Clinical History
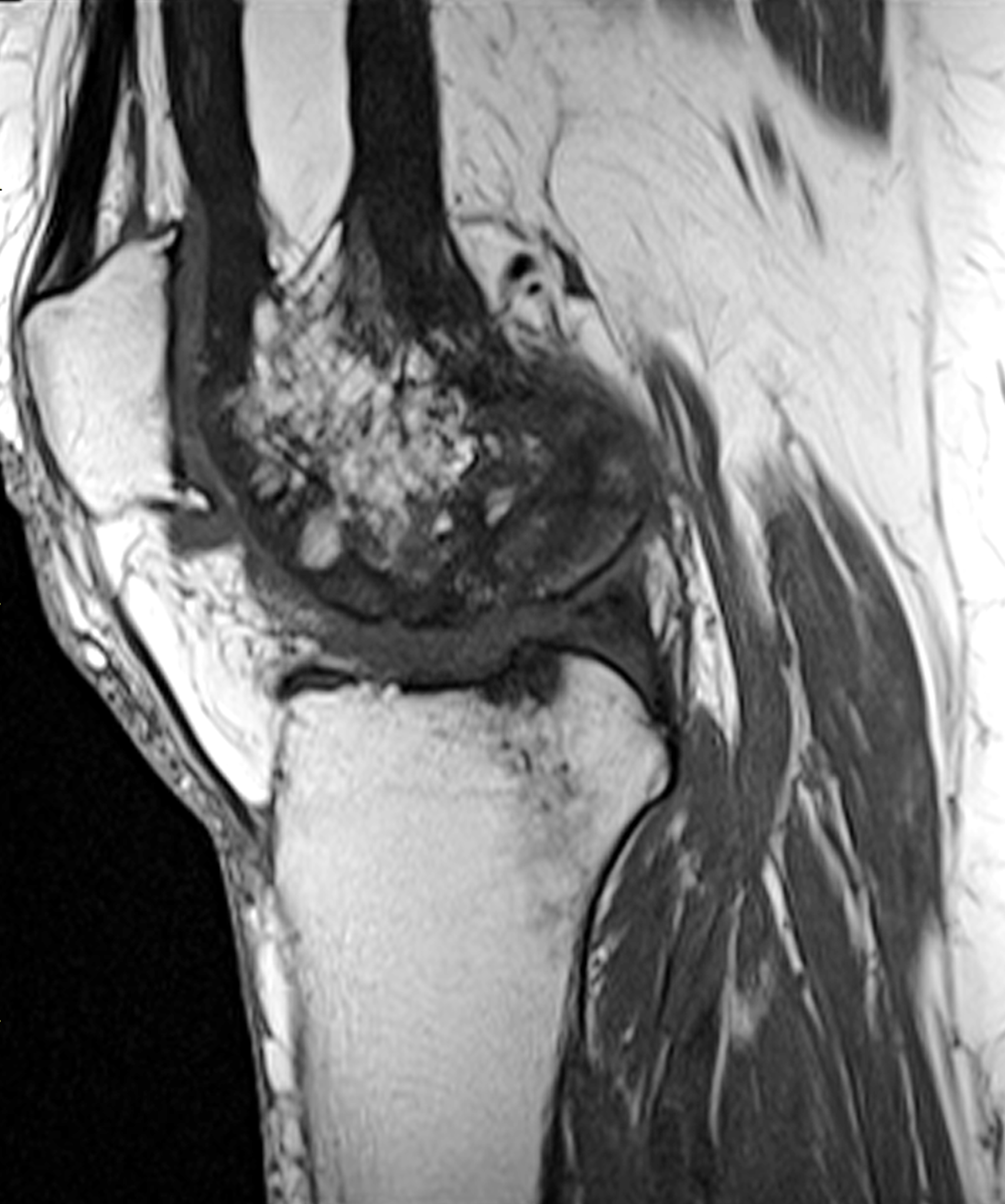
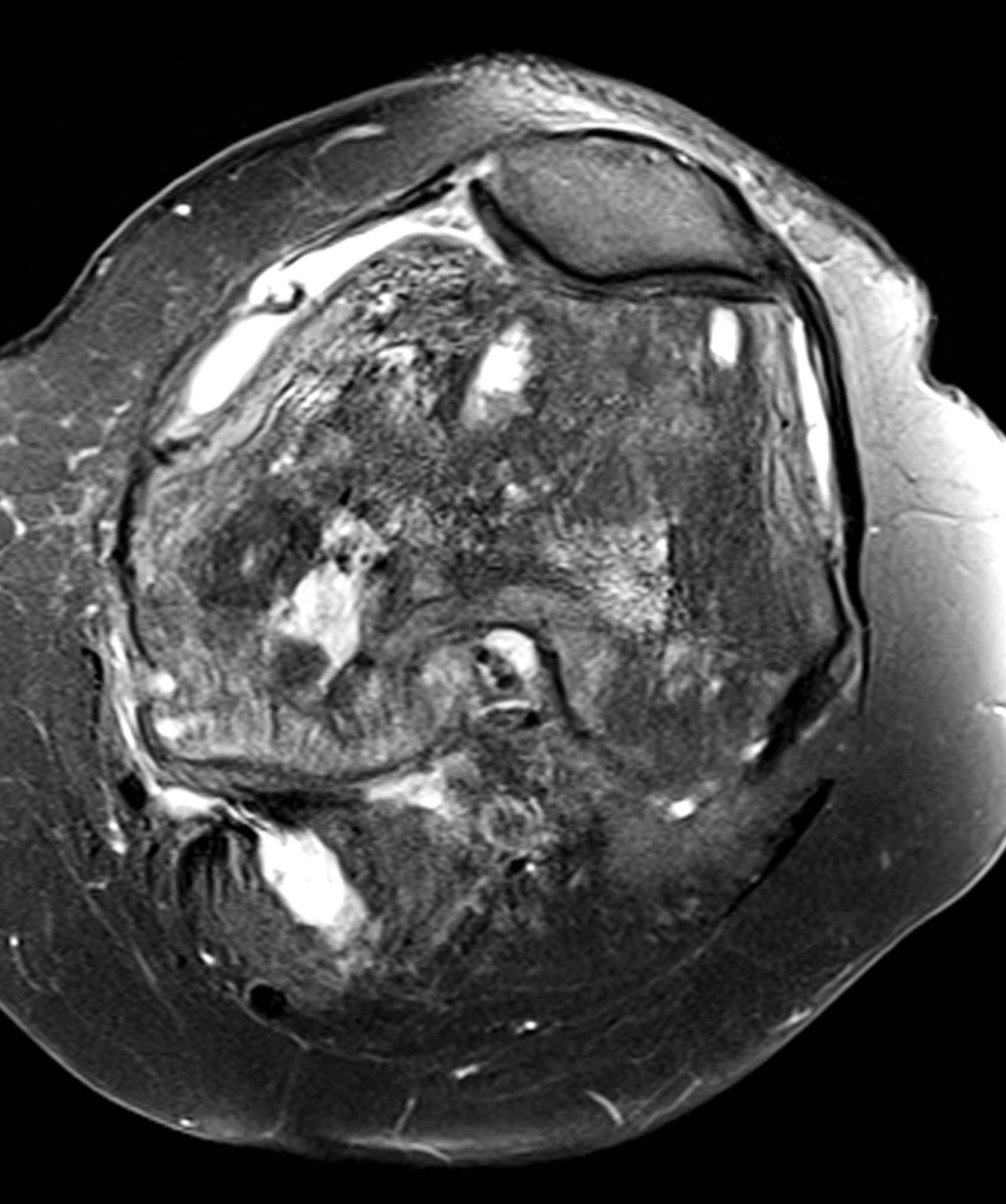
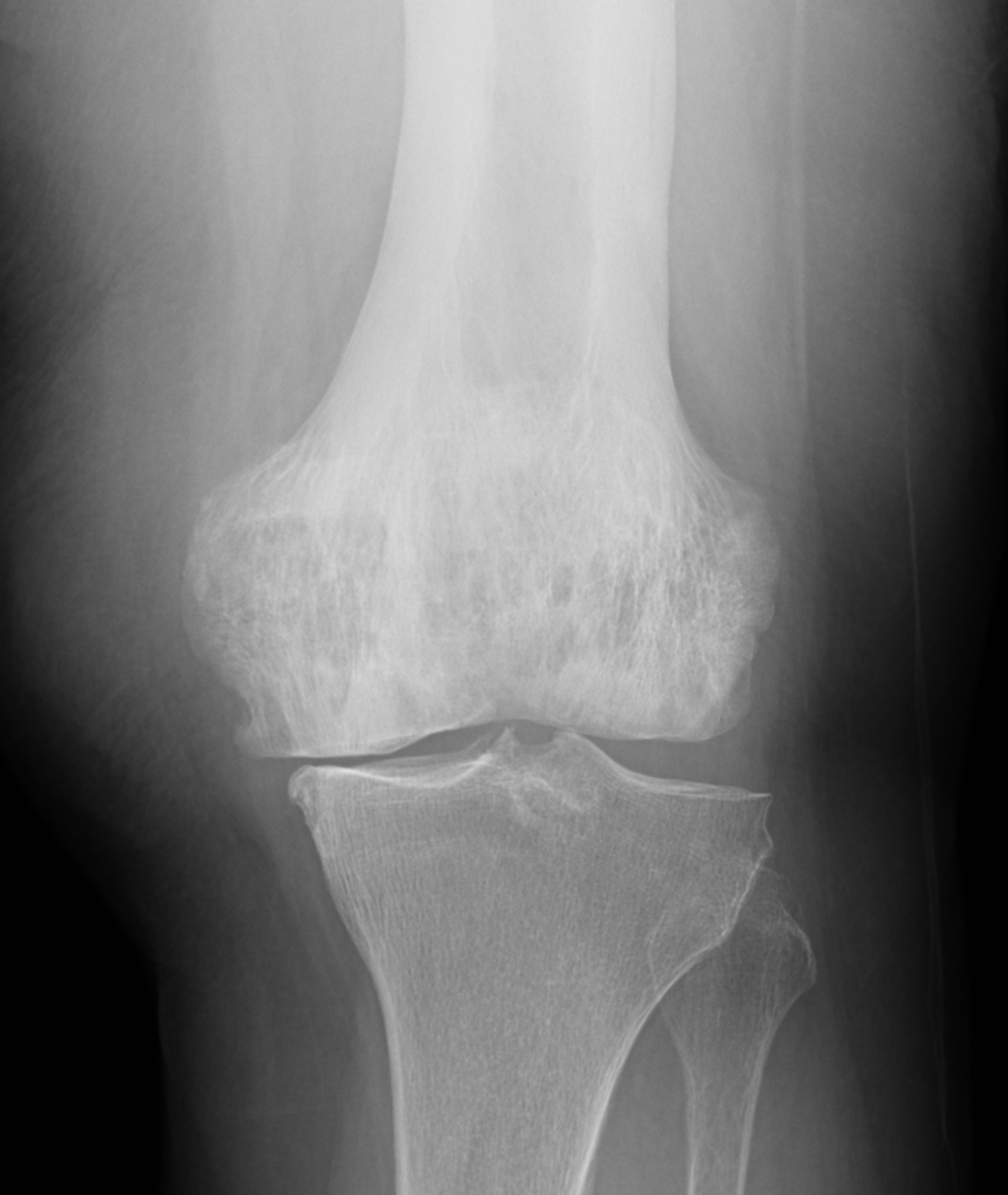
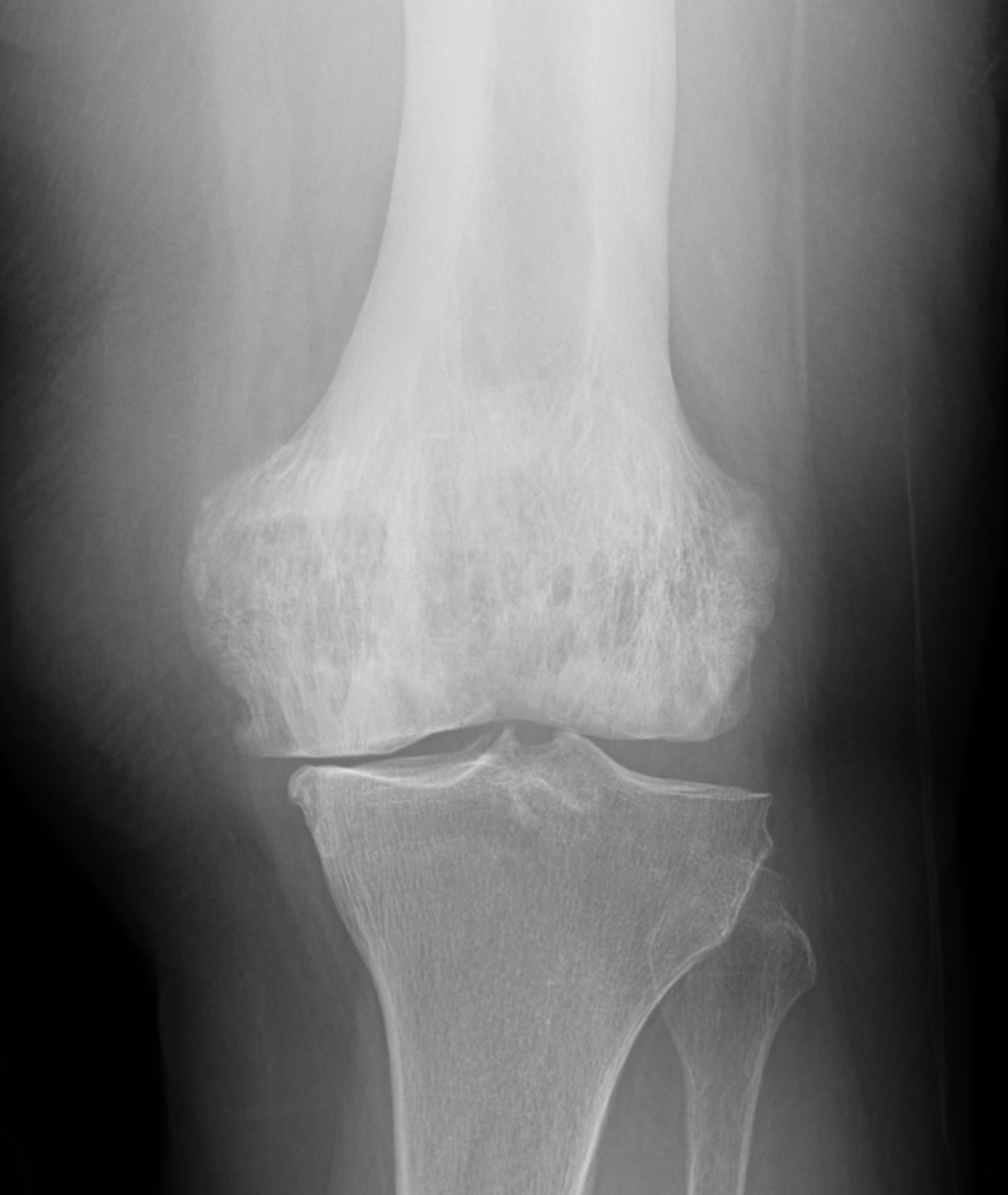
An elderly man presents with generalized knee pain. A sagittal T1-weighted image (1A) of the knee, axial fat-suppressed, fluid sensitive images through the distal femoral shaft (1B) and femoral condyles (1C), and an AP radiograph (1D) of the knee are provided. What are the findings? What is your diagnosis?
Findings
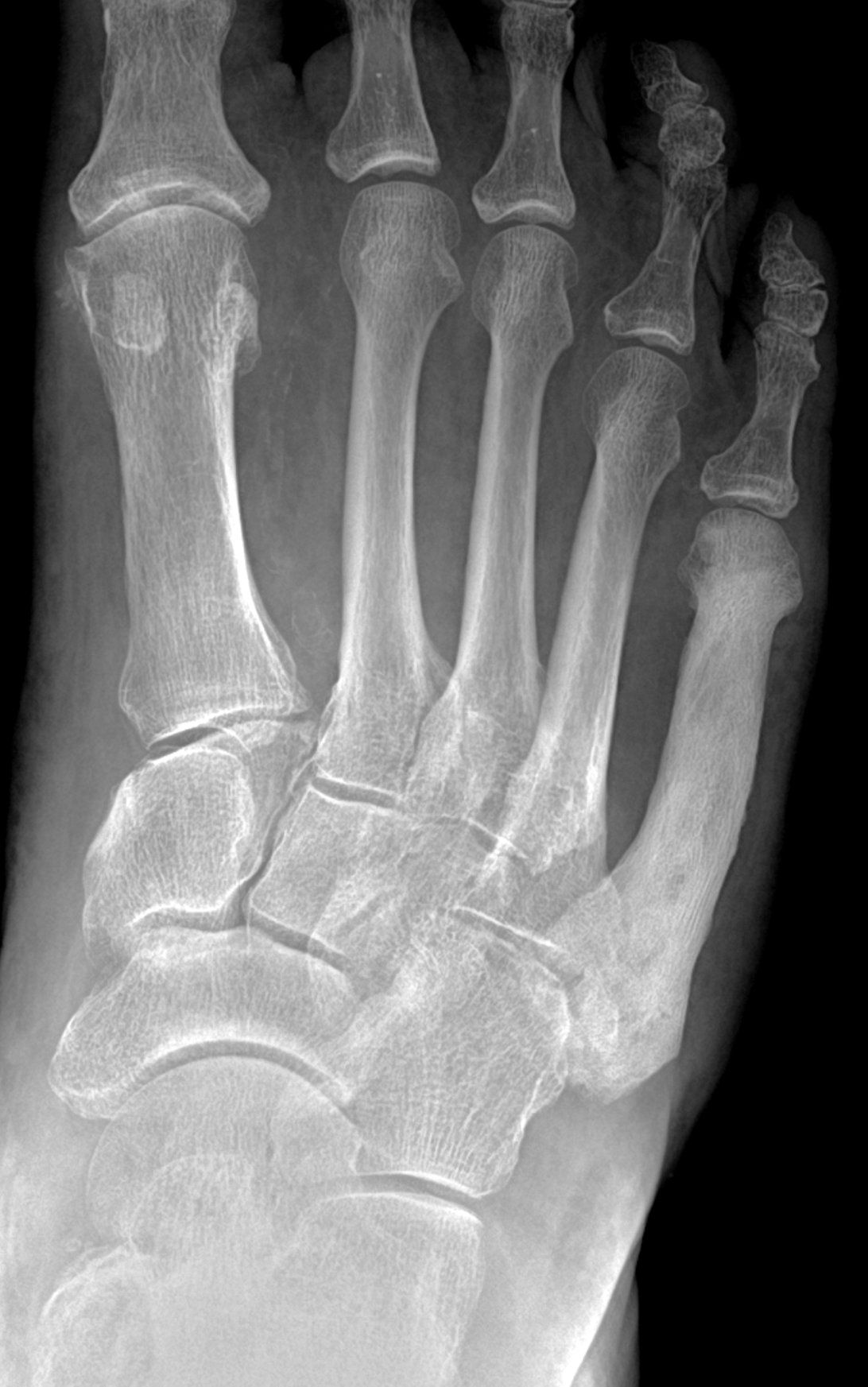
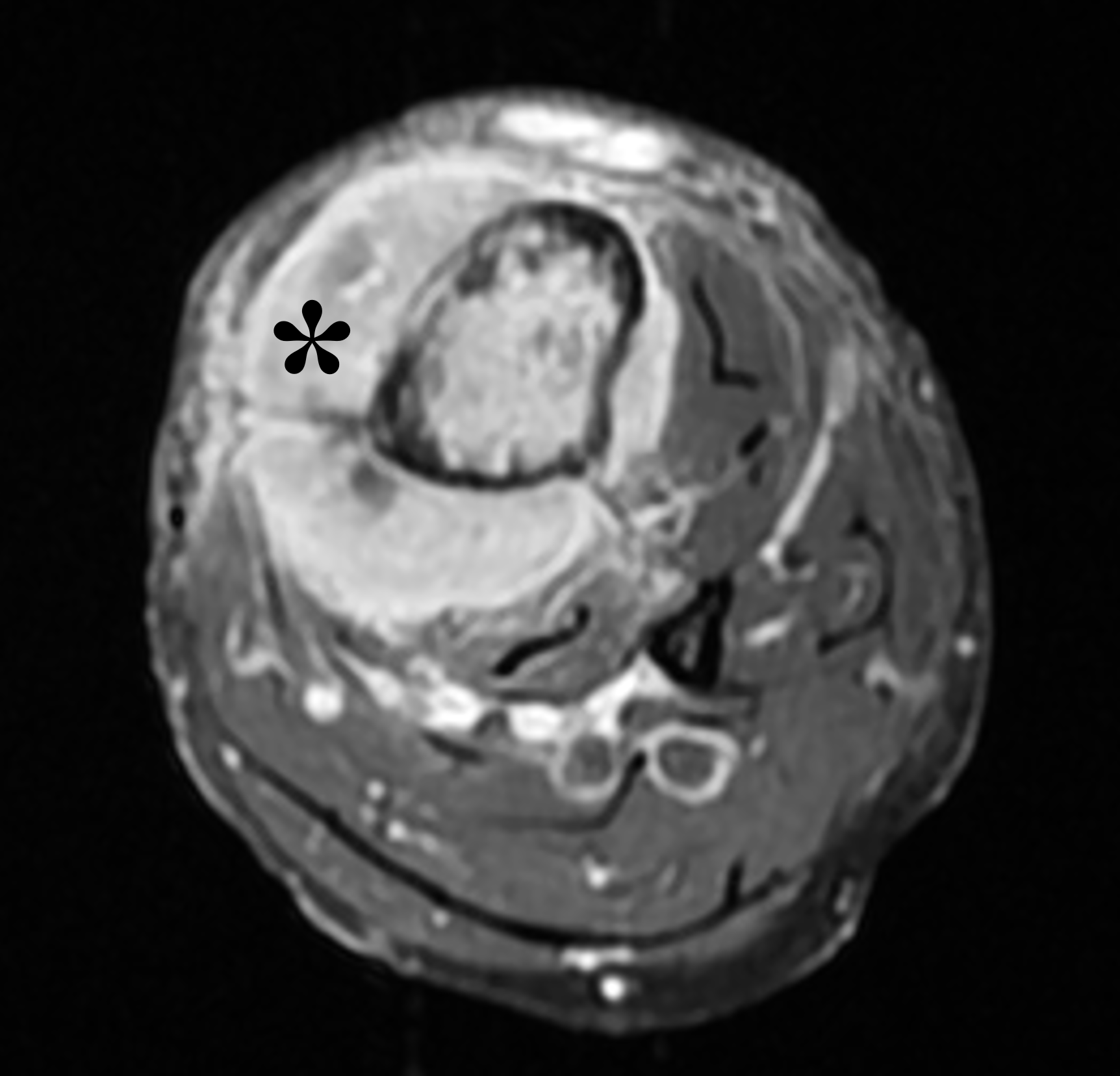
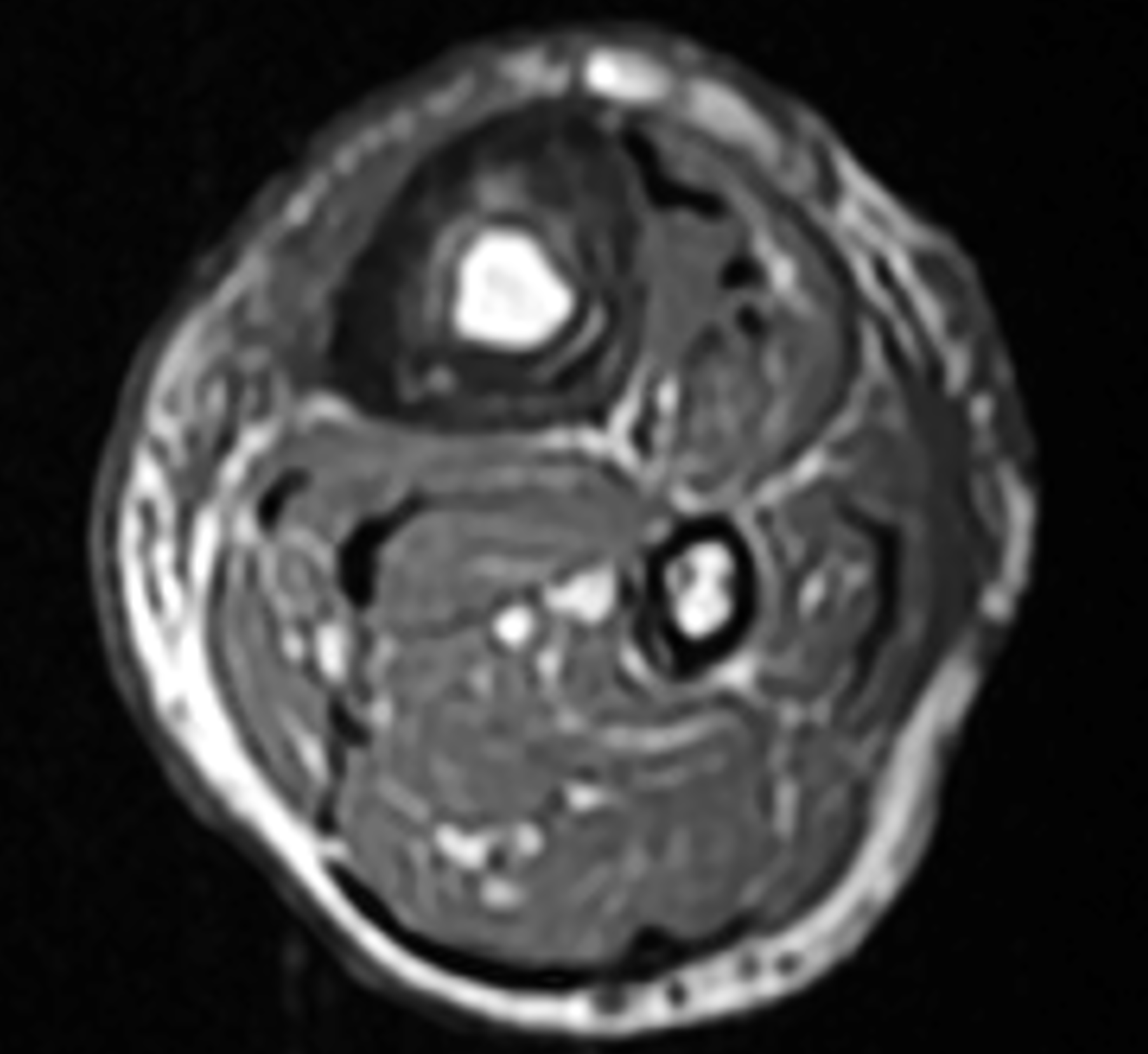
Figure 2: (2A) Sagittal T1-weighted image shows severe thickening of the distal femoral cortex (yellow arrow) and thickening and coarsening of the trabeculae in the femoral condyles (red arrow). Note the high signal intensity fatty marrow in both regions. (2B) Axial fat-suppressed, fluid sensitive sequence through the distal femoral shaft shows severe thickening of the bone cortex, with small round and tubular high-signal intensity foci within the cortex (arrow), likely representing cysts and dilated vascular spaces. (2C) Axial fat-suppressed, fluid sensitive sequence through the femoral epicondyles shows heterogeneous marrow (asterisks), with areas of suppressed marrow fat, mildly increased marrow signal intensity, and small cyst-like lesions. (2D) AP radiograph shows typical findings of mixed phase Paget disease in the distal femur including cortical thickening, course trabeculae, and bone enlargement. Secondary medial compartment predominant osteoarthritis has developed, likely contributing to symptoms.
Diagnosis
Paget disease, mixed phase.
Introduction
Paget disease of bone (osteitis deformans) is a chronic skeletal disorder characterized by accelerated and disorganized bone remodeling in which excessive osteoclastic resorption is followed by exuberant osteoblastic new bone formation. First described in 1877,1 the disease produces structurally abnormal bone with characteristic architectural changes including osseous expansion, cortical thickening, trabecular coarsening, and variable mixtures of lysis and sclerosis. Paget disease affects elderly individuals and is the second most common metabolic bone disease after osteoporosis in older adults.2,3 It most commonly involves the pelvis, spine, skull, and proximal long bones and may be monostotic or polyostotic. Although sometimes asymptomatic and incidentally detected, it may result in pain, deformity, fracture, secondary osteoarthritis, neurologic compromise, and rarely, malignant transformation.2,4,5
Imaging plays a central role in the diagnosis of Paget disease and identification of associated complications. Classical descriptions include lytic, mixed, and blastic phases of disease, although more than one phase may coexist within a single bone or across different sites in the setting of polyostotic Paget disease. Radiographs frequently show pathognomonic findings. CT, bone scintigraphy, and MRI serve complementary roles in confirming the diagnosis, defining disease extent, assessing activity, and evaluating for complications. When interpreted without accompanying radiographs, MRI may be particularly challenging because marrow heterogeneity, contrast enhancement, and at times mass-like features seen in differing stages of Paget disease can mimic malignancy.
Demographics, Etiology, and Causes
Paget disease increases with age. It is estimated to affect 1.5-8.3% of individuals over the age of 40, and 10-11% after the age of 80. However, recent studies report decreasing prevalence over the past 2 decades.6,7,8 There is a mild male predilection (3:2) with the age of onset being slightly younger in men.2-4,9 In the United States, it is the second most common bone disease after osteoporosis. The disease has higher prevalence in some populations of northern European descent, particularly in the United Kingdom, as well as in the United States, New Zealand, and Australia; Paget disease is rare in Asia, the Middle East, Scandinavia, and Africa.6,7
The exact cause of Paget disease remains incompletely understood, but most evidence now supports a multifactorial process involving strong genetic susceptibility with additional environmental triggers. Mutations involving genes that regulate osteoclasts (e.g. SQSTM1, TNFRSF11A, and related pathways) have been detected in both familial and sporadic cases of Paget disease and can be associated with earlier onset and more extensive disease.10,11 Proposed environmental factors include viral infections (especially paramyxovirus) , smoking, arsenic exposure, sedentary lifestyle, diets high in saturated fats, low calcium and low vitamin D levels.11,12
Disease Distribution
Paget disease most often affects the axial skeleton, particularly the pelvis and spine (in 30-75% of cases), sacrum (30-60%), and skull (25-65%). The proximal long bones are also commonly affected, especially the femur (25%–35%) and humerus (approximately 31%). Less common sites include the cervical and thoracic spine, scapula, and clavicle; involvement of the ribs, fibula, patella, and the bones of the hands and feet is uncommon, but any bone can be affected.2,4,5,9,13,14 Monostotic disease accounts for 10%–35% of cases and is more frequently seen in the axial skeleton. Polyostotic disease is more common and tends to involve the lower extremities with a reported right-sided predominance. Pelvic involvement is typically asymmetric, and appendicular disease is often unilateral.4,5
Pathologic Findings and Imaging Correlates
Paget disease reflects a dynamic process of excessive and disorganized bone remodeling. Early disease is dominated by osteoclastic activity, resulting in marked osseous resorption and corresponding lytic change. A reparative phase follows, characterized by increased osteoblastic activity with formation of immature woven bone and disorganized lamellar bone, resulting in the mixed phase of disease. During the more active stages, particularly the lytic and early mixed phases, normal yellow marrow is replaced by fibrovascular tissue, whereas partial or more complete restoration of fatty marrow occurs in the later mixed and inactive phases. These pathologic findings likely account for the variable appearance of the bone marrow seen on MRI. In late disease, sclerosis predominates, with progressive cortical thickening, trabecular coarsening, and osseous enlargement.3,5
Histologically, the hallmark of Paget disease is a mosaic pattern of lamellar bone with prominent cement lines along thickened and coarsened trabeculae. Disruption of normal trabecular architecture and loss of lattice-like interconnections result in structurally weakened bone, often described as “pumice” bone. Remodeling is often most active in the cortex where associated hypervascularity and small vascular channels may explain the intracortical high signal intensity seen on fluid-sensitive MRI sequences in active disease. The radiologic appearance mirrors the pathologic changes: osteolysis in the lytic phase, combined lytic and sclerotic findings with cortical and trabecular thickening in the mixed phase, and diffuse sclerosis and bone expansion in the blastic phase.3,5
Clinical Findings and Complications
Approximately 20% of patients with Paget disease are asymptomatic, with the diagnosis made incidentally on imaging studies obtained for unrelated indications.2,5 9 14 When present, symptoms reflect mechanically weakening of the bone and include localized pain, tenderness, hyperemia, osseous enlargement, deformity, kyphosis, decreased range of motion, pathologic fracture, and secondary osteoarthritis. Bone softening can produce bowing of the long bones, acetabular protrusion, and basilar invagination. Fracture is the most common complication of long-standing Paget disease and typically occurs along the convex surface of weight-bearing long bones. The femur (especially in the subtrochanteric region), tibia, humerus, pelvis, and spine are most often affected.4,5,14,15 Following fracture, substantial pain, deformity, and functional limitation, as well as inefficient healing often require surgical intervention.
Skull disease may lead to progressive enlargement of the head size and cranial neuropathies caused by narrowing of skull base foramina. Platybasia and basilar invagination, reported in up to 30% of cases with skull involvement, may produce narrowing of the foramen magnum. In severe cases, obstructive hydrocephalus, syringomyelia, and brainstem compression have been described. Involvement of the facial bones may produce leontiasis ossea, whereas jaw involvement may be associated with dental abnormalities related to hypercementosis.5,16,17
Vertebral compression fractures and kyphosis are complications of Paget disease in the spine. Central canal or foraminal stenosis (due to osseous enlargement or complications from fracture) can lead to neurologic symptoms. Spinal cord hypoxia has also been reported and has been postulated to result from a vascular steal phenomenon, with blood flow diverted from the spinal cord to hyperemic Pagetic bone.5,16,18
Systemic complications may also occur including high-output heart failure, arterial calcification, and, in severe cases, conduction abnormalities and valvular disease. Less commonly, endocrine, genitourinary, and psychological complications have been described.3,5
Biochemical assessment supports diagnosis, estimates metabolic activity, and helps monitor response to antiresorptive therapy. Total serum alkaline phosphatase (ALP) interpreted together with liver function tests, remains the most widely used and most cost-effective first-line marker and generally reflects overall disease activity and burden, particularly in polyostotic disease. However, total ALP may be normal in monostotic disease, limited skeletal involvement, or inactive/sclerotic disease; accordingly, a normal ALP level does not exclude Paget disease when the imaging findings are characteristic. When total ALP is normal or potentially confounded by hepatobiliary disease, measurement of more specific bone turnover markers, including bone-specific alkaline phosphatase (BSAP) or procollagen type 1 N-terminal propeptide (P1NP), may improve specificity for skeletal turnover and support the diagnosis. Resorption markers such as urinary NTX or serum/urinary CTX may also be used in selected settings, depending on local availability and assay practice.19,20
Imaging Findings Across Modalities and Phases
Radiographs
In the lytic (osteoclastic) phase of disease, osteolysis is the predominant radiographic finding. In the long bones, osteolysis typically begins at one end of the bone and progresses longitudinally at an estimated rate of 1 cm per year. The advancing leading edge forms a sharply marginated radiolucent wedge, producing the classic “blade-of-grass” or “flame-shaped” appearance on radiographs.3,5 14,21 In the skull, large sharply-demarcated radiolucent areas are referred to as osteoporosis circumscripta, and typically occur in the frontal and occipital bones.14,22
As the disease evolves, early trabecular coarsening indicates the transition to the mixed phase, where osteolysis and sclerosis coexist. In the long bones the trabeculae become coarse along stress lines, the cortex thickens, and the bone enlarges.2,3,5,14 In the pelvis, a characteristic finding is along the iliopectineal and ilioischial lines, often asymmetrically, with the right side more commonly affected.9 In the spine circumferential cortical thickening gives rise to the classic “picture-frame” appearance on radiographs along with prominent vertical trabecular thickening.3-5
In the late blastic phase, osteosclerosis predominates. The bones continue to demonstrate cortical thickening and coarsened trabecula in addition to marked enlargement. In some cases, sclerosis becomes so pronounced that the areas of trabecular thickening become obscured and indistinguishable. One example is the “ivory vertebra” where an entire vertebra (including the posterior elements) may appear homogeneously sclerotic and enlarged.23 Although sclerotic, the bones are weak and show signs of softening as well as pathologic fractures.2,14
CT
The CT findings of Paget disease largely mirror the radiographic ones. CT does, however, provide superior cortical and trabecular detail, and more readily illustrates areas of osteolysis, cortical thickening, trabecular coarsening and disorganization, focal areas of sclerosis, and osseous expansion. Where CT is advantageous is in the evaluation of bones with complex anatomy such as the pelvis and spine, and in the work up of suspected complications such as fractures, degenerative arthropathy, narrowing of the foramina of the skull, and spinal stenosis. CT is more sensitive than radiographs for findings associated with malignant degeneration including cortical destruction, aggressive lytic lesions, and soft tissue masses.5,24
MRI
Paget disease is often detected incidentally on MRI, where the variable findings can create a diagnostic dilemma, especially in the absence of available radiographs. Regardless of the marrow signal characteristics, misinterpretation as a marrow-replacing process can often be avoided by recognizing the characteristic imaging features of bone expansion, cortical thickening, and coarsened trabeculae. When available, correlation with radiographs or CT may make it easier to identify the characteristic and often pathognomonic features of Paget disease. In this author’s opinion, whenever possible radiographic correlation should be performed when evaluating an osseous or soft tissue lesion on MRI to avoid misinterpretation and improve diagnostic accuracy.
Across the spectrum of uncomplicated Paget disease, three common marrow patterns have been described, reflecting the balance among fatty marrow, fibrovascular tissue, and sclerosis.5,25
Pattern 1. Fatty marrow is predominantly preserved across pulse sequences (easiest to appreciate on T1-weighted images) corresponding pathologically to atrophic marrow, with associated trabecular coarsening and cortical thickening (Figure 3).26,27 Cortical expansion may narrow the medullary cavity. Post-contrast enhancement, when present, is typically mild and/or patchy. This pattern is most frequently associated with long-standing disease.
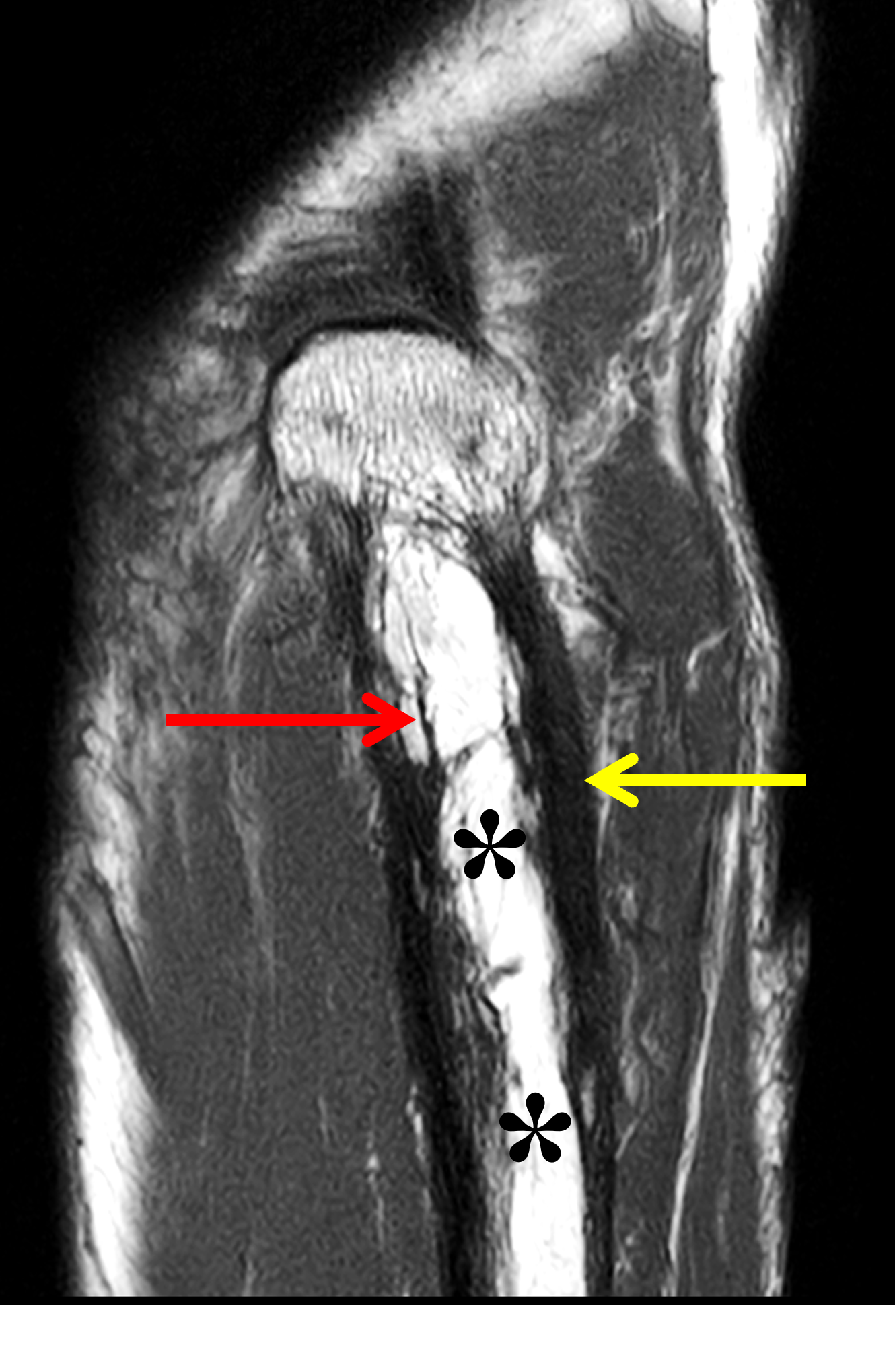
Figure 3: Paget disease of the proximal ulna, MRI pattern 1. (3A) Axial and (3B) coronal T1-weighted images show cortical thickening (yellow arrows) and coarse trabeculae (red arrows) in the ulna compared to the radius (R). Note the preserved high signal intensity fat in the bone marrow (asterisks). (3C) Lateral and (3D) AP radiographs show the typical findings of mixed-phase Paget disease of the ulna with bone enlargement, thick cortex, and prominent trabeculae. Bowing of the ulna indicates bone softening.
Pattern 2. An active fibrovascular marrow replacement pattern is typical of the lytic to early mixed active phases. During this stage, portions of the fatty marrow are progressively replaced by fibrovascular tissue, presumably leading to the low-to-intermediate T1 signal, intermediate-to-high T2/STIR signal, and avid enhancement often referred to as a “speckled” appearance on MRI.5,28,29 The heterogeneous marrow appearance in this phase can be a diagnostic pitfall and lead to misinterpretation as malignancy or malignant degeneration. A key distinguishing feature is the presence of intermixed foci of preserved normal fatty yellow marrow (Figures 4 and 5), supporting active Paget disease and essentially excluding malignancy. Round or tubular areas of T2 hyperintensity may be seen within the thickened cortex, possibly reflecting dilated vascular channels (Figure 6). Findings of bone softening and pathologic fractures can appear during the mixed phase (Figure 7). Marrow signal alterations are frequently relatively mild (Figures 4 and 5), though diffusely abnormal marrow signal may occasionally occur (Figure 8). The importance of correlation with radiographs or CT cannot be overstated: classic bone expansion, cortical thickening, and trabecular coarsening without an aggressive lytic destructive lesion, cortical breakthrough, or a soft tissue mass favors active Paget disease over tumor.
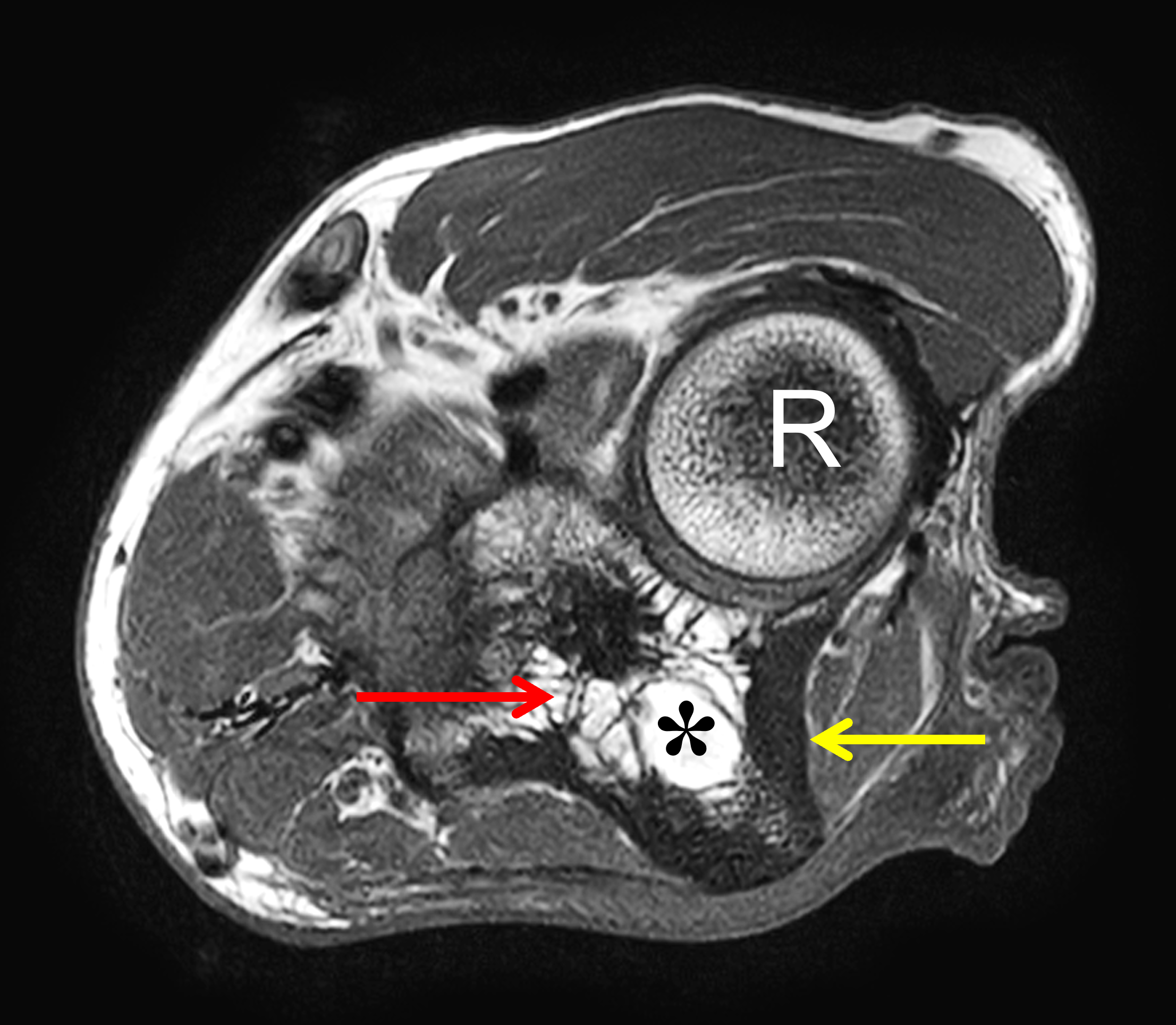
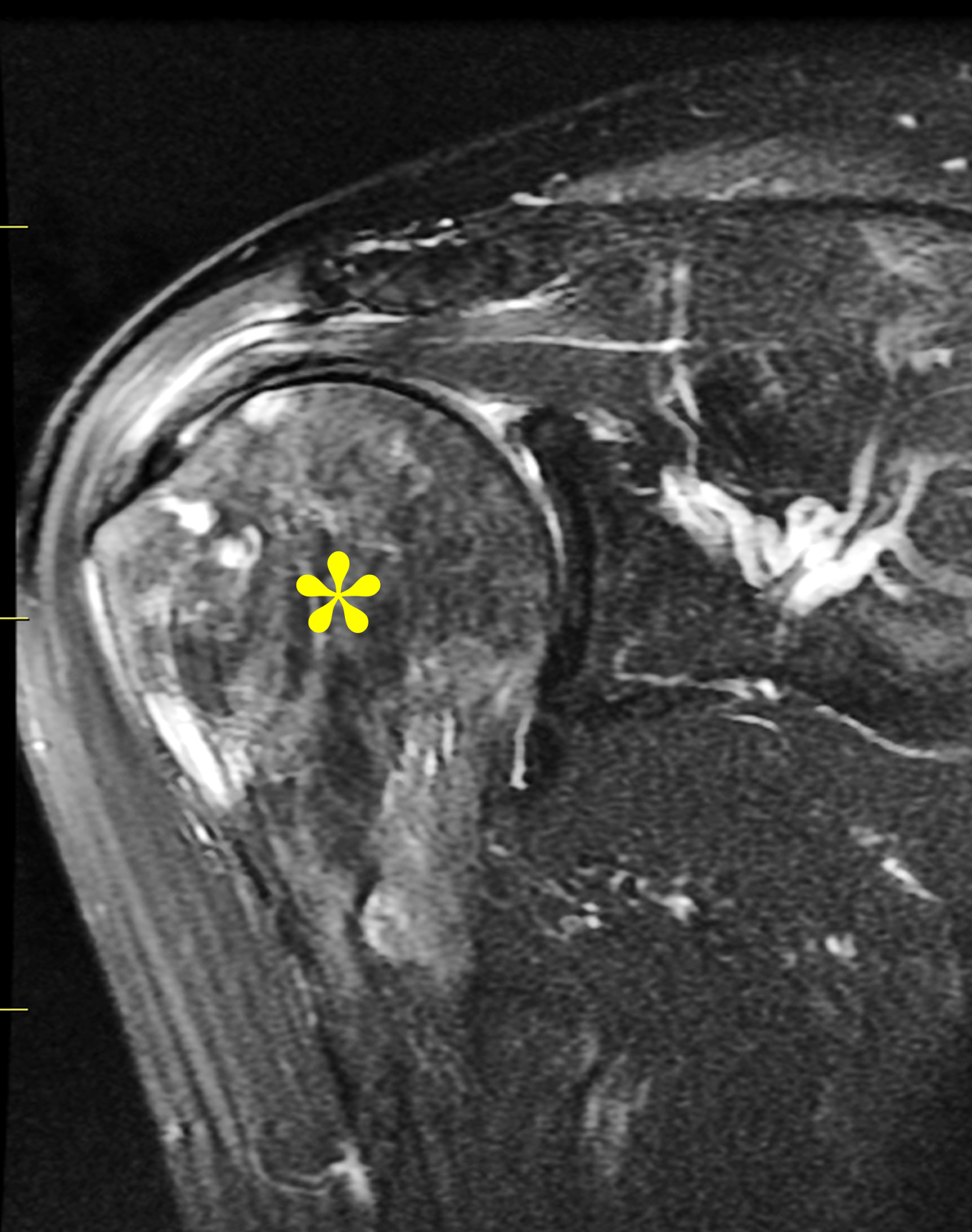
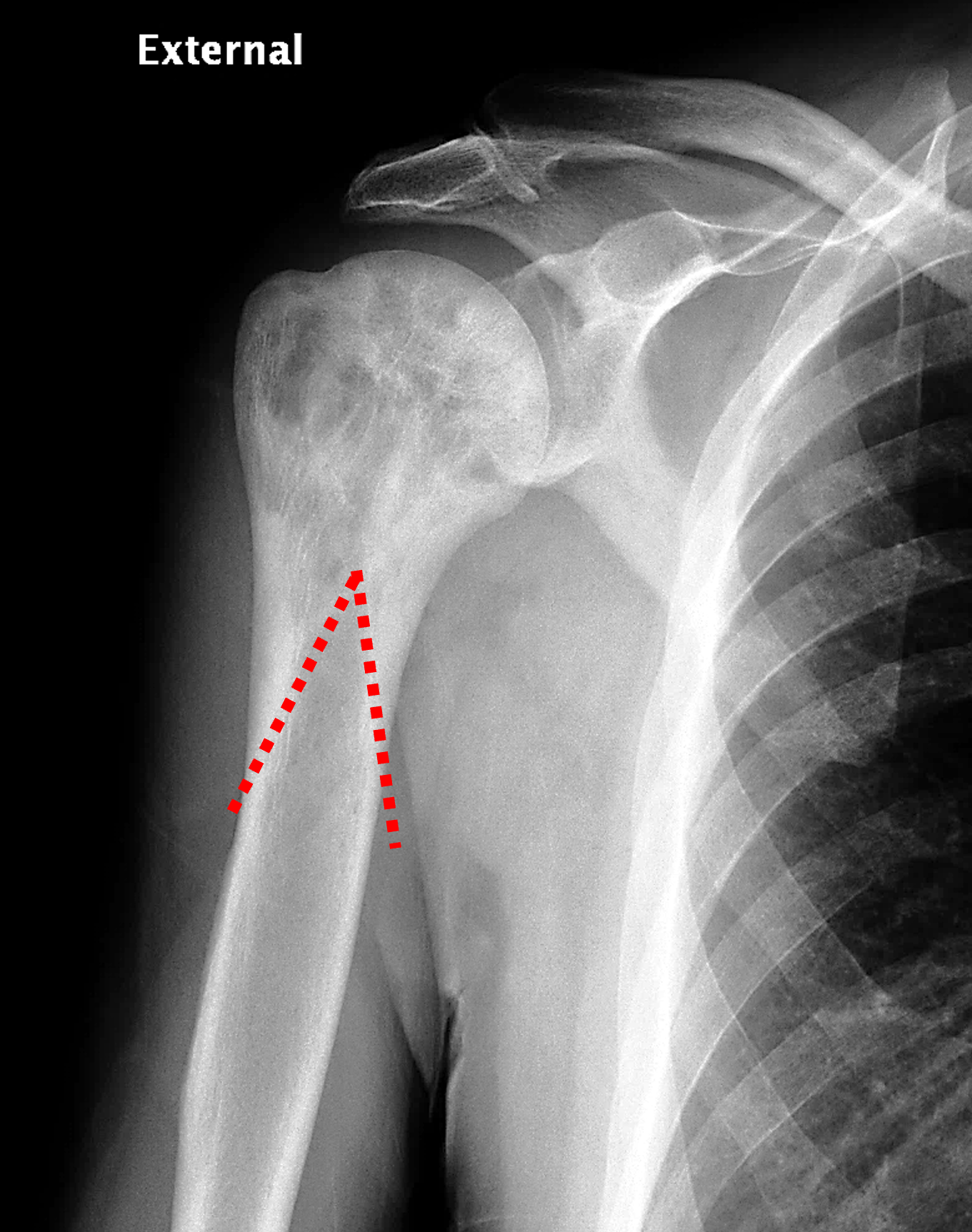
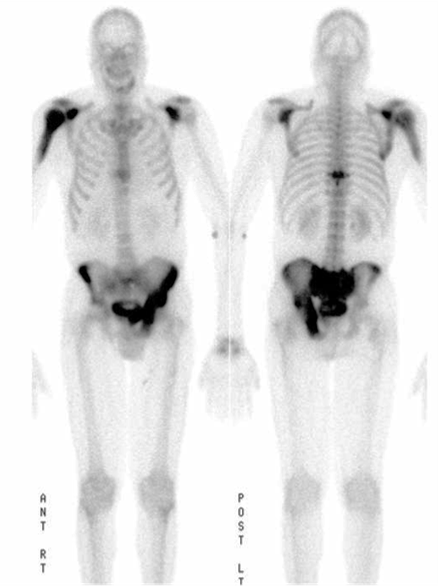
Figure 4: Mixed phase Paget disease of the proximal humerus in a patient with polyostotic disease, MRI pattern 2. (4A) Coronal fat-suppressed, fluid-sensitive image shows heterogeneous marrow (asterisk) with areas of suppressed fat, mildly increased signal, and cystic/tubular high signal intensity areas. (4B) Coronal T1-weighted image shows preserved fat signal (asterisk) within the marrow, as well as thick cortex and trabeculae. (4C) AP radiograph shows typical findings of mixed-phase Paget disease, beginning at the proximal end of the bone and extending distally. Note the sharp demarcation (dotted lines) separating abnormal from normal bone, forming a “flame-shaped” interface. (4D) Bone scintigraphy demonstrates polyostotic disease with intense tracer uptake in the left hemipelvis, sacrum, and a thoracic vertebra in addition to the right proximal humerus.
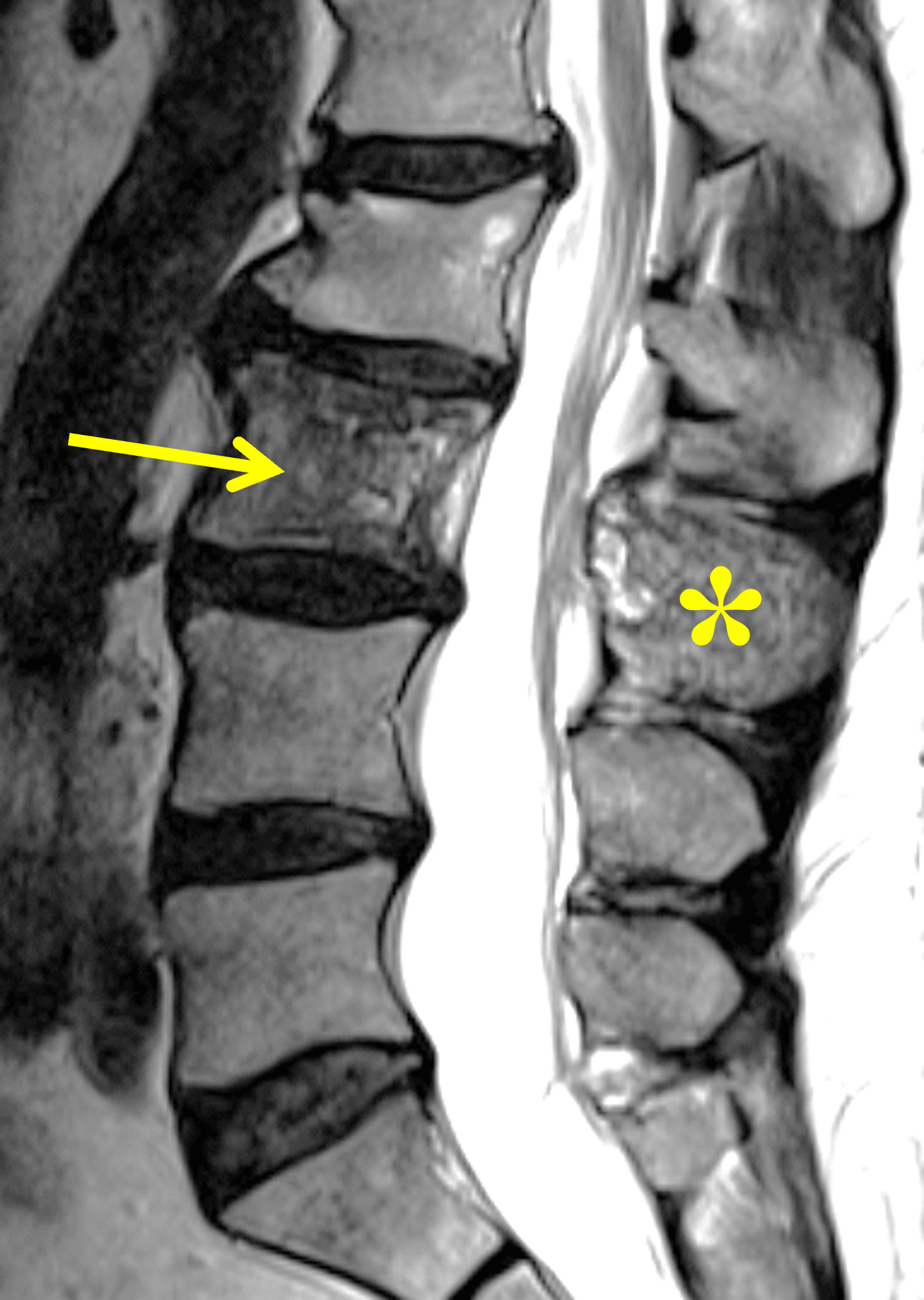
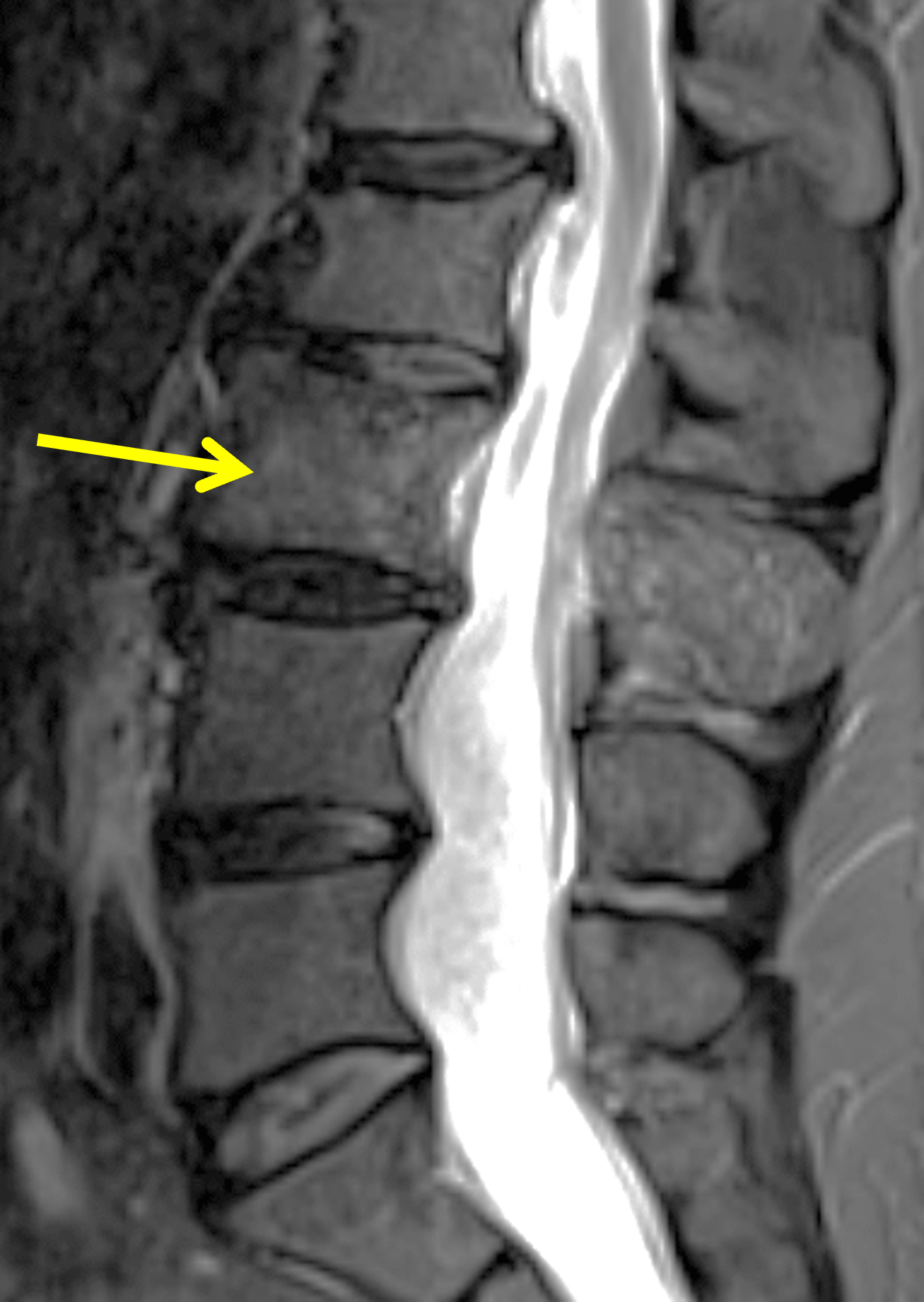
Figure 5. Mixed phase Paget disease of L3, MRI pattern 2. (5A) Sagittal T2-weighted image shows heterogeneous marrow (arrow) in the L3 vertebral body, with regions of both mildly increased and decreased signal intensity. Note the involvement of the spinous process (asterisk), which is characteristic of Paget disease involving the spine. (5B) Sagittal T1-weighted image shows small foci of preserved marrow fat (arrow), a finding that distinguishes Paget disease from malignant infiltration. Note the thickened, vertically-oriented trabeculae. (5C) Sagittal STIR image shows only minimally hyperintense marrow signal (arrow). (5D) Sagitally-reconstructed CT image shows thick, coarse trabeculae in the vertebral body (arrow) and spinous process (asterisk), which can be seen in both Paget disease and hemangioma. The bone is subtly enlarged, but thickened cortex is not present. Because of diagnostic uncertainty, percutaneous biopsy was performed, confirming Paget disease. A healed, benign compression fracture involves the superior endplate of L2.
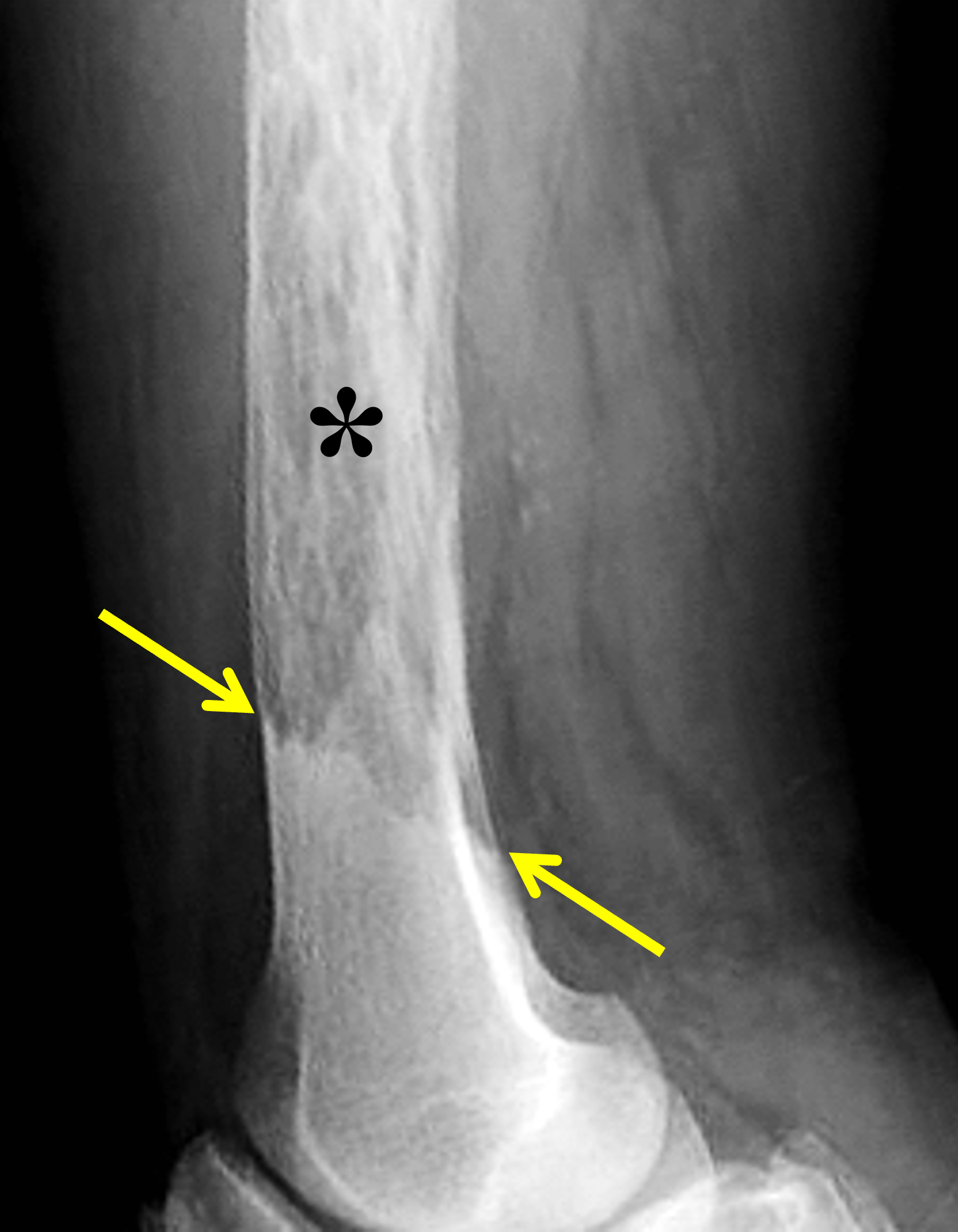
Figure 6: Lytic to early mixed phase Paget disease of the distal femur. (6A) Lateral radiograph shows extensive osteolysis and coarse trabeculae in the femoral shaft (asterisk). It is often difficult to confidently identify thickened cortex during the active phase of osteolysis. Note the sharp transition from Pagetoid bone to normal bone distally (between arrows). (6B) Axial fat-suppressed, fluid-sensitive image demonstrates mildly thickened cortex containing tubular areas of very high signal intensity (arrows), suggesting dilated vascular channels.
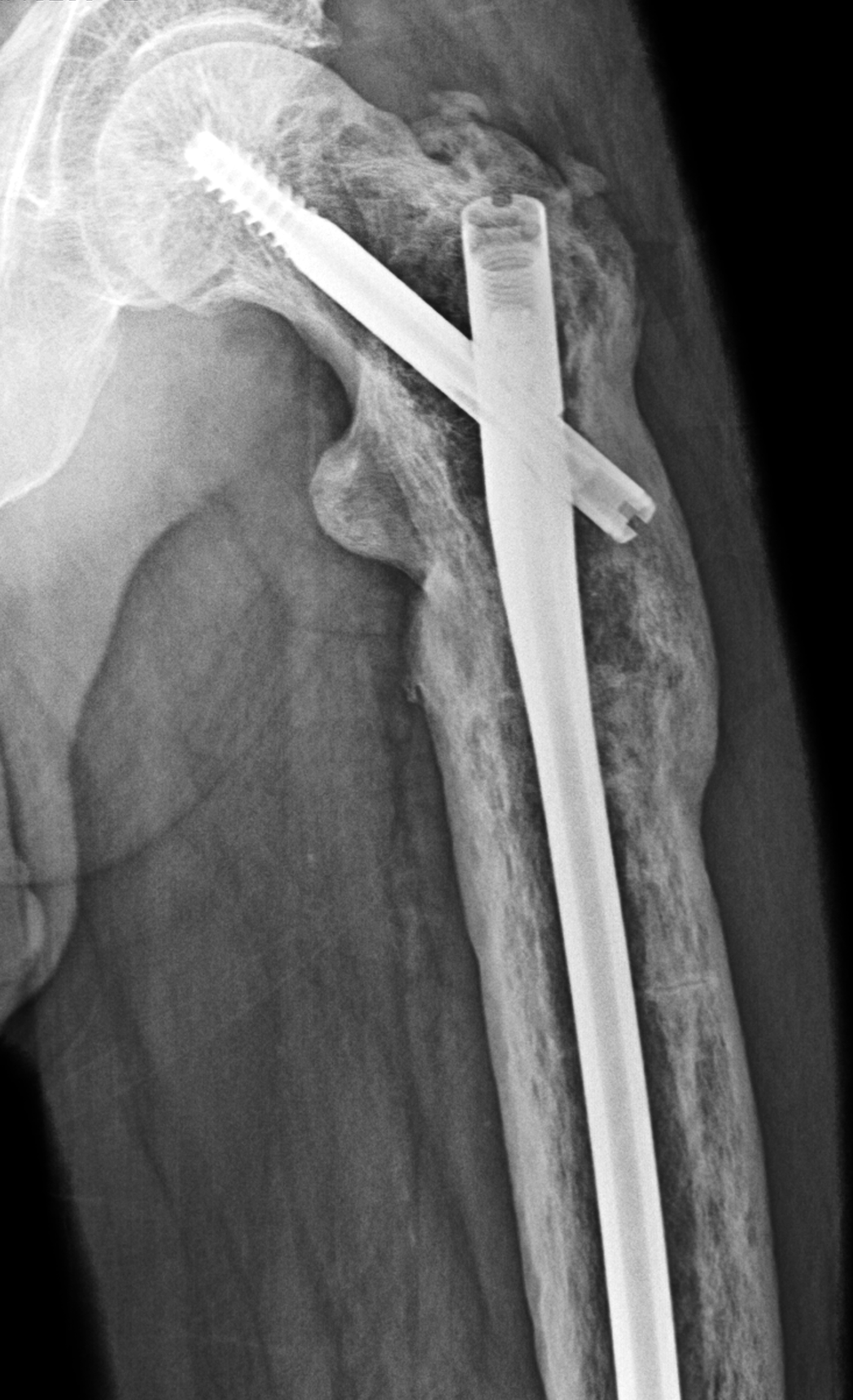
Figure 7: Mixed phase Paget disease of the proximal femur with a developing pathologic fracture. (7A) T1-weighted and (7B) STIR coronal images of the hip show typical MRI pattern 2 findings of Paget disease. Note the mild varus bowing of the femoral neck (red arrows), as well as the developing incomplete, transverse fracture through the lateral cortex (yellow arrows) representing a Pagetic “banana” fracture. (7C) AP radiograph following intramedullary nailing demonstrates fracture healing.
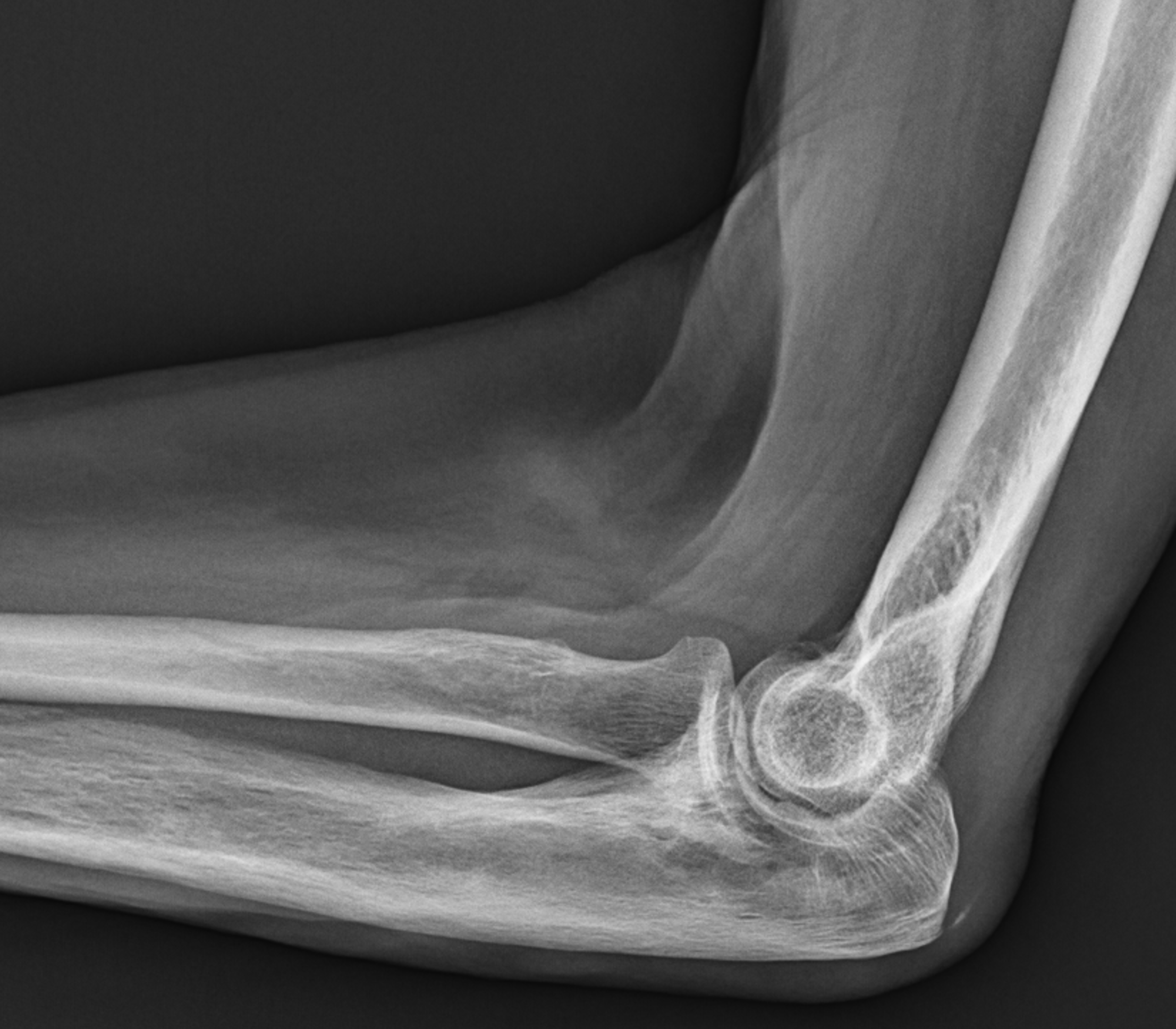
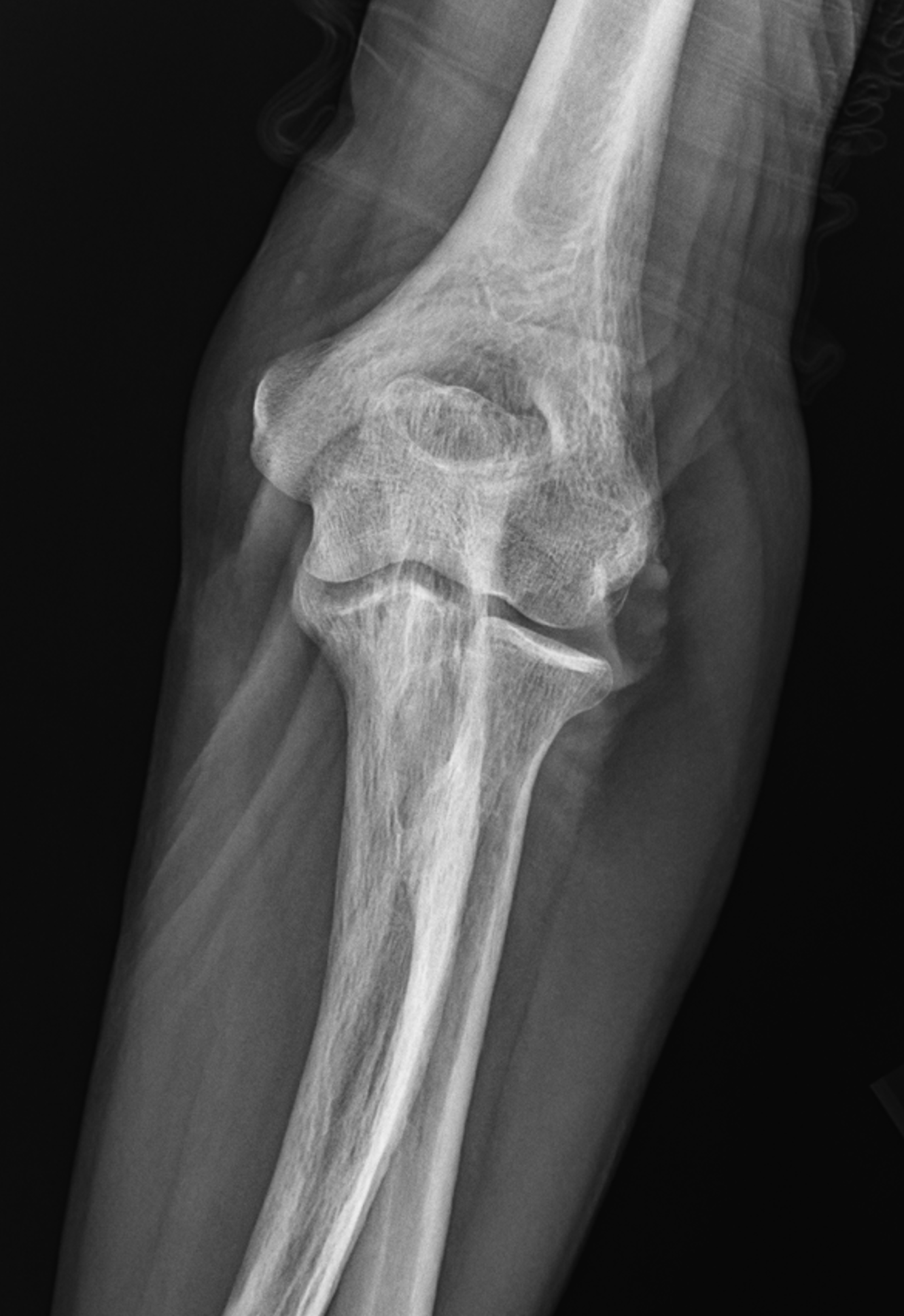
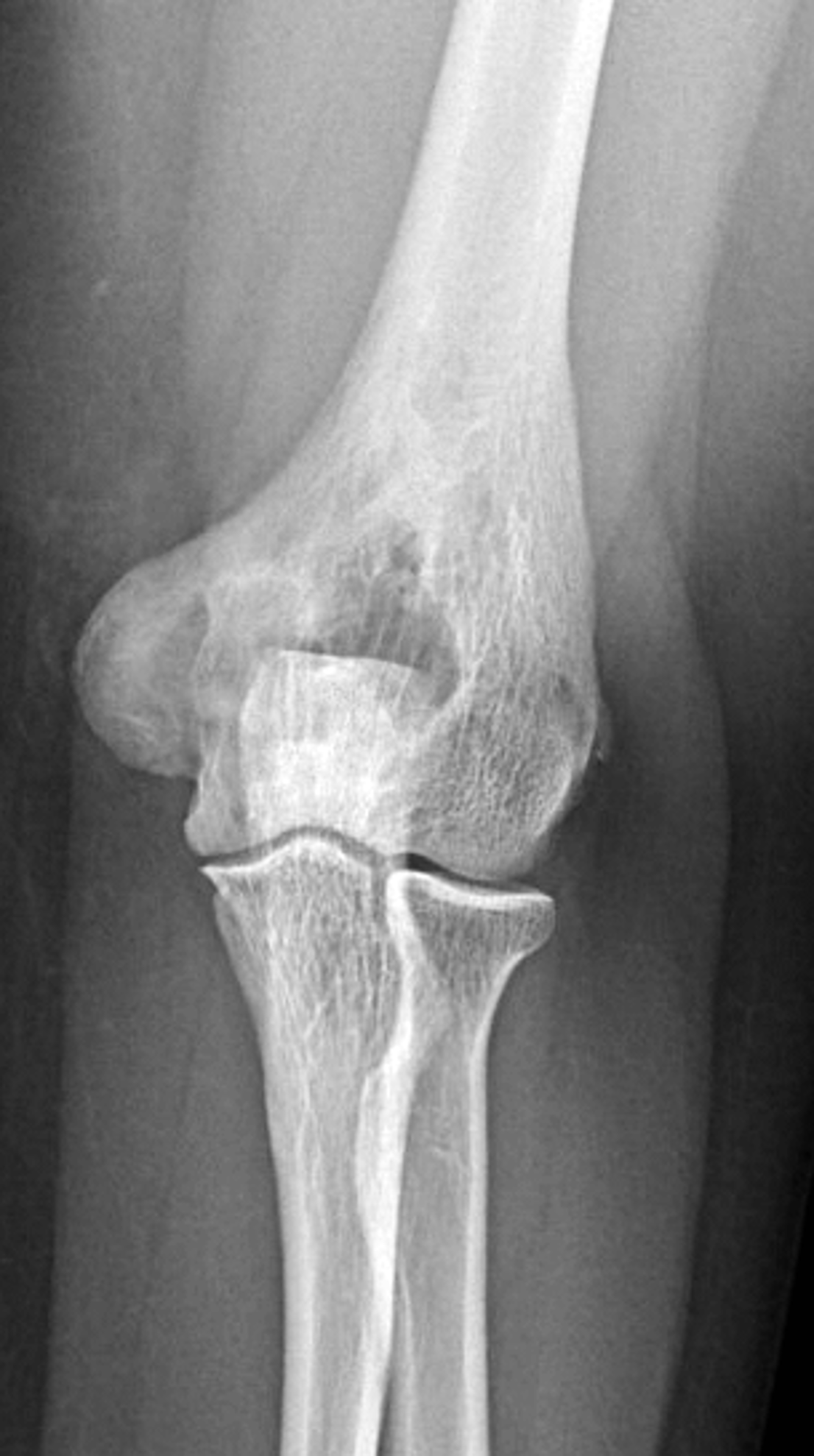
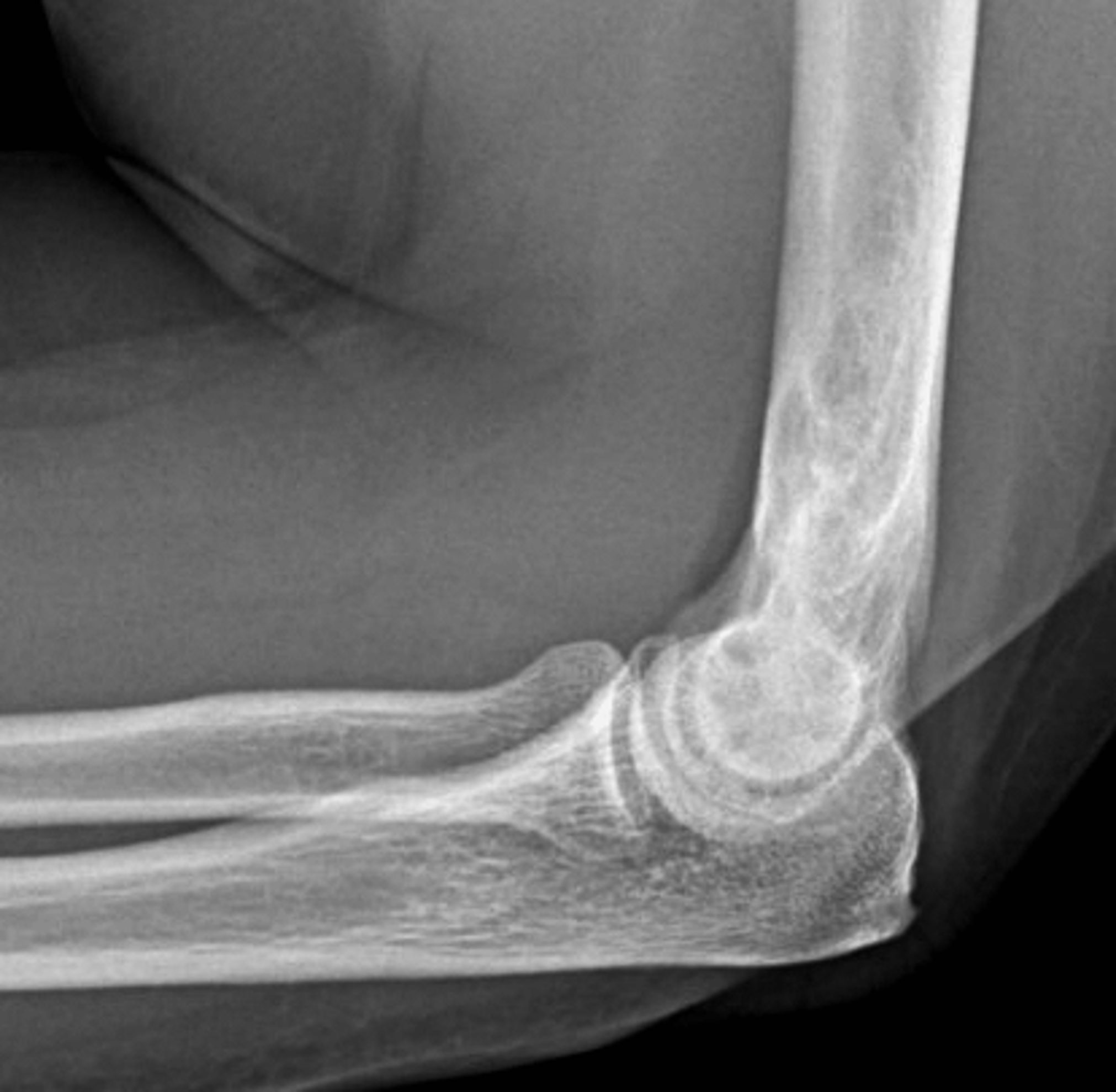
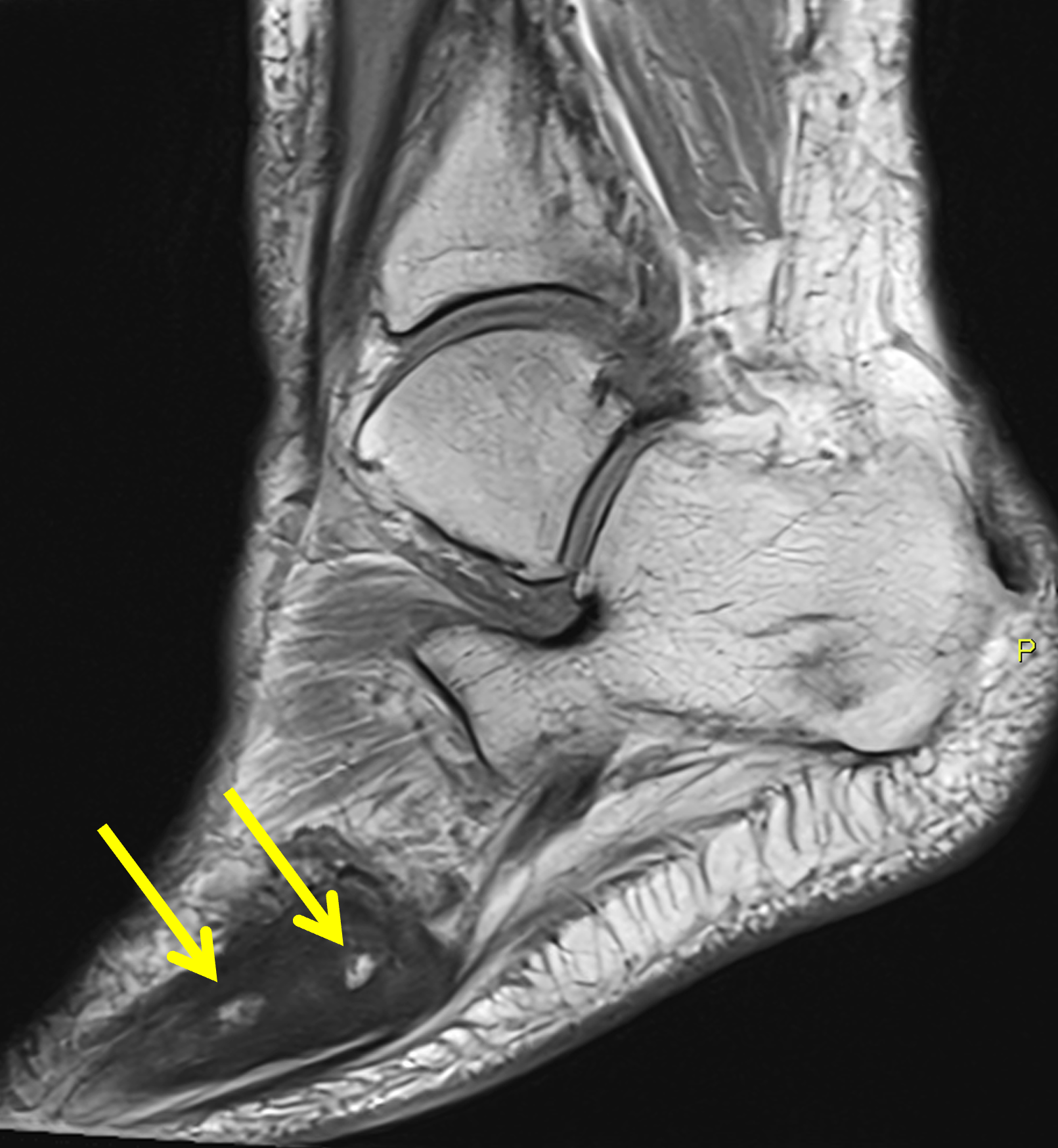
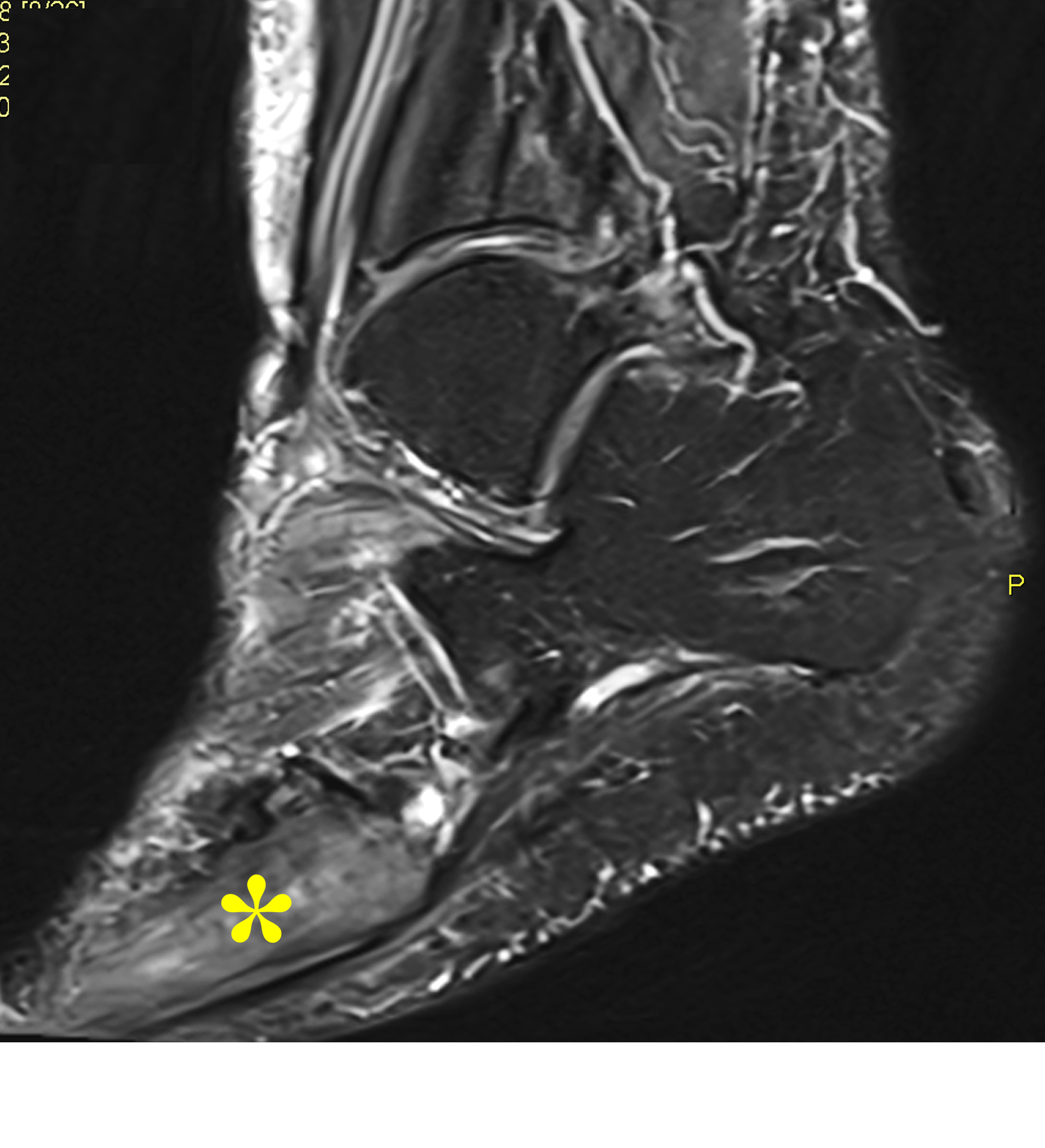
Figure 8: Mixed phase Paget disease in the distal humerus with atypical marrow replacement. (8A) AP and (8B) lateral elbow radiographs show classic findings of Paget disease in the distal humerus, including osseous enlargement, particularly of the medial epicondyle, with associated cortical and trabecular thickening. (8C) T1-weighted and (8D) STIR coronal images show diffusely abnormal marrow signal intensity in the medial humeral epicondyle (asterisks), hypointense compared to skeletal muscle on the T1-weighted sequence and uniformly hyperintense on the STIR sequence. This pattern of marrow replacement is atypical in uncomplicated Paget disease and prompted biopsy, which confirmed the diagnosis. Despite the marrow findings, the radiographic features in addition to the lack of osseous destruction and soft-tissue mass suggest uncomplicated Paget disease and fibrovascular changes. Short interval follow-up to document stability may obviate the need for biopsy in similar cases.
Pattern 3. The least common pattern encountered is during the blastic inactive disease phase with low signal intensity on all pulse sequences suggesting compact bone or fibrous tissue (Figure 9).3,27 Dense sclerosis in the axial skeleton can mimic osteoblastic metastases; accompanying foci of preserved fatty marrow, bone expansion, trabecular coarsening, and stability over time support a diagnosis of Paget disease.
Figure 9: Sclerotic phase Paget disease of the fifth metatarsal, MRI pattern 3. (9A) Sagittal T1-weighted image shows nearly uniform hypointense marrow in the fifth metatarsal, with small islands of preserved marrow fat (arrows), which helps exclude a malignant process. (9B) Sagittal fat-suppressed T2-weighted image shows mildly increased signal intensity throughout the marrow (asterisk). (9C) Dorsoplantar radiograph demonstrates sclerosis throughout the fifth metatarsal, due to cortical and trabecular thickening. Note the bone enlargement and bowing. (9D) Short-axis CT image shows nearly uniform sclerosis throughout the fifth metatarsal.
Bone scintigraphy
Bone scintigraphy is highly sensitive for detecting and mapping the extent of Paget disease and typically demonstrates intense radiotracer uptake at involved sites, particularly in metabolically active disease. Scintigraphy is useful for distinguishing monostotic from polyostotic involvement (Figure 4), identifying additional clinically occult sites, supporting the diagnosis when radiographs/CT are characteristic at a symptomatic site, and monitoring activity alongside biochemical markers. Sarcomatous degeneration may appear as a photopenic “cold” focus within an otherwise hypermetabolic region of Paget disease; however, this finding is nonspecific in the absence of correlative findings on radiographs or cross-sectional imaging.5,26,30
Neoplastic Complications and Neoplastic Mimics
Malignant transformation is the most feared complication of Paget disease. Osteosarcoma is the most common histologic subtype found in Paget sarcoma, followed by chondrosarcoma and undifferentiated pleomorphic sarcoma.2,31 , 32 In contrast to conventional osteosarcoma, the prognosis for patients with sarcomatous degeneration is extremely poor with studies consistently reporting 5-year survival rates of less than 10% despite neoadjuvant chemotherapy and limb salvage surgery.
The true incidence of Paget sarcoma remains uncertain. Although it has been estimated to occur in approximately 1% of patients with long-standing Paget disease, this figure is likely inflated by referral bias as published series are largely based on tertiary-center populations.33 Sarcomatous transformation is slightly more common in polyostotic compared to monostotic disease. Rare cases of multifocal synchronous Paget sarcoma have also been described. Men are affected more often than women, approximately 2:1, and patients are typically 55–80 years old. Most patients present with new focal pain or a rapidly enlarging mass; pathologic fracture occurs in up to one third of cases.3,5,32
The pelvis, femur, and humerus are the most common sites of Paget sarcoma. This distribution differs somewhat from that of Paget disease overall, with the humerus showing a disproportionately high rate of sarcomatous degeneration and the spine, although commonly involved by Paget disease, only rarely undergoing malignant transformation.
Imaging features of Paget sarcoma are those of an aggressive lesion with areas of cortical destruction and an associated soft-tissue mass. In one review of 50 cases, 65% of lesions showed lytic osseous destruction, 17% were mixed lytic-sclerotic, and 17% of cases predominantly sclerotic. Cortical destruction was present in 73%, soft-tissue mass in 58%, osseous expansion in 39%, and pathologic fracture in 32%.34 Aggressive periosteal reaction, a common feature of conventional osteosarcoma, is relatively uncommon in Paget sarcoma, likely reflecting the rapid bone destruction, which allows little time for a reparative response, and the high-grade histologic anaplasia of these tumors.32
On MRI an area of mass-like marrow replacement, often with adjacent cortical destruction and an associated infiltrative soft-tissue mass, is typical (Figure 10).32,34 The involved marrow demonstrates intermediate-to-low signal intensity on T1-weighted images and high signal intensity on fluid-sensitive sequences. Loss of the normal T1-hyperintense fatty marrow signal is one key feature distinguishing sarcomatous degeneration from uncomplicated Paget disease. Of note, during the active phase of uncomplicated Paget disease, the marrow may show focal or more diffuse T2 hyperintense signal intensity in addition to periosteal reaction related to accelerated bone turnover, findings which in isolation should not be confused for malignant degeneration.32,35,36 Additionally, diffuse low T1 signal marrow may be seen in the setting of a pathologic fracture in uncomplicated Paget disease, simulating malignant transformation. In such cases, tissue sampling may be required though biopsy can sometimes be obtained at the time of surgical fixation.34 Contrast enhancement is nonspecific and can be seen in both active disease and sarcomatous degeneration. In Paget sarcoma, MRI is particularly useful for delineating disease extent and for local staging, including evaluation of adjacent joint and neurovascular involvement, subtle or impending pathologic fracture, and the presence of skip metastases.
Figure 10: Paget sarcoma in the tibia. (10A) T1-weighted and (10B) STIR sagittal images show diffuse bone marrow replacement in the mid tibia with deep endosteal scalloping, a pathologic fracture (yellow arrows) and associated soft tissue mass (red arrows). (10C) Axial T1-weighted image shows regions of full-thickness cortical destruction. (10D) Axial fat-suppressed, T1-weighted image obtained following intravenous contrast administration shows a solid, enhancing soft tissue mass surrounding the tibia. Non-contrast axial T1-weighted images (10E) above and (10F) below the tumor demonstrate findings of underlying Paget disease including cortical thickening and fatty bone marrow.
Rarely, benign extraosseous soft-tissue extension with juxtacortical masses may occur in Paget disease, a condition known as tumefactive Paget disease or Paget pseudosarcoma. The appearance may mimic Paget sarcoma, primary bone neoplasm, metastasis, myeloma, plasmacytoma, or infection, often leading to misinterpretation and multiple biopsies.37,38 The entity should be considered when a soft-tissue component arises from Pagetic bone, especially when forming a rind of soft tissue outlining the bone, without evidence of a marrow-replacing lesion or cortical destruction, and against a background of otherwise characteristic imaging features of Paget disease. Identifying areas of preserved normal fatty marrow signal on MRI or CT, is critical for making or at least considering the diagnosis. Equally important is awareness of imaging features highly associated with malignant degeneration, including rapidly progressive osteolysis, cortical destruction, abnormal marrow signal, and a discrete extraosseous mass. Although biopsy is often required for aggressive-appearing lesions including soft tissue masses arising from Pagetic bone, careful radiologic assessment may help identify benign non-neoplastic mimics and, in selected cases, obviate the need for biopsy or re-biopsy.
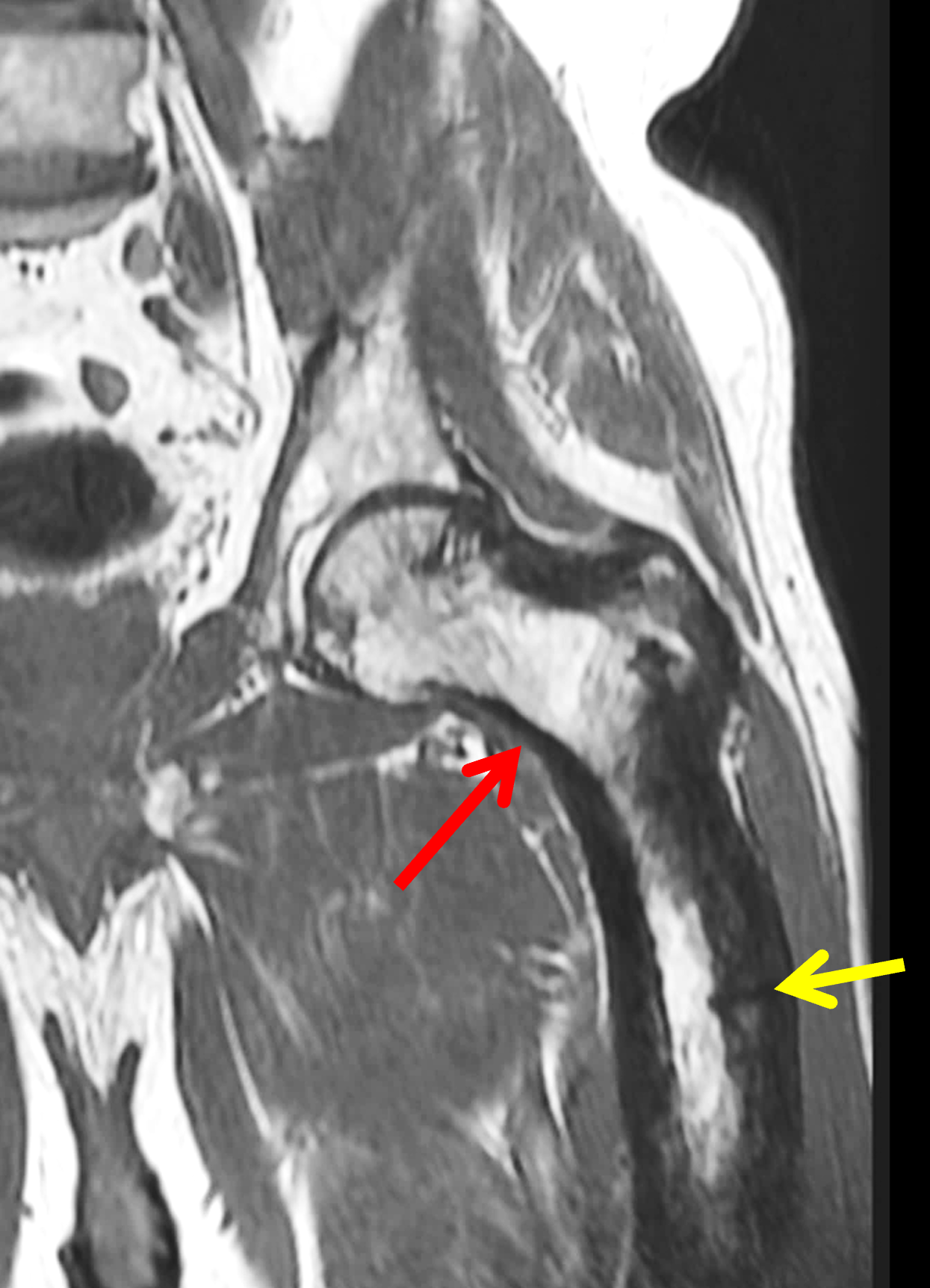
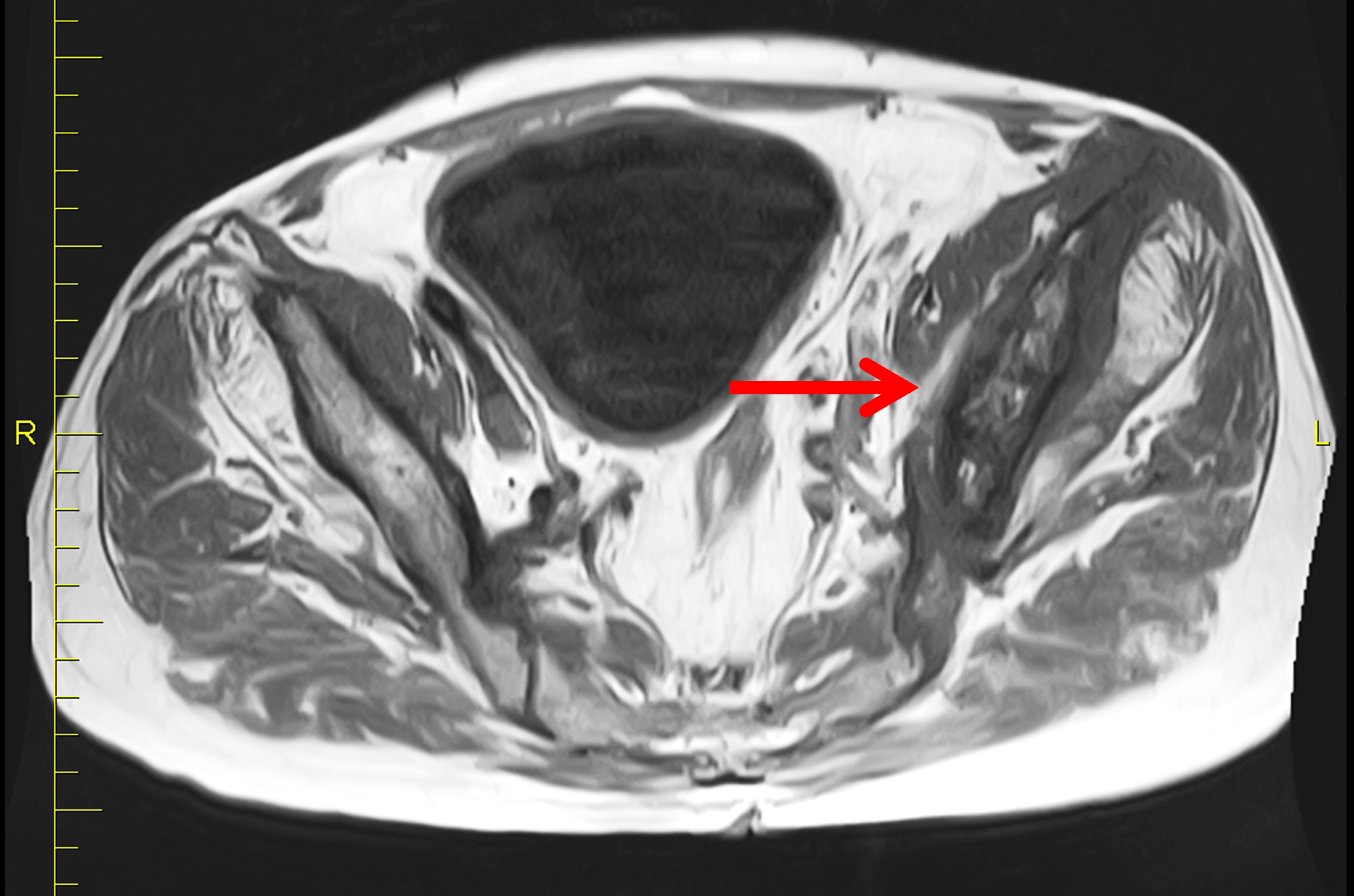
Figure 11. Tumefactive Paget disease of the pelvis. (11A) Axial T1-weighted image shows cortical thickening and thick trabeculae (arrow) in the left hemi-pelvis representing Paget disease. No focal bone destruction is present. High signal intensity foci in the marrow represents areas of preserved marrow fat, which suggests benignity. (11B) Axial fat-suppressed, fluid-sensitive image demonstrates a rind of high signal-intensity soft tissue encasing the iliac bone (arrows). The soft-tissue component prompted a biopsy, which confirmed the diagnosis of tumefactive Paget disease.
Other neoplastic processes may coexist with Paget disease, including giant cell tumor, metastatic disease, lymphoma, leukemia, and multiple myeloma, although this association is generally considered incidental rather than causal. The presence of multiple lytic or sclerotic lesions in bones not affected by Paget disease favors metastases, myeloma, leukemia, or lymphoma rather than sarcomatous transformation arising within Pagetic bone. Interestingly, despite the high prevalence of metastatic disease in the older population commonly affected by Paget disease, metastases involving Pagetic bone have been rarely reported.32,34
Treatment and Management
The goal of treatment has shifted toward symptom improvement rather than intensive biochemical suppression. Pharmacologic therapy is generally recommended for symptomatic active disease, particularly Pagetic bone pain, and may also be considered in selected situations where active disease may lead to neurologic compromise. Medical treatment may also be used before surgery on Pagetic bone to reduce bleeding. Intravenous or oral bisphosphonates are first-line therapy. Although no universal definition of therapeutic response exists, many randomized trials have used normalization of serum ALP or a reduction of at least 75% in excess ALP as evidence of adequate response to nitrogen-containing bisphosphonates. Some authors have further suggested that reducing total ALP, or another chosen bone turnover marker, to below the midpoint of the reference range may prolong remission.19,20
Calcitonin has historically been used when bisphosphonates are contraindicated, although it is generally less potent and is now used infrequently. For Paget disease involving the spine or pelvis, MRI assessment of stenosis and neural compromise can inform surgical decision-making, while preoperative antiresorptive therapy may be considered in selected active cases to reduce metabolic activity and hypervascularity. Follow-up typically includes symptom assessment and serial biochemical markers, with targeted imaging reserved for new symptoms, suspected complications, or discordance between the clinical and laboratory findings.19, 20,39
Conclusion
Paget disease of bone has a broad radiologic spectrum that reflects its underlying pathologic evolution across the lytic, mixed, and blastic phases. Conventional radiography remains the cornerstone of diagnosis. On MRI, the appearance of the disease can be deceiving, especially if only marrow findings are noted; recognizing changes in the osseous elements — bone expansion, cortical thickening, and trabecular coarsening — is essential to avoid misdiagnosis of an aggressive marrow-replacing process. Awareness of its complications, including deformity, fracture, neurologic compromise, and malignant transformation, is critical for appropriate management. While rare, sarcomatous degeneration is heralded by aggressive marrow replacement, cortical destruction, and an associated soft-tissue mass. Familiarity with the imaging spectrum of Paget disease and its complications allows confident diagnosis, helps avoid diagnostic error, and guides appropriate patient care.
References
- Paget J. On a form of chronic inflammation of bones (osteitis deformans). Med Chir Trans (Lond). 1877;6037–64. ↩
- Mirra JM, Brien EW, Tehranzadeh J. Paget’s disease of bone: review with emphasis on radiologic features, Part II. Skeletal Radiol. 1995;24(3):173–184. doi:10.1007/BF00228919 ↩
- Theodorou DJ, Theodorou SJ, Kakitsubata Y. Imaging of Paget disease of bone and its musculoskeletal complications: review. AJR Am J Roentgenol. 2011;196(6 Suppl):S64–75. doi:10.2214/AJR.10.7222 ↩
- Resnick D. Paget disease of bone: current status and a look back to 1943 and earlier. AJR Am J Roentgenol. 1988;150(2):249–256. doi:10.2214/ajr.150.2.249 ↩
- Smith S, Murphey M, Kambiz M, Mulligan M, Resnik C, Gannon F. From the Archives of the AFIP. Radiologic Spectrum of Paget Disease of Bone and Its Complications with Pathologic Correlcation. Radiographics. 2002;221191–1216. ↩
- Collins DH. Paget’s disease of bone; incidence and subclinical forms. Lancet. 1956;271(6933):51–57. doi:10.1016/s0140-6736(56)90422-6 ↩
- Barker DJ, Chamberlain AT, Guyer PB, Gardner MJ. Paget’s disease of bone: the Lancashire focus. Br Med J. 1980;280(6222):1105–1107. doi:10.1136/bmj.280.6222.1105 ↩
- Cooper C, Schafheutle K, Dennison E, Kellingray S, Guyer P, Barker D. The epidemiology of Paget’s disease in Britain: is the prevalence decreasing? J Bone Miner Res. 1999;14(2):192–197. doi:10.1359/jbmr.1999.14.2.192 ↩
- Merkow R, Lane J. Paget’s disease of bone. Orthopedic Clinics of North America. 1990;21171–189. ↩
- Hansen MF, Nellissery MJ, Bhatia P. Common mechanisms of osteosarcoma and Paget’s disease. J Bone Miner Res. 1999;14 Suppl 239–44. doi:10.1002/jbmr.5650140209 ↩
- Rendina D, Falchetti A, Diacinti D, Bertoldo F, Merlotti D, Giannini S, et al. Diagnosis and treatment of Paget’s disease of bone: position paper from the Italian Society of Osteoporosis, Mineral Metabolism and Skeletal Diseases (SIOMMMS). J Endocrinol Invest. 2024;47(6):1335–1360. doi:10.1007/s40618-024-02318-1 ↩
- Roodman GD, Windle JJ. Paget disease of bone. J Clin Invest. 2005;115(2):200–208. doi:10.1172/JCI24281 ↩
- Dalinka M, Aronchick J, Haddad J. Paget’s disese. Orthopedic Clinics of North America. 1983;143–19. ↩
- Mirra J, Brien E, Tehranzadeh J. Paget’s disease of bone: review with emphasis on radiologic features, part I. Skeletal Radiology. 1995;24163–171. ↩
- Melton LJ, 3rd, Tiegs RD, Atkinson EJ, O’Fallon WM. Fracture risk among patients with Paget’s disease: a population-based cohort study. J Bone Miner Res. 2000;15(11):2123–2128. doi:10.1359/jbmr.2000.15.11.2123 ↩
- Poncelet A. The neurologic complications of Paget’s disease. J Bone Miner Res. 1999;14 Suppl 288–91. doi:10.1002/jbmr.5650140218 ↩
- Popper M, Jacobson H, Duff B, Gottlieb C. Basilar impression and platybasia in Paget’s disease of bone. Radiology. 1953;61639–644. ↩
- Llorente MJ, Corts JR, Olmedo FJ, Laguia M. Spinal nerve root compression as onset of monostotic Paget’s disease. Computed tomography and magnetic resonance image findings. Br J Rheumatol. 1994;33(12):1194–1195. doi:10.1093/rheumatology/33.12.1194 ↩
- Singer FR, Bone HG, 3rd, Hosking DJ, Lyles KW, Murad MH, Reid IR, et al. Paget’s disease of bone: an endocrine society clinical practice guideline. J Clin Endocrinol Metab. 2014;99(12):4408–4422. doi:10.1210/jc.2014-2910 ↩
- Ralston SH, Corral-Gudino L, Cooper C, Francis RM, Fraser WD, Gennari L, et al. Diagnosis and Management of Paget’s Disease of Bone in Adults: A Clinical Guideline. J Bone Miner Res. 2019;34(4):579–604. doi:10.1002/jbmr.3657 ↩
- Wittenberg K. The blade of grass sign. Radiology. 2001;221(1):199–200. doi:10.1148/radiol.2211991689 ↩
- Erdheim J. Uber die genese der Paget schenknochenerkrankrung. Beitr Path Anat. 1936;961–7. ↩
- Harris DJ, Fornasier VL. An ivory vertebra: monostotic Paget’s disease of bone. Clin Orthop Relat Res. 1978(136):173–175. ↩
- Kumar A, Poon PY, Aggarwal S. Value of CT in diagnosing nonneoplastic osteolysis in Paget disease. J Comput Assist Tomogr. 1993;17(1):144–146. doi:10.1097/00004728-199301000-00028 ↩
- Dell’Atti C, Cassar-Pullicino VN, Lalam RK, Tins BJ, Tyrrell PN. The spine in Paget’s disease. Skeletal Radiol. 2007;36(7):609–626. doi:10.1007/s00256-006-0270-6 ↩
- Sundaram M. Imaging of Paget’s disease and fibrous dysplasia of bone. J Bone Miner Res. 2006;21 Suppl 2P28–30. doi:10.1359/jbmr.06s205 ↩
- Kaufmann GA, Sundaram M, McDonald DJ. Magnetic resonance imaging in symptomatic Paget’s disease. Skeletal Radiol. 1991;20(6):413–418. doi:10.1007/BF00191082 ↩
- Kelly JK, Denier JE, Wilner HI, Lazo A, Metes JJ. MR imaging of lytic changes in Paget disease of the calvarium. J Comput Assist Tomogr. 1989;13(1):27–29. ↩
- Stacy G, Dxon L. Pitfalls in MR image interpretation prompting referrals to an orthopedic oncolocy clinic. Radiographics. 2007;27805–828. ↩
- Love C, Din AS, Tomas MB, Kalapparambath TP, Palestro CJ. Radionuclide bone imaging: an illustrative review. Radiographics. 2003;23(2):341–358. doi:10.1148/rg.232025103 ↩
- Smith J, Botet J, Yeh S. Bone sarcoma in Paget disease: a study of 85 patients. Radiology. 1984;152583–590. ↩
- Tilden W, Saifuddin A. An update on imaging of Paget’s sarcoma. Skeletal Radiol. 2021;50(7):1275–1290. doi:10.1007/s00256-020-03682-8 ↩
- Mangham DC, Davie MW, Grimer RJ. Sarcoma arising in Paget’s disease of bone: declining incidence and increasing age at presentation. Bone. 2009;44(3):431–436. doi:10.1016/j.bone.2008.11.002 ↩
- Lopez C, Thomas D, Davies A. Neoplastic transformation and tumor-like lesions in Paget’s disease of bone: a pitorial review. European Radiology. 2003;13L151–163. ↩
- Whitten CR, Saifuddin A. MRI of Paget’s disease of bone. Clin Radiol. 2003;58(10):763–769. doi:10.1016/s0009-9260(03)00264-2 ↩
- Jattiot F, Goupille P, Azais I, Roulot B, Alcalay M, Jeannou J, et al. Fourteen cases of sarcomatous degeneration in Paget’s disease. J Rheumatol. 1999;26(1):150–155. ↩
- McNairn JD, Damron TA, Landas SK, Ambrose JL. Benign tumefactive soft tissue extension from Paget’s disease of bone simulating malignancy. Skeletal Radiol. 2001;30(3):157–160. doi:10.1007/s002560000313 ↩
- Tinns B, Davies A, Mangham D. MR imaging of pseudosarcoma in Paget’s disease of bone: a report of two cases. Skeletal Radiology. 2001;30161–165. ↩
- Reid IR, Miller P, Lyles K, Fraser W, Brown JP, Saidi Y, et al. Comparison of a single infusion of zoledronic acid with risedronate for Paget’s disease. N Engl J Med. 2005;353(9):898–908. doi:10.1056/NEJMoa044241 ↩