Clinical History
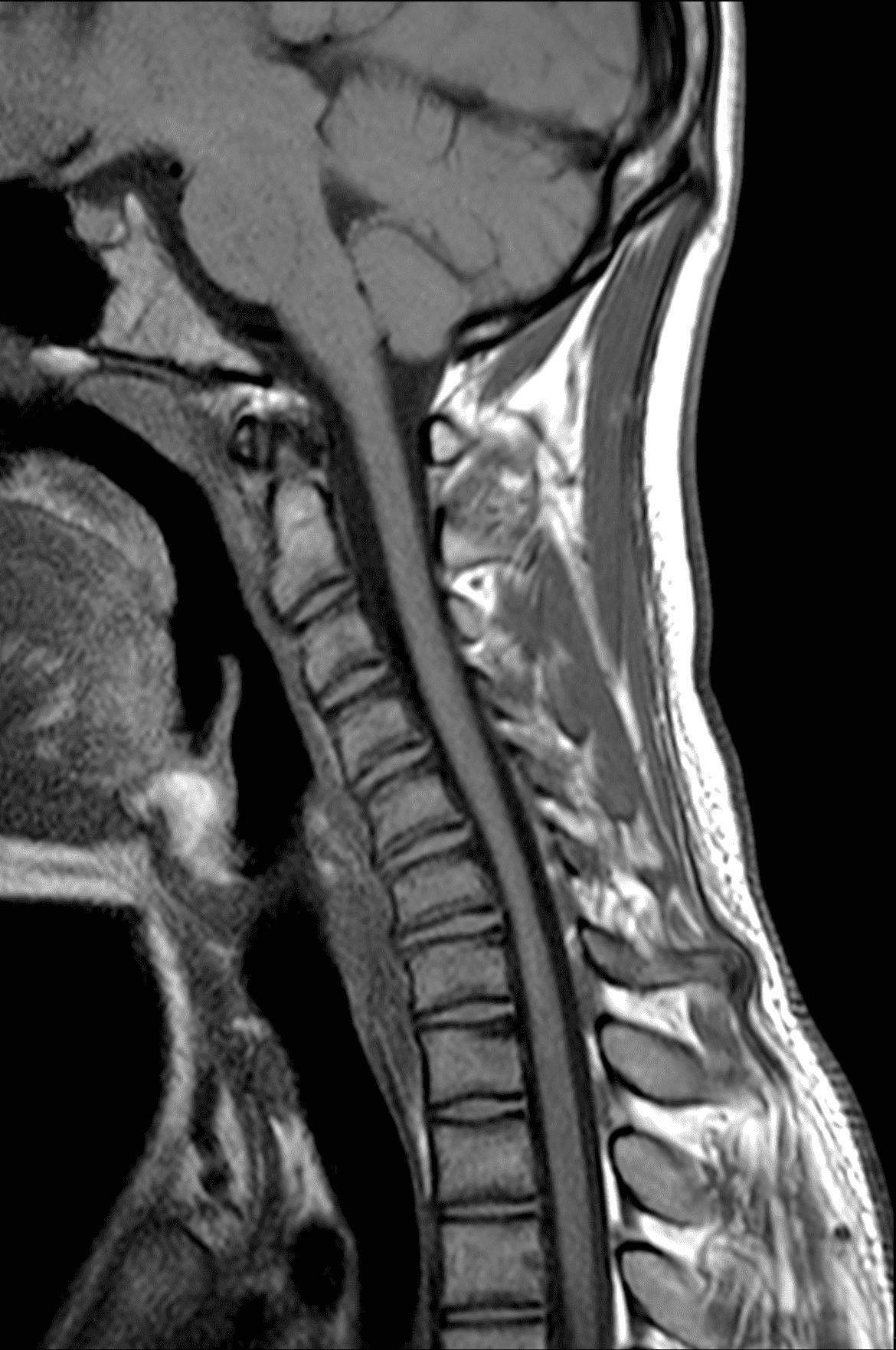
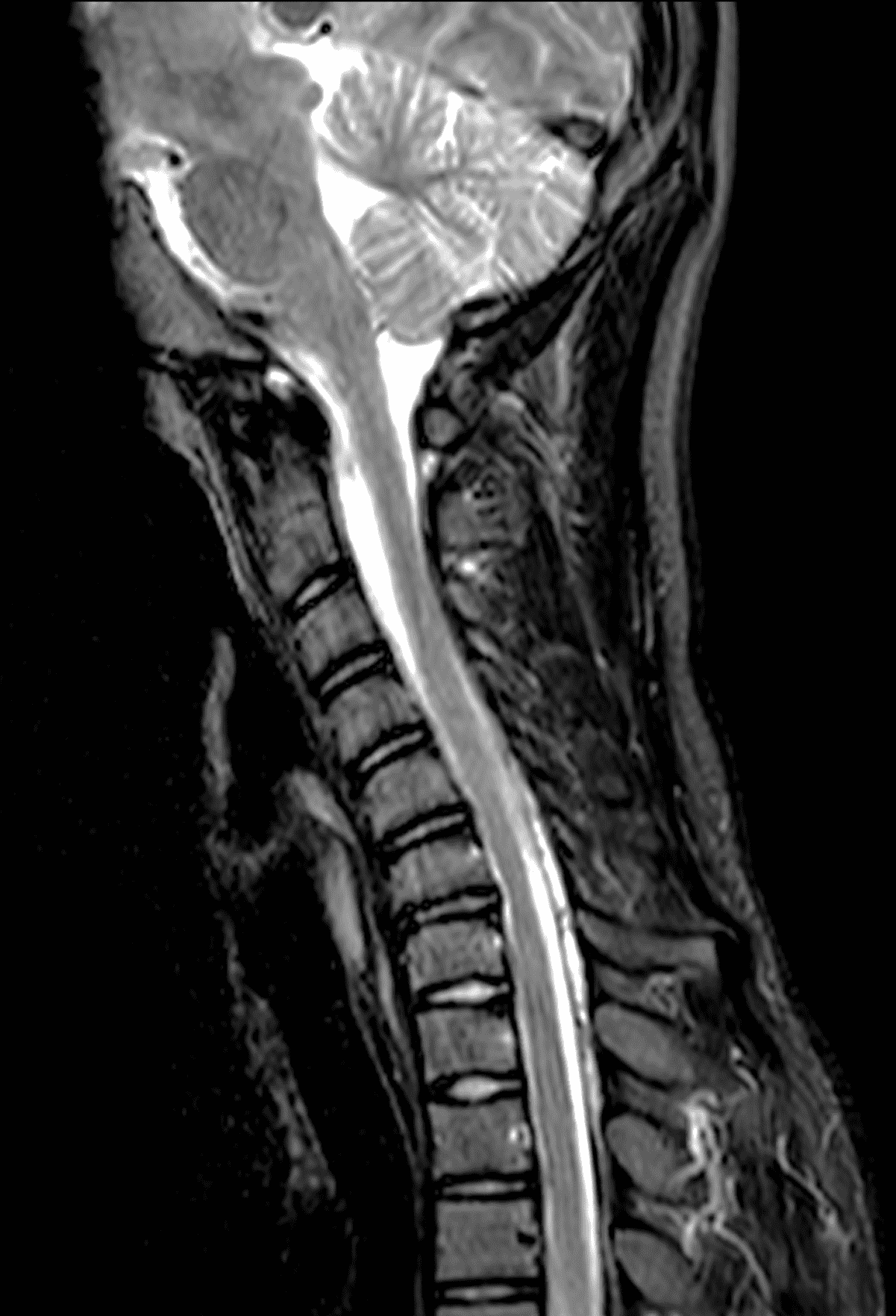
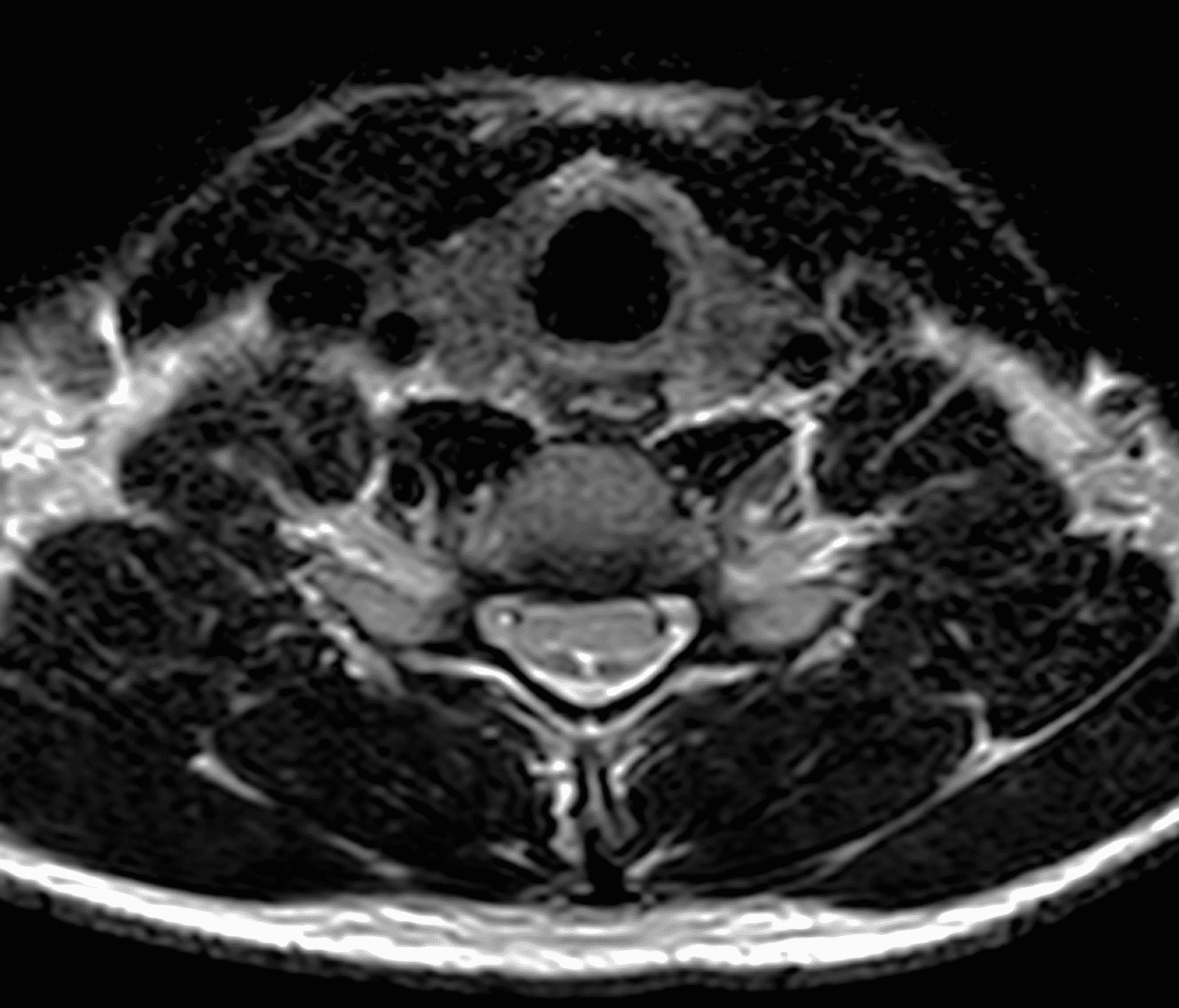
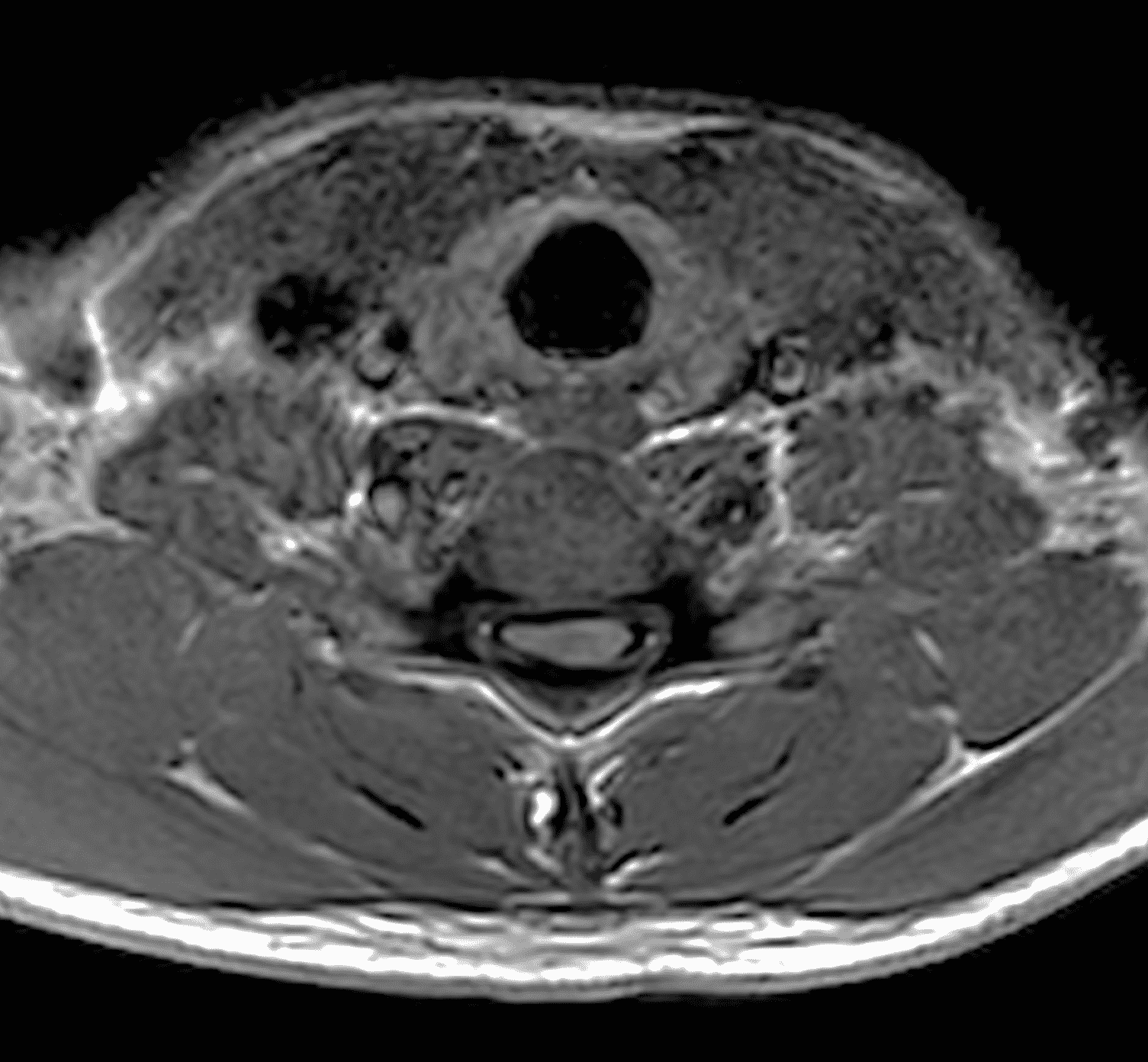
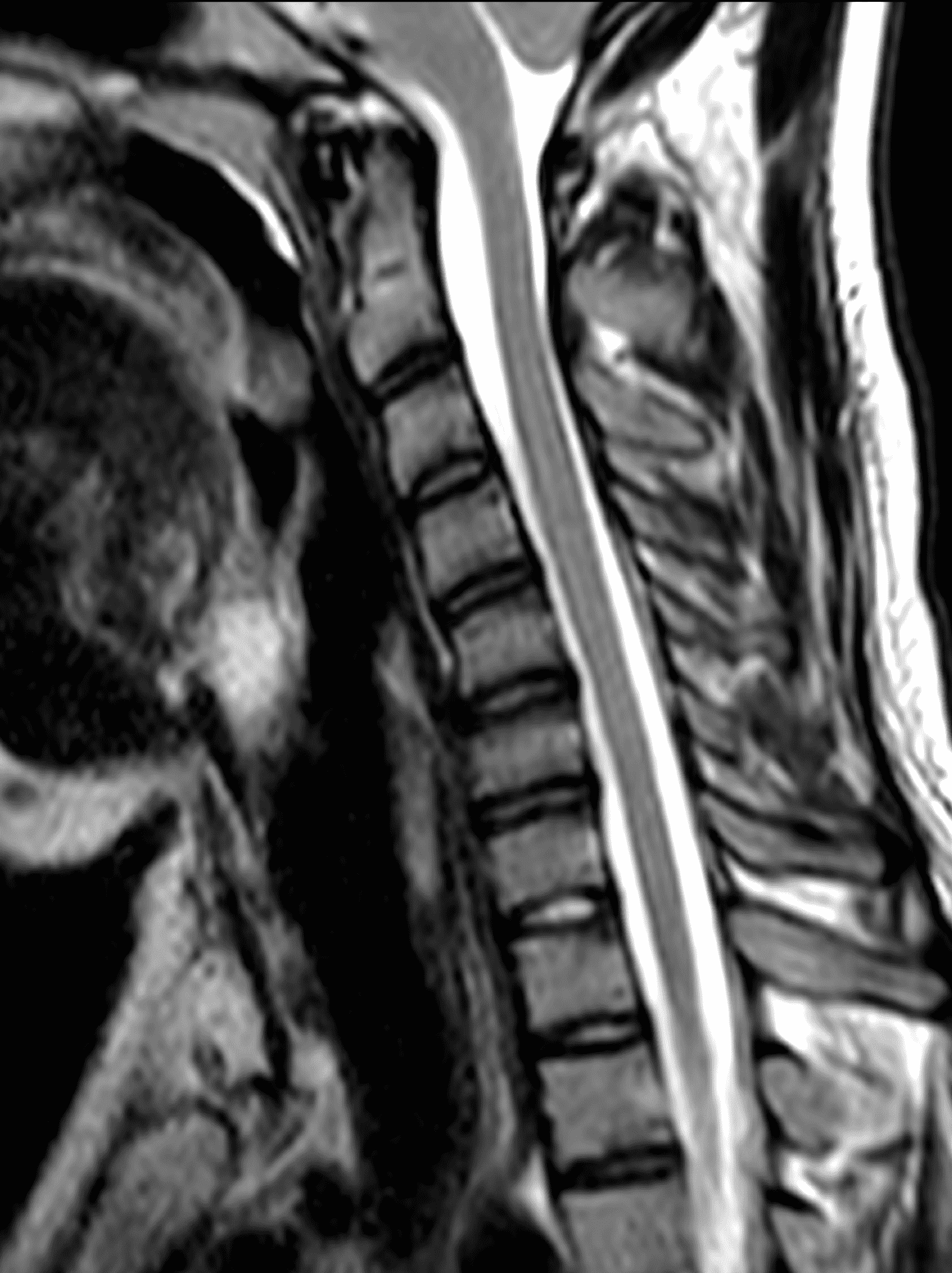
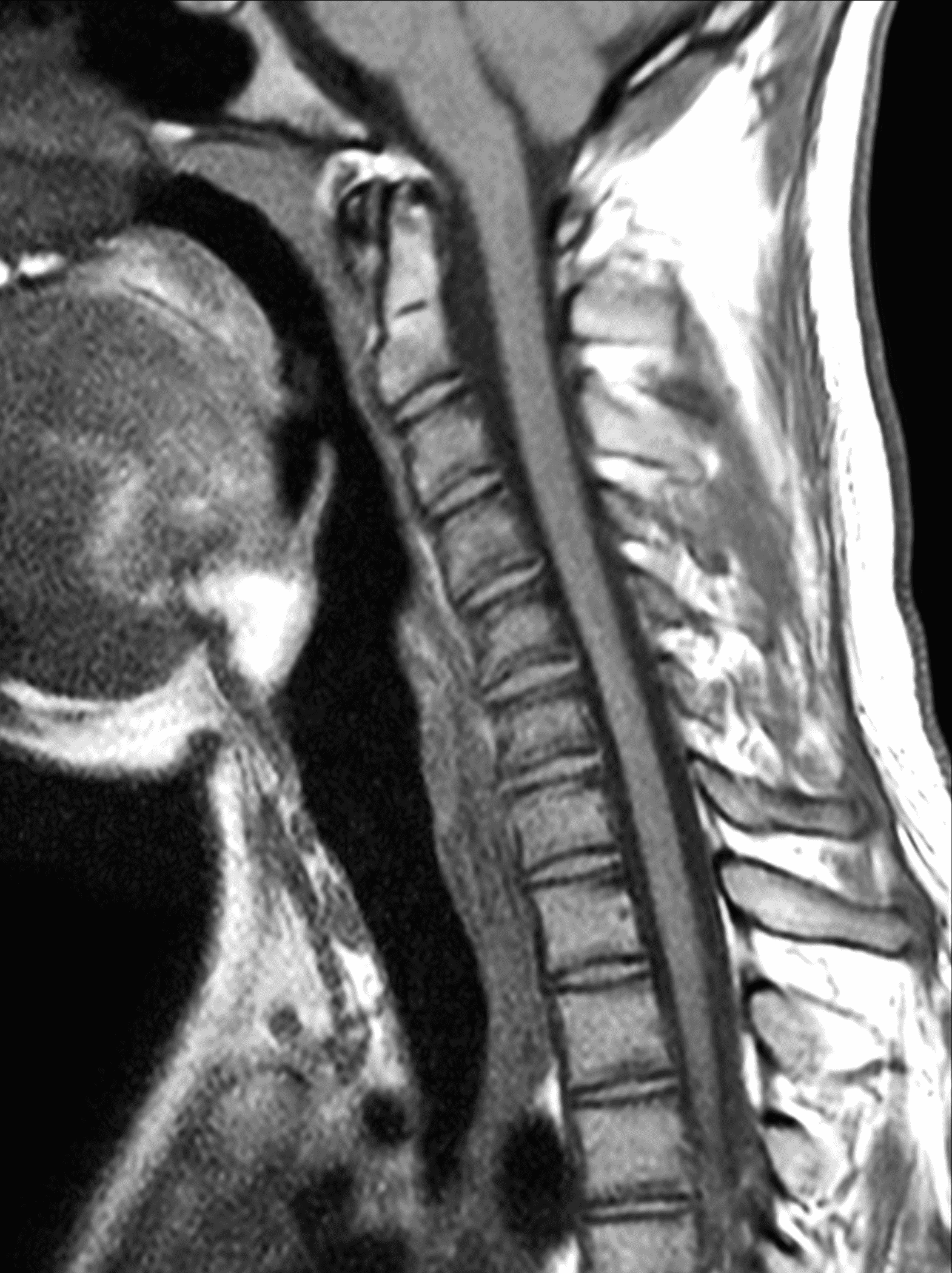
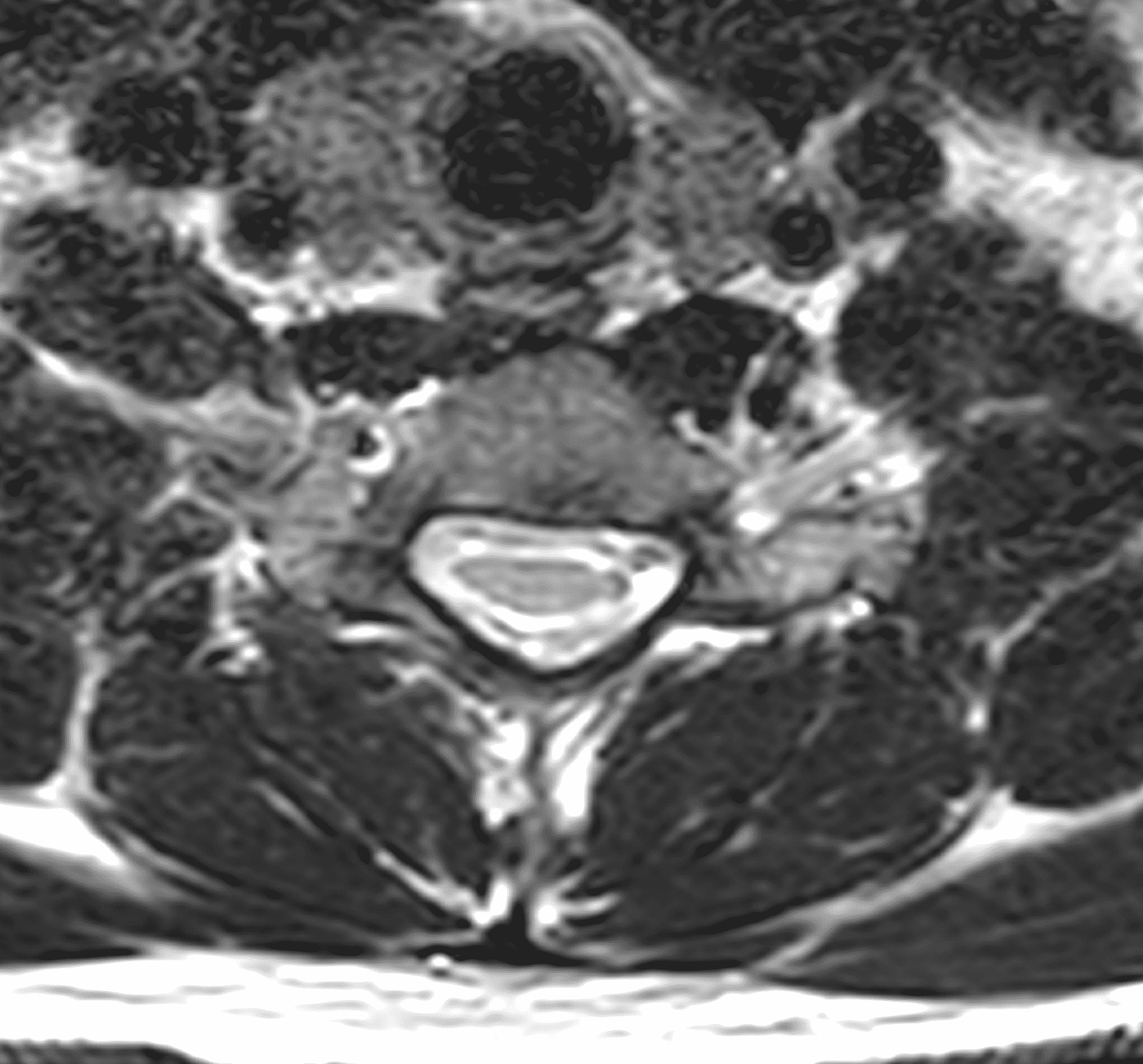
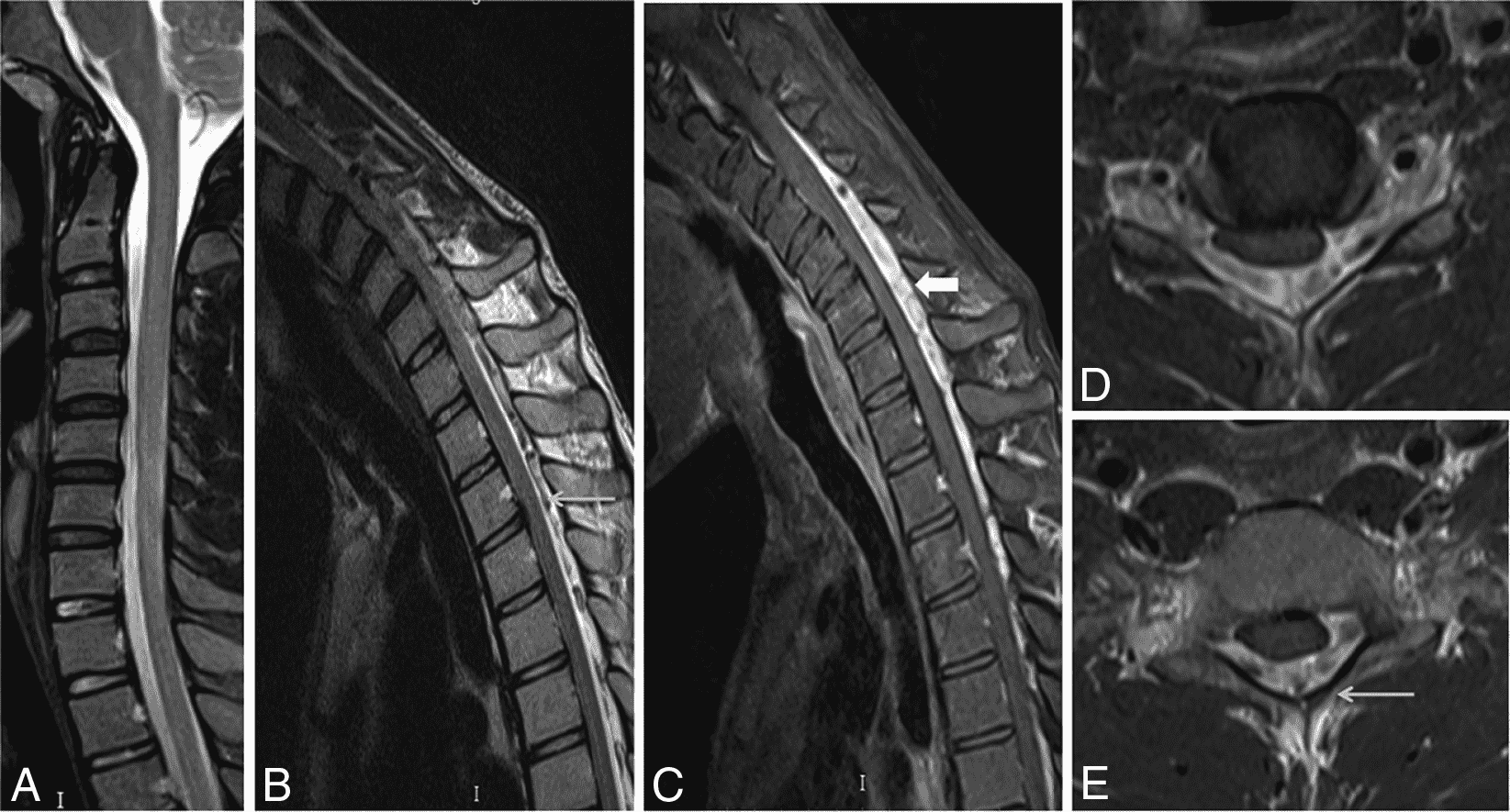
An 18 year old man presents with progressive motor wasting of right hand, progressive right hand weakness, claw-like deformity with flattening of the hand, and inability to straighten the ring and small fingers. He reports no numbness or pain. Sagittal T2-weighted (1A), sagittal T1-weighted (1B), sagittal STIR (1C), axial T2-weighted (1D), and axial T1-weighted (1E) images of the cervical spine obtained around the time of presentation are provided. What are the findings? What is your diagnosis?
Findings
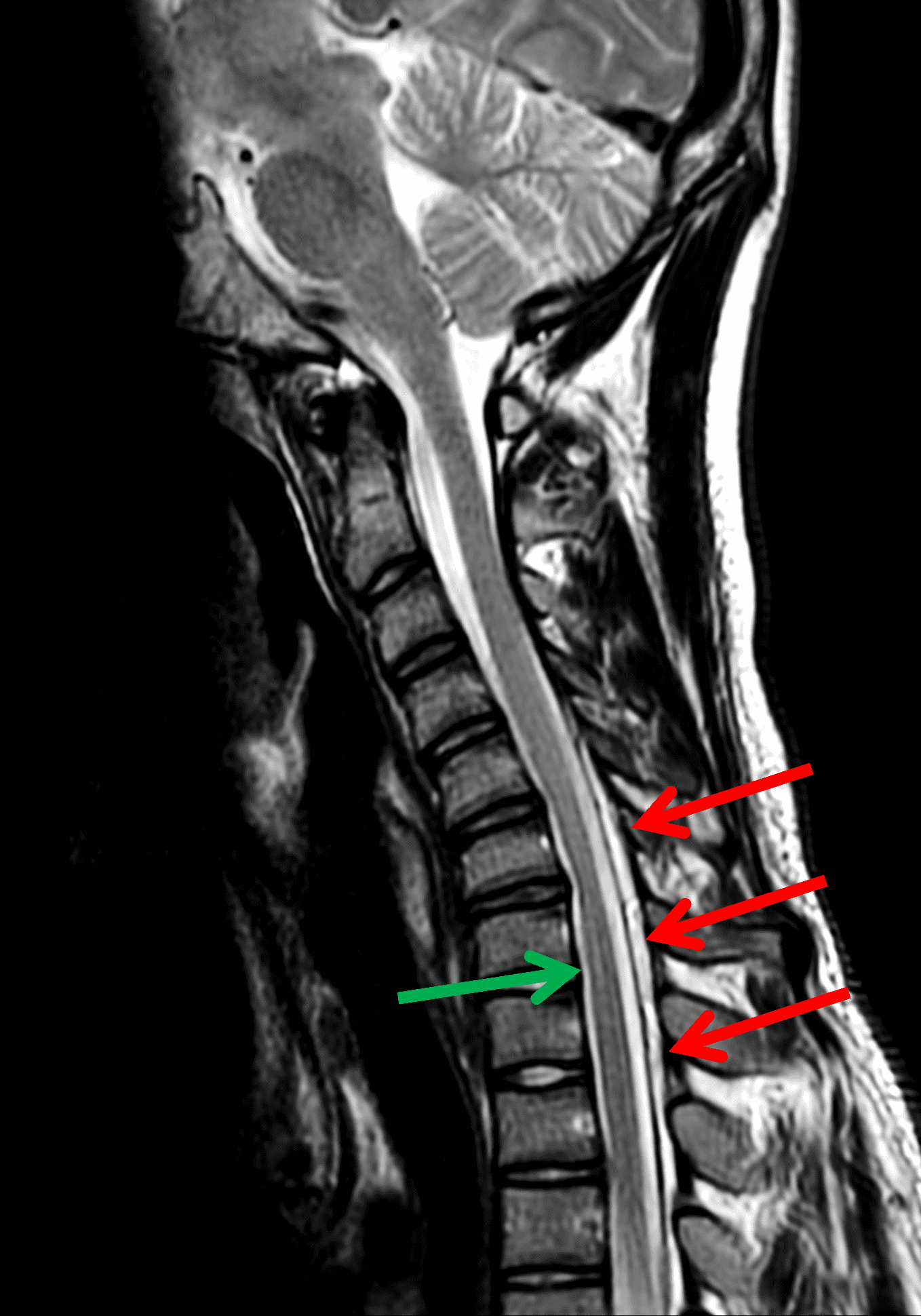
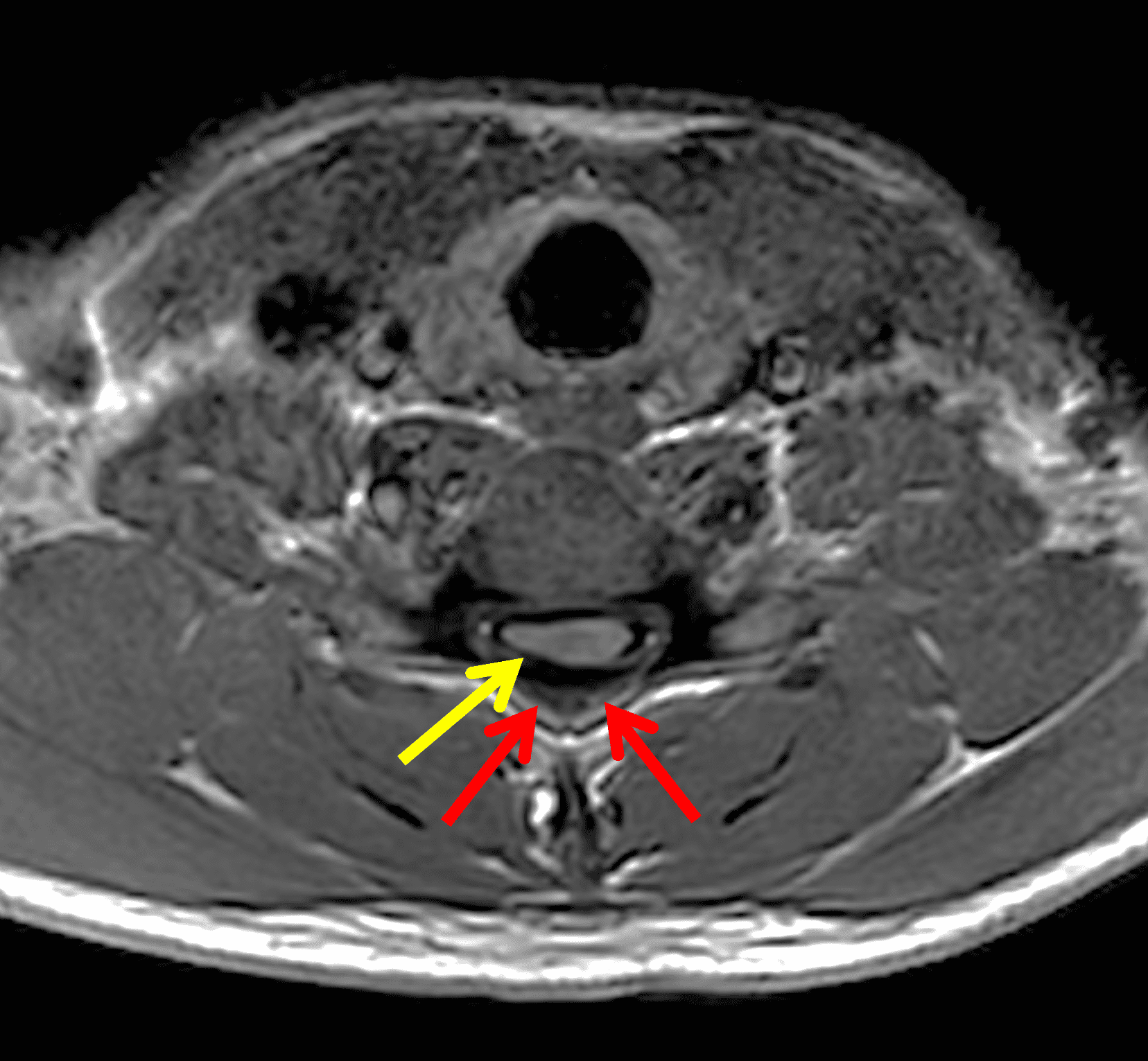
Figure 2: Neutral images of the cervical spine. Sagittal (2A) T2-weighted, (2B) T1-weighted, and (2C) STIR images show mild kyphosis, an enlarged posterior epidural space with loss of attachment of the dura (red arrows), and cord atrophy at the C6 level (yellow arrow). Subtle cord hyperintensity on the water sensitive images at the C7 level (green arrows) likely represents gliosis. (2D) Axial T2-weighted image shows enlargement of the posterior epidural space, right greater than left (red arrows). (2E) Axial T1-weighted image shows an enlarged posterior epidural space (red arrows) and cord atrophy, more severe on the right (yellow arrow).
Diagnosis
Hirayama disease (also known as juvenile benign muscular atrophy of the distal upper extremity, or monomelic amyotrophy).
Introduction
Hirayama disease (HD), also termed juvenile benign muscular atrophy of the upper extremity, is a self-limited cervical myelopathy first described by Hirayama et al.1 in 1959, who presented 12 cases as a new clinical entity distinguished from degenerative and progressive motor neuron disease (i.e. amyotrophic lateral sclerosis [ALS] and spinal progressive muscular atrophy). Additional case studies and case series followed predominantly in Japan and other Asian countries confirming a disease related to flexion movements of the neck.2,3 More recently, this disease has become better recognized by clinicians outside of Asia and several case series have been reported in Europe4 and North America.5
Early case series established that patients are predominantly male in their teens to early twenties with an insidious onset of distal upper limb muscular weakness and wasting in the hand and forearm, sparing the brachioradialis muscle, that may progress over 1 – 3 years before stabilizing.1,2 Early studies focused on clinical features and electrophysiology, but it was not until the first autopsy case was reported,6 that a better understanding of the pathogenesis was formed. The case revealed anterior-posterior flattening of the lower cervical cord with ischemic and atrophic changes of the anterior horn cells, confirming an anterior horn cell disease rather than a subtype of motor neuron disease.
Since MRI became more accessible in the late 1980’s, studies have demonstrated findings on neutral and flexion imaging that have assisted in diagnosis. Imaging reveals dynamic changes with flexion including asymmetric flattening of the cord, forward displacement of the posterior dural sac, and posterior epidural space hyperintensity.7,8,9
Many different names have been used in the literature including monomelic amyotrophy and juvenile spinal muscular atrophy of the distal upper extremity. However, given that there are many overlapping entities when using these other names, Hirayama disease is still the most commonly used term in the literature and more recent authors propose continuing to use Hirayama disease (HD) to label this disease entity.10
Clinical Presentation
The mean age of onset of Hirayama disease is between 15-20 years of age with a significant male predominance.11,12,13 Male-to-female prevalence ranges up to 20:1 in Japan11 and 31.6:1 in China14 to as low as 2.8:1 in India. Several studies noted that disease onset occurs approximately 2 years after the peak velocity of the growth curve.11 Tashiro et al.15 proposed that the male prevalence could be due to more rapid growth spurt in boys compared to girls during puberty, although the inconsistent onset between males and females may also be a factor. Familial cases are quite rare with 9 total cases reported in the literature (Figure 3).16
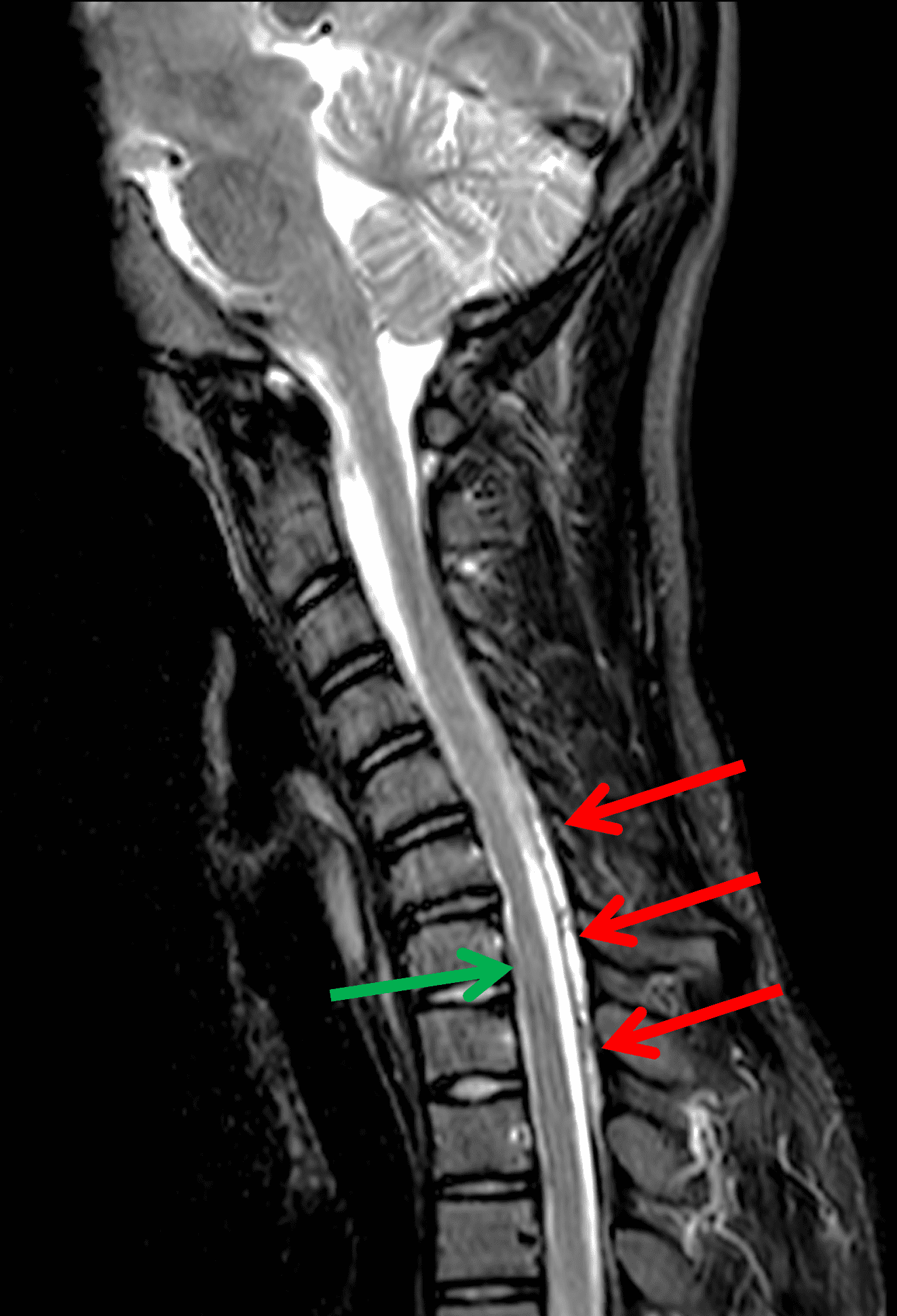
Figure 3: Asymptomatic twin brother of the case shown in Figures 1 and 2, who also underwent work-up. The study was performed in attempted flexion. (3A) Sagittal T2-weighted, (3B) sagittal T1-weighted, (3C) axial T2-weighted, and (3D) axial T1-weighted images show no evidence of cord atrophy/flattening, enlarged posterior epidural space, or forward displacement of the posterior dural sac.
Patients typically present with: 1) insidious onset and progression over 3-5 years followed by spontaneous arrest or a relatively benign period, 2) unilateral or asymmetric bilateral muscle weakness and atrophy involving the distal upper extremities, 3) cold paresis with mild exacerbation of finger weakness on exposure to cold environment, 4) muscle fasciculations with contraction but not at rest, and 5) absence of sensory disturbances.10,17
Muscle weakness and atrophy affect the hand and forearm, including involvement of thenar, hypothenar, interossei muscles, and wrist flexors and extensors, with sparing of brachioradialis muscles. Most patients have more severe ulnar side atrophy,13 a condition called “reverse split hand syndrome,” which may be a feature distinguishing HD from ALS.18 The border of atrophy runs obliquely through volar and dorsal parts of the forearm and had been termed oblique amyotrophy by Hirayama in the past. The right upper limb is more frequently involved regardless of handedness.2 There is commonly unilateral monomelic atrophy, but some patients can present with asymmetric bilateral involvement. Occasionally, there is bilateral symmetric atrophy with an incidence of 3.1% observed in a National Japan Survey,15 and up to 10% in another case series.19 Pradhan et al.19 proposed bilateral symmetric HD to be a more severe variant based on more severe and significant clinical and MR findings.
HD patients experience mild transient worsening of symptoms when exposed to cold environments.11,17 Cold paresis is seen in up to 97% of patients.20 Contraction fasciculation is characterized by fascicular twitching of extensor-side forearm muscles or tremor-like movement of fingers that occurs during stretching but not at rest.
Atypical presentations include: 1) pyramidal signs, 2) atrophy of the proximal upper extremities, 3) long progression, and 4) sensory deficits.10,17 HD is a disease involving anterior horn cells but pyramidal signs, especially Hoffmann sign, appear in some patients, 2.4% in a Japanese study15 and 10.6% in a Chinese study.14 Pyramidal signs are assumed to be the consequence of severe cord injury and whether or not pyramidal signs are present depends on the severity of disease.
Cord compression with flexion in HD usually involves the lower cervical cord, mainly the C7-T1 level, and thus atrophy is primarily observed in the distal upper extremity. However, in a few cases symptoms extend proximally. Functional outcomes are likely worse and more uncertain in patients with proximal symptoms compared to those with the typical distal form of the disease.
Most cases are self-limited with disease progression ending in less than 5 years. While some patients recover function due to reinnervation, most are left with a mild residual disability. In a few instances, the disease progresses over more than 10 years, and the patient’s condition continues to deteriorate after a stable period.21 Additionally, the imaging abnormalities do not improve as the disease progresses, suggesting it is not self-limiting in a small subset of patients.22
The majority of clinical findings are motor related with absence of sensory findings, though a few patients have shown slight hypoesthesia in a localized area of the hand.11,12 One study reported sensory deficits in 19.2% of patients.15 Sensory findings are believed to occur due to compression of the sensory tracts, with deficits becoming more severe with higher stress.
Pathogenesis/Imaging Findings
Pathologic study of HD was not possible prior to 1982 due to the benign clinical course. The first autopsy case was performed in 1982 in a patient who suffered from lung cancer for 3 months and died at the age of 38.6 The patient had onset of HD symptoms in the distal upper extremities predominantly on the left at age 15, which progressed for one year before spontaneous arrest and symptoms remained unchanged. Gross pathology revealed anterior-posterior flattening of the cervical cord, particularly the lower cervical cord. Microscopically, the anterior-posterior diameters of the lower cervical anterior horns were reduced on both sides (left greater than right), most severely at C7 and C8. The anterior horn lesion was characterized by a decreased number of large and small neurons, mild astrogliosis, and central necrosis without cavity formation, findings unlikely in degenerative motor neuron disease. The surviving neurons showed various stages of degenerative change while white matter, posterior horns, and intra- and extramedullary vessels were normal. These findings strongly suggest an ischemic cervical myelopathy localized in the lower cervical cord, and exclude the possibility of motor neuron disease. The neuropathologic findings prompted imaging of the spinal contents, particularly with the improvements in MR imaging.
Early MR studies revealed dynamic changes of the spinal cord and dural sac during neck flexion in HD patients including forward displacement of the posterior dural sac,7 overstretching of the cord,8 a tight dural canal,3 and asymmetric compression and anterior-posterior flattening of the lower cervical cord. The exact cause of HD is unknown, but several mechanisms have been proposed. Hirayama et al.11 speculated that the disease could be related to disproportional growth between the vertebral column and contents of the spinal canal/dural sac during the growth spurt of puberty. This may explain the delay that he noted between the peak age of onset and peak velocity of the growth curve. However, this theory alone cannot explain asymmetric cord involvement. Shinomiya et al.23 proposed a posterior epidural ligament factor. There are two kinds of posterior epidural ligaments between the posterior dura and ligamentum flavum: fine elastic ligaments and large ligaments (approximately 1-3 mm in diameter). These ligaments are abundant at C1-2, decreased below C2, and sparse at C6 and C7. The posterior epidural ligaments may aid in resistance against separation of the posterior dura from the ligamentum flavum. Unequal distribution or lack of these ligaments may be the cause of asymmetric cord compression.
MRI findings with neck flexion
Flexion position MRI of the cervical spine can clearly depict the forward displacement of the posterior dural sac as well as asymmetric compression of the lower cervical cord. 9,19,24,25 Ideally, MRI for the assessment of HD should be performed with 30-40 degrees of flexion. The anterior displacement of the posterior dura is due to a tight dural canal related to disproportionate length of the dural canal and vertebral column during flexion,3 explained by Chen et al.25
The spinal dura is a loose sheath anchored in the vertebral canal by nerve roots and attachment to the periosteum in two places – cranially at the foramen magnum and dorsal surfaces of C2 and C3, and caudally at the coccyx. The remainder of the dura is suspended and cushioned in the spinal canal by epidural fat, venous plexus, and loose connective tissues. In neck extension, the dural sac is slack and thrown into accordion-like transverse folds. With neck flexion, the dura becomes tighter. The difference in length from T1 to the top of the atlas between flexion and extension measures 1.5 cm along the anterior wall and 5 cm along the posterior wall.26 Normally, the slack of the dura can compensate for increased length in flexion. In HD, the dura is no longer slack in extension due to an imbalance in growth and cannot compensate for the increased length of the posterior wall with flexion. The result is anterior shift of the posterior dural wall with flexion that consequently compresses the cervical cord. Compression of the cord may cause microcirculatory disturbances in the anterior spinal artery territory or anterior portion of the spinal cord. Chronic vascular compromise may lead to necrosis of the anterior horns which are most vulnerable to ischemia.
The amount of forward shift of the posterior dural sac in flexion is critical because Lai et al. observed up to 46% of forward shift in healthy subjects though without spinal cord compression.27 Forward shift measured 1.0 to 4.2 mm with a mean of 1 mm in normal control patients, compared with a range of 6.1 to 7.8 mm, with a mean of 6.7 mm in HD patients. Dural shift is measured in the mid sagittal plane. In addition to the anterior displacement of the dura, on axial images the ratio of the anteroposterior size of the cord compared to the transverse dimension decreases with flexion in patients with HD, but not in normal controls.27,28
Contrast-enhanced images demonstrate an associated enhancing crescent-shaped epidural mass in the lower cervical canal containing small flow voids (Figure 4)28 that have been confirmed on venography. The enhancing epidural mass disappears on return to neutral position suggesting this represents congestion of the posterior internal vertebral venous plexus with flow voids indicating a dilated venous plexus. Several physiologic factors contribute to the dilatation of the venous plexus: 1) negative pressure from anterior shift of the dural canal increases flow to the internal vertebral venous plexus,8 2) the anteriorly displaced dural canal compresses the anterior internal vertebral venous plexus and increases burden on the posterior internal vertebral venous plexus,25 and 3) the posture of neck flexion decreases venous drainage from the internal vertebral venous plexus to the jugular veins. This combination of factors causes engorgement of the posterior internal vertebral venous plexus, a striking characteristic finding of HD.
Figure 4: Effect of neck flexion and intravenous contrast for cervical spine MRI in Hirayama Disease. In neutral position (4A), a sagittal T2-weighted image shows a normal appearance of the cervical cord. With neck flexion, (4B) a sagittal T2-weighted image shows an enlarged posterior epidural space with multiple flow voids (arrow), and (4C) a fat-suppressed sagittal T1-weighted image acquired after intravenous contrast administration shows an enhancing posterior epidural venous plexus (block arrow). (4D and 4E) Axial contrast-enhanced T1 fat-suppressed images with neck flexion demonstrate flow voids (4E, arrow) within the enhancing posterior epidural venous plexus, as well as asymmetric flattening of the right hemicord. Reproduced with permission from Boruah DK, Prakash A, Gogoi BB, Yadav RR, Dhingani DD, Sarma B. The Importance of Flexion MRI in Hirayama Disease with Special Reference to Laminodural Space Measurements. AJNR Am J Neuroradiol. 2018 May;39(5):974-980.
MRI findings in neutral position
Although the findings on flexion MRI are characteristic, findings on neutral position cervical MRI are equally, if not more important, to recognize in order to improve detection rate. Many clinicians are not familiar with HD and do not routinely request flexion cervical MRI exams. Several studies have suggested specific findings associated with HD on neutral position MRI or CT including: 1) localized lower cervical cord atrophy, 2) asymmetric cord flattening, 3) noncompressed intramedullary high signal intensity on T2-weighted images, and 4) abnormal cervical curvature (straight or kyphotic).9,19
Chen et al.29 investigated the sensitivity and specificity of these previously suggested neutral position MRI findings and a new proposed finding of loss of attachment (LOA) of the posterior dura from the subjacent lamina. The localized lower cord atrophy and asymmetric cord flattening can be appreciated on axial and sagittal MRI and is most commonly seen at the C4 to C7 levels. This finding is best assessed on axial images since sagittal images may result in erroneous assessment if the cord is not truly at midline. Additionally, comparing axial images to the normal level below is most reliable as the normal cord starts to enlarge at C3, reaches maximum dimension at C5, and tapers below T1. Localized cord atrophy had a sensitivity of 58.7% but specificity of 100% for HD and was not seen in the normal control group.
Asymmetric cord flattening is best assessed on axial T2-weighted images and is defined as cord flattening without narrowed or obliterated adjacent subarachnoid space to avoid confusion with compression due to adjacent spurs or herniated disc. An elliptical spinal cord is considered normal. A pear-shaped cord is seen with asymmetric cord flattening, and a triangular shaped spinal cord is considered symmetric cord flattening. Asymmetric cord flattening was found to have a sensitivity of 69.6% and specificity of 100% for HD.
Noncompressed intramedullary high signal on T2-weighted or STIR sequences occurs in short lengths of 1-3 spinal segments and is also commonly seen at the C4 to C7 levels, with a sensitivity of 28.3% and specificity of 96.1% for HD. Again, this finding is only considered if the surrounding subarachnoid space is patent because abnormal cord signal can be due to compression from adjacent spurs or herniated discs. The hyperintense signal is mainly localized to the anterior horns or anterior and lateral horns of gray matter indicating either ischemia or gliosis.
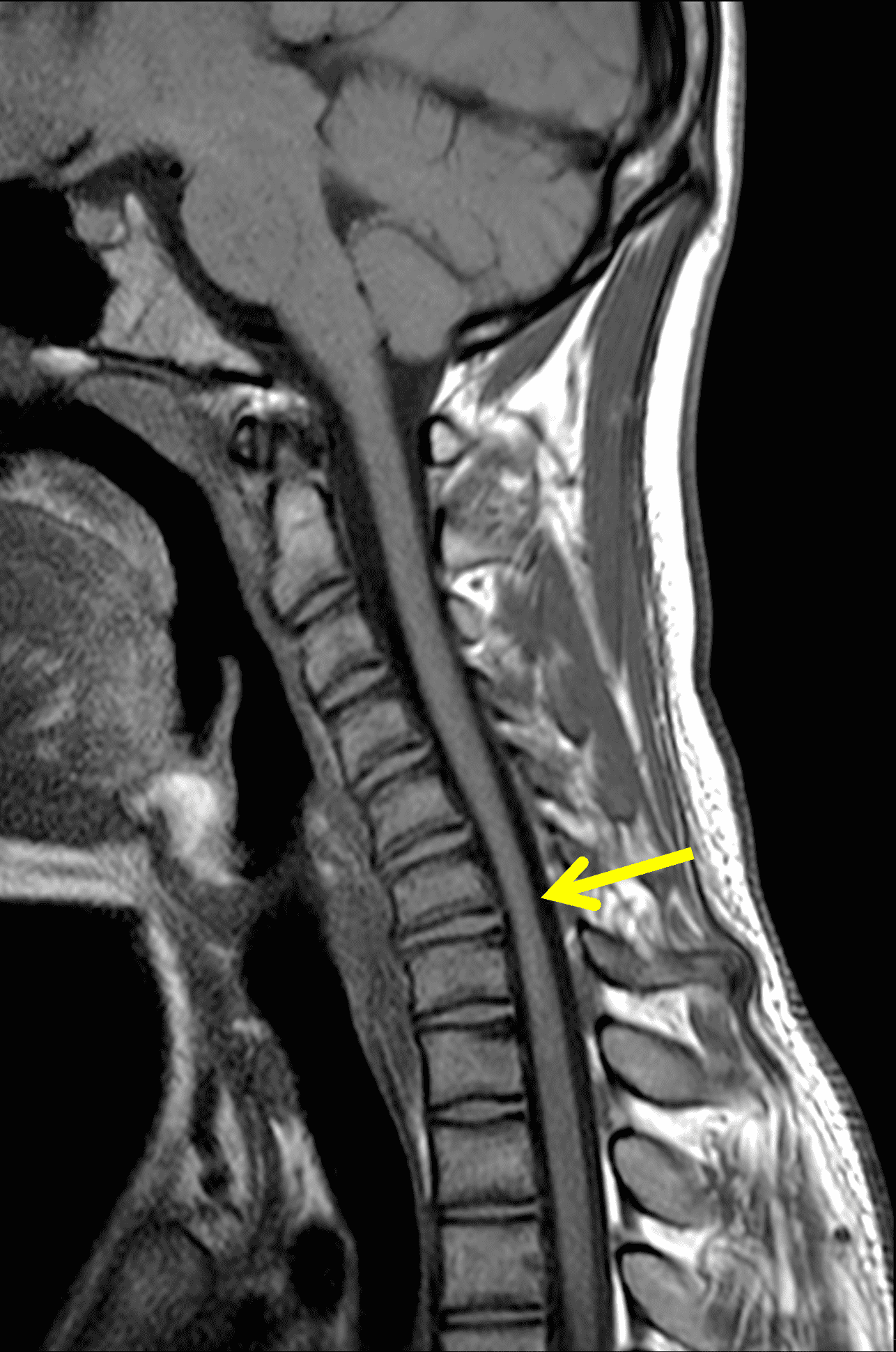
Loss of normal cervical lordosis with straight or kyphotic curvature is a nonspecific but common finding in HD.8,19,29 By definition, normal lordotic curvature is curvature where no part of the dorsal aspect of C3 through C6 vertebral bodies crosses a line drawn from the dorsocaudal aspect of the C2 vertebral body to the dorsocaudal aspect of the C7 vertebral body (Figure 5), based on the principles of Guigui et al.30 and Batzdorf et al.31 Abnormal curvature of the cervical spine demonstrates a sensitivity of 82.6% and specificity of 47.1%.29
Figure 5: Sagittal T2-weighted image of the HD patient in Figure 1 shows loss of normal cervical lordosis (kyphosis). The red line extends from the dorsocaudal C2 vertebral body to the dorsocaudal C7 vertebral body. The C5, C6 and a small portion of C7 vertebral bodies cross posterior to the line.
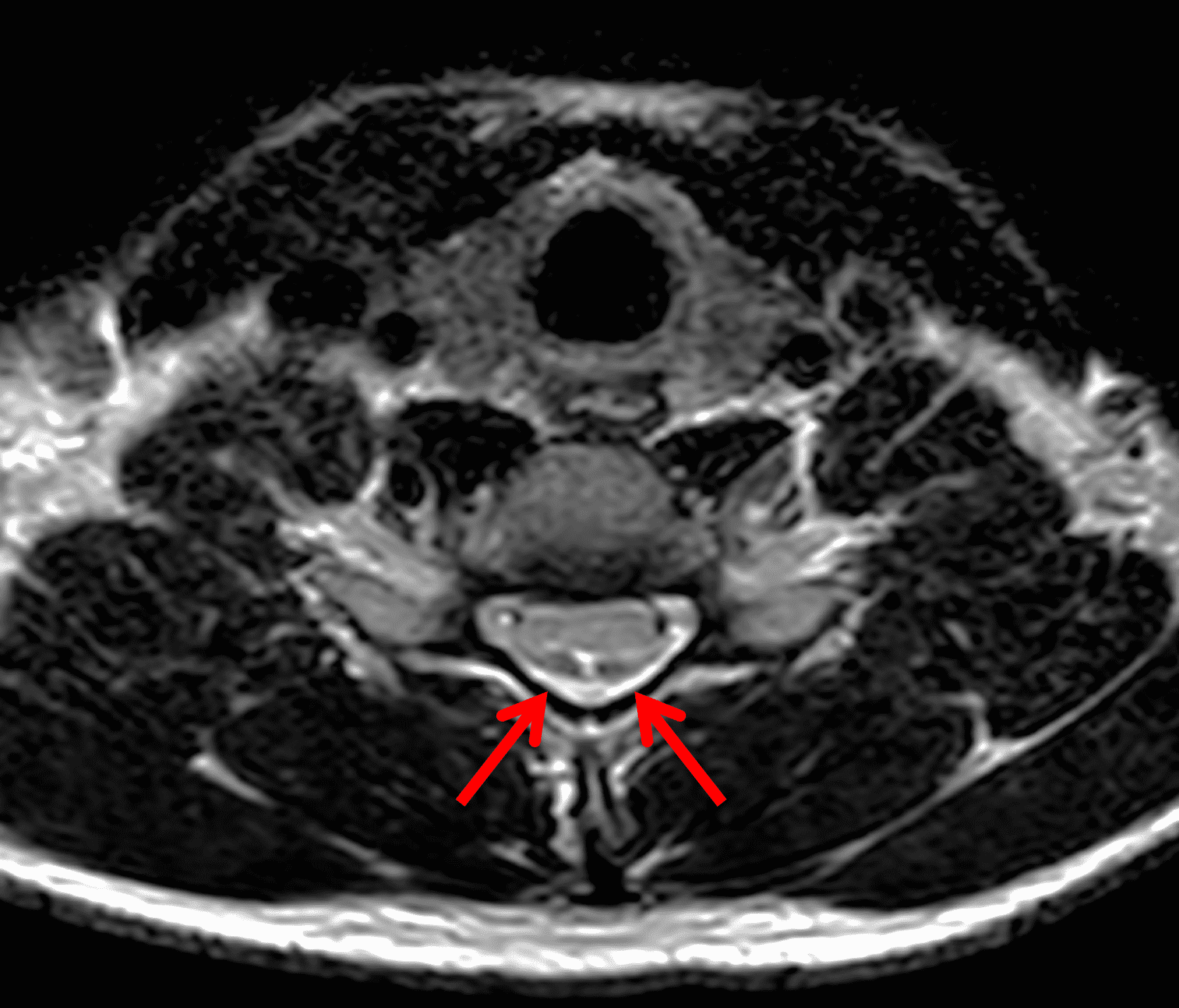
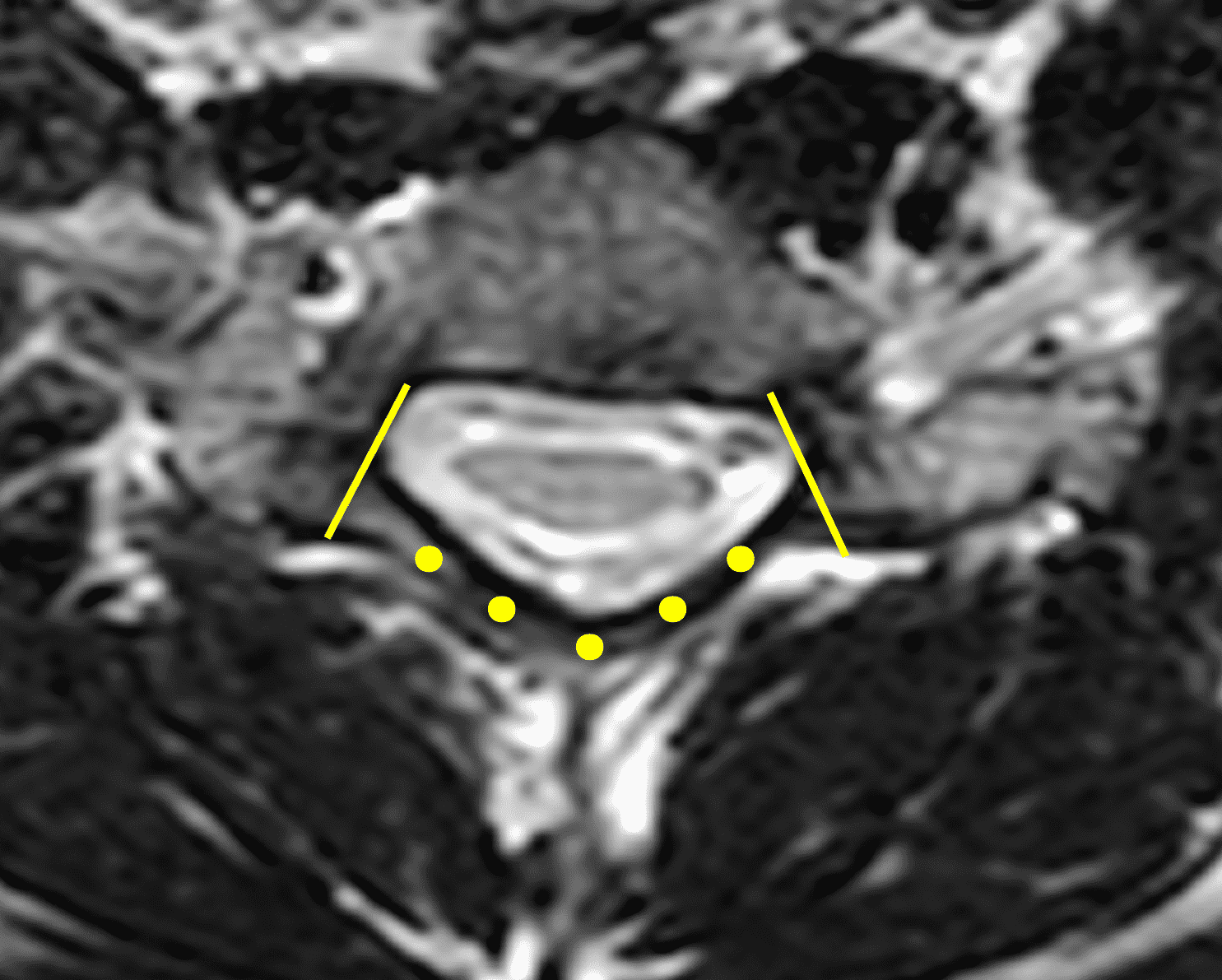
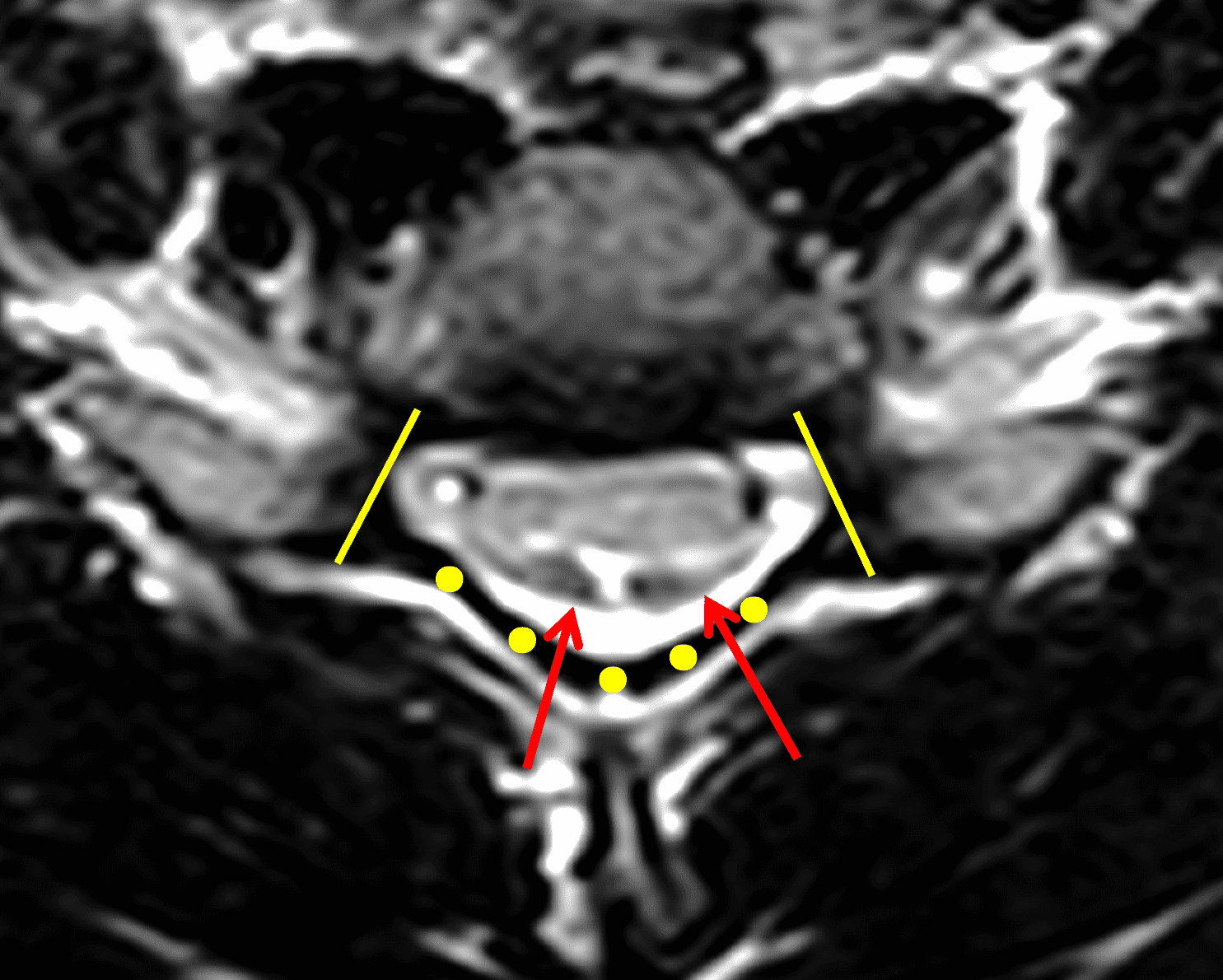
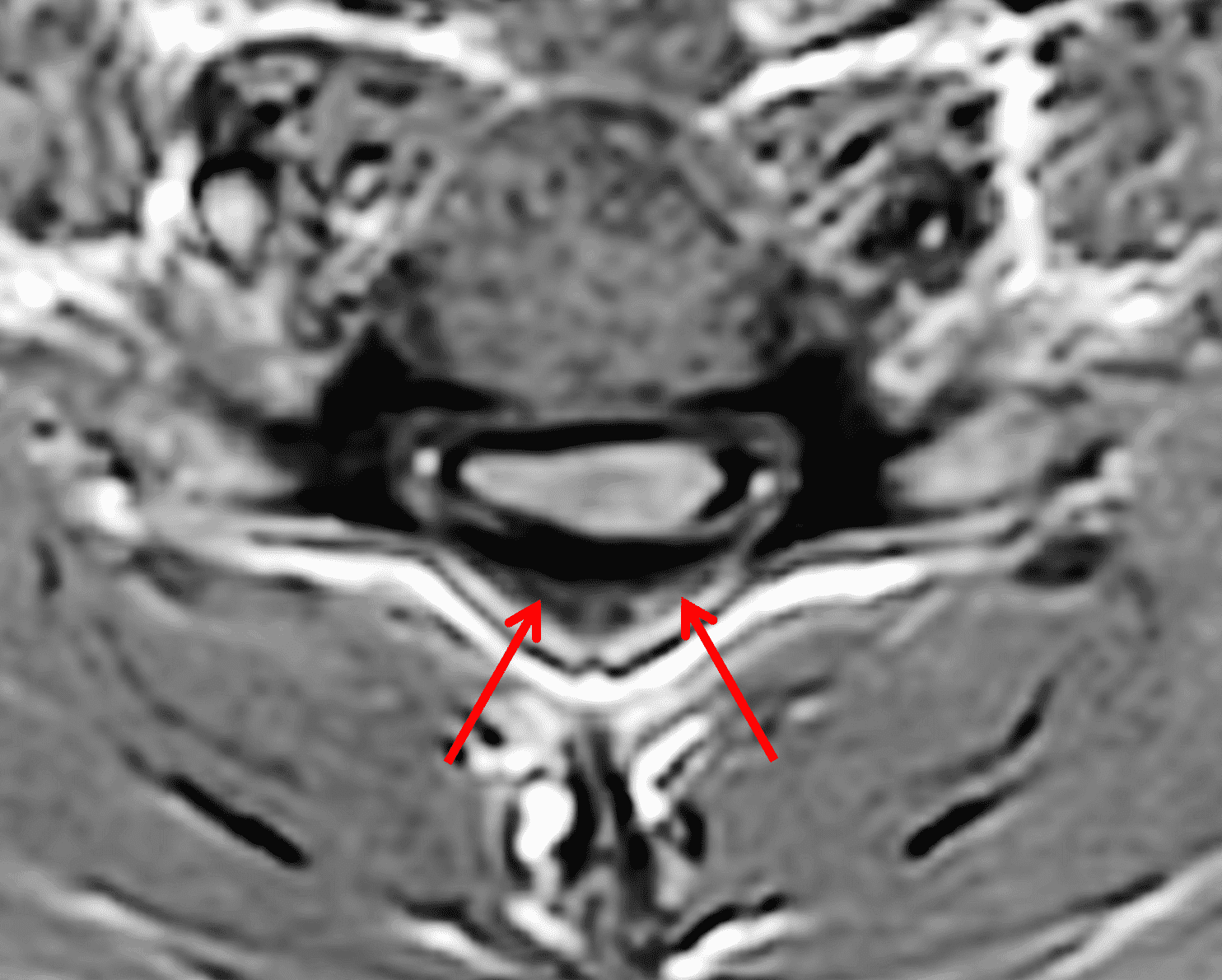
The LOA sign was proposed as a finding suggesting HD on neutral position MRI of the cervical spine.29 The degree of loss of attachment of the posterior dural sac and subjacent lamina are evaluated on each side of the lamina at C4 to C6 levels on axial T2-weighted images, with separation along more than one-third of the lamina on one or both sides considered positive for LOA. To assess the degree of LOA, the lamina is defined medially by the point of junction of the lamina and laterally by a tangential line along the medial aspect of the pedicle and then divided into three equal parts (Figure 6). The LOA sign was found to have a sensitivity of 93.5% and specificity of 98% for HD. The LOA sign was investigated in the first multisite North American study by Lehman et al.5 and found to have a lower sensitivity of 70% but similar specificity of 100%. The authors confirmed that the LOA sign and other findings are often present on neutral position MRI and in the appropriate clinical scenario, should raise suspicion and prompt further assessment by flexion MRI to confirm the diagnosis.
Figure 6: Axial images demonstrating the loss of attachment (LOA) sign. (6A) T2-weighted and (6B) T1-weighted images in a normal spine (same patient as Figure 3). Yellow lines mark the lateral extent of the laminae, which have been divided into thirds (yellow dots). The posterior dura (red arrows) is firmly attached to the lamina. (6C) T2-weighted and (6D) T1-weighted images of a spine with HD (same patient as Figure 1) show the lamina divided into three equal parts (yellow lines and dots) and displacement of the posterior dural sac, right greater than left, with loss of attachment (red arrows). The separation involves more than one-third of the length of the lamina on both sides.
Treatment
Early in the course of HD, the vagueness of the symptoms often contributes to a delay in diagnosis. Although the disease is self-limiting, prompt diagnosis is still important because early intervention can stop the disease progression.11 Conservative treatment with a cervical collar that prevents neck flexion is first line treatment and can shorten the duration of the disease, with some patients reporting increased grip strength and improvement in cold paresis. Patients are advised to wear the collar 24 hours a day for 3-4 years, but many find this intolerable.32 In a study of 73 patients, 80% wore the collar for less than 6 months.
In recent decades, surgery has led to improvement in both subjective and objective symptoms and now plays an important role in management. Indications for surgery could include: 1) progressive course and intolerance of a cervical collar, 2) ineffectiveness of conservative treatment, 3) serious symptoms at presentation, serious degree of spinal cord atrophy, cord atrophy in rare segments, and positive pyramidal signs.10 Various anterior and posterior operative approaches including laminectomy, duraplasty, corpectomy, discectomy, decompression and fusion have all been used for HD. A meta-analysis reviewing the surgical literature revealed that it is still unclear what type of surgery produces optimal results.33 Additionally, not all patients benefit from surgery and clinicians should exercise caution when assessing operative candidates.34
Conclusion
Hirayama disease is a rare benign disease mostly affecting males in their teens and twenties. It is characterized by a slow, progressive course typically involving unilateral or asymmetrically bilateral muscular weakness and atrophy of the distal upper extremity, followed by spontaneous arrest in 3-5 years. The disease is a distinct entity separate from degenerative motor neuron disease. The pathologic findings suggest a focal ischemic cervical myelopathy localized to the anterior horn. The complete pathogenesis is still unknown, but most researchers believe that it is related to a mismatch between the length of the spinal canal and cord compared with the spinal column that occurs related to a growth spurt. MRI findings with neck flexion include cord flattening due to forward displacement of the dural sac, and posterior epidural enhancement related to venous congestion. Asymmetric cord flattening, focal cord atrophy, loss of attachment (LOA) of the dura, cord signal abnormality, and abnormal curvature of the cervical spine on routine, neutral-position MRI are clues to the diagnosis and should prompt further investigation when the appropriate clinical findings are present. Conservative treatment with a cervical collar is the first line therapy. For patients who are unable to tolerate prolonged use of a cervical collar, or who experience short-term disease progression, surgery can be performed, but the optimal operative treatment has not yet been established. Future research could focus on better understanding of the disease and establish more effective treatments with optimization of the surgical approach.
References
- Hirayama K, Toyokura Y, Tsubaki T. Juvenile muscular atrophy of unilateral upper extremity: a new clinical entity. Psychiatric Neurol Jpn 1959;61:2190-2197. ↩
- Sobue I, Saito N, Iida M, Ando K. Juvenile type of distal and segmental muscular atrophy of upper extremities. Ann Neurol 1978;3:429-432. ↩
- Kikuchi S, Tashiro K, Kitagawa K, Iwasaki Y, Abe H. A mechanism of juvenile muscular atrophy localized in the hand and forearm (Hirayama’s disease): flexion myelopathy with tight dural canal in flexion 〈in Japanese〉. Clin Neurol (Tokyo) 1987;27:412-419. ↩
- Cerami C, Valentino F, Piccoli F, La Bella V. A cervical myelopathy with a Hirayama disease-like phenotype. Neurol Sci 2008;29:451-454. ↩
- Lehman VT, Luetmer PH, Sorenson EJ, et al. Cervical spine MR imaging findings of patients with Hirayama disease in North America: a multi-site study. AJNR Am J Neuroradiol 2013;34:451-456. ↩
- Hirayama K, Tomonaga M, Kitano K, et al. The first autopsy case of “juvenile muscular atrophy of unilateral upper extremity”. Shinkei Naika (Neurol Med) 1985;22:85–88. ↩
- Tokumaru Y, Hirayama K. Anterior shift of posterior lower cervical dura mater in patients with juvenile muscular atrophy of unilateral upper extremity. Rinsho Shinkeigaku 1989;29:1237–1243. ↩
- Mukai E, Sobue I, Muto T, et al. Abnormal radiological findings on juvenile-type distal and segmental muscular atrophy of upper extremities. Rinsho Shinkeigaku 1985;25:620–626. ↩
- Biondi A, Dormont D, Weitzner I Jr, et al. MR imagingof the cervical cord in juvenile amyotrophy of distal upper extremity. AJNR Am J Neuroradiol 1989;10:263–268. ↩
- Wang H, Tian Y, Wu J, Luo S, Zheng C, Sun C, Nie C, Xia X, Ma X, Lyu F, Jiang J, Wang H. Update on the Pathogenesis, Clinical Diagnosis, and Treatment of Hirayama Disease. Front Neurol 2022;12:811943. ↩
- Hirayama K. Juvenile muscular atrophy of distal upper extremity (Hirayama disease). Intern Med 2000;39:283–290. ↩
- Huang YC, Ro LS, Chang HS, et al. A clinical study of Hirayama disease in Taiwan. Muscle Nerve 2008; 37:576–582. ↩
- Zhou B, Chen L, Fan DS, et al. Clinical features of Hirayama disease in mainland China. Amyotroph Lateral Scler 2010;11:133–139. ↩
- Wang H, Zheng C, Jin X, Lyu F, Ma X, Xia X, et al. The Huashan diagnostic criteria and clinical classification of Hirayama disease. Chin J Orthop 2019; 39:458–465. ↩
- Tashiro K, Kikuchi S, Itoyama Y, et al. Nationwide survey of juvenile muscular atrophy of distal upper extremity (Hirayama disease) in Japan. Amyotroph Lateral Scler Other Motor Neuron Disord 2006;7: 38–45. ↩
- Atchayaram N, Vasudev MK, Goel G. Familial mono-melic amyotrophy (Hirayama disease): two brothers with classical flexion induced dynamic changes of the cervical dural sac. Neurol India 2009;57:810–812. ↩
- Huang YL, Chen CJ. Hirayama disease. Neuroimaging Clin N Am 2011;21(4):939-950. ↩
- Singh RJ, Preethish-Kumar V, Polavarapu K, Vengalil S, Prasad C, Nalini A. Reverse split hand syndrome: Dissociated intrinsic hand muscle atrophy pattern in Hirayama disease/brachial monomelic amyotrophy. Amyotroph Lateral Scler Frontotemporal Degener 2017;18(1-2):10-16. ↩
- Pradhan S. Bilaterally symmetric form of Hirayama disease. Neurology 2009;72:2083–2089. ↩
- Kijima M, Hirayama K, Nakajima Y. 〈Symptomatological and electrophysiological study on cold paresis in juvenile muscular atrophy of distal upper extremity (Hirayama’s disease)〉. Rinsho Shinkeigaku 2002;42(9):841-848. Japanese. ↩
- Ciceri EF, Chiapparini L, Erbetta A, Longhi L, Cicardi B, Milani N, Solero CL, Savoiardo M. Angiographically proven cervical venous engorgement: a possible concurrent cause in the pathophysiology of Hirayama’s myelopathy. Neurol Sci 2010;31(6):845-848. ↩
- Shao M, Yin J, Lu F, Zheng C, Wang H, Jiang J. The quantitative assessment of imaging features for the study of hirayama disease progression. Biomed Res Int 2015;2015:803148. ↩
- Shinomiya K, Dawson J, Spengler DM, et al. An analysis of the posterior epidural ligament role on the cervical spinal cord. Spine 1996;21:2081–2088. ↩
- Schröder R, Keller E, Flacke S, Schmidt S, Pohl C, Klockgether T, Schlegel U. MRI findings in Hirayama’s disease: flexion-induced cervical myelopathy or intrinsic motor neuron disease? J Neurol 1999;246(11):1069-1074. ↩
- Chen CJ, Chen CM, Wu CL, Ro LS, Chen ST, Lee TH. Hirayama disease: MR diagnosis. AJNR Am J Neuroradiol 1998;19(2):365-368. ↩
- Bland JH. Basic anatomy. In: Bland JH, ed. Disorders of the Cervical Spine: Diagnosis and Medical Management. 2nd ed. Philadelphia, Pa: Saunders; 1994:41–70. ↩
- Lai V, Wong YC, Poon WL, et al. Forward shifting of posterior dural sac during flexion cervical magnetic resonance imaging in Hirayama disease: an initial study on normal subjects compared to patients with Hirayama disease. Eur J Radiol 2011;80:724–728. ↩
- Boruah DK, Prakash A, Gogoi BB, Yadav RR, Dhingani DD, Sarma B. The Importance of Flexion MRI in Hirayama Disease with Special Reference to Laminodural Space Measurements. AJNR Am J Neuroradiol 2018;39(5):974-980. ↩
- Chen CJ, Hsu HL, Tseng YC, Lyu RK, Chen CM, Huang YC, Wang LJ, Wong YC, See LC. Hirayama flexion myelopathy: neutral-position MR imaging findings–importance of loss of attachment. Radiology 2004;231(1):39-44. ↩
- Guigui P, Benoist M, Deburge A. Spinal deformity and instability after multilevel cervical laminectomy for spondylotic myelopathy. Spine 1998; 23:440–447. ↩
- Batzdorf U, Batzdorff A. Analysis of cervical spine curvature in patients with cervical spondylosis. Neurosurgery 1988; 22:827–836. ↩
- Fu Y, Qin W, Sun QL, Fan DS. 〈Investigation of the compliance of cervical collar therapy in 73 patients with Hirayama disease〉. Zhonghua Yi Xue Za Zhi 2016;96(43):3485-3488. ↩
- Bohara S, Garg K, Mishra S, Tandon V, Chandra PS, Kale SS. Impact of various cervical surgical interventions in patients with Hirayama’s disease-a narrative review and meta-analysis. Neurosurg Rev 2021;44(6):3229-3247. ↩
- Zou F, Yang S, Lu F, Ma X, Xia X, Jiang J. Factors Affecting the Surgical Outcomes of Hirayama Disease: A Retrospective Analysis of Preoperative Magnetic Resonance Imaging Features of the Cervical Spine. World Neurosurg 2019;122:e296-e301. ↩