP radiograph demonstrates an 18 mm focus of BCP crystal deposition adjacent to the lateral femoral condyle (red arrow). An involuting lipoma in the lateral femoral condyle (black arrow) was confirmed on MRI. (11B) Coronal T1-weighted image demonstrates multiple foci of low signal intensity corresponding to BCP crystal deposition around the popliteus tendon insertion (arrow). (11C) Coronal fat-suppressed T2-weighted image shows extensive soft tissue edema (arrows) surrounding the calcifications, compatible with active calcific tendinitis.
Calcific Periarthritis about the Wrist
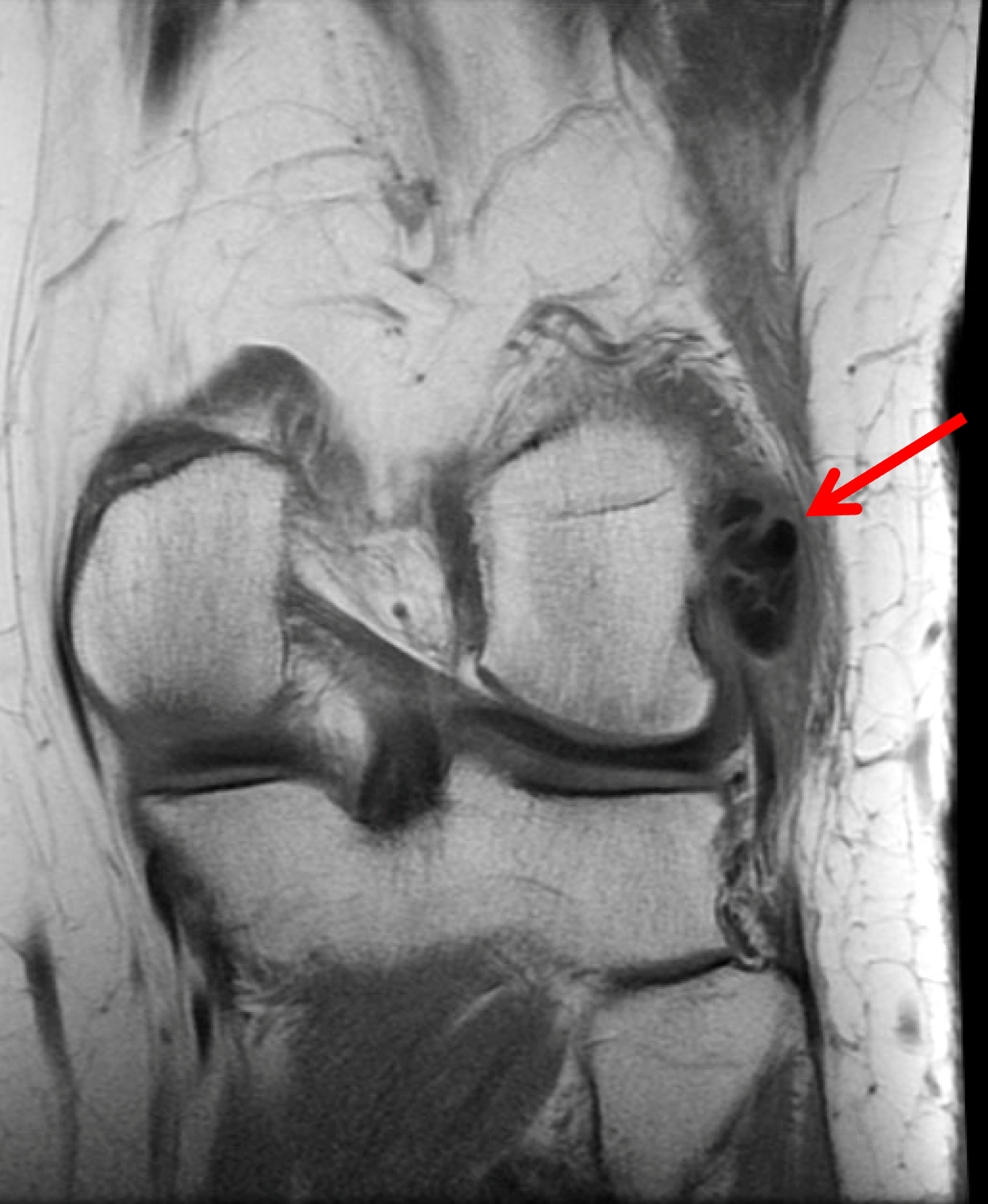
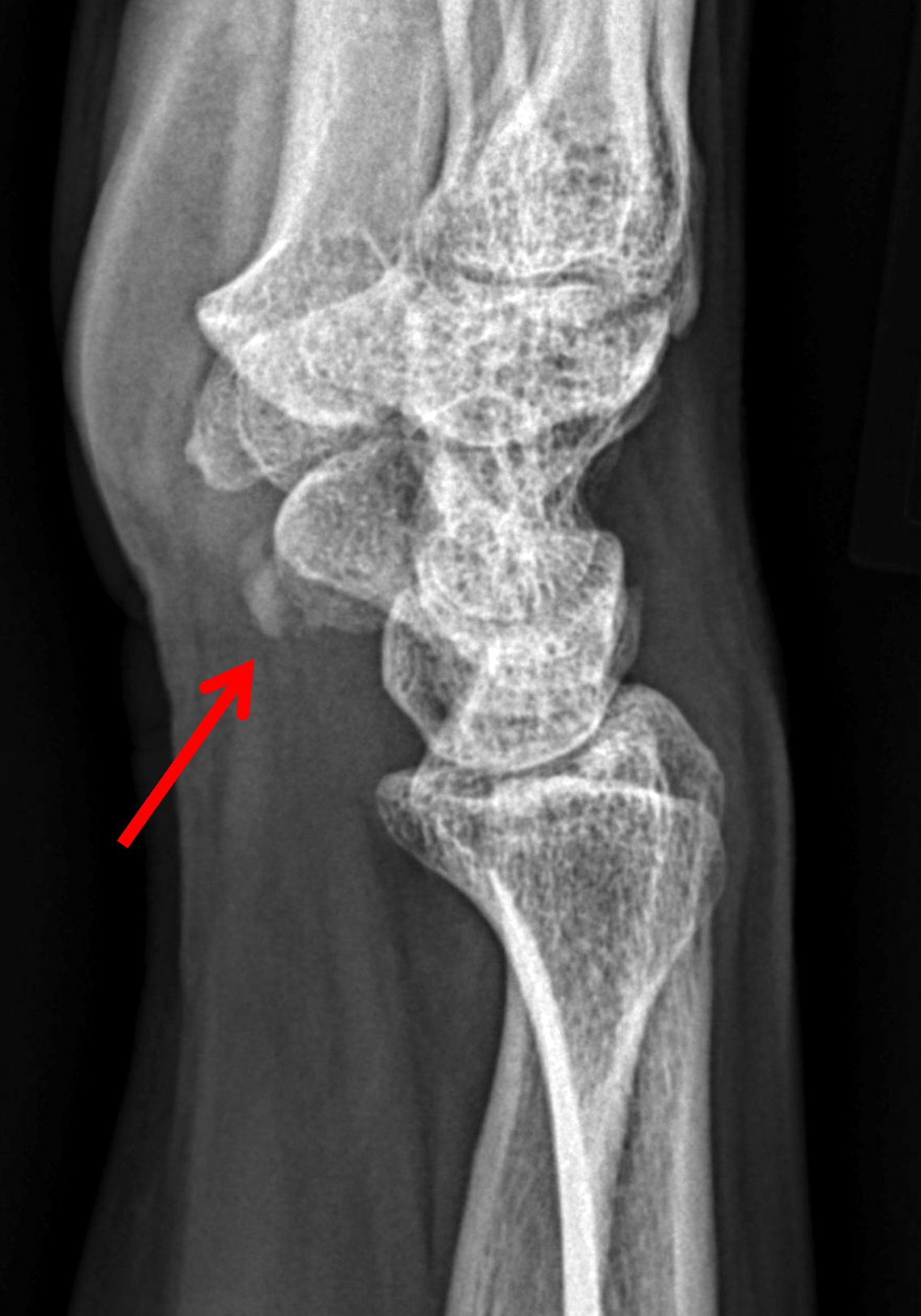
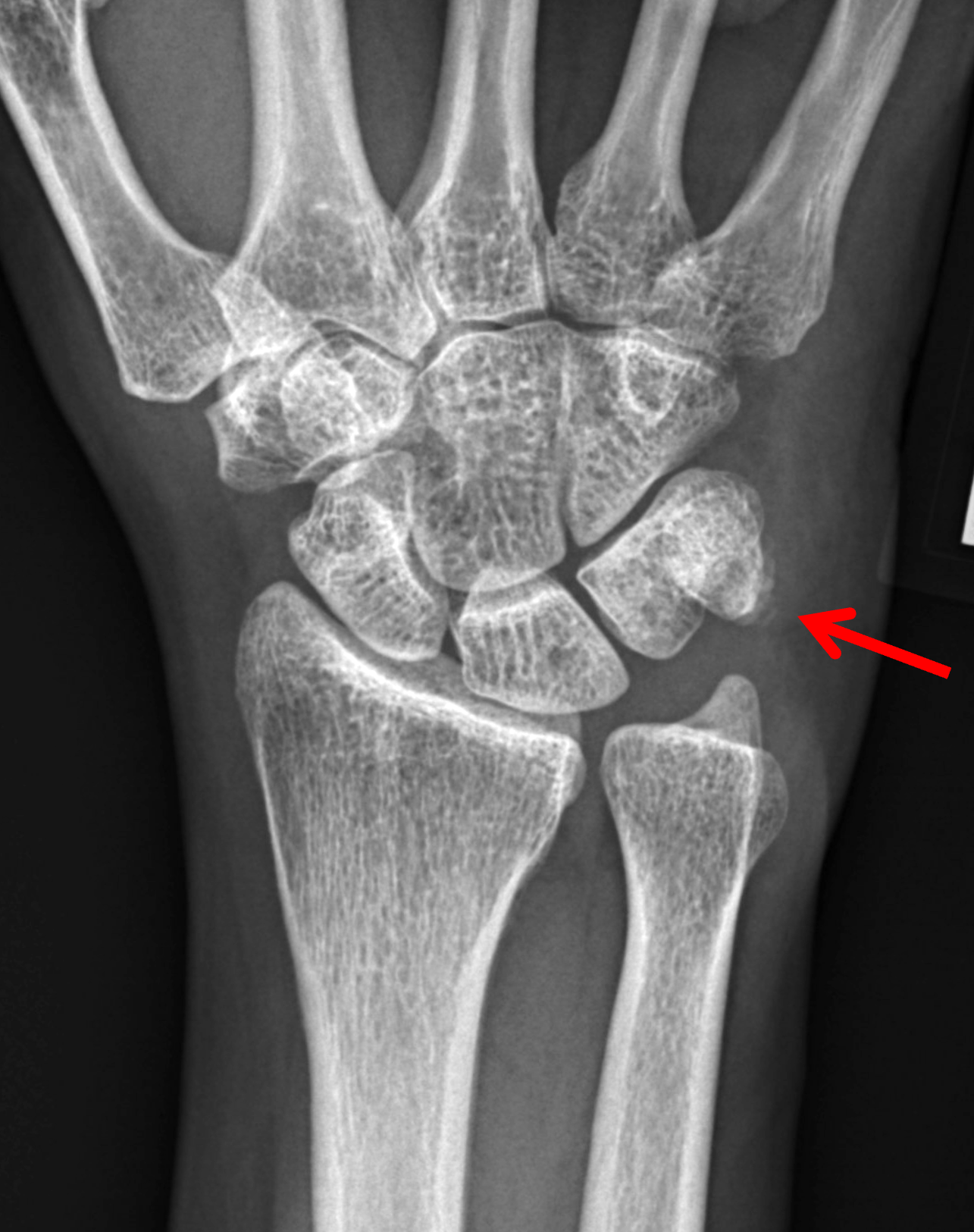
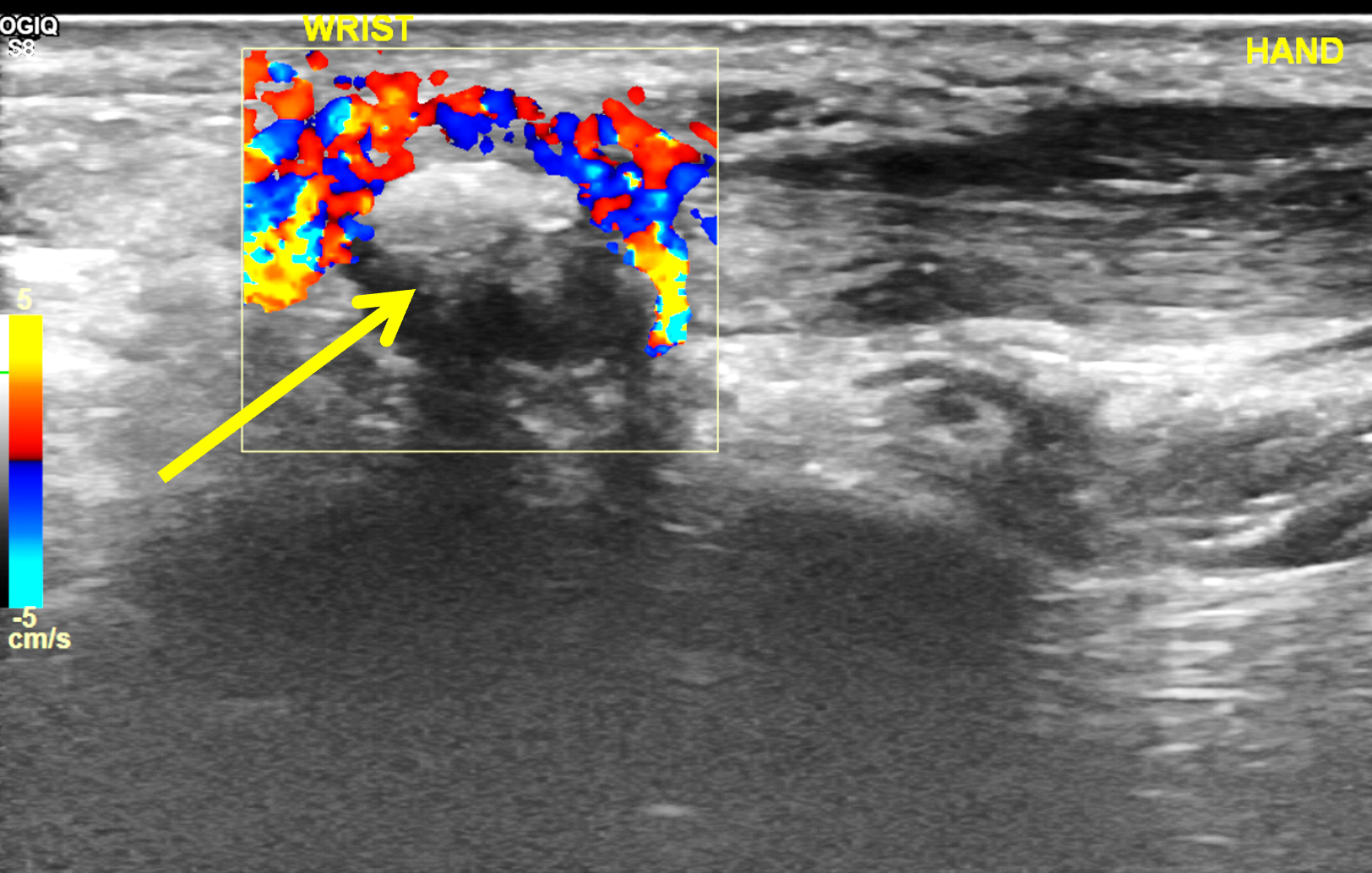
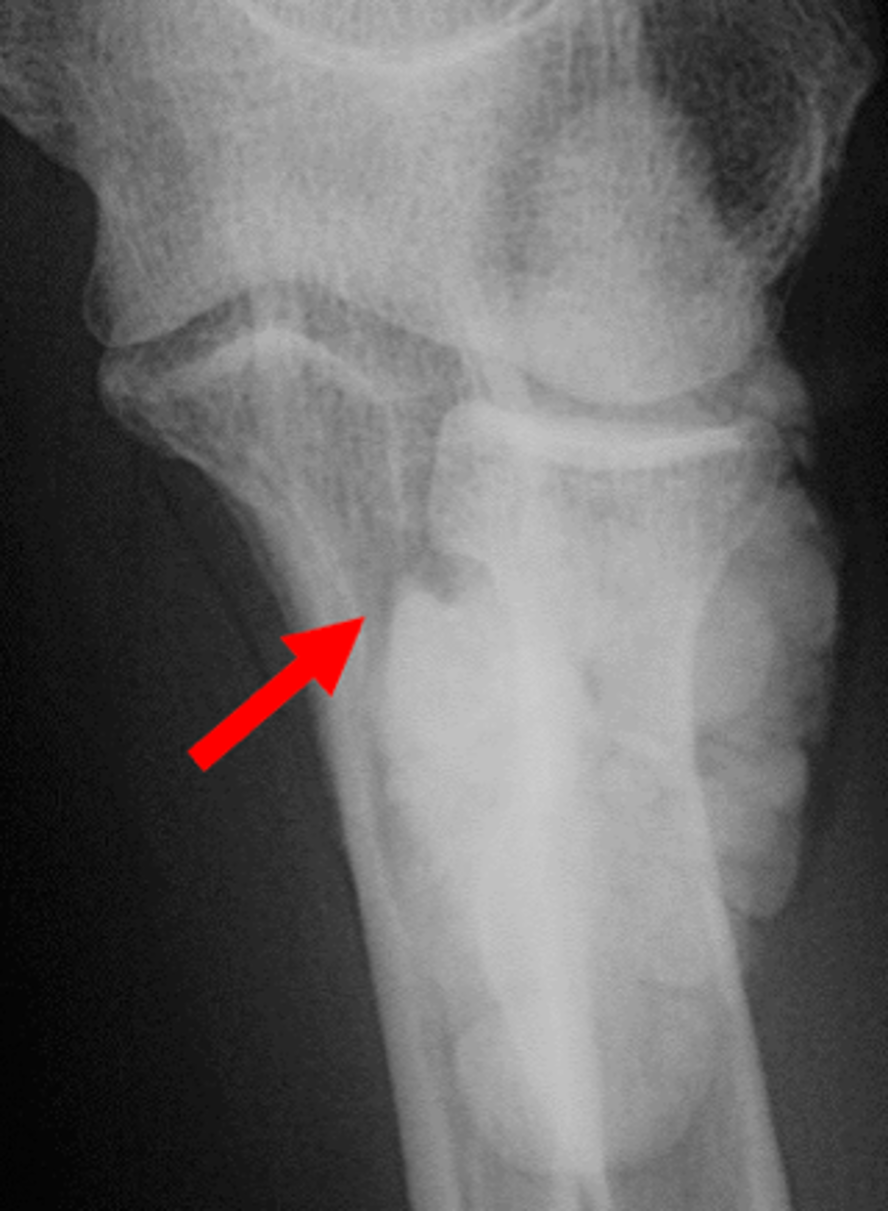
About the wrist, BCP crystal deposition frequently occurs in the flexor carpi ulnaris tendon and can manifest on imaging as calcification just proximal to the pisiform (Figure 12). BCP crystal deposition also commonly affects the flexor carpi radialis tendon, volar to the distal radioulnar joint, as well as the extensor carpi ulnaris tendon, adjacent to the distal portion of the ulna and ulnar styloid process.24
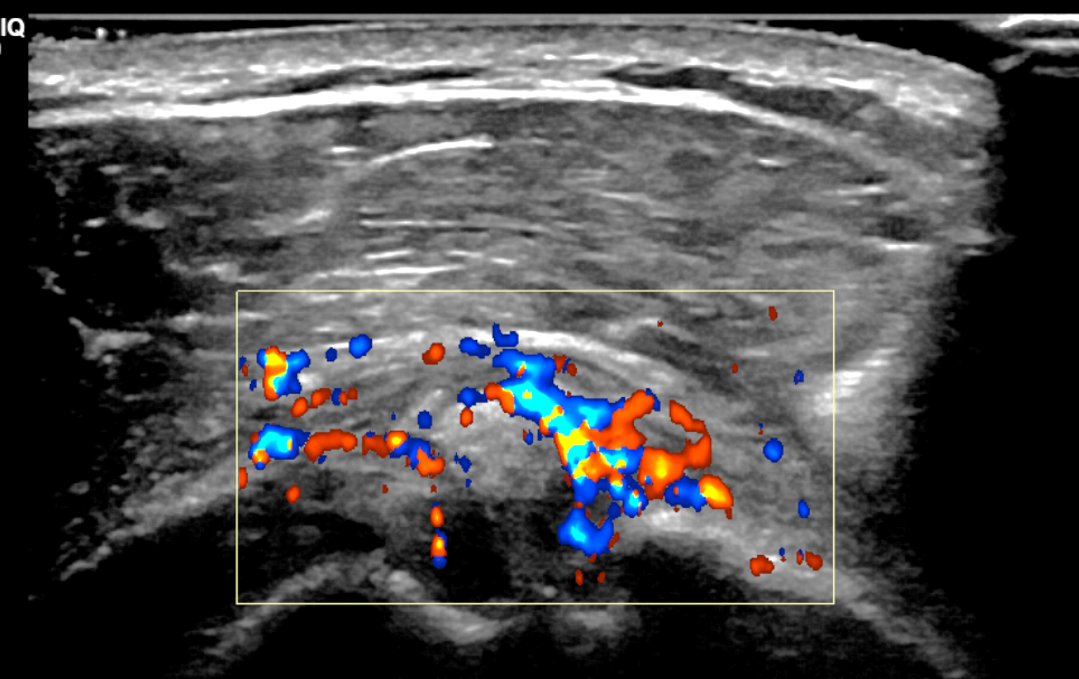
Figure 12: BCP crystal deposition involving the flexor carpi ulnaris tendon at the wrist. (12A) Lateral and (12B) PA radiographs demonstrate BCP crystal deposition at the palmar aspect of the pisiform (arrows), likely within the flexor carpi ulnaris tendon. (12C) Ultrasonography shows hyperechoic BCP crystal deposition within the right flexor carpi ulnaris tendon (arrow) with acoustic shadowing and intense surrounding increased Doppler signal due to inflammation. In a second patient, (12D) lateral radiograph and (12E) coronal fat-suppressed T2-weighted image demonstrate BCP crystal deposition within the flexor carpi ulnaris tendon (arrows).
Calcific Periarthritis about the Elbow
About the elbow, BCP crystals can be deposited in the origins of the common flexor and extensor tendon origins at the medial and lateral humeral epicondyles (Figure 13), respectively, as well as the tendinous insertions of the long and lateral heads of the triceps tendon at the olecranon (Figure 14).24 Crystal migration from the triceps tendon into the olecranon bursa may result in severe olecranon bursitis.
Figure 13: BCP crystal deposition involving the extensor tendon origin in the elbow. (13A) AP radiograph demonstrates BCP crystal deposition in the soft tissues adjacent to the lateral humeral epicondyle (arrow). (13B) Coronal and (13C) axial fat-suppressed T2-weighted images demonstrate inflammatory changes in the common extensor tendon with moderate grade interstitial tearing and soft tissue edema (arrows), related to recent extrusion and inflammation from active calcific periarthritis.
Figure 14. BCP crystal deposition involving the distal triceps tendon at the elbow. (14A) Sagittal and (14B) coronal T1-weighted images demonstrate a low signal intensity focus within the distal triceps tendon at the olecranon (arrows). There was no migration of the calcific deposit into the olecranon bursa or active inflammation.
Calcific Periarthritis in the Neck and Discal Calcification
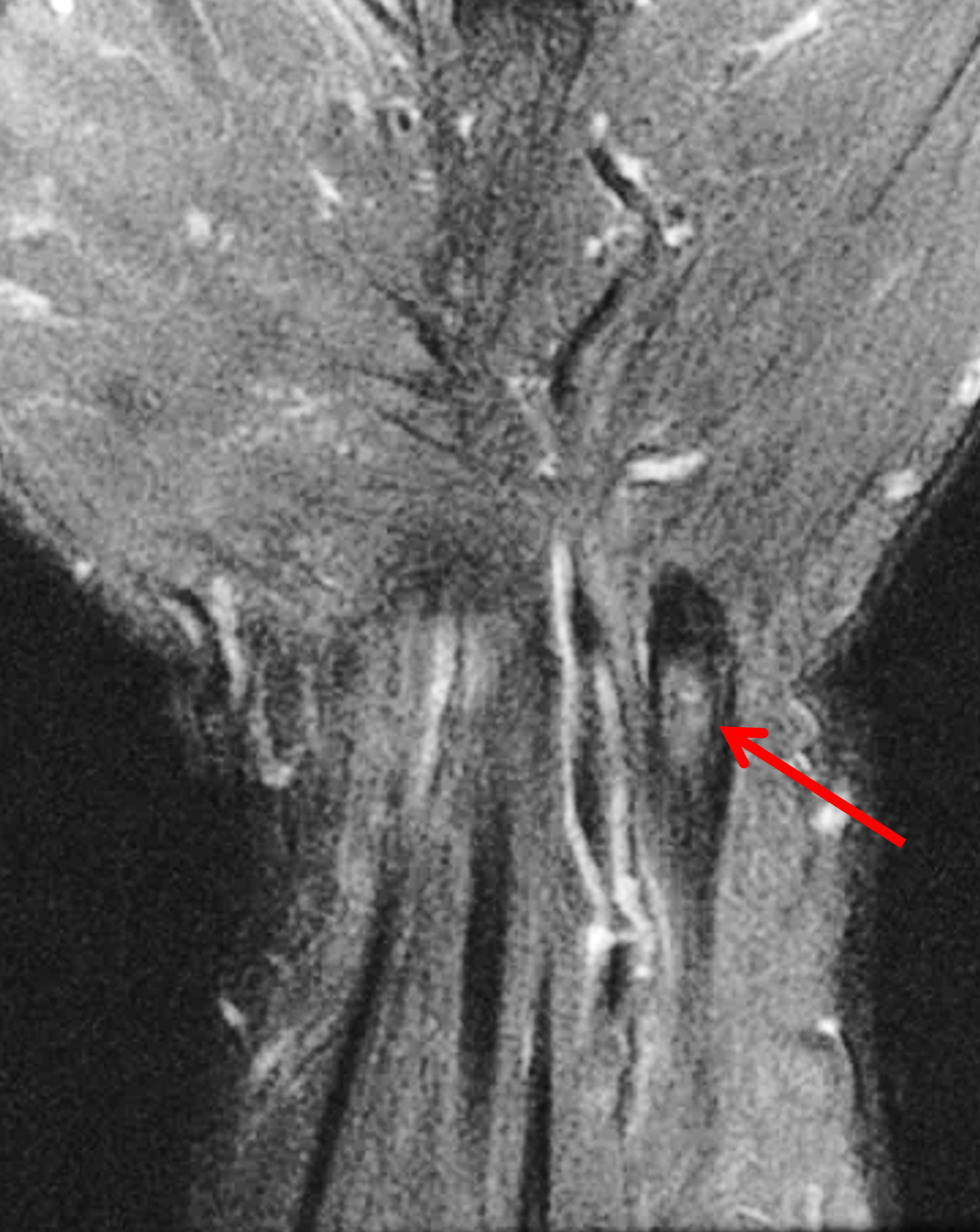
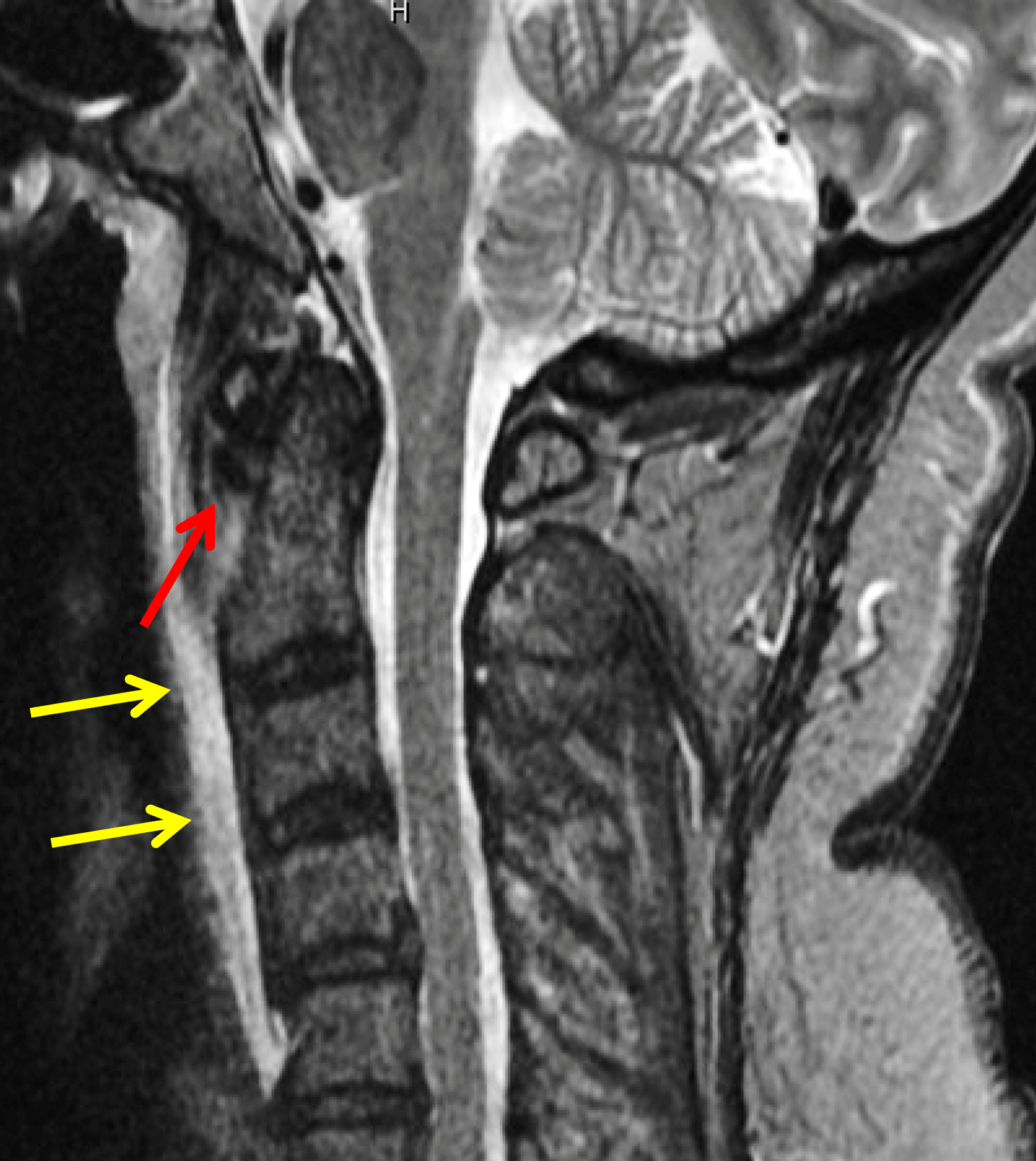
In the neck, globular calcific deposits within the superior tendinous fibers of the longus colli muscle are usually seen in the prevertebral space at the level of the axis,32 and are associated with marked edema appearing as thickening of the prevertebral soft tissues on radiographs and high signal intensity that may extend inferiorly throughout the cervical spine on fluid-sensitive MRI sequences (Figure 15). Although the imaging findings may initially raise the alternative diagnostic possibility of the crowned dens syndrome related to CPPD crystal deposition disease, the location of the calcification and soft tissue prominence in the latter disease are typically posterior to the odontoid process. It is important to distinguish calcific tendinitis of the longus colli muscle from a retropharyngeal abscess, which also is accompanied by prevertebral soft tissue swelling and soft tissue enhancement following intravenous contrast administration. Another differential diagnostic consideration is hyperostosis secondary to prolonged treatment with retinoid-containing medications such as 13-cis-retinoic acid.33
Figure 15: BCP crystal deposition involving the longus colli muscles in the neck. (15A) Sagittal CT reconstructed image demonstrates BCP crystal deposition within the superior tendinous fibers of the longus colli muscle (arrow). (15B) Sagittal fat-suppressed T2-weighted image shows low signal intensity calcifications (red arrow) and associated extensive retropharyngeal/prevertebral soft tissue edema (yellow arrows), likely secondary to calcific tendinitis.
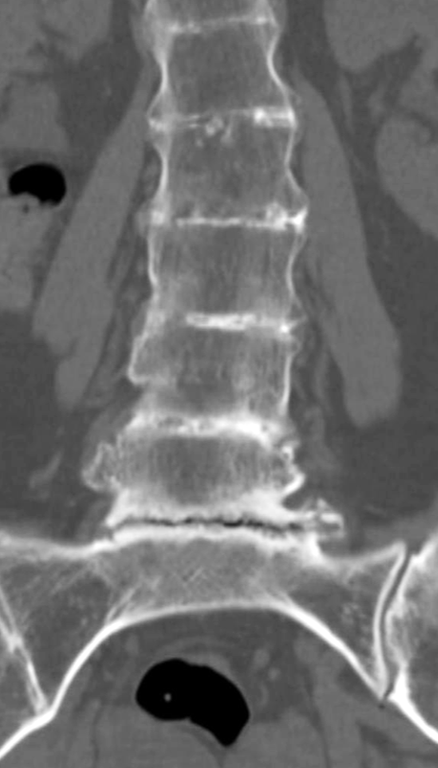
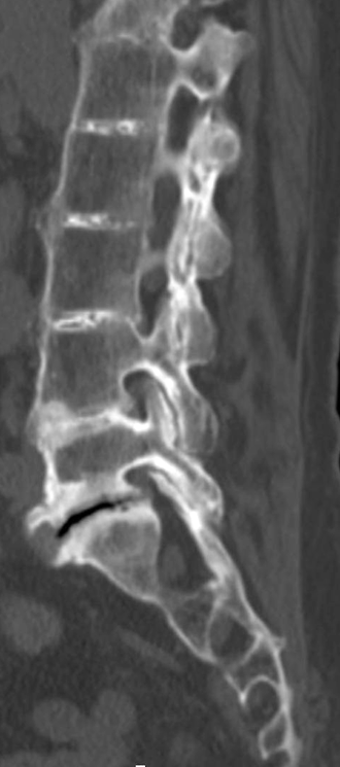
Although calcification of one or more of the intervertebral discs does not fit into the general category of calcific periarthritis, BCP crystal deposition in these discs has been associated with several processes. For example, extensively calcified and displaced intervertebral discs, especially in the thoracic spine, are often very large and are referred to as giant calcified herniated discs (Figure 16), further defined as a herniated disc that occupies more than 40% of the sagittal diameter of the thoracic spinal canal.34 The pattern of calcification in these discs varies in extent, leading to the description of three subtypes: dense, partial, and soft (i.e., developing) calcifications. These giant calcified thoracic discs belong to a subgroup of high-risk herniated thoracic discs,35 requiring challenging surgical treatment.36
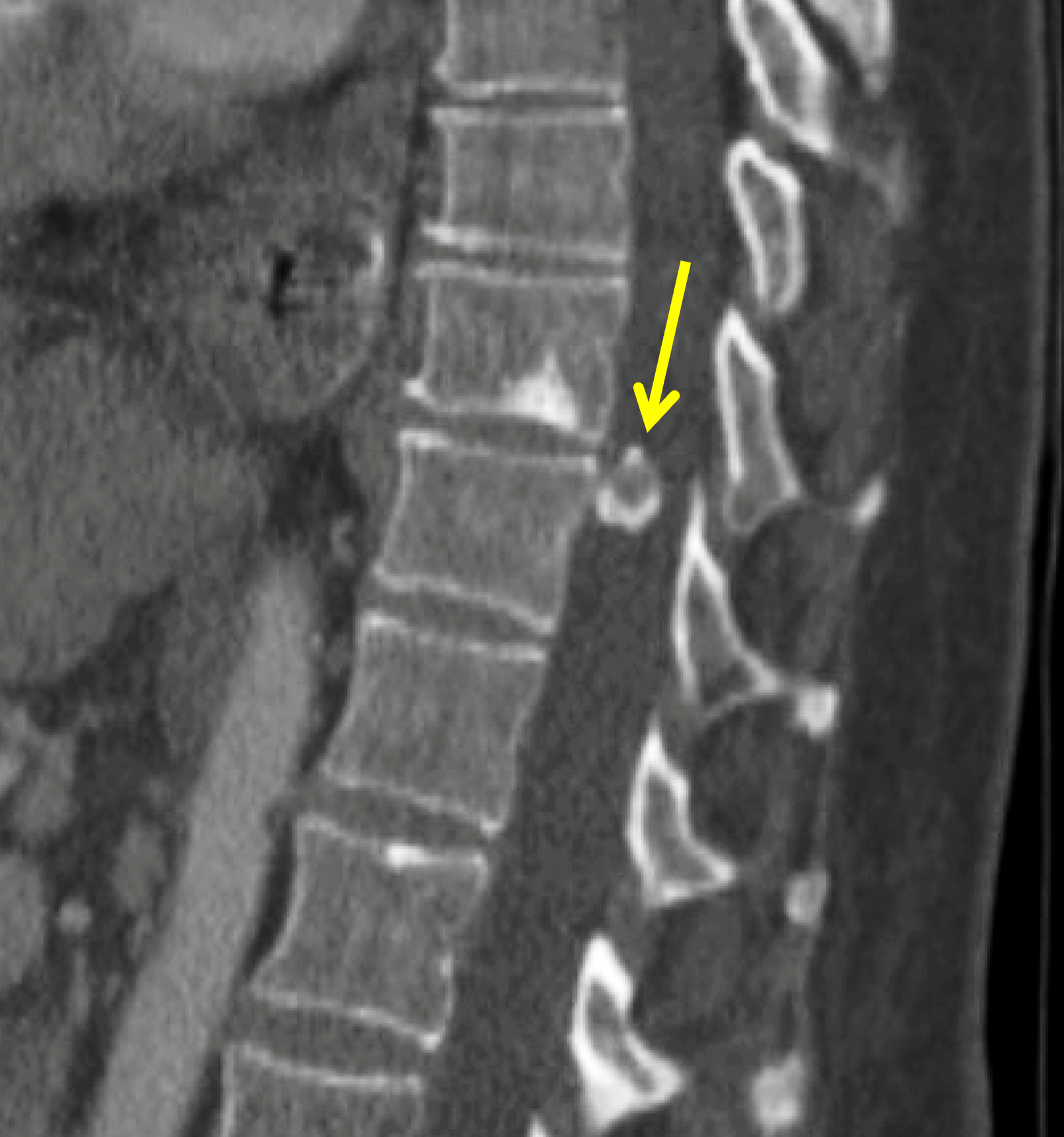
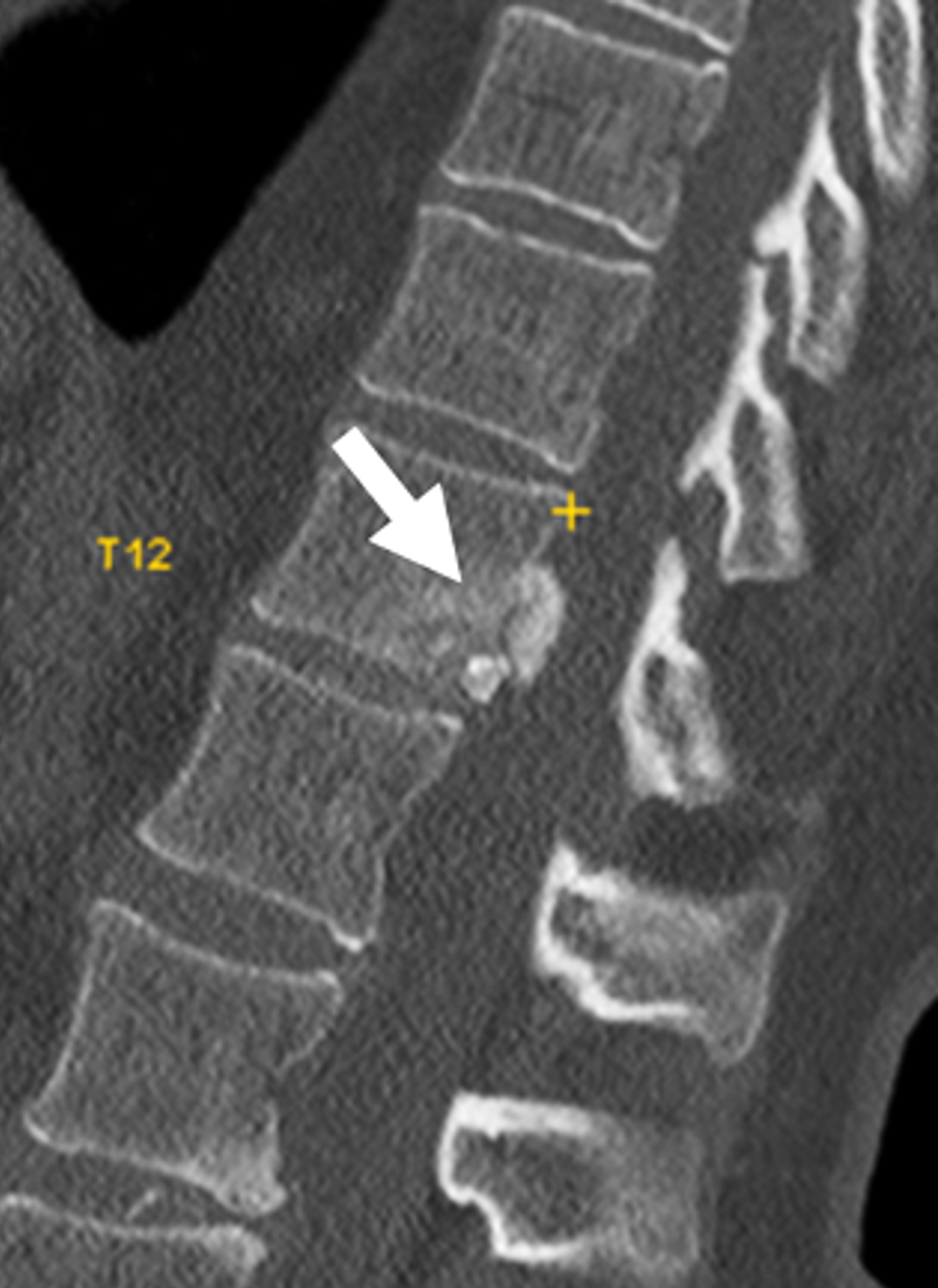
Figure 16: Giant calcified thoracic disc herniations. (16A) Axial and (16B) sagitally-reconstructed CT images demonstrate an approximately 10 mm giant calcified herniated disc fragment at T11 (arrows). In a second patient, (16C) sagitally-reconstructed CT image demonstrates a giant calcified herniated disc fragment (arrow) at T12. (16D) Corresponding sagittal fat-suppressed T2-weighted image shows reactive bone marrow edema within the T12 vertebral body (arrow).
Unlike the peripheral annular calcifications that relate to CPPD crystals (Figure 17), BCP crystals are initially deposited centrally, both within and adjacent to the nucleus pulposus.
Figure 17: CPPD crystal deposition in the intervertebral disc. (17A) AP radiograph and (17B) specimen photograph demonstrate the peripheral annular calcifications (arrows) characteristic of CPPD crystal deposition disease within the spine.
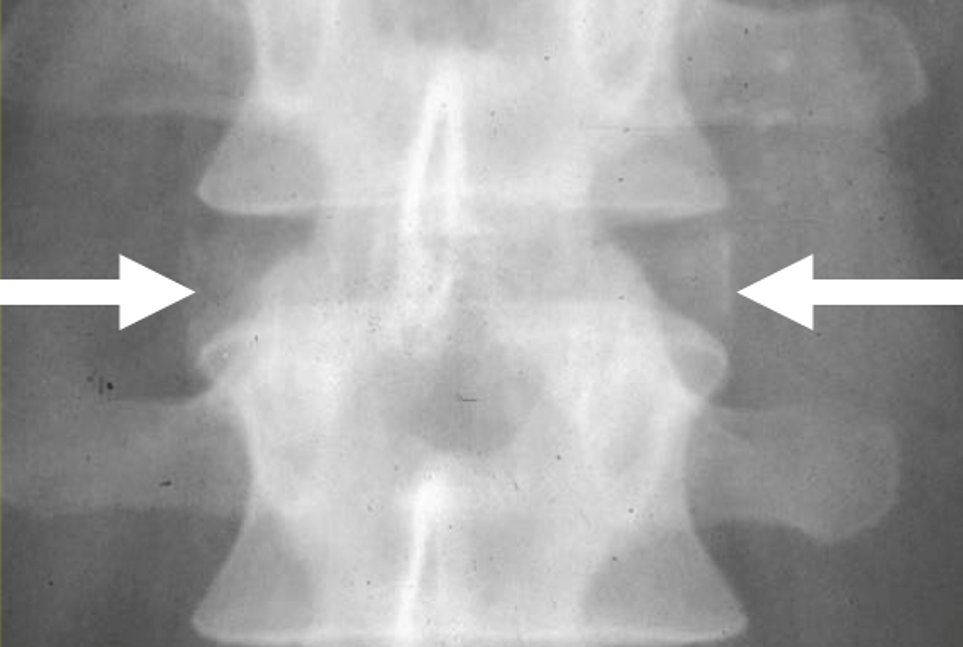
In alkaptonuria, a rare autosomal recessive metabolic disorder that results in deposition of excess homogentisic acid in many tissues due to a deficiency in the enzyme homogentisic oxidase, spinal abnormalities include diffuse vertebral osteopenia, intervertebral disc space narrowing, discal vacuum phenomena, and “wafer-like” intervertebral disc calcifications (Figure 18) related to the deposition of BCP crystals, predominantly affecting the lumbar spine.24,37
Figure 18: BCP crystal deposition in the intervertebral discs in a patient with alkaptonuria. (18A) Coronal and (18B) sagittal reconstructed CT images of the lumbar spine demonstrate the characteristic segmental fusion, diffuse vertebral osteopenia, intervertebral disc space narrowing, discal vacuum phenomena, and “wafer-like” intervertebral disc calcifications related to the deposition of BCP crystals.
Intervertebral disc calcification can also be seen in pediatric patients with the cervical spine more frequently than other levels. Patients may be asymptomatic or may develop symptoms including neck pain and low-grade fever. Severe cases may produce muscle spasms, torticollis, or even dysphagia if there is anterior protrusion of a calcified cervical disc. The precise etiology of intervertebral disc calcifications in children is unclear, with potential causes including idiopathic crystal deposition, an inflammatory or metabolic process, or trauma.38
Tumoral Calcinosis
The term tumoral calcinosis typically refers to mass-like calcific collections that accumulate in the soft tissues, especially around large joints (Figure 19). Radiographs show well- or ill-defined heavily calcified masses that are characterized by low signal intensity on T1-weighted images, and often heterogenous signal intensity on fluid sensitive images. Additionally, with CT or MRI, fluid-calcium levels can be seen secondary to intralesional calcium sedimentation (milk of calcium).39
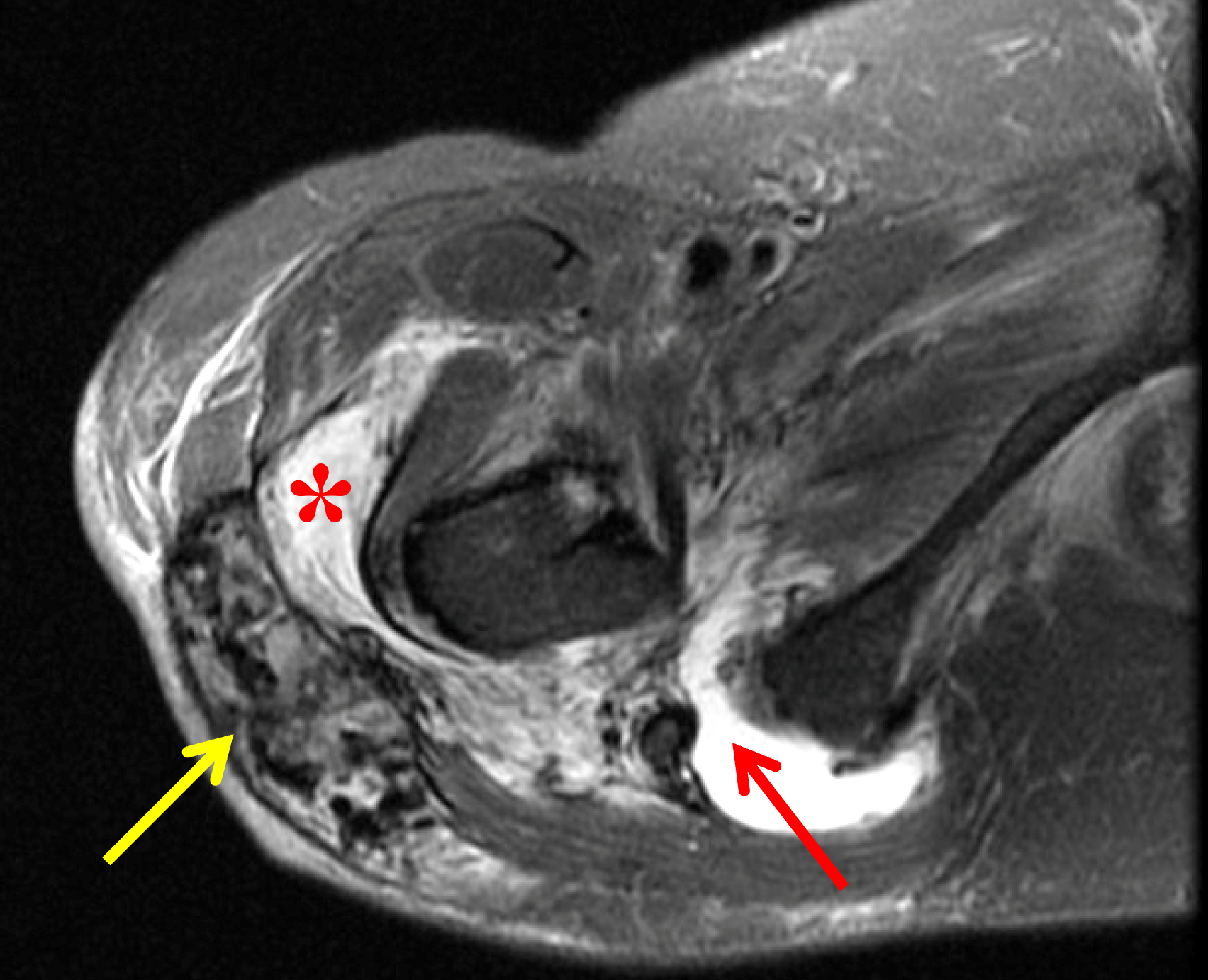
Figure 19: Tumoral calcinosis around both hips in a patient with chronic renal disease. (19A) AP radiograph of the pelvis demonstrates bulky soft tissue calcifications around both greater trochanters (arrows). Surgical clips in the right pelvis are related to a renal transplant. (19B) Axial fat-suppressed, T2-weighted image of the right hip demonstrates a heterogeneous mass containing low signal intensity calcifications (yellow arrow) in the lateral soft tissues and greater trochanteric bursitis (asterisk). Note the proximal hamstring tendon avulsion (red arrow).
There are several causes of tumoral calcinosis related to BCP crystal deposition including two rare, inherited conditions. Primary normophosphatemic tumoral calcinosis is inherited in an autosomal recessive manner and is thought to be secondary to a mutation of the SAMD9 gene. Affected patients have normal serum phosphorus and calcium levels. Primary hyperphosphatemic tumoral calcinosis is also thought to be familial, related to mutations in the Ga1NAc transferase 3 and the fibroblast growth factor 23 genes. These patients have elevated levels of serum phosphorus but normal serum calcium levels.39,40
Far more commonly, tumoral calcinosis relates to an underlying disorder, often metabolic conditions with abnormal calcium-phosphate metabolism, including primary and secondary hyperparathyroidism, collagen vascular diseases (especially scleroderma or systemic sclerosis), milk-alkali syndrome, hypervitaminosis D, and sarcoidosis.39 In patients with hyperparathyroidism, especially secondary hyperparathyroidism related to chronic renal disease (i.e., renal osteodystrophy), there is pathologic deposition of calcium hydroxyapatite, calcium pyrophosphate dihydrate, and calcium oxalate crystals within the soft tissues of the body, including around articulations (Figures 19 and 20) but also in organs such as the heart, stomach, kidneys, and lungs. Crystal deposition can also result in bursitis and synovitis.41
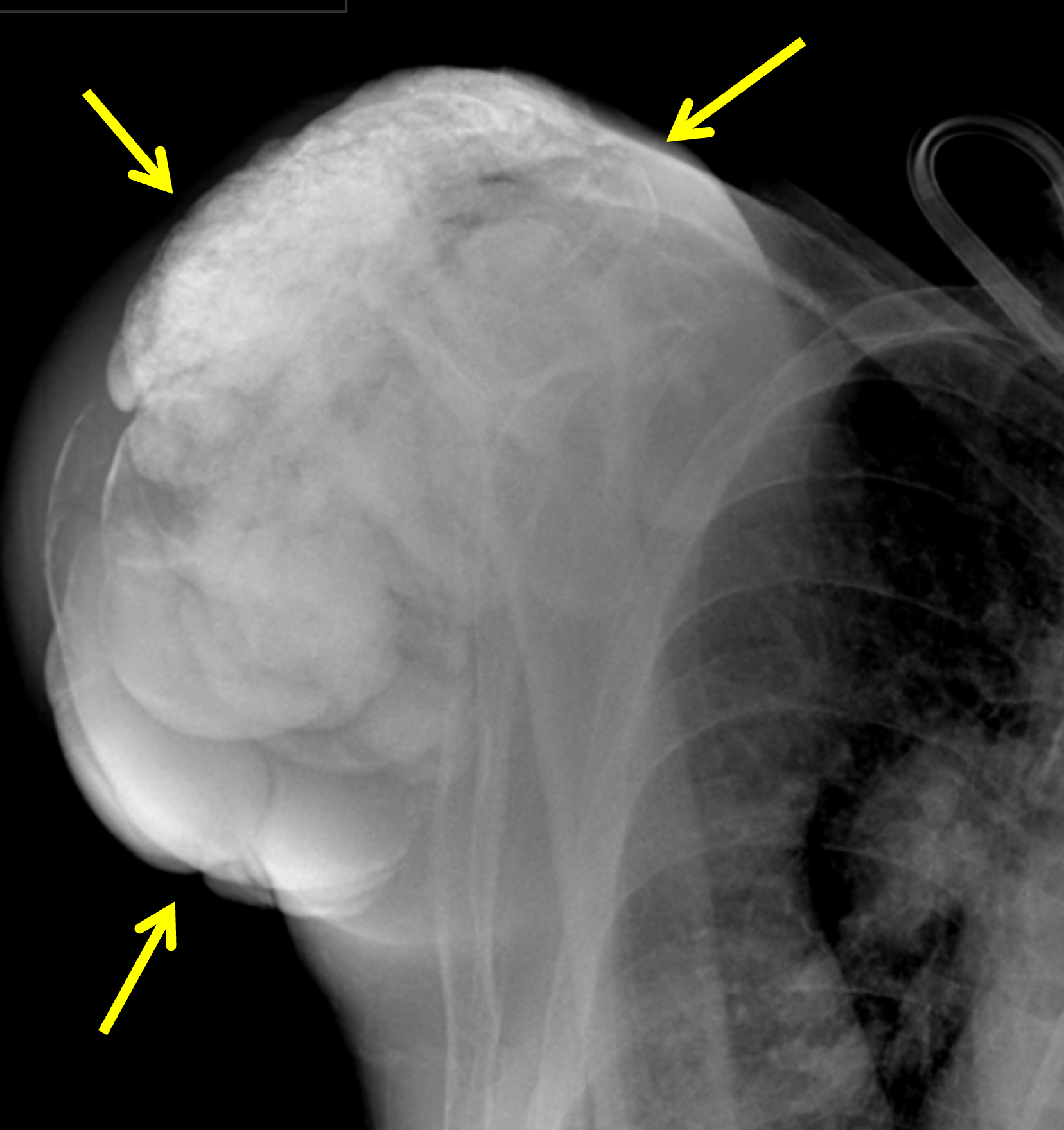
Figure 20: Multifocal tumoral calcinosis in a patient with chronic renal disease. (20A) AP radiograph of the right shoulder and (20B) PA chest radiograph demonstrate tumoral calcinosis about the right shoulder (yellow arrows) and left elbow (white arrow). (20C) PA radiograph of the right wrist and hand demonstrates tumoral calcinosis in the soft tissues of the distal forearm and wrist (arrows). (20D) PA radiograph of the left wrist and hand demonstrates tumoral calcinosis at multiple sites, including the ulnar aspect of the distal forearm (yellow arrow), the fifth metacarpophalangeal joint (red arrow), and the index finger distal phalanx (white arrow). Images courtesy of S. Pe, MD.
In patients with scleroderma, amorphous calcifications can occur in multiple skeletal sites (Figure 21), most commonly in the soft tissues of the finger tips, often with accompanying erosions in the distal phalangeal tufts.24 In patients with systemic sclerosis, tumoral calcinosis is commonly encountered in the spine, especially the cervical spine, sometimes resulting in spinal cord compression or spinal cord injury, as well as compressive peripheral neuropathy.42
Figure 21: Tumoral calcinosis around the proximal radius in a patient with scleroderma. (21A) AP radiograph shows tumoral calcinosis surrounding the proximal radius (arrow). (21B) Coronal T1-weighted image demonstrates low signal intensity bulky calcification in the soft tissues (arrow).
Calcified tumor-like soft tissue deposits may also be seen in cases of CPPD crystal deposition disease, termed tophaceous pseudogout. Although somewhat variable in location, predilection for the temporomandibular joint or paraspinal tissues has been emphasized in some reports.43 Examples of tophaceous pseudogout along with a discussion of differential diagnostic considerations are further detailed in the June 2024 Web Clinic. In MSU crystal deposition disease (gout), tophi of variable size can be encountered and are sometimes accompanied by calcification, simulating the appearance of tumoral calcinosis related to BCP crystal deposition disease.43
Large Joint Destructive Arthropathy
Destructive arthropathy of the glenohumeral joint of the shoulder is a well-recognized entity, often associated with chronic rotator cuff tendon tearing (Figure 22). Two competing etiologic theories have evolved. The first theory was introduced decades ago and championed by Charles Neer and his colleagues at New York Orthopedic Hospital/Columbia-Presbyterian Medical Center. These investigators described findings in 26 patients who all had massive rotator cuff tendon tears secondary to wear and tear rather than trauma, with associated worsening shoulder pain that was exacerbated with activity. They characterized the advanced glenohumeral joint pathology in these patients as cuff-tear arthropathy, believing that the initial event was massive tendon tearing with subsequent atrophy of the glenohumeral articular cartilage, osteopenia, and superior glenohumeral subluxation, leading to a rapidly developing and destructive arthropathy, narrowing of the acromiohumeral distance, and acromial changes that included “spurs” and bone erosion.44 In short, the malalignment related to the torn tendons of the rotator cuff led to the progressive disorganization of the glenohumeral joint and joint instability.44
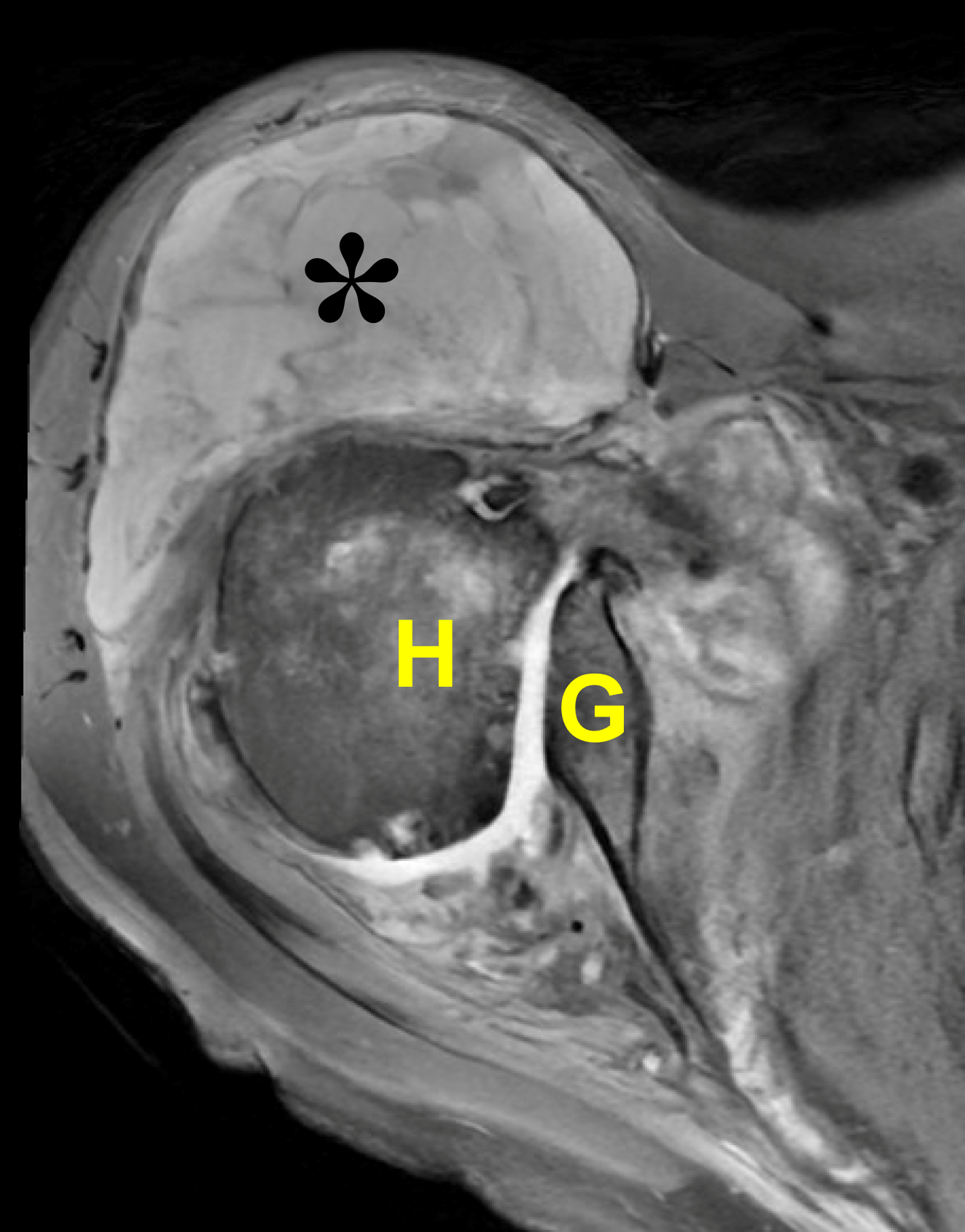
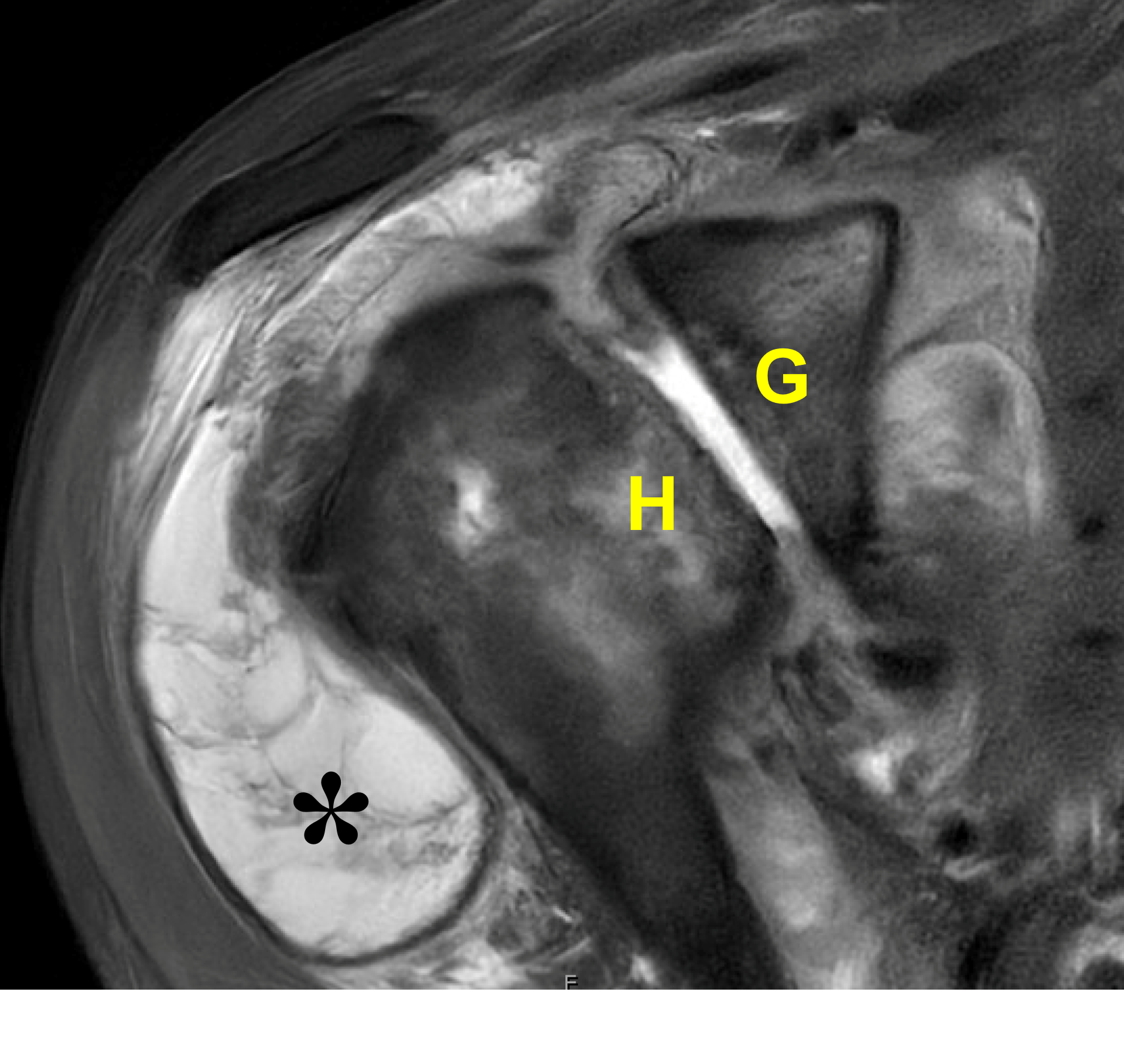
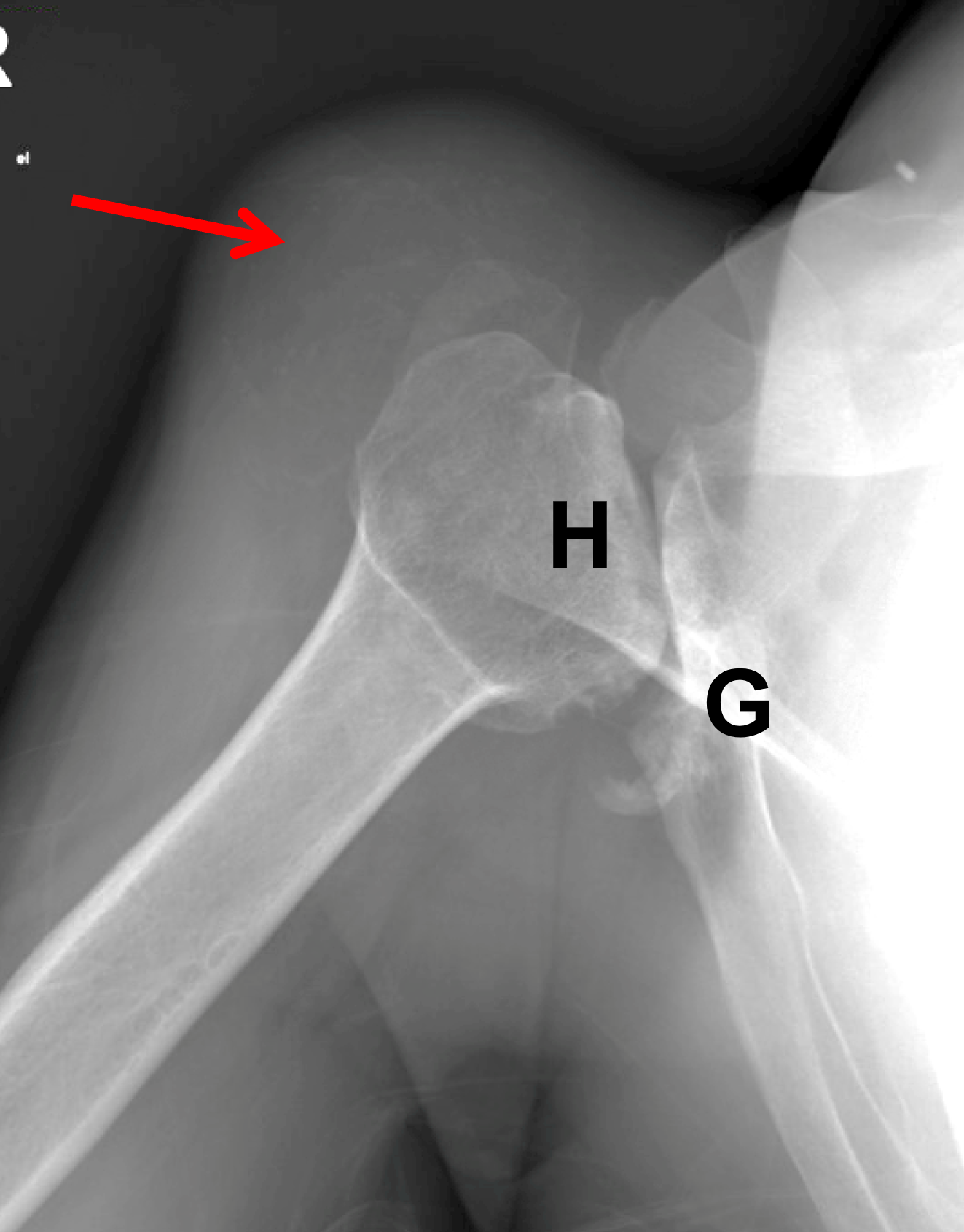
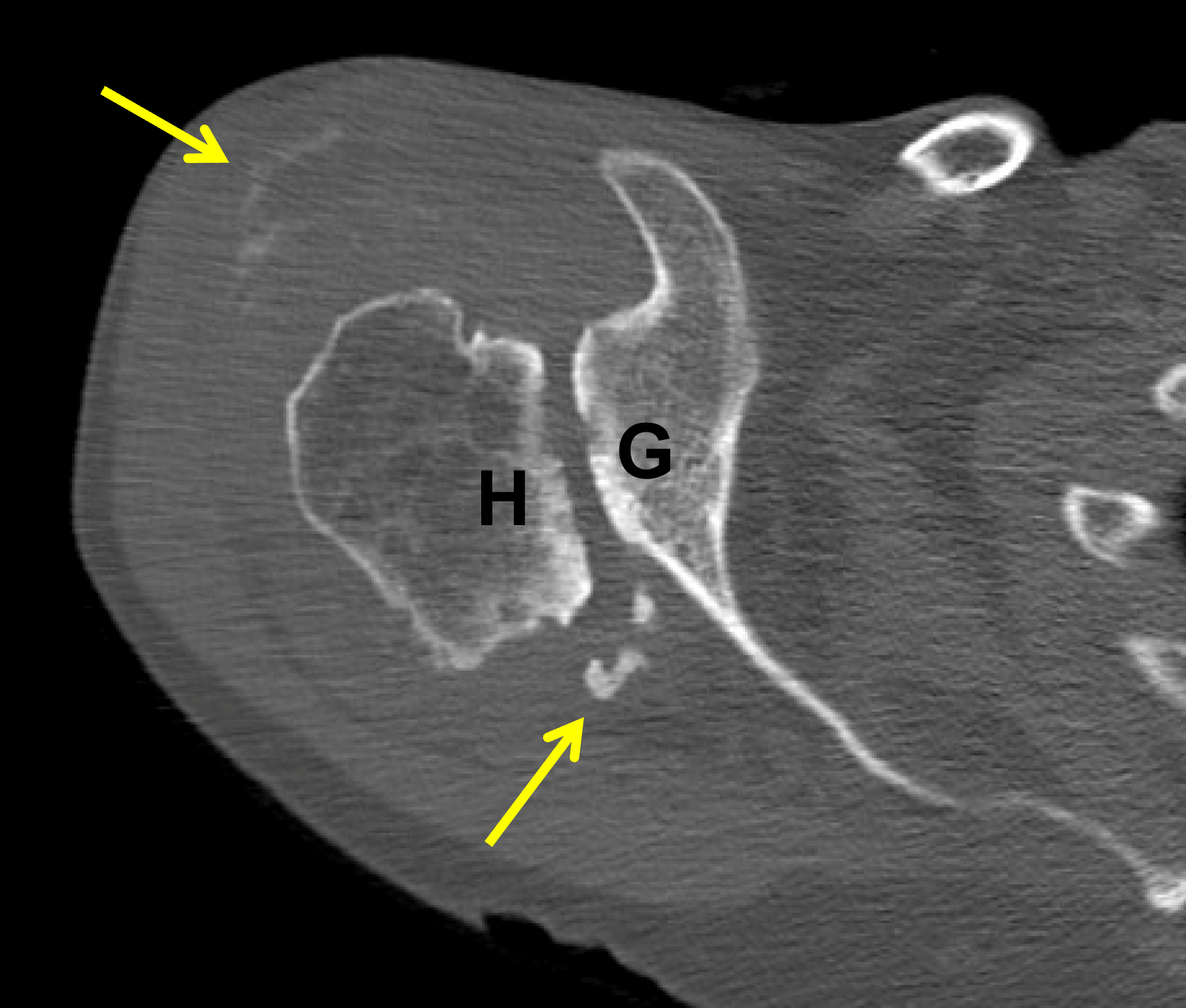
Figure 22: Destructive shoulder arthropathy. (22A) Axial fat-suppressed PD-weighted and (22B) coronal fat-suppressed, T2-weighted images demonstrate advanced destructive arthropathy of the glenohumeral joint, associated with flattening of the humeral head (H), bone loss and acquired retroversion of the glenoid (G), a moderate-size joint effusion, widespread full-thickness cartilage loss, and severe distention of the subacromial-subdeltoid bursa containing synovitis and debris (asterisk). A massive rotator cuff tear is present. (22C) Radiograph and (22D) axial CT show the same findings with severe cartilage loss, flattening of the humeral head (H), bone loss and retroversion of the glenoid (G) and severe bursitis (red arrow). The CT also shows calcifications in the posterior glenohumeral joint and subdeltoid bursa (yellow arrows). Primary diagnostic considerations include Milwaukee shoulder syndrome and cuff tear arthropathy. Similar findings can also be seen due to neuropathic osteoarthropathy, severe posttraumatic arthropathy, chronic infection, or osteoarthrosis related to maldevelopment of the glenoid.
A second competing theory as to the association of rotator cuff tendon tears and destructive arthropathy of the glenohumeral joint was later introduced in 1981 by Dr. Daniel McCarty and his rheumatology colleagues at the University of Wisconsin utilizing the term Milwaukee shoulder after studying 4 elderly women with dysfunctional shoulder joints who were found to have BCP crystals (including carbonate-substituted hydroxyapatite, octacalcium phosphate, and, to a lesser extent, tricalcium phosphate) within the synovial fluid of the involved glenohumeral joints.45 In this initial study, massive rotator cuff tears were present in 7 of the 8 shoulders, and microspheroids that contained calcium hydroxyapatite crystals were present in 12 of 13 synovial fluid samples, regardless of the presence or absence of crystal deposits on radiographs. These investigators proposed that calcium hydroxyapatite crystals develop in diseased joint capsules, synovial tissue, and/or articular cartilage and, when the crystals are released into the synovial fluid, combine with tissue matrix to form microspheroids. After these microspheroids are taken up by the macrophage-like synovial cells, collagenase, neutral protease, and prostaglandin E2 are activated and released. These enzymes and prostaglandin E2 subsequently attack the rotator cuff tendons and other periarticular soft tissues and, in a vicious cycle, induce the release of more crystals, leading to repetition of the entire process that culminates in a destructive arthropathy.46,47,48 In short, tearing of the tendons of the rotator cuff was not the initial event. Rather, it was the accumulation of BCP crystals that led to the release of substances that then attacked the rotator cuff tendons with the later appearance of a destructive arthropathy of the glenohumeral joint.
In a follow-up study in which Halverson, Carrera, and McCarty analyzed 15 additional cases of Milwaukee shoulder, microspheroids were found in the synovial fluid of 13 of the 15 patients, and further analysis of these microspheroids was positive for crystals (CPPD, BCP, or a combination of both). The BCP crystals predominantly consisted of carbonate-substituted hydroxyapatite, but also octacalcium phosphate, and, in one case, tricalcium phosphate. Of note, collagenase activity was detected in some but not all these samples. Based on this follow-up study, collagenase activity was deemed to be the least consistent feature of Milwaukee shoulder syndrome. McCarty and colleagues further investigated symptomatic knee pain in these patients and found that of the 5 synovial fluid samples that were analyzed, one contained BCP crystals, another contained a combination of BCP and CPPD crystals, and the remaining three did not have identifiable crystals. When they analyzed 11 knee radiographs from this set of patients, they found that lateral femorotibial compartment narrowing was just as frequent as medial femorotibial compartment narrowing. Additionally, the frequency of coincidental symptomatic knee pain in patients with Milwaukee shoulder syndrome (53%) and this observation of lateral femorotibial compartment involvement raised the possibility that this syndrome represented a single manifestation of a generalized metabolic condition that can predispose patients to large joint degenerative-like changes. One additional observation was that while the presence of BCP crystals within the synovial fluid correlated with the radiographic severity of the arthropathy, the presence of CPPD crystals correlated only with the patient’s age.47 In 1984, Dr. Paul Dieppe and his colleagues published similar findings in the shoulders and other joints but described it more broadly as “apatite associated destructive arthritis”.49
Although both theories as to the cause of the association of rotator cuff tendon disruption and glenohumeral joint abnormalities are still popular, it is interesting that a similar appearance of a rapidly destructive joint process has been described in other articulations. In an analysis of 27 cases, Dr. S. Zehava Rosenberg and her colleagues described an entity that was termed rapid destructive osteoarthritis of the hip, which mimicked other entities including septic arthritis, seronegative spondyloarthropathy, and neuropathic osteoarthropathy. As radiographic and pathologic examination revealed findings that were compatible with osteoarthrosis, these investigators believed that this entity was a rapidly developing variant of joint degeneration.50 CPPD crystals were noted in only one case, and there was no evidence of infection or neurologic deficit. In a subsequent analysis of 23 patients who presented with hip pain and were found to have rapidly destructive hip disease, Dr. Gerhard Bock and his colleagues proposed that owing to their documentation of involvement of other joints in patients afflicted by this pathologic process of the hip, these manifestations were sequelae of a broader process. Additionally, only one of their 23 patients was found to have CPPD crystals in the synovial fluid. The presence or absence of calcium hydroxyapatite crystals in the synovial fluid samples was not assessed.51 Additional reports of a rapidly destructive arthropathy of the hip have suggested other causative factors including widespread osteonecrosis of the femoral head,52 subtle deficiencies of neurologic function compatible with neuropathic osteoarthropathy, intraarticular corticosteroid injections, and subchondral insufficiency fractures of the femoral head. To our knowledge, a clear link of BCP crystal deposition as the cause of rapidly destructive arthropathy of the hip or other joints has not been definitively established.
In the knee joint, BCP crystal deposition disease predominantly affects the lateral femorotibial compartment, in contrast to primary osteoarthrosis which is typically most pronounced in the medial femorotibial compartment, especially in men, and CPPD arthropathy (i.e., pyrophosphate arthropathy), in which patellofemoral compartment involvement is characteristic.24,47 BCP crystal deposition in the menisci usually manifests as focal deposits, as demonstrated by Dr. Daniel McCarty and his colleagues in their work analyzing pathological calcifications in human cartilage via radiographs and crystallography.53
Treatment
Treatment of BCP crystal deposition disease depends upon the pattern of the disease. For calcific periarthritis, therapy is influenced by the stage and phase of the disease. When symptomatic, non-steroidal anti-inflammatory drugs (NSAIDs) and physiotherapy have been employed.54 Additional noninvasive therapies include ultrasound therapy, acetic acid iontophoresis,55 and extracorporeal shock wave therapy.56,57,58,59 Radiologists can also play a role in treatment of symptomatic deposits by performing ultrasound-guided barbotage (Figure 23).55,60
Figure 23: Ultrasound-guided percutaneous treatment of BCP crystal deposition disease in the shoulder. (23A) Axillary projection radiograph demonstrates BCP crystal deposition (arrow) adjacent to the greater tuberosity. (23B) Color doppler demonstrates marked hypervascularity around the focus of BCP crystal deposition, which was localized to the subacromial/subdeltoid bursa, just anterior to the long head of the biceps tendon at the upper portion of the biceps pulley. (23C) Cine clip demonstrates ultrasound-guided needle placement within the calcifications. (23D) Cine clip obtained during ultrasound-guided aspiration and lidocaine lavage of the calcifications. (23E) Video demonstrating the aspiration technique used during this procedure. The aspirated crystals can be seen within the syringe.
23C: Cine clip demonstrates ultrasound-guided needle placement within the calcifications.
23D: Cine clip obtained during ultrasound-guided aspiration and lidocaine lavage of the calcifications.
23E: Video demonstrating the aspiration technique used during this procedure. The aspirated crystals can be seen within the syringe.
In patients with refractory BCP calcific periarthritis, surgery can be considered (Figure 24), although there is debate regarding the role of subacromial decompression and repair of rotator cuff tendon defects that arise secondary to arthroscopic debridement of the BCP crystals.61,62
Figure 24: Subscapularis BCP crystal deposition managed by surgical debridement. (24A) Grashey radiograph of the right shoulder demonstrates BCP crystal deposition within the subscapularis tendon at its footprint adjacent to the lesser tuberosity (arrow). (24B) Pre-operative axial fat-suppressed PD-weighted image demonstrates a low signal intensity focus corresponding to BCP crystal deposition within the distal subscapularis tendon (arrow). (24C) Post-operative axial fat-suppressed PD-weighted image demonstrates interval resection subscapularis BCP crystal deposition. Note the attenuated tendon (arrow) where the calcification was resected representing an intermediate-grade, partial-thickness tear. The finding is nonspecific given the interval surgery.
Surgical excision (with or without accompanying medical management) is the primary method of treatment for tumoral calcinosis and can improve quality of life. However, complications in cases of incomplete excision can include recurrent mass-like deposits that typically grow more rapidly than the initial deposit.39
In patients with large joint destructive arthropathy, supportive treatment with rest and NSAIDs is typically initially prescribed. Physiotherapy is crucial in maintaining muscle strength and normal range of motion. Arthrocentesis can be therapeutic for patients who have large joint effusions. Severe cases of large joint destructive arthropathy may eventually warrant total joint arthroplasty, in the absence of operative contraindications.63
Conclusion
BCP crystal deposition disease is one of the three major categories of crystal deposition diseases. This Web Clinic details BCP crystal deposition as a precursor to osteoarthrosis, patterns of crystal deposition disease, and potential treatment options. BCP crystal deposition disease can have a variety of imaging manifestations on MRI that can mimic infection, malignancy, and other crystal deposition diseases. Knowledge of the common sites of BCP crystal deposition and correlation of the MRI findings with radiographs and/or CT examinations, can help guide the radiologist toward establishing the correct diagnosis.
References
- Yan JF, Qin WP, Xiao BC, Wan QQ, Tay FR, Niu LN, et al. Pathological calcification in osteoarthritis: an outcome or a disease initiator? Biol Rev Camb Philos Soc. 2020;95(4):960–985. doi:10.1111/brv.12595 ↩
- McCarthy GM, Cheung HS. Point: Hydroxyapatite crystal deposition is intimately involved in the pathogenesis and progression of human osteoarthritis. Curr Rheumatol Rep. 2009;11(2):141–147. doi:10.1007/s11926-009-0020-6 ↩
- Corr EM, Cunningham CC, Helbert L, McCarthy GM, Dunne A. Osteoarthritis-associated basic calcium phosphate crystals activate membrane proximal kinases in human innate immune cells. Arthritis Res Ther. 2017;19(1):23. doi:10.1186/s13075-017-1225-0 ↩
- Yavorskyy A, Hernandez-Santana A, McCarthy G, McMahon G. Detection of calcium phosphate crystals in the joint fluid of patients with osteoarthritis – analytical approaches and challenges. Analyst. 2008;133(3):302–318. doi:10.1039/b716791a ↩
- Grynpas MD, Omelon S. Transient precursor strategy or very small biological apatite crystals? Bone. 2007;41(2):162–164. doi:10.1016/j.bone.2007.04.176 ↩
- Scotchford CA, Ali SY. Magnesium whitlockite deposition in articular cartilage: a study of 80 specimens from 70 patients. Ann Rheum Dis. 1995;54(5):339–344. doi:10.1136/ard.54.5.339 ↩
- Fuerst M, Bertrand J, Lammers L, Dreier R, Echtermeyer F, Nitschke Y, et al. Calcification of articular cartilage in human osteoarthritis. Arthritis Rheum. 2009;60(9):2694–2703. doi:10.1002/art.24774 ↩
- Fuerst M, Niggemeyer O, Lammers L, Schafer F, Lohmann C, Ruther W. Articular cartilage mineralization in osteoarthritis of the hip. BMC Musculoskelet Disord. 2009;10166. doi:10.1186/1471-2474-10-166 ↩
- Nalbant S, Martinez JA, Kitumnuaypong T, Clayburne G, Sieck M, Schumacher HR, Jr. Synovial fluid features and their relations to osteoarthritis severity: new findings from sequential studies. Osteoarthritis Cartilage. 2003;11(1):50–54. doi:10.1053/joca.2002.0861 ↩
- Rachow JW, Ryan LM, McCarty DJ, Halverson PC. Synovial fluid inorganic pyrophosphate concentration and nucleotide pyrophosphohydrolase activity in basic calcium phosphate deposition arthropathy and Milwaukee shoulder syndrome. Arthritis Rheum. 1988;31(3):408–413. doi:10.1002/art.1780310313 ↩
- Shoji K. {Alizarin red S staining of calcium compound crystals in synovial fluid}. Nihon Seikeigeka Gakkai Zasshi. 1993;67(4):201–210. ↩
- Pascual E, Jovaní V. Synovial fluid analysis. Best Pract Res Clin Rheumatol. 2005;19(3):371–386. doi:10.1016/j.berh.2005.01.004 ↩
- Suvorova EI, Buffat PA. Electron diffraction and high resolution transmission electron microscopy in the characterization of calcium phosphate precipitation from aqueous solutions under biomineralization conditions. Eur Cell Mater. 2001;127–42. doi:10.22203/ecm.v001a04 ↩
- Smith DA, Connell SD, Robinson C, Kirkham J. Chemical force microscopy: applications in surface characterisation of natural hydroxyapatite. Analytica Chimica Acta. 2003;479(1):39–57. doi:10.1016/S0003-2670(02)01374-0 ↩
- Hornez JC, Chai F, Monchau F, Blanchemain N, Descamps M, Hildebrand HF. Biological and physico-chemical assessment of hydroxyapatite (HA) with different porosity. Biomol Eng. 2007;24(5):505–509. doi:10.1016/j.bioeng.2007.08.015 ↩
- Eysel HH, Jackson M, Nikulin A, Somorjai RL, Thomson GTD, Mantsch HH. A novel diagnostic test for arthritis: Multivariate analysis of infrared spectra of synovial fluid. Biospectroscopy. 1997;3(2):161–167. doi:10.1002/(SICI)1520-6343(1997)3:2<161::AID-BSPY9>3.0.CO;2-A ↩
- Ziegler CM, Kircher P, Hassfeld S. Analysis of temporomandibular joint synovial fluid using Fourier transform/infrared spectroscopy. J Oral Maxillofac Surg. 2002;60(11):1302–1306. doi:10.1053/joms.2002.35728 ↩
- Shaw RA, Eysel HH, Liu KZ, Mantsch HH. Infrared spectroscopic analysis of biomedical specimens using glass substrates. Anal Biochem. 1998;259(2):181–186. doi:10.1006/abio.1998.2654 ↩
- Shaw RA, Kotowich S, Eysel HH, Jackson M, Thomson GT, Mantsch HH. Arthritis diagnosis based upon the near-infrared spectrum of synovial fluid. Rheumatol Int. 1995;15(4):159–165. doi:10.1007/BF00301774 ↩
- Zaheer A, Lenkinski RE, Mahmood A, Jones AG, Cantley LC, Frangioni JV. In vivo near-infrared fluorescence imaging of osteoblastic activity. Nat Biotechnol. 2001;19(12):1148–1154. doi:10.1038/nbt1201-1148 ↩
- Zilberman Y, Kallai I, Gafni Y, Pelled G, Kossodo S, Yared W, et al. Fluorescence molecular tomography enables in vivo visualization and quantification of nonunion fracture repair induced by genetically engineered mesenchymal stem cells. J Orthop Res. 2008;26(4):522–530. doi:10.1002/jor.20518 ↩
- Swan A, Chapman B, Heap P, Seward H, Dieppe P. Submicroscopic crystals in osteoarthritic synovial fluids. Ann Rheum Dis. 1994;53(7):467–470. doi:10.1136/ard.53.7.467 ↩
- Calafiori AR, Di Marco G, Martino G, Marotta M. Preparation and characterization of calcium phosphate biomaterials. J Mater Sci Mater Med. 2007;18(12):2331–2338. doi:10.1007/s10856-007-3141-3 ↩
- Brower ACF, D.J. Arthritis in Black and White. 3rd Ed. Philadelphia: Elsevier Saunders; 2012. ↩
- Hamada J, Ono W, Tamai K, Saotome K, Hoshino T. Analysis of calcium deposits in calcific periarthritis. J Rheumatol. 2001;28(4):809–813. ↩
- Depalma AF, Kruper JS. Long-term study of shoulder joints afflicted with and treated for calcific tendinitis. Clin Orthop. 1961;2061–72. ↩
- Uhthoff HK, Loehr JW. Calcific Tendinopathy of the Rotator Cuff: Pathogenesis, Diagnosis, and Management. J Am Acad Orthop Surg. 1997;5(4):183–191. doi:10.5435/00124635-199707000-00001 ↩
- Uhthoff HK, Sarkar K, Maynard JA. Calcifying tendinitis: a new concept of its pathogenesis. Clin Orthop Relat Res. 1976(118):164–168. ↩
- Uhthoff HK, Sarkar K. Calcifying tendinitis. Baillieres Clin Rheumatol. 1989;3(3):567–581. doi:10.1016/s0950-3579(89)80009-3 ↩
- Goldman AB. Calcific tendinitis of the long head of the biceps brachii distal to the glenohumeral joint: plain film radiographic findings. AJR Am J Roentgenol. 1989;153(5):1011–1016. doi:10.2214/ajr.153.5.1011 ↩
- Alberts V, Laloo F, Leenknegt B. Tophaceous Gout at the Popliteal Sulcus. J Belg Soc Radiol. 2024;108(1):34. doi:10.5334/jbsr.3523 ↩
- Ring D, Vaccaro AR, Scuderi G, Pathria MN, Garfin SR. Acute calcific retropharyngeal tendinitis. Clinical presentation and pathological characterization. J Bone Joint Surg Am. 1994;76(11):1636–1642. doi:10.2106/00004623-199411000-00006 ↩
- Pennes DR, Ellis CN, Madison KC, Voorhees JJ, Martel W. Early skeletal hyperostoses secondary to 13-cis-retinoic acid. AJR Am J Roentgenol. 1984;142(5):979–983. doi:10.2214/ajr.142.5.979 ↩
- Hott JS, Feiz-Erfan I, Kenny K, Dickman CA. Surgical management of giant herniated thoracic discs: analysis of 20 cases. J Neurosurg Spine. 2005;3(3):191–197. doi:10.3171/spi.2005.3.3.0191 ↩
- Gong M, Liu G, Guan Q, Li L, Xing F, Xiang Z. Surgery for Giant Calcified Herniated Thoracic Discs: A Systematic Review. World Neurosurg. 2018;118109–117. doi:10.1016/j.wneu.2018.06.232 ↩
- Quraishi NA, Khurana A, Tsegaye MM, Boszczyk BM, Mehdian SM. Calcified giant thoracic disc herniations: considerations and treatment strategies. Eur Spine J. 2014;23 Suppl 1(Suppl 1):S76–83. doi:10.1007/s00586-014-3210-5 ↩
- Gil JA, Wawrzynski J, Waryasz GR. Orthopedic Manifestations of Ochronosis: Pathophysiology, Presentation, Diagnosis, and Management. Am J Med. 2016;129(5):536 e531–536. doi:10.1016/j.amjmed.2016.01.010 ↩
- Gerlach R, Zimmermann M, Kellermann S, Lietz R, Raabe A, Seifert V. Intervertebral disc calcification in childhood–a case report and review of the literature. Acta Neurochir (Wien). 2001;143(1):89–93. doi:10.1007/s007010170143 ↩
- King JJ, Brennan KB, Crawford EA, Fox EJ, Ogilvie CM. Surgical complications associated with extensive tumoral calcinosis. Am J Orthop (Belle Mead NJ). 2011;40(5):247–252. ↩
- Chefetz I, Ben Amitai D, Browning S, Skorecki K, Adir N, Thomas MG, et al. Normophosphatemic familial tumoral calcinosis is caused by deleterious mutations in SAMD9, encoding a TNF-alpha responsive protein. J Invest Dermatol. 2008;128(6):1423–1429. doi:10.1038/sj.jid.5701203 ↩
- Chang CY, Rosenthal DI, Mitchell DM, Handa A, Kattapuram SV, Huang AJ. Imaging Findings of Metabolic Bone Disease. Radiographics. 2016;36(6):1871–1887. doi:10.1148/rg.2016160004 ↩
- Avanoglu-Guler A, Campochiaro C, De Luca G, Hughes M, Tufan A, Green L, et al. Calcinosis in systemic sclerosis: An update on pathogenesis, related complications, and management: A heavy burden still waiting to be lifted off patients’ hands. Semin Arthritis Rheum. 2024;66152431. doi:10.1016/j.semarthrit.2024.152431 ↩
- Sadiq M, Nayak M, Farheen A, Digge V. An Unusual Case of Huge Tophaceous Pseudogout Mimicking as a Tumor-Like Lesion around the Ankle Joint: A Case Report and Literature Review. Case Rep Orthop. 2019;20199617184. doi:10.1155/2019/9617184 ↩
- Neer CS, 2nd, Craig EV, Fukuda H. Cuff-tear arthropathy. J Bone Joint Surg Am. 1983;65(9):1232–1244. ↩
- McCarty DJ. Milwaukee shoulder syndrome. Trans Am Clin Climatol Assoc. 1991;102271–283; discussion 283–274. ↩
- McCarty DJ, Halverson PB, Carrera GF, Brewer BJ, Kozin F. “Milwaukee shoulder”–association of microspheroids containing hydroxyapatite crystals, active collagenase, and neutral protease with rotator cuff defects. I. Clinical aspects. Arthritis Rheum. 1981;24(3):464–473. doi:10.1002/art.1780240303 ↩
- Halverson PB, Carrera GF, McCarty DJ. Milwaukee shoulder syndrome. Fifteen additional cases and a description of contributing factors. Arch Intern Med. 1990;150(3):677–682. doi:10.1001/archinte.150.3.677 ↩
- Halverson PB, Cheung HS, McCarty DJ, Garancis J, Mandel N. “Milwaukee shoulder”–association of microspheroids containing hydroxyapatite crystals, active collagenase, and neutral protease with rotator cuff defects. II. Synovial fluid studies. Arthritis Rheum. 1981;24(3):474–483. doi:10.1002/art.1780240304 ↩
- Dieppe PA, Doherty M, Macfarlane DG, Hutton CW, Bradfield JW, Watt I. Apatite associated destructive arthritis. Br J Rheumatol. 1984;23(2):84–91. doi:10.1093/rheumatology/23.2.84 ↩
- Rosenberg ZS, Shankman S, Steiner GC, Kastenbaum DK, Norman A, Lazansky MG. Rapid destructive osteoarthritis: clinical, radiographic, and pathologic features. Radiology. 1992;182(1):213–216. doi:10.1148/radiology.182.1.1727284 ↩
- Bock GW, Garcia A, Weisman MH, Major PA, Lyttle D, Haghighi P, et al. Rapidly destructive hip disease: clinical and imaging abnormalities. Radiology. 1993;186(2):461–466. doi:10.1148/radiology.186.2.8421751 ↩
- Ryu KN, Kim EJ, Yoo MC, Park YK, Sartoris DJ, Resnick D. Ischemic necrosis of the entire femoral head and rapidly destructive hip disease: potential causative relationship. Skeletal Radiol. 1997;26(3):143–149. doi:10.1007/s002560050210 ↩
- McCarty DJ, Jr., Hogan JM, Gatter RA, Grossman M. Studies on pathological calcifications in human cartilage. I. Prevalence and types of crystal deposits in the menisci of two hundred fifteen cadavera. J Bone Joint Surg Am. 1966;48(2):309–325. ↩
- Hongsmatip P, Cheng KY, Kim C, Lawrence DA, Rivera R, Smitaman E. Calcium hydroxyapatite deposition disease: Imaging features and presentations mimicking other pathologies. Eur J Radiol. 2019;120108653. doi:10.1016/j.ejrad.2019.108653 ↩
- Ebenbichler GR, Erdogmus CB, Resch KL, Funovics MA, Kainberger F, Barisani G, et al. Ultrasound therapy for calcific tendinitis of the shoulder. N Engl J Med. 1999;340(20):1533–1538. doi:10.1056/NEJM199905203402002 ↩
- Loew M, Daecke W, Kusnierczak D, Rahmanzadeh M, Ewerbeck V. Shock-wave therapy is effective for chronic calcifying tendinitis of the shoulder. J Bone Joint Surg Br. 1999;81(5):863–867. doi:10.1302/0301-620x.81b5.9374 ↩
- Peters J, Luboldt W, Schwarz W, Jacobi V, Herzog C, Vogl TJ. Extracorporeal shock wave therapy in calcific tendinitis of the shoulder. Skeletal Radiol. 2004;33(12):712–718. doi:10.1007/s00256-004-0849-8 ↩
- Rompe JD, Zoellner J, Nafe B. Shock wave therapy versus conventional surgery in the treatment of calcifying tendinitis of the shoulder. Clin Orthop Relat Res. 2001(387):72–82. doi:10.1097/00003086-200106000-00010 ↩
- Wang CJ, Yang KD, Wang FS, Chen HH, Wang JW. Shock wave therapy for calcific tendinitis of the shoulder: a prospective clinical study with two-year follow-up. Am J Sports Med. 2003;31(3):425–430. doi:10.1177/03635465030310031701 ↩
- de Witte PB, Selten JW, Navas A, Nagels J, Visser CP, Nelissen RG, et al. Calcific tendinitis of the rotator cuff: a randomized controlled trial of ultrasound-guided needling and lavage versus subacromial corticosteroids. Am J Sports Med. 2013;41(7):1665–1673. doi:10.1177/0363546513487066 ↩
- Seil R, Litzenburger H, Kohn D, Rupp S. Arthroscopic treatment of chronically painful calcifying tendinitis of the supraspinatus tendon. Arthroscopy. 2006;22(5):521–527. doi:10.1016/j.arthro.2006.01.012 ↩
- Suzuki K, Potts A, Anakwenze O, Singh A. Calcific tendinitis of the rotator cuff: management options. J Am Acad Orthop Surg. 2014;22(11):707–717. doi:10.5435/JAAOS-22-11-707 ↩
- Nadarajah CV, Weichert I. Milwaukee shoulder syndrome. Case Rep Rheumatol. 2014;2014458708. doi:10.1155/2014/458708 ↩