Clinical History:
A 35 year-old obese female was status post a recent microdiscectomy at L4-5 with resolution of radicular symptoms. Three days post-operatively she developed headaches and episodic recurrent left radicular symptoms. Her headache was positional, and palpation of a subcutaneous bulge deep to the surgical wound closure exacerbated the headache. There was no wound drainage or signs or symptoms of infection. (1a) T2-weighted sagittal, (1b) T2-weighted axial, (1c) T1-weighted sagittal, (1d) and a T1-weighted axial image following contrast administration from a post-operative MRI of the Lumbar Spine are displayed below.
What are the findings? What is your diagnosis?
Findings
Figure 2:
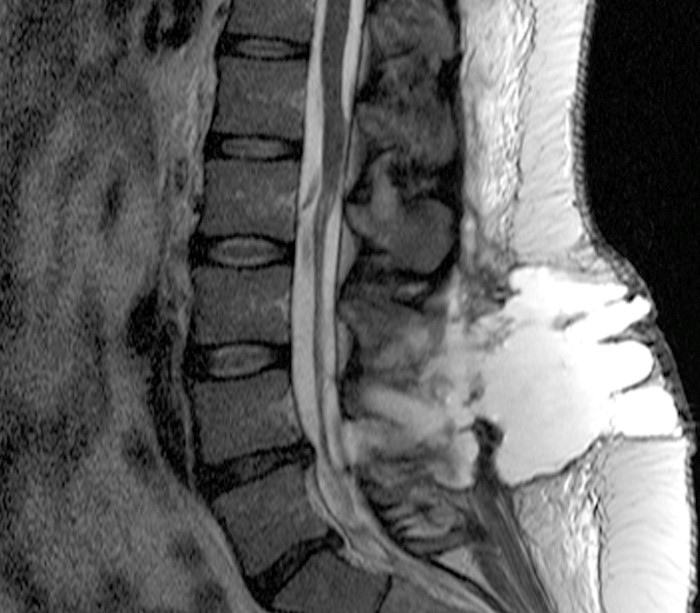
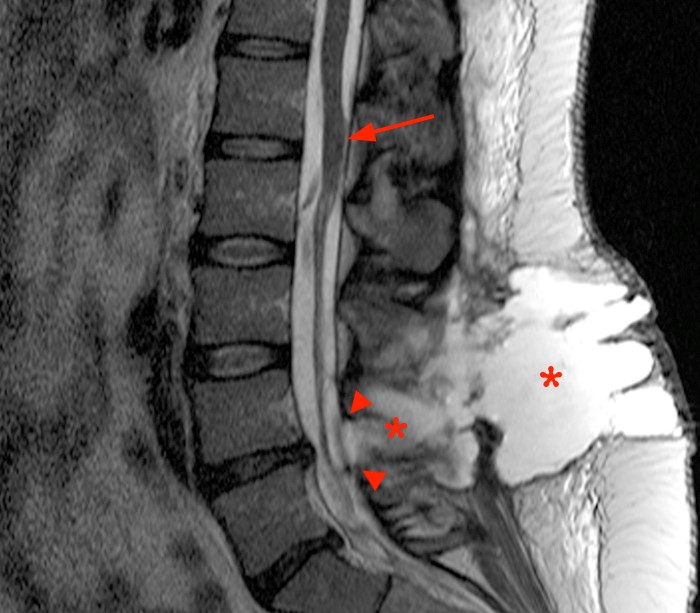
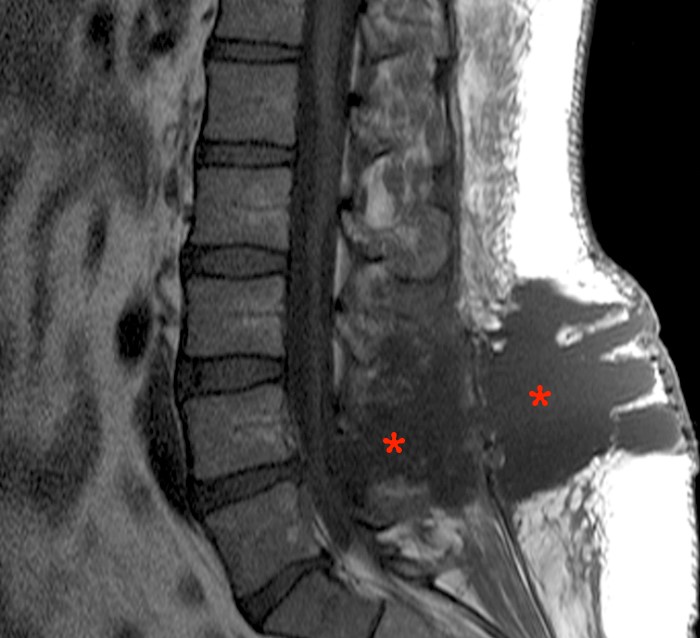
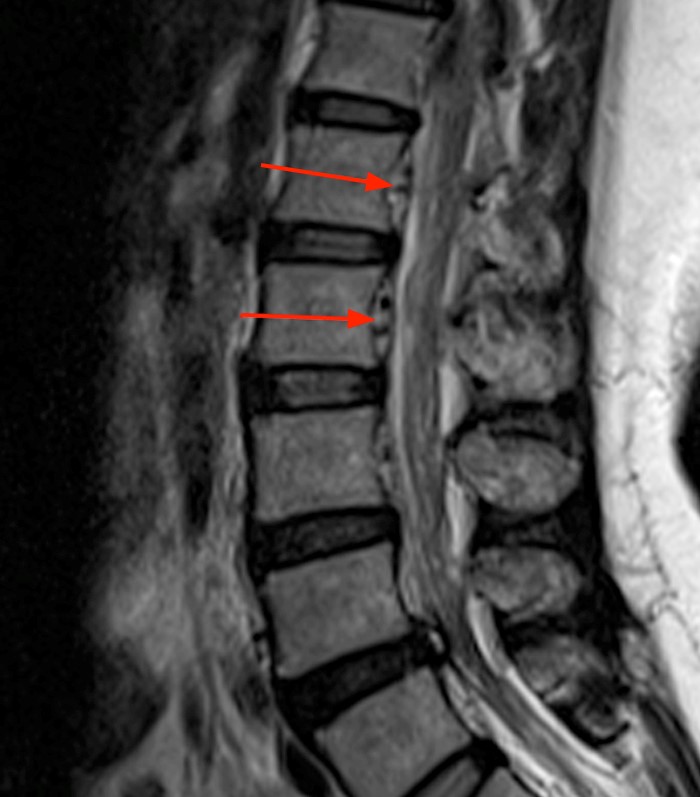
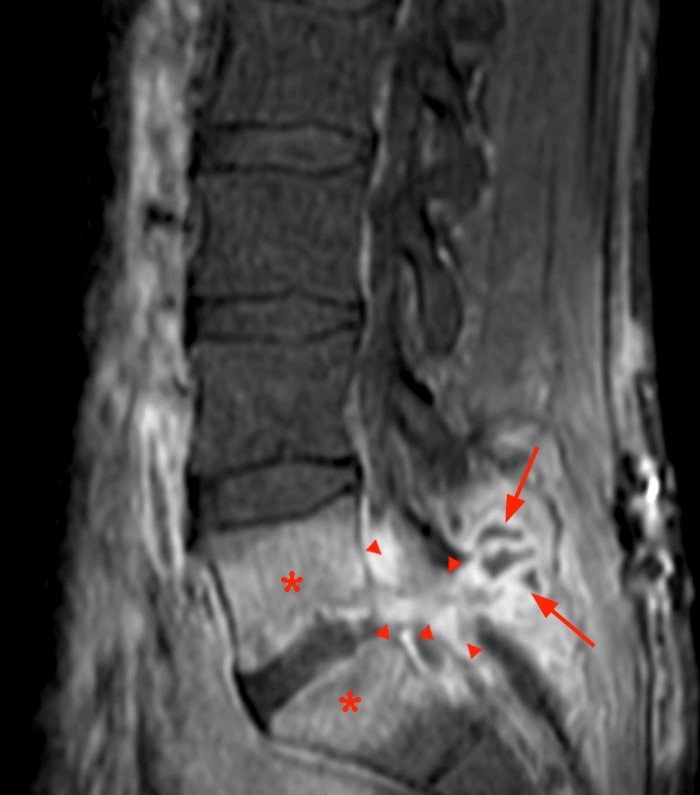
2a: The T2-weighted sagittal image demonstrates a large dorsal extraspinal fluid collection (asterisks) extending from the laminotomy (arrowheads) through the deep muscular fascia into subcutaneous fat. Note lack of signs of discitis or osteomyelitis. There is an unusual appearance to the distal cord and conus which appear displaced (arrow).
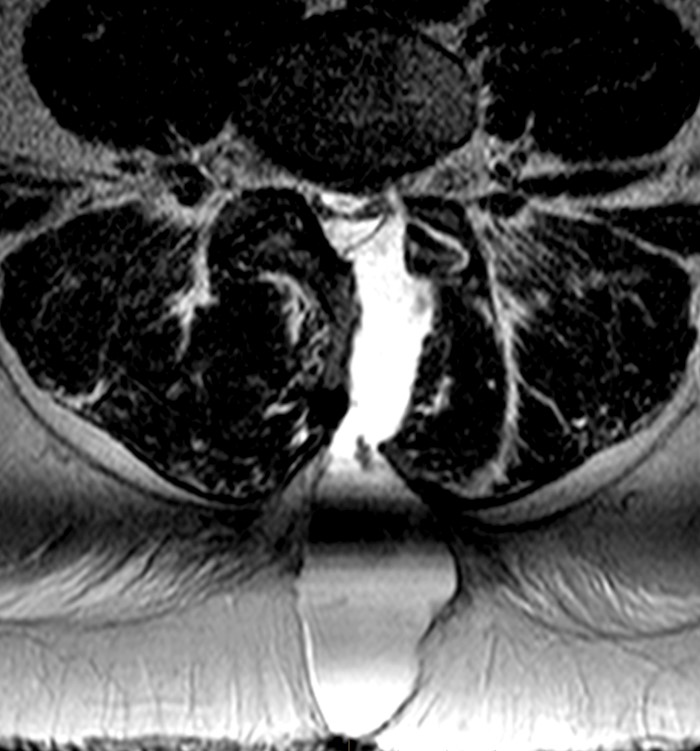
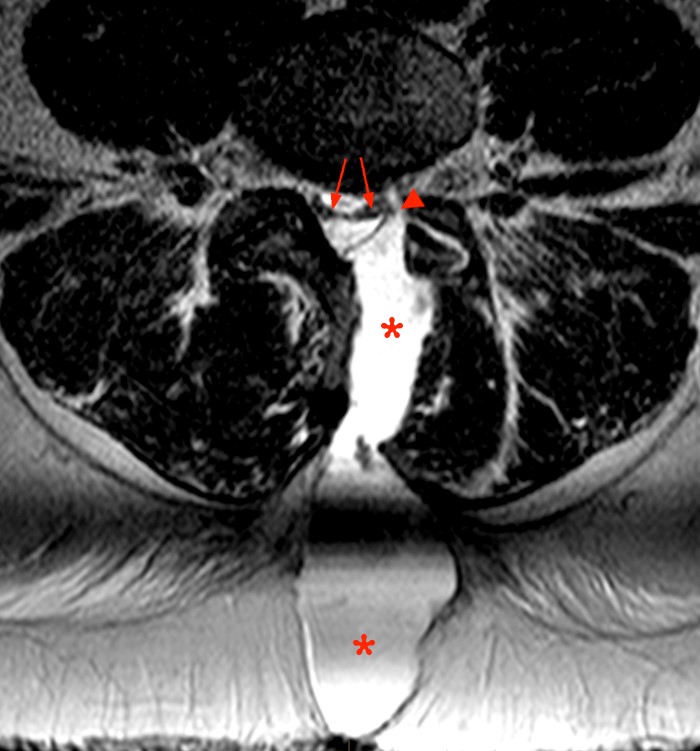
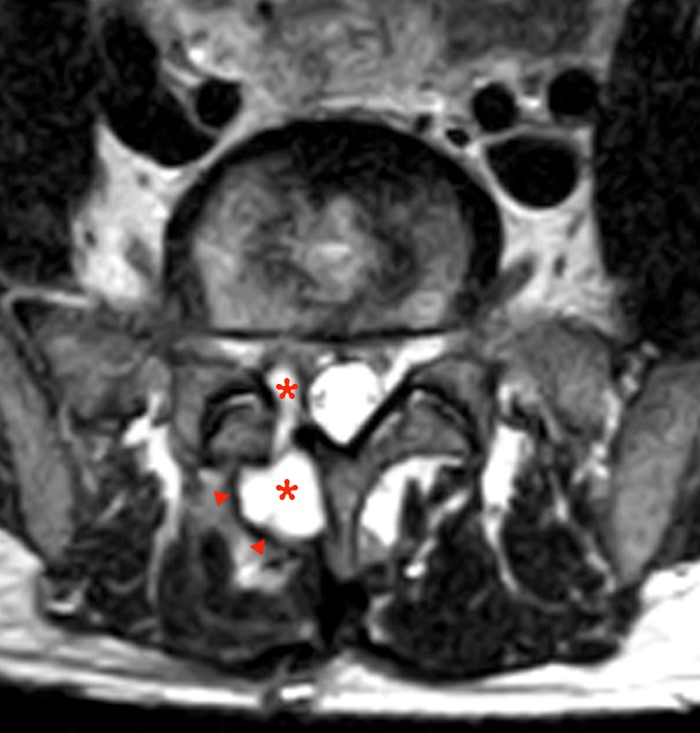
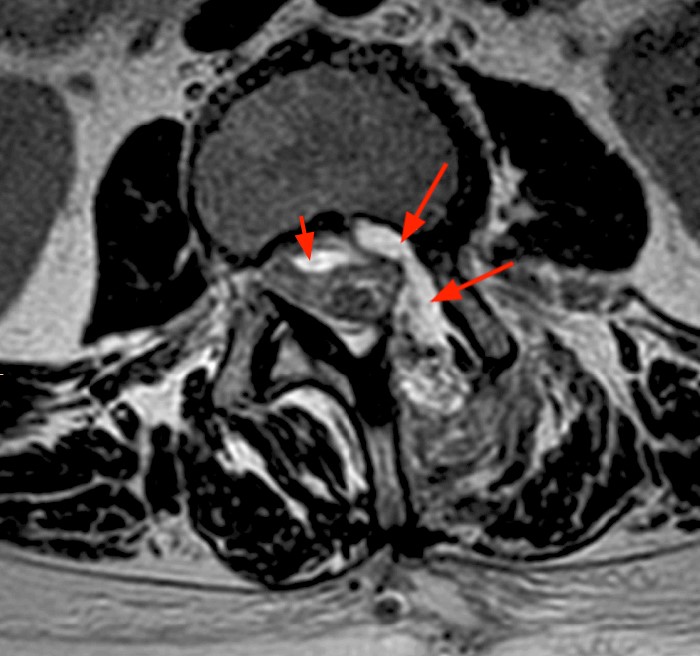
2b: The T2-weighted axial image better demonstrates the bony surgical defect (arrowhead). A large fluid collection (asterisks) extends dorsally from the left lateral recess into the spinal canal and deforms the thecal sac compatible with a pseudomeningocele. Note that there is not edema or inflammation in the dorsal extraspinal musculature beyond that expected for the type of surgical approach. This axial image better demonstrates a deformity of the cauda equina (arrows) compatible with a subdural CSF fluid collection. This unusual constellation of findings is due to leakage and retention of CSF between the dura and the arachnoid.
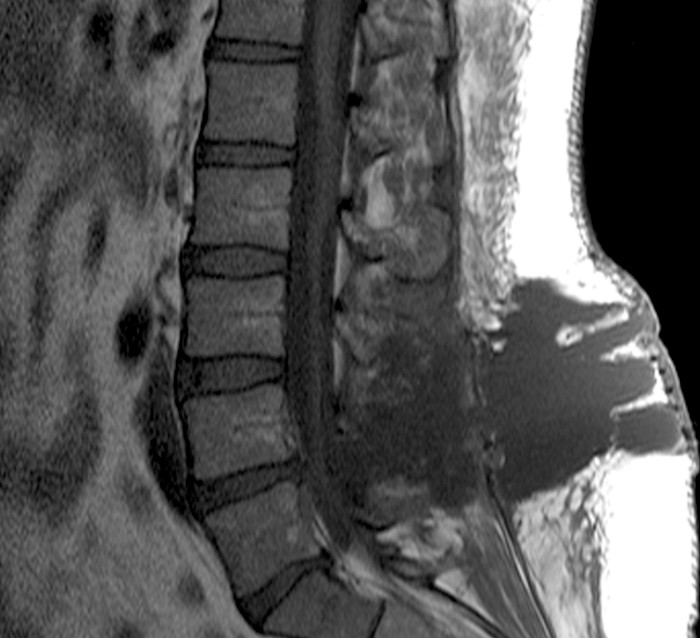
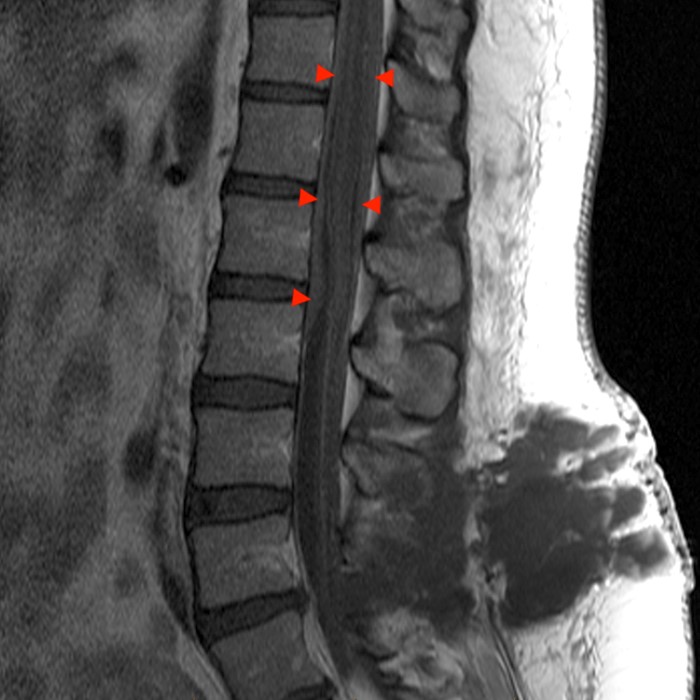
2c: A T1-weighted sagittal image confirms that the fluid/material in question (asterisks) is of CSF signal intensity.
2d: The post-contrast axial image demonstrates continued relative homogeneity and lack of enhancement of this fluid or muscles. The distal cauda equina is “pancaked” (arrowheads) between CSF collections in the subdural space. The sharp margins of the dura (arrows) help properly identify the location of the collection which is otherwise not visible since it is of CSF intensity.
Diagnosis
Post-operative pseudomeningocele.
Follow-up Clinical Information: Given the patient’s obesity, the size of the pseudomeningocele and partial dehiscence of the deep layers of the wound, operative management was elected rather than more conservative treatment. At repeat surgery, CSF seepage was noted through a 2 mm dural disruption. A tiny arachnoid bleb was present extending into this dural rent. This was repaired using 4 interrupted Nurolon sutures and morcellation of a small amount of surrounding fibrous and muscle tissue. Post-operatively the patient’s symptoms completely resolved and follow-up MRI one month later demonstrated complete resolution of the pseudomeningocele and subdural CSF collection.
Introduction
A dural/arachnoid tear during surgery can lead to extravasation of CSF resulting in a fistula or collection of CSF referred to as a pseudomeningocele or meningeal pseudocyst. Symptomatic post-operative pseudomeningoceles are rare complications of spine surgery as most dural rents resolve spontaneously. Clinically, patients may present with a positional headache as a result of intracranial hypotension, non-postural headache, cutaneous CSF drainage at the wound, a palpable subcutaneous bulge, recurrent radicular symptoms and/or low back pain.1,2,3,4 The leak can be directly demonstrated on a myelogram but most commonly is diagnosed with MRI.5,6
Pathogenesis and Incidence
Pseudomeningoceles may be classified as iatrogenic, traumatic or congenital. By far the most common cause is iatrogenic following inadvertent dural injury during surgery. Weinstein reported that pseudomeningocele was the most common complication following surgery for spinal stenosis.7 If a dural tear occurs without an arachnoid tear, an arachnoid bleb may herniate through the dural tear creating a purely arachnoid meningocele without seeping CSF. Usually however, both the dura and arachnoid are injured which can result in a fistula which may cutaneously decompress through the surgical wound. A pseudocyst forms by CSF following the surgical track and expanding surrounding connective tissue with or without developing a fibrous capsule. More rarely the leak may track into the epidural space of the spinal canal or even into the subdural space as in the introductory case. The incidence of durotomy during lumbar spine surgery is reported between 0.3% and 13% but the incidence of fistula or pseudomeningocele is much lower, reported between .07% and 0.3%. The true incidence is unknown as it is felt that most cases resolve spontaneously.1 ,2, 8 The risk of durotomy and pseudomeningocele is increased in a number of conditions and situations including: prior surgery, complicated surgery especially decompression needed for ossification of the posterior longitudinal ligament, prior irradiation, dural scar, congenital malformations including dural ectasia and spinal dysraphism, spinal stenosis, administration of anti-adhesive gels to prevent scarring, intradural surgery and administration of epidural triamcinolone during lumbar decompression.9, 10, 11, 12, 13, 14, 15
The exact cause for persistence of a CSF leak is uncertain with some theories advocating for a ball-valve mechanism and others advocating for volume of the CSF leak depending on the size of the dural tear.1, 2, 16 The ball valve effect, size of the dural defect, CSF pressure, resistance of surrounding soft tissues, neural herniation into the defect, and weakness and bulk of extraspinal musculature all influence the persistence and size of a pseudomeningocele.
Clinical and imaging features
Most patients with a post-operative CSF leaks are unrecognized and asymptomatic and undergo spontaneous resolution.1, 2, 8, 12, 17 Patients can manifest a wide variety of signs and symptoms and presentation can be delayed up to years.18 A cutaneous watery discharge is a prominent feature of a CSF fistula with or without a pseudomeningocele (Figure 4). This drainage may leave a pinkish stain on clothes or bed linens with a light brown halo referred to as the “halo sign.” 1, 2 Less than 1cc of this watery discharge is needed to test it for the presence of beta-2 transferrin, a protein only present in the central nervous system.19 A fluctuant subcutaneous bulge may enlarge with sneezing, coughing or Valsalva maneuver. Palpation of this bulge or the surgical site may be painful and may exacerbate headache. The patient’s headache may or may not be a typical postural headache. Postural headaches are thought to occur from low intracranial and spinal fluid pressure resulting in traction on pain sensitive structures. However, the headache may not be associated with signs of intracranial hypotension, and may not necessarily be related to the size of the durotomy. In headaches such as this, the etiology is uncertain but may be related to venous dilatation to compensate for reduced CSF volume. Recurrent low back pain and or radicular symptoms are common.3 Nerve roots may be entrapped in the dural defect, adhere to the arachnoid opening or become entrapped in the pseudomeningocele.4 There are rare reports of spinal cord herniation16 and a pseudomeningocele has even been described as a cause of chronic meningitis.20
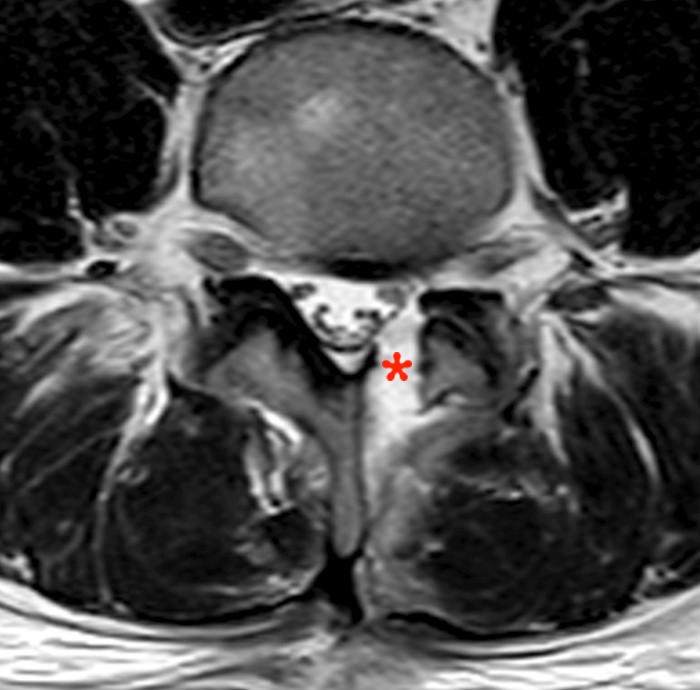
Figure 4:
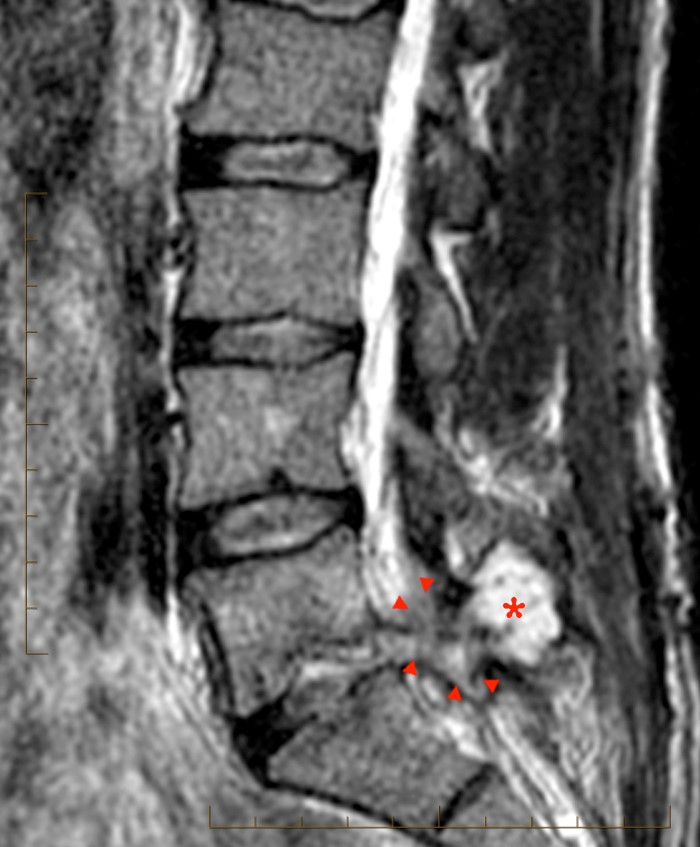
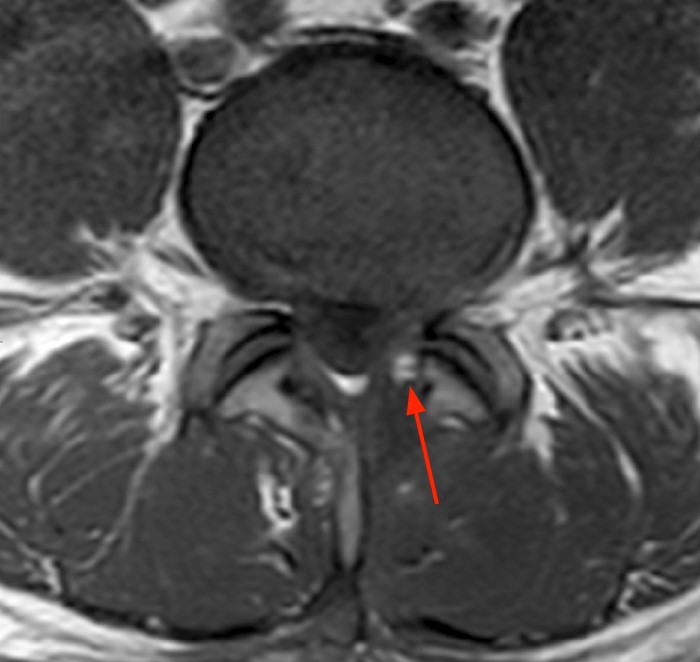
In another patient, a T2-weighted axial image at the laminotomy site reveals a small well-circumscribed fluid collection (asterisk) which exhibits CSF-like signal on all pulse sequences (other images not shown) in this patient with a clinically proven CSF fistula. The collection did not obviously extend superficial to the deep muscular fascia on imaging. Without clinical context this fluid collection cannot be differentiated from a small post-operative seroma.
MRI is the study of choice to examine possible pseudomeningoceles in a post-operative patient particularly when fusion hardware is not in place. MR typically reveals a variable sized CSF intensity fluid collection along the surgical course which may or may not be contained by the deep muscular fascia. This fluid is of homogeneous CSF signal intensity on all pulse sequences including diffusion.6 In the postoperative lumbar spine, the fluid most often extends dorsally from the lateral recess at a nerve root sleeve through the laminotomy. CSF pulsations or the presence of metal hardware may alter its appearance. Usually there is not an enhancing margin or muscle edema except to the degree expected in a recent post-operative condition. A CSF leak may lead to engorgement/enlargement of the anterior epidural venous plexus (Figure 5). Dural thickening and enhancement can be seen in patients with symptomatic intracranial hypotension (Figure 6).
Figure 6:
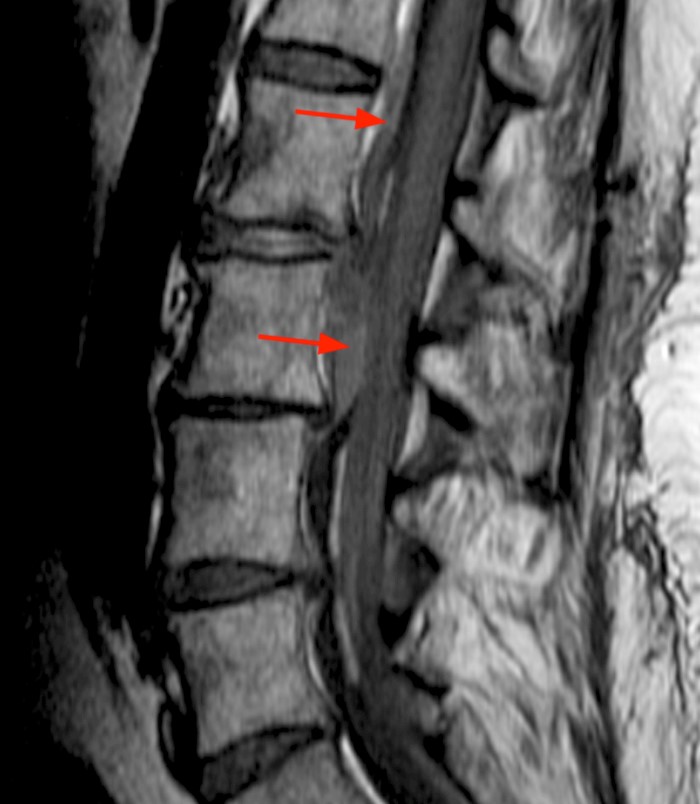
A post-contrast T1-weighted sagittal image from our reference case demonstrates dural thickening and enhancement (arrowheads) which could mimic infectious meningitis, arachnoiditis and subdural empyema without the proper clinical context. All of these findings completely resolved on follow-up MRI.
Correlation with clinical information is imperative in the diagnosis of a post-operative pseudomeningocele as imaging features are shared with a seroma but may also share some features with evolving liquefied hematoma, and to a lesser extent, abscess (Figures 7,8). On MR images, hematoma and abscess both tend to have more complex signal, are inhomogeneous, have thicker enhancing margins or capsule, and in the case of abscess likely accompanying discitis and/or osteomyelitis. A hematoma should “bloom” on T2* (GRE) sequences. Patients with pseudomeningocele typically do not have fever or other signs of infection unless the pseudocyst is secondarily infected.
Myelography, particularly post-myelographic CT is advantageous in demonstrating the actual CSF leak but is usually not performed today in the proper clinical context. While radionuclide myelography can demonstrate the leak, this type of study is generally reserved for cases of occult CSF leaks associated with idiopathic intracranial hypotension or oto/rhinorrhea.
Figure 7:
7a: Post-operative infection with epidural phlegmon and extra-spinal abscess. A T2-weighted axial in a post microdiscectomy patient with recurrent pain and fever. Fluid-like material (asterisks) extends from the lateral recess into the spinal canal through the laminar surgical defect into a small dorsal extra-spinal collection with a relatively thick margin (arrowheads). This could be a pseudomeningocele on this image alone.
7b: On this T2-weighted sagittal image, however, the epidural material in the spinal canal (arrowheads) is not of CSF signal intensity and the extra-spinal material (asterisk) is not homogeneous like CSF.
7c: The post contrast T1-weighted sagittal image with fat suppression reveals signs of osteomyelitis (asterisks), epidural enhancing phlegmon (arrowheads) and a heterogeneous irregularly-enhancing dorsal extraspinal soft tissue abscess (arrows).
Figure 8:
8a: Post-operative hemorrhage. A T2-weighted axial image in a patient who was treated for incidental durotomy during a microdiscectomy. Epidural CSF-like collection is seen in the left lateral recess extending through the laminotomy defect (long arrows). This could mimic a pseudomeningocele on this image alone especially given the history but the fluid is not entirely homogeneous. A second focus of signal disturbance is seen in the anterior aspect of the spinal canal which is subsequently seen to be in the subdural space (short arrow).
8b: A corresponding T1-weighted sagittal midline image demonstrates the pathology is much more complex in signal than CSF (arrows). The findings are compatible with epidural and subdural hematoma. The patient did not have signs or symptoms to indicate an abscess or empyema.
Treatment and Management
Pseudomeningoceles discovered following surgical closure can be treated conservatively or surgically but trends are towards definitive surgical management. Treatment alternatives include: Bed rest in the Trendelenburg position, over-sewing of the incision when a fistula is present, epidural blood patch, and closed subarachnoid drain placement. 1, 2, 6, 12, 17, 21, 22, 23
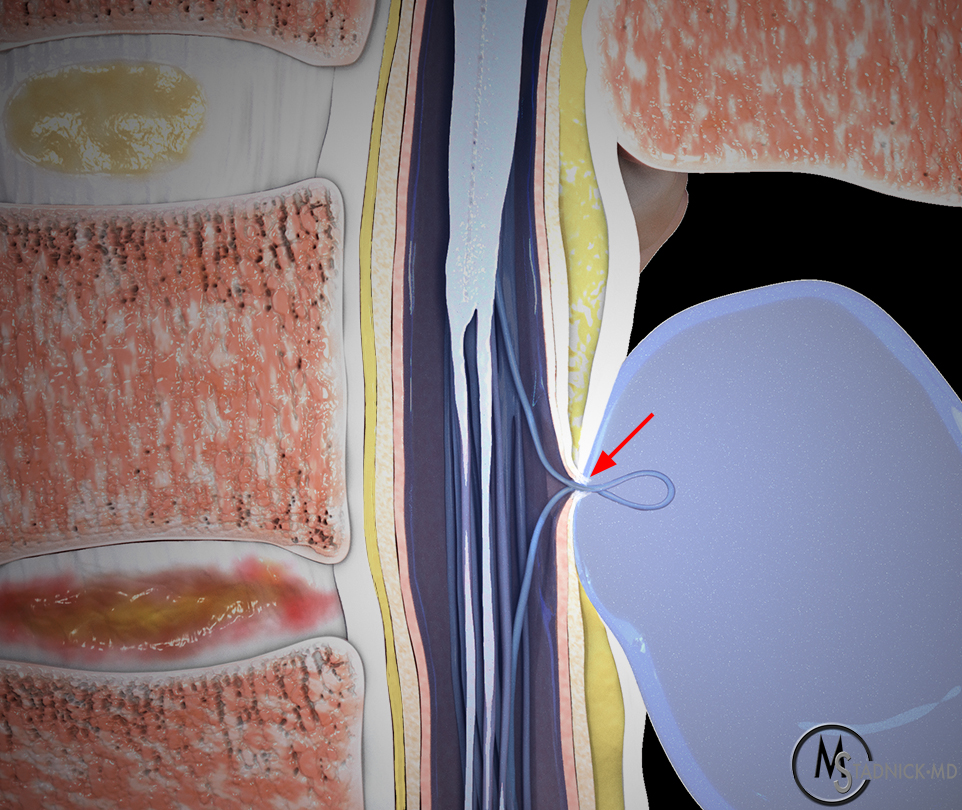
Surgical repair is the definitive treatment and from a practical point of view is often advocated for cases diagnosed by radiologists as these patients tend to be symptomatic. The laminotomy site can be widened if needed to provide greater access to the durotomy and allow better observation of any adherent nerves or bony shards. Primary closure is with fine interrupted monofilament sutures. To assure a watertight seal this is augmented by various adjuvant agents such as DuraSeal or DuraGen, or procedures such as placement of a cadaveric dura, fat or fascial patch or graft using morcellated adjacent extraspinal soft tissues (Figure 9). 1, 2, 12, 17, 21, 24, 25, 26 Rarely, a large defect may require use of a myofascial flap.27
Conclusion
Symptomatic post-operative pseudomeningoceles or fistulae are rare complications of spine surgery. Clinically, patients may present with a variety of signs or symptoms ranging from recurrent radicular symptoms to wound drainage to a positional headache. This complication is most commonly diagnosed with MRI but clinical correlation and careful evaluation of MR imaging findings are imperative as a post-operative pseudomeningocele can have similar features to a post-operative seroma, hematoma, or abscess.
References
- Couture D and Branch CL. Spinal pseudomeningoceles and CSF fistulas. Neurosurg Focus 2003; 15: e6. ↩
- Hawk MW, Kim KD. Review of spinal pseudomeningoceles and cerebrospinal fluid fistulas. Neurosurg Focus 2000; 9: e5. ↩
- Aldrete JA, Ghaly R. Postlaminectomy pseudomeningocele. An unsuspected cause of low back pain. Reg Anesth 1996;20:75-9 ↩
- Hadani M, Findler G, Knoler N, et al. Entrapped lumbar nerve root in pseudomeningocele after laminectomy: report of three cases. Neurosurgery 1986;19:405-07 ↩
- Phillips CD, Kaptain GJ, Razack N. Depiction of a post-operative pseudomeningocele on digital subtraction myelography. AJNR Amer J Neurorad. 2002;23 (2):337-8. ↩
- Jain NK, Dao K, Ortiz AO. Radiologic Evaluation and Management of Postoperative Spine Paraspinal Fluid Collections. Neuroimaging Clinics of North America 2014; 24:2, 375-389.A post-operative CSF fluid collection can have similar imaging features to a post-operative seroma, hematoma, or abscess. ↩
- Weinstein JN, Tosteson TD, Lurie JD, et al. Surgical versus Non Operative Treatment for Lumbar Spinal Stenosis Four-Year Results of the Spine Outcomes Research Trial (SPORT) Spine 2010;35(14):1329-38. ↩
- Solomon P, Sekharappa V, Krishnan V, et al. Spontaneous resolution of postoperative lumbar pseudomeningoceles: A report of four cases. Indian J Orthop 2013; 47(4): 417-21. ↩
- Murphy ME, Kerezoudis P, Ali Alvi M, et al(2017) Risk factors for dural tears: a study of elective spine surgery. Neurological Research 39:2, 97-106. . Online publication date: 1-Feb-2017. ↩
- Scaduto AA, Gamradt SC, Yu WD, et al. Perioperative Complications of Threaded Cylindrical Lumbar Interbody Fusion Devices. J Spinal Disord Tech. 2003; 16(6):502-7. ↩
- Stolke D, Sollman WP, Seifert V. Intra-and postoperative complications in lumbar surgery. Spine. 1989; 14:56-9. ↩
- Wang JC, Bohlman HH, Riew KD. Dural tears secondary to operations on the lumbar spine. Management and results after a two year minimum follow up in eighty-eight patients. J Bone Joint Surg Am 1998; 80:1728-32. ↩
- Stone JG, Bergmann LL, Takamori R, et al. Giant pseudomeningocele causing urinary obstruction in a patient with Marfan syndrome. J Neurosurg Spine 2015; 23:77-80. ↩
- Sellin JN, Vedantam A, Luerssen TG, et al. Safety of epidural triamcinolone acetonide use during lumbar decompression surgery in pediatric patients: an association with delayed pseudomeningocele formation. J Neurosurg Pediatr 2016; 17:667-71. ↩
- Seung-Jae Hyun, Hong-Ki Kim, Ki-Jeong Kim, et al. (2016) Posterior Trans-Dural Repair of Iatrogenic Spinal Cord Herniation after Resection of Ossification of Posterior Longitudinal Ligament. Asian Spine Journal 2016; 10 (2): 355-59. ↩
- Cobb C, Ehni G. Herniation of the spinal cord into an iatrogenic meningocele. Case report. J Neurosurg 1973; 39:533-36. ↩
- Bosacco SJ, Gardner MJ, Guille JT. Evaluation and treatment of dural tears in lumbar spine surgery: a review. Clin Orthop 2001; 389: 238-47. ↩
- Miller PR, Elder FW Jr. Meningeal pseudocysts following laminectomy. Report of ten cases. J Bone Joint Surg (Am) 1968; 50:268-76. ↩
- Reisinger PW, Hochstrasser K. The diagnosis of CSF fistulae on the basis of detection of beta2-transferrin by polyacrylamide gel electrophoresis and immunoblotting. J Clin Chem Clin Biochem 1989; 27: 169-72. ↩
- Koo J, Adamson R, Wagner FC. A new cause of chronic meningitis: infected lumbar pseudomeningocele. Am J Med 1989; 86:103-4. ↩
- Eismont FJ, Wiesel SW, Rothman RH. Treatment of dural tears associated with spinal surgery. J Bone Joint Surg Am 1981; 63: 1132-36. ↩
- Fridley JS, Jea A, Glover CD, et al. Symptomatic postsurgical cerebrospinal fluid leak treated by aspiration and epidural blood patch under ultrasound guidance. J Neurosurg Pediatr 2013; 11:87-90. ↩
- Cornman-Homonoff J, Schweitzer A, Chazen J. (2016) CT-guided epidural blood patch for treatment of CSF leak and pseudomeningocele following tethered cord release in a 3-year-old. Clinical Imaging 2016; 40:6, 1191-1194. ↩
- Kim K, Wright NM. (2011) Polyethylene Glycol Hydrogel Spinal Sealant (DuraSeal Spinal Sealant) as an Adjunct to Sutured Dural Repair in the Spine. Spine 2011; 36:23, 1906-1912. ↩
- Khan MH, Rihn J, Steele G, et al. Postoperative management protocol for incidental dural tears during degenerative lumbar spine surgery: a review of 3, 183 consecutive degenerative lumbar cases. Spine 2006; 31:2609-13. ↩
- Kamenova M, Leu S, Mariani L, et al. Management of incidental dural tear during lumbar spine surgery. To suture or not to suture? World Neurosurg 2016; 87:455-62. ↩
- Misra SN, Morgan HW, Sedler R. Lumbar myofascial flap for pseudomeningocele repair. Neurosurg Focus. 2003; 15(3):1-5. ↩