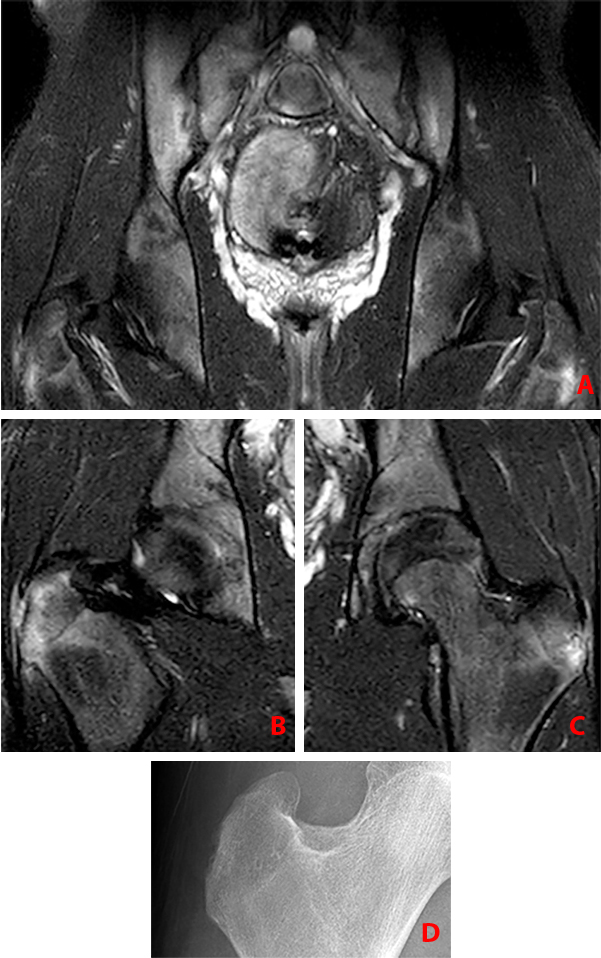
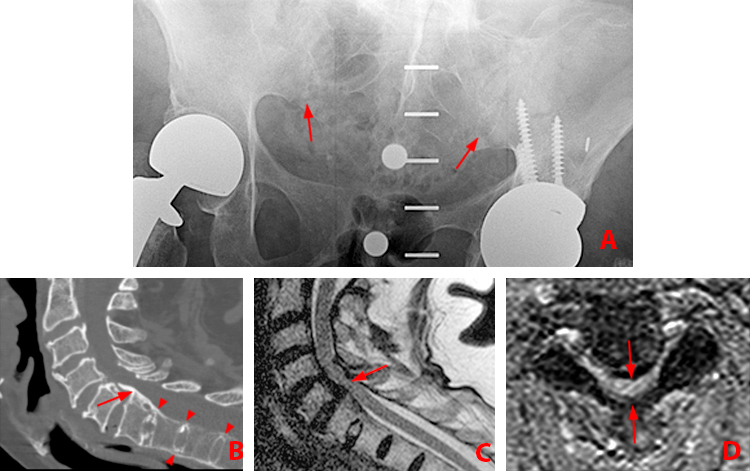
Clinical History: A 16 year-old male presented to his physician complaining of low back pain and stiffness and bilateral hip pain for 2 years. What are the findings? What is your diagnosis?
Findings
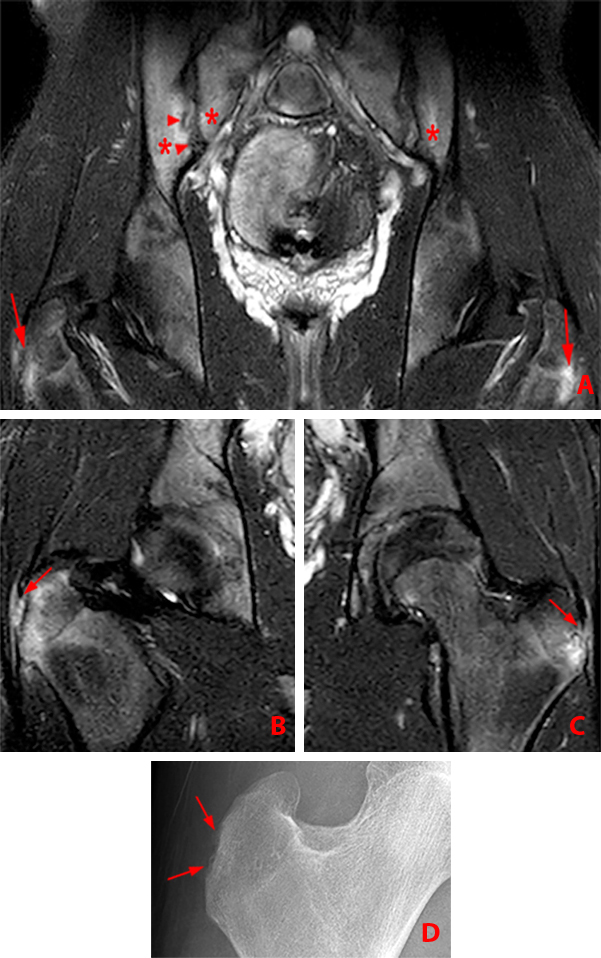
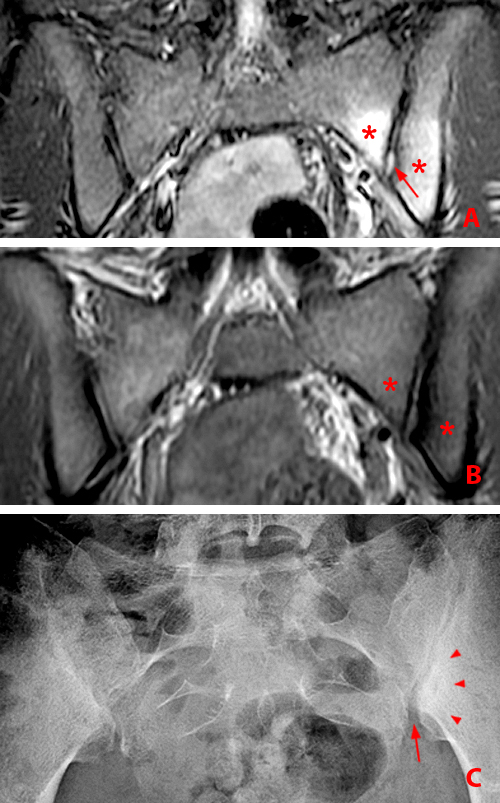
Figure 2:
(2a) Subchondral bone marrow edema (asterisks) is present along the caudal sacroiliac joints, with bilateral joint space widening and erosions on the right (arrowheads), indicative of bilateral sacroiliitis. Subcortical bone marrow edema within the greater trochanters is present (arrows).
(2b,2c) Bilateral enthesitis is present, manifested by subcortical bone marrow edema within each greater trochanter and edema within the entheses of the gluteus medius tendons (arrows).
(2d) Enthesitis of the greater trochanter demonstrated by a combination of cortical erosion and fluffy new bone formation along the greater trochanter (arrows).
(Click image to enlarge)
Diagnosis
Ankylosing spondylitis.
Introduction
Ankylosing spondylitis (AS) is a chronic inflammatory rheumatologic disorder that predominantly affects the axial skeleton and is characterized by sacroiliitis, spondylitis and enthesitis. AS is the most common of the seronegative spondyloarthritides, a group of disorders that are rheumatoid factor negative and are frequently associated with the presence of the HLA-B27 antigen. Ankylosing spondylitis affects both synovial and cartilaginous joints and entheses and is manifested by a combination of osseous erosions and new bone formation, ultimately leading to ankylosis. Men are much more commonly affected than women, with disease onset typically in the teen years to young adulthood. The disease may be associated with significant morbidity, including pain, rigidity, fractures and spinal deformity.1 The mainstay of imaging has been radiographs, which show characteristic changes enabling diagnosis in established disease. MRI can detect the disease much earlier so that appropriate treatment can be instituted quickly and have the greatest chance of altering disease outcome and prevent complications. The early MRI features may be subtle and confused with other entities, however. Radiologists need to be aware of the early disease findings on MRI and to consider ankylosing spondylitis in the differential diagnosis in young patients, especially males, with chronic low back pain.
Pathogenesis
The cause of the disease is unknown, though interaction between an infectious agent, possibly Klebsiella, and the HLA-B27 antigen has been hypothesized.2
Sacroiliac involvement is a hallmark of the disease and is required for diagnosis.3 Plasma cells and lymphocytes infiltrate the sacroiliac joints, both the synovial and ligamentous portions, resulting in subchondral edema, synovitis and joint effusion. The inflammatory infiltrates later lead to bony erosions, healing with new bone formation, which progresses to ankylosis. Tumor necrosis factor-alpha, bone morphogenetic protein and probably prostaglandins contribute to this process.4 The same inflammatory sequence occurs at entheses, the attachments of ligaments, tendons, fascia and capsules to bone.
Within the spine, enthesitis occurs at the junction of the vertebral body endplate and the annulus fibrosis, forming the so-called Romanus lesion. Erosions heal by formation of enchondral bone production, forming a new enthesis, a process associated with abundant nerve fibers and pain. The areas of new bone, syndesmophytes, are ossifications of the outer annulus. When they enlarge, they can involve the anterior longitudinal ligament, the posterior longitudinal ligament and the paravertebral connective tissues.1 Andersson lesions are inflammatory lesions that occur at the junction of the intervertebral disc and the vertebral endplate and can lead to interbody ankylosis. The apophyseal joints and the posterior ligaments are affected by synovitis and enthesitis, respectively, followed by ankylosis. The bamboo spine is the end-result of widespread ankylosis.
AS typically begins at the sacroiliac joints, generally ascending the spine, from lumbar to cervical, although it may skip levels. Lack of sacroiliac involvement has been considered to preclude the diagnosis of ankylosing spondylitis; however, spinal involvement without sacroiliitis has been reported.5 Development of osteoporosis occurs during the course of the disease, and along with the ankylosis places the spine at increased risk of fracture from even mild trauma.
Peripheral joints are less commonly involved, with mainly the large joints, the hips and shoulders developing synovitis, new bone formation and potentially ankylosis in patients with longstanding disease. Small joint disease is uncommon.
Epidemiology
Ankylosing spondylitis is the most frequently diagnosed spondyloarthritis, with a prevalence of 0.1 to 0.86%.1,6 The histocompatibility antigen HLA-B27 gene is present in 90-95% of persons with the disorder. Risk of development of the disease is 5% in those who are HLA-B27 positive.4 Ankylosing spondylitis is more common in Caucasians and Native Americans than in persons of Asian and African descent, related to the prevalence of the HLA-B27 antigen in these populations. Men are more commonly affected than women by a ratio of 4:1 to 10:1. Prevalence in women may be greater than previously thought, however, since women may have an atypical pattern of disease, with more peripheral joint involvement and isolated cervical spine disease in addition to sacroiliitis, resulting in underdiagnosis.1 Ankylosing spondylitis is a disease that begins in young persons, with onset generally in the second through fourth decades. 10% of patients develop the disease after age 39.1
Clinical Presentation
Ankylosing spondylitis is characterized by insidious inflammatory low back pain worse in the morning and after inactivity that improves with exercise, in contrast to mechanical low back pain. Other features include stiffness, limited mobility and pain over bony protuberances secondary to enthesitis. Constitutional symptoms may occur and include fatigue, anorexia, weight loss and low grade fever.1,4 Loss of function and debilitation develop with disease progression. A thoracic kyphotic deformity, with the head and neck protruded forward and gaze directed at the floor, and decreased chest excursion due to ankylosis of the axial joints of chest, characterize advanced disease. Disease progression is not universal, however. In less than 20% of patients with adult onset ankylosing spondylitis does the disease progress to significant disability.1 Young age at onset of symptoms and hip involvement are regarded as poor prognostic signs.4
Anterior uveitis commonly occurs in ankylosing spondylitis.4 Other extraskeletal disease manifestations are rare, and include aortitis, aortic valve insufficiency and pulmonary fibrosis.
Laboratory Tests
In addition to a strong association with HLA-B27, half of patients show elevated C-reactive protein concentrations. A weaker association with elevated ESR has been found.4,7
Imaging
Imaging is essential in the diagnosis of ankylosing spondylitis and depicts both active and chronic phases of the disease. Successive stages of disease can be identified, from early inflammatory involvement to fatty post-inflammatory changes, to bone formation and ankylosis8; different stages may coexist in the same patient.9 Radiographs are usually obtained first but CT and MRI are more sensitive and specific. MRI is able to detect the earliest changes of sacroiliitis: spondylitis, spondylodiscitis, facet arthritis, enthesitis and true ligamentous inflammatory involvement.8,10,11 CT is more sensitive for the changes of erosions, sclerosis and bone formation.9
Radiographs
Radiographs show characteristic changes in longstanding disease but are relatively insensitive in detecting early disease. Radiographic changes usually don’t develop until symptoms have been present for five years.12,13 Hallmark radiographic features are bone erosions, new bone formation and ankylosis. Osteoporosis is also a prominent feature.
Sacroiliac joints: The sacroiliac joint may be difficult to assess radiographically due to the obliquity of the joint and obscuration caused by overlying soft tissues. A modified Ferguson AP view best depicts this articulation. The iliac side of the joint shows changes first due to its thinner cartilage relative to the sacrum. Findings include the loss of definition of the subchondral bone plate, erosions, a widened joint space, and reactive sclerosis around the joint (Fig. 3). Ankylosis is the final stage of this process (Figs. 4, 5). The reactive sclerosis resolves when ankylosis occurs (Fig. 5).14 A key feature is bilateral and symmetric sacroiliac involvement; however, early in the disease, the pattern may be unilateral and asymmetric.
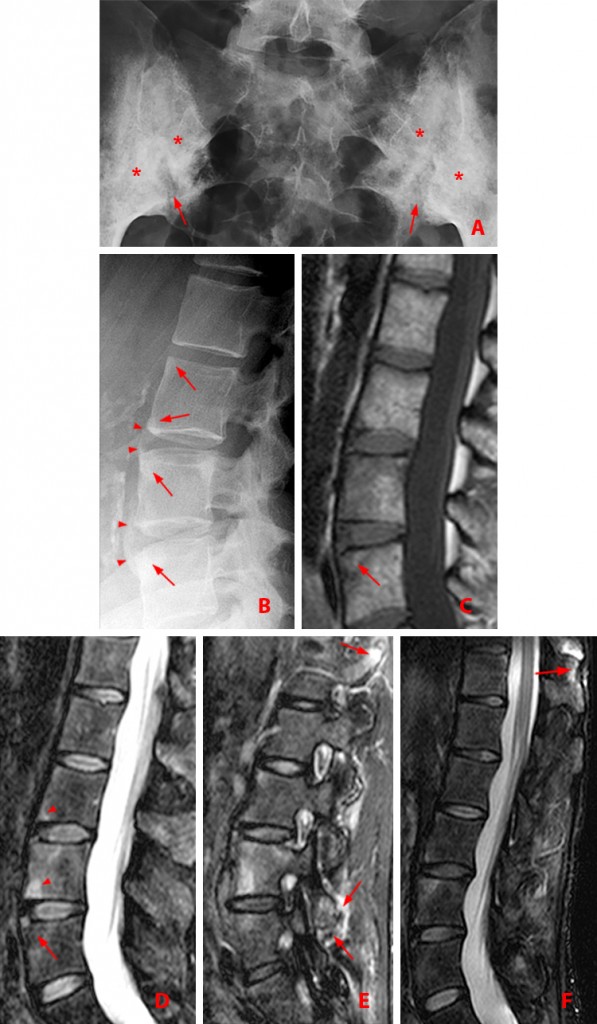
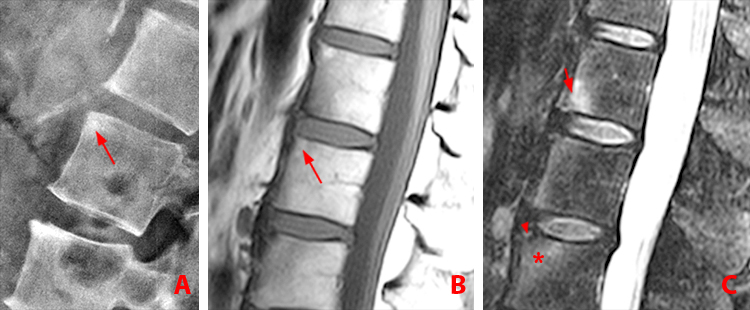
Figure 3:
43 year-old woman with AS and chronic low back and bilateral hip pain, and a combination of active and chronic disease.
(3a) Frontal radiograph of the sacroiliac joints shows bilateral symmetric sacroiliitis with erosions creating a widened appearance of the joints (arrows), and subchondral sclerosis (asterisks).
(3b) Lateral view of the lumbar spine shows squaring of the anterior vertebral bodies with loss of the expected concave anterior surface due to erosion and bone formation, producing “shiny corners” at L3 through L5 (arrows). Syndesmophytes are present at L4 and L5 (arrowheads).
(3c) T1-weighted sagittal MR image fails to show the syndesmophytes. Radiographs better depict the bony proliferative changes of AS. Subtle erosion along the anterior superior corner of the L5 vertebral body indicative of a Romanus lesion is accompanied by post-inflammatory fatty degeneration (arrow).
(3d) T2 fat-suppressed sagittal MR image shows hyperintense Romanus lesions at the anterior L3 and L4 vertebral bodies (arrowheads). The subtle erosion of the Romanus lesion at L5 is accompanied by post-inflammatory fatty degeneration (arrow).
(3e) T2 fat-suppressed sagittal MR image shows facet arthritis, manifested by bone marrow edema and surrounding soft tissue edema (arrows) at T12-L1 and L4-5.
(3f) T2 fat-suppressed sagittal MR image reveals enthesitis of the interspinous ligament at T11-12 (arrow).
(Click image to enlarge)
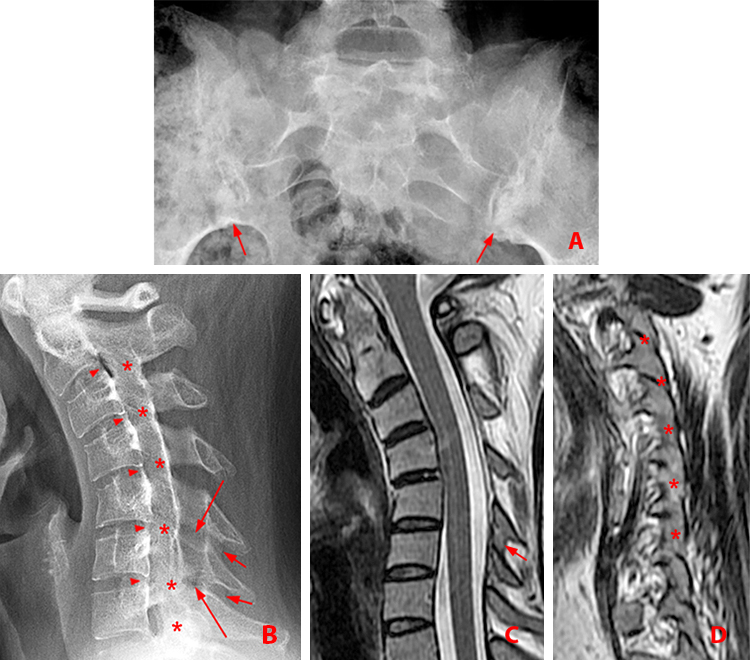
Figure 4:
28 year-old woman with chronic AS involving the sacroiliac joints and cervical spine.
(4a) Frontal view of the sacroiliac joints. Chronic bilateral sacroiliitis has resulted in partial bilateral ankylosis, best seen at the caudal aspects of the joints (arrows).
(4b) Lateral view of the cervical spine depicts multilevel bridging posterior vertebral body syndesmophytes (arrowheads), ankylosis of the facet joints from C2 to T1 (asterisks), and interlaminar (long arrows) and interspinous (short arrows) ankylosis at C5-6 and C6-7. Note the kyphotic deformity and osteopenia.
(4c) Sagittal T2-weighted MR image shows the interspinous ankylosis at C5-6 (short arrow).
(4d) Sagittal T2-weighted MR image shows the multilevel facet ankylosis (asterisks).
Figure 5:
64 year-old woman with chronic AS involving the cervical and thoracic spine and sacroiliac joints, and bilateral hip replacements. The patient complained of chronic neck pain and numbness and tingling in her hands. (Click image to enlarge)
(5a) Frontal view of the hips shows chronic ankylosis of the sacroiliac joints (arrows) without sclerosis.
(5b) Sagittal reconstruction cervical spine CT image demonstrates central spinal stenosis at C5-6 due to posterior syndesmophytosis (arrows). Note syndesmophytes also further caudally (arrowheads).
(5c) Sagittal T2-weighted MR image depicts the compression of the spinal cord caused by the stenosis (arrow). The posterior syndesmophytes at C5-6 are better discriminated on the CT.
(5d) Axial T2*-weighted MR image reveals the marked cord compression (arrows).
Figure 6:
58 year-old woman with AS and chronic back and bilateral hip pain.
(6a) Lateral lumbar radiograph reveals an inactive Romanus lesion at the anterosuperior corner of the L2 vertebral body producing a shiny corner (arrow) and squaring of the vertebral body.
(6b) Note the corresponding, more subtle area of fatty degeneration on the T1-weighted sagittal MR image (arrow).
(6c) Sagittal fat-suppressed T2-weighted image. Note the active Romanus lesion of the anterosuperior L3 vertebral body, with subtle erosion (arrowhead) and bone marrow edema (asterisk). Active Romanus lesion also at caudal L1 vertebral body (short arrow).
Spine: Romanus lesions are manifested by vertebral body corner erosions and bone formation characterized by fluffy amorphous bone sclerosis, forming so-called “shiny corners.” The process results in a squared appearance of the vertebral body with loss of anterior concavity (Figs. 3b, 6a).
As the spine becomes immobilized, the sclerosis may disappear, leaving only the squared appearance of the vertebral body (Fig. 7a). Syndesmophytes are visible as thin, vertically oriented osseous outgrowths along the vertebral body margins that may result in bridging of the vertebral bodies (Figs. 3a, 4b, 5b, 7a). Syndesmophytes are characteristically vertical; however, older age at disease onset may show more laterally displaced excrescences due to a bulging annulus.1 The aseptic spondylodiscitis of AS shows endplate erosions and sclerosis and disc calcification. The disc space is usually preserved. Facet and uncovertebral joint disease manifestations of erosions and subchondral sclerosis may be difficult to detect by radiography.
Figure 7:
72 year old man with chronic AS, severe neck pain, loss of cervical lordosis and forward flexion of the neck.
(7a) Lateral radiograph of the cervical spine shows multilevel facet ankylosis (asterisks) and bridging syndesmophytes (arrows), and forward flexion of the neck.
(7b) Sagittal T1-weighted MR image shows the thin bridging anterior and posterior syndesmophytes due to fatty marrow content (arrows).
(7c) Sagittal T1-weighted MR image. Note the multilevel facet ankyloses (asterisks).
Enthesitis of the posterior ligamentous attachments is manifested by erosions, calcifications and ossification (Fig 4b).
Peripheral ankylosis resulting from syndesmophytes and spinal ligament ossification, and central ankylosis through discs and facet joints result in the appearance of the spine as a fused osseous column, the “bamboo spine”.1 The dagger sign is an osseous vertical stripe created by fusion of the spinous processes on frontal radiographs.
In the cervical spine, erosion of the dens and atlantoaxial subluxation can occur, potentially causing confusion with rheumatoid arthritis.
AS may initially involve the spine at T12-L1; however, it is usually detected radiographically in the lumbar spine.14 The disease then ascends to the thoracic and cervical levels but can skip levels.1 Spinal deformities include loss of lumbar and cervical lordosis (Figs. 4b, 7, 14) and a kyphotic deformity of the thoracic spine (Fig 13).
Hip: Involvement of the hip is usually bilateral and symmetric.1 Joint space loss is variable. When it occurs, it is usually concentric and is associated with axial migration of the femoral head. A collar of osteophytes occurs at the junction of the femoral head and neck, not to be confused with the lateral osseous bump of femoroacetabular impingement. Ankylosis occurs in end stage disease. (Fig. 8)
Figure 8:
22 year-old man with AS and left hip pain.
(8a) Frontal view of the left hip shows marked superior joint space loss, subchondral cysts and prominent osteophyte at the lateral femoral head-neck junction. Superior joint space loss is less common than concentric loss.
(8b) Frontal view including caudal sacroiliac joints shows bilateral sacroiliitis, with left articular surface erosions and sclerosis (arrowheads) and a small erosion on the right (arrow).
MRI
Magnetic resonance imaging represents a major advance in the diagnosis of AS in that it enables early detection of disease, when radiographs are normal. Furthermore, MRI shows abnormalities in patients with clinical disease in whom conventional laboratory markers of disease activity such and CRP and ESR may be normal.15 While active and chronic disease is shown on MRI, the syndesmophytes of chronic disease are more conspicuous on radiographs and CT (Fig. 3b, c).
Sacroiliac joints: T1-weighted, STIR, and T1-weighted fat-suppressed images obtained following gadolinium contrast administration obtained in the axial and oblique coronal planes best assess the sacroiliac joints. The oblique coronal images are obtained parallel to the long axis of the sacrum. STIR is preferred over T2 fat-suppressed images as it is more sensitive in detecting subchondral bone marrow edema.7 The posterior caudal third of the joint is most commonly affected.16
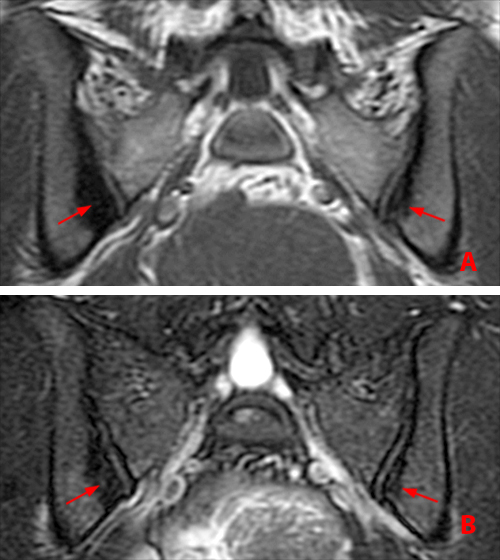
Active disease (Figs. 1, 9) is characterized by inflammation, with subchondral bone marrow edema that is low signal intensity on T1-weighted and hyperintense on STIR/ T2 images, and by gadolinium contrast enhancement. These findings may be associated with articular surface erosions, which are low signal intensity on T1 and STIR/T2 (Figs. 1a, 9c),17 and synovitis and joint effusions (Fig. 10). Enthesitis is characterized by STIR/ T2 hyperintensity at the ligamentous attachments of the pelvis (Fig. 9a) and hips (Fig. 1b, c).
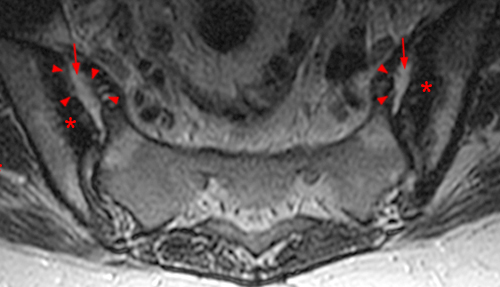
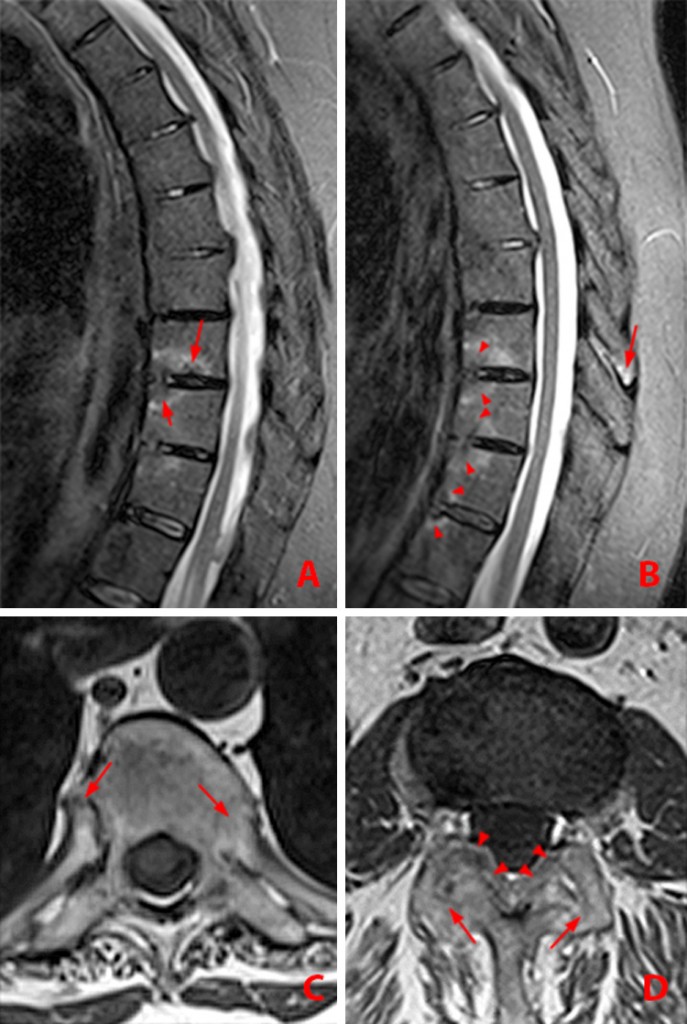
Figure 9:
35 year-old man with AS and sacral pain with active sacroiliitis. Coronal oblique MR images.
(9a) T2-weighted MR image shows bilateral symmetric pattern of subchondral bone marrow edema (asterisks) and enthesitis at the gluteus maximus attachments (arrowheads).
(9b) T1-weighted MR image. Low signal intensity bone marrow edema along the caudal iliac side of the left sacroiliac joint (asterisk) is less conspicuous than the T2 changes.
(9c) T2-weighted image depicts erosions along the superior right sacroiliac joint (arrows).
(Click to enlarge)
Figure 10:
37 year-old man with AS, low back pain and bilateral symmetric sacroiliitis. Axial T2-weighted image from a lumbar spine MRI shows a combination of active sacroiliitis with erosions (arrowheads) and joint effusions (arrows) and the chronic disease finding of subchondral sclerosis (asterisks).
Synovitis and joint effusion are both hyperintense on T2/STIR and cannot be distinguished on these sequences. Gadolinium-enhanced T1-weighted images allow differentiation of the enhancing synovium from the low signal intensity joint effusion. Gadolinium contrast-enhanced images are also more sensitive in detection of active disease, supporting its use.7 The bone marrow edema of active disease on STIR and T2 weighted images may be minimal, overlooked or misdiagnosed as degenerative disease. Such lesions contrast-enhance on gadolinium-enhanced images, which have also been found to show additional disease not visible on STIR/T2, improving diagnostic accuracy. In symptomatic patients, the subchondral edema and enhancement of active disease on MRI is positively correlated with elevated CRP levels.7 The subchondral bone marrow edema and enhancement of active disease is replaced in chronic disease by fatty marrow degeneration, which is T1 hyperintense.
Chronic disease (Fig. 11) is characterized by the enchondral bone formation of the healing response and is low signal intensity on T1 and T2-weighted images, and includes subchondral sclerosis, joint space narrowing, bone bridging and ankylosis. Ankylosis is initially hypointense and later shows T1 signal increase indicative of fatty marrow bridging. Patients with chronic disease may also have active disease findings (Figs. 10, 11). Fatty subchondral bone marrow is seen in patients with both active disease and chronic disease on MRI (Fig. 11).
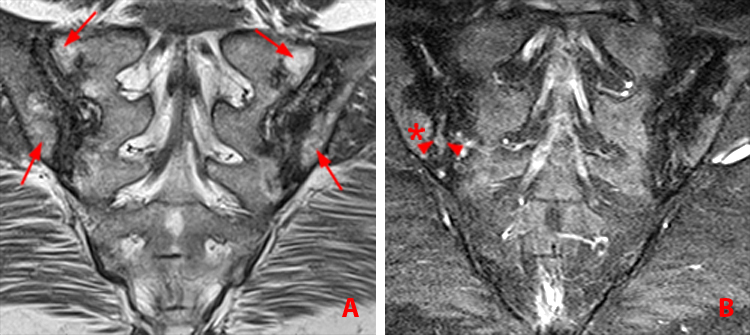
Figure 11:
45 year old woman with AS, 25-year history of low back pain, and chronic sacroiliitis.
(11a) Coronal oblique T1-weighted MR image demonstrates bilateral low signal intensity along the articular surfaces and within the joint, with joint space narrowing and partial bridging. Note fatty marrow degeneration high signal intensity (arrows).
(11b) Coronal oblique T2 fat-suppressed image additionally shows components of active disease with erosions (arrowheads) and subchondral edema (asterisks) caudally on the right.
Spine: Similar to the sacroiliac joints, 3 classes of lesions occur in the spine: acute inflammatory lesions, post-inflammatory fatty bone marrow degeneration and ankylosis.18 The acute inflammatory Romanus lesion is a circumscribed triangular-shaped area of corner edema within the vertebral body that is STIR/ T2-hyperintense and enhances on T1-weighted post-gadolinium images, corresponding to enthesitis at the attachment of the annulus fibrosis to the vertebral body endplate (Figs. 3d, 6c, 12a, b).
Figure 12:
37 year-old man with AS, back pain and active spondylitis. Sagittal MR images.
(12a) STIR image shows hyperintense quadrantic type Romanus lesions anteriorly within the L4 and L5 vertebral bodies and a triangular corner lesion superiorly at L2 (arrows). Note hyperintense posterior ligament enthesitis along the spinous processes (arrowheads).
(12b) Corresponding T1-weighted post-gadolinium image reveals enhancement of the Romanus lesions (arrows). Note enhancement of the interspinous ligament at L3-4 (curved arrow) and enhancement of the spinous processes related to enthesitis (arrowheads).
(12c) STIR image shows facet arthritis with hyperintense subchondral bone marrow edema (arrows).
(12d) Corresponding T1-weighted fat-suppressed post-gadolinium image reveals associated enhancement (arrows).
(Click image to enlarge)
The Romanus lesion is also known as marginal spondylitis8 and the “MRI corner sign”. The finding may be quadrant- shaped rather than triangular.19 Awareness of this sign is important since MRI of the spine is often ordered without accompanying radiographs of the sacroiliac joints, and the finding may be present and suggest the diagnosis when sacroiliac radiographs are inconclusive (19). Romanus lesions are easily overlooked or attributed to the Modic marrow changes of degenerative disease, however. When more than 3 corners are involved in the absence of osteophytes or Schmorl nodes, the specificity of this finding is 81%; the specificity reaches 97% in patients younger than 40.20 Erosions show decreased signal intensity and are more conspicuous on CT.9
Following acute inflammation, fatty degeneration T1 hyperintensity replaces the marrow edema of the Romanus lesion, and only at this stage are shiny corners detected on radiographs8(Fig. 3b-d). Such areas of fatty deposition characterize the inactive Romanus lesion and are predictive of formation of syndesmophytes.20
The syndesmophytes of chronic disease are difficult to see on MRI due to their thinness and low signal intensity. Radiographs and CT are superior in detecting these lesions. Later, when the lesions show fatty marrow signal intensity, they may be more apparent on T1-weighted images (Fig. 7).
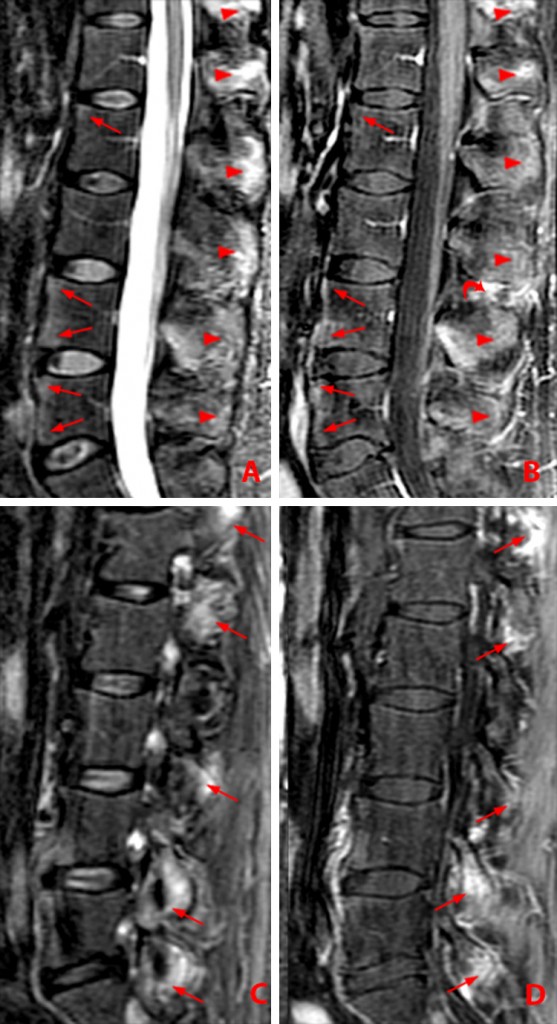
Discovertebral lesions, the so-called Andersson lesions, are the same as Romanus lesions but are more central in location at the discovertebral junction. Erosions are seen later on CT.9 Active lesions can look like Modic type 1 changes of degenerative disc disease, so it is important to search for corner lesions of spondylitis, facet joint involvement, and enthesitis of the posterior ligaments in order to differentiate them (Fig. 13 a, b). Edema and enhancement may also be present in the center or throughout the disc, simulating discitis due to infection. Interbody ankylosis marks chronic disease (Fig 15).
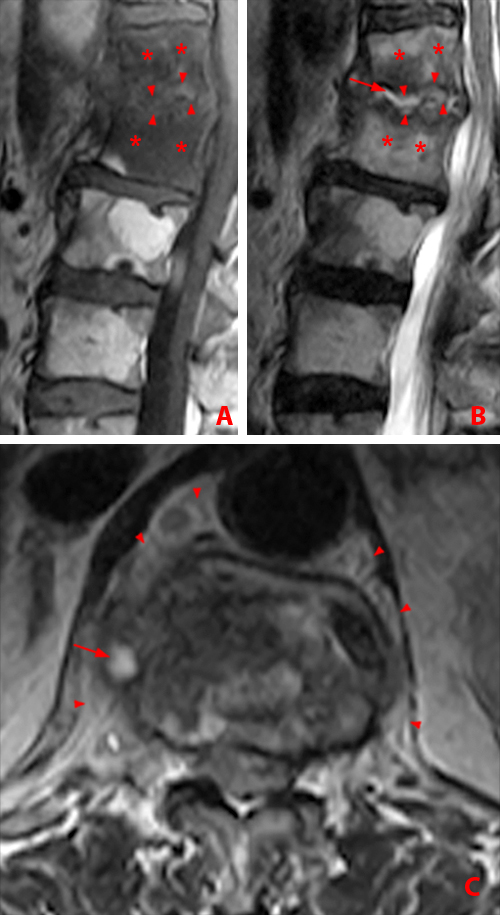
Figure 13:
56 year-old woman with AS. MR images of thoracic spine. Sagittal STIR images, a and b. Axial T1-weighted images, c and d.
(13a) Andersson lesion along inferior endplate of T9 with adjacent bone marrow edema (arrow). Romanus corner lesion within the superior anterior T10 vertebral body (short arrow). Bridging anterior syndesmophytes are present further cephalad.
(13b) Romanus anterior corner lesions at T9-10, T10-11 and T11-12 (arrowheads) and T9-10 spinous process enthesitis (arrow) are shown on adjacent image.
(13c) Axial T1-weighted image demonstrates bilateral costovertebral ankylosis (arrows), more advanced on the left.
(13d) Axial T1-weighted image shows bilateral facet ankylosis (arrows) and ossification of the ligamentum flavum (arrowheads).
(Click image to enlarge)
Facet joint involvement is manifested by subchondral bone marrow and adjacent soft tissue edema and enhancement, which is usually associated with similar changes in the pedicles17 (Fig. 12 c, d). Joint effusions and synovial enhancement are additional manifestations of facet arthritis. Ankylosis may be detected (Fig. 13d) but usually is better seen on radiographs and CT.
In addition to the enthesitis of the anterior and posterior longitudinal ligaments, the posterior ligaments, i.e., the ligamentum flavum (Fig, 13d), supraspinous and interspinous ligaments are typically involved. Posterior enthesitis may be the earliest sign of disease in the lumbar sign and has a specificity of 87% for spondyloarthritis.21 Edema and enhancement occur within the ligaments and at the entheses. The ligaments may be thickened, and there may be associated osteitis of adjacent bone marrow in the spinous processes8 (Figs. 3f, 12a,b and 13b). Ankylosis of the ligamentous joints occurs similar to other affected areas (Figs. 4c, 13d).
The sternocostal and costovertebral joints show arthritis manifested by subchondral bone marrow and adjacent soft tissue edema and enhancement, with ankylosis occurring in chronic disease (Fig. 13c).
As in the sacroiliac joints, coexistence of active inflammatory changes (edema, erosion and enhancement), post-inflammatory fatty degeneration and ankylosis may be seen in the spine (Figs. 3, 13).
Complications
The osteoporotic rigid, fused spine of chronic AS is susceptible to fracture from even minimal trauma. Sometimes fractures may occur without known injury.22 Such fractures are typically horizontal, through the disc or vertebral body, and involve all 3 columns. The fracture may extend as tears of the posterior ligaments rather than involve the neural arch. Radiographs are relatively insensitive due to the coexistent osteoporosis. MDCT and MRI are superior in detecting such fractures and MRI also depicts tears of the longitudinal and posterior ligaments (Fig. 14).
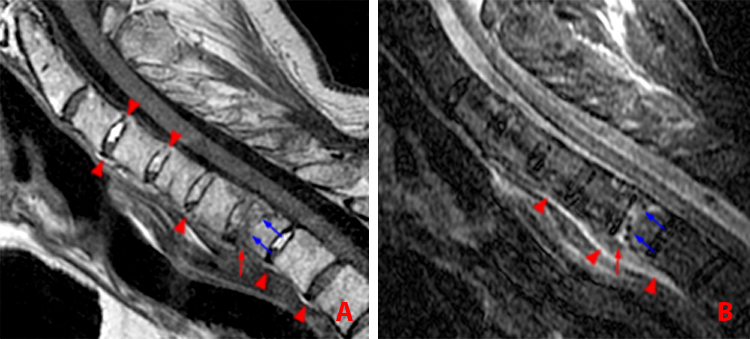
Figure 14:
68 year-old man with AS and neck pain who fell in the shower.
a. Sagittal T1-weighted image shows horizontal fracture of the C7 vertebral body (blue arrows). Note fracture through the ossified anterior longitudinal ligament (red arrow). Bridging syndesmophtes are present anteriorly at multiple levels (arrowheads) and posteriorly at C3-4 and C4-5 (arrowheads).
b. Sagittal STIR image reveals edema surrounding the fracture line (blue arrows), edema of the anterior longitudinal ligament (red arrow) and prevertebral edema (arrowheads).
While spontaneous fracture healing may occur, persistent motion at the fracture site can lead to pseudarthrosis, instability and cord injury. Pseudarthrosis is manifested by irregular osteolysis along the discovertebral junction, sclerosis and vacuum phenomenon in the intervertebral space23 (Fig. 15).
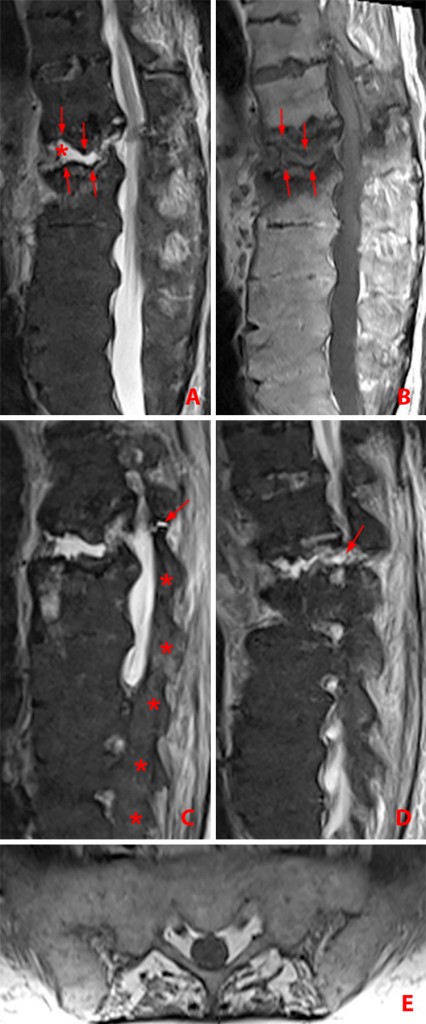
Figure 15:
70 year-old man with AS with history of prior fall. Sagittal MR images show chronic fracture and pseudarthrosis of the L1 vertebral body and neural arch.
(15a) STIR image depicts low signal intensity osseous erosion and sclerosis along the margin of the pseudarthrosis (arrows) with an intervening fluid-filled cleft likely corresponding to avascular necrosis (asterisk).
(15b) Corresponding T1-weighted image demonstrates osseous erosions and sclerosis (arrows). Multilevel interbody ankylosis is present at L2-3 through L5-S1.
(15c) Adjacent STIR image shows the pseudarthrosis also involving the posterior elements (arrow) and multilevel facet ankylosis (asterisks).
(15d) STIR image shows the pseudarthrosis extending through the pedicle (arrow).
(15e) Axial T1 weighted image shows bilateral sacroiliac ankylosis.
(Click image to enlarge)
Spinal stenosis is another potential complication caused by ossification of the posterior longitudinal ligament and ligamentum flavum (Fig. 5b-d). Dural ectasia with arachnoiditis and cauda equina syndrome have been reported in patients with longstanding disease.24
Treatment
NSAIDS and intensive physical therapy are the first line of treatment of AS. While NSAIDs are effective, half of ankylosing spondylitis patients report insufficient control of symptoms with these agents. Furthermore, cardiac and gastrointestinal side effects can restrict their use. The disease modifying anti-rheumatic drugs methotrexate and sulfasalazine have been found to improve the peripheral arthritis associated with AS but not the axial disease.
Tumor necrosis factor (TNF) blocking therapy has been shown to be effective and represents a major advance in the treatment of AS in cases refractory to conventional therapy.4,25 Anti-TNF alpha treatment results in a decrease in spinal inflammation as detected by MRI; almost complete resolution of inflammation was seen in most patients in a study by Braun.5 Clinical improvement has been shown to correlate with reduction in acute inflammatory spinal changes on MRI.26 It is important to begin these agents early for greatest efficacy and to potentially change the course of the disease, underscoring the need for early diagnosis, which is greatly facilitated by MRI. Monitoring disease activity and response to treatment are other important roles of MRI.
Differential Diagnosis
AS is the most common of the seronegative spondyloarthritides. Other subtypes include enteropathic, psoriatic, reactive and undifferentiated.
Enteropathic spondyloarthritis is associated with Crohn disease and ulcerative colitis and has imaging features similar to AS. Psoriatic spondyloarthritis is differentiated by the presence of greater peripheral joint involvement, skin lesions and bilateral asymmetric sacroiliitis. In contrast to the syndesmophytes of AS, pseudosyndesmophytes are thick irregular areas of new bone formation with large implantation bases at vertebral corners that are usually unilateral and asymmetric. Facet involvement is less common in psoriatic spondyloarthritis.
Figure 16:
21 year-old woman with psoriasis, left sacroiliac and hip pain and unilateral sacroiliitis. Sacroiliac involvement in psoriatic arthritis is usually bilateral and asymmetric; however, unilateral disease may occur. Septic arthritis was excluded clinically.
(16a) Coronal STIR image shows sacroiliitis along the caudal left sacroiliac joint, with subchondral edema (asterisks), joint effusion and mild joint widening due to iliac-sided erosion (arrow).
(16b) Corresponding coronal T1-weighted image shows low signal intensity subchondral bone marrow edema and sclerosis (asterisks).
(16c) AP radiographic findings are more subtle, with mild left joint space widening (arrows) and subchondral sclerosis (arrowheads).
(Click image to enlarge)
Reactive spondyloarthritis (formerly Reiter disease) shows bilateral asymmetric sacroiliitis, spinal pseudosyndesmophytes, less common facet joint involvement, peripheral arthritis, usually of the small joints of the lower extremity, and is associated with genitourinary infection.
Undifferentiated spondyloarthritis is diagnosed in persons with spondyloarthritis without definite radiologic signs of sacroiliitis.27
Diffuse idiopathic skeletal hyperostosis (DISH) manifests thick flowing ossifications in contrast to the thin vertical syndesmophtes of AS (Fig. 17). Other differentiating features are lack of facet and sacroiliac joint involvement.
Degenerative disc disease vertebral body osteophytes are differentiated from syndesmophytes by a location slightly further from the disc and directed more horizontally. Andersson and Romanus lesions may be mistaken for Modic changes. The latter are broader in shape and semicircular rather than triangular or quadrantic and associated with disc degeneration, osteophytes and Schmorl’s nodes(Fig. 18).19
Figure 18:
41 year-old man with degenerative disc disease at L5-S1. MR sagittal images.
a. T1-weighted image reveals degenerative loss of disc height, low signal intensity Schmorl’s node (arrow) along the inferior endplate of L5 with low signal intensity broad Modic 1 changes along the L5 and S1 endplates (arrowheads).
b. T2-weighted image depicts degenerative loss of disc signal and height, Schmorl’s node (arrow) and adjacent broad high signal intensity Modic 1 changes (arrowheads).
c. Adjacent T1-weighted image demonstrates associated anterior osteophytes (arrows).
Osteitis condensans ilii is a triangular shaped area of low signal intensity along the anterior and middle third of the ilium adjacent to the sacroiliac joints seen principally in multiparous women due to stress on the joints cause by pregnancy and childbirth (Fig 19).
Figure 19:
30 year-old woman with bilateral osteitis condensans ilii. Coronal T1-weighted (a) and STIR (b) images show the typical triangular-shaped areas of low signal intensity along the iliac sides of the sacroiliac joints (arrows). Note the intact subchondral bone plates and lack of bone marrow edema and joint effusions. (Click image to enlarge)
Septic spondylodiscitis differentiation from Andersson lesions may be difficult (Fig. 20). The presence of paravertebral and epidural soft tissue involvement and circumferential enhancement in the soft tissues anterior to the vertebral body indicate infection. Pseudarthrosis secondary to vertebral body fracture can be mistaken for spondylodiscitis. Detection of fracture of the posterior elements enables the correct diagnosis to be made.
Figure 20:
58 year-old man with back pain for 3 months and septic spondylodiscitis due to staphylococcus aureus at T12-L1. MR images.
(20a) T1-weighted sagittal image demonstrates endplate erosions (arrowheads) and diffuse low signal intensity bone marrow edema within the vertebral bodies (arrows).
(20b) Corresponding T2-weighted sagittal image reveals endplate erosions (arrowheads), bone marrow edema (asterisks) and fluid signal within the joint (arrow).
(20c) Axial T2-weighted shows accompanying small right paravertebral abscess (arrow) and extensive anterior and lateral paravertebral soft tissue edema (arrowheads).
(Click image to enlarge)
Infectious sacroiliitis produces unilateral disease in contrast to AS. Early in the disease, however, AS may be unilateral. Whenever unilateral sacroiliitis is present, septic arthritis needs to be excluded. Involvement of the surrounding soft tissue is a feature of infection but not AS (Fig. 21).
Figure 21:
17 year-old male with left pelvic and buttock pain and septic sacroiliitis from staphylococcus aureus. Coronal oblique STIR MR image shows unilateral sacroiliitis with joint effusion (curved arrow), subchondral edema (asterisk), adjacent soft tissue edema (arrowheads) and abscess caudal to the joint (arrow).
Conclusion
AS is the most common seronegative spondyloarthritis, characterized by predominantly axial involvement of the spine and sacroiliac joints and a symmetric, bilateral pattern of disease. It is painful and potentially debilitating, and may result in a greatly diminished quality of life. New drug therapies can change the course of the disease if begun early, making early detection imperative. MRI allows diagnosis at the beginning of the disease course due to its ability to show the earliest findings of bone and soft tissue edema, joint effusions and synovitis. In patients with low back pain, lumbar spine MRI is often ordered before radiographs, so it is necessary to be aware of the spine manifestations of the disease on MRI, and to remember the included sacroiliac joints in the search pattern. Romanus and Andersson lesions should be considered in the differential diagnosis of endplate marrow changes, especially in young patients without degenerative disc disease. It is important to remember that in chronic disease, active disease may coexist, and such patients may benefit from continued pharmacologic therapy. In addition to disease detection, MRI is useful in monitoring the response to treatment.
References
- Resnick D, Diagnosis of Bone and Joint Disorders. 3rd Ed. Philadelphia: Saunders; 1995 ↩
- Van der Linden S, van der Heijde D. Ankylosing spondylitis: clinical features. Rheum Dis Clin North Am 1998; 24:663-676 ↩
- Van der Linden S, Valkenburg HA, Cats A. Evaluation of diagnostic criteria for ankylosing spondylitis: a proposal for modification of the New York criteria. Arthritis Rheum 1984; 27:361-368 ↩
- Braun J, Sieper J. Ankylosing spondylitis. Lancet 2007; 369:1379-1390. ↩
- Braun J, Landewe R, Hermann K, et al. Major reduction in spinal inflammation in patients with ankylosing spondylitis after treatment with infliximab: results of a multicenter, randomized, double-blind, placebo-controlled MRI study. Arthritis Rheum 2006; 54:1646-1652 ↩
- Braun J, Bollow M, Remlinger G, et al. Prevalence of spondylarthropathies in HLA-B27 positive and negative blood donors. Arthritis Rheum 1998; 41:58-67 ↩
- Bredella M, Steinbach L, Morgan S, et al. MRI of the sacroiliac joints in patients with moderate to severe ankylosing spondylitis. AJR 2006; 187:1420-1426 ↩
- Hermann K, Althoff C, Schneider U, et al. Spinal changes in patients with spondyloarthritis: comparison of MR imaging and radiographic appearances. Radiographics 2005; 253:559-569 ↩
- Lacout A, Rousselin B, Pelage J-P. CT and MRI of spine and sacroiliac involvement in spondyloarthropathy. AJR 2008; 191:1016-1023 ↩
- Ball J. Enthesopathy of rheumatoid and ankylosing spondylitis. Ann Rheum Dis 1971; 30:213-223 ↩
- Hermann K, Bollow M. MRI of the axial skeleton in rheumatoid disease. Best Pract Res Clin Rheumatol 2004; 18:881-907 ↩
- Chary-Valckenaere I, d’Agostino M, Loeuille D. Role for imaging studies in ankylosing spondylitis. Joint Bone Spine 2011; 116:292-301 ↩
- Guglielmi G, Cascavilla A, Scalzo G, et al. Imaging findings of sacroiliac joints in spondyloarthropathies and other rheumatic conditions. Radiol Med (Torino) 2011; 116:292-301 ↩
- Brower A, Flemming D. Arthritis in Black and White. 2nd Ed. Philadelphia: Saunders; 1997 ↩
- Spoorenberg A, van der Heijde D, de Klerk E, et al. Relative value of erythrocyte sedimentation rate and C-reactive protein in assessment of disease activity in ankylosing spondylitis. J Rheumatol 1999; 26:980-984 ↩
- Sieper J, Rudwaleit M, Baraliakos X, et al. The Assessment of Spondyloarthritis International Society (ASAS) handbook: a guide to assess spondyloarthritis. Ann Rheum Dis 2009; 68(suppl 2):1-44 ↩
- Canella C, Schau B, Ribeiro E, et al. MRI in seronegative spondyloarthritis: imaging features and differential diagnosis in the spine and sacroiliac joints. AJR 2013; 200:149-157 ↩
- Hermann KG, Bollow M. Magnetic resonance tomography in spondylarthropathies. Aktuelle Rheumatol 2002; 27:323-331 ↩
- Kim N, Choi J-Y, Hong S, et al. “MR corner sign”: value for predicting presence of ankylosing spondylitis. AJR 2008; 191:124-128 ↩
- Bennett A, Rehman A, Hensor E, et al. The fatty Romanus lesion: a noninflammatory spinal MRI lesion specific for axial spondyloarthropathy. Ann Rheum Dis 2010; 69:891-894 ↩
- Bennett A, Rehman A, Hensor E, et al. Evaluation of the diagnostic utility of spinal magnetic resonance imaging in axial spondyloarthritis. Arthritis Rheum 2009; 60:1331-1341 ↩
- Wang Y, Teng M, Chang C, et al. Imaging manifestations of spinal fractures in ankylosing spondylitis. AJNR 2005; 26:2067-2076 ↩
- Campagna R, Pessis E, Feydy A, et al. Fractures of the ankylosed spine: MDCT and MRI with emphasis on individual anatomic spinal structures. AJR 2009; 192:987-995 ↩
- Grosman H, Gray R, St. Louis E. CT of long-standing ankylosing spondylitis with cauda equine syndrome. AJNR 1983; 4:1077-1080 ↩
- Coates L, Marzo-Ortega H, Bennett A, et al. Anti-TNF therapy in ankylosing spondylitis: insights for the clinician. Ther Adv Musculoskelet Dis 2010; 2(1):37-43 ↩
- Braun J, Baraliakos X, Golder W, et al. MRI examinations of the spine in patients with ankylosing spondylitis, before and after successful therapy with infliximab: evaluation of a new scoring system. Arthritis Rheum 2003; 48:1126-1136 ↩
- Dougados M, van der Linden S, Juhlin R et al. The European Spondyloarthropathy Study Group preliminary criteria for the classification of spondyloarthropathy. Arthritis Rheum 1991; 34:1218-1227 ↩