Clinical History
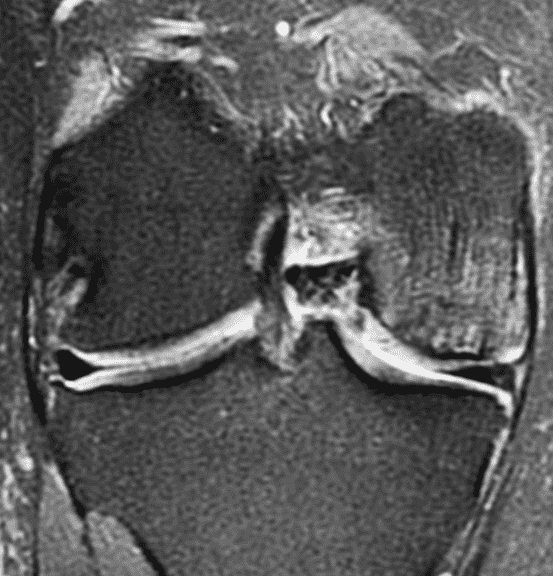
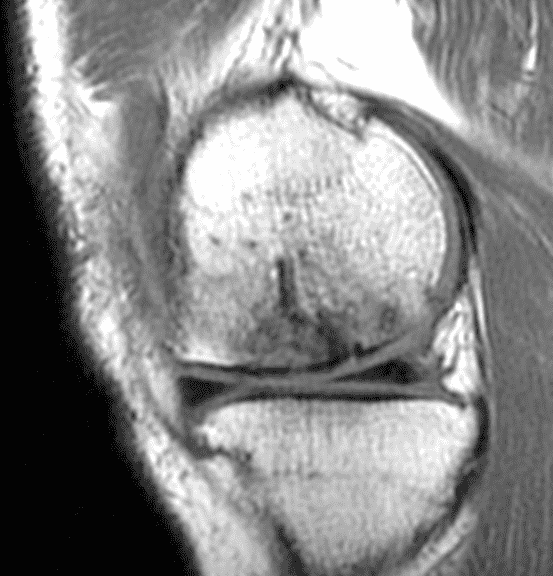
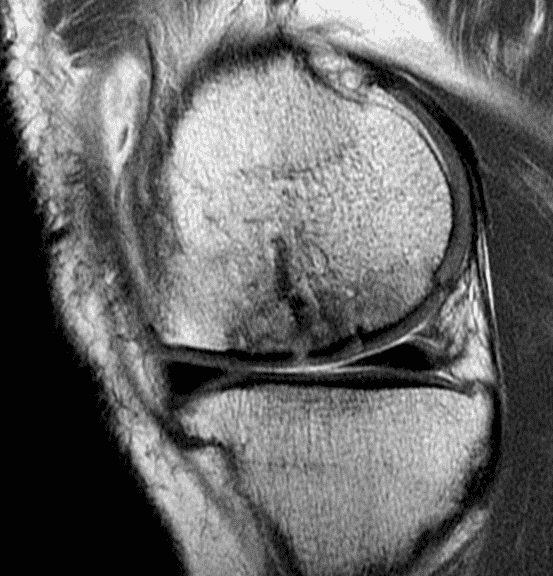
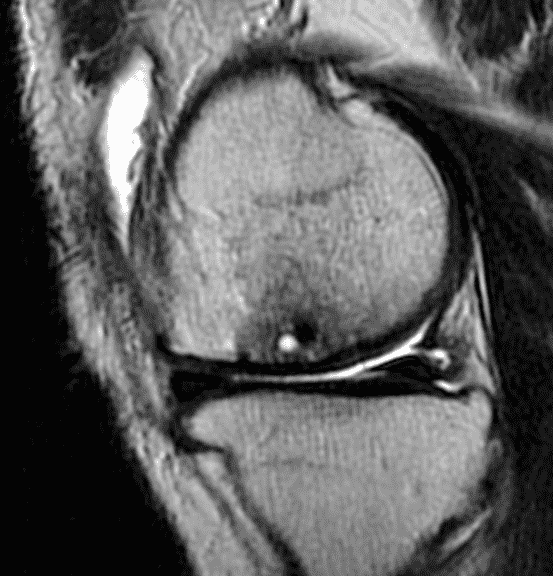
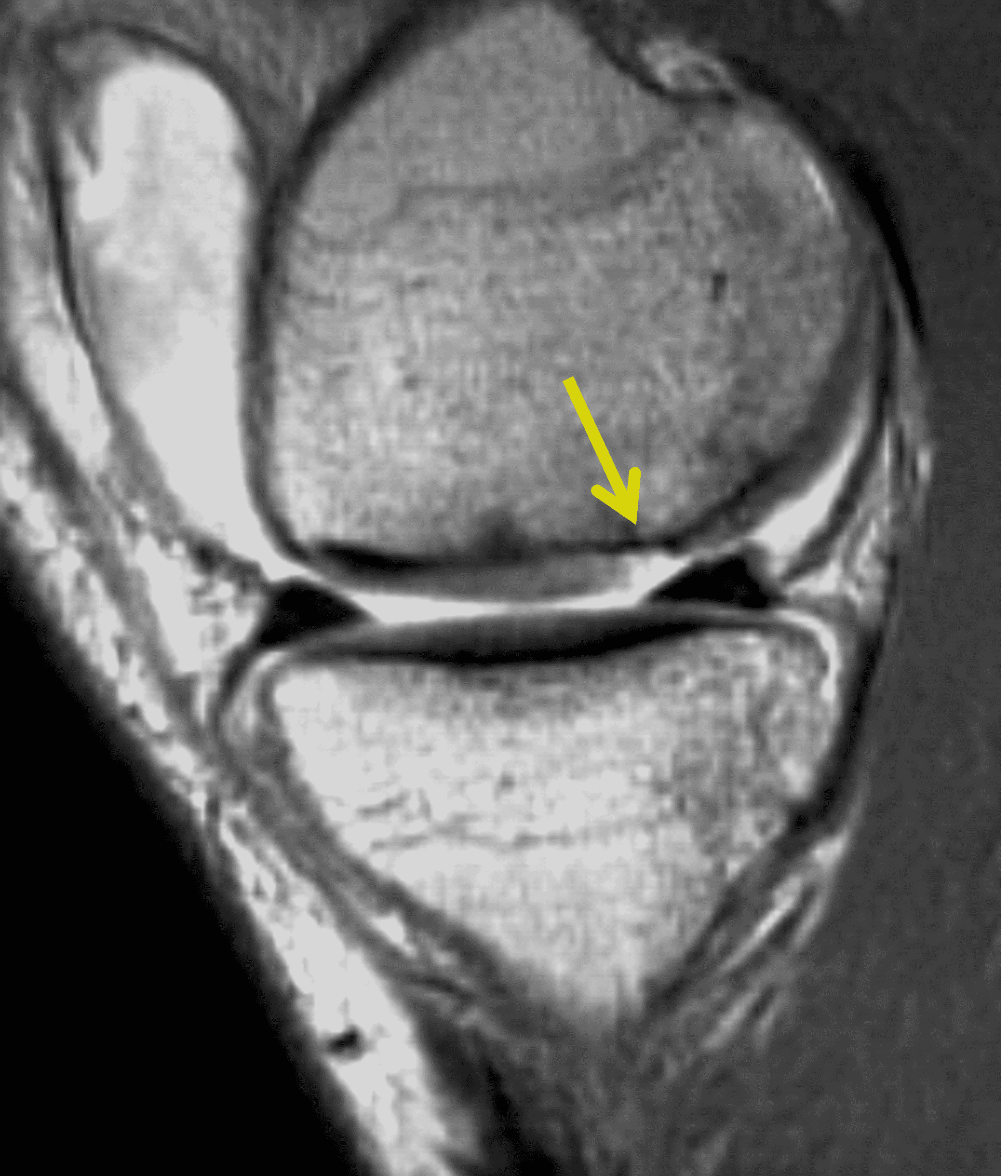
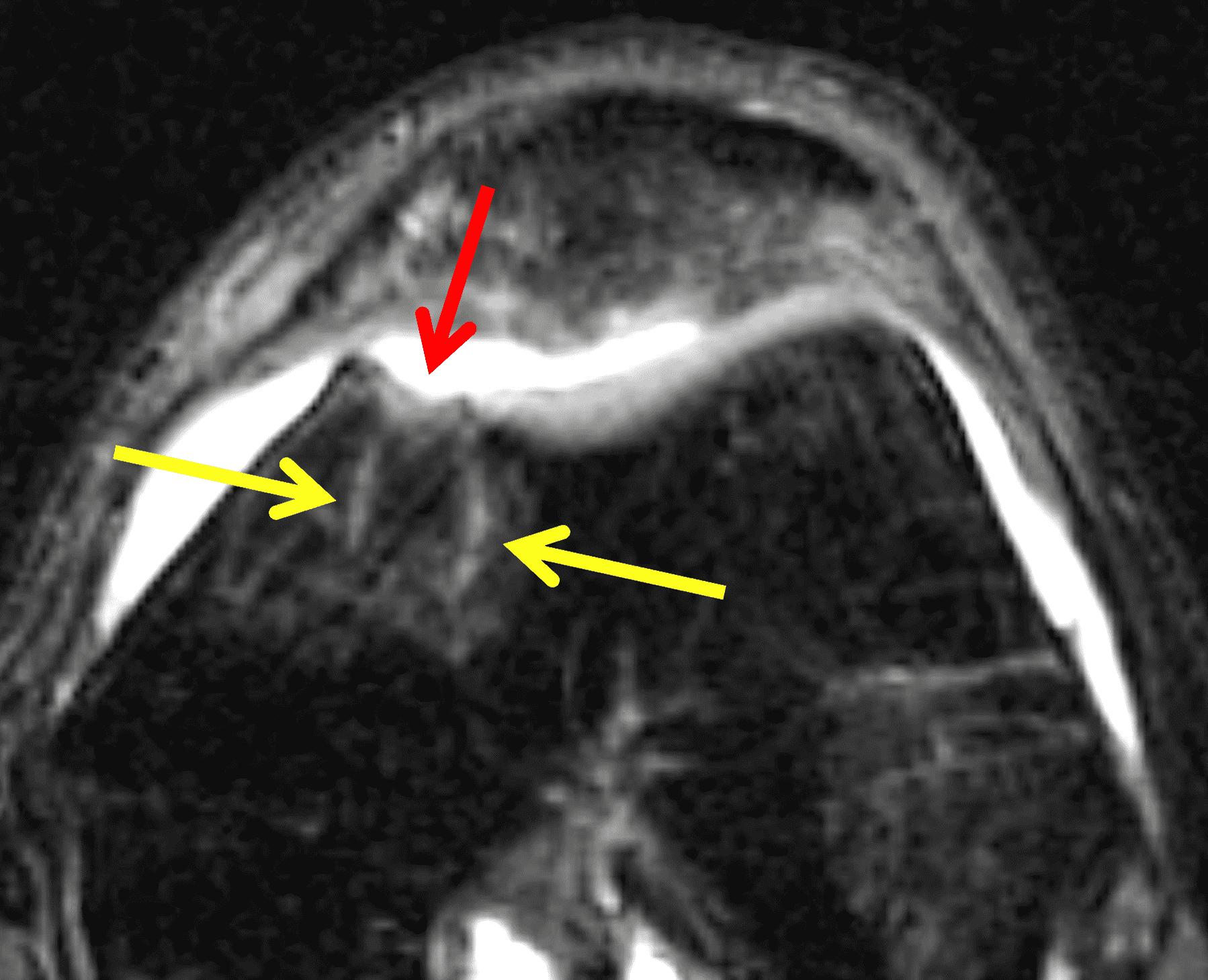
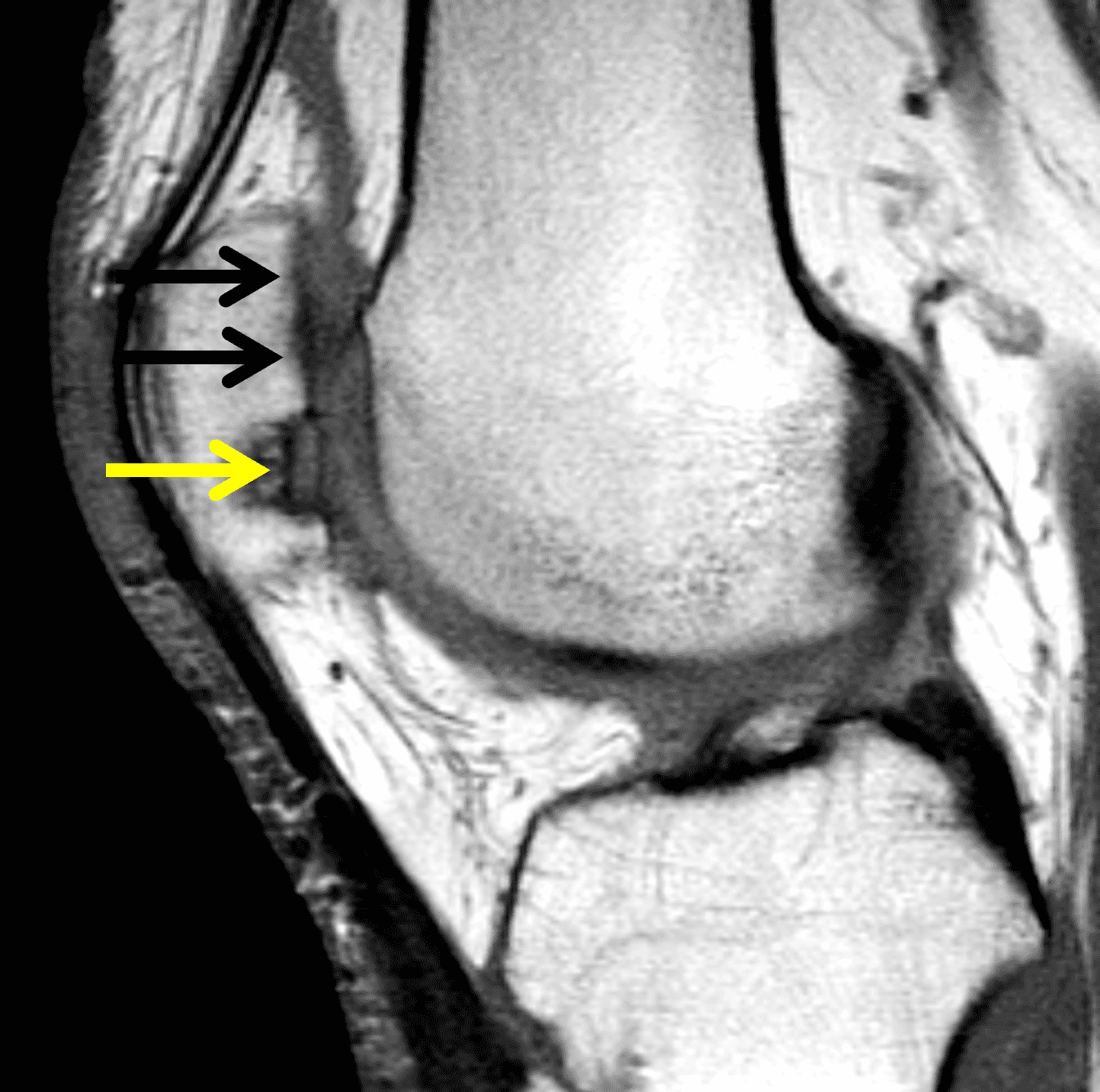
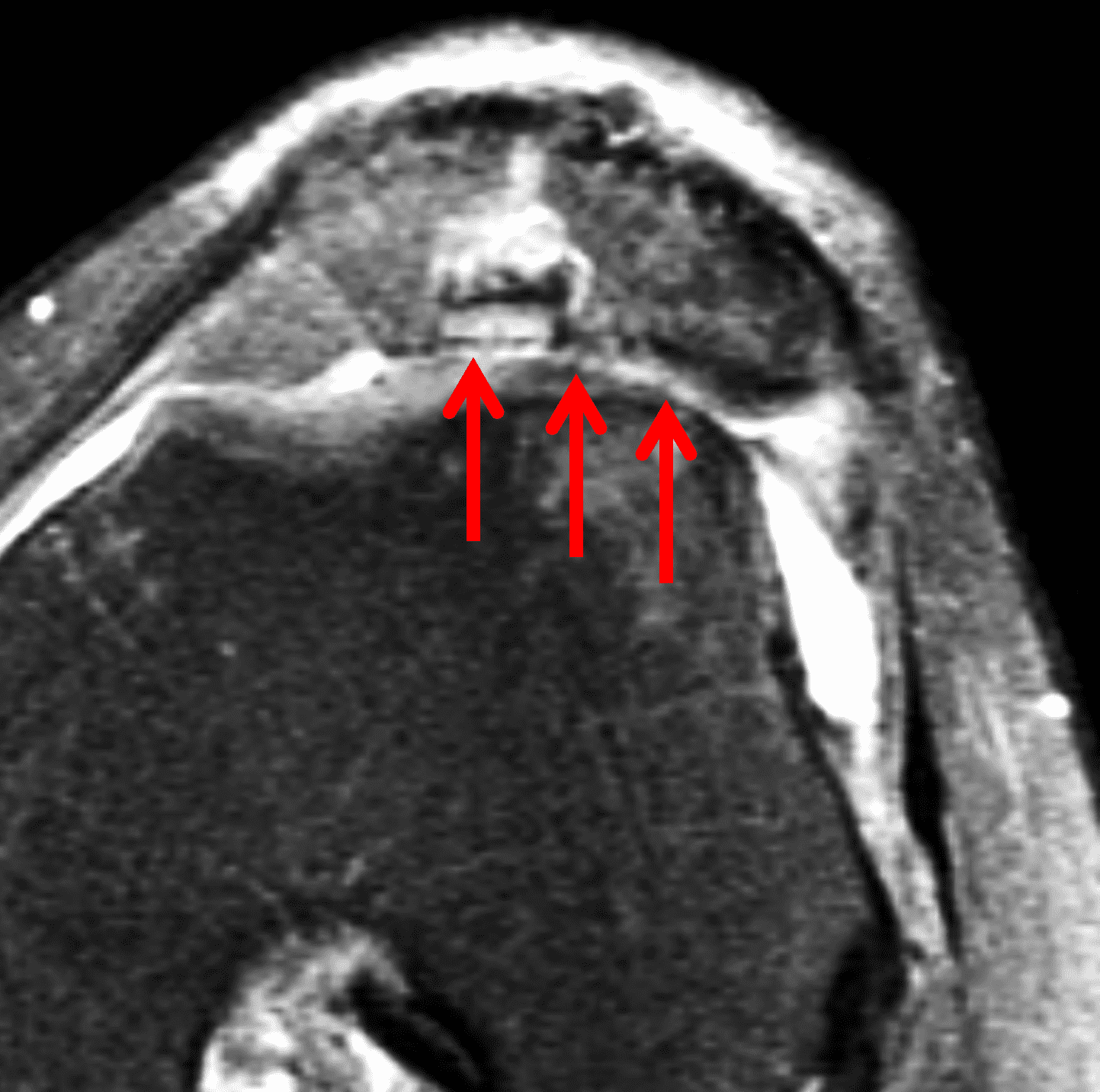
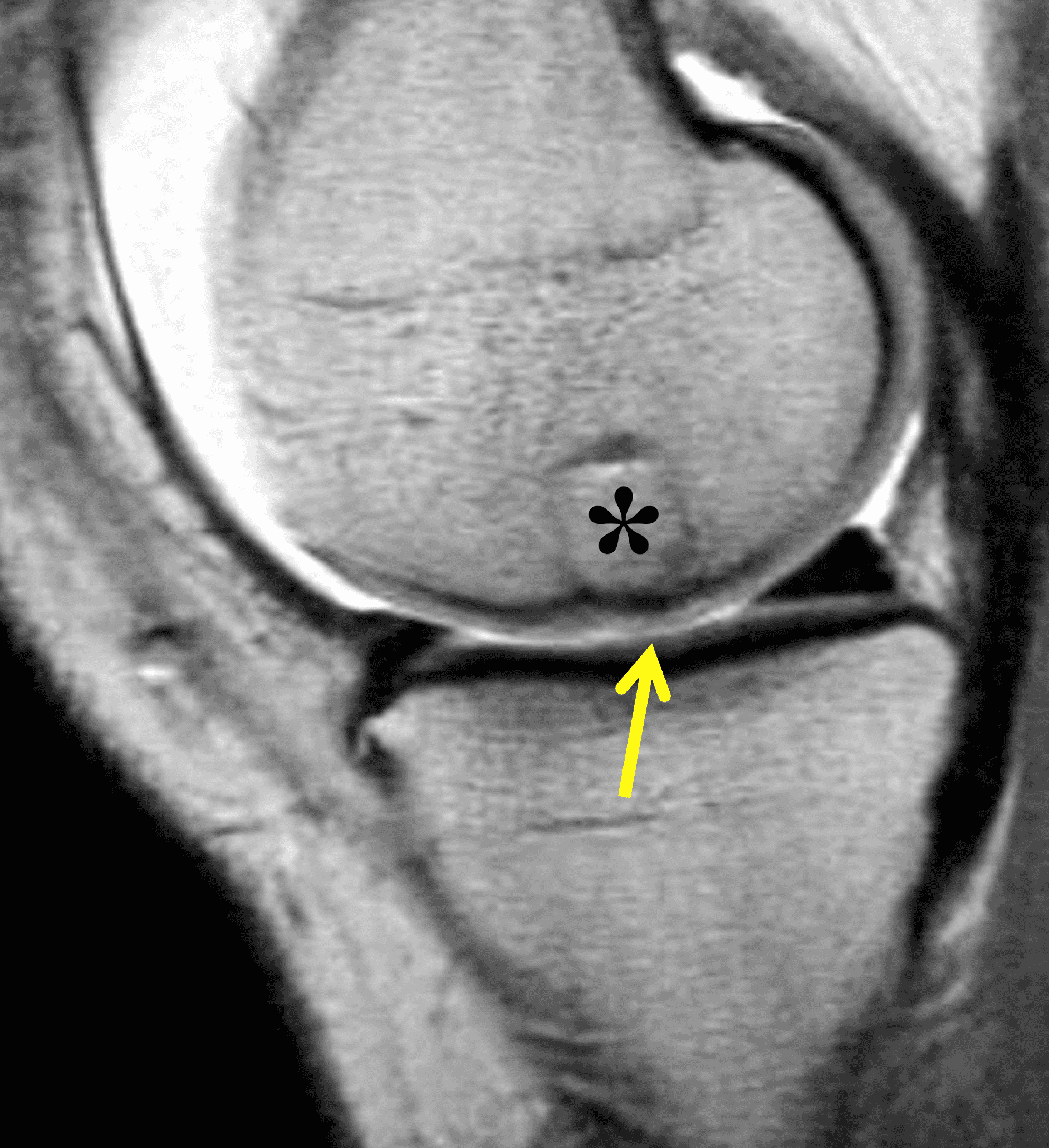
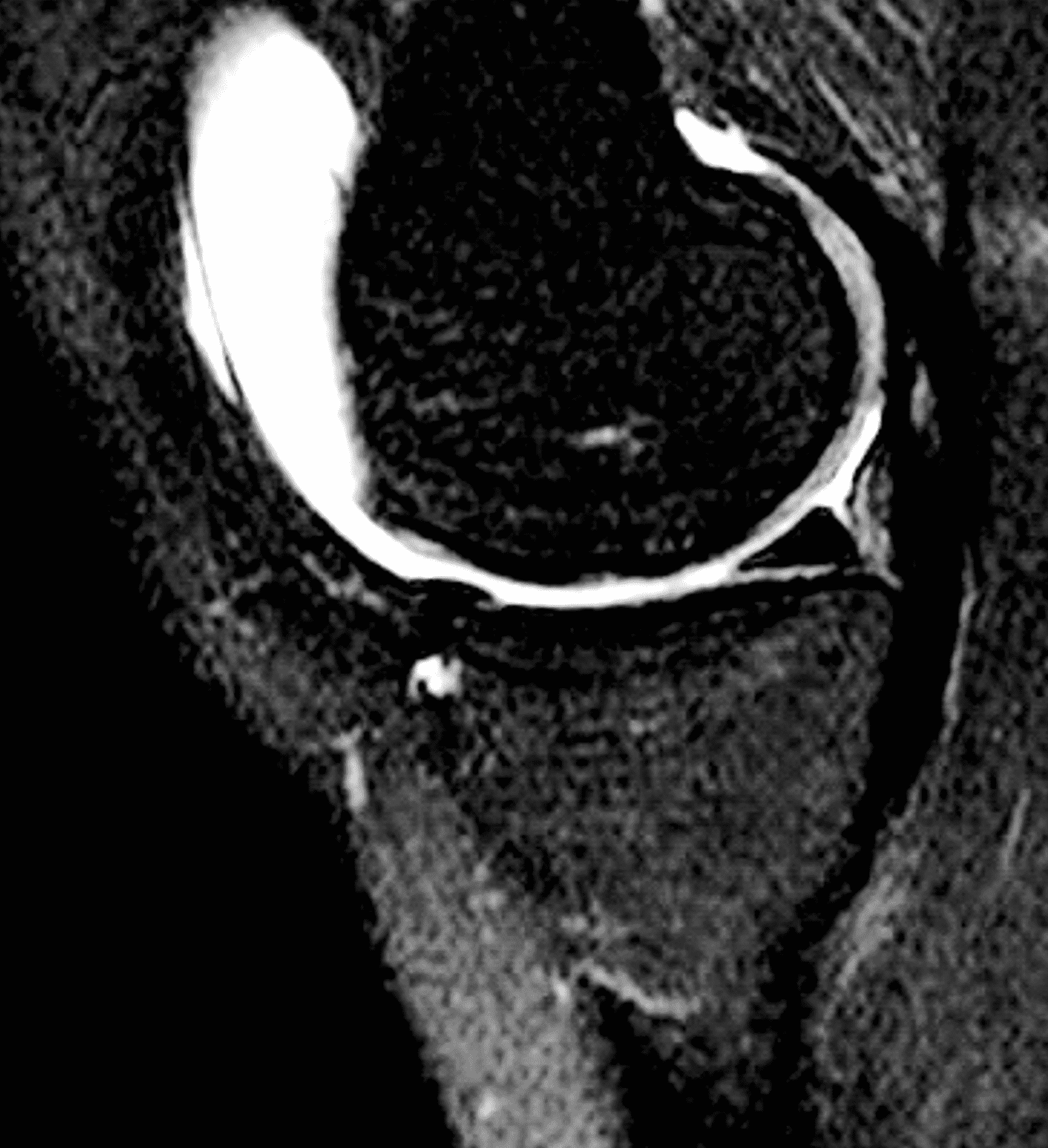
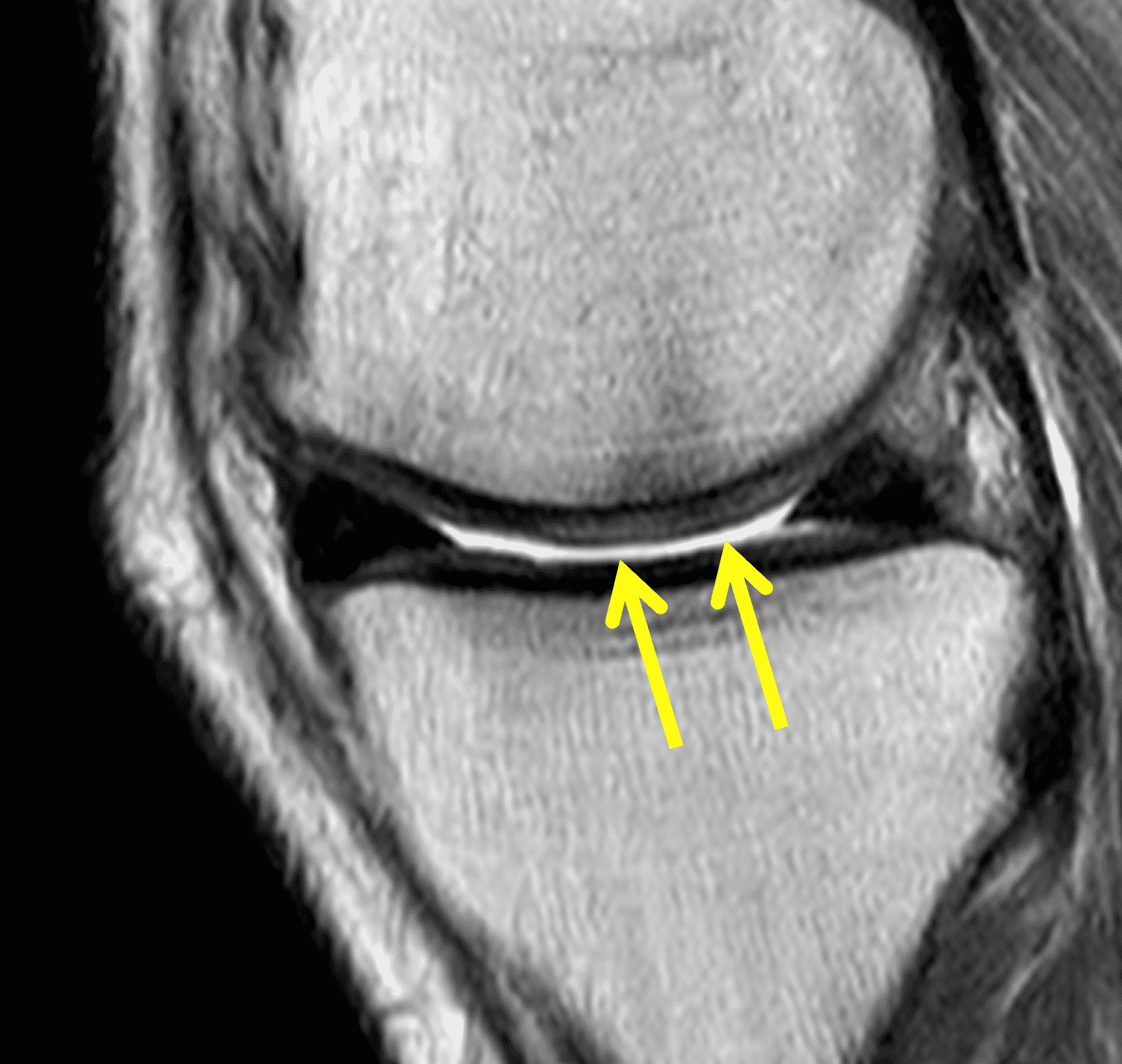
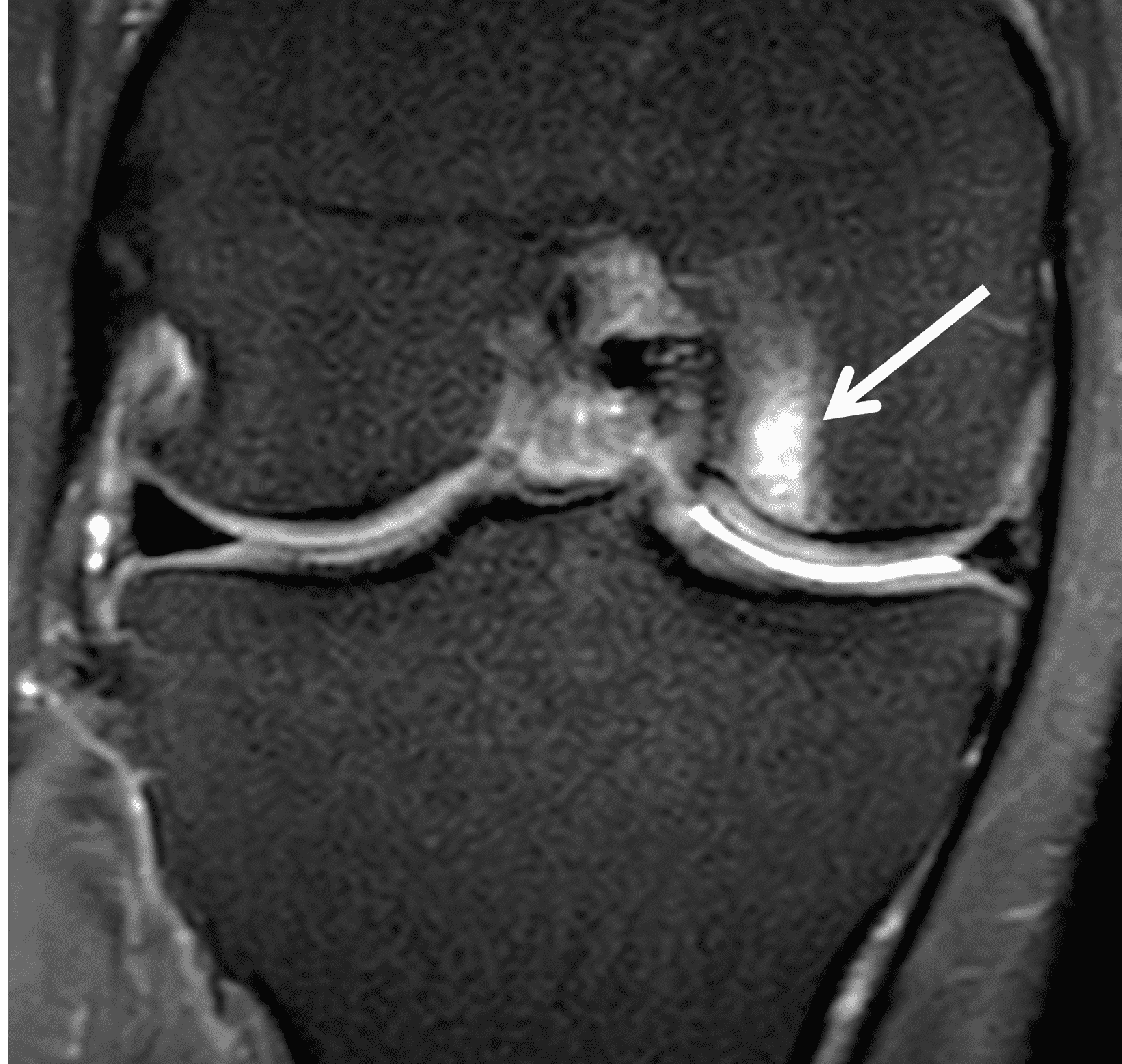
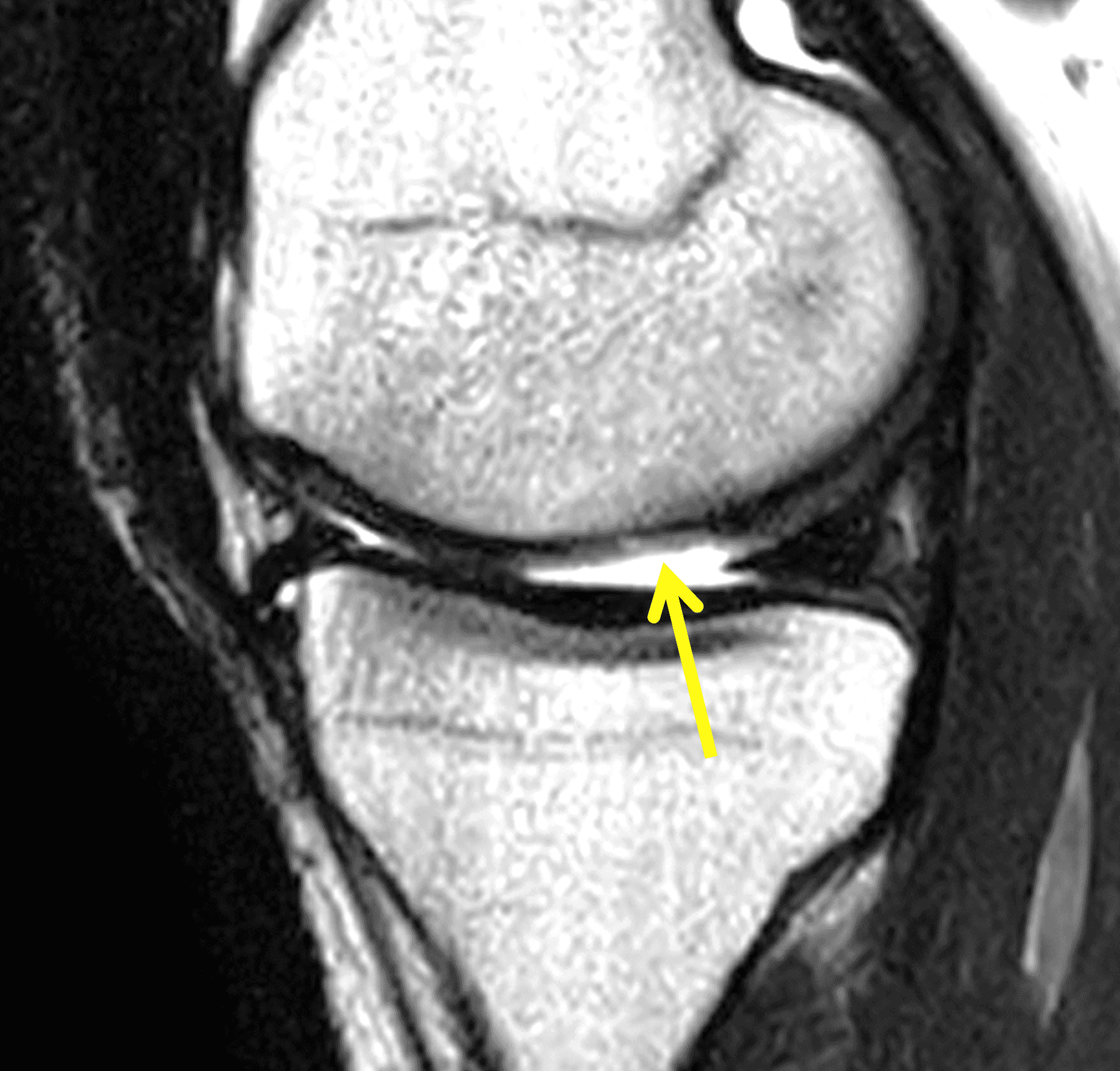
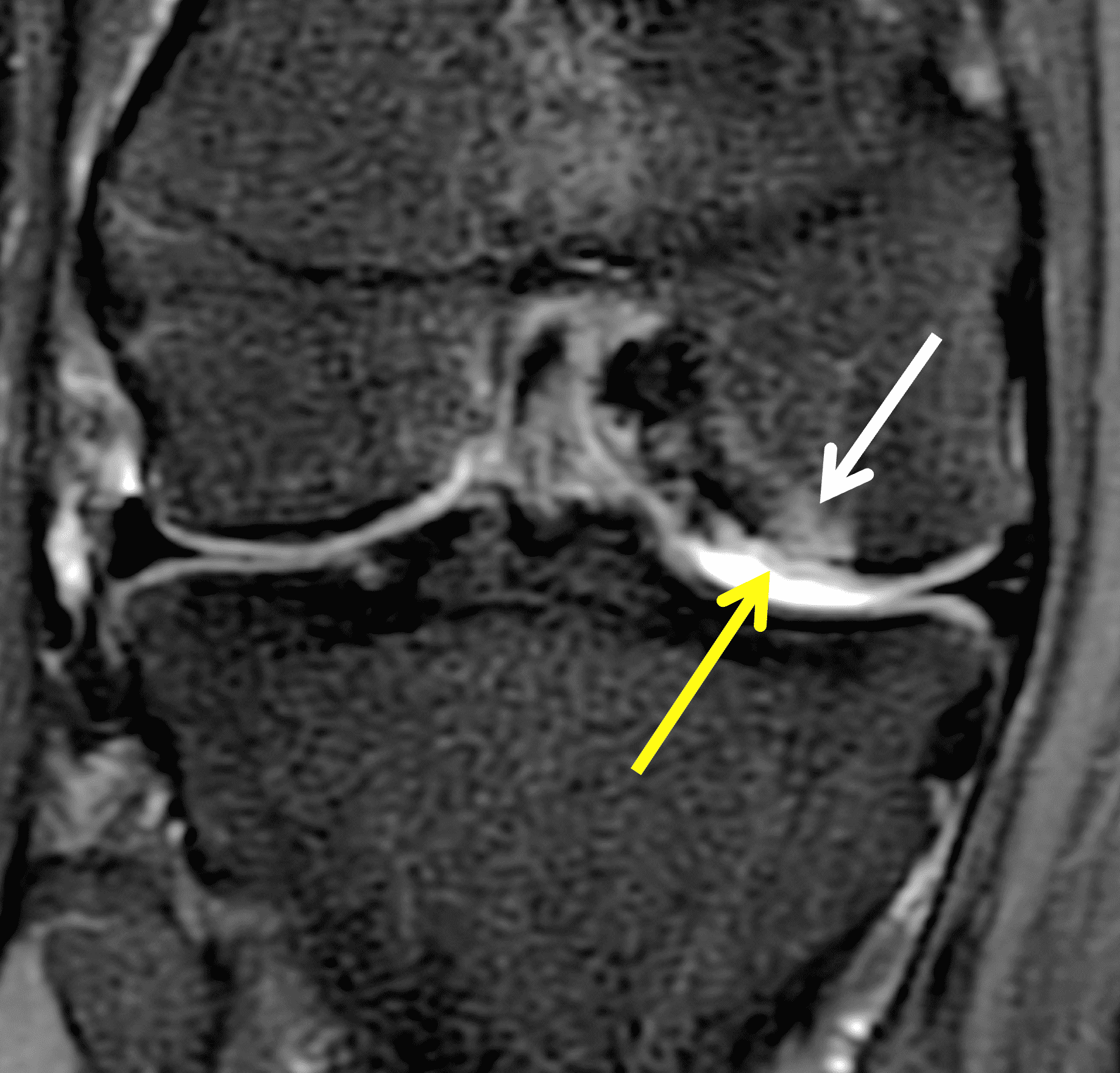
A 32-year-old man suffered a traumatic chondral injury to the medial femoral condyle that was treated with an osteochondral allograft 20 months previously. Representative images from a current knee MRI include (1A) coronal fat-suppressed fluid-sensitive and sagittal (1B) T1-weighted, (1C) proton density-weighted, and (1D) T2-weighted sequences. Are the post-operative MRI findings normal and expected, or abnormal and pathologic? Based on these images, would you characterize the procedure as a success or failure?
Findings
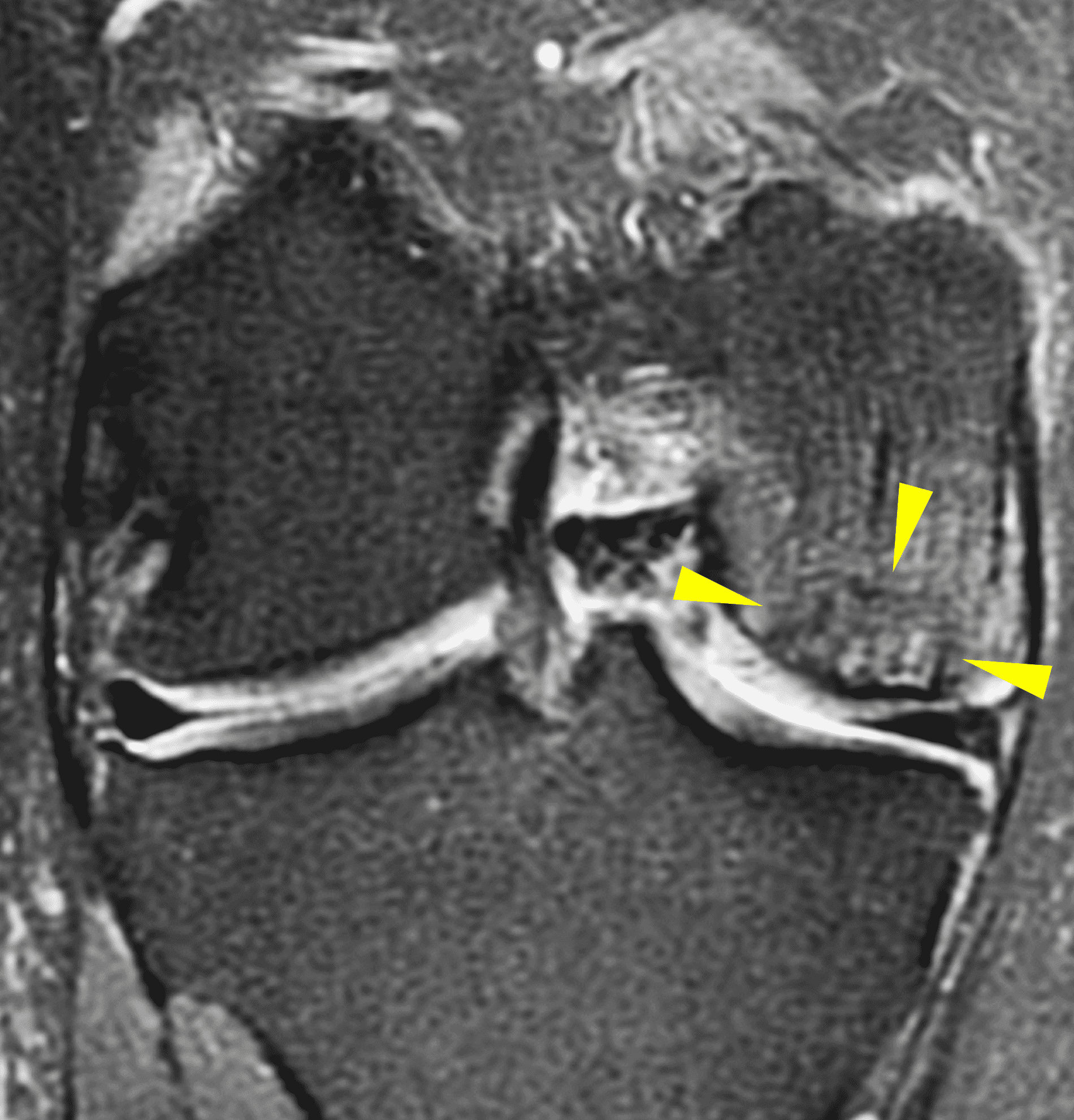
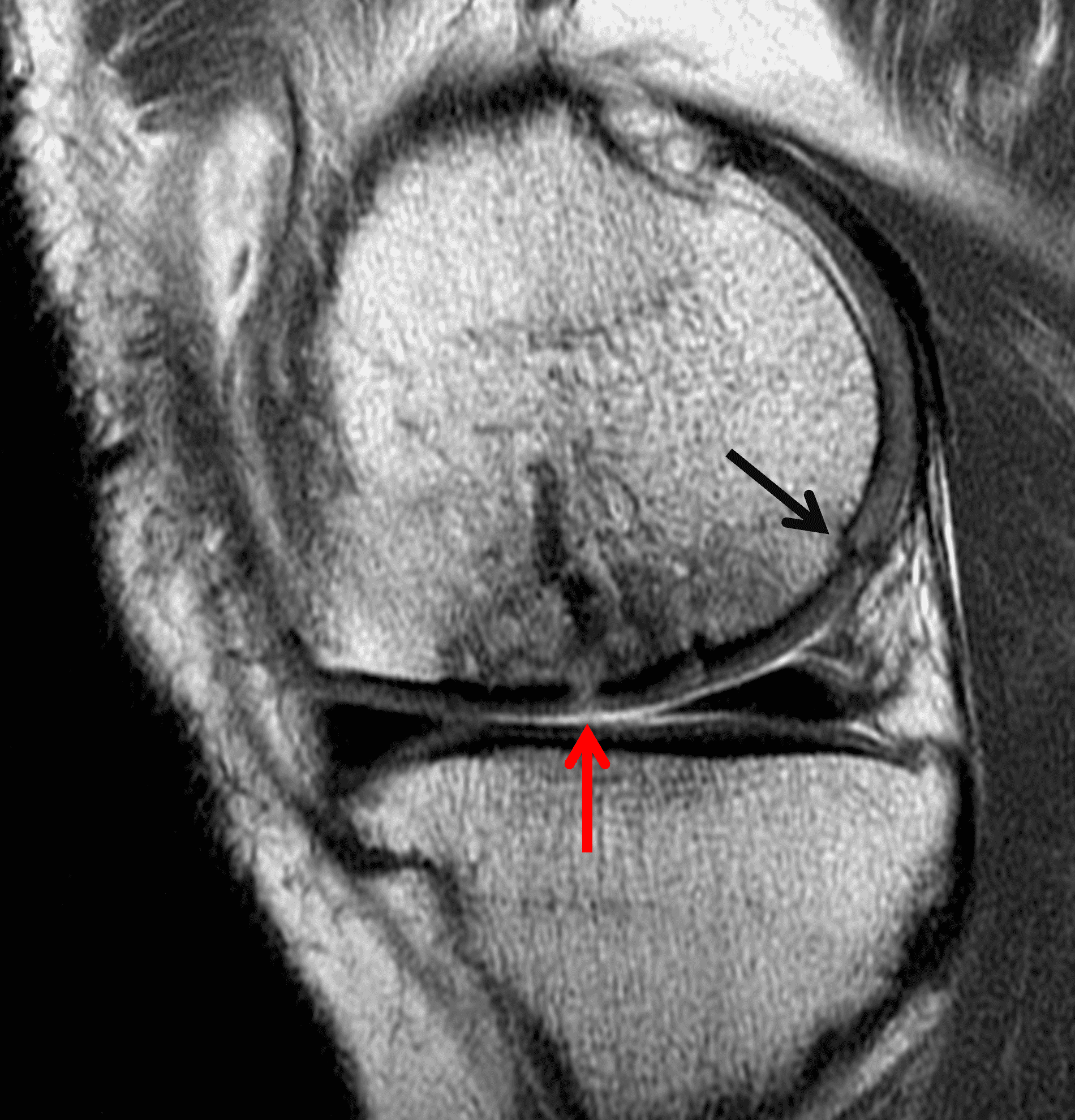
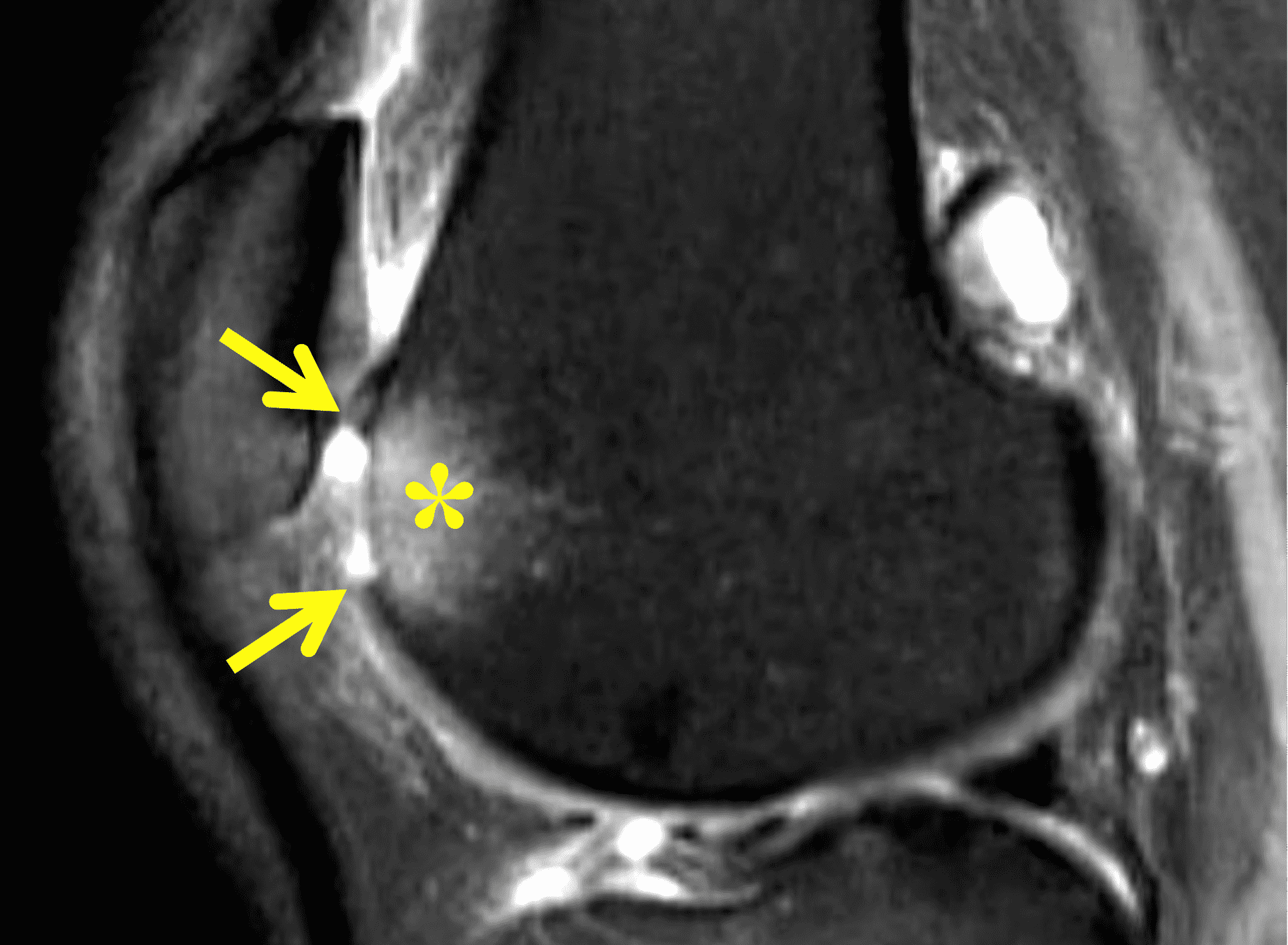
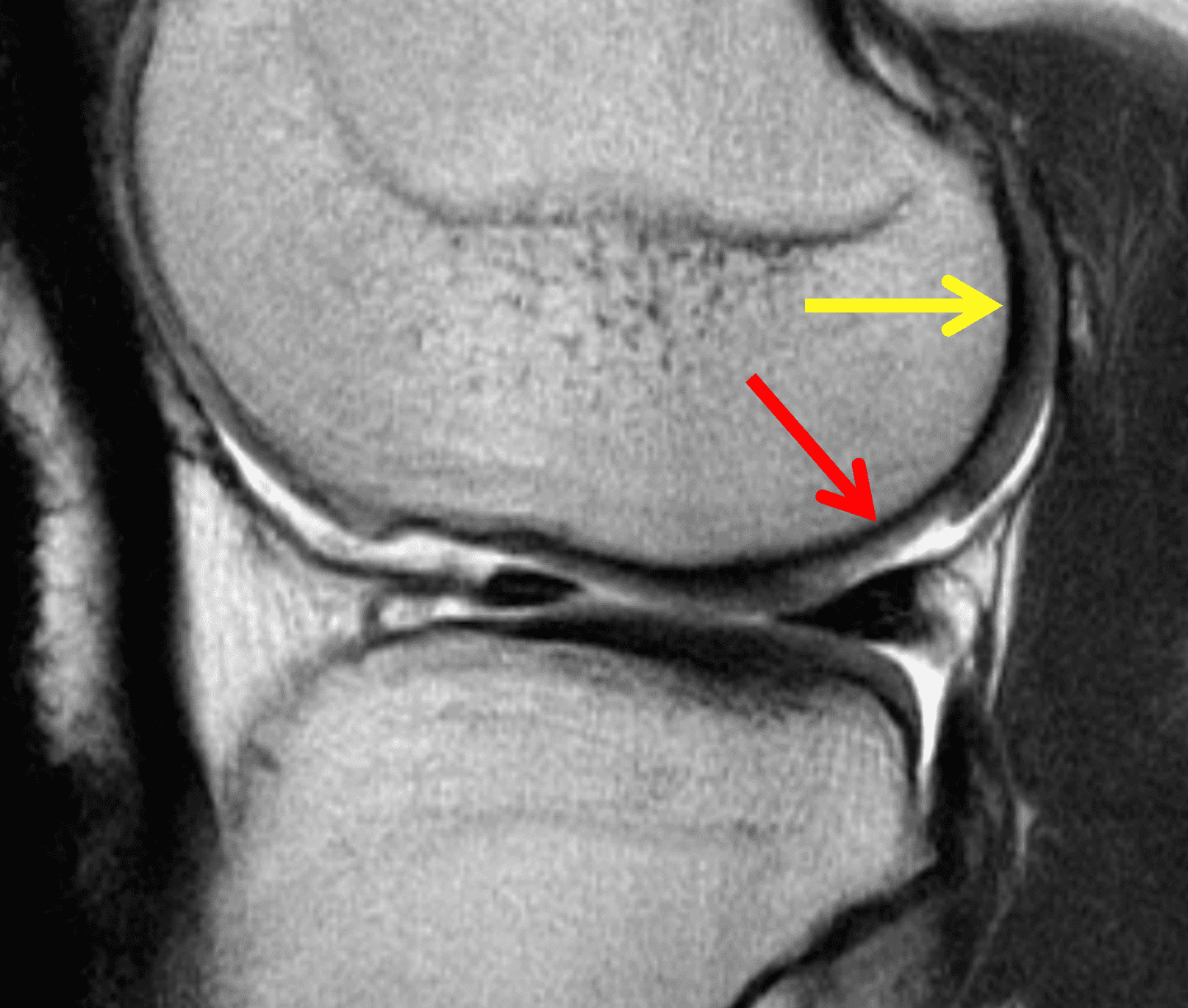
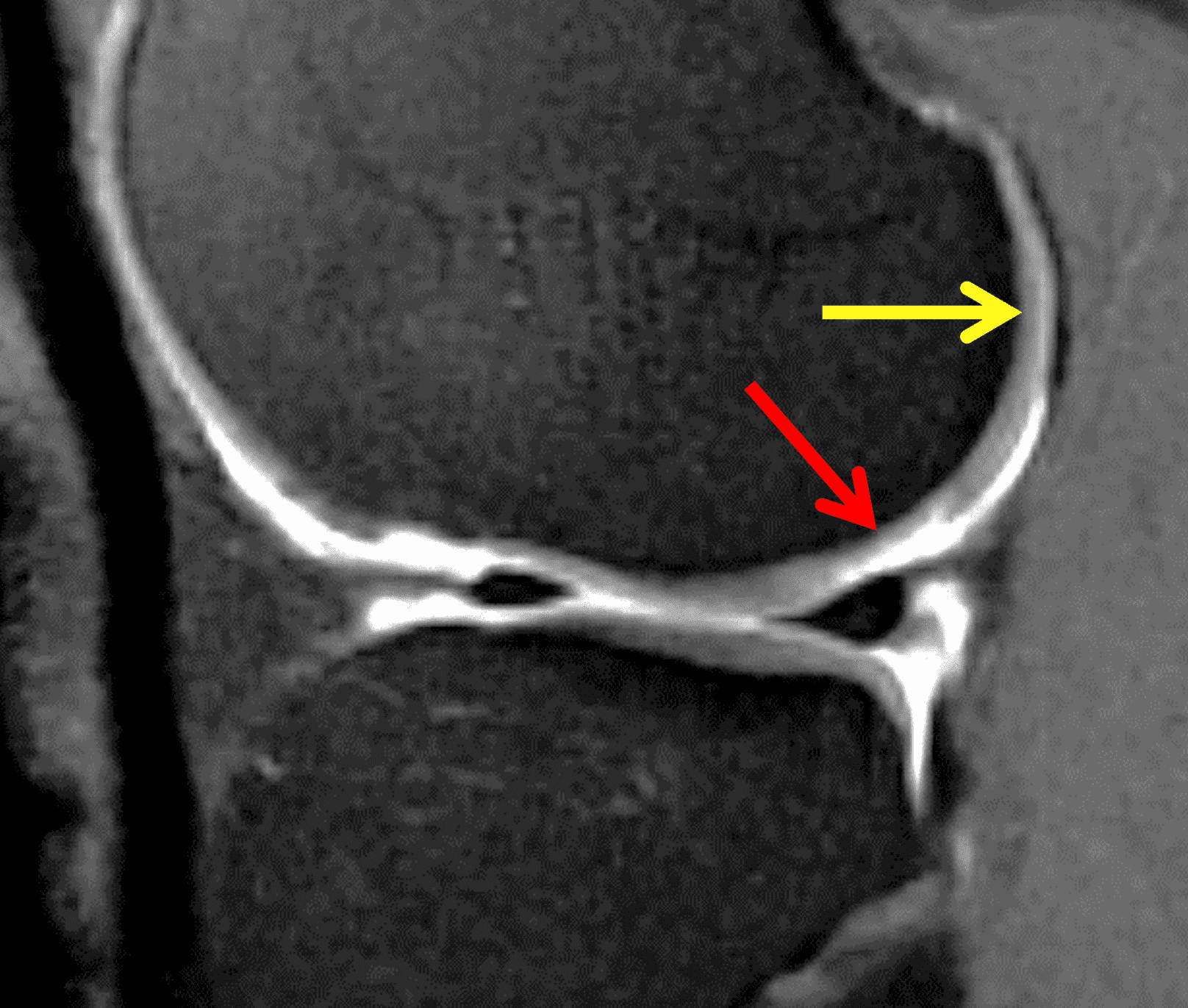
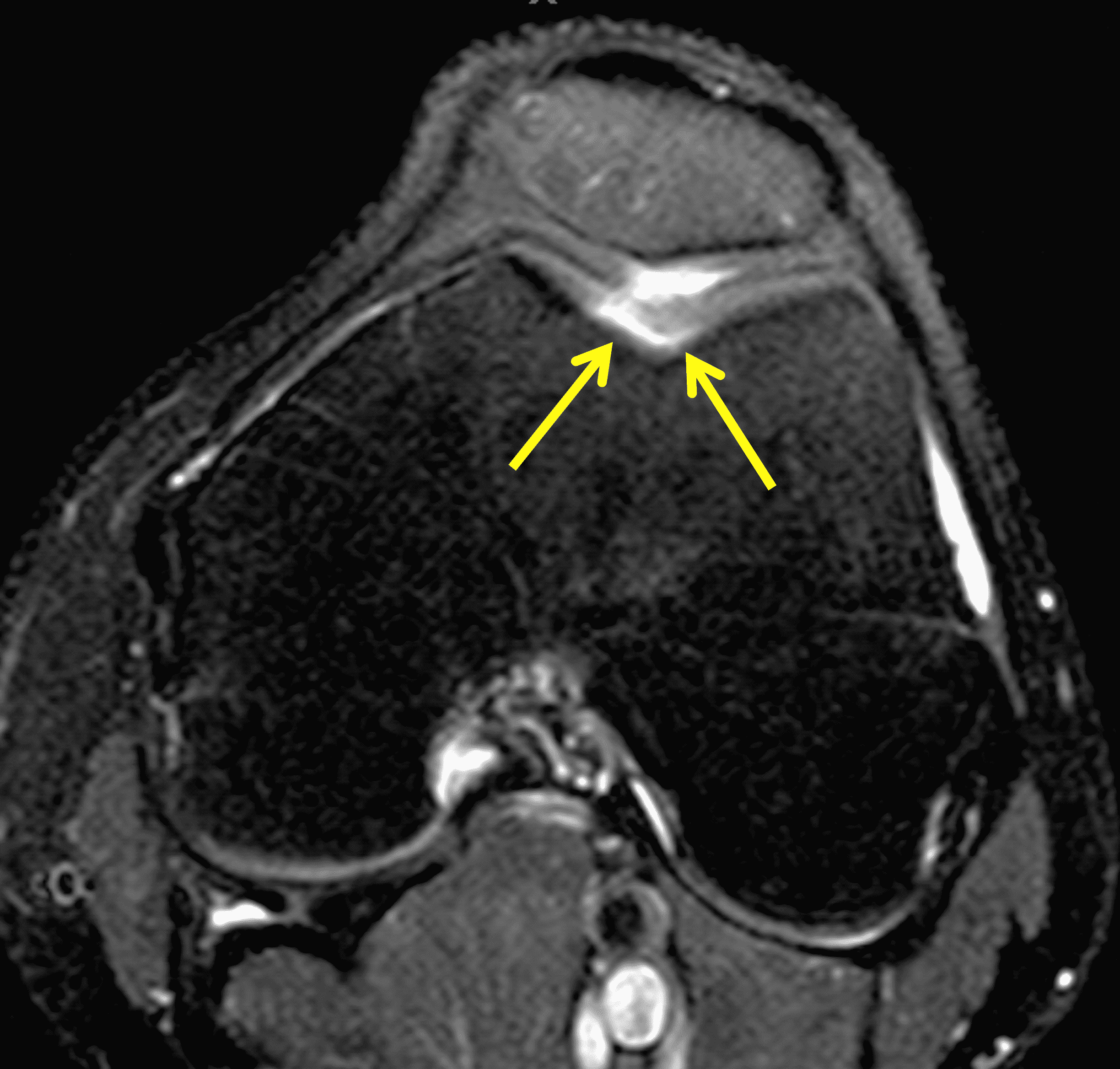
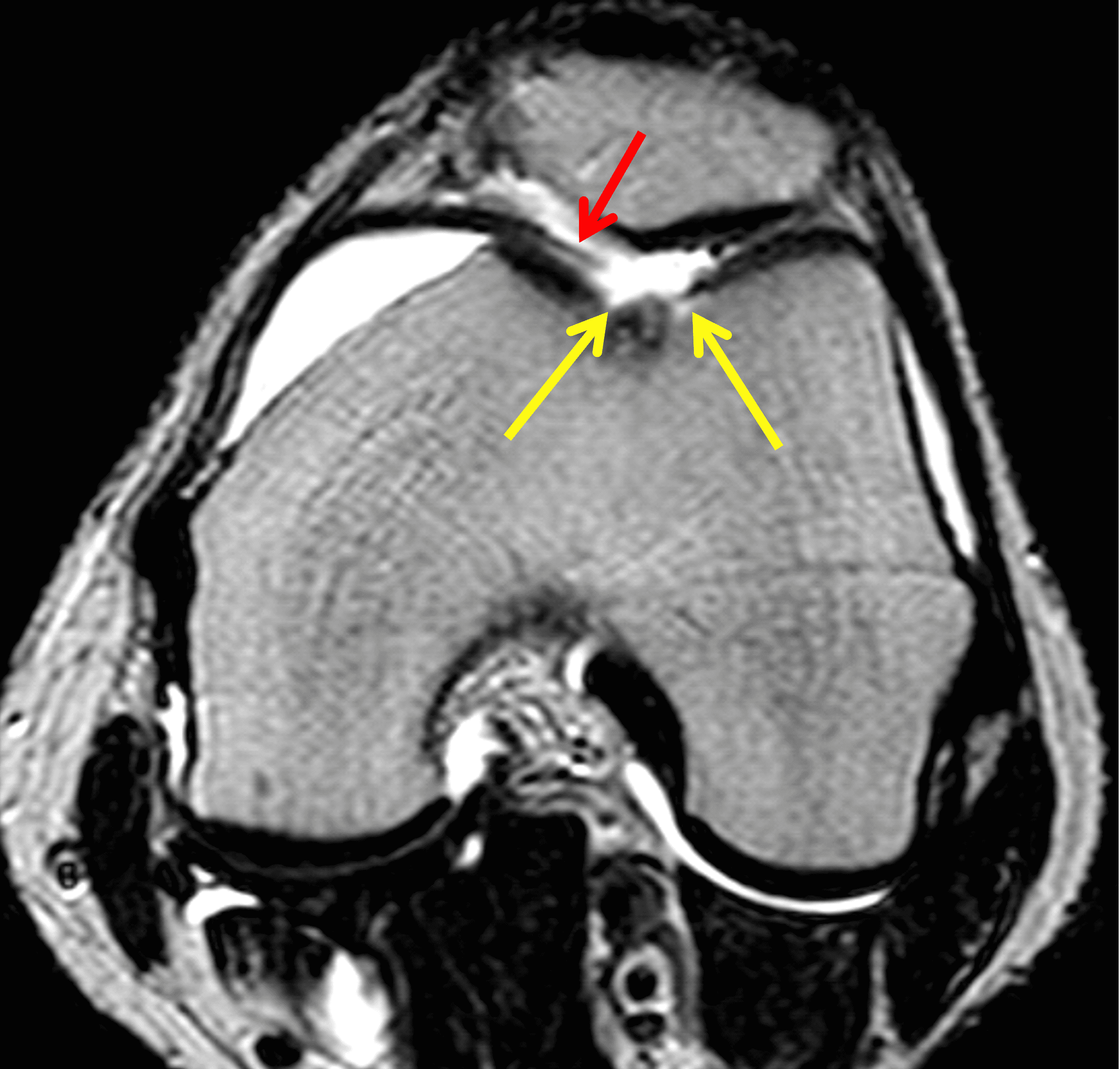
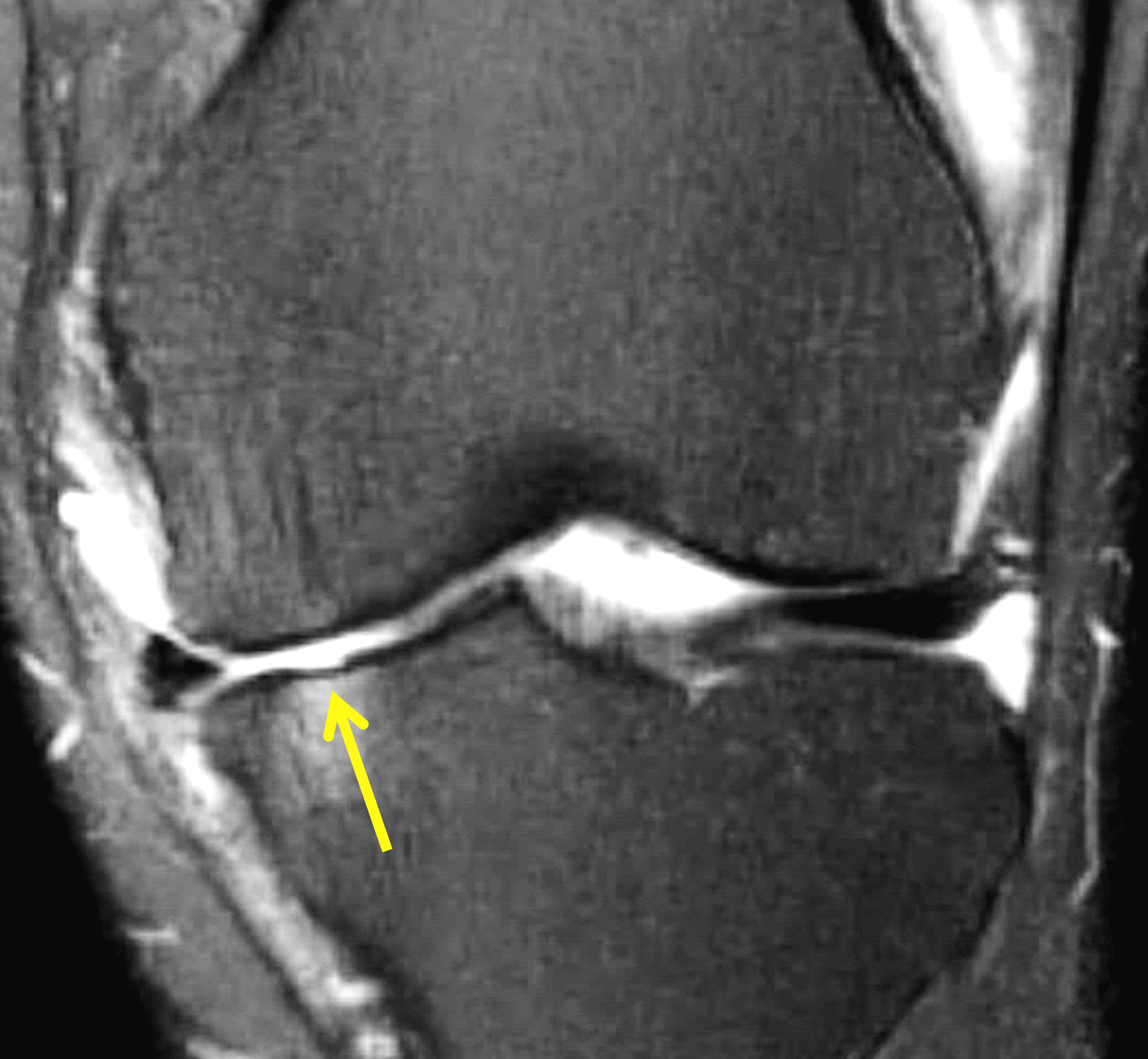
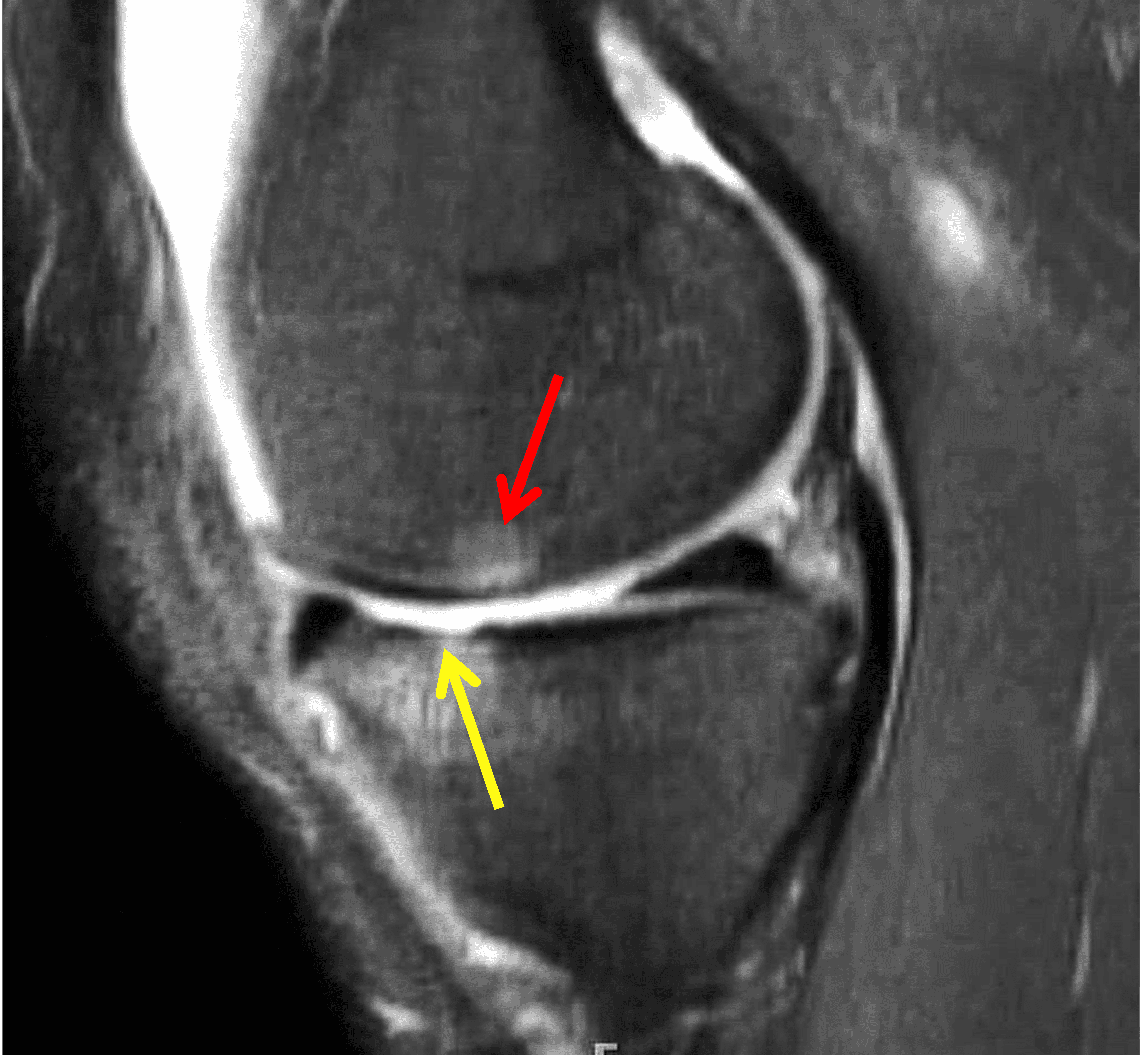
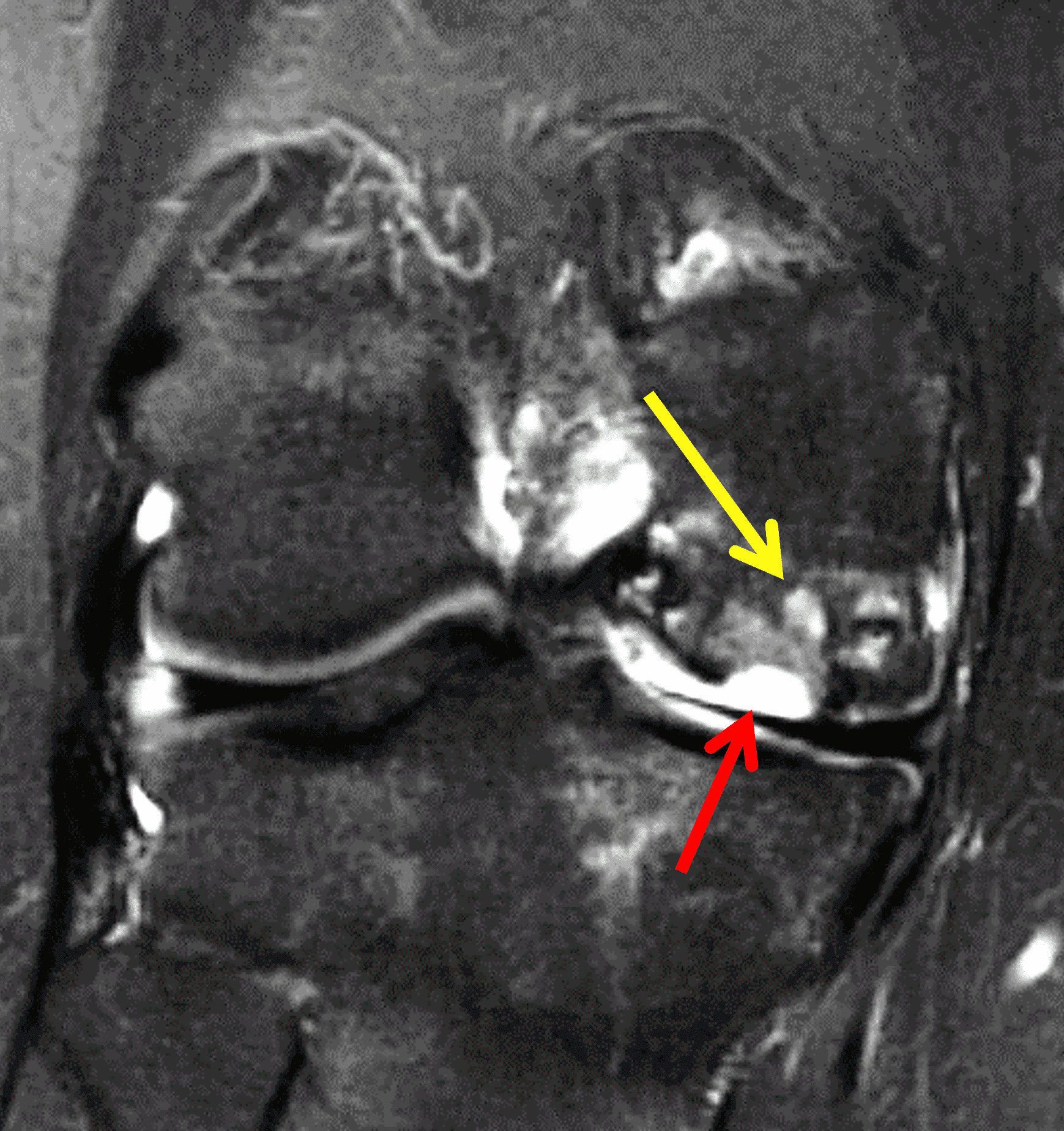
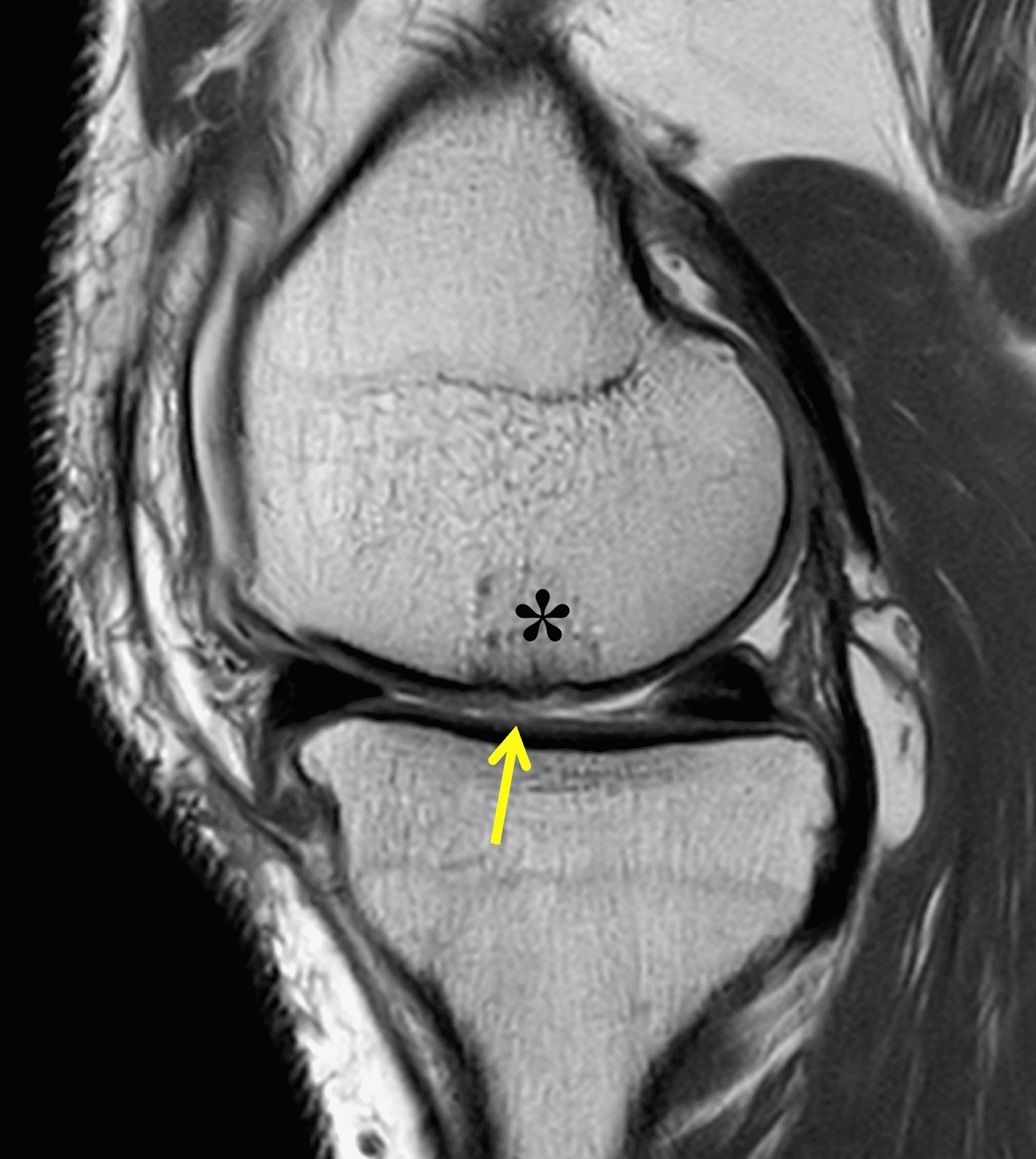
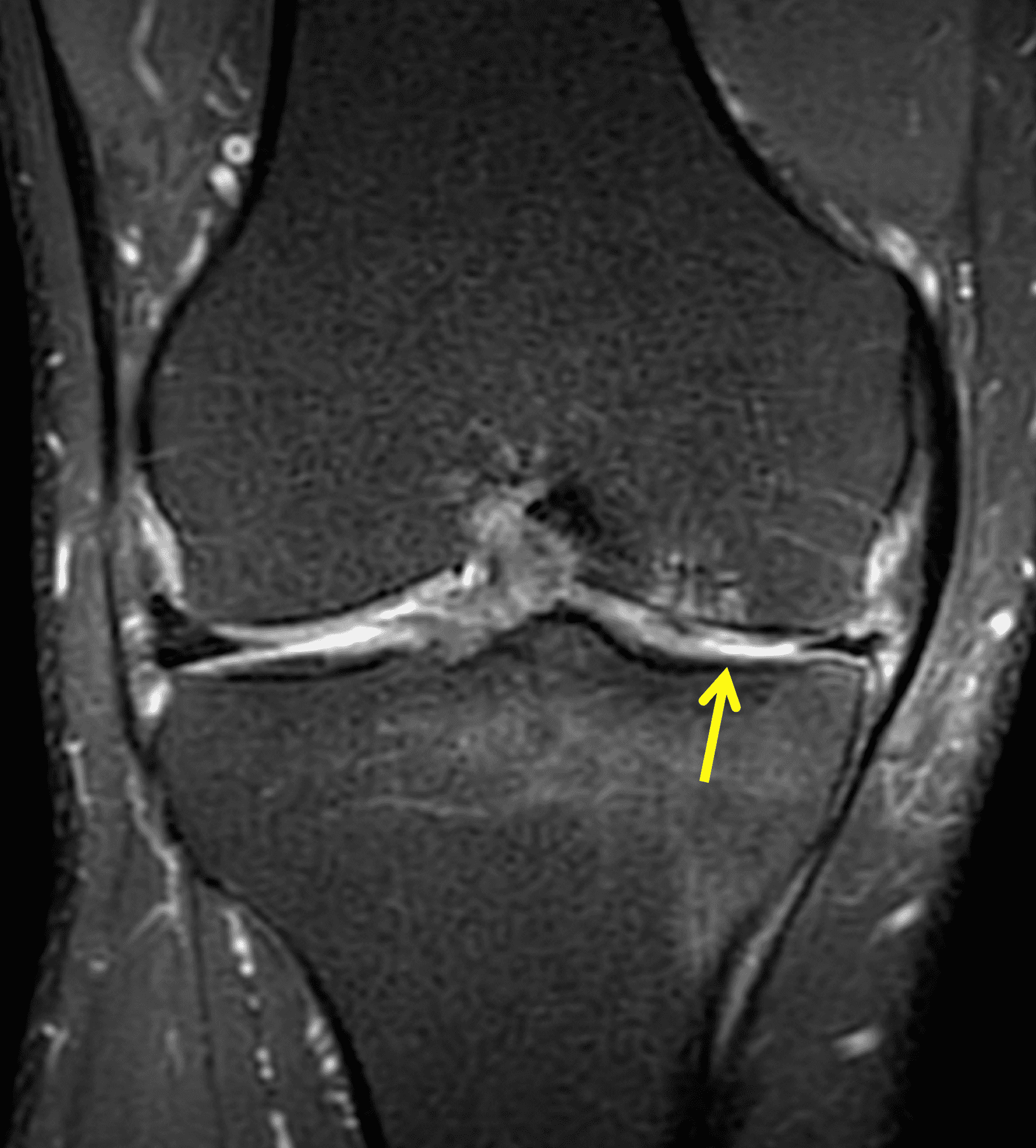
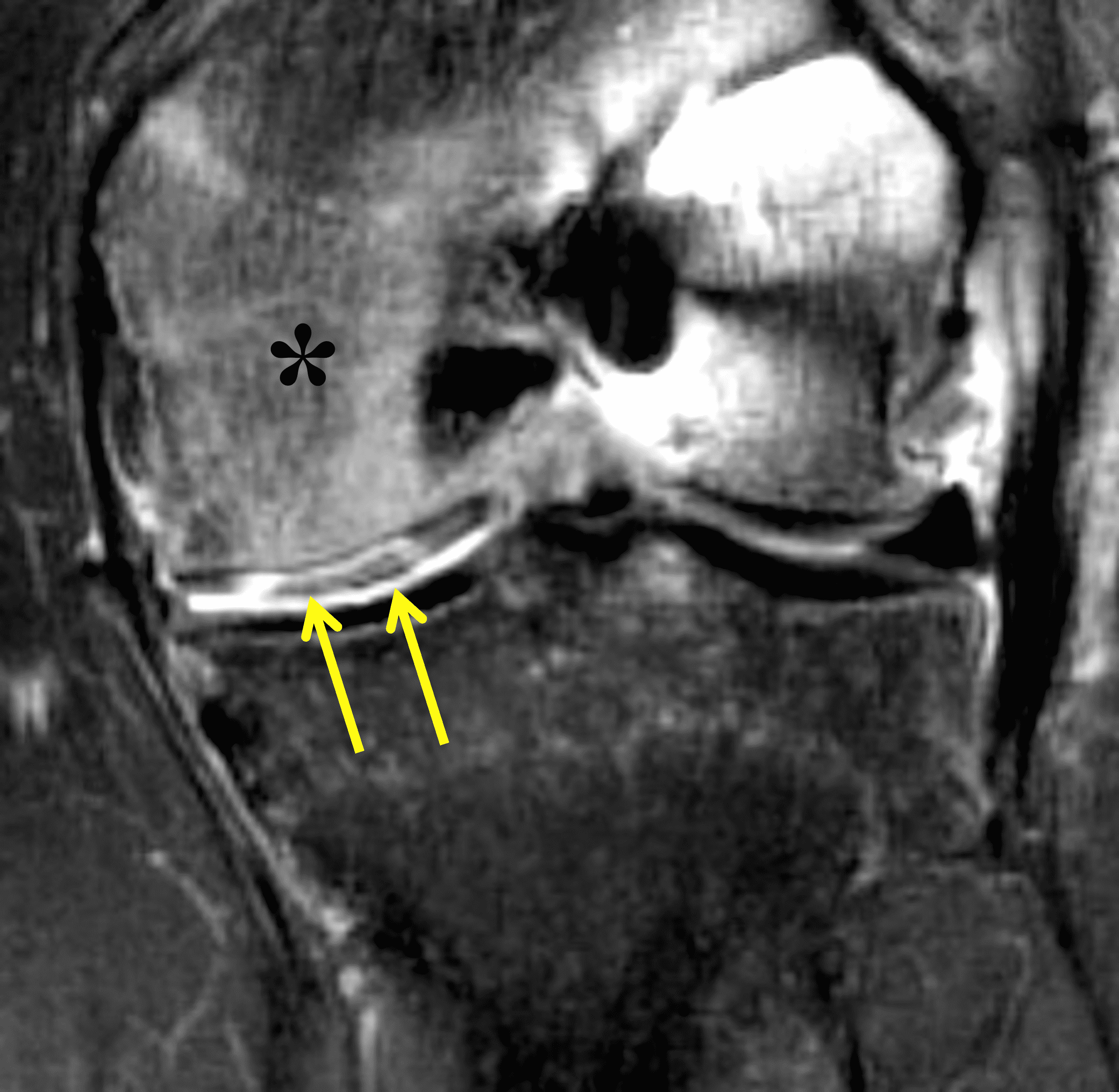
Figure 2: (2A) On the coronal image, the margins of the osteochondral graft are faintly visible (arrowheads). Both the graft and underlying bone show mild marrow edema, an expected finding. (2B) The T1-weighted image shows continuity of the marrow in the graft (asterisks) with the underlying bone, indicating osseous integration. A bioabsorbable pin (arrow) used for graft fixation is partly visible. (2C) The proton density-weighted image shows a low signal intensity seam (black arrow) between the native cartilage and transplant, as well as a defect in the subchondral bone plate of the graft (red arrow) where the pin was drilled, both normal findings. (2D) The T2-weighted image shows a smooth, congruent articular surface (arrows), restoring the normal anatomy, and a tiny subchondral cyst. No findings are present to suggest graft failure. The patient’s current symptoms were attributed to pathology elsewhere in the joint (not shown).
Diagnosis
Successful osteochondral allograft procedure.
Introduction
Articular cartilage injuries are relatively common due to athletic and other trauma, especially in the knee where they may occur as isolated findings or in combination with abnormalities of the menisci, ligaments, and bone.1
In adults, the articular (hyaline) cartilage is separated from the underlying subchondral bone and marrow elements by the subchondral bone plate that includes the deep layer of calcified cartilage and cortical bone with a histologic tidemark between the two.2 This junction normally prevents vascular ingrowth and nutrients from the subchondral bone from reaching the articular cartilage, and also forms a natural cleavage plane along which traumatic chondral injuries can propagate. Additionally, hyaline cartilage is avascular, with no ability to mount an inflammatory response to trauma, nor to heal unaided. Left untreated, articular cartilage injuries can cause pain and mechanical symptoms, and can predispose to premature osteoarthritis.
Orthopedic surgeons have developed several techniques to address symptomatic chondral injuries. Some of these procedures have the potential to restore the damaged articular cartilage. The ultimate goal is to reestablish the articular surface with tissue that has mechanical properties closely mimicking those of native hyaline cartilage (i.e., a low coefficient of friction producing a smooth, gliding surface.)
MRI is the most important noninvasive test to identify and characterize infractions of the articular cartilage. Similarly, following articular cartilage surgery MRI has an important role in evaluating the success or failure of these procedures. This Web Clinic will review the most common surgical procedures designed to address traumatic cartilage lesions along with the expected and abnormal postoperative MRI findings. While the included examples will demonstrate the imaging findings in the knee, similar procedures can be applied to other joints, where the same basic imaging concepts apply.
Types of Treatable Chondral Lesions
Articular cartilage is susceptible to both traumatic and degenerative lesions, but restoration procedures work best for traumatic lesions. Direct impaction, shear, and twisting mechanisms can injure the articular cartilage.3 Traumatic lesions include chondral fractures, which have a characteristic appearance on MRI characterized by a sharp transition between the lesion and surrounding normal cartilage with the walls of the defect oriented perpendicular to the articular surface (Figures 3A and 3B). Defects are often full thickness, extending to the subchondral bone plate. Subchondral marrow edema is common.4 An unstable cartilage fragment may be present in the crater or displaced as a loose body in the joint. Chondral flaps appear similar to chondral fractures but remain attached to the surrounding cartilage along one side of the lesion, which can act as a hinged trap door (Figures 3C and 3D). The two types of traumatic lesion often coexist, with a chondral fracture frequently demonstrating unstable flap(s) in its periphery (Figures 3E and 3F).
Figure 3: Examples of cartilage injuries amenable to restoration procedures. (3A) Drawing and (3B) sagittal fat-suppressed fluid-sensitive image showing a chondral fracture with vertical walls perpendicular to the articular surface (arrows), sharply demarcating the crater from the surrounding normal cartilage, and focal subchondral marrow edema (asterisk). (3C) Drawing and (3D) axial fat-suppressed fluid-sensitive image showing a chondral flap with a vertical wall on one side of the lesion (red arrow) and a intact, hinged bridge of cartilage on the other, with a full-thickness defect in between (yellow arrow). (3E) Sagittal T2-weighted image of a combined chondral fracture and flap. The chondral fracture has sharp margins (yellow arrows) with the surrounding intact cartilage, while a delaminating flap (black arrow) is present along the posterior lesion margin. (3F) Corresponding arthroscopic picture. The subchondral bone plate is exposed at the base of the lesion (asterisk), with a delaminating flap in the lesion periphery (arrow). C = normal surrounding cartilage.
Before skeletal maturity and the formation of a tidemark, a traumatic force can spread through the cartilage and into the subchondral bone resulting in an osteochondral fracture (Figure 4). Osteochondral fractures have the potential to heal spontaneously and can also be treated with internal fixation allowing union of the osseous component to the underlying crater. Unhealed osteochondral fractures can become chronic, unstable osteochondral lesions (osteochondritis dissecans), with formation of a corticated margin at the base of the crater. Specific treatments for both acute and chronic osteochondral lesions exist but will not be addressed in this Web Clinic.
Figure 4: Osteochondral fracture. (4A) Drawing and (4B) sagittal fat-suppressed fluid-sensitive sequence show an acute osteochondral fracture. A fragment composed of both subchondral bone and articular cartilage (yellow arrow) has separated from the underlying bone and is loose within the crater, with associated underlying marrow edema. Note the open growth plate (red arrow) in this adolescent knee.
In contradistinction to traumatic lesions, degenerative chondrosis (osteoarthritis) shows a gradual and wide transition with the surrounding normal cartilage (Figure 5). The walls of the lesion tend to be obliquely oriented rather than vertical. Degenerative cartilage lesions often vary in thickness. Subchondral marrow edema and cyst formation may occur. Cartilage loss on the opposing articular surface is also common, forming “kissing lesions,” which are typically not seen with traumatic defects. In general, cartilage restoration procedures are not indicated for degenerative lesions.
Figure 5: Degenerative arthritis. (5A) Drawing and (5B) sagittal fat-suppressed fluid-sensitive image show a degenerative cartilage lesion (yellow arrows) that varies in depth and has a wide zone of transition with the surrounding cartilage. Note the chondral damage and subchondral edema on the opposing tibial plateau surface (asterisk).
MRI Techniques
Fast (turbo) spin echo sequences with a long TR and intermediate-to-long TE are the mainstay for clinical imaging of both native cartilage and postoperative patients.5 Imaging can be accomplished with or without fat suppression. Fluid is high-signal intensity on these sequences. Normal hyaline articular cartilage appears hypointense compared to joint fluid. The deepest layers of the articular cartilage together with the subchondral bone plate show very low signal intensity with the more superficial layers appearing brighter (Figure 6). The thickness of the very low signal-intensity layer varies with the echo time; the longer the TE, the thicker the deep layer will appear. Direct MR arthrography using T1-weighted sequences following injection of a dilute gadolinium-containing solution into the joint is an alternative approach to imaging articular cartilage.
Figure 6. Clinically useful MRI pulse sequences to evaluate native and treated articular cartilage. Sagittal (6A) T2-weighted fast (turbo) spin echo, (6B) proton density-weighted fast (turbo) spin echo, and (6C) fat-suppressed T1-weighted spin echo post-arthrogram images show the normal layered appearance of articular cartilage (yellow arrows). Note that the deep, low signal intensity band appears thickest on the sequence with the longest TE (6A) and thinnest on the sequence with the shortest TE (6C). This patient underwent prior microfracture of a lateral femoral condyle chondral defect that shows reparative cartilage filling approximately 50% of the depth of the crater (red arrows).
Researchers have developed and investigated multiple specialized MRI sequences to better assess the ultrastructure of hyaline cartilage including the proteoglycans, collagen matrix, and fluid content. Techniques include T2 mapping, sodium MRI, ultrashort TE sequences, diffusion weighted imaging, and dGEMRIC (delayed gadolinium-enhanced MRI of cartilage) approaches. While these pulse sequences can distinguish native from repaired cartilage, their ability to distinguish between repaired cartilage that is health or diseased is currently limited.6 More research is needed before any of these techniques can be recommended for routine clinical assessment.
Similarly, research investigations often apply semi-quantitative scoring systems such as the MR Observation of Cartilage Repair Tissue (MOCART) when assessing postoperative outcomes.7,8 These tools assign scores for anywhere from 7-11 separate variables to each articular surface. While the scoring systems are too cumbersome for routine clinical use, several of the assessed variables are useful to note when interpreting post-operative MRI. The most relevant include the amount of repair tissue filling the original defect, T2 signal of the repair tissue, interface between the base of the lesion and repair tissue, congruence of the articular surface, and changes in the underlying subchondral bone marrow.
Nonrestorative Procedures
While it is sometimes possible to affix a fractured cartilage fragment within its crater using chondral darts or similar fixation, attempts at primary repair of purely cartilaginous lesions risk failure because articular cartilage is avascular and may not heal (Figure 7).9 Chondral defects can be debrided to create smooth edges in a procedure known as chondroplasty. Following debridement, the defect may remain unchanged or partly heal with reparative fibrocartilage (Figure 8). Using continuous passive motion postoperatively may encourage mesenchymal cells to differentiate into cartilage.10 However, compared to hyaline cartilage, fibrocartilage has higher levels of type 1 collagen and less supportive extracellular matrix, factors that make it a less efficient articular surface and lead to future degeneration. In the past, cartilage debridement was employed as a “washout” or “cleanup” procedure in some patients with degenerative arthritis. But controlled studies have shown no advantage of these procedures compared to placebo, lavage, or physical therapy after 1-2 years in knees with osteoarthritis.11,12 For symptomatic, traumatic chondral injuries, chondroplasty can be employed for lesions of any size on any articular surface, and has the advantage of relatively quick recovery compared to restorative procedures. Thus, chondroplasty is often used for elite athletes, especially during their sport’s season. In a study of the National Football League, 67% of athletes were able to return to play following chondroplasty.13 Debridement is also commonly used for traumatic lesions that are found incidentally during arthroscopy that is being performed for meniscus or ligament abnormalities.
Figure 7: Failed attempted chondral repair. (7A) Preoperative axial fat-suppressed fluid-sensitive image shows a full-thickness chondral flap of the central trochlear groove (arrows). (7B) Post-operative sagittal T2-weighted image shows low signal intensity bioabsorbable pins (black arrows) used to transfix the cartilage flap, one of which is fractured (red arrow). (7C) Post-operative axial T2-weighted image shows that the repaired cartilage has separated from the subchondral plate and formed a loose body (red arrow), leaving an empty crater (yellow arrows).
Figure 8: Chondroplasty. Fat-suppressed fluid-sensitive (8A) coronal and (8B) sagittal images approximately 2 years after a medial tibial plateau cartilage defect was treated with arthroscopic chondroplasty. No reparative cartilage is present in the debrided crater (yellow arrows), with underlying subchondral marrow edema. New cartilage loss and marrow edema have developed on the facing medial femoral condyle (red arrow). The patient’s medial knee pain had recurred.
Bone Marrow Stimulation Procedures
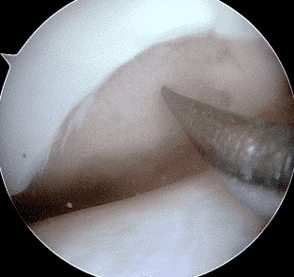
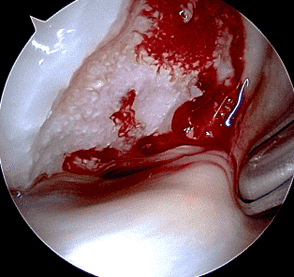
Microfracture procedures that create holes in the subchondral bone plate are commonly used to stimulate fibrocartilaginous repair of chondral defects, typically for lesions less than 4 cm2 in cross-section, in low-demand patients less than 40 years old.14 During arthroscopy, the surgeon uses a drill, pick, or awl to create multiple holes through the base of the cartilage lesion into the subchondral bone.15 The tourniquet is then temporarily released to allow marrow elements including blood and stem cells to fill the crater (Figure 9). Autologous stem cells or platelet rich plasma may be used as adjuncts within the defect. Postoperatively, continuous passive motion enhances metaplasia and remodeling of the subsequent fibrous clot to form reparative fibrocartilage. The quality of the reparative cartilage is variable, often leading to secondary degeneration and breakdown within approximately 5 years.16,17 Reoperation rates are reported to be approximately 25-40%.18,19 Microfracture has the highest success rates for lesions located on the femur and lesions less than 2 cm2.20
Figure 9: Arthroscopic photographs made during microfracture procedure. (9A) The surgeon has debrided the chondral defect and a pick is about to be used to penetrate through the exposed subchondral bone plate. (9B) After several holes are created, the tourniquet is released, allowing marrow elements and blood to fill the crater.
On MRI in the first 6 months following microfracture, the reparative tissue may appear thin and hyperintense compared to hyaline cartilage. In successful procedures, after approximately 2 years the repair tissue will fill the majority of the cartilage defect and become isointense or mildly hypointense compared to the surrounding normal cartilage on fluid-sensitive MR sequences (Figure 10). The degree of defect filling, measured either by volume or depth (Figure 6), is the imaging finding most highly associated with clinical success.8 Subchondral marrow edema should also resolve in the first 1-2 years following microfracture, although decrease in marrow edema, normalization of T2 signal, and the presence of effusion or synovitis weakly correlate with clinical outcomes.
Figure 10: Successful microfracture procedure. (10A) Pre-operative sagittal proton density-weighted image shows a full-thickness chondral fracture in the medial femoral condyle (arrow). (10B) Sagittal proton density-weighted image 3 years after microfracture demonstrates that 100% of the crater is filled with reparative fibrocartilage (yellow arrow), which lacks the normal laminar appearance of the adjacent native cartilage (red arrow). Mild residual subchondral bone changes are present deep to the treated lesion (black arrow).
The main MRI findings indicating failure of microfracture include incomplete filling of the articular cartilage defect, increased subchondral marrow edema, and development of subchondral cysts.13,21 Sometimes overgrowth of the underlying subchondral bone will create an osteophyte within the base of the crater (Figure 11). Subchondral bone overgrowth increases the risk of microfracture failure by 10-fold.22
Figure 11: Failed microfracture procedure. (11A) Sagittal T2-weighted image approximately one year after a chondral defect of the lateral femoral condyle was managed by microfracture shows osteochondral overgrowth forming an intra-cartilaginous osteophyte in the base of the crater (yellow arrow). The result is a stress riser that has led to a new chondral flap along the posterior margin of the treated lesion (red arrows). (11B) Axial fat-suppressed fluid-sensitive image confirms the new, full-thickness chondral flap (arrow).
Osteochondral Transplantation
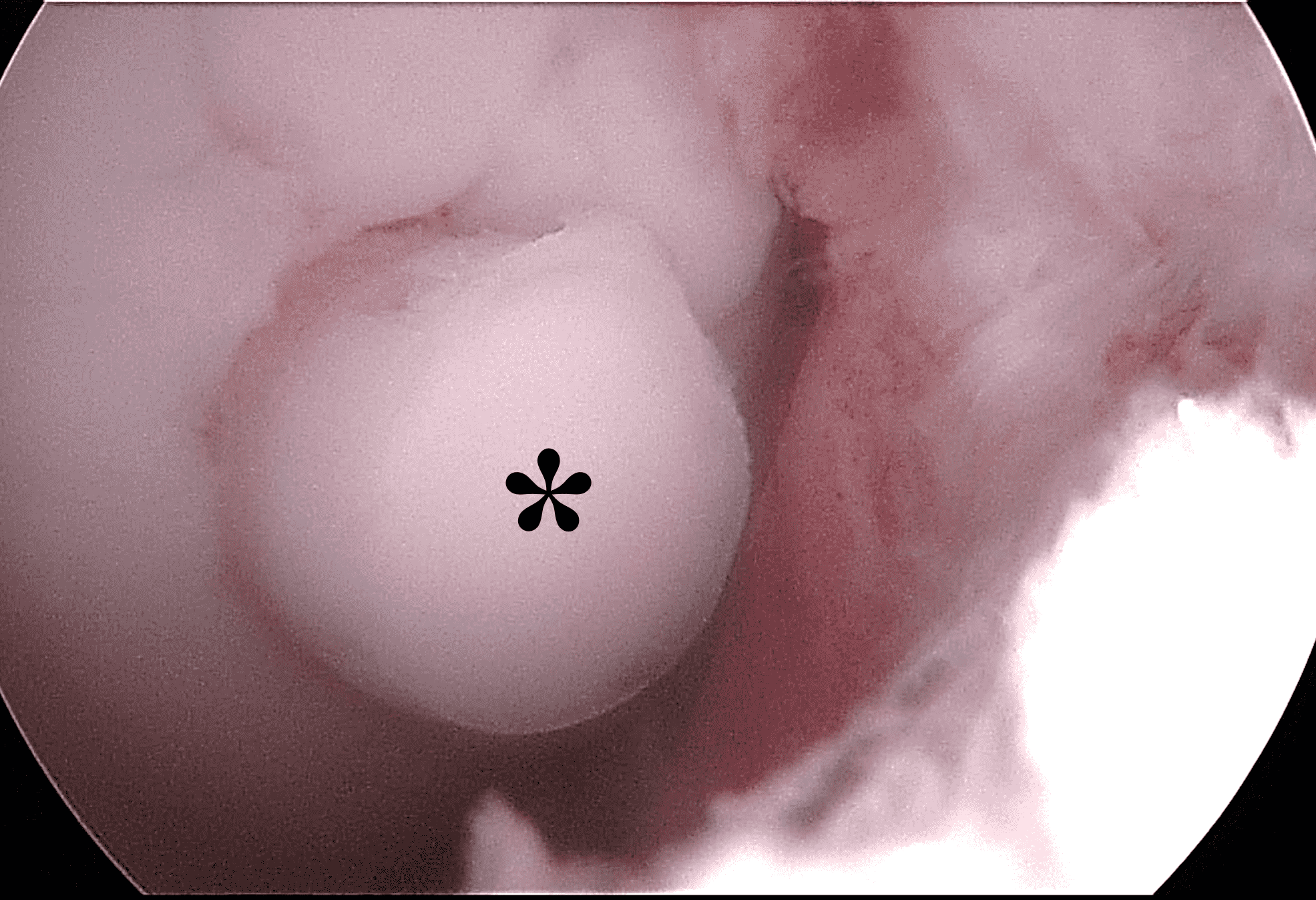
A second method to restore a damaged articular surface is by taking one or more grafts composed of subchondral bone with intact overlying articular cartilage and transferring them into the debrided defect, and then tamping down the graft(s) until the articular surface of the graft tissue is flush with the normal cartilage surrounding the defect (Figure 12). Because the deep aspect of the graft is composed of bone, it has the capacity to heal and incorporate with the surrounding subchondral bone, which stabilizes the graft together with its articular cartilage cap. The transplanted cartilage then functions as the new articular surface. A fibrocartilaginous “weld” typically forms between the cartilage of the grafts with each other and with the surrounding bone, or a thin gap may remain. Autografts are harvested from other sites in the same joint (or sometimes from a different joint in the same patient). Allografts are harvested from cadaver donors.
Figure 12: Arthroscopic photographs during osteochondral autograft procedure. (12A) A tubular cutting instrument (arrow) is used to harvest cylinders of bone plus cartilage from one part of the knee joint. (12B) An osteochondral graft (asterisk) has been placed into the debrided cartilage defect and is about to be pressed into place. Two additional grafts have already been placed adjacent to the current one.
Osteochondral Autograft Procedures
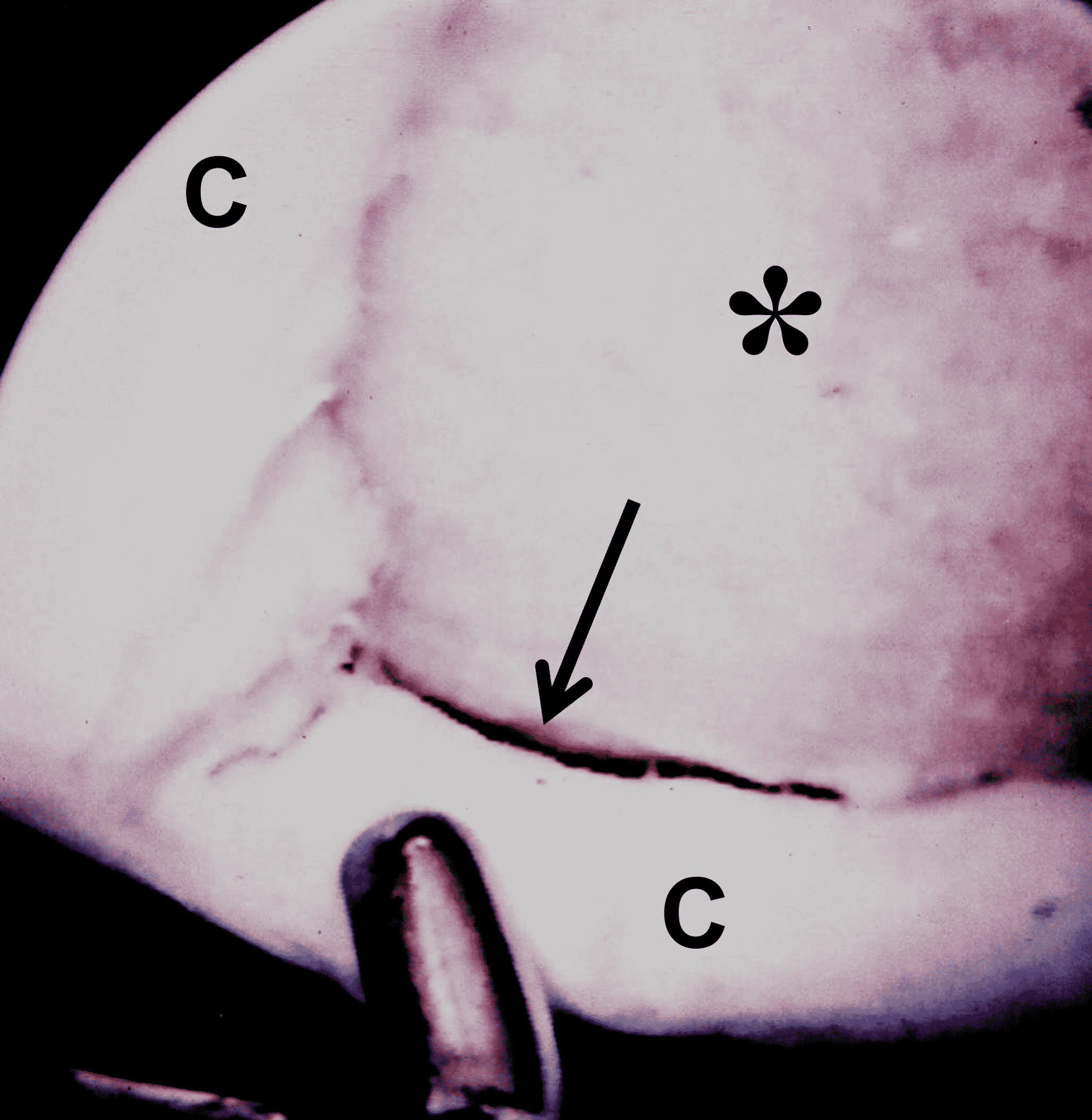
A frequent transfer performed in the knee is an arthroscopic osteochondral autograft, sometimes called a Mosaicplasty™ or OATS™ procedure. The donor sites are typically located along the outer or inner margins of the femoral trochlea, and are harvested with specific instrumentation (Figure 12A), producing cylinders 5-12 mm in diameter and 15-20 mm in length. The most common recipient sites are along the femoral condyles or patella. A donor site will typically backfill with trabeculae and marrow elements at its base and a thin layer of reparative fibrocartilage along its surface (Figure 13). Theoretically, the articular surface at the donor site is exposed to less stress compared to the treated region, but somewhere between 3 and 17% of patients develop symptoms at the donor site, usually relatively early after the procedure.23 Unfortunately, the MRI appearance of the donor site usually does not correlate with whether it is symptomatic or not.24
Figure 13: Osteochondral autograft donor site. (13A) Axial fat-suppressed fluid-sensitive image through a graft harvest site in the medial femoral trochlea. Fatty marrow has backfilled the deep portion of the donor site, which is faintly demarcated by granulation tissue (yellow arrows). Reparative fibrocartilage is present along the articular surface (red arrow). (13B) Arthroscopic image from a subsequent operation shows fibrocartilage capping the donor site (asterisk), which has a distinct color and surface compared to the surrounding normal cartilage.
Lesions amenable to osteochondral allografting are less than 2.5 cm2 limited by donor availability. Patients are usually less than 50 years old and active. The procedure may be performed concurrently with anterior cruciate ligament reconstruction or used as a secondary procedure following failed microfracture. Based on second-look surgery showing an intact articular surface and biopsies showing hyaline cartilage in the transplants, success varies from 92% for femoral condyle lesions to 79% for patellar lesions.25 95% of athletes treated by osteochondral allografts report improvement after 2-3 years.14 Intermediate- to long-term outcomes for OATS procedures are generally better than that for microfracture, including long-term return to play for athletes.26,27,28
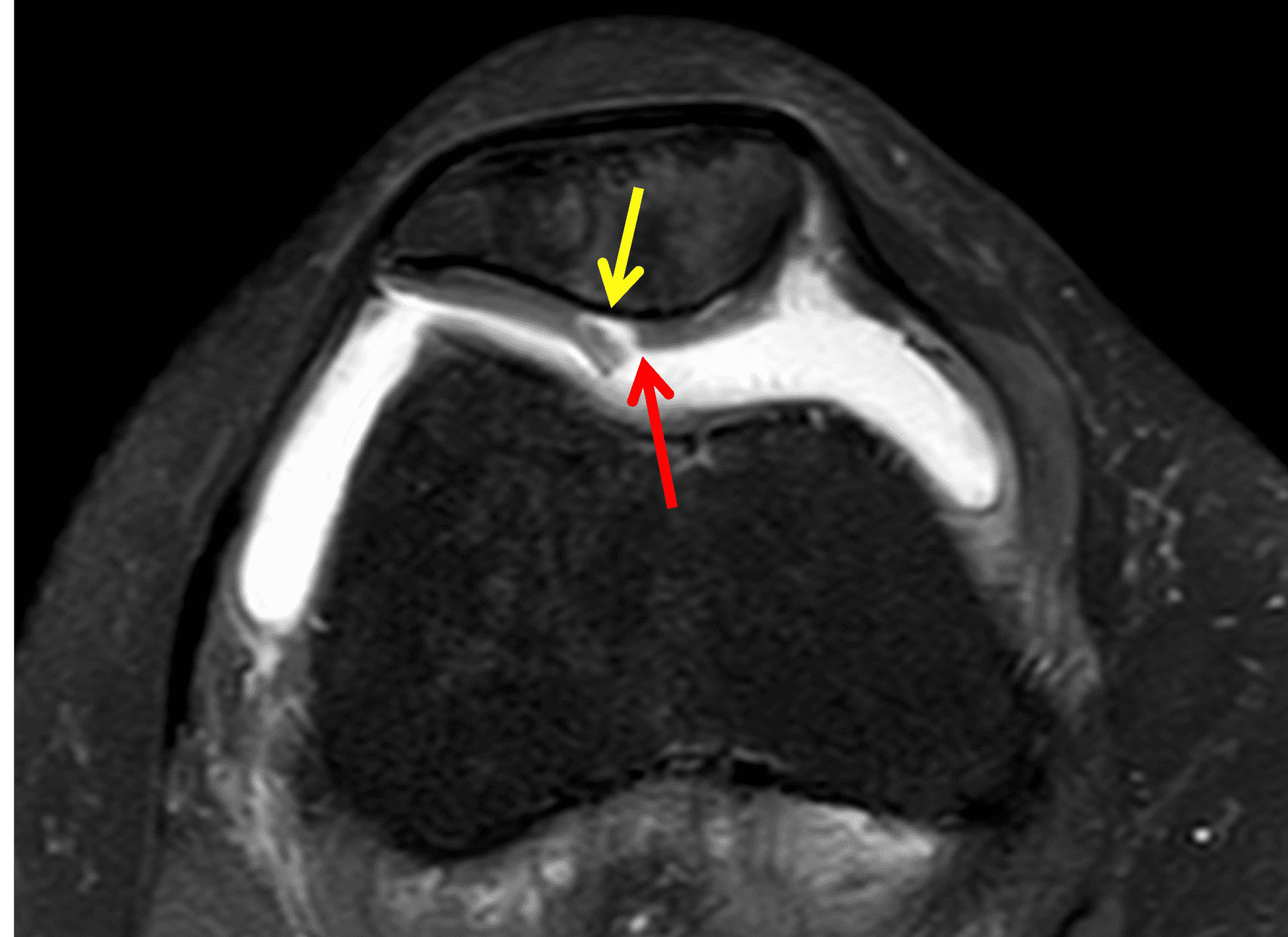
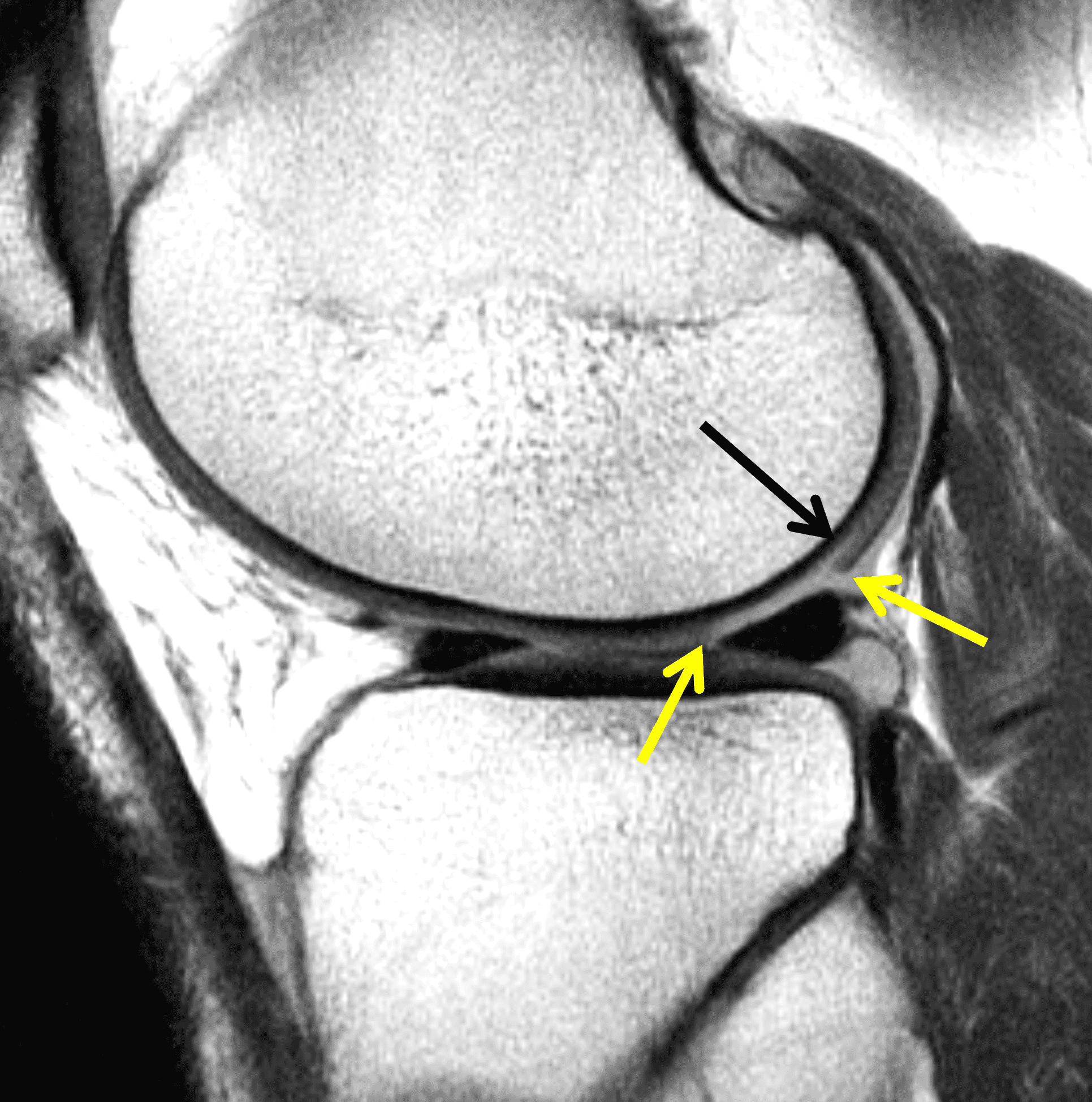
Because the thickness of the transplanted cartilage may differ from that at the recipient site, a step-off in the subchondral bone plate can be visible at the junction between the lesion and graft as a normal finding (Figure 14). The articular surface of the graft, however, should be congruent and flush with the surrounding cartilage on MRI. Marrow edema surrounding the graft is common in the first 3-9 months. The signal intensity within the transplanted cartilage can be variable, and like marrow changes, may take up to 3 years to normalize.29 Successful grafts will show incorporation of the transplanted bone with the surrounding bone (Figure 15). Cyst developmental at the base of graft may be a clue to poor incorporation (Figure 16) but can also be an incidental finding. Other than bone integration and surface congruence, most MRI findings have a weak correlation with outcomes. Osteonecrosis of the graft is a rare complication characterized by low signal intensity in the graft on T1-weighted images and lack of intravenous enhancement; however, graft osteonecrosis does not necessarily correlate with outcomes.24 Over time, osteochondral autografts may fail through development of degenerative arthritis with fissuring of the articular surface and cartilage loss (Figure 17).
Figure 14: Healing osteochondral autograft reconstruction for a patellar chondral defect. (14A) Sagittal proton density-weighted image 6 months after OATS shows a step-off between the subchondral bone plate of the graft (yellow arrow) and adjacent cartilage (black arrows). In this case the donor articular cartilage was thicker compared to the recipient site. (14B) Axial fat-suppressed fluid-sensitive image shows a congruent articular surface across the graft and surrounding cartilage (red arrows) despite the mismatch in the subchondral bone plates. High signal intensity in the grafted cartilage and marrow edema in the grafted bone can be normal findings up to 3 years after the procedure. A second look arthroscopy found that the graft was healing and confirmed a congruent articular surface.
Figure 15: Successful osteochondral autograft procedure for a chondral fracture of the medial femoral condyle. The procedure was performed approximately 7 years previously. (15A) Sagittal proton density-weighted image from a direct MR arthrogram shows osseous integration of the deep portion of the osteochondral graft (asterisk) and congruence of the articular surface (arrow). (15B) Sagittal fat-suppressed T2-weighted image shows normal signal within the graft, which is indistinguishable from the surrounding tissues.
Figure 16: Failed osteochondral allograft procedure. Surgery was performed 4 years previously. (16A) Sagittal T2-weighted and (16B) coronal fat-suppressed fluid-sensitive images show a recurrent full-thickness defect including the articular cartilage and subchondral bone plate of the graft (red arrows), with underlying marrow edema and cyst formation (yellow arrows) at the base of the graft. A loose osteochondral fragment was present in the knee (not shown), which was retrieved during a subsequent salvage procedure.
Figure 17: 20-year follow-up after osteochondral autograft procedure. (17A) Sagittal proton density-weighted and (17B) coronal fat-suppressed fluid-sensitive images show a healed osteochondral graft (asterisk) with mild cartilage loss and fissure of the overlying cartilage (arrows), representing degenerative arthritis.
Osteochondral Allograft Procedures
Larger chondral defects, lesions with extensive underlying bone loss, or previously failed restoration procedures may require an osteochondral allograft. These grafts are harvested from fresh or frozen cadavers and can be as large as the entire femoral condyle. Large grafts may be internally fixated to aid stability while healing occurs. Articular cartilage is immunologically privileged but bone is not, so an immunologic match between the donor and recipient is necessary.21 These procedures are typically performed with open surgery and reserved for relatively younger patients. They are also more costly compared to autograft or microfracture procedures. Graft survivorship is approximately 82% at 10 years.30 Approximately 80% of athletes who undergo osteochondral allografting are able to return to play at their pre-injury level.14 However, high rates of second operations for debridement or loose body removal have been reported.31,32
The postoperative imaging appearance of osteochondral allografts is similar to autografts (Figure 2).33 The subchondral bone plate of the donor and recipient sites do not have to match, but the articular surface should be congruent. Grafts that fail to incorporate by one year have a poor prognosis. Persistent marrow edema (beyond 12 months), a thick graft interface containing cysts or fluid, and subsidence of the graft are associated with poor osseous healing and worse outcomes. Extensive host marrow edema and severe synovitis may be a clue to immunologic rejection of the graft (Figure 18).21, 34
Figure 18: Failed osteochondral allograft. (18A) Sagittal fat-suppressed fluid-sensitive image shows the outline of an osteochondral allograft (red arrows) in the medial femoral condyle. Extensive marrow edema in the graft and underlying bone (asterisk) and severe hypertrophic synovitis (yellow arrow) are findings associated with immunologic rejection. (18B) Sagittal CT image acquired to assess graft healing shows lack of osseous integration (black arrows) with mild subsidence of the graft (yellow arrow) within the host site.
Cellular Repair
A third approach to traumatic chondral injuries is to regenerate hyaline cartilage to replace the lost cartilage. Autologous chondrocyte implantation (ACI) is traditionally performed as a two-step procedure, although recent modifications may change that.35,36 During initial arthroscopy, a sample of the patient’s own chondrocytes are harvested from a healthy part of the knee. These cells are then grown in an in-vitro cell culture, either in suspension or imbedded in a matrix that acts as a scaffolding.37 After 4-8 weeks the cultured chondrocytes are introduced into the original debrided cartilage defect during a second open operation. First generation procedures injected the cartilage cells as a slurry under a collagen or periosteal flap, while second and third generation procedures embed the new cells as a plug within a supporting matrix (matrix-ACI or m-ACI).14 Over time the transplanted chondrocytes should generate new extracellular matrix consisting of collagen and proteoglycans, restoring the normal articular surface.
ACI can be used for both acute and chronic large lesions.38 Like allografting, the procedure is expensive compared to osteochondral autograft and microfracture. Results of ACI are equivalent to microfracture and OATS at 2 years and as good or better than microfracture at 5 years, with femoral lesions faring better than patellar defects. Adhesions between the joint capsule and graft develop in 5-10% of patients and may result in stiffness.39 Approximately 33% of cases fail by 20 years.
On MRI following ACI, the transplanted cartilage may be hyperintense compared to native cartilage on fluid sensitive sequences for up to 3 years.40 Initial underfilling of the defect (due to resorption of the surrounding scaffolding) or graft hypertrophy (due to overgrowth of the cells or periosteal patch) may be present, and is usually not symptomatic, although debridement may be performed if overfilling of the defect results in mechanical symptoms.14,41 Underlying marrow edema is common, but typically resolves by 2-3 years. Unfortunately, other than a persistent joint effusion at 5 years, no postoperative MR findings are correlated with clinical outcomes (Figures 19 and 20).42,43 The one exception is an acute failure of first generation grafts where delamination of the entire graft or periosteal patch results in sudden onset of pain, typically in the first few months after the procedure. In cases of acute delamination, a fluid signal-intensity gap will be visible beneath the grafted tissue (Figure 21).14,44
Figure 19: Successful matrix-associated autologous chondrocyte implantation (m-ACI) procedure. (19A) Sagittal proton density-weighted image 4 years after m-ACI shows regenerated cartilage that is approximately isointense (arrows) compared to the native cartilage, with a smooth, congruent articular surface. (19B) Coronal fat-suppressed fluid sensitive sequence shows mild residual marrow edema (arrow) deep to the m-ACI site. The patient was asymptomatic at this location.
Figure 20: Symptomatic autologous chondrocyte implantation procedure (ACI). (20A) Sagittal T2-weighted image 14 months after ACI shows mild underfilling of the defect with tissue that is hyperintense compared to native cartilage (arrow). (20B) Coronal fat-suppressed fluid sensitive image shows hyperintensity of the articular cartilage (yellow arrow) and underlying subchondral marrow edema (white arrow). These MRI findings can be normal after ACI, but this patient had persistent pain that did not improve following the procedure.
Figure 21: Acute failure of a first generation autologous chondrocyte implantation (ACI) procedure due to acute graft delamination. Coronal fat-suppressed fluid sensitive image acquired 4 months after ACI shows a high signal-intensity interface deep to the grafted cartilage (arrows) representing delamination of the cartilage and overlying periosteal graft, as well as extensive underlying marrow edema (asterisk). The patient presented with atraumatic acute knee pain.
Conclusions
Multiple surgical procedures exist for the treatment of traumatic chondral lesions. An understanding of the expected postoperative imaging findings is necessary for the radiologist to understand which findings to consider abnormal and which do not strongly correlate with clinical results. After microfracture, under-filling of the defect and overgrowth of the subchondral bone are poor prognostic findings. The most common cause for failure of an osteochondral graft is lack of bone incorporation, although immune rejection is a potential complication following cadaveric allografting. However, most MRI findings are not related to clinical outcomes for osteochondral grafting. Similarly, following ACI, most MR findings are not correlated with results, except for acute failures of first generation procedures due to graft delamination.
References
- Flanigan DC, Harris JD, Trinh TQ, Siston RA, Brophy RH. Prevalence of Chondral Defects in Athletes’ Knees: A Systematic Review. Medicine & Science in Sports & Exercise. 2010;42(10). ↩
- Hoemann CD, Lafantaisie-Favreau CH, Lascau-Coman V, Chen G, Guzman-Morales J. The cartilage-bone interface. J Knee Surg. 2012;25(2):85–97. doi:10.1055/s-0032-1319782 ↩
- Marth AA, Link TM. Acute and chronic injury of the knee articular cartilage: prevalence, injury mechanisms, MRI assessment and association with osteoarthritis. Skeletal Radiol. 2025. doi:10.1007/s00256-025-05085-z ↩
- Rubin DA, Harner CD, Costello JM. Treatable chondral injuries in the knee: frequency of associated focal subchondral edema. AJR Am J Roentgenol. 2000;174(4):1099–1106. doi:10.2214/ajr.174.4.1741099 ↩
- Hayashi D, Li X, Murakami AM, Roemer FW, Trattnig S, Guermazi A. Understanding Magnetic Resonance Imaging of Knee Cartilage Repair: A Focus on Clinical Relevance. Cartilage. 2018;9(3):223–236. doi:10.1177/1947603517710309 ↩
- Lansdown DA, Wang K, Cotter E, Davey A, Cole BJ. Relationship Between Quantitative MRI Biomarkers and Patient-Reported Outcome Measures After Cartilage Repair Surgery: A Systematic Review. Orthop J Sports Med. 2018;6(4):2325967118765448. doi:10.1177/2325967118765448 ↩
- Schreiner MM, Raudner M, Marlovits S, Bohndorf K, Weber M, Zalaudek M, et al. The MOCART (Magnetic Resonance Observation of Cartilage Repair Tissue) 2.0 Knee Score and Atlas. Cartilage. 20191947603519865308. doi:10.1177/1947603519865308 ↩
- Blackman AJ, Smith MV, Flanigan DC, Matava MJ, Wright RW, Brophy RH. Correlation between magnetic resonance imaging and clinical outcomes after cartilage repair surgery in the knee: a systematic review and meta-analysis. Am J Sports Med. 2013;41(6):1426–1434. doi:10.1177/0363546513485931 ↩
- Tartibi S, Jackson GR, Boghosian T, Brophy RH, Smith MV, Matava MJ, et al. Fixation Methods, Complications, and Outcomes After Primary Fixation of Isolated Chondral Fragments in the Knee: A Systematic Review. Am J Sports Med. 2025;53(5):1231–1240. doi:10.1177/03635465241254520 ↩
- Salter RB. The biologic concept of continuous passive motion of synovial joints. The first 18 years of basic research and its clinical application. Clin Orthop Relat Res. 1989(242):12–25. ↩
- Kirkley A, Birmingham TB, Litchfield RB, Giffin JR, Willits KR, Wong CJ, et al. A randomized trial of arthroscopic surgery for osteoarthritis of the knee. N Engl J Med. 2008;359(11):1097–1107. doi:10.1056/NEJMoa0708333 ↩
- Moseley JB, O’Malley K, Petersen NJ, Menke TJ, Brody BA, Kuykendall DH, et al. A controlled trial of arthroscopic surgery for osteoarthritis of the knee. N Engl J Med. 2002;347(2):81–88. doi:10.1056/NEJMoa013259 ↩
- Scillia AJ, Aune KT, Andrachuk JS, Cain EL, Dugas JR, Fleisig GS, et al. Return to play after chondroplasty of the knee in National Football League athletes. Am J Sports Med. 2015;43(3):663–668. doi:10.1177/0363546514562752 ↩
- Guermazi A, Roemer F, Alizai H, Winalski CS, Welsch G, Brittberg M, et al. State of the Art: MR Imaging after Knee Cartilage Repair Surgery. Radiology. 2015;277(1):23–43. doi:10.1148/radiol.2015141146 ↩
- Steadman JR, Rodkey WG, Briggs KK. Microfracture: Its History and Experience of the Developing Surgeon. Cartilage. 2010;1(2):78–86. doi:10.1177/1947603510365533 ↩
- Erggelet C, Vavken P. Microfracture for the treatment of cartilage defects in the knee joint – A golden standard? Journal of Clinical Orthopaedics and Trauma. 2016;7(3):145–152. doi:10.1016/j.jcot.2016.06.015 ↩
- Goyal D, Keyhani S, Lee EH, Hui JH. Evidence-based status of microfracture technique: a systematic review of level I and II studies. Arthroscopy. 2013;29(9):1579–1588. doi:10.1016/j.arthro.2013.05.027 ↩
- Gobbi A, Karnatzikos G, Kumar A. Long-term results after microfracture treatment for full-thickness knee chondral lesions in athletes. Knee Surg Sports Traumatol Arthrosc. 2014;22(9):1986–1996. doi:10.1007/s00167-013-2676-8 ↩
- Frank RM, Cotter EJ, Nassar I, Cole B. Failure of Bone Marrow Stimulation Techniques. Sports Med Arthrosc Rev. 2017;25(1):2–9. doi:10.1097/jsa.0000000000000134 ↩
- Gomoll AH. Microfracture and augments. J Knee Surg. 2012;25(1):9–15. doi:10.1055/s-0031-1299654 ↩
- Alparslan L, Winalski CS, Boutin RD, Minas T. Postoperative magnetic resonance imaging of articular cartilage repair. Semin Musculoskelet Radiol. 2001;5(4):345–363. doi:10.1055/s-2001-19044 ↩
- Welton KL, Logterman S, Bartley JH, Vidal AF, McCarty EC. Knee Cartilage Repair and Restoration: Common Problems and Solutions. Clin Sports Med. 2018;37(2):307–330. doi:10.1016/j.csm.2017.12.008 ↩
- Andrade R, Vasta S, Pereira R, Pereira H, Papalia R, Karahan M, et al. Knee donor-site morbidity after mosaicplasty – a systematic review. J Exp Orthop. 2016;3(1):31. doi:10.1186/s40634-016-0066-0 ↩
- Link TM, Mischung J, Wortler K, Burkart A, Rummeny EJ, Imhoff AB. Normal and pathological MR findings in osteochondral autografts with longitudinal follow-up. Eur Radiol. 2006;16(1):88–96. doi:10.1007/s00330-005-2818-6 ↩
- Hangody L, Kish G, Karpati Z, Szerb I, Udvarhelyi I. Arthroscopic autogenous osteochondral mosaicplasty for the treatment of femoral condylar articular defects. A preliminary report. Knee Surg Sports Traumatol Arthrosc. 1997;5(4):262–267. doi:10.1007/s001670050061 ↩
- Pareek A, Reardon PJ, Macalena JA, Levy BA, Stuart MJ, Williams RJ, et al. Osteochondral Autograft Transfer Versus Microfracture in the Knee: A Meta-analysis of Prospective Comparative Studies at Midterm. Arthroscopy: The Journal of Arthroscopic & Related Surgery. 2016;32(10):2118–2130. doi:10.1016/j.arthro.2016.05.038 ↩
- Gudas R, Gudaite A, Pocius A, Gudiene A, Cekanauskas E, Monastyreckiene E, et al. Ten-year follow-up of a prospective, randomized clinical study of mosaic osteochondral autologous transplantation versus microfracture for the treatment of osteochondral defects in the knee joint of athletes. Am J Sports Med. 2012;40(11):2499–2508. doi:10.1177/0363546512458763 ↩
- Lynch TS, Patel RM, Benedick A, Amin NH, Jones MH, Miniaci A. Systematic review of autogenous osteochondral transplant outcomes. Arthroscopy. 2015;31(4):746–754. doi:10.1016/j.arthro.2014.11.018 ↩
- Sanders TG. Imaging of the postoperative knee. Semin Musculoskelet Radiol. 2011;15(4):383–407. doi:10.1055/s-0031-1286018 ↩
- Levy YD, Gortz S, Pulido PA, McCauley JC, Bugbee WD. Do fresh osteochondral allografts successfully treat femoral condyle lesions? Clin Orthop Relat Res. 2013;471(1):231–237. doi:10.1007/s11999-012-2556-4 ↩
- Crawford ZT, Schumaier AP, Glogovac G, Grawe BM. Return to Sport and Sports-Specific Outcomes After Osteochondral Allograft Transplantation in the Knee: A Systematic Review of Studies With at Least 2 Years’ Mean Follow-Up. Arthroscopy. 2019;35(6):1880–1889. doi:10.1016/j.arthro.2018.11.064 ↩
- McCarthy MA, Meyer MA, Weber AE, Levy DM, Tilton AK, Yanke AB, et al. Can Competitive Athletes Return to High-Level Play After Osteochondral Allograft Transplantation of the Knee? Arthroscopy. 2017;33(9):1712–1717. doi:10.1016/j.arthro.2017.03.020 ↩
- Wang T, Wang DX, Burge AJ, Pais M, Kushwaha B, Rodeo SA, et al. Clinical and MRI Outcomes of Fresh Osteochondral Allograft Transplantation After Failed Cartilage Repair Surgery in the Knee. J Bone Joint Surg Am. 2018;100(22):1949–1959. doi:10.2106/JBJS.17.01418 ↩
- Sirlin CB, Brossmann J, Boutin RD, Pathria MN, Convery FR, Bugbee W, et al. Shell osteochondral allografts of the knee: comparison of mr imaging findings and immunologic responses. Radiology. 2001;219(1):35–43. doi:10.1148/radiology.219.1.r01ap0435 ↩
- Brittberg M, Lindahl A, Nilsson A, Ohlsson C, Isaksson O, Peterson L. Treatment of deep cartilage defects in the knee with autologous chondrocyte transplantation. N Engl J Med. 1994;331(14):889–895. doi:10.1056/nejm199410063311401 ↩
- Slynarski K, de Jong WC, Snow M, Hendriks JAA, Wilson CE, Verdonk P. Single-Stage Autologous Chondrocyte-Based Treatment for the Repair of Knee Cartilage Lesions: Two-Year Follow-up of a Prospective Single-Arm Multicenter Study. Am J Sports Med. 2020;48(6):1327–1337. doi:10.1177/0363546520912444 ↩
- Steinwachs M, Kreuz PC. Autologous chondrocyte implantation in chondral defects of the knee with a type I/III collagen membrane: a prospective study with a 3-year follow-up. Arthroscopy. 2007;23(4):381–387. doi:10.1016/j.arthro.2006.12.003 ↩
- Biant LC, Bentley G, Vijayan S, Skinner JA, Carrington RW. Long-term results of autologous chondrocyte implantation in the knee for chronic chondral and osteochondral defects. Am J Sports Med. 2014;42(9):2178–2183. doi:10.1177/0363546514539345 ↩
- Alparslan L, Minas T, Winalski CS. Magnetic resonance imaging of autologous chondrocyte implantation. Semin Ultrasound CT MR. 2001;22(4):341–351. doi:10.1016/s0887-2171(01)90025-3 ↩
- Niethammer TR, Safi E, Ficklscherer A, Horng A, Feist M, Feist-Pagenstert I, et al. Graft maturation of autologous chondrocyte implantation: magnetic resonance investigation with T2 mapping. Am J Sports Med. 2014;42(9):2199–2204. doi:10.1177/0363546514538756 ↩
- Niethammer TR, Pietschmann MF, Ficklscherer A, Gülecyüz MF, Hammerschmid F, Müller PE. Incomplete defect filling after third generation autologous chondrocyte implantation. Arch Med Sci. 2016;12(4):785–792. doi:10.5114/aoms.2016.60967 ↩
- Knutsen G, Drogset JO, Engebretsen L, Grontvedt T, Ludvigsen TC, Loken S, et al. A Randomized Multicenter Trial Comparing Autologous Chondrocyte Implantation with Microfracture: Long-Term Follow-up at 14 to 15 Years. J Bone Joint Surg Am. 2016;98(16):1332–1339. doi:10.2106/JBJS.15.01208 ↩
- Ebert JR, Zheng M, Fallon M, Wood DJ, Janes GC. 10-Year Prospective Clinical and Radiological Evaluation After Matrix-Induced Autologous Chondrocyte Implantation and Comparison of Tibiofemoral and Patellofemoral Graft Outcomes. Am J Sports Med. 2024;52(4):977–986. doi:10.1177/03635465241227969 ↩
- Recht M, White LM, Winalski CS, Miniaci A, Minas T, Parker RD. MR imaging of cartilage repair procedures. Skeletal Radiol. 2003;32(4):185–200. doi:10.1007/s00256-003-0631-3 ↩