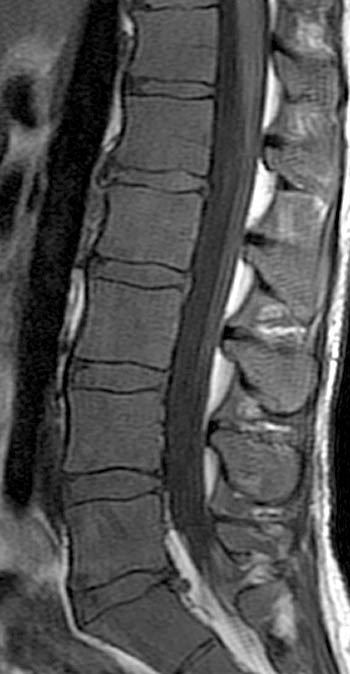
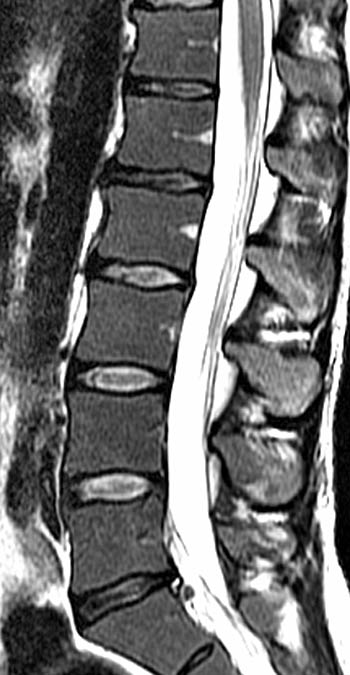
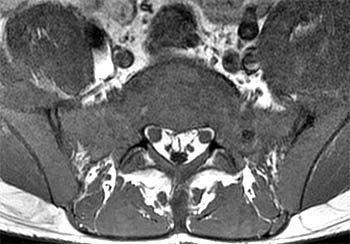
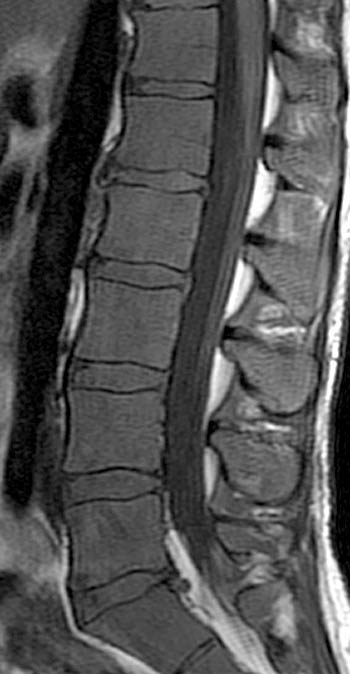
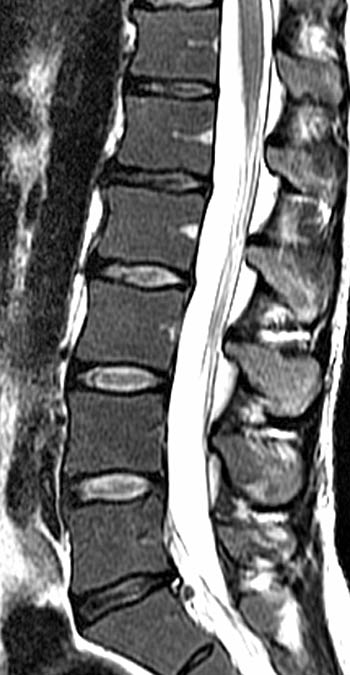
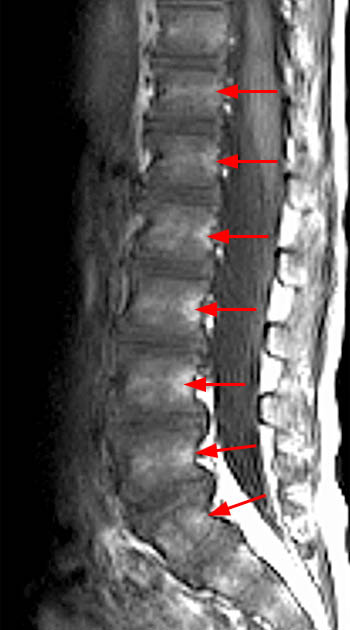
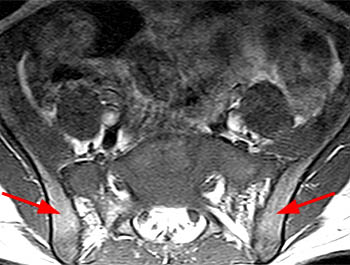
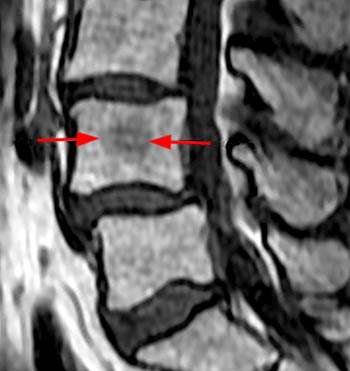
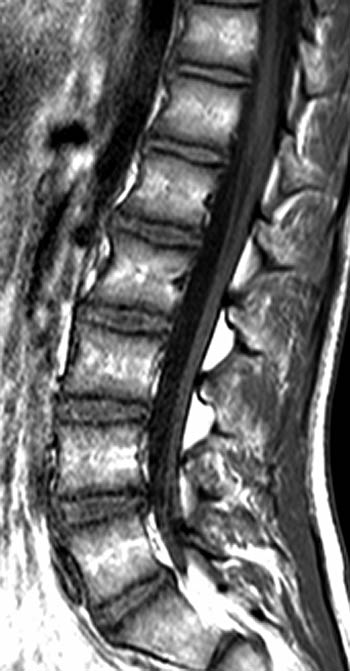
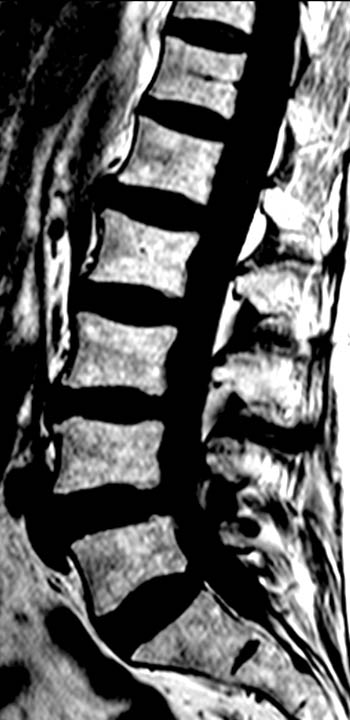
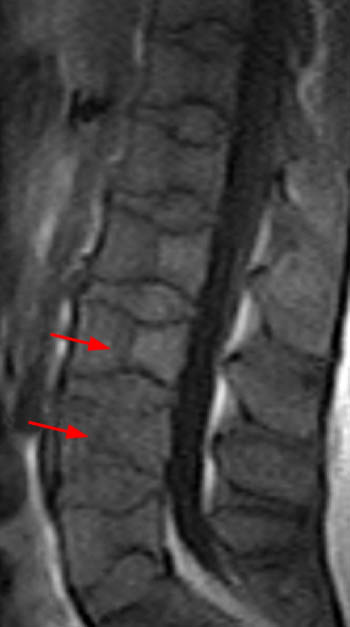
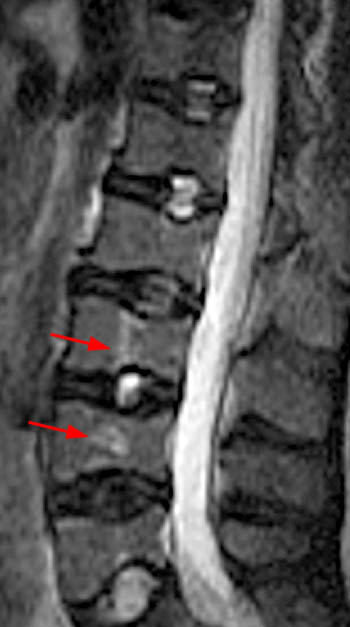
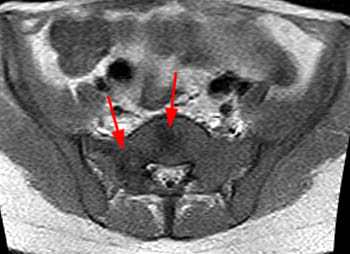
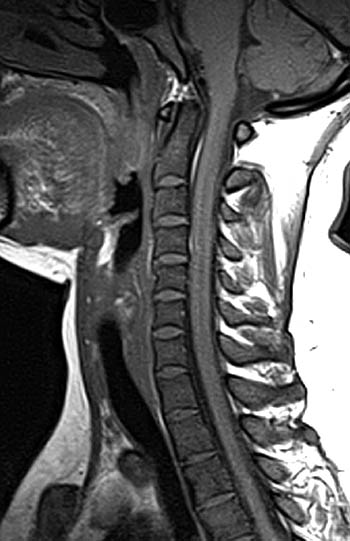
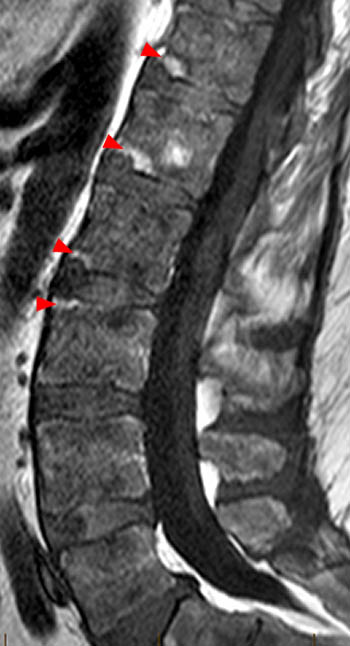
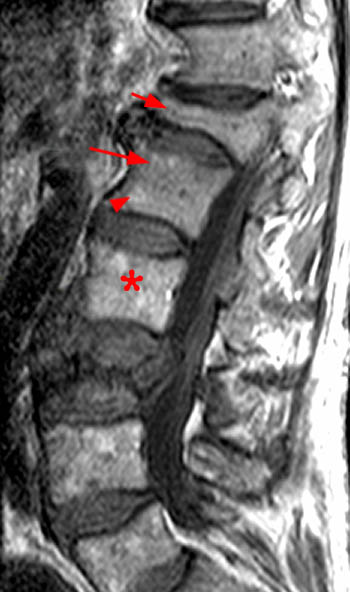
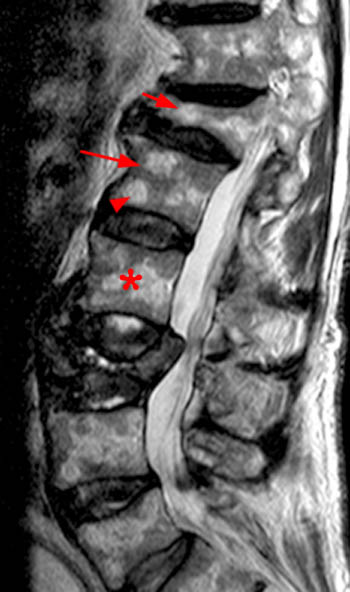
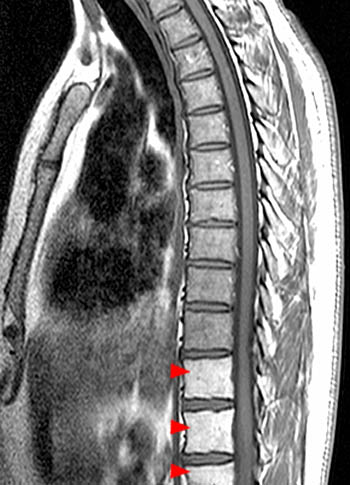
Clinical History: A 43 year old male presents with radicular type pain. Past medical history is non-contributory. (1a) T1- and (1b) T2-weighted sagittal and (1c) T1-weighted axial images are provided. The imaging findings are such that the radiologist feels compelled to place a telephone call to the referring physician. What are the findings? What are the likely diagnoses?
Findings
Diagnosis
Diffuse bone marrow disorder which in this case led to an eventual diagnosis of Chronic Myelogenous Leukemia (CML). Other diffuse marrow diseases and conditions are possible based on the images and will be discussed.
Introduction
This patient exhibits a diffusely abnormal bone marrow pattern due to previously unrecognized leukemia. In adults spinal bone marrow is best imaged with MRI due to the exquisite contrast differentiation between normal marrow and pathologic infiltration on many pulse sequences. Such differentiation is better than x-ray, CT, PET, or even radionuclide bone scan with technetium. The finding of a diffuse marrow abnormality on MRI, however, is non-specific. The most common pathologic infiltration of marrow is metastases from solid organ tumors, but since metastatic disease is much more often multifocal than diffuse in its imaging pattern in the spine, it will only be briefly discussed in this article.
Anatomy of Marrow
Bone marrow can be essentially divided into three parts: red marrow, yellow marrow and supporting structures such as trabecular bone and reticulum. Hematopoietically active marrow is usually referred to as red marrow. It is uniformly found throughout the skeleton at birth. In the adult red marrow is concentrated in the axial skeleton, but may be focally scattered in other parts of the skeleton. Yellow marrow is generally hematopoietically inactive and is concentrated in the adult in the appendicular skeleton. Marrow content and its distribution in the body changes substantially with age and differs by sex1,2,3,4. Red and yellow marrow are not homogeneous tissues. In young children red marrow is approximately 40% water, 40% fat and 20% protein. By 70 years of age that proportion has changed to 60% fat and 10% protein. Yellow marrow is 80% fat1. Red marrow is composed of 60% hematopoietically active cells in the young but only about 30% in the elderly5.
Bone Marrow Conversion
At birth the majority of marrow is hematopoietically active red marrow. Marrow signal is similar or even less than adjacent muscle and disc such that pathologic marrow may be very difficult to ascertain6. Soon thereafter marrow undergoes a predictable pattern of conversion to yellow marrow. This process begins first and is most evident in the appendicular skeleton and proceeds in a centripetal manner (5a,6a,7a). By 25-30 years of age this normal physiologic conversion is complete. Except for the sternum, ribs, proximal aspects of the extremities, and the pelvis, the overwhelming majority of red marrow is in the spine in an adult1,7. However, there is a variable mixture of red and yellow marrow in the spine beyond infancy with progressive increase in fat content of red marrow and increased proportion of yellow marrow with aging2,3.
Appearance of Normal Marrow in the Spine on MRI
Since marrow is not a homogeneous tissue and changes with age, one should expect that its MR appearance will vary depending on the relative proportion of red and yellow marrow, cellularity and density of trabecular bone in the spine and on the type of sequence used for the acquisition. The workhorses of routine spinal imaging are the T1-weighted and T2-weighted Spin Echo (T1w; T2w); the T2-weighted Spin Echo with fat saturation (T2 fatsat) and the short tau inversion recovery (STIR) sequences. Red marrow exhibits intermediate signal and tends toward slightly higher or equal signal compared to muscle on both T1w and T2w sequences. Red marrow has intermediate signal on T2 fatsat and STIR. Yellow marrow will show signal similar to fat on all pulse sequences1,7,8,9.
Fat has short T1 and T2 relaxation times and is hyperintense on T1w sequences and hypointense on true (conventional) T2-weighted sequences. However, newer fast spin echo acquisitions (FSE) do not take advantage of this fact in a trade off for speed so that fat is not as hypointense on the T2wFSE most commonly performed today. Water has a much longer relaxation time compared to fat and is hypointense on T1w and hyperintense on T2w images. Since red marrow has a mixture of water, fat and protein it exhibits intermediate signal intensity on both T1w and T2w sequences1,7,8,9. Importantly, the intensity of marrow in an adult comes with an internal control when viewing T1w sequences. On T1w images in the adult, the signal of the vertebral body should be higher than adjacent intervertebral discs or muscle with few exceptions10,11. Exceptions include calcified discs which have bright T1w signal and particularly islands of red marrow which can be confused with focal pathology such as tumor (8a,9a).
The T2w FSE sequence is substantially less reliable in assessing spinal marrow, particularly in diffuse disease, as comparison with adjacent normal tissues is more variable than on T1w images13. Unless pathologic marrow has a much higher signal (e.g. from increased water content), or lower signal (e.g. from sclerosis) it may not be very conspicuous on T2w views. Visibility can be augmented by suppressing the fat which is present in both red and yellow marrow. STIR uses an inversion pulse to cancel signal from fat. Dixon fat saturation techniques usually nullify the signal from fat by a pre-saturation pulse. The more yellow marrow which is present within a vertebral body the more homogeneously suppressed (hypointense) the body will appear with these techniques. Most pathology will demonstrate relatively higher signal on T2w fatsat or STIR than red or yellow marrow. Images obtained with these sequences tend to be grainier and less distinct due to suppression of signal from lipid protons decreasing the signal to noise ratio9.
Other imaging techniques have been utilized in attempts to differentiate normal from abnormal marrow but are not in clinical use on a day to day basis and are beyond the scope of this article. Some of these techniques include: chemical shift imaging (also known as opposed phase imaging); diffusion; spectroscopy and dynamic contrast enhancement14,15,16,17,18. It is important to note that the introduction of each of these special techniques has met with some success and some disappointment.
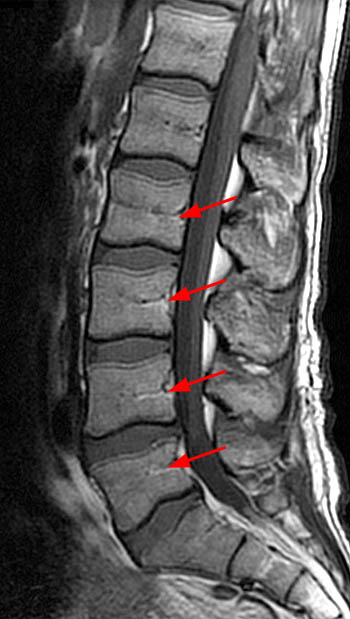
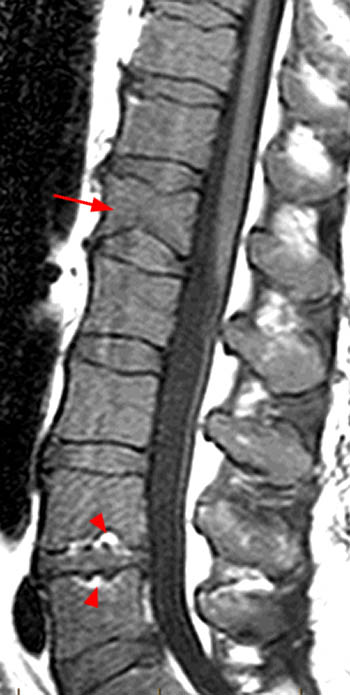
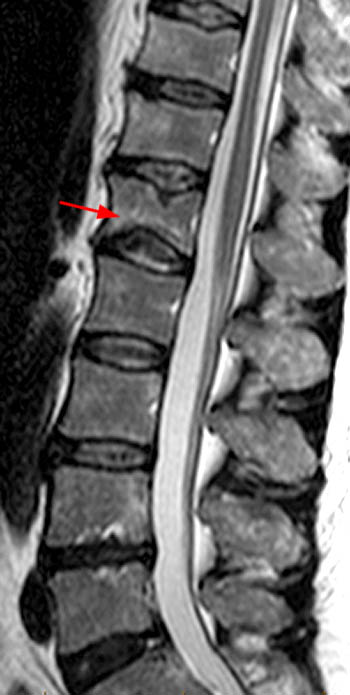
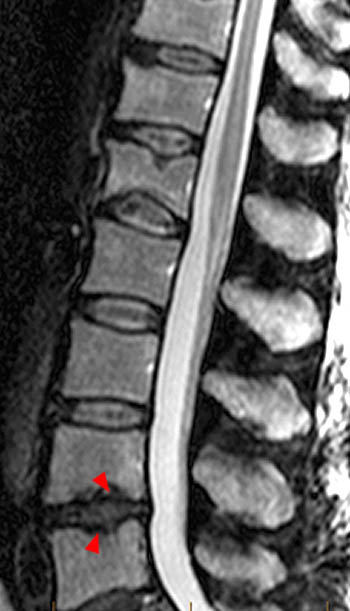
Normal bone marrow conversion that occurs with maturation and aging is rarely uniform. Ricci has described several patterns of normal red marrow and yellow marrow dispersion in the spine after the perinatal period. There is great variability among patients and some differences in the patterns between the cervical, thoracic and lumbar segments. Pattern 1 begins in younger patients where central fat is visible along the basivertebral veins. This pattern may persist for many years (10a,11a). Pattern 2 is represented by band-like and triangular-like foci of fat along the endplates and body “corners.” This pattern may be influenced by adjacent disc degeneration. Pattern 3 (12a) can be referred to as a speckled pattern with tiny foci of interspersed red and yellow marrow. Pattern 4 (13a) is exemplified by larger, rounder areas of yellow marrow and poorly defined areas of red marrow. Except for transition from an infantile pattern of homogeneous red marrow to pattern 1, the patterns do not progress or evolve in a predictable or clinically useful manner3. The patterns may seem to overlap in clinical practice but their descriptions are a useful teaching tool. Usually the human eye creates a “gestalt” of the entirety of the vertebral body signal merging the individual components of red and yellow marrow into a kind of perceptual average19. This subjective assessment can be more accurate at qualifying marrow within bone as normal or abnormal than certain quantifying methods20. Thankfully, this perceptual averaging becomes easier as the patient ages and the percentage of fat within the marrow increases. Additionally, bone trabeculae decrease signal in the vertebral body by creating local field inhomogeneities. This may contribute to the observation that marrow signal can be very high in elderly women on T1w images, whose trabeculae may thin from osteoporosis.
Figure 11:
Ricci Pattern 1 in a 40 year old man demonstrates only a tiny amount of yellow marrow visible along the basivertebral veins (arrows). Red and yellow marrow are evenly dispersed. "Gestalt" of marrow is normal as the vertebral body centrum is of overall higher signal than the adjacent intervertebral discs.
Adult marrow does not appreciably enhance to visual inspection after intravenous gadolinium contrast is administered. Normal marrow in a neonate and young child however may substantially enhance21. Enhancement does occur in pathologic marrow and this difference can be highlighted using post contrast T1w images with fat saturation7.
Pathologic Appearing Bone Marrow
Any system of stratifying pathologies could be criticized but in an attempt at simplicity I divide the diffuse disorders of the spinal marrow into: Reconversion or Hyperplasia; Replacement Disorders; Depletion Disorders; and Reticulum Disorders or disorders of Supporting Structures. The first two categories could even be further simplified into Replacement (Proliferative) Disorders with either normal or abnormal cells. Many diffuse pathologic processes in the spinal marrow have a non-specific appearance with reduced signal on T1w images and intermediate T2w signal. STIR images or post contrast images may or may not differentiate red marrow hyperplasia from pathologic cellular replacement.
1. Reconversion
Physiologic demand for more mature blood cells (especially red blood cells) beyond the body’s normal capacity to produce them acts to reverse the normal pattern of conversion of red marrow to yellow marrow. This stimulated growth of hematopoietically active marrow increases the visibility of red marrow in the axial and appendicular skeleton. This process is referred to as reconversion or sometimes myeloid hyperplasia. Typically this process occurs in a reverse direction from central to peripheral as demands increase. Examples of this physiologic stress include demands on an endurance athlete, particularly training at high altitude22, and cigarette smokers, especially obese women23. Other causes include: hyperplastic anemias such as sickle cell disease, thalassemia and spherocytosis(14a,14b,14c,15a); administration of erythrocyte or granulocyte stimulating agents for therapy and polycythemia vera (16a). Hyperplastic marrow does not differ greatly from a diffuse replacement disorder, sharing T1w characteristics of being iso- or hypointense to muscle. T2w fatsat, STIR and post enhancement characteristics will differ in most but not all cases. In hyperplastic anemias iron accumulation in marrow macrophages related to transfusions or hemolysis may contribute to reduced marrow signal7,8,13,24,25,26,27.
Figure 14:
Sickle cell disease in a 29 year old woman. Erythroid hyperplasia expands the marrow space and thins trabeculae, weakening bone and contributing to the classic endplate deformity. Edema probably from bone infarcts is highlighted by arrows in 14a and 14b. A T1w axial image (14c) demonstrates that marrow signal in sacrum and iliac bones is lower than that of adjacent muscle. Hemosiderosis likely contributes to the decreased marrow signal.
Figure 15:
Spherocytosis in a 56 year old woman. The T1w sagittal image demonstrates diffusely low signal within marrow, not only within the spine but also within the clivus. Marrow signal is abnormally low compared to that of discs and adjacent muscle. Although reports have suggested that clival marrow signal changes are useful in differentiating a benign reconversion process from a more serious replacement (e.g. malignant) process, the appearance of the clivus is not a reliable discriminator20.
Figure 16:
Chronic Renal Failure in a 50 year old woman. A T1w sagittal image demonstrates heterogeneous marrow that is generally reduced in signal. The signal and pattern are abnormal for an adult. There are likely several factors contributing to the marrow appearance including: administration of recombinant erythropoietin, myelofibrosis, iron overload, secondary hyperparathyroidism and aluminum accumulation. It is curious that some areas of degenerative focal fat remain (arrowheads). Only a minority of patients with chronic renal failure have spinal marrow with this appearance.
Polycythemia vera is characterized by a proliferation of a clone of pluripotent erythrocytotic stem cells. This leads to hypercellularity of the marrow which is effectively indistinguishable from a reconversion phenomenon or diffuse marrow replacement by other hematologic malignancies on MR. Fifteen percent of patients eventually develop myelofibrosis with myeloid metaplasia28,29. MR is unable to differentiate that transition.
After successful bone marrow transplantation, hematopoietically active stem cells repopulate the bone marrow in a predictable pattern which has been described by Stevens, et al.30. A few weeks after the transplant, a band-like zone becomes visible in the periphery of the vertebral centrum particularly beneath the endplates. This zone exhibits intermediate signal on both T1w and T2w images, similar to red marrow. The band enlarges over time and is easily distinguished from central fat. Histologically this band of intermediate signal is composed of hematopoietically active cells. The time course is variable but occurs in almost all patients within the first 90 days. Moulopoulos reported that in their experience the marrow repopulation gradually becomes more homogeneous over time25.
2. Marrow Replacement Disorders
Marrow replacement disorders are exemplified by proliferation of abnormal (usually malignant) cells in the bone marrow. Three patterns of proliferation are recognized: a diffuse pattern where the normal bone marrow is completely replaced; a variegated pattern where innumerable tiny clusters of abnormal cells are present; and a focal pattern where larger often spherical lesions predominate. The focal pattern is the classical pattern of metastases from solid organ malignancies and will not be discussed in this review.
The classic example of diffuse replacement is the leukemias, a variety of myeloproliferative disorders which have similar appearances on MR. The normal marrow signal usually is completely replaced by abnormal signal best appreciated as low signal on T1w images. Such an observation is problematic in very young children due to the high percentage of hematopoietically active marrow. An abnormality may not be evident on T2w FSE but often is of greater than normal signal on STIR or T2w fatsat. In early stages of the disease, the MR may appear normal. Diagnosis is made by bone marrow biopsy. A large number of studies have been performed to assess the usefulness of various advanced MR techniques in diagnosing and following patients with leukemia. Using MR to follow treatment is problematic5, 7,8,25,31,32,33,35.
Multiple Myeloma is a plasma cell dyscrasia. Multiple myeloma on MR may appear normal, diffusely abnormal (17a,18a,19a), variegated, multi-focally abnormal or as a solitary lesion (plasmacytoma). The forms may also be “combined.” A visually normal vertebral body may harbor up to 20% abnormal cellular infiltration. The variegated pattern has been described as if “cracked pepper were sprinkled onto the marrow on T1-weighted images”1,8. The multi-focal pattern is the most common pattern and mimics solid organ metastases to the spine. Up to 30% of focal myeloma lesions visible on T1w studies are not distinct on T2w images but may be visible on T2w fatsat or STIR images. The diffuse pattern mimics other replacement disorders such as leukemia and even marrow reconversion. The variegated pattern occurs in less than 5% of patients and is not only attributable to tiny clusters of plasma cells but to inhibition of hematopoiesis creating tiny foci of fat. Around 50% of patients with focal or diffuse MR patterns do not have lytic lesions on conventional x-rays. The use of MR has impacted the staging and follow-up of patients with myeloma and has led to a modification of the classic Durie and Salmon staging system. It has been discovered that patients with a normal or variegated bone marrow appearance tend to Stage I disease. A diffuse pattern of marrow disease is associated with a higher stage and worse prognosis. Whole body MR has found some use in staging and following this disorder7,8,25,36,37,38.
Figure 17:
Multiple myeloma, Diffuse type. A T1w sagittal image demonstrates diffusely low signal in this 56 year-old woman. A mild fracture is present at L1 (arrow) which does not exhibit substantially different signal than that in other marrow. It is curious that some areas suggesting degenerative fat persist (arrowheads) despite complete marrow replacement otherwise.
It is useful to discuss the appearance of post-treatment myeloma on MR as a bridge to understanding the difficulty in providing a clinically useful interpretation of a scan. Vertebral body fractures occur frequently in myeloma and certainly retropulsion of bone and compression of neural elements are important and easy things to assess. However, many such fractures look benign on MR. The development or progression of fractures does not necessarily point to progression of disease as tumor elimination may be followed by loss of structural support. Alternatively a fracture may reflect progression of disease. Hemorrhages may occur within a vertebral body raising the T1w signal which might under some circumstances confuse the evaluation of a segment. Myelofibrosis may develop after treatment. Chemotherapy and radiation therapy change the marrow in predictable ways and may affect its appearance on MR. Initially edema and necrosis occur which can be followed by hypocelluarity if a positive tumoral response is elicited. These findings may be noticeable on MR when compared to a prior study. About two weeks later fatty replacement begins and may be visible on MR and classically occurs around the basivertebral veins. However some focal lesions in myeloma do not change significantly in appearance for up to 5 years. Reconversion can occur depending on the degree to which the therapy has obliterated the marrow. Bone marrow reconversion and the diffuse pattern of myeloma may be similar especially if bone marrow stimulators have been administered. Studies have shown that a pre-treatment intensely, diffusely enhancing focal lesion which does not enhance post-treatment is inactive. Partially or peripherally enhancing lesions are non-specific as post-treatment fibrosis will enhance7,8,25,36,37,39,40. It is interesting that some authors emphasize the STIR and post-contrast differences in appearance between hyperplastic red marrow and malignant infiltration while others do not. I think this relates not only to the variability of pathology affecting marrow but also that the best discrimination after enhancement is obtained through quantitative measurements in the first minute after a dynamic infusion of contrast, something which is not usually performed in day to day practice. Clearly, to interpret a follow-up MR in a patient with a spinal marrow replacement disorder is potentially a complicated task (20a,20b,20c).
Figure 20:
These MRI findings led to a new diagnosis of lymphoma in an 89 year old man with an unusual display of signal abnormalities likely related to a mixture of pathologies. The overall pattern looks "combined." A severe compression fracture is noted at L3. The fracture of T12 (short arrows) looks superficially benign on T1w images (20a) but has abnormal signal on T2w (Fig 20b) and STIR (20c). The T12 and L1 bodies are probably diffusely abnormal for age. The long arrow connotes a slightly hypointense poorly defined lesion on T1w (20a) which is quite conspicuous on the T2w (20b) and STIR (20c) that is typical for focal tumor except in its lack of defined borders on the T1w view. Arrowheads denotes a lesion which has some bright T1w signal that appears substantially larger on the T2w and STIR images, possibly representing a hemorrhagic metastasis. Asterisks denote a larger central area which appears normal on the T1w images but clearly abnormal on the T2w and STIR views. Other areas appear low signal on T1w, T2w and STIR which is not rare in lymphoma. Most of the vertebral bodies have much less high T1w signal within them than normally seen in an 89 year old person.
Bone marrow involvement by malignant lymphoma is much more common with Non-Hodgkin disease than Hodgkin disease and spreads to the marrow 95% of the time hematogenously. Bone marrow involvement upstages the patient and may have prognostic and therapeutic implications. Diffuse or mottled appearing infiltration is much more common than the multi-focal form. Signal intensity can vary considerably with areas of high and low T2w with low T2 signal thought to be attributable to dense clusters with “hypercellularity.” As in the leukemias, follow-up of diffuse forms may be problematic in differentiating hypercellular reactive marrow, fibrosis and residual or recurrent active tumor7,25,41.
3. Marrow Depletion Disorders
Myeloid depletion equates to loss of normal red marrow and thus diffusely markedly fatty signal on all pulse sequences. All of these disorders have the same appearance on MR. Marrow depletion to at least some degree is common after radiation therapy and chemotherapy (21a, 22a) and can be severe enough to notice on MR. In the first few days after initiating radiation, the effects are dominated by necrosis, edema and hemorrhage, especially on STIR. Beginning approximately 3-6 weeks after initiating radiation therapy, the marrow signal begins to increase on T1w images both qualitatively and quantitatively. For doses less than 30Gy the marrow usually regenerates in between one and two years. The recovery of normal red marrow signal has been reported both as patchy and peripheral band-like with band-like recovery apparently more common in the young. For doses greater than 30-40 Gy, the marrow changes are permanent due to ablation of vascular sinusoids. The appearance of marrow depletion disorders on MR can overlap with normal. Often realization that the marrow is depleted is obvious only after observing uninvolved areas of the spine7,25,42,43.
Aplastic anemia is manifested as pancytopenia with numerous causes such as viral infection, drug toxicities and as autoimmune responses to malignancies, although most cases are idiopathic. About half of patients will improve with immunosuppressive therapy and may develop visible foci of hypercellular/active marrow on MR. It is interesting that after bone marrow ablation, stem cell transplantation and marrow regeneration, a typical pattern of reconversion is found beginning in bands along the endplates. That is not the case in the reported therapeutic response in aplastic anemia. Hemosiderosis which may occur after multiple transfusions may reduce marrow signal on MR. A significant percentage of patients with this disorder progress to myelodysplasia and eventually leukemia, if they do not undergo bone marrow transplantation. These observations plus the fact that reconversion tends to be focal has contributed to some MR descriptions of aplastic anemia as heterogeneous. Presumably the transition to preleukemia or leukemia would be heralded by a change in the appearance on the STIR and/or dynamic post contrast study7,25,44,45.
4. Disorders of Supporting Structures and Reticulum
I will just focus on a few of these disorders excluding primary metabolic diseases of bone.
One of the manifestations of HIV infections is a pattern of diffuse loss of signal on T1w and T2w images within spinal marrow46. This finding was reported by Geremia47 in all eleven patients studied and by Steinbach in all 7 of her patients who underwent spinal MR48. This find is felt secondary to hemosiderosis, excessive iron accumulation in marrow macrophages which is histologically characteristic of anemia of chronic disease (23a,24a). In this form of anemia impaired release of iron from macrophages contributes to impaired utilization of iron for hemoglobin production. Hemosiderosis can occur in a variety of other disorders such as hemolytic anemias, hemochromatosis and some malignancies. Plasma cell proliferation in marrow is also well known in HIV and may contribute to the marrow appearance49.
Primary Myelofibrosis, also called myeloid metaplasia, is a rare myeloproliferative disorder of hematopoietic progenitor cells. These cells stimulate the abnormal formation of marrow fibroblasts and incite the release of collagen. Secondary myelofibrosis is much more common, usually resulting from chemotherapy or radiation therapy. Primary myelofibrosis is also a myelodysplasia and therefore predisposes to conversion to leukemia. Some authors describe the MR appearance of myelofibrosis as very low signal on T1w and T2w images, while others do not. This discrepancy is likely due to the degree of fibrosis present, whether the fibrosis is primary or secondary, and whether the primary form is in a preleukemic state7,25,28,50.
Gaucher’s disease is the most common lysosomal storage disorder and can be included in disturbances of the marrow reticulum. This inborn error of metabolism leads to accumulation of glucocerebrosides within the macrophage-monocyte cell line. These so called Gaucher cells replace the normal marrow leading to decreased T1w and T2w signal similar to so many other reconversion and replacement disorders5.
Conclusion
MR is a very sensitive technique for evaluating bone marrow. Opportunities for such evaluation abound as portions of the marrow can be evaluated on nearly every MR study performed. Knowledge of the signal on the variety of pulse sequences and pattern of marrow in the normal spine is essential knowledge in deciding what is potentially pathologic and what is not. The conversion of normal red marrow to yellow marrow and the reconversion back to hematopoietically active marrow under physiologic stress is a common occurrence but highlights one weakness of MR, its lack of specificity without clinical information. In a patient such as the test case who demonstrates diffusely low T1w marrow signal on MR, the differential diagnosis is long and includes many benign reconversion conditions, malignant myeloproliferative disorders and abnormalities of the supporting reticulum.
References
1 Vogler JB, Murphy WA. Bone marrow imaging. Radiology. 1988; 168:679-693
2 Dooms GC, Fisher MR, Hricak H, Richardson M, Crooks LE, Genant HK. Bone marrow imaging: magnetic resonance studies relating to age and sex. Radiology. 1985; 155:429-432.
3 Ricci C, Cova M, Kang YS, Yang A, Rahmouni A, Scott WW, Zerhouni EA: Normal age-related patterns of cellular and fatty bone marrow distribution in the axial skeleton: MR imaging study. Radiology. 1990; 177:83-88.
4 Ishijima H, Ishizaka H, Horikoshi H, et al. Water fraction of lumbar vertebral bone marrow estimated from chemical shift misregistration on MR imaging: normal variations with age and sex. Am J Roentgenol. 1996;167(2):355–358.
5 Burdiles A, Babyn PS. Pediatric Bone Marrow MR Imaging Magnetic Resonance Imaging Clinics of North America. August 2009; Vol. 17, Issue 3, Pages 391-409.
6 Sze G, Baierl P, Bravo S. Evolution of the infant spinal column: evaluation with MR imaging. Radiology 1991;181:819-827
7 Alyas F, Saifuddin A, Connell D: MR Imaging Evaluation of the Bone Marrow and Marrow Infiltrative Disorders of the Lumbar Spine. Magnetic Resonance Imaging Clinics of North America May 2007 .Vol. 15, Issue 2, Pages 199-219.
8 Tall MA, Thompson AK, Vertinsky T, Palka PS: MR Imaging of the Spinal Bone Marrow Magnetic Resonance Imaging Clinics of North America. May 2007 (Vol. 15, Issue 2, Pages 175-198
9 Mirowitz S, Apicella P, Reinus WR, Hammerman AM: MR imaging of bone marrow lesions: Relative conspicuousness on T1-weighted, fat-suppressed T2-weighted, and STIR images. AJR Am J Roentgenol. 1994; 162:215-221
10 Carroll, K. W., Feller, J. F. and Tirman, P. F. J. : Useful internal standards for distinguishing infiltrative marrow pathology from hematopoietic marrow at MRI. Journal of Magnetic Resonance Imaging. 1997; 7: 394-398.
11 Castillo, M, Malko, JA, Hoffman, JC, Jr: The bright intervertebral disk: an indirect sign of abnormal spinal bone marrow on T1-weighted MR images
AJNR Am J Neuroradiol. 1990; 11: 23-26.
12 Schweitzer ME, Levine C, Mitchell DG, et al. Bull’s eyes and halos: useful MR discriminators of osseous metastases. Radiology. 1993;188:249-252.
13 Vande Berg BC, Lecouvet FE, Galant C, et al. Normal variants and frequent marrow alterations that simulate bone marrow lesions at MR imaging. Radiol Clin North Am. 2005;43(4):761-770
14 Zajick DC, Morrison WB, Schweitzer ME, et al. Benign and Malignant Processes: Normal values and Differentiation with Chemical Shift MR Imaging in Vertebral Marrow. Radiology. 2005;237:590-596.
15 W.K. Erly, E.S. Oh, and E.K. Outwater: The Utility of In-Phase/Opposed-Phase Imaging in Differentiating Malignancy from Acute Benign Compression Fractures of the Spine AJNR Am J Neuroradiol. 2006; 27: 1183-1188
16 Swartz, PG, Roberts CC. Radiological Reasoning: Bone Marrow Changes on MRI. Am. J. Roentgenol. Sep 2009; 193: S1-S4.
17 Castillo M. Diffusion-weighted imaging of the spine: is it reliable? AJNR Am J Neuroradiol. 2003;24:1251-1253
18 Griffith JF, Yeung DK, Antonio GE, et al. Vertebral bone mineral density, marrow perfusion, and fat content in healthy men and men with osteoporosis: dynamic contrast-enhanced MR imaging and MR spectroscopy. Radiology. 2005;236(3):945-951.
19 Koontz NA, Gunderman RB. Gestalt Theory: Implications for Radiology Educa tion. AJR Am J Roentgenol. 2008; 190:1156-1160
20 Loevner LA, Tobey JD, Yousem DM, et al. MR imaging characteristics of cranial bone marrow in adult patients with underlying systemic disorders compared with healthy control subjects. AJNR Am J Neuroradiol. 2002; 23:248-254.
21 Sze G, Bravo S, Baierl P, Shimkin PM: Developing spinal column: Gadolinium-enhanced MR imaging. Radiology. 1991; 180:497-502
22 Shellock FG, Morris E, Deutsch AL, Mink JH, Kerr R, Boden SD: Hematopoietic bone marrow hyperplasia: High prevalence on MR images of the knee in asymptomatic marathon runners. AJR Am J Roentgenol. 1992; 158:335-338
23 Poulton TB, Murphy WD, Duerk JL, Chapek CC, Feiglin DH: Bone marrow reconversion in adults who are smokers: MR imaging findings. AJR Am J Roentgenol. 1993; 161:1217-1221
24 Steinbach LS. “MRI in the detection of malignant infiltration of bone marrow”-a commentary. AJR 2007; 188:1443-1445
25 Moulopoulos L A, and Meletios A D. Magnetic Resonance Imaging of the Bone Marrow in Hematologic Malignancies. Blood. 1997; 90: 2127-2147.
26 Fletcher BD, Wall JE, Hanna SL: Effect of hematopoietic growth factors on MR images of bone marrow in children undergoing chemotherapy. Radiology. 1993;189:745-751
27 Hartman RP, Sundaram M, Okuno SH, Sim FH. Effect of granulocyte-stimulating factors on marrow of adult patients with musculoskeletal malignancies: incidence and MRI findings. AJR. 2004; 183:645-653
28 Kaplan KR, Mitchell DG, Steiner RM, Murphy S, Vinitsi S, Rao VM, Burk L, Rifkin MD: Polycythemia vera and myelofibrosis: Correlation of MR imaging, clinical, and laboratory findings. Radiology. 1992; 183:329-334
29 Berlin NI. Polycythemia Vera. Hematol Oncol Clin NA. 2003; 17:1191-1210
30 Stevens SK, Moore SG, Amylon MD: Repopulation of the marrow after transplantation: MR imaging with pathologic correlation. Radiology.1990; 175:213-218
31 McKinstry CS, Steiner RE, Young AT, Jones L, Swirsky D, Aber V: Bone marrow in leukemia and aplastic anemia: MR imaging before, during and after treatment. Radiology. 1987; 162:701-707
32 Rosen BR, Fleming DM, Kushner DC, Zaner KS, Buxton RB, Bennet WP, Wismer GL, Brady TJ: Hematologic bone marrow disorders: Quantitative chemical shift MR imaging. Radiology. 1988; 169:799-804
33 Vande Berg BC, Schmitz PJ, Scheiff JM, Filleul BJ, Michaux JL, Ferrant A, Jamart J, Malghem J, Maldague BE: Acute myeloid leukemia: Lack of predictive value of sequential quantitative MR imaging during treatment. Radiology. 1995; 197:301-305
34 Yu S, Rosenbaum A, Poe LB, Fredrickson BE. The Lumbar Spine In: MRI of the Musculoskeletal System. Chan WP, et al. WB Saunders, 1994, pp 127-172.
35 Vande Berg BC, Michaux L, Scheiff JM, Malghem J, Lecouvet FE, Bourgois MP, Ferrant A, Michaux JL, Maldague BE: Sequential quantitative MR analysis of bone marrow: Differences during treatment of lymphoid versus myeloid leukemia. Radiology. 1996; 201:519-523
36 Hanrahan CJ, Christensen CR, Crim JR. Current Concepts in the Evaluation of Multiple Myeloma with MR Imaging and FDG PET/CT: RadioGraphics. January 2010; 30: 127-142.
37 Angtuaco EJ, Fassas AB, Walker R, Sethi R, Barlogie B. Multiple myeloma: clinical review and diagnostic imaging. Radiology 2004; 231(1):11-23.
38 Moulopoulos LA, Varma DGK, Dimopoulos MA, Leeds NE, Kim EE, Johnston DA, Alexanian R, Libshitz HI: Multiple myeloma: Spinal MR imaging in patients with untreated newly diagnosed disease. Radiology. 1992; 185:833-840
39 Moulopoulos LA, Dimopoulos MA, Alexanian R, Leeds NE, Libshitz HI: Multiple myeloma: MR patterns of response to treatment. Radiology. 1994; 193:441-446
40 Rahmouni A, Divine M, Mathieu D, et al. MR appearance of multiple myeloma of the spine before and after treatment. AJR Am J Roentgenol. 1993; 160:1053-1057
41 Kwee TC, Kwee RM, Verdonck LF, et al. Magnetic resonance imaging for the detection of bone marrow involvement in malignant lymphoma. Br J Haematol
2008;141(1):60-68.
42 Stevens SK, Moore SG, Kaplan ID: Early and late bone-marrow changes after irradiation: MR evaluation. AJR Am J Roentgenol. 1990; 154:745-750
43 Yankelevitz DF, Henschke CI, Knapp PH, et al. Effect of radiation therapy on thoracic and lumbar bone marrow: evaluation with MR imaging. Am J Roentgenol 1991;157(1):87-92.
44 Kaplan PA, Asleson RJ, Klassen LW, Duggan MJ: Bone marrow patterns in aplastic anemia: Observations with 1.5-T MR imaging. Radiology. 1987; 164:441-444
45 Bacigalulpo A, Passweg J. Diagnosis and Treatment of Acquired Aplastic Anemia. Hematol Oncol Clin NA. 2009; 23: 159-170.
46 Burke S, Healy J. Musculoskeletal Manifestations of HIV Infection. Imaging: The British Institute of Radiology. 2002; 14:35-47
47 Geremia GK, McCluney KW, Adler SS, et al. The magnetic Resonance Hypointense Spine of AIDS. JCAT. 1990; 14:785-789.
48 Steinbach LS, Tehranzadeh J, Fleckenstein JL, et al. Human immunodeficiency virus infection: musculoskeletal manifestations. Radiology. 1993; 186:833-838.
49 Tripathi AK, Misra R, Kalra P, et al. Bone marrow abnormalities in HIV Disease. J Assoc Physicians India. 2005; 53:705-710.
50 Barosi G. Myelofibrosis with myeloid metaplasia. Hematol Oncol Clin NA. 2003;17:1211-1226