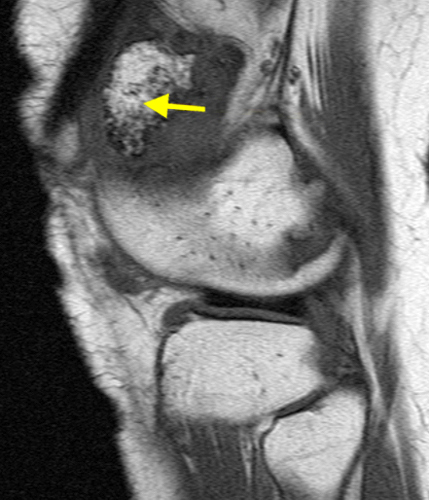
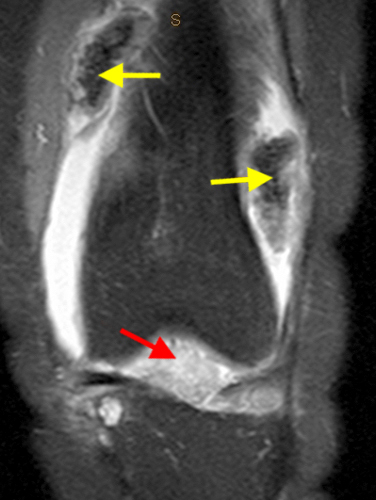
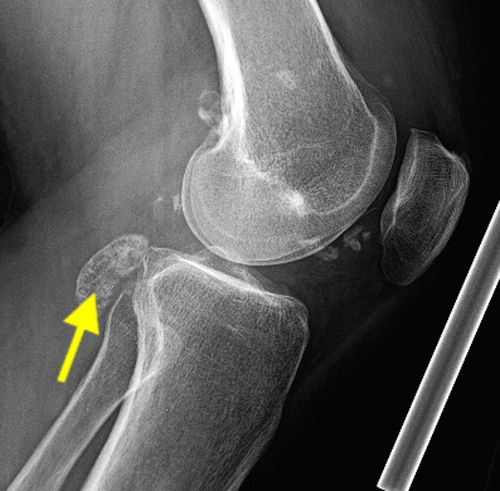
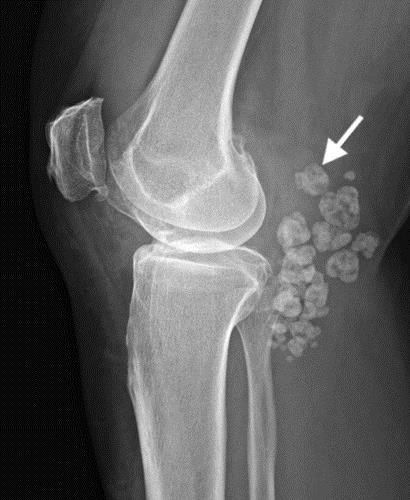
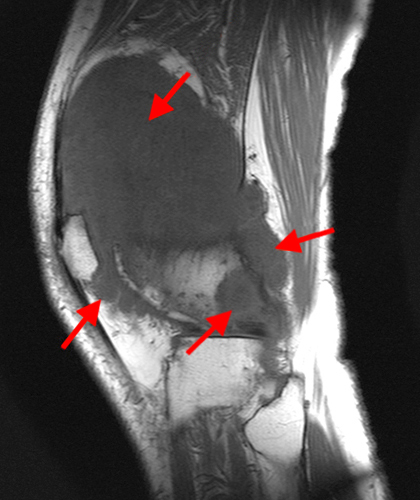
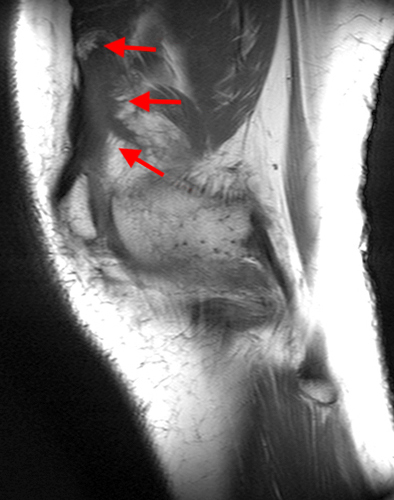
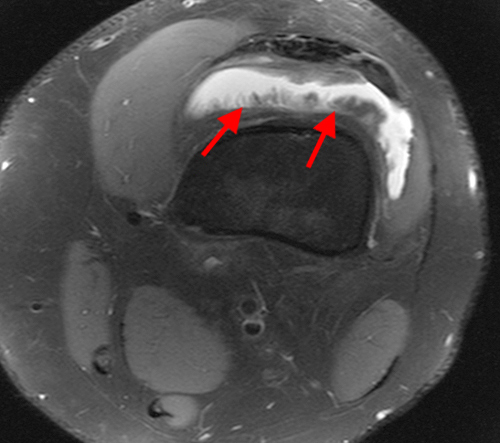
Clinical History: A 51-year-old male presents with slowly progressive swelling, loss of range of motion and intermittent locking of the right knee. What are the findings? What is your diagnosis?
Findings
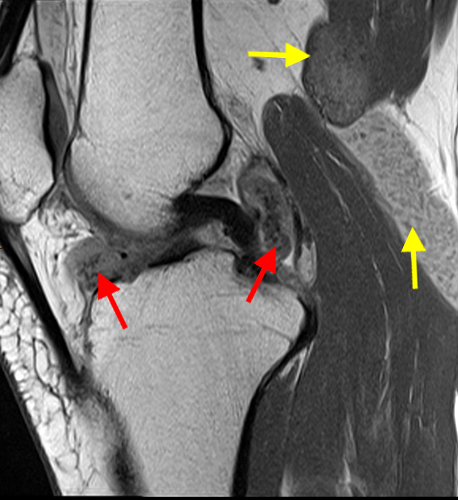
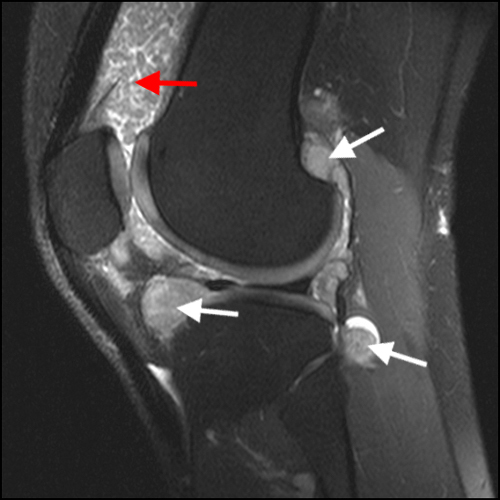
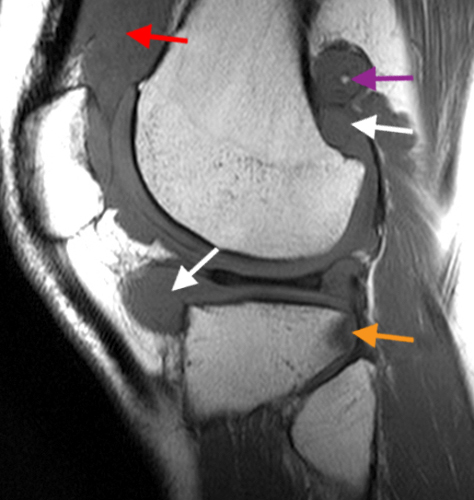
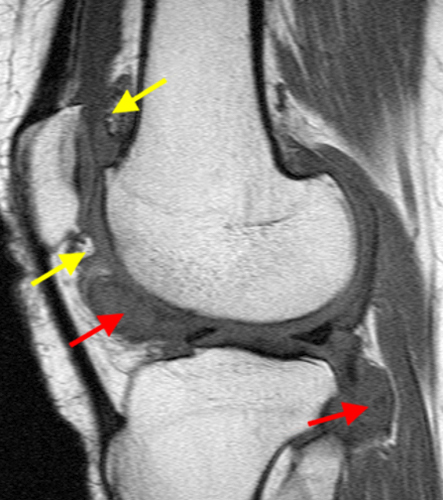
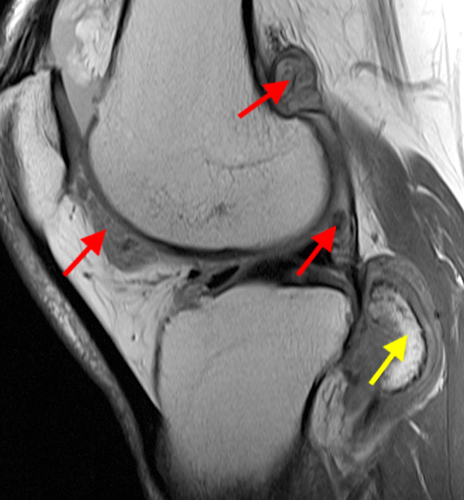
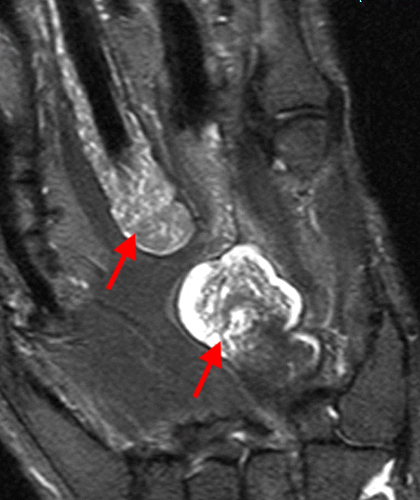
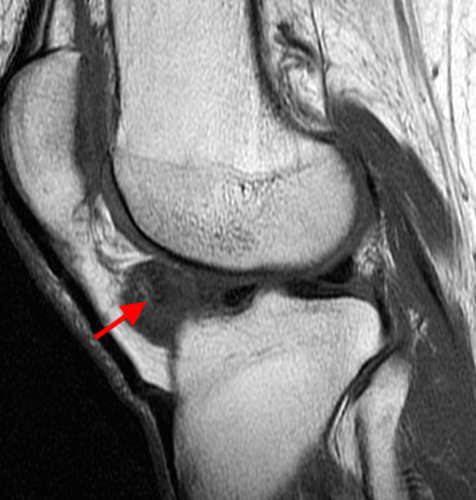
Figure 2:
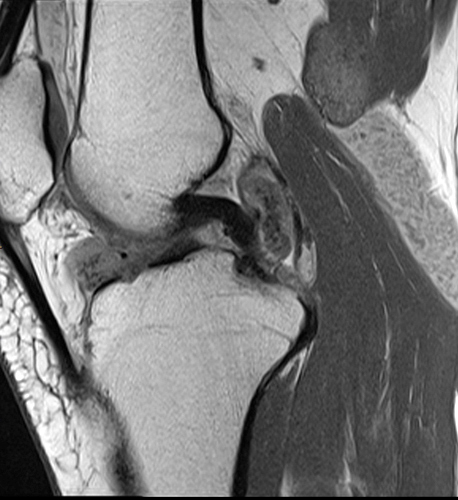
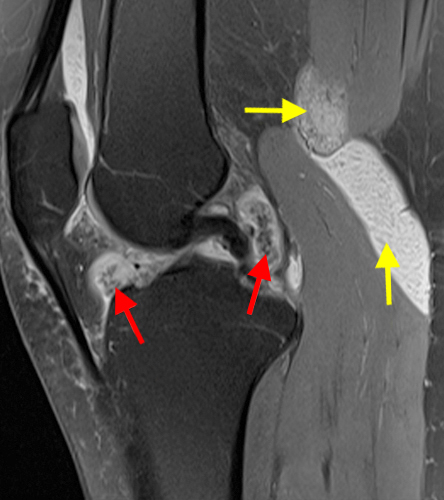
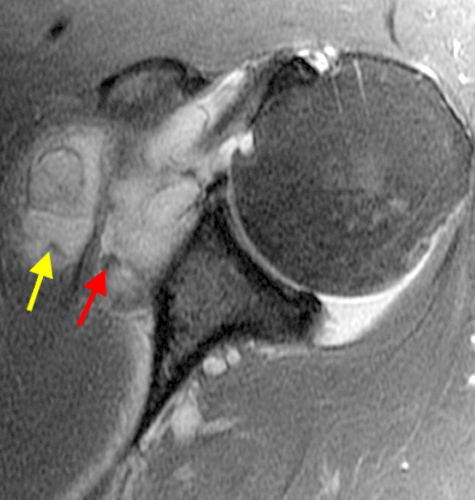
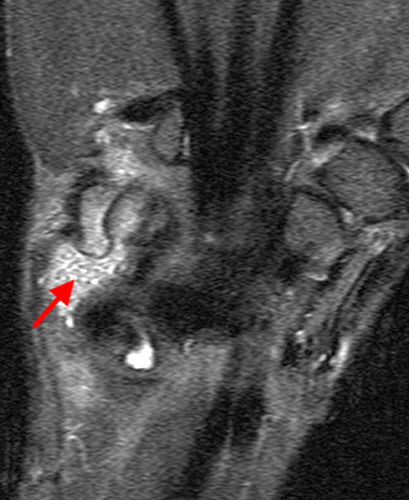
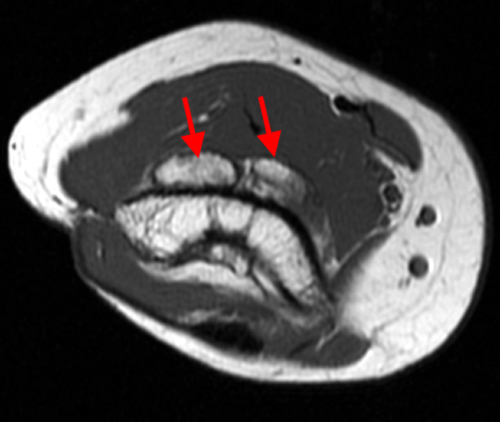
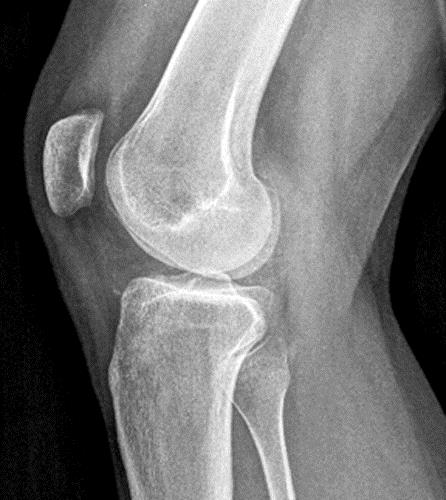
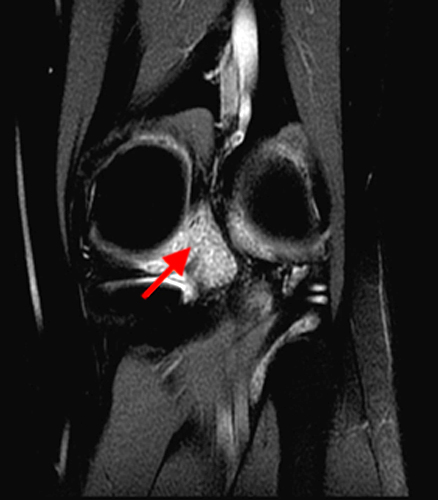
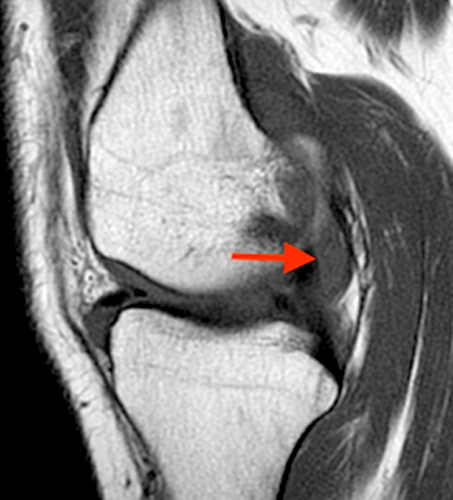
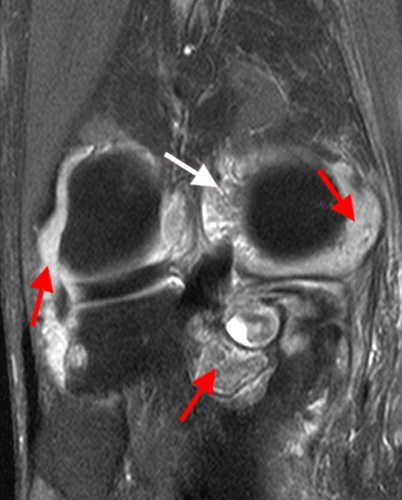
A proton density-weighted sagittal image of the right knee reveals distension of the joint (red arrows) and a popliteal cyst (yellow arrows) by complex material consisting of numerous uniformed sized foci. Several of the intra-articular foci (red arrows) contain central dark signal void. Note the relative preservation of cartilage in the patellofemoral compartment.
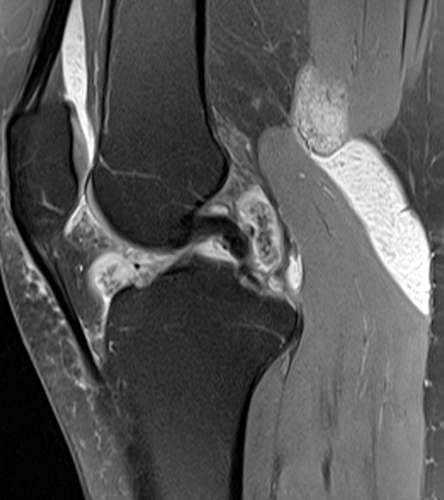
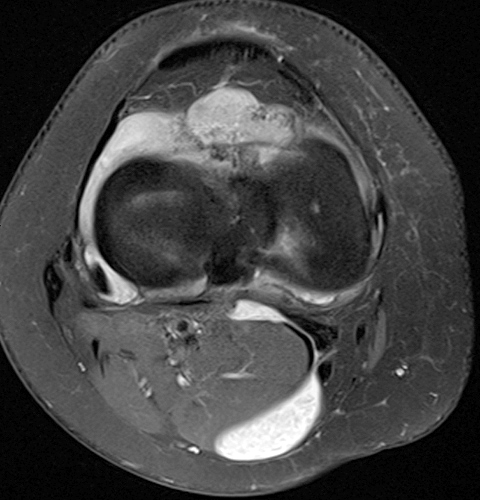
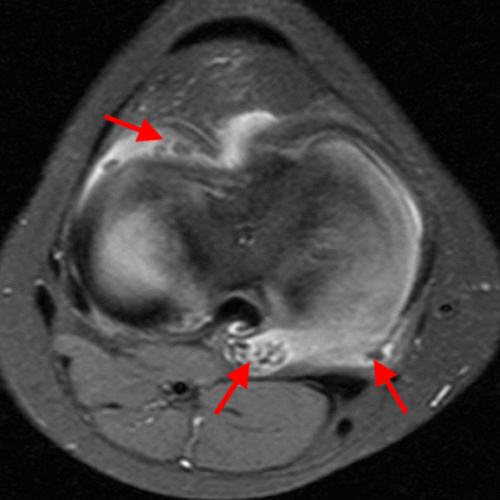
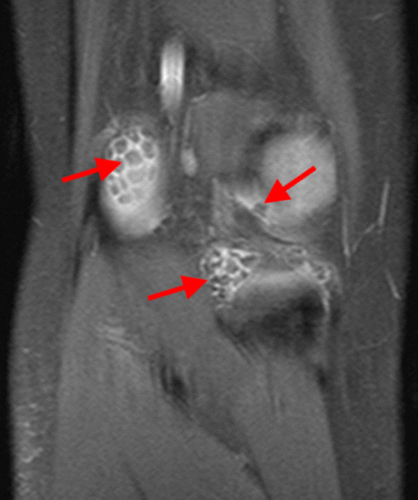
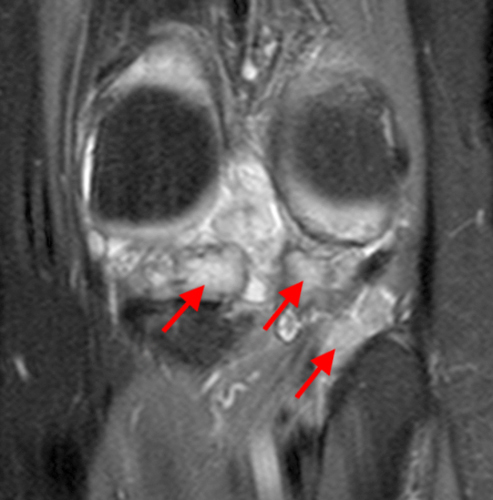
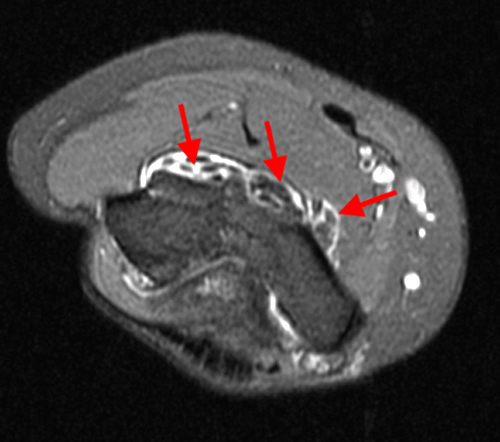
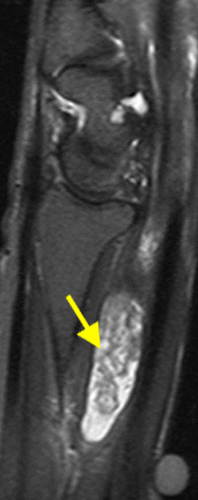
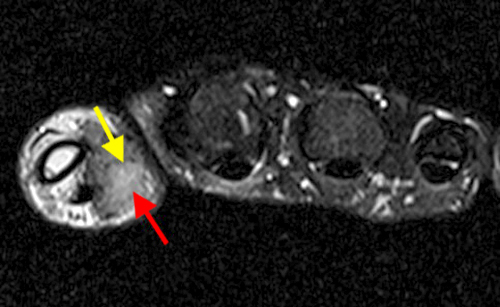
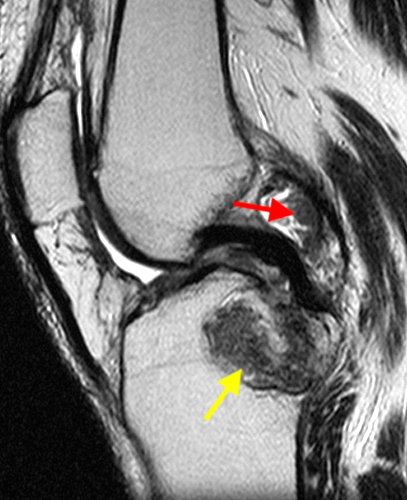
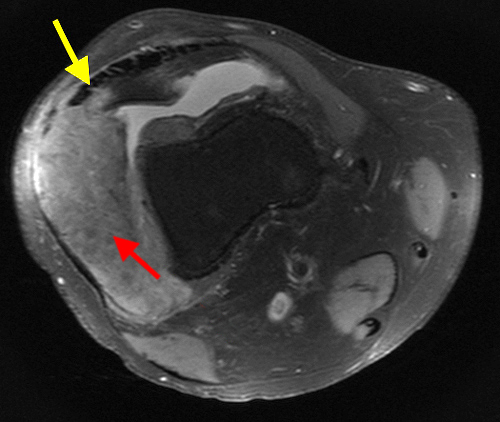
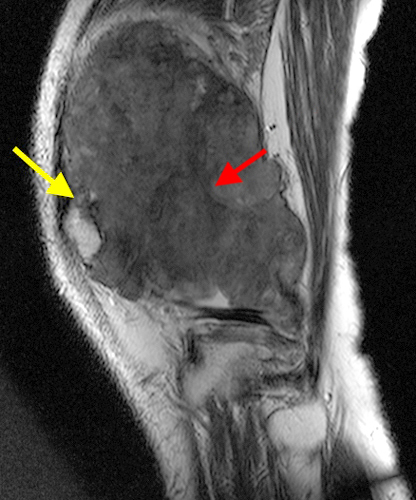
Figure 3:
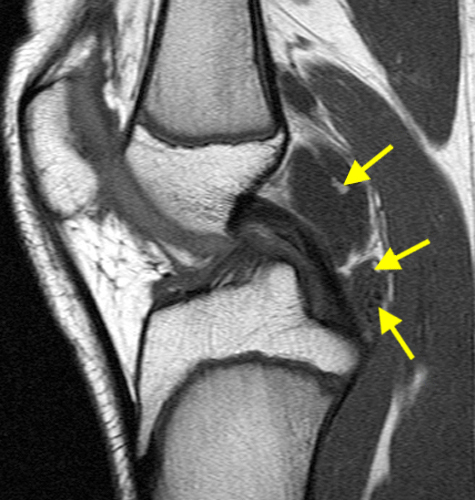
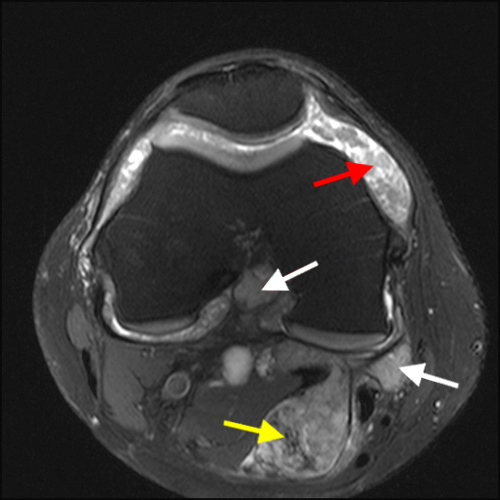
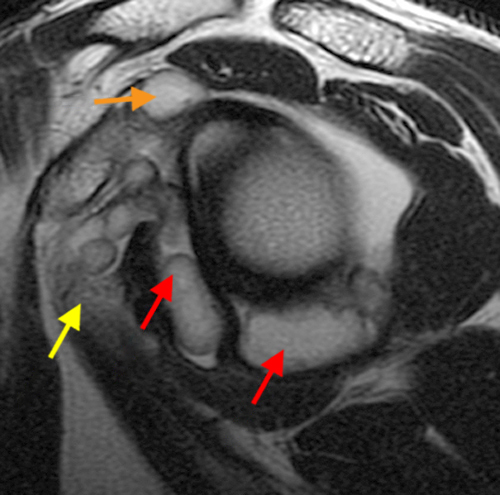
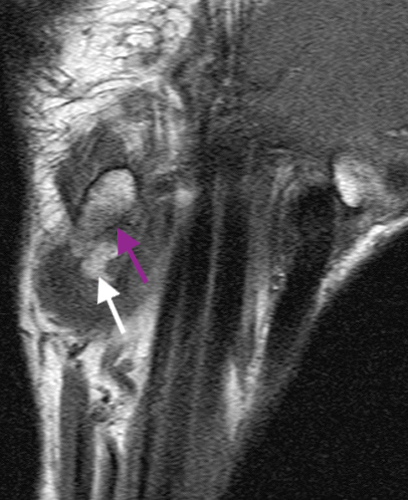
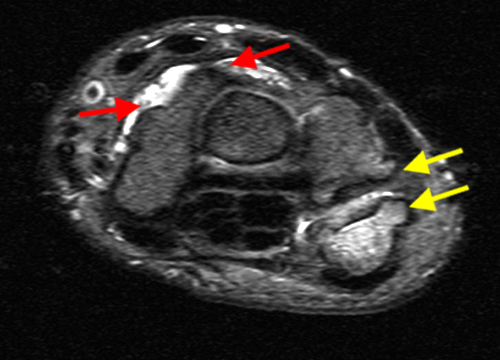
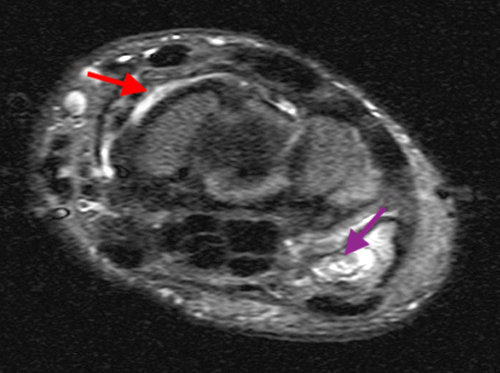
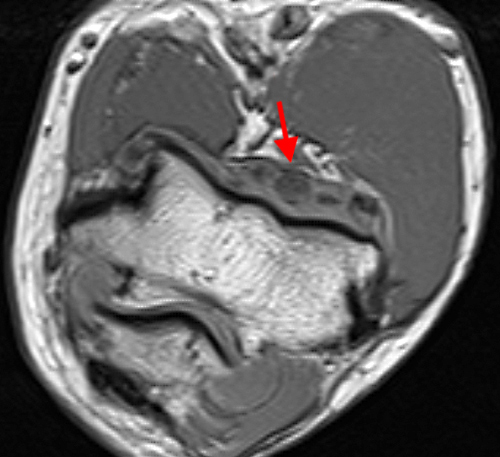
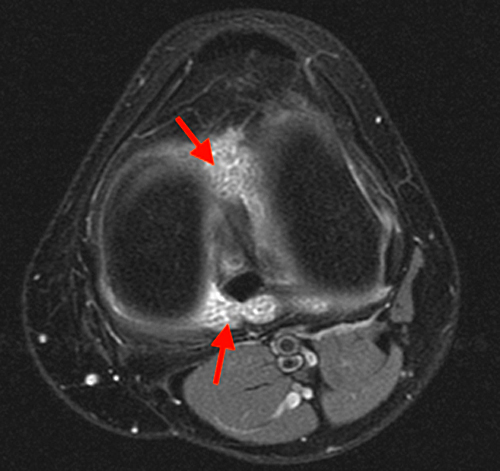
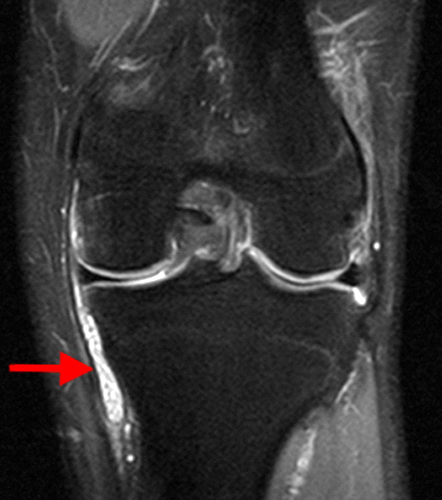
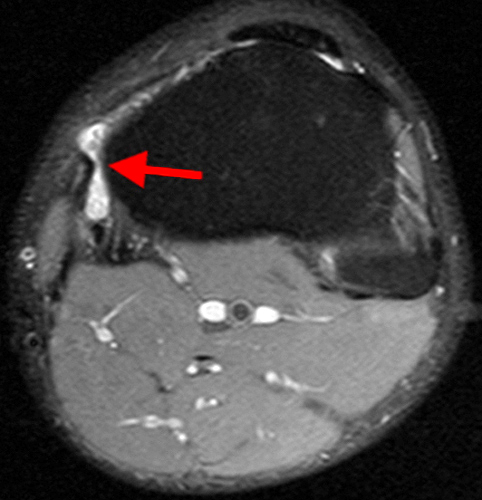
The fat-suppressed proton density-weighted sagittal (3a) and axial (3b) images of the knee reveal distension of the joint (red arrows) and popliteal cyst (yellow arrows) by complex material consisting of numerous uniformed sized signal foci within a background of fluid signal intensity. Several of the intra-articular foci (red arrows (3a)) contain central dark signal void.
Diagnosis
Primary Synovial Chondromatosis (Reichel Syndrome)
Introduction
Synovial chondromatosis is a rare, monoarticular benign process characterized by hyaline cartilaginous bodies proliferating in subsynovial tissue and detaching as a collection of numerous chondral bodies in a joint or less commonly in bursae or tendon sheaths. 70-95% of the bodies undergo mineralization and may eventually undergo ossification. It is typically a self-limiting process, however, it may recur locally and rarely undergo malignant transformation. Imaging plays a key role in the diagnosis and management as the clinical presentation is non-specific and laboratory tests do not exist. Pathology may simulate chondrosarcoma making the MRI characteristic features vital to the correct diagnosis and management of these patients. 1,2,3,4
Epidemiology and Pathogenesis
Primary synovial chondromatosis preferentially affects one joint with predilection for large joints such as the knee, hip, elbow or shoulder. However, it can be seen in any joint, tendon or bursa. It tends to affect males more frequently than females by greater than 2:1 and most commonly in the third to fifth decades. Several case reports describe synovial chondromatosis occurring in children. 1,2,5,6
Three phases of synovial chondromatosis have been described though progression through each phase has not been established. The phase at diagnosis is virtually non-diagnostic without histopathologic correlation. The first phase is the active formation of hyaline cartilaginous bodies within the synovium with no loose intra-articular bodies. The second phase is a combination of both synovial cartilaginous proliferation and shedding of intra-articular loose bodies. The third phase is the complete shedding of numerous intra-articular cartilaginous bodies and no active synovial proliferation. MRI findings are variable though three distinct patterns that correspond to the phase of pathology have been described. The most frequently reported pattern corresponds to phase two pathology followed by phase one and phase three. 1,2,5
The molecular basis of the disease is unclear though high levels of BMP-2 and BMP-4 have been isolated from diseased synovium and loose bodies. Fibroblastic growth factors 2 and 3 and chromosome 6 abnormalities have been found in cases of primary synovial chondromatosis and may also play a factor in metaplasia. These growth factors and cytogenetic abnormalities have not been found in secondary synovial chondromatosis. 1,7,8
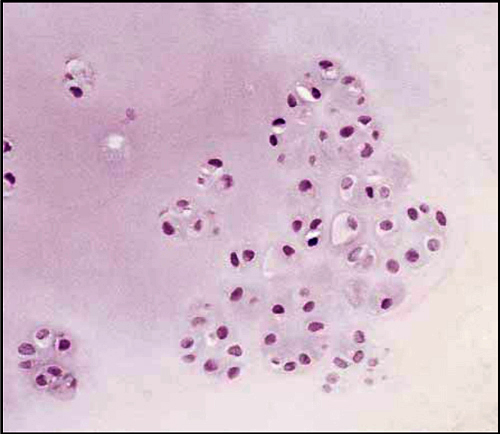
On gross pathology, the synovial bodies tend to range from 2.0 mm to 10 mm in size and are composed of a chalky yellow material consisting of calcification, endochondral ossification or both (Figure 4). Bodies undergo ossification via vascular invasion and endochondral bone formation. Microscopically the hyaline bodies demonstrate greater cellularity than articular cartilage and are clustered rather than evenly distributed. The cells exhibit considerable size range and nuclear chromaticity. Many have atypical features and occasional mitoses can be seen (Figure 5). These atypical features are common findings for this benign disease and thus correlation with characteristic imaging features and clinical presentation is essential for accurately diagnosing this benign condition. 7
Figure 5:
Histology slide of primary synovial chondromatosis: “Typical clustered arrangement of mildly atypical chondrocytes characteristic of synovial chondromatosis (H&E, ×200).” O’Connell JX. Pathology of the Synovium. Am J Clin Pathol 2000;114:773-784. 7. Image courtesy of John X O'Connell, MD.
Secondary synovial chondromatosis is more common than the primary form and presents in the setting of chronic pathology of the joint either secondary to a mechanical or an arthritic process leading to formation of loose chondral bodies. It is more commonly seen in the knee, hip or shoulder in the setting of osteoarthritis, trauma, inflammation, osteonecrosis or neuropathy and found in patients who are older than patients with primary disease. The bodies arise from loose hyaline cartilage implanting in synovium and inducing synovial metaplasia. The subsequent metaplastic chondral bodies that accumulate in the joint tend to be fewer in number and more variable and larger in size suggesting various times of origin. Several concentric rings of calcification may be seen in secondary synovial bodies as opposed to the more typical ‘ring and arc’ pattern of chondroid calcification seen in primary synovial chondromatosis. 1,2,4
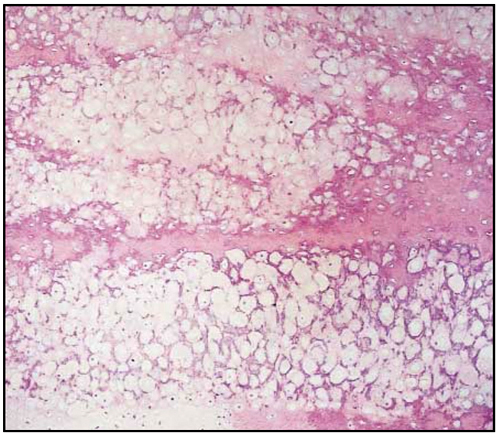
On gross and histologic pathology, the bodies are nodular and bosselated and may contain the osteo-articular fragment that incited the metaplasia. They are composed of reactive and metaplastic features between hyaline and fibrocartilage. Histologically, they are uniform in cellularity and laminar in arrangement as opposed to the clustered appearance seen in primary synovial chondromatosis (Figure 6). They rarely demonstrate atypia or mitotic activity. The primary and secondary forms of synovial chondromatosis are easily differentiated by histopathology. 7
The incidence of synovial chondrosarcoma in the setting of synovial chondromatosis is rare with reported incidence ranging from 1% and most recently reported up to 6.4% 9, and only reported in intra-articular cases. The majority of cases of synovial chondromatosis are self-limiting with case reports of spontaneous resolution. Following synovectomy, the incidence of recurrence in primary synovial chondromatosis is reported as high as 23%. In those patients who present with recurrence, differentiating from a malignant transformation can be difficult. The degree of atypia found in synovial chondromatosis can equal or exceed the atypia found in low and intermediate grade chondrosarcoma making histology not always helpful. To date, there are no reliable immune or histochemical markers that differentiate the two pathologies, therefore, imaging and clinical characteristics play a crucial role. Any patient presenting with an aggressive or debilitating recurrence and/or MRI demonstrating aggressive features of permeative or invasive bone erosions should be regarded as suspicious for malignant transformation to chondrosarcoma. Chondrosarcoma is generally treated by amputation and despite aggressive therapy, up to 56% of patients demonstrate lung metastases as reported in a series by Bertoni et al. 9 Radiation and chemotherapy are ineffective in treating chondrosarcoma and primary synovial chondromatosis. 1,9,10,11,12
Clinical Presentation
Symptoms are often insidious and gradually progress with duration to diagnosis on average of 5 years. Patients most commonly present with pain, followed by swelling, decreased range of motion, and locking of the joint and least commonly present with a palpable mass. They generally do not report acute trauma and do not exhibit signs of infection or illness. Presentation is non-specific and non-diagnostic. Most patients are sent for MRI for more sensitive and specific diagnosis. 1,2
MRI Imaging Characteristics
Depending on the phase of disease at the time of imaging, primary synovial chondromatosis can have a varied MRI appearance but with characteristic and specific diagnostic features. In the setting of synovial metaplasia with no mineralization of bodies, the radiographs may be unremarkable and MRI images demonstrate hyperintense T2 signal intra-articular masses which correspond to T1-weighted intermediate to hypointense masses without areas of internal signal void. The synovial masses will enhance intensely following IV contrast. During the second phase of disease, the synovial bodies are characteristically hyperintense on T2 and low to intermediate signal on T1 with central signal void from calcification or central fat and a low signal rim variably depending on the degree of mineralization. They demonstrate mild enhancement with a peripheral and septal pattern. The bodies tend to be more fusiform in shape. During phase three, there are numerous intra-articular bodies with varying degrees of mineralization characterized by central fat signal intensity with a dark rim from calcification. No synovial lobular masses are seen. 1,2,3,4
CT and x-rays help characterize mechanical erosions and evaluate mineralization of intra-articular bodies. At the time of presentation, up to 70% of patients have radiographically visible calcifications or ossifications. Bone erosions of variable size can be seen more often in joints with a tight capsule such as the hip and are reported in 20-50% of all cases. The calcifications found in primary synovial chondromatosis are the typical ‘ring and arc’ pattern of mineralization rather than concentric rings of calcification seen in secondary synovial chondromatosis. 1,2,3,4
MRI Diagnosis in Primary Synovial Chondromatosis
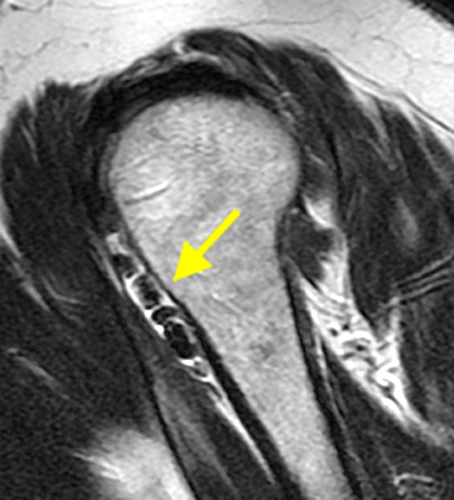
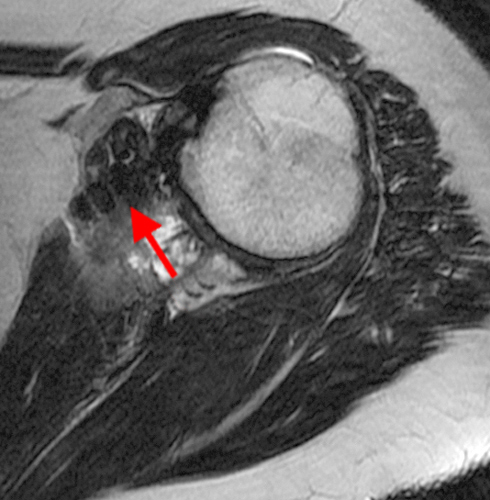
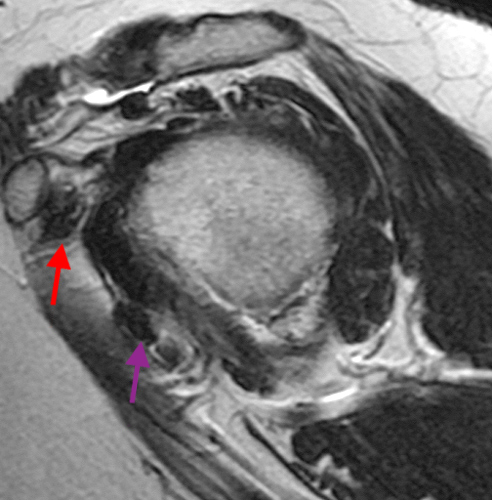
Figure 7:
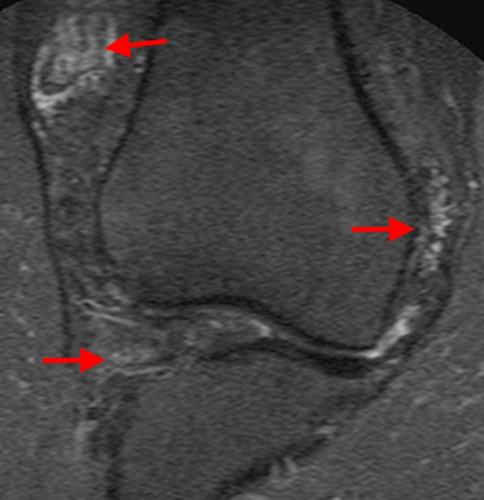
Pediatric Primary Synovial Chondromatosis. A sagittal T1-weighted (7a) and axial (7b) and coronal (7c) fat suppressed proton density-weighted images demonstrate a moderate sized joint effusion containing numerous uniform shape and size intermediate on T1 and intermediate to hyperintense T2 bodies (red arrows). Several of the bodies contain fat signal and signal voids consistent with mineralization are best seen on the T1-weighted sequence (yellow arrows).
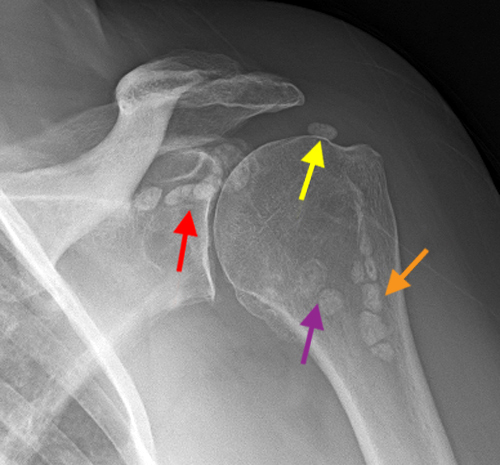
Figure 8:
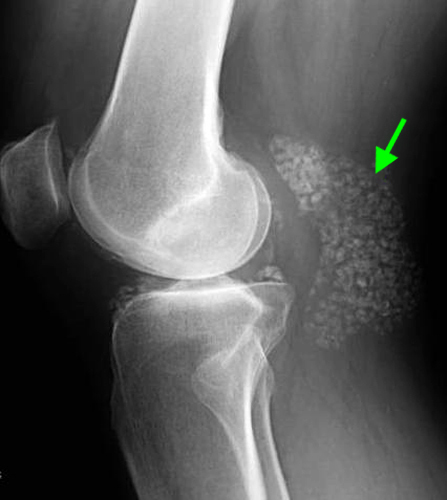
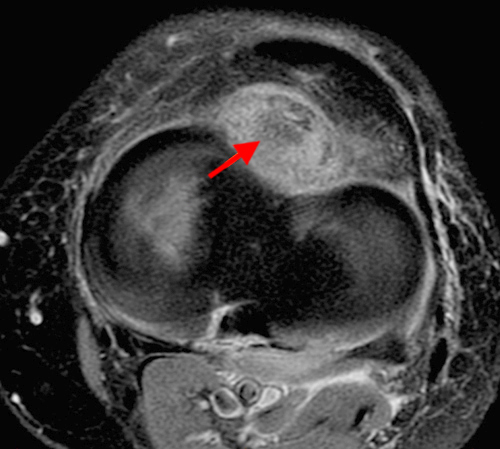
Pediatric Primary Synovial Chondromatosis. A sagittal fat suppressed proton density-weighted (8a), sagittal T1-weighted (8b) and axial fat suppressed proton density-weighted (8c) images of the knee demonstrate a moderate sized joint effusion containing numerous uniform shape and size bodies (red arrows). Several areas of nodular coalescence are noted (white arrows) and a small mechanical erosion is seen at the posterolateral tibial plateau (orange arrow). Several of the bodies contain signal void as seen in the popliteal cyst (yellow arrow) as well as fat signal foci (purple arrow) consistent with mineralization. A plain film radiograph in a different patient (8d) demonstrates the typical ‘ring and arc’ (green arrow) pattern of mineralization seen in numerous uniform shape and sized bodies in the popliteal cyst and is pathognomonic for primary synovial chondromatosis. Note the preservation of cartilage and relative lack of chronic pathology in the joint as is typical for primary synovial chondromatosis.
Figure 9:
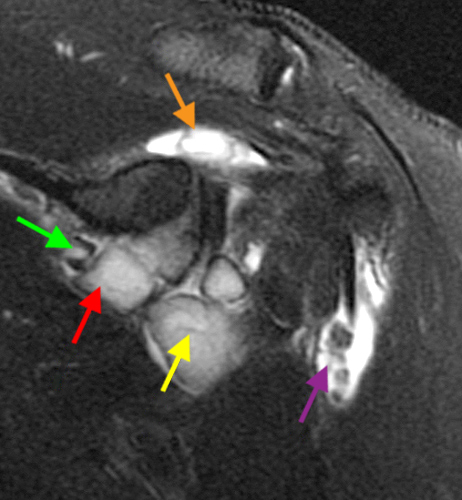
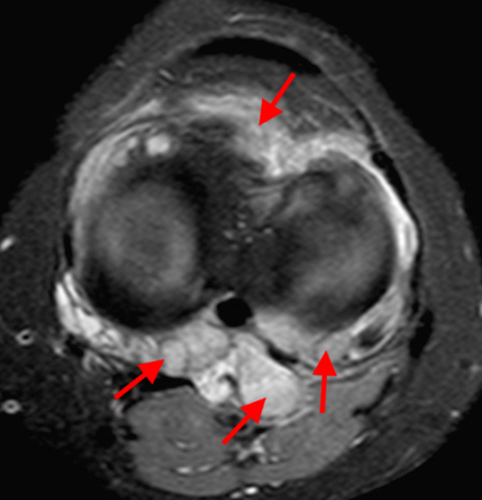
Primary Synovial Chondromatosis in the shoulder. Sagittal T2-weighted (9a, 9d), coronal fat-suppressed T2-weighted (9b), and axial (9c) fat-suppressed proton density-weighted sequences of the shoulder reveal numerous moderate sized lobular hyperintense signal masses throughout the glenohumeral joint, the subscapularis recess, the axillary pouch (red arrows), undermining the supraspinatus tendon (orange arrow), in the subcoracoid bursa (yellow arrow) and in the biceps tendon sheath (purple arrow). Several masses contain faint dark signal mineralization best seen in the biceps tendon sheath. The “ring and arc” pattern of mineralization typically seen in primary synovial chondromatosis is best demonstrated in a body seen in the subscapularis recess (green arrow).
Figure 10:
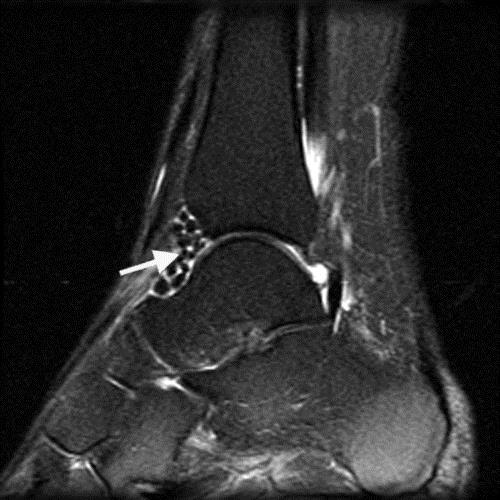
Primary Synovial Chondromatosis in the Ankle. Axial T2-weighted (10a), fat suppressed coronal (10b) and sagittal (10c) T2-weighted sequences demonstrate numerous uniform size and shape dark signal bodies (white arrows) collected in a moderate sized ankle joint effusion. No hyperintense lobular synovial based masses are present and the overall appearance suggests phase three disease.
MRI Diagnosis in Primary versus Secondary Synovial Chondromatosis
Differentiating primary synovial chondromatosis from its more common secondary form may be difficult by MRI as secondary synovial chondromatosis generally presents in the setting of osteoarthritis, and long standing primary synovial chondromatosis predisposes to developing osteoarthritis.1 However, important differences frequently allow the diagnosis to be made.13
Figure 11:
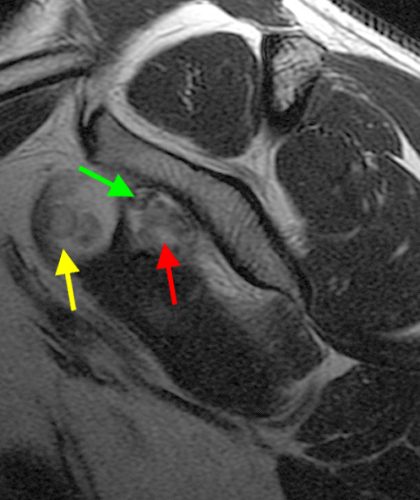
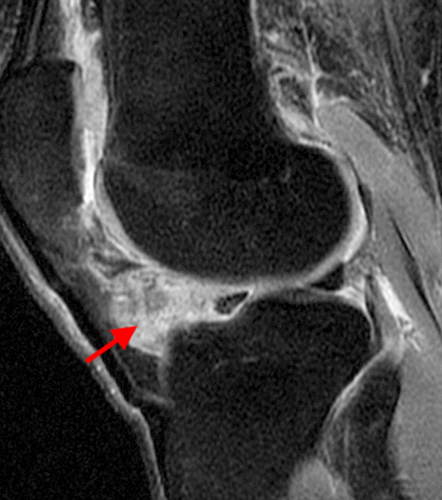
Primary Synovial Chondromatosis in the Knee. Axial (11a), coronal (11c, 11e) fat suppressed proton density-weighted and sagittal (11b, 11d) T1-weighted sequences demonstrate numerous hyperintense T2 signal masses (red arrows), numerous smaller hyperintense T2 signal bodies with central signal void and intermediate signal rind and several ossified masses (yellow arrows) in a background of a moderate sized knee joint effusion. Findings suggest phase two disease process. Uncommonly, bodies may detach as a large conglomerate mass termed a giant synovial chondroma (yellow arrows) and by report have measured up to 20cm.
Figure 12:
Primary Synovial Chondromatosis in the Shoulder. AP radiograph of the shoulder (12a), sagittal (12b, 12d) and axial (12c) T2-weighted sequences of the shoulder reveal multiple uniform shape and size ossified bodies collected throughout the glenohumeral joint, in the subscapularis recess (red arrows), undermining the rotator cuff tendon (yellow arrow), in the subcoracoid bursa (purple arrow) and in the biceps tendon sheath (orange arrow). No hyperintense T2-weighted soft tissue masses are present suggesting phase three disease. Moderate cartilage loss and subchondral degenerative changes are noted in the glenohumeral joint. The bodies are uniform in shape and size which is more common in primary synovial chondromatosis.
Figure 13:
Primary Synovial Chondromatosis in the Wrist with secondary osteoarthritis: AP radiograph (13a), coronal fat suppressed T2 weighted (13b), coronal T1 weighted (13c), and axial fat suppressed T2 weighted (13d, 13e) sequences of the wrist demonstrate multiple generally uniform in size intra-articular bodies, the majority of which demonstrate a hyperintense center and an intermediate signal rim on T2 weighted images (red arrows). Generalized synovial thickening in the setting of osteoarthritis (yellow arrows denoting spurs) is seen on 13d. The plain film radiograph shows soft tissue swelling (green arrow) compatible with joint effusion and the ossified body (white arrow) also seen on the coronal T1 weighted sequence (13c). An erosion (purple arrow) in the pisiform is subtly seen on the coronal T1 weighted sequence (13c) and well seen on the axial fat suppressed T2 weighted sequence (13e).
Figure 14:
Secondary Synovial Chondromatosis in the Knee: Lateral radiograph (14a), axial fat suppressed proton density-weighted (14b) and sagittal proton density-weighted (14c) sequences of the knee demonstrate multiple varying size intra-articular bodies (red arrows), with areas of nodular coalescence and varying degrees of central signal void as well as a large ossified body (yellow arrow). There is moderate osteoarthritis in the joint. A lateral radiograph (14d) in a second patient demonstrates the concentric rings of calcification (white arrow) in chondral bodies, which are fewer in number and variable in size collected in the popliteal cyst and typical for secondary synovial chondromatosis in the setting of chronic osteoarthritis.
Figure 15:
Secondary Synovial Chondromatosis in the Elbow: Axial fat-suppressed proton density-weighted (15a), coronal (15c) fat suppressed T2-weighted and axial T1-weighted (15b, 15d) images of the elbow show multiple varying size intra-articular low signal bodies (red arrows), many of which are bosselated and demonstrate concentric rings of calcification (15c, red arrow). Two large ossified bodies (15b, red arrows) are present. There is moderate osteoarthritis in the joint. These findings are typical for secondary synovial chondromatosis.
Differential Diagnosis on MRI
The bodies of synovial chondromatosis should not be confused for rice bodies which are found in the setting of chronic inflammation of synovium and described in several entities including chronic arthritis, MAI infection, sarcoidosis and most commonly in rheumatoid arthritis. Rice bodies are tiny, uniformly shaped and sized bodies composed of fibrin, which are hypointense on T2, intermediate in signal intensity on T1 and contain no mineralization. It is proposed that synovial cells undergo infarction, are shed into the joint, and become encased by fibrin over a period of time. Others theories suggest the fibrin accumulates in hypertrophied synovium and sheds off as fibrin bodies into the joint. 5,14,15
Figure 16:
Rice Bodies: Lateral radiograph of the knee (16a) demonstrates no calcification or ossification in the joint and no bone erosions. Fat-suppressed T2-weighted axial (16b)and coronal (16c), and T2-weighted sagittal (16d) images reveal distension of the joint by numerous uniform shape and size hypointense loose foci in joint fluid with no matrix and no nodular coalescence. The rice bodies are not readily apparent on the sagittal T1-weighted sequence (16e), as they are relatively isointense to muscle and fluid. In synovial chondromatosis hypointense bodies may be seen on T1-weighted images. The combination of the plain film radiograph demonstrating no mineralization and MRI demonstrating numerous tiny intra-articular bodies makes the diagnosis of rice bodies and excludes the diagnosis of synovial chondromatosis.
Synovial chondromatosis in the tendon sheath or bursa may be referred to as tenosynovial or bursal chondromatosis and is pathologically identical to intra-articular primary synovial chondromatosis. Tenosynovial or bursal chondromatosis most commonly affects adults over 20 years of age with the greatest number cases found in patients in their fifth decade of life. It most commonly affects males, however when diagnosed in older patients there is a female predominance. Extra-articular involvement is most commonly seen in the hands accounting for greater than 50% of cases, followed by feet and wrists. Patients generally present with a painless mass or mass with mild tenderness to palpation. In contrast to intra-articular disease, they seldom present with limited joint motion. 1,3,4,13,16,17
Figure 17:
Figures 17a and b: Synovial bodies in the tibial collateral ligament bursa.
Tibial Collateral Ligament Bursal Synovial Chondromatosis: Fat-suppressed T2-weighted coronal (17a) and axial (17b) images of the knee demonstrate multiple small uniform shape and size bodies distending the tibial collateral ligament bursa. The bodies demonstrate no apparent mineralization (red arrows). In this young 20 y/o male with histology consistent with primary synovial bursal chondromatosis, it is difficult to differentiate synovial bodies, which lack mineralization from rice bodies. Rice bodies more commonly layer dependently in joint fluid as opposed to synovial bodies, which may be attached or partially imbedded within the synovium depending on the phase of the disease process.
Figures 17 c and d: Rice bodies in flexor tendon sheath deep to carpal tunnel tendons in palm and distal forearm and flexor tendon of the long finger.
MAI Tenosynovitis: In contrast to this case of MAI containing rice bodies collected in horseshoe bursa of the wrist, the fat-suppressed T2-weighted coronal (17c) of the hand and sagittal (17d) images of the wrist demonstrate multiple small uniform shape and size bodies. The relative T2 signal is decreased compared to surrounding muscle as compared to the increased T2 signal intensity of the synovial chondromatosis bodies in Figure 17A. The T2 hyperintensity of the chondral bodies is due to the fluid content and hypointensity of the rice bodies is due to fibrin content.
The primary differential diagnosis for bursal or tenosynovial chondromatosis is giant cell tumor of the tendon sheath (GCTTS). It presents as a slow growing painless mass most commonly found in the hand and is pathologically identical to PVNS. MRI shows low to intermediate signal on T1 and T2 weighted sequences due to the presence of hemosiderin. Calcifications are uncommon in GCTTS. 1,2,17,18
Figure 18:
GCTTS: STIR (18a) and T1 weighted (18b) axial images of the thumb reveal a well-defined soft tissue mass along the ulnar aspect of flexor pollicis longus tendon abutting the proximal phalanx of left thumb (red arrows). It is homogenous intermediate to hypointense signal on STIR and T1 weighted images, contains thin linear septa (yellow arrow) and contains no calcifications.
MR’s soft tissue resolution allows differentiation of synovial chondromatosis from other primary synovial pathologies which include pigmented villonodular synovitis (PVNS), hemosiderin arthrosis, nodular synovitis otherwise referred to as localized PVNS, lipoma arborescens and synovial hemangiomas. 1,2,3,4,18,19 PVNS is a benign proliferative synovial lesion, generally mono-articular and presenting in the 3rd to 4th decade of life. PVNS lesions tend to bleed, resulting in characteristic very dark signal hemosiderin deposits. The nodular masses in PVNS typically cause bone erosions, which is helpful in differentiating this disease from hemosiderin arthrosis. PVNS tends to have a more frond-like villous appearance. 1,2,3,4,17,18,19 Hemophilic or hemosiderin arthropathy leads to uniform dark signal thickening of the synovium without nodular masses, villous proliferation or erosions.
Figure 19:
PVNS: Sagittal T2-weighted (19a) and fat suppressed coronal proton density-weighted (19b) images of the knee demonstrate dark signal nodular masses (red arrows) throughout the joint causing large bone erosions in the tibial plateau (yellow arrow). Several areas of nodular and more villous frond-like proliferation of synovium are seen projecting into the joint lateral to the medial femoral condyle (white arrow 19b).
Nodular synovitis, also termed localized pigmented villonodular synovitis, is found in the knee and most commonly in the infrapatellar fat pad. Bodies tend to be ovoid or multilobular, intermediate to high signal on T1 and heterogeneous on T2. Imaging characteristics tend to mimic PVNS with areas of dark signal due to hemosiderin. In addition to being unifocal, nodular synovitis tends to grow outward in a more pedunculated fashion as compared to diffuse PVNS. 2,3,18,19
Figure 21:
Nodular Synovitis: Fat-suppressed proton density-weighted axial (21a), sagittal (21b) and T1-weighted sagittal (21c) images of the knee demonstrate a moderate sized soft tissue mass with mixed signal intensity in the infrapatellar fat pad (red arrows). The lesion demonstrates predominantly hyperintense signal on the proton density-weighted images with central dark signal corresponding to dark signal on the T1-weighted sequence, suggesting hemosiderin within the solid mass.
Figure 22:
Nodular Synovitis: Fat-suppressed proton density-weighted axial (22a), T2 weighted (22b) and T1 weighted sagittal (22c) images of the knee reveal distension of the joint by a large mass (red arrows). The mass demonstrates predominantly low signal intensity on all images. Note the mechanical based erosion of the patella (short yellow arrow). No hyperintense signal or areas of signal void are noted in the mass, which would be expected in a conglomerate mass seen in synovial chondromatosis. Note the mass distends the lateral patellofemoral recess as opposed to filling the entire joint space.
Lipomatosis arborescens is a benign villous proliferation of synovium replaced by mature fat cells and is a reactive response to chronic synovial irritation or inflammation. It is characterized on MRI by frond like proliferation of synovium projecting into the joint with signal on all pulse sequences consistent with fat. 2,19
Figure 23:
Lipoma Arborescens: Sagittal T1-weighted (23a) and fat suppressed axial proton density-weighted (23b) sequences of the knee demonstrate fat signal intensity frond like projections extending into a fluid distended suprapatellar recess of the knee and consistent with lipoma arborescens.
Treatment
Primary synovial chondromatosis tends to be self-limiting, however it can cause severe damage to the joint and lead to osteoarthritis. Early removal of loose bodies in both primary and secondary forms of synovial chondromatosis is generally performed to relieve painful symptoms and to prevent further joint deterioration. 1,2
Synovectomy has generally been reserved for recurrent and persistent synovial metaplasia, however, determining the exact phase of the disease process is based on histologic correlation and therefore most physicians recommend excision of the bodies and synovectomy. Sufficient evidence supports arthroscopic removal of loose bodies with limited synovectomy as first-line surgical therapy. Synovectomy is not necessary in secondary forms of chondromatosis as recurrence is uncommon. Patients who are treated conservatively are generally treated with non-steroidal anti-inflammatories, activity modification, ultrasound and cryotherapy. 1,2,5
Conclusion
Primary synovial chondromatosis is a benign metaplastic process characterized by hyaline cartilage bodies proliferating in subsynovial tissues of the joint, bursa or tendon sheaths. Radiographic and MRI features are pathognomonic when characterized by a collection of numerous mineralized bodies of similar shape and size and seen with relative preservation of articular cartilage of the joint. Secondary synovial chondromatosis may be differentiated from its primary form by the accumulation of variable shape and size bodies, and fewer intra-articular bodies demonstrating concentric rings of calcification. Secondary synovial chondromatosis generally presents in the setting of underlying arthritis of the joint. Treatment of primary synovial chondromatosis most often consists of removal of loose bodies and synovectomy. Malignant transformation of primary synovial chondromatosis to chondrosarcoma is uncommon and can be difficult to distinguish from benign disease. The degree of atypia is similar in both primary disease and chondrosarcoma making pathology not always helpful. Aggressive and debilitating recurrence and/or MRI demonstrating permeative and invasive bone erosions should be regarded as malignant transformation. Understanding the pathology and recognizing the radiographic and MRI appearance of primary synovial chondromatosis and differentiating it from its secondary form and other common synovial pathologies are important in diagnosis and clinical management of these patients. 1,2,5,7
References
- Murphey MD, Vidal JA, Fanburg-Smith JC, Gajewski DA. Imaging of synovial chondromatosis with radiologic-pathologic correlation. Radiographics. 2007;27(5):1465-1488. doi:10.1148/rg.275075116. ↩
- Garner HW, Bestic JM. Benign Synovial Tumors and Proliferative Processes. Semin Musculoskeletal Radiol. 2013;17:177-188. ↩
- McKenzie G, Raby N, Ritchie D. A pictorial review of primary synovial osteochondromatosis. Eur Radiol. 2008;18(11):2662-2669. doi:10.1007/s00330-008-1024-8. ↩
- Ryan RS, Harris AC, O’Connell JX, Munk PL. Synovial osteochondromatosis: the spectrum of imaging findings. Australas Radiol. 2005;49(2):95-100. doi:10.1111/j.1440-1673.2005.01416.x. ↩
- Fuerst M, Zustin J, Lohmann C, Rüther W. (Synovial chondromatosis). Orthopade. Jun 2009;38(6):511-9. ↩
- Kirchhoff C, Buhmann S, Braunstein V, Weiler V, Mutschler W, Biberthaler P. Synovial chondromatosis of the long biceps tendon sheath in a child: a case report and review of the literature. J Shoulder Elbow Surg. May-Jun 2008;17(3):e6-e10. ↩
- O’Connell JX. Pathology of the Synovium. Am J Clin Pathol 2000;114:773-784. ↩
- Nakanishi S, Sakamoto K, Yoshitake H, Kino K, Amagasa T, Yamaguchi A. Bone morphogenetic proteins are involved in the pathobiology of synovial chondromatosis. Biochem Biophys Res Commun. Feb 20 2009;379(4):914-9. ↩
- Evans S, Boffano M, Chaudhry S, Jeys L, Grimer R. Synovial chondrosarcoma arising in synovial chondromatosis. Sarcoma. 2014;2014:647939. ↩
- Bertoni F, Unni KK, Beabout JW, Sim FH. Chondrosarcomas of the synovium. Cancer 1991; 67:155-162. ↩
- Scott Evans, Michele Boffano, Samena Chaudhry, Lee Jeys, and Robert Grimer, “Synovial Chondrosarcoma Arising in Synovial Chondromatosis,” Sarcoma, vol. 2014, Article ID 647939, 4 pages, 2014. doi:10.1155/2014/647939 ↩
- Muramatsu K, Miyoshi T, Moriya A, Onaka H, Shigetomi M, Nakashima D, et al. Extremely rare synovial chondrosarcoma arising from the elbow joint: case report and review of the literature. J Shoulder Elbow Surg. Feb 2012;21(2):e7-11. ↩
- Ho YY, Choueka J. Synovial chondromatosis of the upper extremity. J Hand Surg Am. 2013;38(4):804-810. doi:10.1016/j.jhsa.2013.01.041. ↩
- Rovira SC, Ramos A, Guerrero LE, Vinas DV, et al. ESRA, ECR 2015 symposium. Educational Exhibit. What are Rice Bodies? Differential Diagnosis? ↩
- Chau CLF, Griffith JF, Chan PT, et al. Rice-Body Formation in Atypical Mycobacterial Tenosynovitis and Bursitis: Findings on Sonography and MR Imaging. AJR 2003;180:1455-1459. ↩
- Walker EA, Murphey MD, and Fetsch JF. Imaging characteristics of tenosynovial and bursal chondromatosis. Skeletal Radiol. 2011;40:317-325. ↩
- MRI Web Clinic — June 2013 Palmar Bursae and Flexor Tendon Sheaths of the Wrist and Hand Chad Mann, M.D., Shareef Riad, M.D., Donald Resnick, M.D. See more at: https://radsource.us/palmar-bursae-and-flexor-tendon-sheaths/#sthash.J2zqiOhz.dpuf ↩
- MRI Web Clinic — March 2005 Pigmented Villonodular Synovitis Mark H. Awh, M.D. See more at: https://radsource.us/pigmented-villonodular-synovitis/#sthash.kyhoy2Ht.dpuf ↩
- MRI Web Clinic — May 2013 Lipoma Arborescens Daniel Bodor, M.D. See more at: https://radsource.us/lipoma-arborescens/#sthash.iXum1Q3d.dpuf ↩