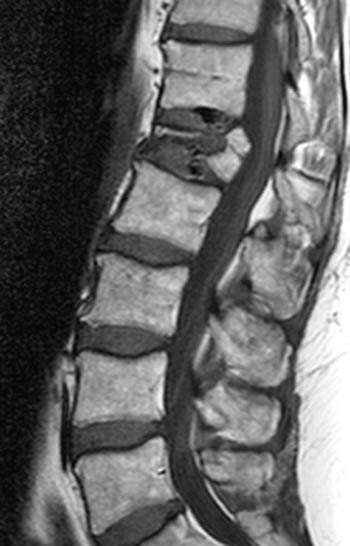
Clinical History: A 58 year-old female with Stage 3A breast cancer presents to her family physician with back pain. A radiograph reveals a severe L1 compression fracture. She is referred to an orthopaedist. On physical exam she exhibits axial type pain without radicular symptoms or myelopathic signs. Concern for possible osseous metastasis to the spine results in performance of an MRI. (1a-c) T1, T2, and fat-suppressed T2-weighted sagittal images, a (1d) T1-weighted axial image, and a (1e) fat-suppressed T1-weighted sagittal image following contrast administration are provided. What are the findings? What is your diagnosis?
Findings
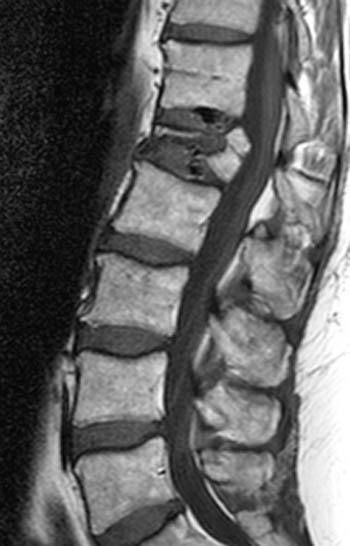
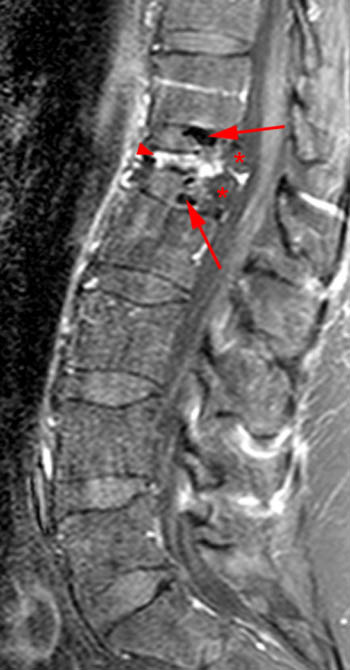
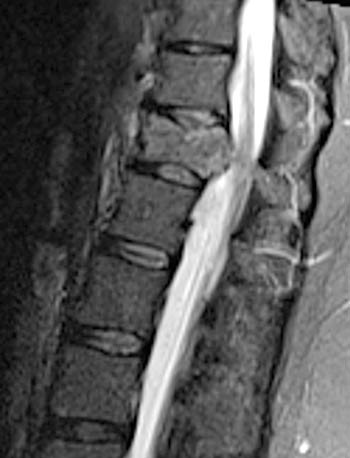
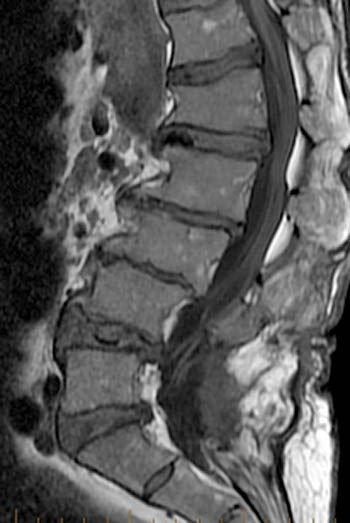
Figure 2:
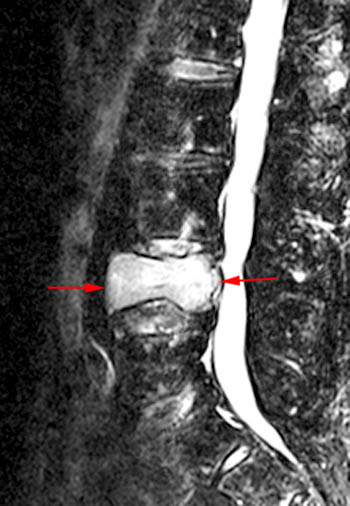
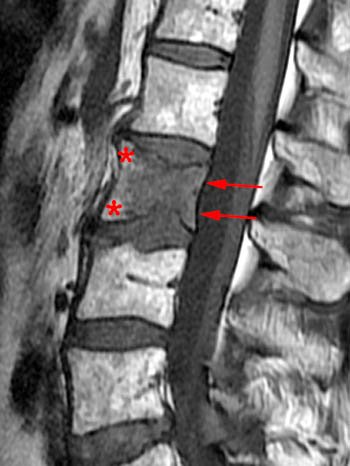
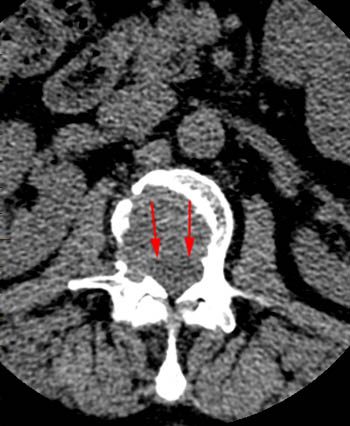
(2a) T1-weighted sagittal, (3a) T2-weighted sagittal, (4a) T2-weighted sagittal with fat-saturation, (5a) T1-weighted axial at the L1 pedicle level and (6a) post-gadolinium T1-weighted sagittal with fat saturation: reveal a moderate compression fracture of L1 with substantial preservation of normal marrow signal, particularly on the T1-weighted images. Retropulsion of bone, low intradiscal and possibly intraosseous signal suggestive of gas, a lack of substantial prevertebral soft tissue mass, a lack of abnormal signal material extending through the posterior vertebral body cortex, sharp or distinct margins of the retropulsed cortex, lack of involvement of the pedicles, and abnormal post-contrast enhancement limited to the compressed portion of the centrum, all of which combine to depict a classic chronic benign fracture.
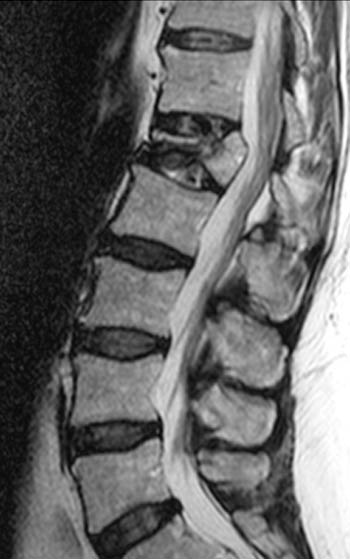
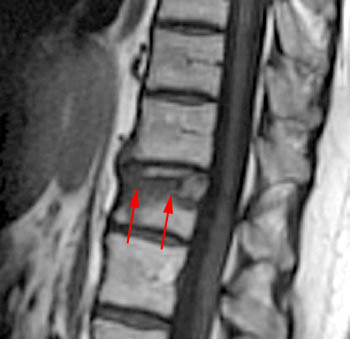
Figure 3:
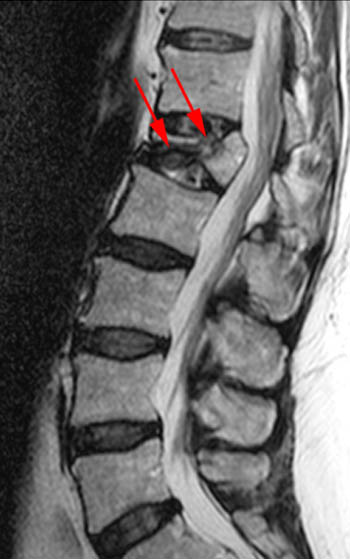
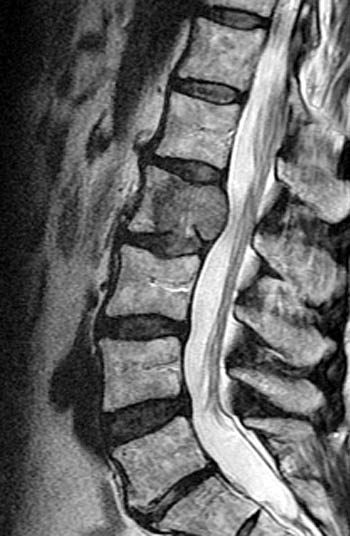
T2-weighted fast spin echo sagittal. Signs of the benign nature of the fracture include preservation of normal T2 marrow signal compared to adjacent segments throughout most of the centrum, a visible fracture line or "cleft" (arrow), and anterior wedging. Although the posterior cortex of the centrum is convex which in some circumstances is a worrisome sign, the cortical margin is sharp and intact and soft tissue does not extend dorsally into the epidural space.
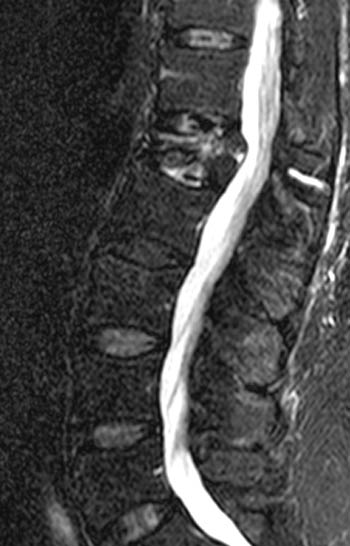
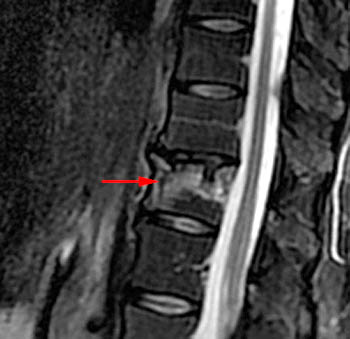
Figure 4:
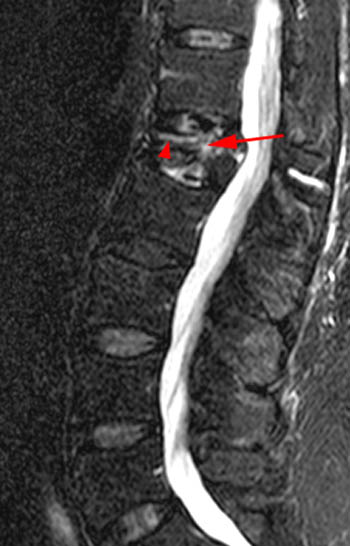
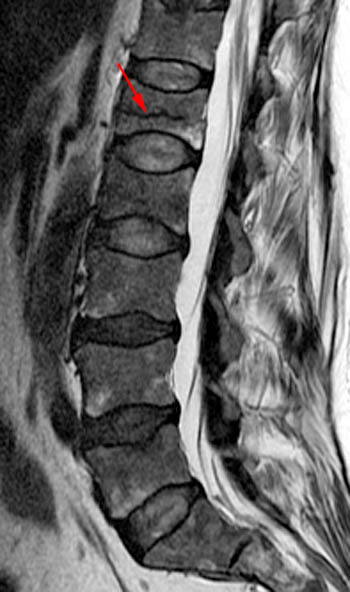
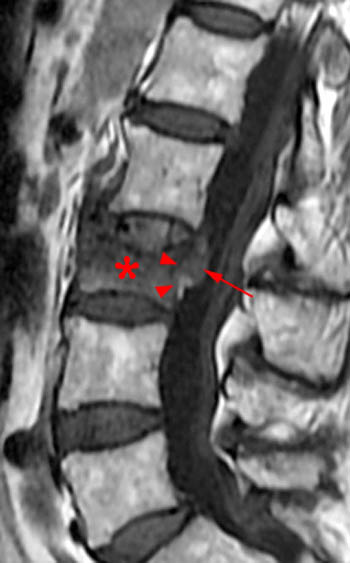
T2-weighted sagittal with application of fat-saturation. Edematous-type or fluid-like signal and hyperemia are bright and more conspicuous on this sequence (as it would be on STIR) compared to the T2-weighted fast spin echo image displayed in 1b. This bright signal is very limited in this chronic fracture. Fluid is found in the fracture cleft (arrow). Linear bright signal under the superior endplate (arrowhead) is most likely hyperemia in this case as the marrow has a normal appearance on the T1-weighted image (2a) but intensely enhances as seen in 1e. The fracture has created a chronic biomechanical instability across this segment leading to signs of persistent "stress" on the segment from axial loading and physiologic motion.
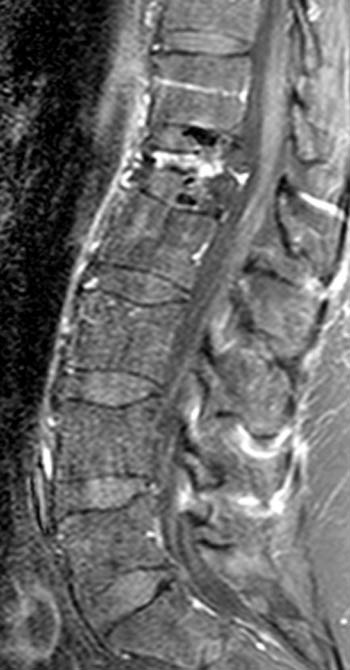
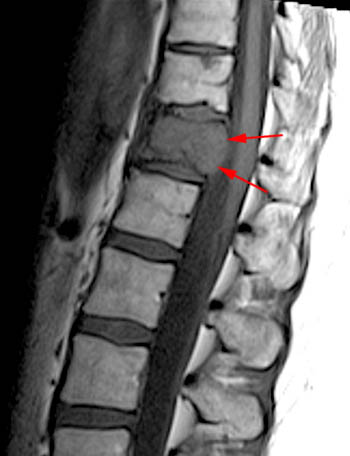
Figure 6:
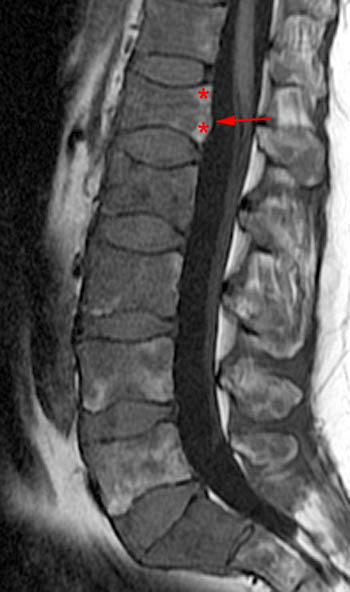
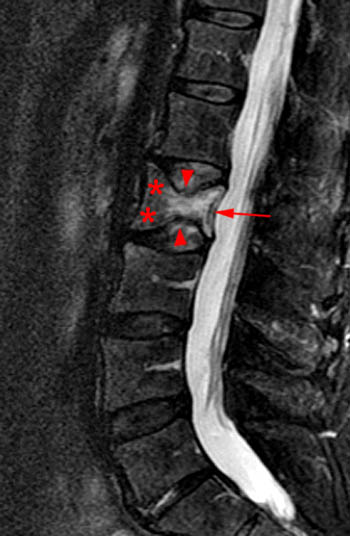
A T1-weighted post contrast sagittal with fat saturation. Enhancement of the compressed anterior aspect of the body (arrowhead) is due to a stress reaction and is also seen in association with the basivertebral vein. The majority of the remaining marrow does not enhance (asterisks), though enhancement on conventional MR is not a good discriminator between benign and malignant fractures. No epidural enhancing lesion is seen. Low signal intradiscal and perhaps intraosseous gas is identified (arrows).
Diagnosis
Benign L1 compression fracture.
Discussion
Deciding whether or not a fracture is benign or pathologic is a common decision confronting a musculoskeletal radiologist. The spine is a very common site for metastases and it has been reported that more than 18,000 patients per year are diagnosed with spinal metastatic disease. Although any systemic malignancy may involve the spine, common solid organ primary tumors include breast, lung, and prostate, whereas common hematopoietic sources include myeloma and lymphoma. Although up to 77% of these cancers have spread to the spine as micrometastases at autopsy, only around 10% become symptomatic. A metastasis to the spine is the initial presentation in up to 20% of cancer patients. Approximately 70% of spine metastases occur in the thoracic spine. Involvement of the cervical spine or the thoracic spine above T5 is rare. Different primary tumors tend to spread to different parts of the spine; for example breast and lung to the thoracic spine and prostate to the lumbar spine and sacrum. Since one third of vertebral fractures in oncology patients are benign and one quarter of fractures in patients with known osteopenia or osteoporosis are malignant, MRI is commonly performed to assist in the diagnosis 1,2,3,4,5.
In acute non-malignant fractures, the bone marrow becomes edematous or fills with fluid. This alters the relaxation time of the marrow leading to hypointensity on T1-weighted images and hyperintensity on T2-weighted or STIR (short inversion-time inversion-recovery) sequences. Usually a hyperemic response occurs leading to proliferation of fibroblastic and granulation tissue all of which evolve over time towards restoration of the primarily fat signal of normal marrow with healing. The time to complete healing is variable especially in the elderly with potentially impeded reparative capacity. Therefore a chronic osteoporotic fracture is more easily distinguished from an acute fracture by nearly completely normal marrow signal especially on T1-weighted and STIR images. Chronic benign fractures especially in the elderly may continue to exhibit imaging signs of “micromotion” or “stress” for months to years by displaying some osseous edema or post contrast enhancement.
Distinguishing an acute osteoporotic fracture from a pathologic fracture is more challenging. Many authors have contributed to our understanding of the expected MR findings in spinal fractures beginning with Yuh in 1989 6 and Baker in 1990 7. Yuh reported that the vertebral body marrow is completely replaced in 88% of malignant fractures and that the accuracy of differentiating a benign from malignant compression fracture was 94% using conventional sequences. There are a myriad of findings on routine MR sequences which are suggestive of a benign origin of a spine fracture 3,4,6,7,8,9,10,11,12,13,14,15,16 and include:
- maintenance of at least some normal marrow signal
- no involvement of the posterior elements
- fluid or gas within the vertebral body
- low intensity band along the fractured endplate
- lack of distinct soft tissue mass within the centrum
- a well defined fracture line
- limited paravertebral soft tissue thickening, swelling or mass
- preservation of the posterior cortex or a non-convex border
- retropulsed bone fragments with sharply marginated or angulated borders
- absence of epidural or foraminal soft tissue mass
- normal marrow signal in adjacent vertebral bodies or ribs
- anterior wedging
- known osteoporosis or osteopenia
- lack of cervical or high thoracic lesion
The above characteristics need to be considered together as no single factor is absolutely reliable in determining if a fracture is benign or malignant. The prevalence or importance of a single feature although reported in the literature can be disputed. For example one author reports that the presence of a fracture line is one of the most reliable signs of benignity 27, while another author reports it is one of the least reliable signs 4. In younger patients usually a non-traumatic pathologic fracture is associated with complete or near complete infiltration of the centrum. However in the elderly, weak bone predisposes to a fracture with only partial marrow replacement. Metastases tend to occur first in the posterolateral aspect of the vertebral body centrum and involve the pedicle late in the process 17. However osteoporotic fractures of the pedicles can occur. Fluid clefts are found in approximately 40% of osteoporotic fractures but can also be present in around 5% of neoplastic fractures 8. Acute benign fractures in the upper thoracic spine are relatively rare but slight presumed developmental concave deformities of the centra at T1, T2 or even T3 are not. Gadolinium contrast is usually not useful in differentiating a benign from a malignant fracture as both types of acute and subacute fractures enhance in unhealed areas. However contrast may be useful in distinguishing between a post traumatic disc herniation or hematoma from an epidural metastasis. Contrast may also be useful in better delineating an intraosseous mass if present. Additionally, some institutions perform advanced techniques such as dynamic contrast-enhanced perfusion imaging in problematic cases 3,18,19.
A classic benign fracture. A 51 year-old female fell down stairs 2 weeks prior to obtaining this MR (7a,8a).
Figure 7:
The T1-weighted sagittal image demonstrates mild anterior wedging with deformity of the superior endplate of T12. Despite low signal edema or hyperemia partially filling the marrow, substantial preservation of fatty marrow signal is seen. A band of low signal horizontally crosses the superior aspect of the centrum (arrow). Other benign signs include the lack of a distinct intraosseous mass, intact posterior cortex, concavity of posterior cortex, lack of abnormalities in adjacent vertebra, and the lack of anterior epidural or prevertebral masses.
A 49 year-old female with back pain and a classic malignant fracture (9a-12a). No known systemic disease was present at the time of MR scanning.
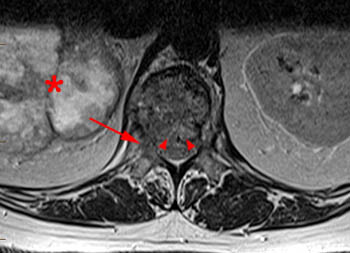
Figure 12:
A T2-weighted axial at the T12 pedicle level. Tumor has extended dorsally into the spinal canal creating mild canal stenosis (arrowheads). The pattern or configuration of extension of bone into the spinal canal is not helpful in differentiating the type of fracture. The posterior elements are involved on the right (arrow). A large mass is present in the right retroperitoneal space (asterisk) which is assumed to be renal in origin. This patient was subsequently found to have extensive systemic metastases including to the brain and skull base, and was treated palliatively.
A 64 year-old male with acute back pain and no history of cancer (13a-15a). Classic pathologic fracture.
Some exceptions to the “rules” (16a,17a). A 52 year-old female with known breast cancer.
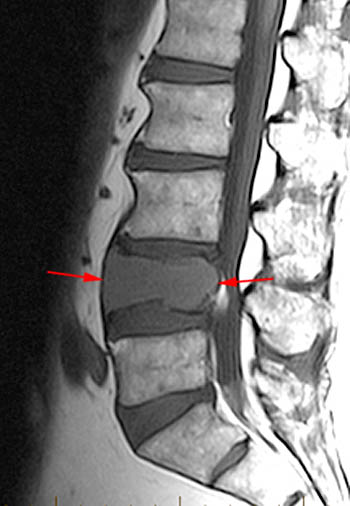
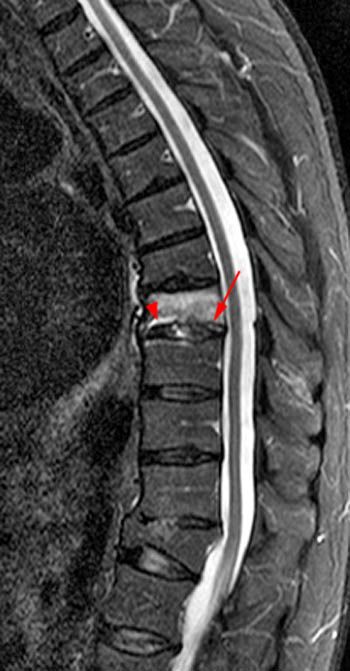
Figure 16:
The T1-weighted Sagittal demonstrates multifocal metastatic tumor. A mild to moderate compression fracture of L1 is visible with preservation of normal marrow signal dorsally (asterisks). If this were an isolated fracture in a patient with a history of acute trauma, its imaging appearance might create some uncertainty. Note that there is slight convexity of the posterior cortex of L1 (arrow) despite adjacent "normal" appearing marrow.
More exceptions to the “rules” (18a-21a). A 58 year-old female with myeloma and a previously treated pathologic L2 fracture diagnosed 3 months earlier.
Figure 18:
A T2-weighted sagittal image with fat-saturation. On this single image, a known pathologic fracture of L2 mimics a fracture with benign characteristics with its substantial preserved marrow (asterisks), lack of a distinct intraosseous mass and concave posterior cortical margin (arrow). The biconcavity of the superior and inferior endplates (arrowheads) is a very common finding in benign osteoporotic fractures and Schmorl's nodes.
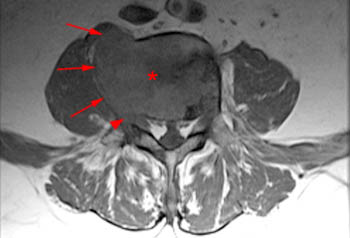
Figure 19:
The corresponding T1-weighted sagittal image demonstrates some preserved marrow signal anteriorly (asterisks). It is included to make another point, however. The posterior cortex of the vertebral body centrum (arrows) appears intact on this sequence, but in actuality it is not, as demonstrated on a CT scan performed 3 days earlier (20a).
Figure 20:
CT image at the L2 pedicle level windowed to exaggerate bone. The posterior cortex is not visible and has been destroyed or demineralized (arrows). Therefore the dark line imitating the posterior cortex of the L2 centrum on MR in (19a) is not cortex but likely a combination of posterior longitudinal ligament and dura mater.
Figure 21:
A pre-treatment T2-weighted sagittal image obtained 3 months earlier than Figs 18a-20a. Signs of malignancy include presence of a distinct mass and convexity of posterior cortex. Progression of spine fracture deformities occur in 50% of patients with myeloma, as in this case when the height of the centrum in 18a is compared to 21a.
Another MR imaging tool which has been developed to aid in diagnosing malignant fractures is Diffusion Weighted Imaging (DWI). Diffusion is based upon the observation that molecules of water in a gradient field undergo phase dispersion due to random motion leading to a loss of measurable signal which can be calculated as the ADC (Apparent Diffusion Coefficient). Generally speaking, extracellular water disperses to a greater extent than intracellular water. Therefore the denser the cellularity in a volume of tissue, the less the water within that tissue will disperse and lose signal. Superficially the qualitative differentiation of a benign versus a malignant vertebral body fracture appears simple enough: a fractured bone filled with cancerous cells should have relatively brighter signal than a non-malignant fracture. Unfortunately, the results of numerous studies have not uniformly concurred with this simple proposition for a variety of reasons including: DWI is very sensitive to patient motion, CSF pulsation and flow in spinal vessels limits some studies, DWI is very sensitive to magnetic susceptibility artifact arising from bone-tissue interactions, the signal to noise ratio has to be balanced with diffusion contrast, and various diffusion sequences suffer from low spatial resolution. At least ten commercially available pulse sequences with inserted diffusion sensitizing gradients have been created to deal with some of these issues. Although fat has a low diffusivity and could theoretically alter either qualitative or quantitative assessment, in the typical clinical scenario much of the fat in the region of interest in the fractured vertebral body has been replaced by edema or cellular infiltration. It is interesting to review the literature on this subject over the last decade 20,21,22,23,24,25,26,27,28,29,30,31,32,33,34. While some authors reported 100% accuracy of this technique in differentiating a benign from a malignant fracture, others felt that the technique was not useful due to cases with false positive or false negative results 20,25,27,28,30,32. Three of five metastases in Castillo’s study were false negatives 20. Zhou found a 50% false positivity and a 27% false negativity in a qualitative review of their 27 patients 25. Some authors have written that a particular sequence provides a very good qualitative evaluation as long as measurements of the actual coefficients are not required while others contend that quantification of ADC is critical for diagnostic accuracy. However, Zhou demonstrated overlap between benign and malignant lesions when measuring their ADCs. An expression that is sometimes used to describe diffusion imaging and its accuracy in differentiating a benign from a pathologic fracture is controversial. There are potentially many reasons for these discrepancies including differences in the type of sequence, differences in the technical aspects such as b values in similar sequences, and differences in the stage or type of cancer. For example, one would not normally expect osteoblastic, osteolytic, treated necrotic, hemorrhagic or mucin producing, or dense hypercellular metastases to exhibit uniform diffusion characteristics. Additionally, the reparative process in an osteoporotic fracture includes proliferation of granulation tissue and fibrosis and a benign fracture may occur in a degenerative or post traumatic sclerotic vertebra, either of which could alter the environment of accumulated edema within the fracture. Use of a single “high” b-value can lead to a false positive artifact referred to as “T2 shine through” particularly on Echo-Planar (EPI) acquisitions, which have been the most common technique utilized in the spine. Since the accuracy of differentiating a benign from a malignant fracture approaches 95% on conventional sequences, confidence in any other imaging technique needs to be quite high to avoid biopsy. Therefore if desired, DWI can complement but not replace conventional sequences for spine fracture evaluation.
False positive DWI. A 73 year old female with acute back pain (22a-24a).
Figure 22:
The T1-weighted sagittal image demonstrates a large amount of edema or pathologic tissue infiltration within the L3 centrum (asterisk). Prevertebral soft tissue swelling is present. Note anterior epidural material of indeterminate origin (arrows). Posterior cortical margin indistinct (arrowheads).
Figure 23:
A corresponding STIR sagittal image reveals lower signal superiorly in the centrum, possibly due to trabecular condensation/ compaction (arrowheads). Hyperintense prevertebral soft tissue extends cranially (asterisks). Anterior epidural material compresses the thecal sac (arrow) and may arise from the disc as an extrusion, from the epidural space as a hematoma or from the vertebral body which has lost its concavity and cortical distinctness. Based on these conventional sequences the fracture could be benign or pathologic.
Hypercellularity restricts diffusion (25a,26a)
Figure 25:
A T1-weighted sagittal image in a 72 year-old male is diffusely abnormal, with normal fatty marrow replaced with low signal indicating increased cellularity. Such a finding may be attributable to a large variety of conditions including hyperplastic anemias or a myeloproliferative disorder such as chronic lymphocytic leukemia.
Benign Fractures. A 70 year-old farmer suffered a recent lifting injury (27a,28a).
Figure 27:
The T1-weighted sagittal images demonstrates a mild to moderate fracture of T8 with near total replacement of marrow signal (arrow). Signs of a benign nature are lack of a distinct osseous mass, adjacent benign older fractures (arrowheads), maintenance of concavity of posterior cortex, and the lack of a prevertebral mass.
Some institutions have studied the usefulness of chemical shift imaging in evaluating the type of spine fracture when the diagnosis is in doubt. Chemical shift imaging, also called in-phase/out-of-phase imaging, is based on the idea that neoplasms replace fat within the marrow in a metastatic fracture. Since both osteoporotic and neoplastic fractures demonstrate a qualitative decline in signal on visual inspection, quantitation is necessary. However overlap in types of fractures still occur, leading to some false positive and false negative results36,37,38,39. Ragab recently advocated increasing the percent of signal drop off from 20% to 35% to increase confidence that a fracture is malignant in origin39.
Conclusion
A need to determine whether a fracture of a vertebral body is benign or malignant is a common task for a radiologist interpreting MRI. Thankfully, differentiation is only occasionally a dilemma, as with proper technique and interpretation skills, the overwhelming majority of such fractures can be differentiated accurately with routine MR imaging sequences.
References
1 Park DK, Munns J, Singh K. Spinal Metastatic Disease: Current Treatment Algorithms. Contemporary Spine Surgery. 2010 Jan;11(1):1-6.
2 Gilbert RW, Kim JH, Posner JB. Epidural spinal cord compression from metastatic tumor: diagnosis and treatment. Ann Neurol. 1978 Jan;3(1):40-51.
3 Kim SH, Smith SE, Mulligan ME. Hematopoietic tumors and metastases involving bone. Radiol Clin North Am. 2011 Nov;49(6):1163-83.
4 Griffith JF, Guglielmi G. Vertebral fracture. Radiol Clin North Am. 2010 May;48(3):519-29.
5 Yuh WT, Quets JP, Lee HJ, Simonson TM, Michalson LS, Nguyen PT, Sato Y, Mayr NA, Berbaum KS. Anatomic distribution of metastases in the vertebral body and modes of hematogenous spread. Spine (Phila Pa 1976). 1996 Oct 1;21(19):2243-50.
6 Yuh WT, Zachar CK, Barloon TJ, Sato Y, Sickels WJ, Hawes DR. Vertebral compression fractures: distinction between benign and malignant causes with MR imaging. Radiology. 1989 Jul;172(1):215-8.
7 Baker LL, Goodman SB, Perkash I, Lane B, Enzmann DR. Benign versus pathologic compression fractures of vertebral bodies: assessment with conventional spin-echo, chemical-shift, and STIR MR imaging. Radiology. 1990 Feb;174(2):495-502.
8 Malghem J, Maldague B, Labaisse MA, Dooms G, Duprez T, Devogelaer JP, Vande Berg B. Intravertebral vacuum cleft: changes in content after supine positioning. Radiology. 1993 May;187(2):483-7.
9 Mirowitz SA, Apicella P, Reinus WR, Hammerman AM. MR imaging of bone marrow lesions: relative conspicuousness on T1-weighted, fat-suppressed T2-weighted, and STIR images. AJR Am J Roentgenol. 1994 Jan;162(1):215-21.
10 An HS, Andreshak TG, Nguyen C, Williams A, Daniels D. Can we distinguish between benign versus malignant compression fractures of the spine by magnetic resonance imaging? Spine (Phila Pa 1976). 1995 Aug 15;20(16):1776-82.
11 Rupp RE, Ebraheim NA, Coombs RJ. Magnetic resonance imaging differentiation of compression spine fractures or vertebral lesions caused by osteoporosis or tumor. Spine (Phila Pa 1976). 1995 Dec 1;20(23):2499-503.
12 Cuénod CA, Laredo JD, Chevret S, Hamze B, Naouri JF, Chapaux X, Bondeville JM, Tubiana JM. Acute vertebral collapse due to osteoporosis or malignancy: appearance on unenhanced and gadolinium-enhanced MR images. Radiology. 1996 May;199(2):541-9.
13 Moulopoulos LA, Yoshimitsu K, Johnston DA, Leeds NE, Libshitz HI. MR prediction of benign and malignant vertebral compression fractures. J Magn Reson Imaging. 1996 Jul-Aug;6(4):667-74.
14 Baur A, Stäbler A, Arbogast S, Duerr HR, Bartl R, Reiser M. Acute osteoporotic and neoplastic vertebral compression fractures: fluid sign at MR imaging. Radiology. 2002 Dec;225(3):730-5.
15 Vanel D. MRI of bone metastases: the choice of the sequence. Cancer Imaging.2004 Mar 23;4(1):30-5.
16 Wiggins MC, Sehizadeh M, Pilgram TK, Gilula LA. Importance of intravertebral fracture clefts in vertebroplasty outcome. AJR Am J Roentgenol. 2007 Mar;188(3):634-40.
17 Algra PR, Heimans JJ, Valk J, Nauta JJ, Lachniet M, Van Kooten B. Do metastases in vertebrae begin in the body or the pedicles? Imaging study in 45 patients. AJR Am J Roentgenol. 1992 Jun;158(6):1275-9.
18 Moulopoulos LA, Maris TG, Papanikolaou N, Panagi G, Vlahos L, Dimopoulos MA. Detection of malignant bone marrow involvement with dynamic contrast-enhanced magnetic resonance imaging. Ann Oncol. 2003 Jan;14(1):152-8.
19 Costa FM, Canella C, Gasparetto E. Advanced magnetic resonance imaging techniques in the evaluation of musculoskeletal tumors. Radiol Clin North Am.2011 Nov;49(6):1325-58.
20 Castillo M, Arbelaez A, Smith JK, Fisher LL. Diffusion-weighted MR imaging offers no advantage over routine noncontrast MR imaging in the detection of vertebral metastases. AJNR Am J Neuroradiol. 2000 May;21(5):948-53.
21 Spuentrup E, Buecker A, Adam G, van Vaals JJ, Guenther RW. Diffusion-weighted MR imaging for differentiation of benign fracture edema and tumor infiltration of the vertebral body. AJR Am J Roentgenol. 2001 Feb;176(2):351-8.
22 Byun WM, Shin SO, Chang Y, Lee SJ, Finsterbusch J, Frahm J. Diffusion-weighted MR imaging of metastatic disease of the spine: assessment of response to therapy. AJNR Am J Neuroradiol. 2002 Jun-Jul;23(6):906-12.
23 Chan JH, Peh WC, Tsui EY, Chau LF, Cheung KK, Chan KB, Yuen MK, Wong ET, Wong KP. Acute vertebral body compression fractures: discrimination between benign and malignant causes using apparent diffusion coefficients. Br J Radiol. 2002 Mar;75(891):207-14.
24 Baur A, Dietrich O, Reiser M. Diffusion-weighted imaging of the spinal column. Neuroimaging Clin N Am. 2002 Feb;12(1):147-60.
25 Zhou XJ, Leeds NE, McKinnon GC, Kumar AJ. Characterization of benign and metastatic vertebral compression fractures with quantitative diffusion MR imaging. AJNR Am J Neuroradiol. 2002 Jan;23(1):165-70.
26 Falcone S. Diffusion-weighted imaging in the distinction of benign from metastatic vertebral compression fractures: is this a numbers game? AJNR Am J Neuroradiol. 2002 Jan;23(1):5-6.
27 Castillo M. Diffusion-weighted imaging of the spine: is it reliable? AJNR Am J Neuroradiol. 2003 Jun-Jul;24(6):1251-3.
28 Maeda M, Sakuma H, Maier SE, Takeda K. Quantitative assessment of diffusion abnormalities in benign and malignant vertebral compression fractures by line scan diffusion-weighted imaging. AJR Am J Roentgenol. 2003 Nov;181(5):1203-9.
29 Raya JG, Dietrich O, Birkenmaier C, Sommer J, Reiser MF, Baur-Melnyk A. Feasibility of a RARE-based sequence for quantitative diffusion-weighted MRI of the spine. Eur Radiol. 2007 Nov;17(11):2872-9.
30 Karchevsky M, Babb JS, Schweitzer ME. Can diffusion-weighted imaging be used to differentiate benign from pathologic fractures? A meta-analysis. Skeletal Radiol. 2008 Sep;37(9):791-5.
31 Oztekin O, Ozan E, Hilal Adibelli Z, Unal G, Abali Y. SSH-EPI diffusion-weighted MR imaging of the spine with low b values: is it useful in differentiating malignant metastatic tumor infiltration from benign fracture edema? Skeletal Radiol. 2009 Jul;38(7):651-8.
32 Dietrich O, Biffar A, Reiser MF, Baur-Melnyk A. Diffusion-weighted imaging of bone marrow. Semin Musculoskelet Radiol. 2009 Jun;13(2):134-44.
33 Bley TA, Wieben O, Uhl M. Diffusion-weighted MR imaging in musculoskeletal radiology: applications in trauma, tumors, and inflammation. Magn Reson Imaging Clin N Am. 2009 May;17(2):263-75.
34 Tanenbaum LN. Diffusion Imaging in the Spine. Applied Radiology. 2011 April;4:9-15.
35 Yu S, Rosenbaum A, Poe LB, Fredrickson BE. The Lumbar Spine In: MRI of the Musculoskeletal System. Chan WP, et al. WB Saunders, 1994, pp 127-172.
36 Zajick DC Jr, Morrison WB, Schweitzer ME, Parellada JA, Carrino JA. Benign and malignant processes: normal values and differentiation with chemical shift MR imaging in vertebral marrow. Radiology. 2005 Nov;237(2):590-6.
37 Erly WK, Oh ES, Outwater EK. The utility of in-phase/opposed-phase imaging in differentiating malignancy from acute benign compression fractures of the spine. AJNR Am J Neuroradiol. 2006 Jun-Jul;27(6):1183-8.
38 Swartz PG, Roberts CC. Radiological reasoning: bone marrow changes on MRI. AJR Am J Roentgenol. 2009 Sep;193(3 Suppl):S1-4.
39 Ragab Y, Emad Y, Gheita T, Mansour M, Abou-Zeid A, Ferrari S, Rasker JJ. Differentiation of osteoporotic and neoplastic vertebral fractures by chemical shift {in-phase and out-of phase} MR imaging. Eur J Radiol. 2009 Oct;72(1):125-33.