Clinical History
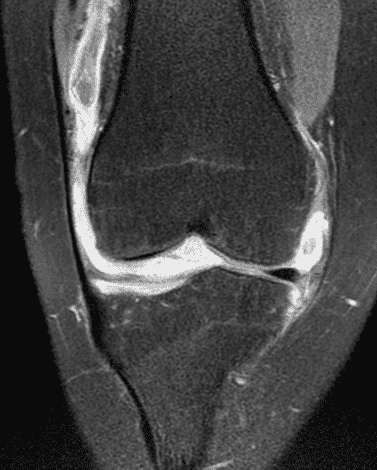
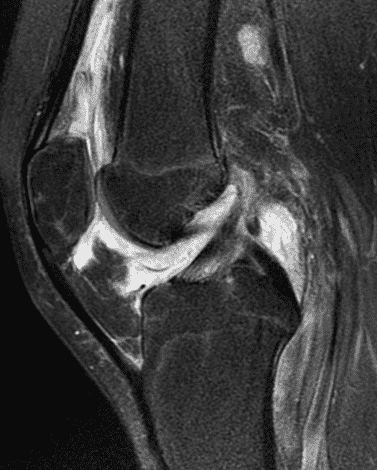
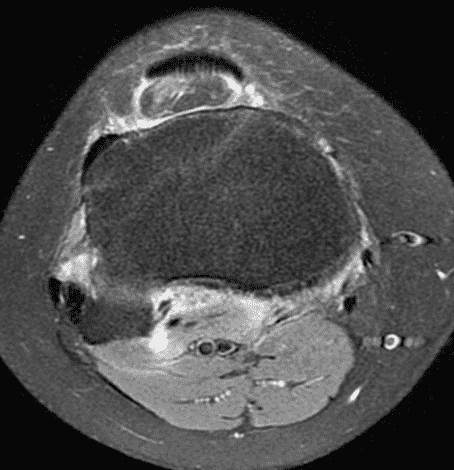
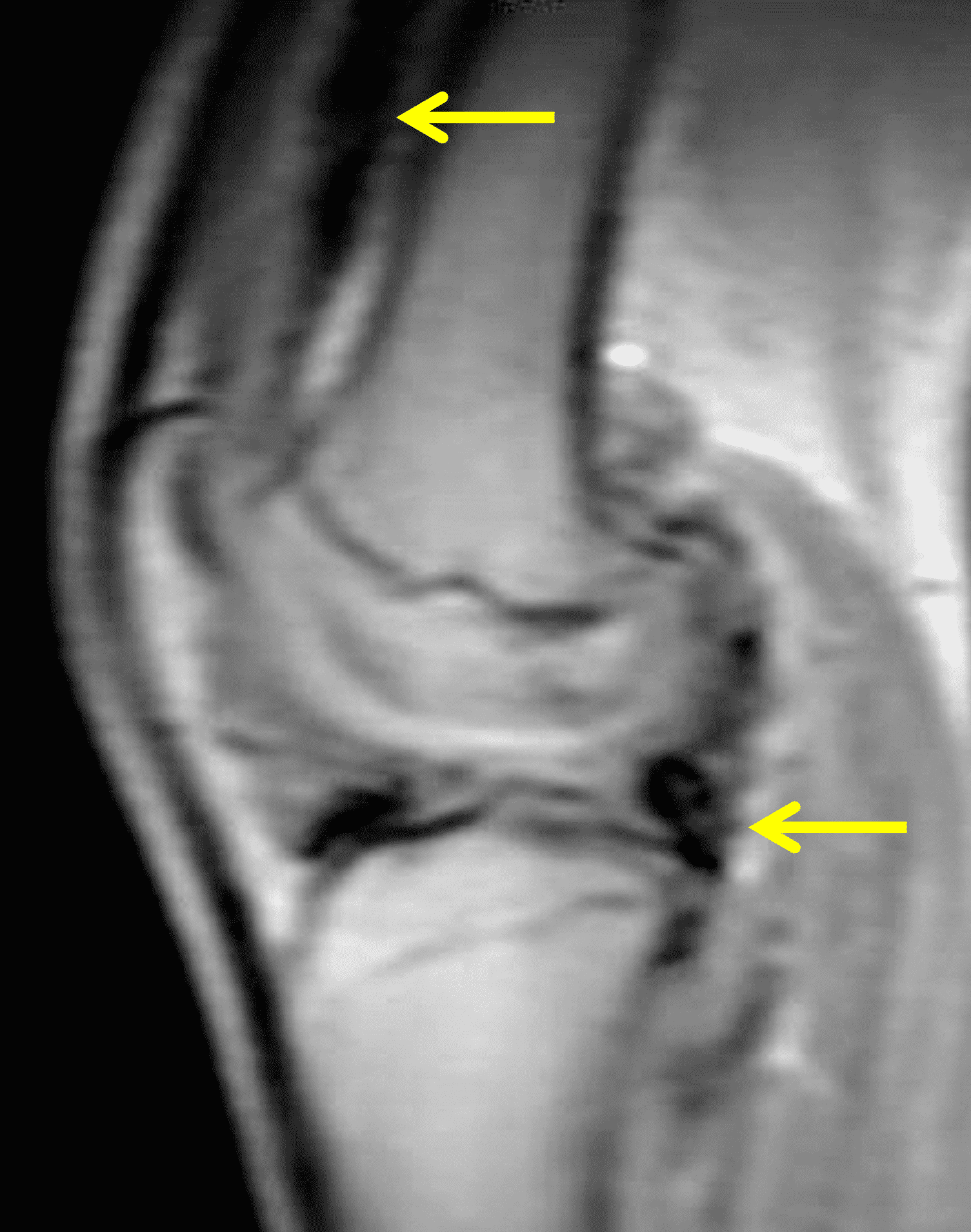
A 19 year-old woman complains of 9 months of unilateral right knee pain and persistent knee swelling without known injury or systemic symptoms. Coronal (1A), sagittal (1B), and transverse (1C) fat-suppressed T2-weighted images of the right knee are provided. What are the findings? What is your differential diagnosis?
Findings
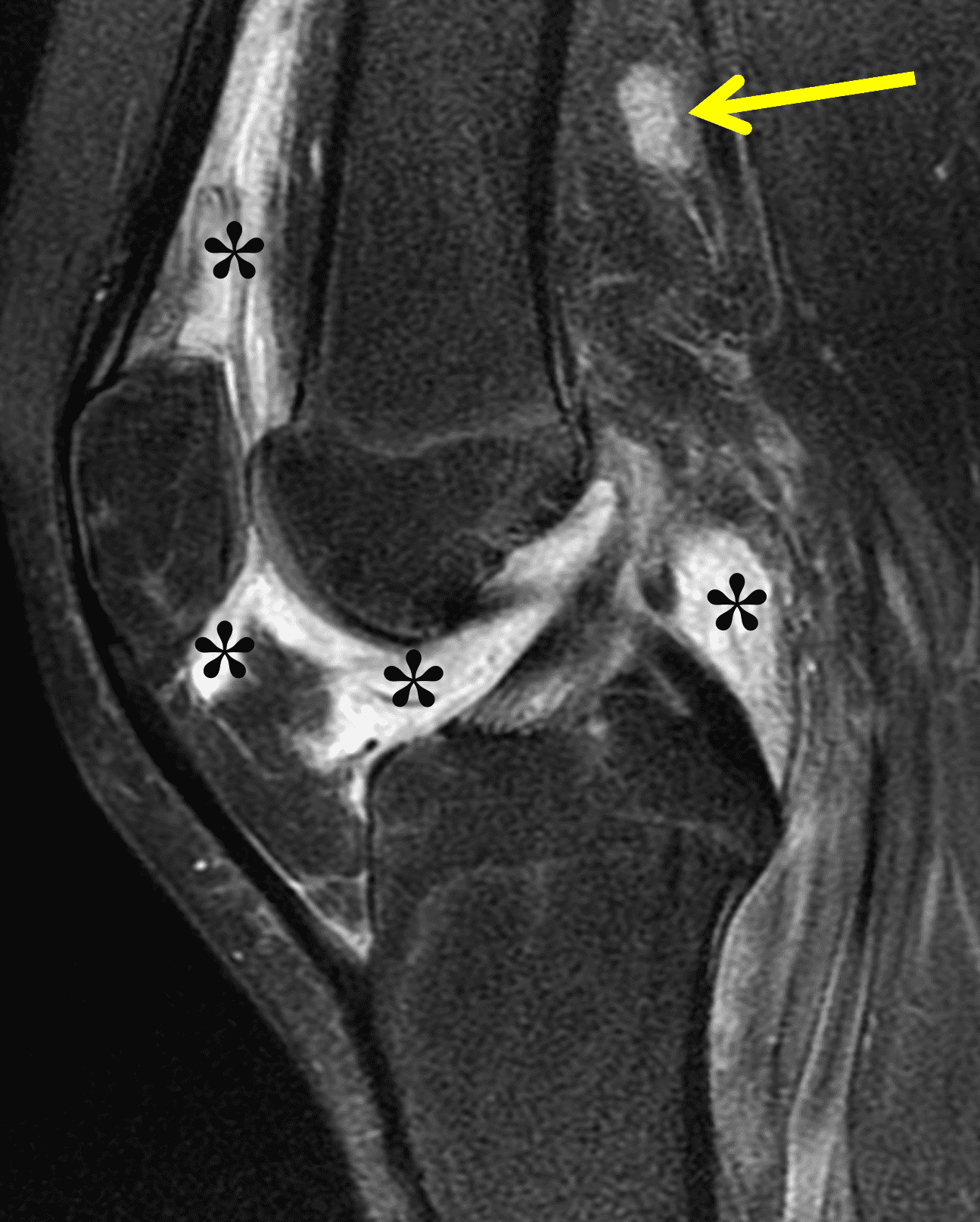
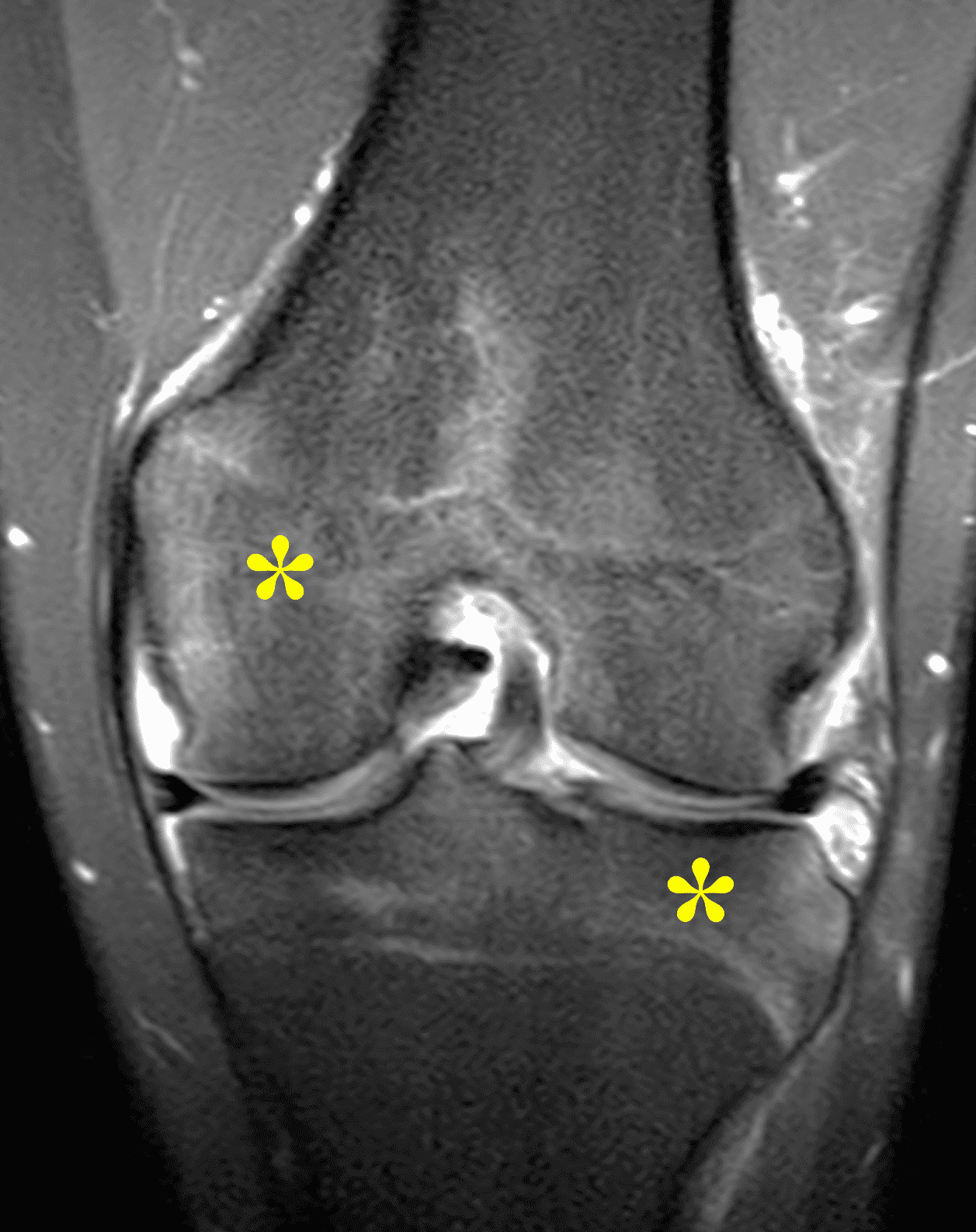
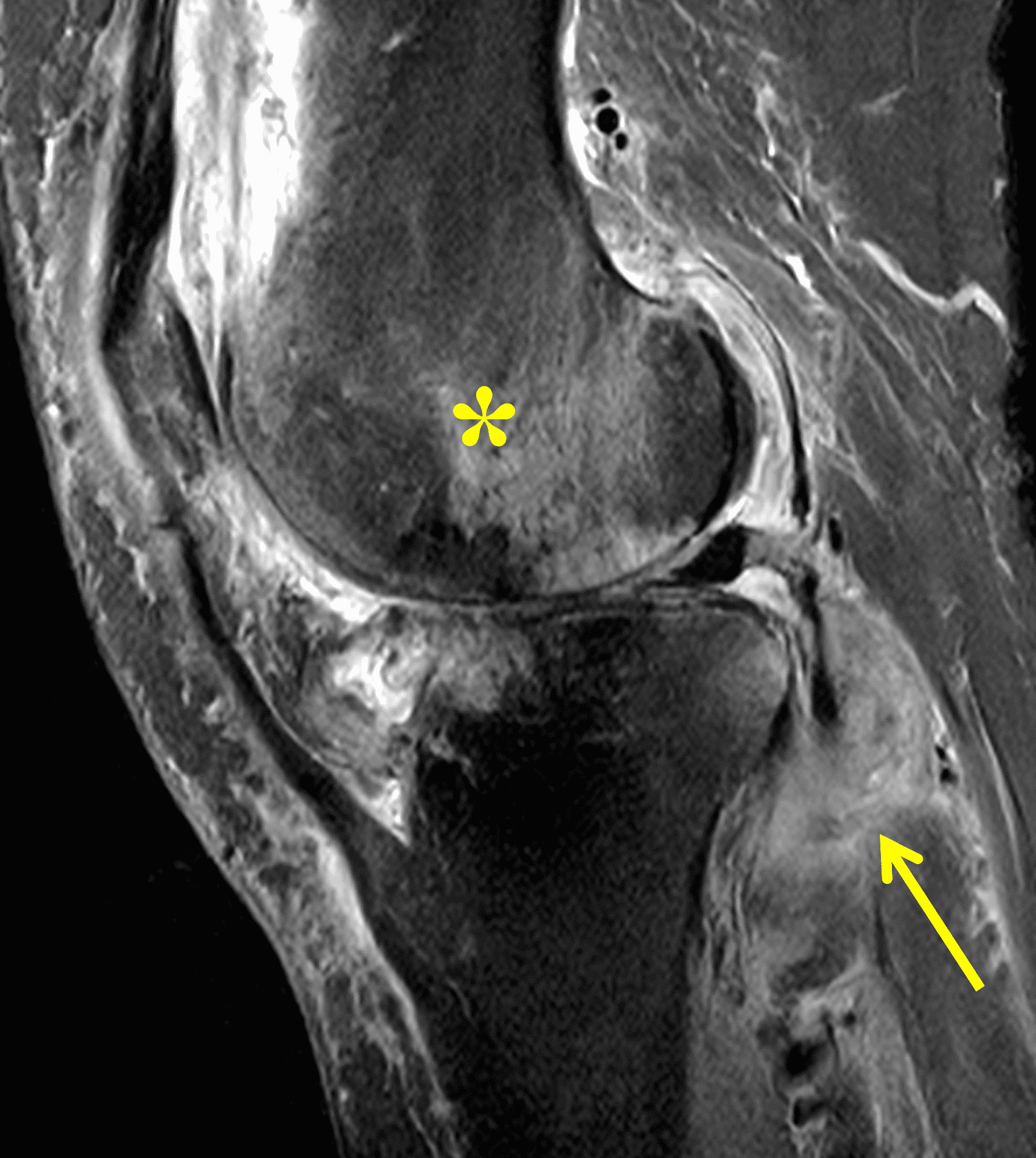
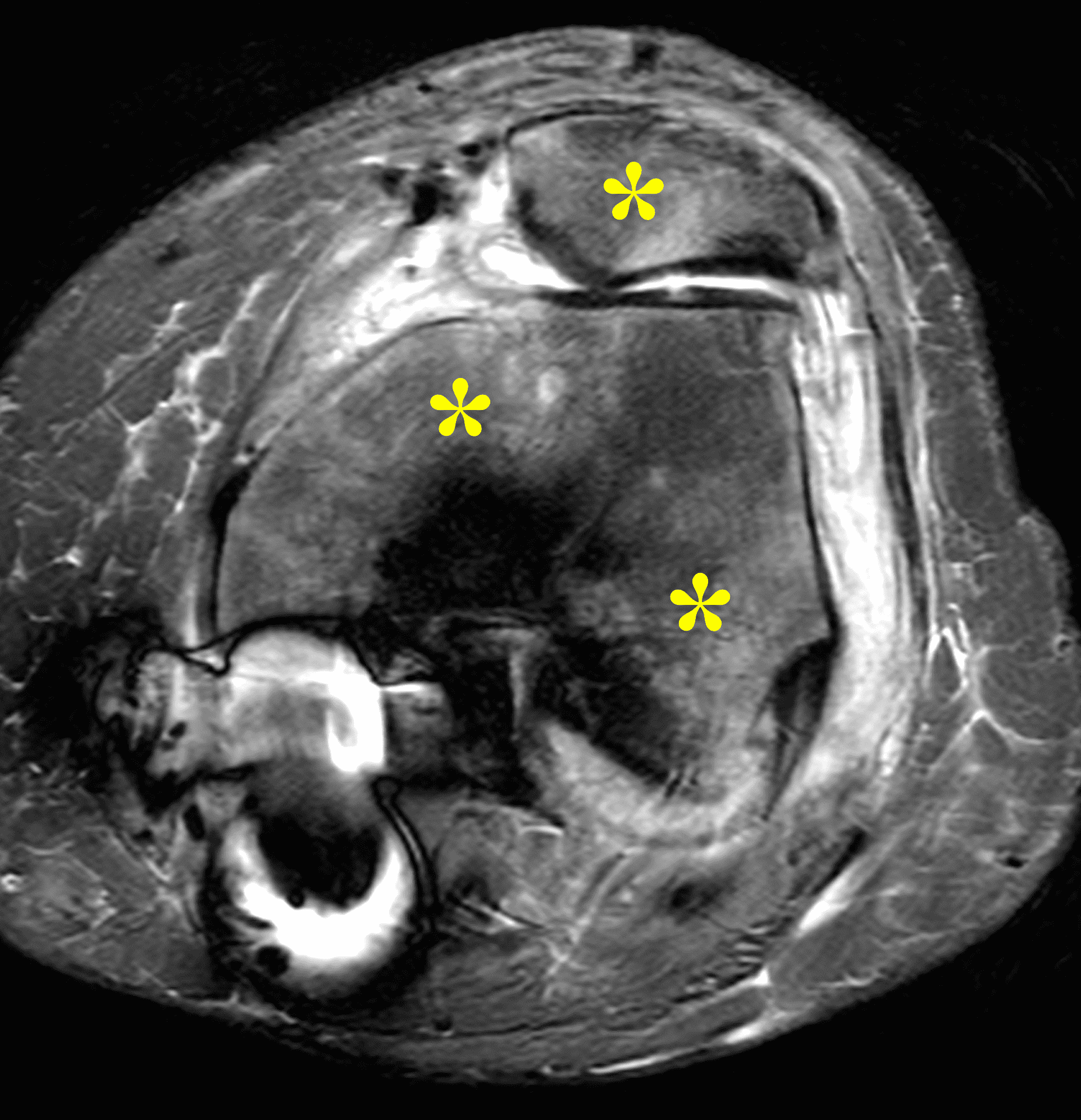
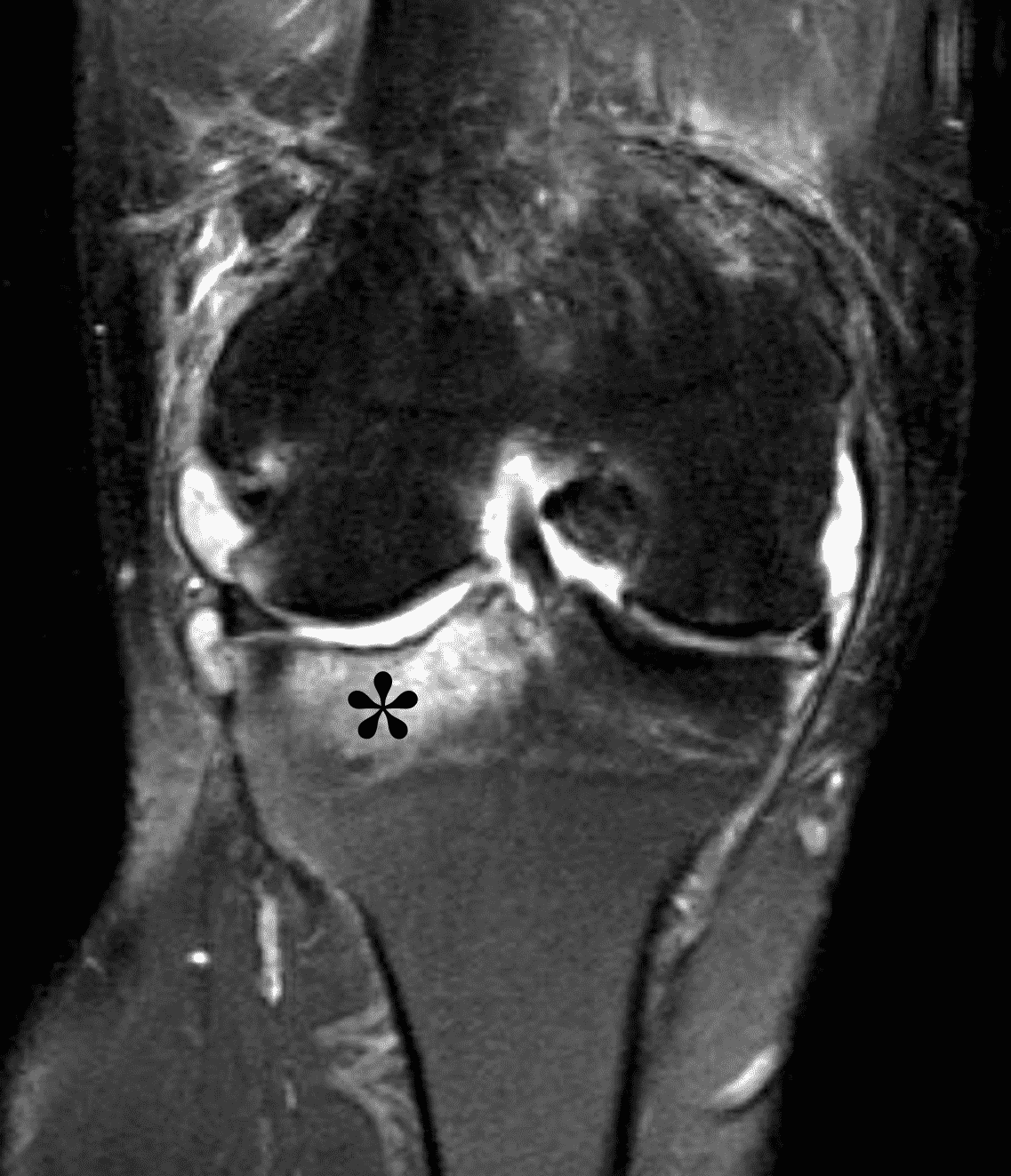
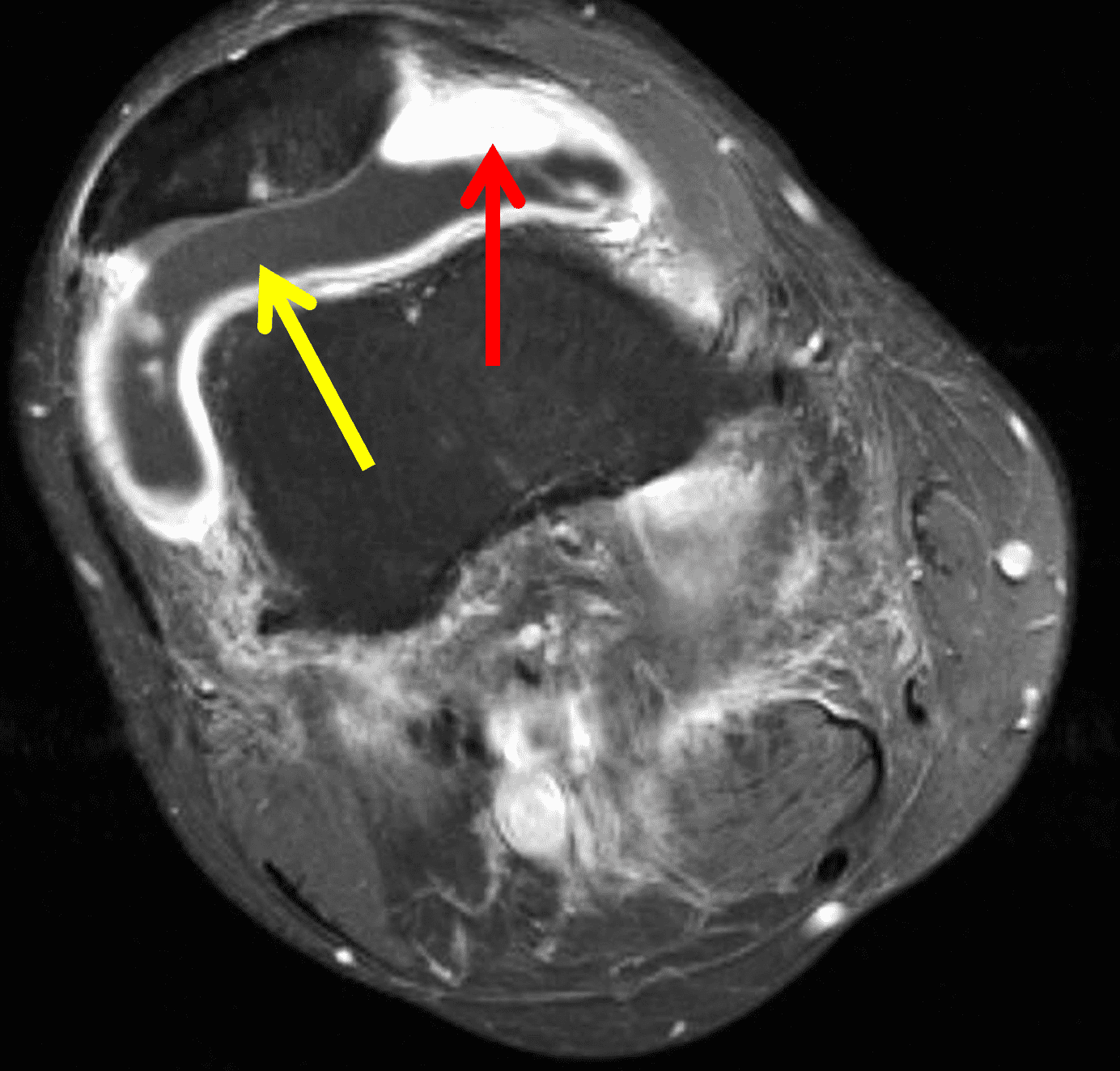
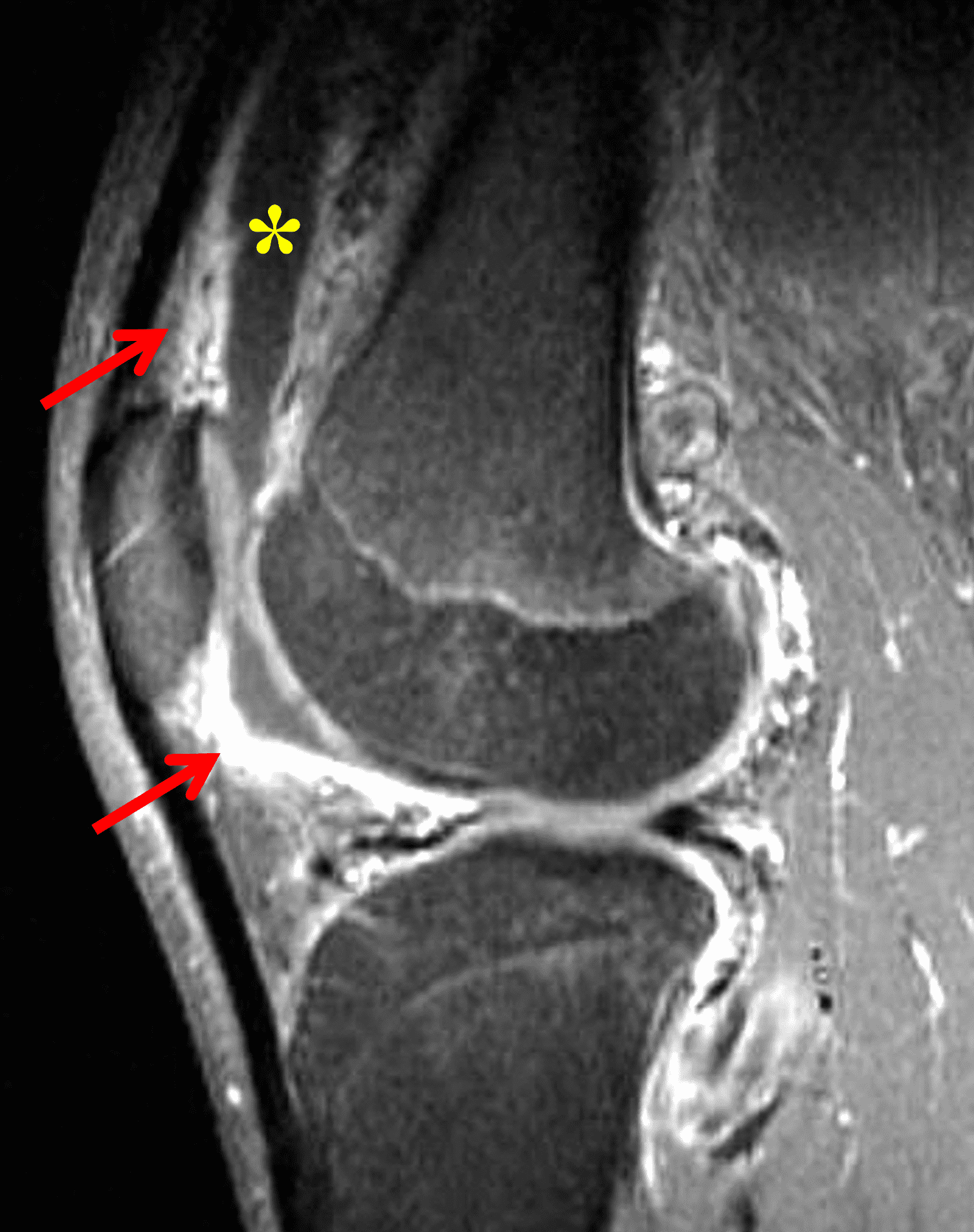
Figure 2: (2A) The coronal image shows a large effusion and severe hypertrophic synovitis (asterisks) in the knee. (2B) On the sagittal image the hypertrophic synovium (asterisks) involves the suprapatellar and infrapatellar recesses as well as the anterior and posterior intercondylar notch. A reactive lymph node (arrow) is present in the popliteal fossa. (2C) The axial image demonstrates myositis (arrows) involving the popliteus and peroneus longus muscles. Note the preserved articular cartilage, lack of erosions, and the absence of subcutaneous or marrow edema.
Diagnosis
Lyme arthritis.
Introduction
Lyme disease is the most common vector-borne zoonotic disease in the United States, with a geographic distribution predominantly involving New England and the mid-Atlantic region, and the upper Midwest.1,2,3 Originally described in the 1970s in Lyme, Connecticut,1,4 it is a multisystem disease caused by the spirochete bacteria Borrelia burgdorferi. Most cases occur between late spring and early summer,5,6 corresponding with the life cycle of its Ixodid tick vectors, predominantly Ixodes scapularis (also known as the deer tick or black-legged tick).1,5,7 The incidence is rising, with nearly 90,000 cases reported nationwide in 2023. The disease is more common in males than females, and has a bimodal age distribution, greatest among children 5-14 years old and adults aged 60-64.6
Clinical Presentation and Diagnosis
Lyme disease progresses through early, early disseminated, and late phases. The most common indication of initial infection is the characteristic erythema migrans rash, a painless, expanding circular or elliptical rash that often resembles a “bulls-eye,” which typically appears within 1-2 weeks at the site of the tick bite (Figure 3).1 However many patients do not develop or may not notice the rash and instead complain of vague constitutional symptoms; especially when there is no recollection of a tick bite, these patients are at risk for delayed diagnosis and treatment.7,8,9 When untreated or inadequately treated, the disease progresses to disseminated and late manifestations that include Lyme arthritis, which commonly manifests months after the initial infection. In such cases magnetic resonance imaging (MRI) may play a useful role in diagnosis and treatment planning.9 Neurologic symptoms, chronic fatigue, and sometimes cardiac issues are additional late complications.
Figure 3: Erythema migrans. Photograph shows the typical rash that occurs shortly after initial exposure. Photo credit: CDC/James Gathany, public domain.
Lyme arthritis most commonly presents as a monoarticular or oligoarticular arthritis, predominantly affecting the knee.5,9,10,11 Clinical, laboratory, and imaging findings often overlap with those of crystalline, inflammatory, and other infectious arthropathies. Definitive diagnosis during the early phases is performed with two-tier serologic testing for antibodies with an enzyme-linked immunosorbent assay (ELISA) followed by confirmatory Western blot testing. In the setting of late-stage disease, such as Lyme arthritis, the absence of IgG and IgM antibodies effectively excludes the diagnosis while the presence of serum IgG and IgM antibodies indicates prior exposure and is not specific for active infection.1,5 Consequently, laboratory testing and arthrocentesis are usually performed for further evaluation; polymerase chain reaction (PCR) testing of synovial fluid is useful as a confirmatory test as it is positive in cases of Lyme arthritis and typically becomes negative following antibiotic treatment.5 However, Lyme serology and PCR testing may not be readily available in non-endemic areas and are time consuming in cases where a diagnosis of septic arthritis and urgent surgical intervention are being considered. In these situations, imaging with MRI may serve as a useful adjunctive decision-making tool.9 Finally, even in more indolent clinical cases and when encountered in non-endemic regions, MRI may be obtained as part of the initial evaluation, especially because the knee symptoms are often nonspecific. In these cases, it is important for the radiologist to be familiar with the imaging features of Lyme arthritis to suggest the diagnosis and provide an appropriate differential diagnosis.
Imaging Findings
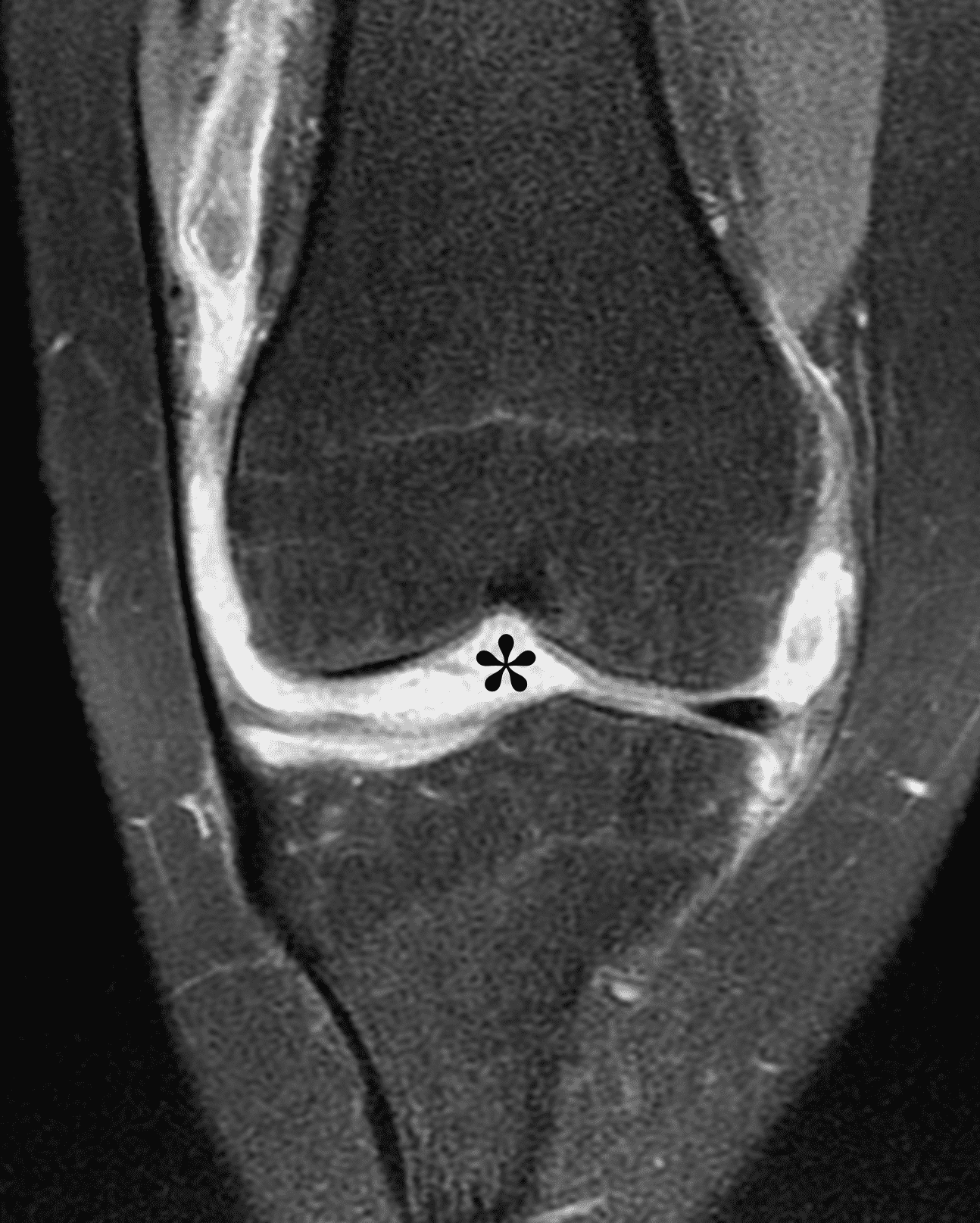
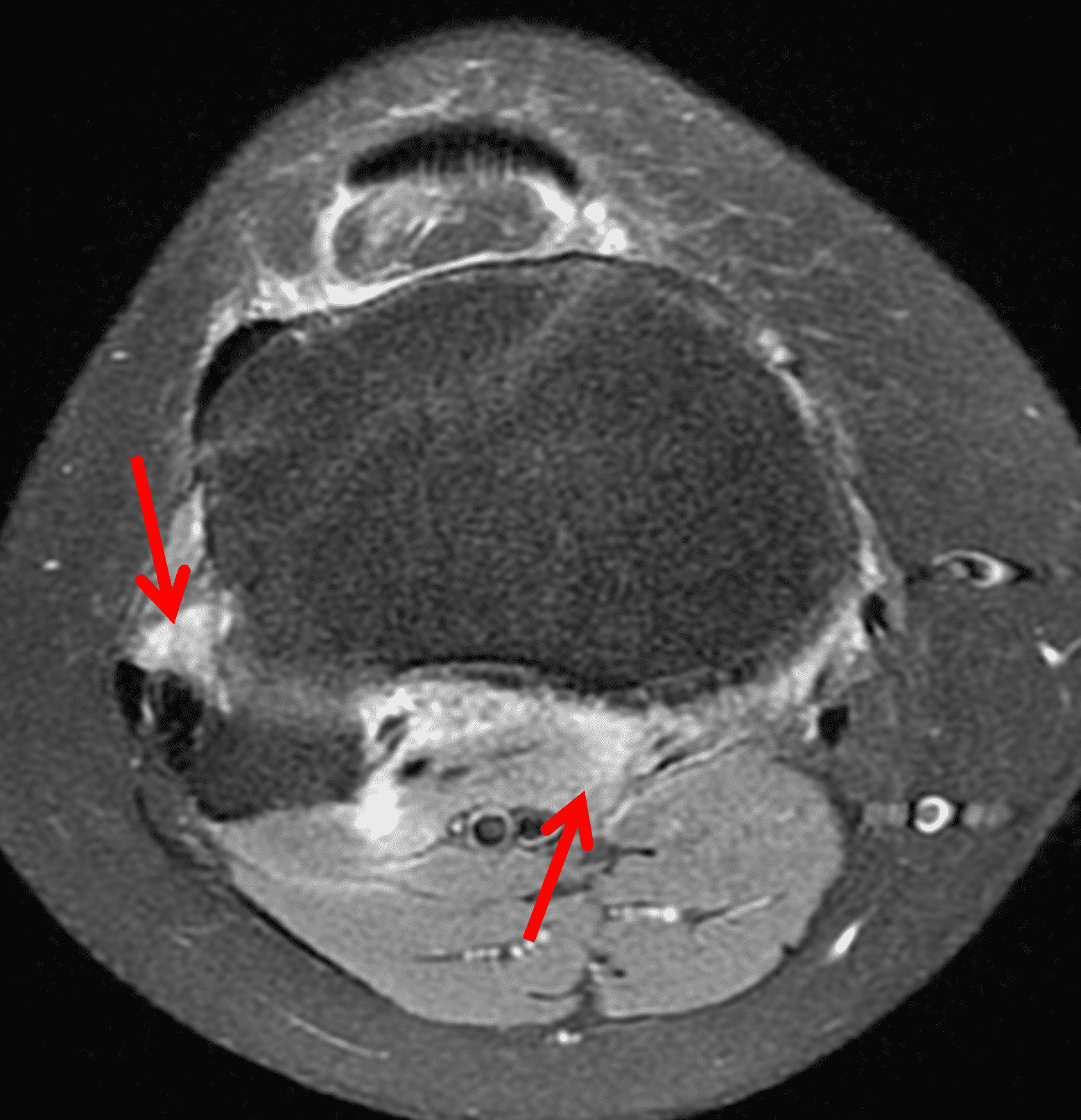
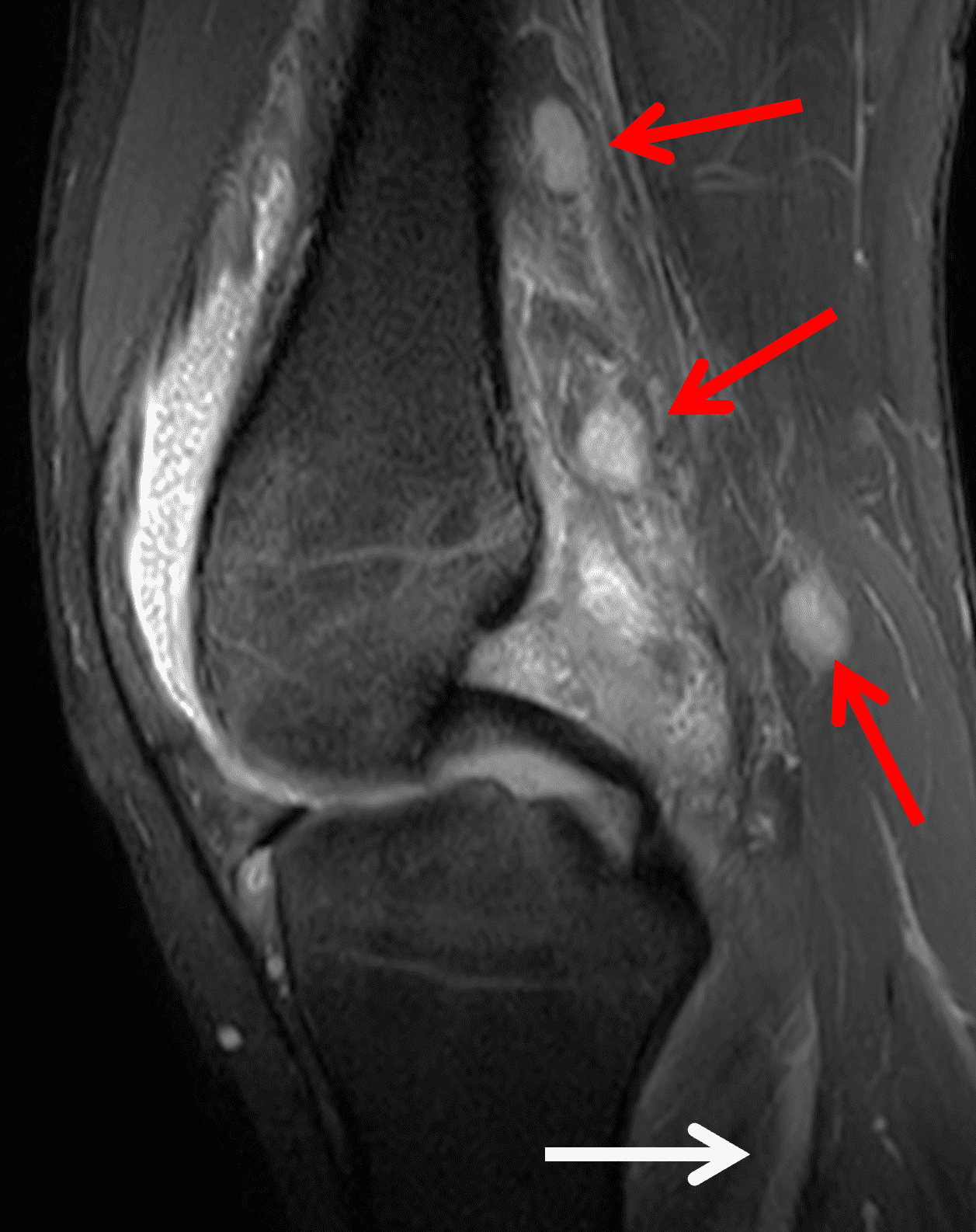
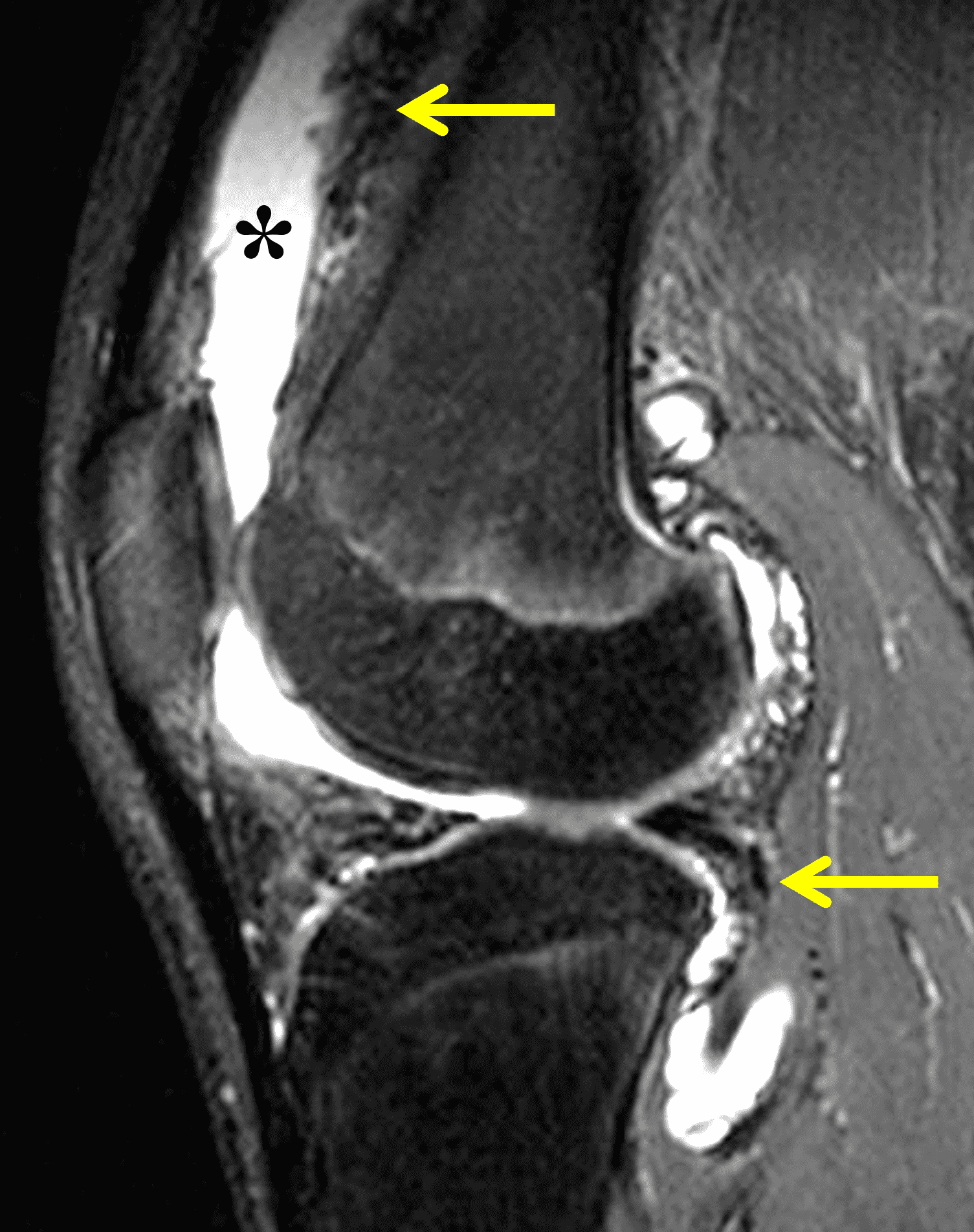
The causative spirochete invades the synovium, triggering a host inflammatory response that leads to synovial hypertrophy and inflammation, resulting in the characteristic clinical symptoms and imaging findings associated with Lyme arthritis.2 Common MR findings associated with Lyme arthritis include synovitis, joint effusion, popliteal lymphadenopathy, and myositis predominantly involving the popliteus (Figure 4).2,3,9 In contrast to acute septic arthritis due to more common bacteria, subcutaneous edema, marrow edema, erosions, and osteomyelitis are uncommon (Figure 4C).
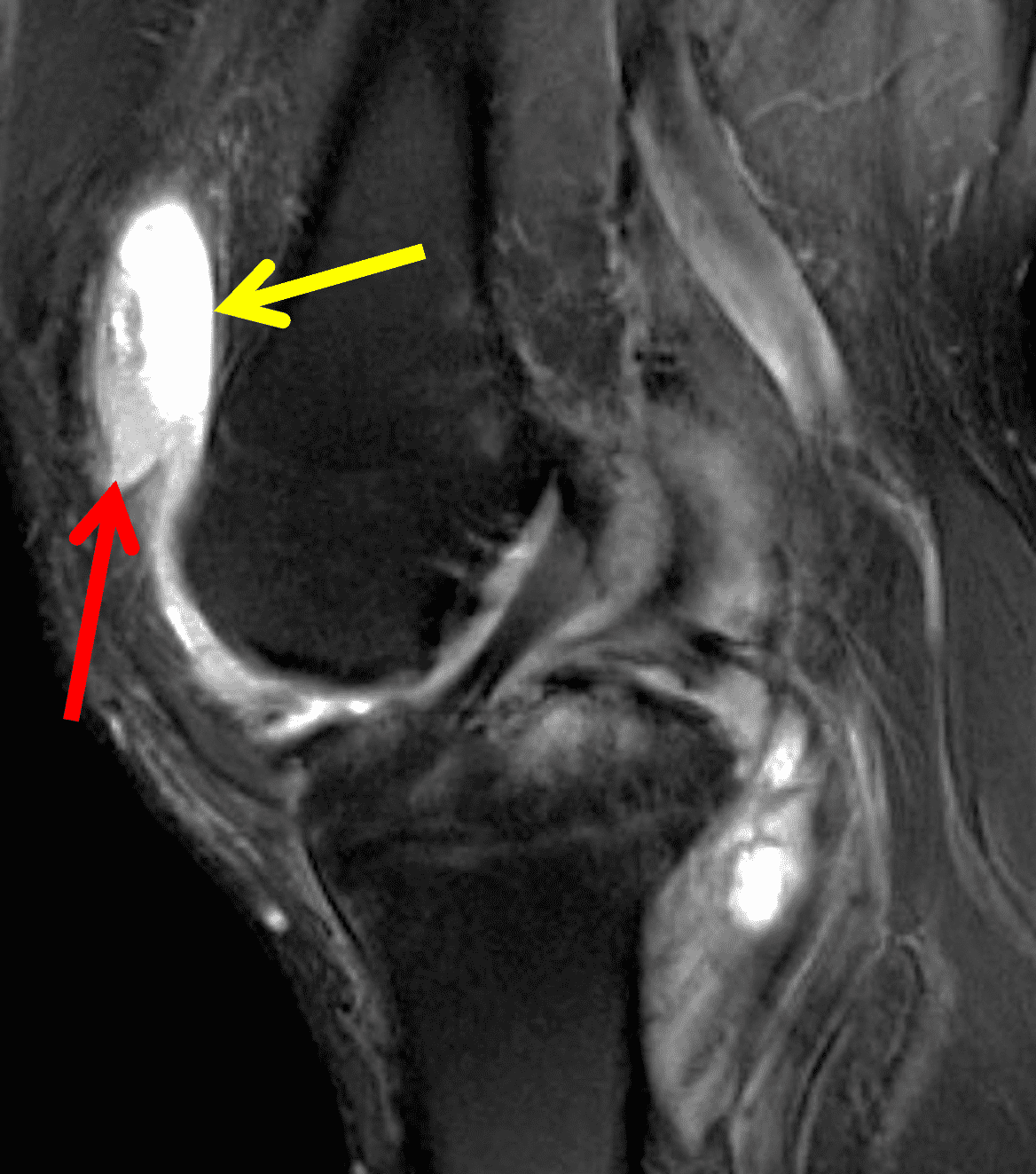
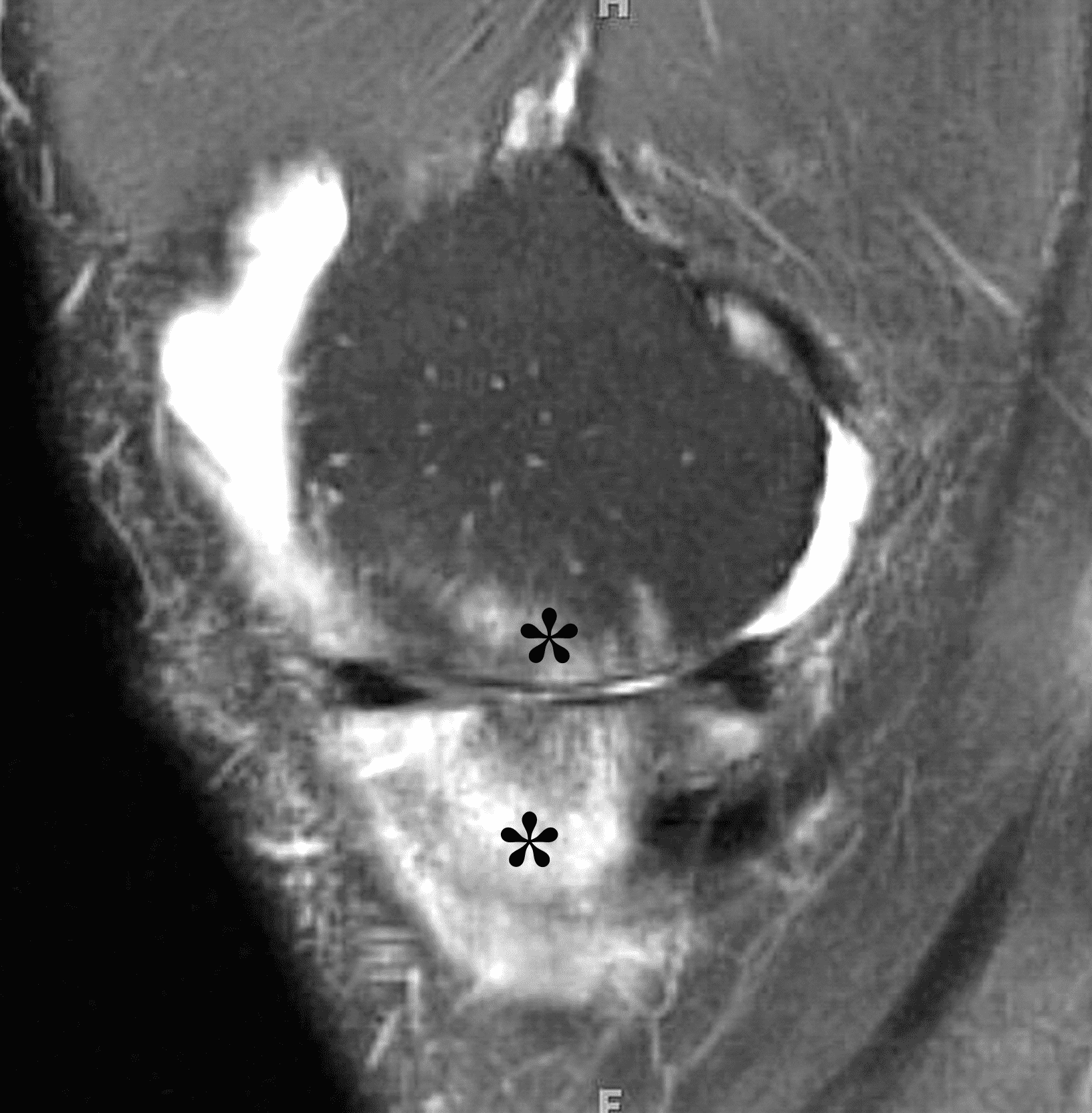
Figure 4: Lyme arthritis. Fat-suppressed, fluid-sensitive images. (4A) Transverse image shows severe proliferative synovitis in the anterior and posterior joint recesses (arrows). Note the lack of surrounding subcutaneous edema. (4B) In addition to synovitis, the sagittal image shows multiple reactive lymph nodes (red arrows) and popliteal myositis (white arrow). (4C) Coronal image also shows patchy non-joint centered marrow edema in the distal femur and proximal tibia (asterisks), which is uncommon in Lyme disease compared to acute septic arthritis.
Differential Diagnosis
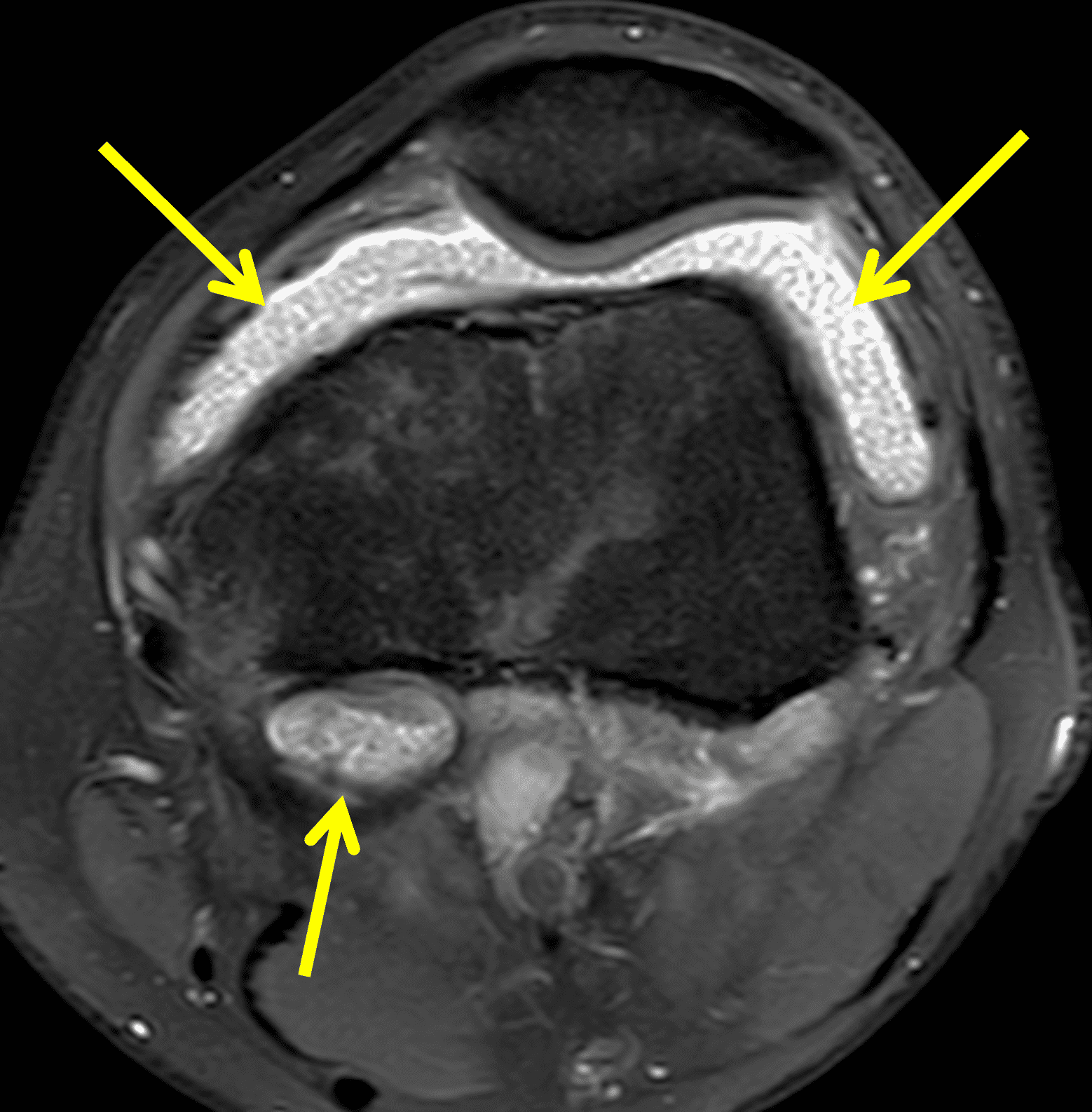
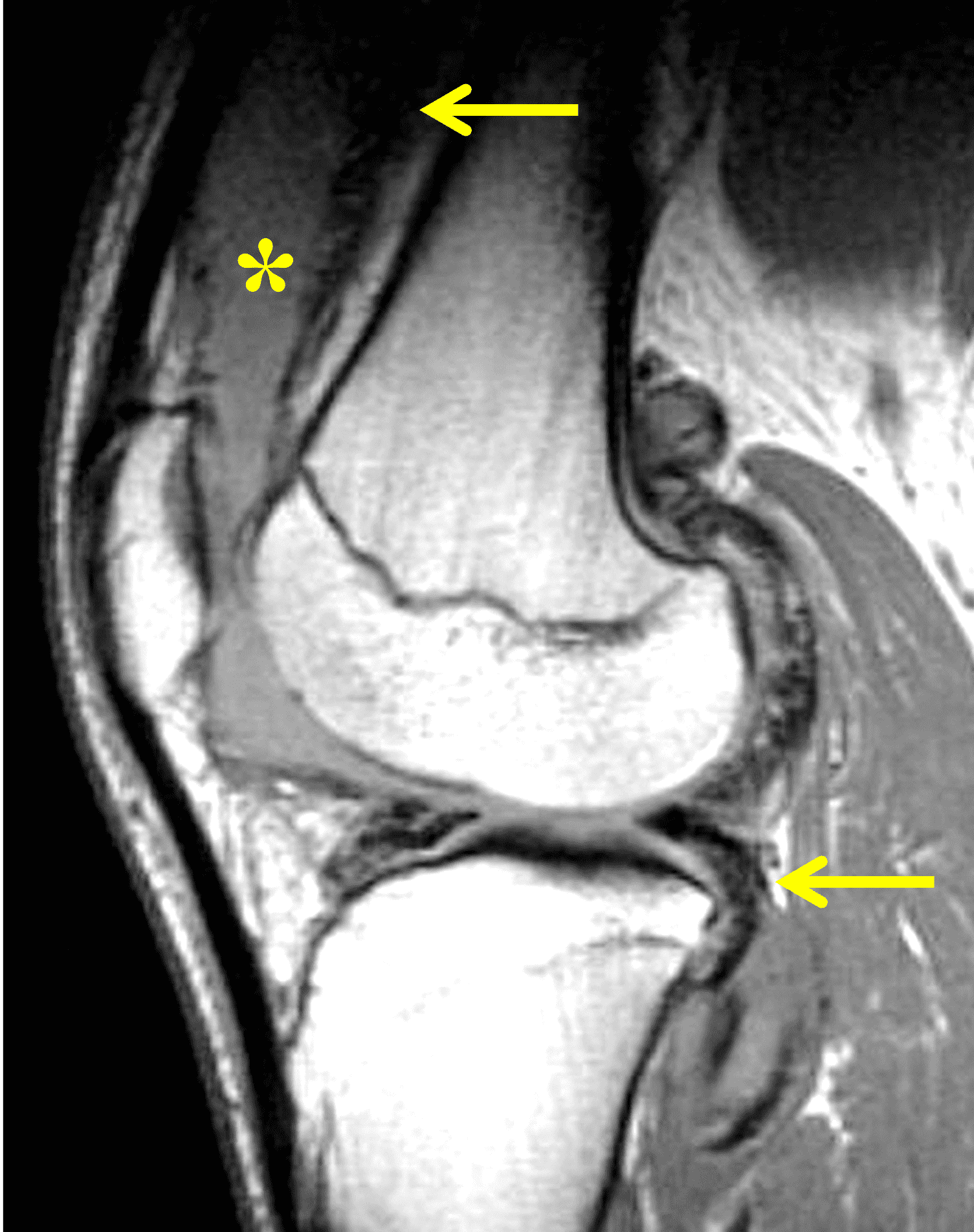
Pediatric patients often present with a more acute clinical picture including fever, difficulty weight-bearing, elevated serum inflammatory markers, and elevated synovial white blood cell count, all of which can also be seen in the setting of acute septic arthritis due to other bacteria.5,9,12 Several studies have identified MR findings that help distinguish Lyme arthritis from septic arthritis for clinical decision-making. Both groups of patients will have synovitis and an effusion related to synovial inflammation and hyperemia. However, subcutaneous edema is highly associated with septic arthritis (Figure 5) and uncommon in Lyme arthritis. Marrow edema and erosions are much more common in septic arthritis, while osteomyelitis – actual infection of the bone – only occurs septic arthritis.2,3,9 Myositis can be seen in both conditions, but severe diffuse myositis is more highly correlated with septic arthritis (Figure 5).2
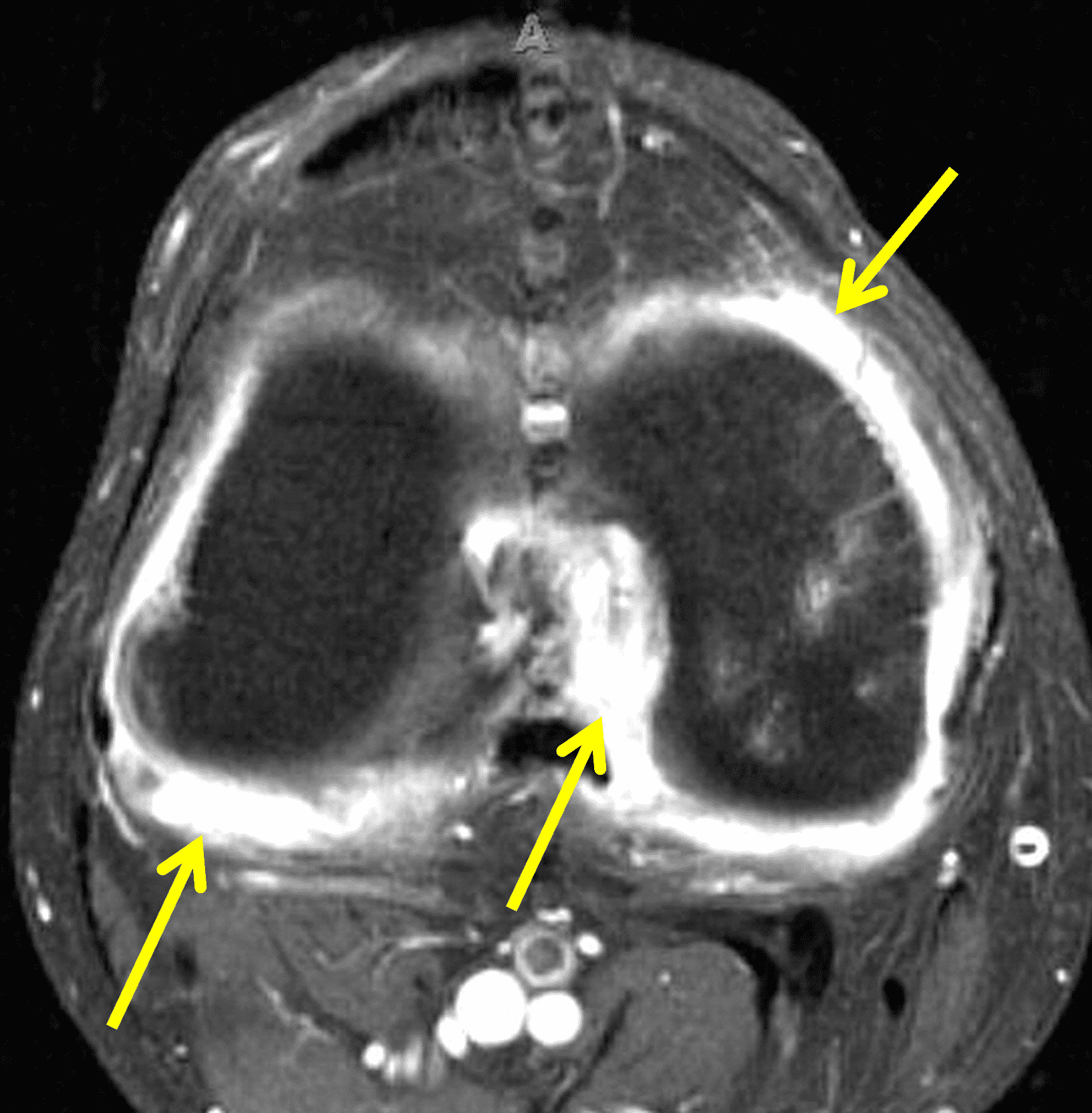
Figure 5: Septic arthritis due to S. aureus. (5A) Sagittal and (5B) axial fat-suppressed fluid sensitive images show severe synovitis and extensive marrow edema (asterisks) in addition to surrounding subcutaneous edema and extensive myositis (arrow). The artifact posteriorly on the axial image is due to a metallic foreign object.
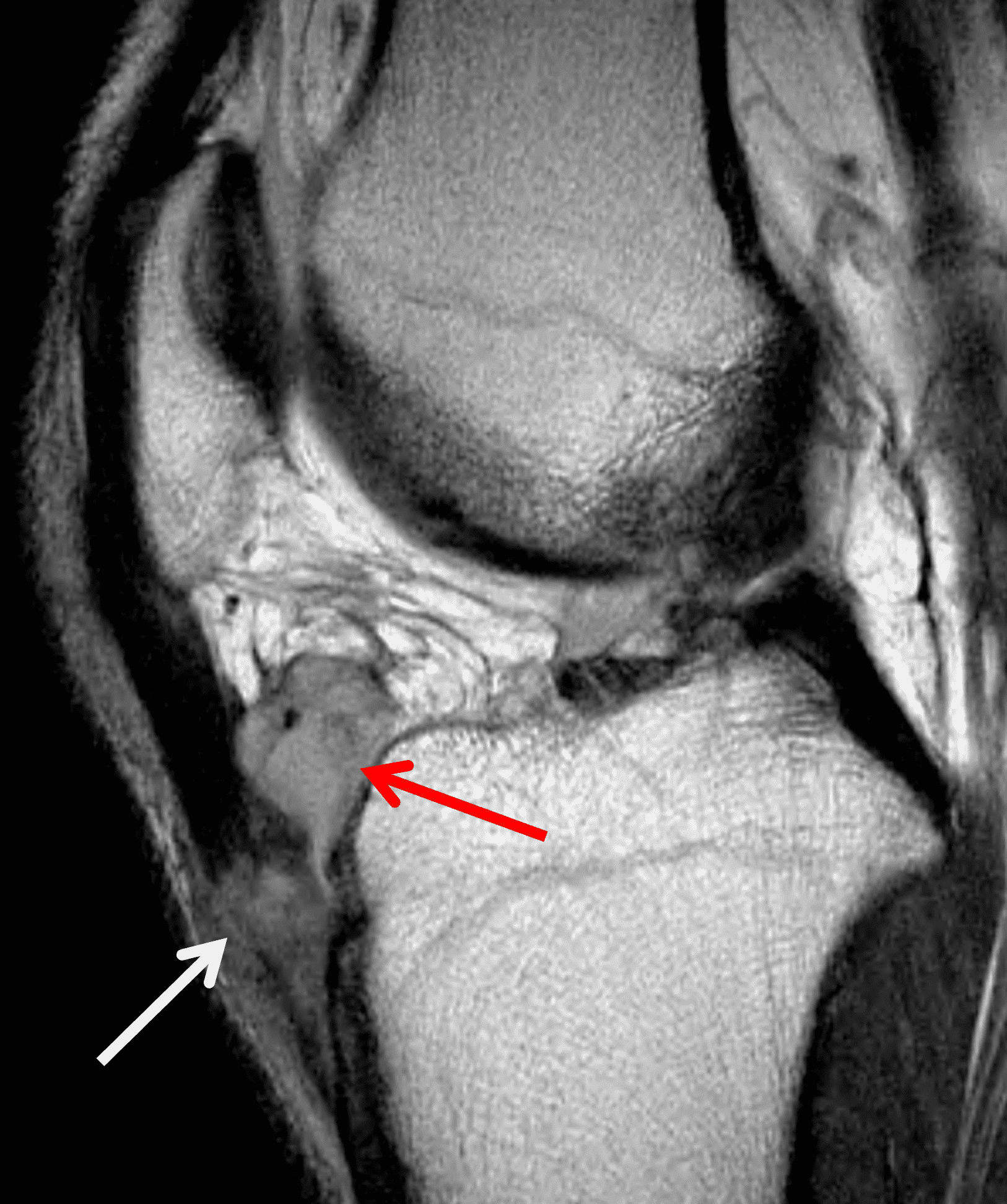
The knee is the most frequently affected joint in juvenile idiopathic arthritis (JIA) and often involved in rheumatoid arthritis (RA) and seronegative spondyloarthritis, with synovial hypertrophy and hyperemia occurring in these conditions, similar to Lyme arthritis.13,14 While a polyarticular presentation is common in inflammatory arthritis and often suggests the correct diagnosis, patients who presenting with oligoarticular disease may be imaged for suspected mechanical or traumatic causes. Similar to Lyme arthritis, these patients’ knees will also demonstrate synovitis and large effusions, but bone marrow edema (osteitis) often occurs in JIA and RA, and is commonly juxta-articular and associated with the weight-bearing joint surfaces (Figure 6).14 Additionally, in advanced inflammatory arthritis, cartilage loss and erosions are common (Figure 7). These findings are rarely seen in the setting of Lyme arthritis.12,13
Figure 6: Rheumatoid arthritis. (6A) Sagittal fat-suppressed T2-weighted image demonstrates an effusion (yellow arrow) and synovitis (red arrow) in the suprapatellar recess. (6B) Coronal fat-suppressed T2-weighted image shows extensive, relatively uniform cartilage loss in the medial and lateral compartments and subchondral osteitis (asterisk) in the medial tibial plateau. (6C) Axial fat-suppressed T1-weighted image obtained following intravenous contrast administration shows the enhancing synovitis (red arrow) and the non-enhancing joint effusion (yellow arrow). Note the uniform cartilage loss of the patella. This patient had polyarticular disease.
Figure 7: Psoriatic arthritis. (7A) Sagittal fat-suppressed T2-weighted image shows severe uniform cartilage loss in the medial compartment with high signal intensity in the subchondral marrow representing osteitis (asterisks). (7B) Axial contrast-enhanced, fat-suppressed T1-weighted image shows extensive, enhancing synovitis throughout the joint (arrows).
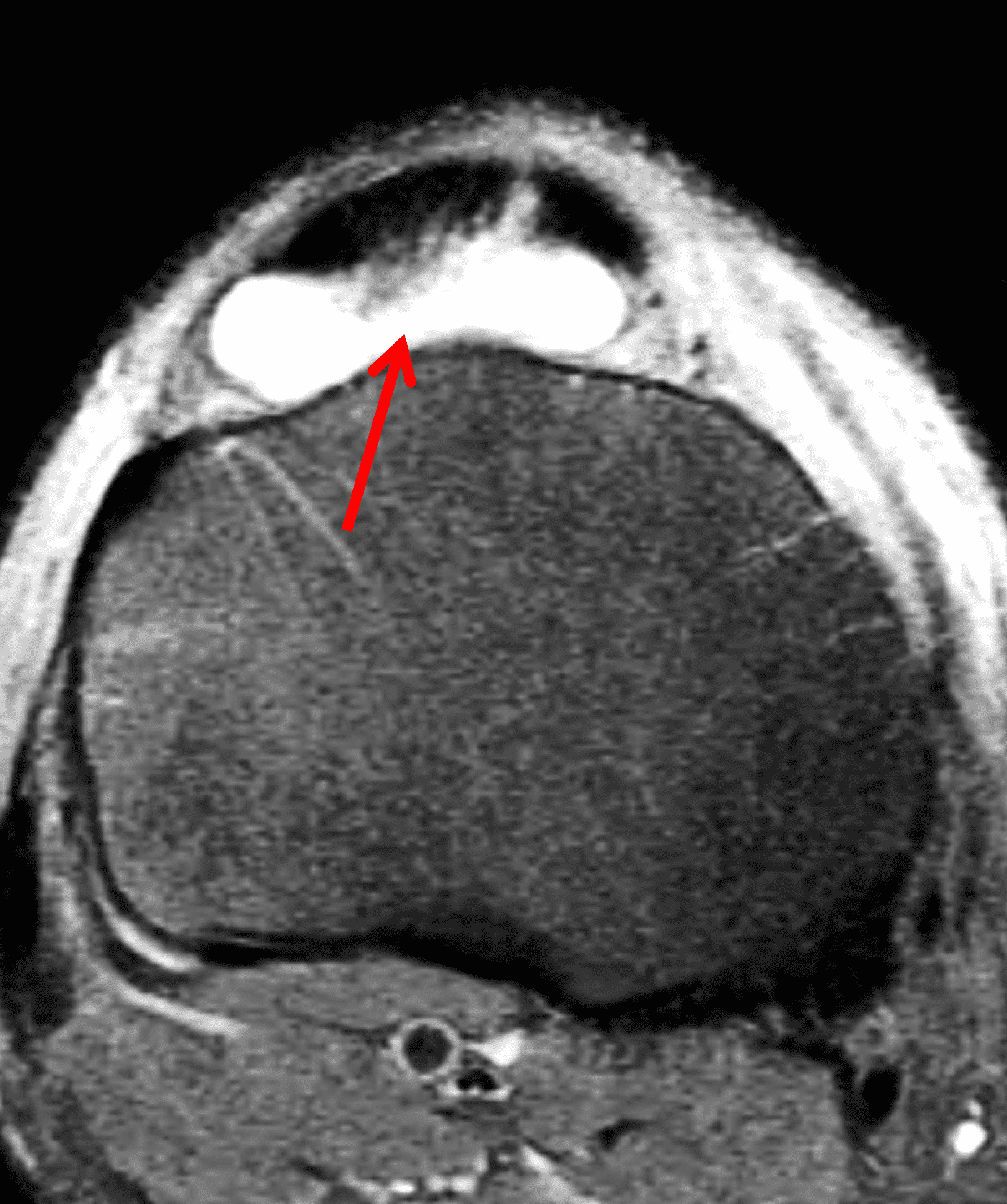
Crystalline arthropathies such as gout commonly present as a monoarticular arthritis with pain and swelling. Diagnosis is usually based on clinical history and synovial fluid analysis without a need for imaging in typical cases. When MRI is performed the findings are variable and may be nonspecific but the distribution of involvement may suggest of the diagnosis. In contrast to Lyme arthritis, erosions are commonly seen in gout involving the knee along with tophaceous involvement of the popliteal and extensor tendons (Figure 8).15
Figure 8: Gout in a 45-year old man. (8A) Sagittal T2-weighted image shows synovial hypertrophy in the deep infrapatellar bursa (red arrow) and soft tissue infiltration of the distal patellar tendon (white arrow). (8B) Axial fat-suppressed T2-weighted image demonstrates the distended deep infrapatellar bursa (red arrow). Biopsy of the patellar tendon confirmed tophaceous gout.
Hemophilia is an X-linked recessive disease in which recurrent spontaneous intra-articular hemorrhage produces joint pain and swelling; recurrent hemarthroses result in hemophilic arthropathy. The knee is commonly affected . Chronic hemosiderin deposition leads to synovial proliferation and chronic inflammation with eventual erosions and cartilage damage. Common MR findings include erosions, synovial hypertrophy, epiphyseal enlargement in children, and hemosiderin deposition within the joint (Figure 9). In later stages, cartilage loss and subchondral cysts may occur.16
Figure 9: Hemophilic arthropathy in a teenage boy. (9A) Sagittal fat-suppressed T2-weighted and (9B) sagittal proton density-weighted images show a joint effusion/synovitis (asterisks) as well as very-low-signal intensity foci lining the suprapatellar recess and posterior synovium (arrows) representing hemosiderin. (9C) Sagittal post-contrast, fat-suppressed T1-weighted image distinguishes between the nonenhancing joint fluid (asterisk) and enhancing synovitis (arrows). (9D) Sagittal gradient-echo recalled sequence obtained as a scout image demonstrates ferromagnetic blooming artifact in the synovial lining (arrows) from hemosiderin deposition.
Treatment
Treatment of Lyme arthritis consists of a 28-day course of oral doxycycline, or amoxicillin if there is a contraindication to doxycycline. Patients may experience an initial flare of symptoms secondary to spirochete death and the Jarisch-Herxheimer reaction.7, 11 In refractory cases intravenous antibiotics may be needed.5, 7 Most patients recover completely, although in rare cases post-antibiotic Lyme arthritis results in chronic symptoms. A persistent presentation should prompt evaluation for a concurrent rheumatologic disorder. If none is identified, additional courses of antibiotics are not beneficial, and management consists of anti-inflammatory medications, steroids, and disease modifying anti-rheumatic drugs (DMARDS) including biologic agents. In refractory cases synovectomy may be necessary with MR serving as a useful preoperative planning tool.7
Conclusion
While it is the most common vector-borne zoonotic disease in the United States, Lyme disease may present a diagnostic challenge, particularly when presenting with late manifestations such as arthritis. A knee effusion and synovitis without underlying mechanical explanation on MR should prompt a search for additional imaging features to help narrow the differential diagnosis. Although not specific, the presence of popliteal lymphadenopathy and myositis should raise the possibility of Lyme arthritis. The absence of extensive surrounding soft tissue edema, erosions, and findings of osteomyelitis favor Lyme arthritis over cases septic arthritis due to other bacteria. A polyarticular presentation, erosions, cartilage loss, and osteitis are uncommon in Lyme disease and should suggest an inflammatory arthritis like RA or JIA. Recognizing the typical MRI findings allows the radiologist to raise the possibility of Lyme arthritis, even in non-endemic areas, to ensure that a proper travel and exposure history are obtained and appropriate testing is performed.
References
- Shapiro ED. Clinical practice. Lyme disease. N Engl J Med. 2014;370(18):1724–1731. doi:10.1056/NEJMcp1314325 ↩
- Powell JE, Lee VK, Parikh SS, Nowalk AJ, Shah AJ. MRI features distinguishing pediatric Lyme arthritis from septic arthritis. Skeletal Radiol. 2025;54(5):1043–1057. doi:10.1007/s00256-024-04804-2 ↩
- Ecklund K, Vargas S, Zurakowski D, Sundel RP. MRI features of Lyme arthritis in children. AJR Am J Roentgenol. 2005;184(6):1904–1909. doi:10.2214/ajr.184.6.01841904 ↩
- Steere AC, Broderick TF, Malawista SE. Erythema chronicum migrans and Lyme arthritis: epidemiologic evidence for a tick vector. Am J Epidemiol. 1978;108(4):312–321. doi:10.1093/oxfordjournals.aje.a112625 ↩
- Arvikar SL, Steere AC. Lyme Arthritis. Infect Dis Clin North Am. 2022;36(3):563–577. doi:10.1016/j.idc.2022.03.006 ↩
- Centers for Disease Control. Lyme Disease Surveillance Data: CDC; 2025. https://www.cdc.gov/lyme/data-research/facts-stats/surveillance-data-1.html. ↩
- Udziela S, Biesiada G, Osiewicz M, Michalak M, Stazyk K, Garlicki A, et al. Musculoskeletal manifestations of Lyme borreliosis – a review. Arch Med Sci. 2022;18(3):726–731. doi:10.5114/aoms.2020.96458 ↩
- Aucott J, Morrison C, Munoz B, Rowe PC, Schwarzwalder A, West SK. Diagnostic challenges of early Lyme disease: lessons from a community case series. BMC Infect Dis. 2009;979. doi:10.1186/1471-2334-9-79 ↩
- Ortiz CD, Barsi J. Differentiating Between Septic Arthritis and Lyme Arthritis in the Pediatric Population. J Pediatr Soc North Am. 2023;5(3):706. doi:10.55275/JPOSNA-2023-706 ↩
- Marcelis S, Vanhoenacker F. Lyme Disease: A Probably Underdiagnosed Cause of Mono-Arthritis. J Belg Soc Radiol. 2021;105(1):80. doi:10.5334/jbsr.2625 ↩
- Stevenson DR, Ghuman A, Jones A, Marks M, Arvikar S, Petridou C, et al. Lyme arthritis: It’s never too late for joint decision making. Clinical Infection in Practice. 2025;25100406. doi:10.1016/j.clinpr.2024.100406 ↩
- Haug L, Slat D, Long J. A Downward Spiral of Knee Pain and Swelling: Use of MRI to Distinguish Lyme Arthritis From Other Pediatric Arthritides. Roentgen Ray Review. 2025;1(1):e25.01003. doi:10.2214/r3j.25.01003 ↩
- Hemke R, Tzaribachev N, Barendregt AM, Merlijn van den Berg J, Doria AS, Maas M. Imaging of the knee in juvenile idiopathic arthritis. Pediatr Radiol. 2018;48(6):818–827. doi:10.1007/s00247-017-4015-6 ↩
- Yang Y, Yuan X, Wang X, Tao R, Jiang T. Preliminary MRI-based investigation of characteristics and prognosis of knee bone marrow edema in children with juvenile idiopathic arthritis. Clin Rheumatol. 2022;41(6):1793–1799. doi:10.1007/s10067-022-06085-3 ↩
- Girish G, Glazebrook KN, Jacobson JA. Advanced imaging in gout. AJR Am J Roentgenol. 2013;201(3):515–525. doi:10.2214/AJR.13.10776 ↩
- Melchiorre D, Manetti M, Matucci-Cerinic M. Pathophysiology of Hemophilic Arthropathy. J Clin Med. 2017;6(7). doi:10.3390/jcm6070063 ↩