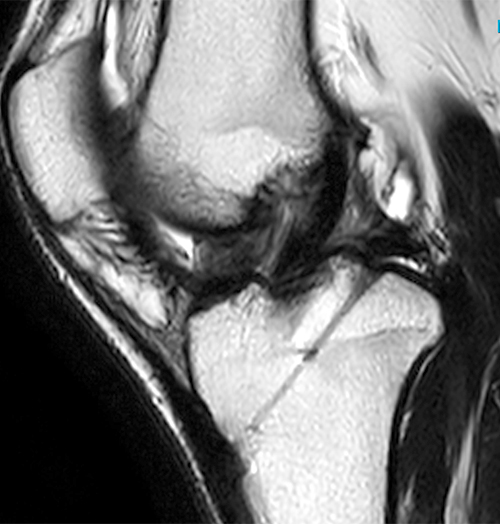
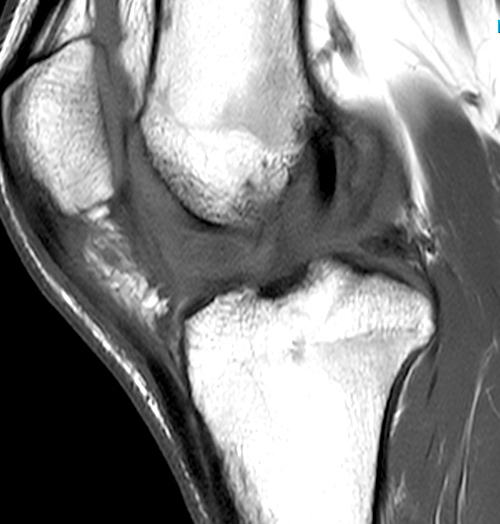
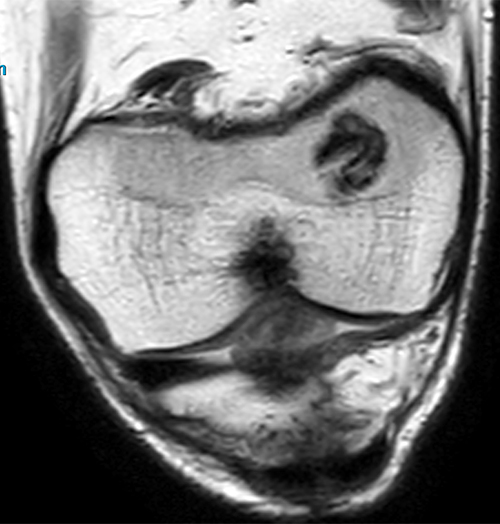
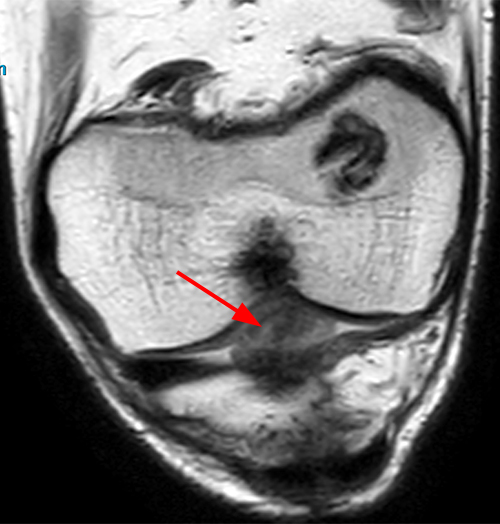
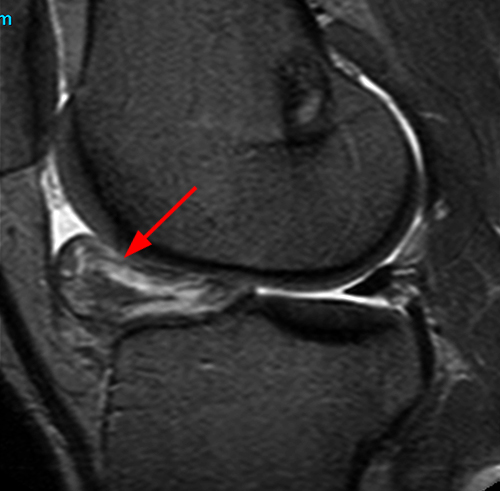
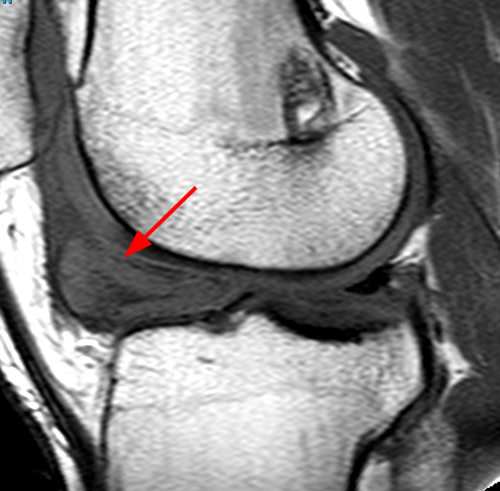
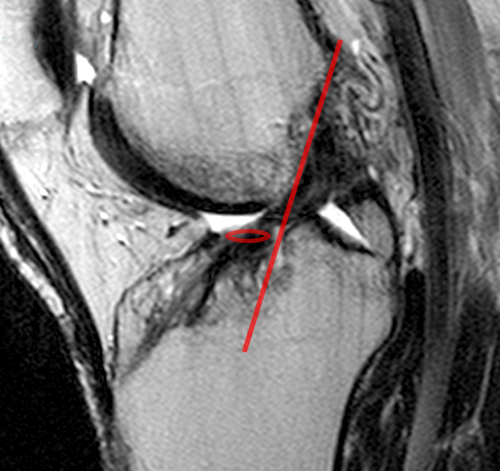
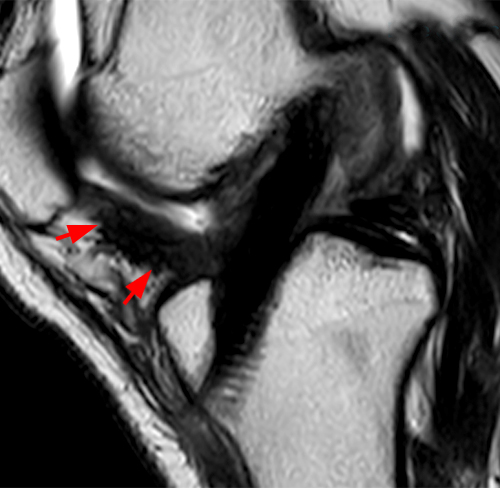
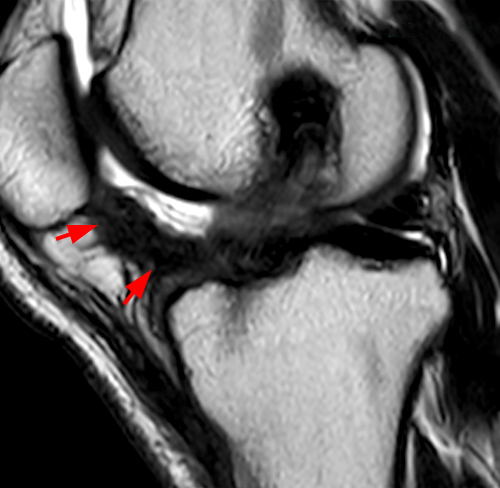
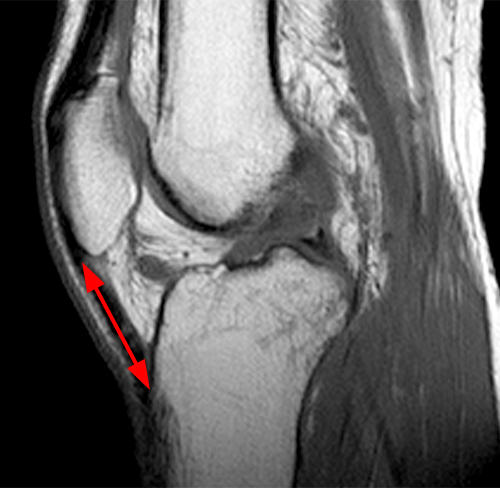
Clinical history: A 19 year-old male presents with limited range of motion of the knee 8 months following anterior cruciate ligament (ACL) reconstruction and a transtibial pullout repair of the posterior root of the lateral meniscus. Sagittal T2-weighted (1A) and T1-weighted (1B) images through the ACL graft and a coronal oblique proton density-weighted (1C) image anterior to the ACL graft are provided. What are the findings? What is your diagnosis?
Findings:
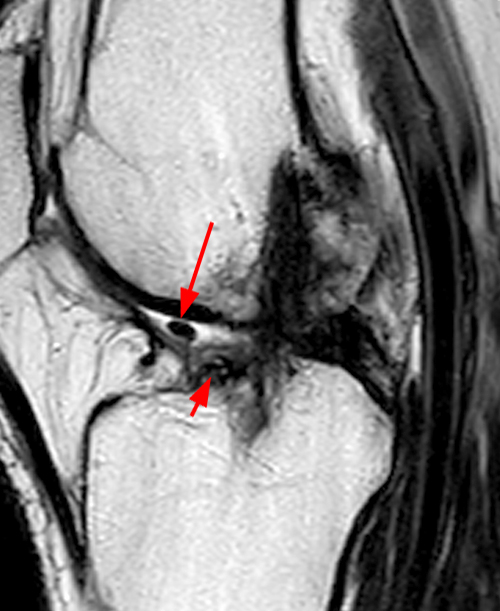
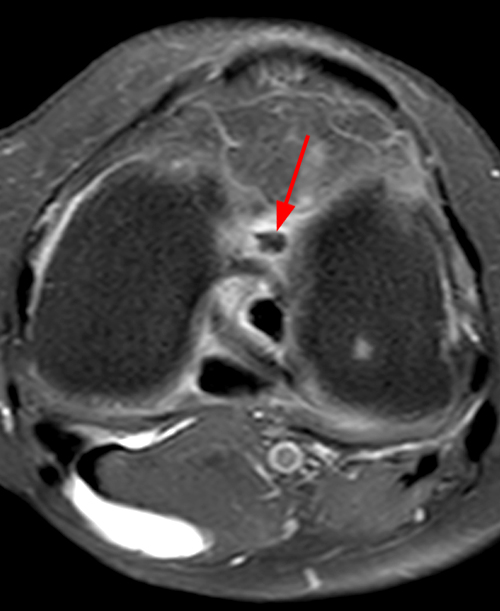
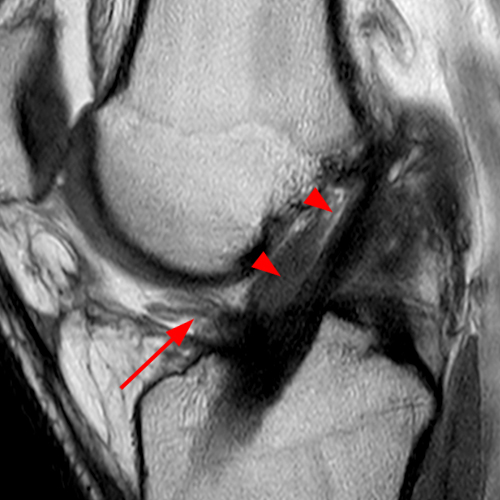
Figure 2:
(2A) The T2-weighted sagittal image demonstrates a nodular heterogeneously low signal mass (arrow) at the anterior margin of the ACL graft. A band of low signal extends over the posterior aspect of the infrapatellar fat pad (short arrows). The tract of the transtibial pullout repair extends obliquely through the tibia (arrowheads). Mild low-signal thickening (arrowhead) is present posterior to the ACL graft, overlying the reattached posterior root of the lateral meniscus. (2B) On the T1-weighted sagittal image, the nodular focus anterior to the ACL (arrow) is heterogeneous but almost isointense to the joint fluid and articular cartilage with subtle central areas of reduced signal. Similar signal characteristics are noted at the posterior margin of the infrapatellar fat pad. (2C) The oblique proton density-weighted image again demonstrates the mass (arrow) anterior to the inferior portion of the central femoral trochlea.
Diagnosis
Arthrofibrosis of the knee with a cyclops lesion anterior to the ACL graft, fibrosis of the anterior interval, and posterior pericapsular fibrosis.
Arthrofibrosis of the knee
Introduction
Arthrofibrosis is the abnormal proliferation of fibrous tissue in a joint leading to loss of motion, pain, muscle weakness, swelling, and functional limitation and is most commonly associated with joint trauma or surgery.1
In the knee, arthrofibrosis most often occurs following anterior cruciate ligament reconstruction and total knee arthroplasty and represents a potentially devastating complication. Clinical evaluation is the mainstay in establishing the diagnosis of arthrofibrosis, however MRI plays an important role in establishing the extent of involvement by fibrosis and to exclude other complications that may have a similar clinical presentation.
Etiology
The cause of arthrofibrosis is multifactorial and incompletely understood. In general, an inciting trauma, surgery, or infection results in a healing response which includes the migration of inflammatory cells and the proliferation of fibroblasts followed by the release of cytokines, growth factors, and reactive oxygen and nitrogen species.1 Failure to terminate the healing response normally results in persistent inflammation of the synovial tissue with increased inflammatory cytokines and certain growth factors that trigger tissue fibrosis via the transformation of fibroblasts.1 Fibroblast proliferation results in the accumulation of increased extracellular matrix which impairs blood flow and results in local hypoxia. Hypoxia acts to stimulate further fibroblast proliferation and extracellular matrix and also induces the metaplastic conversion to fibrocartilage, which can undergo enchondral ossification and result in heterotopic bone formation.1
ACL reconstruction
Arthrofibrosis following ACL reconstruction can present as a focal or diffuse process limiting the mobility of the knee. Other factors that can lead to knee stiffness and restriction in motion after ACL reconstruction may also play a role in the development of arthrofibrotic lesions and include suboptimal femoral or tibial tunnel placement and an overtensioned ACL graft.2
The cyclops lesion, a well-known complication of ACL reconstruction surgery, is an ovoid fibroproliferative nodule found anterior to the ACL graft. The moniker of “cyclops lesion” was given based on the arthroscopic appearance of the fibrous nodule and vessels that resemble an eye. The triggering insult stimulating the formation of a cyclops lesion is unclear with theories including an inflammatory response to drilling debris from the tibial tunnel, remnants of the native ACL, and from scar tissue and piling up of graft fibers arising from repeated graft impingement.3,1,4 No clear difference in the incidence of cyclops lesions is found between bone-patellar tendon-bone and hamstring allografts.5 Muellner et al. described two histologic subtypes.6 The true cyclops is hard and composed of fibrocartilaginous tissue with active central bone formation and no granulation tissue or inflammatory cell infiltration.6 The true cyclops lesions are more likely to be symptomatic.7 The second type, termed a “cyclopoid” lesion, is soft and composed largely of fibrous and granulation tissue with occasional cartilaginous islands.6,4
Both true and cyclopoid types are simply referred to as cyclops lesions, and they are usually indistinguishable by MRI. MRI has an accuracy of 85% in detecting cyclops lesions increasing to over 90% for lesions measuring greater than 1 cm.8 Cyclops lesions are typically small and measure 10-15mm in diameter.8 However, significantly larger lesions may be encountered (Figure 3). On MRI, cyclops lesions are adherent to the ACL graft and are hypointense or isointense to muscle on T1-weighted images and variable in signal intensity on proton density- and T2-weighted images.4 Rarely, areas of ossification within the cyclops lesion are well formed and large enough to be detected on MRI as circumscribed foci with internal signal that mirrors marrow fat signal on T1-weighted and fluid-sensitive sequences (Figure 4).
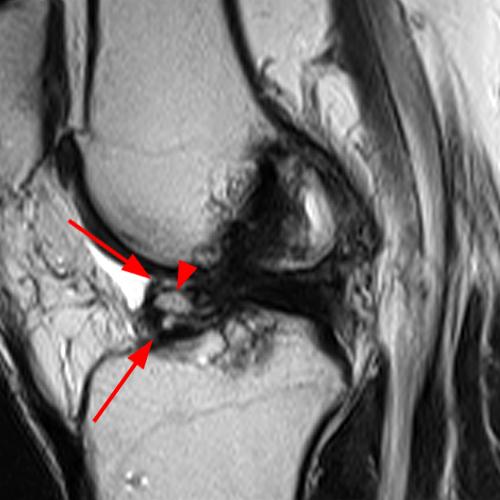
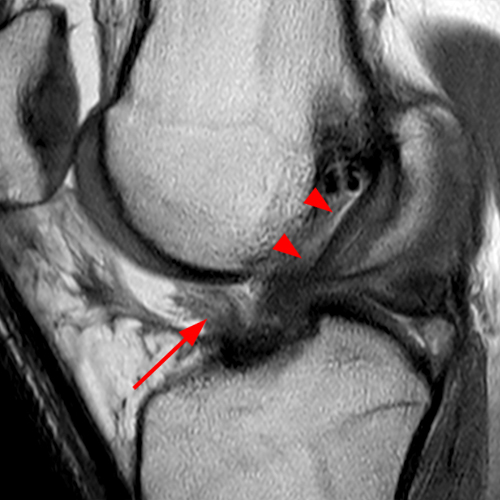
Figure 3:
An 18 year-old female college athlete presents 6 months following ACL reconstruction with locking and catching. Sagittal fat-suppressed proton density-weighted (3A), sagittal T1-weighted (3B), and axial proton density-weighted images demonstrate a large heterogeneous cyclops lesion (arrows) anterior to the ACL graft.
Cyclops lesions can be found in up to 25% of ACL reconstructions at 6 months after surgery. Despite such prevalence, cyclops lesions generally have minimal or no clinical symptoms, and their presence does not portend an inferior clinical outcome, with only 2% of cyclops lesions prompting surgical intervention.9 Symptomatic lesions present with loss of extension, snapping, catching, and painful extension with walking and/or running resulting in the “cyclops syndrome.” 7,8
MRI can assist in distinguishing cyclops lesions from other pathology that may limit knee extension, including roof impingement of the ACL graft (Figure 5), intra-articular bodies (Figure 6), and displaced torn ACL graft fibers. A pseudocyclops lesion (Figure 7) results from anteriorly displaced fibers from a partial tear of the ACL graft which can mimic a cyclops lesion clinically and on MRI.10
Figure 6:
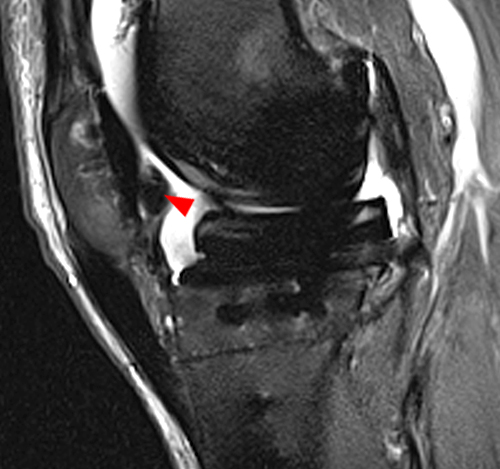
A 66 year-old female 10 years post ACL reconstruction with intermittent locking. Sagittal T2-weighted (5A) and axial fat-suppressed proton density-weighted (5B) images demonstrate a 5 mm intra-articular chondral body (arrows) surrounded by joint fluid anterior to the ACL graft. A focus of soft tissue thickening is compatible with a small cyclops lesion anterior to the graft (arrowhead).
Figure 7:
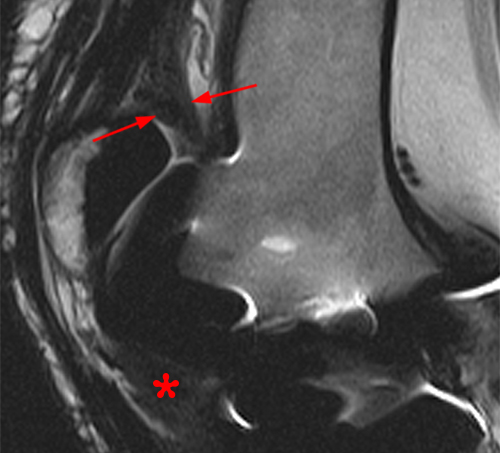
A 17 year-old male 1 year after ACL reconstruction, felt a pop while stepping into a hole with swelling and limited extension at the knee. Sequential sagittal proton-density weighted images demonstrate loss of ligament tissue anteriorly (arrowheads) within the intercondylar notch compatible with a partial tear. Torn anterior ACL graft fibers remain continuous with the graft in the tibial tunnel and are folded anteriorly (arrows) resulting in a pseudocyclops lesion.
Diffuse arthrofibrosis surrounding the ACL graft is rare. Patients may present with decreased range of motion in flexion and extension. Factors that are felt to increase the likelihood of diffuse arthrofibrosis include ACL reconstruction within 4 weeks of the ACL injury, additional ligamentous injuries, and diminished knee flexion preoperatively. Increased preoperative and postoperative inflammation reflected by swelling, effusion, and hyperthermia also plays an important role in the development of this complication.7,11 On MRI, fibrotic tissue encases the ACL graft and can extend anteriorly into the infrapatellar fat pad and suprapatellar bursa or posteriorly to the posterior joint capsule (Figure 8).7
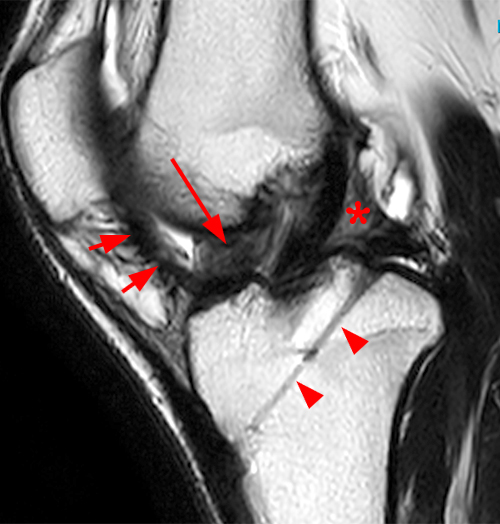
Figure 8:
An 18 year-old female 5 months after ACL reconstruction with pain and diminished range of motion. A sagittal proton density-weighted image demonstrates a diffuse fibrotic reaction encasing the ACL graft with a cyclops lesion anterior to the ACL graft (arrow) and fibrosis posterior to the ACL graft (asterisk) extending to the posterior capsule. Also noted is fibrosis within the infrapatellar fat pad (arrowheads).
Intra-articular fibrosis can occur elsewhere within the knee and may be associated with loss of flexion and/or extension depending on the location. Adhesions in the suprapatellar bursa can form between the capsular elements of the bursa and the medial or lateral gutters. Adhesions can form between the capsule and articular cartilage. Fibrosis in the suprapatellar bursa typically limits knee flexion. Scarring and contraction resulting in a foreshortened suprapatellar bursa leads to further loss of knee flexion.2
Fibrosis of the infrapatellar fat pad appears to be an important cause of pain and stiffness.12,13 The infrapatellar fat pad is susceptible to trauma at the time of the ACL tear, from untreated instability, and from subsequent arthroscopic surgery and ACL reconstruction. The infrapatellar fat pad is richly innervated and is an important pain generator in the knee.14 Surgical and traumatic insults to the infrapatellar fat pad can induce fibrosis and metaplasia resulting in pain (September 2008 Web Clinic – Patellar Fat Pad Abnormalities).13,14
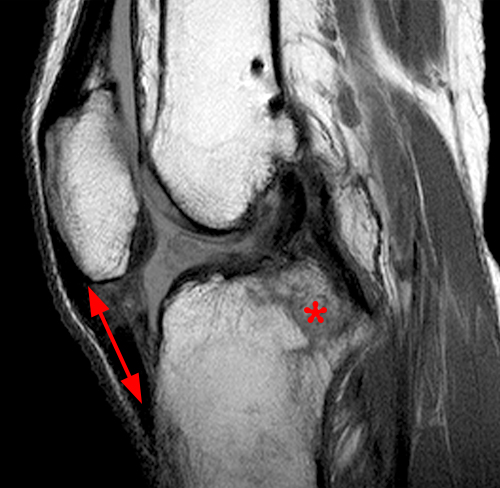
Excessive fibrosis of the infrapatellar fat pad can result in altered biomechanics of the anterior knee. Various terms have been used to describe this pathology including infrapatellar contracture syndrome, synovial fibrosis of the infrapatellar fat pad, scarring of the anterior interval, and patella infera syndrome.12,15,16 Postoperative scarring normally appears as thin linear or spiculated regions of low signal on all sequences with small slightly thickened and more nodular portions found along the route of the arthroscopic portals and at the posterior margin of the fat pad (Figure 9).16 In contrast, symptomatic fibrosis results from more extensive fibrotic changes appearing as thickened and irregular areas of low signal on all sequences, which can greatly reduce the amount of normal fat.
The anterior interval of the knee is found posterior to the patellar fat pad and anterior to the anterosuperior tibial plateau.2 Scarring over the posterior aspect of the infrapatellar fat pad from the patella to the anterior surface of the tibia or the transverse meniscal ligament can bridge the interval and result in restriction of the normal biomechanics of the anterior knee with increased tension on the fat pad, diminished translation of the patellar tendon and patellar entrapment (Figure 10).15
In severe cases of infrapatellar fat pad arthrofibrosis, fibrosis between the patella, patellar tendon, and tibia can result in severe retraction and tethering of the patella leading to patella baja which may become progressive (patella infera). Extracapsular fibrosis may also be seen. This can be a particularly devastating complication that can rapidly lead to osteoarthrosis at the patellofemoral joint if left untreated. Calcification of the fat pad may be present and visible on plain radiographs.1 The MRI findings include severe scarring in the infrapatellar fat pad and progressive patella baja. The development of patella baja is made more apparent by comparing current and prior studies by plain film or MRI (Figure 11).
Figure 11:
48 year-old male with sagittal T1-weighted images at the time of the ACL tear (11A) and 2 years later after a fall (11B) demonstrates the development of severe scarring within the infrapatellar fat pad and posterior to the patellar tendon with interval inferior displacement of the patella. Incidentally noted is a hemarthrosis (11B) (with joint fluid appearing hyperintense to muscle) associated with an intra-articular fracture of the posterior tibia (asterisk).
Total Knee Arthroplasty
Arthrofibrosis associated with total knee arthroplasty (TKA) can result in significant pain and impairment. The incidence of arthrofibrosis following TKA is approximately 4%.17 Arthrofibrosis as the cause for TKA revision ranges from 4.5 to 6.9%.18,19 Multiple factors affect the development of arthrofibrosis following TKA, including surgical technique, component selection, post-operative rehabilitation course, underlying patient-specific disease and genetic factors, and preoperative range of motion.18,19 Some authors suggest a relationship between diffuse arthrofibrosis and chronic infection.18,20,21 Pre-operative range of motion appears to be the most important predictor of postoperative stiffness.18,20,22 Arthrofibrosis associated with TKA most often appears within 5 years of surgery.19 Stiffness and arthrofibrosis developing after 5 years is often associated with other complications such as aseptic loosening, infection, or polyethylene wear.19
With specific techniques and modifications to reduce metal artifacts, MRI is effective in evaluating the complications of TKA including implant loosening, periprosthetic infection, fractures, extensor mechanism injury, polyethylene wear, and arthrofibrosis. MRI can assist in the evaluation of arthrofibrosis in patients with a normal radiographic appearance of the implant but with a limited range of motion.17
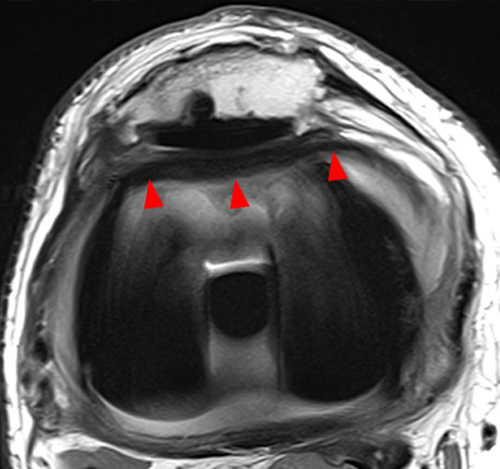
MR imaging findings of diffuse arthrofibrosis include widespread heterogeneous thickening of the synovium. The scarred synovium is hypointense to muscle on proton density-weighted and T2-weighted MR images (Figure 12).17
Focal areas of fibrosis following TKA are often seen in the peripatellar region and can present with mechanical symptoms. On MRI, nodular or band-like synovial thickening or intra-articular masses demonstrate low to intermediate signal on proton-density and T2-weighted images (Figure 13).
Figure 13:
A 60 year-old male 4 years post TKA complains of pain and popping of the knee with walking for the last 6 weeks. On the sagittal inversion recovery image (13A) an abnormal low signal focus is noted posterior to the patella (arrowhead). The axial proton density-weighted image (13B) reveals this structure to be a band-like region of arthrofibrosis (arrowheads) passing posterior to the patella and blending with the synovium medial and lateral to the patella, likely contributing to the patient’s mechanical symptoms.
Patellar clunk syndrome results from localized fibrous tissue forming at the quadriceps insertion on the proximal pole of the patella and can be seen in up to 3.5% of posterior-stabilized TKAs.23 Patients present with a locking sensation or decreased motion during flexion and extension.17 An audible clunk may be observed on physical exam when the knee is extended from the flexed position, presumably from entrapment of the tissue in the intercondylar notch with flexion and abrupt displacement with extension (Figure 14).
Figure 14:
A 40 year-old female who underwent revision TKA 1 year prior presents with catching and locking symptoms anteriorly when going from 90 degrees of flexion to full extension. A sagittal T2-weighted image demonstrates prominent peripatellar scarring in the infrapatellar fat pad (asterisk) and above the patella with a nodular component extending inferiorly at the posterior margin of the superior patella (arrows). The appearance and clinical history are suggestive of patellar clunk syndrome.
Treatment
Concerns of emerging arthrofibrosis should be raised if physical therapy fails to achieve expected range of motion targets following surgery. Initially, a more aggressive physical therapy regimen is attempted along with anti-inflammatory medications. Assessment of the type of deficit is important in directing the therapeutic approach. Splinting or bracing may be used for extension deficits.
Unresolved deficits warrant further intervention including manipulation under anesthesia, arthroscopic debridement, and open debridement. Many authors recommend arthroscopic debridement prior to manipulation under anesthesia to mitigate the risk of fracture, chondral damage, intra-articular hemorrhage, and ligament or tendon rupture. In general, arthroscopic debridement is preferred to open debridement when the pathology is largely intra-articular. Arthroscopic excision is the treatment of choice for cyclops syndrome. Arthroscopic release of anterior interval adhesions is also successful in relieving pain and restoring range of motion. Combinations of arthroscopic debridement of the notch and fat pad, release of scarred fat pad adherent to the retinacular structures and patellar manipulation are used successfully to treat refractory patellofemoral arthrofibrosis.24,25,1,26
Treatment for TKA arthrofibrosis includes manipulation under anesthesia, arthroscopic and open releases, and revision TKA. Of these treatment approaches, revision TKA appears to be least likely to result in clinical improvement.18,20
Conclusion
Arthrofibrosis is a common complication of ACL reconstruction and total knee arthroplasty and can result in a frustrating clinical course and poor functional results. MRI is effective as a tool to evaluate unexplained pain, limited range of motion, and functional limitation in the postoperative patient in whom arthrofibrosis is suspected. MRI can confirm and define the extent of a suspected fibrotic lesion and assist in detecting and differentiating other postoperative complications with a similar clinical presentation.
References
- Haklar U, Ayhan E, Ulku TK, Karaoglu S. Arthrofibrosis of the Knee. In: Doral M, Karlsson J, eds. Sports Injuries. Berlin, Heidelberg: Springer Berlin Heidelberg; 2014:1-20. doi:10.1007/978-3-642-36801-1_100-1 ↩
- Kim DH, Gill TJ, Millett PJ. Arthroscopic treatment of the arthrofibrotic knee. Arthrosc – J Arthrosc Relat Surg. 2004;20(6 SUPPL.):187-194. doi:10.1016/j.arthro.2004.04.036 ↩
- Delincé P, Krallis P, Descamps PY, Fabeck L, Hardy D. Different aspects of the cyclops lesion following anterior cruciate ligament reconstruction: a multifactorial etiopathogenesis. Arthroscopy. 14(8):869-876. http://www.ncbi.nlm.nih.gov/pubmed/9848601. ↩
- Srinivasan R, Wan J, Allen CR, Steinbach LS. Knee Imaging Following Anterior Cruciate Ligament Reconstruction: The Surgeon’s and Radiologist’s Perspectives. Semin Musculoskelet Radiol. 2018;22(4):386-397. doi:10.1055/s-0038-1653953 ↩
- Sanders TL, Kremers HM, Bryan AJ, Kremers WK, Stuart MJ, Krych AJ. Procedural intervention for arthrofibrosis after ACL reconstruction: trends over two decades. Knee Surgery, Sport Traumatol Arthrosc. 2017;25(2):532-537. doi:10.1007/s00167-015-3799-x ↩
- Muellner T, Kdolsky R, Großschmidt K, Schabus R, Kwasny O, Plenk H. Cyclops and cyclopoid formation after anterior cruciate ligament reconstruction: Clinical and histomorphological differences. Knee Surgery, Sport Traumatol Arthrosc. 1999;7(5):284-289. doi:10.1007/s001670050165 ↩
- Bencardino JT, Beltran J, Feldman MI, Rose DJ. MR Imaging of Complications of Anterior Cruciate Ligament Graft Reconstruction. RadioGraphics. 2009;29(7):2115-2126. doi:10.1148/rg.297095036 ↩
- Bradley DM, Bergman AG, Dillingham MF. MR Imaging of Cyclops Lesions. Am J Roentgenol. 2000;174(3):719-726. doi:10.2214/ajr.174.3.1740719 ↩
- Facchetti L, Schwaiger BJ, Gersing AS, et al. Cyclops lesions detected by MRI are frequent findings after ACL surgical reconstruction but do not impact clinical outcome over 2 years. Eur Radiol. 2017;27(8):3499-3508. doi:10.1007/s00330-016-4661-3 ↩
- Simpfendorfer C, Miniaci A, Subhas N, Winalski CS, Ilaslan H. Pseudocyclops: two cases of ACL graft partial tears mimicking cyclops lesions on MRI. Skeletal Radiol. 2015. doi:10.1007/s00256-015-2100-1 ↩
- Mayr HO, Weig TG, Plitz W. Arthrofibrosis following ACL reconstruction – Reasons and outcome. Arch Orthop Trauma Surg. 2004. doi:10.1007/s00402-004-0718-x ↩
- Yoon KH, Tak DH, Ko TS, Park SE, Nam J, Lee SH. Association of fibrosis in the infrapatellar fat pad and degenerative cartilage change of patellofemoral joint after anterior cruciate ligament reconstruction. Knee. 2017;24(2):310-318. doi:10.1016/j.knee.2016.10.018 ↩
- Abreu MR, Chung CB, Trudell D, Resnick D. Hoffa’s fat pad injuries and their relationship with anterior cruciate ligament tears: New observations based on MR imaging in patients and MR imaging and anatomic correlation in cadavers. Skeletal Radiol. 2008;37(4):301-306. doi:10.1007/s00256-007-0427-y ↩
- Flores D V., Mejía Gómez C, Pathria MN. Layered Approach to the Anterior Knee: Normal Anatomy and Disorders Associated with Anterior Knee Pain. RadioGraphics. 2018;38(7):2069-2101. doi:10.1148/rg.2018180048 ↩
- Steadman JR, Dragoo JL, Hines SL, Briggs KK. Arthroscopic Release for Symptomatic Scarring of the Anterior Interval of the Knee. Am J Sports Med. 2008;36(9):1763-1769. doi:10.1177/0363546508320480 ↩
- Paulos LE, Rosenberg TD, Drawbert J, Manning J, Abbott P. Infrapatellar contracture syndrome. Am J Sports Med. 1987;15(4):331-341. doi:10.1177/036354658701500407 ↩
- Fritz J, Lurie B, Potter HG. MR Imaging of Knee Arthroplasty Implants. RadoiGraphics. 2015;35(5):1483-1501. doi:10.1148/rg.2015140216 ↩
- Sharkey PF, Lichstein PM, Shen C, Tokarski AT, Parvizi J. Why Are Total Knee Arthroplasties Failing Today-Has Anything Changed After 10 Years? J Arthroplasty. 2014;29:1774-1778. doi:10.1016/j.arth.2013.07.024 ↩
- Schroer WC, Berend KR, Lombardi A V., et al. Why are total knees failing today? Etiology of total knee revision in 2010 and 2011. J Arthroplasty. 2013. doi:10.1016/j.arth.2013.04.056 ↩
- Kim J, Nelson CL, Lotke PA. Stiffness After Total Knee Arthroplasty:Prevalence of the complication and outcomes of revision. JBJS.2004;86(7):1479-1484. doi: 10.2106/00004623-200407000-00017 ↩
- Su EP, Su SL, Valle AG Della. Stiffness After TKR: How to Avoid Repeat Surgery. orthopaedics. 2010;33(9):658. doi:10.3928/01477447-20100722-48 ↩
- Gandhi R, De Beer J, Leone J, Petruccelli D, Winemaker M, Adili A. Predictive risk factors for stiff knees in total knee arthroplasty. J Arthroplasty. 2006. doi:10.1016/j.arth.2005.06.004 ↩
- Lucas TS, DeLuca PF, Nazarian DG, Bartolozzi AR, Booth RE. Arthroscopic treatment of patellar clunk. Clin Orthop Relat Res. 1999;(367):226-229. http://www.ncbi.nlm.nih.gov/pubmed/10546619. ↩
- Richmond JC, Al Assal M. Arthroscopic Management of Arthrofibrosis of the Knee, Including Infrapatellar Contraction Syndrome. Arthrosc – J Arthrosc Relat Surg. 1991;7(2):144-147. ↩
- Dragoo JL, Johnson C, McConnell J. Evaluation and treatment of disorders of the infrapatellar fat pad. Sport Med. 2012;42(1):51-67. doi:10.2165/11595680-000000000-00000 ↩
- Calloway SP, Soppe CJ, Mandelbaum BR. Clinical Outcomes After Arthroscopic Release of Patellofemoral Arthrofibrosis in Patients With Prior Anterior Cruciate Ligament Reconstruction. Arthrosc – J Arthrosc Relat Surg. 2018;34(5):1603-1607. doi:10.1016/j.arthro.2017.12.011 ↩