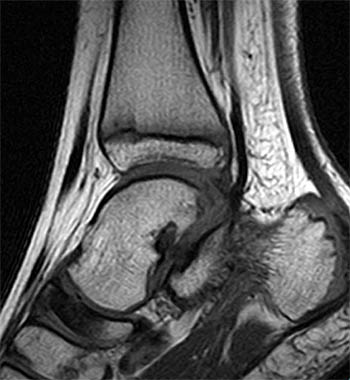
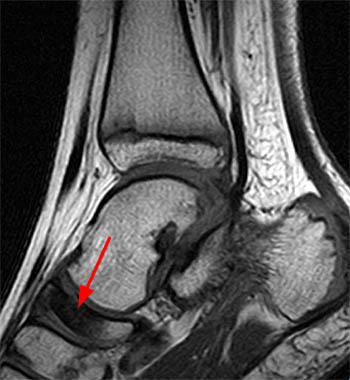
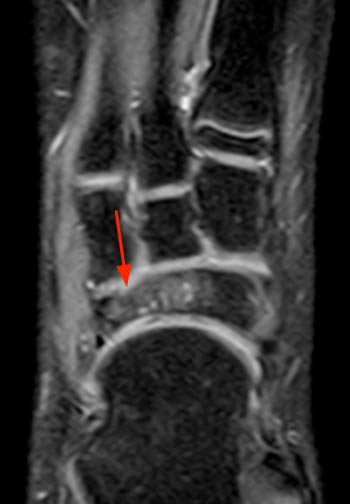
Clinical History: A 9 year old boy presents for imaging with a 4 week history of medial and lateral ankle pain and progressive swelling. (1a) A single sagittal T1-weighted image through the right foot is provided. What are the findings? What is the diagnosis?
Findings
Diagnosis
Articular Navicular Osteochondrosis (Köhler Disease).
Additional images from the case are provided below.
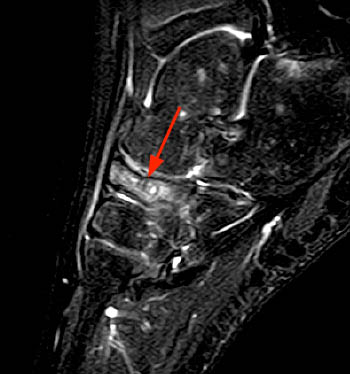
Figure 3:
A sagittal fat-suppressed T2-weighted image centered at the midfoot demonstrates a decreased AP dimension and diffusely and heterogeneously increased signal intensity (arrow) throughout the navicular. Patchy areas of marrow hyperintensity within the hindfoot are likely related to altered biomechanics or disuse.
Discussion
An osteochondrosis is any disease that affects the progress of bone growth involving the centers of ossification. The category of osteochondrosis depends on whether it affects the epiphysis (articular form), apophysis (non-articular form), or the physis.1 The lower extremity articular forms of disease, including Köhler Disease, Freiberg Disease and Legg-Calve-Perthes Disease are detailed in this web clinic.
KOHLER DISEASE (Articular osteochondrosis of the navicular bone)
Köhler Disease, a self-limiting disorder of the navicular bone of the foot in children, occurs from a temporary disruption in the osseous blood supply. The disease is more prevalent in boys, usually occurring between 3-10 years of age. Though more commonly unilateral, the disease may occur bilaterally in up to 25 percent of cases. Patients typically present with swelling and pain that is more common along the medial aspect of the foot, producing a limp with favoring of weight more laterally. Often there is accompanying redness and warmth over the point tender navicular bone. The cause is idiopathic; however, trauma has also been described as a catalyst. One theory is that the delayed ossification of the navicular bone relative to the surrounding ossified bones produces abnormal compression of the navicular and its blood vessels.2 The blood vessels penetrate the cartilaginous portion of the bone but the unossified navicular lacks a mature arterial anastomotic network.
The typical radiologic findings are a combination of flattening, fragmentation, and sclerosis of the navicular bone. Two or more of these usually need to be present to make the diagnosis. Many think that this is not truly a “disease”, but rather an alteration in the sequence of tarsal ossification and some have advocated a requirement to see a change from a previously normal bone in order to make this diagnosis.3
Anatomy
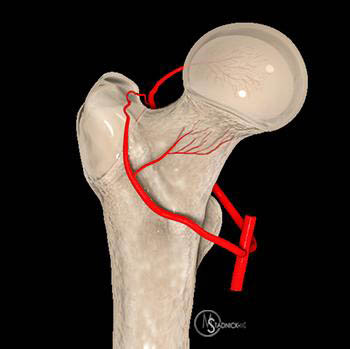
The navicular bone is the last tarsal bone to ossify in the foot. This delayed development is thought to make the bone more susceptible to compression and subsequent vascular damage. If the damage is severe, then the blood supply to the navicular bone can be disrupted, producing avascular necrosis. Typically, the margins of the navicular derive the most blood, supplied by the dorsalis pedis artery laterally and the posterior tibial artery branches medially. The central navicular is the least well supplied, with vascularity originating from end branches.
Radiographic and MRI Evaluation
While Köhler Disease is mostly a clinical diagnosis, standing AP and lateral x-rays may show the characteristic findings seen with avascular necrosis, including fragmentation, sclerosis, and flattening of the bone. The early radiographic changes include collapse and increased density/sclerosis within the navicular, followed by patchy de-ossification and fragmentation. Finally there is healing and the navicular bone is reconstituted.4 MRI typically shows decreased signal on T1-weighting and increased signal on T2-weighting in the early and mid stages of the disease. Osseous flattening may be present. Fragmentation of the navicular ossification center can occur, but the overlying cartilage is generally intact. Infection can mimic the clinical symptoms of Köhler Disease, and MRI can be useful in differentiating these entities, with features characteristic of osteomyelitis such as cortical erosion, adjacent soft tissue abscess, and overlying cellulitis (6a). Nuclear medicine scintigraphy can also be performed, typically showing decreased radiotracer uptake in the setting of Köhler Disease.
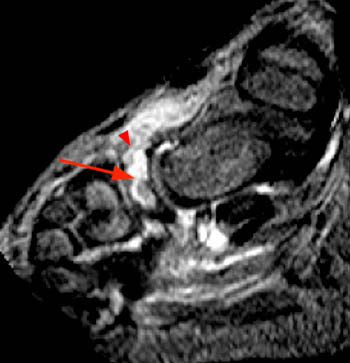
Figure 6:
A sagittal fat suppressed T2-weighted image through the midfoot in a 5 year old girl complaining of pain along the medial side of the foot with limping, swelling, and bruising. The image demonstrates findings of osteomyelitis with severe marrow hyperintensity (arrow) within the ossified portion of the navicular and dorsal erosion of cortex. Soft tissue extension compatible with phlegmon and cellulitis is seen (arrowhead).
Treatment
Treatment is usually conservative and aimed at symptomatic relief. This includes the use of pain medication and arch support. If symptoms do not resolve, a short walking cast is employed for 4-6 weeks with shoe modifications as needed. Long term studies have revealed recovery of function in almost all patients with Köhler Disease, with reconstitution of the navicular.5
FREIBERG DISEASE (ARTICULAR OSTEOCHONDROSIS OF THE FOOT LESSER METARTARSALS)
Freiberg Disease is a painful osteochondrosis involving the metatarsal heads. The term ‘infraction’ was originally coined because of the association of this condition with trauma. The exact etiology remains unknown with major contributing factors including repetitive stress and improper shoe wear including high heels.6 This condition occurs bilaterally in less than 10 percent of cases. Freiberg’s disease typically affects the 2nd more than the 3rd metatarsal but has also been described in rare cases to involve the 4th or 5th metatarsals. This condition occurs most often in adolescent girls who perform ballet and dance.7
Anatomy
There are two centers of ossification for the metatarsals. A primary ossification center is seen for each of the metatarsal shafts, while the lesser metatarsal heads each contain an epiphyseal ossification center. These distal ossification centers begin to develop by the third and fourth years of life and are completely fused with the shafts by the age of 17-20 years. The blood supply to the metatarsal heads is derived from two arterial sources that form a rich extraosseous anastomotic network. These include the dorsal metatarsal arteries (which arise from the dorsalis pedis artery) and the plantar metatarsal arteries (branches of the posterior tibial artery).8 Ischemic necrosis from repetitive trauma likely produces a disruption in the end-arterial blood supply to this region.
Radiographic and MRI Evaluation
Early radiographic findings of Freiberg disease include a joint effusion with widening of the MTP joint space, progressing to avascular necrosis, including sclerosis, cyst development, and finally flattening of the metatarsal head.9 A classification system was devised by Smillie in 1957 for staging.10
Stage 1: subtle fracture line through the epiphysis.
Stage 2: early collapse and central depression.
Stage 3: central depression (resorption).
Stage 4: loose body formation due to freeing of a central fragment.
Stage 5: flattening of the metatarsal head with secondary arthrosis.
MRI is most useful in the early stages of disease when a subtle fracture line may not be evident. In early stages of the disease, a joint effusion and marrow edema may be all that is present. The later stages are analogous with associated cortical flattening and sclerosis (8a,8b).
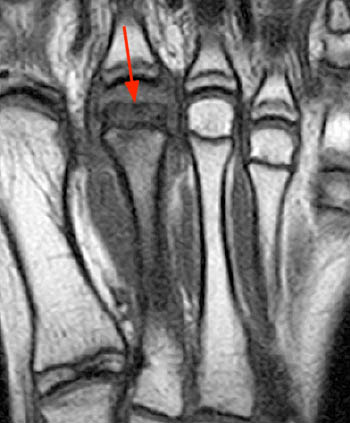
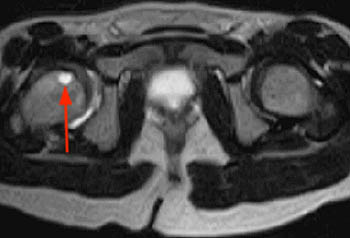
Figure 8:
(8a) A long-axis coronal T1-weighted image through the right foot demonstrates loss of the normal marrow signal throughout the epiphysis and shaft of the 2nd metatarsal, with flattening of the epiphyseal head (arrow). (8b) A corresponding sagittal fat-suppressed T2-weighted image reveals severe marrow edema throughout the 2nd metatarsal with distal flattening of the articular surface (arrow). A reactive joint effusion at the MTP joint is also present.
Treatment
Most early stage cases of Freiberg disease resolve with symptom regression after conservative therapy. This consists of anti-inflammatory medications and activity modification, usually by immobilization of the foot for 4-6 weeks followed by a gradual return to activity with shoe wear modifications including custom orthotics and padding.11 Surgery is necessitated by failing of conservative measures to stem the symptoms. Later stages of the disease may also require surgery due to persistent symptoms from arthrosis and loose bodies within the joint.
LEGG-CALVE-PERTHES DISEASE (ARTICULAR OSTEOCHONDROSIS OF THE FEMORAL HEAD)
Legg-Calve-Perthes Disease (LCP) is a painful osteochondrosis occurring in the hip, producing a limp in the affected child. This is usually noticed in the early avascular phase of the disease process. Boys are five times more likely than girls to develop this condition, with peak incidence at 5-6 years of age. Patients are typically Caucasian, and children of lower socioeconomic status with delayed skeletal age are at greater risk.12 The cause is idiopathic as the exact etiology of the vascular interruption is unknown, with the diagnosis being one of exclusion after other causes of avascular necrosis of the femoral head have been eliminated, such as sickle cell disease, leukemia, prolonged steroid administration and Gaucher’s disease.13 LCP is bilateral in only 15 percent of cases, so a bilateral finding may indicate one of these other systemic conditions. A history of trauma is found in approximately 25% of LCP cases.
Anatomy
The vascular pattern of the femoral head changes over time with development of the secondary ossification center. The ossification center of the femoral head is usually single but occasionally may begin with several nuclei around 4 months of age, supplied by metaphyseal and lateral epiphyseal arteries, the latter of which arise from the medial circumflex artery. By 8 months of age, the fetal ascending metaphyseal arteries begin to regress with enlargement of the ossification center and by 4-7 years of age the main blood supply is seen arising from the lateral epiphyseal arteries, tightly grouped at the lateral aspect of the head.14 Injury to these lateral vessels is surmised to be the cause of LCP. It is only by the age of 9-10 years that the vascular ligamentum teres provides additional vascular supply by way of the medial epiphyseal arteries, forming an epiphyseal anastomotic network. By adolescence the epiphyseal plate is crossed by adult metaphyseal arteries as fusion occurs.
Radiographic and MRI Examination
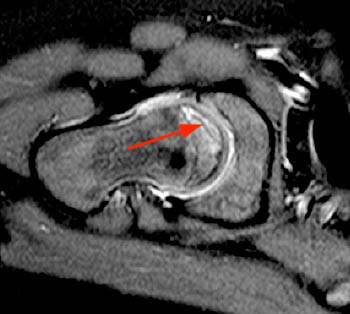
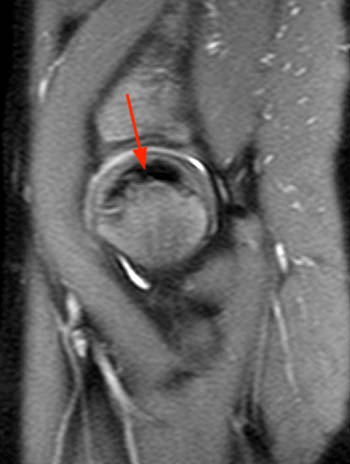
AP and frog leg X-rays in the early avascular stage of the disease may be normal, while MRI is more sensitive in visualizing signal abnormalities in the femoral ossification center. There may be bone marrow edema present, demonstrated by increased signal on T2-weighted or STIR images. An effusion is also usually present, which on X-ray may be seen by recognizing a widened teardrop distance indicating lateral displacement of the ossification center (Waldenstrom’s sign). The ‘crescent’ sign which can be seen on both X-ray and MRI usually indicates a subchondral fracture is present, typically affecting the anterosuperior aspect of the femoral head (10a,11a). This may progress to necrosis of the femoral head, characterized by low signal on all imaging sequences (12a,13a). X-rays and MRI in the later avascular stage may reveal smallness of the femoral ossification nucleus, fissuring/fracturing of the femoral ossification center, flattening and sclerosis of the femoral ossification center or intraepiphyseal gas.13
The reparative and revascular stage of LCP usually occurs over several years and involves resorption of the necrotic bone. Development of metaphyseal cysts/lucencies can occur (14a). Lateral shift of the femoral head may persist, from continuing synovitis and cartilage thickening. Articular surface flattening (coxa plana) is a common later stage finding (15a). Also broadening of the femoral neck and head (coxa magna) can be seen (16a). The physis may also be involved and may exhibit undulation, deepening or bone bridging producing early bone arrest or limb length discrepancies. The incongruence of the femoral head may lead to secondary osteoarthritis.
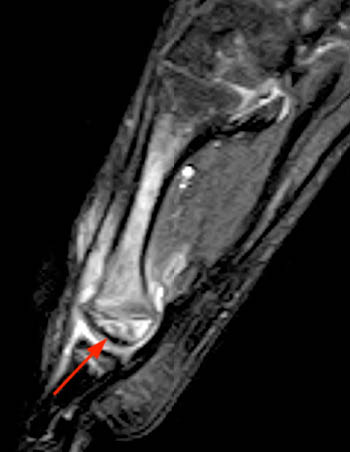
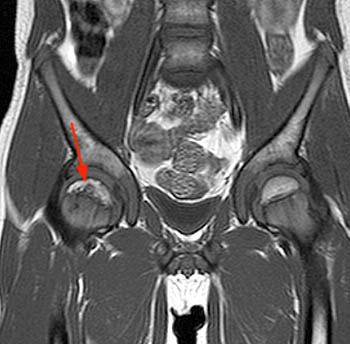
Figure 10:
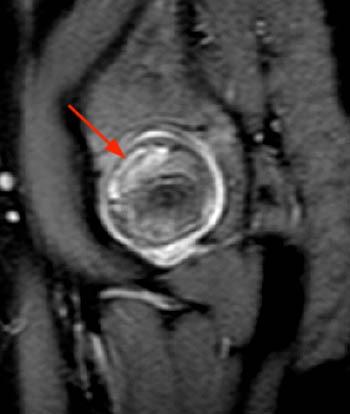
A sagittal fat-suppressed proton density-weighted image in this 10 year-old boy complaining of hip pain reveals a subchondral 'crescent sign' due to a fracture line along the anterosuperior aspect of the femoral ossification center (arrow). There is localized surrounding marrow edema present and a joint effusion is present.
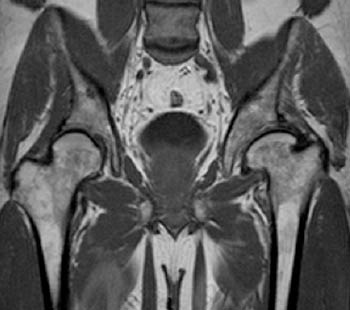
Figure 16:
A coronal fat-suppressed T2-weighted image in a 50 year-old man with left hip pain and a remote history of LCP. Femoral head flattening of the articular surface (coxa plana) and broadening of the femoral neck (coxa magna) are demonstrated. A joint effusion and secondary changes of osteoarthritis are present.
Treatment
It is estimated that 60-70% of hips affected by LCP disease heal spontaneously without functional impairment at maturity; however, a considerable amount of hips progress with pain and secondary degeneration.16 Treatment is variable and based on different criteria. Conservative measures include increasing range of motion utilizing physiotherapy and Botox injections into the adductor muscles. Pain and weight relief measures also are used, including use of variable splints and casts and bed rest. Surgery is typically reserved for older patients with poor range of motion to increase congruity of the hip but no consensus is currently formed on the correct approach to treatment in these patients.17
Conclusion
Articular osteochondroses of the lower extremities can be painful and debilitating disorders, affecting the growing skeleton in children and adolescents. Early recognition by imaging, and in particular MRI, is critical to proper management decisions and preventing long-term complications.
References
1 Siffert RS, Arkin AM. Post-traumatic aseptic necrosis of the distal tibial epiphysis. J Bone Joint Surg. 1950; 32A:691-695.
2 Sakthivel-Wainford, K (2011). Self-Assessment in Musculoskeletal Pathology X-rays. (p. 44) M&K Publishing.
3 Resnick D (1996). Osteochondroses. Bone and Joint Imaging (2nd ed., pp. 960-977) Philadelphia, W.B. Saunders Company.
4 Staheli, LT (2003). Fundamentals of Pediatric orthopaedics. (p.58) Philadelphia, Lippincott Williams and Wilkins.
5 Williams GA, Cowell HR. Köhlers disease of the tarsal navicular. Clin Orthop Relat Res. 1981; 158:53-58
6 Carmont MR, Rees RJ, Blundell CM. Current concepts review: Freiberg’s disease. Foot Ankle Int. 2009;30(2):167-176.
7 Air ME, Rietveld, AB. Freiberg’s Disease as a rare cause of limited and painful revele in dancers. J Dance Med Sci. 2010; 14(1)32-36.
8 Peterson WJ, Lankes JM, Paulsen F, Hassenpflug J. The arterial supply of the lesser metatarsal heads: a vascular injection study in human cadavers. Foot Ankle Intl. 2002 Jun; 23(6) 491-5.
9 Ozonoff MB: The foot. In Ozonoff MB(ed): Pediatric orthopaedic Radiology. Philadelphia, WB Saunders, 1992, pp 304-396
10 Simillie. Treatment of Freiberg’s infraction. Proc R Soc Med. 1967;60:29-31.
11 Mah, CD: Freiberg’s disease. In www.podiatryinstitute.com/pdfs/Update_2008/2008_04.pdf Chapter 4 pp 13-15.
12 Barker DJ, Hall AJ. The epidemiology of Perthes’ disease. Clin Orthop Relat Res 1986; 209:89-94
13 Dillman JR, Hernandez R. MRI of Legg-Calve-Perthes Disease. AJR. 2009 Nov; 193: 1394-1407.
14 Trueta J. The normal vascular anatomy of the human femoral head during growth. The Journal of Bone and Joint Surgery. 1957 May; 39B (2): 358-394
15 Resnick D (1996). Osteochondroses. Bone and Joint Imaging (2nd ed., pp. 960-977) Philadelphia, W.B. Saunders Company.
16 Dillman JR, Hernandez R. MRI of Legg-Calve-Perthes Disease. AJR. 2009 Nov; 193: 1394-1407.
17 Fritz H, Clarke NMP. The management of Legg-Calve-Perthes disease, is there a consensus? J Child Orthop. 2007 March;1(1) 19-25.