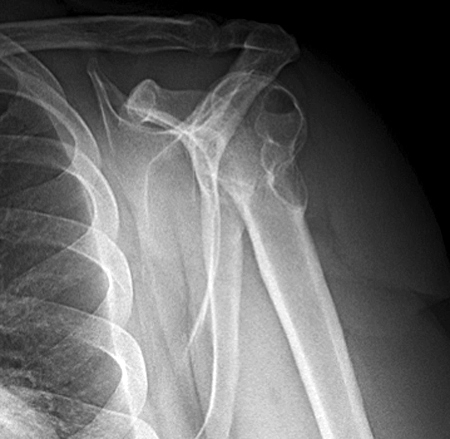
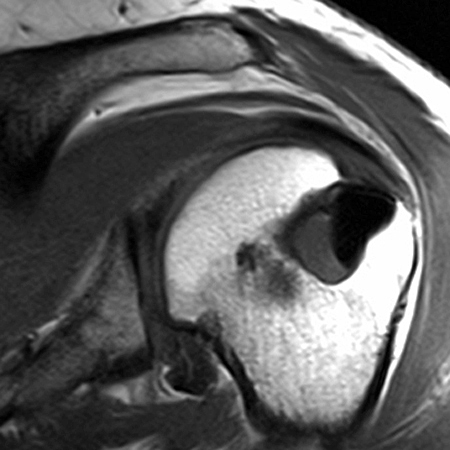
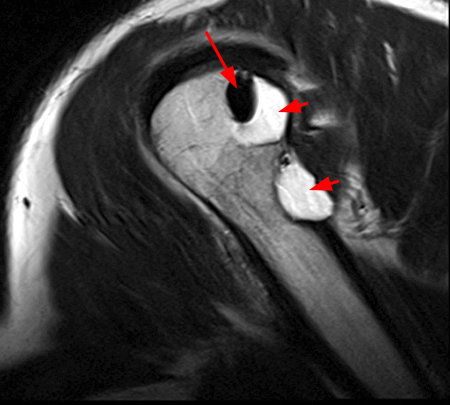
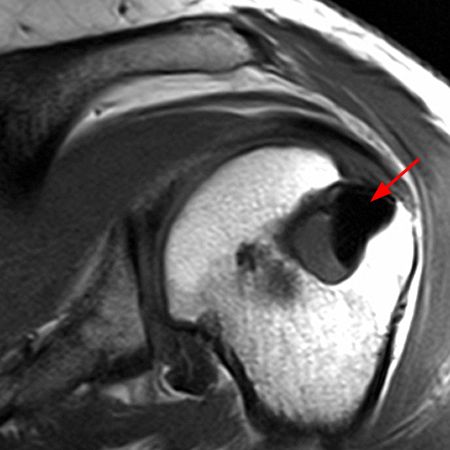
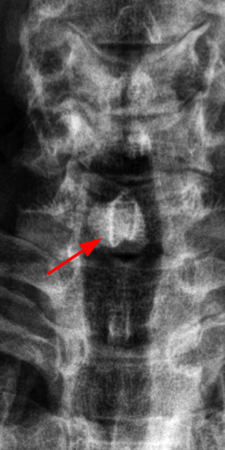
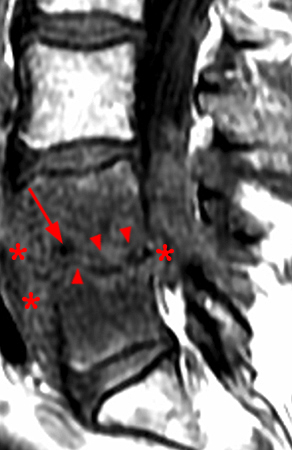
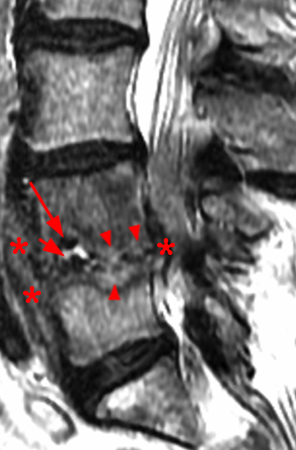
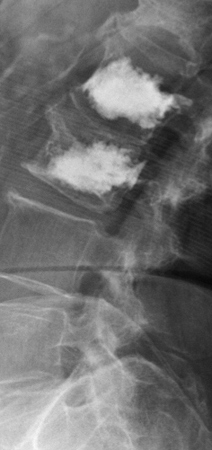
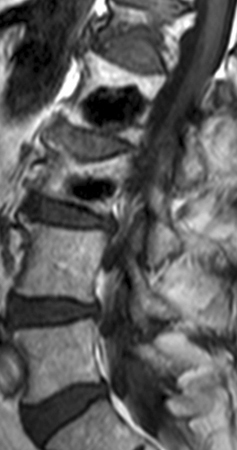
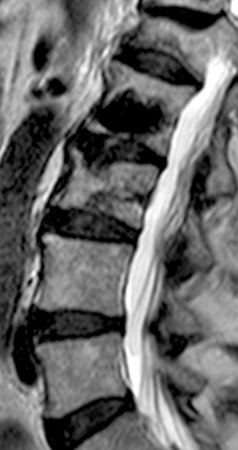
Clinical History: A 50 year-old man was involved in a motor vehicle accident with complaints of shoulder and neck pain. What are the findings? What is your diagnosis?
Findings
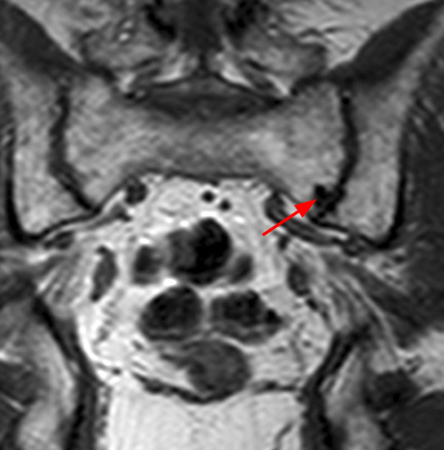
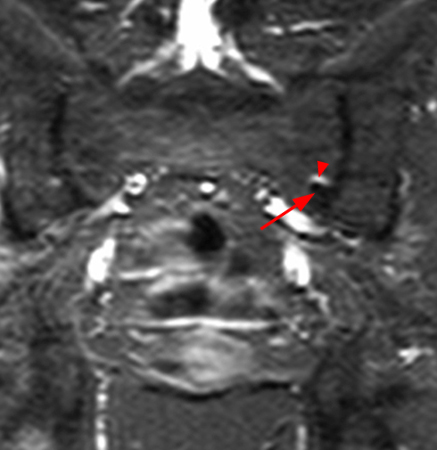
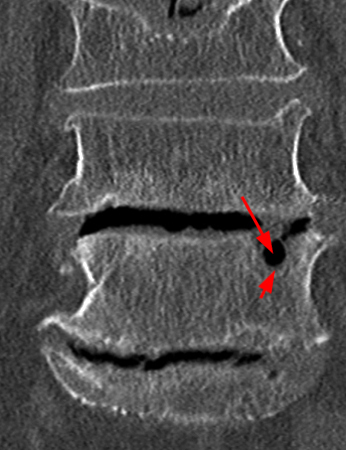
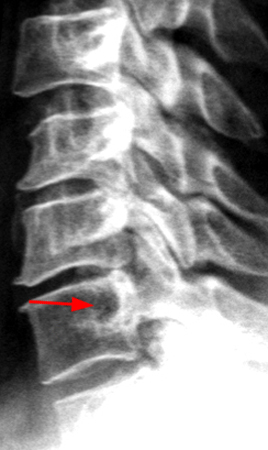
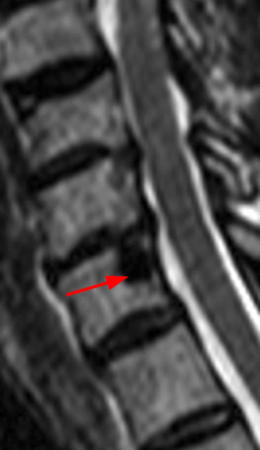
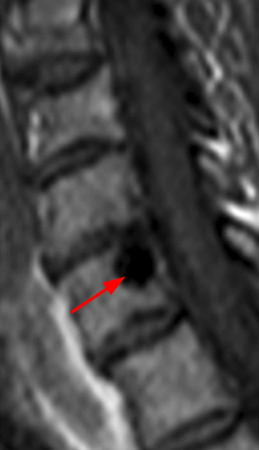
Figure 2:
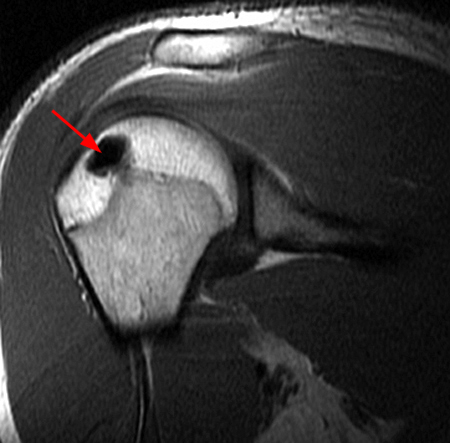
The x-ray (2a) reveals a large, predominantly lytic, non-destructive lesion of the proximal left humeral head and neck with a well-defined sclerotic rim (arrowheads) and narrow zone of transition. The lesion is multilobular and expansile with a more lucent focus at its superior aspect (arrow). The MR images (2b, 2c) demonstrate a cystic lesion (short arrows) with a superior low signal focus (arrow), suggestive of an intracystic focus of air.
Diagnosis
Intraosseous pneumatocyst of the proximal left humerus, associated with an intraosseous ganglion.
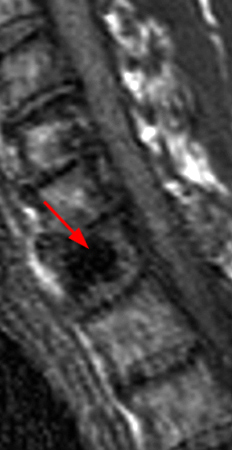
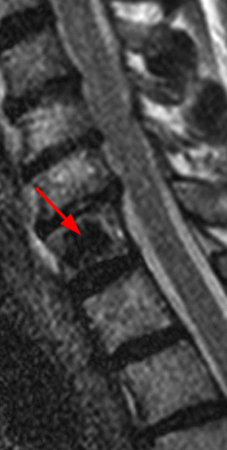
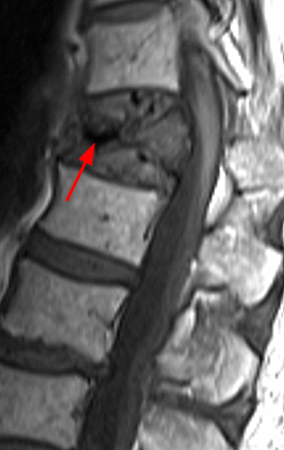
Figure 3:
A second case of a pneumatocyst within the proximal humeral epiphysis in a 16-year old girl who presented with left shoulder pain and possible growth plate injury. The sagittal T1 (3a) and T2-weighted (3b) coronal oblique images through the shoulder reveal a gas-filled lesion demonstrating low signal (arrows) abutting the proximal physis of the humerus. There is a small area of cystic signal present at the distal aspect of the lesion (arrowheads).
Introduction
Intraosseous pneumatocysts are acquired benign lesions. The term was first used in 19841 to describe gaseous lesions within bone. These abnormalities occur fairly often in adults with an incidence of 9 percent2 and are rarely encountered in children. They are classically first imaged by X-ray and subsequently confirmed by CT or MR imaging. These cysts are most commonly found within the vertebral bodies, iliac bones or sacrum. 3,4 More rarely these have been described in other osseous locations such as the humerus and clavicle.5 These lesions are almost always asymptomatic, although there have been reports of pain associated with barotrauma.6 The lesions can enlarge in size.7 There is no determined etiology for these cysts; however, several mechanisms have been postulated.
Proposed Mechanism of Formation
There is no clearly defined cause of pneumatocysts. There may in fact be different etiologies for the presence of air in these lesions depending upon the lesion location, patient age, and presence or absence of degeneration in proximity to the cysts. The “vacuum phenomenon” term may be a misnomer in the great majority of cases as this term implies gas forming from an acute volumetric increase in a space. This has been most recently shown by a team of researchers providing in-vivo imaging proof of the phenomenon of tribonucleation, which refers to gas formation through the action of making and breaking contact between solid surfaces. Utilizing MRI, the study revealed pulling on the knuckles creates a gas cavity at a point when the opposing articular surfaces can no longer avoid separation.8 This effective increase in the volumetric space within the joint causes the dissolved gas (composed mostly of nitrogen within the joint lubricant) to be released, subsequently forming a gas bubble.
In most instances of pneumatocyst there is no acuity and often there is degeneration adjacent to the gas. The reasons for gas formation are multifactorial and can depend upon the duration and intensity of the cavity opening related to intraarticular pressure changes, the permeability of the cavity walls, and the composition of the adjacent tissues. The mechanism for the “vacuum” is thought to be from erosive bone lesions sustaining chronic microtrauma with subsequent migration of gas. For example, in the spine it is thought that chronic stress leads to erosive defects in the osteocartilaginous endplates and extension of the gas into the vertebral body due to migration of the gas from the disc9(Figure 4). In the two shoulder cases above (Figures 1-2, Figure 3), a more appropriate mechanism may be spontaneous involution of mucoid cysts or intraosseous ganglions, hence being replaced by gas in more of a true “vacuum” scenario.
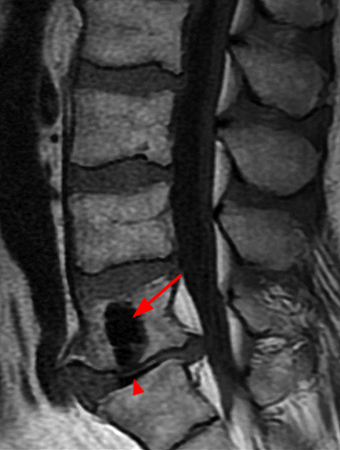
Figure 4:
Sagittal T1- (4a) and T2-weighted (4b) images obtained in a 52 year-old male with low back pain and radiculopathy. There is disc degeneration at L5-S1 with loss of height of the disc, anterior osteophytes, and extrusion of the disc posteriorly. A “vacuum” cleft within the L5-S1 intervertebral disc (arrowhead) and an adjacent large L5 vertebral body pneumatocyst (red arrow) are present. A small gas-fluid level is seen posteriorly (short arrow).
Imaging
Intraosseous pneumatocysts can be appreciated on all imaging modalities. X-rays are often the first study to identify the presence of these lesions resulting in further work-up and confirmation by MRI or CT scanning. The reason for obtaining further imaging is that these rounded and juxta-articular lucent/lytic lesions on radiographs sometimes have a thin sclerotic rim and can simulate more aggressive and sometimes malignant lesions such as metastases and multiple myeloma.10
On CT, pneumatocysts are often incidentally discovered and will contain air, visualized as extremely low density with Hounsfield units, typically ranging from -100 to -1000. The finding of gas in these lesions is pathognomonic for a benign pneumatocyst. The air-filled cavities do not demonstrate enhancement but do sometimes demonstrate communication with the adjacent joint space (Figure 5).
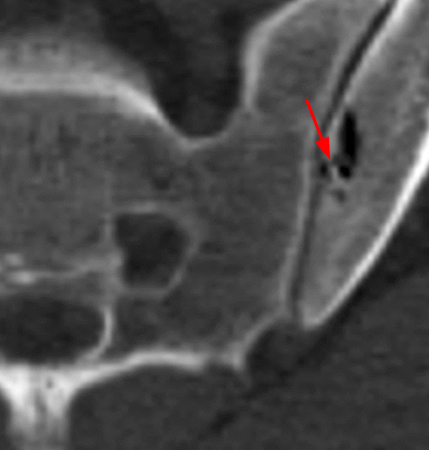
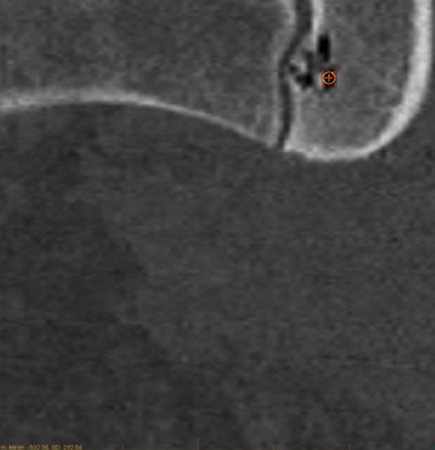
Figure 5:
An axial thin slice (5a) CT image through the pelvis demonstrates an ovoid area of low attenuation within the left iliac bone adjacent to the left sacroiliac joint in a young patient. A small communicating cleft (arrow) to the joint is present. The Hounsfield measurement is -532 (5b), consistent with air within a pneumatocyst.
Air has a characteristic but nonspecific appearance on MRI. T1- and T2-weighted sequences reveal signal void without enhancement (Figure 6).11 Markedly sclerotic benign (Figure 7) and malignant lesions can also have the same appearance of low signal on MRI which is why correlation with the x-ray may be necessary in these circumstances to exclude the differential consideration of osteoblastic metastases.12
Figure 6:
Coronal T1- (6a) and fat suppressed T2-weighted (6b) images obtained through the pelvis reveal the presence of a juxta-articular low signal lesion on both images at the level of the inferior left sacrum (red arrows) consistent with a pneumatocyst. A curvilinear area of high signal along the superior margin in image 6b is likely due to artifact from local magnetic susceptibility differences at the air-bone interface.
Figure 7:
Sagittal T1 (7a) and T2 (7b) weighted MRI images through the cervical spine reveal a low signal lesion (red arrow) centered within the C7 vertebral body without aggressive features. This finding is nonspecific on MRI alone; however, the lesion appears sclerotic on the corresponding AP radiograph (7c). This is a large enostosis (bone island).
Pneumatocysts are most commonly identified at the sacroiliac joints (roughly 2/3 of the time) and mostly along the iliac side of the joint. Interestingly, it has been found that these cysts occur mostly in men. Roughly 1/4 of the pneumatocysts are seen within the spine, mostly within the vertebral bodies and less commonly within the posterior elements and articular processes. Uncommon places for these lesions to occur include the scapula, clavicle, humerus, cervical rib, pubis, and the acetabulum.13
Differential Diagnosis
Osteomyelitis
Bone infection may result in gas within a bone, but is easily distinguished from benign pneumatocysts both clinically and radiographically. Most of the time patients present with pain and fever, and usually have elevated sedimentation rates. Imaging features are more aggressive with osteomyelitis, and can be differentiated by the presence of cortical irregularity with bone destruction, signal changes on MRI in the surrounding bone, and secondary signs within adjacent soft tissue structures (Figure 8).
Figure 8:
Sagittal T1- (8a) and T2-weighted (8b) images in a 57 year-old female with recent surgery presenting with back pain. There are characteristic findings of irregularity of the endplates with vertebral destruction (arrowheads), high-signal within the disc on the T2 sequence from discitis (short arrow), and paraspinous and epidural extension of the infectious process (asterisks). Note the small focus of low signal along the inferior L4 vertebral enplate (arrow), signifying development of air likely from a gas-producing infectious organism.
Osteonecrosis
Osteonecrosis, also called avascular necrosis and bone infarction, results from the death of bone. The causes are multifactorial including vascular, infectious, autoimmune, metabolic, iatrogenic, traumatic, congenital, and infectious. Decreased blood flow to the bone in later stages may cause the bone to collapse. Air at the site of the infarction is unusual, except in cases of vertebral osteonecrosis. In these instances it is thought that the collapse of the vertebral endplate produces ischemia to the bone, often eliciting the “intravertebral cleft” (Figure 9) or “fluid cleft” signs14, both distinguishing this entity from pneumatocyst formation by their more linear configuration and the obligatory association with endplate fracture.
Figure 9:
A 76 year-old with recent lifting injury demostrating a subacute posttraumatic burst fracture of T12. Sagittal T1- (9a) and T2-weighted (9b) images reveal the presence of a linear area of low signal (red arrow) suggestive of air within the collapsed and edematous vertebral body. This is known as the intervertebral cleft sign of vertebral osteonecrosis.
Subchondral cysts
These fluid-filled cysts, also called geodes, occur in the setting of osteoarthritis from loss of overlying cartilage and are predominantly compromised of hyaluronic acid. These lesions may simulate the radiolucent characteristics of pneumatocysts on radiographs, but are easily distinguishable on MR imaging due to the lack of air within the cysts.
Schmorl’s nodes
Schmorl’s nodes result from degeneration of the vertebral endplate and subsequent herniation of the intervertebral disc material into the endplate defect. Typically these herniations result in contour deformity of the vertebral endplate, are not usually completely air-filled, and contain disc material (Figure 10), all features assisting in differentiating from a pneumatocyst. The well-defined margins of the pneumatocyst are seen in the next example for comparison. (Figure 11)
Figure 10:
Coronal (10a) and sagittal (10b) 2D reconstructed CT images in a 64 year old man with low back pain. The CT images demonstrate severe vacuum phenomenon within the L4-5 intervertebral disc communicating with a large partially air-filled Schmorl’s node (arrow) along the superior L5 endplate. Note the cortical depressed margin and the soft tissue density (short arrow) within the cortical defect related to herniated disc material.
Figure 11:
54 year-old female in a motor vehicle accident with mid back numbness. This well circumscribed lucent lesion on sagittal radiograph (13a) within the C7 vertebral body exhibits low signal on T1 (13b) and T2-weighted (13c) images. This is most likely a pneumatocyst given the fairly regular margins and lack of significant endplate contour deformity.
Vertebroplasty/Kyphoplasty
Vertebroplasty and kyphoplasty are two procedures typically used to treat compression and pathologic fractures in the clinical setting of pain. The introduction of bone cement into the vertebral body produces a sclerotic appearance on radiographs and CT, distinguishing this from the lucent appearance of air within pneumatocysts. MR imaging characteristics are similar to those with pneumatocysts; however, a clue to differentiation can often be the tracking of the cement along the course of the procedure posteriorly towards the pedicles.
Figure 12:
Sagittal X-ray (12a), T1- (12b) and T2-weighted (12c) sagittal MR images through the lumbar spine in this 64 year-old patient reveal evidence of multilevel compression fractures treated by kyphoplasty. The radiograph shows ovoid-like density at the L2 and L3 levels within the vertebral bodies indicative of methyl methacrylate cement injection. The corresponding low signal intensity on T1- and T2- weighted images has a similar appearance to pneumatocysts.
Treatment and Prognosis
Intraosseous pneumatocysts are benign lesions that do not require additional workup. Biopsies of these lesions have been performed previously, revealing nonspecific fibrous capsules, sometimes with a myxoid stroma that permits a pathological diagnosis of a ganglion cyst.15 Most of these lesions are discovered incidentally and it is important to recognize the nonaggressive nature of these lesions such that unnecessary treatment does not occur.
Conclusion
Intraosseous pneumatocysts are benign, gas-filled lesions of uncertain etiology, though typically they are associated with degenerative phenomena. Other conditions such as osteomyelitis or osteonecrosis may result in gas within bone, but the imaging findings on MRI allow differentiation of these entities from a simple pneumatocyst. Most pneumatocysts are incidental findings and knowledge of the typical appearance allows an accurate diagnosis to be made and unnecessary treatment can be avoided.
References
- Ramirez H, Blatt ES, Cable HF, McComb BL, Zornoza J, Hibri NS. Intraosseous Pneumatocyst of the ilium findings on radiographs and CT scans. Radiology. 1984;150:503–505. ↩
- Arslan G, Ceken K, Cubuk M et al. Vertebral pneumatocysts. Acta Radiologica, 2001, 42: 20-23. ↩
- Catalano O, De Rosa F, Muto M. Intraosseous pneumatocyst of the ilium: CT findings in two cases and literature review. Eur Radiol. 1997;7: 1449-1451. ↩
- Berenguer J, Pomes J, Bargalle N. Sacral Pneumatocysts:CT appearance. J Comput Assist Tomogr. 1994;18(1):95-97. ↩
- Linker CS, Peterfy CG, Helms CA. Case report 844. Intraosseous pneumatocyst of the clavicle. Skeletal Radiol. 1994;23:315-316 ↩
- Hart BL, Brantly PR, Lubbers PR et al. Compression pain in a diver with Intraosseous pneumatocysts. Undersea Biomedical Research. 1986;13(4):465-468. ↩
- Yamamoto T, Yoshiya S, Kurosaka M, et al. Natural course of an intraosseous pneumatocyst of the cervical spine. AJR Am J Roentgenol. 2002;179:667–669 ↩
- Kawchuk GN, Fryer J, Jaremko JL, Zeng H, Rowe L, Thompson R (2015) Real-Time Visualization of Joint Cavitation. PLoS ONE 10(4): e0119470. doi:10.1371/journal.pone.0119470 ↩
- Coulier B (2004) The spectrum of vacuum phenomenon and gas in spine. Jbr-Btr 87(1):9-16 ↩
- Husain MA, Tetradis S, Mallya SM. Intraosseous pneumatocysts of the cervical spine: a report of four cases and review of literature. Oral Surg Oral Med Oral Pathol Oral Radiol. 2015 Jan;119(1):e49-54.10.1016/j.oooo.2014.09.019. Epub 2014 Nov 4 ↩
- Cosar M, Eser O, Aslan A, et al. Vertebral body pneumatocyst in the cervical spine and review of the literature. Turk Neurosurg. 2008;18:197-199 ↩
- Haithcock JA, Layton KF, Opatowsky MJ. Vertebral pneumatocysts: uncommon lesions with pathognomonic imaging characteristics. Proc (Bayl Univ Med Cent). 2006;19:423-424. ↩
- Ochler, E., Valour, F., and Pascart, T. Intraosseous pneumatocysts: a case based review. Clin Rheumatol. 2013; 32: 129–134 ↩
- Yu CW, Hsu CY, Shih TT et al. Vertebral osteonecrosis: MR imaging findings and related changes on adjacent levels. AJNR, 2007,28:42-47. ↩
- Ramirez H, Blatt ES, Cable HF, McComb BL, Zornoza J, Hibri NS. Intraosseous Pneumatocyst of the ilium findings on radiographs and CT scans. Radiology. 1984;150:503–505. ↩