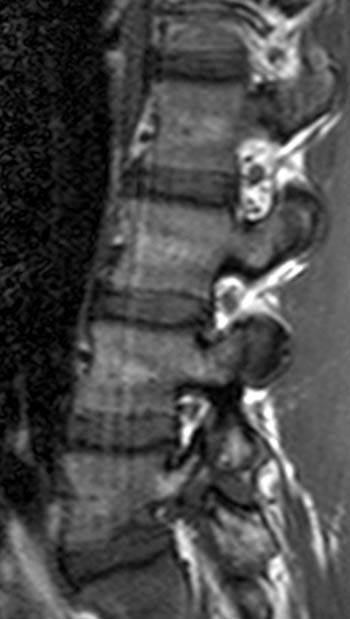
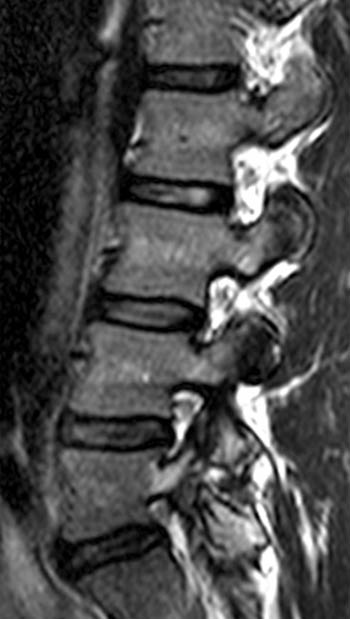
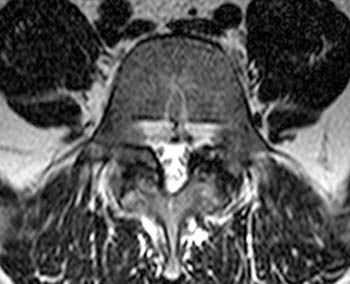
Clinical History: A 22 year-old man presents with right-sided back pain which radiates to his right hip and leg. His symptoms began while running. (1a) T1- and (1b) T2-weighted sagittal and (1c) T2-weighted axial images of the lumbar spine are provided. What are the findings? What is your diagnosis?
Findings
Figure 2:
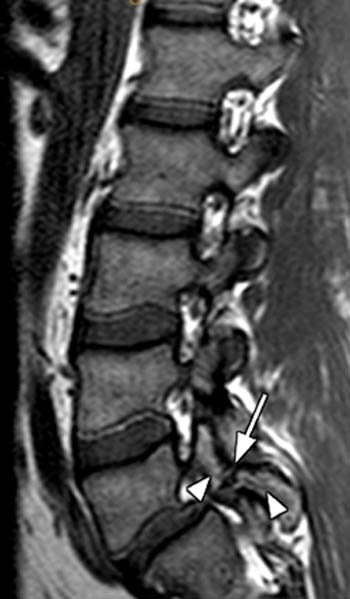
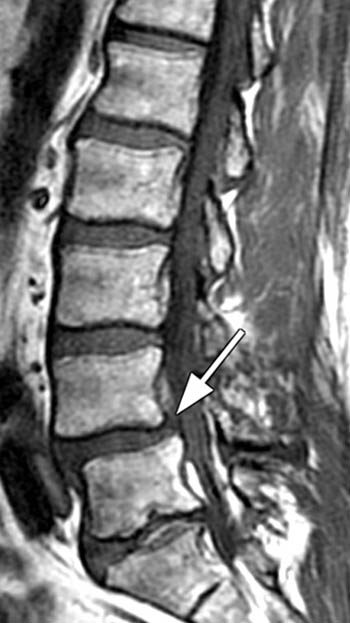
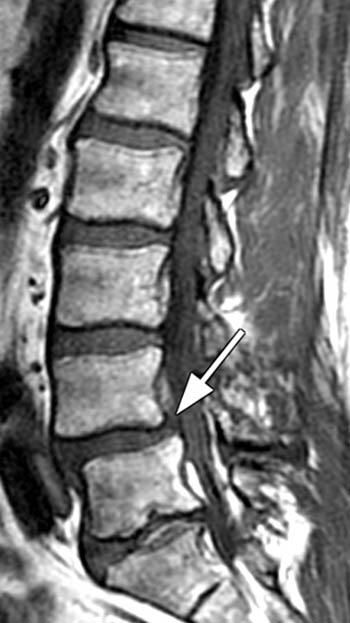
The T1-weighted off-midline sagittal image demonstrates a defect in the L5 pars interarticularis with interruption of the cortex and intermediate signal intensity material in the defect (arrow). Slight anterior subluxation of the L5 vertebral body is seen with respect to the S1 vertebral body. Marrow within the L5 posterior elements is of increased signal intensity on the T1 weighted images (arrowheads).
Figure 3:
The T2-weighted sagittal image obtained at the same location as the image in (2a) again demonstrates the defect in the L5 pars interarticularis (arrow). On this image, the marrow signal in the posterior elements is increased (arrowheads). If the T1-weighted image were not available, this could be mistaken for an acute fracture.
Diagnosis
Bilateral L5 spondylolysis with Grade I spondylolisthesis of L5 on S1.
Discussion
Spondylolysis refers to an osseous defect within the posterior neural arch, most commonly within the pars interarticularis, an isthmus of bone located between the superior and inferior articular processes. Spondylolysis most commonly affects the L5 level (in 85 ” 95% of cases) with the majority of the remaining cases occurring at L4 (5 ” 15% of cases).1 While the exact etiology of spondylolysis is unknown, it is generally believed to represent a stress fracture caused by repetitive loading,2 although there are hereditary and genetic contributing factors.
The prevalence of spondylolysis has been estimated to be between 3% and 6% of the general population.3 The frequency of spondylolysis appears to be higher in athletes, particularly those involved in gymnastics, diving, wrestling, weight-lifting, and throwing.10 Typically, spondylolysis is discovered in childhood or early adulthood, and the frequency of the abnormality does not increase after the age of 20 years.3 The trauma that causes spondylolysis is likely related to the stress of an upright gait in humans, as pars defects are virtually nonexistent in patients who have never walked.11
As in our patient, spondylolysis may lead to spondylolisthesis, a forward (ventral) subluxation of an upper vertebra on a lower vertebra. Wiltse and coworkers have classified spondylolisthesis into five types based upon etiology:12
Type I: Dysplastic. This type is associated with congenital abnormalities of the sacrum and the posterior elements of the lumbar spine.
Type II: Isthmic. Caused by a defect in the pars interarticularis that may be a fatigue fracture, an acute fracture, or an intact but elongated pars.
Type III: Degenerative. Resulting from chronic segmental instability and facet arthrosis.
Type IV: Traumatic. Caused by fractures of the posterior elements not including the pars interarticularis.
Type V: Pathologic. Resulting from localized or diffuse bone disease. Also includes iatrogenic bone loss.
Many patients with spondylolysis are asymptomatic. Of those with symptoms, approximately one-quarter are associated with spondylolisthesis.3 Pain is usually limited to the low back. If the pain radiates, it is usually to the buttocks or the back of the thigh and is often from hamstring tightness rather than lumbar radiculopathy. With associated higher grade spondylolisthesis, however, radiculopathy becomes more common due to associated foraminal stenosis.
MR Imaging Findings
Imaging evaluation of a patient with low back pain typically begins with a series of lumbar spine radiographs. Spondylolysis is usually evident on lateral radiographs, although oblique projections may be useful. On frontal projections, fragmentation of the lamina may be identified.4 If spondylolisthesis is present, it should be graded according to the Myerding system,5 with grade I indicating anterior subluxation of less than 25%; grade II, 25% to 50%; grade III, 50% to 75%; and grade IV, 75% to 100%.
If routine radiographs in a symptomatic patient are unrevealing, further imaging evaluation can include a bone scan, computerized tomography, single-photon-emission computed tomography, or magnetic resonance imaging. As magnetic resonance imaging is one of the primary modalities used to evaluate the lumbar spine, it is important to recognize findings of lumbar spondyolysis.
Spondylolisthesis is readily identified on the midline sagital images. However, as most cases of spondyolysis have normal lumbar vertebral alignment, other findings must be utilized to detect the pars defects. In addition to demonstrating cortical disruption of the pars (A,B), several ancillary findings have been described that may aid in the diagnosis of lumbar spondylolysis.6,7,8 These include a widened anteroposterior diameter of the spinal canal on sagittal images, reactive marrow changes in the posterior elements, and abnormal wedging of the posterior aspect of the vertebral body.
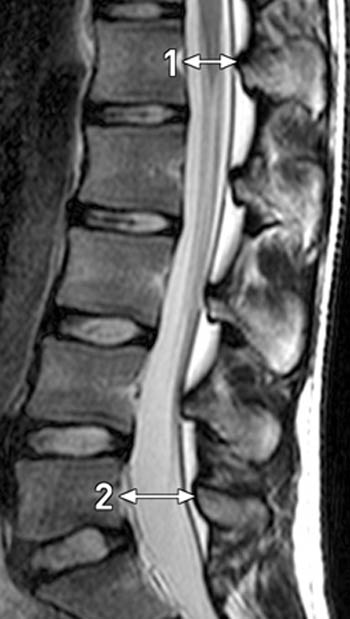
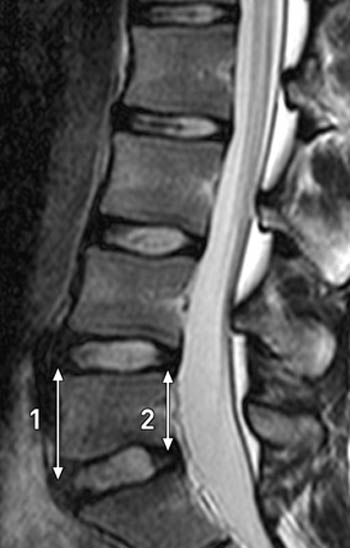
To evaluate the sagittal diameter of the spinal canal, a ratio of the AP diameter at the L5 level to the AP diameter at the L1 level is used. The canal is measured from the posterior cortex of the vertebral body to the anterior aspect of the lamina on a mid-sagittal image. A ratio of 1.25 is normal. This ratio is increased in patients with spondylolysis due to posterior subluxation of the posterior elements, even in cases where no spondylolisthesis is present. Usually, the posterior subluxation of the posterior elements is evident on the mid-sagittal image with a resultant increase in the canal size at the level of the pars defect and actual calculation of a ratio is unnecessary (6a).
Figure 6:
A midline (6a) sagittal T2 weighted image from the same patient in A. The AP diameter of the canal at L1 measures 1.7cm (line 4) and the AP diameter of the canal at L5 measures 2.5cm (line 3). The ratio of L5:L1 is 1.47, which is above the normal value of 1.25. Even without this calculation, it is clear the canal is enlarged at the L5 level with the posterior elements displaced slightly posterior when compared with the posterior elements of the more cranial vertebral bodies.
Reactive marrow changes similar to those observed in patients with degenerative disc disease are identified within the posterior elements adjacent to pars defects in a significant number of patients. In one study, these changes were observed in 40% of patients with spondylolysis and were distributed as a function of age. This observation may be an additional clue that a pars defect is present (E,F) and may be a reflection of the duration of disease. Type I changes (similar to marrow edema – low signal on T1 and high signal on T2 images) are most commonly seen in patients less than 24 years of age. Type II changes (similar to fat signal – high signal on T1 and isointense or high signal on T2 images) are seen in patients with a median age of 35 years. Type III changes (similar to fibrous tissue – low signal on both T1 and T2 images) are seen in patients with a median age of 51 years.7 As both acute and chronic pars defects can demonstrate hyperintensity on routine T2-weighted images, fat-suppressed T2 or STIR sequences are often helpful in order to more easily identify marrow edema suggestive of more recent injury.
Figure 7:
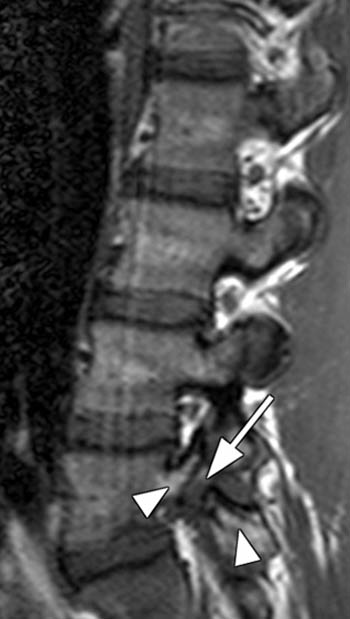
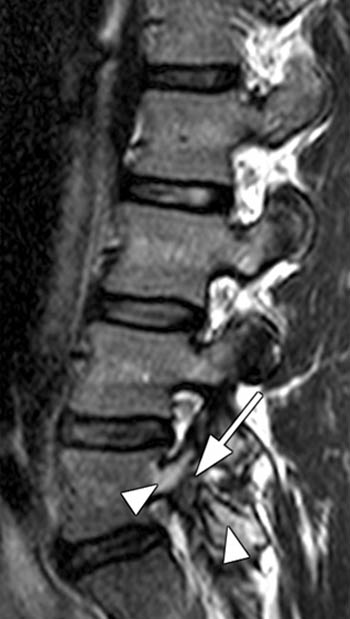
(7a) T1- and (7b) T2- weighted sagittal images of the lumbar spine in a 35 year-old male who presented with 4-5 months of bilateral lower extremity pain are provided. A defect of the pars interarticularis is seen (arrows) with cortical interruption and a resultant grade I spondylolisthesis. Reactive marrow changes are also present adjacent to the pars defect with increased signal within the marrow (arrowheads) on both the T1 and T2 weighted images.
The final ancillary observation that may aid in the detection of spondylolysis is an abnormal wedging of the posterior aspect of the vertebral body at the level of the pars defect. This finding is a well-known radiographic finding that occurs at the level of spondylolisthesis. It is unclear if this finding is an effect of the spondylolisthesis, a predisposing condition, or a combination of both. On sagittal MR images, wedging of the posterior vertebral body is seen both in patients with spondylolisthesis and in those with spondylolysis and no significant subluxation.8 Therefore, such wedging may suggest the presence of pars defects (Figure 8).
Figure 8:
(8a) The same midline sagittal T2 image as seen in Figure 6. In this example, lines have been drawn to show the height of the anterior (line 1) and posterior (line 2) vertebral body. Posterior wedging of the vertebral body is seen, which is an ancillary finding suggesting the presence of spondylolysis at this level.
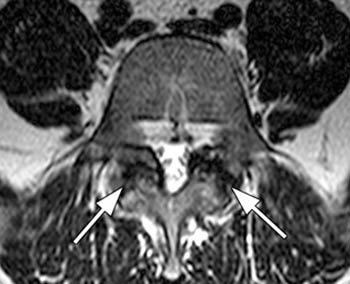
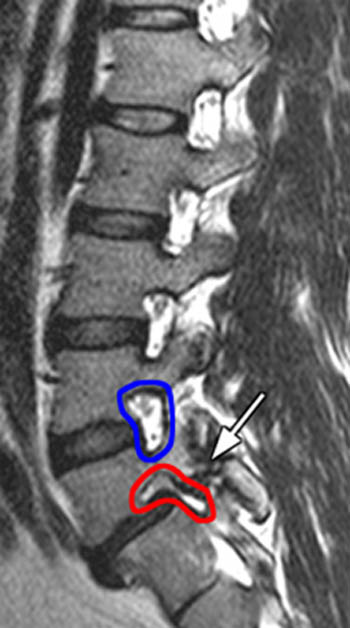
At times, direct visualization of a pars defect is difficult on sagittal MR images, and thus it may be difficult to determine whether a patient with spondylolisthesis has a degenerative origin or if the malalignment is due to spondylolysis. In such cases, characteristic ancillary findings can be utilized to differentiate degenerative spondylolisthesis from isthmic spondylolisthesis. An appearance that we have found to be highly characteristic of isthmic spondylolisthesis is the horizontal neural foramina sign. In patients with spondylolysis, the neural foramina often assume a horizontal configuration on far lateral sagittal images (9a,9b). This feature is not present in degenerative spondylolisthesis, and the configuration also accounts for radiculopathy due to foraminal stenosis in patients with more severe isthmic spondylolisthesis.
Figure 9:
(9a) T1- and (9b) T2-weighted sagittal images in a patient with spondylolisthesis reveal clear pars defects (arrows). A horizontal configuration of the L5 neural foramina is readily apparent (red outline), with resultant foraminal stenosis. Compare this configuration with the normal keyhole appearance of the L4-5 foramina (blue outline). This horizontal configuration is typical in patients with spondylolisthesis due to spondylolysis.
In patients with degenerative spondylolisthesis (10a,11a), the underlying abnormality is intersegmental instability caused by facet arthropathy. These patients will not have horizontal neural foramina, and the associated facet arthropathy, not a feature of isthmic spondylolisthesis, is readily apparent on axial scans.
Treatment and Prognosis
Most patients with spondylolysis or pars stress reactions respond favorably to non-operative treatment. Usually this treatment includes a period of rest or immobilization followed by physical therapy. The role and best type of bracing continue to be debated. In selective cases, epidural steroid injections or selective nerve root blocks help in controlling symptoms. Most authors agree that patients may return to normal activities when they are pain-free, regardless of whether there is radiographic evidence of pars healing.9
After failure of nonoperative management, surgical intervention can be considered. Surgical treatment is more likely in patients with neurologic deficits or spondylolisthesis of Grade III or greater. Various surgical techniques have been utilized, including decompressive laminectomy, posterolateral fusion, interbody fusion, and direct pars repair.10 The patient can usually return to activities once they are pain-free and any fusion has healed.
Conclusion
Spondylolysis is an osseous defect found in both symptomatic and asymptomatic individuals. It predisposes to pathologic intervertebral subluxation or spondylolisthesis, most commonly occurring at the L5-S1 level. The pars defects are thought to represent chronic stress related injuries. Although these often occur during the first decade of life, accompanying vertebral problems develop somewhat later in life. It is important for the interpreter of MR to recognize both the primary and ancillary findings of spondylolysis, and in patients with spondylolisthesis, characteristic MR findings allow differentiation of degenerative versus isthmic causes. MR’s ability to grade disease severity and directly visualize nerve root involvement assists in treatment decisions.
References
1 Standaert CJ, Herring SA, Halpern B, King O. Spondylolysis. Phys Med Rehabil Clin N Am. 11:785, 2000.
2 Jackson DW, Wiltse LL, Dingeman RD, Hayes M. Stress reactions involving the pars interarticularis in young athletes. Am J Sports Med. 9:304 , 1981.
3 Fredrickson BE, Baker D, McHolick WJ, Yuan HA, Lubicky JP. The natural history of spondylolysis and spondylolisthesis. J Bone Joint Surg Am. 66:699, 1984.
4 Amato M, Totty WG, Gilula LA. Spondylolysis of the lumbar spine: demonstration of defects and laminal fragmentation. Radiology.1984; 153:627 -9.
5 Myerding HW. Spondylolisthesis. Surg Gynecol Obstet. 54:371, 1932.
6 Ulmer JL, Mathews VP, Elster AD, King JC. Lumbar spondylolysis without spondylolisthesis: recognition of isolated posterior element subluxation on sagital MR. Am J Neuroradiology. 7:1393, 1995.
7 Ulmer JL, Elster AD, Mathews VP, Allen AM. Lumbar spondylolysis: reactive marrow changes seen in adjacent pedicles on MR images. Am J Roentgen. 164:429, 1995.
8 Ulmer JL, Mathews VP, Elster AD, Mark LP, Daniels DL, Mueller W. MR imaging of lumbar spondylolysis: the importance of ancillary observations. Am J Roentgen. 169:233, 1997.
9 Jackson DW, Wiltse LL, Dingeman RD, Hayes M. Stress reactions involving the pars interarticularis in young athletes. Am J Sports Med. 9:304 , 1981.
10 Bono CM. Low-back pain in athletes. J Bone Joint Surg Am. 86:382, 2004.
11 Rosenberg NJ, Bargar WL, Friedman B. The incidence of spondylolysis and spondylolisthesis in nonambulatory patients. Spine. 6:35, 1981.
12 Wiltse LL, Newman PH, Macnab I. Classification of spondylolysis and spondylolisthesis. Clin Orthop. 117:23, 1976.