Clinical History:
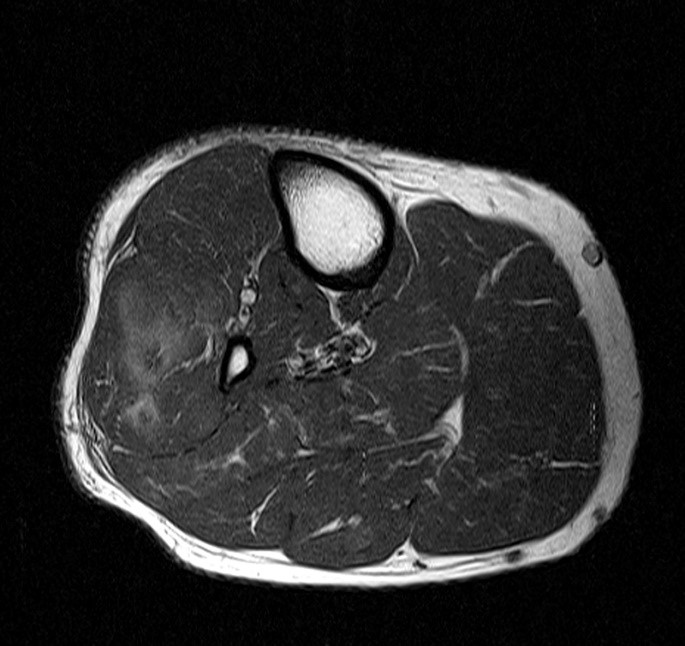
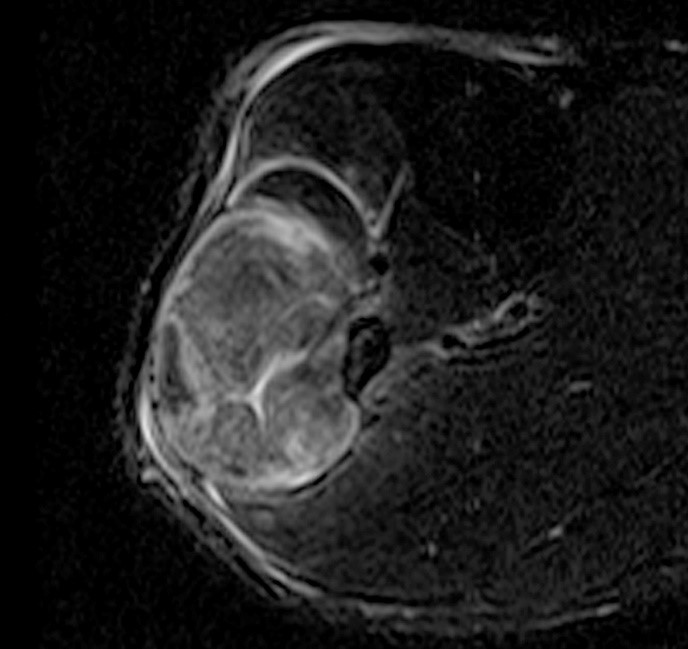
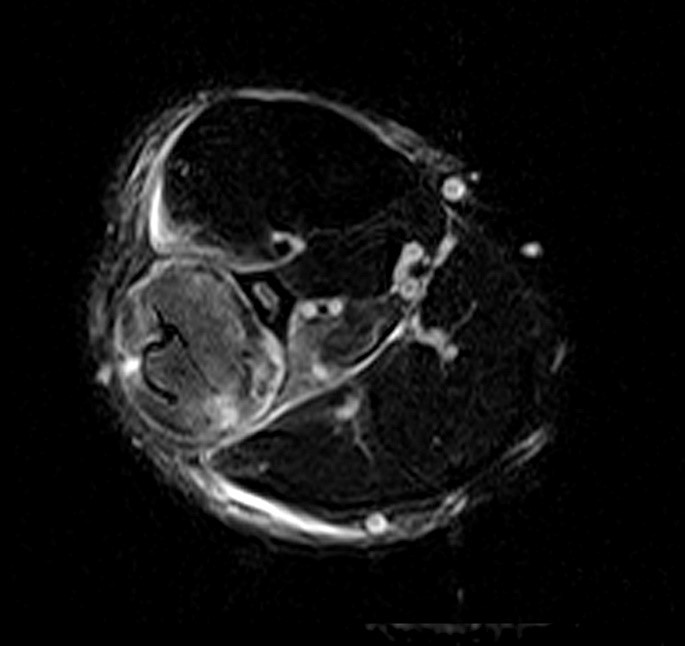
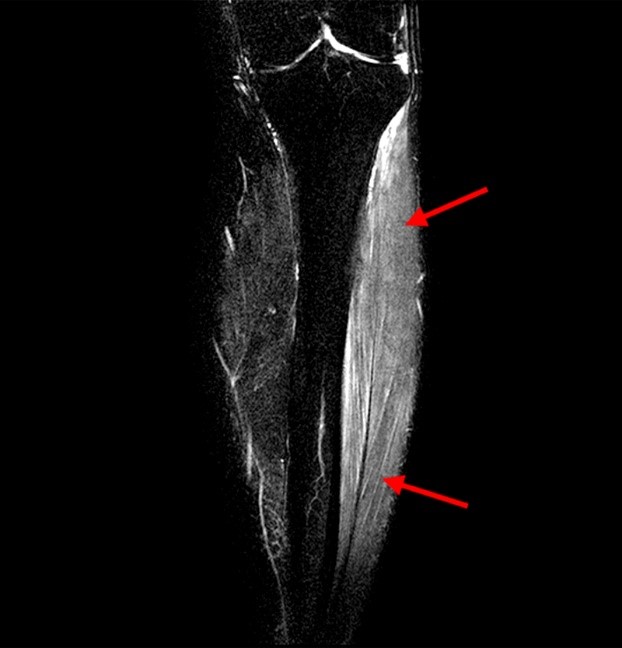
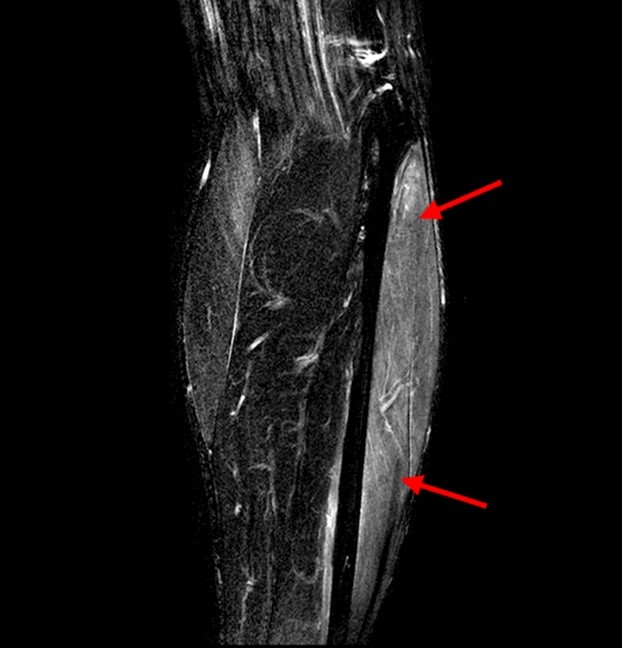
A 32 year-old male presents with right lateral leg pain and swelling. Symptoms began while running a half marathon two weeks ago. (1A) Axial T1-weighted and (1B,C) fat suppressed T2-weighted images are provided. What are the findings? What is your diagnosis?
Findings
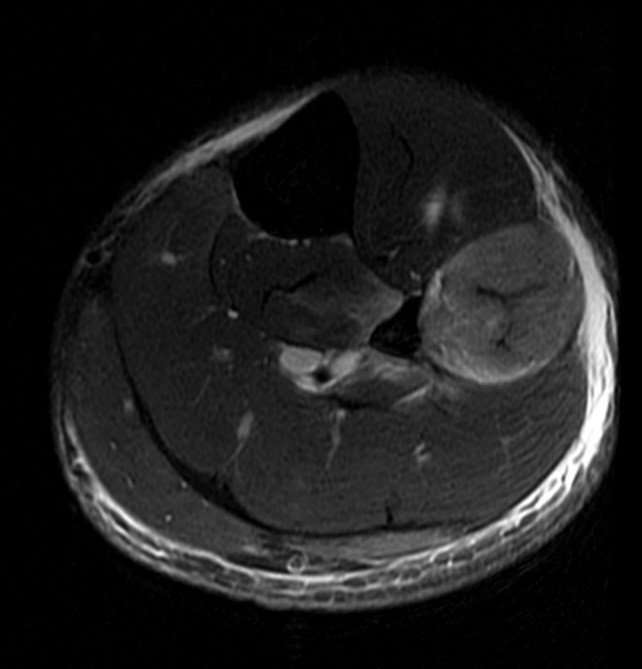
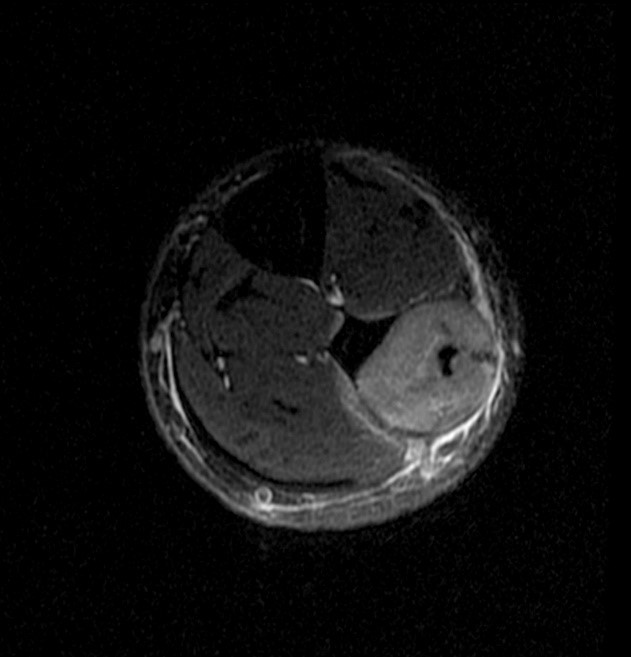
Figure 2:
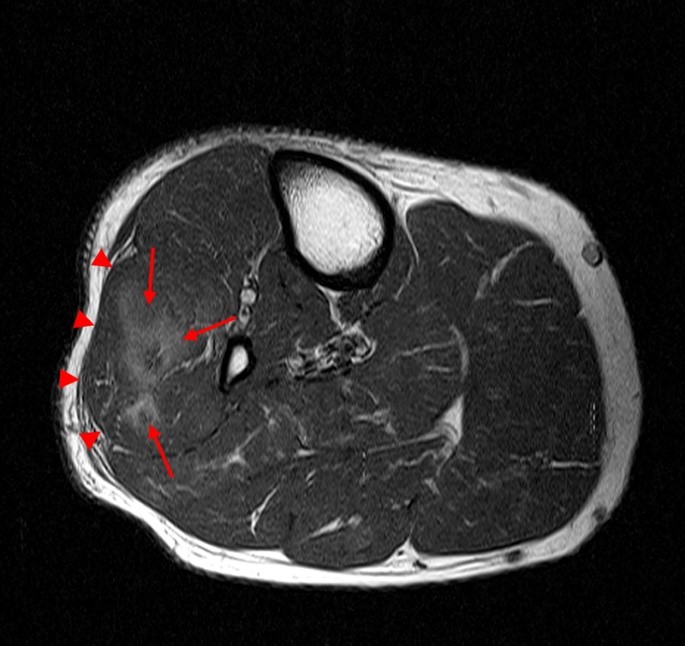
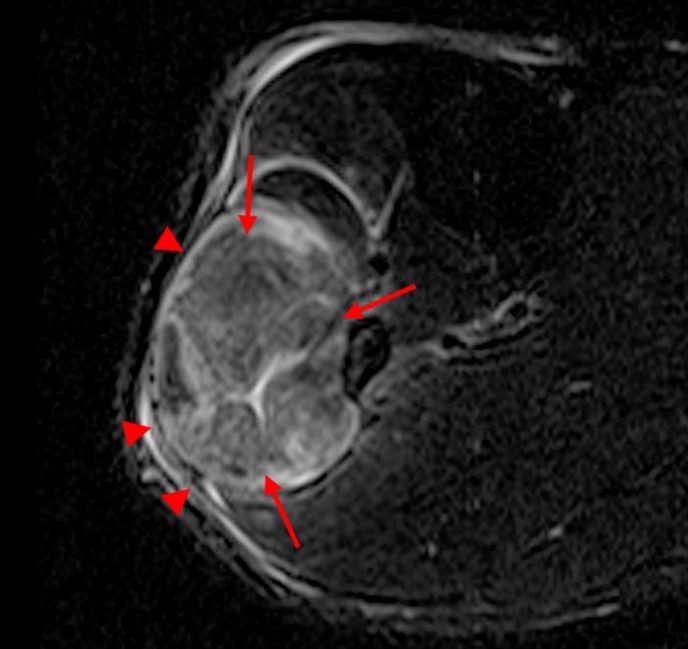
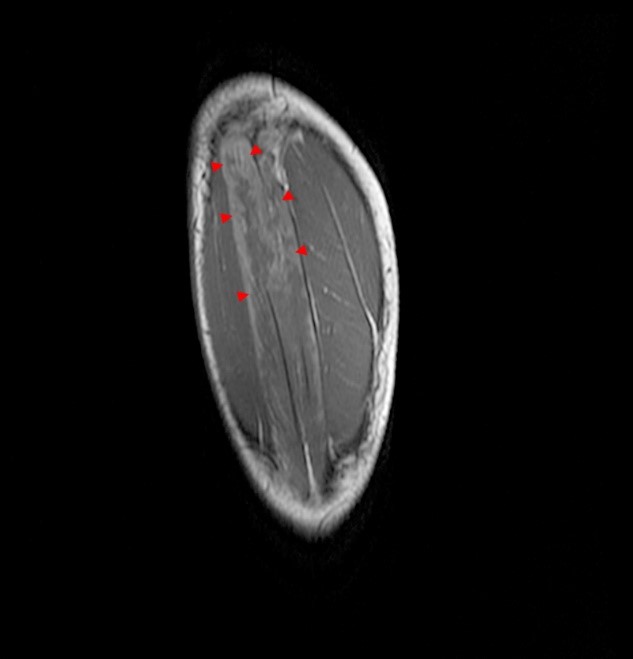
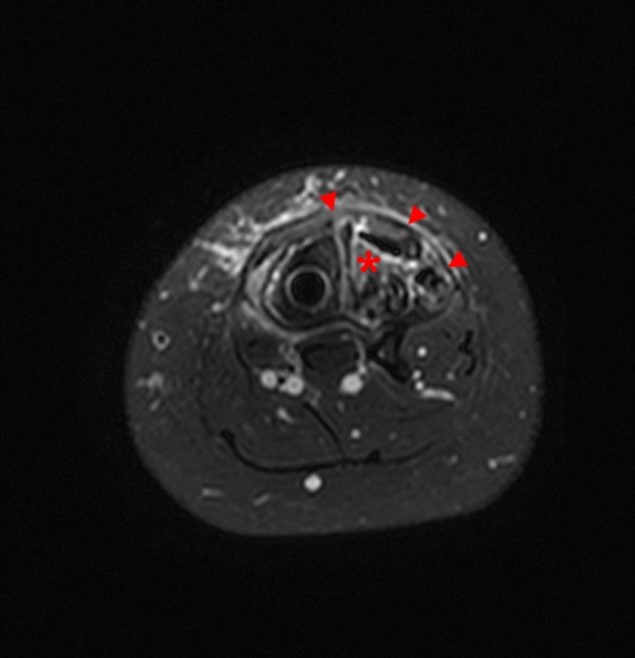
The axial T1-weighted image (2A) demonstrates slight volume increase in the lateral compartment musculature with lateral bowing of the overlying fascia (arrowheads). Areas of diffuse T1 hyperintensity reflect regions of hemorrhage within the muscles (arrows). The more proximal of the axial T2-weighted images (2B) demonstrates diffuse swelling and edema-like signal throughout the lateral compartment (arrows). The fascia is bowed laterally and may be slightly thickened (arrowheads). In addition, edema-like signal is seen adjacent to the fascia within the subcutaneous fat. A more distal T2-weighted image (2C) demonstrates similar findings with more pronounced soft tissue edema and a small fibular fatigue fracture (arrow).
Diagnosis
Acute compartment syndrome involving the lateral compartment of the right leg associated with a fibular fatigue fracture.
Introduction/Background
Compartment syndrome is defined as elevated pressure in a relatively noncompliant anatomic compartment that can cause ischemia, pain and potentially neuromuscular injury, including myonecrosis and rhabdomyolysis.1 If untreated, compartment syndromes may lead to muscle fibrosis and contracture. Muscle ischemia can result when elevated intracompartmental pressure exceeds the intravascular pressure of thin-walled small vessels and these vessels collapse, thereby decreasing the arteriovenous pressure gradient and impeding blood flow. The result is ischemic myoneural necrosis, with increasing edema causing a vicious cycle of increasing compartment pressure ultimately leading to a contracted, useless limb (Volkmann ischemic contracture).
Compartment syndrome is classified generally as acute or chronic. Acute compartment syndrome occurs most commonly in association with fractures, particularly of the tibia, and with soft tissue trauma. Chronic compartment syndrome results most commonly from exertional causes such as exercise or overuse or from nonexertional causes such as a mass lesion, anomalous muscle or infection. Chronic exertional compartment syndrome (CECS) can occur secondary to exertion because chronic muscle activity causes muscle hypertrophy and transiently can swell muscle fibers up to 20 times their resting size, thus causing increased pressure in noncompliant compartments.2
The pathophysiology of CECS is not as clear as that of acute compartment syndrome. Although some studies have provided evidence for muscle ischemia giving rise to the pain associated with CECS, other studies have refuted such a model.3
Additional studies have suggested that patients with CECS have pathologically thickened fascia, which predisposes them to CECS.4
Anatomy
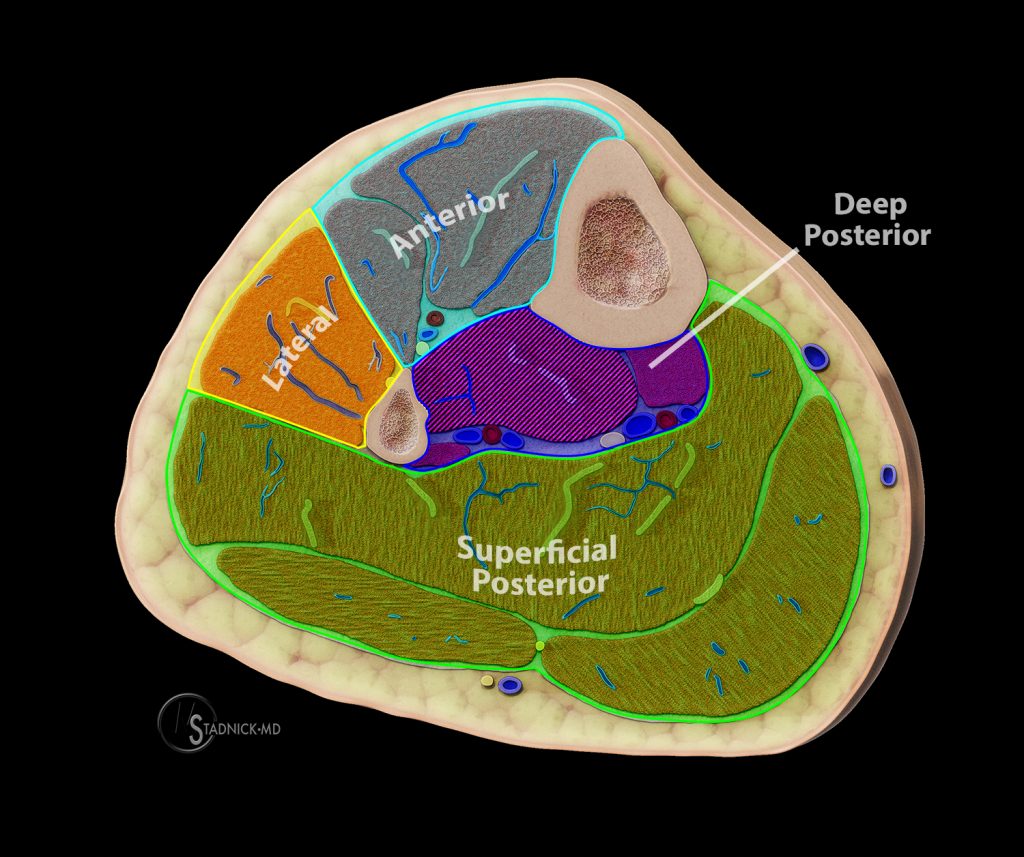
The leg is divided into four anatomic compartments (Figure 3) by fibro-osseous septa which limit the volume and distensibility of each defined compartment: the anterior, lateral, superficial posterior and deep posterior. The tibialis posterior is sometimes classified as its own separate compartment and not grouped with the deep posterior compartment.5
Figure 3:
The leg contains four major myofascial compartments: the anterior compartment, the lateral compartment, the superficial posterior compartment, and the deep posterior compartment. The tibialis posterior (hatched) is sometimes classified as its own separate compartment and not grouped with the deep posterior compartment.
The anterior compartment of the leg is the most common site for acute compartment syndrome. The anterior compartment muscles include the tibialis anterior, the extensor hallucis longus, the extensor digitorum longus and the peroneus tertius. The anterior compartment neurovascular bundle consists of the deep peroneal nerve and the anterior tibial artery. Signs of compartment syndrome affecting the anterior compartment include loss of sensation between the first and second toes and weakness of foot dorsiflexion. Late sequelae include foot drop, claw foot, and deep peroneal nerve dysfunction.
The muscles of the lateral compartment include the peroneus longus and the peroneus brevis. The neurovascular supply to the lateral compartment is provided by the peroneal artery and by branches of the superficial peroneal nerve and the proximal portion of the deep peroneal nerve. Increased pressure in the lateral compartment may produce a deep peroneal nerve deficit, which manifests as weakness in dorsiflexion and inversion of the foot and sensory loss between the first and second toes. As the superficial peroneal nerve also travels in this compartment, sensation to the lower leg and dorsum of the foot may be affected.
The posterior compartment is divided by deep transverse fascia into deep and superficial sections. The superficial posterior compartment muscles include the gastrocnemius, the plantaris and the soleus. No major arteries or nerves are within this compartment. Pain and a palpably tense and tender compartment suggest the diagnosis. The deep posterior compartment muscles include the popliteus, the flexor digitorum longus, the flexor hallucis longus and perhaps the tibialis posterior. The neurovascular supply to the posterior compartment is provided by the tibial nerve and the posterior tibial artery.6 Increased pressure in this compartment may cause plantar foot hypesthesia, weakness of toe flexion and pain with passive extension of the toes.
If a space-occupying lesion, such as neoplasm, edema or hemorrhage occurs in one of these compartments, an increase in intracompartmental pressure develops. When the intracompartmental pressure rises above the capillary pressure, there is impediment to capillary blood flow, resulting in neuromuscular ischemia and severe pain. The anterior compartment is involved 50% to 60% of the time. The deep posterior compartment is the second most commonly involved, occurring 20% to 30% of the time. The remaining 20% of the time, there is involvement of the lateral or superficial posterior compartment. (5) Bilateral leg involvement is common, reportedly occurring in up to 95% of patients with chronic exertional compartment syndrome.7
Clinical Presentation
The initial diagnosis of compartment syndrome is based on the clinical presentation of the syndrome. Patients with acute compartment syndrome present with deep pain or pressure localized to the affected compartment. The history is commonly notable for trauma, fracture, recent surgery, or athletic injury. Classically, physical examination findings associated with acute compartment syndrome have been termed the “6 Ps”:
- pain exacerbated by passive stretch of involved muscle groups
- pallor due to vascular compromise
- paresis due to nerve ischemia
- paresthesias as a result of nerve ischemia
- diminished or absent pulses
- poikilothermia (differing temperatures between the affected limb and the uninjured limb)
Contrary to established dogma regarding pulses in compartment syndrome, distal pulses are usually present in the early stages of the disease because pressures must be very high to occlude flow in the larger arteries.8 The differential diagnosis of acute compartment syndrome includes arterial occlusion, deep vein thrombosis and neuropathy of the affected limb.
Patients with CECS present with chronic, exercise-related pain. Typically, patients are young and athletic; the mean age in one study was 26.9 The pain may be described as ‘pressure’, ‘aching’ or ‘sharp’ and localizes to the involved compartment. Historically, the pain occurs after exercise of the involved muscles and resolves with rest. The pain may progress over time, causing the patient to be able to tolerate progressively less exercise before the onset of pain. Other symptoms of CECS include muscle weakness and paresthesias that are likely related to compromised nerve function.
Physical examination is typically unrevealing in patients with CECS, particularly when they are asymptomatic. Pain to palpation of involved muscles, muscle hypertrophy, and palpation of muscle hernias (which occur in 20% to 60% of patients) are possible physical exam findings.4,9
There is a broad differential diagnosis for CECS, particularly in a patient presenting with leg pain. The differential includes muscle strain injury, delayed onset muscle soreness, medial tibial stress syndrome, stress fracture, tendon pathology, peripheral vascular disease, peroneal nerve apraxia, peripheral neuropathy, deep vein thrombosis, spinal stenosis, and lumbar disc herniation.
Evaluation
Diagnostic tests used to evaluate for compartment syndrome include compartment pressure measurements, near-infrared spectroscopy, and imaging examinations. The gold standard diagnostic modality for acute and chronic compartment syndromes is intracompartmental pressure measurements. In the acute setting, such measurements may be made with a variety of devices. Elevated intracompartmental pressure confirms the diagnosis of acute compartment syndrome. Controversy exists as to what is considered a ‘threshold pressure’ above which a clinically significant compartment syndrome exists and how long such a pressure must be sustained within a compartment for damage to occur. However, studies have shown that the threshold pressure is around 30 to 40 mm Hg.8
There are some initial reports of the promising value of near-infrared spectroscopy in the detection of muscle deoxygenation in acute compartment syndrome.10,11 This is a non-invasive optical technique that can monitor the local muscle oxygenation and perfusion in real time. This method can measure changes in local muscle oxygen saturation and thus has the potential to detect and provide continuous monitoring of intracompartmental ischemia and hypoxia. Near-infrared spectroscopy may, in the future, provide the benefit of a rapid, continuous, noninvasive tool that is sensitive and specific for the early detection of acute compartment syndrome. Technical development and clinical validation of this diagnostic method are currently underway.
Acute compartment syndromes represent a situation in which limbs are acutely threatened. It is for this reason that imaging evaluations are rarely undertaken in this setting. However, magnetic resonance imaging has been shown to be sensitive for detecting nonspecific changes in muscle attributable to compartment syndrome and could therefore be used to evaluate patients for compartment syndrome when clinical doubt exists.12,13
The lack of physical examination findings in patients with CECS has prompted many investigators to validate objective means to make the diagnosis. In contrast to acute compartment syndrome, where routine radiographs are of limited value, radiographs are indicated to evaluate for osseous pathology such as stress fractures. As in acute compartment syndrome, the gold standard diagnostic modality for CECS is intracompartmental pressure measurement. In CECS, investigators have emphasized pressure measurements after exercise, preferably that which reproduces symptoms. The most widely accepted pressure measurement criteria for CECS are those set forward by Pedowitz et al.9 Their study identified three criteria: a pre-exercise resting intracompartmental pressure ≥ 15mm Hg, a 1-minute post-exercise pressure > 30mm Hg, or a 5-minute post-exercise pressure > 20mm Hg. The study suggested that application of these criteria combined with a history suggestive of CECS should result in less than a 5% false-positive rate in the diagnosis. The authors recommend repeat pressure measurements and clinical judgement in equivocal cases.
MR Imaging
Cross-sectional imaging may complement direct pressure measurements by providing noninvasive data on the compartment or compartments that are involved, particularly in the non-acute setting. Proposed indications for MR imaging include the diagnostic assessment of atypical cases (e.g. those with an uncommon location or cause for compartment syndrome or borderline compartment pressure measurements) and evaluation for the possibility of an underlying lesion such as a soft tissue hematoma or neoplasm that may contribute to increased compartment pressure and need to be addressed at surgery.14,15
Furthermore, the lack of abnormal signal intensity in muscle on MR images may help exclude the diagnosis of compartment syndrome.
Magnetic resonance imaging can be used to study the location and extent of ischemic damage to muscle. MR techniques using axial inversion recovery or fat-suppressed T2-weighted or proton density-weighted sequences are sensitive to early changes of muscle compartment edema, which is seen as an infiltrative or feather-like pattern of hyperintensity (Figure 4).
Figure 4:
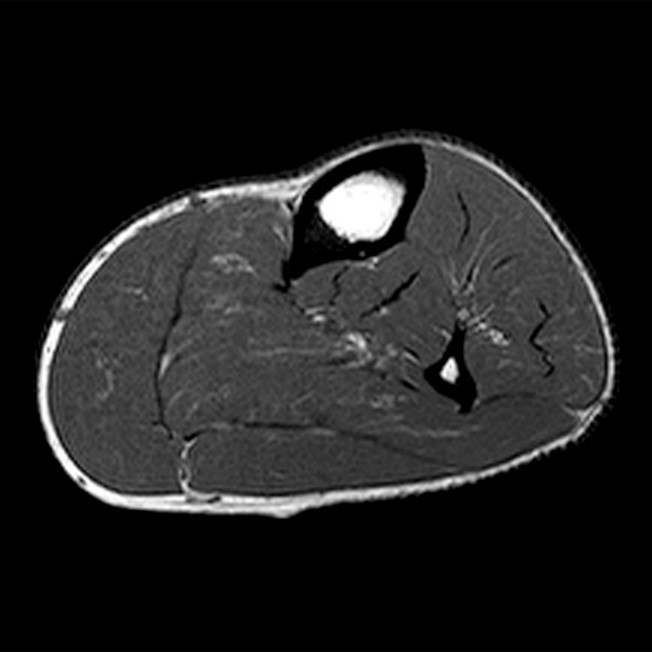
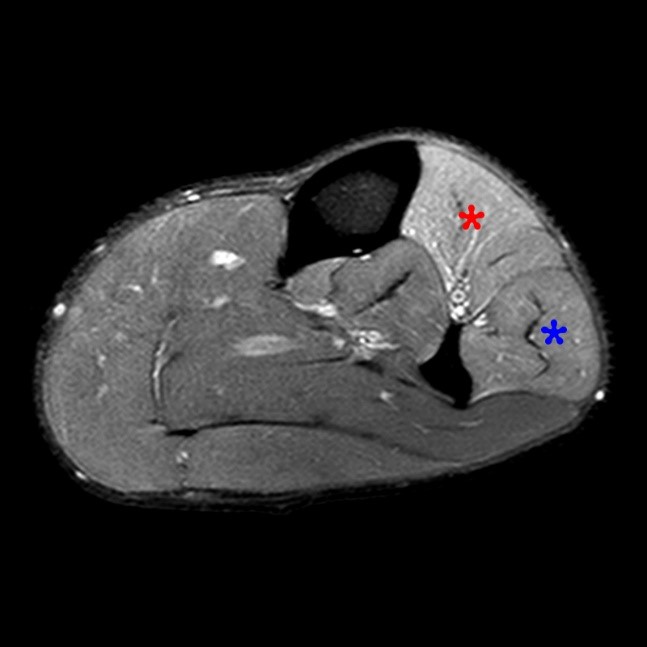
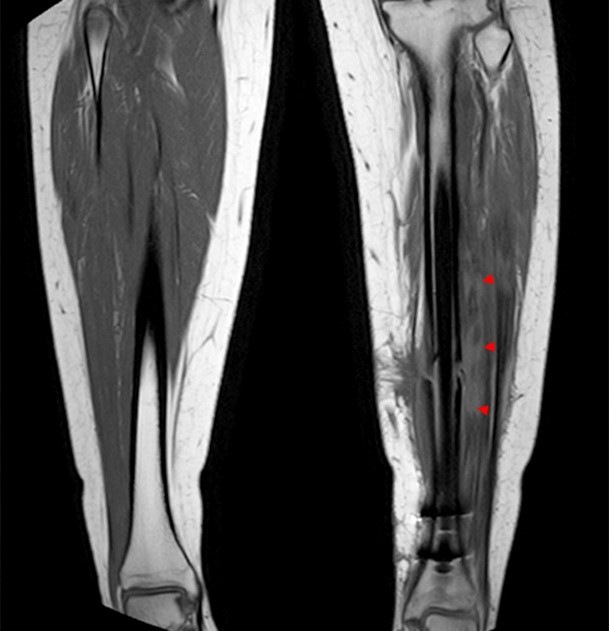
Images from an MR examination of the left leg obtained on a 45 year-old male with pain in the proximal to mid portions of the leg. The axial T1-weighted image (4A) suggests slight bowing of the fascia over the anterior and lateral compartments. However, in this muscular individual with a limited amount of subcutaneous fat, this is uncertain. No intramuscular hemorrhage is identified. The axial fat suppressed proton-density weighted image (4B) demonstrates increased signal within the anterior compartment (red asterisk) and less pronounced signal increase in the lateral compartment (blue asterisk). Coronal fat suppressed T2-weighted images through the anterior (4C) and lateral (4D) compartments confirm the findings (arrows). The findings are compatible with chronic exertional compartment syndrome involving the anterior and lateral compartments of the leg.
Bulging and T2 hyperintensity may also be seen in the adjacent fascia [Figures 1, 5]. Other MR imaging findings observed in the setting of acute and chronic compartment syndrome include the following: 14,16,17
- Increased signal intensity on fat-suppressed T2-weighted images resulting from increased interstitial water or edema (Figures 1, 4) (acute or chronic compartment syndrome)
- Increased signal intensity on T1-weighted images indicating foci of hemorrhage (Figures 1, 5, 6) (acute compartment syndrome) or fatty infiltration (sequela of established compartment syndrome)
- Decreased signal intensity on T1-weighted images caused by fibrosis or dystrophic calcification (Figure 7) (sequela of established compartment syndrome)
- Increased muscle volume caused by muscle hypertrophy, swelling, or both (Figures 1, 5, 6) (acute or chronic compartment syndrome)
- Decreased muscle volume related to atrophy, fibrosis, or both (sequela of established compartment syndrome)
- Fascial thickening (Figures 1, 6) (sequela of established compartment syndrome)
- Herniation of muscle through a tear in the surrounding fascia (Figure 8) (acute or chronic compartment syndrome)
Figure 5:
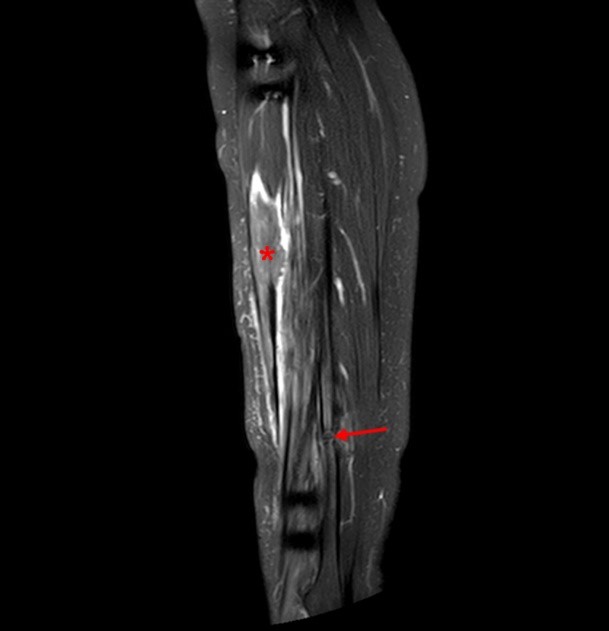
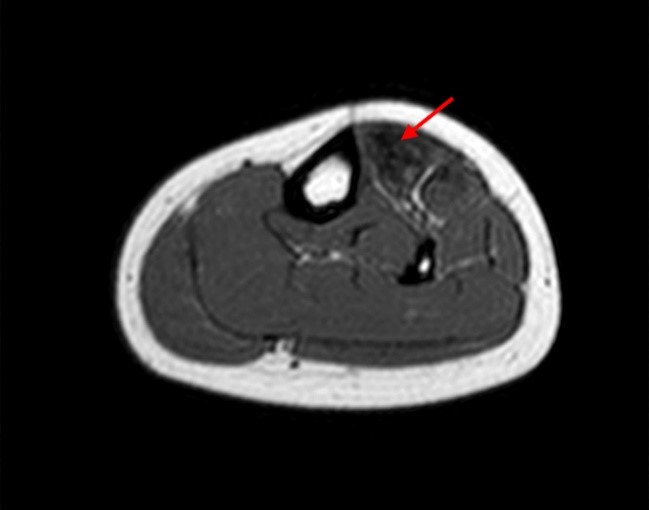
Images from the MR examination of a 24 year-old male with a recent injury playing football. Axial fat suppressed proton density weighted images are presented from the proximal (5A), mid (5B) and distal (5C) aspects of the leg. These demonstrate diffuse swelling and edema within the lateral compartment musculature (arrows) with marked adjacent soft tissue swelling and edema. The sagittal T1-weighted image (5D) demonstrates areas of high signal hemorrhage (arrowheads) within the proximal aspect of the lateral compartment muscles with irregularity of the proximal muscle fibers. A corresponding sagittal inversion recovery image (5C) demonstrates diffuse muscle edema with irregularity of the proximal muscle fibers (arrows). The appearance reflects acute compartment syndrome related to strains of the lateral compartment musculature.
Figure 6:
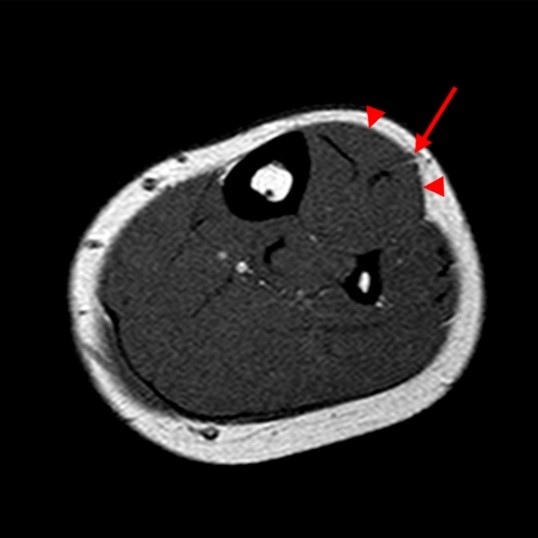
Images from an MR examination performed on a 15 year-old male with left leg pain following treatment of tibial and fibular fractures. The coronal T1-weighted image (6A) reveals a statically locked intramedullary nail within the left tibia. Irregular regions of increased T1 signal in the anterior compartment musculature reflect regions of hemorrhage (arrowheads). The sagittal fat suppressed T2-weighted image (6B) demonstrates a fibular fracture (arrow) and extensive edema within the anterior compartment muscles (asterisk). The axial T1-weighted image (6C) again reveals areas of high signal hemorrhage (arrowheads) within the anterior compartment muscles along with swelling of the anterior compartment. The axial fat suppressed T2-weighted image (6D) demonstrates swelling and edema in the anterior compartment (asterisk). In addition, the overlying fascia is thickened and edematous (arrowheads). The findings are compatible with acute compartment syndrome involving the anterior compartment.
Figure 7:
Images from an MR examination of a 15 year-old female who injured her left leg one month ago playing volleyball. Axial T1-weighted (7A) and inversion recovery (7B) images demonstrate heterogeneous low signal intensity within the anterior compartment (arrows), more significant within the tibialis anterior muscle. This reflects an area of muscle fibrosis in this patient with chronic exertional compartment syndrome.
Figure 8:
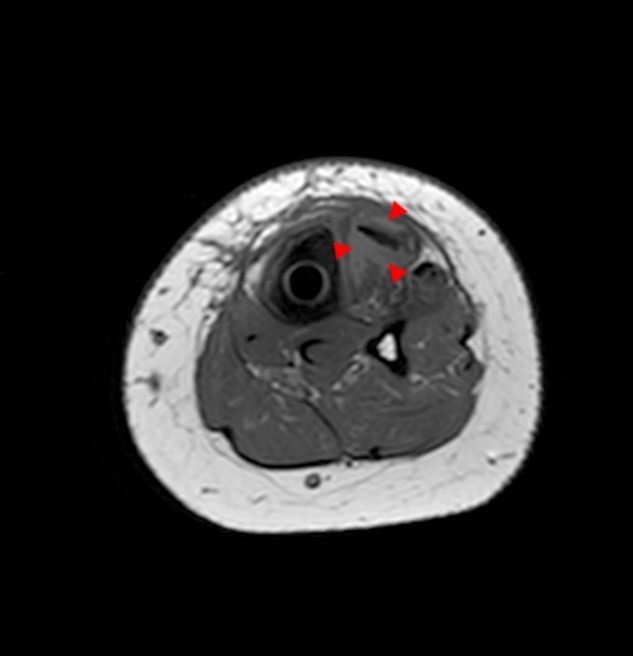
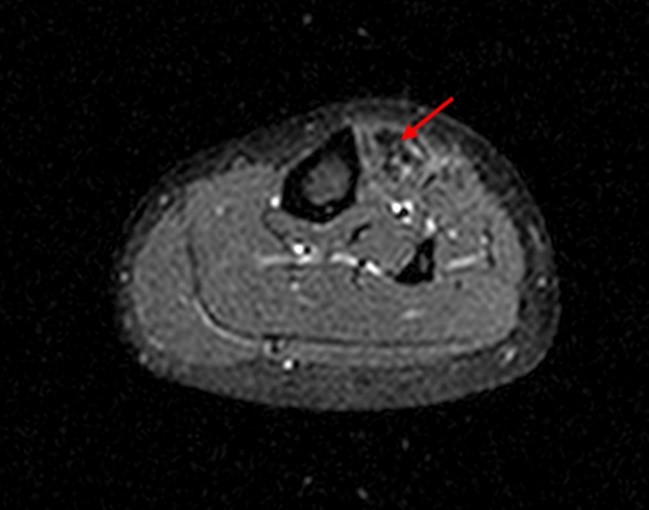
Images from an MR examination of a 16 year-old female with pain and a small lump that occurs after running. The examination was performed following exercise that reproduced the patient’s symptoms. Axial T1-weighted image (8A) demonstrates increased volume of the anterior compartment with bowing of the overlying fascia (arrowheads). In addition, a small fascial hernia (arrow) is seen which corresponds to the patient’s palpable lump. An axial fat suppressed proton-density weighted image (8B) demonstrates swelling and increased signal of the anterior compartment musculature suggesting chronic exertional compartment syndrome (asterisk).
With acute compartment syndrome, diffuse edema-like signal and mildly increased muscle girth are common, occasionally with foci of hemorrhage.
In patients with impending compartment syndrome, gadolinium-enhanced T1-weighted images typically show avid contrast enhancement in the affected muscles.14 This enhancement can be useful in distinguishing between perfused and devitalized muscle.
With chronic exertional compartment syndrome, diffuse edema-like signal and mildly increased muscle girth are common after exercise (Figure 9). The change in T2 signal intensity between pre-exercise and post-exercise images is significantly greater in compartments with post-exercise hypertension.18
For example, in a study of 41 anterior compartments with CECS, T2 signal intensity increased by 28% (range 14-39%) following exercise, compared to an increase of 8% (range 0-10%) in uninvolved superficial posterior compartments.15 Furthermore, the magnitude of the change in signal intensity correlates significantly with the change in pre-exercise and post-exercise pressure measurements, as well as with the absolute post-exercise pressure.
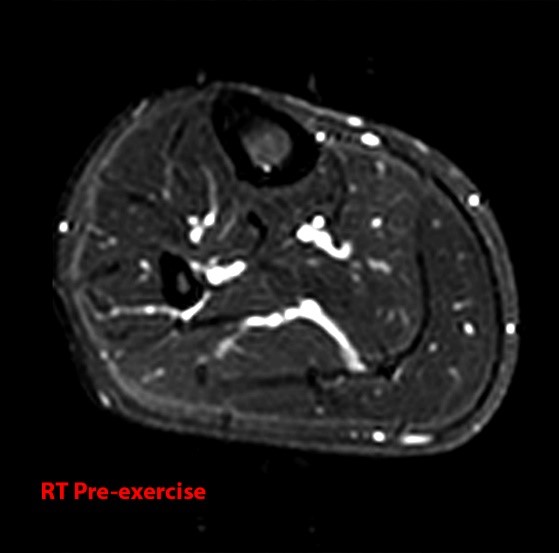
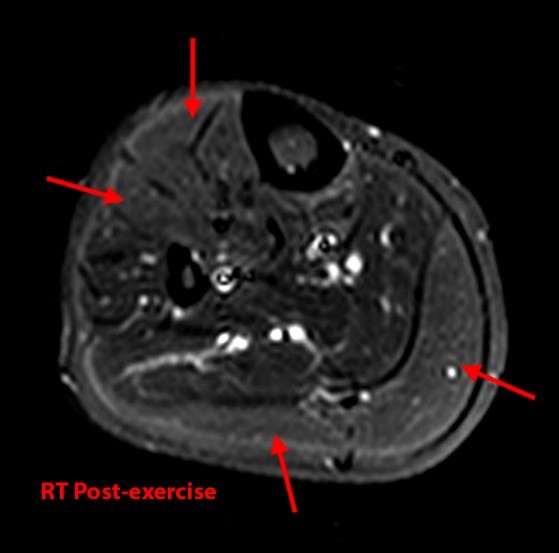
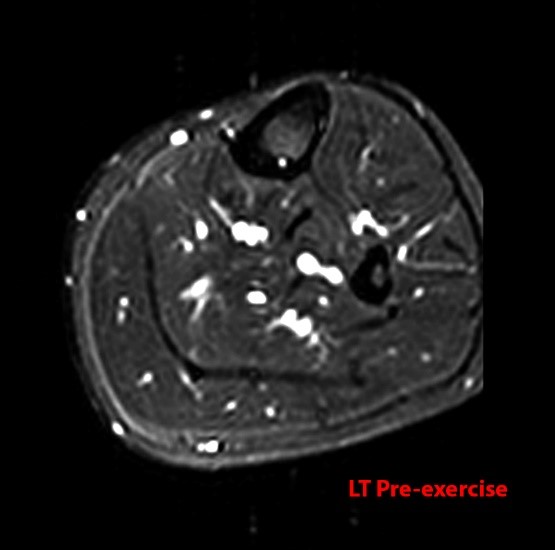
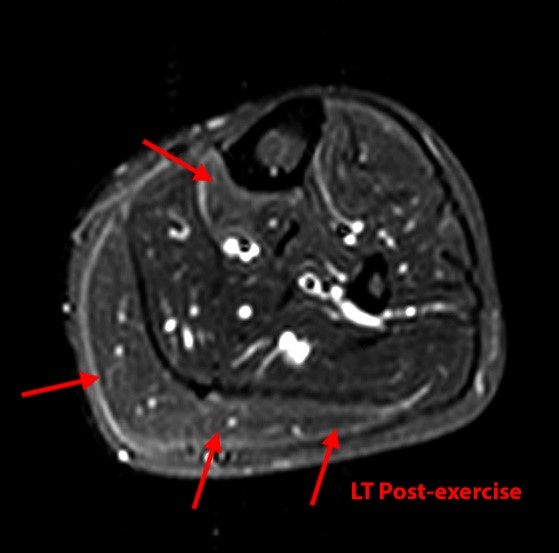
Figure 9:
Images from the MR examination of a 25 year-old male who presented with bilateral lower extremity pain with exercise. Axial inversion recovery images were obtained through both legs both before and after exercise. Image 9A is the right leg pre-exercise and image 9B is the right leg post-exercise. Images 9C and 9D are the pre- and post-exercise images of the left leg. Areas of increased signal intensity are seen within the anterior and superficial posterior compartments of the right leg (arrows). On the left, increased signal is seen within the superficial and deep posterior compartments (arrows). The findings are compatible with chronic exertional compartment syndrome.
Although MR imaging may be sensitive in the evaluation of compartment syndrome, it is not specific. Depending on the clinical context, the imaging differential diagnosis may include other causes of painful, swollen extremities, such as delayed onset muscle soreness, muscle strain, deep venous thrombosis, cellulitis and lymphedema. Like compartment syndrome, deep venous thrombosis may result in muscle edema, particularly in the deep posterior compartment of the leg. However, unlike compartment syndrome, deep venous thrombosis causes venous occlusion and commonly results in subcutaneous edema and skin thickening, which are all findings that can be displayed with MR imaging. Cellulitis and lymphedema show prominent subcutaneous edema and skin thickening on MR images, but swelling and abnormal signal intensity centered in muscle often are absent.
The clinical differential diagnosis of stress-related anterior leg pain commonly includes CECS, bone stress injury and traction periostitis. Interestingly, these three entities may occur separately or together. For example, in a recent study of 44 cases, there was no significant difference in bone findings between patients with and without CECS.19
Treatment and Prognosis
The goal of treatment of acute compartment syndrome is to relieve the pressure and preserve the neuromuscular function of the affected limb. Although the definitive treatment for compartment syndromes is fasciotomy of the affected compartment, some compartment syndromes have been shown to resolve with non-operative management.20 If non-operative management fails, immediate fasciotomy of the affected compartment is indicated to salvage the use of the limb.8
As in acute compartment syndrome, the definitive treatment of CECS is fasciotomy of involved compartments. Although non-operative management may be attempted in CECS patients willing to decrease their exercise routine, the benefits of pain-free exercise generally outweigh the risks of surgery.
Many studies have been performed to examine outcomes of surgical management of CECS. Although the studies are difficult to compare because of different examined outcomes, most report postoperative success over the follow-up period. In one series of 100 patients, of which 70 were treated with fasciotomy, surgical treatment resulted in a 90% rate of cure or significant functional improvement at 4.5 months after surgery.21
In another study, fasciotomy was performed in 53 patients with convincing history and elevated compartment pressure measurements and 18 patients with convincing history only. The study reports a marked reduction in symptoms in 87% of patients at 2 years postoperatively.22
These studies concur with the usual success rates in studies of operative treatment of CECS that ranged from 42% to 92% in a recent review.4
Summary
Familiarity with the MR imaging appearance of compartment syndrome is important, given that imaging may be performed for assessment of pain that initially is thought to be related to other causes, such as stress fracture, muscle strain or soft tissue neoplasm. Potential complications of compartment syndrome not only include contractures, but also myonecrosis, rhabdomyolysis, renal failure and even death. It is therefore important for the radiologist to consider this diagnosis in patients who present for non-specific or exercise-related lower extremity pain.
References
- Elliott KG, Johnstone AJ, Diagnosing acute compartment syndrome. J Bone Joint Surg Br 2003;85:625-32. ↩
- Fraipoint MJ, Adamson GJ. Chronic exertional compartment syndrome. J Am Acad Orthop Surg 2003;11:268-276. ↩
- Amendola A, et al. The use of magnetic resonance imaging in exertional compartment syndromes. Am J Sports Med 1990;18:29-34. ↩
- Shah SN, Miller BS, Kuhn JE. Chronic exertional compartment syndrome. Am J Orthop 2004;33:335-341. ↩
- Davey JR, Rorabeck Ch, Fowler PJ. The tibialis posterior muscle compartment: An unrecognized cause of exertional compartmental syndrome. Am J Sports Med 1984;12:391-397. ↩
- Anderson MW, Temple HT, Dussault RG, et al. Compartmental anatomy: relevance to staging and biopsy of musculoskeletal tumors. Am J Roentgenol 1999;173:1163-1171. ↩
- Reneman RS. The anterior and the lateral compartment syndrome of the leg due to intensive use of muscles. Clin Orthop 1975;113:69-80. ↩
- Mubarak SJ, Pedowitz RA, Hargens AR. Compartment syndromes. Curr Orthop 1989;3:36-40. ↩
- Pedowitz HA, Hargens AR, Mubarak SJ, Gershundi DH. Modified criteria for the objective diagnosis of chronic compartment syndrome of the leg. AM J Sports Med 1990;18:35-40. ↩
- Garr JL, Gentilello LM, Cole PA, et al. Monitoring for compartmental syndrome using near-infrared spectroscopy: a non-invasive continuous transcutaneous monitoring technique. J Trauma 1999;46:613-618. ↩
- Tobias JD, Hoernschemeyer DG. Near-infrared spectroscopy identifies compartment syndrome in an infant. J Pediatr Orthop 2007;27:311-313. ↩
- Boutin RD, Fritz RC, Steinbach LS. Imaging of sports-related muscle injuries. Radiol Clin North Am 2002;40:333-362. ↩
- De Marchi A, Robba T, Ferrarese E, Faletti C. Imaging in musculoskeletal injuries: state of the art. Radiol Med (Torino) 2005;110:115-131. ↩
- Rominger MB, Lukosch CJ, Bachmann GF. MR imaging of compartment syndrome of the lower leg: a case control study. Eur Radiol 2004;14:1432-1439. ↩
- Verleisdonk EJ, van Gils A, van der Werken C. The diagnostic value of MRI scans for the diagnosis of chronic exertional compartment syndrome of the lower leg. Skeletal Radiol 2001;30:321-325. ↩
- May DA, Disler DG, Jones EA, Balkissoon AA, Manaster BJ. Abnormal signal intensity in skeletal muscle at MR imaging: patterns, pearls, and pitfalls. Radiographics 2000;20:S295-S315. ↩
- McDonald S, Bearcroft P. Compartment syndromes. Semin Musculoskelet Radiol 2012;14:236-244. ↩
- Omar HA, Helms CA, Bytomski J. et al. The role of MRI in the diagnosis of chronic exertional compartment syndrome. AJR 2006;186(supl). ↩
- Kiuru MJ, Mantysaari MJ, Pihlajamaki HK, et al. Evaluation of stress-related anterior lower leg pain with magnetic resonance imaging and intracompartmental pressure measurement. Mil Med 2003;168:48-52. ↩
- Robinson D, On E, Halperin N. Anterior compartment syndrome of the thigh in athletes – indications for conservative treatment. J Trauma 1992;32:183-186. ↩
- Detmer DE, Shapre K, Sufit RL, Girdley FM. Chronic compartment syndrome: diagnosis, management, and outcomes. Am J Sports Med 1985;13:162-170. ↩
- Verleisdonk EJ, Schmitz RF, van der Werken C. Long-term results of fasciotomy of the anterior compartment in patients with exercise-induced pain in the lower leg. Int J Sports Med 2004;25:224-229. ↩