Clinical History:
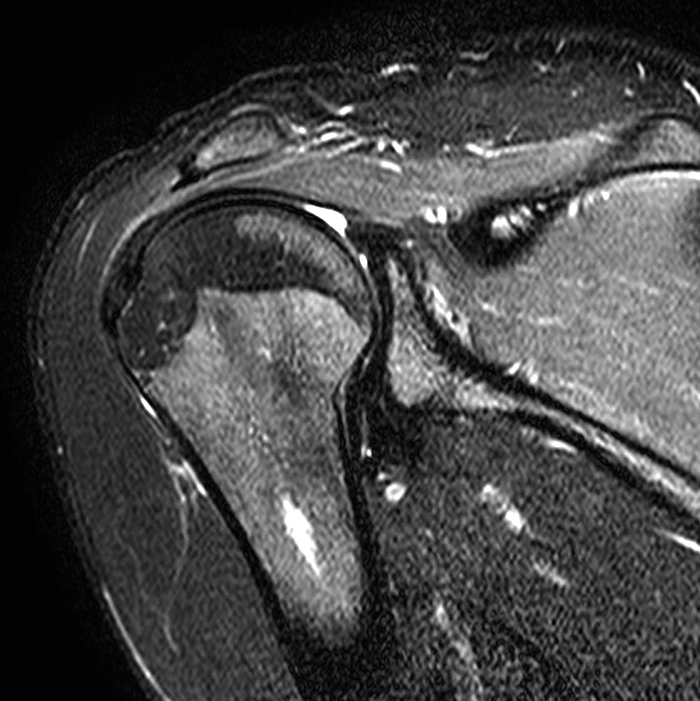
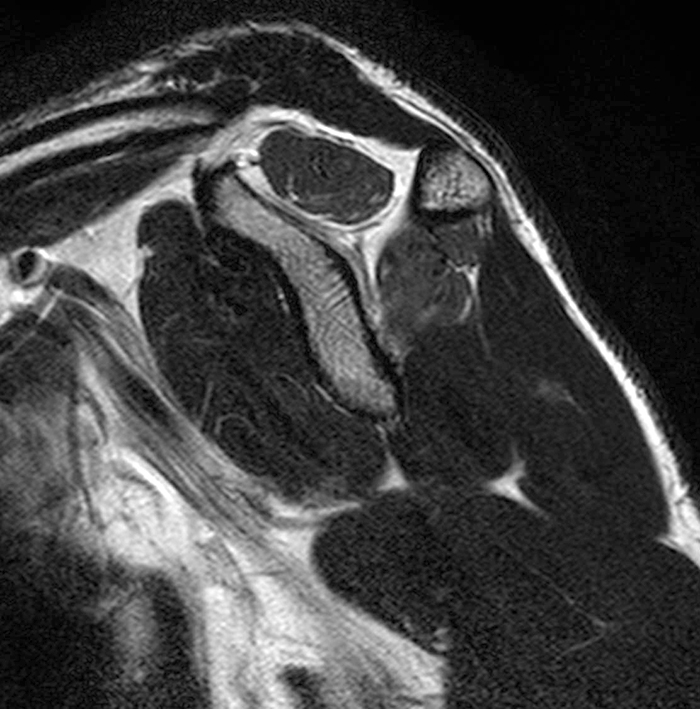
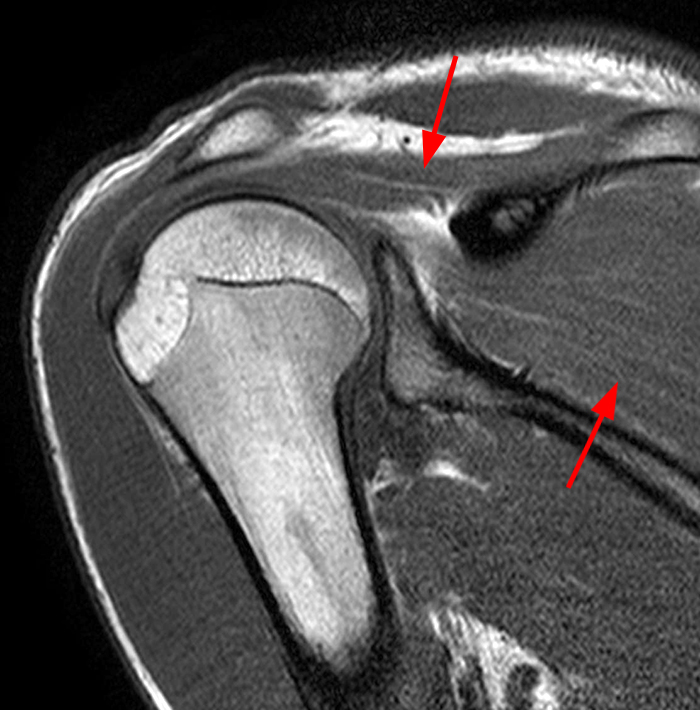
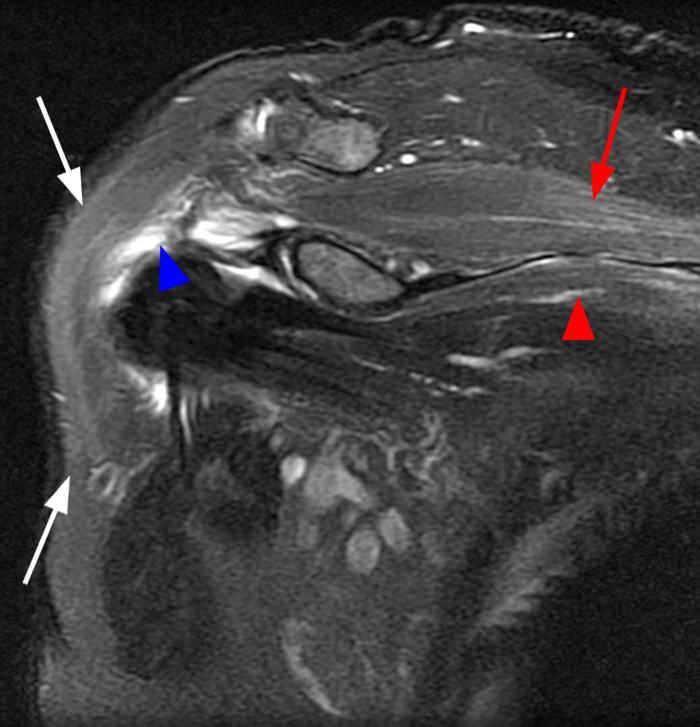
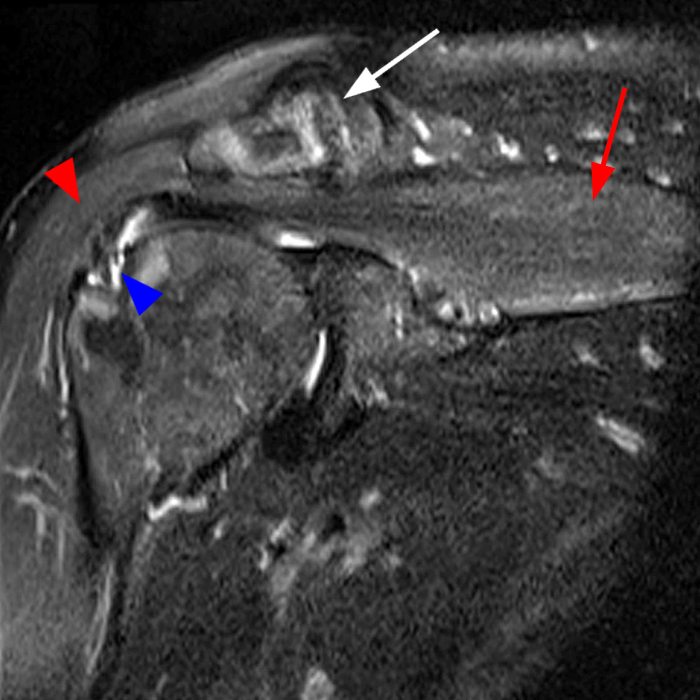
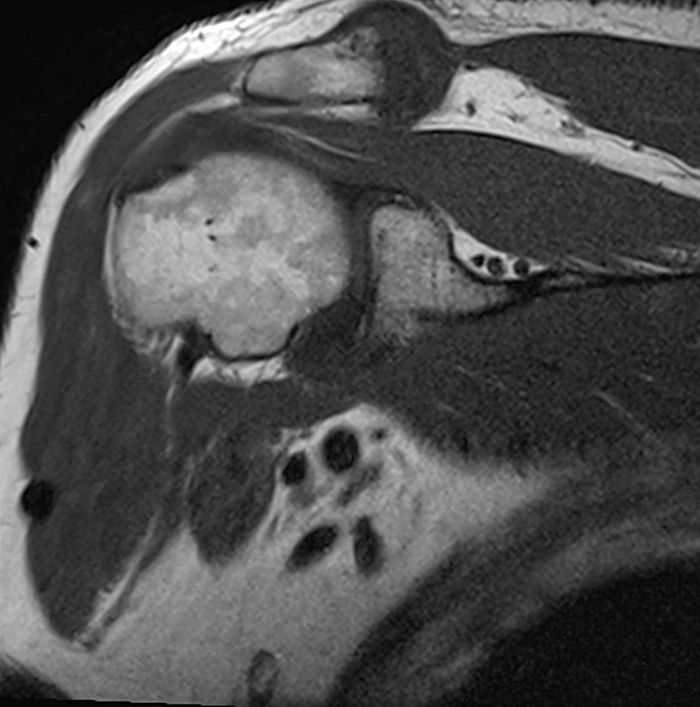
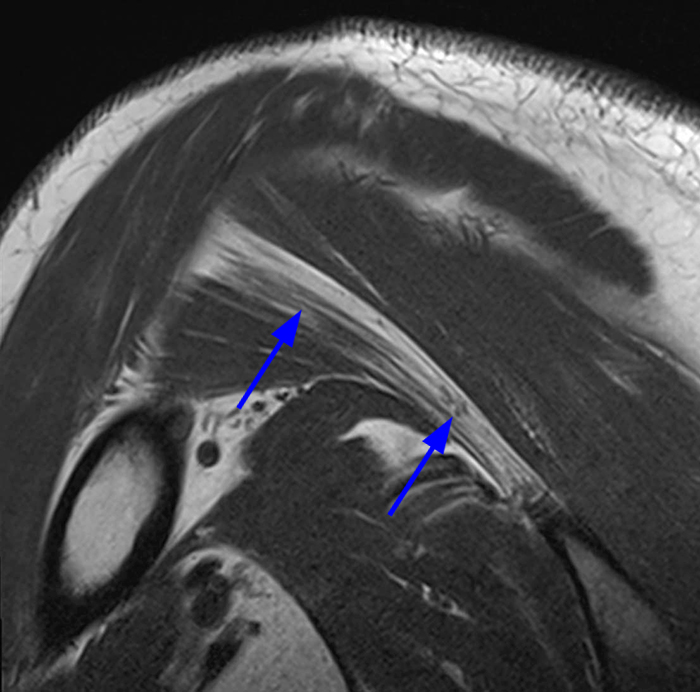
A 16 year-old male presents for MRI with a history as follows: “Shoulder pain, status post subluxation event. External rotation weakness. Evaluate for rotator cuff tear, SLAP lesion, or paralabral cyst.” Fat-suppressed T2-weighted coronal (1A), T1-weighted coronal (1B), and T2-weighted sagittal (1C) images are provided. What are the findings? What is your diagnosis?
Findings:
Figure 2:
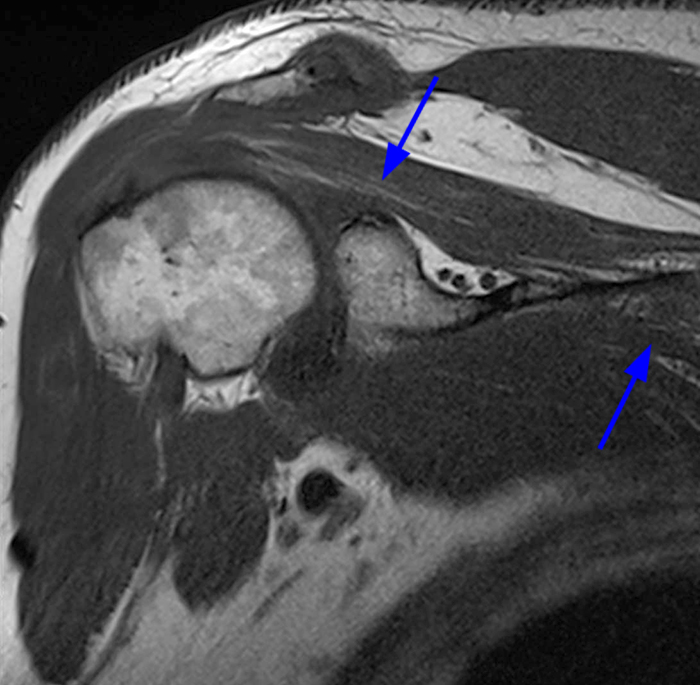
Image 2A demonstrates diffuse homogeneous edema throughout the supraspinatus and infraspinatus muscles, and image 2B reveals mild fatty atrophy of both muscles (arrows). Image 2C shows mild diffuse edema and fatty atrophy of the supraspinatus and infraspinatus muscles (arrows). There is no paralabral cyst in the suprascapular notch (dotted outline) and the remainder of imaged musculature is normal. Note how the order history frequently is, as in this case, incomplete and/or misleading. EMG performed approximately 2 weeks after the MRI showed an incomplete suprascapular neuropathy with evidence of supraspinatus and infraspinatus denervation.
Diagnosis:
Neurogenic edema and mild fatty atrophy in the suprascapular nerve distribution, suspected Parsonage-Turner syndrome.
Introduction:
Parsonage-Turner syndrome (PTS), known by other names such as brachial neuritis and amyotrophic neuropathy, is a clinical syndrome involving the peripheral nervous system, the vast majority involving nerves arising from the brachial plexus. It has protean manifestations that vary widely from patient to patient, most frequently an abrupt onset of severe pain in the shoulder, neck, and/or arm followed by highly variable weakness and sensory deficits in the upper extremity. While PTS is a clinical diagnosis, MRI can be very helpful in suggesting the diagnosis when the presentation is unclear.
Etiology, distribution, and clinical features:
The etiology of PTS is uncertain. It is thought to occur after exposure to an autoimmune trigger that targets the peripheral nervous system in susceptible individuals. 1 There are two forms: idiopathic and hereditary. Hereditary PTS is rare, involving roughly 200 families as an autosomal dominant trait with 80% penetrance and an approximately 75% rate of recurrence.2 The idiopathic form, of which the remainder of this section will detail, is much more common.
The incidence of PTS has been estimated to be 2-4/100,000 patients/year.2 Males are affected more frequently than females in a ratio of 2:1 to 11.5:1.2,3,4,5,6 While individuals affected are usually in the 2nd or 3rd decade2, it can occur in a wide range of ages, from neonates to patients in their 8th decade of life.5 PTS can occur bilaterally in anywhere from 10-33% of patients;2,3 the onset and presentation usually is asymmetric and subclinical in one of the upper extremities.4 Up to 25% have recurrent PTS which often differs from the initial attack.2 Recurrences are usually shorter and milder and may occur within months of the initial episode.4
PTS is felt most likely to be an immune-mediated response to nerves as approximately half of cases are preceded by a potential immune system trigger.2 It is felt that underlying genetic factors play some role as the incidence of recurrence is higher than that of initial episodes in the general population.2
Two of the most frequent associations with PTS include recent viral infection (flu-like illness or upper respiratory infection) and recent immunization,3,7 suggesting a viral infection that directly involves nerves or an autoimmune response to viral infection or viral antigen in immunization.6 Clustered outbreaks of PTS have occurred.8,9 Other associations include recent surgery (even within 24 hours), childbirth, trauma, and strenuous exercise.3,4,6,7,10 Interestingly, HIV and Lyme disease can cause similar symptoms as PTS.2 In addition, neuroborreliosis has been found to cause PTS.11
While most commonly patients have severe pain at onset followed by variable weakness, there may be a more insidious onset.10 Recovery may begin within a month or no recovery may be seen after years.10
The hallmark presentation of PTS, occurring in the vast majority of patients is an abrupt onset of severe, relentless pain in the neck, shoulder, and/or arm, frequently causing awakening from sleep, lasting for 4 weeks on average,10 followed by the rapid onset of multifocal weakness and atrophy, usually in the upper extremity, with variable sensory deficits that are not usually pronounced.2,10 Pain at times can be intermittent.2 Additionally, severe pain at onset can occasionally be absent; in a retrospective study performed over nine years by a hand surgeon practice, 11 out of 38 patients with PTS had no severe pain at onset,1 although only 3.7% of patients had a painless onset in a much larger study.10 The major symptoms are variable. While typically the main symptoms are pain and weakness, some can have any combination of pain, weakness, and numbness or only pain or weakness.5 Frequently after the initial pain subsides, patients can have pain for weeks to months with movement and at the origin or insertion of affected or nonaffected/compensatory muscles, particularly in periscapular, cervical, and/or occipital regions.2 While not usually clinically prominent, the majority have sensory deficits on exam, often causing hypaesthesia and/or paresthesia over the deltoid and lateral upper arm region10 and the lateral cutaneous antebrachial nerve which supplies skin of portions of the forearm.2 Motor and sensory symptoms are completely variable as any portion of the brachial plexus or the nerves derived thereof can be involved, in any combination.2 A major clue to the diagnosis is that sensory symptoms or pain often do not correspond to sites of weakness.2 Alternatively, the clue to the diagnosis may be random upper extremity weakness in those who do not have the constant debilitating pain at onset.2,6 The symptoms and course are highly variable–from relatively mild (e.g. loss of pinch grip) and short to severe with symptoms (e.g. pain and weakness of arm or hand) lasting years.2 The greater the severity and duration of pain, weakness, or both, as well as involvement of multiple nerves usually leads to longer recovery times.4
Any portion of the brachial plexus and any muscle supplied by the plexus from the neck (e.g. sternocleidomastoid, neck extensors) to the hand (e.g. dorsal interossei) can be involved.10 The upper trunk of the brachial plexus (C5 and C6 — periscapular and perihumeral muscles) is most frequent involved (71%), either with (50%) or without (21%) involvement of the long thoracic nerve manifesting as scapular winging or instability.2 In a retrospective review of 27 patients diagnosed with PTS either clinically and/or with EMG using shoulder MRI, the suprascapular nerve was affected in the vast majority (97%; 50% isolated involvement), the axillary nerve affected in 50% (3% isolated involvement), and the subscapular nerve in 3%.3 In another retrospective review of 26 patients diagnosed with PTS either clinically and/or with EMG using shoulder or brachial plexus MRI, the suprascapular nerve was also affected most frequently (89%). 88% showed involvement of muscles in more than one nerve territory as well as a few with involvement of the latissimus dorsi, pectoralis, and rhomboid muscles (which are more completely imaged or, in the case of the rhomboids only imaged, on brachial plexus MRI).5 Women are affected by involvement of a middle or lower brachial plexus distribution twice as frequently (23%) as men (11%).2 In more distal involvement, the anterior and posterior interosseous nerves are some of the most frequently involved.1 While PTS affects the brachial plexus and its peripheral nerves predominantly, nerves outside the plexus (e.g. phrenic nerve, recurrent laryngeal nerve, lumbosacral nerve) can occasionally be involved.10
Diagnosis of Parsonage-Turner Syndrome:
MRI
The exact cause of denervation-related muscle edema is unclear. It is likely due to an increase in extracellular water and/or intramuscular blood volume,12,13,14,15 the extracellular water accumulating due to lack of muscle contractility.15 It is unclear exactly when muscle edema due to denervation can be detected on MRI. Experimentally it has been detected within two hours and clinically within four days.12 However, MRI has been found to inconsistently detect muscle edema in the initial weeks after complete nerve disruption.16 It is generally accepted that muscle edema due to denervation is consistently seen anywhere from two to four weeks following onset.14,17 Muscle atrophy with or without fat infiltration usually quickly follows the presence of intramuscular edema and is seen within months, best detected on T1-weighted or non-fat-suppressed fluid-sensitive sequences. If intravenous contrast is given, there usually is enhancement of the edematous denervated musculature.12,17 At some time (months) following the initial denervation injury the muscle edema will usually resolve or be negligible and the predominant finding is atrophy with or without fat infiltration. The exception are cases where reinnervation occurs rapidly and no residual edema or fatty atrophy is seen. Fat infiltration (and related muscle atrophy) is irreversible; the degree of fatty atrophy is important to describe as it is proportional to permanent loss of strength.
In PTS, edema in affected muscles is typically diffuse, either homogeneous or heterogeneous. Heterogeneous involvement may include “watery” edema, particularly about the musculotendinous junctions (Figure 3).
Figure 3:
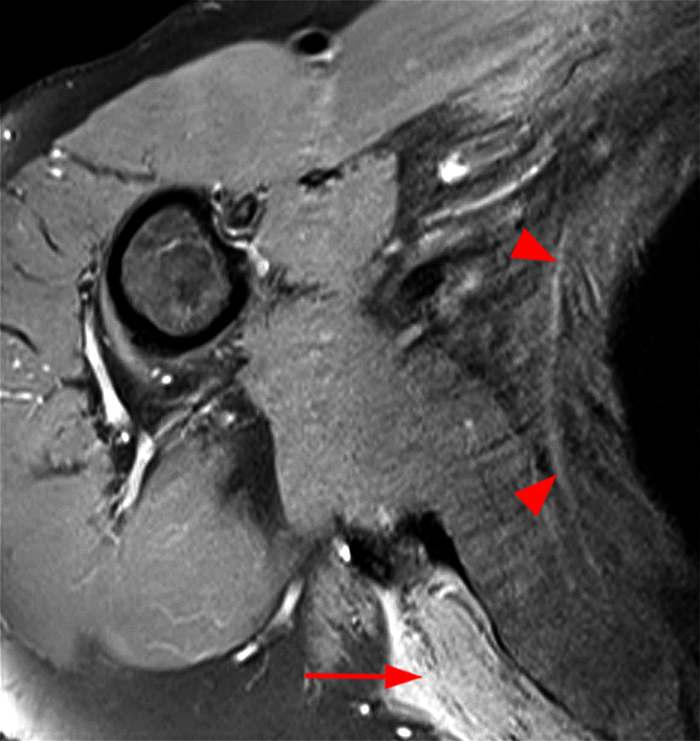
27 year old male with history of shoulder pain with limited range of motion and weakness and no known injury. Fat-suppressed T2-weighted coronal image (3A) reveals heterogeneous edema in the infraspinatus muscle with prominent fluid-like signal about its musculotendinous junction (arrow). The corresponding fat-suppressed proton density-weighted axial image (3B) shows similar findings in the supraspinatus muscle and again in the infraspinatus muscle (arrows). EMG performed 1 week later confirmed suprascapular neuropathy.
While at times the edema may be striking and obvious, at other times it may be relatively mild and may be difficult to detect in studies with inhomogeneous fat suppression. Additionally, muscles in the same nerve distribution may be affected to different degrees, possibly due to collateral nerve supply or fibers that may be affected at different rates after nerve injury16 (Figure 4). Perifascial fluid and/or subcutaneous edema adjacent to affected musculature is not a feature of PTS and should suggest another diagnosis. It may be impossible to differentiate preexisting isolated teres minor denervation edema and/or fatty atrophy (see Radsource Web Clinic – February 2018), or quadrilateral space syndrome with similar findings involving both the teres minor and deltoid muscles from PTS. Clinical history is key in this setting.
Figure 4:
A 77 year-old male presents with severe weakness, pain exacerbated by movements, and limited range of motion after lifting a heavy object 3 months previously, diagnosed clinically with PTS. A fat-suppressed T2-weighted coronal image demonstrates mild heterogeneous edema in the supraspinatus muscle (red arrow) as well as similar edema in the superior subscapularis muscle (red arrowhead); the superior subscapularis edema is presumed due to involvement of the upper subscapular nerve (arising from C5 and C6), or due to collateral innervation from the affected suprascapular nerve. Note the inhomogeneous fat suppression laterally (bracketed by white arrows), interfering with evaluation of deltoid muscle signal. The blue arrowhead indicates subacromial-subdeltoid bursal fluid. Note that it is unclear from the history whether the limited range of motion is due to guarding. Limited passive range of motion is not typical for PTS and should suggest another diagnosis (e.g. adhesive capsulitis).
As the suprascapular nerve is most frequently affected in PTS, any patient with extensive edema in the supraspinatus and infraspinatus muscles without evidence of nerve entrapment or compression (e.g. paralabral cyst in suprascapular notch), history of recent trauma to the brachial plexus, or acute full-thickness tear of the tendons should be suspected to have PTS. PTS should also be high on the differential in any patient with diffuse edema in muscles supplied by different nerves given its propensity for multifocal involvement. If suspected, PTS should be put at the top of the impression to prevent potentially unwarranted surgery (Figures 5 and 6).
Figure 5:
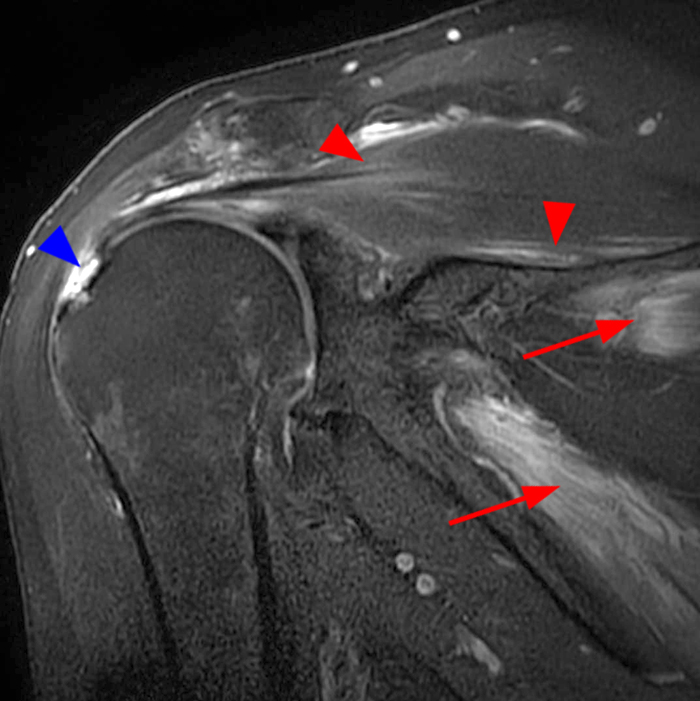
A 59 year-old male presents with a history of shoulder pain and weakness after a lifting injury 19 days prior to MRI--rule out cuff tear. A fat-suppressed T2-weighted coronal image (5A) demonstrates diffuse mildly heterogeneous edema in the supraspinatus (red arrow) and deltoid (red arrowhead) muscles, moderate acromioclavicular osteoarthritis (white arrow), and a partial articular surface tear of the supraspinatus tendon (blue arrowhead). A corresponding T1-weighted coronal image (5B) shows no corresponding muscle atrophy. An additional more posterior fat-suppressed T2-weighted coronal image (5C) reveals similar heterogeneous edema in the infraspinatus (red arrow), deltoid (red arrowhead), and teres minor (short red arrow) muscles. The corresponding T1-weighted coronal image (5D) demonstrates severe fatty atrophy of the superior fibers of the teres minor muscle (blue arrows); the teres minor findings are likely incidental and reflect idiopathic chronic denervation change, particularly given the isolated severe atrophy of its superior fibers as well as having disproportionate atrophy compared to the supraspinatus and deltoid muscles.
Figure 6:
(Figure 6) The patient underwent arthroscopic biceps tenotomy and subacromial decompression with distal clavicle resection just over 2 months later. At postsurgical follow-up the patient had continued weakness with inability to lift the arm. An EMG was performed revealing suprascapular neuropathy. Following the EMG, approximately 3 months following surgery, the patient had repeat MRI. A fat-suppressed T2-weighted coronal image (6A) demonstrates more intense edema in the supraspinatus muscle (red arrow) than on the initial study, new edema in the superior subscapularis muscle (red arrowhead), decreased edema in the deltoid muscle (short red arrow), as well as biceps tenotomy (blue arrow) and acromioplasty (blue arrowhead). There is new fatty atrophy of the supraspinatus and superior subscapularis muscles (blue arrows) on the corresponding T1-weighted coronal image (6B). Similarly, a fat-suppressed T2-weighted coronal image (6C) reveals more intense edema in the infraspinatus muscle (red arrow) than on the former exam. The corresponding T1-weighted coronal image (6D) shows new infraspinatus fatty atrophy (blue arrow). The teres minor muscle (not shown) was unchanged.
On reviewing the clinical notes, the patient presented initially with the sudden onset of constant, severe shoulder pain and weakness—a history much more suspicious for PTS than given in the order history on the initial MRI, confirmed following surgery by EMG. It is felt likely the deltoid muscle was involved, albeit to a lesser degree given its less prominent edema. Perhaps it was not found to be abnormal on EMG due to reinnervation, consistent with the minimal edema seen on the subsequent MRI.
In PTS it should be stressed that denervation edema and/or fatty atrophy seen on shoulder MRI may only image a portion of affected musculature. There may be limited or no inclusion of the serratus anterior muscle on shoulder MRI exams, or it may be unusually thin and/or suboptimally seen due to motion artifact from the adjacent lung or vessel-related phase-encoding artifact (Figure 7). Any combination of these factors is suspected to be the reason why involvement of the long thoracic nerve in PTS is seldom mentioned in MRI studies of PTS.
Figure 7:
Figure 7A.
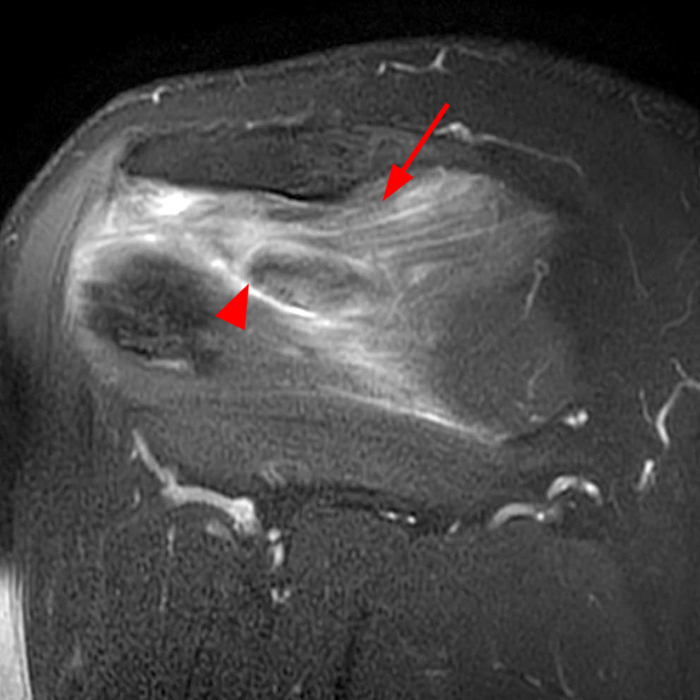
A fat-suppressed proton density axial image from the same patient in Figure 5 and 6 demonstrates linear high signal in a portion of a relatively thin serratus anterior muscle (arrowheads) which is suboptimally imaged due to artifact. Note again the infraspinatus denervation edema (arrow).
More distal upper extremity musculature may be affected in PTS and not included on shoulder MRI. MRI of the brachial plexus includes a larger field of view and can detect affected musculature in PTS patients that will obviously not be detected by shoulder MRI (e.g. rhomboids), as well as potentially subclinical contralateral involvement. As PTS may only affect distal upper extremity musculature the denervation edema and/or fatty atrophy in such cases will not be detected by shoulder MRI. Thus, shoulder MRI may give an incomplete picture of the totality of affected musculature by PTS. While usually thought of as affecting the shoulder girdle musculature, PTS should be considered when neurogenic edema in the absence of an identifiable cause (e.g. mass compression) is seen when imaging the more distal upper extremity. Additionally, although the phrenic nerve is infrequently affected by PTS, it is recommended to pay close attention to the localizer sequence to evaluate for associated diaphragmatic elevation.
MRI can detect reinnervation as well, manifesting as resolution of muscle edema and atrophy.16 Reinnervation is necessary within three months of injury for optimal recovery of denervated musculature.13
It should be stated that the sensitivity of MRI to detect PTS is unknown.3 In one of the largest studies of MRI in PTS patients, those with a clinical diagnosis of PTS but with negative MRI findings were excluded from analysis (number not given).5 There may well be cases that MRI does not detect, likely those imaged very early or late in the course, or in cases of mild denervation edema with poor or absent fat suppression. Later stage PTS may only manifest as fatty muscle atrophy and, as such, the diagnosis may not be considered.
Abnormalities of nerve morphology and signal can be seen in patients with PTS. Nerve constrictions can frequently be seen on brachial plexus MRI.18 In patients with longstanding PTS with minimal or no recovery of function a “bullseye sign” (peripheral hyperintensity and central hypointensity perpendicular to the nerve’s long axis) just proximal to hourglass constrictions of affected nerves can be seen.19 MR neurography may show the affected nerves to be diffusely enlarged and hyperintense.20
EMG
EMG detects electrophysiological abnormalities in denervated muscles and may be used to follow the course of changes to include reinnervation. Electrophysiological changes mirror MRI findings in denervation and reinnervation.12 EMG, like MRI, is unreliable early after the onset of denervation. As such, it is generally accepted to delay EMG evaluation for approximately three weeks following onset.1,7,16
Nerve conduction studies can show reduction in sensory nerve action potential amplitudes in different brachial plexus branches (excluding radiculopathy).2 The needle portion of the exam is key as PTS is believed to be an axonal process.6 The needle portion of the exam allows detection of severity of muscle involvement and can confirm a multifocal plexus distribution as occurs in PTS.2 EMG can help to differentiate PTS from traumatic upper trunk lesions and other plexus lesions, as well as exclude a suspected peripheral nerve compressing lesion.4
EMG is operator-dependent. It can be difficult to sample certain muscles (e.g. the serratus anterior). For obvious reasons denervated muscles will not be detected by EMG if they are not sampled. MRI may detect denervated musculature that is unsuspected and thus may not be sampled by EMG.16
Potential mimics of Parsonage-Turner Syndrome on MRI
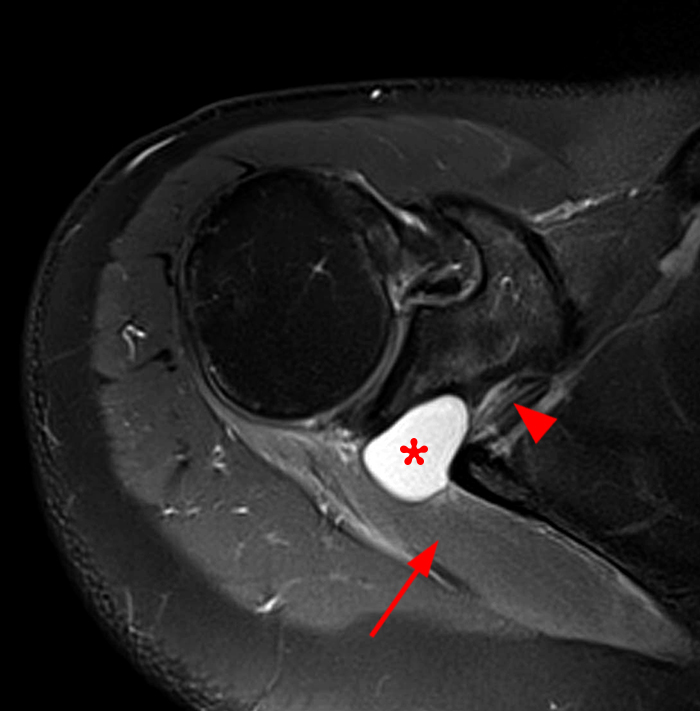
Muscle strains are common and typically show feathery edema on MRI, classically about the musculotendinous junctions but also frequently in the periphery of the muscle (Figure 8). Strains can be fairly extensive, sometimes with perifascial fluid. An acute cuff tear often has extensive and “watery”, fluid-like edema throughout all or a large portion of the muscle (Figure 9).
Figure 8:
Figure 8.
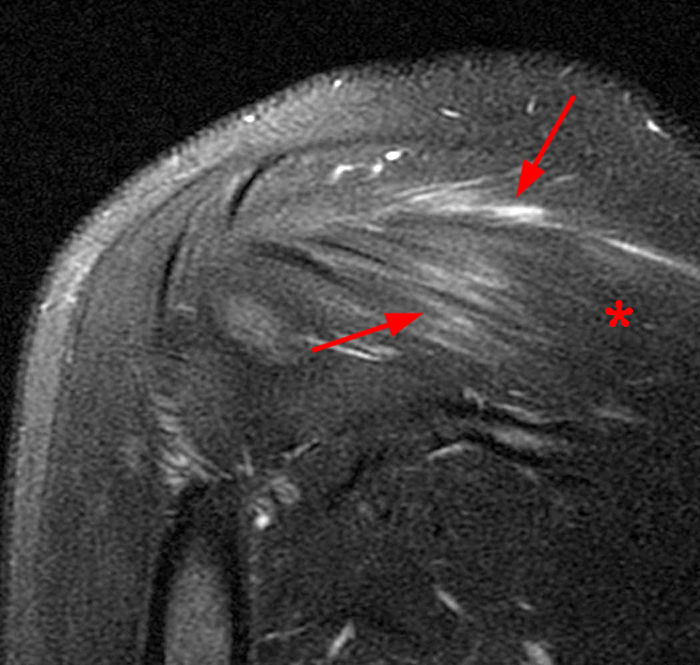
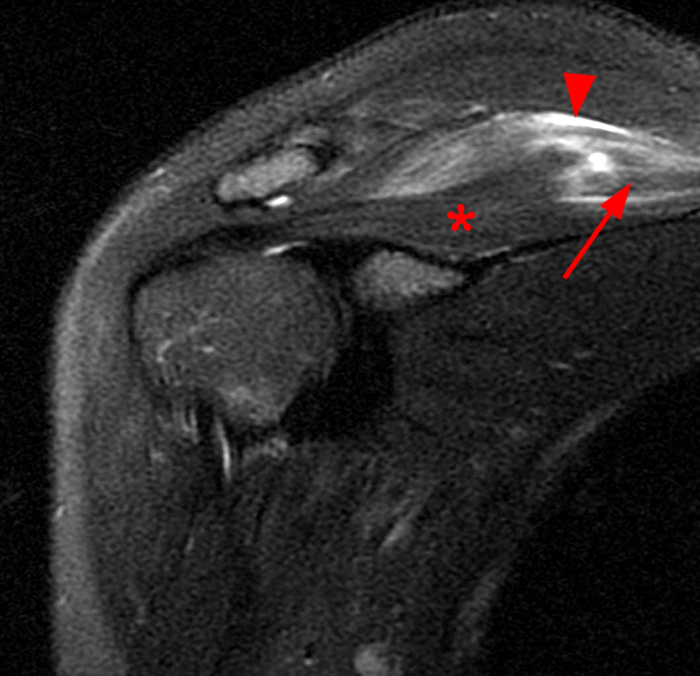
Images from a 28 year-old female with a history of shoulder pain and decreased range of motion. A fat-suppressed T2-weighted coronal image (8A) demonstrates feathery edema centered about the musculotendinous junction and cranial periphery of the infraspinatus muscle (arrows). The medial portion of the muscle is normal (asterisk). There is more extensive feathery edema in the supraspinatus muscle (red arrow), as well as perifascial fluid cranially (arrowhead), but a portion of it too is spared of edema (asterisk) on the fat-suppressed T2-weighted coronal image (8B). A fat-suppressed T2-weighted sagittal image (8C) shows the edema of both muscles largely about the musculotendinous junctions (arrows) with perifascial fluid again seen along the supraspinatus muscle (arrowhead). Edema localized about the musculotendinous junctions as well as peripherally and perifascial fluid is consistent with muscle strains. In contrast, edema will diffusely involve affected musculature without perifascial fluid in individuals with PTS (as in denervation edema in general). This diminutive woman had been participating in hand-to-hand defensive training drills at the Police Academy when she threw an opponent over and felt a pop and pain in the shoulder.
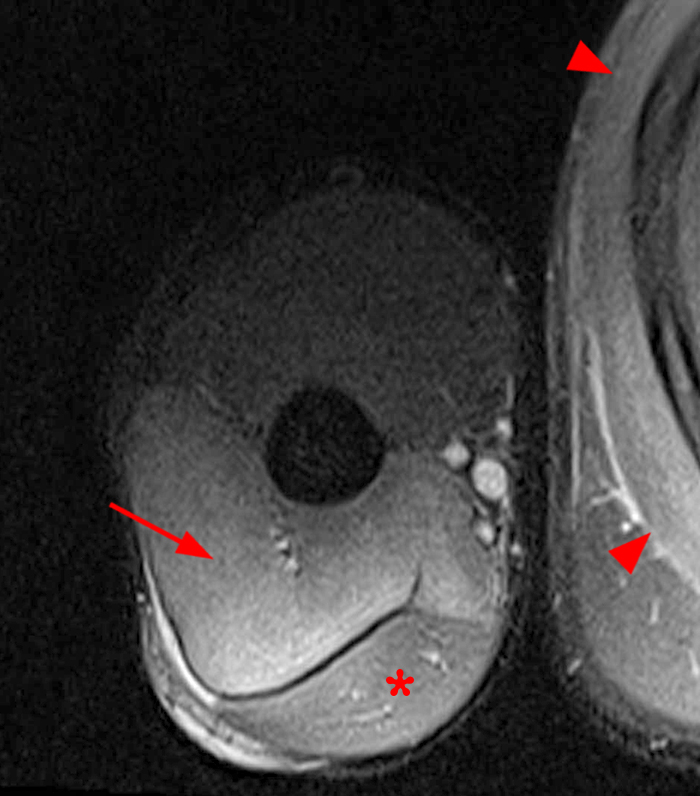
Figure 9:
Figure 9.
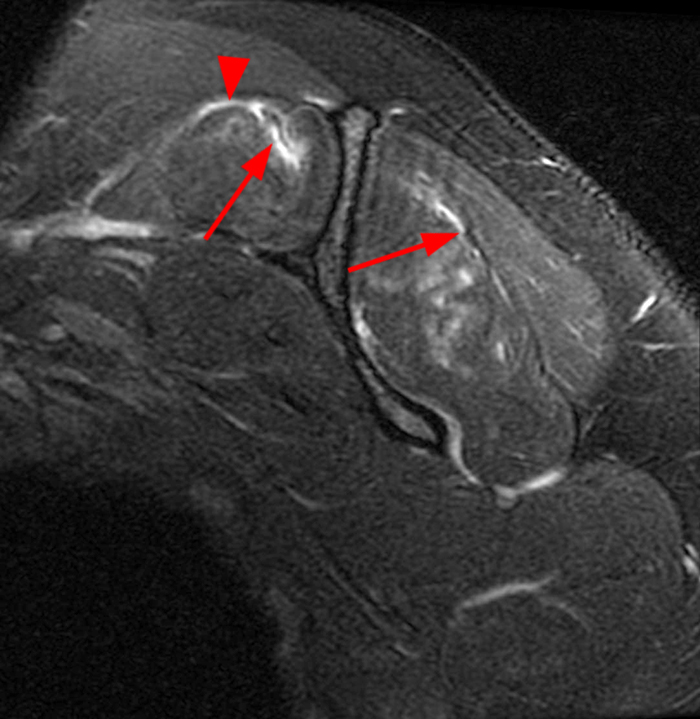
A 60 year-old male presents with shoulder pain, popping, and weakness. Fat-suppressed T2-weighted coronal images are provided. In 9A there is diffuse “watery” infraspinatus muscle edema (red arrow) due to an acute full-thickness retracted tear of its tendon (arrowhead). In 9B there is mild edema about the musculotendinous junction and the caudal periphery of the supraspinatus muscle (red arrowheads), representing a mild strain. Again seen is edema in the infraspinatus muscle (red arrows) with a normal interposed subscapularis muscle. The blue arrowhead points to the insertional component of the infraspinatus tendon tear.
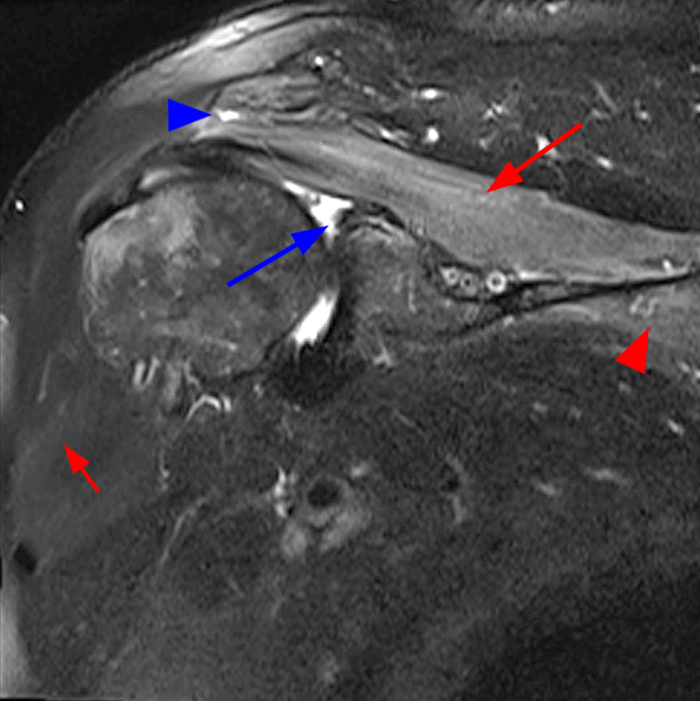
Impingement of the suprascapular nerve in the suprascapular notch causes denervation of both the supraspinatus and infraspinatus muscles, whereas impingement of the nerve more distally in the spinoglenoid notch causes isolated denervation of the infraspinatus muscle as the nerve branch to the supraspinatus muscle exits the suprascapular nerve more proximally. Paralabral cysts, by far and away the most frequent etiology, as well as other masses, varicosities, and vascular malformations can compress the nerve in the suprascapular and/or spinoglenoid notch (Figure 10).
Figure 10:
Figure 10A.
A 61 year-old male presents with shoulder pain, weakness, and decreased range of motion radiating into neck and arm for 2.5 months. A fat-suppressed proton density-weighted axial image demonstrates diffuse edema in the infraspinatus muscle (arrow). A large paralabral cyst is present in the spinoglenoid notch, extending into the posterior portion of the suprascapular notch (asterisk) along the course of the suprascapular neurovascular bundle (arrowhead). The supraspinatus muscle was normal (not shown).
Other causes of suprascapular nerve impingement in the suprascapular notch include a calcified or thick transverse scapular ligament and anterior coracoscapular ligament.3 The axillary nerve can also be compressed by a mass in the quadrilateral space, most frequently a paralabral cyst, manifesting as muscle denervation of the teres minor and deltoid muscles.
Cervical spine disease may be difficult to distinguish from shoulder pathology and at times may masquerade as PTS. As expected, disc impingement on cervical spine root(s) can be seen on shoulder MRI as denervation edema.21 In this setting correlation of findings on both studies should be made to ensure the muscle edema is in a ]correlating to the level of nerve root impingement. Clinically, pain, weakness, and/or sensory deficits should be in the same nerve distribution in cervical radiculopathy, unlike in PTS. Likewise, Spurling’s test is often positive in cervical radiculopathy, unlike in PTS.4
Muscle contusions typically manifest as edema in the periphery of the muscle, often at a muscle-bone interface. Perifascial fluid and overlying subcutaneous edema is seen, not features of PTS.
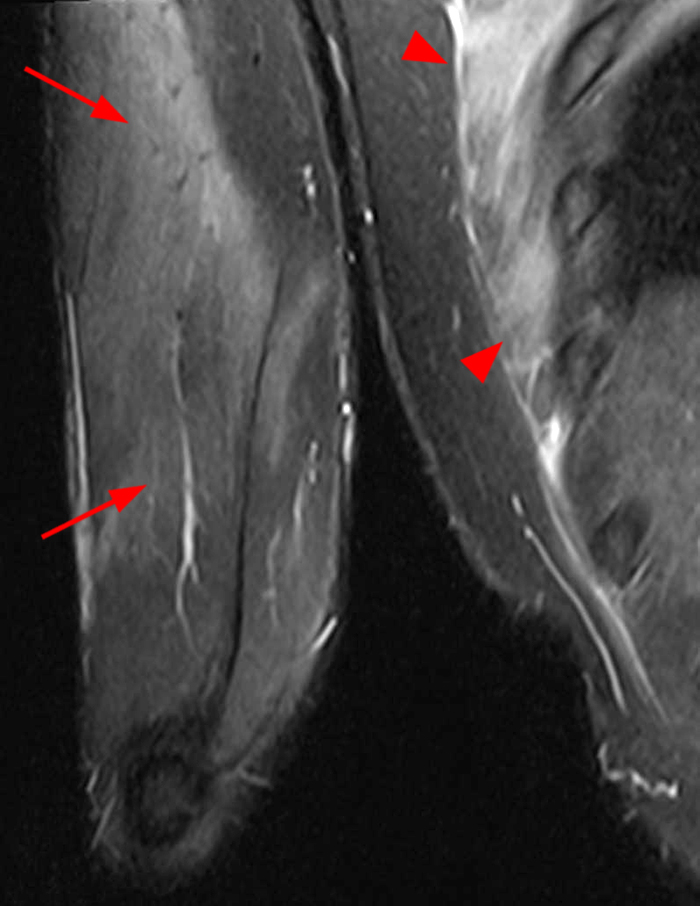
Delayed-onset muscle soreness (DOMS) is seen after strenuous activity involving eccentric muscle loading, often in individuals unaccustomed to the exercise (Figure 11). It is the intensity, not the duration, of exercise that is most closely linked to developing DOMS.22 Muscle pain is delayed, peaking at 24-72 hours after the activity, unlike muscle strains where pain is felt immediately. Maximal muscle edema slightly lags maximal pain, occurring 3-5 days after injury.22,23 Muscle swelling and extensive edema are due to increased intramuscular and interstitial fluid.22 Perifascial fluid can be seen.23
Figure 11:
Figure 11.
A 17 year-old male with clinical history as follows: “4 days status post football injury. No specific trauma, but onset of pain during football drills. Evaluate for intrasubstance tear of the triceps.” A fat-suppressed proton density-weighted axial image (11A) demonstrates extensive edema in the triceps muscle (arrow) with posterolateral perifascial fluid and adjacent subcutaneous edema, largely sparing its long head (asterisk), with similar findings involving the serratus anterior muscle (arrowheads). A STIR coronal image through the posterior upper arm (11B) shows similar findings of the triceps (arrows) and serratus anterior (arrowheads) muscles.
This patient had done an extreme number of burpees during football practice 4 days prior to the MRI. His symptoms improved quickly and resolved 1-2 weeks following the study. His clinical course and presentation 4 days following the strenuous exercise are consistent with DOMS involving the triceps and serratus anterior muscles.
There are many other varied pathologies that could potentially simulate PTS on MRI. Trauma, neoplastic disease, inflammatory processes, and vasculitides can affect the cervical nerve roots, brachial plexus, or peripheral nerves, causing muscle edema and pain. Cervical cord pathology (e.g. syringomyelia) can cause upper limb muscle denervation.12 Infectious myositis can manifest solely as muscle edema.14 Myopathies (inflammatory such as polymyositis, and medication-induced) can occasionally involve the upper extremity, manifesting as muscle edema. Radiation therapy can cause diffuse muscle edema, although history and its straight margin demarcating the therapy field is a major clue to its presence. Compartment syndromes, acute or chronic exertional-related, are rare in the upper extremities; while edema throughout involved muscles can be seen, the muscle swelling with or without perifascial fluid argue against PTS.
Fatty muscle atrophy can occur in many conditions, including chronic tendon tears, severe muscle injury, longstanding disuse, longstanding denervation of any cause, myopathies (e.g. muscular dystrophy, mitochondrial myopathy), and chronic steroid use.24 It can be impossible to distinguish the findings on MRI from the chronic denervation that can be seen in PTS individuals. Clinical history is often paramount in making the distinction.
Treatment:
Treatment for PTS is largely supportive as no treatment has been found to definitively alter its course. Long-acting analgesics and opiates are frequently used, as are gabapentin, carbamazepine, and amitryptyline.1,2 Steroids and immunotherapy may also be utilized.1,6 Physical therapy is a mainstay. Individuals whose symptoms do not improve may need surgical intervention (e.g. tendon transfers).4
Conclusion:
PTS is first and foremost a clinical diagnosis. As van Alfen states, typically the pain is “unlike anything the patient experienced before.”2 Symptoms thereafter are protean and may involve simply the inability to flex or extend fingers, orthopnea due to phrenic nerve involvement, or variable upper extremity weakness and/or numbness. MRI can be key in the diagnosis, particularly in patients with atypical or confusing clinical presentations. It is worth remembering that nerves outside the brachial plexus (e.g. lower cranial nerves) and almost any age (from neonates to the elderly) can be affected. The pattern of edema on MRI is diffuse in affected muscles, either homogeneous or heterogeneous. Edema isolated about musculotendinous junctions and/or perifascial fluid and adjacent subcutaneous edema are findings inconsistent with PTS. PTS should be considered on any MRI of the upper extremity, brachial plexus, or even neck/cervical spine when diffuse muscle edema is present. Corroboration with clinical history is essential. Radiologists may be the first to suggest the diagnosis and proper diagnosis can be crucial to prevent unwarranted surgical interventions.
References
- Milner CS, Kannan K, Iyer VG, et al. Parsonage-Turner Syndrome: clinical and epidemiological features from a hand surgeon’s perspective. Hand. 2016 Jun;11(2):227-31. ↩
- Van Alfen N. The neuralgic amyotrophy consultation. J Neurol. 2007;254:695-704. ↩
- Gaskin CM, Helms CA. Parsonage-Turner Syndrome: MR imaging findings and clinical information of 27 patients. Radiology. 2006;240(2):501-7. ↩
- McCarty EC, Tsairis P, Warren RF. Brachial neuritis. Clin Orthop Relat Res. 1999 Dec;368:37-43. ↩
- Scalf RE, Wenger DE, Frick MA, et al. MRI findings of 26 patients with Parsonage-Turner Syndrome. Am J Roentgenol. 2007 Jul;189(1):W39-44. ↩
- Feinberg JH, Radecki J. Parsonage-Turner Syndrome. HSS J. 2011 Sep;6(2):199-205. ↩
- Hussey AJ, O’Brien CP, Regan PJ. Parsonage-Turner Syndrome-case report and literature review. Hand. 2007(2):218-21. ↩
- Auge WK. Parsonage-Turner Syndrome in the native American Indian. J Shoulder Elbow Surg. 2000;9(2):99-103. ↩
- Bardos V, Somodska V. Epidemiologic study of a brachial plexus neuritis outbreak in northeast Czechoslovakia. World Neurol. 1961;2:973-9. ↩
- Van Alfen N, van Engelen BGM. The clinical spectrum of neuralgic amyotrophy in 246 cases. Brain. 2006;129:438-50. ↩
- Wendling D, Sevrin P, Bouchaud-Chabot A, et al. Parsonage-Turner syndrome revealing Lyme borreliosis. Joint Bone Spine. 2009 Mar;76(2):202-4. ↩
- Kamath S, Venkatanarasimha, Walsh MA. MRI appearance of muscle denervation. Skeletal Radiol. 2008;37:397-404. ↩
- Aralasmak A, Karaali K, Cevikol C, et al. MR imaging findings in brachial plexopathy with thoracic outlet syndrome. Am J Neuroradiol.2010 Mar;31(3):410-7. ↩
- May DA, Disler DG, Jones EA, et al. Abnormal signal intensity in skeletal muscle at MR imaging: patterns, pearls, and pitfalls. RadioGraphics. 2000;20:S295-S315. ↩
- Napier N, Shortt C, Eustace S. Muscle edema: classification, mechanisms, and interpretation. Semin Musculoskeletal Radiol. 2006;10:258-67. ↩
- Fleckenstein JL, Watumull D, Conner KE, et al. Denervated human skeletal muscle: MR imaging evaluation. Radiology. 1993;187:213-8. ↩
- Fischbein NJ, Kaplan MJ, Jackler RK, et al. MR imaging in two cases of subacute denervation change in the muscles of facial expression. Am J Neuroradiol. 2001 May;22:880-4. ↩
- Sneag DB, Rancy SK, Wolfe SW, et al. Brachial plexitis or neuritis? MRI features of lesion distribution in Parsonage-Turner syndrome. Muscle Nerve. 2018 Sep;58(3):359-66. ↩
- Sneag DB, Saltzman EB, Meister DW, et al. MRI bullseye sign: an indicator of peripheral nerve constriction in parsonage-turner syndrome. Muscle Nerve. 2017 Jul;56(1):99-106. ↩
- Chhabra A, Thawait GK, Soldatos T, et al. High-resolution MR neurography of the brachial plexus and its branches, with emphasis on 3D imaging. Am J Neuroradiol. 2013 Mar;34(3):486-97. ↩
- Seo TG, Kim DH, Kim I, et al. Does C5 or C6 radiculopathy affect the signal intensity of the brachial plexus on magnetic resonance neurography? Ann Rehabil Med. 2016;40(2):362-7. ↩
- Flores DV, Gomez CM, Estrada-Castrillon, et al. MR imaging of muscle trauma: anatomy, biomechanics, pathophysiology, and imaging appearance. RadioGraphics. 2018;38:124-48. ↩
- Guermazi A, Roemer FW, Robinson P, et al. Imaging of muscle injuries in sports medicine: sports imaging series. Radiology. 2017 Mar;282(3):646-63. ↩
- Theodorou DJ, Theodorou SJ, Kakitsubata Y. Skeletal muscle disease: patterns of MRI appearances. Br J Radiol. 2012 Dec;85(1020):e1298-308. ↩