Clinical History
An 18 year-old basketball player presents with anterior thigh pain and swelling 4 weeks after being struck in the thigh during a game. (1A) T1-weighted coronal, (1B) fat-suppressed proton density-weighted axial, (1C) fat-suppressed T2-weighted coronal, (1D) post-contrast fat-suppressed T1-weighted axial MR images, and (1E) a lateral radiograph of the thigh are provided. What are the findings? What is your diagnosis?
Findings
Figure 2:
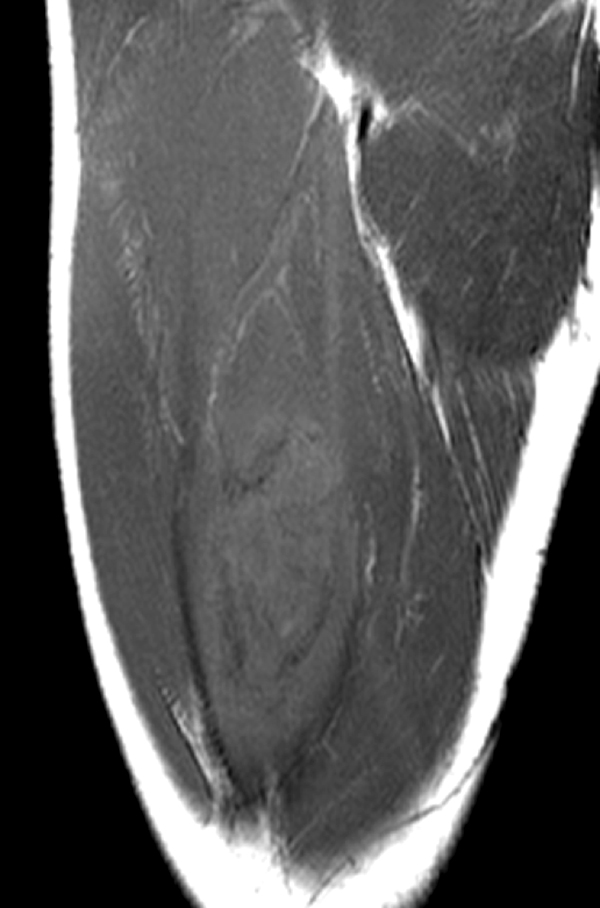
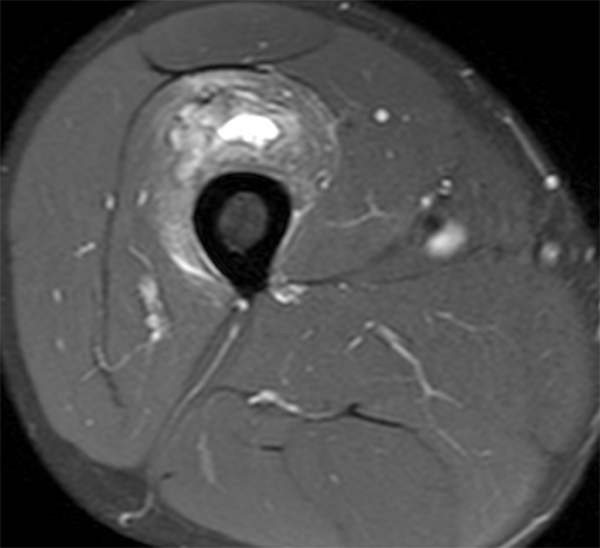
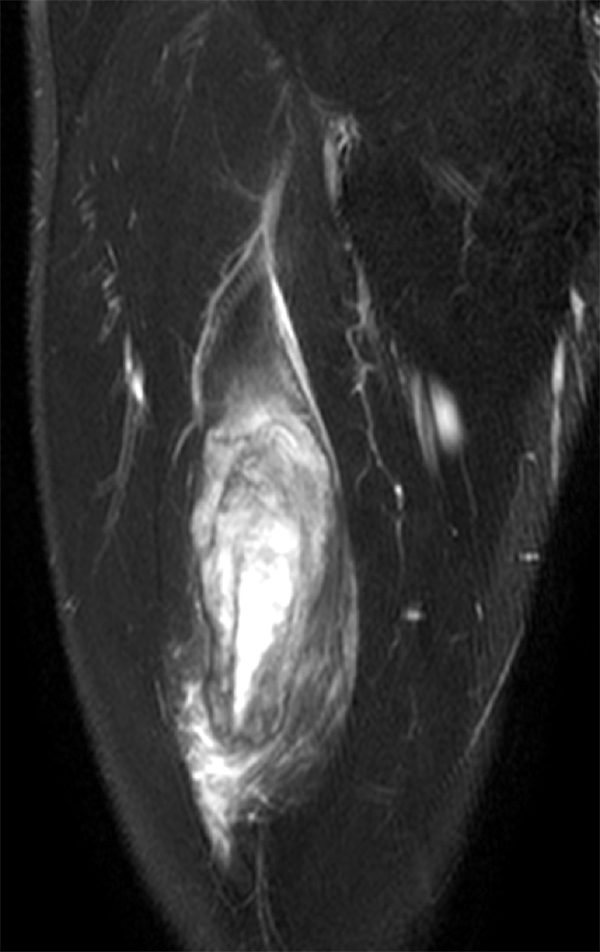
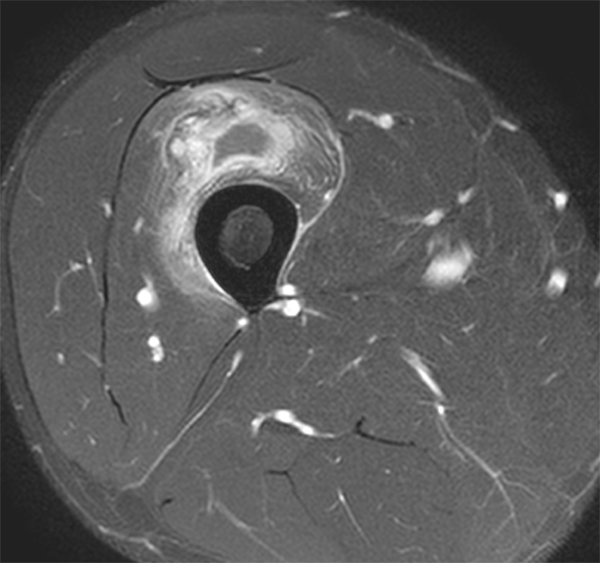
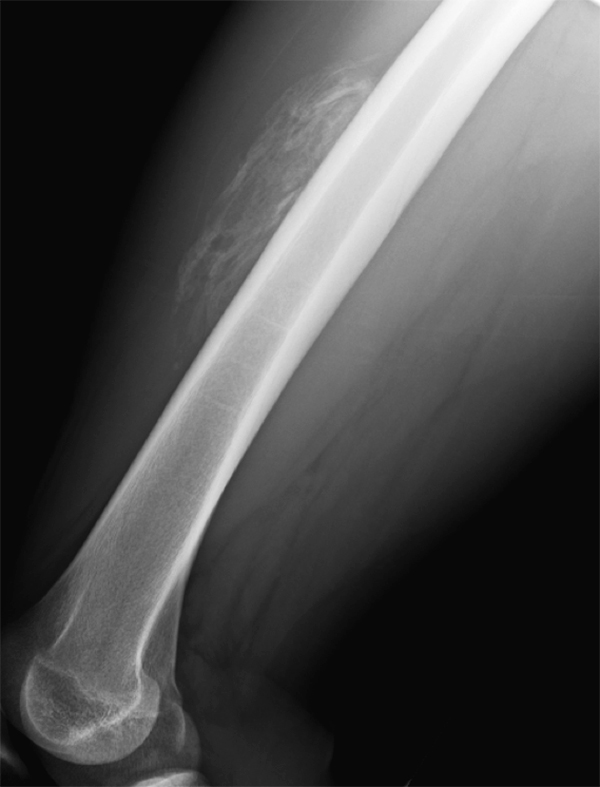
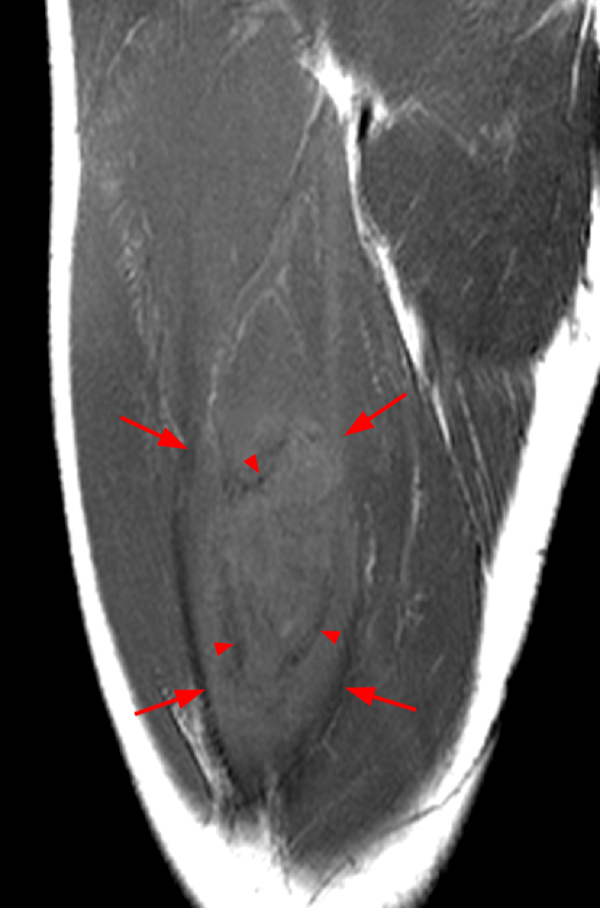
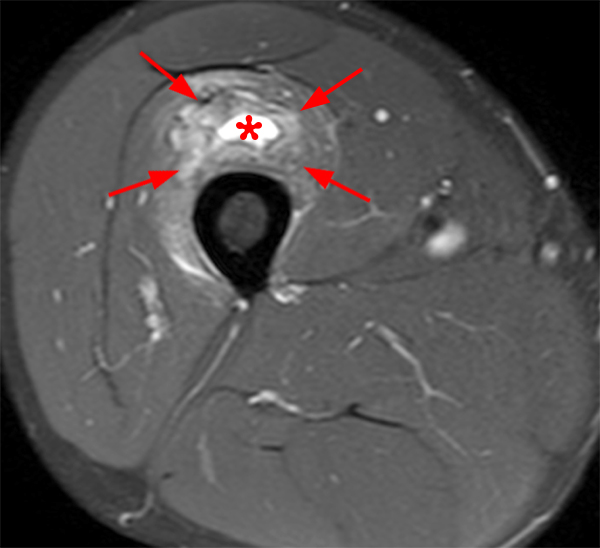
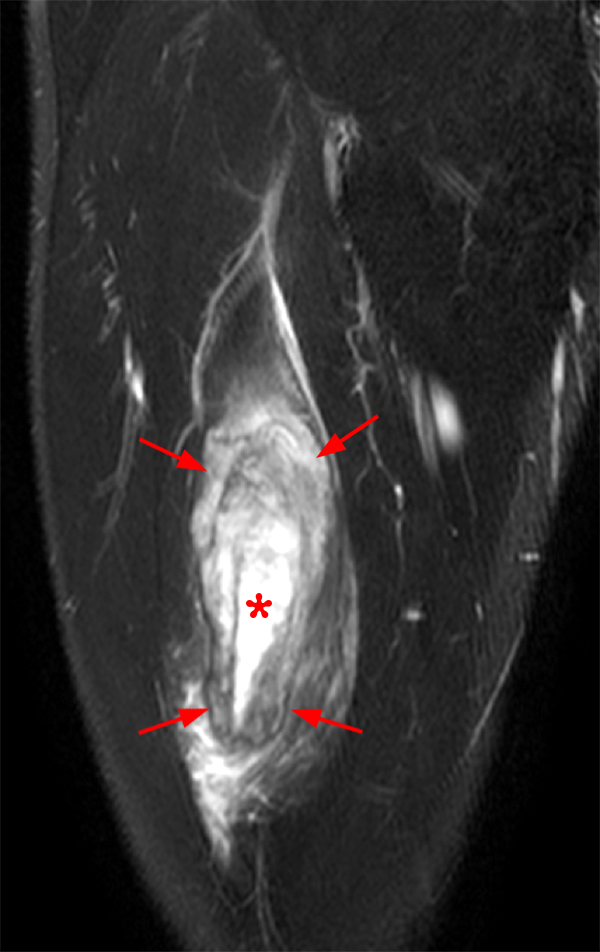
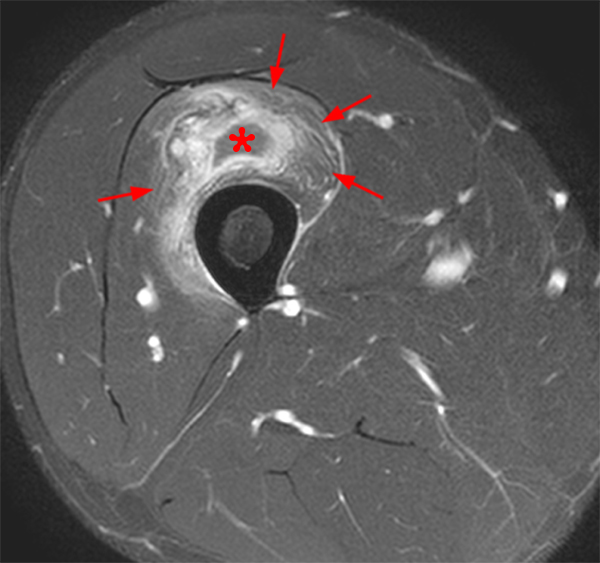
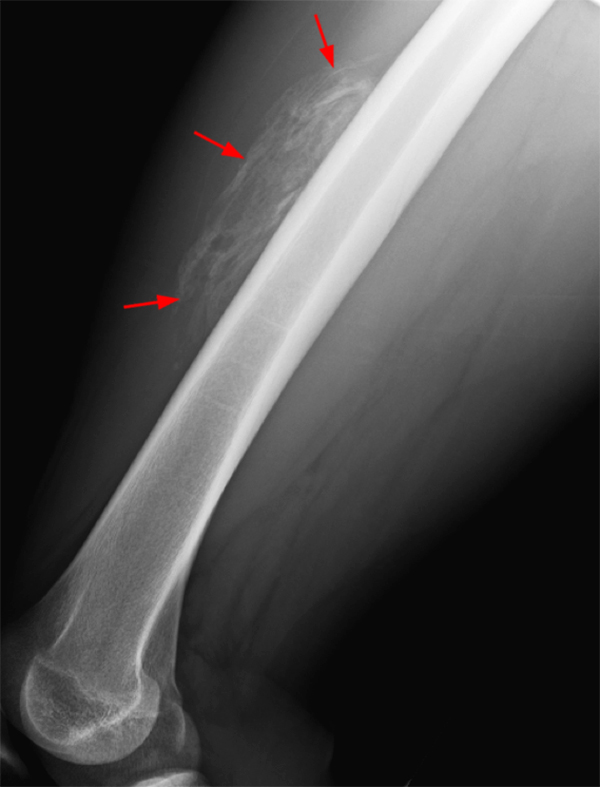
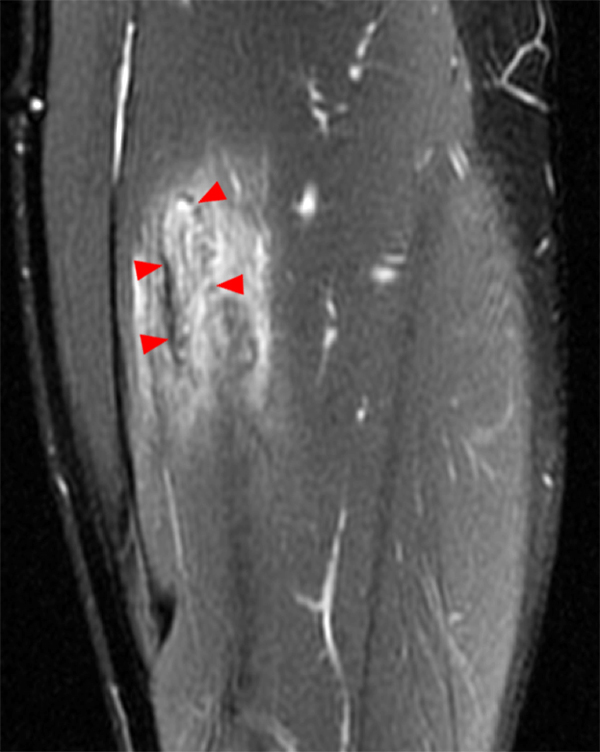
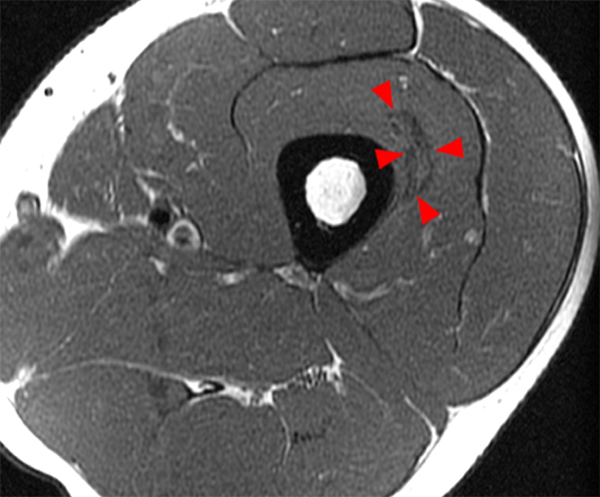
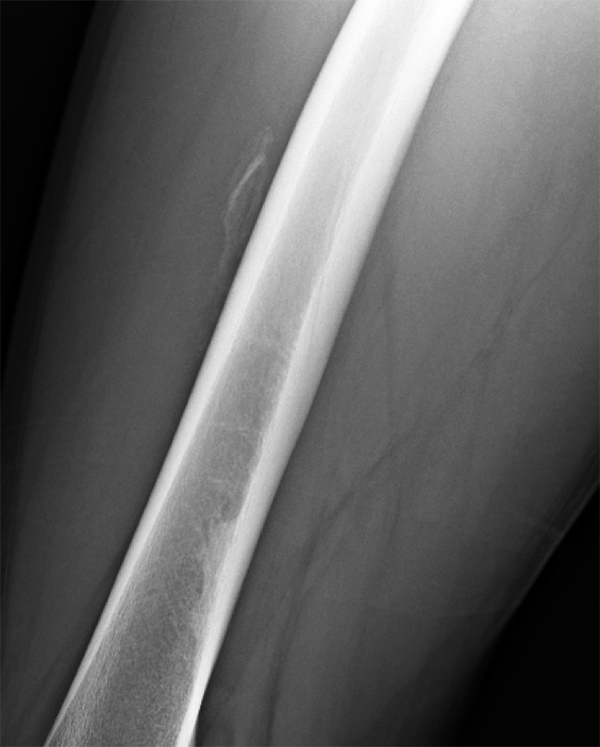
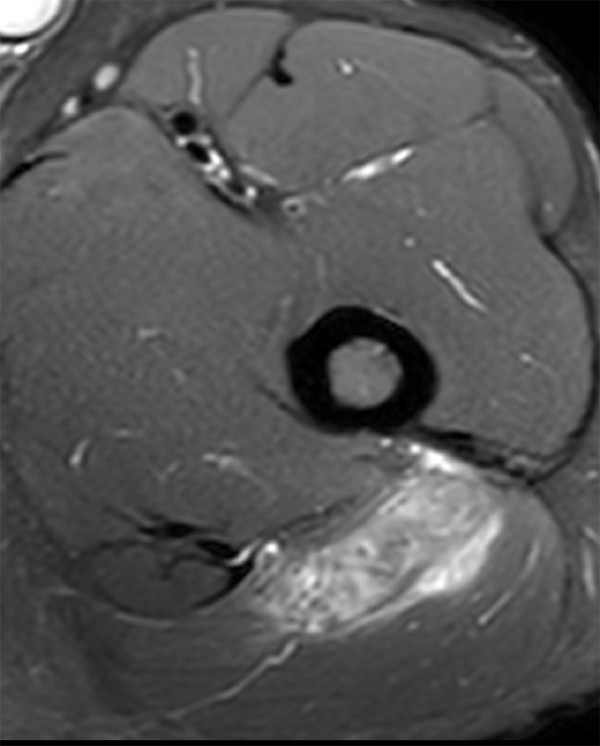
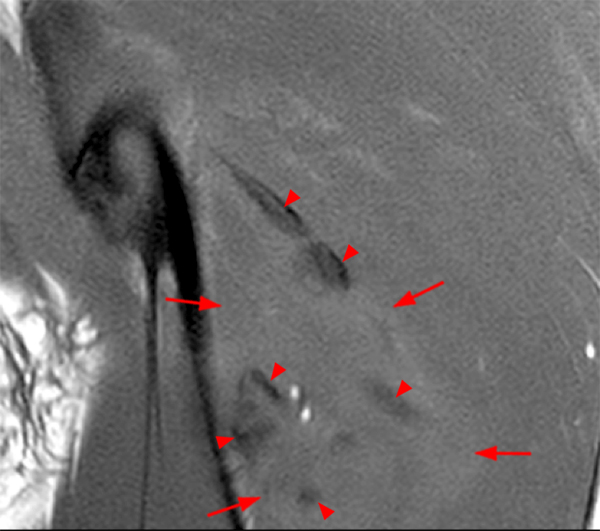
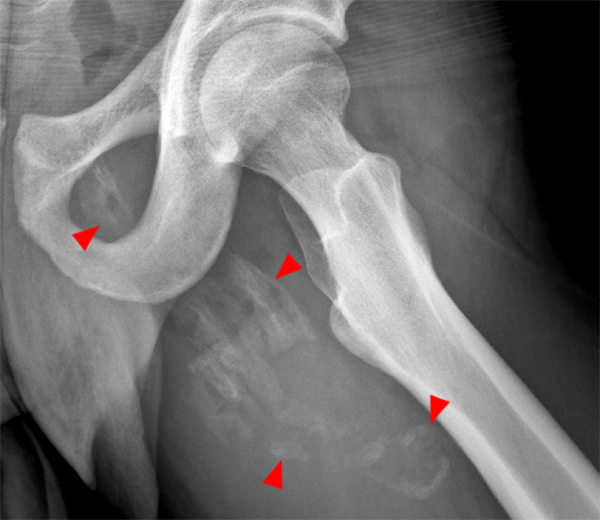
(2A) An ovoid region of soft-tissue swelling slightly hyperintense to muscle (arrows) is seen on the T1-weighted coronal image. Subtle areas of peripheral low signal intensity (arrowheads) are seen within the lesion. (2B,C) The fat-suppressed proton density-weighted axial and T2-weighted coronal images reveal a hyperintense lesion within the vastus intermedius muscle (arrows) with a small region of central fluid signal intensity (asterisks). The abnormality demonstrates lace-like intramuscular enhancement (arrows) with thicker enhancement surrounding the central fluid (asterisk) on the (2D) fat-suppressed T1-weighted post-contrast view. Peripheral calcifications (arrows) are readily apparent on the corresponding (2E) lateral radiograph of the thigh.
Diagnosis
Myositis Ossificans.
Introduction
Although the term myositis ossificans is not without controversy, it is a generally accepted descriptor for a process that results in intramuscular bone proliferation.1 As far back as the early 20th century, descriptions of post-traumatic ossifications in muscles were published in the scientific literature2, and the difficulty in distinguishing these from sarcoma was described.3 In 1991, Kransdorf et al. defined myositis ossificans as a benign, solitary, self-limiting, ossifying soft-tissue mass typically occurring within skeletal muscle4, and this remains a reasonable definition of the term. Other descriptors of the process such as myositis ossificans circumscripta and myositis ossificans traumatica have been used, though such terminology provides little added value. The vast majority of myositis ossificans is acquired, though there is a rare, disabling genetic entity known as fibrodysplasia ossificans progressiva in which similar intramuscular ossifications occur, typically multiple and resulting in progressive deformity and immobility.5
Etiology, distribution, and clinical features
Myositis ossificans can be seen at any age, though it is rare in children under 106 and is most common in adolescents and young adults, with over half of cases occurring in the third decade.7 The most common sites of involvement are the thigh and arm, in particular the quadriceps and brachialis muscles. The majority of patients recall a specific injury or repetitive minor trauma, such as a football player who has sustained a blow to the thigh in a game, and evidence of prior trauma is helpful in making the diagnosis. However, an inciting event may not be recalled in as many as 40% of patients. In the latter case, it has been postulated that many of these patients have simply overlooked a minor injury.8
Clinically, patients frequently report muscle pain persisting longer than expected for a muscle strain or contusion, perhaps due to mechanical irritation of surrounding structures by the process.9 Paresthesias, weakness, lymphedema, and even venous thrombosis have been reported due to compression of neurovascular structures.10 Symptoms tend to improve as the lesions mature, and thus patients who are imaged late in the process may have few or even no associated symptoms.
Pathophysiology
As myositis ossificans develops it passes through three characteristic phases, commonly described as early, intermediate, and mature.11 The early phase lasts from as little as one to as much as four weeks and is characterized by an acute inflammatory period before ossification occurs, typically lacking calcification on plain radiographs. This phase is thought to lead to dysregulation of stem cells in the region of injury and inflammation.12 An inflammatory cascade results in the release of cytokines that act upon skeletal muscle, causing them to undergo transition to mesenchymal stem cells that may differentiate into chondrocytes or osteoblasts. In the intermediate phase, occurring four to eight weeks after inception, osteoid matrix is secreted and calcifications become visible on plain films. The late or mature phase is characterized by bone production at the periphery of the lesion and in the ensuing months fatty metaplasia may appear at the lesion center.13 Over time, regression of the lesion may also occur.
MRI of myositis ossificans
The imaging appearance of myositis ossificans varies in accordance with its stage of evolution. In early myositis ossificans, lesions are typically isointense to muscle on T1-weighted images and heterogeneously hyperintense on T2-weighted images. Edema within surrounding muscles is typically present on T2-weighted images. Diffuse enhancement is most common following administration of paramagnetic contrast, though peripheral enhancement may be encountered, particularly in patients with a central hematoma. Wang et al. recently reported two MR imaging patterns felt to be helpful in the diagnosis of early myositis ossificans14(Figure 3). On T2-weighted images or post contrast T1-weighted images, the increased signal intensity of the lesion in an expanded muscle with intact muscle fibers results in a “striate pattern” when images are obtained in a plane parallel to the muscle fibers. This appearance was found to be the most reliably present pattern on MR images of early myositis ossificans. Less commonly, in planes perpendicular to muscle fibers, the relatively hypointense intact muscle fibers interspersed in the region of edema results in what was described as a “checkerboard-like pattern” on T2-weighted or post-contrast T1-weighted images. Histologically, early myositis ossificans is made up of mesenchymal cells and fibroblasts undergoing numerous mitoses, resulting in a pseudo-sarcomatous appearance both on imaging and histology.15
Figure 3:
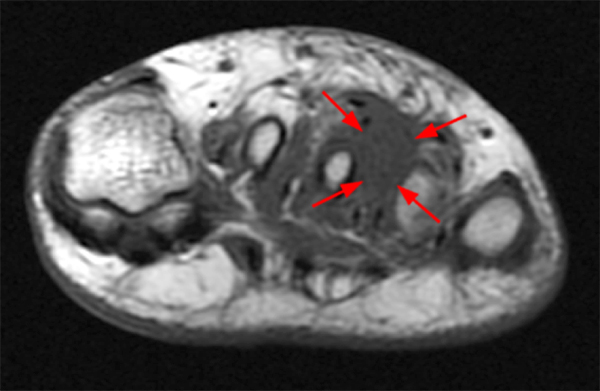
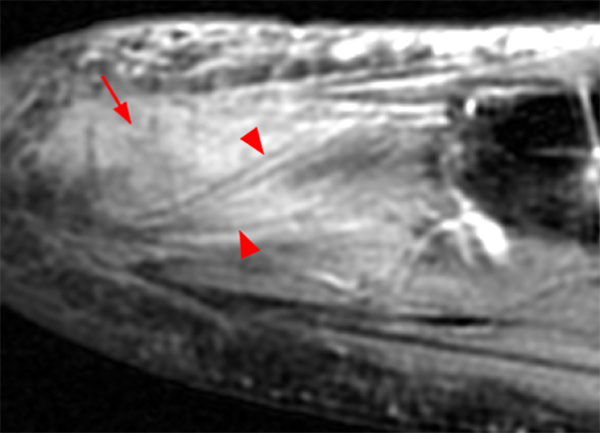
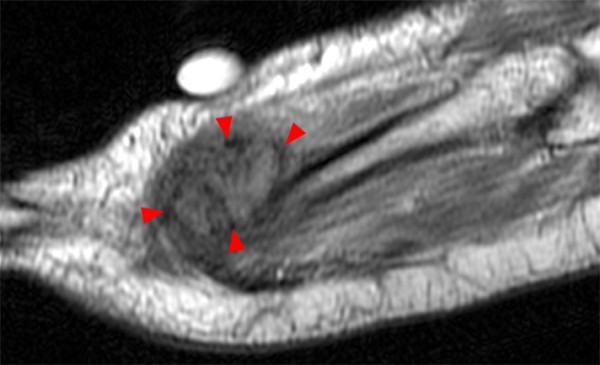
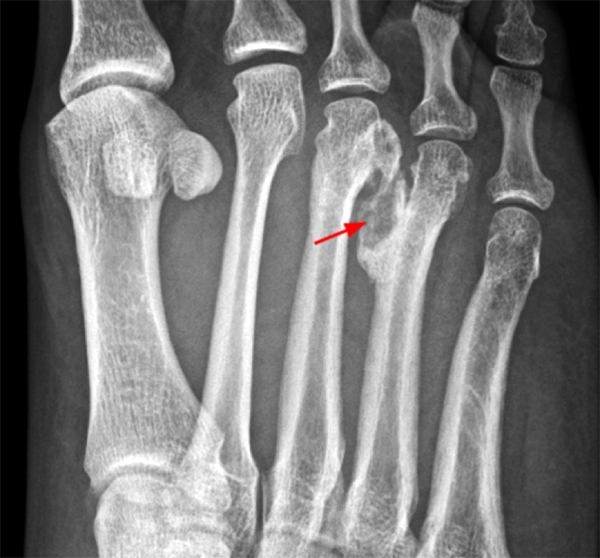
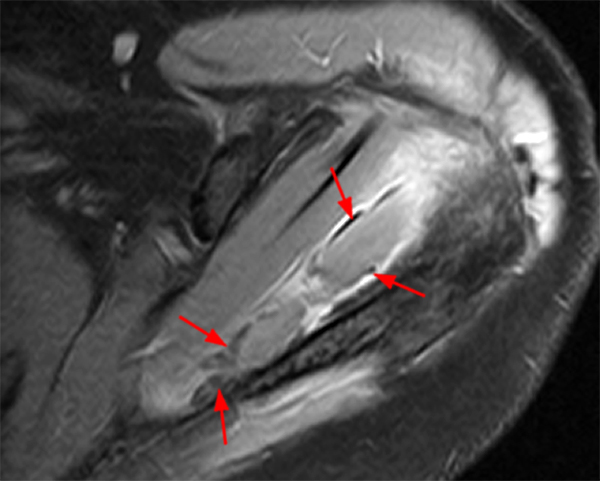
A 43 year-old female presents with forefoot pain and swelling 2 weeks after dropping a laptop on her foot. (3A) A T1-weighted coronal (short axis) image demonstrates nodular soft tissue thickening dorsal to the 3rd interspace (arrows), relatively isointense to normal muscle. A (3B) fat-suppressed proton density-weighted sagittal image reveals the hyperintense nodular lesion anteriorly (arrow). Normal muscle fibers result in a striate pattern of linear low signal intensities (arrowheads) on images obtained parallel to the muscle fibers. On the (3C) post-contrast fat-suppressed T1-weighted coronal view, the central nodular region enhances more intensely than surrounding muscle. In this plane perpendicular to muscle fibers, lower signal intensity normal muscle fibers within the lesion result in a “checkerboard” pattern (inset). Plain radiographs demonstrated no visible calcifications (not shown).
If myositis ossificans is imaged in the intermediate stage (Figures 4,5), T1-weighted images typically demonstrate a lesion that is isointense to normal muscle, though both hypointense and hyperintense lesions may be encountered. T2-weighted images continue to demonstrate heterogeneously increased signal intensity, sometimes with fluid signal intensity centrally due to hemorrhage. Enhancement persists but is often less intense than with acute lesions. At this stage, osteoblasts begin to produce bone and thus calcifications are detectable on plain films, classically peripherally placed. The calcifications are subtle on MRI but may be detectable via a low signal intensity rim, in which case the diagnosis of myositis ossificans becomes highly likely.
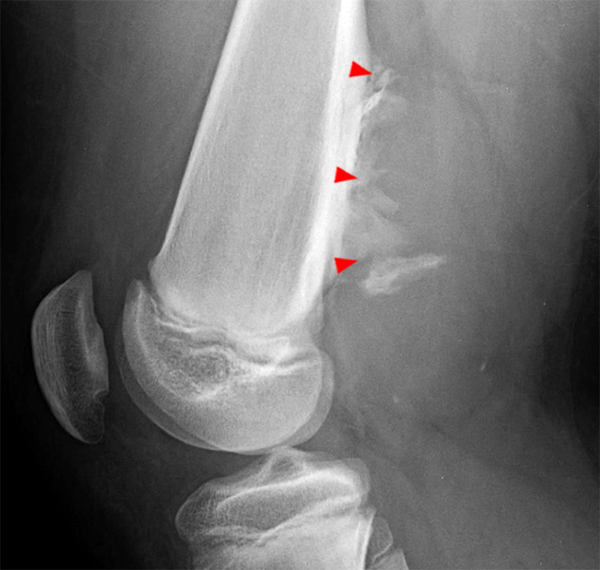
Figure 4:
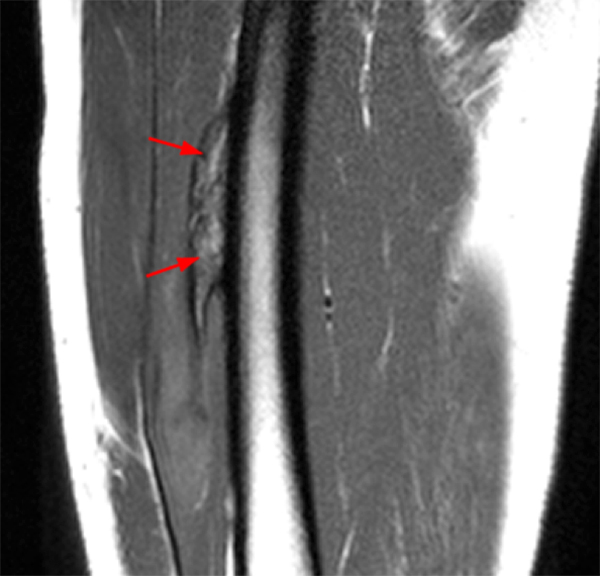
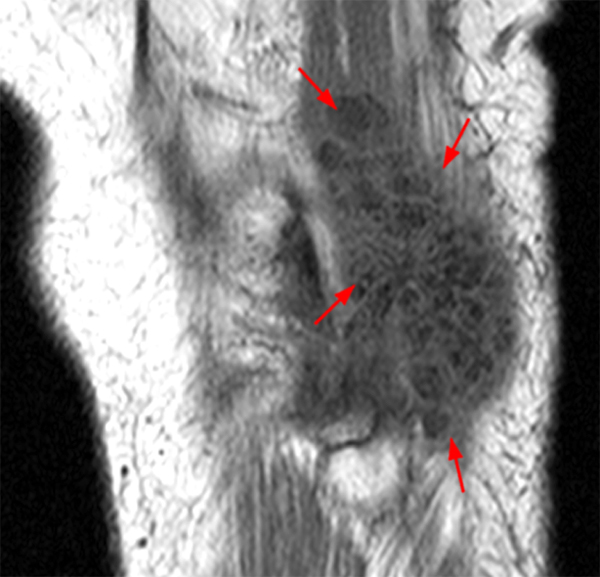
(4A) A fat-suppressed T2-weighted sagittal image of the thigh just medial to the femur from a 17 year-old male injured playing football 6 weeks earlier reveals an edematous lesion within the vastus intermedius muscle with subtle peripheral low signal intensity (arrowheads). (4B) The T1-weighted axial view confirms a peripheral low signal intensity rim (arrowheads). The calcifications are readily apparent on the (4C) corresponding lateral radiograph of the femur.
Figure 5:
A 20 year-old male presents 5 weeks after falling onto the left hip in a football game. (5A) The fat-suppressed proton density-weighted axial image reveals a mildly heterogeneous edematous lesion within the gluteus maximus muscle. On (5B) the T1-weighted coronal image, the lesion (arrow) is mildly hyperintense to surrounding muscle and demonstrates peripheral low signal foci (arrowheads) compatible with calcification. The calcifications (arrowheads) are easily seen on (5C) the corresponding oblique frontal radiograph of the thigh.
In the mature phase of myositis ossificans, edema tends to resolve and a well-corticated peripheral rim of ossification is present.16 In some patients, heterogeneous signal characteristics may persist, though the peripheral ossification typically becomes more defined (Figure 6). In other patients, the maturation of the lesion may progress to the point where the MR appearance is similar to adjacent, native bone (Figure 7).
Figure 6:
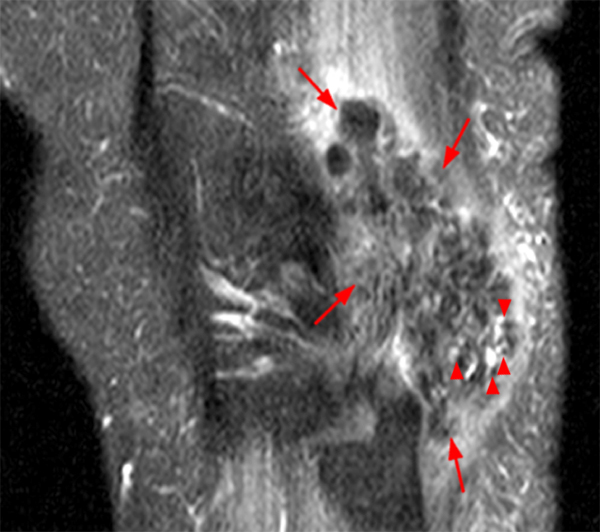
In a follow-up study to Figure 3, the patient returned 6 months after injury for repeat imaging. On (6A) the fat-suppressed T2-weighted coronal image, edema and heterogeneity within the lesion (arrows) persist, though the abnormality appears more well-defined. A (6B) proton density-weighted sagittal view demonstrates clear peripheral low signal intensity (arrowheads) about the lesion. An ossified lesion (arrow) is evident on the (6C) corresponding AP radiograph.
Figure 7:
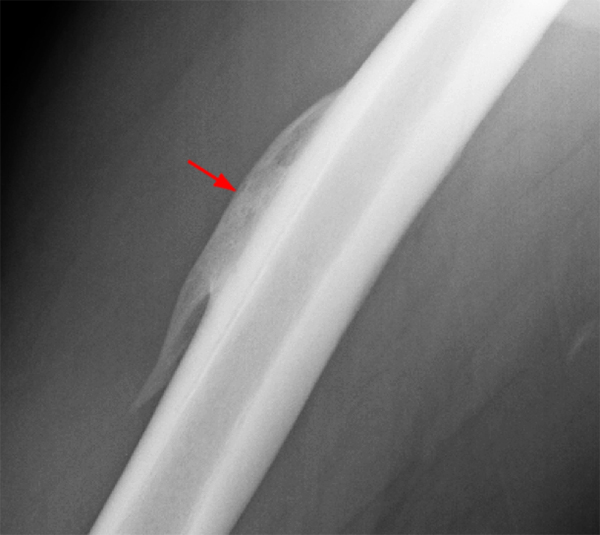
A (7A) T1-weighted sagittal image from a 17 year-old male injured from a fall onto rocks 8 months prior demonstrates an ossified lesion (arrow) along the anterior aspect of the mid shaft of the femur, lying within the vastus intermedius muscle. On (7B) the fat-suppressed proton density-weighted axial image, the lesion (arrow) demonstrates a similar appearance to normal marrow, with mild surrounding intramuscular edema. Mature ossification (arrow) is readily apparent on the (7C) lateral radiograph of the thigh.
Differential diagnosis of myositis ossificans
Given a history of prior trauma, typical MRI and plain film findings, and in particular when lesions are in characteristic locations such as the quadriceps muscles, the diagnosis of myositis ossificans is fairly straightforward. However, a history of trauma is inconsistently present and the imaging findings of myositis ossificans do overlap with other entities that can result in diagnostic confusion.
Hydroxyapatite deposition disease (HADD) is a well-recognized cause of soft tissue calcification, classically seen in the entity known as calcific tendinitis in the shoulder, but the process can affect any extremity. Rarely, the crystal deposition that occurs with HADD can be found in intraosseous and even intramuscular locations (Figure 8), in which MRI findings may appear similar to the intermediate phase of myositis ossificans. HADD differs from myositis ossificans in that the lesions do not undergo ossification and a history of trauma is less common. Although the acute inflammatory phase of HADD can result in edema similar to myositis ossificans, mass-like soft tissue lesions are not a feature, and peripheral calcifications within a muscle are unlikely.
Figure 8:
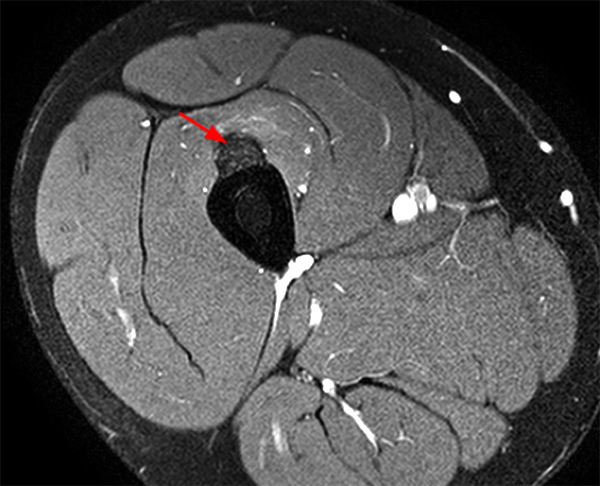
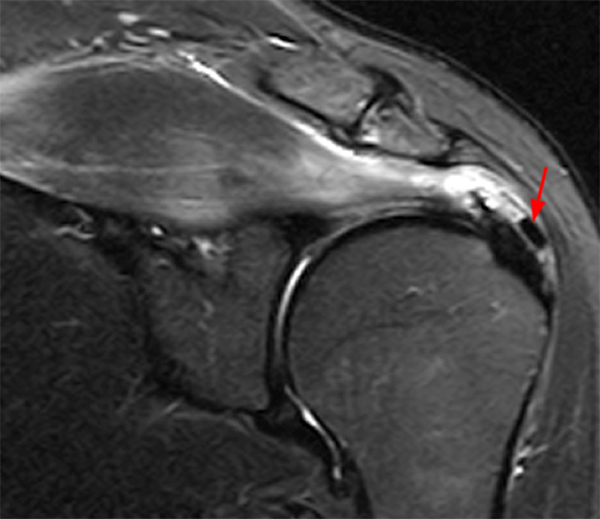
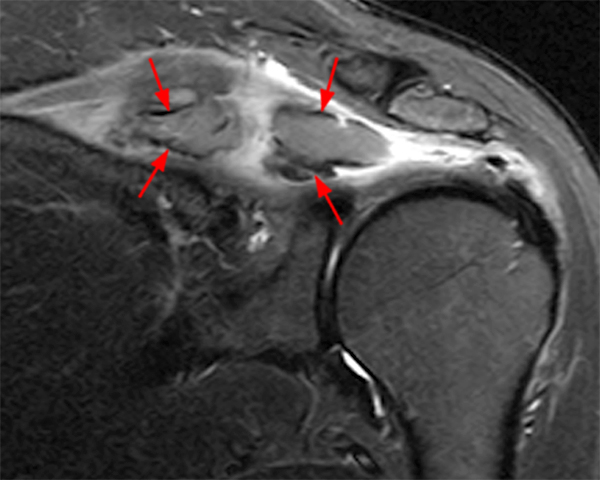
A 52 year-old male with no history of trauma presents with a 2 year history of shoulder pain with recent severe worsening. A (8A) fat-suppressed T2-weighted coronal image demonstrates a low signal intensity focus (arrow) with surrounding edema and bursitis typical in appearance for HADD (calcific peritendinitis). A (8B) more posterior T2-weighted coronal image and a (8C) fat-suppressed proton density weighted axial image reveal large calcifications (arrows) within the posterior supraspinatus muscle belly with florid surrounding edema, in this rare case of intramuscular HADD.
Soft tissue calcifications that appear similar to myositis ossificans can be found in the rare familial disorder known as tumoral calcinosis (Figure 9). An identical appearance is seen much more frequently in patients with renal failure, in which the entity is appropriately termed calcinosis of renal failure. On plain radiographs, the calcifications seen in this entity are frequently denser and more solid appearing than those of myositis ossificans, and the MR appearance differs markedly, as lesions are typically diffusely calcified rather than peripherally calcified, and fluid levels may be seen secondary to the presence of milk of calcium. The distinction is often even easier because the diagnosis of renal failure in these patients is well-established.
Figure 9:
A 37 year-old female with known renal failure presents with a palpable mass at the lateral knee. (9A) An AP radiograph demonstrates multiple dense calcifications at the lateral aspect of the distal femur. (9B) T1-weighted and (9C) fat-suppressed proton density-weighted sagittal images through the mass demonstrate a large calcified lesion (arrows) inseparable from the distal biceps femoris muscle. On the proton density weighted view, small fluid levels (arrowheads) are seen in association with the calcifications at the caudal aspect of the lesion.
By far the most important entity to exclude when evaluating possible myositis ossificans is a soft tissue sarcoma, primarily extraskeletal osteosarcoma or parosteal osteosarcoma. Multiple reports in the literature discuss cases in which myositis ossificans was mistaken for a soft-tissue sarcoma.17,18 Indeed, in the acute phase of myositis ossificans, secretion of osteoid matrix may result in a false positive biopsy for sarcoma, as histologically there may be a pseudo-osteosarcomatous appearance. Luckily, features exist which allow the astute interpreter of MRI to differentiate these entities. The extensive surrounding muscle edema of early myositis ossificans is unusual in most primary neoplasms. Peripheral calcifications/ossifications are typical for myositis ossificans, whereas bone forming sarcomas tend to demonstrate central calcifications that emanate outward (Figure 10). When present, the calcified peripheral rim of myositis ossificans is virtually pathognomonic. Lastly, myositis ossificans usually develops at the diaphysis of a bone, whereas sarcomas are more commonly metaphyseal in location.
Figure 10:
A 12 year-old male presented with knee pain and swelling with running and daily activity. (10A) A T1-weighted sagittal image reveals a large soft tissue mass (arrow) posterior to the distal femoral metaphysis. A low signal intensity region anteriorly (arrowhead) suggests calcification. On (10B) the fat-suppressed T2-weighted sagittal image, the mass is confirmed (arrow). Despite the size of the lesion, note the lack of surrounding soft tissue edema. The corresponding (10C) plain radiograph confirms the calcifications within the lesion (arrowheads). This pattern of calcification which is eccentric and “basal” is felt to be characteristic of surface osteosarcomas. Biopsy results were consistent with low grade parosteal osteosarcoma. Images courtesy of Mini Pathria MD, University of California, San Diego.
Treatment
Conservative treatment of myositis ossificans is often successful, as the process tends to be self-limiting and self-resolving19 It has been suggested that the early use of non-steroidal anti-inflammatory medications may be of benefit in stopping the evolutionary process of myositis ossificans.20 Pamidronate, a drug typically used to treat high serum calcium concentrations, has been reported to be effective in improving clinical and radiographic findings in patients with myositis ossificans.21 Non-pharmacologic treatment usually consists of a short period of immobilization combined with rest, ice, compression, and elevation.22 Physical therapy can then be used to maintain motion and strength. If large hematomas are present, aspiration may be of benefit.
Surgery is generally reserved for persistently symptomatic lesions that have failed conservative therapy. Intractable pain, compression or irritation of adjacent structures, and decreased range of motion are possible indications for operative intervention.23 Classically, surgery is delayed for 6 to 18 months after presentation, as it is thought that surgery does not alter the maturation process and therefore early excision may increase the risk of recurrence.24 However, more recent studies have suggested minimal risk of recurrence with early resection, and it has been suggested that patients who develop myositis ossificans in the setting of unrecognized or minor trauma may benefit from early resection more than those patients who have sustained a major predisposing injury.
Conclusion
Myositis ossificans is a self-limiting, typically post-traumatic cause of bone producing intramuscular lesions. It progresses through characteristic stages both histologically and on imaging studies. Other processes that form calcification or ossification within soft tissue can be confused with myositis ossificans. Of these, the most important distinction to make is to exclude bone forming malignancies such as osteosarcoma. The findings of myositis ossificans on MRI and plain films undergo a typical progression through the time course of the disease, and knowledge of these appearances allows the proper diagnosis to be made.
References
- Flores DV, Gomez CM, Estrada-Castrillon M, et al. MR Imaging of Muscle Trauma: Anatomy, Biomechanics, Pathophysiology, and Imaging Appearance. Radiographics 2018; 38:124-148. ↩
- Jones R, Morgan D: On osseous formations in muscle due to injury (traumatic myositis ossificans). Archive of the Roentgen 1905;9(11):245. ↩
- Coley WB: Myositis ossificans traumatica: A report of three cases demonstrating the difficulties of diagnosis from sarcoma. Ann Surg 1913;57(3):305–337. ↩
- Kransdorf MJ, Meis JM, Jelinek JS: Myositis ossificans: MR appearance with radiologic-pathologic correlation. AJR Am J Roentgenol 1991;157(6):1243–1248. ↩
- Sferopoulos NK, Kotakidou R, Petropoulos AS. Myositis ossificans in children: a review. Eur J Orthop Surg Trumatol, Published online 09 March 2017. ↩
- Howard CR, Porat S, Bar-on E et al. Traumatic myositis ossificans of the quadriceps in infants. J Paed Orthop 1996: 7:80-82. ↩
- Parikh J, Hyare H, Saifuddin A. The imaging features of post-traumatic myositis ossificans with emphasis on MRI. Clinical Radiology 2002; 57:1058-1066. ↩
- Enzinger F.M., Weiss S.W.: Soft tissue tumors. St Louis: Mosby, 2002. ↩
- Walczak BE, Johnson CN, Howe BM. Myositis Ossificans. Journal of the AAOS 2015; 23(10):612-622. ↩
- Mavrogenis AF, Soucacos PN, Papagelopoulos PJ: Heterotopic ossification revisited. orthopaedics 2011;34(3):177. ↩
- Folpe AL, Gown AM: Cartilaginous and osseous soft tissue tumors, in Goldblum JR, Folpe AL, Weiss WS , eds: Enzinger & Weiss’s Soft Tissue Tumors, ed 6. Philadelphia, PA, Elsevier, 2014, pp 917–946. ↩
- Kan L, Liu Y, McGuire TL, et al.: Dysregulation of local stem/progenitor cells as a common cellular mechanism for heterotopic ossification. Stem Cells 2009;27(1):150–156. ↩
- Mirra JM. Osseous soft tumors. In: Mirra JM, Picci P, Gold RH, editors. Bone tumors: Clinical, radiologic and pathologic correlations. London: Lea and Febiger; 1989. P. 1549-86. ↩
- Wang H, Nie P, Li Y, et al. MRI findings of early myositis ossificans without calcification or ossification. BioMed Research International September 2018, Article ID 4186324, 6 pages. ↩
- Mirra JM. Osseous soft tumors. In: Mirra JM, Picci P, Gold RH, editors. Bone tumors: Clinical, radiologic and pathologic correlations. London: Lea and Febiger; 1989. P. 1549-86. ↩
- De Smet AA, Norris MA, Fisher DR. Magnetic resonance imaging of myositis ossificans: analysis of seven cases. Skeletal Radiology 1992: 21:50.-507. ↩
- Luczynska E, Kasperkiewicz H, Domalike A et al. Myositis ossificans mimicking sarcoma, the importance of diagnostic imaging-case report. Pol J Radiol 2014; 79:228-232. ↩
- Grebic D. Pozderac I, Milas I, Domagoj E. A sporadic case of myositis ossificans of the forearm presenting clinically as mesenchymal tumor. Medicina fluminensis 2017; 53(2):225-230. ↩
- Lipscomb AB, Thomas ED, Johnston RK: Treatment of myositis ossificans traumatica in athletes. Am J Sports Med 1976;4(3):111–120. ↩
- Mann D, McCormack B. Commentary: Myositis ossificans. CJEM 1999;1:199. ↩
- Mani-Babu S, Wolman R, Keen R: Quadriceps traumatic myositis ossificans in a football player: Management with intravenous pamidronate. Clin J Sport Med 2014;24(5):e56–e58. ↩
- Järvinen TA, Järvinen TL, Kääriäinen M, Kalimo H, Järvinen M: Muscle injuries: Biology and treatment. Am J Sports Med 2005;33(5):745–764. ↩
- Ellerin BE, Helfet D, Parikh S, et al.: Current therapy in the management of heterotopic ossification of the elbow: A review with case studies. Am J Phys Med Rehabil 1999;78(3):259–271. ↩
- Garland DE: A clinical perspective on common forms of acquired heterotopic ossification. Clin Orthop Relat Res 1991;263:13–29. ↩